Abstract
In the present study, we examined whether substance P (SP) and SP methyl ester (SPME), a selective NK1 agonist, cause biphasic responses consisting of endothelium-dependent relaxation (EDR) and contraction (EDC) in precontracted rabbit intrapulmonary arteries.
In arteries contracted with PGF2α (2×10−6 M), SP as well as SPME caused only EDR at low concentration (10−9 M) and EDR followed by EDC at higher concentrations, indicating the involvement of NK1 receptors. The SP (10−8 M)-induced EDR was abolished in arteries moderately contracted by PGF2α (5×10−7 M) and the EDC in arteries maximally contracted by PGF2α (10−5 M), indicating that EDR and EDC are inversely dependent on preexisting tone.
Indomethacin (10−8–10−6 M), a cyclo-oxygenase inhibitor, and ozagrel (10−8–10−6 M), a TXA2 synthetase inhibitor attenuated the EDC in the SPME (10−7 M)-induced biphasic response and markedly potentiated the EDR. AA-861 (10−8–10−6 M), a 5-lipoxygenase inhibitor, did not affect the EDR or EDC. L-NG-nitro-arginine methyl ester (10−5–10−4 M), a nitric oxide synthase inhibitor, attenuated the EDR and slightly potentiated the EDC.
CP-99994 (10−10–10−8 M), an NK1 antagonist, attenuated the EDC and potentiated the EDR in the SPME (10−7 M)-induced biphasic response, while the NK2 antagonist SR-48968 (10−9–10−7 M) had no effect. CP-99994 attenuated the SPME (10−7 M)-induced EDC under EDR-blockade to a greater extent than the EDR under EDC-blockade, indicating that CP-99994 enhanced the EDR component by preferential inhibition of the EDC component.
In conclusion, NK1 agonists caused a biphasic endothelium-dependent response (EDR and EDC) in submaximally precontracted intrapulmonary arteries. The EDC and EDR mediated by NK1 receptors may play physiological and/or pathophysiological roles in modulation of vascular tone.
Keywords: NK1 receptor, endothelium-dependent contraction, endothelium-dependent relaxation, intrapulmonary artery, nitric oxide, substance P, substance P methyl ester, thromboxane A2
Introduction
The pulmonary circulation is regulated by various neuronal, hormonal and humoral factors including neuropeptides (Barnes & Liu, 1995). The pulmonary arteries are innervated by sensory nerve containing substance P (SP), neurokinin A (NKA) and CGRP (Allen et al., 1989). Treatment with the C-fibre activator capsaicin and electrical stimulation of vagal nerves results in release of SP and NKA in the perfused guinea-pig lung (Saria et al., 1988). SP is also localized in endothelial cells and is released by hypoxia and changes in perfusion flow (Milner et al., 1989; Ralevic et al., 1990). Tachykinin NK1 receptors have been shown to locate on the vascular endothelium (Greeno et al., 1993; Bowden et al., 1996). SP causes only endothelium-dependent relaxation (EDR) via nitric oxide (NO) production in precontracted preparations of guinea-pig and rabbit pulmonary arteries via activation of NK1 receptors (D'Orleans-Just et al., 1986; Emonds-Alt et al., 1993; Floch et al., 1994). In non-contracted preparations of rabbit intrapulmonary arteries, SP evokes endothelium-dependent contraction (EDC) via activation of NK1 receptors and TXA2 production at low concentrations (Shirahase et al., 1995) and endothelium-independent contraction (EIC) via NK2 receptors at higher concentrations (D'Orleans-Just et al., 1986; Shirahase et al., 1995). Neuronal and/or endothelial SP may modulate vascular tone by causing EDR and EDC. However, there have been few reports on the simultaneous occurrence of EDC and EDR under active tone in pulmonary arteries or on their pharmacological nature. In the present study, we demonstrated that SP and SP methyl ester (SPME), a selective NK1 agonist, induce only EDR at low concentrations and a biphasic endothelium-dependent response (EDR followed by EDC) at higher concentrations in isolated submaximally precontracted rabbit intrapulmonary arteries. We also pharmacologically characterized the EDR and EDC components in the biphasic endothelium-dependent response.
Methods
Male Japanese white rabbits (2–3 kg) (Oriental Bio Service, Kyoto, Japan) were fed regular chow (CR-3, Clea Japan, Osaka, Japan) and allowed access to tap water ad libitum. Animals were anaesthetized with sodium pentobarbital (25 mg kg−1, i.v.) and exsanguinated from the common carotid artery. The thoracic cavity was opened and the lungs were excised and placed in aerated Krebs-Henseleit solution of the following composition (mM): NaCl, 120; KCl, 4.7; MgSO4, 1.2; KH2PO4, 1.2; CaCl2, 2.5; NaHCO3, 25; and glucose, 10. The peripheral portions of the intrapulmonary arteries (diameter 0.3–1.0 mm) were isolated from the lungs and carefully cleaned of lung parenchymal, fat and connective tissue. The arteries were helically cut and the strips were fixed vertically between hooks in a 10 ml organ bath containing a nutrient solution maintained at 37±0.5°C and bubbled with a mixture of 95% O2 and 5% CO2. The pH of the solution was 7.4. The end of each strip was attached to the lever of a force-displacement transducer (NEC San-Ei Instrument Co. Ltd, Tokyo, Japan) connected to an ink-writing oscillograph (NEC San-Ei Instrument Co. Ltd and isometric changes in tension were recorded. The applied tension was adjusted to 0.5 g. Each strip was allowed to equilibrate for 1 h, during which time the nutrient solution was changed every 10 min and the applied tension was readjusted. In several experiments, the intact and endothelium-removed strips were prepared from the same artery. The functional integrity of the endothelium in the intact preparations was checked with acetylcholine (ACh), which causes EDR in the presence of active tone (Altiere et al., 1986). The endothelium was removed by intimal rubbing. The rubbed preparations showed no ACh-induced relaxation. The elimination of endothelium was verified morphologically by scanning electron microscopy as described previously (Shirahase et al., 1987).
In the first set of experiments, SP (10−10–10−7 M) and SPME (10−10–10−6 M) were applied non-cumulatively to the arteries with and without endothelium contracted by PGF2α at 2×10−6 M. Relaxation and contraction were expressed as changes in tension from the plateau level of PGF2α-induced contraction. SPME (10−10–10−7 M) was also applied under EDC- and EDR-blockade. To eliminate EDC and EIC, ozagrel (10−5 M), a TXA2 synthetase inhibitor (Iizuka et al., 1981), and SR-48968 (10−7 M), an NK2 antagonist (Emonds-Alt et al., 1992) were pretreated before application of SPME. Ozagrel (10−5 M) and SR-48968 (10−7 M) abolishes the substance P-induced EDC and EIC, respectively, in the isolated rabbit intrapulmonary artery (Shirahase et al., 1995). To eliminate EDR and EIC, L-NAME (10−4 M), a NO synthase inhibitor, and SR-48968 (10−7 M) were pretreated before application of SPME. L-NAME (10−4 M) abolishes the substance P-induced EDR in the isolated rabbit intrapulmonary artery (Shirahase et al., 1997). Then, concentration-response curves of SPME for EDR and EDC were constructed. To examine the effects of magnitude of preexisting tonus, SP (10−8 M) was applied to the intrapulmonary arteries with endothelium contracted by PGF2α at 5×10−7, 2×10−6 and 10−5 M.
In the second set of experiments, the effects of various enzyme inhibitors and receptor antagonists were examined in arteries contracted with PGF2α at 2×10−6 M. Enzyme inhibitors and receptor antagonists were applied 5 min prior to the administration of PGF2α and 20 min prior to the administration of SPME (10−7 M). The effects of CP-99994 (10−10–10−8 M), an NK1 receptor antagonist (Desai et al., 1992), on the SPME-induced EDR in the presence of ozagrel and SR-48968, and on the EDC in the presence of L-NAME and SR-48968 were also examined.
SP (Peptide Institute, Osaka, Japan), SPME (Peptide Institute), indomethacin (Wako Pure Chemical Industries, Ltd, Osaka, Japan), L-NAME (Wako Pure Chemical Industries, Ltd, Osaka, Japan), PGF2α (Cayman Chemical Company, Ann Arbor, MI, U.S.A.), sodium pentobarbitone (Tokyo Kasei Kogyo, Co., Ltd, Tokyo, Japan) and ACh (Daiichi Pharmaceutical Co., Ltd, Tokyo, Japan) were purchased from the sources indicated. Ozagrel hydrochloride (Ono Pharmaceutical Co. Ltd, Osaka, Japan), (+)−(2s,3s) - 3 - (2 - methoxybenzylamino)-2-phenylpiperidine (CP-99994) (Pfizer Inc., Groton, CT, U.S.A.), (s)-N-methyl-N-[4-(4-acetylamino-4-phenylpiperidino)-2-(3,4-dichlorophenyl)butyl] benzamide (SR-48968) (Sanofi Recherche, Cedex, France) and 2-(12-hydroxy-5,10-dodecadiynyl)-3,5,6-trimethyl-1,4-benzoquinone (AA-861) (Takeda Chemical Industries, Ltd, Osaka, Japan) were gifts from the sources indicated. SP, SPME, ozagrel hydrochloride, CP-99994, ACh, sodium pentobarbital and L-NAME were dissolved in distilled water, PGF2α in ethanol, and AA-861 and SR-48968 in dimethylsulphoxide.
Data are expressed as means±s.e.mean. The statistical significance of differences was analysed by Student's t-test for paired data. A P value less than 0.05 was considered significant.
Results
Responses to SP and SPME in endothelium-intact and removed intrapulmonary artery
SP (10−10–10−7 M) and SPME (10−10–10−6 M) were non-cumulatively applied to the endothelium-intact and -removed strips contracted by PGF2α (2×10−6 M). SP and SPME caused only relaxation at 10−9 M and biphasic responses consisting of relaxation followed by contraction at concentrations of 10−8 M and higher in the endothelium-intact strips (Figure 1). These responses were abolished in endothelium-removed strips with the exception of SP (10−7 M), in which partial contraction remained (EIC). Mean values of EDR and EDC induced by SP and SPME are shown in Figure 2.
Figure 1.
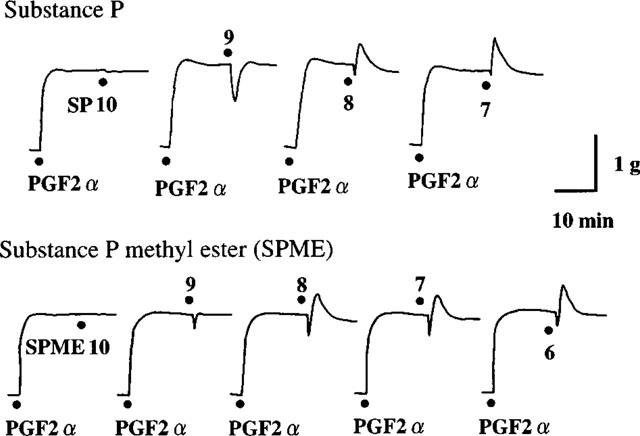
Representative tracings of responses induced by substance P (SP) and substance P methyl ester (SPME) in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). Figures with dots show concentrations of peptides (−log M).
Figure 2.
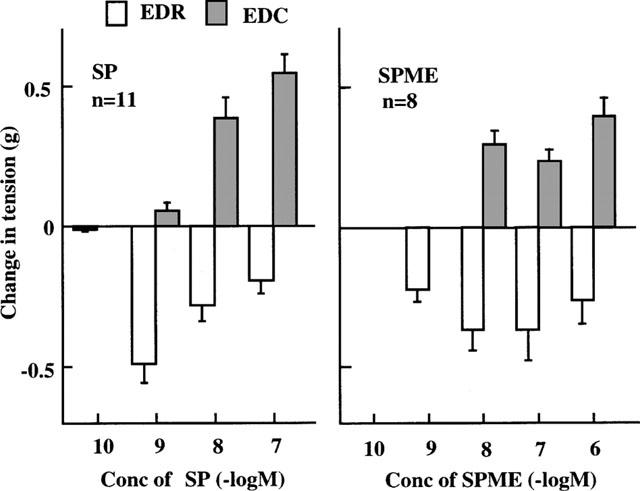
Endothelium-dependent relaxation (EDR) and contraction (EDC) induced by SP and substance P methyl ester (SPME) in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). Data are means±s.e.mean.
EDR and EDC may counteract each other in the biphasic response. To observe the concentration-response relationship for EDR and EDC without this counteraction, SPME (10−10–10−7 M) was applied to strips pretreated with ozagrel (10−5 M) and SR-48968 (10−7 M), or with L-NAME (10−4 M) and SR-48968 (10−7 M), respectively. SPME-induced EDR reached the maximal level at 10−8 M, while EDC did not reach this level even at 10−7 M (Figure 3).
Figure 3.
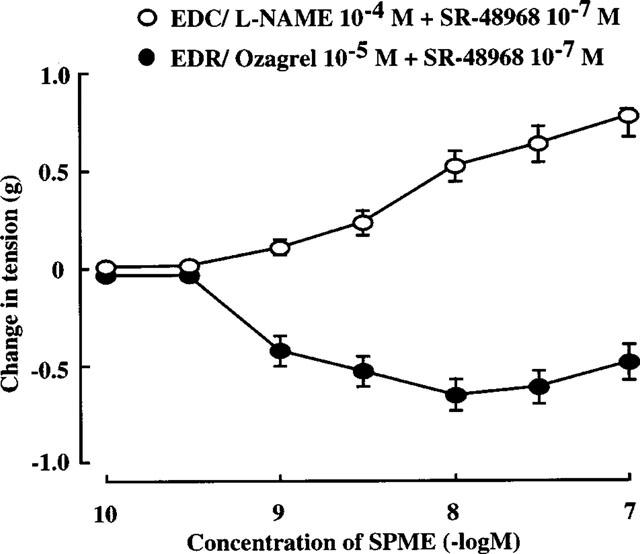
Concentration-response curves of SPME (10−10–10−7 M) for EDR under EDC-blockade and for EDC under EDR-blockade in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). Data are means±s.e.mean (n=9) EDC: SPME was applied in the presence of L-NAME (10−4 M) and SR-48968 (10−7 M), which eliminate EDR and EIC, respectively. EDR: SPME was applied in the presence of ozagrel (10−5 M) and SR-48968 (10−7 M), which eliminate EDC and EIC, respectively.
To examine the effect of magnitude of preexisting tonus, SP (10−8 M) was applied to the endothelium-intact strips contracted by 5×10−7, 2×10−6 and 10−5 M PGF2α, for which the precontraction levels were 0.9±0.1, 1.9±0.1 and 2.5±0.2 g (mean±s.e.mean, n=5), respectively. SP induced biphasic response consisting of EDR (0.5±0.1 g) followed by EDC (0.4±0.1 g) in the arteries contracted by PGF2α at 2×10−6 M. The EDC decreased to 0.1±0.02 g and the EDR increased to 0.7±0.1 g in the arteries contracted by PGF2α at 10−5 M, while the EDC increased to 1.0±0.2 g and the EDR decreased to 0.2±0.1 g in the arteries contracted at 5×10−7 M, indicating that EDR increased and EDC decreased depending on the magnitude of precontraction.
Effects of inhibitors of arachidonic acid metabolism and NO synthesis on the biphasic endothelium-dependent response
The effects of various inhibitors on biphasic endothelium-dependent responses induced by SPME (10−7 M) were examined. The EDC component was attenuated in a concentration-dependent manner by indomethacin (10−8–10−6 M), a cyclo-oxygenase inhibitor (Vane, 1971) and ozagrel (10−8–10−6 M), a TXA2 synthetase inhibitor, resulting in concentration-dependent enhancement of the EDR component (Figure 4). AA-861 (10−8–10−6 M), a specific 5-lipoxygenase inhibitor (Yoshimoto et al., 1982), affected neither EDC nor EDR (Figure 4). The EDR component was attenuated in a concentration-dependent manner by L-NAME (10−5–10−4 M), an inhibitor of NO synthase, resulting in slight enhancement of the EDC component (Figure 4).
Figure 4.
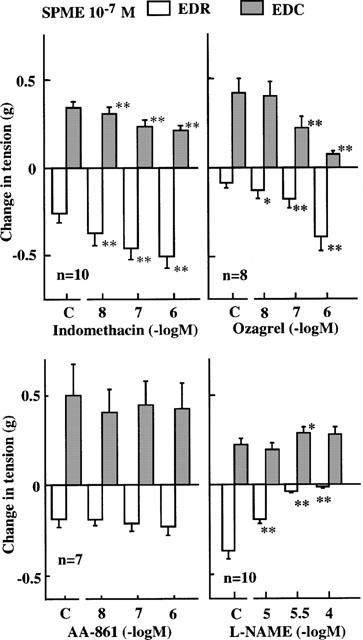
Effects of indomethacin (cyclo-oxygenase inhibitor), ozagrel (TXA2 synthetase inhibitor), AA-861 (5-lipoxygenase inhibitor) and L-NAME (nitric oxide synthase inhibitor) on the EDC and EDR component in the biphasic endothelium-dependent response induced by SPME (10−7 M) in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). C=control. Data are means±s.e.mean. *P<0.05, **P<0.01, Student's t-test for paired data.
Effects of tachykinin NK1 and NK2 receptor antagonists on the biphasic endothelium-dependent response
The effects of NK1 and NK2 antagonists on the biphasic endothelium-dependent response induced by SPME (10−7 M) were examined. CP-99994 (10−10–10−8 M), a tachykinin NK1 receptor antagonist, concentration-dependently attenuated the EDC component but enhanced the EDR component in the biphasic response induced by SPME (10−7 M) (Figure 5). SR-48968 (10−9–10−7 M), an NK2 antagonist, had no effect on the EDC or EDR component (Figure 5). SPME at 10−9 M caused only EDR as shown in Figure 2, which was attenuated by CP-99994 (10−9–10−7 M) in a concentration-dependent manner (data not shown).
Figure 5.
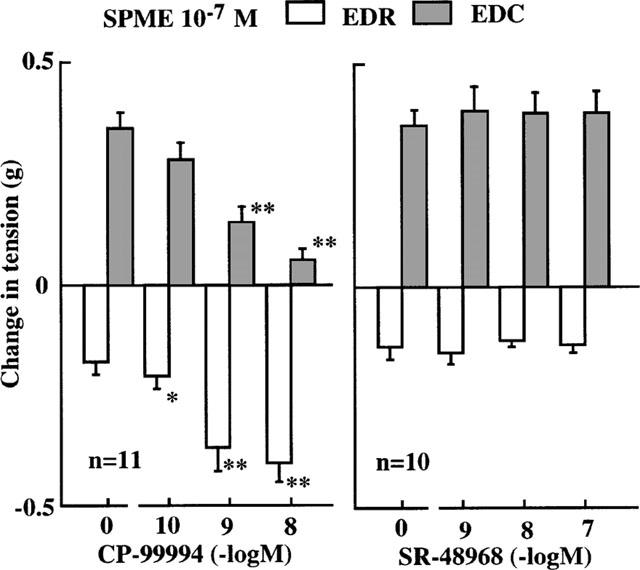
Effects of CP-99994 (tachykinin NK1 receptor antagonist) and SR-48968 (NK2 receptor antagonist) on the EDC and EDR component in the biphasic endothelium-dependent response induced by SPME (10−7 M) in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). C=control. Data are means±s.e.mean. *P<0.05, **P<0.01, student's t-test for paired data.
In strips pretreated with ozagrel (10−5 M) and SR-48968 (10−7 M) to eliminate EDC and EIC components, SPME (10−7 M) evoked only EDR, which was attenuated by CP-99994 (10−7 M) (Figure 6). In strips pretreated with L-NAME (10−4 M) and SR-48968 (10−7 M) to eliminate EDR and EIC components, SPME (10−7 M) evoked only EDC, which was concentration-dependently attenuated by CP-99994 (10−10–10−7 M) (Figure 6). These results showed that the EDC component was more sensitive to NK1 antagonist than the EDR component in the biphasic response induced by SPME (10−7 M).
Figure 6.
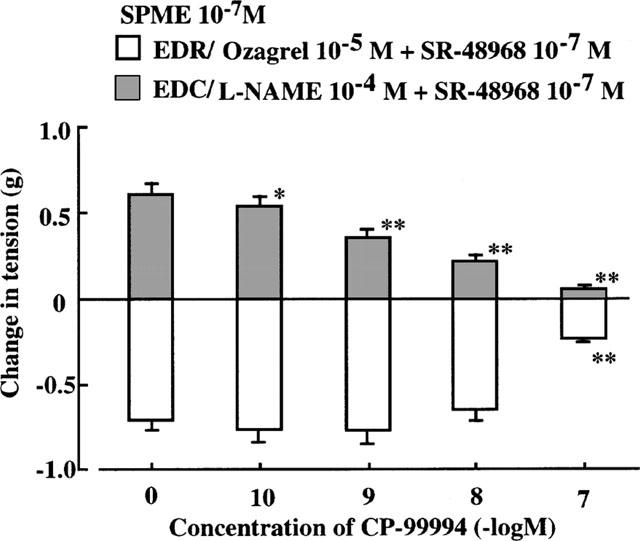
Effects of CP-99994 (tachykinin NK1 receptor antagonist) on the EDR component induced by SPME (10−7 M) under EDC blockade and the EDC component under EDR blockade in endothelium-intact rabbit intrapulmonary arteries precontracted with PGF2α (2×10−6 M). EDR: ozagrel (10−5 M) and SR-48968 (10−7 M) were applied to eliminate EDC and EIC, respectively. EDC: L-NAME (10−4 M) and SR-48968 (10−7 M) were applied to eliminate EDR and EIC, respectively. C=control. Data are mean±s.e.mean (n=11). *P<0.05, **P<0.01, Student's t-test for paired data.
Discussion
SP causes EDR in various peripheral arteries including rabbit pulmonary arteries via NO production in the presence of active tone (Emonds-Alt et al., 1993). We reported previously that SP causes EDC via production of TXA2 in the non-contracted rabbit pulmonary artery (Shirahase et al., 1995). However, there have been few reports on SP-induced EDR and EDC in the same pulmonary arterial preparations. In the present study, we found that SP and SPME, a selective NK1 agonist, caused only EDR at low concentrations and biphasic endothelium-dependent responses (EDR followed by EDC) at concentrations of 10−8 M and higher in the precontracted rabbit intrapulmonary arteries, and that SP (10−8 M)-induced EDC decreased and EDR increased depending on the magnitude of precontraction.
EDR appeared at lower concentrations of SP and SPME in comparison with EDC (Figure 2). EDR did not increase in a concentration-dependent manner since the following EDC counteracted EDR at higher concentrations of SP and SPME. In separate experiments (Figure 3), concentration-response curves of SPME for EDC and EDR were independently constructed using ozagrel to eliminate EDC and L-NAME to eliminate EDR, respectively. The EDR was about 10 fold more sensitive to SPME than the EDC. We speculated that when endothelial cells are exposed to endogenous NK1 agonists, the EDR pathway is first activated at low concentrations and then the EDC pathway is driven at higher concentrations to counteract the EDR as an auto-regulatory mechanism. Although the precise mechanism by which EDR was more sensitive to NK1 activation than EDC is not clear, the nature of endothelial NK1 receptors and/or their signalling process involved in EDC and EDR are considered to be different. The guinea-pig bronchi have been reported to contain unusual septide-selective NK1 receptors (Zeng & Burcher, 1994). Alternatively, sensitivity to second messengers after activation of NK1 receptors may be different between EDC and EDR pathways. NO is produced from arginine by Ca2+-dependent eNOS and TXA2 from arachidonic acid liberated by Ca2+-dependent phospholipase A2. Stimulation of NK1 receptors leads to activation of phospholipase C and to accumulation of IP3, resulting in an increase in intracellular Ca2+ level. eNOS may be activated by lower concentrations of intracellular Ca2+ than phospholipase A2. Similarly to NK1 agonists, Ca2+ ionophores such as A-23187 and ionomycin caused only EDR at low concentrations and a biphasic endothelium-dependent response (EDR followed by EDC) at higher concentrations (unpublished data).
The mechanism by which EDR preceded EDC in the biphasic response also remains to be clarified. Production and/or action of NO are considered to be more rapid than those of TXA2 after stimulation by SP or SPME. The EDC component in the biphasic endothelium-dependent response was decreased and the EDR component increased depending on the magnitude of active tone, suggesting a role of EDC or EDR in auto-regulation of vascular tone.
We reported previously that SP-induced EDC is mediated by production of TXA2 in non-contracted pulmonary arteries (Shirahase et al., 1995). Indeed, cyclo-oxygenase and TXA2 synthetase inhibitors showed concentration-dependent attenuation of the EDC component accompanied by enhancement of the EDR component, indicating that EDR was partially masked by the following EDC in the biphasic endothelium-dependent response. We have also shown that EDC induced by various agonists is mediated by TXA2 in canine cerebral arteries (Shirahase et al., 1987; 1988a,1988b; 1991; Kurahashi et al., 1994). L-NAME concentration-dependently inhibited EDR and slightly enhanced EDC, indicating that NO had little effect on the maximal level of the following EDC. AA-861 affected neither EDR nor EDC, indicating that 5-lipoxygenase metabolites were not involved in the biphasic endothelium-dependent response.
There are three types of tachykinin receptors, NK1, NK2 and NK3. In the pulmonary artery, EDC and EIC are mediated by NK1 and NK2 receptors, respectively (Shirahase et al., 1995). EDR induced by SP is mediated by NK1 receptors in guinea-pig and rabbit pulmonary arteries (Emonds-Alt et al., 1993; Floch et al., 1994). We demonstrated that SPME, a selective NK1 agonist induced only EDR at a low concentration (10−9 M), which was effectively blocked by the selective NK1 antagonist CP-99994. However, CP-99994 showed concentration-dependent attenuation of the EDC component accompanied by enhancement of the EDR component in the biphasic response induced by SPME at 10−7 M. This concentration of SPME was shown to be submaximal for EDC and supramaximal for EDR in the concentration-response curves (Figure 3). Therefore, CP-99994 is considered to inhibit the EDC more effectively than the EDR component, resulting in apparent enhancement of the EDR which had been partially masked by the EDC. Indeed, EDR under EDC-blockade was not enhanced and was attenuated by CP-99994 (Figure 6). The EDC under EDR-blockade was shown to be much more effectively inhibited by the NK1 antagonist than the EDR under EDC-blockade.
The balance between EDC and EDR is important for homeostasis of vascular tone and is impaired in various diseases showing circulatory failure. Indeed, SP-induced EDR is impaired in pulmonary hypertension (Uren et al., 1992; Brett et al., 1996). Thus, inhibitors of the cyclo-oxygenase-TXA2 synthetase pathway are considered to dilate blood vessels by inhibition of EDC and enhancement of EDR in pulmonary hypertension. NK1 antagonists also dilate pulmonary arteries exposed to high concentrations of tachykinins by preferential inhibiton of NK1-mediated EDC accompanied by apparent enhancement of EDR. On the other hand, tachykinins are involved in pulmonary inflammation such as asthma, and cause vasodilation and increases in vascular permeability through activation of NK1 receptors and NO production (Kageyama et al., 1997). TXA2 may be simultaneously produced by tachykinins to counteract NO in an auto-regulatory mechanism. Recently, a number of NK1 antagonists have been synthesized, but none of these have been successfully developed as anti-inflammatory drugs. Under high concentrations of tachykinins, NK1 antagonists may potentiate the proinflammatory effect of NO by preferential inhibition of the EDC (TXA2) pathway. Further studies are needed to clarify the pathophysiological role of tachykinin-induced EDR and EDC and therapeutic significance of their pharmacological modulation.
In conclusion, tachykinin NK1 receptor stimulation causes biphasic endothelium-dependent response (EDR followed by EDC) in submaximally precontracted intrapulmonary arteries. The EDR component in the biphasic endothelium-dependent response is more resistant to NK1 antagonists than the EDC component.
Acknowledgments
This work was supported in part by a Grant-in-Aid from Smoking Research Foundation, Tokyo, Japan.
Abbreviations
- EDC
endothelium-dependent contraction
- EDR
endothelium-dependent relaxation
- EIC
endothelium-independent contraction
- L-NAME
L-NG-nitro-arginine methyl ester
- NKA
neurokinin A
- NO
nitric oxide
- PGF2α
prostaglandin F2α
- SP
substance P
- SPME
substance P methyl ester
- TXA2
thromboxane A2
References
- ALLEN K.M., WHARTON J., POLAK J.M., HAWORTH S.G. A study of nerves containing peptides in the pulmonary vasculature of healthy infants and children and of those with pulmonary hypertension. Br. Heart J. 1989;62:353–360. doi: 10.1136/hrt.62.5.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ALTIERE R.J., KIRITSY-ROY J.A., CATRAVAS J.D. Acetylcholine-induced contractions in isolated rabbit pulmonary arteries: Role of thromboxane A2. J. Pharmacol. Exp. Ther. 1986;236:535–541. [PubMed] [Google Scholar]
- BARNES P.J., LIU S.F. Regulation of pulmonary vascular tone. Pharmacol. Rev. 1995;47:87–131. [PubMed] [Google Scholar]
- BOWDEN J.J., BALUK P., LEFEVRE P.M., VIGNA S.R., MCDONALD D.M. Substance P (NK1) receptor immunoreactivity on endothelial cells of the rat tracheal mucosa. Am. J. Physiol. 1996;270:L404–L414. doi: 10.1152/ajplung.1996.270.3.L404. [DOI] [PubMed] [Google Scholar]
- BRETT S.J., SIMON J., GIBBS R., PEPPER J.R., EVANS T.W. Impairment of endothelium-dependent pulmonary vasodilation in patients with primary pulmonary hypertension. Thorax. 1996;51:89–91. doi: 10.1136/thx.51.1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DESAI M.C., LEFKOWITZ S.L., THADEIO P.F., LONGO K.P., SNIDER R.M. Discovery of a potent substance P antagonist: Recognition of the key molecular determinant. J. Med. Chem. 1992;35:4911–4913. doi: 10.1021/jm00104a018. [DOI] [PubMed] [Google Scholar]
- D'ORLEANS-JUSTE P., DION S., DRAPEAU G., REGOLI D. Different receptors are involved in the endothelium-mediated relaxation and the smooth muscle contraction of the rabbit pulmonary artery in response to substance P and related neurokinins. Eur. J. Pharmacol. 1986;125:37–44. doi: 10.1016/0014-2999(86)90081-6. [DOI] [PubMed] [Google Scholar]
- EMONDS-ALT X., DOUTREMEPUICH J.-D., HEAULME M., NELIAT G., SANTUCCI V., STEINBERG R., VILAIN P., BICHON D., DUCOUX J.-P., PROIETTO V., VAN BROECK D., SOUBRIE P., LE FUR G.L., BRELIERE J.-C. In vitro and in vivo biological activities of SR140333, a novel potent non-peptide tachykinin NK1 receptor antagonist. Eur. J. Pharmacol. 1993;250:403–413. doi: 10.1016/0014-2999(93)90027-f. [DOI] [PubMed] [Google Scholar]
- EMONDS-ALT X., VILAIN P., GOULAOUIC P., PROIETTO V., VAN BROECK D., ADVENIER C., NALINE E., NELIAT G., LE FUR G., BRELIERE J.C. A potent and selective non-peptide antagonist of the neurokinin A (NK2) receptor. Life Sci. 1992;50:101–106. doi: 10.1016/0024-3205(92)90352-p. [DOI] [PubMed] [Google Scholar]
- FLOCH A., FARDIN V., CAVERO I. Characterization of NK1 and NK2 tachykinin receptors in guinea-pig and rat bronchopulmonary and vascular systems. Br. J. Pharmacol. 1994;111:759–768. doi: 10.1111/j.1476-5381.1994.tb14803.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GREENO E.W., MANTYH P., VERCELLOTTI G.M., MOLDOW C.F. Functional neurokinin 1 receptors for substance P are expressed by human vascular endothelium. J. Exp. Med. 1993;177:1269–1276. doi: 10.1084/jem.177.5.1269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- IIZUKA K., AKAHANE K., MOMOSE D., NAKAZAWA M., TANOUCHI T., KAWAMURA M., OHYAMA I., KAJIWARA I., OKADA T., TANIGUCHI K., MIYAMOTO T., HAYASHI M. Highly selective inhibitors of thromboxane synthetase. 1. Imidazole derivatives. J. Med. Chem. 1981;24:1139–1148. doi: 10.1021/jm00142a005. [DOI] [PubMed] [Google Scholar]
- KAGEYAMA N., MIURA M., ICHINOSE M., TOMAKI M., ISHIKAWA J., OHUCHI Y., ENDOH N., SHIRATO K. Role of endogenous nitric oxide in airway microvascular leakage induced by inflammatory mediators. Eur. Respir. J. 1997;10:13–19. doi: 10.1183/09031936.97.10010013. [DOI] [PubMed] [Google Scholar]
- KURAHASHI K., USUI H., SHIRAHASE H., JINO H.Endothelium-dependent contraction of cerebral arteries The Human Brain Circulation 1994167–178.In: Bevane, R.D. & Bevane, J.A. (eds)Humana Press: Totowa pp
- MILNER P., RALEVIC V., HOPWOOD A.M., FEHER E., LINCOLN J., KIRKPATRICK K.A., BURNSTOCK G. Ultrastructural localisation of substance P and choline acetyltransferase in endothelial cells of rat coronary artery and release of substance P and acetylcholine during hypoxia. Experientia. 1989;45:121–125. doi: 10.1007/BF01954843. [DOI] [PubMed] [Google Scholar]
- RALEVIC V., MILNER P., HUDLICKA O., KRISTEK F., BURNSTOCK G. Substance P is released from the endothelium of normal and capsaicin-treated rat hind-limb vasculature, in vivo, by increased flow. Circ. Res. 1990;66:1178–1183. doi: 10.1161/01.res.66.5.1178. [DOI] [PubMed] [Google Scholar]
- SARIA A., MARTLING C.R., YAN Z., THEODORSSON-NORHEIM E., GAMSE R., LUNDBERG J.M. Release of multiple tachykinins from capsaicin-sensitive sensory nerves in the lung by bradykinin, histamine, dimethylphenyl piperazinium, and vagal nerve stimulation. Am. Rev. Respir. Dis. 1988;137:1330–1335. doi: 10.1164/ajrccm/137.6.1330. [DOI] [PubMed] [Google Scholar]
- SHIRAHASE H., KANDA M., KURAHASHI K., NAKAMURA S., USUI H., SHIMIZU Y. Endothelium-dependent contraction in intrapulmonary arteries: mediation by endothelial NK1 receptors and TXA2. Br. J. Pharmacol. 1995;115:1215–1220. doi: 10.1111/j.1476-5381.1995.tb15028.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SHIRAHASE H., KANDA M., MURASE K., NAKAMURA S., JINO H., USUI H., KURAHASHI K.Tachykinin receptor subtypes involved in endothelium-dependent and -independent responses in rabbit intrapulmonary arteries Folia Pharmacol. Jpn. 1997110Suppl 1108P–113P.(Abstract in English) [DOI] [PubMed] [Google Scholar]
- SHIRAHASE H., USUI H., KURAHASHI K., FUJIWARA M., FUKUI K. Possible role of endothelial thromboxane A2 in the resting tone and contractile responses to acetylcholine and arachidonic acid in canine cerebral arteries. J. Cardiovasc. Pharmacol. 1987;10:517–522. doi: 10.1097/00005344-198711000-00004. [DOI] [PubMed] [Google Scholar]
- SHIRAHASE H., USUI H., MANABE K., KURAHASHI K., FUJIWARA M. An endothelium-dependent contraction induced by A-23187, a Ca++ ionophore in canine basilar artery. J. Pharmacol. Exp. Ther. 1988a;247:701–705. [PubMed] [Google Scholar]
- SHIRAHASE H., USUI H., MANABE K., KURAHASHI K., FUJIWARA M. Endothelium-dependent contraction and -independent relaxation induced by adenine nucleotides and nucleoside in the canine basilar artery. J. Pharmacol. Exp. Ther. 1988b;247:1152–1157. [PubMed] [Google Scholar]
- SHIRAHASE H., USUI H., SHIMAJI H., KURAHASHI K., FUJIWARA M. Endothelium-independent and -dependent contractions induced by endothelin-1 in canine basilar arteries. Life Sci. 1991;49:273–281. doi: 10.1016/0024-3205(91)90014-3. [DOI] [PubMed] [Google Scholar]
- UREN N.G., LUDMAN P.F., CRAKE T., OAKLEY C.M. Response of the pulmonary circulation to acetylcholine, calcitonin gene-related peptide, substance P and oral nicardipine in patients with primary pulmonary hypertension. J. Am. Coll. Cardiol. 1992;19:835–841. doi: 10.1016/0735-1097(92)90528-u. [DOI] [PubMed] [Google Scholar]
- VANE J.R. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature New Biol. 1971;231:232–235. doi: 10.1038/newbio231232a0. [DOI] [PubMed] [Google Scholar]
- YOSHIMOTO T., YOKOYAMA C., OCHI K., YAMAMOTO S., MAKI Y., ASHIDA Y., TERAO S., SHIRAISHI M. 2,3,5-Trimethyl-6-(12-hydroxy-5,10-dodecadiynyl)-1,4-benzoquinone (AA-861), a selective inhibitor of the 5-lipoxygenase reaction and the biosynthesis of slow-reacting substance of anaphylaxis. Biochim. Biophys. Acta. 1982;713:470–473. [PubMed] [Google Scholar]
- ZENG X.-P., BURCHER E. Use of selective antagonists for further characterization of tachykinin NK-2, NK-1 and possible ‘septide-selective' receptors in guinea pig bronchus. J. Pharmacol. Exp. Ther. 1994;270:1295–1300. [PubMed] [Google Scholar]


