Abstract
Acoustic experiences significantly shape the functional organization of the auditory cortex during postnatal ‘critical periods’. Here, we investigate the effects of a non-traumatic augmented acoustic environment (AAE) on the central nucleus of the inferior colliculus (ICC) and lower brainstem nuclei in rat during the critical period. Our results show that an AAE during P9–P28 had a persistent effect on the evoked auditory brainstem responses leading to a decreased latency and an increased amplitude of the response at and above the frequency of the stimulus used for the AAE. These findings are correlated with increased numbers of sites in the ICC that responded to the AAE frequency and show higher thresholds. There also were persistent effects in neurons with a best frequency higher than the AAE stimulus. These neurons showed decreased activity at low sound levels in the low frequency tail of the frequency response area. This was at, below and above the AAE stimulus frequency. Less often, increased activity at higher sound levels also was seen. Together, these findings suggest multifaceted interactions between activity-dependent plasticity, homeostasis, and development in the brainstem during the initial stages of hearing. A neonate exposed to an altered auditory environment may experience long-lasting change over the entire network of the auditory system.
Keywords: Inferior Colliculus, Auditory Pathways, Development, Activity-Dependent Plasticity, Augmented Acoustic Environment
INTRODUCTION
An increase in driven activity by a single tone or patterned acoustic stimulus during development causes a change in the auditory cortex that persists into adulthood. Merzenich and coworkers (Zhang et al., 2001, Chang and Merzenich, 2003, de Villers-Sidani et al., 2007) have identified a critical period in rats (P11–P13) for the induction of changes in spectral tuning in the primary auditory cortex (de Villers-Sidani et al., 2007). Exposure to an augmented acoustic environment (AAE) such as a repeated single pure tone, during this critical period has a profound effect that persists into adulthood and increases the area in auditory cortical map for the frequency of the AAE stimulus. Persistent changes in the cortical maps can also be induced by exposure to white noise and band-pass noise during the initial three weeks of hearing (Zhang et al., 2002, Chang and Merzenich, 2003, Chang et al., 2005, de Villers-Sidani et al., 2008). Thus, the acoustic environment has a dramatic and persistent effect at the level of the auditory cortex during the onset of hearing.
Several previous studies have found changes in neurons of the central nucleus of the inferior colliculus (ICC) after AAE in young animals. Early experiments where rats were exposed to frequency modulated sweeps during the first four months of life showed increased activity to FM sweeps but decreased activity to other stimuli (Clopton and Winfield, 1976). When C57BL/6J mice were exposed to 20 Hz click trains from P8–P24, the frequency tuning of ICC neurons was broadened as measured by Q20 (Sanes and Constantine-Paton, 1983, 1985). The ICC neurons of rats exposed to frequency modulated sweeps during the first 5 postnatal weeks showed clusters around that frequency range in the adult (Poon et al., 1990). Furthermore, they also found that exposure to a constant frequency tone during the first 3 postnatal weeks increased the clustering of best frequency around that tone (Poon and Chen, 1992). More recently, AAE was shown to increase prepulse inhibition in mice (Willott and Turner, 2000), and a magnetic resonance imaging study demonstrates a large-scale reorganization of the mouse auditory midbrain tonotopic map induced by a specific sound-rearing pattern (Yu et al., 2007).
Changes in the auditory brainstem during development may contribute to the persistent changes seen in cortex after exposure to AAE. At the synaptic level, long-term potentiation and long-term depression have been found at the brainstem level in dorsal cochlear nucleus (Tzounopoulos et al., 2007, Tzounopoulos and Kraus, 2009) and midbrain in the ICC (Hosomi et al., 1995, Zhang and Wu, 2000, Wu et al., 2002). These effects are measured in minutes or hours, and they take place in brain slices from a developing auditory system during the onset of hearing. These forms of response plasticity could represent the initial changes that lead to a more permanent change.
Here, we investigate the effects of selective AAE stimulation with 60–70 dB SPL narrow-band acoustic stimuli on the responses of neurons of the ICC in the rat. To facilitate comparison to previous cortical studies, we have used the same AAE stimuli during the same developmental stages as used in studies of the cortex (Zhang et al., 2001, Chang and Merzenich, 2003, de Villers-Sidani et al., 2007, de Villers-Sidani et al., 2008). Our results show that this stimulation of neonates during the first three weeks of hearing (P9–P28) has a persistent effect at the levels of the auditory brainstem. This is manifested in changes of the auditory brainstem response, tonotopic maps in the ICC, and frequency response areas of neurons in the IC. Our results suggest a complex interaction between development and brainstem plasticity during the initial stages of hearing that occurs simultaneously with plastic changes in the neocortex. Some preliminary results were presented previously (Oliver et al., 2006, Izquierdo et al., 2007).
MATERIAL AND METHODS
General
These experiments used pigmented Long Evans rats and followed the NIH Guidelines and the Society for Neuroscience Policy for the Use of Animals in Neuroscience Research. The experimental protocols were approved by Animal Care Committees of the University of Salamanca (USAL) and the University of Connecticut Health Center (UCHC). Extracellular recording experiments were conducted at USAL. Detailed of the procedures are given elsewhere (Malmierca, 2003, Malmierca et al., 2003, Hernandez et al., 2005, Malmierca et al., 2005, Malmierca et al., 2008). Animals were anesthetized with urethane (1.5 g/Kg, 20% solution, i.p.; 0.5 g/kg urethane, i.p., for supplements) to preserve an areflexive state. Atropine sulphate (0.05 mg/kg, s.c.) was administered to reduce bronchial secretions and the trachea was cannulated. Lidocaine was injected subdermally around the ears to minimize stimulation from the stereotaxic head holder. During surgery and recording, the body temperature was monitored with a rectal probe and maintained at 38°C with a thermostatically controlled electric blanket. A craniotomy over the occipital cortex was performed, and the dura was reflected. The electrode was advance through the exposed cortex that was covered with 2% agar to prevent drying. Auditory brainstem response experiments were conducted at UCHC when an animal was under isoflurane anaesthesia. Neonatal AAE was conducted in awake animals in litter-filled standard housing with an open grid top to allow AAE from a speaker overhead.
Augmented Acoustic Environment (AAE) in Neonates
All of the AAE protocols were similar to those used previously to induce altered maps of the auditory cortex in the rat. Six litters of animals and their mothers were exposed to an AAE during the first postnatal month. Two litters of rat pups (n=20) were exposed at USAL to a stimulus consisting of 60–70 dB 14 kHz pure tone pips (25 ms duration delivered every 250 ms, 10% duty cycle) for 14–18 hours per day. Animals were exposed from P9, just before the opening of the ears, to age P28. We chose these parameters since Zhang et al. (2001) had used 4 kHz and 19 kHz pure tone pips and a 3 week exposure at this age range to induce postnatal changes in the tonotopic map of auditory cortex. During the AAE exposure, the animals were housed in a custom-built, double-wall sound attenuated chamber where the ambient sound level was less than 20 dB SPL. Recordings were made at ages P24–37, P50–65, and P156–212. In these experiments, adult control animals (n=11; P60 or older) were housed in the animal facility at all times where the ambient sound level was 55–60 dB SPL, and they were joined by the experimental animals when they were not being exposed to the AAE.
One litter at USAL and one litter at UCHC were culled to n=4 animals each and exposed from P9–P17 for 16 hours per day to an AAE of 70 dB, 250 ms sinusoidal amplitude modulation (SAM) tones with a 7 kHz carrier and 40 Hz modulation frequency presented every 750 ms. This identical protocol was shown by de Villers-Sidani et al (2007) to cause reorganization of the tonotopic map in auditory cortex within a critical period between P9 and P11. In this experiment, AAE and control animals at both USAL and UCHC were housed in a sound attenuation chamber at all times. At USAL, AAE animals were housed in a custom built, double-wall sound attenuating chamber. At UCHC, they were housed in a single wall sound-attenuating booth (Industrial Acoustics Co.) and exposed in a double-walled IAC booth. ABR recordings were made at P32 and P112. Single unit recordings in AAE animals were made at P109–125. Single unit control data was obtained over the same time period from adult animals housed in the animal facility.
At USAL, the 14 kHz AAE stimulus was generated by a computer-based system and custom software that controlled a multifunction synthesizer (8904A, Hewlett Packard, Palo Alto CA) (Malmierca et al., 2005) and delivered with a loudspeaker (Realistic, Fort Worth, TX 76102). The sound level in the cage was calibrated with a½″ microphone (B&K 4191) coupled to a DI-2200 spectrum analyzer (Diagnostic Instruments, Livingston, Scotland, UK).
The 7 kHz stimulus was synthesized at UCHC by a computer controlled system TDT System 2 (TDT, Tucker-Davis Technologies Inc, Alachua, FL) and custom software, recorded and edited with Cool Edit (Syntrillium, Phoenix AZ), and saved to a compact disc. Sounds were played with a Philco DVD player (Rutherford NJ), a DRA 395 amplifier (Denon Electronics, Pine Brook NJ), and delivered with a Prologic 80 loudspeaker (Definitive Technologies, Baltimore MD) with a flat response between 2–30 kHz fixed on the ceiling 1 m over the animals. The sound level was calibrated before the beginning of the stimulation sequence by measuring all locations in the cage with a½″ microphone (Bruel & Kjaer North America, Norcross GA) coupled to a measuring amplifier (B&K 4220). The overall level was calibrated with a continuous pure tone that matched the tone pip or SAM carrier frequency. The same CD was used to stimulate animals at USAL using the system above.
Extracellular Recording and Tonotopic Mapping
Single- and multiple units were recorded in animals after selective neonatal AAE and in control animals at USAL using methods described previously (Malmierca et al., 2008). The stimuli were synthesized on a TDT System 2 using custom software, and delivered by two TDT EC1 electrostatic speakers. The ear bars of the stereotaxic device were replaced by hollow specula that accommodated the speakers (Rees et al., 1997, Hernandez et al., 2005). Stimuli were calibrated with a¼″ microphone (B&K 4136) and DI-2200 spectrum analyzer as above. The maximum output of this system was flat from 0.3–5 kHz (~100 ±7 dB SPL) and from 5–40 kHz (90 ±5 dB SPL), and the second and third harmonic components in the signal were 40 dB or more below the level of the fundamental at the highest output level.
For mapping studies of the ICC, neuronal responses were recorded with 1–2 MΩ tungsten-in-glass electrodes (Merrill and Ainsworth, 1972) that were advanced with a Burleigh 6000 microdrive (Fishers NY). Action potentials were amplified (x10000) with a TDT Bioamp amplifier, filtered (0.5 – 3 kHz), processed with a TDT spike discriminator, and then stored as spike times on a computer. The electrode was advanced in 25 or 50 μm increments with the same increment used for all tracks in a single animal. At each recording site, the multiunit responses were monitored by oscilloscope and with an acoustic monitor while we varied the frequency and the level of the stimulus to the ear contralateral to the ICC. By manually sweeping a mouse cursor over a two-dimensional frequency/intensity matrix in our custom software, we could quickly determine the point on the matrix with a response at the lowest sound level, and this was defined as the best frequency (BF) of the site. At the end of each track, electrolytic lesions (5 μA, 5 s) were made using the tungsten electrode for a later histological verification of recording sites.
To analyze the changes in BF at each recording site along the electrode track, the relative change in frequency between adjacent sites was calculated in octaves. Any change in frequency greater than 0.1 octaves was considered significant (see Malmierca et al., 2008); however, the changes in BF were typically larger. We measured the length of the regions of the electrode track with the same BF within this 0.1 octave threshold. We also calculated the mean stimulus level for the threshold response at each stair-step. In the analysis of frequency stair-steps, we compared the stair-steps that contained frequency of the neonatal stimulus with the stair-steps immediately above and below, regardless of BF. In the analysis of multiunit recording sites, we also generated histograms for each animal that represent the proportion of recording sites for each BF, regardless of the stair-step size, location, or sequence. A two-tailed Student’s t test with two samples assuming unequal variances was used to test for differences between the means.
Frequency Response Area (FRA)
Single units usually were collected in separate electrode penetrations after the mapping was complete or in separate animals. Occasionally, a well isolated unit was found during a mapping penetration. The same electrodes, microdrive, and amplifier configurations were used for both mapping and single units. Pure tones with a duration of 75 ms were used to generate FRAs in well-isolated single units following protocols used previously (Hernandez et al., 2005). The frequency and intensity of the stimulus were varied randomly over 2 octave range above and below the BF and a range of 10–100 dB attenuation from the maximum output of the system at 100 dB. Each stimulus was presented 2–5 times.
We used two-tone stimulation in a series of experiments to reliably confirm the occurrence of acoustically evoked inhibition in ICC units while collecting a FRA. In this paradigm, a FRA is collected, while a second tone stimulates the neuron at 5–15 dB above threshold at the BF. By raising the overall excitability with the BF tone, areas of reduced activity in the FRA (presumably due to inhibition) are more easily identified (e.g., Loftus and Sutter, 2001, Kopp-Scheinpflug et al., 2003, Egorova and Ehret, 2008). This approach is fruitful since we can test many of the single neurons that we isolate in the ICC. A probe-tone 75 ms was set at the BF of the unit and presented at intensities 5–15 dB above threshold to induce varying levels of stimulus-evoked, “steady” activity, while varying the frequency and intensity of the stimulus as described above. FRAs from AAE and control animals were normalized and pooled. Data from each single unit was normalized to the maximum response rate in the FRA. We summed the normalized experimental and control FRAs and divided by the number of single units included in the pool. The difference FRA was calculated by subtracting control pooled data from AAE pooled data. In order to determine the effects of the neonatal stimulation on the neuron’s receptive fields, we compared the response at every frequency-intensity bin in the FRA in the experimental and control pooled FRAs with a Mann-Whitney test.
Auditory Brainstem Responses (ABR)
Auditory brainstem responses were recorded on Long Evans rats (n=8) at UCHC. Four animals were exposed to 7 kHz SAM tones, while 4 were age matched, control animals that were raised in an IAC sound booth. Sounds were generated by a TDT System 3 unit (Tucker-Davis Technologies Inc) and delivered to a Bose Mini-Cube speaker (Bose, Boston MA) 10 cm over the animal. The sound delivery system was calibrated for amplitude between 60 – 40,000 Hz with a 1/2″ microphone (Brüel & Kjaer, Norcross, GA) placed in the same position as the animal’s head. Clicks (100 μsec) and pure tone pips (5 ms, 1 ms rise/fall) were presented at 21 Hz and calibrated with reference to 87 dB peak equivalent. At each carrier frequency, we systematically reduce the level of the stimulus in 10 dB steps until the ABR was no longer detectable. The lowest level with a detectable ABR was defined as the animal’s hearing threshold at that frequency. The responses to the stimuli were recorded through subdermal needle electrodes (Integra Neurosupplies, Plainsboro NJ) with the positive electrode placed at the vertex and referenced to the negative electrode placed under the right ear (mastoid). The signal between the vertex and mastoid electrode was differentially amplified (100,000×) with an Isolated Bioelectric amplifier (SA Instrumentation C., San Diego CA), filtered (0.1 Hz – 5 kHz, 12 dB/octave roll off), averaged 100–500 times, and collected with the TDT system using BioSig and SigGen software (Tucker-Davis Technologies Inc). Data was analyzed off line with BioSig, Excel (Microsoft Corp., Redmond WA), and Origin (OriginLab Corp. Northampton MA). Significance difference between means was measured with a two-tailed Student’s t test with two samples assuming unequal variances.
RESULTS
To investigate persistent effects of neonatal AAE on the midbrain, we used assays of single units, multiple units, and brainstem evoked potentials. Single unit analysis, in particular, was applied to multiple animals from multiple litters that were exposed during a similar time-course with slightly different stimuli. Here, we first present the data from tonotopic maps of the ICC since this is the most direct comparison to the analogous studies in the auditory cortex. Secondly, we present the analysis of frequency and intensity responses from single units of ICC. Finally, we present the results of brainstem responses evoked by acoustic stimuli in animals after neonatal AAE in order to investigate brainstem levels where persistent changes may take place.
Tonotopic Maps in ICC after Neonatal AAE
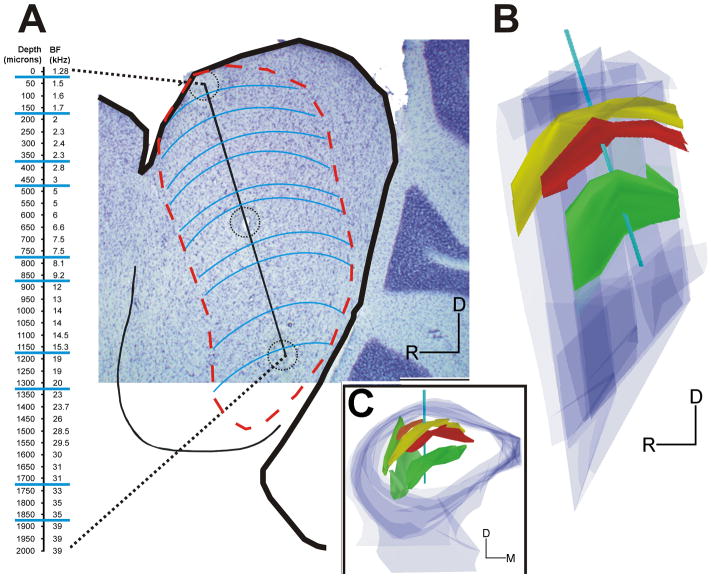
In order to investigate changes in responsiveness in the midbrain, we recorded the frequency that evokes a response at the lowest intensity level (best frequency, BF) from multiunit clusters that every 25 or 50 μm along dorsoventrally oriented microelectrode penetrations in ICC core (Fig. 1A). The electrode entered the ICC at a 10° angle from anterodorsal to posteroventral (Fig. 1B), so the penetration was nearly perpendicular to the fibrodendritic laminae formed by the orientation of afferent axons and dendrites in ICC (Fig. 1B–C, see Oliver, 2005). We previously had shown that in normal rats, when these recordings were made in steps <50 μm apart, there are discrete jumps in the recorded multiunit BF (Malmierca et al., 2008). Thus, the plots of BF vs electrode depth in control rats show a stair-step appearance (Fig. 2A–B). Over the entire ICC, the mean step size was 151 μm and corresponded to a shift in BF of about 1/3 octave (Malmierca et al., 2008).
Figure 1.
Frequency representation in the central nucleus of the inferior colliculus. A, Sagittal section after Nissl staining showing a 2000 μm electrode track (black line) identified with 3 electrolytic lesions (highlighted by circles). The best frequency was recorded at 50 μm intervals. Note that frequency increases as a function of depth, but the best frequencies remain the same for roughly 150 μm then change abruptly. The blue lines show the approximate orientation of the fibrodendritic laminae in ICC. The red lines show the probably boundaries of the fibrodendritic lamina in ICC. B, 3-D reconstruction of 3 axonal laminae (anatomical data from Malmierca el al., 2005 anatomical data from Malmierca el al., 2008) that illustrates how the recording electrode enters the ICC at a 10° angle with respect to the main dorsoventral axis of the ICC. Colors indicate different anatomical laminae formed by afferent axons (yellow, 1.7 kHz lamina; red, 1.8 kHz; green, 4.5 kHz). C, A frontal view of the same 3-D reconstruction seen in B after 90° rotation. The mediolateral location of the track shows that it transverses the ICC. Scale = 2000 μm for electrode track in A (black line); R/D/M orientation arms = 400 μm.
Figure 2.
Electrode penetrations through the ICC in two control animals (A #162 and B #143) and two experimental animals show increased response area at 14 kHz after neonatal AAE (C #83 was age P212 and D #92 was age P30). BFs of multiunit clusters were measured in 50 μm steps along the main dorsoventral axis of the inferior colliculus (Fig. 1) in three cases and in 100 um steps in #92. All ICC penetrations show a stair-step progression. The insets show the expanded 14k Hz stair-steps (horizontal hashed lines), and in most sound exposed cases the stair-step corresponding to 14 kHz covers a larger amount of ICC tissue.
To investigate the effects of early AAE on the tonotopic map in the ICC, two litters of rat pups and their mothers were exposed to 14 kHz pure tone pips at 60–70 dB SPL from ages P9–P28 (after the methods of Zhang et al., 2001). We studied the effects of this augmented acoustic environment on the fine-grained topographic distribution of BF in the ICC of these rats at ages P24–37 and P65–212 and compared them to age-matched control rats that were housed in identical conditions over the same time period except that they were maintained in the animal facility and not exposed to the augmented environment. Data collection from control animals took place on different days from AAE animals. Some of the early control data was published previously (Malmierca et al., 2008).
In comparison to the control, the AAE alters the portion of the tonotopic map in ICC tuned to a BF of 14 kHz. Figure 2C–D shows electrode tracks from two experimental animals. In AAE animals, as in control, there is a stair-step progression of BF in ICC. Despite some difference in the overall length of the electrode tracks at different ages, in most exposed animals (n=6/9) the electrode recorded neurons at the frequency of the early AAE (14 kHz) over a longer distance along the electrode track (e.g., a longer stair-step, Fig. 2C–D, insets) than in control animals (compare with Fig. 2A–B, insets). Multiple electrode penetrations from the same experimental animals (Fig. 2C–D) showed that the 200–300 μm length of the electrode track with a BF of 14 kHz was longer than the length of the track responding to adjacent frequencies. In the ICC of the neonatally exposed animal, the length of the electrode track responding to the AAE frequency of 14 kHz was a mean of 250 μm (Fig. 3B) in comparison to the normal 150 μm.
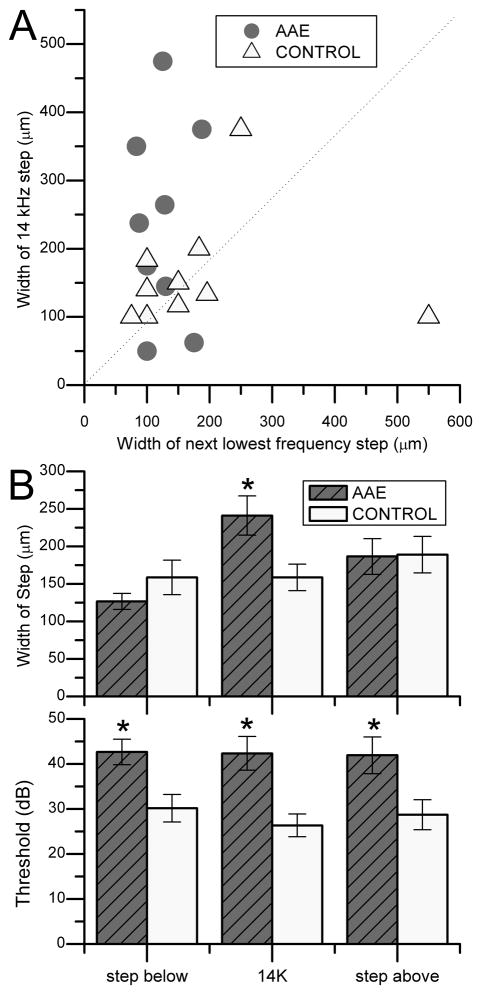
Figure 3.
Comparison of 14 kHz stair-step in control and experimental animals with neonatal AAE. A, Mean thickness (± standard error) of the 14 kHz lamina for each animal versus the next lowest frequency laminae. Note that for the control cases, the points (yellow triangles) lie on the bisecting line, indicating the 14kHz and the next lowest frequency laminae are similar in thickness, however, for the animals after exposure to the augmented acoustic environment (AAE) the points are skewed to the upper left quadrant, indicating that the laminae of the next lowest frequency is smaller than the 14 kHz laminae in this animals. B, The mean width and threshold (± standard error) of the stair-step at 14 kHz (upper) and the stair-steps above and below 14 kHz in tracks pooled across animals in each group. There were 27 electrode tracts in normal animals and 31 electrode tracts in neonatally sound exposed animals. At 14 kHz, the width of the stair-step of the AAE group is significantly greater (*, two tailed T-test, p<0.05). The thresholds for the 14 kHz AAE groups is higher than the control (*, p<0.001).
We compared the mean length of the stair-step that contained 14 kHz to the mean length of the stair-step at the next lowest frequency in each experimental animal (n=9) and each control animal (n=11). The mean stair-step size for each animal is displayed in the scatter plot in Fig. 3A. Most of the data points from the control animals’ cluster around the line of unity, suggesting that these two frequency regions were similar in length. In contrast, most of the data points from animals receiving neonatally AAE are above the line of unity. This shows a relative increase in the length of the 14 kHz stair-step compared to the next lowest frequency stair-step. When measurements of all individual electrode tracks are considered, then significant differences were revealed between the experimental and control groups. The numbers of electrode tracks confined to the ICC ranged from 2–7 per experimental animal (n=31 total for 9 animals) and from 1–5 per control animal (n=27 for 11 animals). The length of the frequency stair-step that included 14 kHz neurons was significantly longer in tracks from AAE animals (241 μm) than in tracks from controls (159 μm) (Fig. 3B, p=0.012). However, there was no difference between experimental and control tracks for the frequency stair-steps in the electrode track to either side of the 14 kHz region (Fig. 3B).
We also measured the threshold for each multiunit recording site. The 14 kHz region and the stair-steps to either side had significantly lower thresholds in control tracks than in experimental tracks (Fig. 3B, bottom, p=0.0009). This suggested that the neonatal AAE had a profound effect on the thresholds beyond the frequency of the AAE stimulus.
In conclusion, early AAE altered the tonotopic map in ICC most often by increasing the size of the anatomical frequency-band lamina and increasing the number of neurons tuned to the BF of the AAE frequency. This was accompanied by an overall increase of the threshold in the experimental animals.
Frequency Response Areas after Neonatal AAE
In order to better understand the spectral changes in single neurons, we studied the frequency tuning of well isolated single neurons from ICC after early AAE in comparison to control. Our sample of FRAs consisted of 51 from well-isolated neurons in animals postnatally exposed to 14 kHz and 120 from equally well isolated control neurons. Our sample of experimental units was enriched for neurons with a BF in the range of 11–16 kHz (n=10 experimental units, 19.61%; n=9 control neurons, 7.5%). For BF <11 kHz, there were similar proportions of neurons (19 experimental neurons, 37.25%, and 45 control neurons, 37.5%). At the higher frequency range BF> 16 kHz, there were relatively more control units with 22 experimental units (43.14%) vs. 66 control units (55%). In 91 control neurons (75.83%), we evaluated two tone stimulation FRAs and found a second peak of activity at 14k Hz in 21.05% (12/57) of the neurons with BF> 14 kHz. In four of those 12 control neurons (33.3%) reduced activity at low sound levels was seen instead at 14 kHz.
Overall, neurons from AAE animals had FRAs and two tone suppressions FRAs similar to normal controls (Hernandez et al., 2005, Egorova and Ehret, 2008). However, many units from experimental animals showed specific changes in the spectral response. There was a peak of activity at the BF and a second higher threshold peak around 14 kHz in 68% of the units from experimental animals (19/28 vs. 21.05% in control units) if the unit had a BF above 14 KHz (BF >14 kHz). The two tone suppression FRA revealed that this second peak of activity was accompanied by less activity at lower intensity levels also at 14 kHz (in 50%, 9/18 units vs. 33.3% in control units). Figures 4B and 4D reveal units with areas of reduced activity that are evident at 14 kHz (Fig. 4B,D, Two Tone, black arrows). For example, the unit in Figure 4B showed a second peak at 14 kHz (FRA, red arrow), and the two-tone FRA revealed reduced activity at lower sound levels at 14 kHz (Two Tone, black arrow) that seems to be a extended part of the region at the BF of the unit at 23 kHz (asterisks). In contrast, another unit showed only reduced activity at 14 kHz and lacked the activity at higher sound levels (Fig. 4C, Two Tone).
Figure 4.
Individual frequency response areas (FRAs) for 4 units (A–D) and their corresponding two tone suppression FRAs from in the ICC neurons of neonates exposed to 14 kHz. A, shows an example of a single unit whose BF is 14 kHz and exhibit clear inhibitory side bands (asterisks). B–D illustrates 3 examples of 3 single units which BF is above 14 kHz and their two tone stimulation indicate that there is an area of low threshold inhibition at 14 kHz (black arrows). These inhibitory thresholds are as low as their excitatory BFs. Color scale represents firing rate in number of spikes after two presentations of the stimuli. Red arrows (left) indicate neonatal stimulus frequency at 14 kHz.
In summary, the data from individual FRAs and two tone suppression FRAs indicate that early AAE to 14 kHz may modify the spectral receptive fields of neurons by altering the response to the AAE frequency. This is often manifested as increased activity at higher sound levels and decreased activity at lower sound levels or both. This is similar to neonatal changes in auditory activity that alter both excitatory and inhibitory responses in the ICC (Vale and Sanes, 2000).
Pooled Frequency Response Areas for Littermates after Neonatal AAE
We compared single unit FRA data from littermates exposed to the same neonatal conditions to similar single units from control animals of the same age. FRAs from AAE and control animals were normalized to maximum spike rate and pooled. Each pool had very similar BFs.
A similar, narrow FRA shape was seen in units from AAE animals (Fig. 5A, AAE, n=8) and control animals (Fig. 5B, CONTROL, n=8) with the BF at 14 kHz, the same frequency of the neonatal AAE. Both groups of units had mean BFs separated by 0.06 octaves (AAE =13.9 ±0.90 kHz; control =13.3 ±0.37 kHz; mean ±SD). The difference FRA (Fig. 5C, DIFF) shows the difference between the relative activity of the AAE and control groups. A modest decrease in activity around 14 kHz was seen at lower intensity levels (Fig. 5C, AAE-CTRL, black arrow), and increased activity was seen above and below the neonatal AAE frequency (Fig. 5C, red). However, these trends did not reach a statistical difference of p=0.05.
Figure 5.
Pooled FRAs from AAE exposed to 14 kHz and control animals. A–C, Pool of neurons with 14 kHz BF (A = 8 AAE; B = 8 control). C is the difference between the FRAs of both groups. The AAE group exhibited an increase in activity for higher intensity stimuli and a decrease of activity for lower intensity stimuli at 14 kHz (black arrow). D–F, Pools of neuron with BF > 14 kHz (D = 17 AAE FRAs; E = 40 control FRAs). F is the FRA difference. Dashed lines delimit the areas in which the responses are significantly different (p<0.05, Mann-Whitney test). A decrease of activity is seen at lower intensity levels above and below the 14 kHz AAE stimulus frequency (black arrow). There was no difference in the mean BF between groups in D and E (mean BF = 23 kHz). Color scale indicates normalized firing rate in number of spikes.
In contrast, units with a BF above 14 kHz showed a significant pattern of reduced activity (Fig. 5D–F). FRA from 17 AAE units (Fig. 5D) and 40 control units (Fig. 5E) had mean BFs separated by 0.15 octaves (AAE =22.32 ±3.68 kHz; control =24.85 ±5.781 kHz; mean ±SD) and were not significantly different (t test, p=0.054). At the neonatal AAE frequency of 14 kHz (black arrow) there were two effects seen in the difference FRA (Fig. 5F). There was decreased activity in experimental units relative to control at low intensity levels and also an increase in relative activity at a high intensity levels. These regions of reduced activity were very consistent across all units and exceeded the significance level (p<0.05; Fig. 5F, dashed black lines). It is important to note that the reduced activity was not limited to the AAE frequency, but extended above and below 14 kHz (Fig. 5F, dashed black lines). This included the BF of the units. These results are consistent with the increased thresholds in the AAE animals. Moreover, these results suggest that profound changes are possible in the spectral coding even when the neonatal stimulus is far from the BF of the neuron.
Replication Experiments at Different AAE Frequencies
To test that the effects described above were not artifactual or due to some unexpected frequency interaction at 14 kHz, we repeated the experiment using a different frequency of AAE. As before, we saw that the AAE modified the activity of units with a BF above the frequency of the AAE stimulus.
In the additional litter, P9–P17 rat pups were exposed to an AAE stimulus consisting of 7 kHz SAM tones (de Villers-Sidani et al., 2007). We compared the normalized FRA (n=29) in three AAE animals with a mean BF=11.9 ±5.0 kHz to control units (19 animals; 32 units) with a mean BF=12.8 ±3.8 kHz, a difference of 0.11 octaves that was not significantly different (p=0.226). In these units, there was a wide-spread, significant decrease (p< 0.05) in the relative activity of AAE units at the AAE frequency of 7 kHz (Fig. 8, black arrow). This extended from 2–10 kHz (Fig. 6, dashed black lines). This pattern was similar to the pattern in the initial cohort of 14 kHz stimulated animals.
Figure 8.
Comparison of mean latency and amplitude of ABR waveform peaks 2 (P2) and 4 (P4) at postnatal day 32. Same animals as in Figure 9. Significant differences between groups indicated by asterisk (*). Significance levels for 7 and 16 kHz as in Fig. 9. The 7 kHz P4 latency (#) p=0.083. The 32 kHz P2 latency p=0.012; P2 amplitude p=0.014. AAE, augmented acoustic environment.
Figure 6.

Difference plots of FRAs from AAE and control animals stimulated with 7 kHz and 40 Hz modulation. The BF of the units was above 7 kHz. Dashed lines delimits the areas in which the responses are significantly different in units from experimental vs. control animals (p<0.05, Mann-Whitney test). Color scale indicates normalized firing rate in number of spikes.
Thus, the spectral responses of units in experimental animals showed decreased activity at the neonatal AAE frequency and surrounding frequencies. This was most pronounced in units where the BF of the units was higher that the frequency of the neonatal stimulation. In our two largest pools of experimental units (14 kHz, n=17 and 7 kHz, n=29) matched to similar control units, the typical response was a decrease in the activity of the units in experimental animals at the frequency of the neonatal AAE. This effect extended to adjacent lower and higher frequencies, up to and including the mean BF of the units. A similar, less significant, decrease was seen for experimental units with a BF at the AAE frequency (14 kHz, n=8).
Auditory Brainstem Responses after Neonatal AAE
In order to investigate whether altered responses to neonatal AAE in the ICC was, in part, due to changes at the lower auditory brainstem nuclei, we examined the ABR of rats exposed to AAE with 7 kHz SAM tones at P9–P17 (40 Hz modulation, after the methods of de Villers-Sidani et al., 2007). The ABR is a far-field recording of potentials evoked by acoustic stimuli in neural structures of the central auditory system in the brainstem. The ABR consisted of at least three prominent peaks with the first peak around 1.5 ms, the second around 2.5 ms, and the last peak around 4.5 ms (Fig. 7). The third peak was typically small, and peaks 4 and 5 merged into a single peak (called peak 4 here for convenience) as shown previously in rat (Blatchley et al., 1987, Newton et al., 1992). These peaks in a tone ABR are associated with the eighth nerve, cochlear nucleus, superior olive, and midbrain, respectively. Note that peak 1 is virtually identical in experimental and control animals suggesting little change in the periphery (Fig. 7).
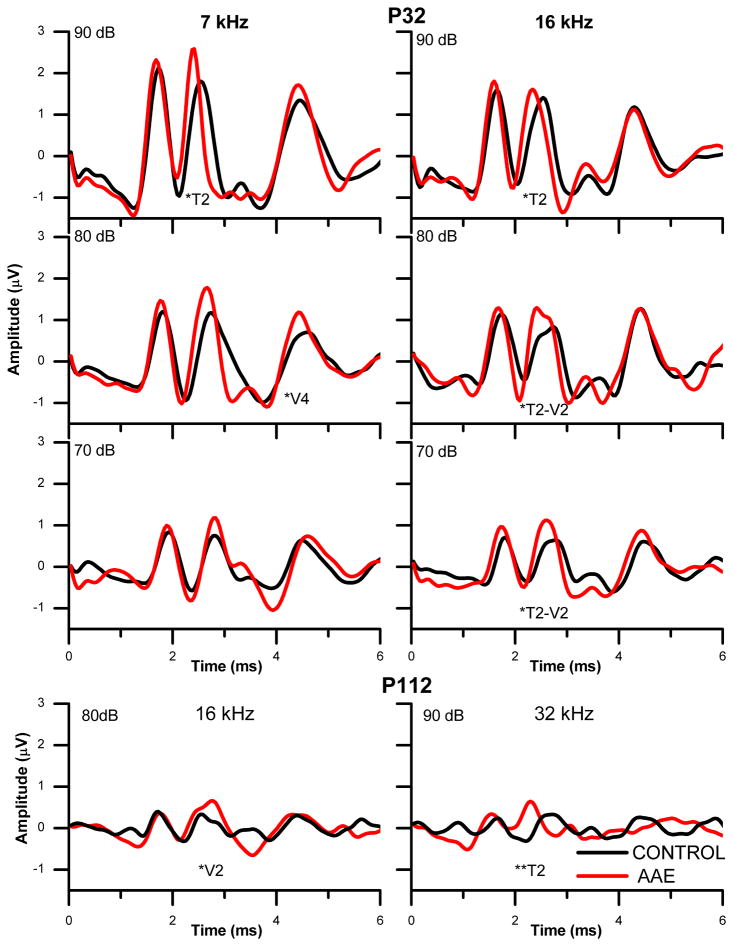
Figure 7.
Mean auditory brainstem responses at postnatal days 32 (P32) and 112 (P112) in animals exposed to and augmented acoustic environment (AAE) of 7 kHz SAM tones (40 Hz modulation) during P9–P17 (n=4) and controls (n=4). Responses of each animal to 500 presentations of a 5 ms pure tone pip were averaged for each group. Positive is an upward deflection. Significant differences between groups in the timing (T) or voltage (V) of peaks are indicated with asterisks (*). At P32 7k Hz, 90dB T2 p=0.0520; 80dB V4 p=0.017. At P32 16 kHz, 90dB T2 p=0.043; 80dB T2 p=0.012 and V2 p=0.046; 70dB T2 p=0.042 and V2 p=0.013. For P112, 16 kHz V2 p=0.032; 32 kHz T2 p=0.009.
Significant changes in the ABR were seen at postnatal day 32, fifteen days after the final day of AAE. For example, Figure 7 shows the averaged responses from the four experimental and four control animals. The animals overstimulated with a 7 kHz SAM tone had a shorter latency second peak (T2) at 90 dB and increased amplitude fourth peak (V4) at 80 dB (Fig. 7, P32). Significant differences in the mean peak timing (Fig. 7, T) or mean peak amplitude (Fig. 7, V) are indicated with asterisk (*, P values in Fig. 7, legend). When responses to 16 kHz were compared to those at the AAE frequency of 7 kHz, the experimental animals showed differences in amplitude and frequency more often. Shorter latencies and increased amplitudes of peak 2 were seen in experimental animals with 90, 80, and 70 dB stimuli. No differences were seen between treatments in the thresholds for clicks or tonal stimuli.
In general, the neonatal AAE changed the second ABR peak (P2, Fig. 8) more often than the fourth peak (P4, Fig. 8). AAE animals had shorter latencies and larger amplitudes at peak 2 evoked by 7, 16, and 32 kHz stimuli (Fig. 8, P2, *). Non-significant increases in amplitude were also seen for peak 2 evoked by 4 kHz tones. On the other hand, the AAE did alter peak 4 in the experimental animals, but only at the AAE frequency of 7 kHz. Peak 4 was increased in the amplitude (Fig. 8, P4, *) and slightly decreased in the latency when evoked by 7 kHz.
The neonatal AAE at P9–P17 produced the most significant changes at P32, and thereafter only changes at peak 2 remained (Fig. 7). When the animals were retested at P112, the ABR amplitudes were smaller perhaps due to the increased head size. Experimental animals showed increased amplitude for peak 2 in response to 16 kHz and a decreased latency in response to 32 kHz.
In summary, the ABR data complement the results described above and suggest that the neonatal AAE altered function at multiple locations in the auditory brainstem. An increase in ABR amplitude was seen at both peaks 2 and 4 at the AAE frequency when presented at higher sound levels. However, there were more widespread changes in both increased amplitude and decreased latency at peak 2, and such changes in peak 2 persisted in older animals. This supports changes at both the cochlear nucleus and midbrain levels in response to neonatal AAE.
DISCUSSION
The present study demonstrates the persistent effects of AAE on the auditory midbrain and lower structures in neonatal rats with narrow band, non-traumatic ~60–70 dB SPL sounds during the first weeks of hearing (P9–P28). Specifically, we show that auditory stimulation led to an expansion of sites that responded to the frequency of the neonatal AAE in multi-unit, tonotopic maps of the ICC. The evoked potential ABR data were consistent with the increased probability of firing of neurons in both the midbrain and cochlear nucleus activated by the neonatal stimulus. In the IC, there was an increase in ABR amplitude for tones at the AAE frequency. However, changes were also seen in the cochlear nucleus where a persistent increase in ABR amplitude and decreased latency were seen at higher frequencies as well as the AAE stimulus.
The tonotopic remapping was accompanied by increased thresholds for multiunit records in the mapping experiments. The decrease in sensitivity was more evident in the FRA of isolated single units. Neurons from experimental animals most often showed a reduction in excitation over a range of frequencies and sound levels, not just the neuronal BF. Such changes were particularly noticeable when the neonatal AAE frequency was in the lower-frequency “tail” of the FRA. These data argue for multiple types of persistent changes at multiple levels of the auditory brainstem induced by early AAE.
The present data are robust and supported by different methodologies, repeated samples with different litters, and different AAE stimuli. The ABR specifically identified the brainstem level as one locus of change. Similar expansions in tonotopic maps in ICC were seen in two litters stimulated with 14 kHz tones. Changes were seen in the frequency response areas from three litters of animals overstimulated with sounds that differed primarily in frequency or carrier frequency. In each litter, the spectral response of well-isolated single units was analyzed with complete FRA or two tone FRA studies rather than simpler threshold tuning curves. The pooled FRA data indicated there was relatively less activity at the AAE frequency in experimental animals relative to controls, although some increased activity to high sound levels was seen. These effects extended over a broad frequency range.
Remapping in the auditory cortex
Tonotopic remapping is well established in the rat’s auditory neocortex after neonatal AAE with a pure tone from P9–P28 -- the same species, stimulus, and time period used here (Zhang et al., 2001). These remapping effects persisted to the oldest age tested (~P70–P114). The critical period appears to be P11–P13 for passive, tone-evoked expansion in tonotopic maps in the auditory cortex in the rat (de Villers-Sidani et al., 2007).
The present mapping data in the IC and the auditory brainstem responses suggest that neonatal stimulation over P9–P28 increases the proportion of neurons responsive to the neonatal stimulus in the IC and other brainstem nuclei. The larger ABR signals are consistent with a larger current source in both the IC and the cochlear nucleus. The suprathreshold stimuli may activate more neurons or it may induce greater synchrony amongst the neurons (Eggermont, 2007). An increase in active neurons in IC is consistent with a manganese-enhanced magnetic resonance imaging study showing that neonatal AAE increases the volume of tissue in the mouse auditory midbrain responsive to the experimental stimuli (Yu et al., 2007). Despite the correlation of the cortical and midbrain reactions to neonatal stimulation, the mechanisms that drive these changes is unclear.
The persistent changes in the auditory brainstem after AAE could be initiated or modulated in a top-down fashion, through local mechanisms, or through an interaction of both afferent and efferent mechanisms (Sanes and Bao, 2009). Corticofugal mechanisms may modulate response properties in the cochlear nucleus (Perrot et al., 2006, Luo et al., 2008), ICC (Suga and Ma, 2003, Zhang et al., 2005), and thalamus (Palmer et al., 2007, Zhang and Yan, 2008). The present experiments focus on the ICC where the lemniscal ascending inputs are relatively strong, as compared to corticofugal inputs (Winer, 2005, Malmierca and Ryugo, 2010). However, the intrinsic IC circuitry (Oliver et al., 1991, Malmierca et al., 1995, Saldaña et al., 1996, Malmierca et al., 2009b) provides a pathway for a cortical influence over the persistent changes in the midbrain. Clearly, a neonate exposed to an altered auditory environment may experience change over the entire network of the auditory system.
Homeostatic control of midbrain plasticity
The present data suggest that neonatal stimulation increased thresholds and diminished responsiveness in the IC. We found a significant increase in thresholds for multiunit clusters in the tonotopic maps at both the frequency of the neonatal stimulus and adjacent frequencies. Likewise, there were profound changes in the FRA of individual neurons, especially those with higher BF than the neonatal stimulus. These changes were most often a decreased activity in the animals exposed to neonatal stimulation. This closely matchs findings in adult animals exposed to moderate, narrow-band sound stimulation who have no hearing loss but show a decreased responsiveness to the stimulating frequency (Norena et al., 2006). Decreased activity at the IC level could lead to decreased activity in the auditory neocortex. Although most cortical mapping studies after neonatal AAE do not report threshold changes, de Villers-Sidani et al. demonstrated systematic increases in the thresholds for experimental animals at all frequencies studied including the 7 kHz neonatal stimulus (Fig. 7B, de Villers-Sidani et al., 2007), a finding similar to the present results.
In addition to a reduced response at low sound levels, we observed an increased responsiveness for suprathreshold stimuli signified by the expanded tonotopic map to the AAE stimulus and the increased ABR responses. This combination suggests a change in the gain control at the auditory brainstem level such that the gain function has a steeper slope than normal. This would lead to a higher threshold and steeply increasing suprathreshold responses. These changes parallel observations of responses in the adult cortex after AAE exposure (Norena et al., 2006), except that here only a single narrow-band stimulus is used. The mechanisms to alter gain control are those also suggested to mediate cortical reorganization (Pienkowski and Eggermont, Syka, 2002, Irvine, 2007). A long-term homeostatic reduction in sensitivity (e.g., LTD) or an habituation to the non-informative AAE stimulus (Pienkowski and Eggermont, Malmierca et al., 2009a, Antunes et al., 2010) would lead to higher thresholds. Simultaneously, a loss of inhibitory side-bands on IC neurons (Wang et al., 1996, Kuwada et al., 1997, LeBeau et al., 2001) and a general reduction of inhibition would unmask excitation and lead to broader response areas. Similar to changes that downregulate inhibition with aging and tinnitus (Caspary et al., 2008, Roberts et al., 2010), a loss of inhibition may lead to hypersensitivity to suprathreshold AAE stimuli. For example, unmasked excitation may lead to increased numbers of neurons responding to the AAE stimulus in multiunit recordings for tonotopic maps. At the same time, increased numbers of neurons or more highly correlated neuronal responses may lead to larger amplitude ABR responses in the IC or cochlear nucleus to suprathreshold stimuli.
Developmental control of midbrain plasticity
The neonatal AAE around the onset of hearing takes place in the context of a still developing system (Friauf and Lohmann, 1999). At the onset of hearing, the entire cochlea does not respond to the full range of sounds (Lippe and Rubel, 1983, Rubel and Ryals, 1983, Harris and Dallos, 1984, Ryals and Rubel, 1985). “The immature [human] auditory system appears to be fully capable of transducing low, but not high frequencies” (Teas et al., 1982) in human babies after birth (Ponton et al., 1992). In the postnatal rat, the high frequency ABR lags relative to the low frequency ABR with only 3 kHz responses observed at P12 (Blatchley et al., 1987). Single unit data in the cochlear nucleus of rat also supports the expansion of the hearing range and lowering of threshold with increasing age (Muller, 1991), and a developmental shift in the tonotopic map in the lateral superior olive in gerbil may be due to postnatal cochlear maturation (Sanes et al., 1989).
High density maps of the developing auditory cortex may reflect the postnatal development of the periphery. For example, at P11, the cortex only responded to 7 kHz tones, at P12 responses included 14 kHz, at P13 responses included 4 kHz, and the tonotopic map expanded to 1.5–30 kHz by P14 (de Villers-Sidani et al., 2007). In the present data, this pattern of non-uniform activity during development could be related to the amounts of reduced activity (threshold shift) seen in litters stimulated with different frequencies. The most profound reductions in excitation relative to controls were found at frequencies that were available over the longest developmental time period (7 kHz, Fig. 8). So, our attempt to impose a uniform duration of neonatal stimulation may have been subverted by the rapid changes still taking place in the cochlea from P9–P14.
Acknowledgments
Financial support was provided by grants from National Institute of Deafness and Communication Disorders (R01-DC000189) to DLO, the Spanish MICINN (BFU2009-07286), EU (EUI2009-04083) and JCYL-UE (GR221) to MSM, and MEC (SAB2004-0076) to MSM and DLO. MAI was supported by a JCYL fellowship.
ABBREVIATIONS
- AAE
Augmented acoustic environment
- ABR
Auditory brainstem response
- BF
Best frequency
- FRA
frequency response area
- IC
inferior colliculus
- ICC
Central nucleus of the inferior colliculus
- SAM
Sinusoidal amplitude modulation
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
LITERATURE CITED
- Antunes FM, Nelken I, Covey E, Malmierca MS. Stimulus-specific adaptation in the auditory thalamus of the anesthetized rat. PLoS One. 2010;5:e14071. doi: 10.1371/journal.pone.0014071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blatchley BJ, Cooper WA, Coleman JR. Development of auditory brainstem response to tone pip stimuli in the rat. Brain Res. 1987;429:75–84. doi: 10.1016/0165-3806(87)90140-4. [DOI] [PubMed] [Google Scholar]
- Caspary DM, Ling L, Turner JG, Hughes LF. Inhibitory neurotransmission, plasticity and aging in the mammalian central auditory system. J Exp Biol. 2008;211:1781–1791. doi: 10.1242/jeb.013581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang EF, Bao S, Imaizumi K, Schreiner CE, Merzenich MM. Development of spectral and temporal response selectivity in the auditory cortex. Proc Natl Acad Sci U S A. 2005;102:16460–16465. doi: 10.1073/pnas.0508239102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang EF, Merzenich MM. Environmental noise retards auditory cortical development. Science. 2003;300:498–502. doi: 10.1126/science.1082163. [DOI] [PubMed] [Google Scholar]
- Clopton BM, Winfield JA. Effect of early exposure to patterned sound on unit activity in rat inferior colliculus. Journal of neurophysiology. 1976;39:1081–1089. doi: 10.1152/jn.1976.39.5.1081. [DOI] [PubMed] [Google Scholar]
- de Villers-Sidani E, Chang EF, Bao S, Merzenich MM. Critical period window for spectral tuning defined in the primary auditory cortex (A1) in the rat. J Neurosci. 2007;27:180–189. doi: 10.1523/JNEUROSCI.3227-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Villers-Sidani E, Simpson KL, Lu YF, Lin RC, Merzenich MM. Manipulating critical period closure across different sectors of the primary auditory cortex. Nat Neurosci. 2008;11:957–965. doi: 10.1038/nn.2144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eggermont JJ. Correlated neural activity as the driving force for functional changes in auditory cortex. Hearing Research. 2007;229:69–80. doi: 10.1016/j.heares.2007.01.008. [DOI] [PubMed] [Google Scholar]
- Egorova M, Ehret G. Tonotopy and inhibition in the midbrain inferior colliculus shape spectral resolution of sounds in neural critical bands. The European journal of neuroscience. 2008;28:675–692. doi: 10.1111/j.1460-9568.2008.06376.x. [DOI] [PubMed] [Google Scholar]
- Friauf E, Lohmann C. Development of auditory brainstem circuitry. Activity-dependent and activity-independent processes. [Review] [88 refs] Cell Tiss Res. 1999;297:187–195. doi: 10.1007/s004410051346. [DOI] [PubMed] [Google Scholar]
- Harris DM, Dallos P. Ontogenetic changes in frequency mapping of a mammalian ear. Science. 1984;225:741–743. doi: 10.1126/science.6463651. [DOI] [PubMed] [Google Scholar]
- Hernandez O, Espinosa N, Perez-Gonzalez D, Malmierca MS. The inferior colliculus of the rat: a quantitative analysis of monaural frequency response areas. Neuroscience. 2005;132:203–217. doi: 10.1016/j.neuroscience.2005.01.001. [DOI] [PubMed] [Google Scholar]
- Hosomi H, Hirai H, Okada Y, Amatsu M. Long-term potentiation of neurotransmission in the inferior colliculus of the rat. Neurosci Lett. 1995;195:175–178. doi: 10.1016/0304-3940(95)11811-a. [DOI] [PubMed] [Google Scholar]
- Irvine DRF. Auditory cortical plasticity: Does it provide evidence for cognitive processing in the auditory cortex? Hearing Research. 2007;229:158–170. doi: 10.1016/j.heares.2007.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izquierdo MA, Malmierca MS, Oliver DL. Early sound exposure alters frequency coding in the inferior colliculus of the rat. 30th Annual Meeting, Assoc Res Otolaryngology; Denver. 2007. [Google Scholar]
- Kopp-Scheinpflug C, Fuchs K, Lippe WR, Tempel BL, Rubsamen R. Decreased temporal precision of auditory signaling in Kcna1-null mice: an electrophysiological study in vivo. J Neurosci. 2003;23:9199–9207. doi: 10.1523/JNEUROSCI.23-27-09199.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuwada S, Batra R, Yin TC, Oliver DL, Haberly LB, Stanford TR. Intracellular recordings in response to monaural and binaural stimulation of neurons in the inferior colliculus of the cat. J Neurosci. 1997;17:7565–7581. doi: 10.1523/JNEUROSCI.17-19-07565.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LeBeau FE, Malmierca MS, Rees A. Iontophoresis in vivo demonstrates a key role for GABA(A) and glycinergic inhibition in shaping frequency response areas in the inferior colliculus of guinea pig. J Neurosci. 2001;21:7303–7312. doi: 10.1523/JNEUROSCI.21-18-07303.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lippe W, Rubel EW. Development of the place principle: tonotopic organization. Science. 1983;219:514–516. doi: 10.1126/science.6823550. [DOI] [PubMed] [Google Scholar]
- Loftus WC, Sutter ML. Spectrotemporal organization of excitatory and inhibitory receptive fields of cat posterior auditory field neurons. Journal of neurophysiology. 2001;86:475–491. doi: 10.1152/jn.2001.86.1.475. [DOI] [PubMed] [Google Scholar]
- Luo F, Wang Q, Kashani A, Yan J. Corticofugal modulation of initial sound processing in the brain. J Neurosci. 2008;28:11615–11621. doi: 10.1523/JNEUROSCI.3972-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmierca MS. The structure and physiology of the rat auditory system: an overview. International review of neurobiology. 2003;56:147–211. doi: 10.1016/s0074-7742(03)56005-6. [DOI] [PubMed] [Google Scholar]
- Malmierca MS, Cristaudo S, Perez-Gonzalez D, Covey E. Stimulus-specific adaptation in the inferior colliculus of the anesthetized rat. J Neurosci. 2009a;29:5483–5493. doi: 10.1523/JNEUROSCI.4153-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmierca MS, Hernandez O, Antunes FM, Rees A. Divergent and point-to-point connections in the commissural pathway between the inferior colliculi. J Comp Neurol. 2009b;514:226–239. doi: 10.1002/cne.21997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmierca MS, Hernandez O, Falconi A, Lopez-Poveda EA, Merchan M, Rees A. The commissure of the inferior colliculus shapes frequency response areas in rat: an in vivo study using reversible blockade with microinjection of kynurenic acid. Experimental brain research Experimentelle Hirnforschung. 2003;153:522–529. doi: 10.1007/s00221-003-1615-1. [DOI] [PubMed] [Google Scholar]
- Malmierca MS, Hernandez O, Rees A. Intercollicular commissural projections modulate neuronal responses in the inferior colliculus. The European journal of neuroscience. 2005;21:2701–2710. doi: 10.1111/j.1460-9568.2005.04103.x. [DOI] [PubMed] [Google Scholar]
- Malmierca MS, Izquierdo MA, Cristaudo S, Hernandez O, Perez-Gonzalez D, Covey E, Oliver DL. A discontinuous tonotopic organization in the inferior colliculus of the rat. J Neurosci. 2008;28:4767–4776. doi: 10.1523/JNEUROSCI.0238-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malmierca MS, Ryugo DK. In: Cortical descending projections to auditory midbrain and brainstem. Winer JA, Schreiner CE, editors. Springer-Verlag; 2011. In Press. [Google Scholar]
- Malmierca MS, Seip KL, Osen KK. Morphological classification and identification of neurons in the inferior colliculus: a multivariate analysis. Anatomy & Embryology. 1995;191:343–350. doi: 10.1007/BF00534687. [DOI] [PubMed] [Google Scholar]
- Merrill EG, Ainsworth A. Glass-coated platinum-plated tungsten microelectrodes. Med Biol Eng. 1972;10:662–672. doi: 10.1007/BF02476084. [DOI] [PubMed] [Google Scholar]
- Muller M. Developmental changes of frequency representation in the rat cochlea. Hear Res. 1991;56:1–7. doi: 10.1016/0378-5955(91)90147-2. [DOI] [PubMed] [Google Scholar]
- Newton EH, Cooper WA, Jr, Coleman JR. Rate and frequency interactions in the auditory brainstem response of the adult rat. Hear Res. 1992;60:73–79. doi: 10.1016/0378-5955(92)90060-z. [DOI] [PubMed] [Google Scholar]
- Norena AJ, Gourevitch B, Aizawa N, Eggermont JJ. Spectrally enhanced acoustic environment disrupts frequency representation in cat auditory cortex. Nat Neurosci. 2006;9:932–939. doi: 10.1038/nn1720. [DOI] [PubMed] [Google Scholar]
- Oliver DL. Neuronal organization in the inferior colliculus. In: Winer JA, Schreiner CE, editors. The Inferior Colliculus. Chapter 2. New York: Springer; 2005. pp. 69–114. [Google Scholar]
- Oliver DL, Izquierdo MA, Malmierca MS. Early sound exposure modifies function in the inferior colliculus (IC) of the rat. Society for Neuroscience; Atlanta, GA: 2006. [Google Scholar]
- Oliver DL, Kuwada S, Yin TC, Haberly LB, Henkel CK. Dendritic and axonal morphology of HRP-injected neurons in the inferior colliculus of the cat. J Comp Neurol. 1991;303:75–100. doi: 10.1002/cne.903030108. [DOI] [PubMed] [Google Scholar]
- Palmer AR, Hall DA, Sumner C, Barrett DJ, Jones S, Nakamoto K, Moore DR. Some investigations into non-passive listening. Hear Res. 2007;229:148–157. doi: 10.1016/j.heares.2006.12.007. [DOI] [PubMed] [Google Scholar]
- Perrot X, Ryvlin P, Isnard J, Guenot M, Catenoix H, Fischer C, Mauguiere F, Collet L. Evidence for corticofugal modulation of peripheral auditory activity in humans. Cereb Cortex. 2006;16:941–948. doi: 10.1093/cercor/bhj035. [DOI] [PubMed] [Google Scholar]
- Pienkowski M, Eggermont JJ. Cortical tonotopic map plasticity and behavior. Neuroscience & Biobehavioral Reviews. doi: 10.1016/j.neubiorev.2011.02.002. In Press, Corrected Proof. [DOI] [PubMed] [Google Scholar]
- Ponton CW, Eggermont JJ, Coupland SG, Winkelaar R. Frequency-specific maturation of the eighth nerve and brain-stem auditory pathway: evidence from derived auditory brain-stem responses (ABRs) J Acoust Soc Am. 1992;91:1576–1586. doi: 10.1121/1.402439. [DOI] [PubMed] [Google Scholar]
- Poon PW, Chen X. Postnatal exposure to tones alters the tuning characteristics of inferior collicular neurons in the rat. Brain Res. 1992;585:391–394. doi: 10.1016/0006-8993(92)91243-8. [DOI] [PubMed] [Google Scholar]
- Poon PW, Chen XY, Hwang JC. Altered sensitivities of auditory neurons in the rat midbrain following early postnatal exposure to patterned sounds. Brain Res. 1990;524:327–330. doi: 10.1016/0006-8993(90)90710-s. [DOI] [PubMed] [Google Scholar]
- Rees A, Sarbaz A, Malmierca MS, Le Beau FE. Regularity of firing of neurons in the inferior colliculus. Journal of neurophysiology. 1997;77:2945–2965. doi: 10.1152/jn.1997.77.6.2945. [DOI] [PubMed] [Google Scholar]
- Roberts LE, Eggermont JJ, Caspary DM, Shore SE, Melcher JR, Kaltenbach JA. Ringing Ears: The Neuroscience of Tinnitus. The Journal of Neuroscience. 2010;30:14972–14979. doi: 10.1523/JNEUROSCI.4028-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubel EW, Ryals BM. Development of the place principle: acoustic trauma. Science. 1983;219:512–514. doi: 10.1126/science.6823549. [DOI] [PubMed] [Google Scholar]
- Ryals BM, Rubel EW. Ontogenetic changes in the position of hair cell loss after acoustic overstimulation in avian basilar papilla. Hear Res. 1985;19:135–142. doi: 10.1016/0378-5955(85)90117-0. [DOI] [PubMed] [Google Scholar]
- Saldaña E, Feliciano M, Mugnaini E. Distribution of descending projections from primary auditory neocortex to inferior colliculus mimics the topography of intracollicular projections. J Comp Neurol. 1996;371:15–40. doi: 10.1002/(SICI)1096-9861(19960715)371:1<15::AID-CNE2>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- Sanes DH, Bao S. Tuning up the developing auditory CNS. Curr Opin Neurobiol. 2009 Jun 15; doi: 10.1016/j.conb.2009.05.014. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanes DH, Constantine-Paton M. Altered activity patterns during development reduce neural tuning. Science. 1983;221:1183–1185. doi: 10.1126/science.6612332. [DOI] [PubMed] [Google Scholar]
- Sanes DH, Constantine-Paton M. The sharpening of frequency tuning curves requires patterned activity during development in the mouse, Mus musculus. J Neurosci. 1985;5:1152–1166. doi: 10.1523/JNEUROSCI.05-05-01152.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanes DH, Merickel M, Rubel EW. Evidence for an alteration of the tonotopic map in the gerbil cochlea during development. J Comp Neurol. 1989;279:436–444. doi: 10.1002/cne.902790308. [DOI] [PubMed] [Google Scholar]
- Suga N, Ma X. Multiparametric corticofugal modulation and plasticity in the auditory system. Nat Rev Neurosci. 2003;4:783–794. doi: 10.1038/nrn1222. [DOI] [PubMed] [Google Scholar]
- Syka J. Plastic Changes in the Central Auditory System After Hearing Loss, Restoration of Function, and During Learning. Physiological Reviews. 2002;82:601–636. doi: 10.1152/physrev.00002.2002. [DOI] [PubMed] [Google Scholar]
- Teas DC, Klein AJ, Kramer SJ. An analysis of auditory brainstem responses in infants. Hear Res. 1982;7:19–54. doi: 10.1016/0378-5955(82)90080-6. [DOI] [PubMed] [Google Scholar]
- Tzounopoulos T, Kraus N. Learning to encode timing: mechanisms of plasticity in the auditory brainstem. Neuron. 2009;62:463–469. doi: 10.1016/j.neuron.2009.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzounopoulos T, Rubio ME, Keen JE, Trussell LO. Coactivation of pre- and postsynaptic signaling mechanisms determines cell-specific spike-timing-dependent plasticity. Neuron. 2007;54:291–301. doi: 10.1016/j.neuron.2007.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vale C, Sanes DH. Afferent regulation of inhibitory synaptic transmission in the developing auditory midbrain. J Neurosci. 2000;20:1912–1921. doi: 10.1523/JNEUROSCI.20-05-01912.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Salvi RJ, Powers N. Plasticity of response properties of inferior colliculus neurons following acute cochlear damage. Journal of neurophysiology. 1996;75:171–183. doi: 10.1152/jn.1996.75.1.171. [DOI] [PubMed] [Google Scholar]
- Willott JF, Turner JG. Neural plasticity in the mouse inferior colliculus: relationship to hearing loss, augmented acoustic stimulation, and prepulse inhibition. Hear Res. 2000;147:275–281. doi: 10.1016/s0378-5955(00)00137-4. [DOI] [PubMed] [Google Scholar]
- Winer JA. Three systems of descending projections to the inferior colliculus. In: Winer JA, Schreiner CE, editors. The Inferior Colliculus. Vol. 1. New York: Springer; 2005. pp. 231–247. Chapter 8. [Google Scholar]
- Wu SH, Ma CL, Sivaramakrishnan S, Oliver DL. Synaptic modification in neurons of the central nucleus of the inferior colliculus. Hear Res. 2002;168:43–54. doi: 10.1016/s0378-5955(02)00375-1. [DOI] [PubMed] [Google Scholar]
- Yu X, Sanes DH, Aristizabal O, Wadghiri YZ, Turnbull DH. Large-scale reorganization of the tonotopic map in mouse auditory midbrain revealed by MRI. Proc Natl Acad Sci U S A. 2007;104:12193–12198. doi: 10.1073/pnas.0700960104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang LI, Bao S, Merzenich MM. Persistent and specific influences of early acoustic environments on primary auditory cortex. Nat Neurosci. 2001;4:1123–1130. doi: 10.1038/nn745. [DOI] [PubMed] [Google Scholar]
- Zhang LI, Bao S, Merzenich MM. Disruption of primary auditory cortex by synchronous auditory inputs during a critical period. Proc Natl Acad Sci U S A. 2002;99:2309–2314. doi: 10.1073/pnas.261707398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Hakes JJ, Bonfield SP, Yan J. Corticofugal feedback for auditory midbrain plasticity elicited by tones and electrical stimulation of basal forebrain in mice. The European journal of neuroscience. 2005;22:871–879. doi: 10.1111/j.1460-9568.2005.04276.x. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Wu SH. Long-term potentiation in the inferior colliculus studied in rat brain slice. Hear Res. 2000;147:92–103. doi: 10.1016/s0378-5955(00)00123-4. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Yan J. Corticothalamic feedback for sound-specific plasticity of auditory thalamic neurons elicited by tones paired with basal forebrain stimulation. Cereb Cortex. 2008;18:1521–1528. doi: 10.1093/cercor/bhm188. [DOI] [PubMed] [Google Scholar]