Abstract
The aromatic amine 4-aminobiphenyl (ABP) is a liver procarcinogen in mice, requiring enzymatic bioactivation to exert its tumorigenic effect. To assess the role of arylamine N-acetyltransferase (NAT)-dependent acetylation capacity in the risk for ABP-induced liver tumors, we compared 1-year liver tumor incidence following the postnatal exposure of wild-type and NAT-deficient Nat1/2(−/−) mice to ABP. At an ABP exposure of 1200 nmoles, male Nat1/2(−/−) mice had a liver tumor incidence of 36% compared to 69% in wild-type males, and at 600 nmoles there was a complete absence of tumors compared to 60% in wild-type mice. Only one female wild-type mouse had a tumor using this exposure protocol. However, levels of N-deoxyguanosin-8-yl-ABP-DNA adducts did not correlate with either the strain or sex differences in tumor incidence. These results suggest that female sex and NAT deficiency reduce risk for ABP-induced liver tumors, but by mechanisms unrelated to differences in DNA-damaging events.
Keywords: aromatic amine, chemical carcinogen, arylamine N-acetyltransferase, DNA adduct, liver cancer, sex differences
1. Introduction
Metabolic bioactivation of chemical carcinogens into DNA damaging agents is a causal factor in a multi-step process leading to the development of tissue-specific tumors [1]. While genotoxic effects are believed to be a necessary prerequisite for chemically-induced carcinogenesis, it has become clear that non-genotoxic effects also contribute to the development of tumors [2,3] by sustaining or promoting a proliferative environment, which is one of the proposed hallmarks of cancer [4].
The aromatic amine 4-aminobiphenyl (ABP), which is found in cigarette smoke and hair dyes, is a recognized human bladder carcinogen (for review see [3]). Since environmental exposure to ABP may thus contribute to the occurrence of some tissue-specific cancers, many studies have been undertaken to measure and correlate potential biomarkers of ABP exposure and cancer risk. ABP-DNA adducts have been detected in the DNA from bladder cancer patients [5], exfoliated ductal epithelial cells isolated from human breast milk [6] and from hepatocytes obtained from hepatocellular carcinoma cases [7], and a higher urinary excretion of ABP is found in smokers versus non-smokers [8]. In addition, ABP can bind to proteins such as hemoglobin and albumin, and thus protein adducts such as ABP hemoglobin adducts have also been used as a surrogate or biomarker for exposure and potential risk [9].
The first step in one proposed bioactivation model for ABP is its N-oxidation to N-hydroxy-ABP (N-OH-ABP). The hepatic cytochrome P450 (CYP) isoform CYP1A2 has been implicated in this reaction in the liver [10], whereas CYP1A1 [11] or peroxidases [12] may be involved in its localized oxidation within other tissues. O-acetylation (OAT) of N-OH-ABP by arylamine N-acetyltransferases (NATs) can yield an acetoxy ester, which is chemically unstable and decomposes into a highly reactive arylnitrenium ion that can bind to and damage DNA. The major DNA adduct formed as a result of ABP bioactivation is N-deoxyguanosin-8-yl-ABP (dG-C8-ABP) which causes primarily G:C → T:A transversion mutations [13] that have been mapped as a common mutation in codon 61 of the H-ras gene of ABP-exposed mice [14]. Competing with N-oxidation in the liver is the protective NAT-mediated N-acetylation which leads to the production of an innocuous acetamide metabolite. Thus, it is unclear whether NAT enzymes facilitate (by O-acetylation) or prevent (by N-acetylation) ABP-induced tumorigenesis.
Two functional NAT isozymes, NAT1 and NAT2, are present in humans and display distinct tissue distributions and substrate specificities [15]. In human liver, both ABP N-acetylation and N-OH-ABP O-acetylation activities are mediated predominantly by NAT2 [16], whereas NAT1 has a widespread tissue distribution and may be involved in the localized activation of aromatic amines within other tissues such as the bladder [17]. In addition, NAT1 is transcribed by alternatively spliced promoters, NATa and NATb, giving rise to multiple transcripts which differ in tissue specificity and the ability to form ABP-DNA adducts and mutations in vitro [18]. Thus, the role that NAT enzymes may have in either the bioactivation or detoxification of aromatic amines such as ABP may ultimately depend on their tissue expression profiles, the balance of competing enzyme pathways, the inducibility of these pathways, the dose and route of exposure, and organ-specific metabolism that can have a significant effect on the rate of clearance [19,20]. Furthermore, NAT enzyme activity can be modulated by environmental factors such as oxidative stress arising from inflammation or environmental exposure [21], and by folate levels which have been shown to affect the methylation status of the mouse Nat2 promoter [22]. Thus, genotype is not the only factor that can influence functional NAT enzyme activity.
Since human NAT1 and NAT2 are both highly polymorphic enzymes (for review see [15,23]), several epidemiological studies have been undertaken to investigate the role of rapid versus slow acetylator phenotype or NAT genotype to tissue-specific cancer risk, particularly in relation to arylamine or heterocyclic amine exposure. NAT1 and/or NAT2 genotypes have been investigated with respect to cancers of the bladder, colon, breast and liver [24,25,26,27]. Although results from epidemiological studies have tended to be contradictory and indicate that there is no clear or consistent correlation between acetylator status and cancer risk, more recent meta-analyses have indicated an increased risk of either breast or bladder cancer in NAT2 slow acetylators that is dependent on smoking intensity [25,26]. Limitations to epidemiological studies include small sample sizes, genotype misclassification errors and lack of quantitative information on chemical exposures due to the complex mixtures of aromatic amines and other chemicals to which individuals may be exposed.
To investigate the role of NAT-dependent acetylation in ABP-induced tumorigenesis, in the present study we have used the neonatal carcinogen bioassay and our Nat1/2(−/−) mice, which are deficient in both mouse Nat1 and Nat2 enzyme activities [28]. The neonatal mouse carcinogen bioassay is an accelerated carcinogenicity assay that has been validated using ABP as a positive control for liver tumors [29]. Using this assay, chemical carcinogens requiring metabolic bioactivation tend to show a restricted tissue tumor sensitivity that is reflective of the distribution of available drug metabolizing enzymes, whereas direct-acting genotoxic agents such as the alkylating agent N-ethyl-N-nitrosourea are more likely to result in a widespread tissue distribution of tumors. On the other hand, non-genotoxic chemical carcinogens that mediate increased lipid peroxidation or oxidative stress do not produce tumors using this bioassay [30]. The assay has also been used to demonstrate that in contrast to the bioactivation model described above, mouse Cyp1a2 may not be the initial enzyme involved in ABP-induced carcinogenesis [31]. ABP N-oxidation activity was still evident in Cyp1a2(−/−) mice, albeit at reduced levels, and ABP-DNA adduct levels were not reduced in these animals [32]. These findings have all challenged the view that mouse Cyp1a2 is involved in the metabolic bioactivation of aromatic amines such as ABP, and reinforces the necessity for conducting bioactivation and carcinogenicity studies in vivo to determine the role that specific biotransformation enzymes such as the NATs may play in determining tissue-specific tumor risk.
2. Materials and methods
2.1. Materials
All animal handling procedures were conducted in accordance with Canadian Council on Animal Care guidelines, and were approved by the University of Toronto Animal Care Committee. ABP, carnitine acetyltransferase, acetyl-DL-carnitine and acetyl-CoA were purchased from Sigma-Aldrich Canada Ltd (Oakville, ON, Canada). Enzymes used for digesting genomic DNA into monomers (DNase I, micrococcal nuclease, nuclease P1, spleen phosphodiesterase, snake venom phosphodiesterase, and alkaline phosphatase) were acquired from Sigma-Aldrich United States (St. Louis, MO). dG-C8-ABP and D5-dG-C8-ABP adduct standards were obtained from Toronto Research Chemicals (Toronto, ON, Canada). Generation of the Nat1/2(−/−) mouse line has been described previously [28], and was backcrossed onto a C57BL/6 background for 10 generations. C57BL/6 mice (referred to subsequently as ‘wild-type’) were purchased from Charles River Canada (Saint-Constant, QC, Canada).
2.2. Neonatal dosing regimen and histopathology
Male and female wild-type and Nat1/2(−/−) mice (n=17–26 per sex, genotype and dose) were injected i.p. on postnatal day 8 (one third of the total dose in a 10 μl volume) and postnatal day 15 (two thirds of the total dose in a 20 μl volume) with either a low dose (200 + 400 = 600 nmol total) or a high dose (400 + 800 = 1200 nmol total) of ABP, or an equivalent volume of vehicle (DMSO). With average animal weights of 4 g and 8 g on postnatal days 8 and 15, respectively, these exposures represent ABP doses of approximately 17 mg/kg. Animals were sacrificed for necropsy at 12 months of age, and inspected for gross lesions of the liver and other tissues. Macroscopically visible liver tumors greater than 1 mm were enumerated. All visible liver nodules were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
2.3. In vitro NAT enzyme activity
Cytosols were prepared from livers of postnatal day 8, postnatal day 15 and adult (8 week) wild-type mice as previously described [28]. NAT activity in liver cytosols was determined using ABP as acceptor substrate (n=3 animals per sex and age). Initial enzyme velocities were measured in duplicate in final reaction volumes of 100 μl, consisting of 10 μl of 1 mM ABP (giving a final concentration of 0.1 mM), 20 μl of 0.5 mM acetyl-CoA (giving a final concentration of 0.1 mM), 20 μl of an acetyl-CoA regenerating system composed of 5 mM acetyl-DL-carnitine and 1 U carnitine acetyltransferase per milliliter of assay buffer (250 mM triethanolamine-HCl, 5 mM EDTA, 5 mM DTT, pH 7.5), and 50 μl of cytosol diluted to a protein concentration of 1 μg/μl. Reactions were initiated with the addition of the cytosol, incubated for 10 min at 37° and terminated by the addition of 10 μl of 15% perchloric acid. After precipitation of the denatured protein, the supernatant fractions were assayed for N-acetylated ABP (AABP) by HPLC, using a Shimadzu LC-2010A system (Shimadzu Scientific Instruments Inc., Columbia, MD) and a reverse-phase Beckman Ultrasphere ODS 5 μm column (25 cm × 4.6 mm I.D.; Beckman Instruments, Fullerton, CA). A mobile phase of 20 mM sodium perchlorate (pH 2.5) and acetonitrile (66:34, v/v) was used at a flow rate of 2.0 ml/min, and eluting compounds were detected by ultraviolet absorbance at 280 nm. The limit of detection for AABP was 0.006 nmol/min/mg.
2.4. Measurement of ABP-DNA adducts
Levels of dG-C8-ABP DNA adducts were measured in DNA from the livers of neonatal wild-type and Nat1/2(−/−) mice exposed to the high-dose ABP regimen (400 nmol on postnatal day 8 and 800 nmol on postnatal day 15) and sacrificed 24 hours later, or from 8 week old age- and sex-matched animals dosed with 20 mg/kg ABP and sacrificed 24 hours later. Genomic DNA was isolated from liver tissue essentially as described [28], and resuspended in buffer containing 5 mM Tris-HCl (pH 7.4), 1 mM CaCl2, 1 mM ZnCl2, 10 mM MgCl2. Approximately 200 μg of genomic DNA was added to 500 pg of deuterated internal standard (D5-dG-C8-ABP) and digested with 10 units of DNase I for 1 hr at 37°C, followed by a 6 hr incubation with 5 units micrococcal nuclease, 5 units nuclease P1, 0.01 units spleen phosphodiesterase and 0.01 units snake venom phosphodiesterase. This was followed by an overnight incubation with 5 units of alkaline phosphatase. Digestions were stopped by addition of two volumes of acetonitrile, and the samples were subsequently filtered, concentrated and subjected to LS-MS/MS as described previously [33].
3. Results
3.1. Tumor incidence and multiplicity
There was no significant difference in mean body weights between wild-type and Nat1/2(−/−) mice of the same treatment group, with the exception of male Nat1/2(−/−) mice treated with the 600 nmol dose of ABP (Table I). These mice weighed less than the corresponding wild-type male mice treated with 600 nmol ABP (p < 0.05). Liver weights were not significantly different between mice of the same treatment group except for male wild-type versus male Nat1/2(−/−) mice and female wild-type versus female Nat1/2(−/−) mice treated with 600 nmol ABP. Mean liver weights were higher in male wild-type than male Nat1/2(−/−) mice for the vehicle control and 1200 nmol ABP groups, but these were not statistically different.
TABLE I.
Final Liver and Body Weights of Wild-type and Nat1/2(−/−) Mice
| Sex/Strain | ABP dose a (nmoles) | N b | Body weight (g) | Liver weight (g) |
|---|---|---|---|---|
| Male | ||||
| wild-type | 0 | 23 | 46.2±2.9 | 2.53±0.55 |
| 600 | 20 | 43.4±5.2 | 2.17±0.54 | |
| 1200 | 26 | 40.8±4.9 | 2.18±0.69 | |
| Nat1/2(−/−) | 0 | 25 | 46.1±7.4 | 1.99±0.61 |
| 600 | 17 | 37.4±3.0* | 1.54±0.17* | |
| 1200 | 22 | 40.4±4.7 | 1.71±0.21 | |
| Female | ||||
| wild-type | 0 | 20 | 33.0±6.5 | 1.32±0.17 |
| 600 | 20 | 31.6±7.1 | 1.47±0.58 | |
| 1200 | 28 | 28.3±5.1 | 1.09±0.17 | |
| Nat1/2(−/−) | 0 | 22 | 32.9±4.0 | 1.20±0.06 |
| 600 | 24 | 30.3±7.5 | 1.06±0.19* | |
| 1200 | 23 | 31.0±4.4 | 1.16±0.18 | |
Mice were dosed on postnatal days 8 and 15 with total doses of 0 (DMSO vehicle), 600 or 1200 nmoles of ABP, and sacrificed at one year of age. Data are shown as mean ± SD.
Number of mice per treatment group.
significantly different from wild-type of the same treatment group (p < 0.05). Statistical differences between group means were determined by one-way ANOVA followed by Bonferroni’s multiple comparison test, using GraphPad Prism (GraphPad Software Inc., San Diego, CA).
Both the incidence and multiplicity of liver tumors were highest in male wild-type mice, and these were moderately dependent on the dose of ABP administered (Table II). Male wild-type mice dosed with 1200 nmol ABP exhibited a 15–16% higher liver tumor incidence and multiplicity than those dosed with 600 nmol ABP. Male Nat1/2(−/−) mice dosed with 1200 nmol ABP had a liver tumor incidence and multiplicity that were 48% and 23% lower than similarly treated male wild-type mice, and male Nat1/2(−/−) mice dosed with 600 nmol ABP had a complete absence of liver tumors compared to the 60% incidence in male wild-type mice.
TABLE II.
Liver Tumor Multiplicity and Incidence
| Sex/Strain | ABP dose a (nmoles) | N b | Tumor No. c | Multiplicity d (mean ± SEM) | Incidence e (%) |
|---|---|---|---|---|---|
| Male | |||||
| wild-type | 0 | 23 | 0 | 0 | 0 |
| 600 | 20 | 39 (12) | 3.25±0.72 | 60 | |
| 1200 | 26 | 68 (18) | 3.78±0.65 | 69.2 | |
| Nat1/2(−/−) | 0 | 25 | 0 | 0 | 0 |
| 600 | 17 | 0 | 0 | 0 | |
| 1200 | 22 | 23 (8) | 2.88±1.33 | 36.4 | |
| Female | |||||
| wild-type | 0 | 20 | 0 | 0 | 0 |
| 600 | 20 | 1 (1) | 1 | 5 | |
| 1200 | 28 | 0 | 0 | 0 | |
| Nat1/2(−/−) | 0 | 22 | 0 | 0 | 0 |
| 600 | 24 | 0 | 0 | 0 | |
| 1200 | 23 | 0 | 0 | 0 | |
Mice were dosed on postnatal days 8 and 15 with total doses of 0 (DMSO vehicle), 600 or 1200 nmoles of ABP, sacrificed at one year of age and liver tumors greater than 1 mm in diameter were enumerated.
Number of mice per treatment group
Total number of tumors counted in the treatment group; number in parentheses is the number of animals in the treatment group with at least one visible tumor
Number of tumors per liver in tumor-bearing mice
Percentage of animals in the treatment group with at least one visible tumor
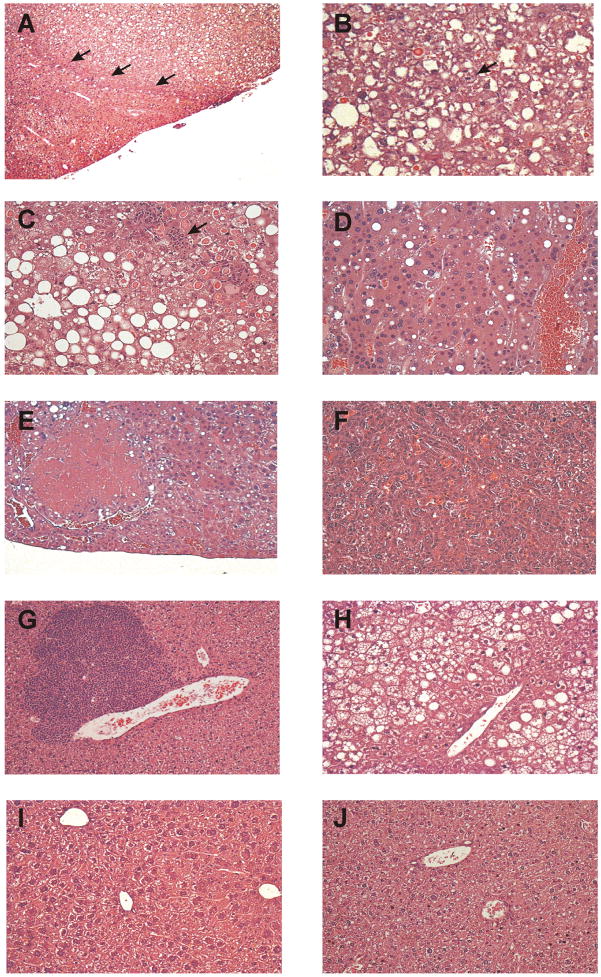
The nodules present in ABP-exposed male mice at 12 months of age were benign adenomas. These nodular adenomas often displayed a distinct margin with a slight compression of overlying hepatocytes (Figure 1A), and were characterized by the presence of mitotic bodies (Figure 1B) and degenerative cytoplasmic changes that included the presence of either cytoplasmic vacuoles, hyaline inclusions and/or the occasional interstitial inflammatory mononuclear infiltrate (Figure 1C). Only one hepatocellular carcinoma was observed in a male Nat1/2(−/−) mouse exposed to 1200 nmol of ABP. This liver tumor replaced large areas of the normal liver and was composed mostly of atypical hepatocytes that displayed notable nuclear pleomorphism and binucleation (Figure 1D). These malignant hepatocytes were grouped as multicellular clusters that showed occasional zonal necrosis despite good overall neovascularization (Figure 1E).
Fig. 1.
Representative H&E liver sections from wild-type or Nat1/2(−/−) mice sacrificed at one year of age after neonatal ABP (dose as indicated) or DMSO vehicle administration. (A) Liver from a representative male wild-type mouse dosed with 1200 nmol of ABP showing compression of hepatocytes at the border of a nodule (x40). (B) Mitotic body (arrowhead) present in the adenoma from a male wild-type dosed with 1200 nmol ABP (x200). (C) Degenerative changes indicated by the presence of intracytoplasmic inclusions and macrovacuoles in an adenoma with inflammatory cells demarcated by an arrow from a representative male wild-type mouse dosed with 1200 nmol of ABP (x200). (D) Nuclear pleomorphism and absence of sinusoidal radial pattern in a hepatocellular carcinoma from a male Nat1/2(−/−) mouse dosed with 1200 nmol ABP (x200). (E) Region of necrosis in a hepatocellular carcinoma from a male Nat1/2(−/−) mouse treated with 1200 nmol of ABP (x200). (F) Presence of erythrocytes in sinsusoidal spaces and disorganized architecture in a female wild-type dosed with 600 nmol ABP (x200). (G) Region of focal periportal inflammation in a liver displaying overall normal architecture from a female wild-type mouse dosed with 1200 nmol of ABP (x100). (H) Steatosis in a male wild-type mouse dosed with 600 nmol ABP (x200). (I) Liver from a representative male Nat1/2(−/−) mouse dosed with 600 nmol of ABP displaying an overall normal architecture (x200). (J) Liver from a representative female Nat1/2(−/−) dosed with 1200 nmol ABP, displaying an overall normal morphology (x200).
Female wild-type and Nat1/2(−/−) mice displayed an almost complete absence of liver tumors at both doses of ABP. The liver from one wild-type female mouse dosed with 600 nmol ABP, however, was highly enlarged with a final liver weight of 3.14 g. This liver exhibited an altered architecture with erythrocyte-filled sinusoidal spaces but with no apparent nodular structure (Figure 1F). Atypical hepatocytes were arranged in a disorganized pattern, suggesting the presence of a hepatocellular carcinoma. No macroscopic liver tumors were detected in animals of either sex or strain dosed with the DMSO vehicle. Focal periportal inflammation or regions of localized mononuclear inflammatory infiltration (Figure 1G) were observed in all groups of animals regardless of dose, sex and strain.
3.2. Fatty changes in the liver
Fatty liver changes were observed most frequently in wild-type male mice. Macroscopically, the livers appeared paler in color and enlarged, although mean liver weights in most cases were not significantly different (see above). Fatty changes were evident microscopically as a diffuse vacuolation with regions of focal vacuolation (Figure 1H). Although the incidence of these changes was highest in DMSO-dosed wild-type male animals (70%), wild-type males administered either the 600 nmol (55% incidence) or 1200 nmol dose of ABP (31% incidence) also displayed diffuse vacuolation. No evidence of fatty changes within the livers of ABP-dosed male Nat1/2(−/−) animals was observed (Figure 1I), while less than 20% of DSMO-dosed male Nat1/2(−/−) mice exhibited a diffuse vacuolation within their livers. This pattern of diffuse vacuolation observed in male mice was not observed in female mice, regardless of dose or strain. In contrast, the livers of female mice typically displayed a normal non-fasted morphology (Figure 1J).
3.3. In vitro liver NAT activity
NAT activity was measured in liver cytosols from postnatal day 8, postnatal day 15, and adult (8 weeks of age) wild-type mice using ABP as acceptor substrate (Figure 2). Activities were 39% and 76% of adult levels by postnatal days 8 and 15, respectively, and did not significantly differ between males and females at any stage of development tested. NAT activity was undetectable in liver cytosols prepared from male or female Nat1/2(−/−) mice at any developmental stage (AABP limit of detection: 0.006 nmol/min/mg).
Fig. 2.

Comparison of NAT activities in wild-type neonatal and adult male and female mice. Liver cytosols prepared from either postnatal day 8 (PND8), postnatal day 15 (PND15) or 8 week old wild-type male and female mice were assayed for NAT activity using 0.1 mM ABP in the presence of 0.1 mM AcCoA, as described in Materials and Methods. N-acetylated ABP (AABP) was separated and quantified by HPLC. Product formation rates represent the means ± SD from n=3 animals per sex and age. There was no statistically significant difference in enzyme activities between males and females at any age.
3.4. dG-C8-ABP adducts
Levels of the major ABP-DNA adduct dG-C8-ABP were measured in neonatal animals exposed to the high-dose ABP regimen (400 nmol on postnatal day 8 and 800 nmol on postnatal day 15). There was no significant difference between males and females of the same strain in animals dosed at the neonatal stage of development. However, there was a significant difference between strains (Figure 3A). Both male and female Nat1/2(−/−) neonatal mice had significantly higher dG-C8-ABP adducts than wild-type male (p < 0.001) and female neonatal mice (p < 0.001), respectively.
Fig. 3.
Levels of dG-C8-ABP adducts in adult (A) and neonatal (B) male and female wild-type and Nat1/2(−/−) mice dosed with ABP. Adult animals were dosed i.p. with 20 mg/kg ABP and sacrificed 24 hours later. Neonatal animals were dosed using the 1200 nmol ABP dosing regimen on postnatal days 8 and 15, and sacrificed 24 hours later. Liver genomic DNA was isolated and levels of dG-C8-ABP were measured by HPLC/MS. *, significantly different from wild-type (p< 0.05).
In ABP-dosed adult mice, adduct levels tended to be higher in female mice than in male mice, as well as higher in Nat1/2(−/−) mice than in wild-type mice (Figure 3B). However, these differences were not statistically significant. Overall, adduct levels were higher in neonatally exposed Nat1/2(−/−) mice than in adult wild-type or Nat1/2(−/−) mice (p < 0.005).
4. Discussion
We have used the neonatal mouse carcinogen bioassay and our Nat1- and Nat2-deficient Nat1/2(−/−) mice to interrogate the role of the NAT enzymes in either facilitating or preventing ABP-induced liver tumorigenesis. In our study, ABP-induced liver tumorigenesis was either attenuated (at the higher ABP dose) or completely absent (at the lower ABP dose) in male Nat1/2(−/−) mice compared to male wild-type mice at the ABP doses used. On the surface, these results would suggest that in the absence of functional Nat enzyme activity, Nat1/2(−/−) mice are protected from ABP-induced liver tumorigenesis due to the loss of bioactivating O-acetylation activity. Female mice, however, displayed a protection from liver tumors regardless of genotype and ABP dose, indicating that sex is a significant factor that can overcome susceptibility to liver carcinogens in mice, and it parallels epidemiological findings in humans in which hepatocellular carcinoma exhibits a significant male bias [34].
However, we saw a paradoxical trend towards higher levels of the major DNA damage adduct dG-C8-ABP in both male and female adult Nat1/2(−/−) mice than in their wild-type counterparts. Although this finding is consistent with the notion that intact ABP N-acetylation in wild-type mice protects against DNA adduct formation, it contradicts the higher liver tumor incidence in male wild-type mice. Moreover, despite the almost complete lack of ABP-induced liver tumors in both wild-type and Nat1/2(−/−) female mice, levels of dG-C8-ABP were generally higher in adult female mice than male mice, although this difference was not statistically significant. Higher dG-C8-ABP adducts in female mice have also been reported in a study using Cyp1a2(−/−) mice dosed with ABP [32], and this did not correlate with liver tumor incidence [31].
Since the mice used in our tumor study were dosed at the neonatal stage of development, we treated neonatal mice with the tumor-inducing high-dose ABP regimen to compare levels of dG-C8-ABP adducts. No sex difference was observed in hepatic dG-C8-ABP adduct levels, which is consistent with a previous study that found no significant difference in the levels of this adduct between neonatally-dosed male and female mice [35]. However, neonatal Nat1/2(−/−) mice had significantly higher adduct levels than neonatal wild-type mice. These results again contrast with the tumor incidences in our carcinogenicity study, where female mice of both strains were protected from liver tumors and male Nat1/2(−/−) mice exhibited both a lower incidence and multiplicity of liver tumors than wild-type males. Thus, surprisingly this biomarker of acute ABP-induced DNA damage did not correlate with the sex and strain differences we observed in liver tumor incidence.
The sex difference in levels of dG-C8-ABP adducts observed in neonatal wild-type animals cannot be attributed to the level of hepatic Nat activity, since this activity in neonatal wild-type mice did not differ between males and females. Rather, the discrepancy in levels of this major adduct may be due to the presence of other bioactivating or detoxifying enzymes present in the liver. Sulfotransferases (SULTs) can catalyze the O-sulfation of N-OH-ABP to form a sulfonyloxy ester intermediate that is also unstable and can bind to DNA, leading to the formation of adducts that facilitate DNA mutation [36]. SULT1A, for instance, has been found to be female predominant in adult mouse liver [37], and is present in neonates at levels similar to or higher than adult levels with no significant sex difference in expression observed until adulthood [38]. A sex difference has also been reported for ABP N-glucuronidation in the livers of C57BL/6 mice, with male mice exhibiting a significantly higher activity [39]. As such, this sex difference in N-glucuronidation activity may also contribute to the lower levels of DNA adducts seen in male mice. Thus, it may be the balance of competing biotransformation pathways and their temporal and/or differential hormonal regulation that determine the levels of dG-C8-ABP adducts formed after acute ABP administration.
While dG-C8-ABP is the major DNA adduct observed following ABP exposure, other minor adducts such as N-deoxyadenosin-8-yl-ABP (dA-C8-ABP) have also been detected [40]. In CD-1 mice exposed neonatally to ABP, A:T → T:A transversion mutations, a molecular feature of dA-C8-ABP adducts, in the H-ras codon 61 were primarily found in contrast to the G:C → T:A transversion mutation in H-ras codon 61 of B6C3F1 mice [41]. More importantly, the developmental stage and the specific mutational spectrum or mutation hotspots induced by ABP in our animals may influence susceptibility to liver tumor formation. Sequence analysis of mutated cII genes from Big Blue transgenic mice indicated the presence of a high frequency of G:C → T:A transversion mutations, but only in animals dosed neonatally with ABP [35]. The induced mutation frequency was also significantly higher in female than in male neonates, although adduct levels were not significantly different. In contrast, the mutational spectra of ABP-dosed adult mice were similar to control mice, suggesting that DNA adducts may be efficiently repaired in adults, whereas the high levels of cell proliferation may facilitate the fixation of unrepaired DNA damage into mutations in neonatal animals. Thus, it will be interesting to determine both the ABP-induced mutational spectra and the frequency of specific mutations, either in the H-ras gene or in the cII gene of Nat1/2(−/−) neonatal animals that have been crossed to transgenic mutation reporter mice. Such studies are currently in progress.
The male predominance of liver tumors in mice following exposure to chemical carcinogens is not restricted to ABP. A higher liver tumor incidence was also found in male than in female mice exposed neonatally to aflatoxin B1 while DNA adduct levels, mutation frequencies and types of mutations showed no sex difference, suggesting that post-initiation effects may be more important determinants of liver tumor development [42]. Diethylnitrosamine (DEN)-induced liver tumorigenesis is also male predominant [43]. Female mice exposed neonatally to a single 25 mg/kg dose of DEN on postnatal day 15 exhibited a lower incidence of liver tumors at 8 months of age than males, which correlated with less hepatotoxicity and an attenuated increase in serum levels of the inflammatory cytokine IL-6 in females in the 24–48 hr period following high-dose (100 mg/kg) DEN administration to adult mice [43]. However, in that study the acute hepatotoxic and inflammatory effects of low-dose DEN in mice exposed neonatally (the tumor-inducing protocol) were not reported. It is possible that ABP may act in a manner similar to DEN by mediating a cytotoxic response in the liver, resulting in the release of inflammatory cytokines which can then promote or facilitate tumor progression. Indeed, cytotoxicity to ABP has been observed in repair-deficient CHO cells co-expressing human NAT2*4 and CYP1A1 [44]. However, we saw no histochemical evidence for significant dose, sex or strain differences in the extent of inflammatory cell infiltration of livers 1 year after mice were dosed neonatally with ABP (Fig. 1). Also, in preliminary experiments we have seen no acute (2–48 hr) increase in either liver damage (serum ALT levels or histological evidence of hepatic necrosis) or serum IL-6 levels in male or female wild-type or Nat1/2(−/−) mice exposed to ABP neonatally using our tumor-inducing protocol (data not shown). It will be important to determine whether and how these and other markers of cell death, proliferation and inflammation following exposure to ABP or chemically unrelated hepatocarcinogens such as DEN are differentially altered in wild-type and Nat1/2(−/−) male and female mice not only acutely but also chronically during the time interval between neonatal chemical exposure and the eventual appearance of tumors in adult mice. Such studies are in progress.
Another interesting finding from our study was that female mice showed no histological signs of fatty liver changes, and male Nat1/2(−/−) mice had a markedly lower incidence of fatty liver changes than wild-type males. Wild-type males showed evidence of both microvesicular and macrovesicular steatosis, which is similar to the pattern of steatosis observed in aromatase-deficient mice, which cannot synthesize estrogen [45]. Obesity or fatty liver has also been shown to promote chronic inflammation and liver tumors in mice [46], whereas caloric restriction in mice exposed neonatally to ABP attenuates liver tumor formation [47]. Thus it will be important to determine whether a chronic inflammation is established subsequent to neonatal ABP exposure, whether it is reduced in Nat1/2(−/−) mice, and its possible relationship to lipid homeostasis. Interestingly, estrogen administration has been shown to protect against the development of fatty liver in aromatase-deficient male mice [45]. The male bias in susceptibility also extends to humans, with men being more predisposed to the development of liver steatosis than women [48].
Because the levels of the putative major DNA damaging adduct do not correlate with liver tumor incidence in our male mice, an alternative mechanism may be occurring that is dependent on an as yet unidentified endogenous function of NATs which is distinct from their ability to influence the degree of reactive metabolite production. For instance, human NAT1 is over-expressed in breast tumors, promoting cellular proliferation and resistance to etoposide [49]. Attenuation of NAT1 activity, either by small molecule inhibition in MDA-MB-231 breast cancer cells [50] or via RNAi-mediated knockdown in colon adenocarcinoma HT-29 cells [51], was found to decrease cell growth and invasiveness of breast cancer cells and to increase cell-cell contact growth inhibition and doubling time in HT-29 cells. In addition, NAT1 knock-down was able to slow tumor formation in vivo. It is possible that the absence of mouse Nat2, the orthologue of human NAT1 [52], is protective in our study by decreasing the rate of cell proliferation and thus slowing tumor growth. Thus, it will be important to determine whether rates of cellular proliferation differ between wild-type and Nat1/2(−/−) mice after ABP administration, and the mechanism by which this occurs.
Another potential protective effect in Nat1/2(−/−) mice may relate to the role of mouse Nat2 in folate catabolism. Dietary folate has been shown to increase CpG methylation in the promoter region of the Nat2 gene leading to a reduction in the use of its core promoter, while functional deletion of the mouse Nat2 gene resulted in an increase in Nat2 methylation [22]. In addition, an inverse correlation has been observed between folate levels and Nat2 enzyme activity in Nat2 rapid and slow acetylator mouse strains [53], and higher folate levels have been detected in erythrocytes from human subjects with lower NAT1 activities [54]. Folate is required for the synthesis of S-adenosylmethionine (SAM), the methyl donor for cellular methylation reactions. Increases in SAM are associated with an increase in glutathione, reduced oxidative stress, a decrease in TNF-α and hepatic inflammation, reduced liver injury in animal models, a restoration of normal methylation reactions, and a pro-apoptotic effect in liver cancer cell lines (for review see [55]). Over-expression of human NAT1 has not been achievable in mice in the presence of normal endogenous levels of its mouse orthologue Nat2 [53], while mice can survive with no apparent pathology with the loss of Nat2 and/or Nat1. This suggests that mouse Nat2 and its human orthologue NAT1 may have endogenous roles in maintaining homeostasis, and that their over-expression is in fact detrimental to the animal. If absence of mouse Nat2 leads to increases in the levels of folate with a corresponding increase in the levels of SAM, this may have a protective effect against liver injury/tumorigenesis induced by ABP and fatty liver changes in male Nat1/2(−/−) animals. Indeed, folic acid supplementation during early hepatocarcinogenesis in rats has a protective effect by reducing hepatic DNA damage and cell proliferation [56], whereas folate deficiency has been associated with increased DNA strand breaks and reduced DNA repair [57]. It is not yet known whether the levels of folate and/or SAM differ in Nat1/2(−/−) mice.
The results from our study indicate that ABP-induced liver tumor incidence in mice does not correspond with the acute DNA damage that occurs in neonatal animals. Female gender and absence of functional Nat1/2 protected against the appearance of both liver tumors and steatosis, in contrast to the observation of higher hepatic DNA adduct levels. This suggests that a novel NAT-dependent mechanism may facilitate the growth of tumors, at least in male mice. In addition, the role of estrogen in mediating what is possibly a mechanistically distinct protective effect against ABP-induced liver tumors warrants further investigation, as does the role of chronic inflammation and potential differences in folate/SAM levels. Thus a convergence of multiple factors including sex, biotransformation capacity, inflammation and NAT status may determine the ultimate outcome following procarcinogen exposure.
Acknowledgments
We thank Christopher Koo, Aldora Ho and Daniel Hanna for their technical assistance. This research was supported by operating grants from the Canadian Institutes of Health Research and the National Cancer Institute of Canada (DMG), and by USPHS grant [R01-CA034627] from the National Cancer Institute (DWH).
Footnotes
Conflicts of interest
The authors have nothing to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Weisburger J. Comments on the history and importance of aromatic and heterocyclic amines in public health. Mutat Res. 2002;506–507:9–20. doi: 10.1016/s0027-5107(02)00147-1. [DOI] [PubMed] [Google Scholar]
- 2.Neumann HG. Aromatic amines in experimental cancer research: tissue-specific effects, an old problem and new solutions. Crit Rev Toxicol. 2007;37:211–236. doi: 10.1080/10408440601028603. [DOI] [PubMed] [Google Scholar]
- 3.Cohen SM, Boobis AR, Meek ME, Preston RJ, McGregor DB. 4-Aminobiphenyl and DNA reactivity: case study within the context of the 2006 IPCS Human Relevance Framework for Analysis of a cancer mode of action for humans. Crit Rev Toxicol. 2006;36:803–819. doi: 10.1080/10408440600977651. [DOI] [PubMed] [Google Scholar]
- 4.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 5.Zayas B, Stillwell SW, Wishnok JS, Trudel LJ, Skipper P, Yu MC, Tannenbaum SR, Wogan GN. Detection and quantification of 4-ABP adducts in DNA from bladder cancer patients. Carcinogenesis. 2007;28:342–349. doi: 10.1093/carcin/bgl142. [DOI] [PubMed] [Google Scholar]
- 6.Ambrosone CB, Abrams SM, Gorlewska-Roberts K, Kadlubar FF. Hair dye use, meat intake, and tobacco exposure and presence of carcinogen-DNA adducts in exfoliated breast ductal epithelial cells. Arch Biochem Biophys. 2007;464:169–175. doi: 10.1016/j.abb.2007.05.018. [DOI] [PubMed] [Google Scholar]
- 7.Wang LY, Chen CJ, Zhang YJ, Tsai WY, Lee PH, Feitelson MA, Lee CS, Santella RM. 4-Aminobiphenyl DNA damage in liver tissue of hepatocellular carcinoma patients and controls. Am J Epidemiol. 1998;147:315–323. doi: 10.1093/oxfordjournals.aje.a009452. [DOI] [PubMed] [Google Scholar]
- 8.Riedel K, Scherer G, Engl J, Hagedorn HW, Tricker AR. Determination of three carcinogenic aromatic amines in urine of smokers and nonsmokers. J Anal Toxicol. 2006;30:187–195. doi: 10.1093/jat/30.3.187. [DOI] [PubMed] [Google Scholar]
- 9.Probst-Hensch NM, Bell DA, Watson MA, Skipper PL, Tannenbaum SR, Chan KK, Ross RK, Yu MC. N-acetyltransferase 2 phenotype but not NAT1*10 genotype affects aminobiphenyl-hemoglobin adduct levels. Cancer Epidemiol Biomarkers Prev. 2000;9:619–623. [PubMed] [Google Scholar]
- 10.Kim D, Guengerich FP. Cytochrome P450 activation of arylamines and heterocyclic amines. Annu Rev Pharmacol Toxicol. 2005;45:27–49. doi: 10.1146/annurev.pharmtox.45.120403.100010. [DOI] [PubMed] [Google Scholar]
- 11.Bendaly J, Metry KJ, Doll MA, Jiang G, States JC, Smith NB, Neale JR, Holloman JL, Pierce WM, Hein DW. Role of human CYP1A1 and NAT2 in 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine-induced mutagenicity and DNA adducts. Xenobiotica. 2009;39:399–406. doi: 10.1080/00498250902748953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Josephy PD. The role of peroxidase-catalyzed activation of aromatic amines in breast cancer. Mutagenesis. 1996;11:3–7. doi: 10.1093/mutage/11.1.3. [DOI] [PubMed] [Google Scholar]
- 13.Melchior WB, Jr, Marques MM, Beland FA. Mutations induced by aromatic amine DNA adducts in pBR322. Carcinogenesis. 1994;15:889–899. doi: 10.1093/carcin/15.5.889. [DOI] [PubMed] [Google Scholar]
- 14.Parsons BL, Beland FA, Von Tungeln LS, Delongchamp RR, Fu PP, Heflich RH. Levels of 4-aminobiphenyl-induced somatic H-ras mutation in mouse liver DNA correlate with potential for liver tumor development. Mol Carcinog. 2005;42:193–201. doi: 10.1002/mc.20083. [DOI] [PubMed] [Google Scholar]
- 15.Sim E, Lack N, Wang CJ, Long H, Westwood I, Fullam E, Kawamura A. Arylamine N-acetyltransferases: structural and functional implications of polymorphisms. Toxicology. 2008;254:170–183. doi: 10.1016/j.tox.2008.08.022. [DOI] [PubMed] [Google Scholar]
- 16.Doll MA, Zang Y, Moeller T, Hein DW. Codominant expression of N-acetylation and O-acetylation activities catalyzed by N-acetyltransferase 2 in human hepatocytes. J Pharmacol Exp Ther. 2010;334:540–544. doi: 10.1124/jpet.110.168567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vaziri SA, Hughes NC, Sampson H, Darlington G, Jewett MA, Grant DM. Variation in enzymes of arylamine procarcinogen biotransformation among bladder cancer patients and control subjects. Pharmacogenetics. 2001;11:7–20. doi: 10.1097/00008571-200102000-00002. [DOI] [PubMed] [Google Scholar]
- 18.Millner LM, Doll MA, Cai J, States JC, Hein DW. NATb/NAT1*4 promotes greater arylamine N-acetyltransferase 1 mediated DNA adducts and mutations than NATa/NAT1*4 following exposure to 4-aminobiphenyl. Mol Carcinog. 2011 doi: 10.1002/mc.20836. Epub Aug 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nebert DW, Dalton TP. The role of cytochrome P450 enzymes in endogenous signalling pathways and environmental carcinogenesis. Nat Rev Cancer. 2006;6:947–960. doi: 10.1038/nrc2015. [DOI] [PubMed] [Google Scholar]
- 20.Shi Z, Dragin N, Galvez-Peralta M, Jorge-Nebert LF, Miller ML, Wang B, Nebert DW. Organ-specific roles of CYP1A1 during detoxication of dietary benzo[a]pyrene. Mol Pharmacol. 2010;78:46–57. doi: 10.1124/mol.110.063438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dairou J, Petit E, Ragunathan N, Baeza-Squiban A, Marano F, Dupret JM, Rodrigues-Lima F. Arylamine N-acetyltransferase activity in bronchial epithelial cells and its inhibition by cellular oxidants. Toxicol Appl Pharmacol. 2009;236:366–371. doi: 10.1016/j.taap.2009.02.010. [DOI] [PubMed] [Google Scholar]
- 22.Wakefield L, Boukouvala S, Sim E. Characterisation of CpG methylation in the upstream control region of mouse Nat2: evidence for a gene-environment interaction in a polymorphic gene implicated in folate metabolism. Gene. 2010;452:16–21. doi: 10.1016/j.gene.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 23.Hein DW. N-acetyltransferase SNPs: emerging concepts serve as a paradigm for understanding complexities of personalized medicine. Expert Opin Drug Metab Toxicol. 2009;5:353–366. doi: 10.1517/17425250902877698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nothlings U, Yamamoto JF, Wilkens LR, Murphy SP, Park SY, Henderson BE, Kolonel LN, Le Marchand L. Meat and heterocyclic amine intake, smoking, NAT1 and NAT2 polymorphisms, and colorectal cancer risk in the multiethnic cohort study. Cancer Epidemiol Biomarkers Prev. 2009;18:2098–2106. doi: 10.1158/1055-9965.EPI-08-1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moore LE, Baris DR, Figueroa JD, Garcia-Closas M, Karagas MR, Schwenn MR, Johnson AT, Lubin JH, Hein DW, Dagnall CL, Colt JS, Kida M, Jones MA, Schned AR, Cherala SS, Chanock SJ, Cantor KP, Silverman DT, Rothman N. GSTM1 null and NAT2 slow acetylation genotypes, smoking intensity and bladder cancer risk: results from the New England bladder cancer study and NAT2 meta-analysis. Carcinogenesis. 2011;32:182–189. doi: 10.1093/carcin/bgq223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Johnson KC, Miller AB, Collishaw NE, Palmer JR, Hammond SK, Salmon AG, Cantor KP, Miller MD, Boyd NF, Millar J, Turcotte F. Active smoking and secondhand smoke increase breast cancer risk: the report of the Canadian Expert Panel on Tobacco Smoke and Breast Cancer Risk (2009) Tob Control. 2011;20:e2. doi: 10.1136/tc.2010.035931. [DOI] [PubMed] [Google Scholar]
- 27.Gelatti U, Covolo L, Talamini R, Tagger A, Barbone F, Martelli C, Cremaschini F, Franceschi S, Ribero ML, Garte S, Nardi G, Donadon V, Donato F. N-Acetyltransferase-2, glutathione S-transferase M1 and T1 genetic polymorphisms, cigarette smoking and hepatocellular carcinoma: a case-control study. Int J Cancer. 2005;115:301–306. doi: 10.1002/ijc.20895. [DOI] [PubMed] [Google Scholar]
- 28.Sugamori KS, Wong S, Gaedigk A, Yu V, Abramovici H, Rozmahel R, Grant DM. Generation and functional characterization of arylamine N-acetyltransferase Nat1/Nat2 double-knockout mice. Mol Pharmacol. 2003;64:170–179. doi: 10.1124/mol.64.1.170. [DOI] [PubMed] [Google Scholar]
- 29.Fu PP, Von Tungeln LS, Hammons GJ, McMahon G, Wogan G, Flammang TJ, Kadlubar FF. Metabolic activation capacity of neonatal mice in relation to the neonatal mouse tumorigenicity bioassay. Drug Metab Rev. 2000;32:241–266. doi: 10.1081/dmr-100100575. [DOI] [PubMed] [Google Scholar]
- 30.Von Tungeln LS, Yi P, Bucci TJ, Samokyszyn VM, Chou MW, Kadlubar FF, Fu PP. Tumorigenicity of chloral hydrate, trichloroacetic acid, trichloroethanol, malondialdehyde, 4-hydroxy-2-nonenal, crotonaldehyde, and acrolein in the B6C3F(1) neonatal mouse. Cancer Lett. 2002;185:13–19. doi: 10.1016/s0304-3835(02)00231-8. [DOI] [PubMed] [Google Scholar]
- 31.Kimura S, Kawabe M, Ward JM, Morishima H, Kadlubar FF, Hammons GJ, Fernandez-Salguero P, Gonzalez FJ. CYP1A2 is not the primary enzyme responsible for 4-aminobiphenyl-induced hepatocarcinogenesis in mice. Carcinogenesis. 1999;20:1825–1830. doi: 10.1093/carcin/20.9.1825. [DOI] [PubMed] [Google Scholar]
- 32.Tsuneoka Y, Dalton TP, Miller ML, Clay CD, Shertzer HG, Talaska G, Medvedovic M, Nebert DW. 4-aminobiphenyl-induced liver and urinary bladder DNA adduct formation in Cyp1a2(−/−) and Cyp1a2(+/+) mice. J Natl Cancer Inst. 2003;95:1227–1237. doi: 10.1093/jnci/djg025. [DOI] [PubMed] [Google Scholar]
- 33.Neale JR, Smith NB, Pierce WM, Hein DW. Methods For Aromatic And Heterocyclic Amine Carcinogen-DNA Adduct Analysis By Liquid Chromatography-Tandem Mass Spectrometry. Polycycl Aromat Compd. 2008;28:402–417. doi: 10.1080/10406630802377773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ruggieri A, Barbati C, Malorni W. Cellular and molecular mechanisms involved in hepatocellular carcinoma gender disparity. Int J Cancer. 2010;127:499–504. doi: 10.1002/ijc.25298. [DOI] [PubMed] [Google Scholar]
- 35.Chen T, Mittelstaedt RA, Beland FA, Heflich RH, Moore MM, Parsons BL. 4-Aminobiphenyl induces liver DNA adducts in both neonatal and adult mice but induces liver mutations only in neonatal mice. Int J Cancer. 2005;117:182–187. doi: 10.1002/ijc.21173. [DOI] [PubMed] [Google Scholar]
- 36.Chou HC, Lang NP, Kadlubar FF. Metabolic activation of the N-hydroxy derivative of the carcinogen 4-aminobiphenyl by human tissue sulfotransferases. Carcinogenesis. 1995;16:413–417. doi: 10.1093/carcin/16.2.413. [DOI] [PubMed] [Google Scholar]
- 37.Alnouti Y, Klaassen CD. Tissue distribution and ontogeny of sulfotransferase enzymes in mice. Toxicol Sci. 2006;93:242–255. doi: 10.1093/toxsci/kfl050. [DOI] [PubMed] [Google Scholar]
- 38.Alnouti Y, Klaassen CD. Mechanisms of gender-specific regulation of mouse sulfotransferases (Sults) Xenobiotica. 2011;41:187–197. doi: 10.3109/00498254.2010.535923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Al-Zoughool M, Talaska G. 4-Aminobiphenyl N-glucuronidation by liver microsomes: optimization of the reaction conditions and characterization of the UDP-glucuronosyltransferase isoforms. J Appl Toxicol. 2006;26:524–532. doi: 10.1002/jat.1172. [DOI] [PubMed] [Google Scholar]
- 40.Hatcher JF, Swaminathan S. Detection of deoxyadenosine-4-aminobiphenyl adduct in DNA of human uroepithelial cells treated with N-hydroxy-4-aminobiphenyl following nuclease P1 enrichment and 32P-postlabeling analysis. Carcinogenesis. 1995;16:295–301. doi: 10.1093/carcin/16.2.295. [DOI] [PubMed] [Google Scholar]
- 41.Manjanatha MG, Li EE, Fu PP, Heflich RH. H- and K-ras mutational profiles in chemically induced liver tumors from B6C3F1 and CD-1 mice. J Toxicol Environ Health. 1996;47:195–208. doi: 10.1080/009841096161898. [DOI] [PubMed] [Google Scholar]
- 42.Woo LL, Egner PA, Belanger CL, Wattanawaraporn R, Trudel LJ, Croy RG, Groopman JD, Essigmann JM, Wogan GN. Aflatoxin B1-DNA adduct formation and mutagenicity in livers of neonatal male and female B6C3F1 mice. Toxicol Sci. 2011;122:38–44. doi: 10.1093/toxsci/kfr087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Naugler WE, Sakurai T, Kim S, Maeda S, Kim K, Elsharkawy AM, Karin M. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science. 2007;317:121–124. doi: 10.1126/science.1140485. [DOI] [PubMed] [Google Scholar]
- 44.Bendaly J, Doll MA, Millner LM, Metry KJ, Smith NB, Pierce WM, Hein DW. Differences between human slow N-acetyltransferase 2 alleles in levels of 4-aminobiphenyl-induced DNA adducts and mutations. Mutat Res. 2009;671:13–19. doi: 10.1016/j.mrfmmm.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chow JD, Jones ME, Prelle K, Simpson ER, Boon WC. A selective estrogen receptor α agonist ameliorates hepatic steatosis in the male aromatase knockout mouse. J Endocrinol. 2011;210:323–334. doi: 10.1530/JOE-10-0462. [DOI] [PubMed] [Google Scholar]
- 46.Park EJ, Lee JH, Yu GY, He G, Ali SR, Holzer RG, Osterreicher CH, Takahashi H, Karin M. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell. 2010;140:197–208. doi: 10.1016/j.cell.2009.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Von Tungeln LS, Bucci TJ, Hart RW, Kadlubar FF, Fu PP. Inhibitory effect of caloric restriction on tumorigenicity induced by 4-aminobiphenyl and 2-amino-1-methyl-6-phenylimidazo-[4,5-b]pyridine (PhIP) in the CD1 newborn mouse bioassay. Cancer Lett. 1996;104:133–136. doi: 10.1016/0304-3835(96)04232-2. [DOI] [PubMed] [Google Scholar]
- 48.Cohen JC, Horton JD, Hobbs HH. Human fatty liver disease: old questions and new insights. Science. 2011;332:1519–1523. doi: 10.1126/science.1204265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Adam PJ, Berry J, Loader JA, Tyson KL, Craggs G, Smith P, De Belin J, Steers G, Pezzella F, Sachsenmeir KF, Stamps AC, Herath A, Sim E, O’Hare MJ, Harris AL, Terrett JA. Arylamine N-acetyltransferase-1 is highly expressed in breast cancers and conveys enhanced growth and resistance to etoposide in vitro. Mol Cancer Res. 2003;1:826–835. [PubMed] [Google Scholar]
- 50.Tiang JM, Butcher NJ, Minchin RF. Small molecule inhibition of arylamine N-acetyltransferase Type I inhibits proliferation and invasiveness of MDA-MB-231 breast cancer cells. Biochem Biophys Res Commun. 2010;393:95–100. doi: 10.1016/j.bbrc.2010.01.087. [DOI] [PubMed] [Google Scholar]
- 51.Tiang JM, Butcher NJ, Cullinane C, Humbert PO, Minchin RF. RNAi-mediated knock-down of arylamine N-acetyltransferase-1 expression induces E-cadherin up-regulation and cell-cell contact growth inhibition. PLoS One. 2011;6:e17031. doi: 10.1371/journal.pone.0017031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kawamura A, Westwood I, Wakefield L, Long H, Zhang N, Walters K, Redfield C, Sim E. Mouse N-acetyltransferase type 2, the homologue of human N-acetyltransferase type 1. Biochem Pharmacol. 2008;75:1550–1560. doi: 10.1016/j.bcp.2007.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cao W, Strnatka D, McQueen CA, Hunter RJ, Erickson RP. N-acetyltransferase 2 activity and folate levels. Life Sci. 2010;86:103–106. doi: 10.1016/j.lfs.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ward A, Hickman D, Gordon JW, Sim E. Arylamine N-acetyltransferase in human red blood cells. Biochem Pharmacol. 1992;44:1099–1104. doi: 10.1016/0006-2952(92)90373-q. [DOI] [PubMed] [Google Scholar]
- 55.Mato JM, Martinez-Chantar ML, Lu SC. Methionine metabolism and liver disease. Annu Rev Nutr. 2008;28:273–293. doi: 10.1146/annurev.nutr.28.061807.155438. [DOI] [PubMed] [Google Scholar]
- 56.Chagas CE, Bassoli BK, de Souza CA, Deminice R, Junior AA, Paiva SA, Dagli ML, Ong TP, Moreno FS. Folic acid supplementation during early hepatocarcinogenesis: Cellular and molecular effects. Int J Cancer. 2011;129:2073–2082. doi: 10.1002/ijc.25886. [DOI] [PubMed] [Google Scholar]
- 57.Cabelof DC, Raffoul JJ, Nakamura J, Kapoor D, Abdalla H, Heydari AR. Imbalanced base excision repair in response to folate deficiency is accelerated by polymerase beta haploinsufficiency. J Biol Chem. 2004;279:36504–36513. doi: 10.1074/jbc.M405185200. [DOI] [PubMed] [Google Scholar]