Abstract
Background
The donation of multiple allografts from a single living donor is a rare practice, and the patient characteristics and outcomes associated with these procedures are not well described.
Methods
Using the SRTR registry, we identified 101 living multi-organ donors and their 133 recipients.
Results
The 49 sequential (donations during separate procedures) multi-organ donors provided grafts to 81 recipients: 21 kidney-then-liver, 15 liver-then-kidney, 5 lung-then-kidney, 3 liver-then-intestine, 3 kidney-then-pancreas, 1 lung-then-liver, and 1 pancreas-then-kidney. Of these donors, 38% donated 2 grafts to the same recipient and 15% donated 2 grafts as non-directed donors. Compared to recipients from first-time, single organ living donors, recipients from second-time living donors had similar graft and patient survival. The 52 simultaneous (multiple donations during one procedure) multi-organ donors provided 2 grafts to 1 recipient each: 48 kidney-pancreas and 4 liver-intestine. Donors had median (IQR) 13.4 [8.3–18.5] years of follow-up for mortality. There was one reported death of sequential donor (2.5 years after second donation). Few post-donation complications were reported over median (IQR) 116 (0–295) days follow-up; however, routine living donor follow-up data were sparse. Recipients of kidneys from second-time living donors had similar graft (p=0.8) and patient survival (p=0.4) when compared to recipients from first-time living donors. Similarly recipients of livers from second-time living donors had similar graft survival (p=0.8) and patient survival (p=0.7) when compared to recipients from first-time living donors.
Conclusions
Careful documentation of outcomes is needed to ensure ethical practices in selection, informed consent, and post-donation care of this unique donor community.
INTRODUCTION
Living donors provide nearly 18% of the organs used for transplantation in the United States (US) each year. 1 Kidney and liver donations are the most common and well-studied forms of living organ donation, but living donors in the US can donate a lung lobe, partial intestine, and even a segment of pancreas with varying degrees of success.2,3 Given the scarcity of organs and the growing transplant waitlist, transplanting multiple grafts from a single living donor might be a potentially useful strategy for a subset of transplant candidates such as pediatric or lower-risk left liver lobe recipients.4 This rare practice is a topic of both clinical and ethical interest; however, there are very few published studies to inform these discussions.
Case series and case reports have documented living multi-organ donation involving a variety of organ pairs, including documentation of simultaneous and sequential liver-kidney, pancreas-kidney, liver-small bowel, and lung-liver living donor transplants from a single donor.4–13 Most case series focus on the recipient, with minimal documentation of donor outcomes. In general, recipients of living donor organs experience advantages, including decreased waiting time, decreased cold ischemia time, increased opportunities for immunological matching, and increased graft survival. However, the paucity of data on the outcomes of living multi-organ donors prevents weighing of risks and benefits for the donor candidate, which is important to comprehensive informed consent.14
The goal of this study was to characterize the landscape of living multi-organ transplantation in the US. Using national registry data, we characterized living multi-organ donors and their recipients and examined outcomes associated with the practice of living multi-organ donation. This study may inform future discussions regarding donor selection, informed consent, and patient education practices.
MATERIALS AND METHODS
Data Source
This study used data from the Scientific Registry of Transplant Recipients (SRTR) external release made available in June 2017. The SRTR data system includes data on all donors, wait-listed candidates, and transplant recipients in the US, submitted by members of the Organ Procurement and Transplantation Network (OPTN), and has been described elsewhere.15 The Health Resources and Services Administration (HRSA), US Department of Health and Human Services, provides oversight to the activities of the OPTN and SRTR contractors.
Study population
The study population consisted of 101 living multi-organ donors and their 133 recipients. Sequential living multi-organ donors were defined as individuals who donated grafts on separate dates (i.e. separate procedures). Simultaneous living multi-organ donors were defined as individuals who donated 2 grafts on the same day, presumably during the same procedure. We studied 49 sequential living multi-organ donors with 81 unique recipients and 52 simultaneous living multi-organ donors with 52 unique recipients between March 1994 and January 2017. We compared recipients of the second graft from sequential living multi-organ recipients to recipients of a graft from first-time living donors. For these analyses, we included 140,501 recipients of first-time single-organ living donor kidneys, 6,056 recipients of first-time living donor livers, 22 recipients of first-time living donor pancreata, and 38 recipients of first-time living donor intestine transplants recorded in the SRTR registry in the same time period.
Statistical analysis
Groups of living multi-organ donors were compared with the Mann-Whitney rank-sum test (continuous variables) or chi-squared test (categorical variables). All-cause graft loss and mortality for recipients of living donor organs were estimated using the Kaplan-Meier method. Differences in recipient patient and graft survival were assessed using the log-rank test of equality. We used 2-sided alpha of 0.05 to indicate a statistically significant difference. All analyses were performed using STATA 14.2/MP for Linux (College Station, Texas).
RESULTS
Sequential Living Multi-organ Donation
Sequential Donor Characteristics and Outcomes
Among the 49 living multi-organ donors who underwent sequential multi-organ donation operations, 21 donated a kidney-then-liver, 15 donated a liver lobe-then-kidney, 5 donated a lung lobe-then-kidney, 3 donated a liver lobe-then-intestine, 3 donated a kidney-then-pancreas segment, 1 donated a lung lobe-then-liver, and 1 donated a pancreas segment-then-kidney (Table 1). These procedures occurred in all 11 United Network for Organ Sharing (UNOS) regions. The majority of sequential living multi-organ donors were women (65.3%) and white (77.6%), with median age at first donation of 38 years (interquartile range (IQR): 28–44). Median time between donations was 3.7 years (IQR: 1.8–7.0). Sequential liver-then-intestine donors had the shortest time between donations (IQR: 2–40 days). With respect to donor-recipient relationships, there were 17 (34.7%) donors who donated both grafts to the same recipient. There were a total of 22 nondirected donations, of which 59% were liver and the remainder were kidney. Of these nondirected donations, 8 were from donors who donated 1 organ to a known recipient and 1 in a nondirected manner. Fourteen recipients received grafts in a nondirected manner from 7 sequential donors, who each donated 2 grafts.
Table 1. Characteristics of living donors who underwent sequential living multi-organ donation.
There were 49 donors who donated organs during separate procedures. Of these donors, 38% donated two grafts to the same recipient and 15% donated two grafts as non-directed donors. The kidney-then-liver group had the smallest proportion of donating to the same recipient and the highest proportion of non-directed donations.
| Kidney-then-liver | Liver-then-kidney | Lung-then-kidney | Liver-then-intestine | Kidney-then-pancreas | Pancreas-then-kidney | Lung-then-liver | |
|---|---|---|---|---|---|---|---|
| N (%) | 21 (42.9) | 15 (30.6) | 5 (10.2) | 3 (6.1) | 3 (6.1) | 1 (2.0) | 1 (2.0) |
| % Female | 61.9 | 66.7 | 60.0 | 100.0 | 66.7 | 100.0 | 0.0 |
| Median age at 1st donation, in years (IQR) | 39.9 (32.8–48.7) | 37.8 (27.9–41.8) | 30.2 (27.9–43.3) | 25.3 (19.3–25.8) | 38.4 (32.4–43.0) | 45.1 (45.1–45.1) | 52.3 (52.3–52.3) |
| Median age at 2nd donation, in years (IQR) | 44.1 (35.4–50.2) | 40.8 (35.8–48.3) | 47.5 (42.1–48.4) | 25.4 (19.3–25.8) | 44.2 (34.3–52.2) | 46.8 (46.8–46.8) | 57.1 (57.1–57.1) |
| Median time between donations, in years (IQR) | 3.0 (2.0–4.2) | 3.7 (0.8–8.6) | 11.0 (7.0–16.2) | 0.0 (0.0–0.1) | 5.7 (1.9–9.1) | 1.7 (1.7–1.7) | 4.9 (4.9–4.9) |
| Median BMI at 2nd donation (IQR) | 25.8 (21.8–27.2) | 26.6 (21.6–28.4) | 23.6 (21.5–24.4) | 23.6 (20.0–37.3) | 23.5 (21.3–25.7) | 22.8 (22.8–22.8) | 25.5 (25.5–25.5) |
| Race/Ethnicity | |||||||
| % White | 76.2 | 80.0 | 100.0 | 66.7 | 33.3 | 100.0 | 100.0 |
| % African-American | 9.5 | 6.7 | 0.0 | 0.0 | 66.7 | 0.0 | 0.0 |
| % Hispanic | 0.0 | 6.7 | 0.0 | 33.3 | 0.0 | 0.0 | 0.0 |
| % Other | 14.3 | 6.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Relationship to 1st recipient | |||||||
| % Related | 28.6 | 80.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| % Directed | 38.1 | 13.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| % Nondirected | 33.3 | 6.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Relationship to 2nd recipient | |||||||
| % Related | 23.8 | 60.0 | 60.0 | 100.0 | 100.0 | 100.0 | 100.0 |
| % Directed | 19.0 | 26.7 | 40.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| % Nondirected | 57.1 | 13.3 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| % NDD for both donations | 28.6 | 6.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| % Same recipient for both donations | 9.5 | 40.0 | 40.0 | 100.0 | 100.0 | 100.0 | 0.0 |
Sequential living donors had a median (IQR) 8.3 (3.1–11.5) years of follow-up for patient survival after their second donation. There was 1 reported death in a sequential donor (kidney-liver) 2.5 years after their second donation. Sequential living donors had median 382 (137–741) days of follow-up for other clinical outcomes, as captured by OPTN reporting. There were no reported intraoperative complications for sequential living multi-organ donors in our study. While follow-up data in the national registry is limited, one kidney-then-liver donor and one liver-then-kidney donor had liver-related complications following their liver graft donations. Four of 49 (8.1%) sequential living multi-organ donors were readmitted between their first donation and their 6-month follow-up, 4 donors (8.1%) were readmitted between their 6-month and 1-year follow-up visit following their first donation, and 2 donors (4%) were readmitted between their second donation and 6-month follow-up visit. Similar to national trends of missing living donor follow-up data, 17 sequential living multi-organ donors had high rates of missing follow-up data. Of the 21 donors who donated a kidney second, follow-up data was complete for 52.4% at 6 months, 33.3% at 6 and 12 months, and 4.8% at 6, 12, and 24 months. None of the 22 living donors who donated a liver lobe second had complete 6- or 12-month follow-up.
Sequential Recipient Characteristics and Outcomes
Among recipients of grafts from sequential living multi-organ donors, 57.2% were women and 76.5% were white, with a median age of (39.5) years (IQR: 32–46.5), although recipient characteristics varied by type of sequential donation (Table 2). For example, each liver-then-intestine donor donated two grafts to the same recipient. These recipients were pediatric patients between 1 and 2 years of age whose indication for liver transplantation was liver failure secondary to total parenteral nutrition (TPN) or hyperalimentation (Table 3); their indication for intestinal transplantation was gastroschisis (66.6%) or intestinal volvulus secondary to malrotation (33.3%) (Table 4). In contrast, only 9.5% of kidney-then-liver sequential living multi-organ donors donated two grafts to the same recipient. The recipients of the first graft (kidney) were 42.9% female and 71.4% white, with median age 48.6 (IQR: 30.8–53.6), whereas recipients of the second graft (liver) were 61.9% female and 86.7% white, with median age 4.3 (IQR: 0.9–44.3) (Table 2). There were 6 kidney-then-liver donors (28.6%) who donated both grafts in a nondirected manner (Table 1).
Table 2. Characteristics of recipients who received grafts from sequential living multi-organ donors.
Recipients of the second graft from a sequential donor are a heterogeneous group, both among and between types of sequential multi-organ donors. BMI could not be calculated for recipients of grafts from liver-then-intestine donors due to small body size (median age 1.2 and 1.3, respectively).
| Kidney-then-liver | Liver-then-kidney | Lung-then-kidney | Liver-then-intestine | Kidney-then-pancreas | Pancreas-then-kidney | Lung-then-liver | |
|---|---|---|---|---|---|---|---|
| First Organ | Kidney | Liver | Lung | Liver | Kidney | Pancreas | Lung |
|
| |||||||
| N (%) | 21 (42.9) | 15 (30.6) | 5 (10.2) | 3 (6.1) | 3 (6.1) | 1 (2.0) | 1 (2.0) |
| % Female | 42.9 | 73.3 | 80.0 | 33.3 | 66.7 | 100.0 | 100.0 |
| Median age, in years (IQR) | 48.6 (30.8–53.6) | 52.6 (36.7–63.3) | 18.8 (15.5–21.2) | 1.2 (1.2–1.9) | 35.7 (30.6–40.8) | 44.5 (44.5–44.5) | 16.4 (16.4–16.4) |
| Median BMI (IQR) | 25.2 (20.4–28.3) | 25.1 (22.2–28.0) | 17.3 (15.7–18.3) | NA | 24.9 (19.4–25.1) | 24.3 (24.3–24.3) | 16.9 (16.9–16.9) |
| Race/Ethnicity | |||||||
| % White | 71.4 | 86.7 | 100.0 | 66.7 | 66.7 | 100.0 | 100.0 |
| % African-American | 19.0 | 6.7 | 0.0 | 0.0 | 33.3 | 0.0 | 0.0 |
| % Hispanic | 0.0 | 6.7 | 0.0 | 33.3 | 0.0 | 0.0 | 0.0 |
| % Other | 9.5 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
|
| |||||||
| Second Organ | Liver | Kidney | Kidney | Intestine | Pancreas | Kidney | Liver |
|
| |||||||
| N (%) | 21 (42.9) | 15 (30.6) | 5 (10.2) | 3 (6.1) | 3 (6.1) | 1 (2.0) | 1 (2.0) |
| % Female | 61.9 | 46.7 | 60.0 | 33.3 | 66.7 | 100.0 | 0.0 |
| Median age, in years (IQR) | 4.3 (0.9–44.3) | 41.3 (20.8–51.6) | 38.1 (34.9–40.8) | 1.3 (1.3–2.0) | 44.8 (32.4–46.4) | 46.3 (46.3–46.3) | 24.8 (24.8–24.8) |
| Median BMI (IQR) | 26.3 (25.7–28.1) | 26.0 (24.2–27.1) | 18.7 (17.8–24.0) | NA | 24.9 (22.1–25.7) | 22.6 (22.6–22.6) | 20.9 (20.9–20.9) |
| Race/Ethnicity | |||||||
| % White | 57.1 | 86.7 | 100.0 | 66.7 | 66.7 | 100.0 | 100.0 |
| % African-American | 9.5 | 6.7 | 0.0 | 0.0 | 33.3 | 0.0 | 0.0 |
| % Hispanic | 9.5 | 6.7 | 0.0 | 33.3 | 0.0 | 0.0 | 0.0 |
| % Other | 23.8 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
Table 3.
Principal diagnosis of recipients who received the first graft donated by a sequential multi-organ donor.
| % | |
|---|---|
| Kidney from a future living liver donor (N=21) | |
| Type II diabetes | 19.0 |
| Focal glomerular sclerosis (FSG) | 14.3 |
| Polycystic kidney disease | 14.3 |
| IGA nephropathy | 9.5 |
| Type I diabetes | 9.5 |
| Alport’s syndrome | 9.5 |
| Chronic pyelonephritis/reflux nephropathy | 4.8 |
| Oxalate nephropathy | 4.8 |
| Renal cell carcinoma | 4.8 |
| Hypertensive nephrosclerosis | 4.8 |
| Chronic glomerulonephritis | 4.8 |
| Liver from a future living kidney donor (N=15) | |
| Primary sclerosing cholangitis | 20.0 |
| Acute hepatic necrosis | 13.3 |
| Cirrhosis (alcoholic) | 13.3 |
| Unknown | 13.3 |
| Cirrhosis (Hepatitis C) | 6.7 |
| Cirrhosis | 6.7 |
| Cirrhosis (steatosis) | 6.7 |
| Biliary atresia | 6.7 |
| Primary hyperoxaluria | 6.7 |
| Polycystic liver disease | 6.7 |
| Lung from a future living kidney donor (N=5) | |
| Cystic fibrosis | 80.0 |
| Obliterative bronchiolitis | 20.0 |
| Liver from a future living intestine donor (N=3) | |
| Liver disease secondary to total parenteral nutrition (TPN) or hyperalimentation | 100.0 |
| Kidney from a future living pancreas donor (N=3) | |
| Type I diabetes | 66.7 |
| Type II diabetes | 33.3 |
| Pancreas from a future living kidney donor (N=1) | |
| Type I diabetes | 100.0 |
| Lung from a future living liver donor (N=1) | |
| Cystic fibrosis | 100.0 |
Table 4.
Principal diagnosis of recipients who received the second graft donated by a sequential multi-organ donor.
| % | |
|---|---|
| Liver from previous living kidney donor (N=21) | |
| Biliary atresia | 38.1 |
| Unknown | 14.3 |
| Cirrhosis | 9.5 |
| Primary biliary cirrhosis | 9.5 |
| Primary sclerosing cholangitis | 9.5 |
| Familial cholestasis | 9.5 |
| Cirrhosis (Hepatitis C) | 4.8 |
| Cirrhosis (alcoholic) | 4.8 |
| Kidney from previous living liver donor (N=15) | |
| Hypertensive nephrosclerosis | 26.7 |
| Unknown | 20.0 |
| Polycystic kidney disease | 13.3 |
| IGA nephropathy | 6.7 |
| Hypoplasia/agenesis | 6.7 |
| Cortical necrosis | 6.7 |
| Congenital obstructive uropathy | 6.7 |
| Type I diabetes | 6.7 |
| Type II diabetes | 6.7 |
| Kidney from previous living lung donor (N=4) | |
| Chronic pyelonephritis/reflux nephropathy | 20.0 |
| Calcineurin inhibitor nephrotoxicity | 20.0 |
| Wegener’s granulomatosis | 20.0 |
| Type I diabetes | 20.0 |
| Unknown | 20.0 |
| Intestine from previous living liver donor (N=3) | |
| Gastroschisis | 66.7 |
| Intestinal volvulus secondary to malrotation | 33.0 |
| Pancreas from previous living kidney donor (N=3) | |
| Type I diabetes | 100.0 |
| Kidney from previous living pancreas donor (N=1) | |
| Type I diabetes | 100.0 |
| Liver from previous living lung donor (N=1) | |
| Cystic fibrosis | 100.0 |
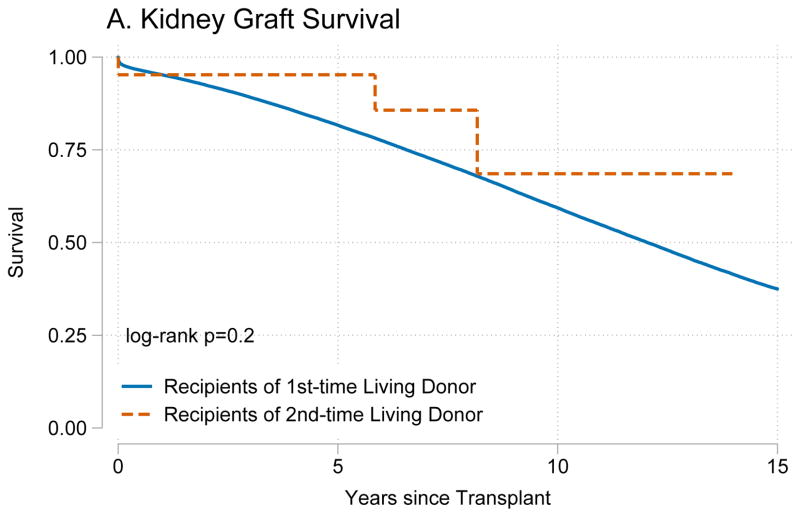
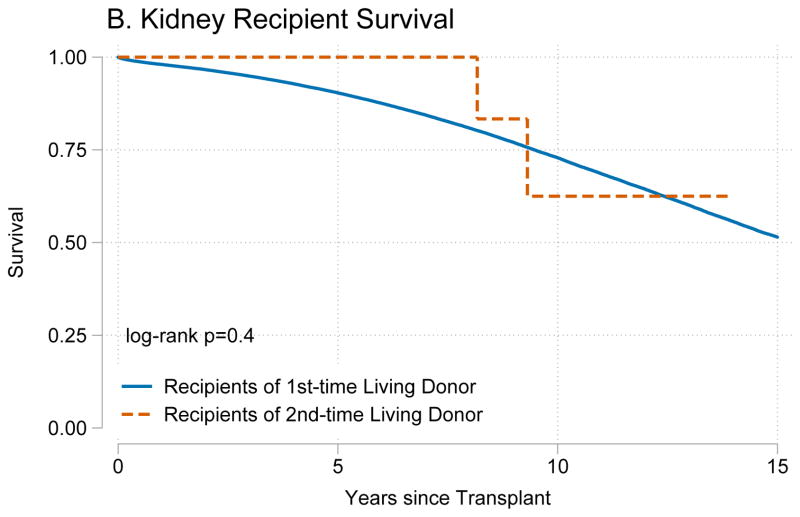
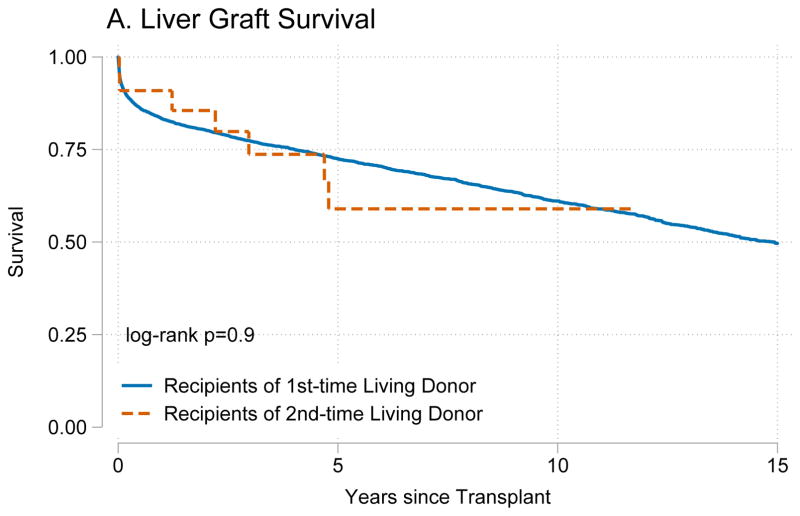
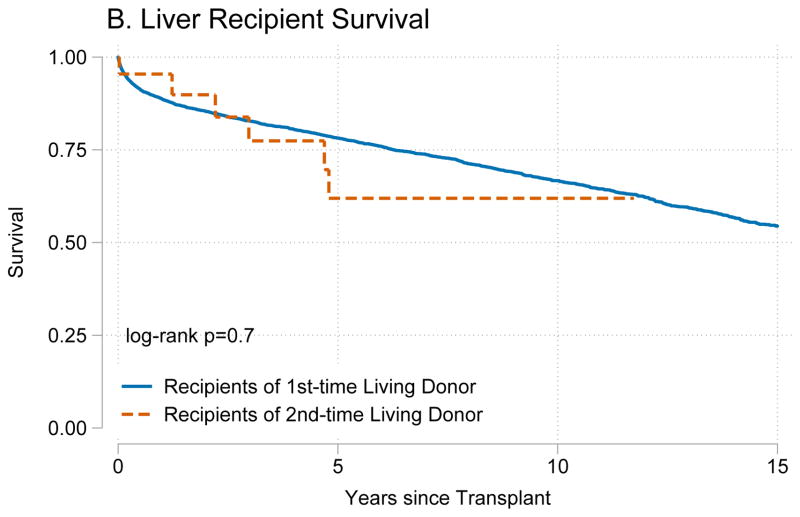
Recipients of kidneys from second-time living donors had similar graft survival (p=0.8) (Figure 1A) and patient survival (p=0.4) (Figure 1B) when compared to recipients of kidneys from first-time living donors. Similarly, recipients of livers from second-time living donors had similar graft survival (p=0.8) (Figure 2A) and patient survival (p=0.7) (Figure 2B) when compared to recipients of livers from first-time living donors.
Figure 1. Outcomes for living donor kidney transplant recipients.
Recipients of kidney graft from first and second time living donors had no differences in (A) death-censored graft failure (p=0.8) or (B) mortality (p=0.4).
Figure 2. Outcomes for living donor liver transplant recipients.
Recipients of a liver graft from first and second time living donors had no differences in (A) death-censored graft failure (p=0.8) or (B) mortality (p=0.7).
Simultaneous Multi-organ Donors
Simultaneous Donor Characteristics and Outcomes
Among the 52 living multi-organ donors undergoing simultaneous donation operations, 48 donated kidney-pancreas and 4 donated liver-intestine grafts. All simultaneous living donation procedures occurred in UNOS Region 7. Of these, kidney-segmental pancreas simultaneous multi-organ donors were 60.4% female and 79.2% white, with median age 42.4 years (IQR: 34.9–48.2; Table 5), while liver-intestine simultaneous multi-organ donors were 75.0% female and 25.0% white, with median age 25.1 (IQR: 21.0–30.3; Table 6). All 52 (100%) donated both grafts to the same recipient (Table 5). The majority (51.1%) of kidney-pancreas simultaneous living multi-organ donors were siblings of the recipient; the remainder of donor-recipient relationships included both biologically related and non-biologically related family as well as directed and nondirected donation. All liver-intestine simultaneous multi-organ donors were parents of the recipient. Only 2 transplant hospitals reported performing simultaneous multi-organ donations. One transplant hospital performed 39 kidney-segmental pancreas procedures and 1 liver-intestine procedure and the second performed 9 kidney-segmental pancreas and 3 liver-intestine procedures.
Table 5. Characteristics of living donors who underwent simultaneous organ donation.
There were 52 donors who donated 2 grafts during the same procedure. These donations only occurred at 2 centers in the US.
| Kidney-Pancreas | Liver-Intestine | |
|---|---|---|
| N (%) | 48 (92.3) | 4 (7.7) |
| % Female | 60.4 | 75.0 |
| Median age, in years (IQR) | 42.4 (34.9–48.2) | 25.1 (21.0–30.3) |
| Median BMI at 2nd donation (IQR) | 24.2 (22.6–25.9) | 26.2 (22.1–31.8) |
| Race/Ethnicity | ||
| % White | 79.2 | 25.0 |
| % African-American | 6.3 | 25.0 |
| % Hispanic | 8.3 | 50.0 |
| % Other | 4.2 | 0.0 |
| Relationship to 1st recipient | ||
| % Related | 91.7 | 100.0 |
| % Directed | 6.3 | 0.0 |
| % Nondirected | 0.0 | 0.0 |
| % Unknown | 2.1 | 0.0 |
| % Same recipient for both donations | 100.0 | 100.0 |
Table 6. Characteristics of recipients who received grafts from a living donor who underwent simultaneous donation.
Each of the 52 simultaneous donors in our study donated 2 organs to a single individual. BMI could not be calculated for recipients of liver-intestine living donations due to small body size (median age 0.8 years).
| B. Simultaneous | Kidney-Pancreas | Liver-Intestine |
|---|---|---|
| N (%) | 48 (92.3) | 4 (7.7) |
| % Female | 58.3 | 75.0 |
| Median age, in years (IQR) | 35.9 (31.3–41.6) | 0.8 (0.8–1.0) |
| Median BMI (IQR) | 23.6 (19.8–26.6) | NA |
| Race/Ethnicity | ||
| % White | 83.3 | 25.0 |
| % African-American | 6.3 | 25.0 |
| % Hispanic | 10.4 | 50.0 |
| % Other | 0.0 | 0.0 |
Simultaneous living donors had a median (IQR) 18.3 (16.2–20.6) years of follow-up for survival after their simultaneous donation. There were no simultaneous multi-organ donor deaths reported in the study period. Simultaneous donors had a median of 0 (0–194) days of follow-up for other clinical outcomes. There were no reported intraoperative or follow-up complications for simultaneous living multi-organ donors. However, 4 of 48 (8.35%) of simultaneous kidney-segmental pancreas donors were readmitted between their donation and 6-month follow-up. One of these donors was also readmitted between their 1-year and 2-year follow-up visits. Like sequential donors, simultaneous living multi-organ donors had high rates of missing follow-up data. None of the 48 kidney donors and the 4 liver donors had complete 6- or 12-month follow-up data.
Simultaneous Recipient Characteristics and Outcomes
Recipients of grafts from simultaneous kidney-segmental pancreas donors were 58.3% female and 83.3% white, with median age 35.9 years (IQR: 31.3–41.6;Table 6). Type I Diabetes was the primary diagnosis of 89.6% of kidney-segmental pancreas recipients; the remainder of kidney-segmental pancreas recipients had type II diabetes (6.3%), hypertensive nephrosclerosis (2.1%), or an unknown primary diagnosis (Table 7). Recipients of grafts from simultaneous liver-intestine donors were 75% female and 25% white, with median age 0.8 years (IQR: 0.8–1.0;Table 6). The primary diagnoses for liver-intestine recipients was liver failure secondary to TPN or hyperalimentation (75%) or unknown (25%).
Table 7.
Principal diagnosis for recipients who received grafts from simultaneous living donation.
| % | |
|---|---|
| Kidney-Pancreas (N=48) | |
| Diabetes type-I | 89.6 |
| Diabetes type-II | 6.3 |
| Hypertensive nephrosclerosis | 2.1 |
| Unknown | 2.1 |
| Liver-Intestine (N=4) | |
| Liver disease secondary to total parenteral nutrition (TPN) or hyperalimentation1 | 75.0 |
| Unknown | 25.0 |
One patient noted both secondary liver disease and intestinal volvulus secondary to malrotation as primary diagnoses (UNOS Code 6002).
Recipients of kidney-pancreas simultaneous living donor grafts had 14.5 years of median kidney graft survival. Recipients of kidneys from second-time living donors had similar graft survival (p=0.8) (Figure 1A) and patient survival (p=0.4) (Figure 1B) when compared to recipients of kidneys from first-time living donors. Similarly, recipients of livers from second-time living donors had similar graft survival (p=0.8) (Figure 2A) and patient survival (p=0.7) (Figure 2B) when compared to recipients of livers from first-time living donors.
Comparison of Select Living Multi-organ Donor Groups
Recipients of kidney-pancreas simultaneous living multi-organ donation were similar to recipients of kidney-then-pancreas and pancreas-then-kidney serial multi-organ donation in sex (p=0.5), race/ethnicity (p=0.3), age at first transplant (p=0.7), body-mass index (BMI) (p=0.9), and primary diagnosis (p=0.4). Recipients of liver-intestine simultaneous multi-organ donation were younger than recipients of liver-then-intestine serial multi-organ donation (p=0.03) but similar in sex (p=0.3), race/ethnicity (p=0.5), and primary diagnosis (p=0.2).
DISCUSSION
In this national registry study, we identified 101 living multi-organ donors and their 133 recipients between 1994 and 2017. Among sequential living donors, 38% donated 2 grafts to the same recipient and 15% donated 2 grafts as nondirected donors. Most sequential living donors donated a kidney followed by a liver segment. Simultaneous donation was limited to 2 transplant hospitals and most simultaneous donors donated a kidney and partial pancreas. Living multi-organ donors had a median 13.4 years of follow-up after their second donation and there was one reported sequential donor death 2.5 years after their second donation. There were very few reported complications for living multi-organ donors and their recipients’ outcomes were comparable to recipients of first-time living donors.
Many disease conditions requiring multi-organ transplantation are dire, notably those in the pediatric population where waitlist mortality exceeds 25%. 9 Intestinal failure followed by TPN induced liver failure is a primary cause of disease in this population, and it is common to use deceased organs in these cases. 18 In 2005, Testa et al reported the first use of living donors to treat this organ failure scenario, and other small series have documented further instances of this practice.11
In our national study, there were only 7 cases of liver-then-intestine donation, 3 sequential and 4 simultaneous. In the available literature, provider preference favors sequential donation, providing the liver segment first to correct the coagulopathy and pathology associated with liver failure, then to provide a partial small intestinal graft of ileum into the improved host environment to allow for cessation of TPN dependence and enteral feeding. 18 This logical treatment explanation does not take into consideration the risks to the donor, undergoing 2 major abdominal operations in sequence in a relatively short timeframe. It also does not allow for an appreciation of the rarity of living donor small bowel transplant itself, let alone in the multi-organ donation setting. Despite the first living donor small bowel transplant being performed 20 years ago, living donors account for <1% of small bowel transplants in the US each year 12 with only 36 documented in the literature prior to 2006.19 In the case of multiple organ donations, Testa et al share that the donor, “underwent double operative stress and was potentially exposed to the complications of 2 major operative procedures.”11 Although limited by incomplete and missing follow-up data, we found no major reported complications from the 2 operative procedures in our study.
A series of 13 patients undergoing liver-kidney sequential multi-organ donation was published, and the authors were lauded for their use of this novel technique to expand the donor pool in a country with limited access to living donation.4 More than half of the recipients in this group were pediatric, and a mean interval between surgeries was 9.6 months. This length of time between donor operations does allow for donor recovery from hepatectomy prior to undergoing nephrectomy, and as the authors argue, should not have increased risks above and beyond the risk of having each major operation separately. However, this small case series may underestimate the occurrence of infrequent complications or those that develop in the long-term as donor follow-up is not well described. We identified 15 liver-kidney sequential multi-organ transplants in the US registry, demonstrating that this is a relatively rare procedure nationwide.
Combined with the case series above and a few individual cases documented in other countries, the volume of liver-kidney sequential living multi-organ donation is insufficient to draw conclusions about donor risk. 2,7 As one author describes, “Is the ethical issue of the risks to the donor a matter of arbitrarily defining an acceptable risk?”7 While we agree that conceptualizing risk is often difficult, the transplant community has an ethical obligation to protect living donors from undue harm.20 These uncommon yet emerging procedures require improved and enhanced donor follow-up to build risk profiles prospectively as surgical science advances.
Kidney-pancreas donation comprised the most common form of simultaneous multi-organ donation, with 48 cases identified in the SRTR registry since 1994. The first living donor simultaneous pancreas-kidney transplant was reported in the US in 1994. 21,22 Much of the literature on donor outcomes after living pancreas-kidney donation has focused on short-term perioperative complications, rather than long-term complications. Consistent with our findings, no cases of perioperative death have been reported in available literature. 21,23 Significant perioperative complications related to pancreatectomy, such as pancreatitis, abscess, or fistula, have been reported in less than 5% of living donors in case series, while reoperation and splenectomy due to bleeding, ischemia, or abscess have been noted in 5% to up to 20%. 23–27 Data on long-term outcomes are limited, but a recent study of 45 living pancreas donors that included 69% simultaneous kidney donations found that over a mean postdonation follow-up period of 16.3 years, 26.7% filled prescriptions for diabetes treatments, compared with 5.9% of kidney-alone living donors (odds ratio 4.13, 95% confidence interval 1.91–8.93; P = 0.0003). 28 These findings suggest a more than 4-fold increase in the incidence of diabetes following living kidney-pancreas donation, a concern that warrants longer follow-up and investigation to adequately understand risks to the donor.
Our study was limited by the small sample size available in the SRTR database, which impacted our ability to measure survival postdonation. Additionally, for certain subgroups, only 2 centers nationally perform these multi-organ donation procedures, making it difficult to draw generalizable inferences. We found follow-up data on living donors to be minimal up to the required 2 years, and even sparser thereafter, which is similar to national trends.17 Particularly for living multi-organ donors who undergo 2 complex surgical procedures, the standardization of long-term follow-up nationwide would help to collect the data necessary to better describe donor risk.
We found that the donation of multiple solid organs from the same living donor is a rare practice in the U.S with only 101 cases over the past two decades. Careful documentation and postdonation follow-up of these living donors is needed to describe donor risk, to inform appropriate informed consent, and to optimize postdonation care for this very unique community of living donors.
Supplementary Material
Acknowledgments
Funding: Funding for this study was provided by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) grant numbers K01DK114388-01 (PI: Henderson), F32DK105600 (PI: DiBrito), 4R01DK096008-04 (PI: Segev), 5K01DK101677-02 (PI: Massie), and 5K24DK101828-03 (PI: Segev), 1F32DK109662-01 (PI: Holscher) and by the Agency for Healthcare Research and Quality (AHRQ) grant number K01HS024600 (PI: Purnell).
The data reported here have been supplied by the Minneapolis Medical Research Foundation (MMRF) as the contractor for the Scientific Registry of Transplant Recipients (SRTR). The interpretation and reporting of these data are the responsibility of the authors and in no way should be seen as an official policy of or interpretation by the SRTR, UNOS/OPTN, or the US Government.
Abbreviations
- BMI
body mass index
- HR
Hazard Ratio
- HRSA
Health Resources and Services Administration
- OPTN
Organ Procurement and Transplantation Network
- SRTR
Scientific Registry of Transplant Recipient
- UNOS
United Network for Organ Sharing
Footnotes
AUTHORSHIP
M.L.H. participated in research design, writing of the paper, and performance of the research.
S.D.R. participated in research design, writing of the paper, and performance of the research.
A.G.T. participated in research design, writing of the paper, and data analysis.
C.M.H. participated in performance of the research and writing of the paper.
A.A.S. participated in data analysis and writing of the paper.
M.G.B. participated in data analysis and writing of the paper.
T.S.P. participated in the writing of the paper and performance of the research.
A.B.M. participated in the research design and data analysis.
J.G.W. participated in the research design and writing of the paper.
M.M.W. participated in writing of the paper.
K.L.L. participated in writing of the paper and performance of the research.
D.L.S. participated in the performance of the research and oversaw the project.
Disclosures: The authors have no conflicts of interest to disclose.
References
- 1.Hart A, Smith J, Skeans M. OPTN/SRTR annual data report 2014. Am J Transplant. 2016;16(S2):11–46. [Google Scholar]
- 2.Astarcioglu I, Karademir S, Gülay H, et al. Primary hyperoxaluria: simultaneous combined liver and kidney transplantation from a living related donor. Liver transplantation. 2003;9(4):433–436. doi: 10.1053/jlts.2003.50072. [DOI] [PubMed] [Google Scholar]
- 3.Date H, Aoyama A, Hijiya K, et al. Outcomes of various transplant procedures (single, sparing, inverted) in living-donor lobar lung transplantation. J Thorac Cardiovasc Surg. 2017;153(2):479–486. doi: 10.1016/j.jtcvs.2016.10.017. [DOI] [PubMed] [Google Scholar]
- 4.Goldaracena N, Selzner N, Selzner M. Living donation to the extreme: Saving a life not once, but twice. Liver Transpl. 2017;23(3):288–289. doi: 10.1002/lt.24718. [DOI] [PubMed] [Google Scholar]
- 5.Humar A, Gruessner RW, Sutherland DE. Living related donor pancreas and pancreas-kidney transplantation. British medical bulletin. 1997;53(4):879–891. doi: 10.1093/oxfordjournals.bmb.a011656. [DOI] [PubMed] [Google Scholar]
- 6.Kim J, Zimmerman MA. Technical aspects for live-donor organ procurement for liver, kidney, pancreas, and intestine. Current opinion in organ transplantation. 2015;20(2):133–139. doi: 10.1097/MOT.0000000000000181. [DOI] [PubMed] [Google Scholar]
- 7.Marujo W, Barros M, Cury R, Pacheco-Silva A, Sette H. Successful combined kidney-liver right lobe transplant from a living donor. The Lancet. 1999;353(9153):641. doi: 10.1016/s0140-6736(98)05658-x. [DOI] [PubMed] [Google Scholar]
- 8.Pacheco-Moreira L, Balbi E, Enne M, et al. One living donor and two donations: Sequential kidney and liver donation with 20-years interval. Trans proc. 2005;37:4337–4338. doi: 10.1016/j.transproceed.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 9.Raofi V, Beatty E, Testa G, et al. Combined living-related segmental liver and bowel transplantation for megacystis-microcolon-intestinal hypoperistalsis syndrome. Journal of pediatric surgery. 2008;43(2):e9–e11. doi: 10.1016/j.jpedsurg.2007.09.073. [DOI] [PubMed] [Google Scholar]
- 10.Tan M, Kandaswamy R, Sutherland DE, Gruessner RW. Laparoscopic donor distal pancreatectomy for living donor pancreas and pancreas–kidney transplantation. American journal of transplantation. 2005;5(8):1966–1970. doi: 10.1111/j.1600-6143.2005.00950.x. [DOI] [PubMed] [Google Scholar]
- 11.Testa G, Holterman M, John E, Kecskes S, Abcarian H, Benedetti E. Combined living donor liver/small bowel transplantation. Transplantation. 2005;79(10):1401–1404. doi: 10.1097/01.tp.0000157280.75015.14. [DOI] [PubMed] [Google Scholar]
- 12.Testa G, Panaro F, Schena S, Holterman M, Abcarian H, Benedetti E. Living related small bowel transplantation: donor surgical technique. Annals of surgery. 2004;240(5):779–784. doi: 10.1097/01.sla.0000143266.59408.d7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zielinski A, Nazarewski S, Bogetti D, et al. Simultaneous pancreas-kidney transplant from living related donor: a single-center experience. Transplantation. 2003;76(3):547–552. doi: 10.1097/01.TP.0000076624.79720.14. [DOI] [PubMed] [Google Scholar]
- 14.Henderson ML, Gross JA. Living Organ Donation and Informed Consent in the United States: Strategies to Improve the Process. The Journal of Law, Medicine & Ethics. 2017;45(1):66–76. doi: 10.1177/1073110517703101. [DOI] [PubMed] [Google Scholar]
- 15.Massie AB, Kucirka LM, Segev DL. Big data in organ transplantation: registries and administrative claims. Am J Transplant. 2014;14(8):1723–1730. doi: 10.1111/ajt.12777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Louis TA, Zeger SL. Effective communication of standard errors and confidence intervals. Biostatistics. 2009;10(1):1–2. doi: 10.1093/biostatistics/kxn014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Henderson ML, Thomas AG, Shaffer A, et al. The National Landscape of Living Kidney Donor Follow-up in the United States. American Journal of Transplantation. 2017 doi: 10.1111/ajt.14356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gangemi A, Tzvetanov IG, Beatty E, et al. Lessons learned in pediatric small bowel and liver transplantation from living-related donors. Transplantation. 2009;87(7):1027–1030. doi: 10.1097/TP.0b013e31819cc3bf. [DOI] [PubMed] [Google Scholar]
- 19.Benedetti E, Holterman M, Asolati M, et al. Living related segmental bowel transplantation: from experimental to standardized procedure. Annals of surgery. 2006;244(5):694–699. doi: 10.1097/01.sla.0000232555.02880.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abecassis M, Adams M, Adams P, et al. Consensus statement on the live organ donor. JAMA. 2000;284(22):2919. doi: 10.1001/jama.284.22.2919. [DOI] [PubMed] [Google Scholar]
- 21.Gruessner R, Kendall DM, Drangstveit MB, Gruessner AC, Sutherland D. Simultaneous pancreas-kidney transplantation from live donors. Annals of surgery. 1997;226(4):471. doi: 10.1097/00000658-199710000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gruessner R, Leone J, Sutherland D. Combined kidney and pancreas transplants from living donors. Paper presented at: Transplantation proceedings; 1998. [DOI] [PubMed] [Google Scholar]
- 23.Reynoso JF, Gruessner CE, Sutherland DE, Gruessner RW. Short-and long-term outcome for living pancreas donors. Journal of hepatobiliary-pancreatic sciences. 2010;17(2):92–96. doi: 10.1007/s00534-009-0147-x. [DOI] [PubMed] [Google Scholar]
- 24.Choi JY, Jung JH, Kwon H, Shin S, Kim YH, Han DJ. Pancreas transplantation from living donors: a single center experience of 20 cases. American Journal of Transplantation. 2016;16(8):2413–2420. doi: 10.1111/ajt.13738. [DOI] [PubMed] [Google Scholar]
- 25.Kenmochi T, Asano T, Maruyama M, et al. Living donor pancreas transplantation in Japan. Journal of hepatobiliary-pancreatic sciences. 2010;17(2):101–107. doi: 10.1007/s00534-009-0148-9. [DOI] [PubMed] [Google Scholar]
- 26.Kirchner VA, Finger EB, Bellin MD, et al. Long-term Outcomes for Living Pancreas Donors in the Modern Era. Transplantation. 2016;100(6):1322–1328. doi: 10.1097/TP.0000000000001250. [DOI] [PubMed] [Google Scholar]
- 27.Sutherland DE, Radosevich D, Gruessner R, Gruessner A, Kandaswamy R. Pushing the envelope: living donor pancreas transplantation. Current opinion in organ transplantation. 2012;17(1):106–115. doi: 10.1097/MOT.0b013e32834ee6e5. [DOI] [PubMed] [Google Scholar]
- 28.Lam NN, Schnitzler MA, Segev DL, et al. Diabetes Mellitus in Living Pancreas Donors: Use of Integrated National Registry and Pharmacy Claims Data to Characterize Donation-Related Health Outcomes. Transplantation. 2017;101(6):1276–1281. doi: 10.1097/TP.0000000000001375. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.