Abstract
A new flavone derivative, chafuroside, has been isolated as a strong anti‐inflammatory compound from oolong tea leaves, and its structure determined to be (2R,3S,4S,4aS,11bS)‐3,4,11‐trihydroxy‐2‐(hydroxymethyl)‐8‐(4‐hydroxyphenyl)‐3,4,4a,11b‐tetrahydro‐2H,10H‐pyrano[2′,3′:4,5]furo[3,2‐g]chromen‐10‐one. To assess its potential to inhibit intestinal carcinogenesis, 2.5, 5 and 10 p.p.m. chafuroside was given in the diet to Apc‐deficient Min mice for 14 weeks from 6 weeks of age. Total numbers of polyps were reduced to 83, 73 and 56% of the control value, respectively. Moreover, dietary administration at 10 and 20 p.p.m. reduced azoxymethane (AOM)‐induced colon aberrant crypt foci (ACF) development in rats to 69% of the AOM‐treated control value with the higher dose. Chafuroside‐associated toxicity was not observed at 2.5–10 p.p.m. in Min mice and 10–20 p.p.m. in AOM‐treated rats. These results suggest that chafuroside might be a good chemopreventive agent for colon cancer. (Cancer Sci 2006; 97: 248–251)
Abbreviations
- AC
aberrant crypt
- ACF
aberrant crypt foci
- AOM
azoxymethane
- COX
cyclooxygenase
- DNFB
2,4‐dinitrofluorobenzene
- EGCG
(–)‐epigallocatechin‐3‐gallate
- NSAID
non‐steroidal anti‐inflammatory drug.
Colon cancer is one of the most common cancers in developed countries( 1 ) and epidemiological studies have shown that a Western‐style diet, high in fat and red meat as well as low in fruits and vegetables, increases the risk.( 2 , 3 ) Thus, foodstuff is a major focus for research, particularly with regard to identification of effective chemopreventive agents.
Epidemiological evidence suggests that drinking green tea (Camellia sinensis) is beneficial for cancer prevention.( 4 , 5 , 6 ) Many animal studies also have shown that tea and its components have anticancer properties.( 7 , 8 ) The major characteristic constituents of green tea are catechins, including EGCG.( 9 ) In black tea, a large proportion of the catechins are converted into theaflavins and thearubigens through oxidation and polymerization. Another tea, oolong tea, is widely consumed in Asia, especially in China and Japan. The difference among green, black and oolong teas lies in fermentation: green tea is unfermented, black tea is completely fermented, and oolong tea is partially fermented.( 7 )
Recently, a strong anti‐inflammatory compound named chafuroside, (2R,3S,4S,4aS,11bS)‐3,4,11‐trihydroxy‐2‐(hydroxymethyl)‐8‐(4‐hydroxyphenyl)‐3,4,4a,11b‐tetrahydro‐2H,10H‐pyrano[2′,3′:4,5]furo[3,2‐g]chromen‐10‐one, was isolated from oolong tea leaves with the aid of an inhibition test with DNFB‐induced contact hypersensitivity in mice, and its total synthesis reported.( 10 , 11 ) The compound was presumed to be produced during the partial fermentation process and showed strong anti‐inflammatory activity in DNFB and 2,4,6‐trinitro‐1‐chlorobenzene‐induced contact hypersensitivity models.( 10 , 11 ) Moreover, a preliminary study demonstrated the effective dose in the DNFB‐induced contact hypersensitivity model to be approximately equal to that of indomethacin, a NSAID, which has been proven to have a cancer‐chemopreventive influence.( 12 )
Although several reports have documented antioxidant, antiallergic and antiobesity activities of oolong tea extracts,( 13 , 14 , 15 ) effects of individual constituents on colon carcinogenesis have hitherto not been described. In the present study, we therefore investigated the impact of chafuroside on intestinal polyp formation in Apc‐deficient Min mice, an animal model of human familial adenomatous polyposis that develops numerous polyps in the intestinal tract.( 16 ) We also investigated the impact of chafuroside on the formation of AOM‐induced aberrant crypt foci, which are putative preneoplastic lesions in the F344 rat colon. In both cases chafuroside reduced the number of lesions, pointing to possible application as a chemopreventive agent for intestinal cancer.
Materials and Methods
Animals and chemicals
Female C57BL/6J‐Apc Min/+ mice (Min mice) were purchased from the Jackson Laboratory (Bar Harbor, ME, USA) at 5 weeks of age and genotyped using a method reported previously.( 16 ) Heterozygotes, as well as wild‐type (C57BL/6J) mice, were acclimated to laboratory conditions for 1 week, along with male F344 rats obtained from Charles River Japan (Atsugi, Japan) at 5 weeks of age. Three to five animals were housed per plastic cage, with sterilized softwood chips as bedding, in a barrier‐sustained animal room, air‐conditioned at 24 ± 2°C and 55% humidity, on a 12:12 h light:dark cycle. Food and water were available ad libitum. The animals were observed daily for clinical signs and mortality. Bodyweights and food consumption were measured weekly. The experiments were conducted according to the ‘Guidelines for Animal Experiments in the National Cancer Center’ of the Committee for Ethics of Animal Experimentation of the National Cancer Center. AOM was purchased from Sigma Chemical Co. (St Louis, MO, USA). Chafuroside was synthesized chemically at the University of Shizuoka (Shizuoka, Japan).( 11 ) Its chemical structure is shown in Fig. 1. The purity of the compound was examined by 1H nuclear magnetic resonance and high‐performance liquid chromatography, and showed no concomitant peaks. The compound was pure enough, estimated to be above 99% (melting point of the compound was 229–232°C). Chafuroside concentrations of 2.5, 5, 10 and 20 p.p.m. were mixed into the powdered basal diet AIN‐76 A (CLEA Japan, Tokyo, Japan) and confirmed to be stable in the diet under the experimental conditions used in the present study. The doses of chafuroside were selected according to our preliminary study in which chafuroside suppressed intestinal polyp formation in Apc gene‐deficient mice.
Figure 1.
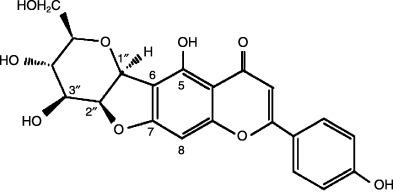
Structure of chafuroside.
Intestinal polyp formation in Min mice
Female Min mice (n = 9–10/group) were fed diets containing 0 (control), 2.5, 5 or 10 p.p.m. chafuroside for 14 weeks from 6 weeks of age. All animals were anesthetized with ether before they were killed. The liver, kidneys and spleen were removed and weighed and the intestinal tract was resected, filled with 10% buffered formalin, and divided into four sections: three segments of small intestine: (1) proximal (4 cm in length from the pylorus ring of the stomach); (2) middle and (3) distal halves of the remainder; and (4) the colon. These segments were opened longitudinally and fixed flat between sheets of filter paper in 10% buffered formalin. Polyp numbers and sizes, and their distributions in the intestine, were determined under a stereoscopic microscope.( 17 )
AOM‐induced ACF development in rats
Male F344 rats, 6 weeks of age, were treated subcutaneously with either AOM in sterile saline at a dose of 15 mg/kg bodyweight or with the saline vehicle, once a week for 2 weeks from 6 weeks of age. From 1 day before the first treatment with AOM, rats were fed control or experimental diets containing chafuroside at 10 or 20 p.p.m. for 4 weeks. At 10 weeks of age, they were killed under ether euthanasia and complete necropsies were carried out. The liver, kidneys and spleen were removed and weighed. The entire colon was resected, filled with 10% buffered formalin, opened longitudinally, and fixed flat between sheets of filter paper in 10% buffered formalin. The colon was then stained with 0.2% methylene blue in saline, and scored under a light microscope for the number of ACF per colon and the mean number of AC per focus.( 18 )
Statistical analysis
The results were expressed as mean ± SD, and statistical analysis was carried out using Dunnett's multiple comparison test. In addition, the linear regression test was also used. Differences were considered to be statistically significant with P‐values less than 0.05.
Results
In Min mice, most polyps were located in the small intestine, with a preponderance in the distal parts, and only a few polyps were observed in the colons (Table 1). Treatment with chafuroside at 2.5, 5 and 10 p.p.m. for 14 weeks clearly decreased the total numbers of polyps to 83, 73 and 56% (P < 0.01) of the untreated control value, respectively (Table 1). The numbers of polyps in the proximal, middle and distal parts of small intestine in the 10 p.p.m. group were 54, 78 and 46% of the untreated control values, respectively (Table 1). Dose‐dependent inhibition was observed in the number of polyps in the proximal (r = –0.9958, P < 0.0005) and distal parts (r = –0.9129, P < 0.01) of the small intestine, and in the total number of polyps (r = –0.9863, P < 0.02). As shown in Fig. 2, administration of chafuroside reduced the number of polyps mainly less than 1.0 mm in diameter. However, the number of polyps measuring ≥1.0 mm in diameter was not affected by chafuroside treatment. Survival rate, general conditions, food consumption and organ weights did not differ among the groups. No significant macroscopic changes were noted in the liver, kidney or spleen. Final body weights in the groups treated with 2.5, 5 and 10 p.p.m. were 103, 105 and 125% of the untreated control value, respectively.
Table 1.
Suppression of intestinal polyp development in Min mice by chafuloside, shown by the number of polyps per mouse
| Group (p.p.m) | No. mice | Small intestine | Colon | Total | ||
|---|---|---|---|---|---|---|
| Proximal | Middle | Distal | ||||
| 0 | 9 | 17.7 ± 9.8 | 39.7 ± 11.6 | 86.8 ± 27.1 | 0.78 ± 0.67 | 144.9 ± 37.7 |
| 2.5 | 7 | 15.3 ± 7.0 (86) | 34.4 ± 7.7 (87) | 70.0 ± 9.9 (81) | 0.86 ± 0.69 (110) | 120.6 ± 21.1 (83) |
| 5.0 | 9 | 13.0 ± 4.3 (73) | 33.4 ± 8.6 (84) | 59.2 ± 18.9 (68)* | 0.56 ± 0.53 (72) | 106.2 ± 26.5 (73) |
| 10.0 | 8 | 9.6 ± 3.1 (54)* | 31.0 ± 19.9 (78) | 40.0 ± 21.8 (46)** | 0.75 ± 0.71 (96) | 81.4 ± 41.9 (56)** |
Data are mean ± SD. Numbers in parentheses are percentages of the control basal diet values. *Significantly different from the basal diet group at P < 0.05. **Significantly different from the basal diet group at P < 0.01.
Figure 2.
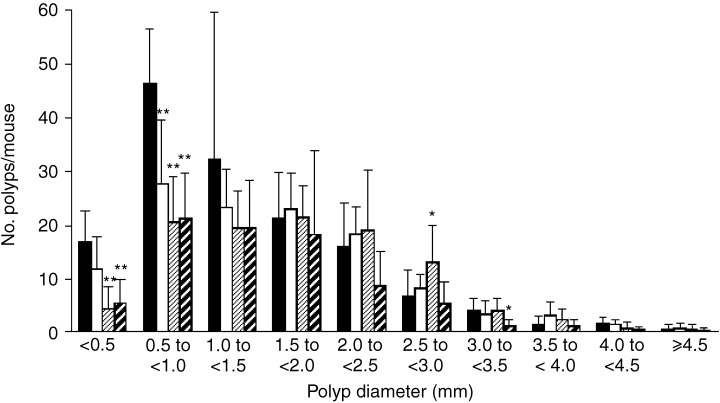
Effects of chafuroside on the size distribution of intestinal polyps in Min mice. Min mice were fed a basal diet (▪) or a diet containing 2.5 (□), 5 (▨) or 10 p.p.m. () chafuroside for 14 weeks. The number of polyps/mouse in each size class is given as a mean value (bars represent SD). *P < 0.05, **P < 0.01.
In AOM‐treated rats, administration of chafuroside at 10 and 20 p.p.m. in the diet for 4 weeks again did not affect general conditions, body weights, food consumption or organ weights. No significant macroscopic changes were observed in the liver, kidney or spleen. ACF were observed in all rats treated with AOM, mainly located in the distal colon. Administration of 10 and 20 p.p.m. chafuroside reduced the total numbers of ACF per colon to 79 (P < 0.05) and 69% (P < 0.01) of the AOM‐treated control value, respectively (Table 2). The total number of AC per colon was also decreased by 16 and 30%, respectively (Table 2). However, treatment with chafuroside did not decrease the mean number of AC per focus (Table 2).
Table 2.
Effects of chafuroside on azoxymethane (AOM)‐induced aberrant crypt focus (ACF) formation in F344 rats
| Group | No. rats with ACF | Total no. ACF/colon (%) | Total no. AC/colon (%) | Mean no. AC/focus |
|---|---|---|---|---|
| AOM treatment | ||||
| Control diet | 9/9 | 278 ± 51 | 618 ± 81 | 2.25 ± 0.18 |
| Chafuroside (10 p.p.m.) | 9/9 | 219 ± 42 (79)* | 522 ± 105 (84) | 2.40 ± 0.27 |
| Chafuroside (20 p.p.m.) | 9/9 | 192 ± 42 (69)** | 435 ± 69 (70)** | 2.29 ± 0.30 |
| Saline treatment | ||||
| Control diet | 0/3 | 0 | 0 | 0 |
| Chafuroside (10 p.p.m.) | 0/3 | 0 | 0 | 0 |
| Chafuroside (20 p.p.m.) | 0/3 | 0 | 0 | 0 |
Data are mean ± SD. Numbers in parentheses are percentages of the control basal diet values. *Significantly different from the basal diet group at P < 0.05. **Significantly different from the basal diet group at P < 0.01. AC, aberrant crypt.
Discussion
In the present study, we obtained clear evidence that a new flavone derivative, chafuroside, suppresses development of intestinal polyps in Min mice and AOM‐induced colon ACF in F344 rats at doses of 5–20 p.p.m. in the diet. Although it is a natural compound found in tea leaves, the doses effective in Min mice were much lower than those reported earlier for well‐known, naturally occurring and synthesized chemopreventive agents. Indeed, the effective dose to reduce numbers of polyps in Min mice was 10 p.p.m. for chafuroside. This value is lower than with (+)‐catechin at 1000 p.p.m.,( 19 ) genistein at 1000 p.p.m.,( 20 ) curcumin at 2000 p.p.m.,( 21 ) and with synthesized aspirin at 250 p.p.m.,( 22 ) piroxicam at 200 p.p.m.( 23 ) and celecoxib at 1500 p.p.m.( 24 ) As treatment with 2.5–10 p.p.m. chafuroside affected only the polyps of smaller size, it might be important to clarify the mechanism by which chafuroside inhibits polyp growth. It has been reported that 100 p.p.m. EGCG is effective for approximately 60% inhibition of AOM‐induced ACF formation in F344 rats,( 25 ) and our results thus suggest that chafuroside possesses a strong potential to inhibit development of putative preneoplastic lesions in the colon.
Because there were no signs of chafuroside‐induced adverse effects in the present study, long‐term consumption for cancer prevention in humans is conceivable. The daily estimated consumption level from a diet containing 10 p.p.m. chafuroside in mice corresponds to approximately 120 mg per day for a 60‐kg adult man. The average concentration of chafuroside in a commercially available oolong tea in Japan is almost 55 µg/L (unpublished data). An adult human might reach an effective dose of chafuroside with consumption of more than 100 L of oolong tea. Therefore, taking a chafuroside supplement or drinking concentrated oolong tea may be useful for cancer prevention.
The strong anti‐inflammatory and chemopreventive effects are presumably related to the two characteristic moieties of chafuroside: the mannose moiety of which the C1 position provides a C‐glycoside linkage with the C6 position of apigenin, and the dihydrofuran moiety, obtained by ring closure via dehydration between hydroxyl groups at the C7 position of apigenin and at the C2 position of mannose.
According to the mechanism of DNFB‐induced contact hypersensitivity, several factors moderated by chafuroside treatment could be assumed. DNFB‐induced skin immune responses are divided into a distinct sensitization phase and an early and late elicitation phase. In the sensitization phase, prostaglandin E2‐EP4 receptor (PGE2‐EP4) signaling pathways are reported to be involved in antigen uptake into Langerhans cells, and in antigen migration to draining lymph nodes.( 26 ) In addition, other colon tumor‐related pathways and mediators, such as ERK1/2 and p38 MAPK signaling pathways, 5‐lipoxygenase and inducible nitric oxide synthase, are also reported to be involved in DNFB‐induced contact hypersensitivity.( 27 , 28 , 29 ) Thus, chafuroside may exhibit its chemopreventive effects by mediating these inflammatory factors. However, the natural function of chafuroside is not well defined and the anti‐inflammatory and chemopreventive mechanisms of this novel flavonoid need to be clarified.
From the present results, we can conclude strong inhibitory effects of chafuroside on intestinal polyp development and ACF formation in Apc‐deficient mice and AOM‐treated rats, respectively, so that chafuroside may be a promising candidate chemopreventive agent for colon cancer.
Acknowledgments
This work was supported by Grants‐in‐Aid for Cancer Research and for the Third‐Term Comprehensive 10‐Year Strategy for Cancer Control from the Ministry of Health, Labour and Welfare of Japan. Dr Katsuhisa Sakano is the recipient of a Research Resident fellowship from the Foundation of Promotion of Cancer Research.
References
- 1. Stewart BW, Kleihues P, eds. World Cancer Report. Lyon: IARC Press, 2003: 12–17. [Google Scholar]
- 2. Slattery ML, Boucher KM, Caan BJ, Potter JD, Ma KN. Eating patterns and risk of colon cancer. Am J Epidemiol 1998; 148: 4–16. [DOI] [PubMed] [Google Scholar]
- 3. Levi F, Pasche C, La Vecchia C, Lucchini F, Franceschi S. Food groups and colorectal cancer risk. Br J Cancer 1999; 79: 1283–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ji BT, Chow WH, Hsing AW et al. Green tea consumption and the risk of pancreatic and colorectal cancers. Int J Cancer 1997; 70: 255–8. [DOI] [PubMed] [Google Scholar]
- 5. Nakachi K, Suemasu K, Suga K, Takeo T, Imai K, Higashi Y. Influence of drinking green tea on breast cancer malignancy among Japanese patients. Jpn J Cancer Res 1998; 89: 254–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sasazuki S, Inoue M, Hanaoka T, Yamamoto S, Sobue T, Tsugane S. Green tea consumption and subsequent risk of gastric cancer by subsite: the JPHC Study. Cancer Causes Control 2004; 15: 483–91. [DOI] [PubMed] [Google Scholar]
- 7. Kuroda Y, Hara Y. Antimutagenic and anticarcinogenic activity of tea polyphenols. Mutat Res 1999; 436: 69–97. [DOI] [PubMed] [Google Scholar]
- 8. Lin JK, Liang YC, Lin‐Shiau SY. Cancer chemoprevention by tea polyphenols through mitotic signal transduction blockade. Biochem Pharmacol 1999; 58: 911–15. [DOI] [PubMed] [Google Scholar]
- 9. Yang GY, Liao J, Li C et al. Effect of black and green tea polyphenols on c‐jun phosphorylation and H2O2 production in transformed and non‐transformed human bronchial cell lines: possible mechanisms of cell growth inhibition and apoptosis induction. Carcinogenesis 2000; 21: 2035–9. [DOI] [PubMed] [Google Scholar]
- 10. Nakatsuka T, Tomimori Y, Fukuda Y, Nukaya H. First total synthesis of structurally unique flavonoids and their strong anti‐inflammatory effect. Bioorg Med Chem Lett 2004; 14: 3201–3. [DOI] [PubMed] [Google Scholar]
- 11. Furuta T, Kimura T, Kondo S et al. Concise total synthesis of flavone C‐glycoside having potent anti‐inflammatory activity. Tetrahedron 2004; 60: 9375–9. [Google Scholar]
- 12. Levy GN. Prostaglandin H synthases, nonsteroidal anti‐inflammatory drugs, and colon cancer. FASEB J 1997; 11: 234–47. [PubMed] [Google Scholar]
- 13. Uehara M, Sugiura H, Sakurai K. A trial of oolong tea in the management of recalcitrant atopic dermatitis. Arch Dermatol 2001; 137: 42–3. [DOI] [PubMed] [Google Scholar]
- 14. Rumpler W, Seale J, Clevidence B et al. Oolong tea increases metabolic rate and fat oxidation in men. J Nutr 2001; 131: 2848–52. [DOI] [PubMed] [Google Scholar]
- 15. Kurihara H, Fukami H, Toyoda Y et al. Inhibitory effect of oolong tea on the oxidative state of low density lipoprotein (LDL). Biol Pharm Bull 2003; 26: 739–42. [DOI] [PubMed] [Google Scholar]
- 16. Moser AR, Pitot HC, Dove WF. A dominant mutation that predisposes to multiple intestinal neoplasia in the mouse. Science 1990; 247: 322–4. [DOI] [PubMed] [Google Scholar]
- 17. Niho N, Mutoh M, Takahashi M, Tsutsumi K, Sugimura T, Wakabayashi K. Concurrent suppression of hyperlipidemia and intestinal polyp formation by NO‐1886, increasing lipoprotein lipase activity in Min mice. Proc Natl Acad Sci USA 2005; 102: 2970–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sutherland LA, Bird RP. The effect of chenodeoxycholic acid on the development of aberrant crypt foci in the rat colon. Cancer Lett 1994; 76: 101–7. [DOI] [PubMed] [Google Scholar]
- 19. Weyant MJ, Carothers AM, Dannenberg AJ, Bertagnolli MM. (+)‐Catechin inhibits intestinal tumor formation and suppresses focal adhesion kinase activation in the Min/+ mouse. Cancer Res 2001; 61: 118–25. [PubMed] [Google Scholar]
- 20. Javid SH, Moran AE, Carothers AM, Redston M, Bertagnolli MM. Modulation of tumor formation and intestinal cell migration by estrogens in the Apc Min/+ mouse model of colorectal cancer. Carcinogenesis 2005; 26: 587–95. [DOI] [PubMed] [Google Scholar]
- 21. Perkins S, Verschoyle RD, Hill K et al. Chemopreventive efficacy and pharmacokinetics of curcumin in the Min/+ mouse, a model of familial adenomatous polyposis. Cancer Epidemiol Biomarkers Prev 2002; 11: 535–40. [PubMed] [Google Scholar]
- 22. Barnes CJ, Lee M. Chemoprevention of spontaneous intestinal adenomas in the adenomatous polyposis coli Min mouse model with aspirin. Gastroenterology 1998; 114: 873–7. [DOI] [PubMed] [Google Scholar]
- 23. Jacoby RF, Marshall DJ, Newton MA et al. Chemoprevention of spontaneous intestinal adenomas in the Apc Min mouse model by the nonsteroidal anti‐inflammatory drug piroxicam. Cancer Res 1996; 56: 710–14. [PubMed] [Google Scholar]
- 24. Jacoby RF, Seibert K, Cole CE, Kelloff G, Lubet RA. The cyclooxygenase‐2 inhibitor celecoxib is a potent preventive and therapeutic agent in the Min mouse model of adenomatous polyposis. Cancer Res 2000; 60: 5040–4. [PubMed] [Google Scholar]
- 25. Ohishi T, Kishimoto Y, Miura N et al. Synergistic effects of (–)‐epigallocatechin gallate with sulindac against colon carcinogenesis of rats treated with azoxymethane. Cancer Lett 2002; 177: 49–56. [DOI] [PubMed] [Google Scholar]
- 26. Kabashima K, Sakata D, Nagamachi M, Miyachi Y, Inaba K, Narumiya S. Prostaglandin E2‐EP4 signaling initiates skin immune responses by promoting migration and maturation of Langerhans cells. Nat Med 2003; 9: 744–9. [DOI] [PubMed] [Google Scholar]
- 27. Matos TJ, Duarte CB, Goncalo M, Lopes MC. DNFB activates MAPKs and upregulates CD40 in skin‐derived dendritic cells. J Dermatol Sci 2005; 39: 113–23. [DOI] [PubMed] [Google Scholar]
- 28. Csato M, Rosenbach T, Czarnetzki BM. Involvement of arachidonate‐dependent lipid mediators and platelet‐activating factor in experimental murine contact dermatitis. Skin Pharmacol 1988; 1: 100–5. [DOI] [PubMed] [Google Scholar]
- 29. Ross R, Gillitzer C, Kleinz R et al. Involvement of NO in contact hypersensitivity. Int Immunol 1998; 10: 61–9. [DOI] [PubMed] [Google Scholar]


