Abstract
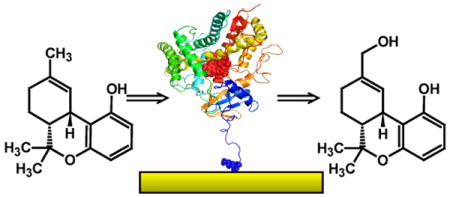
The cytochrome P450 enzymes represent an important class of heme containing enzymes. There is considerable interest in immobilizing these enzymes on a surface so that interactions between a single enzyme and other species can be studied with respect to electron transfer, homodimer or heterodimer interactions, or for construction of biological based chips for standardizing cytochrome P450 metabolism or for high throughput screening of pharmaceutical agents. Previous studies have generally immobilize P450 enzymes in a matrix or on a surface. Here, we have attached CYP2C9 to gold substrates such that the resulting construct maintains the ability to bind and metabolize substrates in the presence of NADPH and cytochrome P450 reductase. The activity of these chips is directly dependent upon the linkers used to attach CYP2C9 and to the presence of key molecules in the active site during enzyme attachment. A novel method to detect substrate-enzyme binding, namely superconducting quantum interference device (SQUID) magnetometry, was used to monitor the binding of substrates. Most significantly, conditions that allow measurable CYP2C9 metabolism to occur have been developed.
The cytochrome P450 enzymes represent an important class of heme containing enzymes. They are responsible for a significant portion of xenobiotic metabolism and have been extensively studied for mechanistic and practical reasons. More recently there has been interest in immobilizing these enzymes on a surface to study the electron transfer in a single enzyme,1 homodimer or heterodimer interactions,2 or for construction of biological based chips for high throughput screening of pharmaceutical agents.
Previous efforts to immobilize P450 enzymes have been made3 including CYP2E1 on gold electrodes1 bacterial P450 BM3 on graphite,4 P450cam immobilized in sol-gel films,5 CYP1A2 and CYP3A4 in polyion films,6 and CYP119 in dimethyldidodecylammonium poly(p-styrene sulfonate).5 In these studies the detection of substrate binding is often made electrochemically by observing shifts in the redox potential upon substrate binding.7 Enzyme-like metabolic reactions have also been observed by the application of an electrochemical current.1 However, to be amenable to study and manipulation at the single molecule level it may be desirable to minimize the surrounding matrix. Also, it is highly desirable for the enzyme to metabolize substrates utilizing endogenous enzymes and co-factors to more closely model the corresponding biological system. To our knowledge, this has not been achieved with cytochrome P450 enzymes.
In this work we have attached CYP2C9 to gold substrates such that the resulting construct maintains the ability to bind and metabolize substrates in the presence of NADPH and cytochrome P450 reductase. The activity of these chips is dependent upon the linkers used to attach CYP2C9 and to the presence of key substrates during the attachment. A novel method to detect substrate binding, namely superconducting quantum interference device (SQUID) magnetometry, was used to monitor the binding of substrates. Most significantly, conditions that allow measurable CYP2C9 metabolism to occur have been identified.
The attachment of CYP2C9 was made by several methods. Utilizing thiols present in the enzyme, CYP2C9 was either directly attached to gold substrates or via a cysteine-maleimide linker.8 The N-terminus is an alternative site though surface lysines may compete. Molecular modeling of CYP2C9, based on its crystal structure,9 shows there are cysteine and lysine residues that border the substrate access channel and the reductase binding site (Figure 1a). Bonding at either of these sites to the substrate may prevent normal enzyme function. However, attachment through the N-terminus methionine is more likely as its pKa is less than lysine (more neutral) and less sterically hindered; a sub-population of CYP2C9 molecules will bond at this site and leave the substrate access channel and reductase binding sites accessible. For N-terminus attachment the gold surface was first treated with a 1:3 mixture of octanethiol (OT) and 11-mercaptoundecanoic acid (MUA). Activation of the carboxylic acid with N-((3-dimethylamino)-propyl)-N-ethyl carbodiimide hydrochloride (EDC) (1.0 mM) and N-hydroxysulfosuccinimide (NHS) followed by reaction with CYP2C9 successfully attached CYP2C9.10 Atomic force microscopy (AFM) images were obtained before and after enzyme attachment. A representative example is shown in Figure 1c. The obvious changes in morphology suggest attachment of the enzyme as features with lateral sizes ranging from about 10-20 nm are clearly visible, consistent with the known dimensions of CYP2C99 if tip/feature convolution is taken into account.
Figure 1.

a) CYP2C9 with the access channel and reductase binding sites, cysteine residues (yellow), and lysines (gray) with lysine nitrogens (blue). b) Cartoon of the attachment of CYP2C9 to a gold surface coated with OT and MUA (3:1) catalyzed by EDC/NHS. c) Corresponding AFM images prior to enzyme attachement (top) and after attachment (bottom).
The issue of whether the CYP2C9 attached to the gold substrate was functional was addressed next. CYP2C9 is a heme enzyme and the iron is known to change spin state when it binds flurbiprofen or a mixture of flurbiprofen and dapsone, but not when dapsone, alone, binds (see supporting information).11 The change in iron spin state should also alter its magnetic susceptibility and therefore may be detected with magnetometry. Samples of gold-CYP2C9, prepared with no substrate, dapsone, flurbiprofen, or a mixture of flurbiprofen and dapsone, were examined. Figure 2 shows the magnetic signal (M) as a function of magnetic field (H) The signal near H=0 may be affected by small amounts of ferromagnetic impurities and is therefore not meaningful. Nevertheless, the slope of the signal (differential susceptibility) at higher fields (dM/dH) only depends on a diamagnetic or paramagnetic signal and is therefore more likely to originate from the CYP2C9. The curves for CYP2C9 without substrate or with dapsone are similar and have slopes that go to zero at higher fields, and therefore the ratio of the spin states for these samples is similar. In contrast, the samples containing flurbiprofen or flurbiprofen and dapsone display positive slopes at higher fields and in solution analogous samples are predominantly high spin. Thus, the change in spin state and the susceptibility at high fields appear to be correlated. Remarkably, this behavior is reversible. The bound flurbiprofen can be washed out with water and the magnetization curve restored to that of CYP2C9 alone.
Figure 2.
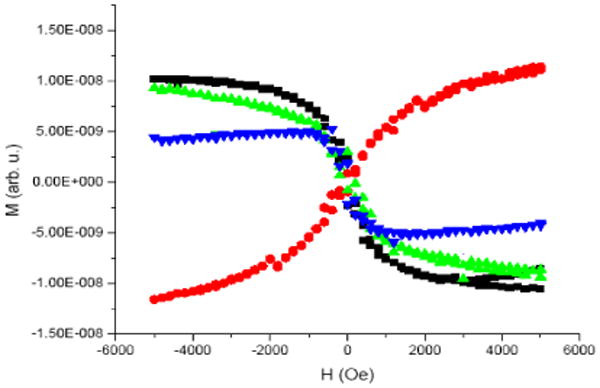
Magnetization data as a function of external magnetic field. Curves correspond to CYP2C9 (black), CYP2C9 and dapsone (green), CYP2C9 and flurbiprofen (red), and CYP2C9 and flurbiprofen/dapsone (blue). Data acquired at a temperature of 300 K in increasing and decreasing fields. The CYP2C9 data at high fields was slightly different for the increasing and decreasing field measurements due to a slight change in the sample position during the last part of the experiment.
The metabolic activity of the gold-CYP2C9 chips was explored next. The metabolism of Δ9-tetrahydrocannibinol (THC) to 11-hydroxy-Δ9-THC by CYP2C9 was selected for two reasons. First, while flurbiprofen and dapsone are substrates for CYP2C9, some oxidation of these substrates may have occurred during chip preparation, potentially producing false positives. Second, the sensitivity of the THC metabolism assay is at least an order of magnitude greater than for flurbiprofen or dapsone metabolism.12 Chips were prepared with CYP2C9 alone and with CYP2C9 in the presence of flurbiprofen alone or flurbiprofen and dapsone. After preparation, the chips were washed with water to remove flurbiprofen or dapsone. Chips were then treated with THC and various combinations of NADPH, reductase, and phospholipid. THC metabolism was only observed for chips that had been prepared with both flurbiprofen and dapsone and to which phospholipid had not been added (Table 1).
Table 1.
Solution and Au bonded CYP2C9 metabolism of THC.a
| No. | Sample | THC-OHb |
|---|---|---|
| 1 | Blank (Solution) | 1.2 (±0.1) |
| 2 | CYP2C9 (Solution) | 616 (±25) |
| 3 | Au (OT/MUA linker) | 0.0 |
| 4 | Au-CYP2C9 (Flurbiprofen or Dapsone) | 0.0 |
| 5 | Au-CYP2C9 (Flurbiprofen and Dapsone) | 20.6 (±1.6) |
Incubations were in THAM buffer (50 mM, pH 7.4). Sample 2 was run 30 min, 1 and 3-5 for 8 hours. Metabolite analysis was by HPLC-MS.12
Values are the average of 3 runs and in ng/pmol protein/hr (±SD).
The conditions required to achieve metabolism of THC by the gold-CYP2C9 chips deserve comment. Only samples prepared with flurbiprofen and dapsone present were metabolically active. CYP2C9 has a large active site that can simultaneously bind two or more molecules as we have shown for flurbiprofen and dapsone.13 The active site of CYP2C9, in samples that do not contain both substrates, may become damaged in the course of preparation in such a way as to prevent metabolism but not prevent binding. However, when both substrates are present they may act as supports to prevent the active site from distorting. Once prepared, the overall enzyme is no longer susceptible to damage as the substrates can be washed out and replaced by THC. Similar observations have been made for CYP1A2 which can only be isolated and purified if 7,8-benzoflavone is bound in the active site.14 Also notable is the dependence of metabolism on phospholipids. While not required for metabolism in solution, phospholipids are generally known to accelerate metabolism. However, with the gold-CYP2C9 constructs, phospholipids are inhibitory.
The cytochrome P450 enzymes are some of the most extensively studied enzymes. Prior to this work they had been successfully attached to solid substrates, shown to bind compounds, and to be redox active, but not metabolically active. Here we have determined a method for attachment and conditions whereby gold-supported CYP2C9 binds and can metabolize substrates. A novel method for detecting binding has also been explored. This work opens the possibility of the construction of nanobiochips of CYP2C9 such that binding and metabolism studies can be standardized. It may also be possible to utilize this system for more basic research on the metabolic effect of homodimers, heterodimers, or complexes of cytochrome P450 with reductase or cytochrome b5.
Supplementary Material
Experimental procedures for the preparation of the Au-CYP2C9 samples, AFM and SQUID magnetometry measurements, and metabolism measurements. The material is available free of charge via the Internet at http://pubs.acs.org.
Acknowledgments
We thank the National Institutes of Health for financial support of the research (RO1 063215), the WVEPSCoR STEM program for fellowship support (to JK), WVNano for postdoctoral and graduate student support (to JG and FP), and Dr. Edwin Rood for his initial support of this work.
References
- 1.Fantuzzi A, Fairhead M, Gilardi G. J Am Chem Soc. 2004;126:5040–5041. doi: 10.1021/ja049855s. [DOI] [PubMed] [Google Scholar]
- 2.Backes WL, Kelley RW. Pharmac Ther. 2004;98:221–233. doi: 10.1016/s0163-7258(03)00031-7. [DOI] [PubMed] [Google Scholar]
- 3.Johnson DL, Lewis BC, Elliot DJ, Miners JO, Martin LL. Biochem Pharmacol. 2005;69:1533–1541. doi: 10.1016/j.bcp.2005.02.020. [DOI] [PubMed] [Google Scholar]
- 4.Udit AK, Hill MG, Bittner VG, Arnold FH, Gray HB. J Am Chem Soc. 2004;126:10218–10219. doi: 10.1021/ja0466560. [DOI] [PubMed] [Google Scholar]
- 5.Iwuoha EI, Kane S, Ania CO, Smyth MR, Ortiz de Montellano PR, Fuhr U. Electroanalysis. 2000;12:980–986. [Google Scholar]
- 6.Sultana N, Schenkman JB, Rusling JF. J Am Chem Soc. 2005;127:13460–13461. doi: 10.1021/ja0538334. [DOI] [PubMed] [Google Scholar]
- 7.Jefcoate CR. Meth Enzymol. 1978;52:258–279. doi: 10.1016/s0076-6879(78)52029-6. [DOI] [PubMed] [Google Scholar]
- 8.Pardo-Yissar V, Katz E, Willner I, Kotlyar AB, Sanders C, Lill H. Faraday Discussions. 2000;116:119–134. doi: 10.1039/b001508n. [DOI] [PubMed] [Google Scholar]
- 9.Wester MR, Yano JK, Schoch GA, Yang C, Griffin KJ, Stout CD, Johnson EF. J Biol Chem. 2004;279:35630–35637. doi: 10.1074/jbc.M405427200. [DOI] [PubMed] [Google Scholar]
- 10.Haes AJ, Chang L, Klein WL, Van Duyne RP. J Am Chem Soc. 2005;127:2264–2271. doi: 10.1021/ja044087q. [DOI] [PubMed] [Google Scholar]
- 11.Hummel MA, Locuson CW, Gannett PM, Rock DA, Mosher C, Rettie AE. Mol Pharmacol. 2005;68:644–651. doi: 10.1124/mol.105.013763. [DOI] [PubMed] [Google Scholar]
- 12.Bland TM, Haining RL, Tracy TS, Callery PS. Biochem Pharmacol. 2005;70:1096–1103. doi: 10.1016/j.bcp.2005.07.007. [DOI] [PubMed] [Google Scholar]
- 13.Tracy TS, Hummel MA, Gannett PM, Aguilar JS. Biochem. 2004;43:7207–7214. doi: 10.1021/bi036158o. [DOI] [PubMed] [Google Scholar]
- 14.Sandhu P, Guo Z, Baba T, Martin MV. Arch Biochem Biophys. 1994;309:168–177. doi: 10.1006/abbi.1994.1099. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Experimental procedures for the preparation of the Au-CYP2C9 samples, AFM and SQUID magnetometry measurements, and metabolism measurements. The material is available free of charge via the Internet at http://pubs.acs.org.


