Protein tyrosine phosphatase-1B (PTP1B) is an abundant, widely expressed non-receptor tyrosine phosphatase, which is thought to be a key negative regulator of insulin signaling (Tonks, 2003). Increased and prolonged tyrosine phosphorylation of the insulin receptor (IR) was observed in mice lacking PTP1B (Elchebly et al., 1999). Global deletion of PTP1B in mice results in increased systemic insulin sensitivity, enhanced glucose uptake into skeletal muscle, and improved glucose tolerance (Elchebly et al., 1999). The increased insulin sensitivity is due to the absence of PTP1B, resulting from failure to dephosphorylate the IR (Elchebly et al., 1999). Liver-specific deletion of PTP1B has been shown to improve metabolic syndrome and attenuates diet-induced endoplasmic reticulum stress (Delibegovic et al., 2009). Neuronal PTP1B also regulates body weight, adiposity and leptin action (Bence et al., 2006). Furthermore, neuronal PTP1B deficiency results in inhibition of hypothalamic 5′ AMP-activated protein kinase (AMPK) and isoform-specific activation of AMPK in peripheral tissues (Xue et al., 2009). We observed that the light-dependent activation of IR in photoreceptors is due to inhibition of PTP1B signaled through photobleaching of rhodopsin (Rajala et al., 2010; Rajala et al., 2007). Our earlier studies also suggest that PTP1B activity is elevated in mice carrying rhodopsin mutation (a mouse model representing retinitis pigmentosa) and a mouse model defect in the photobleaching of rhodopsin (Rajala et al., 2010). On the other hand activation of insulin signaling has been shown to delay the death of cone photoreceptor neurons in mouse models of retinitis pigmentosa (Punzo et al., 2009). Studies from our laboratory over the past decade highlight the neuroprotective role of IR signaling in both rod and cone photoreceptor neurons (Rajala et al., 2013; Rajala et al., 2007). Thus, reducing or blocking the activation of PTP1B could be beneficial to protect the dying retinal cells retinal degenerative diseases. We have previously reported that global PTP1B knockout mice exhibited a significantly lower sensitivity to stress-induced cell death than PTP1B competent mice (Rajala et al., 2010); however, the cell type specific role of PTP1B is not known.
Immunolocalization studies suggest that PTP1B is localized to retinal pigment epithelium, rod inner segments, outer plexiform layer, inner plexiform layer, and ganglion cell layer (Fig. 1 A and 1D) and in rod inner segment it co-localizes with arrestin in dark-adapted retina (Fig. 1C). A weak immunoreactivity of PTP1B is also present in rod outer segments (Fig. 1A and 1B). The adaptability of animals to dark and light conditions is examined with arrestin immunolocalization (Fig. 1B and IE). In dark-adapted retinas, arrestin is localized to the rod inner segments and the outer plexiform layer (Fig. 1B), and upon light illumination arrestin is translocated to photoreceptor outer segments (Fig. 1E). Our immunohistochemical data suggest that PTP1B predominantly localized to rod inner segments irrespective of dark or light adaptation. Since PTP1B is shown to be expressed in various retinal cells including rod photoreceptors (Fig. 1) and it is difficult to understand the contribution of PTP1B from rod cells on photoreceptor cell survival. To determine the functional role of PTP1B in rod photoreceptor cells, we generated a rod cell specific PTP1B knockout mouse line by breeding floxed PTP1B mice to mice expressing Cre-recombinase under the control rod opsin promoter by using the conditional Cre/lox technology.
Figure 1. Localization of PTP1B in mouse retina.
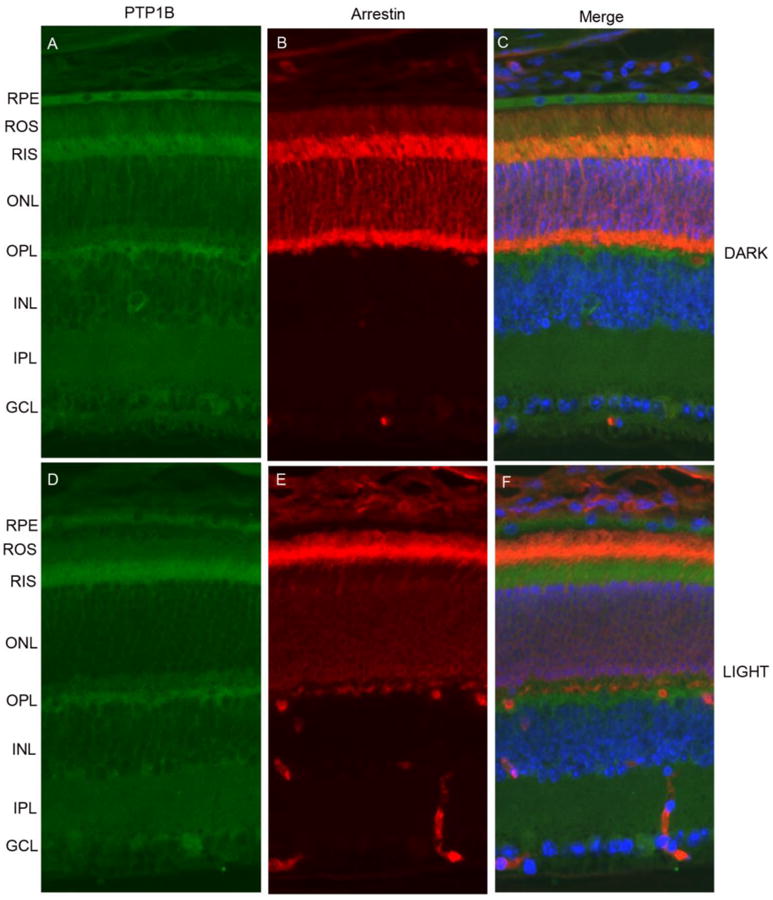
Prefer-fixed sections of dark- (A-C) and light-adapted (D-F) mouse retinas were stained for PTP1B (A, D), arrestin (B, E) and DAPI (C, F) and the immunofluorescence was analyzed by epifluorescence. Panel C and F represent the merge images of PTP1B and arrestin. RPE, retinal pigment epithelium, ROS, rod outer segments; RIS, rod inner segments; ONL, outer nuclear layer; OPL, outer plexiform layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer.
Light microscopic examination of the retinas from wild-type and PTP1B knockout mice at 6-8 weeks of age showed no difference in retinal structure between the two groups when each group was maintained in dim cyclic light (Fig. 2A and 2B). The retinas appeared normal and rod outer segments (ROS) appeared to be well organized (Fig. 2A and 2B). Quantitative analysis of the superior and inferior regions of the outer nuclear layer (ONL) (Rajala et al., 2008) showed no significant differences among the two groups in the average ONL thickness measured at 0.25-mm intervals from the ONL to the inferior and superior ora serrata (Fig. 2E), indicating that rod photoreceptor viability was not different among these mice. Thus, mice lacking PTP1B did not exhibit any structural phenotype when maintained in dim cyclic light.
Figure 2. Morphological analysis of PTP1B knockout mice in dim cyclic light and after light stress.
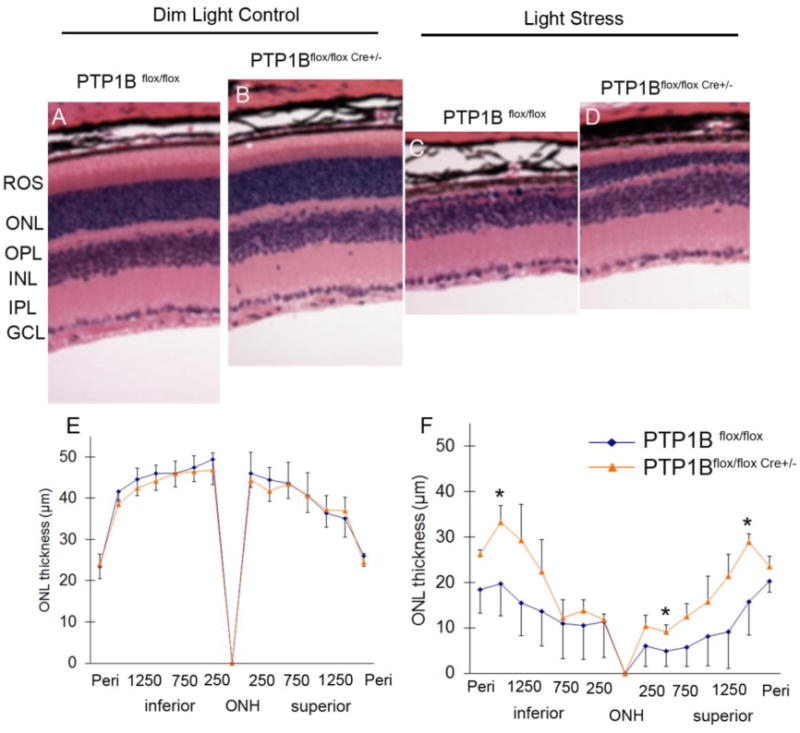
Hematoxylin and eosin-stained (H & E) retinal sections of the retina from the eyes of wild-type (PTP1Bflox/flox) and PTP1B knockout (PTP1Bflox/flox Cre +/-) mice at 6-8 weeks of age under dim cyclic light (A, B) or light-stressed at 14,000 lux for 7 days (C, D). Quantification of morphologic changes in PTP1B knockout mice in dim cyclic light or exposed to light stress. Plots of ONL thickness at 0.25-mm intervals from the optic nerve head (ONH) along with vertical meridian in the superior and interior regions of the retina of dim cyclic light (E) or light stressed (F) wild type and PTP1B knockout mice. Values are mean ± SD, n=3, * p<0.05. ROS, rod outer segments; ONL, outer nuclear layer; INL, inner nuclear layer; IPL, inner plexiform layer; GCL, ganglion cell layer, ONH, optic nerve head.
To understand the neuroprotective potential of the retina, light stress is one of the well accepted readout techniques. The light stress models have received considerable attention in the retina research due to their application in screening drugs and also to study the effect of specific retinal gene mutations or gene ablations (Noell et al., 1966). We exposed wild-type, and PTP1B knockout mice to light stress for 7 days at 14,000 lux. After a seven-day recovery period, we measured the extent of photoreceptor cell loss. The ONL thickness in mice from each group reduced after light exposure, indicating that the number of rod photoreceptors reduced and the greatest reduction was apparent in the retinas of the wild-type mice (Fig. 2C and 2F). When exposed to 14,000 lux, wild-type mice had significantly fewer rod photoreceptors in the superior and inferior regions than the PTP1B knockout mice (Fig. 2D and 2F). These results suggest that rod specific conditional deletion of PTP1B knockout mice were protected from light stress while the wild type mice were not.
Mammalian retinal neurons have a remarkable ability to survive in a hostile environment, being subjected constantly to high levels of oxygen and bombardment by photons. We have proposed earlier that IR signaling is an endogenous neuroprotective pathway that acts as molecular sunglasses against stress-induced retinal degenerations. We have previously reported that loss of IR expression in rod photoreceptors significantly impaired the retinal function and loss of photoreceptors in mice exposed to bright light stress (Rajala et al., 2008), while intravenous injection of an allosteric inhibitor of PTP1B protects rats against light stress-induced retinal degeneration through the protection of IR phosphorylation (Rajala et al., 2010). We also found elevated levels PTP1B activity in mouse models of retinitis pigmentosa, Leber Congenital Amaurosis and streptozotocin-induced diabetic mouse retina (Rajala et al., 2010; Rajala et al., 2009). Our results demonstrate that deletion of PTP1B attenuates light stress-induced photoreceptor degeneration. PTP1B inhibitors and PTP1B antisense oligonucleotides (ASO) have already been recognized as potential therapeutics in the treatment of type-2 diabetes and obesity (Goldstein, 2001; Zinker et al., 2002). However, neither the PTP1B inhibitors nor ASOs has ever been examined in the context of retinal degenerative diseases. Our studies suggest that PTP1B antagonists could be potential therapeutic agents to treat retinal degenerations.
Acknowledgments
This work was supported by grants from the NIH (EY016507; EY00871; EY021725) and by an unrestricted departmental grant from Research to Prevent Blindness, Inc. We thank Dr. Benjamin G. Neel (Ontario Cancer Center, Canada) for pan-PTP1B floxed mice and Dr. Yun Le (OUHSC, Oklahoma City, OK) for providing rod opsin-cre mice. The authors thank Dr. Masaki Tanito for his help in these studies. The technical assistance of Ms. Yu Lee is highly acknowledged.
Footnotes
Compliance with Ethics Guidelines: Conflict of Interest: Raju V.S. Rajala, and Ammaji Rajala declare that they have no conflict of interest
For studies with animals: All institutional and national guidelines for the care and use of laboratory animals were followed.
References
- Bence KK, Delibegovic M, Xue B, Gorgun CZ, Hotamisligil GS, et al. Nat Med. 2006;12:917–924. doi: 10.1038/nm1435. [DOI] [PubMed] [Google Scholar]
- Delibegovic M, Zimmer D, Kauffman C, Rak K, Hong EG, et al. Diabetes. 2009;58:590–599. doi: 10.2337/db08-0913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elchebly M, Payette P, Michaliszyn E, Cromlish W, Collins S, et al. Science. 1999;283:1544–1548. doi: 10.1126/science.283.5407.1544. [DOI] [PubMed] [Google Scholar]
- Goldstein BJ. Curr Drug Targets Immune Endocr Metabol Disord. 2001;1:265–275. doi: 10.2174/1568008013341163. [DOI] [PubMed] [Google Scholar]
- Noell WK, Walker VS, Kang BS, Berman S. Invest Ophthalmol. 1966;5:450–473. [PubMed] [Google Scholar]
- Punzo C, Kornacker K, Cepko CL. Nat Neurosci. 2009;12:44–52. doi: 10.1038/nn.2234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajala A, Anderson RE, Ma JX, Lem J, Al Ubaidi MR, et al. J Biol Chem. 2007;282:9865–9873. doi: 10.1074/jbc.M608845200. [DOI] [PubMed] [Google Scholar]
- Rajala A, Dighe R, Agbaga MP, Anderson RE, Rajala RV. J Biol Chem. 2013;288:19503–19515. doi: 10.1074/jbc.M113.469064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajala A, Tanito M, Le YZ, Kahn CR, Rajala RV. J Biol Chem. 2008;283:19781–19792. doi: 10.1074/jbc.M802374200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajala RV, Tanito M, Neel BG, Rajala A. J Biol Chem. 2010;285:8894–8904. doi: 10.1074/jbc.M109.070854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajala RV, Wiskur B, Tanito M, Callegan M, Rajala A. Invest Ophthalmol Vis Sci. 2009;50:1033–1040. doi: 10.1167/iovs.08-2851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tonks NK. FEBS Lett. 2003;546:140–148. doi: 10.1016/s0014-5793(03)00603-3. [DOI] [PubMed] [Google Scholar]
- Xue B, Pulinilkunnil T, Murano I, Bence KK, He H, et al. Mol Cell Biol. 2009;29:4563–4573. doi: 10.1128/MCB.01914-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinker BA, Rondinone CM, Trevillyan JM, Gum RJ, Clampit JE, et al. Proc Natl Acad Sci U S A. 2002;99:11357–11362. doi: 10.1073/pnas.142298199. [DOI] [PMC free article] [PubMed] [Google Scholar]


