The surface of the eye is an extraordinary and vital component of vision. The smooth wet surface of the cornea is the major refractive surface of the visual system, which, along with corneal transparency, enables light to proceed through the lens and onto the retina for photoreceptor activation. Presence of the smooth wet, refractive ocular surface required for vision comes, however, at a cost. Unlike all other wet-surfaced epithelia of the body, the ocular surface is directly exposed to the outside world where it is especially subject to desiccation, injury and pathogens. As a consequence, numerous protective mechanisms are provided by the Ocular Surface System to ensure vision.
The Ocular Surface System
Maintenance and protection of the smooth refractive surface of the cornea is the function of the Ocular Surface System (Fig. 1).1 The “Ocular Surface System” is defined as the ocular surface, which includes the surface and glandular epithelia of the cornea, conjunctiva, lacrimal gland, accessory lacrimal glands, and meibomian gland, and their apical (tears) and basal (connective tissue) matrices, the eyelashes with their associated glands of Moll and Zeis, those components of the eyelids responsible for the blink, and the nasolacrimal duct. All components of the system are linked functionally by continuity of the epithelia, by innervation, and by the endocrine, vascular and immune systems. The rationale for the use of the term Ocular Surface System is several fold. Firstly, the primary, synergistic function of the system components is to provide, protect and maintain a smooth refractive surface on the cornea. Thus, the name “Ocular Surface System” is linked to its primary function at the ocular surface. Secondly, all the epithelia at the ocular surface are continuous, with no breaks between regions, and all are derived from the surface ectoderm. The corneal and conjunctival epithelia are continuous, through the ductal epithelium, to the lacrimal glandular epithelium, as is the case with the accessory lacrimal glands, the meibomian gland and the nasolacrimal system. The glandular systems are essentially involutions from and specializations of the surface epithelium. Communication along these epithelia occurs through gap junctions and cytokines.2-4 Thirdly, all regions of the ocular surface epithelia produce components of the refractive tear film; the corneal and conjunctival epithelia produce hydrophilic mucins that hold tears onto the surface of the eye; the lacrimal and accessory lacrimal glands secrete water and a host of protective proteins; the meibomian gland provides the superficial tear lipid layer that prevents tear evaporation. The nasolacrimal epithelial system adsorbs tear components and is believed to, through its cavernous vascular system, control/regulate tear outflow, helping to maintain the appropriate tear level, a fine balance between secretion and outflow.5 The functions of the various regions of the continuous epithelia and the eyelid blink are integrated by the nervous, endocrine, circulatory and immune systems, and are supported by the connective tissue with its resident cells and blood vessels.
Figure 1.
Diagrams depicting the Ocular Surface System. (A) Sagittal section through the Ocular Surface System showing that the ocular surface epithelium is continuous (in pink) with regional specializations on/in the cornea, conjunctiva, lacrimal and accessory lacrimal glands, and meibomian gland. Each specialized region of this ocular surface epithelium contributes components of the tear film (in blue). (B) Frontal view of the Ocular Surface System, which includes the surface and glandular epithelia of the cornea, conjunctiva, lacrimal gland, accessory lacrimal glands, and meibomian gland (note enlarged lower lid segment) and their apical (tears) and basal connective tissue matrices, the eye lashes, those components of the eyelids responsible for the blink, and the nasolacrimal duct. The functions of the system's components are integrated or linked by innervation, and the endocrine, vascular and immune systems. We appreciate the artwork of Peter Mallen in the preparation of this figure.
A unit within the Ocular Surface System has been termed “The Lacrimal Functional Unit.6” “The Lacrimal Functional Unit is composed of the lacrimal glands (both main and accessory), the ocular surface and the interconnecting innervation.” This term emphasizes the interplay between the lacrimal gland, the ocular surface, narrowly defined as the corneal and conjunctival epithelia, and the nervous system. The Ocular Surface System is a broader concept in that it recognizes contributions to the tear film of all the regions of the ocular surface epithelia, incorporates the lids and nasolacrimal duct, and recognizes the integrative functions of the endocrine, immune and vascular systems.
Since the functions of all regions of the Ocular Surface System are closely integrated, especially by innervation from the trigeminal nerves, signals from one region of the system influences the blink, goblet cell secretion, lacrimation, and/or lacrimal gland gene expression. For example, a corneal wound induces alteration of the gene expression profile of the lacrimal gland.7 Similarly, dysfunctions of or injury to one or more components of the Ocular Surface System can lead to system-wide sequelae such as cicatrizing diseases and dry eye—the latter being defined as a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance and tear film instability, with potential damage to the ocular surface.8 For example, meibomian or lacrimal gland disease induces dry eye.1
Development of treatments for ocular surface disease may be facilitated by consideration of the Ocular Surface System as a whole, rather than by targeting any one component of the system.
Tear Film and its Interface with the Ocular Surface Epithelium
The smooth refractive tear film is maintained on the cornea and ocular surface by blinking of the eyelid to replenish the tears over the cornea, through continuous constitutive secretion of tear components by all areas of the ocular surface epithelia, and by specializations on the apical surfaces of the corneal and conjunctival epithelia. Early hypotheses of tear film structure separated the secretions from the lacrimal gland, meibomian gland, and conjunctival goblet cell mucins into three separate, distinct layers within the tear film—the lipid, aqueous and mucin layers, respectively. While it is clear that the meibomian gland-derived lipid layer is partitioned on the surface of the aqueous layer, more recent data suggest that the aqueous tears are a mixture of lacrimal fluid and mucins, without a distinct mucin layer within the tears (Fig. 2).9,10 The tear-epithelial cell interface is critical for tear film maintenance on the corneal and conjunctival epithelia. As with all wet-surfaced epithelia of the body, including those of the Ocular Surface System, and the GI, respiratory and reproductive systems, maintenance of fluids on the cell surface is facilitated through membrane specialization on the apical surface membrane, where it abuts the luminal surface. A hydrophilic, heavily glycosylated glycocalyx is present on these apical surface membranes of the epithelia. Recent data demonstrate that a major component of the glycocalyx is a class of newly described membrane-associated mucins.10,11 At the ocular surface, the apical cell membrane adjacent to the tear film interface is thrown into short membrane folds, termed microplicae (Fig. 2). Membrane-associated mucins emanate from the tips of the microplicae and extend up to 500 nm from the membrane to form the glycocalyx.10,12
Figure 2.
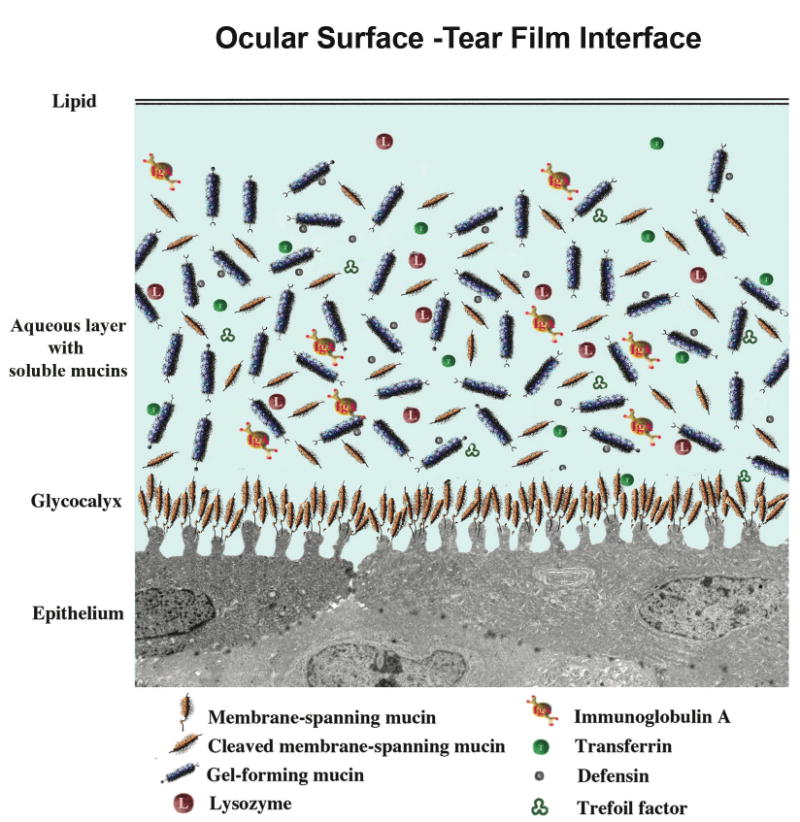
Diagram of the tear film and its interface with the ocular surface epithelium. The outermost lipid layer, the product of the meibomian gland, abuts the aqueous layer, with its soluble mucin components either secreted from conjunctival goblet cells, or shed from apical surfaces of corneal and conjunctival epithelial cells. A multitude of bactericidal and other proteins are present in the aqueous layer. The glycocalyx layer, which extends from the tips of surface ridges known as microplicae, is formed by membrane-associated mucins, which are tethered to the cells by a membrane-spanning domain and short cytoplasmic tail. The extracellular domains of these mucins are constituitively shed into the tear film.
The Search for the H185 Antigen Yields Charcterization of Mucins of the Ocular Surface
Protective mechanisms of the ocular surface epithelium include the ability to heal quickly and adhere tenaciously to underlying connective tissue, especially since the cornea and ocular surface are exposed to the “outside” world, and since blinking and eye rubbing puts abrasive pressure on the epithelium. Early studies on protective mechanisms of the corneal epithelium by our group focused on corneal epithelial wound healing and anchorage in normal, healing, and disease conditions.13-25 Our major contributions in the wound healing area include development of organ culture models of wounded rodent corneas,26 demonstration of increases in specific proteins and glycoproteins during epithelial migration,15,17,19,27 demonstration that an actin “purse string” coordinates epithelial wound closure,24 and demonstration that leading edge cells can be selectively transfected.25
In the epithelial anchorage field, we developed an in vitro system for study of hemidesmosome formation,13 demonstrated that hemidesmosome formation occurs on sites of basal lamina where type VII collagen anchoring fibrils insert,18 demonstrated that hemidesmosome formation requires calcium and is calmodulin mediated,16 showed that diabetes alters epithelial anchorage and epithelia-basement membrane interactions.20,23 and discovered that the α6β4 integrin is a hemidesmosome component.21
To facilitate studies of epithelial anchorage, monoclonal antibodies were developed to use as probes for study of the molecular composition of hemidesmosomes, anchoring structures of basal cells of the corneal epithelium. Serendipitously, during development of these antibodies, an antibody was found that bound to the apical surface of the corneal epithelium of the rat. Upon isolation of the antigen, it was found to have a mucin-like character.28 Because of the dearth of information available about the composition of the apical surface of the human cornea and its potential relevance to surface disease, similar methods were used to develop monoclonal antibodies to the human ocular surface. A human-specific antibody, designated H185, was developed that also recognized a mucin-like molecule on the apical cells of corneal and conjunctival epithelia, that had as its epitope a carbohydrate on the mucin, and that appeared to be membrane associated.29 Subsequent studies of the distribution of the H185 mucin on a series of normal and non-Sjögren's dry eye patients demonstrated that the antibody binding to apical cells of conjunctival epithelium of the dry eye patients was diminished (Fig. 3).30 Finding a clinical alteration of the ocular surface in dry eye patients spurred attempts to isolate and clone the molecule recognized by the H185 antibody and, in so doing, changed the direction of the research in our group from wound healing and epithelial anchorage to that of the role of mucins at the ocular surface in health and disease. The security to make such a major shift in research direction was only possible through 10 years of secure funding provided by an NIH/NEI 10-year Merit Award.
Figure 3.
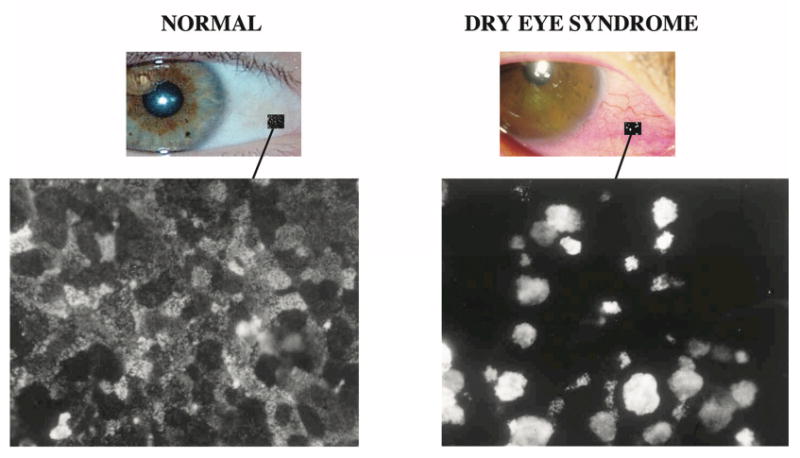
Impression cytology samples from normal subjects and dry eye patients (non-Sjögren's) showing change in localization pattern of binding of an antibody designated H185, which recognizes an O-acetylated sialic acid on the membrane-associated mucin MUC16. On the normal ocular surface, the antibody binds in a “patchwork” pattern, some cells binding the antibody intensely, others to a lesser degree, similar to the light, medium and dark cells seen by scanning electron microscopy. In dry eye patients, apical cell surface binding is lost and goblet cells bind the antibody intensely (see Danjo et al., 1998,30 for details).
Characteristics of Mucins
Mucins are a class of heavily O-glycosylated glycoproteins, the mass of which may reach 80% carbohydrate. As a result of their heavy glycosylation, they have been difficult to characterize, and it was only with the relatively recent application of molecular cloning techniques that mucin genes have been identified.31,32 As a result, there has been an explosion of new information characterizing this class of now 19 unique mucin molecules. Much of this interest has been driven by the fact that a number of mucins are tumor cell markers.32 By definition, a glycoprotein is a mucin if it has within its protein backbone tandem repeats of amino acids that are rich in serine, threonine and proline—the serines and threonines providing sites for O-glycosylation of the molecule. Each mucin gene has a unique tandem repeat amino acid sequence and length, and its alleles vary in numbers of repetitions of the tandem repeat, which lead to polymorphisms within and amongst individuals. Human mucin genes are designated MUC1, 2, etc., in order of discovery; mouse homologues are designated in lower case—muc1, etc. Sequencing of mucin genes has led to the identification of two categories of mucins, secreted and membrane associated (membrane spanning or membrane tethered) (for review see Gendler, 1995 and Hollingsworth, 2004).31,32
Secreted Mucins
Of the seven secreted mucins identified to date, five are the so-called gel-forming mucins (Fig. 4). The gel-forming mucins are the largest glycoproteins known, with genes of 15.7 to 17 kb and deduced proteins of about 600 kDa.33 Their N and C termini have cysteine-rich domains, which allow for homomultimer formation, in turn resulting in large polymers that produce the viscous mucus associated with gastric and respiratory tract secretions. The gel-forming mucins are secreted by goblet or glandular cells in all wet-surfaced epithelia of the body. They are moved about on epithelial surfaces by various mechanisms, including tracheal cilia movement, peristalsis, or, in the case of the ocular surface, by eyelid movement, to clean the surfaces of particulate matter. Secreted mucins' hydrophilic character, which results from its heavy glycosylation, helps to hold fluids on epithelial surfaces. Two small secreted mucins have been identified, MUC7 and MUC9—both of which lack cysteine-rich domains. MUC7 has been shown to have antimicrobial activity.34
Figure 4.
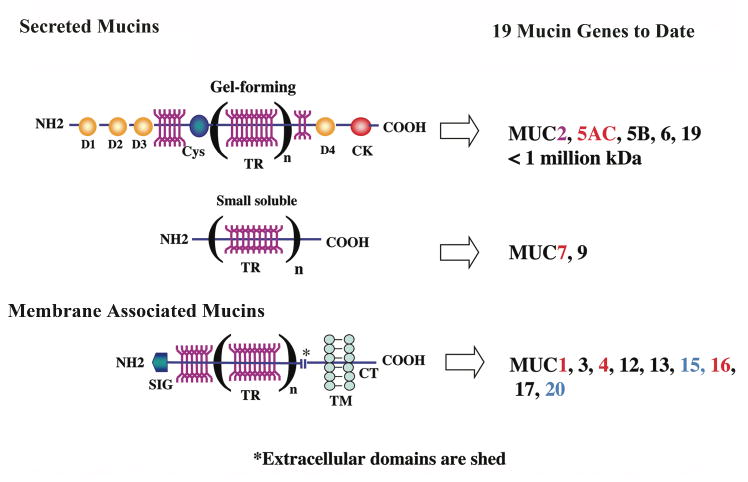
Diagram of the structure of the two classes of mucins—secreted and membrane associated. Nineteen mucins have been identified; those shown in red have been demonstrated to be expressed by the ocular surface epithelium by both in situ hybridization and immunohistochemistry. Those designated in blue have only been found in ocular surface epithelium by PCR and immunoblot analysis, and MUC2, in pink, has been found at low levels by PCR and immunoblot of tears. The characteristic common to both classes of mucins is the presence of tandem repeats (TR) of amino acids in their protein backbones, which may vary in number (n) between individuals (alleles are codominantly expressed). The tandem repeats are rich in serine and threonine, which are sites of O-glycosylation (YYY; in dark pink). Up to 80% of the mass of mucins can be O-glycans. Within the secreted category, two types of mucins are recognized—gel forming and small soluble. Gel-forming mucins are the largest proteins in the body; they have several D domains (D1, D2, etc.) and cysteine-rich domains (Cys, CK). The D domains allow for homomultimerization of individual gel-forming mucin molecules, which allows polymerization to form the viscous mucin gel. Membrane-associated mucins have a single membrane-spanning domain (TM) and a short cytoplasmic tail (CT). Their extracellular domains are constituitively shed into the tear film.9 (Figure after Argueso and Gipson, 2001.57)
Membrane-associated Mucins
To date 9-10 membrane-associated mucins have been identified. All have a short cytoplasmic tail and a single transmembrane domain, and most have a very large, highly O-glycosylated extracellular domain, which may extend up to 500 nm into the glycocalyx (Fig. 4). All wet-surfaced epithelia express several of these mucins, usually in a unique repertoire and with unique glycosylation characteristics.32 Although all probably contribute to the hydrophilic barrier at the epithelial surface, each may have unique functions as well. Recent data suggest that MUCs 1 and 4 have signaling capabilities through the cytoplasmic tail and extracellular EGF-like domains, respectively.32,35 MUC16 has the largest extracellular domain of the membrane-spanning mucins.
Human Ocular Surface Mucins and Their Alterations in Disease
Attempts to clone the H185 antigen (the mucin we found altered in dry eye30) prior to the mapping of the human genome, was a laborious time-consuming process for mucins since it required isolation and deglycosylation of sufficient mucin to make antibodies to the peptide core in order to screen a corneal epithelial cDNA library. During these attempts, human corneal and conjunctival RNA were screened by northern blot analysis for expression of the mucin genes known at that time, and message for several mucins, including the gel-forming mucin MUC5AC and two membrane-associated mucins—MUC1 and MUC4—were identified.36,37 Sites of expression of the mucin genes in ocular surface epithelia were localized by in situ hybridization techniques, demonstrating that MUC5AC is produced by the conjunctival goblet cell, that MUC1 is expressed all along the corneal and conjunctival epithelia, and that MUC4 is highly expressed in the conjunctiva, with decrease in message in the limbus and further decrease toward the central cornea (Fig. 5).
Figure 5.
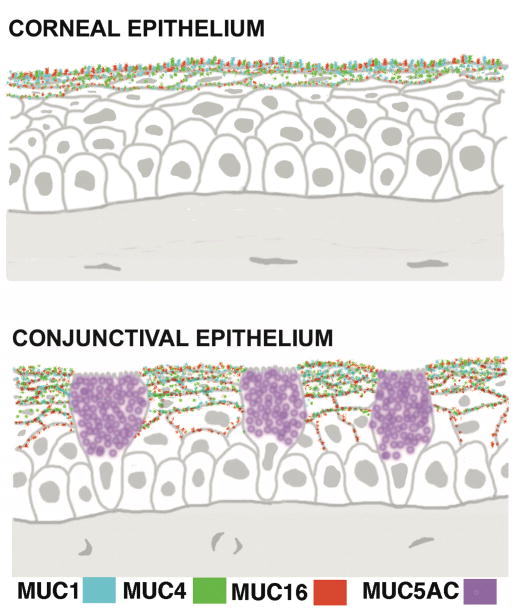
Diagram of sections of corneal and conjunctival epithelium showing the localization of expression of the membrane-spanning mucins MUC1, 4 and 16 in apical cells and the gel-forming mucin MUC5AC in goblet cells. There is very little expression of MUC4 in central epithelia. (Figure after Gipson, 2004.11)
Neither of the two membrane-associated mucins, however, was recognized by the H185 antibody. Attempts at cloning H815 failed in part because of the extreme difficulty in obtaining sufficient mucin from even large-scale, expanded primary cultures of corneal epithelium to allow characterization. Attempts to characterize the H185 mucin were then targeted toward isolation of large quantities of the mucin in order to obtain peptides from immunoprecipitated H185 for subsequent sequencing by conventional or mass spectrometry, followed by cloning from human gene databases. Production of telomerase immortalized corneal and conjunctival cell lines was arranged to obtain larger amounts of mucin starting material.38 Again, during the long laborious process to identify H185, additional work on characterizing MUC5AC expression was done.
After in situ hybridization demonstrated that conjunctival goblet cells express MUC5AC,36 (diagrammed in Fig. 5), peptide antibodies were developed to an unglycosylated N-terminus region of the molecule for use in semi-quantitative western blot and ELISA analyses to measure the amount of the mucin in tears of normal subjects and patients with drying ocular surface disease.39 By comparison to that of normal subjects, the amount of MUC5AC protein in tears of Sjögren's dry eye patients is significantly less. Similarly, there was significantly less MUC5AC message in conjunctival impression cytology samples from the same Sjögren's patients as measured by real time PCR.39 These data demonstrated the feasibility of measuring specific mucins within the tear fluid and demonstrated an alteration in amount of mucin with dry eye.
Western blotting of MUC5AC from agarose gel electrophoresis demonstrated that the size of MUC5AC in tears is smaller than that found in the goblet cells of the native conjunctival epithelium.9 The nature and significance of this alteration is unknown. Perhaps the mucin is modified to prevent gel formation, since an opaque viscous gel could scatter light and interfere with light transmission at the ocular surface.
During the attempts to clone the H185 mucin, new mucins were cloned. Their expression by the ocular surface epithelium was tested, and comparisons to the H185 mucin were made. In 2001, after 20 years of attempting to clone the CA125 antigen, also a mucin-like membrane-associated molecule and an ovarian tumor cell marker, Lloyd et al. were successful.40 CA125 antigen was identified as a membrane-associated mucin and was designated MUC16. Subsequently, by co-immunoprecipitation experiments with several MUC16 antibodies and the H185 antibody, as well as by histochemical colocalization experiments, the ocular surface H185 mucin was identified as MUC16 (Fig. 6).41 Recently, the epitope on MUC16 that the H185 antibody recognizes was identified as an O-acetylated sialic acid.42 Thus ended a 10-year effort to characterize the H185 antigen.
Figure 6.
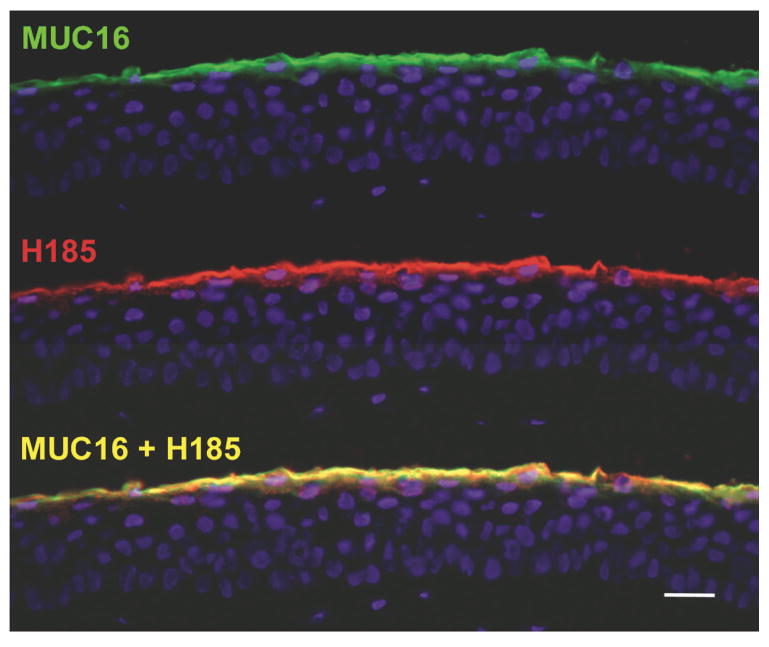
After 10 years of effort, H185 antibody was identified as a carbohydrate antigen on the membrane-associated mucin MUC16.42 The immunofluorescence micrographs of human corneal epithelium show colocalization of MUC16 and H185 antigen. This data and data showing co-immunoprecipitation of the same molecules with MUC16 and H185 antibodies demonstrate their identity. Bar = 25 μm.
During the quest to identify the H185 mucin, we were able to map the mucin gene expression profile of the ocular surface epithelia. We also demonstrated that there are two sources of mucins at the ocular surface, the conjunctival goblet cells and the apical cells of the stratified conjunctival and corneal epithelia. Jumblatt et al. confirmed the MUC5AC expression pattern, found a small amount of MUC2 expression by a yet to be determined cell type at the ocular surface, and found the small secreted mucin MUC 7 to be expressed by the acinar cells of the lacrimal gland.43,44 Recently, MUC16 expression was demonstrated in lacrimal gland ductal epithelium, accessory lacrimal glands, and nasolacrimal duct epithelium.45 Thus the entire ocular surface epithelium produces mucins, and the tears contain goblet cell mucin, as well as shed mucins produced by the corneal and conjunctival epithelia.9
Muc16: Regulation and Function at the Ocular Surface
Having identified the H185 antigen as MUC16, many questions arise regarding its alteration in dry eye. Is the alteration in H185 distribution on the ocular surface of dry eye patients a result of decreased expression of MUC16, increased shedding of its extracellular domain or, since the H185 epitope is a carbohydrate,42 altered glycosylation? Currently studies are ongoing to determine the mechanism of alteration of MUC16 in dry eye. To begin to answer these questions, methods have been developed to quantify membrane-associated mucin mRNA expression and protein in human conjunctival epithelium using impression cytology samples and real-time PCR; methods have also been developed to quantify mucin concentration in tear wash samples.9,39
Studies of membrane-associated mucin function and regulation (especially MUC16) are also ongoing. Major questions remain: “Do membrane-associated mucins each have unique functions?” and “How are the genes regulated at the ocular surface?” To answer these questions, immortalized human corneal and conjunctival epithelial cell lines optimized for mucin expression were developed.38 Several regulators of mucin gene expression have since been identified using these cell lines; serum upregulates expression of MUCs 1, 4 and 16; dexamethasone is a potent upregulator of MUC1 expression; vitamin A is required for MUC4 and MUC16 expression;46 and MUC16 regulation by vitamin A is mediated by secretory phospholipase A2.47 Interestingly, all three of these agents—serum, corticosteroids and vitamin A have been used in treatment of drying eye disease. (For review see Gipson et al., 2004.10)
The corneal epithelial cell line has also been useful in studies of the specific functions of MUC16. As in native human corneal epithelium, the mucin emanates from the tips of microplicae in differentiated cultured cells (Fig. 7). Since it is known that actin filaments insert into microplicae, and since the cytoplasmic tail of MUC1 has been shown to associate with the actin cytoskeleton, experiments were done to determine if the cytoplasmic tail of MUC16 associates with the actin cytoskeleton. Upon analysis of the amino acid sequence, a polybasic sequence in the cytoplasmic tail of the mucin was noted. This sequence is known to associate with the N terminus of ERMs (ezrin, radixin, moesin and merlin), a class of small proteins that link the cytoplasmic tail of membrane-spanning molecules to the actin cytoskeleton. Using pull down experiments, GST (Glutathione S-transferase)-tagged ERM proteins pulled down peptides to the cytoplasmic tail of MUC16. By comparison, the ERMs did not pull down peptides in which the polybasic sequence was substituted. These data indicated that MUC16 is linked to the actin cytoskeleton in the microplicae.48
Figure 7.
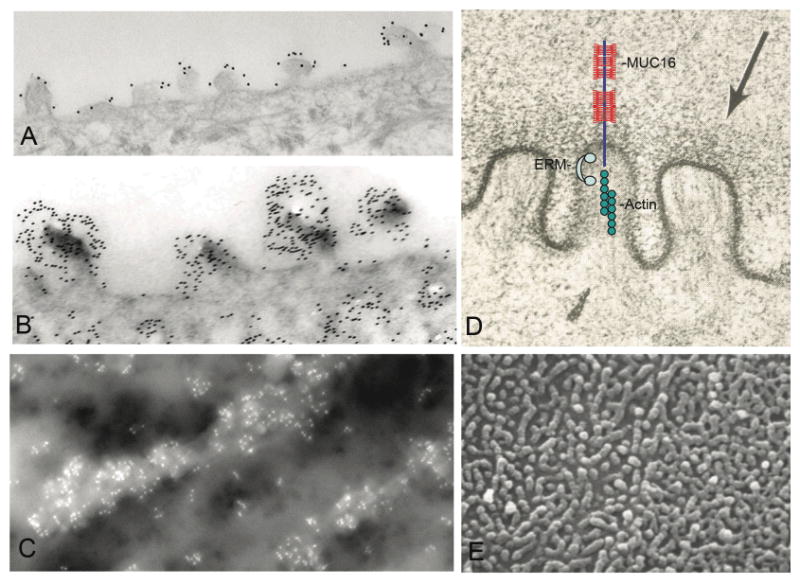
Electron micrographs of corneal epithelial surface microplicae. By transmission electron microscopy, MUC16 (A) and H185 (B) are localized by immunogold methods to the microplicae. Similar methods using field emission scanning electron microscopy were used to show that MUC16 is concentrated on the microplicae (C). Image in (D) summarizes a set of experiments48 in which we demonstrate that the cytoplasmic tail of MUC16 is tethered to the actin cytoskeleton through members of the actin linking family of proteins known as ERMs (ezrin, rodixin moesin, merlin). For reference, a lower magnification scanning electron microscopic view of surface microplicae is shown in (E).
To determine functions of MUC16 on corneal epithelial cells, siRNA methods to knock down expression of the mucin were employed. Greater adherence of Staphylococcus aureus was observed (data not shown), as was greater rose bengal dye penetrance (Fig. 8), indicating MUC16's role in barrier formation at the ocular surface.48
Figure 8.
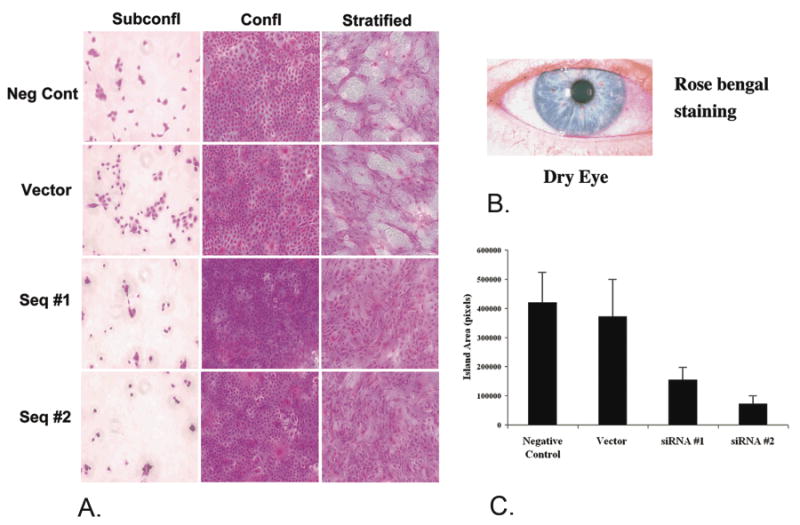
MUC16 provides a barrier to rose bengal dye as indicated by siRNA knockdown of the mucin in a human corneal limbal epithelial cell line. HCLE cells cultured to subconfluent or confluent stages do not express MUC16, and rose bengal dye penetrates all cells. After the cells are cultured seven additional days in serum continuing media, cells stratify and express MUC16. Islands of differentiated cells exclude the rose bengal dye. When MUC16 was knocked down by 80-90% as with Seq. #1 and Seq. #2, the islands that exclude the dye are diminished (A) as quantified in (C). For comparison, rose bengal stains areas of damaged ocular surface epithelium in dry eye (B). (For details of experiments see Blalock et al., In Press.48)
Animal Studies of Mucin Exression at the Ocular Surface
Many questions regarding mucin biology cannot easily answered using human samples or human epithelial cell culture. Thus, animal studies were needed. Homologues for the membrane-associated mucins 1 and 4, and the secreted mucin MUC5AC have been identified in mice and rats, and their presence at the ocular surface has been demonstrated.49-51 Several studies of the mucins in mice have provided information on their appearance during development as well as on their regulation. Developmentally, the appearance of membrane-spanning mucins correlates with eyelid opening at day 14 after birth, with goblet cell mucin MUC5AC appearing earlier—7 days after birth—the stage at which goblet cells first appear in the conjunctiva.52
Vitamin A deficiency is known in humans to cause drying and keratinization of ocular surface epithelium. To determine if specific mucins are lost in vitamin A deficiency, a rat model was used. The expression of rMuc4 and rMuc5AC was completely lost in rats fed a vitamin A-deficient diet, although, interestingly, Muc1 expression was not affected.53 These in vivo data correlate with data from studies of the effect of retinoic acid on regulation of MUC expression in human conjunctival epithelial cells in vitro, showing upregulation of MUCs 4 and 16 with vitamin A and lack of regulation of MUC1.47
In a mouse model of allergic conjunctivitis, repetitive application of allergens to the ocular surface induced a reduction of goblet cells and decreases in Muc5AC and Muc4 mRNA; however, recovery to naïve levels in both occurred after a 24-48 hour period, indicating a rapid recovery of the mucin system.51
The role that calcium ions play in providing cationic shielding to allow tight packaging of highly glycosylated Muc5AC in goblet cells was analyzed using vitamin D receptor knockout mice. The vitamin D receptor controls mineral ion homeostasis and calcium absorption in the intestine, and hypocalcemia occurs in animals in which the receptor is ablated.54 Goblet cells in the hypocalcemic mice, compared to controls, had less Muc5AC antibody binding, an altered granule morphology and less extractable mucin,55 indicating the importance of calcium ions in mucin storage within goblet cells.
Since dry eye in humans is predominant in postmenopausal women, the roles of estrogen and progesterone in the regulation of mucin gene expression at the ocular surface were investigated in mice. The roles of hormones in regulating ocular surface mucins were determined using ovariectomized mice in which estrogen and/or progesterone were replaced. Estrogen and/or progestin were found to have no effect on regulation of mucins in ocular surface epithelium, whereas the hormones were involved in mucin regulation in reproductive tract epithelia.56
Summary
In summary, the search for the identity of a mucin altered in dry eye has led us down a path of discovery that was eventually successful. The studies that were spawned by this search have significantly amplified our understanding of the Ocular Surface System. We have learned about the character of mucins at the ocular surface, how they function and how they are regulated. Their alterations in dry eye have been documented, and methods to understand the alteration developed.
Acknowledgments
It has been an extraordinarily exciting, interesting and rewarding journey. The journey was made possible and truly enjoyable by a group of talented fellows, staff and students involved in the work. Without their efforts, enthusiasm, insights, and skills, the research that led to the Friedenwald award would not have been possible: Pablo Argüeso has been an exceptional fellow and now colleague. I am particularly grateful to the long-term outstanding service of my loyal staff members Sandra Spurr-Michaud, Ann Tisdale, and Gale Unger, who have served with our group, 26, 27 and 17 years, respectively. I am also extraordinarily appreciative of the support of my mentors—Drs. Lynette Feeney-Burns, the late Robert Burns, Claes Dohlman and my colleagues at the Schepens Eye Research Institute. Lastly, but most importantly, my husband and fellow scientist, Dr. Henry Keutmann, has been the most encouraging and supportive spouse one could have—I am deeply grateful to him.
Supported by National Eye Institute grant EY03306 to IKG.
Footnotes
Disclosure: None
Potential reviewers: Linda Hazlett, Dept. of Ophthalmology, Wayne State, lhazlett@med.wayne.edu; Stephen Sugrue, Dept. of Cell Biology, University of Florida, sugrue@anatomy.med.ufl.edu; James Chodosh, University of Oklahoma Health Science Ctr., james-chodosh@ouhsc.edu; Dimitri Azar, Dept. of Ophthalmology, University of Illinois at Chicago, dazar@uic.edu
References
- 1.Research in dry eye: Report of the Research Subcommittee of the International Dry Eye WorkShop in: Report of the International Dry Eye WorkShop (DEWS) Ocul Surf. 2007;5:179–193. doi: 10.1016/s1542-0124(12)70086-1. [DOI] [PubMed] [Google Scholar]
- 2.Williams K, Watsky M. Gap junctional communication in the human corneal endothelium and epithelium. Curr Eye Res. 2002;25:29–36. doi: 10.1076/ceyr.25.1.29.9964. [DOI] [PubMed] [Google Scholar]
- 3.Walcott B, Moore LC, Birzgalis A, et al. Role of gap junctions in fluid secretion of lacrimal glands. Am J Physiol Cell Physiol. 2002;282:C501–507. doi: 10.1152/ajpcell.00004.2001. [DOI] [PubMed] [Google Scholar]
- 4.Kinoshita S, Adachi W, Sotozono C, et al. Characteristics of the human ocular surface epithelium. Prog Retin Eye Res. 2001;20:639–673. doi: 10.1016/s1350-9462(01)00007-6. [DOI] [PubMed] [Google Scholar]
- 5.Paulsen FP, Schaudig U, Thale AB. Drainage of tears: impact on the ocular surface and lacrimal system. Ocul Surf. 2003;1:180–191. doi: 10.1016/s1542-0124(12)70013-7. [DOI] [PubMed] [Google Scholar]
- 6.Stern ME, Beuerman RW, Fox RI, Gao J, Mircheff AK, Pflugfelder SC. The pathology of dry eye: the interaction between the ocular surface and lacrimal glands. Cornea. 1998;17:584–589. doi: 10.1097/00003226-199811000-00002. [DOI] [PubMed] [Google Scholar]
- 7.Fang Y, Choi D, Searles RP, Mathers WD. A time course microarray study of gene expression in the mouse lacrimal gland after acute corneal trauma. Invest Ophthalmol Vis Sci. 2005;46:461–469. doi: 10.1167/iovs.04-0677. [DOI] [PubMed] [Google Scholar]
- 8.Report of the International Dry Eye WorkShop (DEWS) Ocul Surf. 2007;5:67–202. doi: 10.1016/s1542-0124(12)70086-1. [DOI] [PubMed] [Google Scholar]
- 9.Spurr-Michaud S, Argueso P, Gipson I. Assay of mucins in human tear fluid. Exp Eye Res. 2007;84:939–950. doi: 10.1016/j.exer.2007.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gipson IK, Hori Y, Argueso P. Character of ocular surface mucins and their alteration in dry eye disease. Ocul Surf. 2004;2:131–148. doi: 10.1016/s1542-0124(12)70149-0. [DOI] [PubMed] [Google Scholar]
- 11.Gipson IK. Distribution of mucins at the ocular surface. Exp Eye Res. 2004;78:379–388. doi: 10.1016/s0014-4835(03)00204-5. [DOI] [PubMed] [Google Scholar]
- 12.Gipson I, Argueso P. The role of mucins in the function of the corneal and conjunctival epithelia. Int Rev Cytol. 2003;231:1–49. doi: 10.1016/s0074-7696(03)31001-0. [DOI] [PubMed] [Google Scholar]
- 13.Gipson IK, Grill SM, Spurr SJ, Brennan SJ. Hemidesmosome formation in vitro. J Cell Biol. 1983;97:849–857. doi: 10.1083/jcb.97.3.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fujikawa LS, Foster CS, Gipson IK, Colvin RB. Basement membrane components in healing rabbit corneal epithelial wounds: Immunofluorescence and ultrastructural studies. J Cell Biol. 1984;98:128–138. doi: 10.1083/jcb.98.1.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gipson IK, Kiorpes TC, Brennan SJ. Epithelial sheet movement: effects of tunicamycin on migration and glycoprotein synthesis. Dev Biol. 1984;101:212–220. doi: 10.1016/0012-1606(84)90131-3. [DOI] [PubMed] [Google Scholar]
- 16.Trinkaus-Randall V, Gipson IK. Role of calcium and calmodulin in hemidesmosome formation in vitro. J Cell Biol. 1984;98:1565–1571. doi: 10.1083/jcb.98.4.1565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zieske JD, Gipson IK. Protein synthesis during corneal epithelial wound healing. Invest Ophthalmol Vis Sci. 1986;27:1–7. [PubMed] [Google Scholar]
- 18.Gipson IK, Spurr-Michaud SJ, Tisdale AS. Hemidesmosomes and anchoring fibril collagen appear synchronously during development and wound healing. Dev Biol. 1988;126:253–262. doi: 10.1016/0012-1606(88)90136-4. [DOI] [PubMed] [Google Scholar]
- 19.Zieske JD, Bukusoglu G, Gipson IK. Enhancement of vinculin synthesis by migrating stratified squamous epithelium. J Cell Biol. 1989;109:571. doi: 10.1083/jcb.109.2.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Azar DT, Spurr-Michaud SJ, Tisdale AS, Gipson IK. Decreased penetration of anchoring fibrils into the diabetic stroma: A morphometric analysis. Arch Ophthalmol. 1989;107:1520–1523. doi: 10.1001/archopht.1989.01070020594047. [DOI] [PubMed] [Google Scholar]
- 21.Stepp MA, Spurr-Michaud S, Tisdale A, Elwell J, Gipson IK. Alpha 6 beta 4 integrin heterodimer is a component of hemidesmosomes. Proc Natl Acad Sci U S A. 1990;87:8970–8974. doi: 10.1073/pnas.87.22.8970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Azar DT, Spurr-Michaud SJ, Tisdale AS, Moore MB, Gipson IK. Reassembly of the corneal epithelial adhesion structures following human epikeratoplasty. Arch Ophthalmol. 1991;109:1279–1284. doi: 10.1001/archopht.1991.01080090105032. [DOI] [PubMed] [Google Scholar]
- 23.Azar DT, Spurr-Michaud SJ, Tisdale AS, Gipson IK. Altered epithelial-basement membrane interactions in diabetic corneas. Arch Ophthalmol. 1992;110:537–540. doi: 10.1001/archopht.1992.01080160115045. [DOI] [PubMed] [Google Scholar]
- 24.Danjo Y, Gipson IK. Actin “purse string” filaments are anchored by E-cadherin-mediated adherens junctions at the leading edge of the epithelial wound, providing coordinated cell movement. J Cell Sci. 1998;111:3323–3332. doi: 10.1242/jcs.111.22.3323. [DOI] [PubMed] [Google Scholar]
- 25.Danjo Y, Gipson IK. Specific transduction of the leading edge cells of migrating epithelia demonstrates that they are replaced during healing. Exp Eye Res. 2002;74:199–204. doi: 10.1006/exer.2001.1115. [DOI] [PubMed] [Google Scholar]
- 26.Gipson IK, Anderson RA. Effect of lectins on migration of the corneal epithelium. Invest Ophthalmol Vis Sci. 1980;19:341. [PubMed] [Google Scholar]
- 27.Gipson IK, Kiorpes TC. Epithelial sheet movement: protein and glycoprotein synthesis. Dev Biol. 1982;92:259–262. doi: 10.1016/0012-1606(82)90170-1. [DOI] [PubMed] [Google Scholar]
- 28.Gipson IK, Spurr-Michaud SJ, Tisdale AS, Kublin C, Cintron C, Keutmann H. Stratified squamous epithelia produce mucin-like glycoproteins. Tissue Cell. 1995;27:397–404. doi: 10.1016/s0040-8166(95)80060-3. [DOI] [PubMed] [Google Scholar]
- 29.Watanabe H, Fabricant M, Tisdale AS, Spurr-Michaud SJ, Lindberg K, Gipson IK. Human corneal and conjunctival epithelia produce a mucin-like glycoprotein for the apical surface. Invest Ophthalmol Vis Sci. 1995;36:337–344. [PubMed] [Google Scholar]
- 30.Danjo Y, Watanabe H, Tisdale AS, et al. Alteration of mucin in human conjunctival epithelia in dry eye. Invest Ophthalmol Vis Sci. 1998;39:2602–2609. [PubMed] [Google Scholar]
- 31.Gendler SJ, Spicer AP. Epithelial mucin genes. Annu Rev Physiol. 1995;57:607–634. doi: 10.1146/annurev.ph.57.030195.003135. [DOI] [PubMed] [Google Scholar]
- 32.Hollingsworth MA, Swanson BJ. Mucins in cancer: Protection and control of the cell surface. Nat Rev Cancer. 2004;4:45–60. doi: 10.1038/nrc1251. [DOI] [PubMed] [Google Scholar]
- 33.Moniaux N, Escande F, Porchet N, Aubert JP, Batra SK. Structural organization and classification of the human mucin genes. Front Biosci. 2001;6:D1192–1206. doi: 10.2741/moniaux. [DOI] [PubMed] [Google Scholar]
- 34.Situ H, Wei G, Smith CJ, Mashhoon S, Bobek LA. Human salivary MUC7 mucin peptides: effect of size, charge and cysteine residues on antifungal activity. Biochem J. 2003;375:175–182. doi: 10.1042/BJ20030779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Singh PK, Hollingsworth MA. Cell surface-associated mucins in signal transduction. Trends Cell Biol. 2006;16:467–476. doi: 10.1016/j.tcb.2006.07.006. [DOI] [PubMed] [Google Scholar]
- 36.Inatomi T, Spurr-Michaud S, Tisdale AS, Zhan Q, Feldman ST, Gipson IK. Expression of secretory mucin genes by human conjunctival epithelia. Invest Ophthalmol Vis Sci. 1996;37:1684–1692. [PubMed] [Google Scholar]
- 37.Inatomi T, Spurr-Michaud S, Tisdale AS, Gipson IK. Human corneal and conjunctival epithelia express MUC1 mucin. Invest Ophthalmol Vis Sci. 1995;36:1818–1827. [PubMed] [Google Scholar]
- 38.Gipson IK, Spurr-Michaud S, Argueso P, Tisdale A, Ng TF, Russo CL. Mucin gene expression in immortalized human corneal-limbal and conjunctival epithelial cell lines. Invest Ophthalmol Vis Sci. 2003;44:2496–2506. doi: 10.1167/iovs.02-0851. [DOI] [PubMed] [Google Scholar]
- 39.Argueso P, Balaram M, Spurr-Michaud S, Keutmann HT, Dana MR, Gipson IK. Decreased levels of the goblet cell mucin MUC5AC in tears of patients with Sjögren's syndrome. Invest Ophthalmol Vis Sci. 2002;43:1004–1011. [PubMed] [Google Scholar]
- 40.Lloyd KO, Yin BW. Synthesis and secretion of the ovarian cancer antigen CA 125 by the human cancer cell line NIH:OVCAR-3. Tumour Biol. 2001;22:77–82. doi: 10.1159/000050600. [DOI] [PubMed] [Google Scholar]
- 41.Argueso P, Spurr-Michaud S, Russo CL, Tisdale A, Gipson IK. MUC16 mucin is expressed by the human ocular surface epithelia and carries the H185 carbohydrate epitope. Invest Ophthalmol Vis Sci. 2003;44:2487–2495. doi: 10.1167/iovs.02-0862. [DOI] [PubMed] [Google Scholar]
- 42.Argueso P, Sumiyoshi M. Characterization of a carbohydrate epitope defined by the monoclonal antibody H185: sialic acid O-acetylation on epithelial cell-surface mucins. Glycobiology. 2006;16:1219–1228. doi: 10.1093/glycob/cwl041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jumblatt MM, McKenzie RW, Steele PS, Emberts CG, Jumblatt JE. MUC7 Expression in the human lacrimal gland and conjunctiva. Cornea. 2003;22:41–45. doi: 10.1097/00003226-200301000-00010. [DOI] [PubMed] [Google Scholar]
- 44.McKenzie RW, Jumblatt JE, Jumblatt MM. Quantification of MUC2 and MUC5AC transcripts in human conjunctiva. Invest Ophthalmol Vis Sci. 2000;41:703–708. [PubMed] [Google Scholar]
- 45.Jager K, Wu G, Sel S, Garreis F, Brauer L, Paulsen FP. MUC16 in the lacrimal apparatus. Histochem Cell Biol. 2007;127:433–438. doi: 10.1007/s00418-006-0246-6. [DOI] [PubMed] [Google Scholar]
- 46.Hori Y, Spurr-Michaud S, Russo C, Argueso P, Gipson I. Differential regulation of membrane-associated mucins in the human ocular surface epithelium. Invest Ophthalmol Vis Sci. 2004;45:114–122. doi: 10.1167/iovs.03-0903. [DOI] [PubMed] [Google Scholar]
- 47.Hori Y, Spurr-Michaud SJ, Russo CL, Argueso P, Gipson IK. Effect of retinoic acid on gene expression in human conjunctival epithelium: secretory phospholipase A2 mediates retinoic acid induction of MUC16. Invest Ophthalmol Vis Sci. 2005;46:4050–4061. doi: 10.1167/iovs.05-0627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Blalock T, Spurr-Michaud S, Tisdale A, et al. Functions of MUC16 in corneal epithelial cells. Invest Ophthalmol Vis Sci. 2007;48:4509–4518. doi: 10.1167/iovs.07-0430. [DOI] [PubMed] [Google Scholar]
- 49.Inatomi T, Tisdale AS, Zhan Q, Spurr-Michaud S, Gipson IK. Cloning of rat Muc5AC mucin gene: Comparison of its structure and tissue distribution to that of human and mouse homologues. Biochem Biophys Res Commun. 1997;236:789–797. doi: 10.1006/bbrc.1997.7051. [DOI] [PubMed] [Google Scholar]
- 50.Danjo Y, Hazlett LD, Gipson IK. C57BL/6 mice lacking Muc1 show no ocular surface phenotype. Invest Ophthalmol Vis Sci. 2000;41:4080–4084. [PubMed] [Google Scholar]
- 51.Kunert KS, Keane-Myers AM, Spurr-Michaud S, Tisdale AS, Gipson IK. Alteration in goblet cell numbers and mucin gene expression in a mouse model of allergic conjunctivitis. Invest Ophthalmol Vis Sci. 2001;42:2483–2489. [PubMed] [Google Scholar]
- 52.Tei M, Moccia R, Gipson IK. Developmental expression of mucin genes ASGP (rMuc4) and rMuc5AC by the rat ocular surface epithelium. Invest Ophthalmol Vis Sci. 1999;40:1944–1951. [PubMed] [Google Scholar]
- 53.Tei M, Spurr-Michaud SJ, Tisdale AS, Gipson IK. Vitamin A deficiency alters the expression of mucin genes by the rat ocular surface epithelium. Invest Ophthalmol Vis Sci. 2000;41:82–88. [PubMed] [Google Scholar]
- 54.Li YC, Amling M, Pirro AE, et al. Normalization of mineral ion homeostasis by dietary means prevents hyperparathyroidism, rickets, and osteomalacia, but not alopecia in vitamin D receptor-ablated mice. Endocrinology. 1998;139:4391–4396. doi: 10.1210/endo.139.10.6262. [DOI] [PubMed] [Google Scholar]
- 55.Paz HB, Tisdale AS, Danjo Y, Spurr-Michaud SJ, Argueso P, Gipson IK. The role of calcium in mucin packaging within goblet cells. Exp Eye Res. 2003;77:69–75. doi: 10.1016/s0014-4835(03)00084-8. [DOI] [PubMed] [Google Scholar]
- 56.Lange C, Fernandez J, Shim D, Spurr-Michaud S, Tisdale A, Gipson IK. Mucin gene expression is not regulated by estrogen and/or progesterone in the ocular surface epithelia of mice. Exp Eye Res. 2003;77:59–68. doi: 10.1016/s0014-4835(03)00064-2. [DOI] [PubMed] [Google Scholar]
- 57.Argueso P, Gipson IK. Epithelial mucins of the ocular surface: structure, biosynthesis and function. Exp Eye Res. 2001;73:281–289. doi: 10.1006/exer.2001.1045. [DOI] [PubMed] [Google Scholar]



