Abstract
Overweight sedentary individuals are at increased risk for cardiovascular disease, diabetes and some neurological disorders. Beneficial effects of dietary energy restriction (DER) and exercise on brain structural plasticity and behaviors have been demonstrated in animal models of aging and acute (stroke and trauma) and chronic (Alzheimer’s and Parkinson’s diseases) neurological disorders. The findings described below, and evolutionary considerations, suggest brain-derived neurotrophic factor (BDNF) plays a critical role in the integration and optimization of behavioral and metabolic responses to environments with limited energy resources and intense competition. In particular, BDNF signaling mediates adaptive responses of the central, autonomic, and peripheral nervous systems from exercise and DER. In the hypothalamus, BDNF inhibits food intake and increases energy expenditure. By promoting synaptic plasticity and neurogenesis in the hippocampus, BDNF mediates exercise- and DER-induced improvements in cognitive function and neuroprotection. DER improves cardiovascular stress adaptation by a mechanism involving enhancement of brainstem cholinergic activity. Collectively, findings reviewed in this article provide a rationale for targeting BDNF signaling for novel therapeutic interventions in a range of metabolic and neurological disorders.
Keywords: autonomic nervous system, brain-derived neurotrophic factor, cognition, diabetes, exercise, neurogenesis, synaptic plasticity
BDNF and food intake
In the central nervous system (CNS), BDNF and its high-affinity receptor trkB are highly expressed in the hypothalamus where this neurotrophic factor has major regulatory roles in the control of appetite and metabolism.1 Mice that are heterozygous for targeted disruption of BDNF (BDNF+/− mice) show a 50% reduction in BDNF expression in the hypothalamus,2 and consume 47% more food than wild-type mice and are obese.3 Obesity in BDNF+/− mice is prevented by restricting food intake to match that of wild-type counterparts, implying that the loss of BDNF causes hyperphagia, which leads to obesity.4 A reduction in the expression of TrkB also leads to obesity in mice.5 By six months of age, the average weight of male and female BDNF+/− mice are 44% and 33% increased over wild-type counterparts, respectively.3 This age-related obesity and chronic hyperphagia is accompanied by hyperactivity, which is in contrast to lethargy normally associated with obesity.3,6
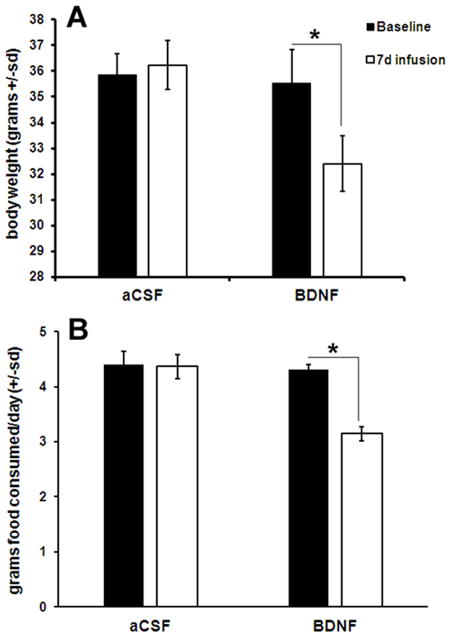
Diminished BDNF signaling results in hyperphagia and obesity, whereas an increase in BDNF signaling has the opposite effect. Intracerebroventricular infusion of BDNF into normal mice results in a significant decrease in food consumption and a loss of body weight (Fig. 1). After 7 days of ICV infusion of BDNF (1.2 μg over 24 h), wild-type mice experience a highly significant (P = 0.005) decrease in food consumption and a significant (P = 0.01) decrease in body weight, whereas mice receiving infusion of artificial cerebrospinal fluid (aCSF) do not. This effect of BDNF also occurs in rats.7 Intracerebroventricular infusion of BDNF into BDNF+/− mice normalizes food intake, body weight, and activity, further implying a physiological role for BDNF in regulating food intake.
Figure 1.
BDNF infusion causes a significant decrease in food consumption and a loss of body weight. Intracerebroventricular infusion of BDNF (1.2μg over 24 h), causes a significant (P = 0.005) decrease in food consumption in wild-type mice as well as a significant (P = 0.01) decrease in body weight. Mice receiving infusion of artificial cerebrospinal fluid (aCSF) do not experience a significant change in either food consumption or body weight.
The precise mechanism by which BDNF signaling suppresses appetite and food intake is not entirely known, although several hypotheses have emerged. The regions of the hypothalamus that regulate food intake include the paraventricular, arcuate (ARC), dorsomedial (DMH), and ventromedial nuclei. BDNF is expressed in some cells in the dorsomedial hypothalamus and at negligible levels in the arcuate; however, it is highly expressed in the ventromedial nuclei (VMH). Bilateral lesions of the VMH cause hypherphagia and obesity implying an important role for this region of the hypothalamus in regulating food intake and energy metabolism.8 Leptin, a polypeptide produced by adipocytes, targets neurons in the ARC nucleus; leptin positively regulates proopiomelanocortin (POMC) neurons, which project to the VMH. In the VMH, expression of the melanocortin receptor 4 (MC4R) regulates expression of BDNF; a reduction in MC4R causes a downregulation of BDNF.5 Further, administration of an MC4R agonist increased the level of BDNF mRNA in food-deprived mice, implying that MC4R signaling regulates alterations in BDNF in response to food deprivation.5 Taken together, these data provide the beginning of a framework by which gut hormones target neurons in the hypothalamus which control expression of BDNF and therefore, food intake and metabolism.
Another region of the brain that may mediate the effects of BDNF signaling on food intake is the brainstem. Specifically, the dorsal vagal complex (DVC) contains insulin and leptin receptors as well as mechanisms that sense glucose levels.9 Intraparenchymal infusion of BDNF to the DVC causes dose-dependent anorexia implying that alterations in BDNF signaling in the DVC, without any alteration in signaling in the hypothalamus, are sufficient to alter feeding and potentially metabolism parameters.10
Experimental manipulations of BDNF levels affect feeding and metabolism, and environmental factors such as food deprivation and stress also cause changes in BDNF expression in the brain. Alternate day fasting stimulates the production of BDNF in neurons in different brain regions including hippocampus, specifically the dentate gyrus.11–13 It is noteworthy that dietary energy restriction restores BDNF levels in BDNF+/− mice and also reverses hyperphagia and obesity in those mice.2 Conversely, food deprivation has an inhibitory effect on BDNF expression in the VMH of the hypothalamus that is partially reversed by administration of a melanocortin receptor 4 antagonist.5 In addition, food deprivation causes a reversible decrease in BDNF protein in the DVC, potentially indicating a role for DVC BDNF signaling in mediating metabolic feedback. The reason for the differential, brain region-specific changes in BDNF expression is likely that the neuronal circuits in the different regions are involved in coordinating complex behavioral and neuroendocrine responses to food intake or deprivation. Because food restriction is stressful, it can cause a rise in corticosterone (cortisol in humans), which has been shown to decrease the expression and production of BDNF in the brain. However the brainstem may respond to food deprivation in a compensatory manner; food deprivation may cause an acute decrease in BDNF signaling, but it is possible that the brainstem upregulates BDNF expression in a compensatory response to a decrease in food intake.
BDNF regulates peripheral energy metabolism
BDNF regulation of food intake is also coupled to the ability of this neurotrophin to regulate peripheral energy metabolism. Low levels of circulating BDNF are noted in individuals with obesity and type 2 diabetes implying a role for this neurotrophin in mediating obesity and metabolism.14 However, patients with type 2 diabetes that are not obese display decreased levels of plasma BDNF, which could indicate that BDNF regulates obesity and metabolism via different mechanisms.15 Mice with BDNF eliminated at birth are hypersensitive to stress, display elevated plasma glucose and insulin levels and are obese.6 Experimental studies show that intracerebroventricular infusion of BDNF increases peripheral insulin sensitivity in normal rodents16 and ameliorates diabetes in mice,17 implying that BDNF signaling in the brain, particularly the hypothalamus, regulates peripheral energy metabolism. Further, BDNF+/− mice display a diabetic phenotype, including elevated levels of circulating glucose, insulin, and leptin.2
Obesity is associated with changes in the serum levels of leptin, insulin, glucose, and corticosterone/cortisol.18 The interplay between these molecules is complex. In normal individuals, insulin is produced in response to increased glucose levels and stimulates glucose uptake by muscle and liver cells; corticosterone induces gluconeogenesis, and leptin production and secretion from adipocytes can be stimulated by insulin; circulating leptin enters the brain and interacts with neurons in the hypothalamus to suppress appetite. While the exact mechanisms by which BDNF regulates metabolism are not entirely known, several studies using leptin receptor mutant (db/db) mice demonstrate an important role of this hormone in BDNF-mediated regulation of metabolism. The db/db mice are hyperglycemic and insulin resistant, and administration of BDNF normalizes glucose levels and insulin sensitivity.16 Further, controlled studies in which both vehicle and BDNF-treated db/db mice consumed the same amount of food showed that BDNF administration significantly reduces blood glucose concentration, indicating that the effects of BDNF on energy metabolism are independent of its effects on appetite. Further proof of a role for leptin in mediating BDNF regulation of energy metabolism lies in data that shows that BDNF+/− mice display increased leptin levels and leptin resistance.3 Conversely, db/db mice that cannot respond to leptin show reduced BDNF expression in the hippocampus and hypothalamus. Systemic BDNF infusion reduces food intake and blood glucose in db/db mice.17,19 Lowering circulating corticosterone levels in db/db mice via adrenalectomy restores BDNF expression in the hippocampus but not the hypothalamus.20
Data show that some of the effects of leptin on energy metabolism may be mediated via changes in body temperature control. Db/db mice show reduced energy metabolism and lower body temperature and may be unable to activate thermogenesis to maintain body heat when their food intake is restricted.16 In fact, administration of BDNF-raised body temperature in food restricted db/db mice compared to ad-libitum mice.17
Other models of diabetes in mice have been used to demonstrate a role for BDNF in regulating energy metabolism. Administration of streptozotocin (STZ) damages pancreatic β cells, leading to impaired insulin production and is therefore a model of type I diabetes. In STZ-treated mice, administration of BDNF causes a reduction in food intake but no change in circulating glucose levels, implying that the mechanism by which BDNF regulates food intake is independent of insulin signaling, but the mechanism by which BDNF regulates energy metabolism is insulin dependent.16
Involvement of BDNF in brainstem control of the cardiovascular system
It is well known that neurotrophic factors such as BDNF are essential for the development of the autonomic nervous system (ANS), particularly in the formation of synaptic connectivity with peripheral targets. For example, BDNF is essential for the survival of arterial baroreceptors during vascular innervation.21 However, recent findings indicate that BDNF also plays a major role in ANS control of cardiovascular function in adults. A role for BDNF in ANS regulation of heart rate is suggested by data showing exercise and dietary energy restriction, both of which increase BDNF levels in the CNS,13,22,23 also decrease heart rate and increase heart rate variability.24–26 The latter effects of exercise and dietary energy restriction are believed to be mediated by a relative increase in parasympathetic activity on the heart. Indeed, Yang et al.27 showed that BDNF could alter the neurotransmitter release properties of sympathetic neurons innervating cardiac myocytes in culture, resulting in a shift from excitatory to inhibitory cholinergic neurotransmission. The cardioprotective effects of dietary energy restriction may result, in part, from the relative increase in parasympathetic activity.28
Studies from our lab further suggest a role for BDNF in regulating parasympathetic and/or sympathetic inputs to the heart. BDNF+/− mice, which exhibit a fifty percent reduction in BDNF mRNA, have significantly elevated heart rates compared to WT mice (Fig. 2A). Further, when exposed to restraint stress, BDNF+/− mice fail to elevate their heart rate to WT levels (Fig. 2B) indicating an impaired cardiovascular stress response. Interestingly, a recent study revealed that humans with a BDNF polymorphism (Val66Met) that results in decreased activity-dependent BDNF secretion, have altered sympathovagal balance leading to sympathetic dominance.29 In addition, carriers of this mutation also exhibit attenuated heart rate responses to stress.30 Taken together with our data, the latter findings suggest that reduced BDNF expression impairs heart rate stress responses and ANS function.
Figure 2.
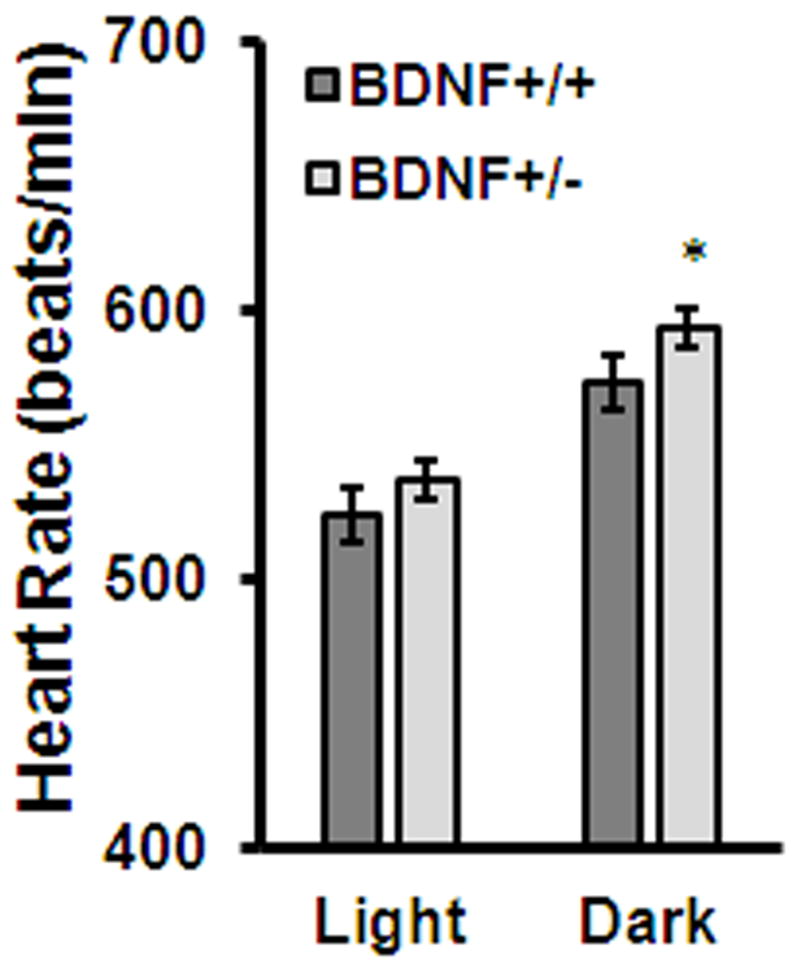
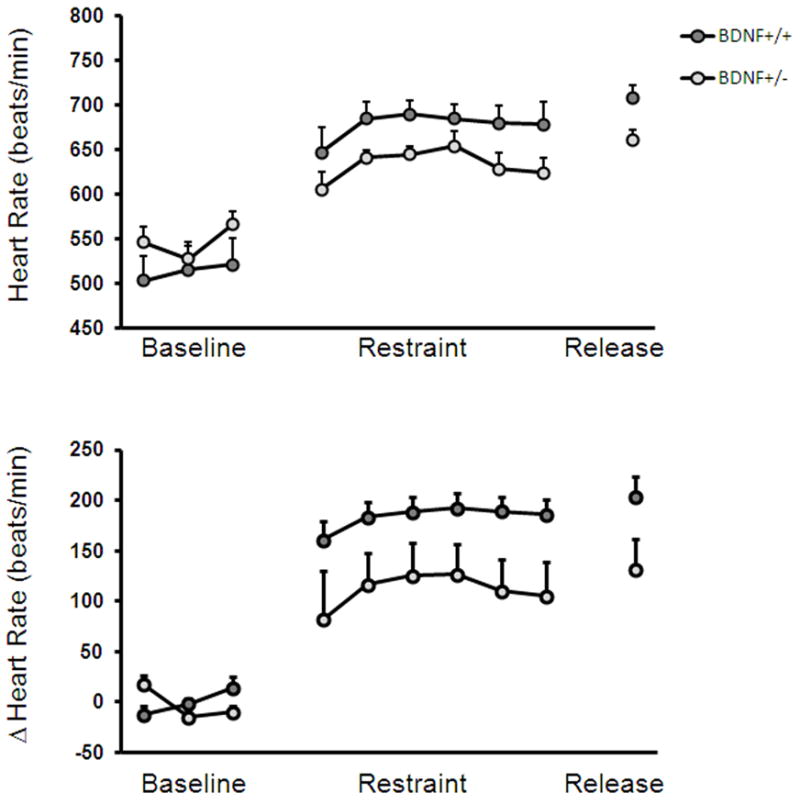
Evidence that BDNF signaling regulates heart rate. (A) BDNF+/− mice have significantly elevated basal heart rates compared to control (BDNF+/+) mice during the dark cycle (P < 0.05). (B) BDNF+/− mice have attenuated heart rates during a 60 min restraint stress and release. Data points represent 10-min bins (P < 0.001). The change in heart rate in BDNF+/− mice is significantly lower than BDNF+/+ mice, both during restraint and release (P < 0.001). Data are represented as mean ± SEM.
BDNF regulation of cardiovascular function likely occurs via signaling in central autonomic nuclei of the brainstem. BDNF is expressed in both baroreceptor and chemoafferents in the nodose and petrosal sensory ganglia, which terminate in the brainstem.21,31 BDNF and TrkB are also produced in central autonomic nuclei of the brainstem,32–35 as well as in higher cardiovascular control areas such as the hypothalamus, forebrain, and amygdala.36–37 Local injection of BDNF into the rostral ventrolateral medulla increases blood pressure,38 and BDNF acutely raises heart rate and blood pressure when applied to the third ventricle.39 Further, BDNF is released in synchrony with baroreceptor activity from baroreceptor afferents onto second order neurons in the nucleus tractus solitarius (NTS), the primary target of afferent cardiovascular input to the brainstem.31,40 Injection of BDNF into the NTS of anesthetized rats increases blood pressure, heart rate, and lumbar sympathetic nerve activity.41 Conversely, inhibition of tonic BDNF signaling in the NTS with a TrkB receptor antagonist decreases blood pressure and heart rate, indicating that BDNF signaling in the NTS tonically modulates cardiovascular regulation.41
BDNF mediates exercise- and energy restriction-induced neuroplasticity
The beneficial effects of exercise on health are unequivocal and, as reviewed previously,42 include profound effects on the brain. Studies performed primarily in laboratory rodents have revealed highly reproducible effects of exercise, particularly aerobic endurance exercise, on the structure and function of the brain. Compared to their more sedentary counterparts, mice or rats that run voluntarily on running wheels (runners), exhibit increased numbers of dendritic spines (i.e., synapses) in hippocampal neurons, increased neurogenesis, and improved performance in some behavioral tests of cognitive function.43–45 Mice and rats will typically run between 2 and 15 kilometers in a 24 h period; there is considerable inter-animal variability in daily running distance, but less day-to-day variability in daily distance run for individual mice.46–48 Where examined, there tends to be a graded positive effect of daily exercise intensity and duration on synaptic plasticity and neurogenesis. In addition, the positive effects of exercise on synaptic strength and learning and memory are not immediately apparent, and instead occur over periods of many days to weeks. The purpose of this section is to describe the evidence that BDNF plays a key role as a mediator of the effects of exercise on synaptic plasticity and neurogenesis (Fig. 3). In keeping with the main theme of this article, we also consider recent evidence that BDNF mediates beneficial effects of intermittent energy restriction (IER) on neuroplasticity and the vulnerability of neurons to injury and disease.49,50
Figure 3.
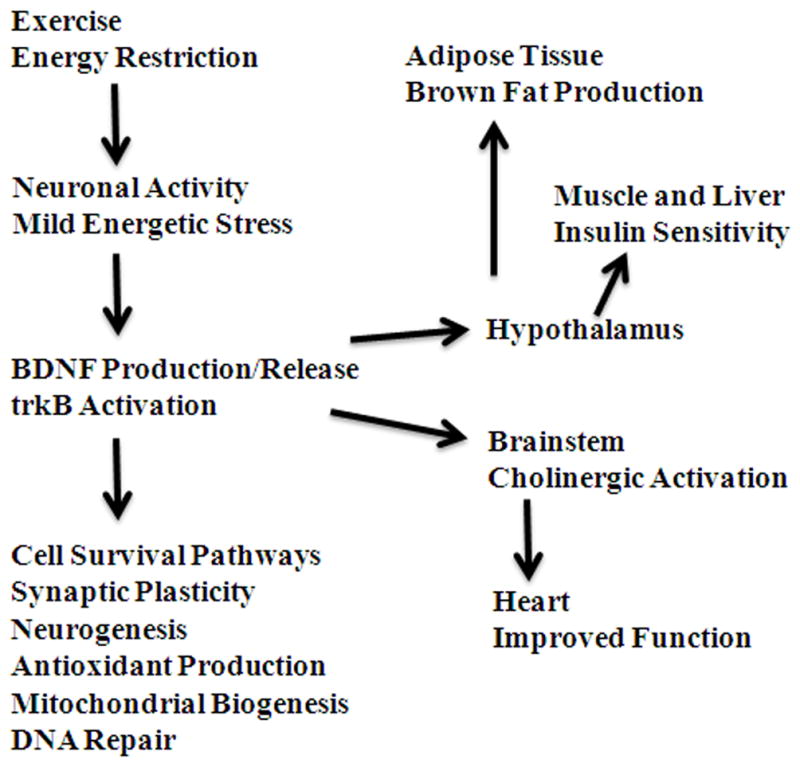
Central roles for BDNF as a mediator of beneficial effects of exercise and dietary energy restriction on neuroplasticity and overall health. Exercise and energy restriction (particular intermittent energy restriction) induce increased activity in neuronal circuits, as well as a mild energetic stress, throughout the nervous system. This mild cellular stress stimulates the production and release of BDNF. BDNF acts on neurons to promote their growth, enhance synaptic plasticity, and increase their resistance to injury and disease. Some of the genes induced in neurons in response to BDNF include those encoding antioxidant enzymes, DNA repair enzymes, and proteins involved in mitochondrial biogenesis. In addition, in the hippocampus BDNF promotes the production and survival of new neurons from stem cells, and the integration of the new neurons into existing neuronal circuits. Acting in brain regions that control neuroendocrine pathways (e.g., hypothalamus) and the autonomic nervous system (e.g., brainstem cholinergic neurons), BDNF mediates beneficial effects of exercise and energy restriction on glucose metabolism, body fat composition, and cardiovascular fitness.
Exercise and BDNF
Mice that are runners can perform better in tests of learning and memory, such as the radial arm maze, compared to non-runners. Interestingly, when mice were provided running wheels for one week and then the running wheels were disabled, they learned the maze fastest when tested one week after cessation of running, but their memory retention was best when tested immediately after the one-week running period.51 Hippocampal BDNF levels were elevated immediately after the one-week running period, remained elevated for two weeks after cessation of running and then returned to baseline by three weeks. There is also evidence that prior exercise can “prime” a BDNF response of hippocampal cells to a subsequent brief period of exercise that would be insufficient to significantly increase BDNF levels in previously sedentary animals.52 The priming effect of exercise is maintained for at least two weeks after the cessation of exercise. The underlying molecular mechanism of the priming effect of exercise on BDNF production remains to be determined. In addition to improving performance in learning and memory tasks, running was shown to enhance the ability of mice to distinguish between objects located close to each other on a touch screen (spatial pattern separation), a process dependent upon hippocampal plasticity.53 Moreover, a study of elderly human subjects showed that a daily aerobic exercise intervention can improve performance in memory tests, with an associated increase in the size of the hippocampus as measured by structural magnetic resonance imaging methods.54
The results of different behavioral tests have revealed that voluntary running and forced treadmill running can differentially affect performance on behavioral tasks. For example, whereas both voluntary and treadmill running improved performance of mice in the water maze, only treadmill running improved performance in a passive avoidance test.55 The latter study further showed that both forced and voluntary running upregulated BDNF–trkB signaling in the hippocampus, while only forced exercise increased BDNF signaling in the amygdala. Inasmuch as forced exercise can be considered to be an aversive (or at least an annoying) stressor, it is understandable that it would engage neuronal circuits in the amygdala, a brain region heavily involved in fear-related learning.56
In rodents, running wheel exercise induces the expression of BDNF at the transcriptional level in multiple brain regions, and in particularly large amounts in dentate gyrus granule neurons and CA1 neurons in the hippocampus, and in neurons in layers II and III of the cerebral cortex.23,57 Elevations in BDNF transcripts can occur within minutes to hours of initiation of vigorous exercise. BDNF production can also be regulated at the translational level in response to exercise and other stimuli, with levels of BDNF protein typically increasing by a greater percentage over baseline compared to the increase in BDNF mRNA levels.58 Compared to their more sedentary control counterparts, runner rats exhibit significantly more LTP in response to theta rhythm-patterned stimulation, and this synaptic strengthening is associated with increased expression of the NR2B NMDA receptor subunit and BDNF.44
Activity-dependent production of nitric oxide may play an important role in the production of BDNF that occurs in response to exercise.59,60 Likely of particular importance for the effects of exercise on synaptic plasticity is the stimulation of BDNF protein production locally from BDNF mRNA associated with ribosomes in dendrites.61–63 In turn, BDNF induces the local (dendritic) translation of mRNAs encoding proteins critical for synaptic plasticity and learning and memory including Arc, NMDA receptor subunits, the postsynaptic density scaffolding protein Homer2, and CamKII.64,65 In neurogenic niches, such as that in the dentate gyrus of the hippocampus and the subventricular zone, BDNF produced by neurons acts upon neural progenitor cells to promote their differentiation into neurons and the survival of those newly-generated neurons.13,59
BDNF is synthesized as a longer protein called proBDNF that has little or no ability to activate TrkB. Biologically active BDNF is generated by enzymatic cleavage of proBDNF; plasmin and matrix metalloprotease 9 are two proteases that have been shown to cleave proBDNF.66,67 Runner rats exhibit elevated activity of tissue-type plasminogen activator (tPA), an enzyme that converts plasminogen to plasmin, in the hippocampus suggesting that exercise increases the generation of mature/active BDNF from proBDNF.68 The latter study further showed that administration of a tPA inhibitor negates the effects of exercise on hippocampal synaptic plasticity. Another study showed that the antidepressant effect of exercise is associated with elevated levels of mature BDNF, and increased expression of tPA in the hippocampus.69
As laboratory rodents age, decrements in performance in learning and memory tasks occur that are associated with reduced LTP and decreased expression of BDNF in the dentate gyrus. These age-related deficits in hippocampal neuroplasticity can be ameliorated by exercise and environmental enrichment.70 In one study, the relative contributions of running and enrichment to the enhancement of neurogenesis in “real world-like” environments was determined by housing C57Bl/6 mice under control, running, enrichment, or enrichment plus running conditions.71 Progenitor cell proliferation and differentiation into neurons, and BDNF expression, were increased significantly only when running wheels were available, suggesting that exercise may be more effective than intellectual challenges alone in stimulating BDNF production and neurogenesis.
Collectively, the available data suggest that exercise enhances synaptic plasticity by increasing activity in neuronal circuits related to the exercise (sensory association cortices, entorhinal cortex, and hippocampus, and motor output-related circuits). Glutamate, the major excitatory neurotransmitter in all of the aforementioned neuronal circuits, activates postsynaptic AMPA and NMDA receptors, resulting in local Ca2+ influx. Ca2+ then binds calmodulin to activate CaMKII, resulting in the activation of nitric oxide synthase and the transcription factor CREB.72 CREB then induces transcription of the Bdnf gene. In addition to glutamatergic signaling, activation of serotonergic and noradrenergic neurons whose cell bodies are located in the brainstem is necessary for a maximum effect of exercise on hippocampal BDNF production.73 BDNF mediates effects of exercise on synaptic plasticity and neurogenesis by activating TrkB. As evidence, ablation of TrkB in hippocampal neural progenitor cells impairs their proliferation and their ability to differentiate into neurons under basal conditions and abolishes exercise-induced neurogenesis.74 Downstream of TrkB, the PI3 kinase–Akt pathway plays a key role in effecting changes in synapses and neural progenitor cells. Thus, when a specific inhibitor of the PI3 kinase–Akt signaling pathway was infused into the brain, the ability of voluntary running to promote synaptic plasticity and the survival of newly generated neurons in the dentate gyrus of the hippocampus was compromised.75
Exercise has been shown to improve functional outcome in animal models of acute brain insults and progressive neurodegenerative disorders, and there is likely a role for BDNF signaling in these beneficial effects of exercise. For example, traumatic brain injury impairs cognitive function in rats, and post-injury exercise can enhance cognitive performance by a BDNF-mediated mechanism.76 Neurogenesis and maze learning were suppressed in a model of systemic infection/encephalitis, and moderate running restored the proliferation and neuronal differentiation of hippocampal neural progenitor cells and also reversed the maze learning deficit.77 In two different models of Alzheimer’s disease (AD), running improved cognitive function with notable reductions in markers of oxidative stress and inflammation in the brain.78,79 Mice and rats that are runners exhibit increased resistance of their dopaminergic neurons to degeneration, and improved motor performance, in dopaminergic toxin-induced experimental Parkinson’s disease (PD).80 In the latter study it was found that levels of glial cell line-derived neurotrophic factor were greater in the striatum of runners compared to sedentary control animals. Exercise can also counteract adverse effects of a high energy diet (a risk factor for stroke, and possibly AD and PD) on synaptic plasticity and cognitive function by a mechanism involving BDNF signaling.81
Dietary energy restriction and BDNF
Dietary energy restriction (DER), both limited daily feeding (sustained caloric restriction) and IER (e.g., alternate day fasting), significantly extend healthspan and lifespan in rats and mice.82 DER can improve cognitive function and delay age-related cognitive impairment in rodents.83,84 Limited daily feeding enhances learning consolidation and synaptic plasticity by a mechanism involving increased expression of the NR2B subunit of the NMDA receptor.85 Recent findings have also demonstrated significant improvement in cognitive performance of nonhuman primates86 and human subjects87 maintained on caloric restriction diets. Hippocampal synapses in rats maintained on DER exhibit enhanced LTP.88 Neurogenesis is increased in mice maintained on an alternate day fasting diet, by a mechanism involving BDNF signaling, and increased survival of newly generated neurons.13 However, whereas alternate day fasting increases expression of BDNF, limited daily feeding has less or no detectable effect on BDNF expression.89 Further work is therefore required to determine how different types of DER affect BDNF signaling in different regions of the nervous system, and if and how those changes in BDNF signaling mediate behavioral and metabolic responses to DER.
DER is remarkable in its ability to protect neurons against dysfunction and degeneration in experimental models of neurological disorders. For example, neuroprotective and/or disease-modifying effects of IER have been demonstrated in animal models of severe epileptic seizures,90 ischemic stroke,91,92 Huntington’s disease,12,90 PD 93,94 and AD.95–98 Rats on a limited daily feeding (40% caloric restriction) diet exhibited recovery of spatial memory function, whereas rats fed ad libitum did not, in a model of cardiac arrest/global cerebral ischemia.99 Rats maintained for four months on 30% caloric restriction diet and then subjected to traumatic brain injury exhibited reduced neuronal degeneration and superior performance in a spatial learning test compared to brain injured rats on a control (ad libitum) diet.100 The improved outcome in the DER mice was associated with elevated levels of BDNF in the region of cerebral cortex around the site of injury and in the adjacent hippocampus. BDNF can protect neurons against oxidative, metabolic, and excitotoxic insults that are relevant to traumatic and ischemic brain injury by inducing the expression of antioxidant enzymes,101 anti-apoptotic Bcl-2 family members,102 and DNA repair enzymes (MPM, unpublished data).103
In a recent study, we maintained young, middle-aged, and old mice for three months on either IER (alternate day fasting) or ad libitum diets, and then subjected them to a focal ischemic stroke. The young and middle-aged mice in the IER group exhibited less brain damage and a better functional outcome, whereas IER had little or no protective effect in the old mice.92 BDNF, the protein chaperones HSP-70 and GRP-78, and the antioxidant enzyme HO-1 were upregulated by IER in the striatum and cortex of young and middle-aged, but not old mice. Moreover, the ability of IER to suppress the production of pro-inflammatory cytokines was attenuated in old mice.92 Thus, the ability of IER to enhance BDNF signaling and protect neurons against injury may be compromised with aging. The latter possibility leads to the conclusion that it is important to initiate DER (and exercise) in young adulthood or mid-life to provide maximal protection of the brain against injury and disease late in life.
BDNF mediates effects of energy intake on cognitive function
The roles of BDNF in synaptic plasticity and learning and memory have been well studied and documented.104–107 BDNF has been shown to stimulate neurogenesis.59, 108 The results of physiological studies also demonstrate that BDNF plays an important role in long-term potentiation (LTP) (see Refs. 109 and 110 for reviews).
Epidemiological studies in human populations suggest that obesity and diabetes may increase risk for developing cognitive impairments and dementia.111–113 Multiple domains of cognitive function can be impaired in patients with diabetes.114 Several studies have provided evidence that diabetes is a risk factor for age-related cognitive impairment and AD.115,116 Studies with laboratory animal models have shown that overeating and obesity promote a range of major diseases including cardiovascular disease, diabetes, and many types of cancer.117 Moreover, higher energy intake induces metabolic disorders and together with a sedentary lifestyle, this may further add risk for development of cognitive impairment.118,119 A study conducted in rats indicated that animals maintained with food elevated in saturated fat or a diet with higher saturated fat and cholesterol committed memory errors compared to controls maintained with normal diets when they were tested in several different types of behavioral tasks.120,121
In a study from our lab using the triple-transgenic mouse model of AD (3xTgAD mice), the mice were maintained for one year (beginning at five months of age) on either ad libitum (control), 40% CR or IER (alternate day fasting) diets. Behavioral testing of the 17-month-old 3xTgAD mice showed that those on the CR and IER diets exhibited higher levels of exploratory behavior, and performed better in both the goal latency and probe trials of the water maze task compared to 3xTgAD mice on a control diet. 3xTgAD mice in the CR group also showed lower levels of Aβ1–40, Aβ1–42, and phospho-Tau in the hippocampus compared to the control diet group.97 At least one other study has reported that long-term IER can enhance cognitive performance in rats and mice.85
Synaptic plasticity and BDNF
The mechanism(s) by which BDNF enables and enforces the changes in synaptic structure and function believed to underlie learning and memory are being elucidated. Although studies have shown that energy restriction affects BDNF expression in multiple brain areas, including cortical and subcortical regions, the hippocampus has receiving the most attention in various studies. The hippocampus has been a focus for studies of cognition-related synaptic plasticity because of the layered structural organization of the neurons and synaptic connections that provides the opportunity to readily perform electrophysiological recordings of synaptic transmission and long-term changes in synaptic strength. In addition, many of the currently well-characterized behavioral tasks used to evaluate memory, particularly for rodents, are hippocampus-dependent. BDNF and LTP have received a great deal of attention in neuroscience research (see 109 and 110 for reviews). BDNF plays a critical role in activity-induced expression of proteins and in generating sustained structural and functional changes at hippocampal synapses. In particular, BDNF is sufficient to induce the transformation of early to late-phase LTP.109 One of our recent studies showed that rats maintained on a high-fat, high-glucose diet exhibited impaired spatial learning ability, reduced hippocampal dendritic spine density, and reduced LTP at Schaffer collateral–CA1 synapses.122 These changes were associated with reductions in levels of BDNF in the hippocampus. Therefore, a diabetogenic high-calorie diet reduces hippocampal synaptic plasticity and impairs cognitive function, possibly by impairing BDNF-mediated effects on dendritic spines. In a mouse model, we also found that excessive energy intake resulted in impaired leptin signaling and hippocampal neurogenesis, and was associated with deficits in synaptic plasticity at the perforant path—dentate granule neuron synapses and impaired hippocampus-dependent memory.45 Other studies reported a complementary result showing that a CR protocol prevents rats from developing age-related deficits in LTP and sustains NMDA and AMPA glutamate receptor levels in the hippocampus.88,123
BDNF and cellular energy metabolism
An additional mechanism by which BDNF may mediate beneficial effects of dietary energy restriction on cognition involves enhancing cellular energy metabolism.124 In a recent study, it was shown that when 3xTgAD mice were fed a diet supplemented with 2-deoxyglucose (2DG) for seven weeks, levels of Aβ in their brains decreased, levels of BDNF increased, and neuronal bioenergetic capacity was increased.125 Mice on the 2DG diet also exhibited elevated levels of ketone bodies, which are fatty acids known to affect neuronal excitability and to protect neurons against excitotoxic and metabolic insults.126 BDNF has been shown to directly modify neuronal energy metabolism. For example, BDNF induces the expression of the monocarboxylate transporter that enables the use of lactate as an alternative energy source.127 The ability of BDNF to induce the monocarboxylate transporter was blocked by inhibitors of PI3 kinase and p42/p44 MAP kinases. Another study showed that BDNF can stimulate glucose utilization and the upregulation of the glucose transporter GLUT3 in cultured neurons.128 BDNF also stimulates sodium-dependent amino acid transport and increases protein synthesis. The abilities of BDNF to increase cellular energy availability and enhance protein synthesis may play important roles in synaptic plasticity because energy substrates (ATP and NAD+) and protein synthesis are critical for synaptic plasticity and learning and memory.129,130
The big picture: BDNF as an integrator of behavioral and neuroendocrine regulation of energy metabolism
Consider mammals, including our primate predecessors and ourselves, humans, in the natural environments in which they evolved. Further consider two very different ends of the “energy equation” with regards to the availability/accessibility of energy (food) resources within the environment (Fig. 4). In one scenario (challenging environment) food is scarce, competition within and among species for the energy is high, and hazards such as predation also enter into the energy equation on both the acquisition and expenditure sides. Such challenging environments demand relatively higher levels of processing and retention (memory) of information from the environment (particularly sights, sounds, and odors). The underlying activity in neuronal circuits involves BDNF production and signaling which promotes synaptic plasticity, the growth of dendrites, and neurogenesis. Individuals in physically challenging environments are at increased risk of injury, including damage to the nervous system. BDNF may provide a survival advantage to those suffering trauma to the nervous system by stimulating mitochondrial biogenesis and resistance of neurons to injury by upregulating the expression of genes encoding cytoprotective proteins. Recent findings suggest that BDNF signaling in the brain mediates the positive effects of a cognitively and physically challenging environment on energy metabolism including increased insulin sensitivity,17 increased genesis of brown fat,131,132 and cardiovascular fitness.133
Figure 4.
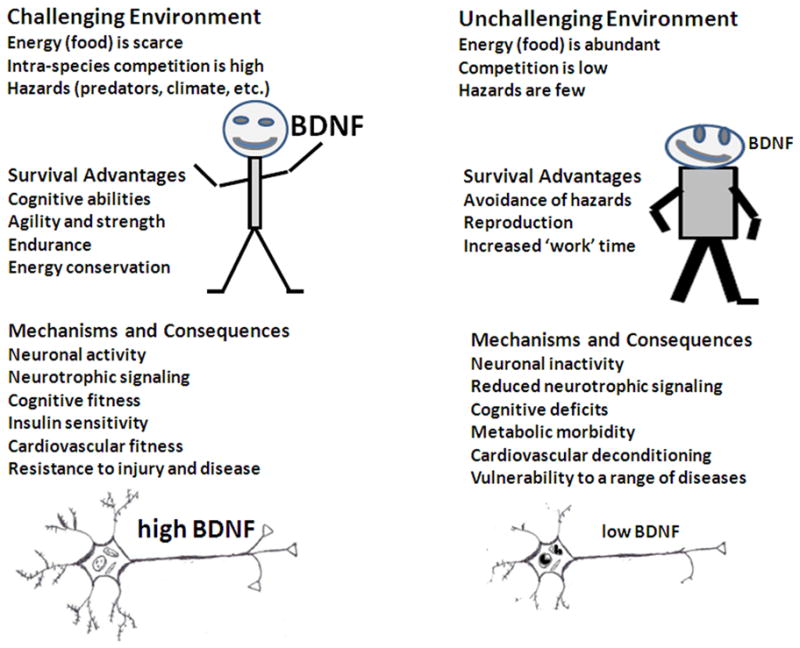
Characteristics of individuals living in natural or self-imposed environments at the two ends of the “energy spectrum.” (Left) An environment where food is scarce and competition and hazards are high, poses major physical and cognitive challenges. From an evolutionary perspective, survival is favored by phenotypes that include superior cognitive abilities, strength and agility, endurance, and the ability to conserve energy. In this setting, neurons are active and the production of neurotrophic factors such as BDNF are elevated. Emerging evidence suggests that BDNF mediates multiple responses of the individual to a Spartan environment, including enhanced neuronal plasticity, increased insulin sensitivity, improved cardiovascular fitness, and resistance of tissues to injury and disease. These different BDNF-mediated responses to a challenging environment can be considered a coordinated adaptive cellular and systems level response to the reduced energy availability and increased energy expenditure required to survive. (Right) In an environment where food is abundant and the need for energy expenditure (exercise) minimal, the individual can increase their attention to reproduction and their occupation within the society. This environment results in reduced activation of adaptive stress response pathways resulting in reduced BDNF signaling, insulin resistance, cardiovascular deconditioning, and susceptibility to a range of diseases, particularly age-related diseases.
In the second scenario (unchallenging environment), energy is readily available and there are few hazards to obtaining the energy (Fig. 4). The time that would otherwise be used for food acquisition is used for reproduction and work to obtain other types of resources that provide an advantage to the individuals and their families. Because energy intake is increased and physical activity reduced in such an environment, BDNF signaling is reduced in neurons in the brain. Reduced BDNF signaling enhances food intake, reduces insulin sensitivity, deconditions the cardiovascular system, reduces cognitive abilities, and increases vulnerability to age-related diseases.
In summary, individuals will be lean, alert, and cognitively “sharp” when living in an environment in which there are major challenges for energy acquisition. This same (healthy) physical and cognitive phenotype can be achieved by self-imposed energy restriction and exercise routines in humans. BDNF signaling plays major roles in mediating adaptive responses of the nervous, cardiovascular, and energy-regulating organ systems in response to exercise and energy restriction. Efforts to develop pharmacological and molecular genetic methods for activating BDNF signaling pathways may result in novel therapeutic treatments for a range of metabolic and neurological disorders.
Acknowledgments
This work was supported by the Intramural Research Program of the National Institute on Aging.
Footnotes
Conflicts of interest
The authors declare no conflicts of interest.
References
- 1.Nawa H, Carnahan J, Gall C. BDNF protein measured by a novel enzyme immunoassay in normal brain and after seizure: partial disagreement with mRNA levels. Eur J Neurosci. 1995;7:1527–1535. doi: 10.1111/j.1460-9568.1995.tb01148.x. [DOI] [PubMed] [Google Scholar]
- 2.Duan W, Guo Z, Jiang H, Ware M, Mattson MP. Reversal of behavioral and metabolic abnormalities, and insulin resistance syndrome, by dietary restriction in mice deficient in brain-derived neurotrophic factor. Endocrinol. 2003b;144:2446–2453. doi: 10.1210/en.2002-0113. [DOI] [PubMed] [Google Scholar]
- 3.Kernie SG, Liebl DJ, Parada LF. BDNF regulates eating behavior and locomotor activity in mice. EMBO J. 2000;19:1290–1300. doi: 10.1093/emboj/19.6.1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coppola V, Tessarollo L. Control of hyperphagia prevents obesity in BDNF heterozygous mice. Neuroreport. 2004;15:2665–2668. doi: 10.1097/00001756-200412030-00022. [DOI] [PubMed] [Google Scholar]
- 5.Xu B, Goulding EH, Zang K, Cepoi D, Cone RD, Jones KR, Tecott LH, Reichardt LF. Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat Neurosci. 2003;6:736–742. doi: 10.1038/nn1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rios M, Fan G, Fekete C, Kelly J, Bates B, Kuehn R, Lechan RM, Jaenisch R. Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol Endocrinol. 2001;15:1748–1757. doi: 10.1210/mend.15.10.0706. [DOI] [PubMed] [Google Scholar]
- 7.Pelleymounter MA, Cullen MJ, Wellman CL. Characteristics of BDNF-induced weight loss. Exp Neurol. 1995;131:229–238. doi: 10.1016/0014-4886(95)90045-4. [DOI] [PubMed] [Google Scholar]
- 8.Shimizu N, Oomura Y, Plata-Salamán CR, Morimoto M. Hyperphagia and obesity in rats with bilateral ibotenic acid-induced lesions of the ventromedial hypothalamic nucleus. Brain Res. 1987;416:153–156. doi: 10.1016/0006-8993(87)91508-3. [DOI] [PubMed] [Google Scholar]
- 9.Grill HJ, Kaplan JM. The neuroanatomical axis for control of energy balance. Front Neuroendocrinol. 2002;23:2–40. doi: 10.1006/frne.2001.0224. [DOI] [PubMed] [Google Scholar]
- 10.Bariohay B, Lebrun B, Moyse E, Jean A. Brain-derived neurotrophic factor plays a role as an anorexigenic factor in the dorsal vagal complex. Endocrinol. 2005;146:5612–5620. doi: 10.1210/en.2005-0419. [DOI] [PubMed] [Google Scholar]
- 11.Lee J, Duan W, Long JM, Ingram DK, Mattson MP. Dietary restriction increases the number of newly generated neural cells, and induces BDNF expression, in the dentate gyrus of rats. J Mol Neurosci. 2000;15:99–108. doi: 10.1385/JMN:15:2:99. [DOI] [PubMed] [Google Scholar]
- 12.Duan W, Guo Z, Jiang H, Ware M, Li XJ, Mattson MP. Dietary restriction normalizes glucose metabolism and BDNF levels, slows disease progression, and increases survival in huntingtin mutant mice. PNAS. 2003a;100:2911–2916. doi: 10.1073/pnas.0536856100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee J, Duan W, Mattson MP. Evidence that brain-derived neurotrophic factor is required for basal neurogenesis and mediates, in part, the enhancement of neurogenesis by dietary restriction in the hippocampus of adult mice. J Neurochem. 2002;82:1367–1375. doi: 10.1046/j.1471-4159.2002.01085.x. [DOI] [PubMed] [Google Scholar]
- 14.Krabbe KS, Nielsen AR, Krogh-Madsen R, Plomgaard P, Rasmussen P, Erikstrup C, Fischer CP, Lindegaard B, Petersen AM, Taudorf S, Secher NH, Pilegaard H, Bruunsgaard HH, Pedersen BK. Brain-derived neurotrophic factor (BDNF) and type 2 diabetes. Diabetologia. 2007;50:431–438. doi: 10.1007/s00125-006-0537-4. [DOI] [PubMed] [Google Scholar]
- 15.Pedersen BK, Pedersen M, Krabbe KS, Bruunsgaard H, Matthews VB, Febbraio MA. Role of exercise-induced brain-derived neurotrophic factor production in the regulation of energy homeostasis in mammals. Exp Physiol. 2009;94:1153–1160. doi: 10.1113/expphysiol.2009.048561. [DOI] [PubMed] [Google Scholar]
- 16.Nakagawa T, Ono-Kishino M, Sugaru E, Yamanaka M, Taiji M, Noguchi H. Brain-derived neurotrophic factor (BDNF) regulates glucose and energy metabolism in diabetic mice. Diabetes Metab Res Rev. 2002;18:185–191. doi: 10.1002/dmrr.290. [DOI] [PubMed] [Google Scholar]
- 17.Nakagawa T, Tsuchida A, Itakura Y, Nonomura T, Ono M, Hirota F, Inoue T, Nakayama C, Taiji M, Noguchi H. Brain-derived neurotrophic factor regulates glucose metabolism by modulating energy balance in diabetic mice. Diabetes. 2000;49:436–444. doi: 10.2337/diabetes.49.3.436. [DOI] [PubMed] [Google Scholar]
- 18.Spiegelman BM, Flier JS. Adipogenesis and obesity: rounding out the big picture. Cell. 1996;87:377–389. doi: 10.1016/s0092-8674(00)81359-8. [DOI] [PubMed] [Google Scholar]
- 19.Ono M, Itakura Y, Nonomura T, Nakagawa T, Nakayama C, Taiji M, Noguchi H. Intermittent administration of brain-derived neurotrophic factor ameliorates glucose metabolism in obese diabetic mice. Metabolism. 2000;49:129–133. doi: 10.1016/s0026-0495(00)90988-0. [DOI] [PubMed] [Google Scholar]
- 20.Stranahan AM, Arumugam TV, Mattson MP. Lowering corticosterone levels reinstates hippocampal brain-derived neurotropic factor and Trkb expression without influencing deficits in hypothalamic brain-derived neurotropic factor expression in leptin receptor-deficient mice. Neuroendocrinol. 2011;93:58–64. doi: 10.1159/000322808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Brady R, Zaidi SI, Mayer C, Katz DM. BDNF is a target-derived survival factor for arterial baroreceptor and chemoafferent primary sensory neurons. J Neurosci. 1999;19:2131–2142. doi: 10.1523/JNEUROSCI.19-06-02131.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee J, Seroogy KB, Mattson MP. Dietary restriction enhances neurotrophin expression and neurogenesis in the hippocampus of adult mice. J Neurochem. 2002b;80:539–547. doi: 10.1046/j.0022-3042.2001.00747.x. [DOI] [PubMed] [Google Scholar]
- 23.Neeper SA, Gomez-Pinilla F, Choi J, Cotman CW. Physical activity increases mRNA for brain-derived neurotrophic factor and nerve growth factor in rat brain. Brain Res. 1996;726:49–56. [PubMed] [Google Scholar]
- 24.Rosenwinkel ET, Bloomfield DM, Arwady MA, Goldsmith RL. Exercise and autonomic function in health and cardiovascular disease. Cardiol Clin. 2001;19:369–387. doi: 10.1016/s0733-8651(05)70223-x. [DOI] [PubMed] [Google Scholar]
- 25.Wan R, Camandola S, Mattson MP. Intermittent fasting and dietary supplementation with 2-deoxy-D-glucose improve functional and metabolic cardiovascular risk factors in rats. Faseb J. 2003a;17:1133–1134. doi: 10.1096/fj.02-0996fje. [DOI] [PubMed] [Google Scholar]
- 26.Wan R, Camandola S, Mattson MP. Intermittent food deprivation improves cardiovascular and neuroendocrine responses to stress in rats. J Nutr. 2003b;133:1921–1929. doi: 10.1093/jn/133.6.1921. [DOI] [PubMed] [Google Scholar]
- 27.Yang B, Slonimsky JD, Birren SJ. A rapid switch in sympathetic neurotransmitter release properties mediated by the p75 receptor. Nat Neurosci. 2002;5:539–545. doi: 10.1038/nn0602-853. [DOI] [PubMed] [Google Scholar]
- 28.Ahmet I, Wan R, Mattson MP, Lakatta EG, Talan M. Cardioprotection by intermittent fasting in rats. Circulation. 2005;112:3115–3121. doi: 10.1161/CIRCULATIONAHA.105.563817. [DOI] [PubMed] [Google Scholar]
- 29.Yang AC, Chen TJ, Tsai SJ, Hong CJ, Kuo CH, Yang CH, Kao KP. BDNF Val66Met polymorphism alters sympathovagal balance in healthy subjects. Am J Med Genet B Neuropsychiatr Genet. 2010;153B:1024–1030. doi: 10.1002/ajmg.b.31069. [DOI] [PubMed] [Google Scholar]
- 30.Alexander N, Osinsky R, Schmitz A, Mueller E, Kuepper Y, Hennig J. The BDNF Val66Met polymorphism affects HPA-axis reactivity to acute stress. Psychoneuroendocrinology. 2010;35:949–953. doi: 10.1016/j.psyneuen.2009.12.008. [DOI] [PubMed] [Google Scholar]
- 31.Martin JL, V, Jenkins K, Hsieh HY, Balkowiec A. Brain-derived neurotrophic factor in arterial baroreceptor pathways: implications for activity-dependent plasticity at baroafferent synapses. J Neurochem. 2009;108:450–464. doi: 10.1111/j.1471-4159.2008.05781.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hofer M, Pagliusi SR, Hohn A, Leibrock J, Barde YA. Regional distribution of brain-derived neurotrophic factor mRNA in the adult mouse brain. EMBO. 1990;9:2459–2464. doi: 10.1002/j.1460-2075.1990.tb07423.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Peiris TS, Machaalani R, Waters KA. Brain-derived neurotrophic factor mRNA and protein in the piglet brainstem and effects of Intermittent Hypercapnic Hypoxia. Brain Res. 2004;1029:11–23. doi: 10.1016/j.brainres.2004.09.024. [DOI] [PubMed] [Google Scholar]
- 34.Schober A, Wolf N, Huber K, Hertel R, Krieglstein K, Minichiello L, Kahane N, Widenfalk J, Kalcheim C, Olson L, Klein R, Lewin GR, Unsicker K. TrkB and neurotrophin-4 are important for development and maintenance of sympathetic preganglionic neurons innervating the adrenal medulla. J Neurosci. 1998;18:7272–7284. doi: 10.1523/JNEUROSCI.18-18-07272.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yan Q, Radeke MJ, Matheson CR, Talvenheimo J, Welcher AA, Feinstein SC. Immunocytochemical localization of TrkB in the central nervous system of the adult rat. J Comp Neurol. 1997;378:135–157. [PubMed] [Google Scholar]
- 36.Altar CA, Cai N, Bliven T, Juhasz M, Conner JM, Acheson AL, Lindsay RM, Wiegand SJ. Anterograde transport of brain-derived neurotrophic factor and its role in the brain. Nature. 1997;389:856–860. doi: 10.1038/39885. [DOI] [PubMed] [Google Scholar]
- 37.Helke CJ, Adryan KM, Fedorowicz J, Zhuo H, Park JS, Curtis R, Radley HE, Distefano PS. Axonal transport of neurotrophins by visceral afferent and efferent neurons of the vagus nerve of the rat. J Comp Neurol. 1998;393:102–117. doi: 10.1002/(sici)1096-9861(19980330)393:1<102::aid-cne10>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- 38.Wang H, Zhou XF. Injection of brain-derived neurotrophic factor in the rostral ventrolateral medulla increases arterial blood pressure in anaesthetized rats. Neuroscience. 2002;112:967–975. doi: 10.1016/s0306-4522(02)00085-4. [DOI] [PubMed] [Google Scholar]
- 39.Nicholson JR, Peter JC, Lecourt AC, Barde YA, Hofbauer KG. Melanocortin-4 receptor activation stimulates hypothalamic brain-derived neurotrophic factor release to regulate food intake, body temperature and cardiovascular function. J Neuroendocrinol. 2007;19:974–982. doi: 10.1111/j.1365-2826.2007.01610.x. [DOI] [PubMed] [Google Scholar]
- 40.Andresen MC, Kunze DL. Nucleus tractus solitarius--gateway to neural circulatory control. Ann Rev Physiol. 1994;56:93–116. doi: 10.1146/annurev.ph.56.030194.000521. [DOI] [PubMed] [Google Scholar]
- 41.Clark CG, Hasser EM, Kunze DL, Katz DM, Kline DD. Endogenous brain-derived neurotrophic factor in the nucleus tractus solitarius tonically regulates synaptic and autonomic function. J Neurosci. 2011;31:12318–12329. doi: 10.1523/JNEUROSCI.0746-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cotman CW, Berchtold NC, Christie LA. Exercise builds brain health: key roles of growth factor cascades and inflammation. Trends Neurosci. 2007;30:464–472. doi: 10.1016/j.tins.2007.06.011. [DOI] [PubMed] [Google Scholar]
- 43.van Praag H, Kempermann G, Gage FH. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999;2:266–270. doi: 10.1038/6368. [DOI] [PubMed] [Google Scholar]
- 44.Farmer J, Zhao X, van Praag H, Wodtke K, Gage FH, Christie BR. Effects of voluntary exercise on synaptic plasticity and gene expression in the dentate gyrus of adult male Sprague-Dawley rats in vivo. Neuroscience. 2004;124:71–79. doi: 10.1016/j.neuroscience.2003.09.029. [DOI] [PubMed] [Google Scholar]
- 45.Stranahan AM, Arumugam TV, Cutler RG, Lee K, Egan JM, Mattson MP. Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons. Nat Neurosci. 2008;11:309–317. doi: 10.1038/nn2055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bruestle DA, Cutler RG, Telljohann RS, Mattson MP. Decline in daily running distance presages disease onset in a mouse model of ALS. Neuromol Med. 2009;11:58–62. doi: 10.1007/s12017-009-8064-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Knab AM, Bowen RS, Moore-Harrison T, Hamilton AT, Turner MJ, Lightfoot JT. Repeatability of exercise behaviors in mice. Physiol Behav. 2009;98:433–440. doi: 10.1016/j.physbeh.2009.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Groves-Chapman JL, Murray PS, Stevens KL, Monroe DC, Koch LG, Britton SL, Holmes PV, Dishman RK. Changes in mRNA levels for brain-derived neurotrophic factor after wheel running in rats selectively bred for high- and low-aerobic capacity. Brain Res. 2011;1425:90–97. doi: 10.1016/j.brainres.2011.09.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mattson MP, Duan W, Guo Z. Meal size and frequency affect neuronal plasticity and vulnerability to disease: cellular and molecular mechanisms. J Neurochem. 2003;84:417–431. doi: 10.1046/j.1471-4159.2003.01586.x. [DOI] [PubMed] [Google Scholar]
- 50.Martin B, Mattson MP, Maudsley S. Caloric restriction and intermittent fasting: two potential diets for successful brain aging. Ageing Res Rev. 2006;5:332–353. doi: 10.1016/j.arr.2006.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Berchtold NC, Castello N, Cotman CW. Exercise and time-dependent benefits to learning and memory. Neuroscience. 2010;167:588–597. doi: 10.1016/j.neuroscience.2010.02.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Berchtold NC, Chinn G, Chou M, Kesslak JP, Cotman CW. Exercise primes a molecular memory for brain-derived neurotrophic factor protein induction in the rat hippocampus. Neuroscience. 2005;133:853–861. doi: 10.1016/j.neuroscience.2005.03.026. [DOI] [PubMed] [Google Scholar]
- 53.Creer DJ, Romberg C, Saksida LM, van Praag H, Bussey T. Running enhances spatial pattern separation in mice. PNAS. 2010;107:2367–2372. doi: 10.1073/pnas.0911725107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, Chaddock L, Kim JS, Heo S, Alves H, White SM, Wojcicki TR, Mailey E, Vieira VJ, Martin SA, Pence BD, Woods JA, McAuley E, Kramer AF. Exercise training increases size of hippocampus and improves memory. PNAS. 2011;108:3017–3022. doi: 10.1073/pnas.1015950108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liu YF, Chen HI, Wu CL, Kuo YM, Yu L, Huang AM, Wu FS, Chuang JI, Jen CJ. Differential effects of treadmill running and wheel running on spatial or aversive learning and memory: roles of amygdalar brain-derived neurotrophic factor and synaptotagmin I. J Physiol. 2009;587:3221–3231. doi: 10.1113/jphysiol.2009.173088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lin TW, Chen SJ, Huang TY, Chang CY, Chuang JI, Wu FS, Kuo YM, Jen CJ. Different types of exercise induce differential effects on neuronal adaptations and memory performance. Neurobiol Learn Mem. 2011 doi: 10.1016/j.nlm.2011.10.006. Epub. [DOI] [PubMed] [Google Scholar]
- 57.Russo-Neustadt AA, Beard RC, Huang YM, Cotman CW. Physical activity and antidepressant treatment potentiate the expression of specific brain-derived neurotrophic factor transcripts in the rat hippocampus. Neuroscience. 2000;101:305–312. doi: 10.1016/s0306-4522(00)00349-3. [DOI] [PubMed] [Google Scholar]
- 58.Adlard PA, V, Perreau M, Engesser-Cesar C, Cotman CW. The timecourse of induction of brain-derived neurotrophic factor mRNA and protein in the rat hippocampus following voluntary exercise. Neurosci Lett. 2004;363:43–48. doi: 10.1016/j.neulet.2004.03.058. [DOI] [PubMed] [Google Scholar]
- 59.Cheng A, Wang S, Cai J, Rao MS, Mattson MP. Nitric oxide acts in a positive feedback loop with BDNF to regulate neural progenitor cell proliferation and differentiation in the mammalian brain. Dev Biol. 2003;258:319–333. doi: 10.1016/s0012-1606(03)00120-9. [DOI] [PubMed] [Google Scholar]
- 60.Chen MJ, Ivy AS, Russo-Neustadt AA. Nitric oxide synthesis is required for exercise-induced increases in hippocampal BDNF and phosphatidylinositol 3′ kinase expression. Brain Res Bull. 2006;68:257–268. doi: 10.1016/j.brainresbull.2005.08.013. [DOI] [PubMed] [Google Scholar]
- 61.Jourdi H, Hsu YT, Zhou M, Qin Q, Bi X, Baudry M. Positive AMPA receptor modulation rapidly stimulates BDNF release and increases dendritic mRNA translation. J Neurosci. 2009;29:8688–8697. doi: 10.1523/JNEUROSCI.6078-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Stranahan AM, Lee K, Martin B, Maudsley S, Golden E, Cutler RG, Mattson MP. Voluntary exercise and caloric restriction enhance hippocampal dendritic spine density and BDNF levels in diabetic mice. Hippocampus. 2009;19:951–961. doi: 10.1002/hipo.20577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wu YC, Williamson R, Li Z, Vicario A, Xu J, Kasai M, Chern Y, Tongiorgi E, Baraban JM. Dendritic trafficking of brain-derived neurotrophic factor mRNA: regulation by translin-dependent and -independent mechanisms. J Neurochem. 2011;116:1112–1121. doi: 10.1111/j.1471-4159.2010.07166.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Yin Y, Edelman GM, Vanderklish PW. The brain-derived neurotrophic factor enhances synthesis of Arc in synaptoneurosomes. PNAS. 2002;99:2368–2373. doi: 10.1073/pnas.042693699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Schratt GM, Nigh EA, Chen WG, Hu L, Greenberg ME. BDNF regulates the translation of a select group of mRNAs by a mammalian target of rapamycin-phosphatidylinositol 3-kinase-dependent pathway during neuronal development. J Neurosci. 2004;24:7366–7377. doi: 10.1523/JNEUROSCI.1739-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pang PT, Teng HK, Zaitsev E, Woo NT, Sakata K, Zhen S, Teng KK, Yung WH, Hempstead BL, Lu B. Cleavage of proBDNF by tPA/plasmin is essential for long-term hippocampal plasticity. Science. 2004;306:487–491. doi: 10.1126/science.1100135. [DOI] [PubMed] [Google Scholar]
- 67.Mizoguchi H, Nakade J, Tachibana M, Ibi D, Someya E, Koike H, Kamei H, Nabeshima T, Itohara S, Takuma K, Sawada M, Sato J, Yamada K. Matrix metalloproteinase-9 contributes to kindled seizure development in pentylenetetrazole-treated mice by converting pro-BDNF to mature BDNF in the hippocampus. J Neurosci. 2011;31:12963–12971. doi: 10.1523/JNEUROSCI.3118-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ding Q, Ying Z, Gómez-Pinilla F. Exercise influences hippocampal plasticity by modulating brain-derived neurotrophic factor processing. Neuroscience. 2011;192:773–780. doi: 10.1016/j.neuroscience.2011.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sartori CR, Vieira AS, Ferrari EM, Langone F, Tongiorgi E, Parada CA. The antidepressive effect of the physical exercise correlates with increased levels of mature BDNF, and proBDNF proteolytic cleavage-related genes, p11 and tPA. Neuroscience. 2011;180:9–18. doi: 10.1016/j.neuroscience.2011.02.055. [DOI] [PubMed] [Google Scholar]
- 70.O’Callaghan RM, Griffin EW, Kelly AM. Long-term treadmill exposure protects against age-related neurodegenerative change in the rat hippocampus. Hippocampus. 2009;19:1019–1029. doi: 10.1002/hipo.20591. [DOI] [PubMed] [Google Scholar]
- 71.Kobilo T, Liu QR, Gandhi K, Mughal M, Shaham Y, van Praag H. Running is the neurogenic and neurotrophic stimulus in environmental enrichment. Learn Mem. 2011;18:605–609. doi: 10.1101/lm.2283011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chen MJ, Russo-Neustadt AA. Running exercise-induced up-regulation of hippocampal brain-derived neurotrophic factor is CREB-dependent. Hippocampus. 2009;19:962–972. doi: 10.1002/hipo.20579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ivy AS, Rodriguez FG, Garcia C, Chen MJ, Russo-Neustadt AA. Noradrenergic and serotonergic blockade inhibits BDNF mRNA activation following exercise and antidepressant. Pharmacol Biochem Behav. 2003;75:81–88. doi: 10.1016/s0091-3057(03)00044-3. [DOI] [PubMed] [Google Scholar]
- 74.Li Y, Luikart BW, Birnbaum S, Chen J, Kwon CH, Kernie SG, Bassel-Duby R, Parada LF. TrkB regulates hippocampal neurogenesis and governs sensitivity to antidepressive treatment. Neuron. 2008;59:399–412. doi: 10.1016/j.neuron.2008.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bruel-Jungerman E, Veyrac A, Dufour F, Horwood J, Laroche S, Davis S. Inhibition of PI3K-Akt signaling blocks exercise-mediated enhancement of adult neurogenesis and synaptic plasticity in the dentate gyrus. PLoS One. 2009;4:e7901. doi: 10.1371/journal.pone.0007901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Griesbach GS, Hovda DA, Gomez-Pinilla F. Exercise-induced improvement in cognitive performance after traumatic brain injury in rats is dependent on BDNF activation. Brain Res. 2009;1288:105–115. doi: 10.1016/j.brainres.2009.06.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wu CW, Chen YC, Yu L, Chen HI, Jen CJ, Huang AM, Tsai HJ, Chang YT, Kuo YM. Treadmill exercise counteracts the suppressive effects of peripheral lipopolysaccharide on hippocampal neurogenesis and learning and memory. J Neurochem. 2007;103:2471–2481. doi: 10.1111/j.1471-4159.2007.04987.x. [DOI] [PubMed] [Google Scholar]
- 78.Parachikova A, Nichol KE, Cotman CW. Short-term exercise in aged Tg2576 mice alters neuroinflammation and improves cognition. Neurobiol Dis. 2008;30:121–129. doi: 10.1016/j.nbd.2007.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.García-Mesa Y, López-Ramos JC, Giménez-Llort L, Revilla S, Guerra R, Gruart A, Laferla FM, Cristòfol R, Delgado-García JM, Sanfeliu C. Physical exercise protects against Alzheimer’s disease in 3xTg-AD mice. J Alzheimers Dis. 2011;24:421–454. doi: 10.3233/JAD-2011-101635. [DOI] [PubMed] [Google Scholar]
- 80.Zigmond MJ, Cameron JL, Leak RK, Mirnics K, Russell VA, Smeyne RJ, Smith AD. Triggering endogenous neuroprotective processes through exercise in models of dopamine deficiency. Parkinsonism Relat Disord. 2009;15(Suppl 3):S42–45. doi: 10.1016/S1353-8020(09)70778-3. [DOI] [PubMed] [Google Scholar]
- 81.Molteni R, Wu A, Vaynman S, Ying Z, Barnard RJ, Gómez-Pinilla F. Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience. 2004;123:429–440. doi: 10.1016/j.neuroscience.2003.09.020. [DOI] [PubMed] [Google Scholar]
- 82.Masoro EJ. Overview of caloric restriction and ageing. Mech Ageing Dev. 2005;126:913–922. doi: 10.1016/j.mad.2005.03.012. [DOI] [PubMed] [Google Scholar]
- 83.Means LW, Higgins JL, Fernandez TJ. Mid-life onset of dietary restriction extends life and prolongs cognitive functioning. Physiol Behav. 1993;54:503–508. doi: 10.1016/0031-9384(93)90243-9. [DOI] [PubMed] [Google Scholar]
- 84.Komatsu T, Chiba T, Yamaza H, Yamashita K, Shimada A, Hoshiyama Y, Henmi T, Ohtani H, Higami Y, de Cabo R, Ingram DK, Shimokawa I. Manipulation of caloric content but not diet composition, attenuates the deficit in learning and memory of senescence-accelerated mouse strain P8. Exp Gerontol. 2008;43:339–346. doi: 10.1016/j.exger.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 85.Fontán-Lozano A, Sáez-Cassanelli JL, Inda MC, de los Santos-Arteaga M, Sierra-Domínguez SA, López-Lluch G, Delgado-García JM, Carrión AM. Caloric restriction increases learning consolidation and facilitates synaptic plasticity through mechanisms dependent on NR2B subunits of the NMDA receptor. J Neurosci. 2007;27:10185–10195. doi: 10.1523/JNEUROSCI.2757-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Dal-Pan A, Pifferi F, Marchal J, Picq JL, Aujard F. Cognitive performances are selectively enhanced during chronic caloric restriction or resveratrol supplementation in a primate. PLoS One. 2011;6:e16581. doi: 10.1371/journal.pone.0016581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Witte AV, Fobker M, Gellner R, Knecht S, Flöel A. Caloric restriction improves memory in elderly humans. PNAS. 2009;106:1255–1260. doi: 10.1073/pnas.0808587106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Eckles-Smith K, Clayton D, Bickford P, Browning MD. Caloric restriction prevents age-related deficits in LTP and in NMDA receptor expression. Mol Brain Res. 2000;78:154–162. doi: 10.1016/s0169-328x(00)00088-7. [DOI] [PubMed] [Google Scholar]
- 89.Newton IG, Forbes ME, Legault C, Johnson JE, Brunso-Bechtold JK, Riddle DR. Caloric restriction does not reverse aging-related changes in hippocampal BDNF. Neurobiol Aging. 2005;26:683–688. doi: 10.1016/j.neurobiolaging.2004.06.005. [DOI] [PubMed] [Google Scholar]
- 90.Bruce-Keller AJ, Umberger G, McFall R, Mattson MP. Food restriction reduces brain damage and improves behavioral outcome following excitotoxic and metabolic insults. Ann Neurol. 1999;45:8–15. [PubMed] [Google Scholar]
- 91.Yu ZF, Mattson MP. Dietary restriction and 2-deoxyglucose administration reduce focal ischemic brain damage and improve behavioral outcome: evidence for a preconditioning mechanism. J Neurosci Res. 1999;57:830–839. [PubMed] [Google Scholar]
- 92.Arumugam TV, Phillips TM, Cheng A, Morrell CH, Mattson MP, Wan RQ. Age and energy intake interact to modify cell stress pathways and stroke outcome. Ann Neurol. 2010;67:41–52. doi: 10.1002/ana.21798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Duan W, Mattson MP. Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease. J Neurosci Res. 1999;57:195–206. doi: 10.1002/(SICI)1097-4547(19990715)57:2<195::AID-JNR5>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 94.Maswood N, Young J, Tilmont E, Zhang Z, Gash DM, Gerhardt GA, Grondin R, Roth GS, Mattison J, Lane MA, Carson RE, Cohen RM, Mouton PR, Quigley C, Mattson MP, Ingram DK. Caloric restriction increases neurotrophic factor levels and attenuates neurochemical and behavioral deficits in a primate model of Parkinson’s disease. PNAS. 2004;101:18171–18176. doi: 10.1073/pnas.0405831102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Patel NV, Gordon MN, Connor KE, Good RA, Engelman RW, Mason J, Morgan DG, Morgan TE, Finch CE. Caloric restriction attenuates Abeta-deposition in Alzheimer transgenic models. Neurobiol Aging. 2005;26:995–1000. doi: 10.1016/j.neurobiolaging.2004.09.014. [DOI] [PubMed] [Google Scholar]
- 96.Qin W, Chachich M, Lane M, Roth G, Bryant M, de Cabo R, Ottinger MA, Mattison J, Ingram DK, Gandy S, Pasinetti GM. Calorie restriction attenuates Alzheimer’s disease type brain amyloidosis in Squirrel monkeys (Saimiri sciureus) J Alzheimers Dis. 2006;10:417–422. doi: 10.3233/jad-2006-10411. [DOI] [PubMed] [Google Scholar]
- 97.Halagappa VK, Guo Z, Pearson M, Matsuoka Y, Cutler RG, Laferla FM, Mattson MP. Intermittent fasting and caloric restriction ameliorate age-related behavioral deficits in the triple-transgenic mouse model of Alzheimer’s disease. Neurobiol Dis. 2007;26:212–220. doi: 10.1016/j.nbd.2006.12.019. [DOI] [PubMed] [Google Scholar]
- 98.Wu P, Shen Q, Dong S, Xu Z, Tsien JZ, Hu Y. Calorie restriction ameliorates neurodegenerative phenotypes in forebrain-specific presenilin-1 and presenilin-2 double knockout mice. Neurobiol Aging. 2008;29:1502–1511. doi: 10.1016/j.neurobiolaging.2007.03.028. [DOI] [PubMed] [Google Scholar]
- 99.Roberge MC, Messier C, Staines WA, Plamondon H. Food restriction induces long-lasting recovery of spatial memory deficits following global ischemia in delayed matching and non-matching-to-sample radial arm maze tasks. Neuroscience. 2008;156:11–29. doi: 10.1016/j.neuroscience.2008.05.062. [DOI] [PubMed] [Google Scholar]
- 100.Rich NJ, Van Landingham JW, Figueiroa S, Seth R, Corniola RS, Levenson CW. Chronic caloric restriction reduces tissue damage and improves spatial memory in a rat model of traumatic brain injury. J Neurosci Res. 2010;88:2933–2939. doi: 10.1002/jnr.22443. [DOI] [PubMed] [Google Scholar]
- 101.Mattson MP, Lovell MA, Furukawa K, Markesbery WR. Neurotrophic factors attenuate glutamate-induced accumulation of peroxides, elevation of intracellular Ca2+ concentration, and neurotoxicity and increase antioxidant enzyme activities in hippocampal neurons. J Neurochem. 1995;65:1740–1751. doi: 10.1046/j.1471-4159.1995.65041740.x. [DOI] [PubMed] [Google Scholar]
- 102.Almeida RD, Manadas BJ, Melo CV, Gomes JR, Mendes CS, Grãos MM, Carvalho RF, Carvalho AP, Duarte CB. Neuroprotection by BDNF against glutamate-induced apoptotic cell death is mediated by ERK and PI3-kinase pathways. Cell Death Differ. 2005;12:1329–1343. doi: 10.1038/sj.cdd.4401662. [DOI] [PubMed] [Google Scholar]
- 103.Yang JL, Tadokoro T, Keijzers G, Mattson MP, Bohr VA. Neurons efficiently repair glutamate-induced oxidative DNA damage by a process involving CREB-mediated up-regulation of apurinic endonuclease 1. J Biol Chem. 2010;285:28191–28199. doi: 10.1074/jbc.M109.082883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lu B, Figurov A. Role of neurotrophins in synapse development and plasticity. Rev Neurosci. 1997;8:1–12. doi: 10.1515/revneuro.1997.8.1.1. [DOI] [PubMed] [Google Scholar]
- 105.Kuczewski N, Porcher C, Lessmann V, Medina I, Gaiarsa JL. Activity-Dependent Dendritic Release of BDNF and Biological Consequences. Mol Neurobiol. 2009;39:37–49. doi: 10.1007/s12035-009-8050-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cowansage KK, LeDoux JE, Monfils MH. Brain-Derived Neurotrophic Factor: A Dynamic Gatekeeper of Neural Plasticity. Curr Mol Pharmacol. 2010;3:12–29. doi: 10.2174/1874467211003010012. [DOI] [PubMed] [Google Scholar]
- 107.Mattson MP, Wan RQ. Neurotrophic Factors in Autonomic Nervous System Plasticity and Dysfunction. Neuromol Med. 2008;10:157–168. doi: 10.1007/s12017-007-8021-y. [DOI] [PubMed] [Google Scholar]
- 108.Schmidt HD, Duman RS. The role of neurotrophic factors in adult hippocampal neurogenesis, antidepressant treatments and animal models of depressive-like behavior. Behav Pharmacol. 2007;18:391–418. doi: 10.1097/FBP.0b013e3282ee2aa8. [DOI] [PubMed] [Google Scholar]
- 109.Lu Y, Christian K, Lu B. BDNF: A key regulator for protein synthesis-dependent LTP and long-term memory? Neurobiol Learn Mem. 2008;89:312–323. doi: 10.1016/j.nlm.2007.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Minichiello L. TrkB signalling pathways in LTP and learning. Nat Rev Neurosci. 2009;10:850–860. doi: 10.1038/nrn2738. [DOI] [PubMed] [Google Scholar]
- 111.Castillo-Quan JI, Barrera-Buenfil DJ, Pérez-Osorio JM, Alvarez-Cervera FJ. Depression and diabetes: from epidemiology to neurobiology. Rev Neurol. 2010;51:347–59. [PubMed] [Google Scholar]
- 112.Nilsson LG, Nilsson E. Overweight and cognition. Scand J Psychol. 2009;50:660–667. doi: 10.1111/j.1467-9450.2009.00777.x. [DOI] [PubMed] [Google Scholar]
- 113.Sabia S, Kivimaki M, Shipley MJ, Marmot MG, Singh-Manoux A. Body mass index over the adult life course and cognation in late midlife: the Whitehall II Cohort Study. Am J Clin Nutr. 2009;89:601–607. doi: 10.3945/ajcn.2008.26482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kodl C, Seaquist ER. Cognitive dysfunction and diabetes mellitus. Endocr Rev. 2008;29:494–511. doi: 10.1210/er.2007-0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hölscher C. Diabetes as a risk factor for Alzheimer’s disease: insulin signalling impairment in the brain as an alternative model of Alzheimer’s disease. Biochem Soc Trans. 2011;39:891–897. doi: 10.1042/BST0390891. [DOI] [PubMed] [Google Scholar]
- 116.Kopf D, Frölich L. Risk of incident Alzheimer’s disease in diabetic patients: a systematic review of prospective trials. J Alzheimers Dis. 2009;16:677–685. doi: 10.3233/JAD-2009-1011. [DOI] [PubMed] [Google Scholar]
- 117.Muoio DM, Newgard CB. Obesity-related derangements in metabolic regulation. Ann Rev Biochem. 2006;75:367–401. doi: 10.1146/annurev.biochem.75.103004.142512. [DOI] [PubMed] [Google Scholar]
- 118.Qiu C, De Ronchi D, Fratiglioni L. The epidemiology of the dementias: an update. Curr Opin Psychiat. 2007;20:380–385. doi: 10.1097/YCO.0b013e32816ebc7b. [DOI] [PubMed] [Google Scholar]
- 119.Stranahan AM, Mattson MP. Impact of energy intake and expenditure on neuronal plasticity. Neuromol Med. 2008;10:209–218. doi: 10.1007/s12017-008-8043-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Greenwood CE, Winocur G. Cognitive impairment in rats fed high-fat diets: a specific effect of saturated fatty-acid intake. Behav Neurosci. 1996;110:451–459. doi: 10.1037//0735-7044.110.3.451. [DOI] [PubMed] [Google Scholar]
- 121.Granholm AC, Bimonte-Nelson HA, Moore AB, Nelson ME, Freeman LR, Sambamurti K. Effects of a saturated fat and high cholesterol diet on memory and hippocampal morphology in the middle-aged rat. J Alzheimers Dis. 2008;14:133–145. doi: 10.3233/jad-2008-14202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Stranahan AM, Norman ED, Lee K, Cutler RG, Telljohann RS, Egan JM, Mattson MP. Diet-induced insulin resistance impairs hippocampal synaptic plasticity and cognition in middle-aged rats. Hippocampus. 2008a;18:1085–1088. doi: 10.1002/hipo.20470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Shi L, Adams MM, Linville MC, Newton IG, Forbes ME, Long AB, Riddle DR, Brunso-Bechtold JK. Caloric restriction eliminates the aging-related decline in NMDA and AMPA receptor subunits in the rat hippocampus and induces homeostasis. Exp Neurol. 2007;206:70–79. doi: 10.1016/j.expneurol.2007.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gomez-Pinilla F, Vaynman S, Ying Z. Brain-derived neurotrophic factor functions as a metabotrophin to mediate the effects of exercise on cognition. Eur J Neurosci. 2008;28:2278–87. doi: 10.1111/j.1460-9568.2008.06524.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Yao J, Chen S, Mao Z, Cadenas E, Brinton RD. 2-Deoxy-D-glucose treatment induces ketogenesis, sustains mitochondrial function, and reduces pathology in female mouse model of Alzheimer’s disease. PLoS One. 2011;2011;6(7):e21788. doi: 10.1371/journal.pone.0021788. Epub 2011 Jul 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Maalouf M, Rho JM, Mattson MP. The neuroprotective properties of calorie restriction, the ketogenic diet, and ketone bodies. Brain Res Rev. 2009;59:293–315. doi: 10.1016/j.brainresrev.2008.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Robinet C, Pellerin L. Brain-derived neurotrophic factor enhances the expression of the monocarboxylate transporter 2 through translational activation in mouse cultured cortical neurons. J Cereb Blood Flow Metab. 2010;30:286–298. doi: 10.1038/jcbfm.2009.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Burkhalter J, Fiumelli H, Allaman I, Chatton JY, Martin JL. Brain-derived neurotrophic factor stimulates energy metabolism in developing cortical neurons. J Neurosci. 2003;23:8212–8220. doi: 10.1523/JNEUROSCI.23-23-08212.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zhang J, Wang Y, Chi Z, Keuss MJ, Pai YM, Kang HC, Shin JH, Bugayenko A, Wang H, Xiong Y, Pletnikov MV, Mattson MP, Dawson TM, Dawson VL. The AAA+ ATPase Thorase regulates AMPA receptor-dependent synaptic plasticity and behavior. Cell. 2011;145:284–299. doi: 10.1016/j.cell.2011.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Abraham WC, Williams JM. LTP maintenance and its protein synthesis-dependence. Neurobiol Learn Mem. 2008;89:260–268. doi: 10.1016/j.nlm.2007.10.001. [DOI] [PubMed] [Google Scholar]
- 131.Wang C, Bomberg E, Billington C, Levine A, Kotz CM. Brain-derived neurotrophic factor in the hypothalamic paraventricular nucleus increases energy expenditure by elevating metabolic rate. Am J Physiol Regul Integr Comp Physiol. 2007;293:R992–1002. doi: 10.1152/ajpregu.00516.2006. [DOI] [PubMed] [Google Scholar]
- 132.Cao L, Choi EY, Liu X, Martin A, Wang C, Xu X, During MJ. White to brown fat phenotypic switch induced by genetic and environmental activation of a hypothalamic-adipocyte axis. Cell Metab. 2011;14:324–338. doi: 10.1016/j.cmet.2011.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Griffioen KJ, Wan R, Brown TR, Okun E, Mughal MR, Camandola S, Phillips TM, Mattson MP. Aberrant heart rate and brainstem BDNF signaling in a mouse model of Huntington’s disease. Neurobiol Aging. 2011 doi: 10.1016/j.neurobiolaging.2011.11.030. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]