Abstract
Background
COVID-19 has affected millions of people, and several chronic medical conditions appear to increase the risk of severe COVID-19. However, our understanding of COVID-19 outcomes in patients with CKD remains limited.
Methods
This was a retrospective cohort study of patients with and without CKD consecutively admitted with COVID-19 to three affiliated hospitals in New York City. Pre-COVID-19 CKD diagnoses were identified by billing codes and verified by manual chart review. In-hospital mortality was compared between patients with and without underlying CKD. Logistic regression was used to adjust this analysis for confounders and to identify patient characteristics associated with mortality.
Results
We identified 280 patients with CKD, and 4098 patients without CKD hospitalized with COVID-19. The median age of the CKD group was 75 (65–84) years, and age of the non-CKD group 62 (48–75) years. Baseline (pre-COVID-19) serum creatinine in patients with CKD was 1.5 (1.2–2.2) mg/dl. In-hospital mortality was 30% in patients with CKD versus 20% in patients without CKD (P<0.001). The risk of in-hospital death in patients with CKD remained higher than in patients without CKD after adjustment for comorbidities (hypertension, diabetes mellitus, asthma, and chronic obstructive pulmonary disease), adjusted OR 1.4 (95% CI,1.1 to 1.9), P=0.01. When stratified by age, elderly patients with CKD (age >70 years) had higher mortality than their age-matched control patients without CKD. In patients with CKD, factors associated with in-hospital mortality were age (adjusted OR, 1.09 [95% CI, 1.06 to 1.12]), P<0.001, baseline and admission serum phosphorus (adjusted OR, 1.5 [95% CI, 1.03 to 2.1], P=0.03 and 1.4 [95% CI, 1.1 to 1.7], P=0.001), serum creatinine on admission >0.3 mg/dl above the baseline (adjusted OR 2.6 [95% CI, 1.2 to 5.4]P=0.01), and diagnosis of acute on chronic kidney injury during hospitalization (adjusted OR 4.6 [95% CI, 2.3 to 8.9], P<0.001).
Conclusions
CKD is an independent risk factor for COVID-19-associated in-hospital mortality in elderly patients. Acute-on-chronic kidney injury increases the odds of in-hospital mortality in patients with CKD hospitalized with COVID-19.
Introduction
The COVID-19 global pandemic has affected millions of people. In spring 2020, New York City (NYC) was the epicenter of the COVID-19 pandemic in the United States. Several chronic medical conditions, such as hypertension, diabetes, and obesity, have been reported as risk factors for severe COVID-19. CKD affects approximately 15% of the adult US population (1). It is associated with immune activation, marked by systemic inflammation, and immune deficiency, manifesting as increased susceptibility to infections (2–4). Patients with CKD have higher mortality from non–COVID-19 associated pneumonia compared with patients with preserved renal function (5). The US Centers for Disease Control and Prevention warns that having CKD of any stage increases the risk for severe illness from COVID-19 (6). However, studies of COVID-19 outcomes in patients with predialysis CKD remain scarce (7–9). Herein, we characterize a cohort of patients with CKD and a control cohort of patients without CKD who were hospitalized with COVID-19 in spring and summer 2020 in NYC.
Materials and Methods
This was a retrospective cohort study that was conducted at a major academic center and three affiliated hospitals in NYC. The study was approved by the Institutional Review Board of Weill Cornell Medicine. Patients with a COVID-19 diagnosis (by reverse-transcriptase PCR of nasopharyngeal swab specimens) that required in-patient admission were included. Patients who were diagnosed with COVID-19 in the emergency department but did not require admission were not included. We included all hospitalized patients with COVID-19 who were discharged by August 31, 2020. Data were abstracted electronically using an institutional reporting database (10). The cohort was screened for patients with CKD using the International Statistical Classification of Diseases tenth revision (ICD-10) diagnostic code of CKD (N18) in electronic medical records at any point since 2015 pre-COVID-19 diagnosis. CKD status was validated by manual chart review. Health care provider-recorded diagnosis of CKD either in prior medical records or in the emergency department was used for validation. Comorbid conditions were identified by their ICD-10 codes.
Statistical Analyses
Continuous variables are presented as median (interquartile range). A t test was used to determine the significance of differences between normally distributed continuous variables and a chi-squared test was used for the proportions. Covariates included in the multivariate analysis of the effect of CKD on mortality included sex, race and ethnicity, and the presence of comorbidities—hyper-tension, diabetes, asthma, and chronic obstructive pulmonary disease. To determine risk factors for in-hospital death in patients with COVID-19 and underlying CKD, we used logistic regression. Adjusted odds ratios (ORs) with 95% confidence intervals (95% CIs) were calculated for variables significant in univariate analysis; adjustment covariates included age, sex, race, and ethnicity. We used STATA for statistical analyses.
Results
Our two cohorts included 280 patients with CKD and 4098 patients without a pre-existing diagnosis of CKD (Table 1). The CKD group was older than the non-CKD group (median age 75 versus 62 years) and had higher percentage of males (63% versus 55%). The percentage of Black patients was higher in the CKD group than in non-CKD group (19% versus 10%), indicating disproportionate vulnerability of Black patients to COVID-19, consistent with previous reports (9). Comorbid conditions included hypertension, diabetes, asthma, chronic obstructive pulmonary disease, and congestive heart failure; all of these conditions were more common in patients with pre-existing CKD than in patients without CKD.
Table 1.
Demographic and clinic characteristics of hospitalized patients with COVID-19, with and without underlying CKD
| Patient characteristics | No CKD (n=4098) | CKD (n=280) | P |
|---|---|---|---|
| Age, yr, median (interquartile range) | 62 (48–75) | 75 (65–84) | <0.001 |
| Male sex, n (%) | 2244 (55) | 176 (63) | 0.009 |
| Borough of residence, n (%) | |||
| Manhattan | 742 (18) | 100 (36) | <0.001 |
| Queens | 2657 (65) | 111 (40) | <0.001 |
| Brooklyn | 454 (11) | 41 (15) | 0.06 |
| The Bronx | 154 (4) | 19 (7) | 0.01 |
| Race and ethnicity, n (%) | |||
| White | 1099 (27) | 87 (31) | 0.11 |
| Black | 415 (10) | 52 (19) | <0.001 |
| Asian | 635 (16) | 42 (15) | 0.83 |
| Hispanic | 1292 (32) | 58 (21) | <0.001 |
| Comorbidities, n (%) | |||
| Hypertension | 1684 (41) | 232 (83) | <0.001 |
| Diabetes mellitus | 1052 (26) | 155 (55) | <0.001 |
| Congestive heart failure | 339 (8) | 109 (39) | <0.001 |
| Asthma | 815 (8) | 38 (14) | 0.003 |
| COPD | 233 (3) | 44 (16) | <0.001 |
| Baseline laboratory characteristics, median (interquartile range) | |||
| Serum creatinine, mg/dl | 0.8 (0.7–1.1) | 1.5 (1.2–2.2) | <0.001 |
| BUN, mg/dl | 16 (12.5–21.2) | 31 (20.2–44) | <0.001 |
| Hemoglobin, g/dl | 12.1 (10.7–13.5) | 11.6 (9.8–13.1) | <0.001 |
| Outcomes, n (%) | |||
| Mechanical ventilation | 329 (8) | 37 (13) | 0.002 |
| Septic shock | 169 (4) | 27 (10) | <0.001 |
| AKI | 892 (22) | 142 (54) | <0.001 |
| In-hospital mortality | 815 (20) | 84 (30) | <0.001 |
COPD, chronic obstructive pulmonary disease.
Patients with CKD required mechanical ventilation more frequently than patients without CKD (Table 1). Patients with CKD were also more frequently diagnosed with septic shock. AKI was diagnosed in 54% of patients with CKD versus in 22% of those without underlying CKD. In-hospital mortality was 30% in patients with CKD and 20% in patients without CKD (unadjusted P<0.001) (Figure 1A, Table 1). After adjustment for demographic characteristics and comorbid conditions (Supplemental Table 1), CKD remained a significant risk factor for in-hospital mortality (adjusted OR, 1.4 [95% CI, 1.1 to 1.9]). Importantly, when both CKD and non-CKD cohorts were stratified by age, patients aged >70 years had higher mortality than their age-matched counterparts without CKD (Figure 1B).
Figure 1. |. Elderly patients with CKD had higher COVID-19-associated in-hospital mortality than their counterparts without CKD.
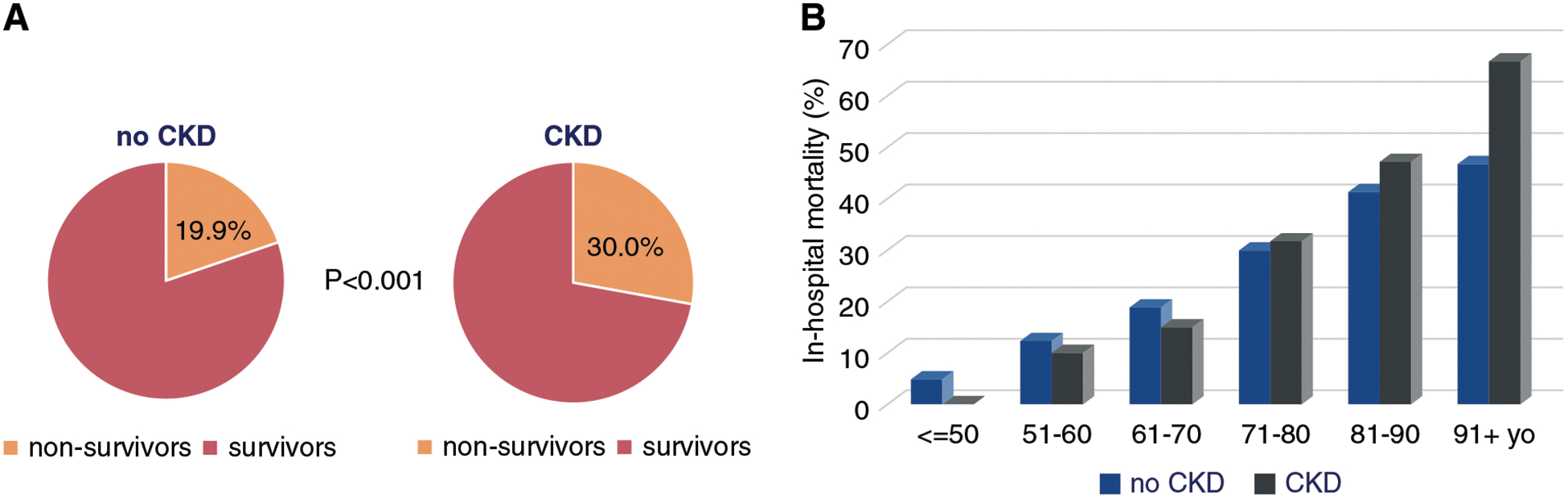
In-hospital mortality in patients with COVID-19 with and without CKD, overall (A) and stratified by age (B).
Within the CKD cohort, the subgroup of patients who died in the hospital was older than the subgroup of those who survived (Figure 2, Table 2). As expected, higher in-hospital mortality was observed with higher CKD stages (Supplemental Figure 1). In univariate analysis, other factors associated with increased odds of mortality were: having primary language other than English (OR, 1.9 [95% CI, 1.1 to 3.1]), body mass index (BMI) <25 kg/m2 (OR, 1.8 [95% CI, 1.1 to 3.0]), pre-COVID-19 BUN (OR, 1.03 [95% CI, 1.01 to 1.05]), serum phosphorus on admission (OR, 1.3 [95% CI, 1.1 to 1.5]), C-reactive protein (OR, 1.1 [95% CI, 1.05 to 1.2]), elevated serum creatinine on admission (OR, 2.9 [95% CI, 1.6 to 5.6]), and diagnosis of AKI during hospitalization (OR, 3.9 [95% CI, 2.2 to 7.1]). Baseline use of angiotensin receptor blockers and higher lymphocyte count on admission were associated with increased odds of survival (OR, 0.5 [95% CI, 0.3 to 0.9] and 0.5 [95% CI, 0.2 to 0.9] respectively). After adjustment for demographic characteristics (Figure 2), variables that remained significantly associated with mortality were age (OR, 1.09 [95% CI, 1.06 to 1.12]), admission serum phosphorus (OR, 1.1 [95% CI, 1.4 to 1.7]), admission C-reactive protein (OR, 1.05 [95% CI, 1.02 to 1.11]), admission serum creatinine above baseline (OR, 2.6 [95% CI, 1.2 to 5.4]), and diagnosis of AKI during hospitalization (OR, 4.6 [95% CI, 2.3 to 8.9]). In addition, in multivariate analysis baseline serum phosphorus was significantly associated with mortality (adjusted OR, 1.5 [95% CI, 1.03 to 2.1]).
Figure 2. |. Analysis of factors associated with in-hospital mortality in COVID-19 patients with CKD.
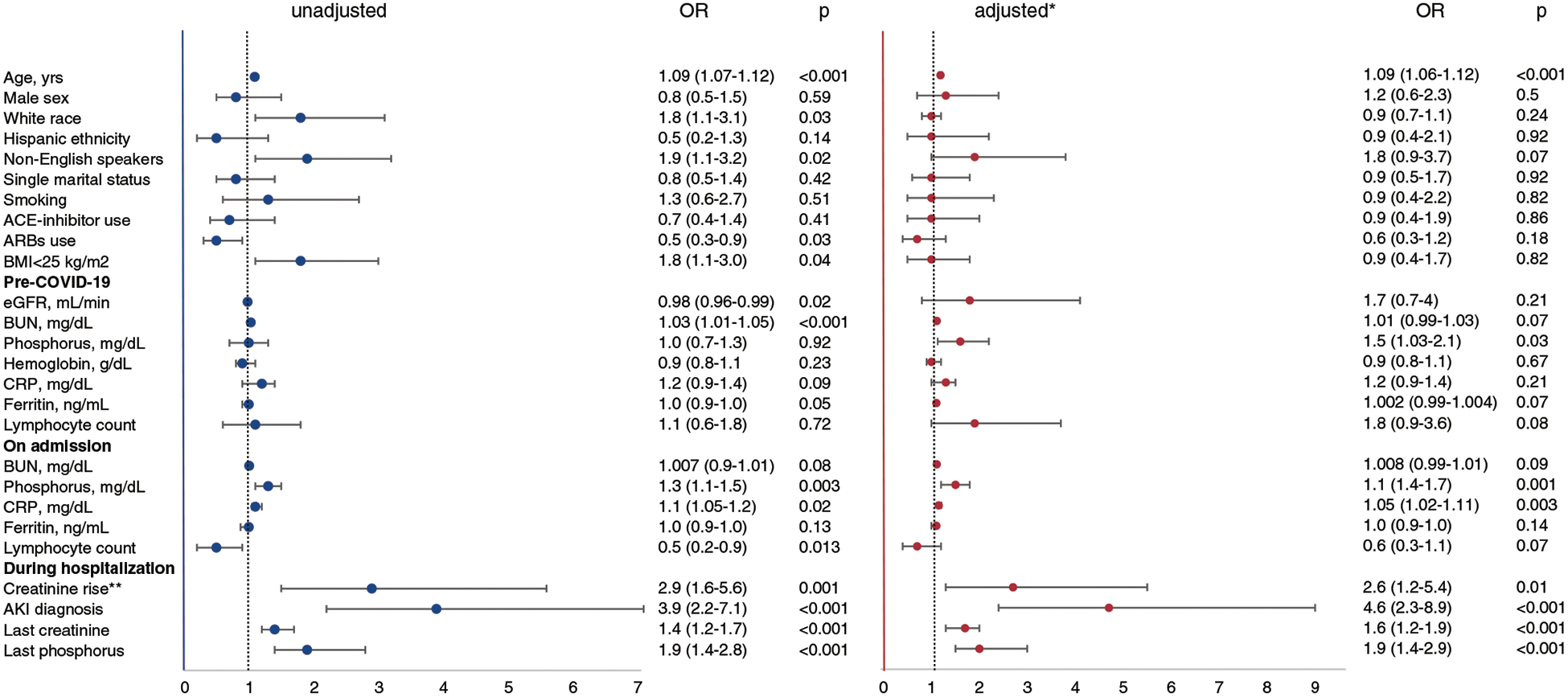
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; CRP, C-reactive protein. *Adjusted by age, sex, race, and ethnicity. **Creatinine rise was defined as admission serum creatinine >0.3 mg/dl above the baseline (pre-COVID-19) serum creatinine.
Table 2.
Demographic and clinical characteristics of hospitalized COVID-19 patients with underlying CKD
| Patient characteristics | All patients (n=280) | Survivors (n=196) | Nonsurvivors (n=84) | P |
|---|---|---|---|---|
| Age, yr, median (interquartile range) | 75 (65–84) | 70 (59–80) | 86 (76–90) | <0.001 |
| Male sex, n (%) | 176 (63) | 125 (64) | 51 (61) | 0.59 |
| Borough of residence: 0.187, n (%) | ||||
| Manhattan | 100 (36) | 73 (37) | 27 (32) | 0.39 |
| Queens | 111 (40) | 69 (35) | 42 (50) | 0.02 |
| Brooklyn | 41 (15) | 31 (16) | 10 (12) | 0.39 |
| The Bronx | 19 (7) | 15 (8) | 4 (5) | 0.37 |
| Race and ethnicity: 0.036, n (%) | ||||
| White | 87 (31) | 53 (27) | 34 (41) | 0.02 |
| Black | 52 (19) | 40 (20) | 12 (14) | 0.22 |
| Asian | 42 (15) | 25 (13) | 17 (20) | 0.11 |
| Hispanic | 58 (21) | 45 (23) | 13 (16) | 0.15 |
| Maintenance medications (pre-COVID-19), n (%) | ||||
| ACE-inhibitors | 62 (22) | 46 (24) | 16 (19) | 0.41 |
| Angiotensin receptor blockers | 93 (33) | 73 (37) | 20 (24) | 0.02 |
| Diuretics | 118 (42) | 82 (42) | 36 (43) | 0.87 |
| Sevelamer | 23 (8) | 15 (8) | 8 (10) | 0.61 |
| Calcitriol | 28 (10) | 21 (11) | 7 (8) | 0.53 |
| Antihyperlipidemic agents | 186 (66) | 133 (68) | 53 (63) | 0.43 |
| Comorbidities, n (%) | ||||
| Hypertension | 232 (83) | 162 (83) | 70 (83) | 0.89 |
| Diabetes mellitus | 155 (55) | 112 (57) | 43 (51) | 0.36 |
| Congestive heart failure | 109 (39) | 72 (37) | 37 (44) | 0.25 |
| Asthma | 38 (14) | 29 (15) | 9 (11) | 0.36 |
| COPD | 44 (16) | 31 (16) | 13 (15) | 0.94 |
| BMI, kg/m2, median (interquartile range) | 25.7 (21.7–29.2) | 26 (22.4–30.5) | 24.2 (21.3–29.2) | 0.40 |
| Baseline (pre–COVID-19) laboratory characteristics, median (interquartile range) | ||||
| Creatinine, mg/dl, n=192 | 1.5 (1.2–2.2) | 1.4 (1.1–2.0) | 1.6 (1.3–2.7) | 0.21 |
| BUN, mg/dl, n = 189 | 31 (21–44) | 29 (19–42) | 35 (25–53) | 0.01 |
| eGFR (ml/min per 1.73 m2), n=192 | 44 (27–59) | 46 (29–60) | 40.5 (23–52) | 0.02 |
| Hemoglobin, g/dl, n=173 | 11.6 (9.8–13.1) | 11.8 (10–13.2) | 11.3 (9.4–12.5) | 0.23 |
| Ferritin, ng/ml, n=64 | 160.5 (60–357.5) | 104.1 (52.8–349) | 262.9 (139–473) | 0.02 |
| Phosphorus, mg/dl, n=188 | 3.6 (3.2–4.2) | 3.6 (3.2–4.1) | 3.7 (3.2–4.3) | 0.91 |
| PTH, pg/ml, n=49 | 81.9 (57.8–120.5) | 80.8 (60.1–96.7) | 92.1 (46.2–197.9) | 0.46 |
| CRP, mg/dl, n=74 | 3.6 (0.8–4.8) | 2.2 (0.6–4.6) | 4.2 (2.4–5.6) | 0.02 |
| Lymphocyte count, ×103 mm−3, n=173 | 1.2 (0.9–1.7) | 1.2 (0.9–1.7) | 1.2 (0.8–1.8) | 0.72 |
| Laboratory characteristics on admission | ||||
| Creatinine, mg/dl, n=268, median (interquartile range) | 1.98 (1.4–3.2) | 1.9 (1.3–3.1) | 2.1 (1.6–3.4) | 0.42 |
| Creatinine >0.3 mg/dl from baseline, n=185 (%) | 84 (45.4) | 45 (36.8) | 38 (63.3) | 0.001 |
| BUN, mg/dl, n=228, median (interquartile range) | 43 (27–69) | 38 (24–67) | 46 (32–74) | 0.07 |
| Hemoglobin, g/dl, n=270, median (interquartile range) | 11.8 (9.8–13.6) | 11.9 (9.8–13.7) | 11.6 (9.5–13.2) | 0.29 |
| Ferritin, ng/ml, n=185, median (interquartile range) | 688.9 (351.2–1318.0) | 688.9 (328.5–1258.0) | 695 (437.4–1392.0) | 0.10 |
| Phosphorus, mg/dl, n=188, median (interquartile range) | 3.85 (3.3–5) | 3.75 (3.3–4.8) | 4.5 (3.3–7) | 0.0015 |
| CRP, mg/dl, n=181, median (interquartile range) | 10.4 (4.9–19.3) | 10.2 (4.6–16.5) | 11.7 (5.8–23.7) | 0.02 |
| Lymphocyte count, ×103 mm−3, n=248, median (interquartile range) | 0.8 (0.5–1.2) | 0.9 (0.6–1.3) | 0.7 (0.5–1.0) | 0.01 |
| Laboratory characteristics at discharge, median (interquartile range) | ||||
| Creatinine, mg/dl, n=153 | 1.8 (1.2–3.0) | 1.4 (1.1–2.4) | 3.0 (1.8–4.7) | <0.001 |
| Phosphorus, mg/dl, n=85 | 4.1 (3.1–5.2) | 3.7 (3.1–4.6) | 6.4 (4.2–9.6) | <0.001 |
ACE, angiotensin-converting enzyme; COPD, chronic obstructive pulmonary disease; BMI, body mass index; PTH, parathyroid hormone.
Discussion
Identification and stratification of risk factors for severe COVID-19 disease are important for the development of effective preventative strategies and interventions. Although emerging data support the role of hypertension, diabetes, and coronary artery disease as risk factors for severe COVID-19 (11), the significance of CKD as an underlying condition for severe COVID-19 remains less well understood. In a meta-analysis of early reports from China, no study individually found CKD as significant predictor of severe COVID-19. However, when data of individual studies were pooled, a significant association between CKD and severe COVID-19 was observed (OR, 3.03 [95% CI, 1.09 to 8.47]) (7). In a cohort of 5700 patients from the NYC area hospitalized with COVID-19, the reported prevalence of CKD was 5% and ESKD 3.5% on the basis of the available ICD-10 diagnostic codes in medical history (12). COVID-19 in patients on dialysis received close attention, mainly from the standpoint of developing preventative guidelines, given frequent aggregation of these patients in hemodialysis units (13). However, data on the outcomes of COVID-19 in patients who are predialysis with CKD remain scarce (8). In a recent multicenter study of patients admitted to the intensive care units across the United States, the presence of pre-existing kidney failure treated by maintenance dialysis was strongly associated with in-hospital death, whereas pre-existing nondialysis CKD had an intermediate association, compared with no pre-existing CKD (9).
In this study, we reported the role of CKD in COVID-19-associated in-hospital mortality on the basis of the cohort of 4378 patients hospitalized with COVID-19, 280 of whom carried the diagnosis of CKD before the COVID-19 pandemic. Patients with CKD had approximately 50% higher in-hospital mortality than patients without CKD. In a cohort of 1603 patients admitted with COVID-19 in Spain, underlying CKD was a risk factor for in-hospital death with a hazard ratio 1.59 (1.06–2.37) (14). In a multicenter study of 4264 patients with COVID-19 admitted to the intensive care units in the United States, CKD had a slightly lower risk of in-hospital death, hazard ratio 1.25 (1.08–1.44) (9). Although in both studies associations were adjusted for age, a stratification of mortality by age was not reported. It has been well established that COVID-19 disproportionately affects the elderly within the general population, whereas the role of age in patients with CKD and COVID-19 remained less well understood. In our cohort, the overall difference in mortality between CKD and non-CKD cohorts was most pronounced in elderly patients. In the subgroup of patients younger than 70, we did not observe differences in mortality between patients with and without underlying CKD. Our results highlight that CKD may be a particularly significant risk factor for mortality in elderly patients with COVID-19 and warrant further analysis of the role of CKD as risk factor for adverse outcomes in COVID-19 separately in the younger, middle age, and elderly population.
CKD-specific patient characteristics that may be responsible for adverse outcomes of COVID-19 have received little attention to date. In addition to advanced age, several CKD complications were associated with in-hospital death in patients with CKD in our cohort. It has been well established that obesity is a risk factor for adverse outcomes of COVID-19 (15,16). In our univariate analysis, BMI appeared to follow the reverse epidemiology in patients with CKD. Indeed, patients with a BMI <25 kg/m2 were at higher risk for mortality. This phenomenon has been described for all-cause mortality in patients with CKD (17). Our findings warrant further investigation of nutritional parameters in patients with CKD and COVID-19 and their role in outcomes.
Although most patients in our CKD cohort did not have severe hyperphosphatemia, pre-COVID-19 and admission serum phosphorus were independently associated with in-hospital death in patients with CKD. Distorted phosphorus homeostasis, a hallmark of CKD-mineral and bone disorder, has profound vascular effects in patients with CKD (18). Severe COVID-19 frequently leads to endothelial injury (19). In future studies, it would be of interest to clarify if vascular component of CKD mineral and bone disorder predisposes patients with CKD to more severe vascular injury from COVID-19.
AKI is a common complication of COVID-19 in hospitalized patients (20). It was suggested that early evaluation of renal reserve in the course of COVID-19 may inform therapeutic interventions (21). In patients with CKD, episodes of acute on chronic kidney injury have been characterized as novel risk factors for disease progression (22). At the same time, the role of acute on chronic kidney injury in outcomes of COVID-19 in patients with CKD has not been fully elucidated. In our cohort of patients hospitalized with COVID-19, patients with CKD were diagnosed with AKI 2.5 times more frequently than patients without CKD. Patients with CKD who died in the hospital had AKI more frequently than those patients with CKD who survived. The diagnosis of AKI during hospitalization was the strongest predictor of in-hospital death in patients with CKD and COVID-19 among the potential risk factors that we analyzed (adjusted OR, 4.6 [95% CI, 2.3 to 8.9]). In future studies, it is important to investigate the mechanisms of acute on chronic kidney injury in patients with COVID-19 and underlying CKD, and to test the effectiveness of preventative measures (e.g., early and aggressive fluid resuscitation in patients without oliguria) in improving COVID-19 outcomes in patients with CKD.
The strengths of our study included analysis of a large single-center cohort of consecutively hospitalized patients with COVID-19 who had underlying CKD, and comparing the outcomes with a control group of patients without CKD. Our study has limitations. Baseline laboratory data were not available for all patients. Our analysis was focused on in-hospital mortality and we did not follow patients after discharge. Electronic data abstraction may not have provided the degree of granularity that can be achieved in a fully manual chart review. Although we presented the largest single-center cohort of patients with COVID-19 and CKD to date, an even larger sample size would be required to render more precise estimates of the effects that baseline CKD-specific patient characteristics may have on outcomes.
In conclusion, our findings indicate predialysis CKD is an independent risk factor for in-hospital death in elderly patients with COVID-19. Ongoing control of CKD complications, and early and aggressive measures to prevent the development of acute-on-chronic kidney injury may serve as opportunities to improve the outcomes of COVID-19 in elderly patients with CKD. Further evaluation of the role that CKD may play in COVID-19 outcomes in different age groups is warranted.
Supplementary Material
Supplemental Table 1. Role of CKD in COVID-19–associated in-hospital mortality: multivariate analysis.
Supplemental Figure 1. In hospital mortality in patients with COVID-19 with pre-existing CKD by CKD stages.
Acknowledgments
We thank the staff at Weill Cornell Medicine and affiliated New York Presbyterian hospitals for their care of patients with COVID-19. Weill Cornell Medicine Institutional COVID-19 research data repository received support from the New York Presbyterian Hospital and Weill Cornell Medical College, including the Clinical and Translational Science Center (UL1 TR000457) and Joint Clinical Trials Office. We greatly appreciate the assistance of Sajjad Abedian, Weill Cornell Medicine research informatics analyst, and the help of Weill Cornell medical students, Heba Shaaban and Rachel Abramson with reviewing medical records. This work was partially presented at the 2020 American Society of Nephrology virtual Kidney Week meeting.
Funding
O. Akchurin reported receiving research support from NIH National Institute of Diabetes and Digestive and Kidney Diseases grant K08-DK114558 and is a recipient of the Rohr Family Clinical Scholar Award (Weill Cornell Medicine). M. Choi reports receiving research support from NIH grants R01 HL133801, R01 HL055330, and R01 HL132198. This work was supported by the Weill Cornell Medical College Clinical and Translational Science Center (funded through NIH NCATS grant UL1 TR000457).
Disclosures
J. Choi reports receiving research support from National Institutes of Health (NIH), National Center for Advancing Translational Sciences (NCATS) grant KL2-TR-002385, Roche Diagnostics, and Allergan, outside the submitted work. The spouse of M. Choi is a cofounder and shareholder, and serves on the Scientific Advisory Board of Proterris, Inc. All remaining authors have nothing to disclose.
Footnotes
Supplemental Material
This article contains the following supplemental material online at http://kidney360.asnjournals.org/lookup/suppl/doi:10.34067/KID.0004142020/-/DCSupplemental.
References
- 1.United States Renal Data System, 2019. USRDS Annual Data Report: Epidemiology of Kidney Disease in the United States. Available at https://www.usrds.org/media/2371/2019-executive-summary.pdf. Accessed September 20, 2020 [Google Scholar]
- 2.Kurts C, Panzer U, Anders H-J, Rees AJ: The immune system and kidney disease: Basic concepts and clinical implications. Nat Rev Immunol 13: 738–753, 2013. 10.1038/nri3523 [DOI] [PubMed] [Google Scholar]
- 3.Pahl MV, Vaziri ND: Immune function in chronic kidney disease In: Chronic Renal Disease, Elsevier, 2020, pp 503–519 [Google Scholar]
- 4.Akchurin OM, Kaskel F: Update on inflammation in chronic kidney disease. Blood Purif 39: 84–92, 2015. 10.1159/000368940 [DOI] [PubMed] [Google Scholar]
- 5.James MT, Quan H, Tonelli M, Manns BJ, Faris P, Laupland KB, Hemmelgarn BR; Alberta Kidney Disease Network: CKD and risk of hospitalization and death with pneumonia. Am J Kidney Dis 54: 24–32, 2009. 10.1053/j.ajkd.2009.04.005 [DOI] [PubMed] [Google Scholar]
- 6.Centres for Disease Control and Prevention: Coronavirus (COVID-19), 2020. Available at: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html#chronic-kidney-disease
- 7.Henry BM, Lippi G: Chronic kidney disease is associated with severe coronavirus disease 2019 (COVID-19) infection. Int Urol Nephrol 52: 1193–1194, 2020. 10.1007/s11255-020-02451-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Alberici F, Delbarba E, Manenti C, Econimo L, Valerio F, Pola A, Maffei C, Possenti S, Piva S, Latronico N, Focà E, Castelli F, Gaggia P, Movilli E, Bove S, Malberti F, Farina M, Bracchi M, Costantino EM, Bossini N, Gaggiotti M, Scolari F; Brescia Renal COVID Task Force: Management of patients on dialysis and with kidney transplantation during SARS-COV-2 (COVID-19) pandemic in Brescia, Italy. Kidney Int Rep 5: 580–585, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Flythe JE, Assimon MM, Tugman MJ, Chang EH, Gupta S, Shah J, Sosa MA, Renaghan AD, Melamed ML, Wilson FP, Neyra JA, Rashidi A, Boyle SM, Anand S, Christov M, Thomas LF, Edmonston D, Leaf DE; STOP-COVID Investigators: Characteristics and outcomes of individuals with pre-existing kidney disease and COVID-19 admitted to intensive care units in the United States [published online ahead of print September 19, 2020]. Am J Kidney Dis 10.1053/j.ajkd.2020.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sholle ET, Kabariti J, Johnson SB, Leonard JP, Pathak J, Varughese VI, Cole CL, Campion TR Jr: Secondary use of patients’ electronic records (super): An approach for meeting specific data needs of clinical and translational researchers. AMIA Annu Symp Proc 2017: 1581–1588, 2017 [PMC free article] [PubMed] [Google Scholar]
- 11.CDC COVID-19 Response Team: Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019-United States, February 12-March 28, 2020. MMWR Morb Mortal Wkly Rep 69: 382–386, 2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, Barnaby DP, Becker LB, Chelico JD, Cohen SL, Cookingham J, Coppa K, Diefenbach MA, Dominello AJ, Duer-Hefele J, Falzon L, Gitlin J, Hajizadeh N, Harvin TG, Hirschwerk DA, Kim EJ, Kozel ZM, Marrast LM, Mogavero JN, Osorio GA, Qiu M, Zanos TP; the Northwell COVID-19 Research Consortium: Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area [published correction appears in JAMA 323: 2098, 2020 10.1001/jama.2020.7681]. JAMA 323: 2052–2059, 2020. 10.1001/jama.2020.6775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kliger AS, Cozzolino M, Jha V, Harbert G, Ikizler TA: Managing the COVID-19 pandemic: International comparisons in dialysis patients. Kidney Int 98: 12–16, 2020. 10.1016/j.kint.2020.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Portolés J, Marques M, López-Sánchez P, de Valdenebro M, Muñez E, Serrano ML, Malo R, García E, Cuervas V: Chronic kidney disease and acute kidney injury in the COVID-19 Spanish outbreak. Nephrol Dial Transplant 35: 1353–1361, 2020. 10.1093/ndt/gfaa189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, Satlin MJ, Campion TR Jr, Nahid M, Ringel JB, Hoffman KL, Alshak MN, Li HA, Wehmeyer GT, Rajan M, Reshetnyak E, Hupert N, Horn EM, Martinez FJ, Gulick RM, Safford MM: Clinical characteristics of covid-19 in New York city. N Engl J Med 382: 2372–2374, 2020. 10.1056/NEJMc2010419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sattar N, McInnes IB, McMurray JJV: Obesity is a risk factor for severe COVID-19 infection: Multiple potential mechanisms. Circulation 142: 4–6, 2020. 10.1161/CIRCULATIONAHA.120.047659 [DOI] [PubMed] [Google Scholar]
- 17.Kalantar-Zadeh K, Kovesdy CP, Derose SF, Horwich TB, Fonarow GC: Racial and survival paradoxes in chronic kidney disease. Nat Clin Pract Nephrol 3: 493–506, 2007. 10.1038/ncpneph0570 [DOI] [PubMed] [Google Scholar]
- 18.Voelkl J, Lang F, Eckardt K-U, Amann K, Kuro-O M, Pasch A, Pieske B, Alesutan I: Signaling pathways involved in vascular smooth muscle cell calcification during hyperphosphatemia. Cell Mol Life Sci 76: 2077–2091, 2019. 10.1007/s00018-019-03054-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, Ruschitzka F, Moch H: Endothelial cell infection and endotheliitis in COVID-19. Lancet 395: 1417–1418, 2020. 10.1016/S0140-6736(20)30937-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee JR, Silberzweig J, Akchurin O, Choi ME, Srivatana V, Lin J, Liu F, Malha L, Lubetzky M, Dadhania DM, Shankaranarayanan D, Shimonov D, Neupane S, Salinas T, Bhasin A, Varma E, Leuprecht L, Gerardine S, Lamba P, Goyal P, Caliendo E, Tiase V, Sharma R, Park JC, Steel PAD, Suthanthiran M, Zhang Y: Characteristics of acute kidney injury in hospitalized COVID-19 patients in an urban academic medical center [published online ahead of print September 18, 2020]. Clin J Am Soc Nephrol 10.2215/CJN.07440520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cantaluppi V, Guglielmetti G, Dellepiane S, Marengo M, Mehta RL, Ronco C: A call to action to evaluate renal functional reserve in patients with COVID-19. Am J Physiol Renal Physiol 319: F792–F795, 2020. 10.1152/ajprenal.00245.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hannan M, Ansari S, Meza N, Anderson AH, Srivastava A, Waikar S, Charleston J, Weir MR, Taliercio J, Horwitz E, Saunders MR, Wolfrum K, Feldman HI, Lash JP, Ricardo AC; CRIC Study Investigators; Chronic Renal Insufficiency Cohort (CRIC) Study Investigators: Risk factors for CKD progression: Overview of findings from the CRIC study [published online ahead of print November 11, 2020]. Clin J Am Soc Nephrol 10.2215/CJN.07830520 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Table 1. Role of CKD in COVID-19–associated in-hospital mortality: multivariate analysis.
Supplemental Figure 1. In hospital mortality in patients with COVID-19 with pre-existing CKD by CKD stages.


