Abstract
In this study, we describe a simple and efficient method for mapping the distribution and localization of all sialylated sphingoglycolipids present in coronal mouse brain sections using a conventional axial matrix-assisted laser desorption/ionization time of flight. A single scan of a histological tissue section gives a complete profile of ganglioside species without derivatization or labeling. We have developed and tested a new matrix preparation (2,6-dihydroxyacetophenone [DHA]/ammonium sulfate/heptafluorobutyric acid [HFBA]) to maximize the detection of all ganglioside species; the ammonium sulfate limits the formation of salt adducts, while the addition of HFBA increases the stability of DHA in a vacuum, thus facilitating imaging applications. Our results, in both extracted samples and whole tissue sections using negative ion reflectron and linear modes, show differences in localization in several brain regions depending on the sialic acids and the ceramide-associated core gangliosides.
Keywords: brain, gangliosides, imaging, MALDI, mass spectrometry
Introduction
Until recently, tissue lipid analysis was a work-intensive and time-consuming technique, where lipids were extracted from tissue, derivatized, then analyzed by gas chromatography mass spectrometry or liquid chromatography mass spectrometry. In the past 5 years, lipid analysis has undergone a renaissance with direct lipid profiling (Jackson et al. 2005; Woods and Jackson 2006) or imaging (Cornett et al. 2007; Jackson, Ugarov, et al. 2007; Jackson, Wang, et al. 2007) from tissue, as it turned out to be fairly easy to apply matrix to a thin tissue section, which is then mass analyzed by matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) with no additional preparation. Protein imaging was single handedly brought to the fore by the Caprioli group (Caprioli et al. 1997; Stoeckli et al. 2001). Simplicity was only one of many advantages. Imaging does not only give a molecular profile, it also shows the spatial distribution of the molecules of interest at the tissue surfaces and allows the mapping of all the molecules within the mass range interrogated.
One could say that the meteoric rise of this technique could be due to the ease with which certain lipids such as phosphatidylcholines and sphingomyelins are desorbed from tissue. However, that is not the case with other classes such as ceramides, cerebrosides and gangliosides. Nevertheless, the mass spectrometry community has been hard at work finding alternative MALDI matrices that have improved our likelihood of detecting these hard to visualize compounds.
Gangliosides are a family of glycosphingolipids (GSLs) synthesized on a scaffold of ceramide and characterized by the presence of an oligosaccharide chain containing one or more sialic acid residues (Figure 1, Kolter et al. 2002). They are amphiphilic molecules found in the outer layer of the plasma membranes of all vertebrate cells and are particularly abundant in the central nervous system (CNS) where they account for about 6% of the weight of lipids from the brain and the peripheral nervous system; they are also one of the main constituents of lipid rafts (Simons and Toomre 2000; Sonnino et al. 2007) and play a major role in cell recognition and signaling (Hakomori and Igarashi 1995; Lopez and Schnaar 2009). Alterations in ganglioside metabolism caused by a deficiency of degrading enzymes may result in serious metabolic pathologies due to the accumulation of GSLs in lysosomes mainly in the CNS in lysosomal diseases (Kolter and Sandhoff 2006) including GM1 and GM2 gangliosidosis, Niemann-Pick C and Gaucher disease types II and III. They are also implicated in Alzheimer disease pathogenesis by binding with β-amyloid and amyloid precursor protein (Ariga et al. 2008) and play an important role in many immune-mediated neurological disorders, such as Guillain–Barré syndrome (Yu et al. 2006).
Fig. 1.
Scheme of ganglioside biosynthesis (Svennerholm 1964). Mouse brain ganglioside species that are detected using DHA/ammonium sulfate/HFBA 0.05% matrix are represented by structural forms with d18:1/C18:0 ceramide moiety. All sialylated ganglioside species of the a- and can be detected in mouse brain. Asialoganglioside species and the other minor sialylated ganglioside species cannot be detected because they are not present or do not contain sialic acid. Cer, ceramide; GlcCer, glucosylceramide; LacCer, lactosylceramide. ▲, glucose; ●, galactose; ▪, N-acetylgalactosamine; ◊, sialic acid.
Several ganglioside distribution studies using methyl esterification for derivatization (Zarei et al. 2008) or gold nanoparticle labeling (Nagahori et al. 2009) to increase signal detection by MALDI-time of flight (TOF) were done. However, these methods add steps that result in loss of the biomolecules which are often present in very low concentrations in the tissue. Many matrices have been tested for the study of gangliosides by MALDI (Ivleva et al. 2004, 2005; Sugiura et al. 2008; Zarei et al. 2008; Chen et al. 2008) where sodium and potassium adducts often represent most of the detected species. We developed and tested a new matrix solution (2,6-dihydroxyacetophenone [DHA]/ammonium sulfate 3 mM for extracted gangliosides and DHA/ammonium sulfate 125 mM/heptafluorobutyric acid [HFBA] 0.05% for imaging applications) dissolved in 50% ethanol, which maximized detection of [M-H]− ganglioside species (GM1, GD1, GT1, GM2, GM3, GD3, GT3 and the elusive GQ1s that had yet to be imaged). Distribution of all sialylated ganglioside species in mouse brain tissue sections were analyzed using MALDI mass spectrometry in negative ion mode (O'Connor et al. 2002; Zarei et al. 2008) with a conventional axial MALDI-TOF.
Results and discussion
Extracted ganglioside distribution in mouse brain tissue sections
Previous studies have shown that due to the sublimation of DHA under high vacuum (Jackson, Ugarov, et al. 2007; Jackson, Wang, et al. 2007; Franck et al. 2009), acquisition of data cannot exceed 1 h. So the chemical properties of the matrix have been changed to fit imaging conditions. This matrix preparation limits the formation of salt adducts and increases the signal detection due to the presence of ammonium sulfate, while the HFBA increases the DHA stability in a vacuum.
We have analyzed the effectiveness of our DHA matrix mixture by comparing the spectra we obtained to those acquired with the commonly used 2,5-dihydroxybenzoic acid (DHB) in 0.1% trifluoroacetic acid (TFA) matrix in both linear and reflectron negative ion modes (Figure 2). The ganglioside distribution obtained in linear (Figure 2A) and reflectron (Figure 2B) modes using DHB in 0.1% TFA shows that only four or five different ganglioside species [M-H]− could be identified, and the spectra were dominated by sodium and potassium adducts especially for GD1 species. Many species of gangliosides such as GM3, GD3, GD2, GT1, GQ1 and their O-acetylated forms could not be detected. Linear and reflectron ion modes gave somewhat similar results. However, the signal to noise (S/N) ratio was decreased, and the resolution improved in reflectron ion mode.
Fig. 2.
Mass spectra of ganglioside species by MALDI/MS in negative ion mode using DHB in 0.1% TFA matrix in linear mode (A) and reflectron mode (C).Detected ganglioside species by saturated DHA/ammonium sulfate 3 mM/HFBA 0.05% were represented using linear mode (B) and reflectron mode (D).
The use of DHA matrix in negative linear ion mode improved the detection of ganglioside species such as O-acetylated GT1 forms (Figure 2C). The reflectron ion mode allows the detection of more species of sialylated gangliosides (especially GD1 and GQ1 and their acetylated forms), as well as a decrease in the signal to noise (S/N) ratio and an increase in resolution (Figure 2D). In addition, ganglioside species such as GM2, GM3 and GD3 are also detected. Salt adducts of the major species detected when using DHB in 0.1% TFA are clearly minor species with our DHA matrix due to the addition of ammonium sulfate. Moreover, the loss of CO2 reported with the use of the ATT matrix is nonexistent using a conventional axial MALDI-TOF (Zarei et al. 2008). The presence of O-acetyl forms of GD1, GT1 and GQ1 gangliosides obtained by MALDI/MS using our DHA matrix is consistent with the results obtained with 2D thin-layer chromatography (TLC) by Sonnino’s group (Scandroglio et al. 2009).
Ganglioside mapping by imaging mass spectrometry
For imaging, the matrix was applied to the tissue sections using a chemical ink jet printer. Mouse brain sections were cut into thin sections (18 μm thickness) using a cryostat and directly deposited onto the MALDI target plate. Matrix solution was applied as a homogenous grid of spots with a known amount of matrix (28 nL of matrix per spot) in a cool environment (~4°C) to limit the rapid evaporation of ethanol and crystallized matrix clumps found when using a traditional TLC reagent sprayer or an airbrush for the deposition of saturated DHA matrix.
We have studied and imaged four different sections of mouse brain representing four regions, corresponding respectively to levels 46, 67, 80 and 82 of coronal locations in Allen’s Brain Mouse Atlas. Only [M-H]− ganglioside species and no salt adducts were imaged (Figure 3). The GM3 (d18:1/C18:0) ganglioside species (m/z 1179.7) are present mainly in the lateral reticular nucleus, hippocampus formation and hypothalamus. In-source fragmentation of sialic acids is often alluded to in the literature (O'Connor et al. 2002; Ivleva et al. 2004), and our data show that it occurs in reflectron ion mode, resulting in GD1 and GT1 losing their sialic acid moieties and being detected as GM1 ganglioside species. However, the comparison of GM1 and GD1 species shows a clear difference between the GM1 species itself and the GM1 species resulting from the fragmentation of GD1 and GT1 species. The GM1 (d18:1/C18:0) ganglioside species (m/z 1544.7) appears clearly in the corpus callosum, thalamus and midbrain, unlike GD1 (d18:1/C18:0) ganglioside species (m/z 1835.7). However, the presence of GM1 in the cortex is due to GD1 fragmentation. The GM1 (d20:1/C18:0) ganglioside species (m/z 1572.7) mapping is also slightly different from results obtained with the GD1 (d20:1/C18:0) ganglioside species (m/z 1863.7). These results confirm recent studies which show that eicosasphingosine species of GM1 and GD1 gangliosides (d20:1 sphingoïd base) are mainly located in the molecular layer of the dentate gyrus in the mouse brain hippocampus (Sugiura et al. 2008; Chan et al. 2009). Interestingly, GT1 ganglioside mapping also differs in several brain regions depending on their ceramide core structure. GT1 (d18:1/C18:0) ganglioside species (m/z 2126.7) are not expressed in the corpus callosum and part of the midbrain region and are mainly present in the hippocampus and cortex, while GT1 (d20:1/C18:0) ganglioside species (m/z 2154.7) are mostly located in the cortex, midbrain and part of the hippocampus formation. The elusive GQ1b (d18:1/C18:0) ganglioside specie (m/z 2417.8) was mapped, for the first time, by MALDI imaging, in the periaqueductal gray, hippocampus and hypothalamus regions only in the two last brain areas.
Fig. 3.
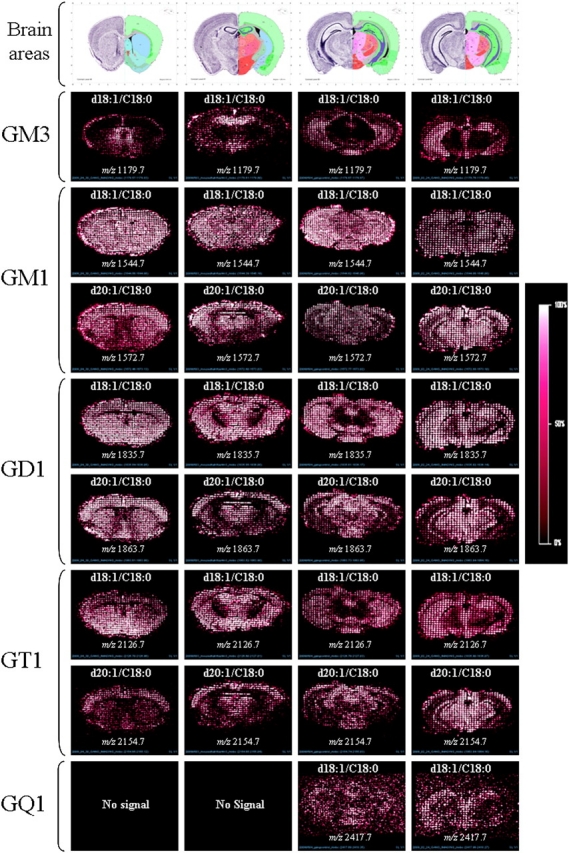
MALDI imaging mass spectrometry of gangliosides using saturated DHA/ammonium sulfate 125 mM/HFBA 0.05% in negative ion reflectron mode. Major gangliosides species are detected such as GM3, GM1, GD1, GT1 and GQ1. These results show differences in localization in several brain regions depending on the sialic acids and the ceramide core associated gangliosides. All other sialylated glycosphingolipids present in these mice brain sections can be mapped (data not shown).
The use of linear ion mode (Figure 4) for mapping GM1, GD1 and GT1 ganglioside species gave the same results obtained using reflectron ion mode. In-source fragmentation of gangliosides was also observed. However, the comparison of the mapped areas confirms the presence of GM1 in the corpus callosum.
Fig. 4.
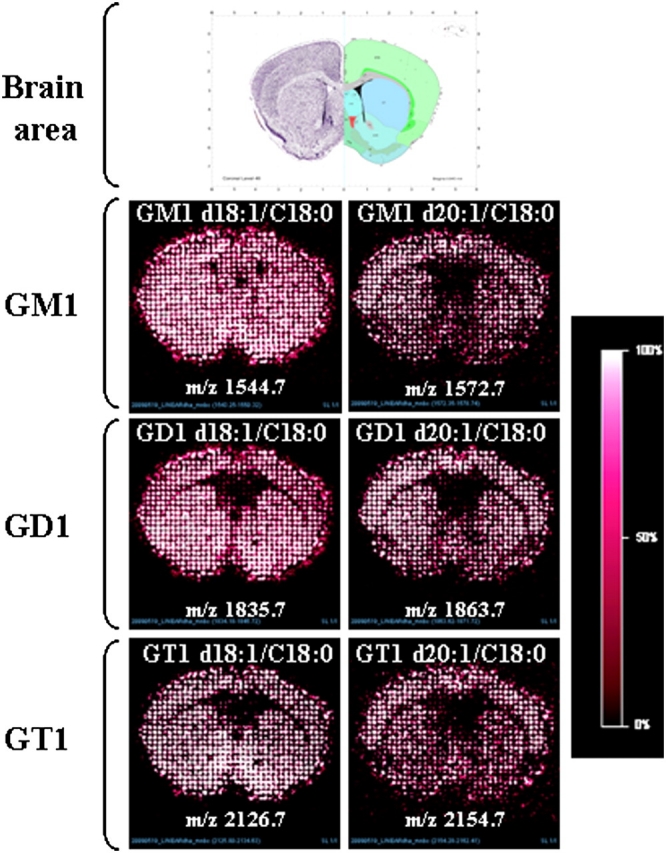
MALDI imaging of ganglioside species detected in mouse brain section using negative linear ion mode. The use of linear ion mode for mapping GM1, GD1 and GT1 ganglioside species gave the same results obtained using reflectron ion mode for the two species d18:1/C18:0 and d20:1/C18:0. Fragmentation of gangliosides was also observed. However, the comparison of the mapped areas confirms the presence of GM1 in the corpus callosum.
Other ganglioside species such as GD3, GT3, GM2, GD2 and O-acetylated forms of GD1, GT1 and GQ1 were imaged (data not shown).
Antibodies or lectins are widely used to detect ganglioside species in histological sections of a mouse brain (Sun et al. 2004; Heffer-Lauc et al. 2005) through their oligosaccharide moieties and requires the use of multiple antibodies to visualize all species. The direct localization of sialylated ganglioside using MALDI/MS is a choice method, as the analysis of a single tissue section gives an almost complete profile of these molecules and a fast and simple assay for complementing immunohistochemistry investigations that have already been done for the detection of ganglioside isoforms in function of their sphingoid base and acetylation of their sialic acids.
Our matrix preparation enabled the development of a new method for the detection of all sialylated sphingoglycolipids present in a tissue section by MALDI imaging as well as from tissue extracts without having to derivatize or label the samples, which implies that this matrix could also be used to detect gangliosides from biological fluid extracts and deletes steps in the sample preparation, thus making the process more efficient as it saves time and sample.
Our data demonstrate that this technique with a single scan using a conventional MALDI-TOF efficiently detects all sialylated ganglioside species present in a sample. Even though in-source fragmentation of gangliosides (GD1 and GT1 to GM1) is only partially eliminated, GM1 mapping is possible by comparison with GD1 mapping. This approach could be useful for comparing controls and animal models of neurological diseases or ganglioside “Knock-out” with a high level of structural information and histological accuracy.
Materials and methods
Chemicals
HFBA, DHA and DHB were purchased from Fluka (St. Louis, MO). Ethanol was purchased from the Winner-Graham Company (Cockeysville, MD). Distilled water was obtained from a Picopure2 system (Hydro, Rockville, MD). Ammonium sulfate and TFA were obtained from J.T. Baker (Phillipsburg, NJ).
Experimental animals
All the animal use and handling in this work abide by the Guide for the Care and Use of Laboratory Animals (NIH).
Tissue preparation
Brain tissues were obtained from adult wild-type C57BL/6 mice. Total lipids were extracted from three mouse brain sections (≈ 2 mg) using modified Folch extraction method (Folch et al. 1957). Briefly, 2 mg of mouse brain sections were extracted with 40 μL chloroform/methanol (2:1; v/v), sonicated and vortexed during 2 h. Distilled water (8 μL) was added, and the mixture was vortexed during 1 min then centrifuged 10 min at 3000 rpm. The upper phase (aqueous phase) containing enriched ganglioside species was dried under a gentle stream of nitrogen and re-dissolved with methanol then kept at −20°C until mass spectrometry analysis. The matrix used for purified gangliosides is saturated DHA dissolved in 50% ethanol with the addition of ammonium sulfate (3 mM) or saturated DHB with 0.1% of TFA dissolved in 50% ethanol.
For imaging, the frozen brains are put on top of dry ice before sectioning. The tissue samples were attached to the cryostat specimen disk using ice slush made from distilled water. Frozen brain tissue was cut into thin sections (18 μm thickness) using a cryostat (Leica Microsystems CM3050S, Bannockburn, IL) at −25°C (cryochamber temperature) and −16°C (specimen cooling temperature). Coronal tissue slices were directly deposited on the MALDI target plate and brought to room temperature before matrix coating (Jackson et al. 2005).
Matrix application system
A CHIP-1000 Chemical Inkjet Printer with a piezoelectric head (Shimadzu-Biotech, Columbia, MD) was used for matrix deposition. Saturated DHA dissolved in 50% ethanol with the addition of ammonium sulfate (125 mM) and 0.05% of HFBA was used as a matrix for tissue imaging applications. Matrix (28 nL) was deposited per spot by the chemical inkjet printer, and the distance between two spots was 240 μm. Optimized conditions use a dwell voltage at 29 V and a dwell time at 34 μs, and the quantity of a drop is 100 pL using an air flow at −0.25 kPa. Two bottles of ice water were placed on each side of the piezoelectric head to limit the rapid evaporation of ethanol.
Imaging mass spectrometry
MALDI mass spectra were obtained using a 4700 MALDI-TOF/TOF mass spectrometer (Applied Biosystems, Foster City, CA) with a 355-nm Nd:YAG laser (200 Hz, ~100 μm) using delayed extraction for linear and reflectron mode. The mass spectrometer was calibrated with a total ganglioside mixture (porcine brain). Our MALDI coupled to a 4000 series MSI image acquisition software was used for data acquisition. The target plate stepping distance was set to 80 μm for both the X and Y directions by the MSI image acquisition software. MS analysis was conducted in negative ion mode. Each spot (spectral pixel) in an image run was an average of 50 shots. Data processing was performed using Tissue View 1.0 in order to generate ion intensity maps (images) for the various ganglioside species.
Acknowledgments
This research was supported by the Intramural Research Program of the National Institute on Drug Abuse, NIH. We thank the Office of National Drug Control Policy (ONDCP) for instrumentation funding, without which this and other projects could not have been accomplished. We also would like to thank Steve Wishnies (Shimadzu Scientific Instruments) for technical support.
Abbreviations
ATT, 6-azo-2-thiothymine; CNS, central nervous system; DHA, 2,6-dihydroxyacetophenone; DHB, 2,5-dihydroxybenzoic acid; GCMS, gas chromatography mass spectrometry; GSL, glycosphingolipid; HFBA, heptafluorobutyric acid; MALDI, matrix-assisted laser desorption/ionization; MS, mass spectrometry; TFA, trifluoroacetic acid; TLC, thin-layer chromatography; TOF, time of flight.
References
- Ariga T, McDonald MP, Yu RK. Role of ganglioside metabolism in the pathogenesis of Alzheimer’s disease—a review. J Lipid Res. 2008;49(6):1157–1175. doi: 10.1194/jlr.R800007-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caprioli RM, Farmer TB, Gile J. Molecular imaging of biological samples: localization of peptides and proteins using MALDI-TOF MS. Anal Chem. 1997;69(23):4751–4760. doi: 10.1021/ac970888i. [DOI] [PubMed] [Google Scholar]
- Chan K, Lantier P, Liu X, Sandhu JK, Stanimirovic D, Li J. MALDI mass spectrometry imaging of gangliosides in mouse brain using ionic liquid matrix. Anal Chim Acta. 2009;639(1-2):57–61. doi: 10.1016/j.aca.2009.02.051. [DOI] [PubMed] [Google Scholar]
- Chen Y, Allegood J, Liu Y, Wang E, Cachón-Gonzalez B, Cox TM, Merrill AH, Jr, Sullards MC. Imaging MALDI mass spectrometry using an oscillating capillary nebulizer matrix coating system and its application to analysis of lipids in brain from a mouse model of Tay-Sachs/Sandhoff disease. Anal Chem. 2008;80(8):2780–2788. doi: 10.1021/ac702350g. [DOI] [PubMed] [Google Scholar]
- Cornett DS, Reyzer ML, Chaurand P, Caprioli RM. MALDI imaging mass spectrometry: molecular snapshots of biochemical systems. Nat Meth. 2007;4(10):828–833. doi: 10.1038/nmeth1094. [DOI] [PubMed] [Google Scholar]
- Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957;226(1):497–509. [PubMed] [Google Scholar]
- Franck J, Arafah K, Barnes A, Wisztorski M, Salzet M, Fournier I. Improving tissue preparation for matrix-assisted laser desorption ionization mass spectrometry imaging. Part 1: using microspotting. Anal Chem. 2009;81(19):8193–8202. doi: 10.1021/ac901328p. [DOI] [PubMed] [Google Scholar]
- Hakomori S, Igarashi Y. Functional role of glycosphingolipids in cell recognition and signaling. J Biochem. 1995;118(6):1091–1103. doi: 10.1093/oxfordjournals.jbchem.a124992. [DOI] [PubMed] [Google Scholar]
- Heffer-Lauc M, Lauc G, Nimrichter L, Fromholt SE, Schnaar RL. Membrane redistribution of gangliosides and glycosylphosphatidylinositol-anchored proteins in brain tissue sections under conditions of lipid raft isolation. Biochim Biophys Acta. 2005;1686(3):200–208. doi: 10.1016/j.bbalip.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Ivleva VB, Elkin YN, Budnik BA, Moyer SC, O'Connor PB, Costello CE. Coupling thin-layer chromatography with vibrational cooling matrix-assisted laser desorption/ionization Fourier transform mass spectrometry for the analysis of ganglioside mixtures. Anal Chem. 2004;76(21):6484–6491. doi: 10.1021/ac0491556. [DOI] [PubMed] [Google Scholar]
- Ivleva VB, Sapp LM, O’Connor PB, Costello CE. Ganglioside analysis by thin-layer chromatography matrix-assisted laser desorption/ionization orthogonal time-of-flight mass spectrometry. J Am Soc Mass Spectrom. 2005;16(9):1552–1560. doi: 10.1016/j.jasms.2005.05.003. [DOI] [PubMed] [Google Scholar]
- Jackson SN, Ugarov M, Egan T, Post JD, Langlais D, Schultz JA, Woods AS. MALDI-ion mobility-TOFMS imaging of lipids in rat brain tissue. J Mass Spectrom. 2007;42(8):1093–1098. doi: 10.1002/jms.1245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson SN, Wang HJ, Woods AS. In situ structural characterization of glycerophospholipids and sulfatides in brain tissue using MALDI-MS/MS. J Am Soc Mass Spectrom. 2007;18(1):17–26. doi: 10.1016/j.jasms.2006.08.015. [DOI] [PubMed] [Google Scholar]
- Jackson SN, Wang HY, Woods AS, Ugarov M, Egan T, Schultz JA. Direct tissue analysis of phospholipids in rat brain using MALDI-TOFMS and MALDI-ion mobility-TOFMS. J Am Soc Mass Spectrom. 2005;16(2):133–138. doi: 10.1016/j.jasms.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Kolter T, Proia RL, Sandhoff K. Combinatorial ganglioside biosynthesis. J Biol Chem. 2002;277(29):25859–25862. doi: 10.1074/jbc.R200001200. [DOI] [PubMed] [Google Scholar]
- Kolter T, Sandhoff K. Sphingolipid metabolism diseases. Biochim Biophys Acta. 2006;1758(12):2057–2079. doi: 10.1016/j.bbamem.2006.05.027. [DOI] [PubMed] [Google Scholar]
- Lopez PHH, Schnaar RL. Gangliosides in cell recognition and membrane protein regulation. Curr Opin Struct Biol. 2009;19(5):549–557. doi: 10.1016/j.sbi.2009.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagahori N, Abe M, Nishimura S. Structural and functional glycosphingolipidomics by glycoblotting with an aminooxy-functionalized gold nanoparticle. Biochemistry. 2009;48(3):583–594. doi: 10.1021/bi801640n. [DOI] [PubMed] [Google Scholar]
- O'Connor PB, Mirgorodskaya E, Costello CE. High pressure matrix-assisted laser desorption/ionization Fournier transform mass spectrometry for minimization of ganglioside fragmentation. J Am Soc Mass Spectrom. 2002;13(4):402–407. doi: 10.1016/S1044-0305(02)00351-3. [DOI] [PubMed] [Google Scholar]
- Scandroglio F, Loberto N, Valsecchi M, Chigorno V, Prinetti A, Sonnino S. Thin layer chromatography of gangliosides. Glycoconj J. 2009;26(8):961–973. doi: 10.1007/s10719-008-9145-5. [DOI] [PubMed] [Google Scholar]
- Simons K, Toomre D. Lipid rafts and signal transduction. Nat Rev Mol Cell Biol. 2000;1(1):31–39. doi: 10.1038/35036052. [DOI] [PubMed] [Google Scholar]
- Sonnino S, Mauri L, Chigorno V, Prinetti A. Gangliosides as components of lipid membrane domains. Glycobiology. 2007;17(1):1R–13R. doi: 10.1093/glycob/cwl052. [DOI] [PubMed] [Google Scholar]
- Stoeckli M, Chaurand P, Hallahan DE, Caprioli RM. Imaging mass spectrometry: a new technology for the analysis of protein expression in mammalian tissues. Nat Med. 2001;7(4):493–496. doi: 10.1038/86573. [DOI] [PubMed] [Google Scholar]
- Sugiura Y, Shimma S, Konishi Y, Yamada MK, Setou M. Imaging mass spectrometry technology and application on ganglioside study; visualization of age-dependent accumulation of C20-ganglioside molecular species in the mouse hippocampus. PLoS ONE. 2008;3(9):e3232. doi: 10.1371/journal.pone.0003232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun J, Shaper NL, Itonori S, Heffer-Lauc M, Sheikh KA, Schnaar RL. Myelin-associated glycoprotein (Siglec-4) expression is progressively and selectively decreased in the brains of mice lacking complex gangliosides. Glycobiology. 2004;14(9):851–857. doi: 10.1093/glycob/cwh107. [DOI] [PubMed] [Google Scholar]
- Svennerholm L. The gangliosides. J Lipid Res. 1964;5:145–155. [PubMed] [Google Scholar]
- Woods AS, Jackson SN. Brain tissue lipidomics: direct probing using matrix-assisted laser desorption/ionization mass spectrometry. AAPS Journal. 2006;8(2):E391–E395. doi: 10.1007/BF02854910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu RK, Usuki S, Ariga T. Ganglioside molecular mimicry and its pathological roles in Guillain–Barré syndrome and related disease. Infect Immun. 2006;74(12):6517–6527. doi: 10.1128/IAI.00967-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarei M, Bindila L, Souady J, Dreisewerd K, Berkenkamp S, Muthing J, Peter-Katalinic J. A sialylation study of mouse brain gangliosides by MALDI a-TOF and o-TOF mass spectrometry. J Mass Spectrom. 2008;43(6):716–725. doi: 10.1002/jms.1367. [DOI] [PubMed] [Google Scholar]




