Abstract
OBJECTIVES:
To explore the longitudinal association of neonatal adiposity (fat mass percentage) with BMI trajectories and childhood overweight and obesity from ages 2 to 6 years.
METHODS:
We studied 979 children from the Healthy Start cohort. Air displacement plethysmography was used to estimate fat mass percentage. Child weight and recumbent length or standing height were abstracted from medical records. Overweight and obesity were defined as BMI levels ≥85th percentile for age and sex. Mixed-effects models were used to examine the association between neonatal fat mass percentage and BMI trajectories from age 2 to 6 years. We tested for effect modification by sex, race and/or ethnicity, and breastfeeding duration. We estimated the proportion of children classified as overweight or obese at specific levels of neonatal fat mass percentage (mean ± SD).
RESULTS:
The mean neonatal adiposity level was 9.1% ± 4.0%. Child BMI levels differed by neonatal adiposity. Each SD increase in neonatal adiposity resulted in a 0.12 higher overall BMI level between ages 2 to 6 years (95% confidence interval: 0.03 to 0.20; P < .01), and this association was not modified by offspring sex, race and/or ethnicity, or breastfeeding duration. Increasing neonatal adiposity was associated with an increasing proportion of childhood overweight and obesity by age 5 years (P = .02).
CONCLUSIONS:
We provide novel evidence that higher neonatal adiposity is significantly associated with higher overall BMI levels and an increased likelihood of overweight or obesity from ages 2 to 6 years. Because various prenatal exposures may specifically influence offspring fat accretion, neonatal adiposity may be a useful surrogate end point for prenatal interventions aimed at reducing future childhood overweight and obesity.
The Healthy Start Study has been registered at www.clinicaltrials.gov (identifier NCT02273297).
Obesity and metabolic disorders among children have become increasingly common. Despite various public health initiatives in the United States, the prevalence of childhood overweight and obesity has increased steadily since 1988.1 Estimates from the 2015 to 2016 NHANES indicate that 1 in 3 children have overweight or obesity.2 The increasing proportion of obesity is closely tracked by a similar rise in the prevalence of metabolic diseases among children.3
Pregnancy may be an ideal time for prevention because obesity and metabolic diseases are primed during fetal development.4 Numerous trials have been focused on providing dietary and physical activity counseling to pregnant women classified as overweight or obese,5–7 with offspring birth weight as one of the main end points. Although the risk of macrosomia was reduced in one trial,6 most had no effects on birth weight.5,7 Although birth weight is generally associated with having obesity later in life,8 it may not capture more specific effects of prenatal exposures on offspring fat accretion. For example, in the Healthy Start cohort, we found that both maternal diet9 and third trimester energy expenditure10 had specific effects on neonatal fat mass but did not influence neonatal fat-free mass or birth weight. However, there are limited data on whether neonatal adiposity is in fact associated with childhood BMI later in life. In an Ethiopian population, Admassu et al11 used air displacement plethysmography to directly measure fat mass at birth and at 4 years of age. Increased fat mass at birth was associated with an increased fat mass index at 4 years of age.11 There is a need to conduct a longitudinal analysis of the relationship between neonatal adiposity and childhood BMI trajectories.12 In addition, childhood BMI growth differs by sex and race and/or ethnicity of the child13 and modifiable factors, such as breastfeeding duration.14 The association between neonatal adiposity and childhood BMI growth may vary across subgroups, but this has yet to be explored.
We assessed the longitudinal associations between neonatal adiposity (defined as the proportion of the fat mass divided by total mass) and childhood BMI levels between 2 and 6 years of age as well as the prevalence of overweight and obesity in an ethnically diverse prebirth cohort in Colorado.
METHODS
Study Population
The Healthy Start Study recruited 1410 pregnant women aged ≥16 years with singleton pregnancies before 24 weeks’ gestation from the obstetrics clinics at the University of Colorado Hospital between 2010 and 2014. Enrolled women had no history of previous stillbirth or extremely preterm birth (<25 weeks’ gestation) and no self-reported diagnosis of diabetes, cancer, asthma managed with steroids, or psychiatric illness. Participants completed 2 research visits during pregnancy (median of 17 and 27 weeks’ gestation) and at delivery (median of 1-day postdelivery). Approximately 1297 offspring are now being followed. Participants were eligible for the current analysis if they had complete body composition measures at birth and had ≥3 length and height and weight measurements from well-child visits from age 2 to 6 years. The Healthy Start Study protocol was approved by the Colorado Multiple Institutional Review Board. All women provided written informed consent before the first study visit. The Healthy Start Study was registered as an observational study at www.clinicaltrials.gov as NCT02273297.
Neonatal Size and Body Composition
Fat mass and fat-free mass were measured by using whole-body air displacement plethysmography (PEA POD; COSMED, Rome, Italy) within 72 hours of delivery. PEA POD is used to measure body mass and volume, calculate body density, and then estimate fat mass (grams) and fat-free mass (grams). Neonatal adiposity (fat mass percentage) was calculated as a proportion of the fat mass divided by total mass. Plethysmography has excellent validity and reliability in infants.15–17 Trained research personnel measured fat mass and fat-free mass twice, with a third measurement taken when the percent of fat mass differed by >2.0%. The average of the 2 closest readings was used for analysis.
Growth Data
We abstracted weight, recumbent length (generally until 24 months), and standing height (generally after 24 months) from medical records at pediatric visits. These measurements were generally recorded at well-child visits, which occur at 24, 30, 36, 48, 60, and 72 months of age. BMI was calculated by dividing the weight in kilograms by height in meters squared. BMI-for-age z scores were calculated by using the 2006 World Health Organization (WHO) growth charts.18 Overweight or obesity was defined as having a BMI ≥85th percentile for age and sex.
Demographic Data
Household income, maternal education, maternal race and/or ethnicity, and active smoking during pregnancy were self-reported via study questionnaires. Maternal age at delivery was calculated by subtracting the participant’s date of birth from the date of delivery. Maternal height was measured by using a stadiometer during the first pregnancy research visit. Prepregnancy weight was obtained from medical records (91%) or self-reported at the first pregnancy research visit (9%). Prepregnancy BMI was calculated as prepregnancy weight (kilograms) divided by height squared (square meters). Gestational weight gain was calculated as the difference between the last available weight measurement during pregnancy (measured by research staff or medical personnel) and prepregnancy weight.
Data Preparation
Before our analyses, we removed implausible values of the longitudinal data. We used WHO growth standards19,20 to calculate z scores for BMI, length, weight, and weight-for-length by sex and age. If an observation exceeded a WHO defined extreme z score (weight: <−6 or >5; length: <−6 or >6; weight-for-length: <−5 or >5; and BMI: <−5 or >5), it was removed from the data set. We then removed observations in which childhood height appeared to be retained across pediatric visits or decreased over time or childhood weight increased or decreased by 15 kg in 1 year (a threshold we based on the 2000 Centers for Disease Control and Prevention growth charts).21 Finally, we developed an anomaly algorithm that was used to identify paired peaks and dips in height or weight across time. The algorithm was used to determine if a peak or dip was an outlying value. Identified outliers were then removed.
Statistical Analysis
Mixed-effects models were used to assess whether neonatal adiposity was associated with childhood BMI trajectories, from ages 2 to 6 years. Because the relationship between age and BMI is not linear,12 we used a polynomial approach to model childhood BMI trajectory as a function of age. On the basis of the deviance information criteria,22 the best-fit trajectory included a quadratic term in age. Normality assumptions were verified via examination of the Jackknifed-studentized residuals. We used Wald tests with Kenward-Roger degrees of freedom and an α level of .05 to assess the significance of the association between the neonatal adiposity and childhood BMI over time.23,24 Age, age squared, neonatal adiposity, and sex were included as predictors in the first model. We tested whether the association between neonatal adiposity and childhood BMI changed over time by introducing a product term with age into the mixed-effects model. The interaction term was not statistically significant (P = .27), suggesting that the relationship between neonatal adiposity and childhood BMI was constant over time. We also tested for interactions of neonatal adiposity with child sex, maternal race and/or ethnicity (non-Hispanic white, non-Hispanic Black, Hispanic, or other), and duration of exclusive breastfeeding (<6 or ≥6 months) by introducing product terms into separate models and found no evidence of effect modification. Therefore, we adjusted for child sex, maternal race and/or ethnicity (non-Hispanic white, non-Hispanic Black, Hispanic, or other), maternal education (less than high school, high school diploma, or some college), and, in the final model, the duration of exclusive breastfeeding (<6 or ≥6 months) to control for these potential confounders.
We estimated the proportion of children who were classified as overweight or obese by level of neonatal adiposity (mean ± SD). We calculated the respective proportions at ages 2 to 3, 3 to 4, 4 to 5, and 5 to 6 years.
All statistical analyses were performed by using SAS version 9.4 (SAS Institute, Inc, Cary, NC).
RESULTS
Of the 1297 children being followed, 979 children had at least 2 weight and length and height measurements abstracted from medical records between ages 2 and 6 years. Of these children, 946 had fat mass and fat-free mass measured at birth. There were no substantial differences in maternal or child characteristics for the analytic sample included in this report compared with the entire cohort (Supplemental Table 4). Participants had an average of 4.1 BMI measurements between the ages of 2 and 6 years (range: 1–14), with an average of 1.7 BMI measurements each year (range: 1–7).
The maternal and child characteristics of participants included in this report are shown in Table 1. Fifty-four percent of the participants were non-Hispanic white, 16% were non-Hispanic Black, 24% were Hispanic, and 6% were other. At the time of birth, mothers were, on average 28 years of age (SD: 6). Mothers had a prepregnancy BMI of 26 (SD: 6) and gained 14 kg during pregnancy (SD: 6 kg). The mean gestational age at birth was 39 weeks (SD: 1 week). Less than 4% were born before 37 weeks’ gestation. The mean birth weight was 32 380 ± 482 g, mean neonatal fat mass was 293 ± 150 g, mean fat-free mass was 2829 ± 348 g, and mean percent fat mass was 9.1% ± 4.0%. Approximately 6% of children were born at a low birth weight (<2500 g), and 5% were born macrosomic (>4000 g). In total, 40 women were diagnosed with gestational diabetes.
TABLE 1.
Characteristics of Participants in the Healthy Start Study (n = 979)
| Mean ± SD or Proportion | |
|---|---|
| Maternal Characteristics | |
| Maternal age, y, mean ± SD | 28 ± 6 |
| Gestational wt gain, kg, mean ± SD | 14 ± 6 |
| Prepregnancy BMI, mean ± SD | 26 ± 6 |
| Gestational age at birth, wk, mean ± SD | 39 ± 1 |
| Maternal race and/or ethnicity, % | |
| Non-Hispanic white | 54 |
| Non-Hispanic Black | 16 |
| Hispanic | 24 |
| Other | 6 |
| Household income, % | |
| <$40 000 | 28 |
| $40 001–$70 000 | 18 |
| >$70 000 | 34 |
| Do not know | 20 |
| Mother’s highest level of education, % | |
| <12 y | 13 |
| High school degree | 18 |
| College classes or college degree | 68 |
| Maternal smoking during pregnancy, % | |
| Yes | 8 |
| No | 92 |
| Child characteristics, mean ± SD | |
| Birth wt, g | 3230 ± 482 |
| Fat mass (n = 946), g | 293 ± 150 |
| Fat-free mass (n = 946), g | 2829 ± 348 |
| Adiposity (n = 946), % fat mass | 9.1 ± 4.0 |
| Birth wt category, % | |
| Low birth wt (<2500 g) | 6 |
| Normal birth wt (2500–4000 g) | 89 |
| Macrosomia (>4000 g) | 5 |
The indicators of neonatal body composition were correlated. The strongest correlation was between fat mass and adiposity (Pearson correlation coefficient of 0.96). Birth weight was strongly associated with fat-free mass (Pearson correlation coefficient of 0.93) and moderately correlated with fat mass and adiposity (Pearson correlation coefficients of 0.66 and 0.49, respectively). Fat-free mass was weakly correlated with fat mass and adiposity (Pearson correlation coefficients of 0.45 and 0.24, respectively).
The mixed-effects models indicated that neonatal adiposity was significantly associated with higher overall BMI levels from ages 2 to 6 years (Table 2). Model 1 shows that each SD increase in neonatal adiposity was associated with 0.12 higher BMI between ages 2 and 6 years (95% confidence interval [CI]: 0.03 to 0.20; P < .01). This association persisted after further adjustment for maternal race and/or ethnicity and education (Model 2; adjusted β-coefficient: 0.10; 95% CI: 0.2 to 0.19; P = .02) and duration of exclusive breastfeeding (Model 3; adjusted β-coefficient: 0.12; 95% CI: 0.03 to 0.22; P < .01). Figure 1 depicts the predicted BMI levels from ages 2 to 7 years by neonatal adiposity among girls and boys. At age 2 years, the predicted BMI levels among girls were at their highest (Fig 1A; 15.9 within the low neonatal adiposity group, 16.0 within the mean adiposity group, and 16.1 within the high neonatal adiposity group). BMI then declined until age 4.7 years, when the BMI trajectory changed direction. By age 7 years, the predicted BMI levels were approaching the predicted levels at age 2 years. The growth pattern was similar among boys, although boys had higher overall BMI levels (Fig 1B).
TABLE 2.
The Association Between Neonatal Adiposity and Childhood BMI From Sequentially Adjusted Mixed-Effects Regression Models
| Adjusted β (95% CI) | P | |
|---|---|---|
| Model 1 | ||
| Age, y | −0.60 (−0.73 to −0.46) | <.01 |
| Age squared | 0.06 (0.04 to 0.08) | <.01 |
| Neonatal adiposity, SD increase | 0.12 (0.03 to 0.20) | <.01 |
| Sex, male | 0.32 (0.14 to 0.49) | <.01 |
| Model 2 | ||
| Age, y | −0.60 (−0.73 to −0.47) | <.01 |
| Age squared | 0.06 (0.05 to 0.08) | <.01 |
| Neonatal adiposity, SD increase | 0.10 (0.02 to 0.19) | .02 |
| Sex, male | 0.34 (0.17 to 0.51) | <.01 |
| Maternal education | −0.22 (−0.30 to −0.15) | <.01 |
| Race and/or ethnicity | −0.25 (−0.30 to −0.33) | <.01 |
| Model 3 | ||
| Age, y | −0.50 (−0.73 to −0.47) | <.01 |
| Age squared | 0.05 (0.03 to 0.07) | <.01 |
| Neonatal adiposity, SD increase | 0.12 (0.03 to 0.22) | <.01 |
| Sex, male | 0.34 (0.17 to 0.53) | <.01 |
| Maternal education | −0.21 (−0.30 to −0.12) | <.01 |
| Race and/or ethnicity | −0.21 (−0.32 to −0.10) | <.01 |
| Exclusive breastfeeding | −0.02 (−0.01 to 0.02) | .34 |
FIGURE 1.
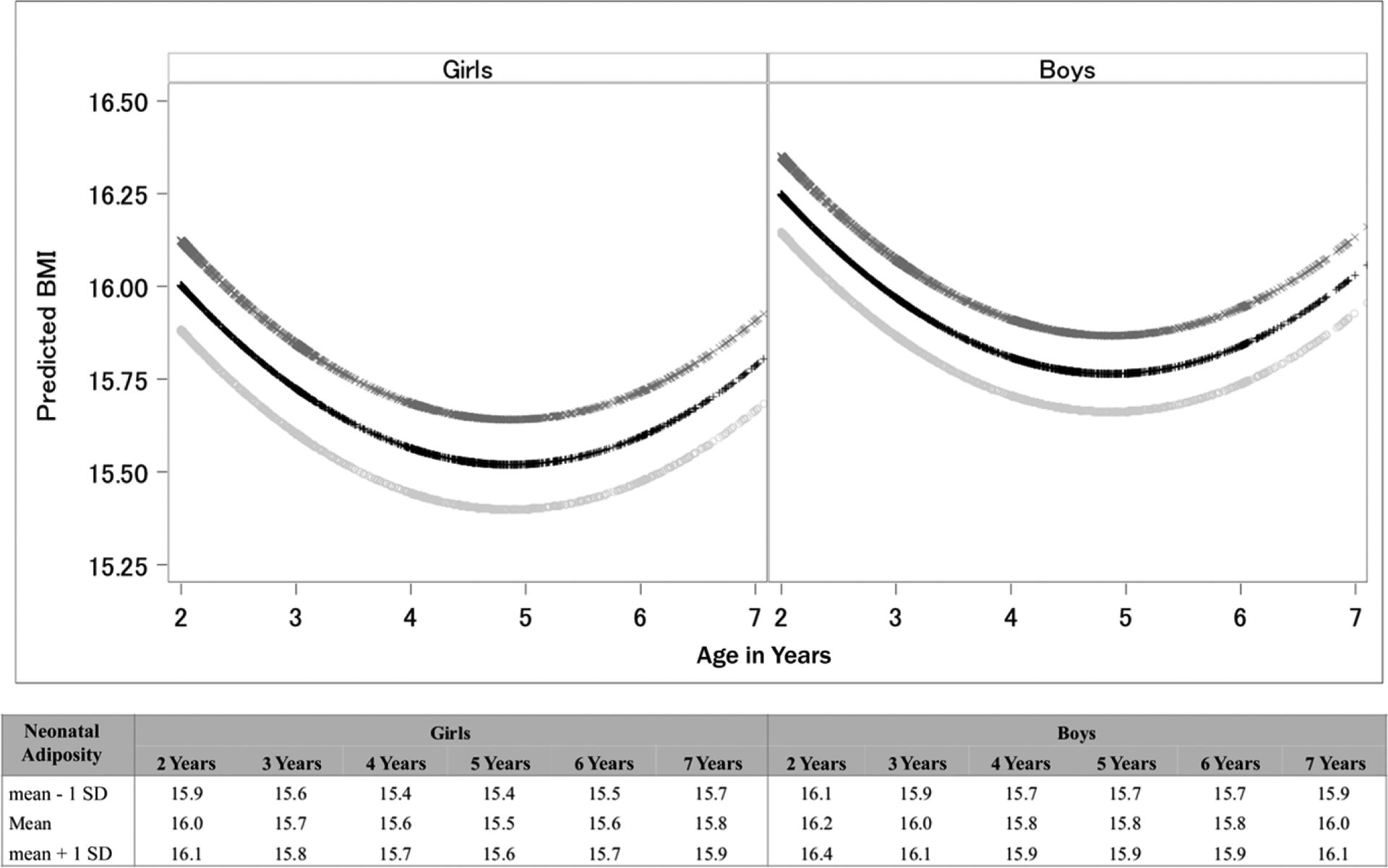
Predicted BMI levels according to neonatal adiposity.
Table 3 presents the proportion of overweight and obesity by levels of neonatal adiposity (mean ± SD; 9.1% ± 4.0%). In general, increased neonatal adiposity was associated with an increase in the proportion of children having overweight or obesity. For instance, at age 2 to 3 years, overweight or obesity was noted in 15% (95% CI: 9% to 21%) of the children born with 5.1% neonatal adiposity (mean − 1 SD), 18% (95% CI: 15% to 21%) of the children born with 9.1% neonatal adiposity, and 23% (95% CI: 15% to 30%) of the children born with 13.1% neonatal adiposity (mean + 1 SD). By age 5 to 6 years, overweight or obesity was observed in only 3% (95% CI: 0% to 8%) of the children born with 5.1% neonatal adiposity, 15% (95% CI: 9% to 21%) of the children born at 9.1% neonatal adiposity and 23% (95% CI: 9% to 39%) of the children born with 13.1% neonatal adiposity.
TABLE 3.
Proportion of Children by Age Who Were Classified as Overweight or Obese According to Neonatal Adiposity
| Neonatal Adiposity | 2–3 y | 3–4 y | 4–5 y | 5–6 y | ||||
|---|---|---|---|---|---|---|---|---|
| n | Percent (95% CI) | n | Percent (95% CI) | n | Percent (95% CI) | n | Percent (95% CI) | |
| 5.1% (mean − 1 SD) | 132 | 15 (9 to 21) | 87 | 13 (6 to 20) | 82 | 10 (3 to 16) | 36 | 3 (0 to 8) |
| 9.1% (mean) | 527 | 18 (15 to 21) | 362 | 19 (15 to 23) | 386 | 17 (13 to 21) | 136 | 15 (9 to 21) |
| 13.1% (mean + 1 SD) | 120 | 23 (15 to 30) | 69 | 22 (12 to 32) | 97 | 14 (7 to 21) | 30 | 23 (9 to 39) |
| P for trend | — | P = .08 | — | P = .08 | — | P = .26 | — | P = .02 |
—, not applicable.
DISCUSSION
Higher neonatal adiposity is associated with higher overall BMI levels and higher prevalence of overweight and obesity. This association was similar in boys and girls and across racial and ethnic groups and did not differ according to breastfeeding duration. These findings are important because various prenatal exposures, such as maternal diet and physical activity during pregnancy, may have specific effects on offspring fat accretion that are not identified by simply measuring birth weight.9,10 Thus, our results suggest that neonatal adiposity is a potentially useful surrogate end point for prenatal trials, designed to reduce future risk of child overweight and obesity. Furthermore, our data may help to identify at-risk children, particularly those born macrosomic or to mothers with diabetes, who could benefit from proven obesity interventions.
Our study is the first to conduct a longitudinal analysis of neonatal adiposity and childhood BMI trajectories. Exploring childhood BMI trajectories is important because single estimates of BMI may not capture complex growth patterns.12 Yet, our study demonstrates a constant relationship between neonatal adiposity and childhood BMI through age 6 years, in which the relationship did not change over time. We observed that each SD increase in neonatal adiposity resulted in a 0.12 higher overall BMI between ages 2 and 6 years. Although this association is modest, a 0.12 increase in BMI represents a 2% to 4% increase in BMI percentile on the basis of the Centers for Disease Control and Prevention BMI-for-age growth charts for boys and girls.21 The implications of our findings are important because even small increases in childhood percentiles could significantly contribute to early systemic inflammation and subsequent cardiometabolic diseases.25
The association between neonatal adiposity and BMI did not vary by subgroups. Although a shorter duration of breastfeeding is associated with rapid BMI growth in early childhood,14 we did not detect an interaction between neonatal adiposity and breastfeeding duration on BMI trajectories. Although there are many reasons for promoting breastfeeding exclusivity for at least 6 months,26 our results do not indicate that breastfeeding duration modifies the association between neonatal adiposity and childhood BMI levels. Similarly, we did not observe that sex or race and/or ethnicity modified this association, despite their individual effects on BMI growth patterns.13
Increased neonatal adiposity was associated with an increased proportion of childhood overweight and obesity across early childhood. These associations tended to be stronger between the ages of 2 to 3 and 5 to 6 years. Conversely, at ages 4 to 5 years, the highest proportion of childhood overweight and obesity was present among children with mean neonatal adiposity levels. It is unclear why the association between neonatal adiposity and childhood overweight and obesity was not sequential at this age. One possible explanation is that growth may have shifted slightly upwards among the mean neonatal adiposity group during BMI rebound (when the BMI trajectory changed direction), which occurred at 4.7 years.
This study had some limitations. The quality of the weight and height measures abstracted from medical records is an important consideration. However, we developed a sophisticated technique for removing statistical anomalies, which likely reduced measurement errors. After removing statistical anomalies, our final data set included height and weight data from 979 participants, with between 1 to 7 pediatric visits per year (mean: 1.7).
A second limitation is the extent to which findings are generalizable to other populations. The mean neonatal adiposity (9.1% ± 4.0%) in our study is comparable to that elsewhere in the United States,27,28 United Kingdom,29 and Australia,30 although slightly higher than what has been reported in an Ethiopian population.31 Furthermore, our cohort was relatively well educated, with a household income exceeding $40 000. Thus, our results may be generalizable but only to other populations with similar distributions of neonatal adiposity, maternal education, household income, race and/or ethnicity, and other early-life factors, such as the duration of exclusive breastfeeding.
Finally, the use of BMI to approximate childhood adiposity is an inherent limitation. BMI is a crude approximation of excess fat mass32 and may contribute to misclassification of overweight or obesity. For instance, large BMI values in childhood may be a result of large amounts of fat-free mass, rather than fat mass.33 Conversely, childhood BMI values within the “normal” range may fail to detect excess fat mass for size, particularly if fat-free mass is low.34 Despite the potential for misclassification, BMI is strongly correlated with adiposity33 and is used to identify children at future risk of adverse cardiometabolic outcomes as well as fat mass.34 Thus, BMI continues to be a useful tool for screening children at risk for excess adiposity,33 which further supports the clinical utility of our results.
CONCLUSIONS
Childhood overweight and obesity is linked to many adverse outcomes, including metabolic syndrome and type 2 diabetes.35 Obesity is primed during prenatal development, beginning as early as midgestation,36 which suggests that pregnancy may be an ideal time to intervene. In the current study, we provide novel evidence that higher neonatal adiposity is associated with a higher BMI and increased likelihood of overweight or obesity from ages 2 to 6 years. Because prenatal factors may have a specific effect on offspring fat accretion,9,10 our results suggest that neonatal adiposity may be a useful surrogate end point for prenatal trials designed to reduce future child overweight and obesity.
Supplementary Material
WHAT’S KNOWN ON THIS SUBJECT:
Higher birth weight is associated with increased childhood BMI levels. However, in no previous studies have researchers examined the longitudinal association between neonatal adiposity (fat mass percentage) and childhood BMI or overweight and obesity status at age 2 to 6 years.
WHAT THIS STUDY ADDS:
Higher neonatal adiposity predicts higher BMI and overweight and obesity in childhood. Because certain prenatal factors have specific effects on neonatal adiposity without influencing birth weight, neonatal adiposity may be a useful surrogate end point for prenatal interventions aimed at reducing childhood overweight and obesity.
FUNDING:
Supported by the National Institutes of Health (R01DK076648, UL1TR001082, and R01GM121081). Funded by the National Institutes of Health (NIH).
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
ABBREVIATIONS
- CI
confidence interval
- WHO
World Health Organization
Footnotes
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
REFERENCES
- 1.Ogden CL, Carroll MD, Lawman HG, et al. Trends in obesity prevalence among children and adolescents in the United States, 1988–1994 through 2013–2014. JAMA. 2016;315(21):2292–2299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Skinner AC, Ravanbakht SN, Skelton JA, Perrin EM, Armstrong SC. Prevalence of obesity and severe obesity in US children, 1999–2016. [published correction appears in Pediatrics. 2018; 142(30):e20181916]. Pediatrics. 2018; 141(3):e20173459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Al-Hamad D, Raman V. Metabolic syndrome in children and adolescents. Transl Pediatr. 2017;6(4):397–407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Oken E. Maternal and child obesity: the causal link. Obstet Gynecol Clin North Am. 2009;36(2):361–377, ix–x [DOI] [PubMed] [Google Scholar]
- 5.Mustila T, Raitanen J, Keskinen P, Luoto R. A pragmatic controlled trial to prevent childhood obesity within a risk group at maternity and child health-care clinics: results up to six years of age (the VACOPP study). BMC Pediatr. 2018;18(1):89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gillman MW, Oakey H, Baghurst PA, Volkmer RE, Robinson JS, Crowther CA. Effect of treatment of gestational diabetes mellitus on obesity in the next generation. Diabetes Care. 2010;33(5): 964–968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tanvig M, Vinter CA, Jørgensen JS, et al. Anthropometrics and body composition by dual energy X-ray in children of obese women: a follow-up of a randomized controlled trial (the Lifestyle in Pregnancy and Offspring [LiPO] study). PLoS One. 2014;9(2): e89590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schellong K, Schulz S, Harder T, Plagemann A. Birth weight and long-term overweight risk: systematic review and a meta-analysis including 643,902 persons from 66 studies and 26 countries globally. PLoS One. 2012;7(10): e47776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Crume TL, Brinton JT, Shapiro A, et al. Maternal dietary intake during pregnancy and offspring body composition: the Healthy Start Study. Am J Obstet Gynecol. 2016;215(5): 609.e1–609.e8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harrod CS, Chasan-Taber L, Reynolds RM, et al. Physical activity in pregnancy and neonatal body composition: the Healthy Start Study. Obstet Gynecol. 2014;124(2, pt 1):257–264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Admassu B, Wells JCK, Girma T, et al. Body composition during early infancy and its relation with body composition at 4 years of age in Jimma, an Ethiopian prospective cohort study. Nutr Diabetes. 2018;8(1):46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wen X, Kleinman K, Gillman MW, Rifas-Shiman SL, Taveras EM. Childhood body mass index trajectories: modeling, characterizing, pairwise correlations and socio-demographic predictors of trajectory characteristics. BMC Med Res Methodol. 2012;12:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Min J, Wen X, Xue H, Wang Y. Ethnic disparities in childhood BMI trajectories and obesity and potential causes among 29,250 US children: findings from the Early Childhood Longitudinal Study-Birth and Kindergarten Cohorts. Int J Obes (Lond). 2018;42(9):1661–1670 [DOI] [PubMed] [Google Scholar]
- 14.Rzehak P, Oddy WH, Mearin ML, et al. ; WP10 working group of the Early Nutrition Project. Infant feeding and growth trajectory patterns in childhood and body composition in young adulthood. Am J Clin Nutr. 2017;106(2): 568–580 [DOI] [PubMed] [Google Scholar]
- 15.Ellis KJ, Yao M, Shypailo RJ, Urlando A, Wong WW, Heird WC. Body-composition assessment in infancy: air-displacement plethysmography compared with a reference 4-compartment model. Am J Clin Nutr. 2007;85(1):90–95 [DOI] [PubMed] [Google Scholar]
- 16.Ma G, Yao M, Liu Y, et al. Validation of a new pediatric air-displacement plethysmograph for assessing body composition in infants. Am J Clin Nutr. 2004;79(4):653–660 [DOI] [PubMed] [Google Scholar]
- 17.Yao M, Nommsen-Rivers L, Dewey K, Urlando A. Preliminary evaluation of a new pediatric air displacement plethysmograph for body composition assessment in infants. Acta Diabetol. 2003;40(suppl 1):S55–S58 [DOI] [PubMed] [Google Scholar]
- 18.World Health Organization. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development. Geneva, Switzerland: World Health Organization; 2006 [Google Scholar]
- 19.Yang H, de Onis M. Algorithms for converting estimates of child malnutrition based on the NCHS reference into estimates based on the WHO Child Growth Standards. BMC Pediatr. 2008;8:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.de Onis M, Onyango AW, Borghi E, Siyam A, Nishida C, Siekmann J. Development of a WHO growth reference for school-aged children and adolescents. Bull World Health Organ. 2007;85(9):660–667 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kuczmarski RJ, Ogden CL, Guo SS, et al. 2000 CDC growth charts for the United States: methods and development. Vital Health Stat 11 2002;(246):1–190 [PubMed] [Google Scholar]
- 22.Royston P, Sauerbrei W. Multivariable Model-Building: A Pragmatic Approach to Regression Analysis Based on Fractional Polynomials for Modelling Continuous Variables. Hoboken, NJ: John Wiley & Sons, Inc.; 2008 [Google Scholar]
- 23.Kenward MG, Roger JH. An improved approximation to the precision of fixed effects from restricted maximum likelihood. Comput Stat Data Anal. 2009; 53(7):2583–2595 [Google Scholar]
- 24.Kenward MG, Roger JH. Small sample inference for fixed effects from restricted maximum likelihood. Biometrics. 1997;53(3):983–997 [PubMed] [Google Scholar]
- 25.Singer K, Eng DS, Lumeng CN, Gebremariam A, Lee JM. The relationship between body fat mass percentiles and inflammation in children. Obesity (Silver Spring). 2014; 22(5):1332–1336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Section on Breastfeeding. Breastfeeding and the use of human milk. Pediatrics. 2012;129(3). Available at: www.pediatrics.org/cgi/content/full/129/3/e827 [DOI] [PubMed] [Google Scholar]
- 27.Josefson JL, Zeiss DM, Rademaker AW, Metzger BE. Maternal leptin predicts adiposity of the neonate. Horm Res Paediatr. 2014;81(1):13–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hull HR, Dinger MK, Knehans AW, Thompson DM, Fields DA. Impact of maternal body mass index on neonate birthweight and body composition. Am J Obstet Gynecol. 2008;198(4):416.e1–6 [DOI] [PubMed] [Google Scholar]
- 29.Crozier SR, Inskip HM, Godfrey KM, et al. ; Southampton Women’s Survey Study Group. Weight gain in pregnancy and childhood body composition: findings from the Southampton Women’s Survey. Am J Clin Nutr. 2010;91(6):1745–1751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Au CP, Raynes-Greenow CH, Turner RM, Carberry AE, Jeffery H. Fetal and maternal factors associated with neonatal adiposity as measured by air displacement plethysmography: a large cross-sectional study. Early Hum Dev. 2013;89(10): 839–843 [DOI] [PubMed] [Google Scholar]
- 31.Andersen GS, Girma T, Wells JCK, Kæstel P, Michaelsen KF, Friis H. Fat and fat-free mass at birth: air displacement plethysmography measurements on 350 Ethiopian newborns. Pediatr Res. 2011;70(5):501–506 [DOI] [PubMed] [Google Scholar]
- 32.Javed A, Jumean M, Murad MH, et al. Diagnostic performance of body mass index to identify obesity as defined by body adiposity in children and adolescents: a systematic review and meta-analysis. Pediatr Obes. 2015;10(3): 234–244 [DOI] [PubMed] [Google Scholar]
- 33.Maynard LM, Wisemandle W, Roche AF, Chumlea WC, Guo SS, Siervogel RM. Childhood body composition in relation to body mass index. Pediatrics. 2001; 107(2):344–350 [DOI] [PubMed] [Google Scholar]
- 34.Lawlor DA, Benfield L, Logue J, et al. Association between general and central adiposity in childhood, and change in these, with cardiovascular risk factors in adolescence: prospective cohort study. BMJ. 2010;341:c6224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Biro FM, Wien M. Childhood obesity and adult morbidities. Am J Clin Nutr. 2010; 91(5):1499S–1505S [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Symonds ME, Pope M, Sharkey D, Budge H. Adipose tissue and fetal programming. Diabetologia. 2012;55(6): 1597–1606 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


