Abstract
There is currently a strong interest among both audiologists and hearing researchers to find a physiological measure that can be used as a marker of how amplified sounds are processed by the brain (i.e., hearing aid fitting) or how the brain changes with exposure to amplified sounds (i.e., hearing aid acclimatization). Currently, auditory evoked potentials are used, or proposed to be used, for both of these purposes to some degree. It is clear from the literature that some of these uses are potentially useful clinically while others are quite problematic. The current state of aided cortical auditory evoked potentials will be discussed relative to their application to hearing aid fitting/verification and in understanding hearing aid acclimatization. Future areas of promise as well as current gaps in the literature will also be addressed.
Keywords: Auditory evoked potentials (AEPs), Event-related potentials (ERPs), hearing aids, acclimatization
1. Introduction
A primary goal of fitting hearing aids on an individual is to increase the audibility of various speech cues and to improve speech understanding. One also hopes that as a result of better speech understanding, the quality of life will improve. There are a host of potential variables that can affect the outcome of a hearing aid fitting. Factors specific to the outcome of speech understanding might include variables specific to the listening environment (e.g., signal type, presence of background noise, etc.), the hearing aid (e.g., algorithms, settings, etc.), and the patient (e.g., integrity of the auditory system, age, motivation, lifestyle, etc.). Souza & Tremblay (2006) introduced a schematic to help illustrate some of the variables that may contribute to speech understanding in hearing aid users. Figure 1 shows a modified version of that schematic which includes four stages: (1) the original acoustic signal, (2) the modifications made by the hearing aid, (3) the processing of the peripheral and central auditory systems, and finally (4) the higher order processes, including the behavioral response.
Figure 1.

A schematic representing four stages of the listening experience for hearing aid users. Stage 1 includes the incoming signal. Stage 2 addresses the hearing aid and how it modifies the signal. Stage 3 begins when the amplified signal is delivered to the ear and includes transduction and processing by the auditory system. Stage 4 includes higher order processes up to and including a behavioral response of some kind.
Stage 1 includes the incoming signal and the acoustic properties associated with it. This could include the context in which the signal is presented.
Stage 2 addresses the hearing aid and how it modifies the acoustic signal including the hearing aid settings such as compression, gain, and frequency response.
Stage 3 begins when the signal is delivered to the ear. Acoustic waveforms are transduced and processed through several parallel processing streams by the peripheral and central auditory systems.
Stage 4 includes the higher order processes such as those that modulate attention and memory, and all processes leading up to, and including, a behavioral response of some kind.
It should be noted that these divisions are somewhat arbitrary and each stage is not independent of the others; in reality, there is considerable interaction and overlap between stages. The purpose of the schematic is to help place the content of this review paper into the context of the listening experience. The focus will be primarily on Stage 2 and Stage 3 and the interaction between these two aspects of the hearing aid experience. The purpose of this paper is to review the literature that addresses how measures of the brain can contribute to successful rehabilitation of hearing impairment and inform our general understanding of the neural contributions to the process. After providing a brief introduction to evoked potentials, the paper reviews the evidence from two main perspectives that are present in the literature: (a) the process of hearing aid fitting and verification, and (b) the characterization of hearing aid acclimatization effects.
2. A Measure of Neural Encoding
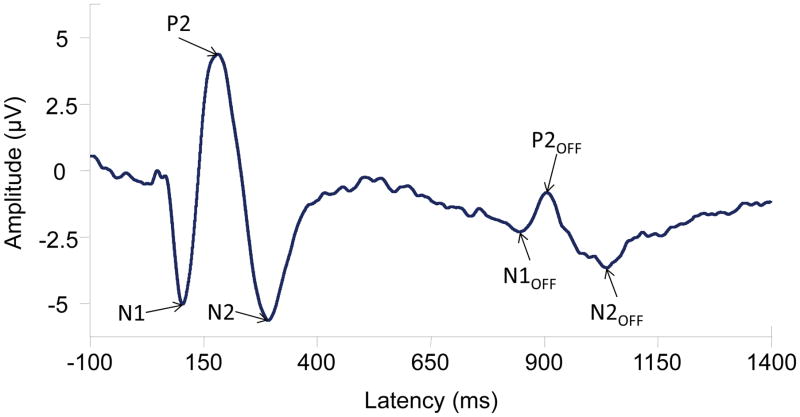
In the hearing aid literature, the most common method of measuring neural processing has been electroencephalography (EEG), or the measurement of electrical activity at the level of the scalp. Auditory evoked potentials (AEPs) are a type of EEG that reflect neural activity in response to auditory stimuli, thus allowing the characterization of how the brain encodes sounds. AEPs consist of negative and positive peaks in voltage ranging in time from a few milliseconds to several hundred milliseconds post stimulus onset. These peaks include the auditory brainstem response (ABR) occurring in the first 10 ms after stimulus onset, middle latency responses occurring between 10 and 50 ms, and late latency responses that occur beyond 50 ms. In addition to these transient AEPs, sustained AEPs can also be considered, such as the auditory steady state response (ASSR) or the related frequency follow response (FFR; for a review see Picton, 2011). AEPs are one tool that can be used to understand how the auditory system is encoding cues important for speech understanding. As a relatively inexpensive tool, they are a clinically relevant way to understand how the brain is encoding amplified sounds and can also help us to determine how the auditory system changes with exposure to hearing-aid-processed sounds. Although some mention is made of ABR and ASSR literature, this paper focuses primarily on cortical AEPs given that most of the current research activity uses these potentials. Figure 2 illustrates a cortical AEP waveform and the notable peaks, including N1, P2, and N2. Not only do stimulus onsets evoke these peaks, but acoustic changes within a stimulus and stimulus offsets can also result in similar responses shown as N1OFF, P2OFF, and N2OFF (Picton, 2011; Martin et al., 2008).
Figure 2.
An example of the CAEP including N1, P2, and N2 waves to a 756-ms, 1000-Hz pure tone. Also displayed are subsequent N1OFF, P2OFF, and N2OFF waves that are elicited by an acoustic change in the stimulus; in this case the change was the offset of the signal.
Variations of the term “aided AEPs” have been used in the literature in different ways. For the purposes of this manuscript, we define the term as AEP responses elicited from an individual using stimuli that have been processed by a hearing aid. There are at least four methods for recording aided AEPs. The most common method found in the literature involves recording evoked potentials while an individual is wearing the hearing aid and stimuli are presented in the sound field. This sound-field approach is most intuitive and representative of how clinical aided AEPs would be measured; however, when testing monaurally in the sound field it is important to minimize contribution of the non-test ear, often done with an ear plug of some kind. Another method is to record the hearing aid output offline either in a coupler or a mannequin such as the Knowles Electronics Manikin for Acoustic Research (KEMAR) or the Brüel and Kjær Head and Torso Simulator (e.g., Billings et al., 2012; Easwar, Glista, et al., 2012). The output recording is than presented through insert earphones to the participant. In the third method, the hearing is is worn by the individual, and stimuli are presented through the hearing aid using direct audio input (e.g., Glista et al. 2012). Finally, in the fourth method used in the hearing literature, hearing aid processing is simulated with a master hearing aid approach, in which hearing aid processing is applied digitally to a signal using Matlab (Mathworks, Natick, MA) or other signal processing software. The modified stimuli are then presented through insert earphones. While less real-world, the last three methods have the advantage of minimizing extraneous variables related to sound field testing such as the effects of head movement and standing waves. In addition, variability in how the hearing aid processes sound from presentation to presentation is minimized. In addition, when testing monaurally the contribution of the non-test ear is controlled through earphone presentation. A distinct approach to recording aided AEPs is to make measurements without the hearing aid and apply results to our understanding of the aided condition. This has often been the approach in studying hearing aid acclimatization as will be discussed below.
3. Hearing Aid Fitting: Estimating Unaided and Aided Thresholds
Hearing aid fitting and verification has been the primary focus of the aided AEP literature to date. The usefulness of a physiological measure to assist the clinician in the fitting process would be especially beneficial for hard-to-test populations. Unaided use of AEPs continues today as a means of threshold estimation. Historically, cortical AEPs have been used successfully to estimate behavioral thresholds (approximately within 10 dB of behavioral threshold) of both normal-hearing and hearing-impaired populations (e.g., Rapin et al., 1970; Prasher et al., 1993; Yeung et al., 2007; Ikeda et al., 2010); however, in some cases unaided cortical AEP thresholds can exceed behavioral thresholds by more than 20 dB (e.g., Glista et al., 2012; Van Maanen et al., 2005). This wide range of variability in threshold estimation has the potential to cause obvious challenges in the hearing aid fitting process. Other AEPs, such as the ABR and ASSR have also been used for unaided threshold estimation especially in infants as they can be recorded effectively while the patient is sleeping (for a review see Korczak et al., 2012; and Hood, 1998).
Cortical AEPs have also been used to estimate aided thresholds to assist with the hearing aid fitting process with hard-to-test populations such as infants. Similar to the rationale of an aided behavioral threshold, the purpose of aided cortical AEPs is to verify that the amplified signal is being processed by the brain. Rapin & Graziani (1967) were the first to publish aided cortical AEP data, presenting eight case studies of infants/children. Five of the eight individuals demonstrated a 20 dB improvement in aided cortical AEP response threshold compared to unaided AEP threshold, two cases showed no improvement in cortical AEP threshold, and one had insufficient data to determine benefit. This initial report was followed by several papers presenting a number of case studies that suggested the potential for aided cortical AEPs to assist in the hearing aid fitting process (Gravel et al., 1989; Kurtzberg, 1989; Kraus & McGee, 1994; Gatehouse & Robinson, 1996).
In parallel with aided cortical AEP investigations, researchers explored aided ABRs as a possible measure to assist with the hearing aid fitting (Beauchaine, Gorga, Reiland, et al., 1986; Brown, Klein, & Snydee, 1999; Gerling, 1991; Gorga, Beauchaine, & Reiland, 1987; Hecox, 1983; Kileny, 1982). However, brief transient stimuli (clicks and tone pips) are used to elicit the ABR, and these stimuli are not ideal for measuring hearing aid function. Researchers concluded that such brief-duration stimuli were problematic because they did not effectively and consistently activate hearing aid circuitry (Brown, Klein, & Snydee, 1999; Gorga, Beauchaine, & Reiland, 1987).
The recent development of HearLab by Frye Electronics (Tigard, USA) and National Acoustic Laboratories (Sydney, Australia) has increased interest in aided cortical AEPs and encouraged their use for hearing aid fitting in infants (e.g., Purdy et al., 2005; Frye, 2010; NAL, 2010; Golding, 2007; Dillon, 2005). Although approved for use clinically in some countries, it has not yet received clinical approval in Canada or the United States. While the potential need for such a physiological measure is apparent, it is only recently that systematic research questioning the use of aided AEPs has emerged. A determination of whether aided AEPs are of value clinically remains unresolved because of varied results across these studies. The main obstacle to clinical implementation is that under certain conditions, no effect of amplification is demonstrated (Tremblay, Billings, et al., 2006; Billings et al., 2007; Billings et al., 2011; Marynewich et al., 2012; Jenstad et al., 2012). That is, there is no statistical difference between CAEP waveforms elicited in an unaided condition and those elicited in an aided condition, despite the additional gain provided by the hearing aid and perceptual measures verifying increased gain. These data are quite problematic from a clinical perspective.
Inconsistent research findings present an important clinical challenge; if aided AEPs can only be used for some subset of subjects or only a specific group of hearing aids, then the clinical utility is greatly diminished. The wide range of variability is a problem that must be addressed before aided AEPs can completely transition into the clinic. It may be that some of the variability can be explained by the stimulus level used relative to the threshold of the individual or group being tested. Reexamination of one of the first larger scale parametric studies of aided AEPs provides some insight into why an amplification effect may not always be present (Korczak et al., 2005). Korczak and colleagues (2005) varied signal level in two different groups of hearing impaired participants. They demonstrated that the effect of amplification measured using aided CAEPs is level dependent. That is, amplification effects (i.e., differences between unaided and aided conditions) were more likely to occur near threshold than at suprathreshold levels. This illustrates an important aspect of the early aided AEP literature: isolating an effect of amplification by comparing a barely audible or inaudible unaided evoked response with a suprathreshold aided evoked response often resulted in significant changes to waveform morphology.
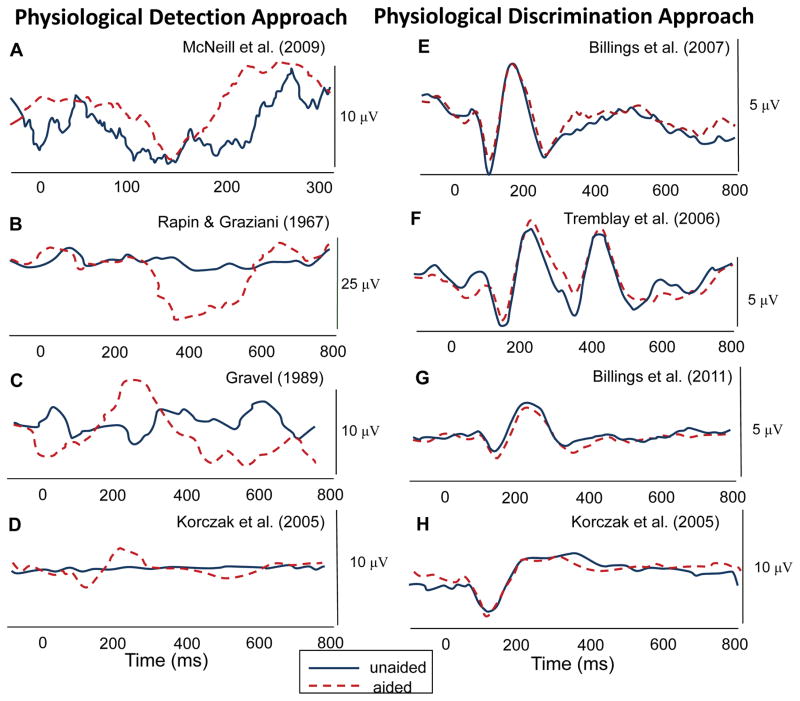
Billings and colleagues (2012) revisited the literature with Korczak’s (2005) results in mind and proposed a perspective of thinking about these results that may help understand the current clinical utility of aided AEPs until more can be understood about their limitations. Many of the early studies demonstrated significant changes in waveform morphology in the aided condition relative to the unaided condition, but usually the unaided stimulus was barely audible or not audible (e.g., Gravel et al., 1989; McNeill et al., 2009; Rapin et al., 1967). In contrast, several studies failed to show an effect when both aided and unaided stimuli were tested at suprathreshold levels. Figure 3 and Table 1, taken from Billings et al. (2012), show the possible effects of testing near threshold (i.e., a physiological detection approach) as opposed to testing at suprathreshold levels (i.e., a physiological discrimination approach). Amplification effects are more likely to be found when the detection approach is used. A related factor to the physiological detection/discrimination perspective in the aided AEP literature that should be considered carefully is the hearing status of the participants. Many of the studies that demonstrate absent amplification effects were completed in individuals with normal hearing as a means to control interactions between an impaired auditory system and AEP results, while many studies that show amplification effects were completed in individuals with hearing impairment. In an attempt to bridge these findings, Billings et al. (2012) elicited aided AEPs from noise-masked normal-hearing individuals and demonstrated that a detection approach resulted in amplification effects and a discrimination approach did not. Additional well-controlled studies in hearing impaired individuals are needed to verify the validity of a detection approach where the aided AEP threshold is found. It appears that the aided AEP discrimination approach remains problematic clinically.
Figure 3.
Examples of physiological detection (a–d) and physiological discrimination (e–h) approaches from the aided CAEP literature. Results across these studies demonstrate significant amplification effects (unaided versus aided) for physiological detection, but very limited amplification effects for physiological discrimination. All figures were modified from published figures; the appropriate citation is indicated for each panel (see Table 1 for details). This figure was original published in Billings et al., 2012.
Table 1.
Experimental conditions used in the studies cited in Figure 3. Note that the parameters listed below are specific to the conditions used to generate the waveform data presented in Figure 3 and do not necessarily represent all conditions presented in each study. The eight studies included represent aided CAEP data in the literature for which unaided and aided waveforms were able to be reproduced. Table was originally published in Billings et al., 2012.
| Subjects
|
Experimental Design
|
Source | ||||||
|---|---|---|---|---|---|---|---|---|
| n | Characteristics | Stimulus | Duration (ms) | ISI (ms) | Signal Level | Conditions | ||
| Fig 3 Detection Approach Examples | ||||||||
| (A) McNeil et al., 2009 | 1 | 68 yo male; severe to profound SNHL | /ba/ | 115 | 750* | 60 dB nHL | Unaided and aided | Fig. 1, pg. 81: unaided and left hearing aid |
| (B) Rapin & Graziani, 1967 | 1 | 21 mo female; rubella, sedated | 500-Hz tone | Not Specified | Not Specified | 109 dB re: 0.0002 dynes/cm2 | Unaided and aided | Fig. 4, pg. 892: 109 dB with and 109 dB without aid |
| (C) Gravel, 1989 | 1 | 7 mo male; severe to profound SNHL | /da/ | Not Specified | Not Specified | Not Specified | Unaided and aided (aid set to user settings) | Fig. 3, pg. 271 |
| (D) Korczak et al., 2005 | 7 | Adults; severe to profound SNHL | /ba/&/da/ | 150 | 950 | 80 dB ppeSPL | Unaided and aided (aid set to MCL) | Fig. 3, pg. 176: Standard responses in lower panel, left |
| Fig 3 Discrimination Approach Examples | ||||||||
| (E) Billings et al., 2007 | 13 | Young adults; normal hearing | 1000-Hz tone | 757 | 1910 | 50 dB SPL | Unaided and aided (20 dB gain) | Fig. 2, pg. 238: 50 dB aided and 50 dB unaided waveforms |
| (F) Tremblay et al., 2006 | 7 | Young adults; normal hearing | /si/ | 655 | 1910 | 64 dB SPL | Unaided and aided (average gain of 19 dB) | Fig. 7, pg. 99: Top panel |
| (G) Billings et al., 2011 | 9 | Young adults; normal hearing | 1000-Hz tone | 756 | 1910 | 40 dB SPL | Unaided and aided (gain of 20 dB) | Fig. 6, pg. 7: Panel (a) unaided waveform, panel (b) aided waveform |
| (H) Korczak et al., 2005 | 4 | Adults; moderate SNHL | /ba/&/da/ | 150 | 950 | 85 dB HL | Unaided and aided (aid set to MCL) | Fig. 3, pg. 176: Standard responses in lower panel, center |
Reference does not state whether this value refers to onset to onset, or offset to onset. All other inter-stimulus intervals (ISIs) refer to offset to onset. HL, hearing level; ISI, inter-stimulus interval; MCL, most comfortable loudness; ppeSPL, peak-to-peak equivalent sound pressure level; SNHL, sensorineural hearing loss; SPL, sound pressure level.
There is also an emerging interest in other electrophysiological measures. The ASSR and the FFR may be promising as clinical tools to measure the physiological aided threshold. Several researchers have demonstrated good potential for their use in measuring aided thresholds and demonstrating the effect of amplification (Picton et al., 1998; Dimitrijevic et al., 2004; Stroebel et al., 2007; Shemesh et al., 2012; Damarla & Manjula; 2007; Anderson & Kraus, 2013). An advantage of these responses may be that they are elicited by longer-duration stimuli than the ABR and are analyzed in the frequency domain, effectively reducing dependence on the size of a particular peak. However, similar difficulties to those experienced with the ABR may be present given that the ASSR is evoked primarily by the initial portions of a stimulus (Korczak, 2012; also see Clinard & Tremblay in this issue). It remains unknown whether the spectral analyses used to interpret these steady-state potentials will allow this measure to overcome challenges that have been demonstrated with aided CAEPs and aided ABRs. Additional research is needed to determine the extent of the interactions between hearing-aid-processed stimuli and the neural responses.
In summary, a portion of the amplification effect variability that exists in the literature may be explained by differences in approach across studies. Where a detection approach is taken, there is more likely to be significant amplification effects; where a discrimination approach is taken, an absent amplification effect is more likely to result.
4. What are the limitations of aided CAEPs?
Why have various studies employing aided CAEPs found no effect of amplification? As indicated above, in some cases the absence of effects may be due to testing at suprathreshold levels. At threshold, the specifics of the hearing aid processing lose their relevance to some degree because an absent response in the unaided condition is often compared to a present response in the aided condition; in that situation, the overall level is the robust cue being encoded (i.e., a detection task). However, when two suprathreshold conditions are compared, as in a discrimination task, the more subtle hearing aid processing characteristics complicate the picture. At least two major cues have emerged in the literature as contributing to the absent amplification effects at suprathreshold stimulus levels: signal-to-noise ratio (SNR) and stimulus onset characteristics (e.g., rise time). Billings and colleagues (2007, 2011) demonstrated that SNR plays an important role in aided CAEP testing. Specifically, they found that when amplification effects were absent, the SNR ratio between the unaided and aided conditions was very similar. Furthermore, they demonstrated that CAEPs have a strong sensitivity to SNR rather than absolute signal level (Billings et al., 2009). This could be problematic for testing amplification effects at suprathreshold levels as shown in Figure 3. It may be that when signal and noise are present at suprathreshold levels that amplification effects would not be expected. However, while SNR is an important factor, it is also clear that SNR does not account for all aided CAEP results (Billings et al., 2011; Jenstad et al., 2012).
Onset characteristics of the stimulus are also critical to CAEP morphology, and it has been demonstrated that hearings aids can dramatically modify the signal onset in ways that will affect AEPs (Jenstad et al., 2012). It is worth noting that the onset modifications were not consistent across hearing aids, signal levels, or signal types. The two digital hearing aids that were tested varied dramatically in the resulting modifications to onsets. Jenstad pointed out that SNR and onset modifications together do not account for the lack of amplification effects that they found. In addition, even when rise characteristics such as overshoot and rise time are modified by the hearing aid, there is not always an effect on CAEPs (Easwar, Glista et al, 2012). Additional research is needed to determine what other acoustic properties of the hearing-aid-processed stimuli are affecting the AEP.
Aided CAEPs can be reliably recorded for some speech cues and not in other cases. For example aided CAEPs showed reliable differences between the syllables ‘see’ and ‘shee’ (Tremblay et al., 2006a, 2006b), but did not distinguish between the syllables ‘ma’, ‘ga’, and ‘ta’ (Munro et al., 2011). Additional research will need to determine how sensitivities to various cues differ in unaided and aided conditions. That information may help to determine what cues affecting AEPs, in addition to SNR and onset cues, are being modified by the hearing aid.
It is important to note that an absent AEP is usually nondiagnostic on its own (Stapells, 2002); that is, an absent response does not conclusively indicate that the person cannot hear the stimulus. AEPs represent the neural synchrony of some subset of auditory neurons at a given point in time and provide only a glimpse into the complex central auditory nervous system. Furthermore, the absence of a response is determined either by visual inspection or by some automated detection algorithm, and does not necessarily indicate that there is not neural response present. It may be that biological noise, subject state or a host of other factors have prevented our ability to detect a response. For this reason, both the unaided and aided AEP literature has examples of absent evoked potentials to stimuli that are clearly detectable behaviorally. As mentioned above, while visual CAEP detection thresholds are within 10 dB of behavioral threshold in most cases, they have been found to exceed 20 dB in some studies and individuals.
In summary, at suprathreshold levels, hearing aid processing and AEPs interact in unexpected ways. We are beginning to understand some of these confounds (i.e., SNR and signal onset modifications); however, additional research is needed to determine what other factors are contributing. Furthermore, it appears that the causes of an absent amplification effect vary from individual to individual and hearing aid to hearing aid, a challenge that will also need to be addressed further in the laboratory.
5. Evidence of Hearing Aid Acclimatization
Hearing aid acclimatization has been studied from both behavioral and physiological perspectives. Behaviorally, the evidence for perceptual changes over time is quite mixed, with some studies reporting significant increases in benefit and others reporting no significant change (for a review see Turner et al., 1996). This discrepancy in the literature was part of the motivation behind the 1995 Eriksholm Workshop that focused on the deprivation and acclimatization effects associated with hearing impairment and hearing aid use. It was clear that some of the discrepancy in the literature could be due to how acclimatization was being defined. As part of this workshop, researchers defined acclimatization as a change in performance over time that is linked to the available acoustic information and that cannot be attributed to task, procedural, or training effects (Arlinger et al., 1996). Probably one of the largest available datasets, and most robust in terms of outcome measures, is a longitudinal study completed by Humes and colleagues (2002, 2003) in which a battery of speech perception measures and self-report measures were used to investigate changes over time in more than 134 individuals for one year, 49 individuals for two years, and 9 individuals for three years. Results demonstrated little evidence for acclimatization. A review of the behavioral acclimatization literature is beyond the scope of this manuscript and has been addressed by others (Turner et al., 1996; Palmer et al., 1998; Kuk et al., 2003; Munro et al., 2008); however, even 18 years after the Eriksholm Workshop, conflicting results remain unexplained and one could conclude as they did in 1995, that while there is evidence for acclimatization, it is difficult to specify under what conditions, the size of the effect, or its time course (Arlinger et al., 1996).
The mixed behavioral evidence surrounding hearing aid acclimatization has led some to explore physiological measures as a marker of neural changes throughout the auditory system. Gatehouse and Robinson (1996) applied a clever methodology that had been initially used behaviorally (Gatehouse, 1989) to study acclimatization using AEPs; unilaterally aided individuals were tested for acclimatization effects by comparing results for the hearing aid fitted ear compared to the non-fitted ear, effectively using the non-fitted ear as a control. In this early case study they reported improved intensity discrimination and increases in cortical AEP amplitudes at the highest intensity tested. This result has been followed by a series other studies from Munro and colleagues as well as others, demonstrating some physiological acclimatization effects using acoustic reflex thresholds, the ABR, and cortical AEPs (Munro et al. 2007a; Munro et al. 2007b; Bertoli et al., 2011). In contrast, a recent doctoral dissertation began to explore acclimatization in the aided condition, but found little evidence for any acclimatization effect as measured using cortical AEPs (McCullough, 2010). Given these mixed results, it remains unclear how the physiological effects relate to perception. It is noteworthy that these physiological acclimatization measures were made without the hearing aid on the individual. Many of the same difficulties and variability that are associated with measuring aided thresholds are present when measuring changes over time as well; however, one might hypothesize that measures would be most sensitive to acclimatization if testing were completed in the aided condition. Several studies by Sharma and colleagues have demonstrated neural change over time with the device being worn during testing (Sharma et al., 2004; Sharma et al., 2005); unfortunately details about the hearing aid settings from test to retest and the acoustic modifications resulting from amplification were not reported making it difficult to judge how these results fit into the literature.
In summary, while acclimatization does occur, the extent of that effect remains unclear. Furthermore, physiological measures have not helped to clarify these issues. It appears that before acclimatization can be demonstrated in the aided condition, the interaction between hearing aid processing and the resulting evoked potentials needs to be better understood.
6. Future Research Needs and Recommendations
An area not addressed in this review, but important to the future of understanding the effects of hearing aids on brain and behavior is the effect of auditory training in hearing aid users (for a review of physiologic effects of training, see Tremblay et al., 2007). This area will be important for future research as training programs become more user friendly and self administered. Data have been mixed on the efficacy of these programs, and more studies are needed to clarify what neural changes, if any, are occurring as a result of these programs.
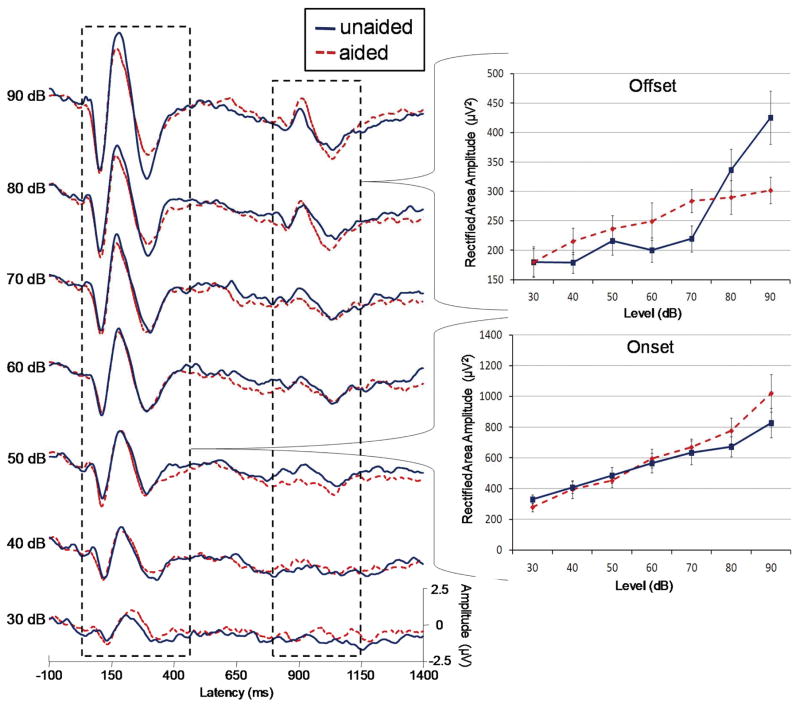
In the aided evoked potential literature, there is emerging interest in the use of aided change responses (Hinchey et al., 2009; Kirby et al., 2012). A change response consists of a repeated N1 and P2 peaks that result from changes within a stimuli. Also called the acoustic change complex (ACC) in the literature (Martin et al., 1999; 2000), the change response has been used with aided AEPs with the syllables ‘see’ and ‘shee’ (Tremblay, Billings et al., 2006; Tremblay, Kalstein et al., 2006). The neural response the change associated with stimulus offset also demonstrates promise. Figure 4 shows data from a previous paper (Billings et al., 2007). We found previously that there was no significant effect of amplification on the N1-P2 onset. We reanalyzed the onset and offset response (shown in the right dotted box) using a rectified area measure rather than peak values (offset peaks were very difficult to identify given the size of the physiological noise relative to the response). The rectified area was calculated over the range of the onset and offset response (60 to 450 and 790 to 1140 ms, respectively) after baseline correction was applied over the same range (baseline correction was used as a means to control for any drift that occurred later in the waveform). While neural onset amplification effects remained absent as previously shown (Billings et al., 2007), offset amplification effects were significant (F=19.8(6,72), p=<.001) and an interaction between level and amplification (F=5.6(6,72), p=.003) was found using a repeated measures analysis of variance (ANOVA). Subsequent posthoc testing using paired samples t-test revealed significant effects and trends of amplification at 40, 60, 70, and 80 dB (p=.023, .074, .003, .008, respectively). It may be that some of the hearing aid modifications to stimulus rise characteristics (Jenstad et al., 2012) are not as problematic for offset aided AEPs. In addition, perhaps area measures can help improve sensitivity to some of the subtle effects of amplification in a discrimination approach paradigm.
Figure 4.
Grand averages (n=13) and area measures for unaided (dotted) and aided (solid) conditions as a function of signal level. A minimal effect of amplification is seen for the onset at most signal levels, similar to peak results published previously (Billings et al., 2007). However, offset area measures demonstrate a significant difference between unaided and aided conditions. It is possible that an area measure or an acoustic change peak such as the offset is sensitive to amplification effects unlike the onset response.
One inherent difficulty with aided AEPs is that results using one hearing aid may not necessarily translate to other hearing aids, or even to the same hearing aid with different settings. If aided CAEPs are to be used successfully in the clinic on a larger scale, this difficulty of variability needs to be understood. A key to improving understanding will be to characterize the interaction between the hearing aid signal processing, and resulting modification to the stimulus, and the AEP. To move our understanding of aided AEPs forward, it is important that researchers record and characterize the output of the hearing aid. This can be done with in-the-canal recordings using a probe tube microphone (such as the Etymotic ER7c) to verify what acoustic cues are reaching the ear, or at the very least in a manniquim or coupler. The interaction between hearing aid and EPs is the main contributor to problems currently.
The aided AEP literature has progressed dramatically in the last decade, mostly as a result of moving from case study reports to group data with more statistical power. Additional large sample studies will be needed to determine what subtle variables are important to understanding aided AEPs. Once the contributing factors are identified, translation back to the clinical application in individuals will need to be completed.
In conclusion, the aided CAEP literature demonstrates considerable difficulties in application to the clinic especially when trying to differentiate two present responses; the variability within and across hearing aids, individuals, and studies is quite problematic clinically. However, when used in a physiological detection approach (i.e., comparing an absent response to a response that is present) in conjunction with the battery of other audiological tests, aided AEPs may be useful in a detection approach to determine whether aided input is reaching the auditory cortex. It should be kept in mind, however, that physiological detection of an aided stimulus does not mean that the hearing aid is fit appropriately; in fact, a present response compared to an absent one indicates only that the signal, or some portion of the signal, is being encoded physiologically. In addition, as with unaided AEPs, visual detection thresholds can be quite elevated when compared with behavioral thresholds; this limitation in the aided domain will have to be more completely determined for clinical use of aided AEPs to be of great benefit.
Learning Objectives.
The learner will be able to identify the potential uses and challenges associated with using aided cortical auditory evoked potentials.
Acknowledgments
Special thanks to Tina Penman, Lucas Baltzell, Angela Eilbes, and Melissa Papesh for assistance with and comments about this manuscript. This work was supported by grants from the National Institute on Deafness and Other Communication Disorders (R03DC10914) and the Veterans Affairs Rehabilitation Research and Development Service (C8006W).
Abbreviations
- CAS
central auditory system
- SNR
signal-to-noise ratio
- EEG
electroencephalography
- CAEPs
cortical auditory evoked potentials
- AEPs
auditory evoked potentials
- ABR
auditory brainstem response
- ASSR
auditory steady state response
- FFR
frequency following response
References
- Anderson S, Kraus N. The potential role of the cABR in assessment and management of hearing impairment. Int J of Otolaryngology. 2013:1–10. doi: 10.1155/2013/604729. http://dx.doi.org/10.1155/2013/604729. [DOI] [PMC free article] [PubMed]
- Anderson S, Parbery-Clark A, White-Schwoch T, Drehobl S, Kraus N. Effects of hearing loss on the subcortical representation of speech cues. J Acoust Soc Am. 2013;133(5):3030–3038. doi: 10.1121/1.4799804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arlinger S, Gatehouse S, RA, Byrne D, et al. Report of the Eriksholm workshop on auditory depreivation and acclimatization. Ear Hear. 1996;17(3):87S–98S. doi: 10.1097/00003446-199617031-00009. [DOI] [PubMed] [Google Scholar]
- Beauchaine KA, Gorga MP, Reiland JK, Larson LL. Application of ABRs to the hearing-aid selection process: preliminary data. J Speech Hear Res. 1986;29(1):120–128. doi: 10.1044/jshr.2901.120. [DOI] [PubMed] [Google Scholar]
- Bertoli S, Probst R, Bodmer D. Late auditory evoked potentials in elderly long-term hearing-aid users with unilateral or bilateral fittings. Hearing Research. 2011;280:58–69. doi: 10.1016/j.heares.2011.04.013. [DOI] [PubMed] [Google Scholar]
- Billings CJ, Tremblay KL, Souza PE, Binns MA. Effects of hearing aid amplification and stilulus intensity on cortical auditory evoked potentials. Audiol Neurotol. 2007;12:234–246. doi: 10.1159/000101331. [DOI] [PubMed] [Google Scholar]
- Billings CJ, Tremblay KL, Strecker GC, Tolin WM. Human evoked cortical activity to signal-to-noise ration and absolute signal level. Hearing Research. 2009;254:15–24. doi: 10.1016/j.heares.2009.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billings CJ, Bennett KO, Molis MR, Leek MR. Cortical encoding of signal in noise: effects of stimulus type and recording paradigm. Ear Hear. 2011;32(1):53–60. doi: 10.1097/AUD.0b013e3181ec5c46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billings CJ, Papesh MA, Penman TM, Baltzell LS, Gallun FJ. Clinical use of aided cortical auditory evoked potentials as a measure of physiological detection or physiological discrimination. Int J of Otolaryngology. 2012:1–14. doi: 10.1155/2012/365752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown E, Klein AJ, Snydee KA. Hearing-aid-processed tone pips: electroacoustic and ABR characteristics. J Am Acad Audiol. 1999;10(4):190–197. [PubMed] [Google Scholar]
- Damarla VKS, Manjula P. Application of ASSR in the hearing aid selection process. Australian and New Zealand Journal of Audiology. 2007;29(2):89–97. [Google Scholar]
- Dillon H. So, baby, how does it sound? Cortical assessment of infants with hearing aids. Hear J. 2005;58(10):10–17. [Google Scholar]
- Dimitrijevic A, John MS, Picton TW. Auditory steady-state responses and word recognition scores in normal-hearing and hearing-impaired adults. Ear Hear. 2004;25(1):68–84. doi: 10.1097/01.AUD.0000111545.71693.48. [DOI] [PubMed] [Google Scholar]
- Easwar V, Glista D, Purcell DW, Scollie SD. Hearing aid processing changes tone burst onset: effect on cortical auditory evoked potentials in individuals with normal audiometric thresholds. Am J Audiol. 2012;21:82–90. doi: 10.1044/1059-0889(2012/11-0039). [DOI] [PubMed] [Google Scholar]
- Easwar V, Purcell DW, Scollie SD. Electroacoustic comparison of hearing aid output of phonemes in running speech versus isolation: implications for aided cortical auditory evoked potentials testing. Int J of Otolaryngology. 2012:1–10. doi: 10.1155/2012/518202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frye Electronics Inc, HEARLab System. Operator’s Manual. 2010 http://frye.com/manuals/hearlab.pdf.
- Gatehouse S. Apparent auditory deprivation effects of late onset: The role of presentation level. J Acoust Soc Am. 1989;86:2103–2106. doi: 10.1121/1.398469. [DOI] [PubMed] [Google Scholar]
- Gatehouse S, Robinson K. Acclimatization to monaural hearing aid fitting-effects on loudness functions and preliminary evidence for parallel electrophysiological and behavioural effects. In: Kollmeier B, editor. Psychoacoustics, Speech and Hearing Aids. Singapore: World Scientific; 1996. pp. 319–330. [Google Scholar]
- Gerling IJ. In search of a stringent methodology for using ABR audiometric results. Hear J. 1991;44(26):28–30. [Google Scholar]
- Glista D, Easwar V, Purcell DW, Scollie S. A pilot study on cortical auditory evoked potentials in children: aided CAEPs reflect improved high-frequency audibility with frequency compression hearing aid technology. Int J of Ototlaryngology. 2012:1–12. doi: 10.1155/2012/982894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Golding M, Pearce W, Seymour J, Cooper A, Ching T, Dillon H. The relationship between obligatory cortical auditory evoked potentials (CAEPS) and functional measure in young infants. J Am Acad Audiol. 2007;18(2):117–125. doi: 10.3766/jaaa.18.2.4. [DOI] [PubMed] [Google Scholar]
- Gorga MP, Beauchaine KA, Reiland JK. Comparison of onset and steady-state responses of hearing aids: implications for use of the auditory brainstem response in the selection of hearing aids. J Speech Hear Res. 1987;20(1):130–136. doi: 10.1044/jshr.3001.130. [DOI] [PubMed] [Google Scholar]
- Gravel JS, Kurtzberg D, Stapells DR, Vaughan HG, Wallace IF. Case studies. Seminars in Hearing. 1989;10(3):272–287. [Google Scholar]
- Hecox KE. Role of auditory brain stem response in the selection of hearing aids. Ear Hear. 1983;4(1):51–55. doi: 10.1097/00003446-198301000-00009. [DOI] [PubMed] [Google Scholar]
- Hinchey T, Korczak P, Martin BA, Pallett S. Effects of hearing aid gain on slow cortical auditory evoked potentials. Association for Research in Otolaryngology Mid-Winter Meeting; 2009. [Google Scholar]
- Hood LJ. Clinical Applications of the Auditory Brainstem Response. Clifton Park, NY: Delmar Cengage Learning; 1998. [Google Scholar]
- Humes LE, Wilson DL, Barlow NN, Garner C. Changes in hear-aid benefit following 1 or 2 year of hearing-aid use by older adults. J of Speech, Language, and Hearing Research. 2002;45:772–782. doi: 10.1044/1092-4388(2002/062). [DOI] [PubMed] [Google Scholar]
- Humes LE, Wilson DL. An examination of changes in hearing-aid performance and benefit in the elderly over a 3-year period of hearing-aid use. J of Speech, Language, and Hearing Research. 2003;46:137–145. doi: 10.1044/1092-4388(2003/011). [DOI] [PubMed] [Google Scholar]
- Ikeda K, Hayashi A, Matsuda O, Sekiguchi T. An ignoring task improves validity of cortical evoked response audiometry. NeuroReport. 2010;21:709–715. doi: 10.1097/WNR.0b013e32833b502a. [DOI] [PubMed] [Google Scholar]
- Jenstad LM, Marynewich S, Stapells DR. Slow cortical potentials and amplification – Part II: acoustic measures. Int J of Ototlaryngology. 2012:1–14. doi: 10.1155/2012/386542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kileny P. Auditory brainstem responses as indicators of hearing aid performance. Ann Otol Rhinol Laryngol. 1982;91(1 Pt 1):61–64. doi: 10.1177/000348948209100114. [DOI] [PubMed] [Google Scholar]
- Kirby B, Abbas PJ, Brown CJ. Verification of frequency compression parameters using the auditory change complex. Poster presentation at American Auditory Society Scientific and Technology Meeting; Scottsdale. March 2012. [Google Scholar]
- Korczak PA, Kurtzberg D, Stapells DR. Effects of sensorineural hearing loss and personal hearing aids on cortical event-related potential and behavioral measure of speech-sound processing. Ear Hear. 2005;26:165–185. doi: 10.1097/00003446-200504000-00005. [DOI] [PubMed] [Google Scholar]
- Korczak P, Smart J, Delgado R, Strobel TM, Bradford C. Auditory steady-state responses. J Am Acad Audiol. 2012;23:146–170. doi: 10.3766/jaaa.23.3.3. [DOI] [PubMed] [Google Scholar]
- Kraus N, McGee TJ. Mismatch negativity in the assessment of central auditory function. Am J Audiol. 1994:39–51. doi: 10.1044/1059-0889.0302.39. [DOI] [PubMed] [Google Scholar]
- Kuk FK, Potts L, Valente M, Lee L, Picirrillo J. Evidence of acclimatization in persons with severe-to-profound hearing loss. J Am Acad Audiology. 2003;14(2):84–99. doi: 10.3766/jaaa.14.2.4. [DOI] [PubMed] [Google Scholar]
- Kurtzberg D. Cortical event-related potential assessment of auditory system function. Seminars in Hearing. 1989;10(3):252–261. [Google Scholar]
- Martin BA, Boothroyd A. Cortical, auditory, event related potentials in response to periodic and aperiodic stimuli with the same spectral envelope. Ear Hear. 1999;20:33–44. doi: 10.1097/00003446-199902000-00004. [DOI] [PubMed] [Google Scholar]
- Martin BA, Boothroyd A. Cortical, auditory, evoked potentials in response to changes of spectrum and amplitude. Journal of the Acoustical Society of America. 2000;107:2155–2161. doi: 10.1121/1.428556. [DOI] [PubMed] [Google Scholar]
- Martin BA, Tremblay KL, Korczak P. Speech evoked potentials: from the laboratory to the clinic. Ear Hear. 2008;29(3):285–313. doi: 10.1097/AUD.0b013e3181662c0e. [DOI] [PubMed] [Google Scholar]
- Marynewich S, Jenstad LM, Stapells DR. Slow cortical potentials and amplification – Part I: N1-P2 measures. Int J of Otolaryngology. 2012:1–11. doi: 10.1155/2012/921513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCullagh JP. PhD thesis. University of Connecticut; Mansfield, Conn, USA: 2010. An investigation of central auditory nervous system plasticity following amplification. [Google Scholar]
- McNeill C, Sharma M, Purdy SC. Are cortical auditory evoked potentials useful in the clinical assessment of adults with cochlear implants? Cochlear Implants Int. 2009;10(S1):78–84. doi: 10.1002/cii.391. [DOI] [PubMed] [Google Scholar]
- Munro KJ, Pisareva NY, Parker DJ, Purdy SC. Asymmetry in the auditory brainstem response following experience of monaural amplification. NeuroReport. 2007;18(17):1871–1874. doi: 10.1097/WNR.0b013e3282f1b003. [DOI] [PubMed] [Google Scholar]
- Munro KJ, Walker AJ, Purdy SC. Evidence for adaptive plasticity in elderly monaural hearing aid users. NeuroReport. 2007;18(12):1237–1240. doi: 10.1097/WNR.0b013e32822025f4. [DOI] [PubMed] [Google Scholar]
- Munro KJ. Reorganization of the adult auditory system: perceptual and physiological evidence from monaural fitting of hearing aids. Trends in Amplification. 2008;12(3):254–271. doi: 10.1177/1084713808323483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munro KJ, Purdy SC, Ahmed S, Begum R, Dillon H. Obligatory cortical auditory evoked potential waveform detection and differentiation using a commercially available clinical system: HEARLab™. Ear Hear. 2011;32(6):782–786. doi: 10.1097/AUD.0b013e318220377e. [DOI] [PubMed] [Google Scholar]
- National Acoustics Laboratories, HEARLab System. Operator’s Manual. 2010 http://hearlab.nal.gov.au/pdf/HEARLab-Manual.pdf.
- Palmer C, Nelson C, Lindley G. The functionally and physiologically plastic adult auditory system. J Acoust Soc Amer. 1998;103(4):1705–1721. doi: 10.1121/1.421050. [DOI] [PubMed] [Google Scholar]
- Picton TW, Durieux-Smith A, Champagne SC, et al. Objective evaluation of aided thresholds using auditory steady-state responses. J Am Acad Audiol. 1998;9:315–331. [PubMed] [Google Scholar]
- Picton TW. Human Auditory Evoked Potentials. Plural Publishing, Inc; 2011. [Google Scholar]
- Prasher D, Mula M, Luxon L. Cortical evoked potential criteria in the objective assessment of auditory threshold: a comparison of noise induced hearing loss with Meniere’s disease. J Laryngol Otol. 1993;107(9):780–786. doi: 10.1017/s0022215100124429. [DOI] [PubMed] [Google Scholar]
- Purdy SC, Katsch R, Dillon H, Storey L, Sharma M, Agung K. Aided cortical auditory evoked potentials for hearing instrument evaluation in infants. In: Seewald RC, Bamford JM, editors. A Sound Foundation Through Early Amplification. Phonak AG; Basel, Switzerland: 2005. pp. 115–127. [Google Scholar]
- Rapin I, Graziani LJ. Auditory-evoked responses in normal, brain damaged, and deaf infants. Neurology. 1967;17:881–894. doi: 10.1212/wnl.17.9.881. [DOI] [PubMed] [Google Scholar]
- Rapin I, Ruben RJ, Lyttle M. Diagnosis of hearing loss in infants using auditory evoked responses. Laryngoscope. 1970;80(5):712–22. doi: 10.1288/00005537-197005000-00003. [DOI] [PubMed] [Google Scholar]
- Sharma A, Tobey E, Dorman MF, et al. Central auditory maturation and babbling development in infants with cochlear implants. Arch Otolaryngol Head Neck Surg. 2004;130(5):511–516. doi: 10.1001/archotol.130.5.511. [DOI] [PubMed] [Google Scholar]
- Sharma A, Dorman MF, Kral A. The influence of a sensitive period on central auditory development in children with unilateral and bilateral cochlear implants. Hearing Research. 2005;203:134–143. doi: 10.1016/j.heares.2004.12.010. [DOI] [PubMed] [Google Scholar]
- Shemesh R, Attias J, Magdoub H, Nageris BI. Prediction of aided and unaided audiograms using sound-field auditory steady-state evoked responses. Int J of Audiol. 2012;51:746–753. doi: 10.3109/14992027.2012.700771. [DOI] [PubMed] [Google Scholar]
- Souza PE, Trmblay KL. New perspectives on assessing amplification effects. Trends Amplif. 2006;10:119–143. doi: 10.1177/1084713806292648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stapells DR. Cortical event-related potentials to auditory stimuli. In: Katz J, Medwetsky L, Burkard R, editors. Handbook of Clinical Audiology. 5. Baltimore, MD: Lippingcott, Williams & Wilkins; 2002. [Google Scholar]
- Stroebel D, Swanepoel D, Groenewald E. Aided auditory steady-state responses in infants. Int J of Audiology. 2007;46(6):287–292. doi: 10.1080/14992020701212630. [DOI] [PubMed] [Google Scholar]
- Tremblay KL, Billings CJ, Friesen LM, Souza PE. Neural representation of amplified speech sounds. Ear Hear. 2006;27(2):93–103. doi: 10.1097/01.aud.0000202288.21315.bd. [DOI] [PubMed] [Google Scholar]
- Tremblay KL, Kalstein L, Billings CJ, Souza PE. The neural representation of consonant-vowel transitions in adults who wear hearing aids. Trends in Amplification. 2006;10(3):155–162. doi: 10.1177/1084713806292655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tremblay KL. Training-related changes in the brain: evidence from human auditory-evoked potentials. Seminars in Hearing. 2007;28(2):120–132. [Google Scholar]
- Turner CW, Humes LE, Bentler RA, Cox RM. A review of past research on changes in hearing aid benefit over time. Ear Hear. 1996;17(3):14S–28S. doi: 10.1097/00003446-199617031-00003. [DOI] [PubMed] [Google Scholar]
- Van Maanen A, Stapells DR. Comparison of multiple auditory steady-state responses (80 versus 40 Hz) and slow cortical potentials for threshold estimation in hearing-impaired adults. Int J of Audiol. 2005;44:613–624. doi: 10.1080/14992020500258628. [DOI] [PubMed] [Google Scholar]
- Yeung K, Wong L. Prediction of hearing thresholds: comparison of cortical evoked response audiometry and auditory steady state response audiometry techniques. Int J of Audiol. 2007;46(1):17–25. doi: 10.1080/14992020601102238. [DOI] [PubMed] [Google Scholar]