Abstract:
The use of an arterial line filter (ALF) within the pediatric cardiopulmonary bypass (CPB) circuit is not a new concept. It has always presented the perfusionist with a circuit component that while valuable, increased prime volume. The purpose of this study was to evaluate the change in prime volume and emboli between a conventional oxygenator with separate ALF and a new generation oxygenator with integral arterial filter (AF). We performed a clinical, non-randomized retrospective evaluation of the Terumo Capiox® RX15 (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI) (n = 10) in conjunction with the Terumo Capiox® AF125X ALF or the Capiox ® AF02 vs. the Terumo Capiox® FX15 oxygenator with integral AF (n = 10). The above circuit components, in combination with the LUNA EDAC® (emboli detection and classification) Quantifier (LUNA Innovations, Blacksburg, VA) were placed at various locations within each patient’s CPB circuit to establish and quantify the presence and volume of gaseous emboli during all phases of cardiopulmonary bypass. The EDAC® system is available/used for all patients undergoing CPB at this institution. When compared to a more conventional CPB circuit, the Capiox® FX15 primes more easily as it does not require a carbon dioxide flush while still providing a 32 μ AF. There was no statistical difference in air handling between the tested oxygenators and their associated circuits. During this review it was determined that use of the Capiox® FX15 simplifies the arterial limb of the pediatric CPB circuit. Removal of the separate ALF led to the removal of several, now unnecessary, arterial connectors and additional tubing (arterial line filter bypass). Removal of these components led to a reduction in prime volume and decreased the hemodilutional effect. The FX15 provided a safe, simplified pediatric CPB circuit and was as effective in gaseous microemboli removal as was the more traditional RX15 with separate ALF during this review.
Keywords: EDAC®, gaseous microemboli, arterial line filter, bypass
Arterial line filters (ALF) represent safety within the cardiopulmonary bypass (CPB) circuit. Many clinicians believe the ALF prevents transmission of gaseous micro-emboli (GME) and macroemboli to the patient. While the use of an ALF may assist in reducing GME, stopping transmission to the patient is another problem as Riley has shown (1). It has been the practice of some centers to eliminate the ALF from their pediatric circuits in an effort to reduce the overall circuit prime volume.
GME are generated through a multitude of sources including: pulsatile perfusion, low venous reservoir level, vacuum assisted venous drainage, venous line air, field sucker return, medication administration, blood sampling, and cavitation (2–10). Reducing or eliminating any of these GME sources should reduce the total embolic load potentially delivered to the patient. While elimination of these sources of GME is certainly desirable, the practicality of achieving this appears unreachable. One is then left with reducing the total embolic load as much as possible, which, to date, has been best achieved through the use of an ALF. The separate ALF as a safety device has associated shortcomings. There is additional time required for setup (carbon dioxide [CO2] flush) and an associated increase in CPB prime volume. Terumo has introduced a new oxygenator series with an integral arterial filter (AF), known as the Capiox® FX series (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI). The Capiox® FX15 has an integral 32 μ arterial screen filter surrounding the oxygenator fiber bundle and has a similar static prime volume as the Terumo Capiox® RX15 (144 mL vs. 135 mL). Comparatively, the FX15 and RX15 have identical maximum blood flow rates (5 L/min), heat exchanger surface area and composition (.14 m2, stainless steel), and fiber bundle surface and material (∼1.5 m2, microporous polypropylene).
The purpose of this retrospective nonrandomized study was to evaluate GME handling, by means of emboli count and volume, as well as the change in circuit prime volumes between the Terumo Capiox® FX15 with integral arterial filter and the Terumo Capiox® RX15 with separate ALF using either the Terumo Capiox® AF125X or AF02.
MATERIALS AND METHODS
Circuit Design
All patients received a standard CPB circuit configuration with either the Capiox® RX15 oxygenator and separate ALF or the Capiox® FX15 oxygenator. Purge lines from the top of all separate ALF were run in the open position during all periods of bypass. FX15 purge lines were run in the closed position during all periods of bypass, as GME trapped by the screen filter is exhausted via the gas outlet port per the manufacturer. The CPB hardware consisted of: Maquet HL-20 heart-lung machine and HCU-30 heater-cooler (Maquet Cardiopulmonary, Hirrlingen, Germany) and CDI 500” blood parameter monitoring system (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI). The CPB circuit consisted of arterial/venous line with internal diameter tubing sizes of 1/4″ × 3/8″ or 3/8″ × 3/8″ or 5/16″ × 3/8″ respectively, raceway tubing in either 3/8″ or 1/2″, and a hard-shell venous reservoir (Terumo Cardiovascular System Corporation, Ann Arbor, MI). The Capiox ® RX15 circuit contained a Capiox® AF02 with a 1/4? arterial line or Capiox® AF125X with a 3/8″ arterial line. Two different blood cardioplegia systems were used within the circuits at a 1:1 ratio with blood:cardioplegia solution. The Sorin CSC14 blood cardioplegia system (Sorin Group USA, Inc., Arvada, CO) or, for patients >50 kg, the Terumo BC50 (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI) system was used. Each circuit contained a Terumo Capiox® HC05S (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI) hemofilter. CDI 500” (Terumo Cardiovascular Systems Corporation, Ann Arbor, MI) arterial blood gas and venous saturation/hematocit sensors were incorporated into the arterial and venous lines respectively. Operative field suction return was via two 1/4? lines to the hard shell reservoir for all cases. The standard priming solution used was Normosol-R® (Hospira, Inc., Lake Forest, IL).
The Capiox® RX15 circuit contained three EDAC” sensor cuvettes at the following locations: 1) 6 inches pre-oxygenator, 2) 6 inches post oxygenator, and 3) 6 inches post-ALF. The Capiox® FX15 circuit contained two EDAC® sensor cuvettes. One cuvette was placed 6 inches pre-oxygenator and one was placed 6 inches post-oxygenator within the arterial line. The EDAC® sensors were connected to each cuvette and to the EDAC® Quantifier (Luna Innovations, Inc., Blacksburg, VA) for continuous data collection. Data collection included emboli size and count, yielding total embolic load of GME in real time through non-invasive ultrasound technology during all periods of CPB. Once primed, all circuits were recirculated through a .2 μ pre-bypass filter (Pall Corp., East Hills, NY) at a temperature of 37°C for a minimum of 15 minutes. Anticoagulation was managed via heparin concentration, Hemostasis Management System (HMS Plus) (Medtronic, Minneapolis, MN) and activated clotting time, maintained at >480 seconds as measured with the Hemochron® Signature Elite (International Technidyne Corporation, Edison, NJ) and the ACT+ cuvette.
Experimental Design
After receiving Institutional Review Board approval, the records of 20 consecutive patients requiring a 1.5 m2 oxygenator were reviewed. Ten patients underwent CPB with the Capiox® RX15 circuit and 10 with the Capiox® FX15 (Table 1). Each patient underwent continuous, non-pulsatile perfusion during data collection. The EDAC® recording and associated data collection began prior to retrograde autologous prime (RAP) and continued through termination of modified ultrafiltration (MUF) on all reviewed records. Standard operating perfusion procedure was followed regarding conduct of CPB. Events related to CPB were recorded throughout the surgical case highlighting significant events during the CPB period. The events within the EDAC® record in conjunction with the perfusion record were then compared to assist with defining “considerable” GME events related to CPB.
Table 1.
Patient demographics, circuit, and oxygenator used.
| Circuit | Oxygenator | Procedure | Age (yrs) | Sex | Wt (Kg) | ALF | CPB (min) | X-clamp (min) |
|---|---|---|---|---|---|---|---|---|
| 1/4″ × 3/8″ | RX15 | ASD | 8 | Male | 20.0 | AF-02 | 35 | 18 |
| 3/8″″ × 3/8″ | RX15 | PA plasty, PVR, VSD, LPA/RPA stent | 16 | Male | 65.9 | AF-125 | 212 | 85 |
| 3/8″″ × 3/8″ | RX15 | RV-PA conduit | 18 | Male | 53.8 | AF-125 | 85 | 0 |
| 1/4″″ × 3/8″ | RX15 | VSD | 4 | Male | 18.9 | AF-02 | 56 | 32 |
| 3/8″″ × 3/8″ | RX15 | PVR | 18 | Female | 58.3 | AF-125 | 73 | 0 |
| 3/8″″ × 3/8″ | RX15 | ASD, common atrium repair | 16 | Male | 43.0 | AF-125 | 64 | 40 |
| 3/8″″ × 3/8″ | RX15 | PVR, cryoablation | 34 | Female | 89.0 | AF-125 | 131 | 0 |
| 1/4″″ × 3/8″ | RX15 | ASD secundum | 9 | Male | 30.7 | AF-02 | 64 | 36 |
| 1/4″″ × 3/8″ | RX15 | Fenestrated Fontan | 5 | Male | 19.1 | AF-02 | 60 | 0 |
| 3/8″″ × 3/8″ | RX15 | PVR, Redo | 27 | Female | 65.5 | AF-125 | 70 | 0 |
| 1/4″″ × 3/8″ | FX15 | PA plasty | 7 | Male | 29.6 | IAF | 55 | 0 |
| 3/8″″ × 3/8″ | FX15 | ASD, sinus venosus | 39 | Male | 65.8 | IAF | 74 | 37 |
| 1/4″″ × 3/8″ | FX15 | Aortic valvuloplasty | 6 | Male | 22.1 | IAF | 158 | 77 |
| 3/8″″ × 3/8″ | FX15 | PVR, RVOT reconstruction | 6 | Male | 58.7 | IAF | 140 | 0 |
| 3/8″″ × 3/8″ | FX15 | AVR, aortic root replacement | 28 | Male | 72.2 | IAF | 269 | 189 |
| 5/16″ × 3/8″ | FX15 | PVR, RVOT reconstruction, PA plasty | 26 | Female | 79.7 | IAF | 125 | 0 |
| 3/8″″ × 3/8″ | FX15 | AVR, mechanical | 21 | Male | 78.6 | IAF | 154 | 122 |
| 1/4″″ × 3/8″ | FX15 | sub-aortic membrane resection | 5 | Male | 19.3 | IAF | 52 | 34 |
| 3/8″″ × 3/8″ | FX15 | PVR, cryoablation | 21 | Male | 60.6 | IAF | 65 | 0 |
| 1/4″″ × 3/8″ | FX15 | RV-PA conduit, Redo | 6 | Male | 21.5 | IAF | 126 | 0 |
AVR, aortic valve replacement; ASD, atrial septal defect; VSD, ventricular septal defect; PA, pulmonary artery; LPA, left pulmonary artery; RPA, right pulmonary artery; PVR, pulmonary valve replacement; RV-PA, right ventricle-to-pulmonary artery conduit; RVOT, right ventricular outflow tract; Redo, re-operation; INT, internal arterial filter.
Statistical Methods
Each channel (pre-oxygenator, post-oxygenator, and post-ALF) was totaled for GME count, count by size in 20 micron increments up to 1,000 microns, and volume during CPB. The data were further divided into two GME size groups: less than 40 μ and greater than 40 μ. Post-arterial filter GME count and size were subtracted from pre-oxygenator count and size to determine GME transmission across each system. The LUNA Innovations EDAC® Quantifier software was used for all data collection. Statistical significance was considered p < .05. All analyses were performed using Microsoft Excel ” (Microsoft, Redmond, WA).
RESULTS
The number and volume of GME entering or exiting either circuit was not statistically different (Table 2). Within each oxygenator type there was considerable variance in the number and volume of GME as demonstrated by the large standard deviations. On average, the RX15 circuit removed 84% (±20) of the number of GME compared to 93% (±5) for the FX15 circuit, which was not statistically significant. Likewise, the RX15 circuit removed 89% (±19) of the volume of GME compared to 86% (±22) for the FX15 circuit, and did not achieve statistical significance.
Table 2.
Total embolic load assessed entering and exiting the
| Pre-Oxygenator |
Post-Filter |
|||
|---|---|---|---|---|
| Mean | ±SD | Mean | ±SD | |
| GME Count | ||||
| RX15 (n = 10) | 130,955 | 154,589 | 9,405 | 10,315 |
| FX15 (n = 10) | 254,941 | 126,341 | 20,426 | 21,905 |
| GME Volume (mL) | ||||
| RX15 (n = 10) | .000797 | .000635 | .000044 | .000053 |
| FX15 (n = 10) | .006047 | .015326 | .003934 | .012140 |
No statistical difference between the number of emboli entering or exiting either system; SD, standard deviation.
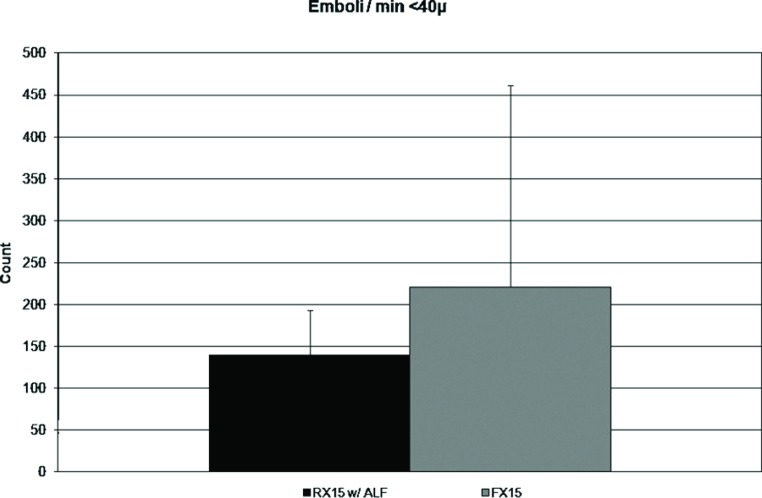
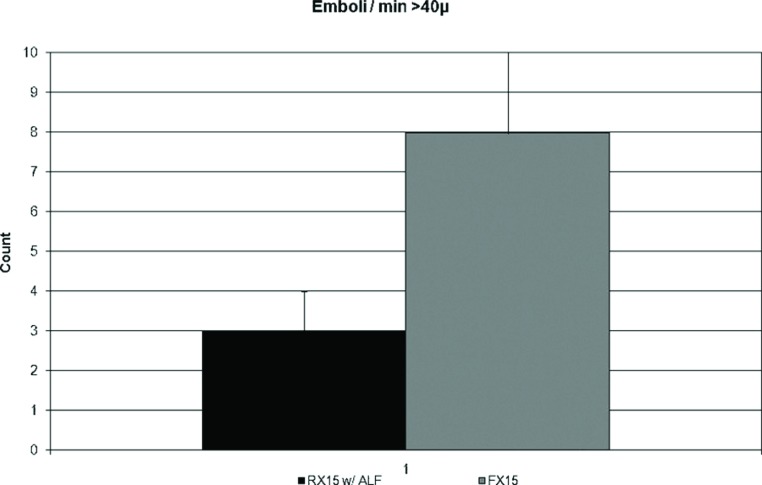
The average CPB time for the RX15 circuit was 85 minutes (±51) and 122 minutes (±66) for the FX15 circuit. Due to the variation in CPB time, and the fact that emboli continue to pass, it was necessary to evaluate GME handling per unit time of bypass. Figures 1 and 2 show the number of emboli post filter per minute of bypass time for the two circuits which were not statistically significant.
Figure 1.
GME/min of CPB (<40 μ) measured by the post-filter EDAC® sensor. Not statistically significant.
Figure 2.
GME/min of CPB (>40 μ) measured by the post-filter EDAC® sensor. Not statistically significant.
The FX15 system allowed for the elimination of eight post-oxygenator connections through the removal of the ALF. Removal of the Capiox ® AF02 32 μ, 40 mL prime ALF from our 1/4? × 3/8? arterial-venous (AV) loop and associated ALF bypass loop resulted in a 56 mL reduction in prime volume for this circuit. Within the next larger circuit (3/8″ × 3/8″ AV loop) the FX15 allowed for the removal of the Capiox® 37 μ 125 mL prime AF125X ALF and associated filter bypass loop. This resulted in a net reduction in prime volume of 183 mL, yielding a total circuit prime volume of 499 mL for the 3/8 × 3/8″ circuit (Table 3). CO2 flushing is not required with the new FX series oxygenator and therefore decreased the time required for priming by approximately 5 minutes. While not significant, the elimination of the CO2 flush is important. Forgetting or choosing to omit this step while priming a conventional ALF does increase time required to de-air the circuit.
Table 3.
Volume (mL) required for circuit prime.
| Volume (mL) Comparison | ||||
|---|---|---|---|---|
| Oxygenator | ALF | AV-Loop | Circuit Prime mL | Difference |
| Capiox® RX15 | AF02 | 1/4″ × 3/8″ | 431 | — |
| Capiox® RX15 | AF125X | 3/8″ × 3/8″ | 82 | — |
| Capiox® FX15 | * | 1/4″ × 3/8″ | 375 | −56 mL† |
| Capiox® FX15 | * | 3/8″ × 3/8″ | 99 | −183 mL‡ |
Denotes integral arterial filter system.
13% reduction in circuit prime volume.
27% reduction in prime volume. Not statistically significant.
DISCUSSION
GME during CPB and its associated neurologic sequelae have been reported by Diegeler et al. (11). Unfortunately, there are currently not any data suggesting a certain volume or quantity of GME that should be avoided to protect the patient from these sequelae. It therefore becomes imperative for the perfusionist to be familiar with the many sources of GME including but not limited to: circuit design and circuit access, field sucker return, venous reservoir volume, cannulation/decannulation, and various other surgical maneuvers (1–10). This knowledge will allow the perfusionist to protect the patient to the best of their ability, which, until recently meant the utilization of a separate ALF. This component of the arterial limb was responsible for a considerable quantity of the overall prime volume (15–28%), making bloodless surgery more difficult in the pediatric population. The advent of the Capiox® FX15 has satisfied the desire and postulated need for arterial filtration while significantly decreasing the overall circuit volume, thereby maximizing the opportunity of performing bloodless surgery in this patient group (Table 3).
While comparing these oxygenator systems with the EDAC®, specific bypass events were attributed to large increases in GME generation leading to the reported large standard deviations. The most significant event identified was “sucker bypass” which resulted in the largest increase in GME count. This increase was similar within both oxygenator systems. Despite these large increases in GME count, very few (16% for the RX15 with ALF, 7% for the FX15) of the emboli entering either oxygenator system were delivered to the patient. In fact, of the GME delivered, the majority were less than 40 μ (>92%). This fact is important when one considers the volume of emboli associated with a count. While it has been shown that exceeding a threshold of emboli on transcranial doppler (TCD) may increase the risk of stroke in adults undergoing CPB, the long term effects of GME in the pediatric population have not been determined (12). At Nationwide Children’s Hospital there are on-going studies correlating GME with TCD and pre-/post-operative MRI. In the patients within this study, no patient had an overt neurological event.
CONCLUSION
The Terumo Capiox® FX15 oxygenator with integral AF primed faster when compared to the Capiox® RX15 with separate ALF as the time required to prime the separate ALF was omitted. Removal of the separate ALF yields a prime volume reduction without sacrificing safety. The use of an integral arterial filter in the oxygenator yields a reduction in total connections located within the arterial limb of the CPB circuit from nine to one, further decreasing potential sites of failure, clot, or disconnect. The Capiox ® FX15 with integral arterial filter effectively reduces GME when compared to either the RX15 with the 32 μ AF02 or the 37 μ AF125X ALF and appears to be an excellent choice in protecting the patient from GME.
REFERENCE
- 1.Riley JB.. Arterial line filters ranked for gaseous micro-emboli separation performance: An in vitro study. J Extra Corpor Technol. 2008;40:21–6. [PMC free article] [PubMed] [Google Scholar]
- 2.Butler B.. Gaseous micro emboli: Concepts and considerations. J Extra Corpor Technol. 1983;15:148–55. [PMC free article] [PubMed] [Google Scholar]
- 3.Lynch JE, Riley JB.. Microemboli detection on extracorporeal bypass circuits. Perfusion. 2008;23:23–32. [DOI] [PubMed] [Google Scholar]
- 4.Merkle F, Bottcher W, Hetzer R.. Prebypass filtration of cardiopulmonary bypass circuits: An outdated technique? Perfusion. 2003;18: 81–8. [DOI] [PubMed] [Google Scholar]
- 5.Myers G.. Preventing gaseous microemboli during blood sampling and drug administration:An in vitro investigation. J Extra Corpor Technol. 2007;39:192–8. [PMC free article] [PubMed] [Google Scholar]
- 6.Norman MJ, Sistino JJ, Acsell JR.. The effectiveness of low-prime cardiopulmonary bypass circuits at removing gaseous emboli. J Extra Corpor Technol. 2004;36:336–42. [PubMed] [Google Scholar]
- 7.Schreiner RS, Rider AR, Myers JW, et al. Microemboli detection and classification by innovative ultrasound technology during simulated neonatal cardiopulmonary bypass at different flow rates, perfusion modes, and perfusate temperatures. ASAIO J. 2008;54:316–24. [DOI] [PubMed] [Google Scholar]
- 8.Undar A, Ji B, Kunselman AR, Myers JL.. Detection and classification of gaseous microemboli during pulsatile and nonpulsatile perfusion in a simulated neonatal CPB model. ASAIO J. 2007;53:725–9. [DOI] [PubMed] [Google Scholar]
- 9.Wang S, Win KN, Kunselman AR, Woitas K, Myers JL, Undar A.. The capability of trapping gaseous microemboli of two pediatric arterial filters with pulsatile and nonpulsatile flow in a simulated infant CPB model. ASAIO J. 2008;54:519–22. [DOI] [PubMed] [Google Scholar]
- 10.Win KN, Wang S, Undar A.. Microemboli generation, detection and characterization during CPB procedures in neonates, infants, and small children. ASAIO J. 2008;54:486–90. [DOI] [PubMed] [Google Scholar]
- 11.Diegeler A, Hirsch R, Schneider F, et al. Neuromonitoring and neurocognitive outcome in off-pump versus conventional coronary bypass operation. Ann Thorac Surg. 2000;69:1162–6. [DOI] [PubMed] [Google Scholar]
- 12.Russell D.. Cerebral microemboli and cognitive impairment. J Neurol Sci. 2002;203–204:211–4. [DOI] [PubMed] [Google Scholar]