Abstract
Long QT syndrome (LQTS) is a detrimental arrhythmia syndrome mainly caused by dysregulated expression or aberrant function of ion channels. The major clinical symptoms of ventricular arrhythmia, palpitations, and syncope vary among LQTS subtypes. Susceptibility to malignant arrhythmia is a result of delayed repolarization of the cardiomyocyte action potential. There are 17 distinct subtypes of LQTS linked to 15 autosomal dominant genes with monogenic mutations. However, due to the presence of modifier genes, the identical mutation may result in completely different clinical manifestations in different carriers. In this review, we describe the roles of various ion channels in orchestrating action potentials and discuss molecular etiologies of various types of LQTS. We highlight the usage of patient-specific induced pluripotent stem cell (iPSC) models in characterizing fundamental mechanisms associated with LQTS. To mitigate the outcomes of LQTS, treatment strategies are initially focused on small molecules targeting ion channel activities. Next-generation treatments will reap the benefits from development of LQTS patient-specific iPSC platform, which is bolstered by the state-of-the-art technologies including whole-genome sequencing, CRISPR genome editing, and machine learning. Deep phenotyping and high-throughput drug testing using LQTS patient-specific cardiomyocytes herald the upcoming precision medicine in LQTS.
Keywords: Congenital long QT syndrome (LQTS), Ion channels, Patient-specific iPSCs, Precision medicine
Introduction
Long QT syndrome (LQTS) is an inherited or acquired arrhythmia syndrome that may co-occur with malignant sudden cardiac death. It is characterized by the prolongation of QT interval and an enhanced threat of ventricular arrhythmia (1, 2). LQTS is primarily divided into congenital and acquired types. Congenital LQTS is mainly attributed to mutations in ion channels and their accessory proteins. Acquired LQTS is primarily caused by various pharmacological agents (e.g., antibiotics, antidepressants, antihistamines, antineoplastics).
Seventeen genes have been reported to be associated with congenital LQTS. Mutations in three common genes (KCNQ1, KCNH2, and SCN5A) account for more than 75% of clinically validated congenital LQTS cases, whereas less than 5% have been attributed to mutations in other genes (3). Genetic etiologies for the remaining approximately 20% of hereditary LQTS are unknown. Intriguingly, a clinical study revealed that the identical LQTS-relevant mutations are not necessarily linked to the same disease phenotype (4). Currently, effective treatment options for LQTS are beta-blockers (propranolol, nadolol, etc.), sodium channel blockers (e.g., mexiletine, flecainide, etc.), and surgical interventions that include left cardiac sympathetic denervation (LCSD) and implantable cardioverter-defibrillator (ICD) (5).
In this review, we introduce the clinical manifestations and diagnosis of LQTS. We discuss the molecular basis of ion channels underlying congenital LQTS and explore the potential of modeling LQTS using patient-specific iPSCs. We then summarize the current treatment strategies for LQTS and speculate on the future of precision medicine in LQTS through utilizing patient-specific iPSCs and whole-genome sequencing.
Clinical manifestations and diagnosis of LQTS
Clinical symptoms of LQTS may be present at birth or develop later in life. Severe symptoms may occur because of an irregular heartbeat that can lead to fainting, seizures, or sudden cardiac death. LQTS patients show high risks of cardiac arrhythmia events, including ventricular tachycardia and torsades de pointes, which predisposes transient syncope and further deteriorates into ventricular fibrillation (6). Most symptomatic LQTS patients suffer from primary arrhythmic events with a notable difference in gender, age, and health differences (7). An array of stimuli is correlated with LQTS, including running, swimming, unforeseen noises, and being scared. Patients with LQTS1 are threatened with upregulated synergistic activities, emotional dysregulation, and physical burdens (7, 8). LQTS2 patients are susceptible to low plasma potassium levels, with female patients in the postpartum period are vulnerable to sleep disturbance (9). For LQTS3 patients, most cardiac arrhythmic events occur during rest or sleep (10).
To monitor the electrical activity within each heartbeat, an electrocardiogram (ECG) of the electrical signal traveling through the heart is recorded. A normal heartbeat has five separate electrical waves (Figure 1A): P, Q, R, S, and T. The P wave corresponds to the process of atrial depolarization that extends from the SA node throughout the atria. Subsequent QRS wave combination represents the ventricular depolarization process. The subsequent ST-segment is considered the isoelectric period in which the T wave indicates the process of ventricular repolarization. The occasionally exhibited U wave is a tiny, circular deflection that follows the waves of depolarization (QRS) and repolarization (T) (11). U waves reflect the last step of ventricular repolarization from a functional standpoint. Although the origin of the U wave is still a matter of debate, most clinicians and researchers associate it with “afterdepolarizations” in the ventricle.
Figure 1. Characteristic summary of ion channels in LQTS.
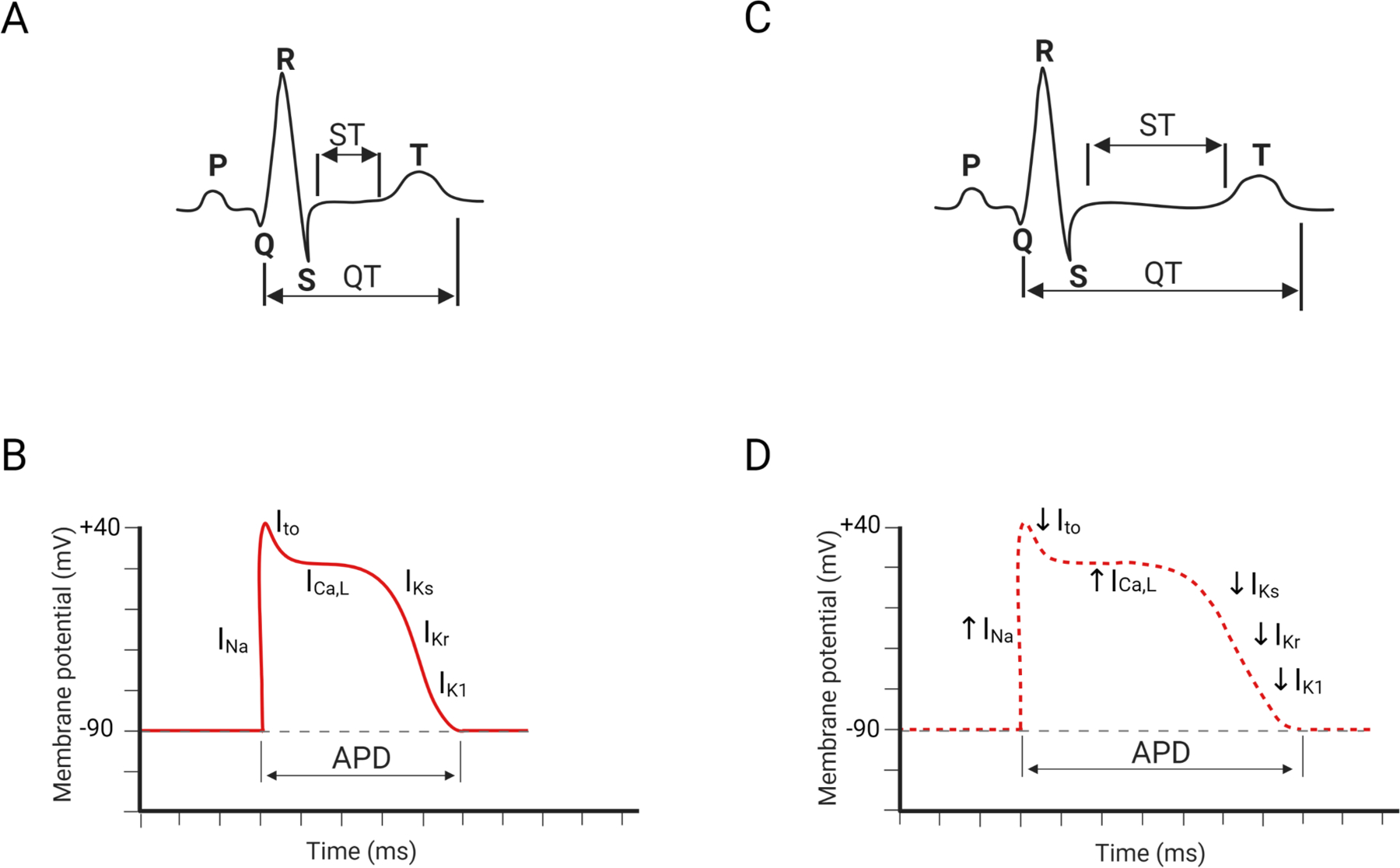
(A) A representative pattern of normal ECG. (B) A schematic view of action potential duration in normal ventricular cardiomyocytes. (C) A representative pattern of LQTS-specific ECG. (D) A schematic view of action potential duration in LQTS ventricular cardiomyocytes. INa: sodium currents; Ito: transient outward potassium currents; ICa,L: L-type calcium current; IKs: slow delayed rectifier potassium current; IKr: rapid delayed rectifier potassium current; IK1: inward rectifier potassium current.
The QT interval is the overall duration of the ventricular depolarization and repolarization processes, which is inversely proportional to the heart rate. Compared to those with a normal heart rhythm, ECGs of LQTS patients show a prolonged QT interval (Figure 1B). The overall ECG pattern is largely determined by electrophysiological profiles of each ventricular cardiomyocyte in the heart. In a ventricular cardiomyocyte, an action potential (AP) is produced by the coordinated activation (open) and inactivation (close) of several membrane-embedded ion channels (Figure 1C) (12). The initial upstroke of the cardiac AP is orchestrated by the activation and inactivation of sodium channels, followed by the repolarization that is conducted by many distinctive genres of potassium channels. Initial repolarization is directed by the efflux of transient outward potassium currents (Ito). Subsequently, a plateau stage is achieved by the flow of outward currents through opening delayed rectifier potassium channels (ultra-rapid: IKur, rapid: IKr, slow: IKs) and the counteracting force driven by L-type calcium channels. The terminal stage of the repolarization is operated by rapid delayed rectifier potassium currents (IKr) and inward rectifier potassium currents (IK1). The combined dysregulated activities of several ion channels extend the action potential duration (APD), resulting in the QT interval prolongation (Figure 1D).
Diagnosis of long QT syndrome depends on a broad spectrum of factors including heart rate corrected QT intervals (QTc) and electrocardiographic parameters (13). The upper values of normal QTc are 440ms for males and 460ms for females, respectively (14). To diagnose LQTS, accurate measurements of QTc duration at rest are required (14). Regarding electrocardiographic parameters, the abnormal shape of the T-wave is a common phenomenon in LQTS patients. Precordial leads are particularly instructive in reflection of biphasic or notched T-wave. From clinical perspective, patients with different types of LQTS exhibit distinct patterns of ST-T segment (15, 16). ECG of LQTS1 is characterized by a broad T wave and prolonged QT interval (17). ECG in LQTS2 present the notched asymmetrical T wave and low amplitude (17). LQTS3 patients are predisposed to encounter cardiac episodes and QT during rest (7). LQTS3 may present as a prolonged isoelectric interval following a normal T wave shape on the ECG (17).
Based on QTc duration, precordial leads, and a series of criteria, Schwartz et al. and Priori et al. established the scoring system to stratify the risks associated with LQTS (18, 19). In addition, genetic screening is beneficial to both diagnosis of asymptomatic LQTS and identification of the proband along with their familial members carrying causative mutations (14). Even acquired LQTS has been shown to be associated with underlying genetic factors (20). Therefore, genetic screening is essential for deciphering the relationship between genotype and phenotype, which can potentially explore the unidentified LQTS relevant mutations.
Although QT interval prolongation is the defining characteristic of LQTS, roughly 40% of genetically verified LQTS patients have a normal QTc at rest (21). It is crucial to distinguish individuals with LQTS from those with a normal QTc to properly diagnose the illness and put effective LQTS preventative measures into place. Another confounding factor in diagnosing LQTS is the subjective determination of Q and T wave boundaries. To overcome these above-mentioned obstacles, recent studies using machine learning (ML) and deep learning (DL) have shown an improvement in the diagnosis of LQTS (21, 22). Prifti et al. developed an artificial intelligence (AI)-driven approach to identify ECG patterns alternations that are indicatives of drug-induced LQTS as well as congenital LQTS (23). Their algorithm identified ECGs of LQTS2 patients with excellent accuracy; ECGs of LQTS1 and LQTS3 patients were also detected, albeit with lower accuracy than LQTS2. It is possible that rule-based expert systems may be eventually replaced by AI someday since deep learning methodologies can delineate the intricate patterns of ECGs. One challenging aspect of using DL to implement LQTS diagnosis is the need for large-scale training datasets to generate high-quality and unbiased results. Another major obstacle is the interpretability of the findings as the non-linear characteristics of deep neural networks are often difficult to comprehend.
Identification of disease-specific mutations enables clinicians to accumulate invaluable data for future diagnosis and precision treatment. A clinical trial (NCT04581408) is currently underway to test the mutation-specific treatment for LQTS2. The most common genetic causes of LQTS are associated with mutations in KCNQ1, KCNH2, and SCN5A (24). Identification of specific locations of genetic mutations within ion channels has proven beneficial in predicting the onset of LQTS (25). From prognostic studies, clinicians can explicitly educate LQTS patients to avoid exposure to LQTS triggering factors as mentioned before. Therefore, genotyping may aid in devising alternative strategies in combination with other treatment options (26). Because it is likely to identify irrelevant genetic variants of LQTS-related genes in asymptomatic populations, symptomatic individuals and those with a familial history of LQTS should be prioritized to receive genetic testing. Clinical manifestations have the potential to become a reliable way to classify pathogenic variants in LQTS. For instance, using ECG T waves morphology, we can classify the pathogenic genetic variants through unique pattern recognition. A broad-based T wave is frequently observed in LQTS1 patients carrying KCNQ1 variants. Low-amplitude notched T waves and late-onset peaked/biphasic T waves are ECG features of LQTS2 individuals bearing KCNH2 variants and LQTS3 subjects bearing SCN5A variants, respectively. CALM1–3 mutation carriers exhibit bradycardia or atrioventricular block along with severe QT prolongation, which is seldom observed in other LQTS subtypes. A patient with a loss-of-function variant in TRDN exhibited negative T waves and QT prolongation in precordial leads (27). KCNJ2 mutations were found in patients with prominent U-waves without prolonged QT, suggesting a phenotypic manifestation of Anderson-Tawil Syndrome. Extracardiac signs of ATS include dysmorphic features, recurrent paralysis, and neurocognitive deficits (28). Pediatric patients with certain CACNA1C variants demonstrated a spectrum of electrophysiological abnormalities including bradycardia, atrioventricular block, QT prolongation, and polymorphic ventricular arrhythmias, which is correlates with Timothy Syndrome (29). Nine (AKAP9, ANK2, CAV3, KCNE1, KCNE2, KCNJ2, KCNJ5, SCN4B, SNTA1) of the seventeen genes previously described as pathogenic LQTS variants were recently classified to have insufficient or disputed evidence for disease causation (30). Due to the complexity of the genetic architecture of LQTS (30–32), it is not recommended to solely utilize genetic variation for clinical decision-making until they are validated by solid clinical evidence.
Molecular basis of ion channel abnormalities underlying congenital LQTS
1. Major LQTS Genes
LQTS1 is the most common type of congenital LQTS, accounting for 30%−35% of all LQTS cases. KCNQ1 encodes α-subunit of voltage-gated potassium channel Kv7.1, generating slow delayed rectifier potassium currents (IKs) (33). Missense mutations in Kv7.1 lead to reduced IKs, thus prolonging cardiac repolarization. Mutations in voltage-sensing domains of KCNQ1 are frequently observed in LQTS1 (34). The dynamic balance of this membrane protein is achieved by forward trafficking and retrieval transport. Due to lack of proper forward trafficking, KCNQ1 mutants exhibit reudced presence on the plasma membrane (Figure 2A) (35). Moreover, incomplete removal of the extracellular loop and helix S6 by heterozygous exon 7 deletion mutation in KCNQ1 leads to defective trafficking and APD prolongation (36). IKs is upregulated in response to beta-adrenergic stimulation through the activation of protein kinase A (PKA). Missense mutant A341V-Kv7.1 exhibited attenuated upregulation of IKs after stimulation due to insensitivity to PKA activation (37). Furthermore, missense variants (G189R, R190Q, R243C, and V254M) located in the cytoplasmic loops were found to disrupt PKA regulation of Kv7.1 activity in the context of beta-adrenergic stimulation (38). The extremely rare autosomal recessive Jervell and Lange-Nielsen Syndrome 1 is caused by homozygous or compound heterozygous mutations in KCNQ1 and is characterized by prominent QT prolongation and bilateral sensorineural hearing loss, which is a consequence of complete loss of IKs (39, 40).
Figure 2. LQTS pathogenic hotspots in the subunits of representative ion channels including Kv7.1, Kv11.1, Nav1.5, and Cav1.2.
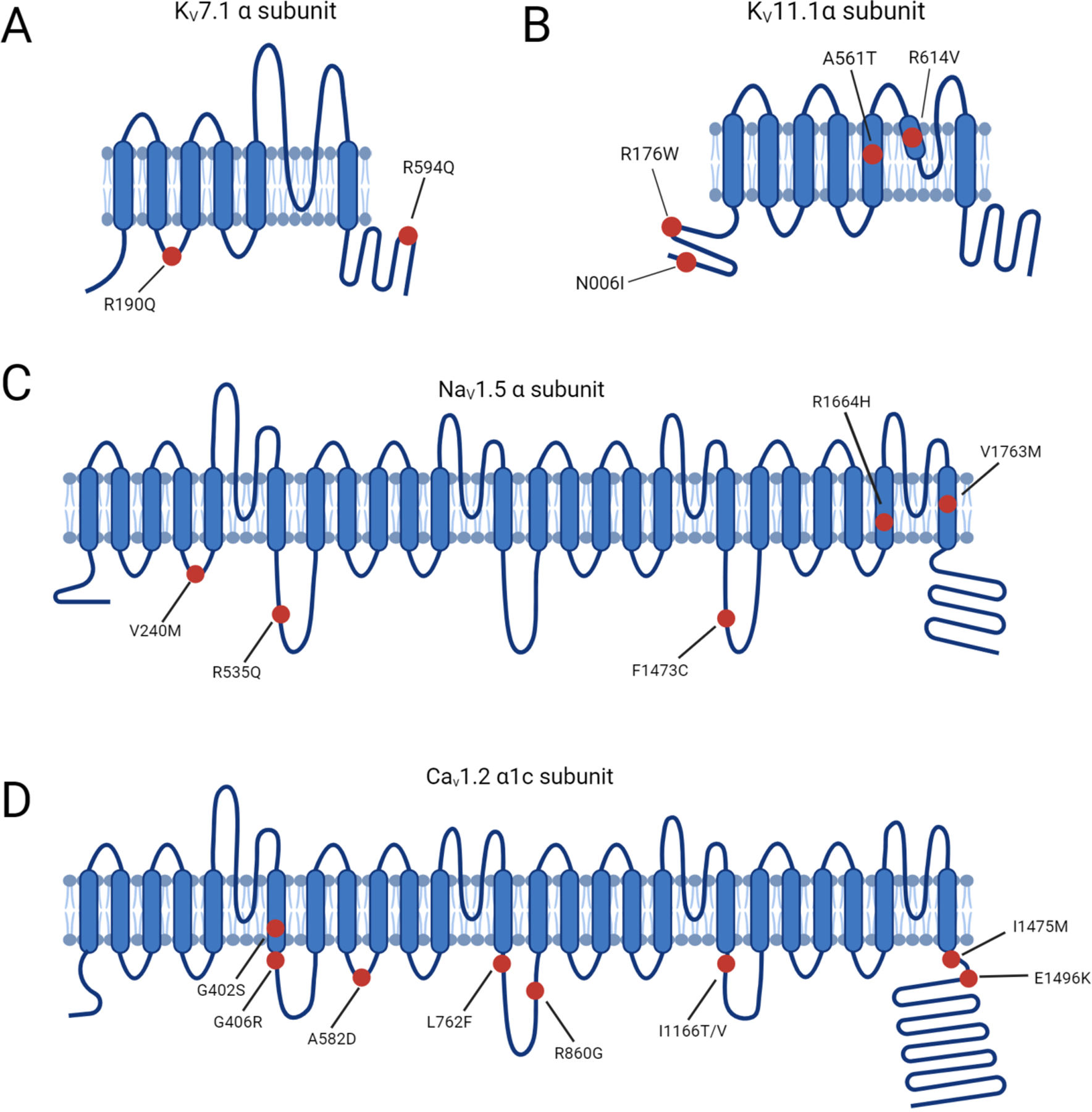
(A) Predicted topology of Kv7.1 α subunit with the location of mutations associated with LQTS1. (B) Predicted topology of Kv11.1 α subunit with mutation sites related to LQTS2. (C) Predicated topology of Nav1.5 α subunit with the location of genetic mutations associated with LQTS3. (D) Predicted topology of Cav1.2 α1c subunit with the mutation positions related to LQTS8. Red dots indicate potential mutation positions in the respective ion channel proteins.
LQTS2 accounts for 25%−30% of all LQTS cases and is caused by mutations in KCNH2, which encodes α-subunit of voltage-gated potassium channel Kv11.1. KCNH2 contributes to the rapid delayed rectifier potassium currents (IKr) (33). KCNH2 mutations are categorized according to the molecular mechanism by which they reduce IKr (41). Class 1 is comprised of nonsense mutations that impair the production of the Kv11.1 channel subunits by nonsense mediated RNA decay (42). Class 2 consists of missense mutations that disrupt the intracellular transport of full-length Kv11.1 to the cell plasma membrane (43). Abnormal Kv11.1 gating kinetics are identified in class 3 mutations, which are predicted to decrease the open probability in repolarization phase (44). Permeation of Kv11.1 is impaired in class 4 mutations due to a presumable blockade of K+ selectivity pore, leading to a diminished IKr (43). Heterozygous KCNH2 mutations exert a dominant-negative effect on KCNH2 channel-associated IKr by interfering with trafficking and impairing the heterodimer assembly (Figure 2B) (45). Mutations within the Kv11.1 lead to reduced IKr, thus prolonging the repolarization process.
LQTS3 is attributed to gain-of-function mutations in SCN5A, which encodes α-subunit of cardiac sodium channel Nav1.5 and mediates the sodium current underlying the depolarization phase of the cardiac AP (46). A broad spectrum of auxiliary proteins acts as part of a macromolecular complex that controls Nav1.5 activity (47). Gain-of-function mutations in SCN5A primarily impair cardiac sodium channel inactivation and are associated with LQTS3 (Figure 2C) (48).
2. Minor LQTS Genes
A-kinase anchoring proteins (AKAPs) are scaffolding proteins that propagate signaling molecules and deliver them to downstream sites for modulating phosphorylation. AKAP9-S1570L mutation has been identified in LQTS patients and interferes with KCNQ1 phosphorylation and reduces the sensitivity of IKs channel to cAMP stimulation (49). In the heart, macromolecular complexes controlled by AKAPs coordinate the phosphorylation status of L-type calcium channel (50) and slow delayed rectifier IKs channel (51).
CACNA1C mutations outside of exon 8 have been reported in non-syndromic LQTS patients (52). Substitutions at positions 582, 762, and 860 slow the inactivation process of Cav1.2, thus increasing the amplitude of ICa,L (Figure 2D) (53, 54). Changes in the activation of Cav1.2 triggered by mutations at 1166, 1475, and 1496 positions also enlarge the ICa,L (Figure 2D) (55). Gain-of-function mutations upregulate Cav1.2 activities and delay the repolarization phase, leading to prolonged APD in LQTS.
CAV3 is responsible for caveolin 3 biosynthesis and mutations in CAV3 are seen in LQTS. Caveolin 3 is a vital constituent of caveolae that are embedded in the plasma membrane and involved in endocytosis and lipid homeostasis. Caveolin 3 is coupled to the α subunit of Nav1.5, which regulates the depolarizing INa current. Disruption of the association between caveolin 3 and Nav1.5 leads to CAV3-LQTS and sudden infant death (56–58). Previous studies on the downregulation of KCNH2-mediated IKr under low extracellular K+ suggest that caveolin proteins are required in the endocytosis of KCNH2 (59).
Mutations in KCNE1 accounts for less than 1% of known LQTS cases (60). KCNE1 regulates both the voltage-gated slowly activating potassium channel and the rapidly activating potassium channel (61, 62). KCNE1 mutations may cause sensorineural hearing loss in Jervell and Lange-Nielsen Syndrome 2 (63). A common variant (D85N) in KCNE1 (64) delays IKs channel opening in Xenopus oocytes and displays severe loss-of-function effects on IKs and IKr in CHO cells (65). A considerable extension of QT interval is also associated with the KCNE1 variant D85N. Moreover, the D85N variant is found in severe LQTS3 patients and some individuals with drug-induced LQTS (66).
KCNE2 encodes an accessory subunit (β-subunit) that acts as a modulator of a variety of voltage-gated potassium channels (67). Recent studies indicate that rare loss-of-function KCNE2 variants are likely to confer proarrhythmic susceptibility instead of inducing LQTS6 phenotype (68).
KCNJ5 encodes cardiac G-protein-coupled inward rectifier potassium channel 4 (Kir3.4) (69). LQTS patients with KCNJ5 mutations show peak QT interval prolongation, a right-skewed T wave, and abnormal repolarization durations (70). Kir3.4 forms a heterodimer with Kir3.1 to maintain the resting membrane potential and contributes to the last repolarization phase. Intriguingly, loss-of-function in atria-specific Kir3.4 induces atrial fibrillation and supraventricular arrhythmia (71).
SCN4B encodes the sodium voltage-gated channel β-subunit 4, which is required for the regulation of Nav1.5 sarcolemmal expression and gating. SCN4B mutations in LQTS can significantly increase the late sodium current in vitro (72).
SNTA1 encodes the membrane adaptor protein α-1-syntrophin. Two SNTA1 mutants (A390V and A257G) can increase the late sodium current and are associated with LQTS phenotypes (73, 74). Nav1.5 is regulated by a dystrophin complex that involves the α-1-syntrophin (75). Early studies uncover a functional complex including α-1-syntrophin, neuronal nitric oxide synthase (nNOS), and calcium ATPase subtype 4b (PMCA4b) (76). The following studies demonstrate that α-1-syntrophin connects nNOS and PMCA4b to Nav1.5 in the heart (77). These studies indicate that α-1-syntrophin is a novel regulator of sodium current.
TRDN encodes Triadin that is involved in calcium release regulation and excitation-contraction coupling in cardiomyocytes. Loss-of-function mutations in TRDN are linked to LQTS phenotypes in five subjects with homozygous or compound heterozygous frameshift mutations (27).
3. Multisystem LQTS genes
Ankyrin-B syndrome is caused by heterozygous loss-of-function mutations in ANK2 (78) which encodes the ubiquitous ankyrin-B adaptor protein. Ankyrins are essential for the cellular organization of various ion channels and transporters (79). Mutations in ANK2 that cause both QT prolongation and sinus bradycardia are found in both large families and sporadic patients with LQTS3 (80, 81).
Anderson-Tawil Syndrome is associated with mutations in KCNJ2, which encodes Kir2.1 and contributes to IK1 (82). The majority of KCNJ2 mutations in Anderson-Tawil Syndrome result in dominant-negative inhibition of Kir2.1 and diminished IK1 amplitude (83, 84).
Timothy Syndrome results from rare single amino acid substitutions in the exon 8 or alternatively spliced exon 8A of Cav1.2 which is encoded by CACNA1C. Specifically, mutations that replace either glycine residue 402 or 406 with large bulky side chains interfere with normal deactivation process (Figure 2D) (85).
Severe recurrent infantile cardiac arrest syndrome is associated with mutations in calmodulin. Calmodulin is a sensor for the intracellular calcium level, which is encoded by three genes: CALM1, CALM2, and CALM3. Calmodulin activities play a significant role in the modulation of Cav1.2 function. Dysfunctional calmodulin activities caused by mutations in CALM genes are associated with a type of arrhythmogenic syndromes in LQTS (86). Patients with LQTS were shown to have a variety of mutations (D130G, F142L, and E141G in CALM1; D96V, N98I, N98S, D130G, D130V, D132E, D134H, and Q136P in CALM2; and D130G and N138K in CALM3) (86–88). Most mutations are located in calmodulin’s C-terminal Ca2+-binding domains (EF-hands III and IV), particularly in the residues directly involved in Ca2+ binding. A plethora of evidence indicate that LQTS relevant calmodulin mutations attenuate Ca2+ binding affinity and result in a defective Ca2+-dependent inactivation of Cav1.2, causing an increased and uncontrolled Ca2+ influx (88, 89). The mutation locations in CALM1–3 reveals special topological domains that are susceptible to genetic variation and imply the significance of Ca2+-binding affinity for proper function of calmodulin. Table1 summarizes the key findings for all-abovementioned LQTS associated genes.
Table 1.
Genetic basis of LQTS subtypes.
| Gene (Genotype) | Gene loci | Protein | Gain/Loss Function | Frequency | Functional effect | References |
|---|---|---|---|---|---|---|
| Long QT syndrome (Major) | ||||||
| KCNQ1 (LQTS1) | 11p15.5 | Kv7.1 α subunit | Loss | 30%−35% | Reduced IKs | (39, 162) |
| KCNH2 (LQTS2) | 7q35–36 | Kv11.1 α subunit | Loss | 25%−30% | Reduced IKr | (163) |
| SCN5A (LQTS3) | 3p21–3p | Nav1.5 α subunit | Gain | 5%−10% | Increased INa | (159) |
| Long QT syndrome (Minor) | ||||||
| AKAP9 (LQTS11) | 7q21–22 | Yotiao | Loss | <1% | Reduced IKs | (49) |
| CACNA1C (LQTS8) | 12p13.3 | Cav1.2 α1c subunit | Gain | <1% | Increased ICa,L | (52) |
| CAV3 (LQTS9) | 3p25 | Caveolin 3 | Loss | <1% | Increased INa | (57) |
| KCNE1 (LQTS5) | 21q22.1 | Kv7.1 β subunit | Loss | <1% | Reduced IKs | (164) |
| KCNE2 (LQTS6) | 21q22.1 | Kv11.1 β subunit | Loss | <1% | Reduced IKr | (165) |
| KCNJ5 (LQTS13) | 11q24.3 | Kir3.4 α subunit | Loss | <1% | Reduced IK,ACh | (69) |
| SCN4B (LQTS10) | 11q23.3 | Nav1.5 β4-subunit | Loss | <1% | Increased INa | (72) |
| SNTA1 (LQTS12) | 20q11.2 | Syntrophin-α1 | Loss | <1% | Increased INa | (73) |
| TRDN | 6q22.31 | Triadin | Loss | Vary rare | N/A | (27) |
| Ankyrin-B Syndrome | ||||||
| ANKB (LQTS4) | 4q25–27 | Ankyrin B | Loss | <1% | Abnormal ion channel localization | (80) |
| Anderson-Tawil Syndrome | ||||||
| KCNJ2 (LQTS7) | 17q23 | Kir2.1 α subunit | Loss | <1% | Reduced IK1 | (82) |
| Jervell and Lange-Nielsen Syndrome | ||||||
| KCNQ1 | 11p15.5 | Kv7.1 α subunit | Loss | Vary rare | Reduced IKs | (40) |
| KCNE1 | 21q22.1 | Kv7.1 β subunit | Loss | Vary rare | Reduced IKs | (166) |
| Timothy Syndrome | ||||||
| CACNA1C | 12p13.3 | Cav1.2 α1c subunit | Gain | Vary rare | Increased ICa,L | (29) |
| Recurrent Infantile Cardiac Arrest Syndrome | ||||||
| CALM1 | 14q24-q31 | Calmodulin | Gain | <1% | Abnormal Ca2+ signaling | (87) |
| CALM2 | 2p21.1-p21.3 | Calmodulin | Gain | <1% | Abnormal Ca2+ signaling | (87) |
| CALM3 | 19q13.32 | Calmodulin | Gain | <1% | Abnormal Ca2+ signaling | (167) |
Modeling LQTS using patient-specific iPSCs
Before the advent of the iPSC techniques, researchers mainly relied on heterologous expression systems using transformed cell lines (e.g., HEK293, CHO cells) with transgenic expression of human ion channel genes. Because these transformed cells are not relevant to human cardiomyocytes, phenotypes in heterologous expression systems could be significantly different from cardiac phenotypes in clinical manifestations. In addition, genotype-phenotype correlation is challenged due to the splicing variance in human cardiomyocytes (90, 91) and absence of genetic modifiers in the heterologous system (92, 93). Human iPSC-derived cardiomyocytes (iPSC-CMs) can overcome these challenges as they are patient-specific and clinically relevant. Human iPSC-CMs can be used to customize medical treatment and quickly test hundreds of new therapeutic molecules for cardiovascular disease (94, 95). A large population of human iPSC-CMs were exploited to serve as a reliable electrophysiological model for Comprehensive in vitro proarrhythmia assay (96). Moreover, patient-specific iPSCs could be employed to stratify subpopulations who may respond to treatment and receive the benefits of medicine while experiencing minimal side effects (97).
As most LQTS is caused by the cell-autonomous pathology of cardiomyocytes, iPSC-CMs are able to recapitulate abnormal electrophysiological features of LQTS-associated ion channels in the dish and provide a versatile platform for cardiac toxicity testing and novel drug discovery. Combined with CRISPR/Cas9 genome editing tools, LQTS-causing genetic variants can be corrected in diseased iPSC-CMs to study phenotype-genotype interactions using patient-specific cardiomyocytes (98). Table 2 summarizes the cellular phenotypes of major subtypes of LQTS that have been recapitulated using patient-specific cardiomyocytes.
Table 2.
Summary of studies on modeling LQTS using patient-specific iPSCs.
| Gene (Genotype) | Cellular Phenotype | References |
|---|---|---|
| KCNQ1 (LQTS1) | Reduced IKs, APD prolongation, relief of the diseased phenotype by β blockers, restored IKs function by LUF7346 | (35, 110) |
| KCNH2 (LQTS2) | Reduced IKr, APD prolongation, EADs, LUF7346 restored the IKr activation | (102–104, 110) |
| SCN5A (LQTS3) | APD prolongation, delayed INa inactivation, expedited recovery from INa inactivation | (46, 48, 106, 168) |
| KCNE1 | Reduced IKs | (64, 65, 169) |
| KCNE2 | Reduced IKr | (165, 170) |
| KCNJ2 (Anderson-Tawil Syndrome) | Severe arrhythmic issues, abnormal intracellular Ca2+ release, flecainide attenuated the symptoms of arrhythmia and recovered the Ca2+ handling | (171) |
| CACNAC1 (Timothy Syndrome) | APD prolongation, aberrant Ca2+ release, unusual and slow contraction | (107) |
| KCNJ5 | Reduced IK,ACh | (69) |
| CALM1 (Recurrent Infantile Cardiac Arrest Syndrome) | APD prolongation, deficient ICa,L inactivation, verapamil rescued the abnormal repolarization, altered rate-dependency, and response to isoproterenol. | (108) |
| CALM2 (Recurrent Infantile Cardiac Arrest Syndrome) | APD prolongation, irregular ICa,L inactivation, descending Ca2+/Calmodulin-dependent inactivation of ICa,L | (109, 172) |
Patient-specific iPSCs have been utilized to create a platform where pathogenesis of LQTS is investigated in vitro. One advantage of patient-specific iPSC-CMs is the ability to evaluate the impact of genetic abnormality on the phenotypic reflection of an individual. For instance, iPSC-CMs could recapitulate most of electrophysiological characteristics seen in LQTS patients. The opportunity to investigate functions of mutated proteins directly in LQTS patient-derived cardiomyocytes could provide novel insights into molecular basis of LQTS for novel drug discovery.
Moretti et al. established a patient iPSC-based platform for modeling LQTS1 (35). In iPSC-CMs derived from LQTS1 patients carrying KCNQ1-R190Q mutations, ventricular-, atrial-, and nodal-like cells were categorized by specific myocyte lineage markers MLC2v, MLC2a, and HCN4, respectively. The KCNQ1-R190Q mutation significantly reduced IKs peak and tail currents, thus prolonging APD in iPSC-CMs. Mutations in Arginine 190 led to abnormal expression of Kv7.1 in the plasma membrane in the KCNQ1-R190Q carrier (99, 100). In addition, KCNQ1 mutation altered the IKs amplitude by adjusting the sensitivity to adrenergic activation (35). β-adrenergic agonist isoproterenol increased the IKs current in healthy iPSC-CMs, which was abrogated in LQTS1 cardiomyocytes. Early after-depolarizations (EADs) were elicited in LQTS1 cardiomyocytes in the presence of isoproterenol, which recapitulated clinical observations in LQTS1 patients who are vulnerable to emotional arousal or exercise (7). As β-blockers are predominantly prescribed to treat arrhythmic symptoms, LQTS-specific iPSC-CMs in the dish also phenocopy the beneficial effects of β-blockers in suppressing arrhythmias (101).
Itzhaki et al. generated an iPSC model from dermal fibroblasts of a female LQTS2 patient with the KCNH2-A614V mutation (102). KCNH2-A614V iPSC-CMs had prolonged repolarization and higher susceptibility to the development of EADs. IKr amplitude was reduced in KCNH2-A614V iPSC-CMs compared to healthy cardiomyocytes. Matsa et al. reported an iPSC model of a symptomatic and an asymptomatic carrier of the KCNH2-A561T mutation (103). A prolonged AP was found in atrial-like and ventricular-like cardiomyocytes but not in pacemaker cells differentiated from KCNH2-A561T iPSCs. Lahti et al. generated iPSC-CMs from an asymptomatic carrier with R176W mutation in KCNH2 and ventricular-like cardiomyocytes showed prolonged APD (104). Point mutations (A614V, A561T, and R176W) in KCNH2 interfered with channel complex assembly and chaperone interaction, resulting in abnormal degradation or defective transport to the plasma membrane (102–104). Additionally, Chai et al. identified modifier genes responsible for differential severity in an LQTS2 family (105). Lower IKr and augmented ICa,L amplitude were observed in iPSC-CMs derived from symptomatic patients with a KCNH2-R752W mutation.
Fatima et al. reported the generation of iPSCs from two LQTS3 patients with V240M or R535Q mutations in SCN5A (46). APD was longer in LQTS3 iPSC-CMs compared to healthy controls. Electrophysiological profiling revealed that Nav1.5 took a longer time to reach inactivation in LQTS3 iPSC-CMs, which contributed to the prolonged APD. Terrenoire et al. derived iPSC-CMs from a proband with an F1473C mutation in SCN5A, which was located in the channel inactivation gate (106). The F1473C mutation increased late sodium current and delayed repolarization, leading to an extended QT interval and a higher susceptibility to arrhythmia. They also found that the sodium channel blocker mexiletine exhibited stronger inhibitory activities when applying a higher pacing rate to iPSC-CMs. Together, these findings support patient-specific iPSC models for studying pathophysiology of LQTS1–3 that are associated with mutations in potassium and sodium channels in cardiomyocytes, and for developing patient- and disease-specific drug treatment.
Yazawa et al. examined iPSC-CMs originally derived from two LQTS8 patients (107). LQTS8 iPSC-CMs showed an abnormal delay in ICa,L inactivation, and prolonged Ca2+ transients. The APs were three times longer in LQTS8 ventricular-like iPSC-CMs than healthy controls. Roscovitine enhanced the inactivation of the voltage-dependent Ca2+ channel and restored the regular Ca2+ transients in LQTS8 iPSC-CMs (107). Dysfunction in CALM1-F142L is a major cause of LQTS14 which is characterized by relapsed cardiac arrests (108). Rocchetti et al. explored effects of heterozygous CALM1-F142L mutations on repolarization frequency, membrane currents, and intracellular Ca2+ oscillations using patient-specific iPSC-CMs. The prolonged APD and altered repolarization frequency were linked to a perturbation of Ca2+-dependent inactivation of ICa,L. In addition, another group created iPSCs from an LQTS15 patient with CALM2-D130G mutation (109). Intracellular Ca2+ handling in LQTS15 iPSC-CMs were abrogated and Ca2+-dependent inactivation of ICa,L was reduced. Furthermore, they employed CRISPR genome editing to selectively inhibit the mutant allele and rescued Ca2+ handing abnormalities in LQTS15 iPSC-CMs.
In summary, LQTS patient-specific iPSCs can be used for testing drug efficacy and toxicity in a disease-specific context. Numerous regimens with the potential to rectify the disease phenotype have been investigated using LQTS2-specific iPSC-CMs. Sala et al. exploited patient-specific iPSC-CMs to examine the effects of KCNH2 selective activator on both acquired and congenital LQTS. The small chemical LUF7236 restored IKr by delaying the KCNH2 channel deactivation (110). In some instances, nicorandil, PD118057, and ICA-105574 treatment in LQTS2 iPSC-CMs resulted in APD shortening and EAD elimination (111, 112), which is consistent with clinical regimens. In addition, LQTS-specific iPSC-CMs have tremendous potential in evaluating the likelihood of regimens on restoring cardiac abnormalities, allowing for quick identification of unanticipated toxicity and laying the foundation for risk prediction. LQTS iPSC-CMs from an individual carrying a KCNH2-R165W mutation exhibited more sensitive to arrhythmogenic medications compared to healthy controls (104). Significant APD elongation and decreased IKs amplitude were observed in LQTS1 iPSC-CMs exposed to E-4031, which was manifested by the formation of new EADs (35). These findings suggest greater sensitivity to malignant arrhythmias in response to culprit drugs in LQTS, which might substantially suppress the remaining IKr level (102). Therefore, modeling LQTS and testing new drugs using patient-specific iPSC-CMs could be a big step forward in the realm of personalized health care.
Treatment strategies for LQTS
Beta-blockers are the mainstream drugs of choice for LQTS. A study conducted on LQTS1 and LQTS2 patients showed that not all beta-blockers are equally effective (113). One possible explanation is that ion channels exhibit varying degrees of pharmacological response to beta-blocker therapies. Propranolol is widely considered as the most effective drug, owing to sodium channel inhibitory activity with a meager impact on potassium channels (113). Additionally, the precise nature of the genetic mutation in ion channel genes may be a determinant of the efficacy of beta-blocker therapy for LQTS (114). For example, LQTS1 patients who have a missense mutation appear to benefit more from beta-blocker medication than those who have other genetic variants. Besides beta-blockers, the sodium channel blocker mexiletine may also lessen the recurrence of cardiac events in LQTS2 patients (115). Combination treatment with mexiletine resulted in a significant reduction in polymorphic ventricular tachycardia and no syncope episodes occurred after the treatment. LQTS3 patients rely on pharmacological treatment (e.g. beta-blockers, sodium channel blockers), ICD, and pacemaker treatment (116). Specifically, beta-blockers are routine regimens, and ICD is suggested for those at a higher risk. Pacemaker treatment is for LQTS3 patients with symptomatic bradycardia as well as QT lengthening who don’t respond to regular therapeutics.
In patients with Anderson-Tawil Syndrome, beta-blockers are commonly prescribed even without reducing the frequency of ventricular ectopy. Flecainide can mitigate the arrhythmic symptoms of certain Anderson-Tawil Syndrome patients by activating IK1 (117, 118). However, amiodarone and acetazolamide are more effective in Anderson-Tawil Syndrome patients with a heterozygous R218W mutation in KNCJ2 (119). Verapamil, a Cav1.2 blocker, is shown to effectively restrain the bidirectional ventricular tachycardia in Anderson-Tawil Syndrome (120). Increasing the potassium concentration in the blood is beneficial in controlling ventricular arrhythmias in Anderson-Tawil Syndrome (121).
ICD implantation is a standard approach for CACNA1C-LQTS patients. Numerous medications with the potential to prevent CACNA1C-LQTS arrhythmias have been explored, including nifedipine, pinacidil, and ranolazine (122). Owing to Ca2+ influx through L-type Ca2+ channels, suppression of ICa,L by nifedipine was postulated to be anti-arrhythmic. One alternative strategy to alleviate arrhythmia is to raise the repolarization currents with the KATP channel opener pinacidil. Both therapies reduced AP and eliminated the proclivity to EAD. Ranolazine, a sodium channel blocker, suppressed EADs without affecting the APD. Inhibition of the activity of L-type Ca2+ channels by verapamil can attenuate ventricular arrhythmia in LQTS8 through shortening the repolarization period (123).
Left cardiac sympathetic denervation (LCSD) is an effective, minimally invasive anti- fibrillatory treatment for individuals with LQTS (124). LCSD is advised for individuals with LQTS who are suffering breakthrough cardiac episodes. Patients who are unable to tolerate the adverse effects of beta blockers may benefit from supplementary treatment with LCSD. Recent studies on LCSD revealed that LCSD monotherapy exhibited superior efficacy, which is supposed to push it forward into a guideline-directed therapy (125)
Novel drug discovery using the patient-specific iPSC platform would drive the next-generation precision treatment regime for LQTS. Voltage-sensitive dyes can be applied to 2D iPSC-CMs monolayer to assess action potentials under certain drug treatments. To evaluate whether a new drug could cause potential ventricular arrhythmia, comprehensive in vitro proarrhythmia assay (CiPA) using patient-specific iPSC-CMs has been used for testing drug-induced arrhythmias (126, 127). A non-invasive platform called multi-electrode array (MEA) is also available for extensive electrophysiological profiling of human iPSC-CMs under different drug conditions (128, 129). Taking advantage of patient-specific iPSC-CMs and cutting-edge techniques would facilitate novel drug screening and discovery process for treating LQTS.
Precision medicine in LQTS
Precision medicine is a multidisciplinary approach to the prevention and treatment of cardiovascular disease while integrating various individuals’ genetic profiles, environmental cues, and lifestyles that are factors influencing disease phenotypes (130). This approach addresses the constraints of reductionism in medicine, which assumes that all patients with the same symptoms have the identical phenotype. Precision medicine utilizes omics approaches (proteomics, transcriptomics, metabolomics, etc.) to interpret clinical data and further characterize the phenotype (131, 132). These phenotypic data can subsequently be evaluated using molecular interaction frameworks to identify previously unknown clinical phenotypes and disease connections and to choose the most effective therapeutics.
Precision medicine is involved in the accurate diagnosis approach of LQTS. The recently developed AI-ECG model was able to differentiate individuals with electrocardiographically concealed LQTS from the healthy cohorts. Several limitations have been considered in applying AI-enabled tools for a better interpretation of clinical data, such as ECG. The strongest illustration of how errors may occur in an AI-driven approach is overfitting, which occurs when there are excessive features compared to the quantity of data in the training set (133). The AI-driven approach performs extremely well on the training set but badly on the test set with low generalizability because of overfitting. There are additional clinical and technical obstacles to the implementation of AI–ECG in patient care. AI analysis depends on standardized digital ECG sampling, which is not ubiquitously accessible even in high-resource clinical situations (134). AI essentially relies on the integrity of data. Poor-quality data will disable appropriate training and validation on AI algorithms, precluding the development of AI-enabled techniques (135).
To implement precision medicine in cardiovascular disease field, it is necessary to establish a modeling platform to test various strategies. Human iPSC-CMs act as a disease model in a dish by keeping the patient’s genetic traits and recapitulating the disease phenotypes in vitro. Human iPSCs are capable of being differentiated into cardiomyocytes (136, 137). Current protocols provide simplified roadmaps for generating a variety of cardiomyocytes to characterize the channelopathies (138, 139). Consequently, translational approaches using iPSC technology have implemented these cellular models for elucidating novel mechanisms of disease (102, 111, 140–143). Emerging CRISPR/Cas9 genome editing techniques enable researchers to distinguish the impact of variants for monogenic disorders, such as cardiac channelopathies (144). Two strategies were developed to resolve the confounding factors behind the mutation: (1) Insert the variant allele into a wild-type cell line to validate whether the disease phenotype is attributed to the variant; (2) Correct the sequence of the variant in a patient-specific iPSC line to determine whether a corrected genetic variant reverses the diseased phenotype (Figure 3). In these complementary approaches, each genome-edited cell line serves as the isogenic control compared to the unedited parental line to identify the causative variant (145). Additionally, isogenic controls are beneficial in eliminating the intrinsic variability displayed by iPSC-CMs in various healthy and diseased individuals (145). iPSC-CMs provide an exceptional patient-specific model to study the underlying mechanisms of multiple diseases. However, several disadvantages have yet to be overcome. Human iPSC-CMs exhibit similar properties to neonatal stage cardiomyocytes in terms of morphology and protein expression profiles (146, 147). Although iPSC-CMs sufficiently recapitulate the phenotype of many inherited heart defects, they may not mature enough to understand the fundamental mechanisms underpinning inherited LQTS. In addition, epigenetic modifications are altered in the iPSC reprogramming process (148) regardless of the rudimentary epigenetic memory of the original somatic cells, which eliminate the contribution of personal lifestyles, or medication usage vital to reconstruct the manifestation of disease phenotypes.
Figure 3. Patient-specific iPSCs take the lead for precision medicine in LQTS.
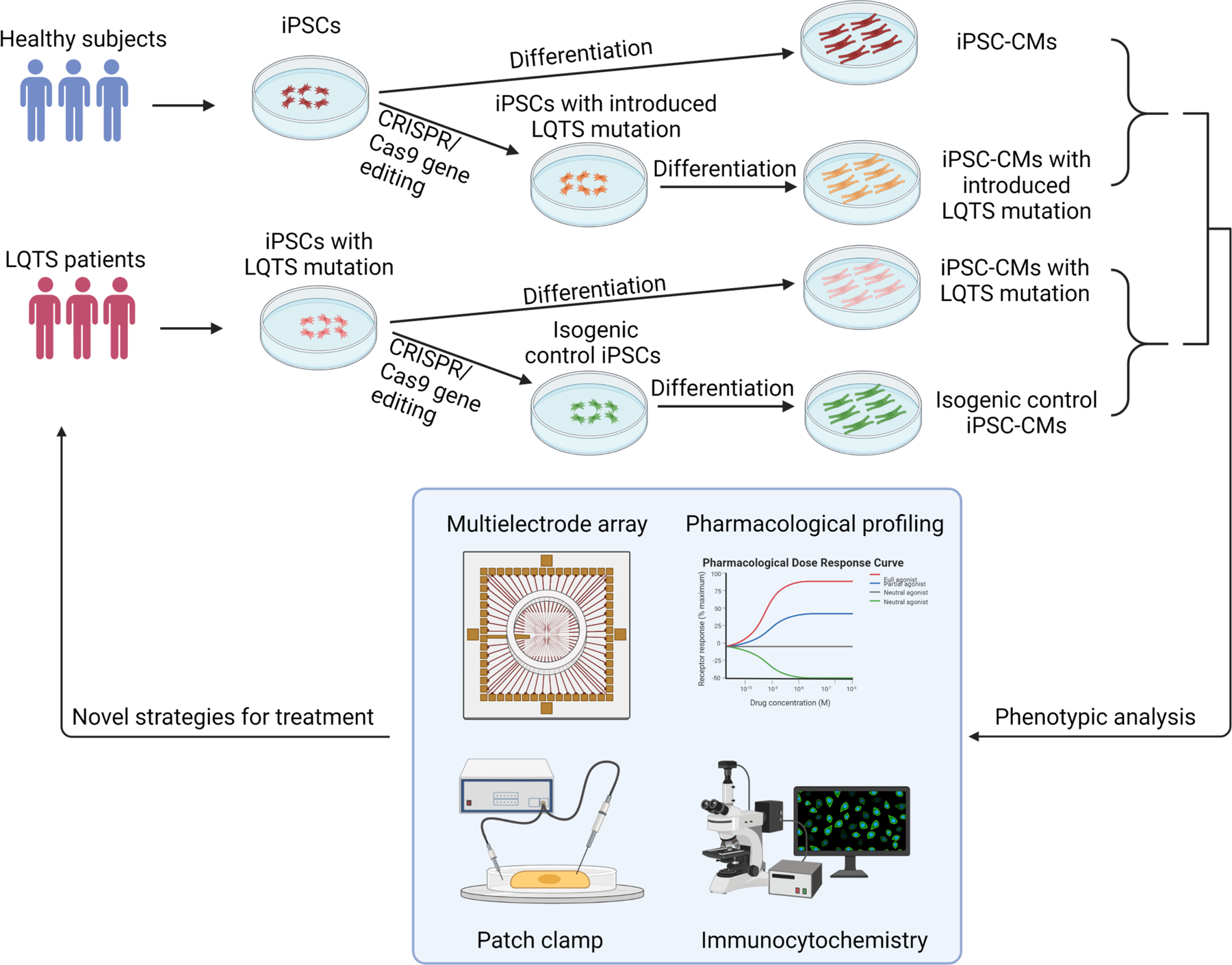
Blood samples are collected for generation of patient-specific iPSCs. Pathogenic genetic variants are identified through whole-genome sequencing. CRISPR/Cas9 mediated genome editing can either introduce or correct a mutation in patient-specific iPSCs to study genotype-phenotype interactions. Deep phenotyping of LQTS iPSC-derived cardiomyocytes include multielectrode arrays, pharmacological profiling, electrophysiological characterization, and immunocytochemistry. Preclinical testing in LQTS patient-specific cardiomyocytes will guide clinicians for precise stratification and treatment of LQTS.
Precision medicine aims to administer the most appropriate treatment strategy for an individual based on their unique personal profiles. To incorporate complex data sets (i.e., next-generation sequencing, omics, clinical tests) into precision medicine, advanced biological systems are essential to interpret data and further provide guidance for identifying physiological and pathological factors. Precision medicine approaches to treating LQTS patients should consider various factors, including age, gender, electrolytes, QT-prolonging drugs, etc. Among LQTS1 patients, male adolescents are prone to develop initial cardiac issues earlier than female adolescents (149). However, female adults are at higher risk than male adults in encountering the first cardiac event. In LQTS2 pathogenic subjects, the mortality rate remains higher in women than men (24). Extracellular K+ concentration contributes to the regulation of QT duration in healthy individuals. Low serum K+ levels serve as a highly susceptible factor in predisposing arrhythmic death (150). Increased serum K+ concentration by oral uptake and intravenous injection is beneficial in shortening QT interval in LQTS patients (151, 152). The QT interval measures the duration of cardiac repolarization, which is primarily determined by equilibration of influx of sodium and calcium ions as well as efflux of potassium ions. QT prolongation is caused by an increase in inward current or a decrease in outward current. The suppression of the outward IKr, which prolongs individual cardiac ventricular APDs and ECG, is thought to be the mechanism by which medications prolong the QT interval (153). Ongoing pharmacogenetics (154) and pharmacogenomics (155) studies on drug-induced LQTS will probably lead to a stratified and customized strategy for treating acquired LQTS.
RNA interference (RNAi) and CRISPR genome editing have been used for treating LQTS in vitro. To circumvent the dominant-negative effect of KCNQ1 mutations in LQTS1, researchers developed a dual-component RNAi method that allows to knock down the mutant KCNQ1 transcript while leaving the wild-type KCNQ1 transcript intact (156). They further examined this RNAi-mediated strategy in rescuing KCNQ1 function in both TSA201 cell line and human iPSC-CMs to pave the way for completely repairing dysfunctional mutants in LQTS1. Lu et al. demonstrated that siRNA knockdown of KCNH2-E637K mutation restored normal KCNH2 localization to the membrane in cells co-expressing the KCNH2-E637K mutant and recovered the normal amplitudes of maximum current and tailed current (157). In another study, impaired channel trafficking to plasma membrane was found in LQTS2 iPSC-CMs with a KCNH2-A561T mutation, which could be rescued by a mutant allele-specific siRNA knockdown (103). In addition, CRISPR/Cas9-based genome editing was employed to treat LQTS. Limpitikul et al. first generated iPSC-CMs from an LQTS patient carrying CALM2-D130G mutation and subsequently characterized the electrophysiological properties of these iPSC-CMs (109). Moreover, they utilized CRISPR interference (CRISPRi) to specifically knockdown the mutant CALM2-D130G transcript and achieved a functional rescue in patient-specific iPSC-CMs. In summary, both RNAi and CRISPRi hold great promise for precisely treating LQTS patients.
The combination of patient-specific iPSC-CMs and genome editing has advanced the identification of purported modifier genes involved in shaping the phenotype of LQTS1 (158) and LQTS2 (105). However, numerous intrinsic restrictions exist when searching for a single causative gene in certain LQTS cases. The primary challenge is to establish the genotype-phenotype link. For instance, mutations in SCN5A were previously identified as a single causative gene for congenital LQTS (159). However, additional abnormalities associated with SCN5A mutations have been identified in Brugada syndrome (160) and dilated cardiomyopathy (161). The second challenge is the low penetrance in pedigrees with familial LQTS (4). When determining penetrance, mutation screening in the family members of the proband is the most effective method. Low penetrance could implicate other factors including genetic variants or environmental cues on LQTS pathogenesis. Therefore, an increasing number of LQTS-associated variants poses potential challenges to developing precision medicine for each LQTS subtype. Enhanced understanding of LQTS pathophysiology has led to the development and production of genotype-specific therapies (156). RNAi and CRISPRi mediated approaches have been shown to be effective in vitro to shorten the prolonged APD (109, 156, 157). Although development of genotype-specific pharmacotherapies may overcome underlying molecular defects of LQTS, the complexities of gene-specific therapeutic approaches may exceed the initial expectations and may not become available to all LQTS patients. With enough scientific input, a balance between “one therapy fits all” and “personalized treatment for each individual patient” is likely to be accomplished using patient-specific iPSC-CMs. This “personalized treatment” strategy contrasts with the “one therapy fits all” approach, in which diseases treatment and preventive measures are established based on the outcomes of randomized trials without taking individual differences into account.
Future perspectives
Patient-specific iPSCs and next-generation genome sequencing have significantly advanced the implementation of precision medicine in LQTS, which strives to improve the stratification of the patient population and to identify patient-specific interventions through examining large population datasets and genetic traits of LQTS patients. To implement the precision treatment in LQTS, researchers and doctors need to work together to complete this unprecedented mission. In the future, the genome of individual LQTS patients will be sequenced to identify potential genetic variants associated with QT prolongation (Figure 3). In parallel, their blood samples will be collected for generation of patient-specific iPSC lines and cardiomyocyte differentiation. Together with genomic information and CRISPR genome editing, deep phenotyping using patient-specific iPSC-CMs will be utilized to stratify LQTS patients into multiple subgroups based on their phenotypes in the petri dish. In addition, potential candidate drugs will be administered to patient-specific iPSC-CMs and identify the best candidate for each group of LQTS patients to restore normal QT prolongation. Finally, machine learning of deep phenotyping of patient-specific cardiomyocyte data, large and diverse population-based clinical database, and genetic traits of LQTS will eventually assign the optimal treatment regime to each LQTS patient for the best outcomes. The future precision medicine will be on the horizon which would be heralded by the forward clinical trial and novel drug testing in a petri dish using LQTS patient-specific iPSCs.
Acknowledgements
We would like to thank Dr. Dennis Lewandowski for critically reading and editing this manuscript. Research in Dr. Ming-Tao Zhao’s lab was supported by the American Heart Association (AHA) Career Development Award 18CDA34110293, NIH/NHLBI R01HL155282, R21HL165406, Additional Ventures Innovation Fund (AVIF), and Single Ventricle Research Fund (SVRF).
Footnotes
Conflict of Interest
None.
References
- 1.Moss AJ (2003) Long QT Syndrome. JAMA 289(16), 2041–4 [DOI] [PubMed] [Google Scholar]
- 2.Roden DM (2008) Clinical practice. Long-QT syndrome. N Engl J Med 358(2), 169–76 [DOI] [PubMed] [Google Scholar]
- 3.Tester DJ and Ackerman MJ (2014) Genetics of long QT syndrome. Methodist Debakey Cardiovasc J 10(1), 29–33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Priori SG, Napolitano C and Schwartz PJ (1999) Low penetrance in the long-QT syndrome: clinical impact. Circulation 99(4), 529–33 [DOI] [PubMed] [Google Scholar]
- 5.Etheridge SP and Cohen MI (2017) An Overview of Diagnosis and Management Strategies for Long QT Syndrome. J Innov Card Rhythm Manag 8(6), 2750–57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schwartz PJ, Crotti L and Insolia R (2012) Long-QT syndrome: from genetics to management. Circ Arrhythm Electrophysiol 5(4), 868–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schwartz PJ, et al. (2001) Genotype-phenotype correlation in the long-QT syndrome: gene-specific triggers for life-threatening arrhythmias. Circulation 103(1), 89–95 [DOI] [PubMed] [Google Scholar]
- 8.Ackerman MJ, Tester DJ and Porter CJ (1999) Swimming, a gene-specific arrhythmogenic trigger for inherited long QT syndrome. Mayo Clin Proc 74(11), 1088–94 [DOI] [PubMed] [Google Scholar]
- 9.Khositseth A, et al. (2004) Identification of a common genetic substrate underlying postpartum cardiac events in congenital long QT syndrome. Heart Rhythm 1(1), 60–4 [DOI] [PubMed] [Google Scholar]
- 10.Perez-Riera AR, et al. (2018) The congenital long QT syndrome Type 3: An update. Indian Pacing Electrophysiol J 18(1), 25–35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ritsema van Eck HJ, Kors JA and van Herpen G (2005) The U wave in the electrocardiogram: a solution for a 100-year-old riddle. Cardiovasc Res 67(2), 256–62 [DOI] [PubMed] [Google Scholar]
- 12.Kanno S and Saffitz JE (2001) The role of myocardial gap junctions in electrical conduction and arrhythmogenesis. Cardiovasc Pathol 10(4), 169–77 [DOI] [PubMed] [Google Scholar]
- 13.Wilde AAM, Amin AS and Postema PG (2022) Diagnosis, management and therapeutic strategies for congenital long QT syndrome. Heart 108(5), 332–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schwartz PJ, et al. (2020) Inherited cardiac arrhythmias. Nat Rev Dis Primers 6(1), 58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moss AJ, et al. (1995) ECG T-wave patterns in genetically distinct forms of the hereditary long QT syndrome. Circulation 92(10), 2929–34 [DOI] [PubMed] [Google Scholar]
- 16.Van Langen IM, et al. (2003) The use of genotype-phenotype correlations in mutation analysis for the long QT syndrome. J Med Genet 40(2), 141–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Modi S and Krahn AD (2011) Sudden cardiac arrest without overt heart disease. Circulation 123(25), 2994–3008 [DOI] [PubMed] [Google Scholar]
- 18.Schwartz PJ and Ackerman MJ (2013) The long QT syndrome: a transatlantic clinical approach to diagnosis and therapy. Eur Heart J 34(40), 3109–16 [DOI] [PubMed] [Google Scholar]
- 19.Priori SG, et al. (2013) HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes: document endorsed by HRS, EHRA, and APHRS in May 2013 and by ACCF, AHA, PACES, and AEPC in June 2013. Heart Rhythm 10(12), 1932–63 [DOI] [PubMed] [Google Scholar]
- 20.Itoh H, et al. (2016) The genetics underlying acquired long QT syndrome: impact for genetic screening. Eur Heart J 37(18), 1456–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bos JM, et al. (2021) Use of Artificial Intelligence and Deep Neural Networks in Evaluation of Patients With Electrocardiographically Concealed Long QT Syndrome From the Surface 12-Lead Electrocardiogram. JAMA Cardiol 6(5), 532–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hermans BJM, et al. (2020) Improving long QT syndrome diagnosis by a polynomial-based T-wave morphology characterization. Heart Rhythm 17(5 Pt A), 752–58 [DOI] [PubMed] [Google Scholar]
- 23.Prifti E, et al. (2021) Deep learning analysis of electrocardiogram for risk prediction of drug-induced arrhythmias and diagnosis of long QT syndrome. Eur Heart J 42(38), 3948–61 [DOI] [PubMed] [Google Scholar]
- 24.Priori SG, et al. (2003) Risk stratification in the long-QT syndrome. N Engl J Med 348(19), 1866–74 [DOI] [PubMed] [Google Scholar]
- 25.Moss AJ, et al. (2007) Clinical aspects of type-1 long-QT syndrome by location, coding type, and biophysical function of mutations involving the KCNQ1 gene. Circulation 115(19), 2481–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Makita N, et al. (2008) The E1784K mutation in SCN5A is associated with mixed clinical phenotype of type 3 long QT syndrome. J Clin Invest 118(6), 2219–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Altmann HM, et al. (2015) Homozygous/Compound Heterozygous Triadin Mutations Associated With Autosomal-Recessive Long-QT Syndrome and Pediatric Sudden Cardiac Arrest: Elucidation of the Triadin Knockout Syndrome. Circulation 131(23), 2051–60 [DOI] [PubMed] [Google Scholar]
- 28.Perez-Riera AR, et al. (2021) Andersen-Tawil Syndrome: A Comprehensive Review. Cardiol Rev 29(4), 165–77 [DOI] [PubMed] [Google Scholar]
- 29.Splawski I, et al. (2004) Ca(V)1.2 calcium channel dysfunction causes a multisystem disorder including arrhythmia and autism. Cell 119(1), 19–31 [DOI] [PubMed] [Google Scholar]
- 30.Adler A, et al. (2020) An International, Multicentered, Evidence-Based Reappraisal of Genes Reported to Cause Congenital Long QT Syndrome. Circulation 141(6), 418–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ingles J and Semsarian C (2020) Time to Rethink the Genetic Architecture of Long QT Syndrome. Circulation 141(6), 440–43 [DOI] [PubMed] [Google Scholar]
- 32.Roberts JD, et al. (2020) An International Multicenter Evaluation of Type 5 Long QT Syndrome: A Low Penetrant Primary Arrhythmic Condition. Circulation 141(6), 429–39 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen L, Sampson KJ and Kass RS (2016) Cardiac Delayed Rectifier Potassium Channels in Health and Disease. Card Electrophysiol Clin 8(2), 307–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Huang H, et al. (2018) Mechanisms of KCNQ1 channel dysfunction in long QT syndrome involving voltage sensor domain mutations. Sci Adv 4(3), eaar2631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moretti A, et al. (2010) Patient-specific induced pluripotent stem-cell models for long-QT syndrome. N Engl J Med 363(15), 1397–409 [DOI] [PubMed] [Google Scholar]
- 36.Ma D, et al. (2015) Characterization of a novel KCNQ1 mutation for type 1 long QT syndrome and assessment of the therapeutic potential of a novel IKs activator using patient-specific induced pluripotent stem cell-derived cardiomyocytes. Stem Cell Res Ther 6(1), 39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Heijman J, et al. (2012) Dominant-negative control of cAMP-dependent IKs upregulation in human long-QT syndrome type 1. Circ Res 110(2), 211–9 [DOI] [PubMed] [Google Scholar]
- 38.Barsheshet A, et al. (2012) Mutations in cytoplasmic loops of the KCNQ1 channel and the risk of life-threatening events: implications for mutation-specific response to β-blocker therapy in type 1 long-QT syndrome. Circulation 125(16), 1988–96 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Giudicessi JR and Ackerman MJ (2013) Prevalence and potential genetic determinants of sensorineural deafness in KCNQ1 homozygosity and compound heterozygosity. Circ Cardiovasc Genet 6(2), 193–200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Splawski I, et al. (1997) Molecular basis of the long-QT syndrome associated with deafness. N Engl J Med 336(22), 1562–7 [DOI] [PubMed] [Google Scholar]
- 41.Delisle BP, et al. (2004) Biology of cardiac arrhythmias: ion channel protein trafficking. Circ Res 94(11), 1418–28 [DOI] [PubMed] [Google Scholar]
- 42.Gong Q, et al. (2007) Nonsense mutations in hERG cause a decrease in mutant mRNA transcripts by nonsense-mediated mRNA decay in human long-QT syndrome. Circulation 116(1), 17–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Anderson CL, et al. (2006) Most LQT2 mutations reduce Kv11.1 (hERG) current by a class 2 (trafficking-deficient) mechanism. Circulation 113(3), 365–73 [DOI] [PubMed] [Google Scholar]
- 44.Chen J, et al. (1999) Long QT syndrome-associated mutations in the Per-Arnt-Sim (PAS) domain of HERG potassium channels accelerate channel deactivation. J Biol Chem 274(15), 10113–8 [DOI] [PubMed] [Google Scholar]
- 45.Mehta A, et al. (2014) Re-trafficking of hERG reverses long QT syndrome 2 phenotype in human iPS-derived cardiomyocytes. Cardiovasc Res 102(3), 497–506 [DOI] [PubMed] [Google Scholar]
- 46.Fatima A, et al. (2013) The disease-specific phenotype in cardiomyocytes derived from induced pluripotent stem cells of two long QT syndrome type 3 patients. PLoS One 8(12), e83005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Abriel H (2010) Cardiac sodium channel Na(v)1.5 and interacting proteins: Physiology and pathophysiology. J Mol Cell Cardiol 48(1), 2–11 [DOI] [PubMed] [Google Scholar]
- 48.Ma D, et al. (2013) Modeling type 3 long QT syndrome with cardiomyocytes derived from patient-specific induced pluripotent stem cells. Int J Cardiol 168(6), 5277–86 [DOI] [PubMed] [Google Scholar]
- 49.Chen L, et al. (2007) Mutation of an A-kinase-anchoring protein causes long-QT syndrome. Proc Natl Acad Sci U S A 104(52), 20990–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hulme JT, et al. (2006) Phosphorylation of serine 1928 in the distal C-terminal domain of cardiac CaV1.2 channels during beta1-adrenergic regulation. Proc Natl Acad Sci U S A 103(44), 16574–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Marx SO, et al. (2002) Requirement of a macromolecular signaling complex for beta adrenergic receptor modulation of the KCNQ1-KCNE1 potassium channel. Science 295(5554), 496–9 [DOI] [PubMed] [Google Scholar]
- 52.Boczek NJ, et al. (2013) Exome sequencing and systems biology converge to identify novel mutations in the L-type calcium channel, CACNA1C, linked to autosomal dominant long QT syndrome. Circ Cardiovasc Genet 6(3), 279–89 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fukuyama M, et al. (2014) Long QT syndrome type 8: novel CACNA1C mutations causing QT prolongation and variant phenotypes. Europace 16(12), 1828–37 [DOI] [PubMed] [Google Scholar]
- 54.Landstrom AP, et al. (2016) Novel long QT syndrome-associated missense mutation, L762F, in CACNA1C-encoded L-type calcium channel imparts a slower inactivation tau and increased sustained and window current. Int J Cardiol 220, 290–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wemhoner K, et al. (2015) Gain-of-function mutations in the calcium channel CACNA1C (Cav1.2) cause non-syndromic long-QT but not Timothy syndrome. J Mol Cell Cardiol 80, 186–95 [DOI] [PubMed] [Google Scholar]
- 56.Cronk LB, et al. (2007) Novel mechanism for sudden infant death syndrome: persistent late sodium current secondary to mutations in caveolin-3. Heart Rhythm 4(2), 161–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vatta M, et al. (2006) Mutant caveolin-3 induces persistent late sodium current and is associated with long-QT syndrome. Circulation 114(20), 2104–12 [DOI] [PubMed] [Google Scholar]
- 58.Arnestad M, et al. (2007) Prevalence of long-QT syndrome gene variants in sudden infant death syndrome. Circulation 115(3), 361–7 [DOI] [PubMed] [Google Scholar]
- 59.Massaeli H, et al. (2010) Involvement of caveolin in low K+-induced endocytic degradation of cell-surface human ether-a-go-go-related gene (hERG) channels. J Biol Chem 285(35), 27259–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Splawski I, et al. (2000) Spectrum of mutations in long-QT syndrome genes. KVLQT1, HERG, SCN5A, KCNE1, and KCNE2. Circulation 102(10), 1178–85 [DOI] [PubMed] [Google Scholar]
- 61.Sanguinetti MC, et al. (1996) Coassembly of K(V)LQT1 and minK (IsK) proteins to form cardiac I(Ks) potassium channel. Nature 384(6604), 80–3 [DOI] [PubMed] [Google Scholar]
- 62.Barhanin J, et al. (1996) K(V)LQT1 and lsK (minK) proteins associate to form the I(Ks) cardiac potassium current. Nature 384(6604), 78–80 [DOI] [PubMed] [Google Scholar]
- 63.Tyson J, et al. (1997) IsK and KvLQT1: mutation in either of the two subunits of the slow component of the delayed rectifier potassium channel can cause Jervell and Lange-Nielsen syndrome. Hum Mol Genet 6(12), 2179–85 [DOI] [PubMed] [Google Scholar]
- 64.Tesson F, et al. (1996) Exclusion of KCNE1 (IsK) as a candidate gene for Jervell and Lange-Nielsen syndrome. J Mol Cell Cardiol 28(9), 2051–5 [DOI] [PubMed] [Google Scholar]
- 65.Nishio Y, et al. (2009) D85N, a KCNE1 polymorphism, is a disease-causing gene variant in long QT syndrome. J Am Coll Cardiol 54(9), 812–9 [DOI] [PubMed] [Google Scholar]
- 66.Westenskow P, et al. (2004) Compound mutations: a common cause of severe long-QT syndrome. Circulation 109(15), 1834–41 [DOI] [PubMed] [Google Scholar]
- 67.Abbott GW (2015) The KCNE2 K+ channel regulatory subunit: Ubiquitous influence, complex pathobiology. Gene 569(2), 162–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Roberts JD, et al. (2017) Loss-of-Function KCNE2 Variants: True Monogenic Culprits of Long-QT Syndrome or Proarrhythmic Variants Requiring Secondary Provocation? Circ Arrhythm Electrophysiol 10(8) [DOI] [PubMed] [Google Scholar]
- 69.Yang Y, et al. (2010) Identification of a Kir3.4 mutation in congenital long QT syndrome. Am J Hum Genet 86(6), 872–80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wang F, et al. (2013) The phenotype characteristics of type 13 long QT syndrome with mutation in KCNJ5 (Kir3.4-G387R). Heart Rhythm 10(10), 1500–6 [DOI] [PubMed] [Google Scholar]
- 71.Calloe K, et al. (2007) Characterizations of a loss-of-function mutation in the Kir3.4 channel subunit. Biochem Biophys Res Commun 364(4), 889–95 [DOI] [PubMed] [Google Scholar]
- 72.Medeiros-Domingo A, et al. (2007) SCN4B-encoded sodium channel beta4 subunit in congenital long-QT syndrome. Circulation 116(2), 134–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ueda K, et al. (2008) Syntrophin mutation associated with long QT syndrome through activation of the nNOS-SCN5A macromolecular complex. Proc Natl Acad Sci U S A 105(27), 9355–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wu G, et al. (2008) alpha-1-syntrophin mutation and the long-QT syndrome: a disease of sodium channel disruption. Circ Arrhythm Electrophysiol 1(3), 193–201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gavillet B, et al. (2006) Cardiac sodium channel Nav1.5 is regulated by a multiprotein complex composed of syntrophins and dystrophin. Circ Res 99(4), 407–14 [DOI] [PubMed] [Google Scholar]
- 76.Williams JC, et al. (2006) The sarcolemmal calcium pump, alpha-1 syntrophin, and neuronal nitric-oxide synthase are parts of a macromolecular protein complex. J Biol Chem 281(33), 23341–8 [DOI] [PubMed] [Google Scholar]
- 77.Oceandy D, et al. (2007) Neuronal nitric oxide synthase signaling in the heart is regulated by the sarcolemmal calcium pump 4b. Circulation 115(4), 483–92 [DOI] [PubMed] [Google Scholar]
- 78.Mohler PJ, et al. (2007) Defining the cellular phenotype of “ankyrin-B syndrome” variants: human ANK2 variants associated with clinical phenotypes display a spectrum of activities in cardiomyocytes. Circulation 115(4), 432–41 [DOI] [PubMed] [Google Scholar]
- 79.Mohler PJ, Gramolini AO and Bennett V (2002) Ankyrins. J Cell Sci 115(Pt 8), 1565–6 [DOI] [PubMed] [Google Scholar]
- 80.Mohler PJ, et al. (2003) Ankyrin-B mutation causes type 4 long-QT cardiac arrhythmia and sudden cardiac death. Nature 421(6923), 634–9 [DOI] [PubMed] [Google Scholar]
- 81.Mohler PJ, et al. (2004) A cardiac arrhythmia syndrome caused by loss of ankyrin-B function. Proc Natl Acad Sci U S A 101(24), 9137–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Plaster NM, et al. (2001) Mutations in Kir2.1 cause the developmental and episodic electrical phenotypes of Andersen’s syndrome. Cell 105(4), 511–9 [DOI] [PubMed] [Google Scholar]
- 83.Preisig-Müller R, et al. (2002) Heteromerization of Kir2.x potassium channels contributes to the phenotype of Andersen’s syndrome. Proc Natl Acad Sci U S A 99(11), 7774–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ai T, et al. (2002) Novel KCNJ2 mutation in familial periodic paralysis with ventricular dysrhythmia. Circulation 105(22), 2592–4 [DOI] [PubMed] [Google Scholar]
- 85.Depil K, et al. (2011) Timothy mutation disrupts the link between activation and inactivation in Ca(V)1.2 protein. J Biol Chem 286(36), 31557–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Boczek NJ, et al. (2016) Spectrum and Prevalence of CALM1-, CALM2-, and CALM3-Encoded Calmodulin Variants in Long QT Syndrome and Functional Characterization of a Novel Long QT Syndrome-Associated Calmodulin Missense Variant, E141G. Circ Cardiovasc Genet 9(2), 136–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Crotti L, et al. (2013) Calmodulin mutations associated with recurrent cardiac arrest in infants. Circulation 127(9), 1009–17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kato K, et al. (2022) Novel CALM3 Variant Causing Calmodulinopathy With Variable Expressivity in a 4-Generation Family. Circ Arrhythm Electrophysiol 15(3), e010572. [DOI] [PubMed] [Google Scholar]
- 89.Su J, et al. (2020) The LQT-associated calmodulin mutant E141G induces disturbed Ca(2+)-dependent binding and a flickering gating mode of the Ca(V)1.2 channel. Am J Physiol Cell Physiol 318(5), C991–C1004 [DOI] [PubMed] [Google Scholar]
- 90.Tsuji-Wakisaka K, et al. (2011) Identification and functional characterization of KCNQ1 mutations around the exon 7-intron 7 junction affecting the splicing process. Biochim Biophys Acta 1812(11), 1452–9 [DOI] [PubMed] [Google Scholar]
- 91.Gong Q, et al. (2008) A splice site mutation in hERG leads to cryptic splicing in human long QT syndrome. J Mol Cell Cardiol 44(3), 502–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Watanabe H, et al. (2011) Striking In vivo phenotype of a disease-associated human SCN5A mutation producing minimal changes in vitro. Circulation 124(9), 1001–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Schwartz PJ, Crotti L and George AL Jr. (2018) Modifier genes for sudden cardiac death. Eur Heart J 39(44), 3925–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Del Alamo JC, et al. (2016) High throughput physiological screening of iPSC-derived cardiomyocytes for drug development. Biochim Biophys Acta 1863(7 Pt B), 1717–27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sharma A, et al. (2018) Use of human induced pluripotent stem cell-derived cardiomyocytes to assess drug cardiotoxicity. Nat Protoc 13(12), 3018–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yim DS (2018) Five years of the CiPA project (2013–2018): what did we learn? Transl Clin Pharmacol 26(4), 145–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Klaren WD and Rusyn I (2018) High-Content Assay Multiplexing for Muscle Toxicity Screening in Human-Induced Pluripotent Stem Cell-Derived Skeletal Myoblasts. Assay Drug Dev Technol 16(6), 333–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Schwartz PJ (2021) 1970–2020: 50 years of research on the long QT syndrome-from almost zero knowledge to precision medicine. Eur Heart J 42(11), 1063–72 [DOI] [PubMed] [Google Scholar]
- 99.Wilson AJ, et al. (2005) Abnormal KCNQ1 trafficking influences disease pathogenesis in hereditary long QT syndromes (LQT1). Cardiovasc Res 67(3), 476–86 [DOI] [PubMed] [Google Scholar]
- 100.Pan N, et al. (2009) A hydrophobicity-dependent motif responsible for surface expression of cardiac potassium channel. Cell Signal 21(2), 349–55 [DOI] [PubMed] [Google Scholar]
- 101.Shah M and Carter C (2008) Long QT syndrome: A therapeutic challenge. Ann Pediatr Cardiol 1(1), 18–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Itzhaki I, et al. (2011) Modelling the long QT syndrome with induced pluripotent stem cells. Nature 471(7337), 225–9 [DOI] [PubMed] [Google Scholar]
- 103.Matsa E, et al. (2014) Allele-specific RNA interference rescues the long-QT syndrome phenotype in human-induced pluripotency stem cell cardiomyocytes. Eur Heart J 35(16), 1078–87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lahti AL, et al. (2012) Model for long QT syndrome type 2 using human iPS cells demonstrates arrhythmogenic characteristics in cell culture. Dis Model Mech 5(2), 220–30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chai S, et al. (2018) Physiological genomics identifies genetic modifiers of long QT syndrome type 2 severity. J Clin Invest 128(3), 1043–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Terrenoire C, et al. (2013) Induced pluripotent stem cells used to reveal drug actions in a long QT syndrome family with complex genetics. J Gen Physiol 141(1), 61–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yazawa M, et al. (2011) Using induced pluripotent stem cells to investigate cardiac phenotypes in Timothy syndrome. Nature 471(7337), 230–4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Rocchetti M, et al. (2017) Elucidating arrhythmogenic mechanisms of long-QT syndrome CALM1-F142L mutation in patient-specific induced pluripotent stem cell-derived cardiomyocytes. Cardiovasc Res 113(5), 531–41 [DOI] [PubMed] [Google Scholar]
- 109.Limpitikul WB, et al. (2017) A Precision Medicine Approach to the Rescue of Function on Malignant Calmodulinopathic Long-QT Syndrome. Circ Res 120(1), 39–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sala L, et al. (2016) A new hERG allosteric modulator rescues genetic and drug-induced long-QT syndrome phenotypes in cardiomyocytes from isogenic pairs of patient induced pluripotent stem cells. EMBO Mol Med 8(9), 1065–81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Matsa E, et al. (2011) Drug evaluation in cardiomyocytes derived from human induced pluripotent stem cells carrying a long QT syndrome type 2 mutation. Eur Heart J 32(8), 952–62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Meng J, et al. (2013) Compound ICA-105574 prevents arrhythmias induced by cardiac delayed repolarization. Eur J Pharmacol 718(1–3), 87–97 [DOI] [PubMed] [Google Scholar]
- 113.Chockalingam P, et al. (2012) Not all beta-blockers are equal in the management of long QT syndrome types 1 and 2: higher recurrence of events under metoprolol. J Am Coll Cardiol 60(20), 2092–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Bhuiyan ZA, et al. (2013) Congenital Long QT Syndrome: An Update and Present Perspective in Saudi Arabia. Front Pediatr 1, 39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bos JM, et al. (2019) Mexiletine Shortens the QT Interval in Patients With Potassium Channel-Mediated Type 2 Long QT Syndrome. Circ Arrhythm Electrophysiol 12(5), e007280. [DOI] [PubMed] [Google Scholar]
- 116.Wilde AAM and Remme CA (2018) Therapeutic approaches for Long QT syndrome type 3: an update. Europace 20(2), 222–24 [DOI] [PubMed] [Google Scholar]
- 117.Miyamoto K, et al. (2015) Efficacy and safety of flecainide for ventricular arrhythmias in patients with Andersen-Tawil syndrome with KCNJ2 mutations. Heart Rhythm 12(3), 596–603 [DOI] [PubMed] [Google Scholar]
- 118.Caballero R, et al. (2010) Flecainide increases Kir2.1 currents by interacting with cysteine 311, decreasing the polyamine-induced rectification. Proc Natl Acad Sci U S A 107(35), 15631–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Junker J, et al. (2002) Amiodarone and acetazolamide for the treatment of genetically confirmed severe Andersen syndrome. Neurology 59(3), 466. [DOI] [PubMed] [Google Scholar]
- 120.Tristani-Firouzi M and Etheridge SP (2010) Kir 2.1 channelopathies: the Andersen-Tawil syndrome. Pflugers Arch 460(2), 289–94 [DOI] [PubMed] [Google Scholar]
- 121.Sansone V and Tawil R (2007) Management and treatment of Andersen-Tawil syndrome (ATS). Neurotherapeutics 4(2), 233–7 [DOI] [PubMed] [Google Scholar]
- 122.Casini S, Verkerk AO and Remme CA (2017) Human iPSC-Derived Cardiomyocytes for Investigation of Disease Mechanisms and Therapeutic Strategies in Inherited Arrhythmia Syndromes: Strengths and Limitations. Cardiovasc Drugs Ther 31(3), 325–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Jacobs A, et al. (2006) Verapamil decreases ventricular tachyarrhythmias in a patient with Timothy syndrome (LQT8). Heart Rhythm 3(8), 967–70 [DOI] [PubMed] [Google Scholar]
- 124.Niaz T, et al. (2020) Left Cardiac Sympathetic Denervation Monotherapy in Patients With Congenital Long QT Syndrome. Circ Arrhythm Electrophysiol 13(12), e008830. [DOI] [PubMed] [Google Scholar]
- 125.Schwartz PJ and Ackerman MJ (2022) Cardiac sympathetic denervation in the prevention of genetically mediated life-threatening ventricular arrhythmias. Eur Heart J 43(22), 2096–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hortigon-Vinagre MP, et al. (2016) The Use of Ratiometric Fluorescence Measurements of the Voltage Sensitive Dye Di-4-ANEPPS to Examine Action Potential Characteristics and Drug Effects on Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes. Toxicol Sci 154(2), 320–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Blinova K, et al. (2017) Comprehensive Translational Assessment of Human-Induced Pluripotent Stem Cell Derived Cardiomyocytes for Evaluating Drug-Induced Arrhythmias. Toxicol Sci 155(1), 234–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Millard D, et al. (2018) Cross-Site Reliability of Human Induced Pluripotent stem cell-derived Cardiomyocyte Based Safety Assays Using Microelectrode Arrays: Results from a Blinded CiPA Pilot Study. Toxicol Sci 164(2), 550–62 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Qu Y and Vargas HM (2015) Proarrhythmia Risk Assessment in Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes Using the Maestro MEA Platform. Toxicol Sci 147(1), 286–95 [DOI] [PubMed] [Google Scholar]
- 130.Leopold JA and Loscalzo J (2018) Emerging Role of Precision Medicine in Cardiovascular Disease. Circ Res 122(9), 1302–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Lindsey ML, et al. (2015) Transformative Impact of Proteomics on Cardiovascular Health and Disease: A Scientific Statement From the American Heart Association. Circulation 132(9), 852–72 [DOI] [PubMed] [Google Scholar]
- 132.Shah SH and Newgard CB (2015) Integrated metabolomics and genomics: systems approaches to biomarkers and mechanisms of cardiovascular disease. Circ Cardiovasc Genet 8(2), 410–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Krittanawong C, et al. (2019) Deep learning for cardiovascular medicine: a practical primer. Eur Heart J 40(25), 2058–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Siontis KC, et al. (2021) Artificial intelligence-enhanced electrocardiography in cardiovascular disease management. Nat Rev Cardiol 18(7), 465–78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Goldstein BA, Navar AM and Carter RE (2017) Moving beyond regression techniques in cardiovascular risk prediction: applying machine learning to address analytic challenges. Eur Heart J 38(23), 1805–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zwi L, et al. (2009) Cardiomyocyte differentiation of human induced pluripotent stem cells. Circulation 120(15), 1513–23 [DOI] [PubMed] [Google Scholar]
- 137.Lian X, et al. (2012) Robust cardiomyocyte differentiation from human pluripotent stem cells via temporal modulation of canonical Wnt signaling. Proc Natl Acad Sci U S A 109(27), E1848–57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Gnecchi M, Stefanello M and Mura M (2017) Induced pluripotent stem cell technology: Toward the future of cardiac arrhythmias. Int J Cardiol 237, 49–52 [DOI] [PubMed] [Google Scholar]
- 139.van den Berg CW, et al. (2015) Transcriptome of human foetal heart compared with cardiomyocytes from pluripotent stem cells. Development 142(18), 3231–8 [DOI] [PubMed] [Google Scholar]
- 140.Gnecchi M and Schwartz PJ (2012) The unstoppable attraction for induced pluripotent stem cells: are they the magic bullet for modeling inherited arrhythmogenic diseases? J Am Coll Cardiol 60(11), 1001–4 [DOI] [PubMed] [Google Scholar]
- 141.Bellin M, et al. (2013) Isogenic human pluripotent stem cell pairs reveal the role of a KCNH2 mutation in long-QT syndrome. EMBO J 32(24), 3161–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zhang M, et al. (2014) Recessive cardiac phenotypes in induced pluripotent stem cell models of Jervell and Lange-Nielsen syndrome: disease mechanisms and pharmacological rescue. Proc Natl Acad Sci U S A 111(50), E5383–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Mura M, et al. (2017) The KCNH2-IVS9–28A/G mutation causes aberrant isoform expression and hERG trafficking defect in cardiomyocytes derived from patients affected by Long QT Syndrome type 2. Int J Cardiol 240, 367–71 [DOI] [PubMed] [Google Scholar]
- 144.Garg P, et al. (2018) Genome Editing of Induced Pluripotent Stem Cells to Decipher Cardiac Channelopathy Variant. J Am Coll Cardiol 72(1), 62–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Sala L, Bellin M and Mummery CL (2017) Integrating cardiomyocytes from human pluripotent stem cells in safety pharmacology: has the time come? Br J Pharmacol 174(21), 3749–65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Karbassi E, et al. (2020) Cardiomyocyte maturation: advances in knowledge and implications for regenerative medicine. Nat Rev Cardiol 17(6), 341–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Veerman CC, et al. (2015) Immaturity of human stem-cell-derived cardiomyocytes in culture: fatal flaw or soluble problem? Stem Cells Dev 24(9), 1035–52 [DOI] [PubMed] [Google Scholar]
- 148.Kim K, et al. (2010) Epigenetic memory in induced pluripotent stem cells. Nature 467(7313), 285–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zareba W, et al. (2003) Modulating effects of age and gender on the clinical course of long QT syndrome by genotype. J Am Coll Cardiol 42(1), 103–9 [DOI] [PubMed] [Google Scholar]
- 150.Etheridge SP, Asaki SY and Niu MC (2019) A personalized approach to long QT syndrome. Curr Opin Cardiol 34(1), 46–56 [DOI] [PubMed] [Google Scholar]
- 151.Etheridge SP, et al. (2003) A new oral therapy for long QT syndrome: long-term oral potassium improves repolarization in patients with HERG mutations. J Am Coll Cardiol 42(10), 1777–82 [DOI] [PubMed] [Google Scholar]
- 152.Compton SJ, et al. (1996) Genetically defined therapy of inherited long-QT syndrome. Correction of abnormal repolarization by potassium. Circulation 94(5), 1018–22 [DOI] [PubMed] [Google Scholar]
- 153.Kannankeril P, Roden DM and Darbar D (2010) Drug-induced long QT syndrome. Pharmacol Rev 62(4), 760–81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Niemeijer MN, et al. (2015) Pharmacogenetics of Drug-Induced QT Interval Prolongation: An Update. Drug Saf 38(10), 855–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Strauss DG, et al. (2017) Common Genetic Variant Risk Score Is Associated With Drug-Induced QT Prolongation and Torsade de Pointes Risk: A Pilot Study. Circulation 135(14), 1300–10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Dotzler SM, et al. (2021) Suppression-Replacement KCNQ1 Gene Therapy for Type 1 Long QT Syndrome. Circulation 143(14), 1411–25 [DOI] [PubMed] [Google Scholar]
- 157.Lu X, et al. (2013) RNA interference targeting E637K mutation rescues hERG channel currents and restores its kinetic properties. Heart Rhythm 10(1), 128–36 [DOI] [PubMed] [Google Scholar]
- 158.Lee YK, et al. (2021) MTMR4 SNVs modulate ion channel degradation and clinical severity in congenital long QT syndrome: insights in the mechanism of action of protective modifier genes. Cardiovasc Res 117(3), 767–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Wang Q, et al. (1995) SCN5A mutations associated with an inherited cardiac arrhythmia, long QT syndrome. Cell 80(5), 805–11 [DOI] [PubMed] [Google Scholar]
- 160.Chen Q, et al. (1998) Genetic basis and molecular mechanism for idiopathic ventricular fibrillation. Nature 392(6673), 293–6 [DOI] [PubMed] [Google Scholar]
- 161.McNair WP, et al. (2004) SCN5A mutation associated with dilated cardiomyopathy, conduction disorder, and arrhythmia. Circulation 110(15), 2163–7 [DOI] [PubMed] [Google Scholar]
- 162.Wang Q, et al. (1996) Positional cloning of a novel potassium channel gene: KVLQT1 mutations cause cardiac arrhythmias. Nat Genet 12(1), 17–23 [DOI] [PubMed] [Google Scholar]
- 163.Curran ME, et al. (1995) A molecular basis for cardiac arrhythmia: HERG mutations cause long QT syndrome. Cell 80(5), 795–803 [DOI] [PubMed] [Google Scholar]
- 164.Splawski I, et al. (1997) Mutations in the hminK gene cause long QT syndrome and suppress IKs function. Nat Genet 17(3), 338–40 [DOI] [PubMed] [Google Scholar]
- 165.Abbott GW, et al. (1999) MiRP1 forms IKr potassium channels with HERG and is associated with cardiac arrhythmia. Cell 97(2), 175–87 [DOI] [PubMed] [Google Scholar]
- 166.Duggal P, et al. (1998) Mutation of the gene for IsK associated with both Jervell and Lange-Nielsen and Romano-Ward forms of Long-QT syndrome. Circulation 97(2), 142–6 [DOI] [PubMed] [Google Scholar]
- 167.Chaix MA, et al. (2016) Novel CALM3 mutations in pediatric long QT syndrome patients support a CALM3-specific calmodulinopathy. HeartRhythm Case Rep 2(3), 250–54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Malan D, et al. (2016) Human iPS cell model of type 3 long QT syndrome recapitulates drug-based phenotype correction. Basic Res Cardiol 111(2), 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Bianchi L, et al. (1999) Cellular dysfunction of LQT5-minK mutants: abnormalities of IKs, IKr and trafficking in long QT syndrome. Hum Mol Genet 8(8), 1499–507 [DOI] [PubMed] [Google Scholar]
- 170.Millat G, et al. (2006) Spectrum of pathogenic mutations and associated polymorphisms in a cohort of 44 unrelated patients with long QT syndrome. Clin Genet 70(3), 214–27 [DOI] [PubMed] [Google Scholar]
- 171.Kuroda Y, et al. (2017) Flecainide ameliorates arrhythmogenicity through NCX flux in Andersen-Tawil syndrome-iPS cell-derived cardiomyocytes. Biochem Biophys Rep 9, 245–56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Yamamoto Y, et al. (2017) Allele-specific ablation rescues electrophysiological abnormalities in a human iPS cell model of long-QT syndrome with a CALM2 mutation. Hum Mol Genet 26(9), 1670–77 [DOI] [PubMed] [Google Scholar]


