Abstract
The aim of this review is to describe recent advances and topics in the surgical management of bile duct cancer. Radical resection with a microscopically negative margin (R0) is the only way to cure cholangiocarcinoma and is associated with marked survival advantages compared to margin-positive resections. Complete resection of the tumor is the surgeon’s ultimate aim, and several advances in the surgical treatment for bile duct cancer have been made within the last two decades. Multidetector row computed tomography has emerged as an indispensable diagnostic modality for the precise preoperative evaluation of bile duct cancer, in terms of both longitudinal and vertical tumor invasion. Many meticulous operative procedures have been established, especially extended hepatectomy for hilar cholangiocarcinoma, to achieve a negative resection margin, which is the only prognostic factor under the control of the surgeon. A complete caudate lobectomy and resection of the inferior part of Couinaud’s segment IV coupled with right or left hemihepatectomy has become the standard surgical procedure for hilar cholangiocarcinoma, and pylorus-preserving pancreaticoduodenectomy is the first choice for distal bile duct cancer. Limited resection for middle bile duct cancer is indicated for only strictly selected cases. Preoperative treatments including biliary drainage and portal vein embolization are also indicated for only selected patients, especially jaundiced patients anticipating major hepatectomy. Liver transplantation seems ideal for complete resection of bile duct cancer, but the high recurrence rate and decreased patient survival after liver transplant preclude it from being considered standard treatment. Adjuvant chemotherapy and radiotherapy have a potentially crucial role in prolonging survival and controlling local recurrence, but no definite regimen has been established to date. Further evidence is needed to fully define the role of liver transplantation and adjuvant chemo-radiotherapy.
Keywords: Bile duct cancer, Cholangiocarcinoma, Surgery, Liver transplantation, Hepatectomy, Pancreaticoduodenectomy, Adjuvant chemotherapy, Adjuvant radiation
INTRODUCTION
Although bile ducts are anatomically classified as either intrahepatic or extrahepatic and cholangiocarcinoma refers to malignant tumors originating from epithelial cells lining both the intrahepatic and extrahepatic biliary trees[1], the term “bile duct cancer” usually refers to extrahepatic bile duct cancer. Histologically speaking, both extrahepatic and intrahepatic cholangiocarcinoma could be considered together, but they are usually discussed as separate entities based on critical differences in their clinical manifestations[2,3] and current prevalent staging systems describe these entities by completely different classifications[4,5]. Additionally, the term “extrahepatic cholangiocarcinoma/bile duct cancer” encompasses hilar cholangiocarcinoma and distal bile duct cancer[1-3].
In this review article, we describe the current diagnosis and treatments of “bile duct cancer”, referring to extrahepatic cholangiocarcinoma, and focusing on the surgical treatment.
CLASSIFICATION OF EXTRAHEPATIC CHOLANGIOCARCINOMA
Anatomic classification
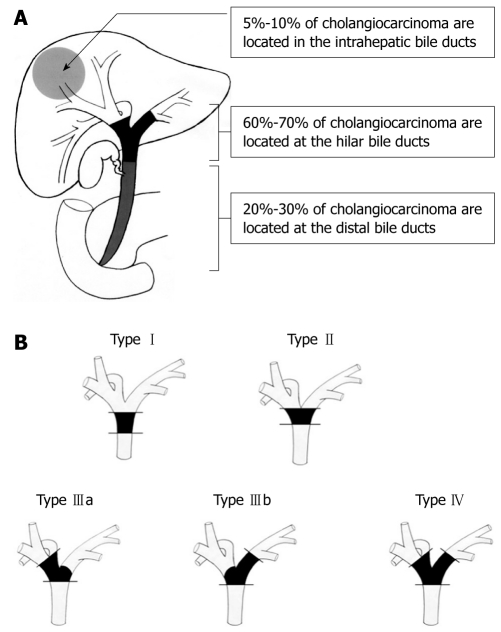
Extrahepatic bile duct cancer is further classified as hilar or distal. Hilar cholangiocarcinoma, also called a Klatskin tumor[6], is located within 2 cm of the bifurcation of the common bile duct and is further divided into four types by Bismuth and Corlette based on the anatomic location of the tumor[1,7]. Approximately 60% to 70% of cholangiocarcinoma is reported to be located at the hilum, 20% to 30% at the distal bile duct, and 5% to 10% intrahepatic bile duct[1,8,9]. The anatomic schema is presented in Figure 1. This classification, based on the longitudinal location of the bile duct cancer, defines the surgical strategy, including the operability and curability.
Figure 1.
Anatomic classification of cholangiocarcinoma. A: The majority of cholangiocarcinoma (60%-70%) develop in the hilar bile duct and are called Klatskin tumors. The distal bile duct is involved in 20% to 30% of cases, while intrahepatic cholangiocarcinomas represent 5% to 10% of the tumors originating from the biliary tract; B: Bismuth-Corlette classification of hilar bile duct cancer. Type I, cholangiocarcinoma confined to the common bile duct; Type II, cholangiocarcinoma involves the bifurcation of the common bile duct; Type IIIa, cholangiocarcinoma involves the bifurcation and the right hepatic duct; Type IIIb, cholangiocarcinoma involves the bifurcation and the left hepatic duct; Type IV, cholangiocarcinoma involves the bifurcation and extends to both the right and left hepatic ducts.
Pathologic classification
Most cholangiocarcinomas are well-to-moderately differentiated adenocarcinomas with a tendency to develop desmoplastic reactions and early perineural invasion[1,2,10]. Macroscopically, extrahepatic cholangiocarcinoma develops sclerosing strictures, nodular lesions, or papillary growth[11]. The sclerosing type is the most common, while the papillary pattern is rare but associated with a more favorable prognosis[12,13].
MODE OF CANCER SPREAD AND INFILTRATION
Understanding the patterns of anatomic spread and infiltration of bile duct cancer is critical for planning treatment. Infiltration by bile duct cancer includes both longitudinal extension and vertical invasion. Longitudinal extension refers to the longitudinal spread of the tumor along the biliary tree, and vertical invasion refers to direct invasion of the surrounding pancreas or duodenum, infiltration into the hepatoduodenal ligament including the adjacent hepatic artery and portal vein, and direct invasion of the hepatic parenchyma[14-16]. Distant metastasis and lymph node invasion could be an extension of vertical invasion.
Longitudinal extension
The longitudinal spread determines the type of radical operation, including pancreaticoduodenectomy (PD), extended hepatectomy and hepatopancreaticoduodenectomy, and apparent infiltration beyond the secondary branches on both sides of the biliary tree is generally considered unresectable.
Microscopic extension of bile duct cancer beyond the tumor margin visualized by present diagnostic modalities or the margin observed macroscopically is often encountered. Longitudinal extension consists of superficial and submucosal infiltration depending on the tumor growth pattern, and sometimes includes direct infiltration along lymphatic and perineural tissues[13]. Sakamoto et al[17] reported a correlation between the gross tumor type and the pattern of infiltration, and demonstrated that mucosal extension predominantly occurs with papillary (intraductal) and nodular (mass-forming) tumors, while submucosal extension mainly occurs with sclerosing (infiltrating) tumors. Tumor spread beyond the macroscopic margin is determined by the type of invasion, with a mean length of 6-10 mm for submucosal spread and 10-20 mm for mucosal spread[18]. Therefore, a gross surgical margin of more than 1 cm in the infiltrating type and more than 2 cm in the papillary and nodular types is recommended to obtain microscopically negative margins.
Vertical infiltration
The evaluation of vertical infiltration is critical to the patient’s prognosis, because it usually defines resectability and curability. Apparent distant metastasis and para-aortic lymph node metastasis are absolute contraindications for radical surgery[19-21], while direct invasion of major vessels, regardless of the prognosis, present surgical challenges whose indication and operative procedures continue to be debated[22-28].
Patients with cancer invasion confined to the fibromuscular layer of the extrahepatic bile duct have a better postoperative survival rate (80%-100% 5-year survival), while those with cancer that extends beyond the fibromuscular layer have a poor prognosis[29-31].
Perineural invasion beyond the bile duct wall is a unique characteristic of cholangiocarcinoma that is observed in 75% of cases and has proved to be a significant prognostic factor for poor outcome[14,16,32].
Hilar cholangiocarcinoma easily infiltrates into the hepatic parenchyma and the hepatoduodenal ligament, in which the hepatic artery and portal vein are located adjacent to the bile duct, while distal bile duct cancer directly invades the pancreas or duodenum[33,34]. Up to 80% of hilar cholangiocarcinoma directly infiltrates the liver parenchyma and surrounding connective tissues of the hepatoduodenal ligament[34,35], thus necessitating meticulous three-dimensional knowledge of the hepatic hilum and challenging operations for this disease entity[36]. One of the most important points of anatomic consideration for the vertical invasion of hilar cholangiocarcinoma is the need for hepatectomy with complete caudal lobectomy. Hilar cholangiocarcinoma spreads not only longitudinally along the right and left hepatic ducts, but also in the cranial and dorsal directions along the thin bile ducts. Hence, it is necessary to remove the liver parenchyma adjacent to the hepatic hilum together with the hilar plate to achieve a complete curative resection. In this sense, complete caudate lobectomy and resection of the inferior part of Couinaud’s segment IV coupled with right or left hemihepatectomy (according to the predominant tumor location) is the main goal of surgical resection of hilar cholangiocarcinoma[37-41]. Major hepatectomy with caudate lobectomy for hilar cholangiocarcinoma is associated with improved outcome[26,41-45].
PREOPERATIVE EVALUATION OF BILE DUCT CANCER
Preoperative evaluation of bile duct cancer in terms of radical resection consists of a multidisciplinary approach with ultrasonography (US), helical-computed tomography, magnetic resonance imaging (MRI) including MR cholangiography (MRC), direct cholangiography via endoscopic retrograde cholangiography (ERC) or percutaneous transhepatic biliary drainage (PTBD), intraductal US (IDUS), and bile cytology or biopsy[1].
Among these, dynamic multidetector row computed tomography (MDCT) is now widely used for the preoperative evaluation and staging of bile duct cancer, as it provides not only qualitative diagnosis and indicates the location of the tumor, but also shows the relationship between adjacent tissues, such as the hepatic artery, portal vein, and liver parenchyma[46-48].
Bile duct cancer is often revealed as a focal thickening of the ductal wall with various enhancement patterns[49,50]. The accuracy of the differential diagnosis of a malignant lesion from benign stenosis is reported to be over 90%[48], with satisfactory accuracy in evaluating major vessel involvement and liver parenchyma invasion[49-55]. Yet, lymph node metastasis is still difficult to diagnose preoperatively, even with the recent higher resolution of MDCT[51-54]. Some authors report that MDCT is effective for evaluating even longitudinal spread along the bile duct, demonstrating that the efficacy is equivalent to that of evaluation via MRC or direct cholangiography[56-62]. We reported that the evaluation of longitudinal tumor spread by MDCT was even superior to that of direct cholangiography and equivalent to histologic assessment of the specimen[62]; however, in cases with hilar cholangiocarcinoma, it seems still difficult to estimate the ductal spread precisely, even with the recent advancements of MDCT[53,63].
Additional important information obtained from MDCT and its three-dimensional reconstruction for surgeons is the precise arterial/portal/venous anatomy around the hepatic hilum/hepatoduodenal ligament/pancreas head/duodenum in relation to the tumor. Many authors have reported that MDCT is effective for preoperative planning and for navigation during the operation[46,59,62]. Figure 2 shows a multiple fusion image of a three-dimensional reconstruction of MDCT in a case of hilar cholangiocarcinoma.
Figure 2.
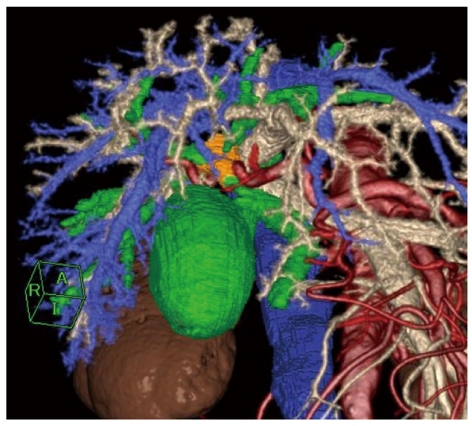
Hilar cholangiocarcinoma (Bismuth-Corlette type IIIa). Comprehensive multiphase fusion images of the tumor (orange), bile duct (green), and surrounding vessels including hepatic artery (red), portal vein (light yellow), and hepatic vein (blue).
MRI with concurrent MRC provides three-dimensional reconstruction of the biliary tree, and the diagnostic accuracy in evaluating cholangiocarcinoma is reported to be comparable to that of invasive cholangiography via ERC or PTBD[64-68]. MRI also facilitates the evaluation of vertical invasion of the tumor, similar to MDCT[65,69,70]. To exclude artifacts of biliary instrumentation and obtain precise images of ductal wall thickening and luminal stenosis/dilatation, both MDCT and MRI are strongly recommended before decompressing the biliary tree.
Despite the controversy regarding the necessity for preoperative biliary drainage to reduce surgical morbidity, direct cholangiography via endoscopic naso-biliary drainage (ENBD) tube or PTBD tube remains the gold standard for preoperative evaluation of ductal spread, especially in Japan[71-74]. A drawback of these invasive procedures is the risk of complications such as post-ERC pancreatitis[75], bacterobilia and cholangitis[76], bleeding, sepsis, catheter tract seeding, and even death[77]. With the recent improvements in the diagnostic accuracy of other noninvasive imaging modalities, as mentioned above, these invasive procedures might soon not be essential, at least as preoperative diagnostic tools. On the other hand, ERC and PTBD have the advantage of enabling IDUS or choledochoscopy and of providing bile cytology, brushing cytology, and biopsy, which can confirm the diagnosis of cholangiocarcinoma[78]. Unfortunately, these histopathologic examinations yield low sensitivity, and non-diagnostic cytology or biopsy results may not rule out cholangiocarcinoma in the presence of appropriate radiologic findings[79,80]. In the absence of other explainable causes of biliary strictures, patients should be assumed to have cancer and operated on as such, accepting that 10% to 15% might prove to have a benign lesion on the final histologic investigation[81,82]. Fukuda et al[83] reported 100% sensitivity of bile duct biopsy by adding choledochoscopy to ERC, and a new technology such as SpyGlass®, which is currently under investigation, may soon improve the diagnostic accuracy of preoperative malignancy confirmation[84,85].
PREOPERATIVE TREATMENTS
Biliary drainage
The efficacy of preoperative biliary drainage for patients with obstructive jaundice remains controversial. Based on previous reports emphasizing the adverse effects of biliary drainage, such as pancreatitis, cholangitis, and tract seeding[76,86-91], and the recent meta-analysis comparing surgery with preoperative biliary drainage to that without drainage, which showed no beneficial effect of preoperative drainage[77], routine biliary drainage is not recommended for all jaundiced patients, especially for distal bile duct cancer without any complications[92]. Furthermore, preoperative biliary drainage seems to increase the risk of perioperative infections and a longer postoperative hospital stay[76,77]. Preoperative biliary drainage has proved to be beneficial, however, in the presence of cholangitis, severe malnutrition, and coagulation abnormalities[93,94], and it is absolutely indicated for patients requiring major hepatic resection for curative surgery[36,71]. Prolonged preoperative jaundice is associated with increased postoperative morbidity and mortality after hepatic resection due to severe cholestatic liver dysfunction[95,96]. When biliary drainage is performed for patients awaiting major hepatectomy, radical surgery should be postponed for several weeks until the serum total bilirubin is less than 2.0 mg/dL to allow for sufficient restoration of hepatic function[36]. The use of sequential liver volumetric analyses and hepatic functional studies is warranted when anticipating extended hepatectomy to secure a sufficient volume and function of the future remnant liver, thereby minimizing the risk of postoperative liver failure[97].
Portal vein embolization
As discussed above, hilar cholangiocarcinoma usually requires extended hepatectomy (i.e. extended right hepatectomy, and right or left trisegmentectomy), which is related with a rather high rate of perioperative mortality (0% to 19%)[98]. This is partly due to the increased rate of postoperative liver failure with major hepatic resection[99-101]. Portal vein embolization, which was first indicated for hilar cholangiocarcinoma by Makuuchi et al[102], is now widely accepted as a valuable preoperative measure in anticipation of extensive liver resection with a subsequent small liver remnant volume[103-108]. Compensatory hypertrophy of the remnant liver parenchyma, usually an increase of 8% to 20% within 2 to 6 wk, is induced in association with atrophy of the future resected liver by selectively occluding the main portal branch to the liver parenchyma to be removed. In general, portal vein embolization can benefit patients requiring a future liver remnant volume of less than 25% to 35% of the original liver volume, yet the indication is still controversial, especially for patients with normal liver function. Currently, there seems to be no objection to portal vein embolization for potentially resectable patients with normal liver function when the anticipated future liver remnant volume is less than 20% of the total liver volume, or in patients with compromised liver function when the anticipated future liver remnant volume is less than 40% of the total liver volume[98]. Once portal vein embolization is performed, sequential evaluation of liver function and volumetry is mandatory not to miss the optimal timing for radical surgery[109].
SURGICAL TREATMENT OF BILE DUCT CANCER
Considering that radical resection with a microscopically negative margin (R0) is the only way to cure bile duct cancer and is associated with marked survival advantages compared to margin-positive resections (R1, microscopically positive; R2, macroscopically positive), achieving complete resection of the tumor is the most critical mission for surgeons. Many authors have reported meticulous operative procedures and their outcomes during last two decades with an attempt to achieve the most radical and safe resection of bile duct cancer.
Pancreaticoduodenectomy for distal bile duct cancer
Pancreaticoduodenectomy (PD), including pylorus preserving PD (PPPD) coupled with porta hepatis lymphadenectomy, is the standard treatment of choice for the complete removal of distal bile duct cancer, but extended lymphadenectomy including the para-aortic nodes is not justified because it does not provide a survival advantage and is associated with increased perioperative morbidity[20,21,110,111]. Both PD and PPPD provide equal outcome for distal bile duct cancer[20,21,112-114], while segmental bile duct excision is rarely an option because only 10% of patients undergoing bile duct excision alone obtain curative resection margins on final pathology[110,115]. Based on the equivalent outcome between segmental bile duct resection and PD recently reported by Lee et al[116], segmental bile duct resection with excision of surrounding lymph nodes and connective tissues seems to be a possible strategy[117], yet it is not accepted as a standard operation for distal bile duct cancer for obtaining a negative surgical margin[118].
Reports from high-volume centers during the last decade are summarized in Table 1[21,33,116,119-131]. The overall 3- and 5-year survival rates after radical surgery ranged from 33% to 63% and 16% to 52%, respectively. A recent meta-analysis by Japanese Biliary Tract Cancer Statistics Registry revealed that the overall 3- and 5-year survival rates of distal bile duct cancer after radical resection were 58% and 44%, respectively, among 779 patients who received PD or PPPD between 1998 and 2004 in Japan[132,133].
Table 1.
Review of the literature on pancreaticoduodenectomy for distal bile duct cancer
| Author | Yr | Resections (n) | R0 resection (%) | Overall 3-yr survival (%) | Overall 5-yr survival (%) | R0 5-yr survival (%) | Operative mortality (%) |
| Kayahara et al[119] | 1999 | 50 | 72 | 47 | 35 | 48 | 2 |
| Suzuki et al[120] | 2000 | 99 | 52 | 50 | 37 | 52 | 3 |
| Yeo et al[21] | 2002 | 49 | NA | 38 | 16 | NA | 33 |
| Yoshida et al[121] | 2002 | 27 | 85 | 37 | 37 | 44 | 4 |
| Sakamoto et al[122] | 2005 | 55 | 84 | 52 | 26 | NA | 7 |
| Jang et al[123] | 2005 | 103 | 84 | 38 | 30 | NA | 5 |
| Cheng et al[124] | 2007 | 112 | 87 | 51 | 25 | 27 | 3 |
| Murakami et al[125] | 2007 | 36 | 81 | 54 | 50 | 62 | 0 |
| Sasaki et al[126] | 2007 | 77 | 92 | NA | 37 | 362 | NA |
| DeOliveria et al[127]1 | 2007 | 239 | 78 | 35 | 23 | 27 | 3 |
| Allen et al[128] | 2008 | 98 | 85 | 45 | 43 | 422 | 3 |
| Bahra et al[129] | 2008 | 95 | 81 | 36 | 29 | 34 | 4 |
| Lee et al[116] | 2009 | 149 | NA | 46 | 38 | NA | NA |
| Hong et al[33] | 2009 | 147 | 90 | 33 | 18 | NA | NA |
| Nomura et al[130] | 2009 | 57 | 61 | NA | 36 | NA | 2 |
| Kawai et al[131] | 2010 | 62 | 77 | 63 | 52 | 59 | 0 |
1Including palliative operations;
Including hilar cholangiocarcinoma;
Including operations for pancreatic cancer. NA: Not available; R0: Negative surgical margin.
Extended hepatectomy for hilar cholangiocarcinoma
Based on the mode of tumor extension and the radicality and simplicity of the procedure, extended right- or left-hemihepatectomy is regarded as the standard radical operation for hilar cholangiocarcinoma[37-39,134], even for patients with Bismuth and Corlette type I or II[118]. Extended right hemihepatectomy consists of the resection of the right liver, the inferior part of segment IV, the hilar plate, and the entire caudate lobe, while extended left hemihepatectomy consists of resection of the left liver, the hilar plate of the right paramedian sector, and most of the caudate lobe, both coupled with complete resection of the extrahepatic bile duct and porta hepatis lymphadenectomy. “Right” or “left” is dependent on the predominance of the tumor, but an extended right-hemihepatectomy is preferentially indicated for even centrally located tumors, because of the length of each hepatic duct, location of the hilar common bile duct in the hepatoduodenal ligament, facility of complete caudate lobectomy, and the ease of portal vein reconstruction[39,135]. When the tumor spreads diffusely into the intrapancreatic bile duct, PD should be added to extended hemihepatectomy simultaneously[136-138].
Right or left trisegmentectomy is one of the most extensive hepatic resection procedures because of the massive volume loss of the liver parenchyma, and hilar cholangiocarcinoma invading the hepatic hilum sometimes requires trisegmentectomy for curative resection. Trisegmentectomy is advantageous in terms of obtaining a negative margin of the bile ducts[139,140]. A negative ductal margin is achieved in 75% and 88% of cases with left and right trisegmentectomy, respectively, both of which are higher than the negative rate obtained with extended hemihepatectomy[139,140]. On the other hand, minimally invasive procedures, including parenchyma-preserving hepatectomy and bile duct segmental resection without hepatectomy, could be an option for Bismuth-Corlette type I and II or cancer of the middle bile duct, but in terms of obtaining R0, those procedures are indicated for strictly localized tumors or for patients with a poor general condition or high-risk factors[36,41,116,117]. Many authors report improved survival with complete caudate lobectomy and major hepatectomy[26,41-45].
Metastasis to regional lymph nodes is one of the most important prognostic factors influencing survival after resection for hilar cholangiocarcinoma[45,141]. Patients with nodal involvement beyond the hepatoduodenal ligament, including para-aortic nodal metastases, were shown to have dismal prognosis with a 5-year survival of 0% to 12%[141-144]. Therefore, routine lymph node dissection beyond the hepatoduodenal ligament is not recommended. On the other hand, portal vein resection and reconstruction has been performed for hilar cholangiocarcinoma with conflicting results[145]. With recent technical advances, several retrospective series have shown no difference in surgical mortality between patients undergoing major hepatectomy with portal vein resection and without it, advocating routine resection of the portal vein for en-bloc, “no-touch” resection[25,26], but the impact of portal vein resection on long-term survival seems to be less clear. Moreover, other studies have shown equivalent or worse survival in patients undergoing portal vein resection[23,27,28]. When there is severe adhesion between the tumor and portal vein, combined resection and reconstruction is necessary to obtain a possible negative surgical margin, yet routine resection of the portal vein might not be recommended unless supported by findings from a randomized clinical trial.
Reports from high-volume centers during the last decade are summarized in Table 2[26,45,71,73,128,135,142,146-160]. The overall 3- and 5-year survival rates after radical surgery ranged from 37% to 60% and 20% to 42%, respectively. A recent meta-analysis by the Japanese Biliary Tract Cancer Statistics Registry revealed that the overall 3- and 5-year survival rates of hilar bile duct cancer after radical resection were 47% and 39%, respectively, among 255 patients who received major hepatectomy during 1998 and 2004 in Japan[132,133].
Table 2.
Review of the literature on hepatectomy for hilar bile duct cancer
| Author | Yr | Resections (n) | Major hepatectomy (%) | R0 resection (%) | Overall 3-yr survival (%) | Overall 5-yr survival (%) | R0 5-yr survival (%) | Operative mortality (%) |
| Neuhaus et al[26] | 1999 | 80 | 85 | 61 | NA | 22 | 37 | 8 |
| Todoroki et al[142] | 2000 | 101 | 58 | 14 | NA | 28 | 38 | 4 |
| Lee et al[73] | 2000 | 111 | 99 | 78 | 52 | 22 | NA | 6 |
| Nimura et al[71] | 2000 | 142 | 90 | 61 | 43 | 26 | 27 | 9 |
| Capussotti et al[146] | 2002 | 36 | 83 | 89 | 41 | 27 | 29 | 3 |
| Seyama et al[147] | 2003 | 87 | 67 | 64 | 55 | 40 | 46 | 0 |
| Kawasaki et al[135] | 2003 | 79 | 87 | 68 | NA | 22 | 40 | 1 |
| Rea et al[148] | 2004 | 46 | 100 | 80 | 39 | 26 | 30 | 9 |
| Hemming et al[149] | 2005 | 53 | 98 | 80 | 60 | 35 | 45 | 9 |
| Dinant et al[150] | 2006 | 99 | 38 | 31 | 37 | 27 | 33 | 15 |
| Sano et al[151] | 2007 | 126 | 100 | 56 | 44 | 35 | NA | 4 |
| Baton et al[152] | 2007 | 59 | 100 | 46 | 45 | 20 | 22 | 5 |
| Hasegawa et al[153] | 2007 | 49 | 90 | 78 | 50 | 40 | 50 | 2 |
| Allen et al[128] | 2008 | 106 | 82 | 77 | 45 | 29 | 421 | 4 |
| Ito et al[45] | 2008 | 38 | 53 | 63 | 65 | 33 | 60 | 0 |
| Hirano et al[154] | 2009 | 146 | 90 | 90 | 53 | 36 | NA | 3 |
| Lee et al[155] | 2009 | 302 | 89 | 71 | 41 | 33 | 47 | 2 |
| Young et al[156] | 2009 | 51 | 92 | 57 | 36 | 20 | 40 | 8 |
| Igami et al[157] | 2009 | 298 | 98 | 74 | 49 | 42 | 52 | 2 |
| Miyazaki et al[158] | 2009 | 107 | 91 | 59 | 45 | 28 | 33 | 2 |
| Unno et al[159] | 2009 | 125 | 100 | 63 | 37 | 35 | 46 | 8 |
| Murakami et al[160] | 2009 | 42 | 86 | 74 | 42 | 30 | NA | 7 |
Including distal bile duct cancer. NA: Not available; R0: Negative surgical margin.
Liver transplantation for hilar cholangiocarcinoma
For the treatment of locally advanced hilar cholangiocarcinoma beyond the indication for resection, orthotopic liver transplantation (OLT) may offer the advantage of resection of all structures involved by the tumor, including vessels within the hepatoduodenal ligament, all intra- and extrahepatic bile ducts, and whole liver parenchyma. Patients requiring a total hepatectomy to achieve a negative margin and those with underlying liver failure precluding hepatic resection are possible candidates for OLT, but early experience with OLT for hilar cholangiocarcinoma was disappointing with early recurrence rates of more than 50% and a 5-year survival rate of 10% to 20%[161-164]. Recently, in highly selected patients undergoing neoadjuvant protocols, improved survivals were reported with a 5-year survival rate of 30% to 45%[165-167]. More recently, the so-called “Mayo protocol” was reported with the intent of treating a highly selected group of patients with cholangiocarcinoma with a strict regimen of preoperative staging and neoadjuvant chemoradiation followed by OLT[168-170]. The inclusion criteria of this protocol are as follows, (1) locally advanced unresectable disease with positive intraluminal cytology or biopsy, or CA19-9 >100 with radiographic features of malignancy; (2): primary sclerosing cholangitis with resectable bile duct cancer; and (3): absence of medical contraindication for OLT. Eligible patients receive neoadjuvant chemoradiation therapy comprising external radiation of 4500 cGy with concomitant fluorouracil, and transcatheter Iridium-192 brachytherapy of 2000-3000 cGy, followed by oral capecitabine as tolerated until OLT. Additionally, patients should undergo a staging laparotomy to rule out metastatic disease prior to OLT. In 38 patients who underwent this protocol, an 82% 5-year disease free survival was reported. Currently, the indication for OLT for the treatment of cholangiocarcinoma is reserved for highly selected patients in specialized centers.
PROGNOSTIC FACTORS AFTER SURGICAL RESECTION
A variety of clinicopathologic factors have been reported as prognostic factors in previous studies after curative intent surgery for bile duct cancer. Among those, the most frequently reported factors with a negative impact on patient survival are as follows: positive surgical margin[26,34,45,96,119,121,126,127,129,130,135,141,144,149,152,153,155,157,159], nodal involvement (including numbers of metastatic lymph node)[19,23,26,32,34,45,110,111,117,119,121,123-127,130,131,135,141-144,147,150,152,153,155,171], histologic morphology of the tumor[12,13,23,26,34,45,96,123,127,141,142,144,148,159], perineural invasion[14,26,29-33,117,122,131,159], and limited resection of the bile duct[34,41-45,150]. Of these, complete surgical resection with a microscopically negative margin is the only factor under the control of the surgeon and is therefore the most important goal of surgical treatment. A recent study emphasized the significance and accuracy of intraoperative assessment of bile duct margin[172], while a positive bile duct margin itself seems to have minimum impact on patient survival[12,173,174]. As noted above, there is a close association between the extent of the hepatic resection and the rate of negative margins in hilar cholangiocarcinoma[175]. These factors all warrant an aggressive surgical approach to bile duct cancer.
ADJUVANT THERAPY
Because of the high rate of recurrence and poor survival after radical surgery, postoperative chemotherapy, radiotherapy, and chemoradiation have been evaluated in terms of improving patient survival after resection of bile duct cancer[176].
Adjuvant chemotherapy
A previous randomized trial revealed that chemotherapy significantly improved survival and quality of life compared to best supportive care for unresectable cholangiocarcinoma[177]. The most extensively studied agents in unresectable cholangiocarcinoma are fluorouracil (5-FU) and gemcitabine, which have been investigated as a single agent[177-179] and in combination with other drugs, such as mitomycin C[178], leucovorin[180], cisplatin[181], capecitabine[182], epirubicin[183], and oxaliplatin[184]. Eckel et al[185] conducted a pooled analysis of 104 chemotherapy studies in advanced bile duct cancers, which suggested that gemcitabine combined with cisplatin or oxaliplatin resulted in the best response without significantly improved survival. On the other hand, reports on adjuvant chemotherapy after resection are scarce (Table 3). A recent multicenter randomized trial evaluated the effect of adjuvant chemotherapy with mitomycin C and 5-FU versus surgery alone for patients with pancreato-biliary malignancies, in which no survival benefit was seen for 139 patients with R0 resection for cholangiocarcinoma[186]. Recent institutional retrospective experiences found that gemcitabine-based adjuvant chemotherapy after curative-intent surgery significantly improved patient survival[160,187]. In summary, gemcitabine in combination with cisplatin or oxaliplatin seems to be the most efficacious regimen in cholangiocarcinoma, but adjuvant chemotherapy alone cannot yet be considered standard therapy after resection.
Table 3.
Review of the literature on adjuvant therapy for surgically resected bile duct cancer
| Author | Yr | Background | P-value | Comments | ||
| Chemotherapy | Without chemotherapy | With chemotherapy | ||||
| Takada et al[186] | 2002 | Curative-resection | 28% 5-yr OS | 41% 5-yr OS | 0.48 | Two courses of mitomycin C plus infusional 5-FU, followed by oral 5-FU until tumor progression; 40% received a non-curative operation |
| Non-curative resection | 16% 5-yr OS | 8% 5-yr OS | 0.300 | |||
| Yubin et al[187] | 2008 | Hilar cholangiocarcinoma | 37 mo MS | 43 mo MS | < 0.05 | Infusional gemcitabine based; no discrimination between curative and palliative resection; some of patients received radiation but the contribution of radiation was not analyzed |
| Murakami et al[160] | 2009 | Hilar cholangiocarcinoma | 23% 5-yr OS | 57% 5-yr OS | 0.026 | Infusional gemcitabine and oral S-1, every 2 wk |
| Radiation | Without radiation | With radiation | ||||
| Pitt et al[188] | 1995 | Hilar cholangiocarcinoma | 20 mo MS | 20 mo MS | NS | |
| Cameron et al[189] | 1990 | Hilar cholangiocarcinoma | 21% 3-yr OS | 21% 3-yr OS | NS | |
| Zlotecki et al[190] | 1998 | Bile duct cancer | 19% 5-yr OS | 35% 5-yr OS | NS | |
| Heron et al[195] | 2003 | Distal cholangiocarcinoma | 63 mo MS | MS not reached (> 129 mo) | NS | |
| Gonzalez et al[194] | 1999 | Hilar cholangiocarcinoma Hilar cholangiocarcinoma | 0.023 | |||
| 8 mo MS | 19 mo MS | 0.001 | ||||
| Schoenthaler et al[193] | 1994 | R1 resection | 11 mo MS | 22 mo MS, X-rays | 0.010 | |
| 61 mo MS, charged particles | 0.001 | |||||
| Cheng et al[192] | 2007 | Hilar cholangiocarcinoma | HR 4.3, 95% CI 3.6-4.9 | < 0.01 | Radiation was significantly helpful for R1/2 resection patients | |
| Todoroki et al[191] | 2000 | Bile duct cancer | 14% 5-yr OS | 34% 5-yr OS | 0.010 | Radiation was significantly helpful for R1 resection patients |
| Gerhards et al[197] | 2003 | Hilar cholangiocarcinoma | 8 mo MS | 24 mo MS | < 0.05 | |
| Chemoradiation | Without chemoradiation | With chemoradiation | ||||
| Nakeeb et al[198] | 2002 | Bile duct cancer | 11 mo MS (Chemotherapy alone) | 16 mo OS | 0.020 | Chemotherapy; 5-FU |
| 8 mo MS (Radiation alone) | ||||||
| Kim et al[199] | 2002 | Bile duct cancer | 9% 5-yr OS (Radiation alone) | 41% 5-yr OS | 0.0005 | Chemotherapy; 5-FU, P = 0.14 in multivariate analysis |
| Serafini et al[200] | 2001 | Bile duct cancer | 29 mo MS (Surgery alone) | 42 mo MS | 0.07 | Chemotherapy; 5-FU; when stratified by location, only distal tumors significantly benefited (41 mo MS vs 25 mo MS, P < 0.05) |
| Hughes et al[201] | 2007 | Distal cholangiocarcinoma | 22 mo MS (Surgery alone) | 37 mo MS | 0.04 | Chemotherapy; 5-FU |
MS: Mean survival; NS: Not significant; OS: Overall survival; R1: Microscopically positive margin; R2: Macroscopically positive margin; HR: Hazard ratio; CI: Confidential interval.
Adjuvant radiotherapy
Several studies have described adjuvant external beam radiotherapy, with or without dose escalation by intraluminal brachytherapy (Table 3). One prospective study and some retrospective studies found no survival benefit of adjuvant radiotherapy in patients who received a curative intent resection for cholangiocarcinoma[188-190]. In contrast, several large retrospective series suggested a survival benefit with adjuvant radiation. Todoroki et al[191] demonstrated a significantly higher 5-year survival of 34% in patients with R1 resections with adjuvant radiotherapy (intraoperative and extra beam) compared to 14% with surgery alone. Similarly, Cheng et al[192] and Schoenthaler et al[193] reported the efficacy of adjuvant radiation for the survival of patients with R1 or R2 resections. Other authors have corroborated the improved survival with adjuvant radiotherapy[194-197]. Although the bulk of retrospective data suggest that improved survival may be achieved with the use of adjuvant radiation, one prospective study was negative for survival benefit in a selected group of patients with hilar cholangiocarcinoma. Further prospective investigation is required to clarify the role of adjuvant radiation after resection.
Adjuvant chemoradiation therapy
The radiosensitization effect of chemotherapy has led to the investigation of concurrent chemoradiation as an adjuvant for resected cholangiocarcinoma. The most commonly utilized agent is 5-FU. Previous retrospective data revealed favorable outcomes of adjuvant chemoradiation (Table 3). Adjuvant chemoradiation has a survival benefit over adjuvant chemotherapy, adjuvant radiation, and surgery alone, especially in distal bile duct cancer[198-201]. Recently, three retrospective studies emphasized a comparable outcome of adjuvant chemoradiation for patients with non-curative surgery when compared to patients with curative surgery alone[202-204].
CONCLUSION
Several advances have been made in the surgical management of bile duct cancer within the last two decades. Surgical morbidity and mortality have been dramatically decreased, but the long-term outcome remains poor. Among several generally accepted prognostic factors, a negative resection margin is the only factor under the control of the surgeon, which has resulted in many meticulous endeavors to establish safe curative procedures and to achieve improved outcome. Further evidence is needed to fully define the role of liver transplantation. Additionally, advances in adjuvant therapy are warranted for improvement of the long-term outcome.
Footnotes
Peer reviewer: Jean-Nicolas Vauthey, MD, Professor of Surgery, Chief, Liver Service, The University of Texas M. D. Anderson Cancer Center, 1515 Holcombe Boulevard, Unit 444, Houston, TX 77030, United States
S- Editor Cheng JX L- Editor O’Neill M E- Editor Ma WH
References
- 1.Aljiffry M, Walsh MJ, Molinari M. Advances in diagnosis, treatment and palliation of cholangiocarcinoma: 1990-2009. World J Gastroenterol. 2009;15:4240–4262. doi: 10.3748/wjg.15.4240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.de Groen PC, Gores GJ, LaRusso NF, Gunderson LL, Nagorney DM. Biliary tract cancers. N Engl J Med. 1999;341:1368–1378. doi: 10.1056/NEJM199910283411807. [DOI] [PubMed] [Google Scholar]
- 3.Lazaridis KN, Gores GJ. Cholangiocarcinoma. Gastroenterology. 2005;128:1655–1667. doi: 10.1053/j.gastro.2005.03.040. [DOI] [PubMed] [Google Scholar]
- 4.Greene FL, Page DL, Flemming ID. AJCC (American Joint Committee on Cancer) Cancer staging Manual. 6th ed. New York: Splinger-Verlag; 2002. [Google Scholar]
- 5.Japanese Society of Biliary Surgery. Classification of biliary tract carcinoma. 2nd English ed. Tokyo: Kanehara; 2004. [Google Scholar]
- 6.Klatskin G. Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis. an unusual tumor with distinctive clinical and pathological features. Am J Med. 1965;38:241–256. doi: 10.1016/0002-9343(65)90178-6. [DOI] [PubMed] [Google Scholar]
- 7.Bismuth H, Corlette MB. Intrahepatic cholangioenteric anastomosis in carcinoma of the hilus of the liver. Surg Gynecol Obstet. 1975;140:170–178. [PubMed] [Google Scholar]
- 8.Patel T. Cholangiocarcinoma. Nat Clin Pract Gastroenterol Hepatol. 2006;3:33–42. doi: 10.1038/ncpgasthep0389. [DOI] [PubMed] [Google Scholar]
- 9.Ishak KG, Anthony PP, Sobin LH. World Health Organization International Histologic Classification of Tumors: Histological Typing of Tumors to the Liver. 2nd ed. Berlin: Springer Verlag; 1994. [Google Scholar]
- 10.Lim JH, Park CK. Pathology of cholangiocarcinoma. Abdom Imaging. 2004;29:540–547. doi: 10.1007/s00261-004-0187-2. [DOI] [PubMed] [Google Scholar]
- 11.Weinbren K, Mutum SS. Pathological aspects of cholangiocarcinoma. J Pathol. 1983;139:217–238. doi: 10.1002/path.1711390210. [DOI] [PubMed] [Google Scholar]
- 12.Jarnagin WR, Bowne W, Klimstra DS, Ben-Porat L, Roggin K, Cymes K, Fong Y, DeMatteo RP, D'Angelica M, Koea J, et al. Papillary phenotype confers improved survival after resection of hilar cholangiocarcinoma. Ann Surg. 2005;241:703–712; discussion 712-714. doi: 10.1097/01.sla.0000160817.94472.fd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Igami T, Nagino M, Oda K, Nishio H, Ebata T, Yokoyama Y, Shimoyama Y. Clinicopathologic study of cholangiocarcinoma with superficial spread. Ann Surg. 2009;249:296–302. doi: 10.1097/SLA.0b013e318190a647. [DOI] [PubMed] [Google Scholar]
- 14.Bhuiya MR, Nimura Y, Kamiya J, Kondo S, Fukata S, Hayakawa N, Shionoya S. Clinicopathologic studies on perineural invasion of bile duct carcinoma. Ann Surg. 1992;215:344–349. doi: 10.1097/00000658-199204000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ogura Y, Takahashi K, Tabata M, Mizumoto R. Clinicopathological study on carcinoma of the extrahepatic bile duct with special focus on cancer invasion on the surgical margins. World J Surg. 1994;18:778–784. doi: 10.1007/BF00298931. [DOI] [PubMed] [Google Scholar]
- 16.Yamaguchi K, Chijiiwa K, Saiki S, Shimizu S, Takashima M, Tanaka M. Carcinoma of the extrahepatic bile duct: mode of spread and its prognostic implications. Hepatogastroenterology. 1997;44:1256–1261. [PubMed] [Google Scholar]
- 17.Sakamoto E, Nimura Y, Hayakawa N, Kamiya J, Kondo S, Nagino M, Kanai M, Miyachi M, Uesaka K. The pattern of infiltration at the proximal border of hilar bile duct carcinoma: a histologic analysis of 62 resected cases. Ann Surg. 1998;227:405–411. doi: 10.1097/00000658-199803000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ebata T, Watanabe H, Ajioka Y, Oda K, Nimura Y. Pathological appraisal of lines of resection for bile duct carcinoma. Br J Surg. 2002;89:1260–1267. doi: 10.1046/j.1365-2168.2002.02211.x. [DOI] [PubMed] [Google Scholar]
- 19.Kitagawa Y, Nagino M, Kamiya J, Uesaka K, Sano T, Yamamoto H, Hayakawa N, Nimura Y. Lymph node metastasis from hilar cholangiocarcinoma: audit of 110 patients who underwent regional and paraaortic node dissection. Ann Surg. 2001;233:385–392. doi: 10.1097/00000658-200103000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Riall TS, Cameron JL, Lillemoe KD, Campbell KA, Sauter PK, Coleman J, Abrams RA, Laheru D, Hruban RH, Yeo CJ. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma--part 3: update on 5-year survival. J Gastrointest Surg. 2005;9:1191–1204; discussion 1204-1206. doi: 10.1016/j.gassur.2005.08.034. [DOI] [PubMed] [Google Scholar]
- 21.Yeo CJ, Cameron JL, Lillemoe KD, Sohn TA, Campbell KA, Sauter PK, Coleman J, Abrams RA, Hruban RH. Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma, part 2: randomized controlled trial evaluating survival, morbidity, and mortality. Ann Surg. 2002;236:355–366; discussion 366-368. doi: 10.1097/00000658-200209000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nagino M, Nimura Y, Nishio H, Ebata T, Igami T, Matsushita M, Nishikimi N, Kamei Y. Hepatectomy with simultaneous resection of the portal vein and hepatic artery for advanced perihilar cholangiocarcinoma: an audit of 50 consecutive cases. Ann Surg. 2010;252:115–123. doi: 10.1097/SLA.0b013e3181e463a7. [DOI] [PubMed] [Google Scholar]
- 23.Ebata T, Nagino M, Kamiya J, Uesaka K, Nagasaka T, Nimura Y. Hepatectomy with portal vein resection for hilar cholangiocarcinoma: audit of 52 consecutive cases. Ann Surg. 2003;238:720–727. doi: 10.1097/01.sla.0000094437.68038.a3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shimada H, Endo I, Sugita M, Masunari H, Fujii Y, Tanaka K, Misuta K, Sekido H, Togo S. Hepatic resection combined with portal vein or hepatic artery reconstruction for advanced carcinoma of the hilar bile duct and gallbladder. World J Surg. 2003;27:1137–1142. doi: 10.1007/s00268-003-6801-6. [DOI] [PubMed] [Google Scholar]
- 25.Hirano S, Kondo S, Tanaka E, Shichinohe T, Tsuchikawa T, Kato K. No-touch resection of hilar malignancies with right hepatectomy and routine portal reconstruction. J Hepatobiliary Pancreat Surg. 2009;16:502–507. doi: 10.1007/s00534-009-0093-7. [DOI] [PubMed] [Google Scholar]
- 26.Neuhaus P, Jonas S, Bechstein WO, Lohmann R, Radke C, Kling N, Wex C, Lobeck H, Hintze R. Extended resections for hilar cholangiocarcinoma. Ann Surg. 1999;230:808–818; discussion 819. doi: 10.1097/00000658-199912000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hemming AW, Kim RD, Mekeel KL, Fujita S, Reed AI, Foley DP, Howard RJ. Portal vein resection for hilar cholangiocarcinoma. Am Surg. 2006;72:599–604; discussion 604-605. [PubMed] [Google Scholar]
- 28.Muñoz L, Roayaie S, Maman D, Fishbein T, Sheiner P, Emre S, Miller C, Schwartz ME. Hilar cholangiocarcinoma involving the portal vein bifurcation: long-term results after resection. J Hepatobiliary Pancreat Surg. 2002;9:237–241. doi: 10.1007/s005340200025. [DOI] [PubMed] [Google Scholar]
- 29.Cha JM, Kim MH, Lee SK, Seo DW, Lee SS, Lee JH, Lee SG, Jang SJ. Clinicopathological review of 61 patients with early bile duct cancer. Clin Oncol (R Coll Radiol) 2006;18:669–677. doi: 10.1016/j.clon.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 30.Mizumoto R, Ogura Y, Kusuda T. Definition and diagnosis of early cancer of the biliary tract. Hepatogastroenterology. 1993;40:69–77. [PubMed] [Google Scholar]
- 31.Kurosaki I, Tsukada K, Watanabe H, Hatakeyama K. Prognostic determinants in extrahepatic bile duct cancer. Hepatogastroenterology. 1998;45:905–909. [PubMed] [Google Scholar]
- 32.He P, Shi JS, Chen WK, Wang ZR, Ren H, Li H. Multivariate statistical analysis of clinicopathologic factors influencing survival of patients with bile duct carcinoma. World J Gastroenterol. 2002;8:943–946. doi: 10.3748/wjg.v8.i5.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hong SM, Pawlik TM, Cho H, Aggarwal B, Goggins M, Hruban RH, Anders RA. Depth of tumor invasion better predicts prognosis than the current American Joint Committee on Cancer T classification for distal bile duct carcinoma. Surgery. 2009;146:250–257. doi: 10.1016/j.surg.2009.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jarnagin WR, Fong Y, DeMatteo RP, Gonen M, Burke EC, Bodniewicz BS J, Youssef BA M, Klimstra D, Blumgart LH. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507–517; discussion 517-519. doi: 10.1097/00000658-200110000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nagorney DM, Kendrick ML. Hepatic resection in the treatment of hilar cholangiocarcinoma. Adv Surg. 2006;40:159–171. doi: 10.1016/j.yasu.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 36.Seyama Y, Makuuchi M. Current surgical treatment for bile duct cancer. World J Gastroenterol. 2007;13:1505–1515. doi: 10.3748/wjg.v13.i10.1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mizumoto R, Suzuki H. Surgical anatomy of the hepatic hilum with special reference to the caudate lobe. World J Surg. 1988;12:2–10. doi: 10.1007/BF01658479. [DOI] [PubMed] [Google Scholar]
- 38.Shimizu H, Sawada S, Kimura F, Yoshidome H, Ohtsuka M, Kato A, Miyazaki M. Clinical significance of biliary vascular anatomy of the right liver for hilar cholangiocarcinoma applied to left hemihepatectomy. Ann Surg. 2009;249:435–439. doi: 10.1097/SLA.0b013e31819a6c10. [DOI] [PubMed] [Google Scholar]
- 39.Hirano S, Tanaka E, Shichinohe T, Suzuki O, Hazama K, Kitagami H, Okamura K, Yano T, Kondo S. Treatment strategy for hilar cholangiocarcinoma, with special reference to the limits of ductal resection in right-sided hepatectomies. J Hepatobiliary Pancreat Surg. 2007;14:429–433. doi: 10.1007/s00534-006-1190-5. [DOI] [PubMed] [Google Scholar]
- 40.Tsuzuki T, Ogata Y, Iida S, Nakanishi I, Takenaka Y, Yoshii H. Carcinoma of the bifurcation of the hepatic ducts. Arch Surg. 1983;118:1147–1151. doi: 10.1001/archsurg.1983.01390100021006. [DOI] [PubMed] [Google Scholar]
- 41.Dinant S, Gerhards MF, Busch OR, Obertop H, Gouma DJ, Van Gulik TM. The importance of complete excision of the caudate lobe in resection of hilar cholangiocarcinoma. HPB (Oxford) 2005;7:263–267. doi: 10.1080/13651820500372376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sugiura Y, Nakamura S, Iida S, Hosoda Y, Ikeuchi S, Mori S, Sugioka A, Tsuzuki T. Extensive resection of the bile ducts combined with liver resection for cancer of the main hepatic duct junction: a cooperative study of the Keio Bile Duct Cancer Study Group. Surgery. 1994;115:445–451. [PubMed] [Google Scholar]
- 43.Nimura Y, Hayakawa N, Kamiya J, Kondo S, Shionoya S. Hepatic segmentectomy with caudate lobe resection for bile duct carcinoma of the hepatic hilus. World J Surg. 1990;14:535–543; discussion 544. doi: 10.1007/BF01658686. [DOI] [PubMed] [Google Scholar]
- 44.Ogura Y, Mizumoto R, Tabata M, Matsuda S, Kusuda T. Surgical treatment of carcinoma of the hepatic duct confluence: analysis of 55 resected carcinomas. World J Surg. 1993;17:85–92; discussion 92-93. doi: 10.1007/BF01655714. [DOI] [PubMed] [Google Scholar]
- 45.Ito F, Agni R, Rettammel RJ, Been MJ, Cho CS, Mahvi DM, Rikkers LF, Weber SM. Resection of hilar cholangiocarcinoma: concomitant liver resection decreases hepatic recurrence. Ann Surg. 2008;248:273–279. doi: 10.1097/SLA.0b013e31817f2bfd. [DOI] [PubMed] [Google Scholar]
- 46.Uchida M, Ishibashi M, Tomita N, Shinagawa M, Hayabuchi N, Okuda K. Hilar and suprapancreatic cholangiocarcinoma: value of 3D angiography and multiphase fusion images using MDCT. AJR Am J Roentgenol. 2005;184:1572–1577. doi: 10.2214/ajr.184.5.01841572. [DOI] [PubMed] [Google Scholar]
- 47.Zech CJ, Schoenberg SO, Reiser M, Helmberger T. Cross-sectional imaging of biliary tumors: current clinical status and future developments. Eur Radiol. 2004;14:1174–1187. doi: 10.1007/s00330-004-2327-z. [DOI] [PubMed] [Google Scholar]
- 48.Choi SH, Han JK, Lee JM, Lee KH, Kim SH, Lee JY, Choi BI. Differentiating malignant from benign common bile duct stricture with multiphasic helical CT. Radiology. 2005;236:178–183. doi: 10.1148/radiol.2361040792. [DOI] [PubMed] [Google Scholar]
- 49.Seo H, Lee JM, Kim IH, Han JK, Kim SH, Jang JY, Kim SW, Choi BI. Evaluation of the gross type and longitudinal extent of extrahepatic cholangiocarcinomas on contrast-enhanced multidetector row computed tomography. J Comput Assist Tomogr. 2009;33:376–382. doi: 10.1097/RCT.0b013e318184f3f7. [DOI] [PubMed] [Google Scholar]
- 50.Han JK, Choi BI, Kim AY, An SK, Lee JW, Kim TK, Kim SW. Cholangiocarcinoma: pictorial essay of CT and cholangiographic findings. Radiographics. 2002;22:173–187. doi: 10.1148/radiographics.22.1.g02ja15173. [DOI] [PubMed] [Google Scholar]
- 51.Watadani T, Akahane M, Yoshikawa T, Ohtomo K. Preoperative assessment of hilar cholangiocarcinoma using multidetector-row CT: correlation with histopathological findings. Radiat Med. 2008;26:402–407. doi: 10.1007/s11604-008-0249-4. [DOI] [PubMed] [Google Scholar]
- 52.Unno M, Okumoto T, Katayose Y, Rikiyama T, Sato A, Motoi F, Oikawa M, Egawa S, Ishibashi T. Preoperative assessment of hilar cholangiocarcinoma by multidetector row computed tomography. J Hepatobiliary Pancreat Surg. 2007;14:434–440. doi: 10.1007/s00534-006-1191-4. [DOI] [PubMed] [Google Scholar]
- 53.Akamatsu N, Sugawara Y, Osada H, Okada T, Itoyama S, Komagome M, Shin N, Cho N, Ishida T, Ozawa F, et al. Diagnostic accuracy of multidetector-row computed tomography for hilar cholangiocarcinoma. J Gastroenterol Hepatol. 2010;25:731–737. doi: 10.1111/j.1440-1746.2009.06113.x. [DOI] [PubMed] [Google Scholar]
- 54.Park MS, Lee DK, Kim MJ, Lee WJ, Yoon DS, Lee SJ, Lim JS, Yu JS, Cho JY, Kim KW. Preoperative staging accuracy of multidetector row computed tomography for extrahepatic bile duct carcinoma. J Comput Assist Tomogr. 2006;30:362–367. doi: 10.1097/00004728-200605000-00003. [DOI] [PubMed] [Google Scholar]
- 55.Lee HY, Kim SH, Lee JM, Kim SW, Jang JY, Han JK, Choi BI. Preoperative assessment of resectability of hepatic hilar cholangiocarcinoma: combined CT and cholangiography with revised criteria. Radiology. 2006;239:113–121. doi: 10.1148/radiol.2383050419. [DOI] [PubMed] [Google Scholar]
- 56.Chen HW, Pan AZ, Zhen ZJ, Su SY, Wang JH, Yu SC, Lau WY. Preoperative evaluation of resectability of Klatskin tumor with 16-MDCT angiography and cholangiography. AJR Am J Roentgenol. 2006;186:1580–1586. doi: 10.2214/AJR.05.0008. [DOI] [PubMed] [Google Scholar]
- 57.Kim HJ, Kim AY, Hong SS, Kim MH, Byun JH, Won HJ, Shin YM, Kim PN, Ha HK, Lee MG. Biliary ductal evaluation of hilar cholangiocarcinoma: three-dimensional direct multi-detector row CT cholangiographic findings versus surgical and pathologic results--feasibility study. Radiology. 2006;238:300–308. doi: 10.1148/radiol.2381041902. [DOI] [PubMed] [Google Scholar]
- 58.Aloia TA, Charnsangavej C, Faria S, Ribero D, Abdalla EK, Vauthey JN, Curley SA. High-resolution computed tomography accurately predicts resectability in hilar cholangiocarcinoma. Am J Surg. 2007;193:702–706. doi: 10.1016/j.amjsurg.2006.10.024. [DOI] [PubMed] [Google Scholar]
- 59.Endo I, Shimada H, Sugita M, Fujii Y, Morioka D, Takeda K, Sugae S, Tanaka K, Togo S, Bourquain H, et al. Role of three-dimensional imaging in operative planning for hilar cholangiocarcinoma. Surgery. 2007;142:666–675. doi: 10.1016/j.surg.2007.05.018. [DOI] [PubMed] [Google Scholar]
- 60.Cho ES, Park MS, Yu JS, Kim MJ, Kim KW. Biliary ductal involvement of hilar cholangiocarcinoma: multidetector computed tomography versus magnetic resonance cholangiography. J Comput Assist Tomogr. 2007;31:72–78. doi: 10.1097/01.rct.0000230013.24091.8e. [DOI] [PubMed] [Google Scholar]
- 61.Park HS, Lee JM, Choi JY, Lee MW, Kim HJ, Han JK, Choi BI. Preoperative evaluation of bile duct cancer: MRI combined with MR cholangiopancreatography versus MDCT with direct cholangiography. AJR Am J Roentgenol. 2008;190:396–405. doi: 10.2214/AJR.07.2310. [DOI] [PubMed] [Google Scholar]
- 62.Akamatsu N, Sugawara Y, Osada H, Okada T, Itoyama S, Komagome M, Shin N, Ishida T, Ozawa F, Hashimoto D. Preoperative evaluation of the longitudinal spread of extrahepatic bile duct cancer using multidetector computed tomography. J Hepatobiliary Pancreat Surg. 2009;16:216–222. doi: 10.1007/s00534-009-0045-2. [DOI] [PubMed] [Google Scholar]
- 63.Senda Y, Nishio H, Oda K, Yokoyama Y, Ebata T, Igami T, Sugiura T, Shimoyama Y, Nimura Y, Nagino M. Value of multidetector row CT in the assessment of longitudinal extension of cholangiocarcinoma: correlation between MDCT and microscopic findings. World J Surg. 2009;33:1459–1467. doi: 10.1007/s00268-009-0025-3. [DOI] [PubMed] [Google Scholar]
- 64.Hünerbein M, Stroszczynski C, Ulmer C, Handke T, Felix R, Schlag PM. Prospective comparison of transcutaneous 3-dimensional US cholangiography, magnetic resonance cholangiography, and direct cholangiography in the evaluation of malignant biliary obstruction. Gastrointest Endosc. 2003;58:853–858. doi: 10.1016/s0016-5107(03)02302-2. [DOI] [PubMed] [Google Scholar]
- 65.Lopera JE, Soto JA, Múnera F. Malignant hilar and perihilar biliary obstruction: use of MR cholangiography to define the extent of biliary ductal involvement and plan percutaneous interventions. Radiology. 2001;220:90–96. doi: 10.1148/radiology.220.1.r01jl3990. [DOI] [PubMed] [Google Scholar]
- 66.Varghese JC, Farrell MA, Courtney G, Osborne H, Murray FE, Lee MJ. A prospective comparison of magnetic resonance cholangiopancreatography with endoscopic retrograde cholangiopancreatography in the evaluation of patients with suspected biliary tract disease. Clin Radiol. 1999;54:513–520. doi: 10.1016/s0009-9260(99)90848-6. [DOI] [PubMed] [Google Scholar]
- 67.Rösch T, Meining A, Frühmorgen S, Zillinger C, Schusdziarra V, Hellerhoff K, Classen M, Helmberger H. A prospective comparison of the diagnostic accuracy of ERCP, MRCP, CT, and EUS in biliary strictures. Gastrointest Endosc. 2002;55:870–876. doi: 10.1067/mge.2002.124206. [DOI] [PubMed] [Google Scholar]
- 68.Park MS, Kim TK, Kim KW, Park SW, Lee JK, Kim JS, Lee JH, Kim KA, Kim AY, Kim PN, et al. Differentiation of extrahepatic bile duct cholangiocarcinoma from benign stricture: findings at MRCP versus ERCP. Radiology. 2004;233:234–240. doi: 10.1148/radiol.2331031446. [DOI] [PubMed] [Google Scholar]
- 69.Hänninen EL, Pech M, Jonas S, Ricke J, Thelen A, Langrehr J, Hintze R, Röttgen R, Denecke T, Winter L, et al. Magnetic resonance imaging including magnetic resonance cholangiopancreatography for tumor localization and therapy planning in malignant hilar obstructions. Acta Radiol. 2005;46:462–470. doi: 10.1080/02841850510021625. [DOI] [PubMed] [Google Scholar]
- 70.Fulcher AS, Turner MA. HASTE MR cholangiography in the evaluation of hilar cholangiocarcinoma. AJR Am J Roentgenol. 1997;169:1501–1505. doi: 10.2214/ajr.169.6.9393153. [DOI] [PubMed] [Google Scholar]
- 71.Nimura Y, Kamiya J, Kondo S, Nagino M, Uesaka K, Oda K, Sano T, Yamamoto H, Hayakawa N. Aggressive preoperative management and extended surgery for hilar cholangiocarcinoma: Nagoya experience. J Hepatobiliary Pancreat Surg. 2000;7:155–162. doi: 10.1007/s005340050170. [DOI] [PubMed] [Google Scholar]
- 72.Kondo S, Hirano S, Ambo Y, Tanaka E, Okushiba S, Morikawa T, Katoh H. Forty consecutive resections of hilar cholangiocarcinoma with no postoperative mortality and no positive ductal margins: results of a prospective study. Ann Surg. 2004;240:95–101. doi: 10.1097/01.sla.0000129491.43855.6b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lee SG, Lee YJ, Park KM, Hwang S, Min PC. One hundred and eleven liver resections for hilar bile duct cancer. J Hepatobiliary Pancreat Surg. 2000;7:135–141. doi: 10.1007/s005340050167. [DOI] [PubMed] [Google Scholar]
- 74.Sano T, Shimada K, Sakamoto Y, Yamamoto J, Yamasaki S, Kosuge T. One hundred two consecutive hepatobiliary resections for perihilar cholangiocarcinoma with zero mortality. Ann Surg. 2006;244:240–247. doi: 10.1097/01.sla.0000217605.66519.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kumar M, Prashad R, Kumar A, Sharma R, Acharya SK, Chattopadhyay TK. Relative merits of ultrasonography, computed tomography and cholangiography in patients of surgical obstructive jaundice. Hepatogastroenterology. 1998;45:2027–2032. [PubMed] [Google Scholar]
- 76.Hochwald SN, Burke EC, Jarnagin WR, Fong Y, Blumgart LH. Association of preoperative biliary stenting with increased postoperative infectious complications in proximal cholangiocarcinoma. Arch Surg. 1999;134:261–266. doi: 10.1001/archsurg.134.3.261. [DOI] [PubMed] [Google Scholar]
- 77.Sewnath ME, Karsten TM, Prins MH, Rauws EJ, Obertop H, Gouma DJ. A meta-analysis on the efficacy of preoperative biliary drainage for tumors causing obstructive jaundice. Ann Surg. 2002;236:17–27. doi: 10.1097/00000658-200207000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Domagk D, Wessling J, Reimer P, Hertel L, Poremba C, Senninger N, Heinecke A, Domschke W, Menzel J. Endoscopic retrograde cholangiopancreatography, intraductal ultrasonography, and magnetic resonance cholangiopancreatography in bile duct strictures: a prospective comparison of imaging diagnostics with histopathological correlation. Am J Gastroenterol. 2004;99:1684–1689. doi: 10.1111/j.1572-0241.2004.30347.x. [DOI] [PubMed] [Google Scholar]
- 79.Harewood GC, Baron TH, Stadheim LM, Kipp BR, Sebo TJ, Salomao DR. Prospective, blinded assessment of factors influencing the accuracy of biliary cytology interpretation. Am J Gastroenterol. 2004;99:1464–1469. doi: 10.1111/j.1572-0241.2004.30845.x. [DOI] [PubMed] [Google Scholar]
- 80.Domagk D, Poremba C, Dietl KH, Senninger N, Heinecke A, Domschke W, Menzel J. Endoscopic transpapillary biopsies and intraductal ultrasonography in the diagnostics of bile duct strictures: a prospective study. Gut. 2002;51:240–244. doi: 10.1136/gut.51.2.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gerhards MF, Vos P, van Gulik TM, Rauws EA, Bosma A, Gouma DJ. Incidence of benign lesions in patients resected for suspicious hilar obstruction. Br J Surg. 2001;88:48–51. doi: 10.1046/j.1365-2168.2001.01607.x. [DOI] [PubMed] [Google Scholar]
- 82.Nakayama A, Imamura H, Shimada R, Miyagawa S, Makuuchi M, Kawasaki S. Proximal bile duct stricture disguised as malignant neoplasm. Surgery. 1999;125:514–521. [PubMed] [Google Scholar]
- 83.Fukuda Y, Tsuyuguchi T, Sakai Y, Tsuchiya S, Saisyo H. Diagnostic utility of peroral cholangioscopy for various bile-duct lesions. Gastrointest Endosc. 2005;62:374–382. doi: 10.1016/j.gie.2005.04.032. [DOI] [PubMed] [Google Scholar]
- 84.Tsuyuguchi T, Fukuda Y, Saisho H. Peroral cholangioscopy for the diagnosis and treatment of biliary diseases. J Hepatobiliary Pancreat Surg. 2006;13:94–99. doi: 10.1007/s00534-005-1064-2. [DOI] [PubMed] [Google Scholar]
- 85.Fishman DS, Tarnasky PR, Patel SN, Raijman I. Management of pancreaticobiliary disease using a new intra-ductal endoscope: the Texas experience. World J Gastroenterol. 2009;15:1353–1358. doi: 10.3748/wjg.15.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pitt HA, Gomes AS, Lois JF, Mann LL, Deutsch LS, Longmire WP Jr. Does preoperative percutaneous biliary drainage reduce operative risk or increase hospital cost? Ann Surg. 1985;201:545–553. doi: 10.1097/00000658-198505000-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lai EC, Mok FP, Fan ST, Lo CM, Chu KM, Liu CL, Wong J. Preoperative endoscopic drainage for malignant obstructive jaundice. Br J Surg. 1994;81:1195–1198. doi: 10.1002/bjs.1800810839. [DOI] [PubMed] [Google Scholar]
- 88.Povoski SP, Karpeh MS Jr, Conlon KC, Blumgart LH, Brennan MF. Association of preoperative biliary drainage with postoperative outcome following pancreaticoduodenectomy. Ann Surg. 1999;230:131–142. doi: 10.1097/00000658-199908000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cortes A, Sauvanet A, Bert F, Janny S, Sockeel P, Kianmanesh R, Ponsot P, Ruszniewski P, Belghiti J. Effect of bile contamination on immediate outcomes after pancreaticoduodenectomy for tumor. J Am Coll Surg. 2006;202:93–99. doi: 10.1016/j.jamcollsurg.2005.09.006. [DOI] [PubMed] [Google Scholar]
- 90.Cherqui D, Benoist S, Malassagne B, Humeres R, Rodriguez V, Fagniez PL. Major liver resection for carcinoma in jaundiced patients without preoperative biliary drainage. Arch Surg. 2000;135:302–308. doi: 10.1001/archsurg.135.3.302. [DOI] [PubMed] [Google Scholar]
- 91.Sakata J, Shirai Y, Wakai T, Nomura T, Sakata E, Hatakeyama K. Catheter tract implantation metastases associated with percutaneous biliary drainage for extrahepatic cholangiocarcinoma. World J Gastroenterol. 2005;11:7024–7027. doi: 10.3748/wjg.v11.i44.7024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.van der Gaag NA, Kloek JJ, de Castro SM, Busch OR, van Gulik TM, Gouma DJ. Preoperative biliary drainage in patients with obstructive jaundice: history and current status. J Gastrointest Surg. 2009;13:814–820. doi: 10.1007/s11605-008-0618-4. [DOI] [PubMed] [Google Scholar]
- 93.Aly EA, Johnson CD. Preoperative biliary drainage before resection in obstructive jaundice. Dig Surg. 2001;18:84–89. doi: 10.1159/000050105. [DOI] [PubMed] [Google Scholar]
- 94.Nakeeb A, Pitt HA. The role of preoperative biliary decompression in obstructive jaundice. Hepatogastroenterology. 1995;42:332–337. [PubMed] [Google Scholar]
- 95.Kennedy TJ, Yopp A, Qin Y, Zhao B, Guo P, Liu F, Schwartz LH, Allen P, D'Angelica M, Fong Y, et al. Role of preoperative biliary drainage of liver remnant prior to extended liver resection for hilar cholangiocarcinoma. HPB (Oxford) 2009;11:445–451. doi: 10.1111/j.1477-2574.2009.00090.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Su CH, Tsay SH, Wu CC, Shyr YM, King KL, Lee CH, Lui WY, Liu TJ, P'eng FK. Factors influencing postoperative morbidity, mortality, and survival after resection for hilar cholangiocarcinoma. Ann Surg. 1996;223:384–394. doi: 10.1097/00000658-199604000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Maguchi H, Takahashi K, Katanuma A, Osanai M, Nakahara K, Matuzaki S, Urata T, Iwano H. Preoperative biliary drainage for hilar cholangiocarcinoma. J Hepatobiliary Pancreat Surg. 2007;14:441–446. doi: 10.1007/s00534-006-1192-3. [DOI] [PubMed] [Google Scholar]
- 98.Ito F, Cho CS, Rikkers LF, Weber SM. Hilar cholangiocarcinoma: current management. Ann Surg. 2009;250:210–218. doi: 10.1097/SLA.0b013e3181afe0ab. [DOI] [PubMed] [Google Scholar]
- 99.Kubota K, Makuuchi M, Kusaka K, Kobayashi T, Miki K, Hasegawa K, Harihara Y, Takayama T. Measurement of liver volume and hepatic functional reserve as a guide to decision-making in resectional surgery for hepatic tumors. Hepatology. 1997;26:1176–1181. doi: 10.1053/jhep.1997.v26.pm0009362359. [DOI] [PubMed] [Google Scholar]
- 100.Shirabe K, Shimada M, Gion T, Hasegawa H, Takenaka K, Utsunomiya T, Sugimachi K. Postoperative liver failure after major hepatic resection for hepatocellular carcinoma in the modern era with special reference to remnant liver volume. J Am Coll Surg. 1999;188:304–309. doi: 10.1016/s1072-7515(98)00301-9. [DOI] [PubMed] [Google Scholar]
- 101.Gerhards MF, van Gulik TM, de Wit LT, Obertop H, Gouma DJ. Evaluation of morbidity and mortality after resection for hilar cholangiocarcinoma--a single center experience. Surgery. 2000;127:395–404. doi: 10.1067/msy.2000.104250. [DOI] [PubMed] [Google Scholar]
- 102.Makuuchi M, Thai BL, Takayasu K, Takayama T, Kosuge T, Gunvén P, Yamazaki S, Hasegawa H, Ozaki H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: a preliminary report. Surgery. 1990;107:521–527. [PubMed] [Google Scholar]
- 103.Ribero D, Abdalla EK, Madoff DC, Donadon M, Loyer EM, Vauthey JN. Portal vein embolization before major hepatectomy and its effects on regeneration, resectability and outcome. Br J Surg. 2007;94:1386–1394. doi: 10.1002/bjs.5836. [DOI] [PubMed] [Google Scholar]
- 104.Hemming AW, Reed AI, Howard RJ, Fujita S, Hochwald SN, Caridi JG, Hawkins IF, Vauthey JN. Preoperative portal vein embolization for extended hepatectomy. Ann Surg. 2003;237:686–691; discussion 691-693. doi: 10.1097/01.SLA.0000065265.16728.C0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Shoup M, Gonen M, D'Angelica M, Jarnagin WR, DeMatteo RP, Schwartz LH, Tuorto S, Blumgart LH, Fong Y. Volumetric analysis predicts hepatic dysfunction in patients undergoing major liver resection. J Gastrointest Surg. 2003;7:325–330. doi: 10.1016/s1091-255x(02)00370-0. [DOI] [PubMed] [Google Scholar]
- 106.Nagino M, Kamiya J, Nishio H, Ebata T, Arai T, Nimura Y. Two hundred forty consecutive portal vein embolizations before extended hepatectomy for biliary cancer: surgical outcome and long-term follow-up. Ann Surg. 2006;243:364–372. doi: 10.1097/01.sla.0000201482.11876.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Imamura H, Shimada R, Kubota M, Matsuyama Y, Nakayama A, Miyagawa S, Makuuchi M, Kawasaki S. Preoperative portal vein embolization: an audit of 84 patients. Hepatology. 1999;29:1099–1105. doi: 10.1002/hep.510290415. [DOI] [PubMed] [Google Scholar]
- 108.Farges O, Belghiti J, Kianmanesh R, Regimbeau JM, Santoro R, Vilgrain V, Denys A, Sauvanet A. Portal vein embolization before right hepatectomy: prospective clinical trial. Ann Surg. 2003;237:208–217. doi: 10.1097/01.SLA.0000048447.16651.7B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yokoyama Y, Nagino M, Nishio H, Ebata T, Igami T, Nimura Y. Recent advances in the treatment of hilar cholangiocarcinoma: portal vein embolization. J Hepatobiliary Pancreat Surg. 2007;14:447–454. doi: 10.1007/s00534-006-1193-2. [DOI] [PubMed] [Google Scholar]
- 110.Fong Y, Blumgart LH, Lin E, Fortner JG, Brennan MF. Outcome of treatment for distal bile duct cancer. Br J Surg. 1996;83:1712–1715. doi: 10.1002/bjs.1800831217. [DOI] [PubMed] [Google Scholar]
- 111.Sasaki R, Takahashi M, Funato O, Nitta H, Murakami M, Kawamura H, Suto T, Kanno S, Saito K. Prognostic significance of lymph node involvement in middle and distal bile duct cancer. Surgery. 2001;129:677–683. doi: 10.1067/msy.2001.114555. [DOI] [PubMed] [Google Scholar]
- 112.Tran KT, Smeenk HG, van Eijck CH, Kazemier G, Hop WC, Greve JW, Terpstra OT, Zijlstra JA, Klinkert P, Jeekel H. Pylorus preserving pancreaticoduodenectomy versus standard Whipple procedure: a prospective, randomized, multicenter analysis of 170 patients with pancreatic and periampullary tumors. Ann Surg. 2004;240:738–745. doi: 10.1097/01.sla.0000143248.71964.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Seiler CA, Wagner M, Bachmann T, Redaelli CA, Schmied B, Uhl W, Friess H, Büchler MW. Randomized clinical trial of pylorus-preserving duodenopancreatectomy versus classical Whipple resection-long term results. Br J Surg. 2005;92:547–556. doi: 10.1002/bjs.4881. [DOI] [PubMed] [Google Scholar]
- 114.Diener MK, Knaebel HP, Heukaufer C, Antes G, Büchler MW, Seiler CM. A systematic review and meta-analysis of pylorus-preserving versus classical pancreaticoduodenectomy for surgical treatment of periampullary and pancreatic carcinoma. Ann Surg. 2007;245:187–200. doi: 10.1097/01.sla.0000242711.74502.a9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Wade TP, Prasad CN, Virgo KS, Johnson FE. Experience with distal bile duct cancers in U.S. Veterans Affairs hospitals: 1987-1991. J Surg Oncol. 1997;64:242–245. doi: 10.1002/(sici)1096-9098(199703)64:3<242::aid-jso12>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 116.Lee HG, Lee SH, Yoo DD, Paik KY, Heo JS, Choi SH, Choi DW. Carcinoma of the middle bile duct: is bile duct segmental resection appropriate? World J Gastroenterol. 2009;15:5966–5971. doi: 10.3748/wjg.15.5966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sakamoto Y, Shimada K, Nara S, Esaki M, Ojima H, Sano T, Yamamoto J, Kosuge T. Surgical management of infrahilar/suprapancreatic cholangiocarcinoma: an analysis of the surgical procedures, surgical margins, and survivals of 77 patients. J Gastrointest Surg. 2010;14:335–343. doi: 10.1007/s11605-009-1072-7. [DOI] [PubMed] [Google Scholar]
- 118.Ikeyama T, Nagino M, Oda K, Ebata T, Nishio H, Nimura Y. Surgical approach to bismuth Type I and II hilar cholangiocarcinomas: audit of 54 consecutive cases. Ann Surg. 2007;246:1052–1057. doi: 10.1097/SLA.0b013e318142d97e. [DOI] [PubMed] [Google Scholar]
- 119.Kayahara M, Nagakawa T, Ohta T, Kitagawa H, Tajima H, Miwa K. Role of nodal involvement and the periductal soft-tissue margin in middle and distal bile duct cancer. Ann Surg. 1999;229:76–83. doi: 10.1097/00000658-199901000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Suzuki M, Unno M, Oikawa M, Endo K, Katayose Y, Matsuno S. Surgical treatment and postoperative outcomes for middle and lower bile duct carcinoma in Japan--experience of a single institute. Hepatogastroenterology. 2000;47:650–657. [PubMed] [Google Scholar]
- 121.Yoshida T, Matsumoto T, Sasaki A, Morii Y, Aramaki M, Kitano S. Prognostic factors after pancreatoduodenectomy with extended lymphadenectomy for distal bile duct cancer. Arch Surg. 2002;137:69–73. doi: 10.1001/archsurg.137.1.69. [DOI] [PubMed] [Google Scholar]
- 122.Sakamoto Y, Kosuge T, Shimada K, Sano T, Ojima H, Yamamoto J, Yamasaki S, Takayama T, Makuuchi M. Prognostic factors of surgical resection in middle and distal bile duct cancer: an analysis of 55 patients concerning the significance of ductal and radial margins. Surgery. 2005;137:396–402. doi: 10.1016/j.surg.2004.10.008. [DOI] [PubMed] [Google Scholar]
- 123.Jang JY, Kim SW, Park DJ, Ahn YJ, Yoon YS, Choi MG, Suh KS, Lee KU, Park YH. Actual long-term outcome of extrahepatic bile duct cancer after surgical resection. Ann Surg. 2005;241:77–84. doi: 10.1097/01.sla.0000150166.94732.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Cheng Q, Luo X, Zhang B, Jiang X, Yi B, Wu M. Distal bile duct carcinoma: prognostic factors after curative surgery. A series of 112 cases. Ann Surg Oncol. 2007;14:1212–1219. doi: 10.1245/s10434-006-9260-0. [DOI] [PubMed] [Google Scholar]
- 125.Murakami Y, Uemura K, Hayashidani Y, Sudo T, Ohge H, Sueda T. Pancreatoduodenectomy for distal cholangiocarcinoma: prognostic impact of lymph node metastasis. World J Surg. 2007;31:337–342; discussion 343-344. doi: 10.1007/s00268-006-0224-0. [DOI] [PubMed] [Google Scholar]
- 126.Sasaki R, Takeda Y, Funato O, Nitta H, Kawamura H, Uesugi N, Sugai T, Wakabayashi G, Ohkohchi N. Significance of ductal margin status in patients undergoing surgical resection for extrahepatic cholangiocarcinoma. World J Surg. 2007;31:1788–1796. doi: 10.1007/s00268-007-9102-7. [DOI] [PubMed] [Google Scholar]
- 127.DeOliveira ML, Cunningham SC, Cameron JL, Kamangar F, Winter JM, Lillemoe KD, Choti MA, Yeo CJ, Schulick RD. Cholangiocarcinoma: thirty-one-year experience with 564 patients at a single institution. Ann Surg. 2007;245:755–762. doi: 10.1097/01.sla.0000251366.62632.d3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Allen PJ, Reiner AS, Gonen M, Klimstra DK, Blumgart LH, Brennan MF, D'Angelica M, Dematteo R, Fong Y, Jarnagin WR. Extrahepatic cholangiocarcinoma: a comparison of patients with resected proximal and distal lesions. HPB (Oxford) 2008;10:341–346. doi: 10.1080/13651820802276630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Bahra M, Jacob D, Langrehr JM, Neumann UP, Neuhaus P. Carcinoma of the distal and middle bile duct: surgical results, prognostic factors, and long-term follow-up. J Hepatobiliary Pancreat Surg. 2008;15:501–507. doi: 10.1007/s00534-007-1308-4. [DOI] [PubMed] [Google Scholar]
- 130.Nomura T, Tsuchiya Y, Nashimoto A, Yabusaki H, Takii Y, Nakagawa S, Sato N, Kanbayashi C, Tanaka O. Prognostic factors for radical resection of middle and distal bile duct cancer. Hepatogastroenterology. 2009;56:294–298. [PubMed] [Google Scholar]
- 131.Kawai M, Tani M, Kobayashi Y, Tsuji T, Tabuse K, Horiuchi T, Oka M, Yamaguchi K, Sakata Y, Shimomura T, et al. The ratio between metastatic and examined lymph nodes is an independent prognostic factor for patients with resectable middle and distal bile duct carcinoma. Am J Surg. 2010;199:447–452. doi: 10.1016/j.amjsurg.2009.01.019. [DOI] [PubMed] [Google Scholar]
- 132.Miyakawa S, Ishihara S, Horiguchi A, Takada T, Miyazaki M, Nagakawa T. Biliary tract cancer treatment: 5,584 results from the Biliary Tract Cancer Statistics Registry from 1998 to 2004 in Japan. J Hepatobiliary Pancreat Surg. 2009;16:1–7. doi: 10.1007/s00534-008-0015-0. [DOI] [PubMed] [Google Scholar]
- 133.Ishihara S, Miyakawa S, Takada T, Takasaki K, Nimura Y, Tanaka M, Miyazaki M, Nagakawa T, Kayahara M, Horiguchi A. Status of surgical treatment of biliary tract cancer. Dig Surg. 2007;24:131–136. doi: 10.1159/000101901. [DOI] [PubMed] [Google Scholar]
- 134.Shimizu H, Kimura F, Yoshidome H, Ohtsuka M, Kato A, Yoshitomi H, Furukawa K, Miyazaki M. Aggressive surgical resection for hilar cholangiocarcinoma of the left-side predominance: radicality and safety of left-sided hepatectomy. Ann Surg. 2010;251:281–286. doi: 10.1097/SLA.0b013e3181be0085. [DOI] [PubMed] [Google Scholar]
- 135.Kawasaki S, Imamura H, Kobayashi A, Noike T, Miwa S, Miyagawa S. Results of surgical resection for patients with hilar bile duct cancer: application of extended hepatectomy after biliary drainage and hemihepatic portal vein embolization. Ann Surg. 2003;238:84–92. doi: 10.1097/01.SLA.0000074984.83031.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Ebata T, Nagino M, Nishio H, Arai T, Nimura Y. Right hepatopancreatoduodenectomy: improvements over 23 years to attain acceptability. J Hepatobiliary Pancreat Surg. 2007;14:131–135. doi: 10.1007/s00534-006-1106-4. [DOI] [PubMed] [Google Scholar]
- 137.Hirono S, Tani M, Kawai M, Ina S, Uchiyama K, Yamaue H. Indication of hepatopancreatoduodenectomy for biliary tract cancer. World J Surg. 2006;30:567–573; discussion 574-575. doi: 10.1007/s00268-005-0380-7. [DOI] [PubMed] [Google Scholar]
- 138.Miyagawa S, Makuuchi M, Kawasaki S, Hayashi K, Harada H, Kitamura H, Seki H. Outcome of major hepatectomy with pancreatoduodenectomy for advanced biliary malignancies. World J Surg. 1996;20:77–80. doi: 10.1007/s002689900014. [DOI] [PubMed] [Google Scholar]
- 139.Nagino M, Kamiya J, Arai T, Nishio H, Ebata T, Nimura Y. "Anatomic" right hepatic trisectionectomy (extended right hepatectomy) with caudate lobectomy for hilar cholangiocarcinoma. Ann Surg. 2006;243:28–32. doi: 10.1097/01.sla.0000193604.72436.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Shimada K, Sano T, Sakamoto Y, Kosuge T. Safety and effectiveness of left hepatic trisegmentectomy for hilar cholangiocarcinoma. World J Surg. 2005;29:723–727. doi: 10.1007/s00268-005-7704-5. [DOI] [PubMed] [Google Scholar]
- 141.Klempnauer J, Ridder GJ, von Wasielewski R, Werner M, Weimann A, Pichlmayr R. Resectional surgery of hilar cholangiocarcinoma: a multivariate analysis of prognostic factors. J Clin Oncol. 1997;15:947–954. doi: 10.1200/JCO.1997.15.3.947. [DOI] [PubMed] [Google Scholar]
- 142.Todoroki T, Kawamoto T, Koike N, Takahashi H, Yoshida S, Kashiwagi H, Takada Y, Otsuka M, Fukao K. Radical resection of hilar bile duct carcinoma and predictors of survival. Br J Surg. 2000;87:306–313. doi: 10.1046/j.1365-2168.2000.01343.x. [DOI] [PubMed] [Google Scholar]
- 143.Kitagawa Y, Nagino M, Kamiya J, Uesaka K, Sano T, Yamamoto H, Hayakawa N, Nimura Y. Lymph node metastasis from hilar cholangiocarcinoma: audit of 110 patients who underwent regional and paraaortic node dissection. Ann Surg. 2001;233:385–392. doi: 10.1097/00000658-200103000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kosuge T, Yamamoto J, Shimada K, Yamasaki S, Makuuchi M. Improved surgical results for hilar cholangiocarcinoma with procedures including major hepatic resection. Ann Surg. 1999;230:663–671. doi: 10.1097/00000658-199911000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Miyazaki M, Kato A, Ito H, Kimura F, Shimizu H, Ohtsuka M, Yoshidome H, Yoshitomi H, Furukawa K, Nozawa S. Combined vascular resection in operative resection for hilar cholangiocarcinoma: does it work or not? Surgery. 2007;141:581–588. doi: 10.1016/j.surg.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 146.Capussotti L, Muratore A, Polastri R, Ferrero A, Massucco P. Liver resection for hilar cholangiocarcinoma: in-hospital mortality and longterm survival. J Am Coll Surg. 2002;195:641–647. doi: 10.1016/s1072-7515(02)01481-3. [DOI] [PubMed] [Google Scholar]
- 147.Seyama Y, Kubota K, Sano K, Noie T, Takayama T, Kosuge T, Makuuchi M. Long-term outcome of extended hemihepatectomy for hilar bile duct cancer with no mortality and high survival rate. Ann Surg. 2003;238:73–83. doi: 10.1097/01.SLA.0000074960.55004.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Rea DJ, Munoz-Juarez M, Farnell MB, Donohue JH, Que FG, Crownhart B, Larson D, Nagorney DM. Major hepatic resection for hilar cholangiocarcinoma: analysis of 46 patients. Arch Surg. 2004;139:514–523; discussion 523-525. doi: 10.1001/archsurg.139.5.514. [DOI] [PubMed] [Google Scholar]
- 149.Hemming AW, Reed AI, Fujita S, Foley DP, Howard RJ. Surgical management of hilar cholangiocarcinoma. Ann Surg. 2005;241:693–699; discussion 699-702. doi: 10.1097/01.sla.0000160701.38945.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Dinant S, Gerhards MF, Rauws EA, Busch OR, Gouma DJ, van Gulik TM. Improved outcome of resection of hilar cholangiocarcinoma (Klatskin tumor) Ann Surg Oncol. 2006;13:872–880. doi: 10.1245/ASO.2006.05.053. [DOI] [PubMed] [Google Scholar]
- 151.Sano T, Shimada K, Sakamoto Y, Esaki M, Kosuge T. Changing trends in surgical outcomes after major hepatobiliary resection for hilar cholangiocarcinoma: a single-center experience over 25 years. J Hepatobiliary Pancreat Surg. 2007;14:455–462. doi: 10.1007/s00534-006-1194-1. [DOI] [PubMed] [Google Scholar]
- 152.Baton O, Azoulay D, Adam DV, Castaing D. Major hepatectomy for hilar cholangiocarcinoma type 3 and 4: prognostic factors and longterm outcomes. J Am Coll Surg. 2007;204:250–260. doi: 10.1016/j.jamcollsurg.2006.10.028. [DOI] [PubMed] [Google Scholar]
- 153.Hasegawa S, Ikai I, Fujii H, Hatano E, Shimahara Y. Surgical resection of hilar cholangiocarcinoma: analysis of survival and postoperative complications. World J Surg. 2007;31:1256–1263. doi: 10.1007/s00268-007-9001-y. [DOI] [PubMed] [Google Scholar]
- 154.Hirano S, Kondo S, Tanaka E, Shichinohe T, Tsuchikawa T, Kato K, Matsumoto J, Kawasaki R. Outcome of surgical treatment of hilar cholangiocarcinoma: a special reference to postoperative morbidity and mortality. J Hepatobiliary Pancreat Sci. 2010;17:455–462. doi: 10.1007/s00534-009-0208-1. [DOI] [PubMed] [Google Scholar]
- 155.Lee SG, Song GW, Hwang S, Ha TY, Moon DB, Jung DH, Kim KH, Ahn CS, Kim MH, Lee SK, et al. Surgical treatment of hilar cholangiocarcinoma in the new era: the Asan experience. J Hepatobiliary Pancreat Sci. 2010;17:476–489. doi: 10.1007/s00534-009-0204-5. [DOI] [PubMed] [Google Scholar]
- 156.Young AL, Prasad KR, Toogood GJ, Lodge JP. Surgical treatment of hilar cholangiocarcinoma in a new era: comparison among leading Eastern and Western centers, Leeds. J Hepatobiliary Pancreat Sci. 2010;17:497–504. doi: 10.1007/s00534-009-0203-6. [DOI] [PubMed] [Google Scholar]
- 157.Igami T, Nishio H, Ebata T, Yokoyama Y, Sugawara G, Nimura Y, Nagino M. Surgical treatment of hilar cholangiocarcinoma in the "new era": the Nagoya University experience. J Hepatobiliary Pancreat Sci. 2010;17:449–454. doi: 10.1007/s00534-009-0209-0. [DOI] [PubMed] [Google Scholar]
- 158.Miyazaki M, Kimura F, Shimizu H, Yoshidome H, Otuka M, Kato A, Yoshitomi H, Furukawa K, Takeuchi D, Takayashiki T, et al. One hundred seven consecutive surgical resections for hilar cholangiocarcinoma of Bismuth types II, III, IV between 2001 and 2008. J Hepatobiliary Pancreat Sci. 2010;17:470–475. doi: 10.1007/s00534-009-0207-2. [DOI] [PubMed] [Google Scholar]
- 159.Unno M, Katayose Y, Rikiyama T, Yoshida H, Yamamoto K, Morikawa T, Hayashi H, Motoi F, Egawa S. Major hepatectomy for perihilar cholangiocarcinoma. J Hepatobiliary Pancreat Sci. 2010;17:463–469. doi: 10.1007/s00534-009-0206-3. [DOI] [PubMed] [Google Scholar]
- 160.Murakami Y, Uemura K, Sudo T, Hayashidani Y, Hashimoto Y, Nakamura H, Nakashima A, Sueda T. Gemcitabine-based adjuvant chemotherapy improves survival after aggressive surgery for hilar cholangiocarcinoma. J Gastrointest Surg. 2009;13:1470–1479. doi: 10.1007/s11605-009-0900-0. [DOI] [PubMed] [Google Scholar]
- 161.Goss JA, Shackleton CR, Farmer DG, Arnaout WS, Seu P, Markowitz JS, Martin P, Stribling RJ, Goldstein LI, Busuttil RW. Orthotopic liver transplantation for primary sclerosing cholangitis. A 12-year single center experience. Ann Surg. 1997;225:472–481; discussion 481-483. doi: 10.1097/00000658-199705000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Meyer CG, Penn I, James L. Liver transplantation for cholangiocarcinoma: results in 207 patients. Transplantation. 2000;69:1633–1637. doi: 10.1097/00007890-200004270-00019. [DOI] [PubMed] [Google Scholar]
- 163.Jeyarajah DR, Klintmalm GB. Is liver transplantation indicated for cholangiocarcinoma? J Hepatobiliary Pancreat Surg. 1998;5:48–51. doi: 10.1007/pl00009950. [DOI] [PubMed] [Google Scholar]
- 164.Schwartz JJ, Hutson WR, Gayowski TJ, Sorensen JB. Liver transplantation for cholangiocarcinoma. Transplantation. 2009;88:295–298. doi: 10.1097/TP.0b013e3181adc9e5. [DOI] [PubMed] [Google Scholar]
- 165.Sudan D, DeRoover A, Chinnakotla S, Fox I, Shaw B Jr, McCashland T, Sorrell M, Tempero M, Langnas A. Radiochemotherapy and transplantation allow long-term survival for nonresectable hilar cholangiocarcinoma. Am J Transplant. 2002;2:774–779. doi: 10.1034/j.1600-6143.2002.20812.x. [DOI] [PubMed] [Google Scholar]
- 166.Becker NS, Rodriguez JA, Barshes NR, O'Mahony CA, Goss JA, Aloia TA. Outcomes analysis for 280 patients with cholangiocarcinoma treated with liver transplantation over an 18-year period. J Gastrointest Surg. 2008;12:117–122. doi: 10.1007/s11605-007-0335-4. [DOI] [PubMed] [Google Scholar]
- 167.Robles R, Figueras J, Turrión VS, Margarit C, Moya A, Varo E, Calleja J, Valdivieso A, Valdecasas JC, López P, et al. Spanish experience in liver transplantation for hilar and peripheral cholangiocarcinoma. Ann Surg. 2004;239:265–271. doi: 10.1097/01.sla.0000108702.45715.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Heimbach JK, Gores GJ, Haddock MG, Alberts SR, Nyberg SL, Ishitani MB, Rosen CB. Liver transplantation for unresectable perihilar cholangiocarcinoma. Semin Liver Dis. 2004;24:201–207. doi: 10.1055/s-2004-828896. [DOI] [PubMed] [Google Scholar]
- 169.Rea DJ, Heimbach JK, Rosen CB, Haddock MG, Alberts SR, Kremers WK, Gores GJ, Nagorney DM. Liver transplantation with neoadjuvant chemoradiation is more effective than resection for hilar cholangiocarcinoma. Ann Surg. 2005;242:451–458; discussion 458-461. doi: 10.1097/01.sla.0000179678.13285.fa. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Rosen CB, Heimbach JK, Gores GJ. Liver transplantation for cholangiocarcinoma. Transpl Int. 2010;23:692–697. doi: 10.1111/j.1432-2277.2010.01108.x. [DOI] [PubMed] [Google Scholar]
- 171.Ito K, Ito H, Allen PJ, Gonen M, Klimstra D, D'Angelica MI, Fong Y, DeMatteo RP, Brennan MF, Blumgart LH, et al. Adequate lymph node assessment for extrahepatic bile duct adenocarcinoma. Ann Surg. 2010;251:675–681. doi: 10.1097/SLA.0b013e3181d3d2b2. [DOI] [PubMed] [Google Scholar]
- 172.Endo I, House MG, Klimstra DS, Gönen M, D'Angelica M, Dematteo RP, Fong Y, Blumgart LH, Jarnagin WR. Clinical significance of intraoperative bile duct margin assessment for hilar cholangiocarcinoma. Ann Surg Oncol. 2008;15:2104–2112. doi: 10.1245/s10434-008-0003-2. [DOI] [PubMed] [Google Scholar]
- 173.Hernandez J, Cowgill SM, Al-Saadi S, Villadolid D, Ross S, Kraemer E, Shapiro M, Mullinax J, Cooper J, Goldin S, et al. An aggressive approach to extrahepatic cholangiocarcinomas is warranted: margin status does not impact survival after resection. Ann Surg Oncol. 2008;15:807–814. doi: 10.1245/s10434-007-9756-2. [DOI] [PubMed] [Google Scholar]
- 174.Shingu Y, Ebata T, Nishio H, Igami T, Shimoyama Y, Nagino M. Clinical value of additional resection of a margin-positive proximal bile duct in hilar cholangiocarcinoma. Surgery. 2010;147:49–56. doi: 10.1016/j.surg.2009.06.030. [DOI] [PubMed] [Google Scholar]
- 175.Petrowsky H, Hong JC. Current surgical management of hilar and intrahepatic cholangiocarcinoma: the role of resection and orthotopic liver transplantation. Transplant Proc. 2009;41:4023–4035. doi: 10.1016/j.transproceed.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 176.Anderson C, Kim R. Adjuvant therapy for resected extrahepatic cholangiocarcinoma: a review of the literature and future directions. Cancer Treat Rev. 2009;35:322–327. doi: 10.1016/j.ctrv.2008.11.009. [DOI] [PubMed] [Google Scholar]
- 177.Rao S, Cunningham D, Hawkins RE, Hill ME, Smith D, Daniel F, Ross PJ, Oates J, Norman AR. Phase III study of 5FU, etoposide and leucovorin (FELV) compared to epirubicin, cisplatin and 5FU (ECF) in previously untreated patients with advanced biliary cancer. Br J Cancer. 2005;92:1650–1654. doi: 10.1038/sj.bjc.6602576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Kornek GV, Schuell B, Laengle F, Gruenberger T, Penz M, Karall K, Depisch D, Lang F, Scheithauer W. Mitomycin C in combination with capecitabine or biweekly high-dose gemcitabine in patients with advanced biliary tract cancer: a randomised phase II trial. Ann Oncol. 2004;15:478–483. doi: 10.1093/annonc/mdh096. [DOI] [PubMed] [Google Scholar]
- 179.Penz M, Kornek GV, Raderer M, Ulrich-Pur H, Fiebiger W, Lenauer A, Depisch D, Krauss G, Schneeweiss B, Scheithauer W. Phase II trial of two-weekly gemcitabine in patients with advanced biliary tract cancer. Ann Oncol. 2001;12:183–186. doi: 10.1023/a:1008352123009. [DOI] [PubMed] [Google Scholar]
- 180.Choi CW, Choi IK, Seo JH, Kim BS, Kim JS, Kim CD, Um SH, Kim JS, Kim YH. Effects of 5-fluorouracil and leucovorin in the treatment of pancreatic-biliary tract adenocarcinomas. Am J Clin Oncol. 2000;23:425–428. doi: 10.1097/00000421-200008000-00023. [DOI] [PubMed] [Google Scholar]
- 181.Ducreux M, Van Cutsem E, Van Laethem JL, Gress TM, Jeziorski K, Rougier P, Wagener T, Anak O, Baron B, Nordlinger B. A randomised phase II trial of weekly high-dose 5-fluorouracil with and without folinic acid and cisplatin in patients with advanced biliary tract carcinoma: results of the 40955 EORTC trial. Eur J Cancer. 2005;41:398–403. doi: 10.1016/j.ejca.2004.10.026. [DOI] [PubMed] [Google Scholar]
- 182.Iyer RV, Gibbs J, Kuvshinoff B, Fakih M, Kepner J, Soehnlein N, Lawrence D, Javle MM. A phase II study of gemcitabine and capecitabine in advanced cholangiocarcinoma and carcinoma of the gallbladder: a single-institution prospective study. Ann Surg Oncol. 2007;14:3202–3209. doi: 10.1245/s10434-007-9539-9. [DOI] [PubMed] [Google Scholar]
- 183.Ellis PA, Norman A, Hill A, O'Brien ME, Nicolson M, Hickish T, Cunningham D. Epirubicin, cisplatin and infusional 5-fluorouracil (5-FU) (ECF) in hepatobiliary tumours. Eur J Cancer. 1995;31A:1594–1598. doi: 10.1016/0959-8049(95)00323-b. [DOI] [PubMed] [Google Scholar]
- 184.André T, Reyes-Vidal JM, Fartoux L, Ross P, Leslie M, Rosmorduc O, Clemens MR, Louvet C, Perez N, Mehmud F, et al. Gemcitabine and oxaliplatin in advanced biliary tract carcinoma: a phase II study. Br J Cancer. 2008;99:862–867. doi: 10.1038/sj.bjc.6604628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Eckel F, Schmid RM. Chemotherapy in advanced biliary tract carcinoma: a pooled analysis of clinical trials. Br J Cancer. 2007;96:896–902. doi: 10.1038/sj.bjc.6603648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Takada T, Amano H, Yasuda H, Nimura Y, Matsushiro T, Kato H, Nagakawa T, Nakayama T. Is postoperative adjuvant chemotherapy useful for gallbladder carcinoma? A phase III multicenter prospective randomized controlled trial in patients with resected pancreaticobiliary carcinoma. Cancer. 2002;95:1685–1695. doi: 10.1002/cncr.10831. [DOI] [PubMed] [Google Scholar]
- 187.Yubin L, Chihua F, Zhixiang J, Jinrui O, Zixian L, Jianghua Z, Ye L, Haosheng J, Chaomin L. Surgical management and prognostic factors of hilar cholangiocarcinoma: experience with 115 cases in China. Ann Surg Oncol. 2008;15:2113–2119. doi: 10.1245/s10434-008-9932-z. [DOI] [PubMed] [Google Scholar]
- 188.Pitt HA, Nakeeb A, Abrams RA, Coleman J, Piantadosi S, Yeo CJ, Lillemore KD, Cameron JL. Perihilar cholangiocarcinoma. Postoperative radiotherapy does not improve survival. Ann Surg. 1995;221:788–797; discussion 797-798. doi: 10.1097/00000658-199506000-00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Cameron JL, Pitt HA, Zinner MJ, Kaufman SL, Coleman J. Management of proximal cholangiocarcinomas by surgical resection and radiotherapy. Am J Surg. 1990;159:91–97; discussion 97-98. doi: 10.1016/s0002-9610(05)80612-9. [DOI] [PubMed] [Google Scholar]
- 190.Zlotecki RA, Jung LA, Vauthey JN, Vogel SB, Mendenhall WM. Carcinoma of the extrahepatic biliary tract: surgery and radiotherapy for curative and palliative intent. Radiat Oncol Investig. 1998;6:240–247. doi: 10.1002/(SICI)1520-6823(1998)6:5<240::AID-ROI6>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 191.Todoroki T, Ohara K, Kawamoto T, Koike N, Yoshida S, Kashiwagi H, Otsuka M, Fukao K. Benefits of adjuvant radiotherapy after radical resection of locally advanced main hepatic duct carcinoma. Int J Radiat Oncol Biol Phys. 2000;46:581–587. doi: 10.1016/s0360-3016(99)00472-1. [DOI] [PubMed] [Google Scholar]
- 192.Cheng Q, Luo X, Zhang B, Jiang X, Yi B, Wu M. Predictive factors for prognosis of hilar cholangiocarcinoma: postresection radiotherapy improves survival. Eur J Surg Oncol. 2007;33:202–207. doi: 10.1016/j.ejso.2006.09.033. [DOI] [PubMed] [Google Scholar]
- 193.Schoenthaler R, Phillips TL, Castro J, Efird JT, Better A, Way LW. Carcinoma of the extrahepatic bile ducts. The University of California at San Francisco experience. Ann Surg. 1994;219:267–274. doi: 10.1097/00000658-199403000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.González González D, Gouma DJ, Rauws EA, van Gulik TM, Bosma A, Koedooder C. Role of radiotherapy, in particular intraluminal brachytherapy, in the treatment of proximal bile duct carcinoma. Ann Oncol. 1999;10 Suppl 4:215–220. [PubMed] [Google Scholar]
- 195.Stein DE, Heron DE, Rosato EL, Anné PR, Topham AK. Positive microscopic margins alter outcome in lymph node-negative cholangiocarcinoma when resection is combined with adjuvant radiotherapy. Am J Clin Oncol. 2005;28:21–23. doi: 10.1097/01.coc.0000139017.90599.f5. [DOI] [PubMed] [Google Scholar]
- 196.Heron DE, Stein DE, Eschelman DJ, Topham AK, Waterman FM, Rosato EL, Alden M, Anne PR. Cholangiocarcinoma: the impact of tumor location and treatment strategy on outcome. Am J Clin Oncol. 2003;26:422–428. doi: 10.1097/01.COC.0000026833.73428.1F. [DOI] [PubMed] [Google Scholar]
- 197.Gerhards MF, van Gulik TM, González González D, Rauws EA, Gouma DJ. Results of postoperative radiotherapy for resectable hilar cholangiocarcinoma. World J Surg. 2003;27:173–179. doi: 10.1007/s00268-002-6434-1. [DOI] [PubMed] [Google Scholar]
- 198.Nakeeb A, Tran KQ, Black MJ, Erickson BA, Ritch PS, Quebbeman EJ, Wilson SD, Demeure MJ, Rilling WS, Dua KS, et al. Improved survival in resected biliary malignancies. Surgery. 2002;132:555–563; discission 563-564. doi: 10.1067/msy.2002.127555. [DOI] [PubMed] [Google Scholar]
- 199.Kim S, Kim SW, Bang YJ, Heo DS, Ha SW. Role of postoperative radiotherapy in the management of extrahepatic bile duct cancer. Int J Radiat Oncol Biol Phys. 2002;54:414–419. doi: 10.1016/s0360-3016(02)02952-8. [DOI] [PubMed] [Google Scholar]
- 200.Serafini FM, Sachs D, Bloomston M, Carey LC, Karl RC, Murr MM, Rosemurgy AS. Location, not staging, of cholangiocarcinoma determines the role for adjuvant chemoradiation therapy. Am Surg. 2001;67:839–843; discussion 843-844. [PubMed] [Google Scholar]
- 201.Hughes MA, Frassica DA, Yeo CJ, Riall TS, Lillemoe KD, Cameron JL, Donehower RC, Laheru DA, Hruban RH, Abrams RA. Adjuvant concurrent chemoradiation for adenocarcinoma of the distal common bile duct. Int J Radiat Oncol Biol Phys. 2007;68:178–182. doi: 10.1016/j.ijrobp.2006.11.048. [DOI] [PubMed] [Google Scholar]
- 202.Nelson JW, Ghafoori AP, Willett CG, Tyler DS, Pappas TN, Clary BM, Hurwitz HI, Bendell JC, Morse MA, Clough RW, et al. Concurrent chemoradiotherapy in resected extrahepatic cholangiocarcinoma. Int J Radiat Oncol Biol Phys. 2009;73:148–153. doi: 10.1016/j.ijrobp.2008.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Borghero Y, Crane CH, Szklaruk J, Oyarzo M, Curley S, Pisters PW, Evans D, Abdalla EK, Thomas MB, Das P, et al. Extrahepatic bile duct adenocarcinoma: patients at high-risk for local recurrence treated with surgery and adjuvant chemoradiation have an equivalent overall survival to patients with standard-risk treated with surgery alone. Ann Surg Oncol. 2008;15:3147–3156. doi: 10.1245/s10434-008-9998-7. [DOI] [PubMed] [Google Scholar]
- 204.Park JH, Choi EK, Ahn SD, Lee SW, Song SY, Yoon SM, Kim YS, Lee YS, Lee SG, Hwang S, et al. Postoperative Chemoradiotherapy for Extrahepatic Bile Duct Cancer. Int J Radiat Oncol Biol Phys. 2010:Epub ahead of print. doi: 10.1016/j.ijrobp.2009.12.031. [DOI] [PubMed] [Google Scholar]