Summary
Many signaling proteins change their location within cells in response to external stimuli. In photoreceptors, this phenomenon is remarkably robust. The G protein of rod photoreceptors and rod transducin concentrates in the outer segments (OS) of these neurons in darkness. Within ~30 minutes after illumination, rod transducin redistributes throughout all of the outer and inner compartments of the cell. Visual arrestin concurrently relocalises from the inner compartments to become sequestered primarily within the OS. In the past several years, the question of whether these proteins are actively moved by molecular motors or whether they are redistributed by simple diffusion has been extensively debated. This review focuses on the most essential works in the area and concludes that the basic principle driving this protein movement is diffusion. The directionality and light dependence of this movement is achieved by the interactions of arrestin and transducin with their spatially restricted binding partners.
Keywords: G protein-mediate signaling, transducin, arrestin, diffusion, photoreceptors, rod, cone, retina, rhodopsin
INTRODUCTION
Photoreceptor Cells and Signaling Molecules
Photoreceptors are terminally differentiated sensory neurons that have a very distinct morphology. These ~100-µm-long cells consist of 1) a dendritic structure called the outer segment (OS), 2) the inner segment (IS), 3) the nucleus [the photoreceptor nuclei form a distinct layer in the retina, called the outer nuclear layer (ONL)] and 4) an axon ending with a glutaminergic synapse. Phototransduction occurs in the OS in which the visual pigment rhodopsin is localized in a stack of membrane discs that fill essentially the entire volume of the OS. In rods, these discs originate as invaginations of the plasma membrane at the base of the OS, then detach from it in a process akin to endocytosis and continuously move along the length of the OS. In a few days, they reach the distal end and are phagocytozed by cells of the retinal pigment epithelium. Rhodopsin, and other members of the phototransduction pathway are synthesized in the IS, which contains ribosomes, mitochondria and other organelles.
Rhodopsin is a G protein-coupled receptor, which activates the heterotrimeric G protein, transducin, by catalyzing the binding of GTP to its α a subunit (Gαt). Gαt dissociates from its Gβγ subunit and then stimulates an effector enzyme, cGMP phosphodiesterase. The resulting hydrolysis of cGMP leads to the closure of cGMP-gated ion channels in the OS plasma membrane, which hyperpolarizes the cell and suppresses the glutamate release at the synaptic terminal. The active state of this pathway is terminated by the following molecular mechanisms. Rhodopsin is inactivated by phosphorylation by rhodopsin kinase (GRK1) and binding of a soluble protein arrestin, which blocks rhodopsin-transducin interaction. The G protein is inactivated by GTP hydrolysis and reassociation of the inactive Gαβγ complex. GTP hydrolysis is accelerated by a GTPase-activating protein, RGS9. Cyclic GMP is resynthesized by a retinal guanylate cyclase, which is regulated by Ca2+. To achieve high efficiency of photon capture, the OS contains a high concentration (~3 mM) of rhodopsin; other signaling proteins are also expressed at concentrations higher than their homologues in nonphotoreceptor cells [reviews: (1–3)].
In addition to their ability to convert photon capture into an electric signal, photoreceptors also have the ability to adapt to different levels of illumination. They are more sensitive to light of after a prolonged exposure to darkness. In the presence of background illumination, the sensitivity is reduced by several orders of magnitude, which allows the visual system to remain responsive to the environment. Rods are more sensitive than cones and are responsible for vision in dim light, but saturate and do not function in daylight. Cones function well in bright light and essentially do not saturate. Rod and cones have distinct morphology and express different gene products for opsins, transducin subunits, GRKs, arrestins, and phosphodiesterases.
Light-dependent Relocalization of Arrestin and Transducin
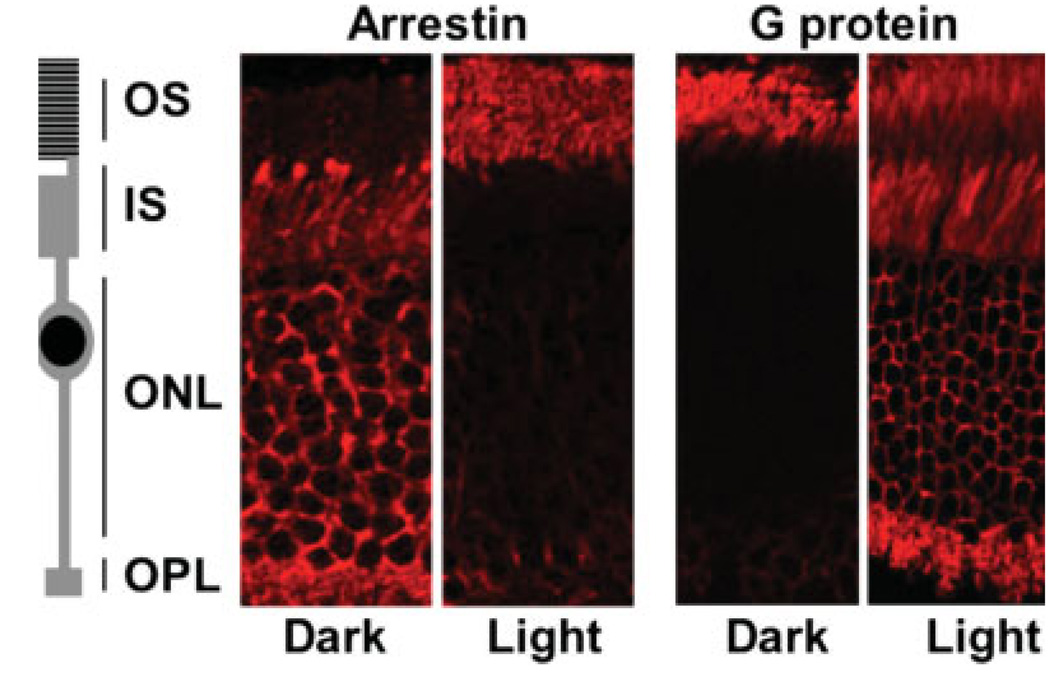
Two decades ago, several researchers observed a dramatic change in localization of arrestin and transducin in rods (4–6) (illustrated in Fig. 1). In dark-adapted cells, arrestin localized primarily in the inner compartments and redistributed to the OS in light. In contrast, transducin concentrated in the OS in darkness and dispersed throughout the cell in light. These original discoveries came back into the focus of vision researchers in 2002, when Arshavsky and colleagues proved that this phenomenon represents protein migration rather than an artifact related to epitope masking or an alternative process such as protein degradation and resynthesis (7). The crucial technical approach in that landmark work was tangential sectioning of the retina. Flattened frozen retinas were sliced on a microtome perpendicular to the longitudinal axis of the rods, and the sections corresponding to the portions of the OS and other compartments were analyzed by western blot. Because the transferred proteins are denatured and therefore equally available to the antibodies, this method potentially permits better quantitative analysis than in situ immunostaining. Sokolov et al. also correlated transducin departure from the OS with the concurrent reduction of rod light sensitivity, hypothesizing that the reduction of transducin concentration in the OS contributes to light adaptation in rods (7). This work sets the stage for further questions. First, what is the mechanism that drives this robust protein redistribution—is it active transport or diffusion? Second, what is the biological importance of protein redistribution in photoreceptors?
Figure 1.
Light-dependent redistribution of arrestin and transducin. The schematic drawing of a rod cell is adjacent to immunofluorescence images of retinal slices immunostained for arrestin and Gαt. The retinas were obtained from light- or dark-adapted mice. OS, outer segments containing the rhodopsin-harboring discs; IS, inner segments; ONL, outer nuclear layer; OPL, outer plexiform layer, which contains photoreceptor synaptic termini.
Recent reviews, discussing potential mechanisms and significance of this movement, analyzed existing literature and evaluated the strengths of both the molecular motor and diffusion hypotheses (8–10). However, until recently, there was not enough experimental evidence available to determine which of the two hypotheses was more likely to be correct. In this article, we discuss important recent investigations of the light-induced movement of arrestin and transducin in vertebrate photoreceptors.
ACTIVE TRANSPORT OR DIFFUSION?
The Relationship Between Translocation and Signal Transduction
Several studies have investigated how light-induced protein redistribution is influenced by the disruption of signal transduction. The genes encoding rhodopsin kinase, arrestin (11, 12), rod transducin α subunit (12), guanylate cyclase (13) and RGS9 (14, 15) each have been inactivated by homologous recombination. The influence of these mutations on protein redistribution in rods has been investigated thoroughly. Transgenic studies that altered phosphorylation sites on rhodopsin (11), that led to the expression of cone transducin in rods (15) and that manipulated the nucleotide-binding status of rod Gαt (14) also have been evaluated for their influence on protein redistribution. For example, it was shown that a GTPase-deficient Gαt subunit (Q200L mutant) distributed throughout the rod cell even after many days in darkness. Accordingly, the increased lifetime of the GTP-bound state of transducin in the RGS9 knockout also facilitates its translocation (14, 15), whereas overexpression of RGS9 impeded movement of rod transducin from the OS (16). The effect of the RGS9 knockout and the GTP-ase-deficiency mutations are similar to the effect of increasing light intensity. In simplistic terms, intensifying signaling leads to more robust translocation.
The studies in which genetic manipulations were used to investigate relationships between signaling and protein distribution established that the localization of transducin and arrestin are influenced by phototransduction. However, the underlying mechanism that determines localization of these proteins could not be gleaned from those studies because altered signaling could influence either the ability of a protein to diffuse or its interactions with active transport machinery.
What Is the Role of Molecular Motors?
Marszalek et al. used CreloxP mutagenesis to prevent expression of the kinesin-II subunit, KIF3A, in mouse photoreceptors (17). Both arrestin and opsin accumulated within the inner segments of the mutant photoreceptors. The localization of transducin and the processing of opsin were unaffected. This study suggested that the active transport could be involved in arrestin migration. Similar findings were reported for trafficking of arrestin and Gαq in drosophila (18, 19). In that case, trafficking was disrupted in flies that lacked NINAC, a form of myosin that is normally enriched in eyes. These reports have been interpreted as an indication that active transport is needed for protein redistribution in photoreceptors, for example, “arrestin is carried into the light-sensitive microvilli by phosphoinositide-enriched vesicles driven by a myosin motor” (9).
Does the Cytoskeleton Play an Important Role in Protein Redistribution in Photoreceptors?
The second component that could be essential for active transport is the “tracks” on which molecular motors move their cargo: microtubules and actin filaments. Indeed, there appears to be a relationship between the cytoskeleton and light-dependent redistribution of both arrestin and transducin. First, McGinnis et al. exploited the relatively large dimensions of rods in Xenopus laevis retinas to study the subcellular localization of arrestin by confocal microscopy. They demonstrated that a fraction of arrestin that does not move to the inner compartments in dark is colocalized with microtubules in the connecting cilium, the axoneme, and the microtubules associated with the disc incisures (20). In another study, the Smith laboratory incubated live tadpoles in water-containing drugs known to affect the cytoskeleton. They reported that thiabendazole, a drug known to perturb microtubules, slowed translocation of both transducin and arrestin from OS to IS. Cytochalasin D and latrunculin B, which disrupt microfilaments, selectively slowed transducin movement from inner to outer segments (21). Although this allowed Peterson et al. to conclude that “ light-driven translocation of visual proteins at least partially relies on an active motor-driven mechanism, ” these results should be interpreted with caution. Purified arrestin was shown to bind to microtubules directly, without participation of molecular motors (22, 23). Therefore, it is possible that the disruption of microtubes did not disrupt active transport, but rather, prevented arrestin sequestration by microtubules in the inner compartments. Furthermore, subjecting live tadpoles to days in the drugs could lead to changing the localization of other proteins, subtle changes in cell morphology or other indirect effects.
Nonetheless, the effects of molecular motor knockouts and manipulations with cytoskeleton strengthened the idea that arrestin and transducin move by active transport. The existence of a novel pathway that engages active transport was suggested in a number of articles [e.g.,(11, 24)]. However, based on the theoretical calculations of the rate and amount of protein to be carried across the photoreceptor compartments pointed to a problem: such a transport would have to be nearly 1,000 times more efficient than transport of rhodopsin and, as a matter of fact, it would have to be significantly more efficient than any known active transport mechanism (10).
Is ATP Required for Protein Redistribution in Photoreceptors?
In addition to a motor protein and the cytoskeleton, which are the “engine” and the “track” in the active transport, this process requires “fuel:” metabolic energy in the form of ATP. If molecular motors drive arrestin and transducin, this process should be halted in the absence of ATP. However, this is not the case (15, 25). Both arrestin and transducin can relocalize in a light-dependent manner to and from the OS in photoreceptors deprived of metabolic energy sources. To deplete retinas of ATP, explanted retinas or eyecups were incubated in a medium lacking glucose and supplemented with deoxyglucose and potassium cyanide. The completeness of ATP depletion under these conditions was demonstrated by three independent assays: (i) direct measurement of ATP concentration, (ii) lack of rhodopsin phosphorylation, and (iii) impairment of rod transducin movement from the OS to IS. This later observation was due to the depletion of GTP, which requires ATP to be synthesized. The extent and kinetics of transducin and arrestin redistribution in ATP-depleted cells were similar to those observed in live animals. Taken together, these results argue against an essential role of active transport or any other process requiring metabolic energy, in protein redistribution in photoreceptors. The findings point to a mechanism solely based on diffusion, the process utilizing the “free” energy of Brownian motion.
THE “SINKS”
It is obvious that a random process such as diffusion by itself cannot support a unidirectional movement of a protein. However, the directionality of protein migration can be achieved if the protein is trapped by a binding site (“sink”) localized in a specific cellular compartment. The migrating protein will relocalize in the direction of its immobile-binding partner along the concentration gradient created by the interaction. The protein must be soluble while in transit, but insoluble (e.g., membrane bound) at the destination. Both rod transducin and arrestin switch from soluble to membrane-bound states depending on photoreceptor activation. The sink must be as abundant as the migrating protein, and the affinity of the sink to the migrating protein must be regulated by light. Rhodopsin and disc membranes were suggested as the OS sinks of arrestin and transducin, respectively.
Transducin
Rod transducin in its inactive heterotrimeric state has a high affinity for membranes. Overall, the complex has two lipid modifications: the N-acyl of Gαt and S-prenyl-of Gγt. Upon light-induced dissociation into Gα and Gβγ, each entity has only one lipid anchor and readily detaches from the membrane. The dissociated state also can be induced in the dark by a synthetic peptide that binds to Gβ and competes with Gα-GDP-for binding to Gβγ. The peptide induces separation of the subunits and causes them to dissociate from membranes and redistribute throughout the entire photoreceptor cell. This occurs even without the activation of transducin, that is, without GTP binding (15). Thus, subunit dissociation and subsequent detachment from the membranes are the necessary and sufficient events that are responsible for the redistribution on of rod transducin. Obviously, transducin can reanchor to the disc when its active state is terminated by GTP hydrolysis, and so it appears to require a “ threshold of activation” (16). If transducin is locked into the activated state by the Q200L mutation (14) or by GTPγS binding (15), it cannot return to the OS even in darkness.
Cone transducin does not redistribute from the OS even in direct sunlight when the bulk of cone pigment is bleached. It primarily stays in the OS even upon GTP GTPγS binding. Accordingly, activated cone transducin remains associated with the membranes. The differences between cone and rod transducins have been explained by the stronger affinity of Gβγ for Gαt in the cone G protein, even in the GTP GTPγS-bound form (15). A more recent report showing that in rat cone transducin can be induced to distribute at a high-light intensity (26). At the moment, the reason for the difference between the observations in mice (13, 15, 24) and fish (22, 27) versus rat is unknown. It has not been tested whether rat cone transducin can detach from the membranes upon activation, but that is certainly what one should expect if it diffuses to the IS upon activation.
Arrestin
Arrestin is a soluble protein that, according to the suggested model (25), shuttles between two sinks: rhodopsin in the OS and microtubules in the inner compartments. Microtubules are particularly abundant in the inner segment (28). Biochemical fractionation of the retina showed that more arrestin is bound to tubulin in dark than in light. Furthermore, addition of rhodopsin-containing vesicles facilitated arrestin dissociation from an isolated cytoskeleton fraction (25). Another experiment supporting the presence of binding sites for arrestin in the inner compartments showed that an EGFP fusion of arrestin concentrated in the inner compartments to a greater extent than EGFP alone (29).
The knockout of a motor protein causes mislocalization of arrestin but only indirectly. Rhodopsin and membranes are not delivered normally to the OS in these mutant photoreceptors (17). The normal distribution of arrestin is disrupted because the sink for binding arrestin in the outer segment is mislocalized.
Does a protein require a sink in the inner compartment to move from the OS? The volume of cytosol is higher in the inner compartments than in the OS in which most of the space is taken by the discs. Therefore, once the protein is free to diffuse, it would be expected to disperse and equalize its concentration throughout the entire rod cytosol. Based on this notion, it might seem that an inner compartment sink is not necessary. However, such a sink would decrease the cytosol volume necessary to accommodate the protein arriving from the OS. A small cell volume would be advantageous because it increases the density of photoreceptors in the retina, which allows higher resolution. Therefore, it is tempting to speculate that a sink in the inner compartments is more important in vertebrates than in insects in which the volume of microvili is much less than that of the rest of the cell. It has been suggested that the “second sink” for arrestin is tubulin (25). For transducin, the identity and the role of sink in the inner segment are less clear. Several candidates have been suggested, including centrin (30, 31), cytoskeleton (21), LGN (32, 33) and phosducin (34). However, direct experimental evidence that evaluates these ideas is not yet available.
It is likely that diffusion limited by interaction with sinks is a common process occurring in various types of cells and processes. For example, diffusion would rapidly and effectively carry soluble protein kinases across the cell toward their emerging substrates and anchoring proteins. It is also worth noting that diffusion is not limited to cytosolic molecules. Membrane proteins can diffuse within the two-dimensional space of the membrane toward a binding partner immobilized, for example, in a synapse.
ENERGY, SPEED, AND SPACE
Metabolic Energy
The diffusion model implies that virtually unlimited quantities of proteins can relocalize without energy consumption. However, to make this process work, cells must continuously spend energy on (i) maintaining cell polarity (e.g., outer segments), (ii) nonrandom localization of the sinks, and (iii) supporting the signal-dependent changes in affinity between the sink and the migrating molecule (e.g., synthesis of GTP and posttranslational modifications). The cell must keep up with the basic metabolic demands of maintaining unidirectional trafficking of the discs, rhodopsin, RGS9 and all other proteins that do not redistribute in light versus dark. If redistribution of transducin and arrestin was driven by active transport, the sudden energy demand associated with that process might cripple the cell’s ability to maintain the steady concentration of ATP.
Translocation Kinetics
The rate of light-dependent protein migration in rods is different depending on the direction (to or from the OS), and it is also different for arrestin versus transducin. These differences were discussed previously as an argument for the existence of distinct trafficking mechanisms, including the participation of distinct molecular motors (20, 21). Can the diffusion model explain the distinct translocation kinetics?
One of the most important points in the diffusion model is that bona fide “propulsion” of an individual protein molecule does not limit the rate of protein redistribution between the compartments. Theoretical calculations show that unimpeded diffusion is several fold faster than the reported rates of arrestin or transducin relocalization (10, 35). Experiments showed that GFP diffusion is indeed rapid both in frog (29) and in mouse photoreceptors (25). The rate of GFP redistribution measured by FRAP in live mouse rods is about 2 minute at room temperature (~20°C) and should be even faster in a live mammal (~37°C). Indeed, diffusion of hemoglobin microinjected into muscle cells was up to 1.8 times faster at 37°C than at 21°C (36, 37). In the interaction-restricted diffusion model, protein interactions, not diffusion itself, determine relocalization rates. It is very likely that a path of a freely diffusing protein molecule transverses the cell more than once before “finding” its immobile binding partner.
The return of transducin to the OS at the onset of darkness is slower than the movement from the OS in light (7). We think that change in size of this protein upon subunit dissociation can substantially contribute to the difference in these rates. In addition to directly affecting the diffusion coefficient, the molecular weight (Stokes radius) of the molecule also determines how it is impeded by the meshwork of tubules and filaments. Experiments measuring diffusion of various proteins in different muscle cell types show that cytoskeleton progressively reduces the rate of diffusion of larger proteins when compared with the smaller ones (38).
According to Sokolov et al. (2004), Gβγ travels about two-fold slower in the photoreceptors of mice lacking phosducin. In other words, phosducin accelerates light-induced redistribution of Gβγ. Because the diffusion coefficient for a larger (~80 kDa) Gβγ-phosducin complex is less than that of Gβγ. (~45 kDa), this result may appear to contradict the diffusion model. However, as we have discussed earlier, transducin relocalization is limited by its dissociation from discs, not by diffusion of the free molecule. Therefore, the effect of phosducin makes sense because it is known to facilitate both the dissociation of the G protein subunits and detachment of Gβγ from the membrane (39, 40). Furthermore, coimmunoprecipitation in wild-type mice indicated that Gβγ travels from the OS as the complex with phosducin (34). Diffusion of the larger complex should be slower than that of free monomeric Gαt (39 kDa), which can at least partially explain why Gβγ lags behind Gαt while migrating from the OS (7).
For arrestin, the rate of light-induced relocalization to the OS can be determined by the rate of accumulation of forms of rhodopsin that are capable or incapable of arrestin binding. For example, the dark-induced movement of arrestin from the OS corresponds to the rate of opsin dephosphorylation (25). Simply saying, because this biochemical reaction is slower than rhodopsin bleaching and phosphorylation, the rate of arrestin departure from OS is slower than its recruitment in light. The rhodopsin functional cycle involves mechanisms such as photoisomerization of 11-cis retinal, phosphorylation by GRK1, dephosphorylation and opsin formation upon the release of all-trans retinal. It is a complex process [recent review: (41)]. Although the importance of each of the these steps needs to be understood, it appears that in principle, different kinetics of movement from and to OS can an be explained in terms of kinetics of arrestin:rhodopsin interaction, without involvement of active transport.
Cell Morphology
An argument used against diffusion as the mechanism driving transducin and arrestin relocalization is that proteins must cross the narrow cilium connecting the IS to OS [e.g., (24)] and “move around the hundreds of flat discs that span the width of the outer segment” (35). However, diffusion rate only depends on three parameters: size of the particle, viscosity of the medium, and temperature. Therefore, while intuitively it may seem that narrow spaces should impede diffusion, they do not, as long as the protein can actually “fit through the hole.” The diameter of the cilium is at least 20 times wider than the size of transducin or arrestin. Random movements of water molecules are sufficient to propel protein diffusion regardless of how far away—10 nm or 10 lm—the “walls of the tunnel” are. The membranes of the cilium and the discs are impenetrable for proteins; they cannot act as a molecular sieves and do not alter viscosity and they should not affect free diffusion. Only if a protein binds to membranes for significant time before dissociating, its movement along the length of the photoreceptor cell will be affected. Experiments with GFP, which is an inert cytosolic protein, showed that its diffusion across the cilium is as fast as is in other cells (25, 29). It should also be pointed out that an active transport process would face the same “space” problem moving across the cell as passive diffusion. If the cilium or the discs impede movement of a single protein molecule, it seems unlikely that a much larger molecular motor complex can circumvent these obstacles.
QUANTITATIVE ASPECTS OF RHODOPSIN-MEDIATED RECRUITMENT OF ARRESTIN TO THE OS
The process of interaction-restricted diffusion should result in accurate targeting because protein keeps moving until it “settles down” on its target. Another attribute of this model is that both arrestin and transducin can distribute in a precise manner without the need for “oversight” by any sort of specialized regulatory process. Both GTP binding to Gαt and arrestin binding to rhodopsin are directly dependent on the amount of light-activated rhodopsin. If the signal directly regulates the release or sequestration of a diffusing molecule, the mechanism is simple and reliable. In contrast, regulation of active trafficking for the four independently migrating molecules—arrestin, Gαt, Gβγ, and recoverin—via hypothetic signaling pathways would be very complex and prone to errors.
A recent report has described a quantitative analysis of arrestin translocation in which the authors argued against a simple relationship between rhodopsin activation and arrestin recruitment to the OS (42). They measured the amount of arrestin and rhodopsin (Rh) present in rods and found that Rh:arrestin ratio is close to 1:1, an estimate consistent with findings of other groups (4, 43). At high levels of illumination one active Rh molecule sequestered one arrestin, which confirmed that arrestin translocates to the OS by virtue of its binding to active rhodopsin. However, the authors of the study also made another, unexpected observation. A small amount of active rhodopsin (Rh*), which is produced in rods at low levels of bleaching, caused recruitment of a disproportionately large pool of arrestin to the OS. This puzzling effect was referred to as “superstoichiometry.” According to calculations presented in that work, one Rh* could cause relocalization of up to 30 arrestin molecules. The authors also reported that a critical threshold of rhodopsin activation is required for redistribution of arrestin: 1.5% bleaching did not cause any arrestin movement in their experiments, but 3% did trigger redistribution of nearly 40% of arrestin. They interpreted these results as the indication that there is a light-triggered process other than simple diffusion and binding of arrestin to rhodopsin.
Another argument that the simple diffusion model is inadequate was based on the observation that arrestin translocation is altered in R9AP or Gat knockouts. Mice lacking Gαt reportedly do not have the same illumination threshold for arrestin movement, and in R9AP knockout mice arrestin redistributed at a lower light level. The authors concluded that arrestin translocation is triggered downstream of phototransduction, and that it is regulated by the “amount signaling” rather than the amount of rhodopsin bleaching. In other words, rhodopsin does not act merely as a sink, but rather sends a G protein-mediated signal to release arrestin from its binding sites in the inner com-compartments partments [discussed in (35)].
A reasonable mechanism for superstoichiometry and threshold-associated arrestin redistribution has not been proposed (35, 42). However, there may not be a real need for such a mechanism because all the reported experimental findings can be reconciled with the interaction-restricted diffusion model. We interpret the data reported by Strissel et al. (2006) as follows. The results were presented as bar graphs or scatter plots, depicting the percentage of arrestin signal in the OS. The values were determined by immunoblot analyses of tangential sections of the retinas. Our concern about the interpretation of these data focuses on the way in which the curve fitting was done. Assigning an exponential function versus a linear or biphasic was based in some cases on as few as four data points. The difference between wild type (which was fit with a biphasic equation) and Gαt−/− mice (which was fit with an exponential equation) was based on just two crucial data points corresponding to low illumination. In our opinion, a statistical analysis at this level for data that relies solely on tangential sectioning is not sufficient. In this sophisticated technique, variables may include the curvature of individual retinas as they are flattened before sectioning, the actual thickness of the sections, as well as immunoblot scans. At low light levels, the condition when the superstoichiometry effect was observed, arrestin concentration in the sections corresponding to OS is low. According to the presented calibration, the western blot signal is linear only within a narrow (three to fivefold) range. The relative amounts of arrestin in OS under these conditions would have been underestimated or would have fallen below the detection level. Therefore, the detection threshold for the western blot analysis could have been confused with, or misinterpreted as, a biological threshold. The authors discussed this problem, but quantification of arrestin content in the OS under dim light was not adequately addressed and remains a major limitation of this study. In addition to the artifacts associated with the measurements of arrestin, another caveat is likely to contribute to the observation of superstoichiometry: an underestimation of the concentration of arrestin binding-competent forms of rhodopsin. Strissel et al. evaluated the concentration of Rh* only by spectroscopy. They did not take into account the accumulation of phosphorylated opsin (P-Op) and phosphorylated dark rhodopsin (P-Rh), both of which elude spectroscopic detection but certainly contribute to the arrestin sink.
The phenomena observed in R9AP and Gat knockout mice could have resulted from their long-term effects on rods, which develop under intensified (R9AP R9AP2/2) or absent (Gαt−/−) transducin-mediated signaling. For example, compensatory effects could alter the lifetimes and abundance of activated and/or phosphorylated states of rhodopsin.
If the measurements and interpretation of mouse phenotypes presented by Strissel et al. are correct, we have to assume the existence of a G protein-mediated pathway that causes the release of arrestin from its binding sites in the inner compartments (35). However, until this mechanism is unraveled, this hypothesis remains a speculation. It is particularly challenging to explain how up to 40% of arrestin can stay in the OS without binding to rhodopsin and without leaking back to the IS. It appears to us that the model in which arrestin movement is determined by its binding to rhodopsin does not have disagreements with currently available experimental evidence.
WHAT IS THE SIGNIFICANCE OF PROTEIN REDISTRIBUTION IN PHOTORECEPTORS?
It has been suggested that “protein translocation is likely to contribute to photoreceptor light adaptation by adjusting the sensitivity and speed of photoresponse to ever-changing conditions of ambient illumination” (8). This idea is based on the observation that both adaptation and protein redistribution can occur simultaneously in a light-dependent manner (7). However, there is no experimental evidence that there is a link between the two phenomena. The only experiment addressing the cause-and-effect relationship is an important study by Fukada and colleagues (44). These researchers produced a mouse model with a knockin of the Gγ mutant that geranylgeranylated at its C-terminus instead of the usual farnesylation. The resulting rod Gβγ complex tightly binds to membranes and does not translocate to the inner compartments. Rod photoresponses to dim-light flashes in these mice were essentially intact, but the ability to adapt to background illumination was impaired. This experiment strongly supports the possibility that the movement of transducin plays a role in adaptation. However, relocalization of rod transducin requires light levels that a higher than the range where rods can adapt. Furthermore, in cones transducin does not translocate at all, but these cells adapt much better than rods, indicating that the protein redistribution is not essential and that photoreceptors can utilize a host of other adaptive mechanisms.
With respect to arrestin, it has been pointed out that if arrestin moves to OS simply by virtue of its binding to rhodopsin, this mechanism cannot be adaptive (42). Indeed, arrestin binds after rhodopsin has already been bleached and therefore it does not help the cell to “ anticipate” further activation. It should also be noted that rods saturate at light levels that do not yet induce arrestin movement. We think, therefore, that arrestin translocation does not play a role in light adaptation, but simply shuts rods off. Because arrestin knockout leads to light-dependent death of rods (26, 45), it is likely that its ability to keep rhodopsin inactive during the day is a critical function of arrestin.
At the moment, the experimental evidence is not sufficient to unambiguously establish functional role of light-dependent protein redistribution. An appealing hypothesis is that the relocalization of both arrestin and transducin contributes to the protection of rods from light-induced cell death. Experiments utilizing mutants of arrestin and transducin that are not capable of translocation, but retain other essential functions are needed to understand the true significance of this remarkably robust phenomenon.
ACKNOWLEDGEMENTS
We thank V. Gurevich (Vanderbilt University) for helpful discussions. This work was supported by NIH grants GM 060019 (VZS), EY 06641 (JBH) and a grant from Hope-for-Vision Foundation (VZS).
Abbreviations
- OS
outer segments
- IS
inner segment
- ONL
outer nuclear layer
REFERENCES
- 1.Arshavsky VY, Lamb TD, Pugh EN., Jr G proteins and phototransduction. Annu. Rev. Physiol. 2002;64:153–187. doi: 10.1146/annurev.physiol.64.082701.102229. [DOI] [PubMed] [Google Scholar]
- 2.Fain GL, Matthews HR, Cornwall MC, Koutalos Y. Adaptation in vertebrate photoreceptors. Physiol. Rev. 2001;81:117–151. doi: 10.1152/physrev.2001.81.1.117. [DOI] [PubMed] [Google Scholar]
- 3.Makino CL, Wen XH, Lem J. Piecing together the timetable for visual transduction with transgenic animals. Curr. Opin. Neurobiol. 2003;13:404–412. doi: 10.1016/s0959-4388(03)00091-6. [DOI] [PubMed] [Google Scholar]
- 4.Broekhuyse RM, Tolhuizen EF, Janssen AP, Winkens HJ. Light induced shift and binding of S-antigen in retinal rods. Curr. Eye Res. 1985;4:613–618. doi: 10.3109/02713688508999993. [DOI] [PubMed] [Google Scholar]
- 5.Philp NJ, Chang W, Long K. Light-stimulated protein movement in rod photoreceptor or cells of the rat retina. FEBS Lett. 1987;225:127–132. doi: 10.1016/0014-5793(87)81144-4. [DOI] [PubMed] [Google Scholar]
- 6.Whelan JP, McGinnis JF. Light-dependent subcellular movement of photoreceptor proteins. J. Neurosci. Res. 1988;20:263–270. doi: 10.1002/jnr.490200216. [DOI] [PubMed] [Google Scholar]
- 7.Sokolov M, Lyubarsky AL, Strissel KJ, Savchenko AB, Govardovskii VI, Pugh EN, Jr, Arshavsky VY. Massive light-driven translocation of transducin between the two major compartments of rod cells: a novel mechanism of light adaptation. Neuron. 2002;34:95–106. doi: 10.1016/s0896-6273(02)00636-0. [DOI] [PubMed] [Google Scholar]
- 8.Arshavsky VY. Protein translocation in photoreceptor light adaptation: a common theme in vertebrate and invertebrate vision. Sci. STKE. 2003;204:PE43. doi: 10.1126/stke.2003.204.pe43. [DOI] [PubMed] [Google Scholar]
- 9.Strissel KJ, Arshavsky VY. Myosin III illuminates the mechanism of arrestin translocation. Neuron. 2004;43:2–4. doi: 10.1016/j.neuron.2004.06.026. [DOI] [PubMed] [Google Scholar]
- 10.Strissel KJ, Sokolov M, Arshavsky VY, Williams DS, editors. Recent Advances in Human Biology. World Scientific; 2004. Light-driven translocation of signaling proteins in vertebrate and invertebrate photoreceptors; pp. 163–193. [Google Scholar]
- 11.Mendez A, Lem J, Simon M, Chen J. Light-dependent translocation of arrestin in the absence of rhodopsin phosphorylation and transducin signaling. J. Neurosci. 2003;23:3124–3129. doi: 10.1523/JNEUROSCI.23-08-03124.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang H, Huang W, Zhu X, Craft CM, Baehr W, Chen CK. Light-dependent redistribution of visual arrestins and transducin subunits in mice with defective phototransduction. Mol. Vis. 2003;9:231–237. [PubMed] [Google Scholar]
- 13.Coleman JE, Semple-Rowland SL. GC1 deletion prevents light-dependent arrestin translocation in mouse cone photoreceptor or cells. Invest. Ophthalmol. Vis. Sci. 2005;46:12–16. doi: 10.1167/iovs.04-0691. [DOI] [PubMed] [Google Scholar]
- 14.Kerov V, Chen D, Moussaif M, Chen YJ, Chen CK, Artemyev NO. Transducin activation state controls its light-dependent translocation in rod photoreceptors. J. Biol. Chem. 2005;280:41069–41076. doi: 10.1074/jbc.M508849200. [DOI] [PubMed] [Google Scholar]
- 15.Rosenzweig DH, Nair KS, Wei J, Wang Q, Garwin G, Saari JC, Chen CK, Smrcka AV, Swaroop A, Lem J, Hurley JB, Slepak VZ. Subunit dissociation and diffusion determine the subcellular localization of rod and cone transducins. J. Neurosci. 2007;27:5484–5894. doi: 10.1523/JNEUROSCI.1421-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lobanova ES, Finkelstein S, Song H, Tsang SH, Chen CK, Sokolov M, Skiba NP, Arshavsky VY. Transducin translocation in rods is triggered by saturation of the GTPase-activating complex. J. Neurosci. 2007;27:1151–1160. doi: 10.1523/JNEUROSCI.5010-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marszalek JR, Liu X, Roberts EA, Chui D, Marth JD, Williams DS, Goldstein LS. Genetic evidence for selective transport of opsin and arrestin by kinesin-II in mammalian photoreceptors. Cell. 2000;102:175–187. doi: 10.1016/s0092-8674(00)00023-4. [DOI] [PubMed] [Google Scholar]
- 18.Lee SJ, Montell C. Light-dependent translocation of visual arrestin regulated by the NINAC myosin III. Neuron. 2004;43:95–103. doi: 10.1016/j.neuron.2004.06.014. [DOI] [PubMed] [Google Scholar]
- 19.Cronin MA, Diao F, Tsunoda S. Light-dependent subcellular translocation of Gqαin Drosophila photoeceptors is facilitated by the photoreceptor-specific myosin III NINAC. J. Cell Sci. 2004;117(Pt 20):4797–4806. doi: 10.1242/jcs.01371. [DOI] [PubMed] [Google Scholar]
- 20.McGinnis JF, Matsumoto B, Whelan JP, Cao W. Cytoskeleton participation in subcellular trafficking of signal transduction proteins in rod photoreceptor cells. J. Neurosci. Res. 2002;67:290–297. doi: 10.1002/jnr.10120. [DOI] [PubMed] [Google Scholar]
- 21.Peterson JJ, Orisme W, Fellows J, McDowell JH, Shelamer CL, Dugger DR, Smith WC. A role for cytoskeletal elements in the light-driven translocation of proteins in rod photoreceptors. Invest. Ophthalmol. Vis. Sci. 2005;46:3988–3998. doi: 10.1167/iovs.05-0567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nair KS, Hanson SM, Kennedy MJ, Hurley JB, Gurevich VV, Slepak VZ. Direct binding of visual arrestin to microtubules determines the differential subcellular localization of its splice variants in rod photoreceptors. J. Biol. Chem. 2004;279:41240–41248. doi: 10.1074/jbc.M406768200. [DOI] [PubMed] [Google Scholar]
- 23.Hanson SM, Francis DJ, Vishnivetskiy SA, Klug CS, Gurevich VV. Visual arrestin in binding to microtubules inv involves a distinct conformational change. J. Biol. Chem. 2006;281:9765–9772. doi: 10.1074/jbc.M510738200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Elias RV, Sezate SS, Cao W, McGinnis JF. Temporal kinetics of the light/dark translocation and compartmentation of arrestin and α-transducin in mouse photoreceptor cells. Mol. Vis. 2004;10:672–681. [PubMed] [Google Scholar]
- 25.Nair KS, Hanson SM, Mendez A, Gurevich EV, Shestopalov VI, Vishnivetskiy SA, Kennedy MJ, Chen J, Hurley JB, Gurevich VV, Slepak VZ. Light-dependent re-distribution of arrestin in vertebrate rods is an energy-independent process governed by protein-protein interactions. Neuron. 2005;46:555–567. doi: 10.1016/j.neuron.2005.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chan S, Rubin WW, Mendez A, Liu X, Song X, Hanson SM, Craft CM, Gurevich VV, Burns ME, Chen J. Functional comparisons of visual arrestins in rod photoreceptors of transgenic mice. Invest. Ophthalmol. Vis. Sci. 2007;48:1968–1975. doi: 10.1167/iovs.06-1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kennedy MJ, Dunn FA, Hurley JB. Visual pigment phosphorylation but not transducin translocation can contribute to light adaptation in zebrafish cones. Neuron. 2004;41:915–928. doi: 10.1016/s0896-6273(04)00086-8. [DOI] [PubMed] [Google Scholar]
- 28.Eckmiller MS. Microtubules in a rod-specific cytoskeleton associated with outer segment incisures. Vis. Neurosci. 2000;17:711–722. doi: 10.1017/s0952523800175054. [DOI] [PubMed] [Google Scholar]
- 29.Peet JA, Bragin A, Calvert PD, Nikonov SS, Mani S, Zhao X, Besharse JC, Pierce EA, Knox BE, Pugh EN., Jr Quantification of the cytoplasmic spaces of living cells with EGFP reveals arrestin-EGFP to be in disequilibrium in dark adapted rod photoreceptors. J. Cell Sci. 2004;117(Pt 14):3049–3059. doi: 10.1242/jcs.01167. [DOI] [PubMed] [Google Scholar]
- 30.Wolfrum U, Giessl A, Pulvermuller A. Centrins, a novel group of Ca2+-binding proteins in vertebrate photoreceptor cells. Adv. Exp. Med. Biol. 2002;514:155–178. doi: 10.1007/978-1-4615-0121-3_10. [DOI] [PubMed] [Google Scholar]
- 31.Giessl A, Pulvermuller A, Trojan P, Park JH, Choe HW, Ernst OP, Hofmann KP, Wolfrum U. Differential expression and interaction with the visual G-protein transducin of centrin isoforms in mammalian photoreceptor cells. J. Biol. Chem. 2004;279:51472–51481. doi: 10.1074/jbc.M406770200. [DOI] [PubMed] [Google Scholar]
- 32.Nair KS, Mendez A, Blumer JB, Rozenzveig D, Slepak VZ. The presence of Leu-Gly-Asn repeat enriched protein, LGN, a putative binding partner for transducin, in photoreceptors. Invest. Ophthalmol. Vis. Sci. 2005;46:383–389. doi: 10.1167/iovs.04-1006. [DOI] [PubMed] [Google Scholar]
- 33.Kerov VS, Natochin M, Artemyev NO. Interaction of transducin-α with LGN, a G-protein modulator expressed in photoreceptor cells. Mol. Cell Neurosci. 2005;28:485–495. doi: 10.1016/j.mcn.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 34.Sokolov M, Strissel KJ, Leskov IB, Michaud NA, Govardovskii VI, Arshavsky VY. Phosducin facilitates light-driven transducin translocation in rod photoreceptors. Evidence from the phosducin knockout mouse. J. Biol. Chem. 2004;279:19149–19156. doi: 10.1074/jbc.M311058200. [DOI] [PubMed] [Google Scholar]
- 35.Calvert PD, Strissel KJ, Schiesser WE, Pugh EN, Jr, Arshavsky VY. Light-driven translocation of signaling proteins in vertebrate photoreceptors. Trends Cell Biol. 2006;16:560–568. doi: 10.1016/j.tcb.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 36.Papadopoulos S, Jurgens KD, Gros G. Diffusion of myoglobin in skeletal muscle cells—dependence on fibre type, contraction and temperature. Pflugers Arch. 1995;430:519–525. doi: 10.1007/BF00373888. [DOI] [PubMed] [Google Scholar]
- 37.Papadopoulos S, Endeward V, Revesz-Walker B, Jurgens KD, Gros G. Radial and longitudinal diffusion of myoglobin in single living heart and skeletal muscle cells. Proc. Natl. Acad. Sci. USA. 2001;98:5904–5909. doi: 10.1073/pnas.101109798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Papadopoulos S, Jurgens KD, Gros G. Protein diffusion in living skeletal muscle fibers: dependence on protein size, fiber type, and contraction. Biophys. J. 2000;79:2084–2094. doi: 10.1016/S0006-3495(00)76456-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Loew A, Ho YK, Blundell T, Bax B. Phosducin induces a structural change in transducin βγ. Structure. 1998;6:1007–1019. doi: 10.1016/s0969-2126(98)00102-6. [DOI] [PubMed] [Google Scholar]
- 40.Savage JR, McLaughlin JN, Skiba NP, Hamm HE, Willardson BM. Functional roles of the two domains of phosducin and phosducin-like protein. J. Biol. Chem. 2000;275:30399–30407. doi: 10.1074/jbc.M005120200. [DOI] [PubMed] [Google Scholar]
- 41.Bartl FJ, Vogel R. Structural and functional properties of metarhodopsin III: recent spectroscopic studies on deactivation pathways of rhodopsin. Phys. Chem. Chem. Phys. 2007;9:1648–1658. doi: 10.1039/b616365c. [DOI] [PubMed] [Google Scholar]
- 42.Strissel KJ, Sokolov M, Trieu LH, Arshavsky VY. Arrestin translocation is induced at a critical threshold of visual signaling and is superstoichiometric to bleached rhodopsin. J. Neurosci. 2006;26:1146–1153. doi: 10.1523/JNEUROSCI.4289-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hanson SM, Gurevich EV, Vishnivetskiy SA, Ahmed MR, Song X, Gurevich VV. Each rhodopsin molecule binds its own arrestin. Proc. Natl. Acad. Sci. USA. 2007;104:3125–3128. doi: 10.1073/pnas.0610886104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kassai H, Aiba A, Nakao K, Nakamura K, Katsuki M, Xiong WH, Yau KW, Imai H, Shichida Y, Satomi Y, Takao T, Okano T, Fukada Y. Farnesylation of retinal transducin underlies its translocation during light adaptation. Neuron. 2005;47:529–539. doi: 10.1016/j.neuron.2005.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu J, Dodd RL, Makino CL, Simon MI, Baylor DA, Chen J. Prolonged photoresponses in transgenic mouse rods lacking arrestin. Nature. 1997;389:505–509. doi: 10.1038/39068. [DOI] [PubMed] [Google Scholar]