Abstract
Combinations of voriconazole, fluconazole, and itraconazole with caspofungin were evaluated against 50 Candida glabrata isolates by the time-kill, disk diffusion, and Etest methods. The majority of antifungal combinations were indifferent. By the time-kill method, synergistic activity was detected with eight (16%) of the caspofungin-voriconazole and seven (14%) of the caspofungin-fluconazole combinations, but synergy was not seen with the caspofungin-itraconazole combination. Further comparisons of the Etest and disk diffusion synergy techniques with the time-kill method are warranted.
Candida glabrata infections are common in immunocompromised hosts and difficult to treat since they are often resistant to azole antifungal agents (2, 10, 12). The echinocandin caspofungin (CAS) inhibits the synthesis of 1,3-β-d-glucan, an essential cell wall compound (19). Voriconazole (VORI), fluconazole (FLU), and itraconazole (ITRA) are triazole antifungal agents and inhibit the synthesis of ergosterol by inhibiting the enzyme lanosterol 14α-demethylase (3). As these drugs act on different targets, it is important to look for combinations of drugs that might be synergistic. The main objective of our study was to test for synergistic activities of VORI, FLU, and ITRA combined with CAS against C. glabrata isolates. The other objectives were to compare results from different synergy testing methods, namely, the time-kill method, Etest, and disk diffusion method, and to evaluate the CLSI and Etest methods in terms of categorical agreement.
Fifty clinical isolates were tested. Candida parapsilosis ATCC 22019 was included for quality control (6). Stock solutions of VORI (Pfizer, Barcelona, Spain), CAS (Merck & Co., Inc., West Point, PA), FLU (Pfizer, Barcelona, Spain), and ITRA (Jansen-Cilag) were prepared with the appropriate solvent (dimethyl sulfoxide for VORI and ITRA and distilled water for CAS and FLU). The final concentrations were 0.03 to16 μg/ml for VORI, 0.0625 to 64 μg/ml for CAS and FLU, and 0.5 to 8 μg/ml for ITRA. MICs of drugs were determined by the CLSI broth microdilution method (BMD) (6) and corresponded to the lowest concentration that showed prominent (≥50%) growth inhibition. MICs were read after 24 and 48 h of incubation and were evaluated (5, 6, 9). The Etest was performed on RPMI 1640 agar plates as recommended by the manufacturer, and MICs were compared to the reference BMD MICs (AB Biodisk, Solna, Sweden) (1).
Synergy testing was performed using the time-kill, Etest, and disk diffusion methods. All testing was carried out in duplicate.
For the time-kill studies, each isolate was tested against drugs alone and in combination at concentrations equal to each drug's MIC to correlate with the Etest. The numbers of CFU were determined at 0, 2, 6, and 24 h. The limit of detection was 50 CFU/ml. Synergy and antagonism were defined, respectively, as a ≥100-fold increase or decrease in killing compared with the killing of the most active single agent. If the change was less than 100-fold, the interaction was considered indifferent (3).
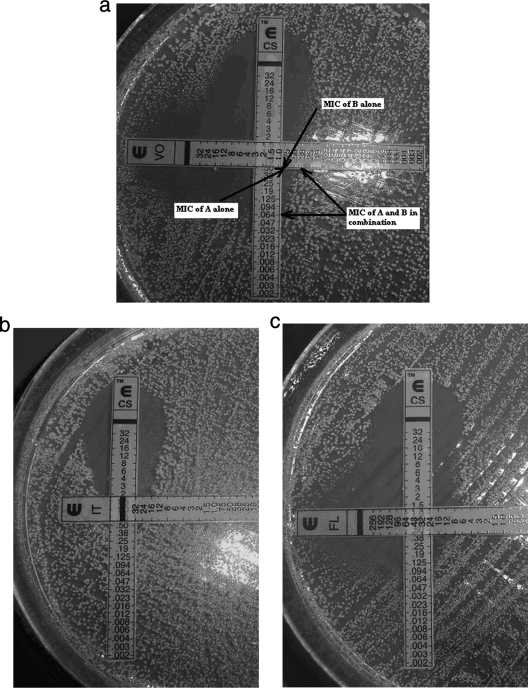
Synergy testing by the Etest was performed by placing E strips on the agar plates in a cross formation, with the strips at a 90° angle where the sections of agar containing the drugs at their MICs meet (Fig. 1) (11, 22). The plates were incubated at 35°C for 48 h. The fractional inhibitory concentration (FIC) index is calculated as ΣFIC = FIC A + FIC B, where FIC A is the MIC of the combination divided by the MIC of drug A alone, and FIC B is the MIC of the combination divided by the MIC of drug B alone. An FIC of ≤0.5 indicated a synergic effect, an FIC of >0.5 to 4.0 an indifferent effect, and an FIC of >4.0 an antagonistic effect (20).
FIG. 1.
Representation by the Etest synergy method of caspofungin (CS) in combination with voriconazole (VO) (a), itraconazole (IT) (b), or fluconazole (FL) (c) (strains 4, 6, and 20).
The disk diffusion method was performed as described in CLSI document M44-A2 (7). Disks were embedded with a drug alone or with drugs in combination. The final concentrations for CAS, VORI, FLU, and ITRA were 2.5, 1.0, 25, and 8 μg/disk, respectively (4, 7, 14, 21, 23). To determine combined effects, each sterile paper disc was impregnated with 10 μl of the prepared sample containing the mixture of both drugs. Inhibition zone diameters were measured after incubation for 48 h at 35°C (3).
For statistical analysis, the in vitro results were analyzed by Student's t test, and a value of <0.05 was considered significant.
The median MICs are presented in Table 1. In general, 24-h MIC readings were similar to those at 48 h for all antifungal agents. CAS was determined to be the most effective antifungal. The levels of agreement between (within ±2 dilutions) the BMD and Etest methods were 92%, 84%, 90%, and 58% for FLU, VORI, CAS, and ITRA, respectively.
TABLE 1.
In vitro susceptibilities of 50 C. glabrata isolates to caspofungin, fluconazole, itraconazole, and voriconazole as determined by two different methods and with two incubation times
| Drug | Incubation time (h) | Methoda | MIC (μg/ml) |
% of isolates by categoryb at 48 h |
||||
|---|---|---|---|---|---|---|---|---|
| Range | 50% | 90% | S | S-DD | I or R | |||
| CAS | 24 | BMD | 0.125-2 | 0.125 | 0.5 | |||
| 48 | 0.125-2 | 0.125 | 0.5 | 98 | 0 | 2 | ||
| 24 | Etest | 0.125-2 | 0.25 | 0.5 | ||||
| 48 | 0.125-2 | 0.25 | 0.5 | 100 | 0 | 0 | ||
| FLU | 24 | BMD | 2-32 | 16 | 32 | |||
| 48 | 4-64 | 32 | 64 | 0 | 74 | 26 | ||
| 24 | Etest | 4-128 | 8 | 16 | ||||
| 48 | 6->256 | 32 | 32 | 4 | 74 | 22 | ||
| ITRA | 24 | BMD | 0.125-2 | 0.25 | 0.5 | |||
| 48 | 0.125-2 | 0.25 | 0.5 | 0 | 62 | 38 | ||
| 24 | Etest | 0.125-16 | 0.06 | 1 | ||||
| 48 | 0.125-32 | 1 | 2 | 0 | 62 | 38 | ||
| VORI | 24 | BMD | 0.125-2 | 0.125 | 1 | |||
| 48 | 0.125-4 | 0.25 | 1 | 78 | 20 | 2 | ||
| 24 | Etest | 0.25-2 | 0.5 | 1 | ||||
| 48 | 0.25-4 | 2 | 2 | 78 | 20 | 2 | ||
For BMD, the CLSI M27-A3 reference method (6) was used. For the Etest, MICs were rounded up to the next even log2 concentration.
S, susceptible; S-DD, susceptible-dose dependent; I, intermediate; R, resistant.
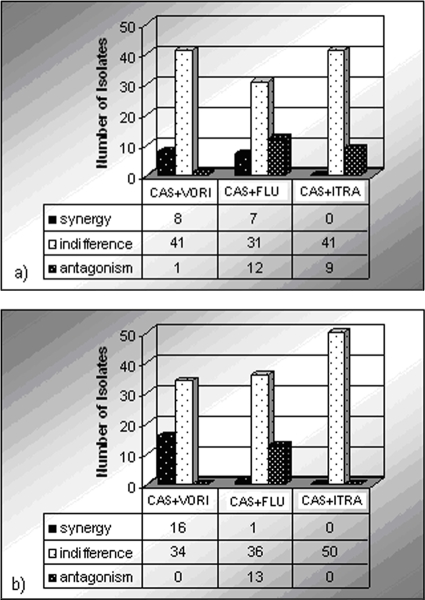
Results obtained from antifungal combinations by the time-kill and Etest methods are reported in Fig. 2. By the time-kill method, synergistic activity was detected with eight (16%) of the CAS-VORI and seven (14%) of the CAS-FLU combinations. For CAS plus ITRA, no synergy was detected. The majority of antifungal combinations were found to be indifferent. The highest antagonistic activity was shown for the CAS-FLU (24%) and CAS-ITRA (18%) combinations. The CAS-VORI combination showed antagonism against only one strain (2%).
FIG. 2.
Results obtained with antifungal combinations by the time-kill (a) and Etest (b) methods.
For the CAS plus VORI, CAS plus FLU, and CAS plus ITRA combinations, the concordance of the Etest synergy method and time-kill assay was demonstrated for 33 of 50 (66%), 43 of 50 (86%), and 41 of 50 (82%) isolates, respectively.
The disk diffusion results are reported in Table 2. For the CAS-VORI combination, the zone diameters obtained with the whole-drug combination were not smaller than those obtained when a drug was used alone. However, with two isolates and CAS plus FLU and with six isolates and CAS plus ITRA, the inhibition zone diameters decreased compared with that of the drug used alone.
TABLE 2.
In vitro activities of VORI, FLU, ITRA, and CAS, alone and in combination with CAS, against 50 C. glabrata isolates as determined by disk diffusion assaya
| Drug | Mean halo diamb ± SD (mm) |
|---|---|
| CAS alone | 24 ± 2.7 a |
| VORI alone | 24 ± 4.0 b |
| CAS + VORI | 41 ± 6.3 |
| FLU alone | 23 ± 4.0 c |
| CAS + FLU | 32 ± 3.8 |
| ITRA alone | 16 ± 5.9 d |
| CAS + ITRA | 27 ± 2.6 |
Each isolate was tested in duplicate.
With treatment of 2.5 μg/disk of CAS, 1 μg/disk of VORI, 25 μg/disk of FLU, or 8 μg/disk of ITRA. a, b, c, and d, P was <0.05 versus results for CAS, VORI, FLU, and ITRA alone, respectively.
So far, synergy data for echinocandins and azoles are very limited; however, these studies were concentrated mostly on Aspergillus species (3, 8, 13, 15, 16, 18). Therefore, as synergy testing methods are not yet standardized for reproducibility and interpretation, it is very difficult to compare our results with those of different studies (17). Our results demonstrated that the type of interaction is method dependent. The Etest method appears promising, and further investigations are warranted.
Synergy, indifference, or antagonism could not be defined exactly by the disk diffusion method, so comparison with the time-kill test cannot be done relatively. However, when we examined the disk diffusion results for isolates showing synergy with the results of the time-kill method, there were marked increases in the zone diameters of drugs in combination for those isolates. If results of the disk diffusion test are compared with different drug concentrations, it may be useful for standardization.
In conclusion, our results indicate that a combination of caspofungin and voriconazole might be effective in infections due to C. glabrata, but there is no evidence of in vivo synergy. Further studies are needed to determine the reliability of these methods and the correlation of in vitro and in vivo results.
Acknowledgments
This work was supported by a grant from Eskisehir Osmangazi University (project 20051104).
Footnotes
Published ahead of print on 1 March 2010.
REFERENCES
- 1.Alexander, B., T. C. Byrne, K. L. Smith, K. E. Hanson, K. J. Anstrom, J. R. Perfect, and L. B. Reler. 2007. Comparative evaluation of Etest and Sensititre YeastOne panels against the Clinical and Laboratory Standards Institute M27-A2 reference broth microdilution method for testing Candida susceptibility to seven antifungal agents. J. Clin. Microbiol. 45:698-706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baltch, A. L., L. H. Bopp, R. P. Smith, W. J. Ritz, and P. B. Michelsen. 2008. Anticandidal effects of voriconazole and caspofungin, singly and in combination, against Candida glabrata, extracellularly and intracellularly in granulocyte-macrophage colony stimulating factor (GM-CSF)-activated human monocytes. J. Antimicrob. Chemother. 62:1285-1290. [DOI] [PubMed] [Google Scholar]
- 3.Barchiesi, F., E. Spreghini, M. Maracci, A. W. Fothergill, I. Baldassarri, M. G. Rinaldi, and G. Scalise. 2004. In vitro activities of voriconazole in combination with three other antifungal agents against Candida glabrata. Antimicrob. Agents Chemother. 48:3317-3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barchiesi, F., E. Spreghini, S. Tomassetti, D. Giannini, and G. Scalise. 2007. Caspofungin in combination with amphotericin B against Candida parapsilosis. Antimicrob. Agents Chemother. 51:941-945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Canton, E., A. Espinel-Ingroff, and J. Peman. 2009. Trends in antifungal susceptibility testing using CLSI reference and commercial methods. Expert Rev. Anti Infect. Ther. 7:107-119. [DOI] [PubMed] [Google Scholar]
- 6.Clinical and Laboratory Standards Institute. 2008. Reference method for broth dilution antifungal susceptibility testing of yeast; approved standard—third edition. Document M27-A3. Clinical and Laboratory Standards Institute, Wayne, PA.
- 7.Clinical and Laboratory Standards Institute/NCCLS. 2004. Method for antifungal disk diffusion susceptibility testing of yeasts; approved guideline—second edition. Document M44-A2. Clinical and Laboratory Standards Institute/NCCLS, Wayne, PA.
- 8.Cuenca-Estrella, M., A. Gomez-Lopez, G. Garcia-Effron, L. Alcazar-Fuoli, E. Mellado, J. M. Buitrago, and J. L. Rodriguez-Tudela. 2005. Combined activity in vitro of caspofungin, amphotericin B, and azole agents against itraconazole-resistant clinical isolates of Aspergillus fumigatus. Antimicrob. Agents Chemother. 49:1232-1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dias, A. L. T., F. E. Matsumoto, M. S. C. Melhem, E. G. Silva, M. E. Auler, A. M. Siqueira, and C. R. Paula. 2006. Comparative analysis of Etest and broth microdilution method (AFST-EUCAST) for trends in antifungal drug susceptibility testing of Brazilian Cryptococcus neoformans isolates. J. Med. Microbiol. 55:1693-1699. [DOI] [PubMed] [Google Scholar]
- 10.Fidel, P. L., J. A. Vazquez, and J. D. Sobel. 1999. C. glabrata. Review of epidemiology, pathogenesis and clinical disease with comparison to Candida albicans. Clin. Microbiol. Rev. 12:80-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kiraz, N., I. Dag, M. Yamac, A. Kiremitci, N. Kaçsifoğlu, and Y. Akgun. 2009. Antifungal activity of caspofungin in combination with amphotericin B against Candida glabrata: comparison of disk diffusion, Etest, and time-kill methods. Antimicrob. Agents Chemother. 53:788-790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Koehler, A. P., K. C. Chu, E. T. Houang, and A. F. Cheng. 1999. Simple, reliable, and cost-effective yeast identification scheme for the clinical laboratory. J. Clin. Microbiol. 37:422-426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.López-Ciudad, V., M. J. Castro-Orjales, C. León, et al. 2006. Successful treatment of Candida parapsilosis mural endocarditis with combined caspofungin and voriconazole. BMC Infect. Dis. 6:73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lozano-Chiu, M., P. W. Nelson, V. L. Paetznick, and J. H. Rex. 1999. Disk diffusion method for determining susceptibilities of Candida spp. to MK-0991. J. Clin. Microbiol. 37:1625-1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mariné, M., C. Serena, F. J. Pastor, and J. Guarro. 2006. Combined antifungal therapy in a murine infection by Candida glabrata. J. Antimicrob. Chemother. 58:1295-1298. [DOI] [PubMed] [Google Scholar]
- 16.Oliveira, E. R., A. W. Fothergill, W. R. Kirkpatrick, B. J. Coco, T. F. Patterson, and S. W. Redding. 2005. In vitro interaction of posaconazole and caspofungin against clinical isolates of Candida glabrata. Antimicrob. Agents Chemother. 49:3544-3545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pankey, G. A., and D. S. Ashcraft. 2005. In vitro synergy of ciprofloxacin and gatifloxacin against ciprofloxacin-resistant Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 49:2959-2964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Perea, S., G. Gonzalez, A. W. Fothergill, W. R. Kirkpatrick, M. G. Rinaldi, and T. F. Patterson. 2002. In vitro interaction of caspofungin acetate with voriconazole against clinical isolates of Aspergillus spp. Antimicrob. Agents Chemother. 46:3039-3041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pfaller, M. A., L. Boyken, R. J. Hollis, S. A. Messer, S. Tendolkar, and D. J. Diekema. 2006. In vitro susceptibilities of Candida spp. to caspofungin: four years of global surveillance. J. Clin. Microbiol. 44:760-763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rodríguez, M. M., M. Ruiz, F. J. Pastor, G. Quindós, A. Carrillo, and G. Guarro. 2007. In vitro interaction of micafungin and fluconazole against Candida. J. Antimicrob. Chemother. 60:188-190. [DOI] [PubMed] [Google Scholar]
- 21.Silici, S., N. A. Koc, D. Ayangil, and S. Cankaya. 2005. Antifungal activities of propolis collected by different races of honeybees against yeasts isolated from patients with superficial mycoses. J. Pharmacol. Sci. 99:39-44. [DOI] [PubMed] [Google Scholar]
- 22.White, R. L., D. S. Burgess, M. Manduru, and J. A. Bosso. 1996. Comparison of three different in vitro methods of detecting synergy: time-kill, checkerboard, and E-test. Antimicrob. Agents Chemother. 40:1914-1918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yan, L., M. Li, Y. Cao, P. Gao, Y. Wang, and Y. Jiang. 2009. The alternative oxidase of Candida albicans causes reduced fluconazole susceptibility. J. Antimicrob. Chemother. 64:764-773. [DOI] [PubMed] [Google Scholar]