Abstract
Vancoymycin causes red man syndrome, an itchy erythematous eruption involving the face, neck and upper torso. Atopic dermatitis also manifests itch and erythema, and staphylococcus δ-toxin contributes to this process. The antibiotic and toxin each provoke mast cell degranulation but the mechanism had not been understood. We have determined that these compounds evoke degranulation via interaction with the same receptor, MRGPRX2, on mast cells. A receptor antagonist inhibits this process. Antagonists of this receptor may have therapeutic potential.
MRGPRX2 and staphylococcus δ-toxin
Mast cells have long been implicated in the pathogenesis of atopic dermatitis, an intensely pruritic inflammatory skin disease(1, 2). As opposed to healthy individuals, more than 90% of atopic dermatitis patients are colonized with Staphylococcus aureus (S. aureus) in the affected skin areas(3). Culture supernatants of S. aureus demonstrate potent mast cell degranulation activity and δ-toxin has been identified as the main degranulation inducing factor(4). Skin colonization with S. aureus, but not a mutant deficient in δ-toxin, promotes IL-4 production and allergic skin disease(4). These effects are IgE-independent, are diminished in KitW-sh/W-sh mast cell-deficient mice, and are restored by mast cell reconstitution suggesting a pivotal role for IgE-independent mast cell degranulation in δ-toxin-induced allergic skin disease(4). Despite these findings, the mechanism by which δ-toxin induces mast cell degranulation remained unknown(4). Given the role of MRGPRX2 in itch(5, 6) and IgE-independent mast cell degranulation(7), we evaluated the interaction of δ-toxin with MRGPRX2 using ratiometric Fura-2 imaging in a HEK293 cell line stably expressing MRGPRX2 (Figure 1A). This activation is inhibited by QWF, a MRGPRX2 antagonist(5) (Figure 1A). QWF also inhibits δ-toxin-induced degranulation in the human LAD2 mast cell line (Figure 1B).
Figure 1. Staphylococcus δ-toxin and vancomycin activate human MRGPRX2 to induce mast cell degranulation.
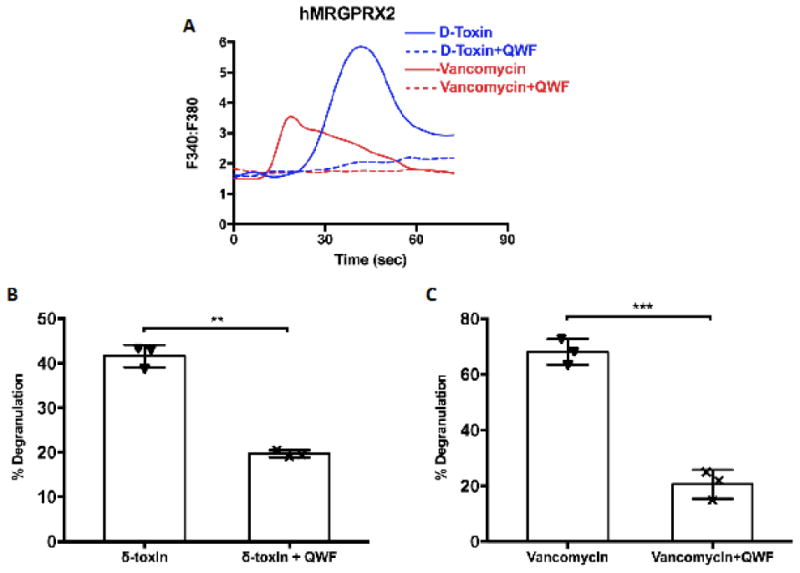
A. The interaction of staphylococcus δ-toxin and vancomycin with a HEK-293 cell line stably expressing MRGPRX2. Intracellular calcium [Ca2+]i was determined by ratiometric Fura-2 imaging as an indicator of receptor activation after addition of staphylococcus δ-toxin (10 μM) and vancomycin (100 μM). The MRGPRX2 antagonist QWF was used at equal concentrations of the ligands. The traces represent responses in individual cells. B and C. Staphylococcus δ-toxin (10 μM) and vancomycin (100 μM) induce mast cell degranulation in the human LAD2 mast cell line. The level of mast cell degranulation was assessed by the release of β-hexosaminidase in mast cell granules, quantified by the level of its substrate p-nitrophenyl N-acetyl-β-D-glucosamide (PNAG) digested in a colorimetric assay(5, 14). QWF significantly decreases the mast cell degranulation induced by staphylococcus δ-toxin (p=0.0022) and vancomycin (p=0.0003).
MRGPRX2 and red man syndrome
Human MRGPRX2 and a mouse orthologue, MrgprB2, mediate basic secretagogue-inudced mast cell degranulation(7). Pseudo-allergic reactions associated with medications such as fluoroquinolone antibiotics and curare derived neuromuscular agents are diminished in MrgprB2-/- mice(7). Vancomycin is a glycopeptide antibiotic frequently used to treat nosocomial infections and the drug of choice to treat methicillin-resistant staphylococci(8). Two types of hypersensitivity reactions are associated with vancomycin: One type is IgE- mediated anaphylaxis. The second type is red man syndrome; an IgE- independent mast cell degranulation associated with a pruritic erythematous rash involving the face, neck, and upper torso(8). Clinical features similar to red man syndrome are observed frequently with ciprofloxacin and atracurium but surprisingly not with teicoplanin, a glycopeptide antibiotic similar to vancomycin(9). Given the wide clinical implications of glycopeptides and the burden of red man syndrome, we investigated the interaction of vancomycin and teicoplanin with MRGPRX2. Teicoplanin does not activate MRGPRX2 and does not induce mast cell degranulation (Figure 2). In contrast, we demonstrate that vancomycin activates human MRGPRX2 and that this activation is inhibited by QWF (Figure 1A). QWF significantly decreases vancomycin-induced degranulation in the human LAD2 mast cell line (Figure 1C).
Figure 2. Interaction of teicoplanin with human MRGPRX2 and mast cells.
A. Teicoplanin does not activate human MRGPRX2 stably expressed in HEK-293 cells. Intracellular calcium [Ca2+]i was determined by ratiometric Fura-2 imaging as an indicator of receptor activation after addition of teicoplanin (100 μM). Each trace represents a unique cell. B. Teicoplanin does not induce degranulation in the human LAD2 mast cell line (P>0.05, ns; non-significant).
Conclusion
Activation of MRGPRX2 on mast cells induces the release of several pro-inflammatory mediators including histamine, interleukins, tumor necrosis factor and prostaglandin D2(7, 10). These findings are consistent with data that has previously implicated MRGPRX2 in itch. The data presented here link MRGPRX2 to two specific itchy conditions, red man syndrome associated with vancomycin, and atopic dermatitis, at least within the context of staphylococcus δ-toxin.
Red man syndrome is an acute IgE-independent reaction mediated by mast cells, the symptoms of which are controlled partially by antihistamines. This observation is consistent with the release of histamine from mast cells contributing to red man syndrome although antihistamines do not prevent its occurrence(8). Atopic dermatitis is a chronic inflammatory skin disease with a complex pathogenesis involving the microbiome, barrier defects, and different inflammatory cells including mast cells. Antihistamines have limited efficacy in the treatment or control of the symptoms of atopic dermatitis(11). This observation is consistent with the contribution of additional mast cell-derived mediators, for example IL-4, to the pathogenesis of atopic dermatitis.
MRGPRX2 has been detected on mast cells, sensory neurons and, most recently, keratinocytes, where it leads to the release of IL-6(12, 13). Substance P, a neuropeptide associated with atopic dermatitis, activates MRGPRX2 and induces scratching in mice via Mrgprs(5). The data reported here extend the contributions of this receptor to inflammatory conditions associated with itch and erythema with the demonstration that vancomycin and staphylococcus δ-toxin activate mast cells via MRGPRX2. Antagonists of MRGPRX2 have the potential to relieve the symptoms of or to treat red man syndrome and atopic dermatitis.
References
- 1.Kawakami T, Ando T, Kimura M, Wilson BS, Kawakami Y. Mast cells in atopic dermatitis. Curr Opin Immunol. 2009;21(6):666–78. doi: 10.1016/j.coi.2009.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu FT, Goodarzi H, Chen HY. IgE, mast cells, and eosinophils in atopic dermatitis. Clin Rev Allergy Immunol. 2011;41(3):298–310. doi: 10.1007/s12016-011-8252-4. [DOI] [PubMed] [Google Scholar]
- 3.Rudikoff D, Lebwohl M. Atopic dermatitis. Lancet. 1998;351(9117):1715–21. doi: 10.1016/S0140-6736(97)12082-7. [DOI] [PubMed] [Google Scholar]
- 4.Nakamura Y, Oscherwitz J, Cease KB, Chan SM, Munoz-Planillo R, Hasegawa M, et al. Staphylococcus delta-toxin induces allergic skin disease by activating mast cells. Nature. 2013;503(7476):397–401. doi: 10.1038/nature12655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Azimi E, Reddy VB, Shade KC, Anthony RM, Talbot S, Pereira PJ, et al. Dual action of neurokinin-1 antagonists on Mas-related GPCRs. JCI Insight. 2016;1(16):e89362. doi: 10.1172/jci.insight.89362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reddy VB, Sun S, Azimi E, Elmariah SB, Dong X, Lerner EA. Redefining the concept of protease-activated receptors: cathepsin S evokes itch via activation of Mrgprs. Nat Commun. 2015;6:7864. doi: 10.1038/ncomms8864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McNeil BD, Pundir P, Meeker S, Han L, Undem BJ, Kulka M, et al. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature. 2014 doi: 10.1038/nature14022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sivagnanam S, Deleu D. Red man syndrome. Crit Care. 2003;7(2):119–20. doi: 10.1186/cc1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wilson AP. Comparative safety of teicoplanin and vancomycin. Int J Antimicrob Agents. 1998;10(2):143–52. doi: 10.1016/s0924-8579(98)00025-9. [DOI] [PubMed] [Google Scholar]
- 10.Gaudenzio N, Sibilano R, Marichal T, Starkl P, Reber LL, Cenac N, et al. Different activation signals induce distinct mast cell degranulation strategies. J Clin Invest. 2016;126(10):3981–98. doi: 10.1172/JCI85538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Buddenkotte J, Maurer M, Steinhoff M. Histamine and antihistamines in atopic dermatitis. Adv Exp Med Biol. 2010;709:73–80. doi: 10.1007/978-1-4419-8056-4_8. [DOI] [PubMed] [Google Scholar]
- 12.Kiatsurayanon C, Niyonsaba F, Chieosilapatham P, Okumura K, Ikeda S, Ogawa H. Angiogenic peptide (AG)-30/5C activates human keratinocytes to produce cytokines/chemokines and to migrate and proliferate via MrgX receptors. J Dermatol Sci. 2016;83(3):190–9. doi: 10.1016/j.jdermsci.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 13.Bader M, Alenina N, Andrade-Navarro MA, Santos RA. MAS and its related G protein-coupled receptors, Mrgprs. Pharmacological reviews. 2014;66(4):1080–105. doi: 10.1124/pr.113.008136. [DOI] [PubMed] [Google Scholar]
- 14.Kuehn HS, Radinger M, Gilfillan AM. Measuring mast cell mediator release. Curr Protoc Immunol. 2010 doi: 10.1002/0471142735.im0738s91. Chapter 7:Unit7 38. [DOI] [PMC free article] [PubMed] [Google Scholar]



