Abstract
We reviewed response to immune checkpoint inhibitors (ICI) of 207 patients with diagnoses of lung or head and neck cancer treated with chemotherapy/ICI combination therapy and ICI monotherapy between 2015 and 2020 at one of three clinical pavilions associated with the Dan L. Duncan Comprehensive Cancer Center at Baylor College of Medicine. Two of these pavilions (Harris Health System and the Michael E. DeBakey Veterans Affairs Medical Center) serve large minority populations and provide equal access to care regardless of means. 174 patients had a diagnosis of lung cancer (non-small cell or small cell) and 33 had a diagnosis of head and neck squamous cell carcinoma (HNSCC). 38% self-identified as Black, 45% as non-Hispanic White, and 18% as Hispanic. The objective response rate (ORR) was similar for lung cancer (35.057%) and HNSCC patients (30.3%) (p=0.894). The ORR for Hispanic and Black patients was lower compared to non-Hispanic White patients (H 27.0%, B 32.5%, W 38.7%; H vs. W p=0.209; B vs. W p=0.398). When considering only patients treated with ICI monotherapy, the ORR for Hispanic patients dropped further to 20.7% while the ORR of Black and non-Hispanic White patients remained about the same (B 29.3% and W 35.9%, H vs. W p=0.133; B vs. W p=0.419). Immune related adverse events were the lowest in the Hispanic population occurring in only 30% of patients compared to 40% of patients in the Black cohort and 50% of the non-Hispanic White cohorts.
Keywords: immune checkpoint inhibitors, lung cancer, race, head and neck cancer
Introduction
Since the approval of the first immune checkpoint inhibitor (ICI) to treat melanoma in 20111, ICIs targeting the programmed death (PD)-1 – programmed death ligand (PDL)-1 signaling axis between tumor cells and infiltrating T-lymphocytes have demonstrated activity in multiple solid tumor types.2,3,4,5 ICIs targeting PD-1 (e.g., nivolumab and pembrolizumab) and PD-L1 (e.g., atezolizumab, durvalumab) have been shown to improve survival in non-small cell lung cancer (NSCLC) patients, and have been established as part of standard-of-care therapy since 2015.5–14 Nivolumab and pembrolizumab were approved to treat head and neck squamous cell carcinoma (HNSCC), another smoking related malignancy, in 2016 and 2019, respectively.15,16
Although the potential of ICIs to improve disease control and survival is now well established, whether the benefit of ICIs can be realized at a population level remains an open question given persistent disparities in cancer diagnosis and treatment delivery for patients with advanced stage and metastatic disease.17–22 Unfortunately, despite persistent efforts, <25% of cancer-related clinical trial participants are racial minorities.23–25,26 This inclusion disparity is particularly obvious in clinical trials that led to the approval of ICIs for the treatment of NSCLC (Checkmate 017, 90% White7 ; Checkmate 057, 91% White6 ; Keynote 001, 82% White9; OAK, 71% White13; Impower 150, 82% White14; Keynote 010, 73% White10) and in HNSCC (Checkmate 141, 83% White27; Keynote 048, 73% White16). Compounding this deficit is the fact that most cancer-related phase III clinical trials fail to report ethnicity.23
Given the limited data available regarding ICI effectiveness in minority patients, some investigators have attempted to address this issue by comparing the safety and efficacy of ICI treatment for NSCLC in real world populations. Ayers et al. found a trending survival benefit in African Americans treated with ICI in a cohort consisting of 30% African Americans (HR 0.6; P=0.062)25, while Nazha et al. reported no difference in overall survival with ICI treatment in African Americans (P=0.84).28 Data on Hispanic patients, now the largest minority population in the United States is lacking from all studies to date, whether prospective or retrospective.29 The Harris Health System (HHS) and the Michael E. DeBakey Veterans Affairs Medical Center (MEDVAMC) in Houston, Texas provide tertiary cancer care for a diverse patient population (e.g. HHS- 57% Hispanic, 25% African American, 10% White, and 8% other) providing equal access to Harris County residents or Veterans, respectively, regardless of insurance status or financial means. We retrospectively analyzed oncologic outcomes for NSCLC and HNSCC patients treated with PD-1 and PDL-1 ICIs at these equal access institutions in order to determine whether racial and/or ethnic disparities are present.
Methods
Patient Population.
Following approval from Baylor College of Medicine (BCM), HHS and MEDVAMC Institutional Review Boards, we performed a retrospective analysis of patients with a diagnosis of NSCLC or HNSCC between 2015 and 2020. Patients had either locally recurrent disease for which definitive local therapy (surgery or radiation) was no longer an option or metastatic disease. Patients receiving adjuvant immunotherapy as part of a definitive treatment strategy were excluded from the analysis. All collection and analysis of the current data was performed in a manner consistent with existing standards for clinical research (Declaration of Helsinki, US Federal Policy for the Protection of Human Subjects). Patient race/ethnicity (self-identified and listed in the electronic medical record), age, gender, tobacco and alcohol exposure, tumor characteristics, treatment history, ICI toxicities, response to ICI, PDL-1 status, ECOG performance status, and BMI were collected and analyzed. Tumor histology, stage (T, N, M- classifications), number of metastases, radiation treatment, and chemotherapy treatment were collected through review of the electronic medical records. Inclusion criteria included: 1) primary NCSLC or HNSCC, 2) tissue diagnosis at the participating institutions, 3) treatment delivery at the participating institutions, 4) treatment with ICI or ICI plus conventional cytotoxic chemotherapy. Patient response to ICI was determined by review of surveillance imaging (PET/CT, CT, or MRI; using official radiologist/nuclear medicine reading) following ICI treatment to determine complete response, partial response, stable disease, or progressive disease. Routine images were reviewed for each patient up to death or loss to follow up (LTFU) to determine best overall response. Best overall objective response rate (ORR) was determined by calculating the number of patients attaining a complete response or a partial response and dividing by the total patient population. PD-L1 status was ascertained from clinical records using companion testing for ICIs for those lung cancer patients for which data was available and reported as percentage of viable tumor cells expressing the protein.
Statistical analysis.
In order to assess the efficacy of ICIs with as few patient and disease intrinsic confounders as possible, we chose ORR as the primary endpoint for comparison. Associations between identified clinical, biological and pathologic variables were determined by two-sided Chi-square tests. Statistical calculations were performed with Prism (Graphpad Software LLC Version 9.1.2). For all statistics, p-values were considered to be statistically significant if below a threshold of 0.05 (two-sided).
Data Availability Statement
The data generated in this study are available within the article and its supplementary data files.
Results
Patient and disease characteristics.
We analyzed data for 174 lung and 33 head and neck cancer patients receiving either ICI therapy alone or in combination with chemotherapy across all three treatment pavilions at BCM. As the goal of our study was to compare treatment responses of the Black and Hispanic patient population to those of the White patient population, Table 1 details the patient demographics and disease characteristics categorized by these three different racial/ethnic cohorts (Table 1, Supplementary Table 1). A slight majority of the patients were non-Hispanic White (45%) followed closely by Black patients (37%) while the Hispanic cohort constituted 18% of the study population. The three cohorts were generally evenly matched among the detailed patient and disease characteristics with significant differences found in three categories among the entire cohort and two categories among the ICI monotherapy cohort. Specifically, the Hispanic population consisted of fewer smokers compared to the White population (p=0.0002) and had more sites of metastatic disease compare to the White patients (p=0.032). The White population reported more alcohol consumption than Black patients (p=0.0378) (Supplementary Figure 1A–C). These differences were also present in the cohort of patients that received ICI monotherapy, except for alcohol intake, which was similar between the various racial/ethnic cohorts (Supplementary Figure 2A & B).
Table 1.
Patient and treatment characteristics
| Black | White | Hispanic | ||
|---|---|---|---|---|
| N | 77 | 93 | 37 | |
| Age at diagnosis | 61.4 | 62.4 | 60.1 | |
| Age at ICI initiation | 62.8 | 64.3 | 61.1 | |
| Sex | ||||
| Male | 58 (75.3) | 69 (74.2) | 22 (59.5) | |
| Female | 19 (24.7) | 24 (25.8) | 15 (40.5) | |
| Smoking status * | ||||
| Yes | 66 (85.7) | 85 (91.4) | 24 (64.7) | |
| Never smoker | 10 (13.0) | 8 (8.6) | 13 (35.1) | |
| Smoker at diagnosis | 21 (27.3) | 34 (36.6) | 7 (18.9) | |
| Unknown | 1 (1.3) | 0 (0) | 0 (0) | |
| Alcohol ¥ | ||||
| Yes | 21 (27.3) | 39 (41.9) | 10 (27.0) | |
| No | 50 (64.9) | 44 (47.3) | 26 (68.4) | |
| Unknown | 6 (7.8) | 10 (10.8) | 1 (2.7) | |
| Cancer | ||||
| Lung | 68 (88.3) | 75 (80.6) | 31 (83.8) | |
| Adenocarcinoma | 51 (75.0) | 44 (58.7) | 22 (71.0) | |
| Squamous | 15 (22.1) | 21 (28.0) | 7 (22.6) | |
| Small cell | 2 (2.9) | 8 (10.7) | 2 (6.5) | |
| NSCLC (NOS) | 0 (0) | 2 (2.7) | 0 (0) | |
| Head and neck SCC | 9 (11.7) | 18 (19.4) | 6 (16.2) | |
| ECOG | ||||
| 0 | 9 (11.7) | 13 (14.0) | 7 (18.9) | |
| 1 | 33 (42.9) | 40 (43.0) | 19 (51.4) | |
| 2 | 23 (29.9) | 27 (29.0) | 7 (18.9) | |
| 3 | 3 (3.9) | 7 (7.5) | 3 (8.1) | |
| 4 | 1 (1.3) | 0 (0) | 0 (0) | |
| Unknown | 8 (10.4) | 6 (6.5) | 1 (2.7) | |
| Number of Metastasis * | ||||
| 0 | 10 (13.0) | 12 (12.9) | 5 (13.5) | |
| Contralateral lung only | 7 (9.0) | 4 (4.3) | 6 (16.2) | |
| 1 | 20 (26.0) | 41 (44.1) | 4 (10.8) | |
| 2 | 30 (39.0) | 16 (17.2) | 15 (40.5) | |
| 3 | 6 (7.8) | 16 (17.2) | 4 (10.8) | |
| 4+ | 4 (5.2) | 4 (4.3) | 3 (8.1) | |
| Any brain metastasis | 13 (16.9) | 25 (26.9) | 13 (35.1) | |
| Prior systemic therapy (all) | ||||
| 0 | 19 (24.7) | 24 (25.8) | 10 (27.0) | |
| 1 | 33 (42.9) | 46 (49.5) | 17 (45.9) | |
| 2 | 18 (23.4) | 15 (16.1) | 5 (13.5) | |
| 3 | 4 (5.2) | 4 (4.3) | 2 (5.4) | |
| 4+ | 3 (3.9) | 4 (4.3) | 3 (8.1) | |
| Prior systemic therapy (concurrent and adjuvant chemo excluded) | ||||
| 0 | 29 (37.7) | 39 (41.9) | 17 (45.9) | |
| 1 | 33 (42.9) | 41 (44.1) | 13 (35.1) | |
| 2 | 10 (13.0) | 6 (6.5) | 2 (5.4) | |
| 3 | 2 (2.6) | 3 (3.2) | 2 (5.4) | |
| 4+ | 2 (2.6) | 4 (4.3) | 3 (8.1) | |
| Concurrent chemo with ICI | ||||
| Yes | 19 (24.7) | 15 (16.1) | 8 (21.6) | |
| No | 58 (75.3) | 78 (83.9) | 29 (78.3) | |
| ICI treatment | ||||
| Nivolumab | 36 (46.8) | 47 (50.5) | 16 (43.2) | |
| Pembrolizumab | 41 (59.7) | 46 (49.5) | 21 (56.8) | |
| Prior radiation anywhere* ¥ | ||||
| Yes | 49 (63.6) | 73 (78.5) | 22 (59.5) | |
| No | 28 (36.4) | 20 (21.5) | 15 (40.5) | |
| PD-L1 (NSCLC only) | ||||
| 0% | 7 (10.6) | 8 (11.9) | 4 (12.9) | |
| 1–49% | 17 (25.8) | 8 (11.9) | 6 (19.4) | |
| >50% | 16 (24.2) | 18 (26.9) | 10 (32.3) | |
| Unknown | 26 (39.4) | 41 (61.2) | 11 (35.5) |
p<0.05 for Hispanic vs. White
p<0.05 for Black vs. White
Treatment characteristics.
Treatment characteristics were well matched among the racial/ethnic cohorts (Table 1). About 20% of patients among each cohort received ICI in combination with chemotherapy. 40% of the patients in each cohort received ICI as first line treatment for recurrent or metastatic disease. Both Hispanic and Black patients were subject to less radiation treatment (definitive or palliative) than White patients (p=0.032 and p=0.027, respectively) (Supplementary Figure 3A).
The ICI monotherapy patient population also showed similar treatment characteristics among the three racial/ethnic groups (Supplementary Table 1). Only 20% of these patients received ICI monotherapy in the first-line setting. Also, only the Hispanic population was subject to significantly less radiation therapy (definitive or palliative) than the White population in this treatment cohort (p=0.026) (Supplementary Figure 3B).
Overall population responses.
Treatment responses were first calculated for the entire cohort and compared by disease type. ORR was approximately 30% for the entire 207 patient cohort and was similar between the NSCLC and HNSCC patient cohorts (Figure 1a). Overall, the response rates were similar between the cohorts that received ICI monotherapy and ICI in combination with chemotherapy (Figure 1b). PD-L1 status was available for slightly more than half of NSCLC patients reviewed (Table 1). As expected, a PD-L1 status of 50% or more was associated with a significantly (p=0.0086) higher ORR compared to a PD-L1 less than 50% in the population of patients receiving ICI monotherapy (Figure 1c). Given some significant differences in patient, disease, and treatment characteristics between the racial cohorts noted above, we calculated ORRs for each of these differences separately to assure they did not confound the ORR analysis of the racial cohorts detailed below. ORRs did not differ significantly for any of the disparate characteristics described in either the entire study cohort or the ICI monotherapy cohort (Supplementary Figures 4 and 5). p16 status of oropharyngeal cancers had no discernible impact on response as only one out of nine cancers showed a response to therapy (Supplementary Table 2).
Figure 1. Best overall response.
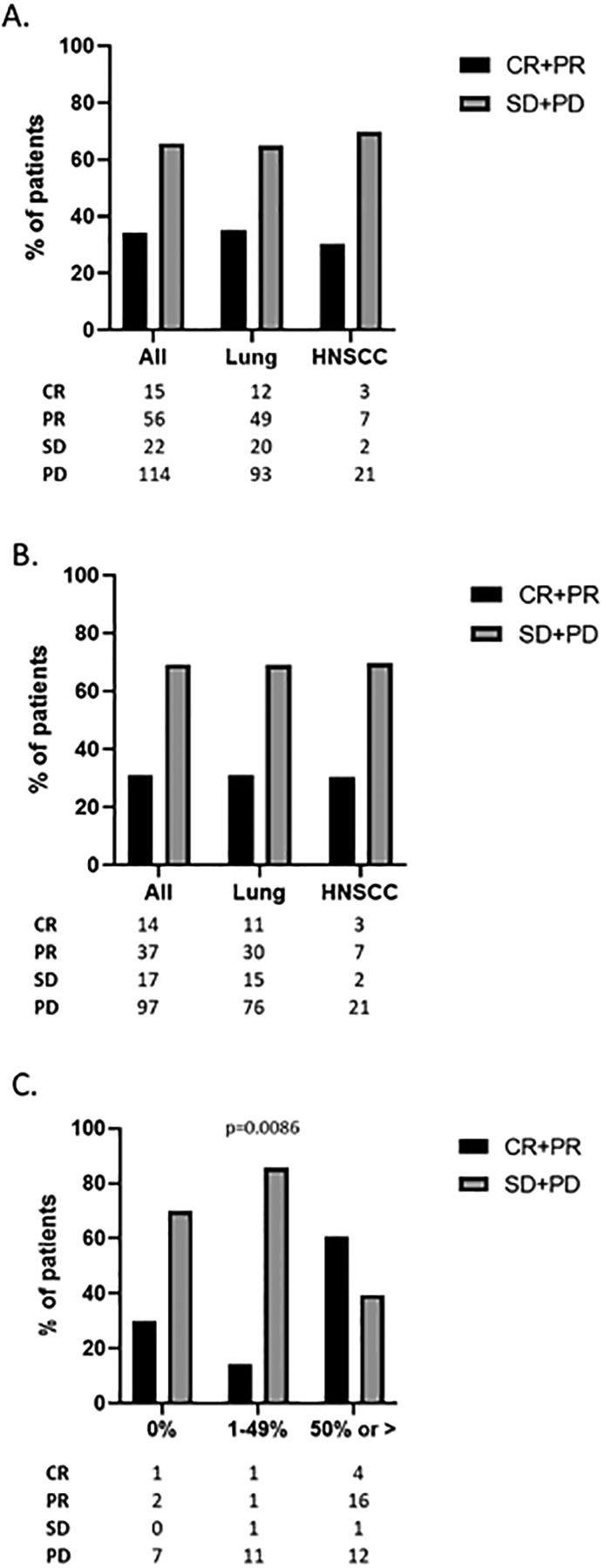
A) ORR data for all patients and by disease category for patients treated with either ICI monotherapy or chemo/ICI combination therapy. B) ORR data for all patients and by disease category for patients treated with ICI monotherapy. C) ORR data for NSCLC patients stratified by PD-L1 status treated with ICI monotherapy. Only patients for whom PD-L1 was known were included. CR- complete response; PR- partial response; SD- stable disease; PD- progressive disease.
Responses by race/ethnicity.
ORR in Hispanic patients was lower than that of Black and non-Hispanic White patients (27.0% compared to 32.5.% and 38.7%, respectively) though these differences were not statistically significant compared to the non-Hispanic White patient population (Figure 2a). When patients receiving chemo and ICI combination therapy were removed from the analysis, the ORR of the Hispanic cohort decreased further while the response rate of Black and White patients remained about the same. Only 20.7% of Hispanic patients treated with ICI monotherapy had a response while the ORR for Black and non-Hispanic White were 29.3% and 35.9% respectively. (Figure 2b). Over 70% of Hispanic patients’ tumors were refractory to ICI monotherapy while lack of any clinical benefit (complete response, partial response, or stable disease) to ICI monotherapy was seen in only 55.2% and 56.4% of Black and non-Hispanic White patients, respectively. No complete responses (CR) were observed in the Hispanic ICI monotherapy cohort and only one patient treated with combination immunotherapy and chemotherapy achieved a CR. Black and non-Hispanic White patients treated with ICI monotherapy had a CR rate of 5.2% and 14.1%, respectively (Figure 2b). Interestingly, all the CRs in the Black and non-Hispanic White populations were in patients treated with ICI monotherapy.
Figure 2. Best overall response by race/ethnic group.
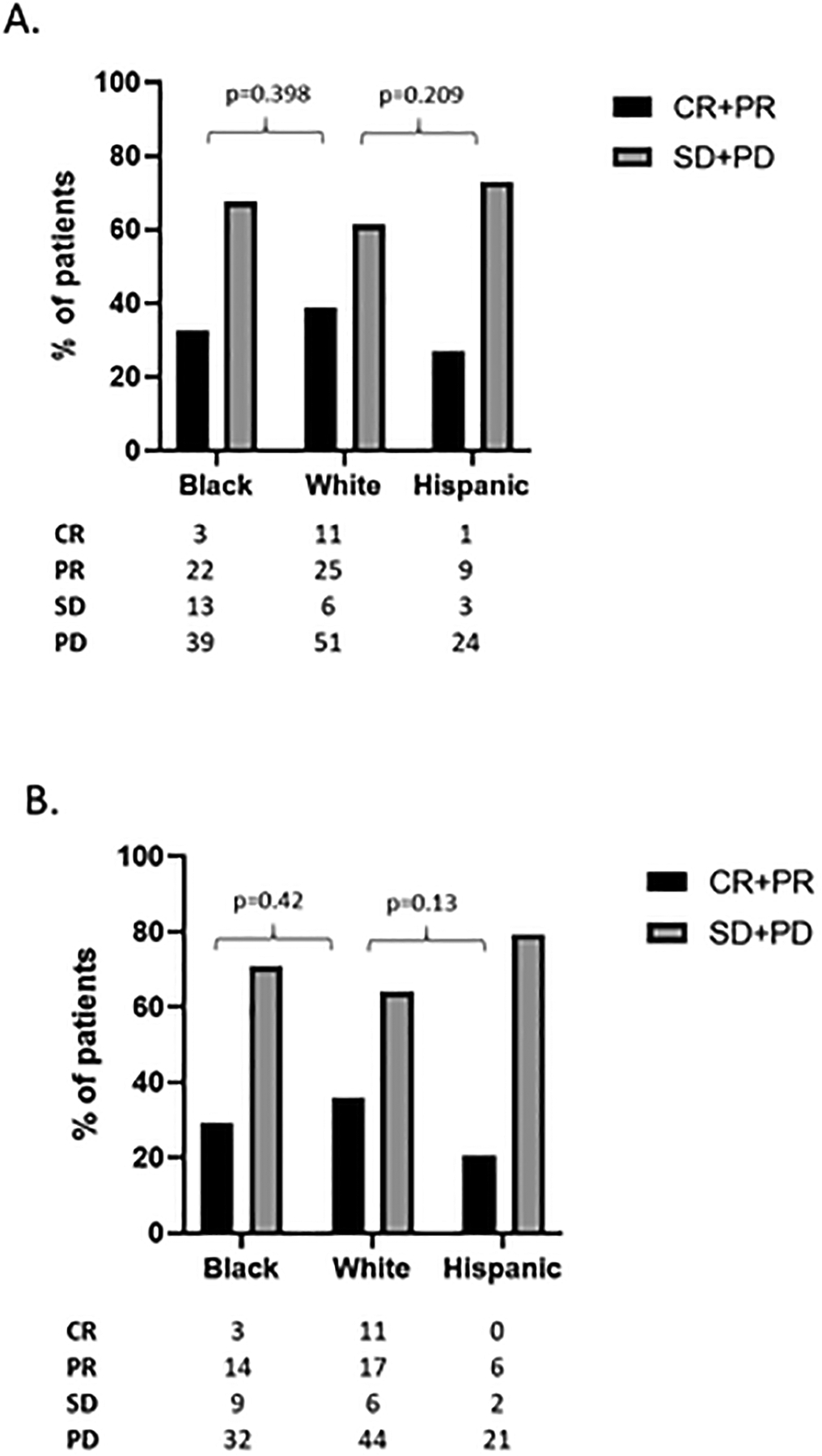
A) ORR data for all patients and by race/ethnic group for patients treated with either ICI monotherapy or chemo/ICI combination therapy. B) ORR data for all patients and by race/ethnic group for patients treated with ICI monotherapy. CR- complete response; PR- partial response; SD- stable disease; PD- progressive disease.
Responses by PD-L1 in NSCLC.
We assessed ORR in NSCLC patients using a combination of PD-L1 status and race/ethnicity. Again, as expected, Black and non-Hispanic White patients with a PD-L1 of 50% or greater exhibited a high ORR of over 65% when treated with ICI monotherapy while only 29% of Hispanic patients responded. (Figure 3a). This ORR dropped drastically to approximately 20% or below for tumors with PD-L1 less than 50% (Figures 3b, c). Paradoxically, 50% of Hispanic patients with a PD-L1 0% exhibited a partial response. (Figure 3).
Figure 3. Best overall response by PD-L1.
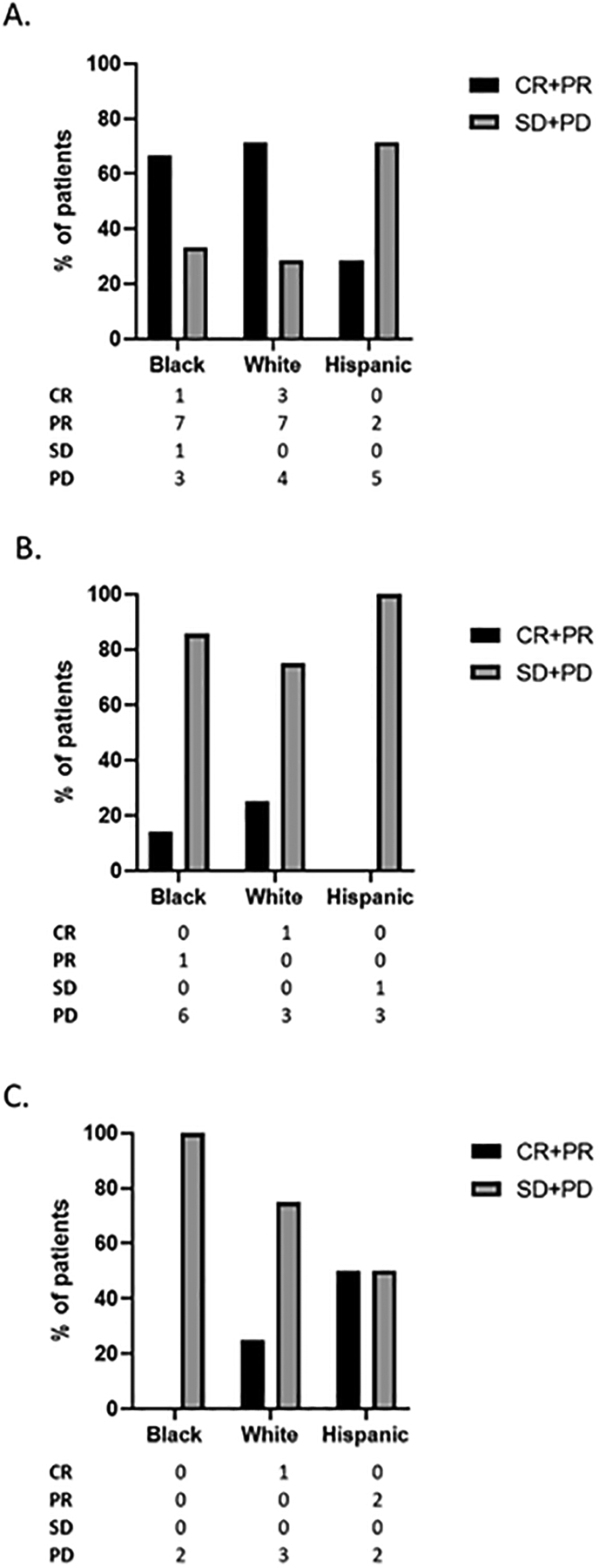
ORR data by race/ethnic group for NSCLC patients treated with ICI monotherapy stratified by PD-L1 A) ≥50%, B) 1–49% and C) 0% . CR- complete response; PR- partial response; SD- stable disease; PD- progressive disease.
Immune-related Adverse Events.
Adverse events thought to be related to immunotherapy (irAEs) were analyzed for the ICI monotherapy cohort. Approximately 40% of Black and 50% non-Hispanic White patients experienced irAEs of any grade. However, less than 30% of Hispanic patients developed any irAEs which was significantly less (p=0.0109) than non-Hispanic White patients (Figure 4a). We assessed the frequency of severe irAEs defined as toxicities resulting in systemic steroid administration, temporary cessation of ICI therapy, or permanent cessation of therapy among the three racial/ethnic cohorts. Approximately 15–20% of patients, regardless of race/ethnicity, experienced severe irAEs with about 10% of patients, again regardless of race/ethnicity, requiring a complete cessation of therapy (Supplementary Table 3). Given the shared mechanism of ICI irAEs and anti-tumor immunity, we hypothesized that the presence of toxicity would predict positive response to ICIs. While patients experiencing at least one irAE in the ICI monotherapy cohort had a higher ORR than those patients exhibiting no irAEs (36% vs. 27%, respectively) this difference was not statistically significant (Figure 4b) Finally, while a higher percentage of Hispanic patients was treated with pembrolizumab compared to White patients, and patients treated with pembrolizumab exhibited less irAEs, neither of these differences were statistically significant suggesting that the observed irAE disparity was unrelated to ICI type (Supplementary Figures 6 and 7).
Figure 4. Immune-related adverse events (irAEs) within the entire treatment cohort.
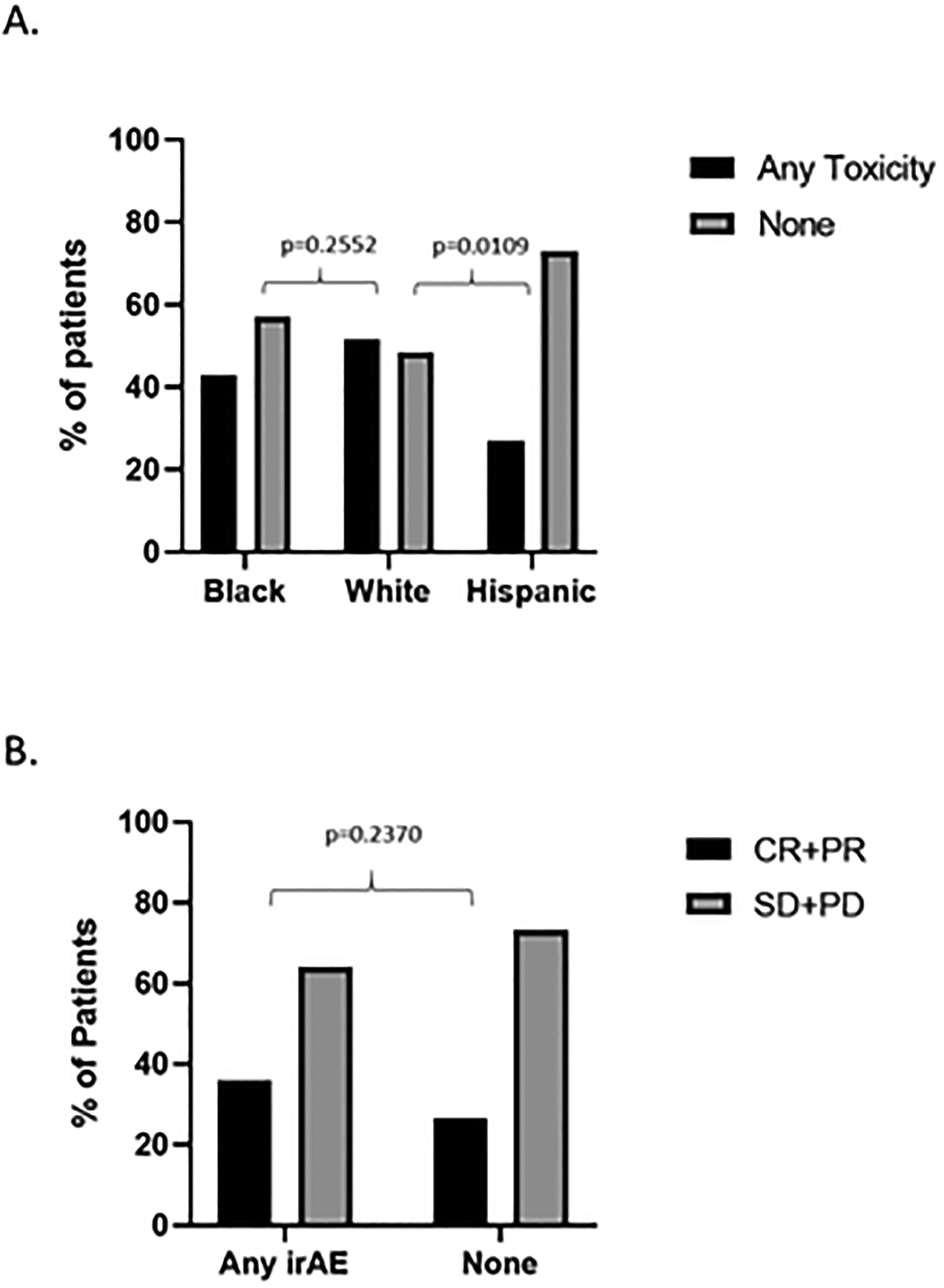
A) Overall irAE rate as a function of race/ethnic group. B) ORR as a function of irAE for the ICI monotherapy cohort. CR: Complete response; PR: Partial response; SD: stable disease; PD: Progressive disease.
Discussion
Although racial disparities persist across multiple solid tumor histologies in the US population at large, individual institutional studies have been able to demonstrate equivalence in the setting of equal access. Our previous work in HNSCC has shown that both HHS and the MEDVAMC are able to generate equivalent oncologic outcomes for patients which is consistent with data from other institutions.30–34 These data would suggest that cancer outcome disparities at a population level are likely driven primarily by socioeconomic status, unequal access to timely and high-quality healthcare, and other social factors. Indeed, retrospective studies have shown that race strongly correlates to HNSCC disease stage at presentation, with Blacks presenting more often with advanced locoregional disease or with metastatic disease.19 Similarly, for HNSCC some have suggested that reduced survival in Black patients is a function of differential distribution of low-risk and high-risk disease (as a function of human papillomavirus (HPV)) status.18 However, contradicting these assertions are data which indicate that even when HPV status is accounted for, Black HNSCC patients and to a lesser degree Hispanic patients demonstrate reduced survival compared to their counterparts.35 Other biological factors have also been considered. Ramakodi et al. identified ancestry related single nucleotide polymorphisms (SNPs) in the DNA polymerase beta gene which may impact response to conventional chemo-radiation strategies used for both HNSCC and NCSLC in the curative intent setting.36 Differential SNP distribution may be compounded by differential somatic mutation distribution in some HNSCC patients to further impede treatment response and reduce survival.37 Transcriptional data from African American patients demonstrates race-related shifts in both tumor metabolism and DNA repair across multiple disease sites including HNSCC and other smoking related malignancies.38
Taken together these data highlight an outstanding question for clinicians and patients alike: are Black and Hispanic patients likely to derive the same benefit from ICIs once access is established? Certainly, the sparsity of minority patients in prospective clinical trials and the lack of consistently reported race-based response data prevents a definitive answer using level I data. It is therefore critical that continued attempts to answer this question be made in the real-world, post-approval setting. This study offers retrospective data from our academic institution experience that represents the more diverse patient population that may be encountered in an urban center. Overall, our data are encouraging for Black patients. Not only are response rates comparable with those of White patients, but overall toxicity appears to be similar. Despite reports that show ICI enhances OS in Hispanics when compared to chemotherapy,39 our findings are concerning, particularly given the low rate of complete response. This lower response rate paralleled by a lower rate of irAEs suggest the potential for reduced effectiveness of this agent class. This report is the first showing potential decreased ICI effectiveness in a substantial Hispanic NSCLC and HNSCC patient population and our findings will need to be validated in additional series. Specifically, to better understand this finding, future studies will need to be enriched for patients treated with ICI monotherapy. Furthermore, it will be essential to determine what other factors might contribute to the altered effectiveness of ICI in Hispanics. Studies have suggested that the efficacy of ICI in various cancer types may be impacted by multiple factors including the use of antibiotics,40,41 the gut microbiome, mutational burden, infections, and exercise.42,43 Thus, future studies will also be needed to assess the impact of additional factors on racial/ethnic disparities in ICI therapy.
We acknowledge the limitations of our study which include its retrospective nature, cohort size, limited availability of PD-L1 data for the NSCLC patients, and absence of detailed socio-economic/cultural cohort data. Sample size in particular limits our ability to identify confounders using multivariate analysis, though we did identify significant differences among our racial/ethnic cohorts and have shown that these do not impact response rates (Supplementary Figures 1–5). Sample size may also be responsible for the high response rate noted in our HNSCC cohort which is almost double what was reported in two landmark clinical trials (15% trial cohort vs. 30% study cohort) whereas the lung cohort was only slightly higher than trial results (25% trial cohort vs. 31% study cohort).7,16,44,45 However, the cohort spans two distinct equal access institutions with significant minority populations, affiliated with a tertiary academic institution and an NCI-designated comprehensive cancer center (CCC), helping control for at least one important factor contributing to oncologic outcome disparities among racial minorities: access to care. To further evaluate the outcomes of this study, a concerted effort is needed to enact specific policies and funding opportunities focused on minority recruitment in order to encourage enrollment of minority patients into clinical trials focused on ICIs in NSCLC and HNSCC in a manner representative of current demographic shifts. As such, we consider it imperative that NCI-designated CCCs which serve predominantly minority populations form a closer network designed for data sharing, specimen banking and research integration. Within such a network, it is important to support biobanking and genotyping efforts that will allow us to develop biologically focused analyses of interactions between ancestry and treatment response. These analyses may help to elucidate the effects of socio-economic/cultural/environmental factors compared to biological differences which even in the context of equal access to care is not adequately parsed out. Furthermore, patient diversity in these efforts is especially important as major existing bio-response databases predominantly consist of White population of European ancestry.46 Combined with national efforts such as those exemplified by the Million Veteran Program which seeks to elucidate the health impacts of genetics, lifestyle, and military exposure, such a network could help better understand how to serve and advance the health of our patients
In conclusion, this retrospective cohort study identifies a potential signal of decreased ICI-response in Hispanic lung cancer and HNSCC patients. Coupled with a significantly lower irAE rate for Hispanic patients, the data suggest a possible underlying mechanistic reason for this disparity. However, the exact causes remain unclear and may be linked to differences equally as disparate ranging from variations in intrinsic tumor biology and immunology, genetics and epigenetics to extrinsic socio-economic and cultural (e.g. diet, exercise, medication exposure, etc.) factors. Expanding the investigation of this outcome and its causes is an imperative next step in the effort to improve outcomes for Hispanic lung and head and neck cancer patients and will serve to broaden our understanding of factors impacting ICI response.
Supplementary Material
Statement of Significance.
To our knowledge this report is the first to compare ICI effectiveness within a diverse patient population with a substantial Black and Hispanic NSCLC and HNSCC patient population treated in the context of equal access to care. The data presented in this manuscript suggests reduced effectiveness of ICI monotherapy in Hispanic patients and thereby underscores the need for improved access and representation of racial/ethnic minority patients in ICI clinical trials.
Funding Acknowledgment:
This material is the result of work supported with resources and the use of facilities of the Michael E. DeBakey VA Medical Center and was specifically supported by an institutional pilot grant (V.C.S). This work is supported in part by a Career Development Award from the Veterans Administration Clinical Science Research and Development division (1IK2CX001953) (V.C.S.). The project was supported by the Ruth L. Kirschstein National Research Service Award (NRSA) Individual Predoctoral Fellowship to Promote Diversity in Health-Related Research (F31-Diversity- 1F31HL156500) and the American Society of Hematology Minority Hematology Graduate Student Fellowship (2532420504) (M.A.F.). Contents do not represent the views of the US Department of Veterans Affairs or the US Government.
Footnotes
Conflicts of interest: The authors declare no potential conflicts of interest.
References
- 1.Heidi. Melanoma drug wins US approval. Nature Ledford; 471. [DOI] [PubMed] [Google Scholar]
- 2.Tang J, Shalabi A, Hubbard-Lucey VM. Comprehensive analysis of the clinical immuno-oncology landscape. Ann Oncol 2018; 29:84–91. [DOI] [PubMed] [Google Scholar]
- 3.Esfahani K, Roudaia L, Buhlaiga N, Del Rincon SV, Papneja N, Miller WH Jr. A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol 2020; 27:S87–S97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kao HF, Lou PJ. Immune checkpoint inhibitors for head and neck squamous cell carcinoma: Current landscape and future directions. Head Neck 2019; 41 Suppl 1:4–18. [DOI] [PubMed] [Google Scholar]
- 5.Onoi K, Chihara Y, Uchino J, Shimamoto T, Morimoto Y, Iwasaku M et al. Immune Checkpoint Inhibitors for Lung Cancer Treatment: A Review. J Clin Med 2020; 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE et al. Nivolumab versus Docetaxel in Advanced Nonsquamous Non-Small-Cell Lung Cancer. N Engl J Med 2015; 373:1627–1639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brahmer J, Reckamp KL, Baas P, Crino L, Eberhardt WE, Poddubskaya E et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med 2015; 373:123–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gandhi L, Rodriguez-Abreu D, Gadgeel S, Esteban E, Felip E, De Angelis F et al. Pembrolizumab plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer. N Engl J Med 2018; 378:2078–2092. [DOI] [PubMed] [Google Scholar]
- 9.Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015; 372:2018–2028. [DOI] [PubMed] [Google Scholar]
- 10.Herbst RS, Baas P, Kim DW, Felip E, Perez-Gracia JL, Han JY et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016; 387:1540–1550. [DOI] [PubMed] [Google Scholar]
- 11.Paz-Ares L, Luft A, Vicente D, Tafreshi A, Gumus M, Mazieres J et al. Pembrolizumab plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N Engl J Med 2018; 379:2040–2051. [DOI] [PubMed] [Google Scholar]
- 12.Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csoszi T, Fulop A et al. Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N Engl J Med 2016; 375:1823–1833. [DOI] [PubMed] [Google Scholar]
- 13.Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet 2017; 389:255–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N Engl J Med 2018; 378:2288–2301. [DOI] [PubMed] [Google Scholar]
- 15.Ferris RL, Blumenschein G Jr., Fayette J, Guigay J, Colevas AD, Licitra L et al. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med 2016; 375:1856–1867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Burtness B, Harrington KJ, Greil R, Soulieres D, Tahara M, de Castro G Jr. et al. Pembrolizumab alone or with chemotherapy versus cetuximab with chemotherapy for recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-048): a randomised, open-label, phase 3 study. Lancet 2019; 394:1915–1928. [DOI] [PubMed] [Google Scholar]
- 17.Clarke JA, Despotis AM, Ramirez RJ, Zevallos JP, Mazul AL. Head and Neck Cancer Survival Disparities by Race and Rural-Urban Context. Cancer Epidemiol Biomarkers Prev 2020; 29:1955–1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mazul AL, Naik AN, Zhan KY, Stepan KO, Old MO, Kang SY et al. Gender and race interact to influence survival disparities in head and neck cancer. Oral Oncol 2021; 112:105093. [DOI] [PubMed] [Google Scholar]
- 19.Megwalu UC, Ma Y. Racial Disparities in Oropharyngeal Cancer Stage at Diagnosis. Anticancer Res 2017; 37:835–839. [DOI] [PubMed] [Google Scholar]
- 20.Russo DP, Tham T, Bardash Y, Kraus D. The effect of race in head and neck cancer: A meta-analysis controlling for socioeconomic status. Am J Otolaryngol 2020; 41:102624. [DOI] [PubMed] [Google Scholar]
- 21.Sheth S, Farquhar DR, Lenze NR, Mazul A, Brennan P, Anantharaman D et al. Decreased overall survival in black patients with HPV-associated oropharyngeal cancer American Journal of Otolaryngology 2021; 42:102780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Voltzke KJ, Lee YA, Zhang ZF, Zevallos JP, Yu GP, Winn DM et al. Racial differences in the relationship between tobacco, alcohol, and the risk of head and neck cancer: pooled analysis of US studies in the INHANCE Consortium. Cancer Causes Control 2018; 29:619–630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Grant SR, Lin TA, Miller AB, Mainwaring W, Espinoza AF, Jethanandani A et al. Racial and Ethnic Disparities Among Participants in US-Based Phase 3 Randomized Cancer Clinical Trials. JNCI Cancer Spectr 2020; 4:pkaa060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Loree JM, Anand S, Dasari A, Unger JM, Gothwal A, Ellis LM et al. Disparity of Race Reporting and Representation in Clinical Trials Leading to Cancer Drug Approvals From 2008 to 2018. JAMA Oncol 2019; 5:e191870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ayers KL, Mullaney T, Zhou X, Liu JJ, Lee K, Ma M et al. Analysis of Real-World Data to Investigate the Impact of Race and Ethnicity on Response to Programmed Cell Death-1 and Programmed Cell Death-Ligand 1 Inhibitors in Advanced Non-Small Cell Lung Cancers. Oncologist 2021; 26:e1226–e1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen MS Jr., Lara PN, Dang JH, Paterniti DA, Kelly K Twenty years post-NIH Revitalization Act: enhancing minority participation in clinical trials (EMPaCT): laying the groundwork for improving minority clinical trial accrual: renewing the case for enhancing minority participation in cancer clinical trials. Cancer 2014; 120 Suppl 7:1091–1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Saba NF, Blumenschein G Jr., Guigay J, Licitra L, Fayette J, Harrington KJ et al. Nivolumab versus investigator’s choice in patients with recurrent or metastatic squamous cell carcinoma of the head and neck: Efficacy and safety in CheckMate 141 by age. Oral Oncol 2019; 96:7–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nazha B, Goyal S, Chen Z, Engelhart A, Carlisle JW, Beardslee TJ et al. Efficacy and safety of immune checkpoint blockade in self-identified Black patients with advanced non-small cell lung cancer. Cancer 2020; 126:5040–5049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Miller KD, Goding Sauer A, Ortiz AP, Fedewa SA, Pinheiro PS, Tortolero-Luna G et al. Cancer Statistics for Hispanics/Latinos, 2018. CA Cancer J Clin 2018; 68:425–445. [DOI] [PubMed] [Google Scholar]
- 30.Zevallos JP, Sandulache VC, Hamblin J, Skinner HD, Kramer J, Hartman CM et al. Impact of race on oropharyngeal squamous cell carcinoma presentation and outcomes among veterans. Head Neck 2016; 38:44–50. [DOI] [PubMed] [Google Scholar]
- 31.Sandulache VC, Kubik MW, Skinner HD, Malsky JA, Gelbard AH, Zevallos JP. Impact of race/ethnicity on laryngeal cancer in patients treated at a Veterans Affairs Medical Center. Laryngoscope 2013; 123:2170–2175. [DOI] [PubMed] [Google Scholar]
- 32.Fullmer TM, Shi J, Skinner HD, Hernandez DJ, Huang AT, Mai W et al. Early glottic cancer in a veteran population: Impact of race on management and outcomes. Laryngoscope 2019. [DOI] [PubMed] [Google Scholar]
- 33.Fullmer T, Wilde DC, Shi JW, Wagner T, Skinner H, Eicher SA et al. Demographic and Tumor Characteristic Impact on Laryngeal Cancer Outcomes in a Minority Underserved Patient Population. Otolaryngol Head Neck Surg 2020; 162:888–896. [DOI] [PubMed] [Google Scholar]
- 34.Chen LM, Li G, Reitzel LR, Pytynia KB, Zafereo ME, Wei Q et al. Matched-pair analysis of race or ethnicity in outcomes of head and neck cancer patients receiving similar multidisciplinary care. Cancer Prev Res (Phila) 2009; 2:782–791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Faraji F, Rettig EM, Tsai HL, El Asmar M, Fung N, Eisele DW et al. The prevalence of human papillomavirus in oropharyngeal cancer is increasing regardless of sex or race, and the influence of sex and race on survival is modified by human papillomavirus tumor status. Cancer 2019; 125:761–769. [DOI] [PubMed] [Google Scholar]
- 36.Ramakodi MP, Devarajan K, Blackman E, Gibbs D, Luce D, Deloumeaux J et al. Integrative genomic analysis identifies ancestry-related expression quantitative trait loci on DNA polymerase beta and supports the association of genetic ancestry with survival disparities in head and neck squamous cell carcinoma. Cancer 2017; 123:849–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ramakodi MP, Kulathinal RJ, Chung Y, Serebriiskii I, Liu JC, Ragin CC. Ancestral-derived effects on the mutational landscape of laryngeal cancer. Genomics 2016; 107:76–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Piyarathna DWB, Balasubramanian A, Arnold JM, Lloyd SM, Karanam B, Castro P et al. ERR1 and PGC1alpha associated mitochondrial alterations correlate with pan-cancer disparity in African Americans. The Journal of clinical investigation 2019; 129:2351–2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cardona AF, Ruiz-Patiño A, Arrieta OG, Martin C, Raez L, Barrón ZLZ et al. EP1.04–46 Immunotherapy at Any Line Improves Survival in Hispanic Patients with Advanced Metastatic NSCLC Compared to Chemotherapy (Quijote-CLICaP). Journal of Thoracic Oncology 2019; 14:S984–S985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ruiz-Patino A, Barron F, Cardona AF, Corrales L, Mas L, Martin C et al. Antibiotics impair immune checkpoint inhibitor effectiveness in Hispanic patients with non-small cell lung cancer (AB-CLICaP). Thorac Cancer 2020; 11:2552–2560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Derosa L, Hellmann MD, Spaziano M, Halpenny D, Fidelle M, Rizvi H et al. Negative association of antibiotics on clinical activity of immune checkpoint inhibitors in patients with advanced renal cell and non-small-cell lung cancer. Ann Oncol 2018; 29:1437–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yan X, Zhang S, Deng Y, Wang P, Hou Q, Xu H. Prognostic Factors for Checkpoint Inhibitor Based Immunotherapy: An Update With New Evidences. Front Pharmacol 2018; 9:1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Deshpande RP, Sharma S, Watabe K. The Confounders of Cancer Immunotherapy: Roles of Lifestyle, Metabolic Disorders and Sociological Factors. Cancers (Basel) 2020; 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ferris RL, Blumenschein G Jr., Fayette J, Guigay J, Colevas AD, Licitra L et al. Nivolumab vs investigator’s choice in recurrent or metastatic squamous cell carcinoma of the head and neck: 2-year long-term survival update of CheckMate 141 with analyses by tumor PD-L1 expression. Oral Oncol 2018; 81:45–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 2019; 393:1819–1830. [DOI] [PubMed] [Google Scholar]
- 46.Carrot-Zhang J, Chambwe N, Damrauer JS, Knijnenburg TA, Robertson AG, Yau C et al. Comprehensive Analysis of Genetic Ancestry and Its Molecular Correlates in Cancer. Cancer Cell 2020; 37:639–654 e636. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data generated in this study are available within the article and its supplementary data files.


