Abstract
Many non-mammalian vertebrates produce hair cells throughout life and recover from hearing and balance deficits through regeneration. In contrast, embryonic production of hair cells declines sharply in mammals where deficits from hair cell losses are typically permanent. Hair cell density estimates recently suggested that the vestibular organs of mice continue to add hair cells after birth, so we undertook comprehensive counting in murine utricles at different ages. The counts show that 51 % of the hair cells in adults arise during the 2 weeks after birth. Immature hair cells are most common near the neonatal macula’s peripheral edge and striola, where anti-Ki-67 labels cycling nuclei in zones that appear to contain niches for supporting-cell-like stem cells. In vivo lineage tracing in a novel reporter mouse where tamoxifen-inducible supporting cell-specific Cre expression switched tdTomato fluorescence to eGFP fluorescence showed that proteolipid-protein-1-expressing supporting cells are an important source of the new hair cells. To assess the contributions of postnatal cell divisions, we gave mice an injection of BrdU or EdU on the day of birth. The labels were restricted to supporting cells 1 day later, but by 12 days, 31 % of the labeled nuclei were in myosin-VIIA-positive hair cells. Thus, hair cell populations in neonatal mouse utricles grow appreciably through two processes: the progressive differentiation of cells generated before birth and the differentiation of new cells arising from divisions of progenitors that progress through S phase soon after birth. Subsequent declines in these processes coincide with maturational changes that appear unique to mammalian supporting cells.
Keywords: ear, hair cell, regeneration, proliferation, vestibular, sensory
Introduction
Mechanoreceptive hair cells are produced throughout life in fish, amphibians, reptiles, and birds, and they can be regenerated from neighboring supporting cells to restore hearing and balance function (Corwin 1981, 1985; Corwin and Cotanche 1988; Jorgensen and Mathiesen 1988; Ryals and Rubel 1988; Popper and Hoxter 1990; Lanford et al. 1996; Warchol 2011). In contrast, regenerative responses in mammalian ears are limited and appear largely ineffective (Forge et al. 1993; Warchol et al. 1993; Brigande and Heller 2009; Kawamoto et al. 2009). Consistent with this, early investigations in rodents reported that the production of hair cells and supporting cells peaks during the second half of embryogenesis and declines precipitously before birth (Ruben 1967; Sans and Chat 1982; Lee et al. 2006). Yet, recent statistical density estimates have suggested that hair cell numbers in mouse utricles continue to increase in the weeks after birth (Rusch et al. 1998; Kirkegaard and Nyengaard 2005).
Those reports attributed the statistical increases to postnatal hair cell differentiation and the direct conversion of supporting cells into hair cells (Rusch et al. 1998; Kirkegaard and Nyengaard 2005). However, we suspected that previously undetected populations of postnatally dividing progenitors might continue to produce progeny that would become new hair cells, thus contributing to the growth of the utricle’s sensory epithelium.
As a first step, we sought to accurately measure how utricular hair cell populations change with age by counting all the hair cells in a series of utricles from embryonic, neonatal, juvenile, and adult mice. The counts showed that numbers equivalent to 51 % of the hair cells in the average adult utricle first appear during the 2 weeks after birth. To trace cell lineages, we generated a novel strain of Cre-Lox reporter mice where tamoxifen administration switches supporting cells that express proteolipid protein 1 (PLP) and all the progeny in their subsequent lineages from red (tdTomato) to green (eGFP) fluorescent protein expression. In the utricles of those mice, the numbers of eGFP-positive hair cells increased with increasing time after a tamoxifen injection, indicating that PLP-expressing cells are an important source of new hair cells.
To test the hypothesis that postnatal proliferation contributes to growth in the utricle’s hair cell population, we gave mice the thymidine analogs, 5-bromo-2-deoxyuridine (BrdU) and 5-ethynyl-2′-deoxyuridine (EdU), in vivo. Analysis of the utricles from those mice showed that substantial numbers of cells enter S phase of the cell cycle soon after birth, with over a quarter of the progeny from the postnatal cell divisions eventually differentiating as hair cells. Other DNA labeling results showed that S phase entry in the macula declines exponentially through the second day after birth. Cell production and hair cell differentiation both occur most often at the postnatal macula's peripheral edge, with their greatest incidence at its lateral edge. Those processes occur with a somewhat lower incidence in another growth zone near the medial striola. In those regions, basal nuclei in supporting cells or supporting-cell-like progenitors label strongly for Ki-67, a marker of actively cycling cells. Thus, for a short period after birth, the mouse utricle’s hair cell population grows via two processes: the postnatal proliferation of supporting cells or supporting-cell-like progenitors and the differentiation of the progeny produced from the divisions of those cells and other progeny produced from progenitors that last replicated their DNA before birth, with both of these processes occurring most often in a peripheral growth zone (PGZ) and somewhat less frequently in a striolar growth zone (SGZ).
Materials and methods
Animals and dissection of utricles
All animal experiments were performed according to protocols approved by the Animal Care and Use Committee of the University of Virginia. B6.129(Cg)-Gt(ROSA)26Sortm4(ACTB-tdTomato,-EGFP)Luo/J mice (Muzumdar et al. 2007) were from the Jackson Laboratory (stock no. 007676; Bar Harbor, Maine). The B6.129(Cg)-Gt(ROSA)26Sortm4(ACTB-tdTomato,-EGFP)Luo/J mice are referred to as membrane tdTomato/membrane eGFP mice (mT/mG mice) in this report. B6.Cg-Tg(Plp1-cre/ERT)3Pop/J mice (Doerflinger et al. 2003) were obtained from The Jackson Laboratory (stock no. 005975). B6.Cg-Tg(Plp1-cre/ERT)3Pop/J are referred to as PLP-Cre mice. Swiss Webster mice were obtained from Charles River (Wilmington, MA). Math1/nGFP mice were obtained from J. Johnson at the University of Texas Southwestern Medical Center (Lumpkin et al. 2003).
To perform lineage tracing, we injected tamoxifen (1.5 mg/40 g body weight, i.p.; Invitrogen, Carlsbad, CA) into mT/mG;PLP-Cre pups once a day on postnatal day 0 (P0) and P1, euthanizing and fixing their ears at P2, P4, P9, or P13. To label proliferating cells and their progeny, BrdU was injected into Swiss Webster pups (50 mg/ kg body weight, s.c.; Sigma-Aldrich, St. Louis, MO) at 12-h intervals for 3 days, and pups were euthanized on the morning of the fourth day and their utricles fixed. Age intervals for the 12-h BrdU injections were P0–P2, P3–P5, P6–P8, P9–P11, and P12–P14, with the mice euthanized and utricles fixed 12 h later. Other pups received EdU (50 mg/ kg body weight, s.c. injection; Invitrogen) once a day at P0, P1, or P2 and were euthanized and fixed 24 h later at P1, P2, or P3, respectively. To trace the lineages and fates of cells that entered S phase on P0, EdU was injected into additional pups one time on P0 (50 mg/ kg body weight, s.c. injection). These mice were euthanized and their utricles fixed at P5, P8, P12, or P18.
Pups were anesthetized on ice and then decapitated. Juvenile and adult mice were killed by carbon dioxide asphyxiation. Labyrinths were dissected from temporal bones in ice-cold DMEM/F-12 (Invitrogen), the utricles were isolated, and the roof and the otoconia were dissected away. Vestibular organs were fixed in fresh 4 % paraformaldehyde in phosphate-buffered saline (PBS) or Shandon Glyo-Fixx (Thermo Scientific, Waltham, MA) either for 15 min at room temperature (RT) or overnight at 4 °C. For culture experiments, utricles from mT/mG mice were adhered to glass bottom dishes (Mat-Tek, Ashland, MA) coated with Cell-Tak (BD Biosciences, San Jose, CA) and cultured in DMEM/F12 with 5 % FBS (Invitrogen), 1.5 μM tamoxifen, 0.25 μg/mL Fungizone (Invitrogen), and 10 μg/mL ciprofloxacin (Bayer, Berlin, Germany) at 37 °C and 5 % CO2. Both right and left utricles were used in this study. In the figures, images of right utricles shown have been horizontally rotated, so all appear in the orientation of left utricles.
Immunocytochemistry
Rabbit anti-myosin VIIA (1:200, cat. no. 25-6790; Proteus Biosciences, Ramona, CA) and mouse anti-alpha II spectrin (1:50, cat. no. MAB1622; Millipore, Temecula, CA) were used to label hair cell somata and cuticular plates, respectively. Mouse anti-protocadherin-15-CD2 (1:200; generous gift from Drs. Thomas B. Friedman, NIDCD; Inna Belyantseva, NIDCD; and Zubair Ahmed, Cincinnati Children’s Hospital Medical Center) and rabbit anti-espin (1:10; generous gift from Dr. James Bartles, Northwestern University) were used to label stereocilia. Mouse anti-BrdU (1:50, cat. no. 347580; BD Biosciences) was used to label cells in lineages that had entered S phase. Rabbit anti-Ki-67 (1:200, cat. no. RM-9106-S0; Lab Vision, Kalamazoo, MI) was used to label cells in the active late G1, S, G2, and M phases of the cell cycle. Rabbit anti-GFP (1:200, cat. no. A-6455; Invitrogen) was used to label GFP in utricles from Math1/nGFP transgenic mice. Mouse anti-E-cadherin (1:200, cat. no. 610181; BD Biosciences) was used to label intercellular junctions in the epithelia.
Fixed utricles were washed in PBS and then pre-incubated for 1 h at RT in blocking solution: PBS containing 0.2 % Triton X-100 (PBS-T) and 10 % normal goat serum (NGS; Vector Laboratories, Burlingame, CA). Samples to be labeled with anti-BrdU were incubated in DNase I (0.5 Kunitz units/μL; Sigma) at 37 °C for 1 h before incubation in the blocking solution. For the detection of EdU, utricles were rinsed in PBS after the blocking step and a copper-catalyzed Click-iT reaction performed using a kit as per the manufacturer’s instructions (Invitrogen). Samples were next incubated overnight in the appropriate primary antibodies in PBS-T with 2 % NGS, followed by three rinses in PBS-T and incubation with Alexa-conjugated secondary antibodies (1:200, Invitrogen) and/or phalloidin (5 U/mL, Invitrogen) in PBS-T for 2 h at RT. Then, utricles were rinsed in PBS three times and mounted in SlowFade (Invitrogen) for imaging using Zeiss LSM 510 or 700 confocal microscopes.
To localize protocadherin-15-CD2, utricles were fixed in Shandon Glyo-Fixx, washed in PBS, and pre-incubated in the blocking solution for 30 min at RT. They were then rinsed three times in PBS and incubated with the anti-protocadherin-15-CD2 in PBS with 2 % NGS overnight. After three rinses in PBS, they were incubated with Alexa-conjugated secondary antibodies (1:200) and phalloidin (5 U/mL) in PBS for 2 h at RT. Then, they were rinsed in PBS three times, mounted, and imaged as described above.
Quantification of cellular numbers, macular areas, and spatial cell densities
Mouse utricles were fixed at embryonic day 18.5 (E18.5), P0, P2, P4, P8, P12, P16, P52, and P80. In one utricle per mouse and four mice per age, all of the hair cells were counted manually using the Cell Counter plug-in of ImageJ (U.S. National Institutes of Health, Bethesda, MD). Only the cells that were labeled by both anti-myosin VIIA and anti-alpha II spectrin were counted as hair cells. None contained spectrin-positive cuticular plates in the absence of myosin VIIA labeling, and <1 % of the hair cells showed positive labeling for myosin VIIA without detectable antibody labeling of spectrin protein in the developing hair cell’s cuticular plate. Most of these myosin-VIIA-positive, alpha-II-spectrin-negative hair cells had small apical surfaces and small hair bundles that were stained by phalloidin labeling of F-actin. Since we suspected that such cells were not far along in the process of developing as hair cells, and because <1 % of the potential hair cells exhibited just the myosin VIIA labeling, our criteria allowed only the doubly labeled spectrin-positive, myosin-VIIA-positive cells to be included in the total hair cell counts used in the quantitative analyses.
The area of the macula in each utricle was measured from confocal images using ImageJ to trace the outline of the macula’s outer boundary. The spatial densities of hair cells were calculated by dividing the mean number of hair cells by the mean macular area for utricles from each age group.
Quantification of S phase entry and mitotic hair cell production
In all applicable experiments, the BrdU- or EdU-labeled nuclei were counted manually using the Cell Counter plug-in in ImageJ. EdU-positive/myosin-VIIA-positive hair cells were counted separately. For each DNA-labeled specimen, a Z-stack of confocal images of the utricle was acquired using a 20x/0.75-NA objective (slice thickness = 0.88 μm, satisfying the Nyquist criterion). The Z-axis sampling when combined with the phalloidin and/or anti-E-cadherin labeling, allowed the cells in the sensory epithelium to be reliably distinguished from cells in the underlying stroma, ensuring that no labeled cells in the stroma were included in the counts.
ImageJ was used to trace the border between the sensory epithelium and the surrounding non-sensory epithelium and for recording the x–y image coordinates of the counted nuclei and their positions in relation to the macula's lateral edge. Using a custom program written in Matlab R2009b (Mathworks, Natick, MA), the minimum distance between each labeled cell and the lateral edge was calculated using those coordinates and the Euclidean distance formula. To create the dot plots in Figures 6 and 8, images were overlaid from each of the utricles in which the S-phase-labeled cells had been counted. Each of the images was aligned with the macular outlines of the other S-phase-labeled utricles. Then, the translation and rotation values that were used to achieve the alignment of macular outlines were used to identically translate and rotate the images generated from the Cell Counter plug-in that contained a dot at the location of each labeled nucleus. Finally, the aligned dot images were overlaid on a diagram of the macula showing the hair bundle reversal line and a shaded area representing the striola. The position of the reversal line shown on the diagram was estimated from the measurements in Figure 3; the width of the striola was derived from published measurements (Desai et al. 2005; Li et al. 2008).
FIG. 6.
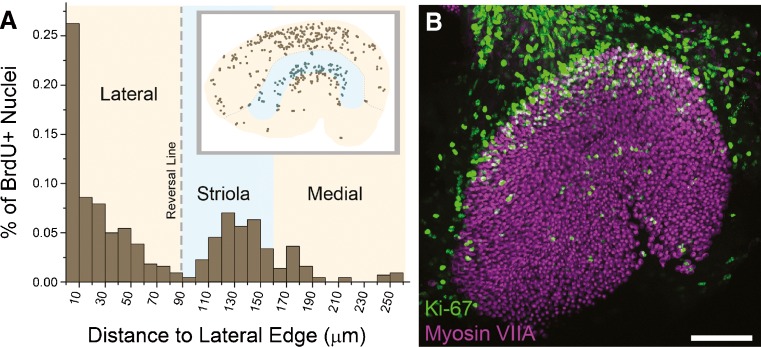
BrdU-positive nuclei are most common at the lateral edge and the medial striola in utricles from neonatal mice. A Histogram of the percentage of BrdU-positive nuclei in 10-μm intervals along the lateral–medial axis shows two spatially separate populations of proliferating cells in the macula of the neonatal mouse utricle. The largest population is at the lateral edge, and a smaller population is centered near the medial edge of the striola. The inset is a dot-plot showing pooled locations of BrdU-positive nuclei from P3 mouse utricles overlaid on a diagram of the macula (tan), with a dashed line marking the reversal line and the striola marked in light blue shading (n = 4). B Confocal image of a utricle from a P0 mouse labeled with antibodies to myosin VIIA and Ki-67. The pattern of Ki-67-positive cells is similar to the BrdU labeling pattern, although more cells are labeled. Scale bar, 100 μm.
FIG. 8.
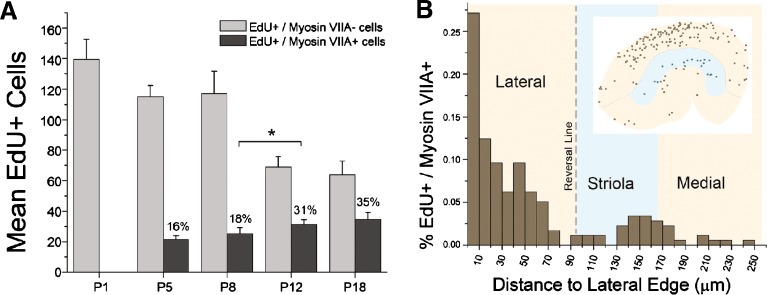
Temporal and spatial analyses show that significant numbers of hair cells differentiate from EdU-labeled cells or their progeny near the lateral edge of the utricle during the 2 weeks after birth. A Graph showing the absence of EdU-positive/myosin-VIIA-positive cells in EdU-labeled utricles harvested at P1 and the progressive increase in the percentage of EdU-positive cells (from 16 to 35 %) that became myosin-VIIa-positive in utricles harvested from mice at P5–P18, following EdU injection on P0. A significant increase in the percentage of EdU-positive/myosin-VIIA-positive cells occurred from P8 to P12 (*p < 0.05, one-way ANOVA with Bonferonni’s multiple comparisons test). Data are expressed as the mean ± SEM. B Histogram showing the percentage of EdU-positive/myosin-VIIA-positive hair cells in 10-μm intervals along the lateral–medial axis of utricles from P8 mice that received one EdU injection at P0. Dot-plot (inset) showing the pooled locations of newly produced (EdU-positive/myosin-VIIA-positive) hair cells overlaid on a diagram of the P8 macula (tan shading), with a dashed line marking the reversal line and the striola marked by light blue shading, as in the dot-plot of BrdU-labeled cells in Fig. 6 (n = 7 utricles). The postnatal production of hair cells occurs most frequently in a growth zone at the macula's lateral edge and another near its medial striola.
FIG. 3.
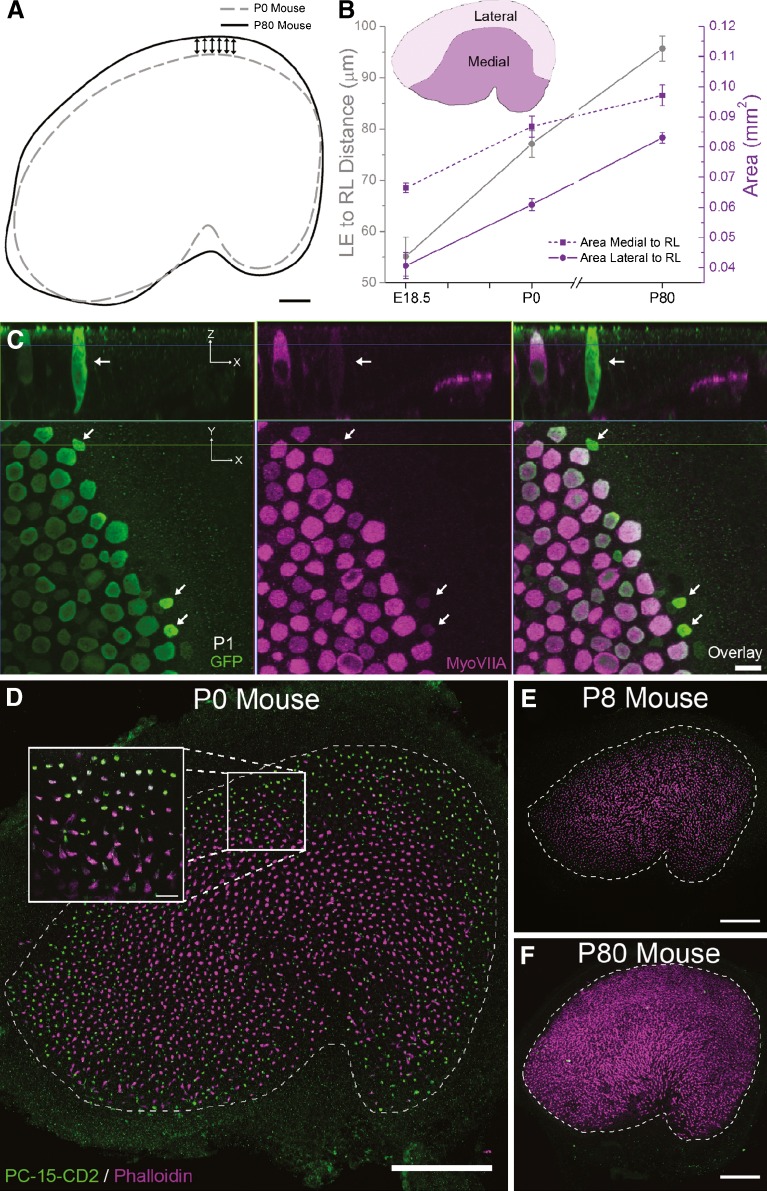
In neonatal mouse utricles, the macula expands along its peripheral edge and most noticeably at its lateral edge, a region containing many new hair cells. A The diagram illustrating the results of Fourier shape descriptor analysis for maculae from P0 and P80 mice suggests that the macula expands along its peripheral edge as mice mature, but with the most significant expansion occurring at the lateral edge (arrows). In these overlays, the lateral edge expands 30.5 ± 1 μm, n = 6 utricles per age. Scale bar, 50 μm. B The area of the utricle that is lateral to the reversal line expands substantially between birth and maturity. The graph shows area measurements of the regions of the macula lateral (solid purple line) and medial (dashed purple line) to the reversal line in E18.5, P0, and P80 mice. The representative macula in the inset shows the location of these two regions. The gray line on the graph shows the average distance between the lateral edge (LE) and reversal line (RL) at the same ages. Spectrin labeling was used to identify the line of reversal and the lateral edge (n = 6 utricles per age). Data are expressed as the mean ± SEM. C Confocal images show that GFP-expressing, Atoh1-positive, myosin VIIA-negative differentiating hair cells are present at the lateral edge of a utricle from a P1 Math1/nGFP transgenic mouse. Top x–z views parallel to the long axis of the hair cells. Arrow marks an Atoh1-GFP-positive/myosin VIIA-negative cell that appears to be in contact with the basal lamina. Bottom x–y view showing the apical surfaces of hair cells. Arrows mark three Atoh1-GFP-positive (green) cells that express considerably lower levels of myosin VIIA than the neighboring hair cells (purple). Scale bar, 10 μm. D Confocal image of a P0 mouse utricle labeled with phalloidin (purple) and an antibody to protocadherin-15-CD2 (PC-15-CD2, green), a marker for immature hair bundles. Scale bar, 100 μm. Zoomed inset shows the predominance of PC-15-CD2-labeled hair bundles at the macula's lateral edge. Scale bar, 10 μm. In utricles from P8 mice, fewer hair bundles are labeled with antibodies to protocadherin-15-CD2 (E) and no hair bundles are labeled in utricles from adult mice (F). Scale bars for (E, F), 100 μm.
Measurements of macular expansion
ImageJ was also used to record the pixel coordinates of lines drawn along the macula's entire boundary, lateral edge, and reversal line in the images of utricles labeled with spectrin antibodies. The pixel coordinates of the macula’s boundary and the reversal line were used within ImageJ to calculate the areas of the macular regions that were lateral and medial to the reversal line. To calculate the distance from the lateral edge to the reversal line, the pixel coordinates were inserted into a custom Matlab program, which calculated the Euclidean distance from each pixel coordinate on the lateral edge line to the nearest pixel coordinate on the reversal line.
Morphometric analysis
Fourier shape descriptor analysis was used to calculate and plot the average outlines of the macula from newborn mice (at P0) and adults (at P80). For this, ImageJ was used to trace the macular boundary in the images of utricles labeled with antibodies to myosin VIIA and spectrin. Spectrin labeling of the cuticular plates showed where hair bundle polarity reversed. To eliminate discrepancies in rotation and translation, the outlines for the maculae from six P0 and six P80 mice were overlaid using the reversal line as a fiduciary. Then, the Fourier plug-in (written by Thomas Boudier and Ben Tupper) in ImageJ was used to compute the first ten coefficients of the Fourier expansion series that best fit each outline. The coefficients generated from each outline were averaged and reinserted into the Fourier series using the “rEfourier” function (written by David Thomas) in Matlab to obtain the average macular outline for each age. Finally, the average outlines for the two ages were overlaid just posterior to the hilus, where the macula's medial edge is slightly indented. In P0 mice, the hilus region exhibited minimal protocadherin-15-CD2 labeling and, for the purpose of the morphometric analysis, was therefore assumed to vary less with age and postnatal growth.
Statistics
All statistical analyses were performed using OriginPro 7.5 or GraphPad Prism 5.0. To calculate nonlinear least squares fits, OriginPro uses Levenberg–Marquardt chi-squared minimization with automatic parameter initialization. To fit equations to the data, 200 iterations of this minimization routine were performed. Confidence intervals and coefficients of determination were computed using built-in functions in the software. For statistical comparisons, Prism was used to conduct ANOVAs or a two-way ANOVA followed by Bonferonni’s multiple comparisons tests (α level = 0.05 in all cases). All descriptive statistics are presented as the mean ± SEM.
Results
Half of the total hair cells in the murine utricle first appear after birth
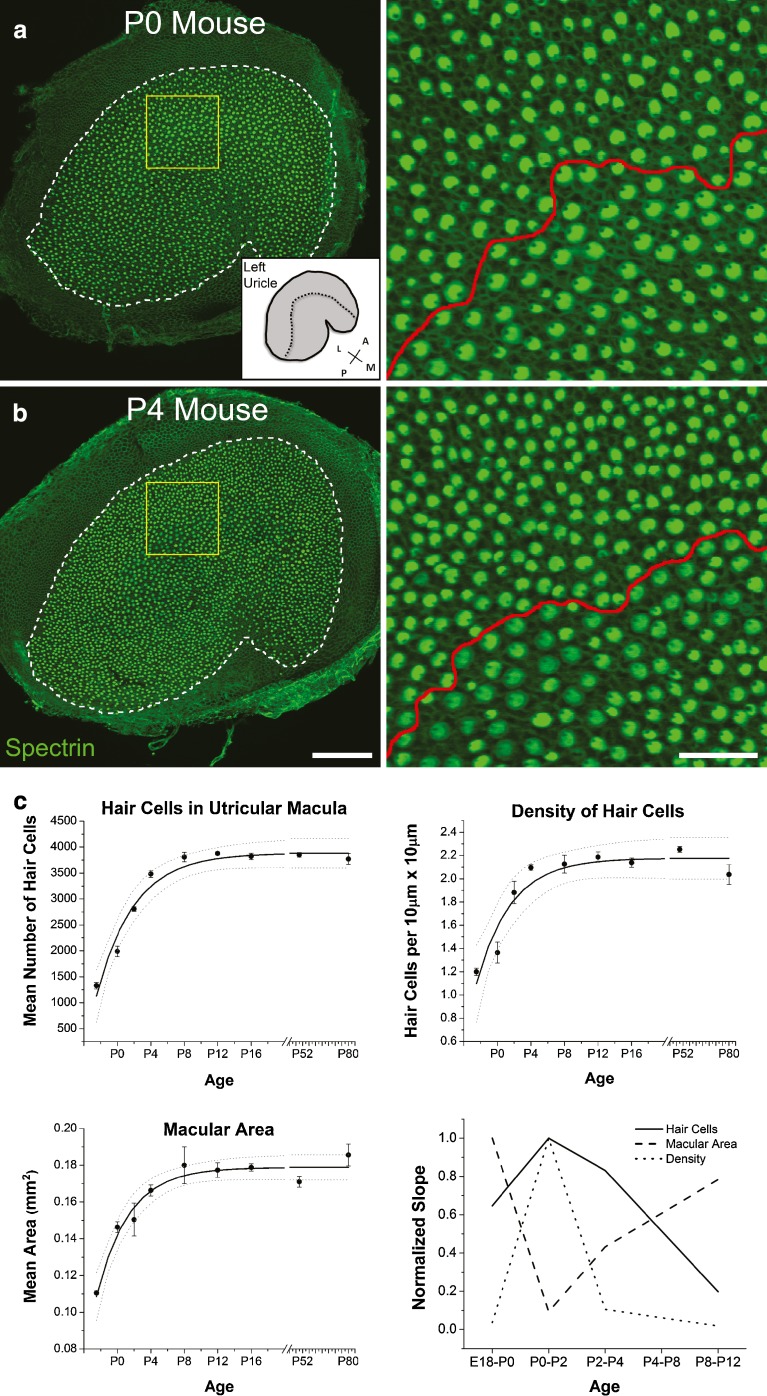
To accurately assess the magnitude and time course of an apparent increase in the number of hair cells in the mouse utricle, we fixed utricles from embryonic day 18.5 (E18.5), postnatal day 0 (P0), P2, P4, P8, P12, P16, P50, and P80 mice (n = 4 per age) and immunostained them with antibodies to myosin VIIA and alpha II spectrin, which label the hair cell soma and cuticular plate, respectively (Fig. 1; Hasson et al. 1997; Deans et al. 2007; Jensen-Smith and Hallworth 2007). Using confocal images of those whole-mount utricles, we counted all the hair cells that were positive for both labels. The counts show that between birth and P12, the utricle's hair cell population grows by an average of 1,888 ± 118 hair cells, which is equivalent to 51 % of the mean total hair cells present in the mature mouse utricle (Fig. 1 and Table 1). The hair cell counts approached a plateau with an average that was similar to numbers that have been reported in other studies of the mouse utricle (Fig. 1 and Table 1; Desai et al. 2005; Kirkegaard and Nyengaard 2005; Li et al. 2008).
FIG. 1.
In the utricles of mice, 51 % of the hair cells first appear after birth. A, BLeft Confocal images show utricles from newborn (P0) (A) and 4-day-old (P4) (B) mice labeled with antibodies to spectrin (green). Spectrin labeling facilitates the identification of individual hair cells when imaged with a low magnification, high numerical aperture objective (image taken with ×20/0.75-NA objective). Scale bar, 100 μm. Inset diagram (bottom right in A) shows the macula and its anatomical orientation (A anterior, M medial, P posterior, L lateral; a dashed line marks the line of hair bundle polarity reversal). Right Zoomed regions within the yellow boxes on the left show hair cell cuticular plates individually resolved for counting. A red line marks the line of polarity reversal. Note the presence of hair cells with small cuticular plates distributed throughout the images from the P0 and P4 utricles and the higher spatial density of hair cells in the P4 utricle. Scale bar, 25 μm. C Graphs show hair cell numbers, hair cell density, macular area, and rates of change in those measures versus mouse age. Solid lines indicate von Bertalanffy equation fits; dashed lines indicate 95 % confidence intervals. Data are expressed as the mean ± SEM, n = 4 utricles per data point.
TABLE 1.
Data values from the graphs in Figure 1C which show the mean number of hair cells in the utricular macula, the mean macular area, and the mean hair cell density (n = 4 utricles from different mice)
| Age | Hair cells | Macular area (mm2) | Hair cell density (hair cells/10 μm2) |
|---|---|---|---|
| E18.5 | 1,329 ± 64 | 0.111 ± 0.002 | 1.20 ± 0.03 |
| P0 | 1,988 ± 101 | 0.146 ± 0.003 | 1.36 ± 0.09 |
| P2 | 2,805 ± 44 | 0.150 ± 0.009 | 1.88 ± 0.10 |
| P4 | 3,484 ± 68 | 0.166 ± 0.003 | 2.10 ± 0.03 |
| P8 | 3,806 ± 91 | 0.180 ± 0.010 | 2.13 ± 0.07 |
| P12 | 3,876 ± 28 | 0.177 ± 0.004 | 2.19 ± 0.04 |
| P16 | 3,820 ± 61 | 0.179 ± 0.002 | 2.14 ± 0.04 |
| P50 | 3,849 ± 46 | 0.171 ± 0.003 | 2.25 ± 0.03 |
| P80 | 3,770 ± 106 | 0.186 ± 0.006 | 2.04 ± 0.09 |
Postembryonic hair cell addition in the ray has been modeled as a von Bertalannfy growth function (Bertalanffy 1934; Corwin 1983). The number of hair cells counted across ages in mouse utricles here also were fit to a von Bertalannfy growth function with r2 = 0.96 (Fig. 1C and Table 2), showing that the hair cell population increases logarithmically between P0 and P12. The forward difference formula was used to estimate the time rate of change for hair cell numbers, and the plots of the estimated derivative revealed that the most rapid postnatal production of hair cells occurs between birth and P2 (Fig. 1C).
TABLE 2.
Coefficients from von Bertalannfy curve fits in Figure 1C
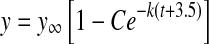 |
|||
|---|---|---|---|
| y∞ | C | k | |
| Hair cells | 3,909 ± 115 | 0.87 ± 0.08 | 0.22 ± 0.04 |
| Macular area | 0.180 ± 0.003 | 0.49 ± 0.05 | 0.25 ± 0.05 |
| Hair cell density | 2.18 ± 0.07 | 0.61 ± 0.10 | 0.24 ± 0.07 |
y is the number of hair cells (i.e., the dependent variable), y∞ is the number of hair cells at infinity, C is the scaling parameter, k is the curvature parameter, and t is the age in days (i.e., the independent variable); −3.5 represents the first time point (E18.5)
The macular area expands as the spatial density of hair cells increases
In the utricles where we counted hair cells, we measured the macular area and found that a 1.23-fold mean increase occurs between P0 and P8 (P0 = 0.146 ± 0.003 mm2, P8 = 0.180 ± 0.010 mm2; Fig. 1C and Table 1). After P8, appreciable increases were no longer detected. The increase in macular area was fit by a von Bertalannfy growth curve with r2 = 0.96 (Fig. 1C and Table 2), indicating that the macula expands logarithmically as mice mature during the first 8 days of postnatal life. The timing of the change in macular area differed, however, from that for hair cell addition, with the most rapid expansion in area occurring between P4 and P8 (Fig. 1C). This suggests that the growth in macular area results not only from the addition of new hair cells but also from growth in cell size and the possible addition of new supporting cells.
In the ears of some non-mammalian vertebrates, postnatal growth of the otic sensory epithelia occurs primarily through an appositional addition of new hair cells that accumulate in a growth zone at the epithelium's peripheral edge, in a manner analogous to the growth of tree trunks. In the ears of other non-mammals, interstitial addition is prevalent, with new hair cells arising throughout the macula in a scattered pattern (Corwin 1981, 1983, 1985; Popper and Hoxter 1984). To determine whether postnatal addition in the mouse utricle occurs though apposition, interstitial addition, or a combination, we divided the total hair cells by the macular area measured at each age. The values show that the spatial density of hair cells increases logarithmically from P0 to P12 (r2 = 0.90; Fig. 1C and Tables 1 and 2), with the rate of density change mirroring the increase in hair cell number (Fig. 1C). If new hair cells were added only at the macula’s peripheral edge, then, spatial density would have remained constant or decreased, so we conclude that the growth occurs through a combination of interstitial and appositional addition of new hair cells.
PLP-expressing supporting cells are an important source of new hair cells
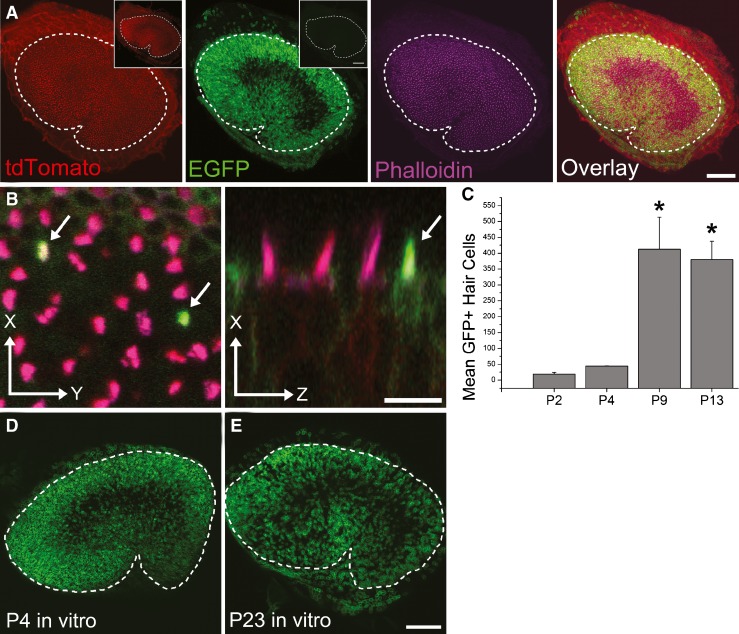
To trace cell lineages, we generated a novel mouse model. In mT/mG mice, membrane-targeted tdTomato encoded by a floxed allele is under control of the chicken beta-actin promoter, producing strong red fluorescence in all cell types (Muzumdar et al. 2007). A membrane-targeted eGFP sequence follows the second of the loxP sites that bracket the tdTomato sequence. To create a line of mice that could be used for lineage tracing in the vestibular epithelia, we crossed the mT/mG mice with a tamoxifen-inducible PLP-Cre strain in which Cre recombinase is expressed under control of the PLP promoter in a large subset of the ear’s supporting cells. Tamoxifen-induced, cell-specific expression of Cre recombinase leads to excision of the tdTomato sequence and expression of eGFP, resulting in a switch from strong red fluorescence to green fluorescence at the membranes of the PLP-Cre-expressing cells and cells that arise subsequently in their lineage.
In the utricular sensory epithelium of PLP-Cre mice, Cre expression has been shown to be restricted to supporting cells (Gomez-Casati et al. 2010). Prior to tamoxifen injection, all the cells in the utricular sensory epithelium of mT/mG;PLP-Cre mice exhibited bright red membrane fluorescence and no green fluorescence (Fig. 2A, insets). Giving the mT/mG;PLP-Cre mice an injection of tamoxifen on P0 and another on P1 resulted in the appearance of a mosaic pattern of red fluorescent cells and green fluorescent cells in the utricles of mice that were killed at P2 (Fig. 2A). As expected, the majority of the eGFP-expressing cells were supporting cells that extended from the epithelium’s apical surface to the basal lamina. However, eGFP-expressing hair cells were observed in those specimens (Fig. 2B), indicating that some PLP-expressing supporting cells or progeny had converted into hair cells between the initial tamoxifen injection at P0 and tissue fixation at P2.
FIG. 2.
Lineage tracing in mice where tamoxifen injections switch PLP-expressing supporting cells from expressing tdTomato (red) to expressing eGFP (green) indicates that new hair cells arise from supporting cells. A Confocal images of a utricle from a P4 mT/mG;PLP-Cre mouse that received tamoxifen at P0 and P1. PLP does not appear to be expressed in the supporting cells within the striola. Hair bundles are labeled with fluorescent phalloidin (purple). The insets show low-magnification confocal images from a utricle of a P4 mT/mG;PLP-Cre mouse that did not receive tamoxifen. Note that there is no eGFP expression in the inset image on the right. Scale bars, 100 μm. B Zoomed confocal images of the utricle in (A). Left x–y view at the level of the hair cell bundles. All hair bundles are labeled by fluorescent phalloidin (purple), and most appear magenta because those hair cells also express tdTomato (red). Arrows point to two hair cells that express eGFP in their bundles, which indicates that each arose from a PLP-expressing supporting cell or its lineage. Right x–z view parallel to the long axis of the hair cells. Arrow indicates an eGFP-positive hair cell. Scale bar, 10 μm. C Graph of the mean number of eGFP-positive hair bundles in utricles from mT/mG;PLP-Cre mice that were fixed at various ages after they all received tamoxifen at P0 and P1. Asterisks indicate a significant difference compared to the P2 and P4 time points (p < 0.05, one-way ANOVA with Bonferonni’s multiple comparisons test). D, E Confocal images of utricles from P4 (D) and P23 (E) mT/mG;PLP-Cre mice that were harvested and cultured in the presence of 1.5 μM tamoxifen for 4 days. Utricles were fixed and eGFP (green) expression was imaged without antibody enhancement. Scale bar, 100 μm.
eGFP expression was largely absent from the striolar region in utricles from mT/mG;PLP-Cre mice that received tamoxifen at P0 and P1 (Fig. 2A), and when tamoxifen was administered at older ages, the amount of eGFP expression in the extrastriolar supporting cells appeared to decline (data not shown). To rule out the possibility that differences in tamoxifen’s access to the striola and extrastriola regions might have accounted for the in vivo results, we treated other utricles with tamoxifen in vitro. Those results confirmed the differential striola–extrastriola eGFP expression patterns of eGFP (Fig. 2D, E). Our results suggest that the PLP promoter is normally inactive in most of the supporting cells that reside within the striola of the mouse utricle and that the incidence of PLP-expressing supporting cells in the extrastriolar regions declines with age.
By fixing utricles after 1, 3, 8, and 12 days and counting the number of eGFP-positive hair bundles that were also labeled with fluorescent phalloidin, we tracked the increase in the eGFP-positive hair cells (P0/P1 tamoxifen induction, n = 2–4 utricles per time point). Between P2 and P4, eGFP-positive hair bundles increased 2.3-fold, and by 9.3-fold between P4 and P9 (p<0.05), but no significant increase occurred between P9 and P13 (p > 0.05, one-way ANOVA with Bonferonni’s multiple comparisons test; Fig. 2C). Thus, PLP-expressing supporting cells or supporting-cell-like stem cells appear to be an important source of the new hair cells that arise in neonatal mouse utricles.
The macula expands at its lateral edge
During embryonic development, the murine utricle expands at its anterior, posterior, and medial edges (Denman-Johnson and Forge 1999). To determine where the postnatal expansion occurs, we traced the outline of the macula in myosin-VIIA-immunolabeled utricles from P0 and P80 mice. Then, using Fourier shape descriptor analysis, we calculated and plotted the average shape for the macula at the two ages sampled (n = 6). Overlays of the average outlines suggest that postnatal growth occurs throughout the sensory epithelium's lateral edge, with the location of that edge moving 30.5 ± 1.0 μm outward between P0 and P80 (Fig. 3A). Expansion also appeared to occur at the posterior and medial edges, particularly in the hilar region, but the expansion along those edges did not appear as consistent as the expansion at the lateral edge.
Confocal images of spectrin-labeled utricles allowed clear identification of hair cell polarity directions and localization of the reversal line (Figs. 1A, B and 3B, inset). Between E18.5 and P0, the macular area medial to the reversal line increased 31 %, but expansion there slowed after birth (12 % increase between P0 and P80, n = 4–6; dashed purple line in Fig. 3B). In comparison, the area of the macula lateral to the reversal line showed greater percent expansions in the days just prior to birth as well as after birth (a 50 % increase between E18.5 and P0 and a 37 % increase between P0 and P80, n = 4–6; solid purple line in Fig. 3B). For quantitative assessments, we focused on the outward expansion along the lateral–medial axis by measuring the distance from the macula's reversal line to its lateral edge (Fig. 1A, B). In a study of the embryonic growth that occurs between E14.5 and E18.5, that distance has been found to remain relatively constant at ∼50 μm (Denman-Johnson and Forge 1999), and our measurements showed a comparable mean of 55.1 ± 7.6 μm in E18.5 Swiss Webster mice (n = 4, gray line in Fig. 3B). In addition, our measurements show that the lateral edge to reversal line distance also increases 40 % between E18.5 and P0 (mean distance at P0 = 77.1 ± 6.4 μm, n = 6; Fig. 3B) and by an additional 24 % between P0 and P80 (mean distance at P80 = 95.7 ± 6.1 μm, n = 6; Fig. 3B). At that point, the distance we measured was similar to adult values reported previously (Desai et al. 2005; Li et al. 2008). Taken together, the data suggest that embryonic and postnatal patterns of macular expansion differ, with outward expansion at the lateral edge predominant after birth and lesser expansion occurring at the posterior and medial edges.
New hair cells are found most often near the peripheral edge of the macula
To pinpoint individual new hair cells in the postnatal epithelia, we analyzed the spatiotemporal expression patterns of hair cell differentiation markers. The basic helix-loop-helix transcription factor, Atoh1 (formerly Math1), is the earliest protein known to be expressed specifically in differentiating hair cells (Bermingham et al. 1999; Zheng and Gao 2000). To map the locations of differentiating hair cells, we used an antibody against myosin VIIA and used anti-GFP to enhance detection in utricles from mice that carry an Atoh1/nGFP transgene. The onset of Atoh1 expression in hair cells precedes the expression of myosin VIIA. Cells that were strongly positive for both GFP and myosin VIIA were present throughout the sensory epithelium, but GFP-positive cells that labeled weakly or not at all with anti-myosin VIIA were concentrated near the lateral edge, where many were the outermost hair cell and some had not yet lost their basal process (Fig. 3C, arrow in x–z view). Similar cells also were observed in utricles from Atoh1/nGFP mice that were not immunostained with the antibody to GFP.
A cytoplasmic domain splice variant of protocadherin-15, protocadherin-15-CD2, is expressed as hair cells develop immature hair bundles and is downregulated when hair cells become more mature (Ahmed et al. 2006). Anti-protocadherin-15-CD2 brightly labeled the majority of hair bundles in a band at the peripheral edge of the P0 macula and labeled some hair bundles that were scattered more widely through the central regions of the macula (Fig. 3D). At P8, that antibody labeled fewer bundles than at P0, and it labeled none at P80 (Fig. 3E, F). These immune labeling results are in agreement with results from the morphometric analyses in identifying the macula’s peripheral edge and, in particular, the lateral edge as the predominant sites of postnatal hair cell addition and macular expansion.
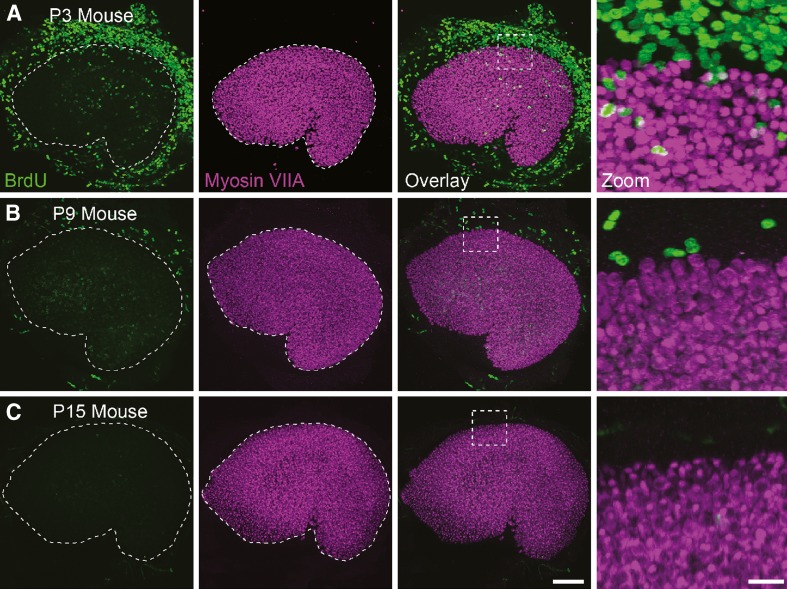
BrdU labels substantial numbers of cells in the postnatal utricular macula
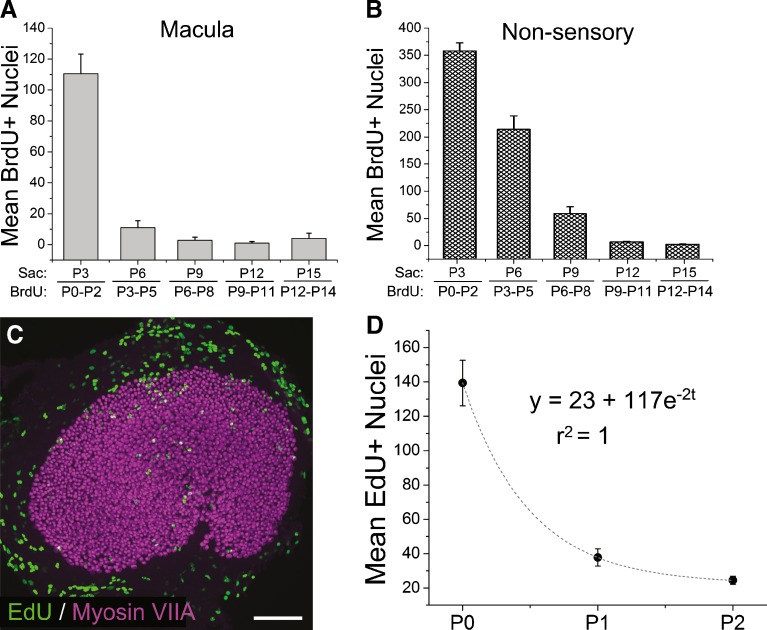
To determine whether some or all of the new hair cells arose from cells that replicated their DNA and divided after birth, we gave mice subcutaneous injections of BrdU at 12-h intervals from P0 through P2 and fixed their utricles at P3. The sensory epithelium in these utricles contained on average 114 ± 13 BrdU-positive nuclei (n = 4 utricles; Figs. 4A and 5A). Extension of this labeling protocol through the subsequent 2 weeks of postnatal development (i.e., 2 days of label administered in vivo followed by fixation of the utricles 12 h after the last injection) revealed that the proliferation in the sensory epithelium decreases early as neonates mature (11 ± 4 BrdU-positive nuclei per utricle at P6 after P3–P5 labeling; 3 ± 2 at P9 after P6–P8 labeling; 1 ± 1 at P12 after P9–P11 labeling; and 4 ± 2 at P15 after P12–P14 labeling, n = 4 utricles per age group; Figs. 4B, C and 5A).
FIG. 4.
In vivo BrdU labeling in the sensory and non-sensory epithelia of the utricle in mice during the 2 weeks after birth. For 3 days, BrdU was injected every 12 h, and on the morning of the fourth day, utricles were fixed and labeled with antibodies to BrdU (green) and myosin VIIA (purple). A The confocal image of the utricle from a mouse that was given BrdU from P0 to P2 shows many labeled cells in the non-sensory epithelium and smaller numbers of labeled cells within the macula. Zoomed-in regions (dashed boxes) show BrdU labeling within the macula (delineated by myosin VIIA labeling). B Fewer BrdU-positive nuclei are present in the non-sensory epithelium of a mouse that received BrdU from P6 to P8. Minimal labeling was detected in that macula. C Confocal image of a utricle from a mouse that received BrdU from P12 to P14 shows almost no BrdU labeling in the non-sensory epithelium and the macula. Scale bar, 100 μm. Scale bar in zoomed images, 20 μm.
FIG. 5.
Quantification of BrdU labeling revealed that early in life, hundreds of cells are dividing in the utricles of mice, but in the sensory epithelium most cells exit the cell cycle early in the week after birth. A Graph of the average number of BrdU-positive nuclei per macula in utricles from mice that received injections of BrdU every 12 h for 3 days prior to the indicated age. Sac age at sacrifice, BrdU age interval over which BrdU was administered. B Graph of the average number of BrdU-labeled nuclei in the band of non-sensory epithelium that is within 30 μm of the macula's edge. Note that the BrdU labeling in the non-sensory epithelium declines more slowly than the labeling in the macula (n = 4 utricles per age). Data are expressed as the mean ± SEM. C EdU incorporated into cycling cells and their progeny in vivo is still detectable (green) 1 week after injection. A confocal image of a utricle harvested at P1 from a mouse that received one injection of EdU at P0 shows a pattern similar to the utricles labeled by multiple daily injections of BrdU (Fig. 4). D Graph showing the rapid postnatal decrease in the number of EdU-positive nuclei per macula that were detected in utricles harvested 24 h after mice received a single injection of EdU. Dashed line is the fit of exponential equation shown. Data are expressed as the mean ± SEM, n = 4–5 utricles per age.
To characterize the decline with better temporal resolution, we gave additional mice a single injection of EdU at P0, P1, or P2 and fixed their utricles 24 h later. EdU detection does not require antibodies, so one injection can give more specific and intense labeling of S-phase nuclei (Fig. 5C; Salic and Mitchison 2008). The number of EdU-positive cells that could be labeled in this way declined exponentially from P0 to P3 (Fig. 5D), indicating that the greatest amount of postnatal proliferation occurs on the day of birth. This time course is similar to what was observed in Ruben's classic investigation of terminal cell divisions (Ruben 1967), but our results show many more cells enter the S phase after birth than previously reported.
The non-sensory epithelium in the utricles of mice that received injections of BrdU during the P0–P2 period and in others that had received BrdU from P3 to P5 exhibited a much higher incidence of BrdU-positive nuclei than in the sensory epithelium (Fig. 4). There, a large population of dividing cells adjacent to the sensory epithelium also appeared to decline or cease proliferating with age (731 ± 65 BrdU-positive nuclei at P3 following P0–P2 label administration; 330 ± 29 at P6 following P3–P5 administration; 87 ± 12 at P9 after P6–P8 administration; 12 ± 1 at P12 after P9–P11administration; and 6 ± 1 at P15 after P12–P14 administration; Fig. 5B). The frequency of DNA labeling in the non-sensory epithelium suggested the possibility that some of the new hair cells might arise as progeny produced from progenitors located just outside the peripheral edge of the sensory epithelium.
The macula's lateral edge and medial striola are sites of postnatal cell division
The images of whole-mount utricles from P3 mice suggested that BrdU-positive nuclei might be most commonly found near the macula's lateral edge and its striola (Fig. 4A). To determine whether that was the case, we assessed the spatial distribution quantitatively by measuring the shortest distance between each BrdU-positive nucleus in the macula and the lateral edge. When we plotted the fraction that fell within each 10-μm interval of distance, the histogram showed a half-normal distribution of BrdU-positive nuclei that peaked within the outermost 10-μm zone at the lateral edge and decayed to a minimum that was 80–90 μm more medial (Fig. 6A). The half-normal distribution contained 61 % of the macula's BrdU-positive nuclei. A second smaller peak contained 37 % of the BrdU-positive nuclei within a normal distribution that was 100–200 μm medial to the edge (Fig. 6A). Our measurements of the distance between the reversal line and the lateral edge in maculae from P0 and P80 mice suggest that the width of a Peripheral Growth Zone (PGZ) at the lateral edge would be 83 μm at P3. Previous studies have shown that the striola extends ∼72 μm medially from the reversal line, positioning it 83–155 μm from the lateral edge (Desai et al. 2005; Li et al. 2008). Thus, quantitative measurements and overlays of the outline of the macula are in agreement in showing that the half-normal distribution of BrdU-positive nuclei falls within a presumptive PGZ at the lateral edge. A lesser peak and normal distribution of BrdU-positive nuclei appears to be a Striolar Growth Zone (SGZ) that is centered in the medial striolar region. The PGZ and SGZ appear to be the primary sites of postnatal cell production.
When we immunostained utricles from P0 mice with an antibody to the Ki-67 antigen, which labels cells in the active phases of the cell cycle, a band of strong labeling in the basal supporting cell nuclear layer at the lateral edge of the macula was revealed (Kee et al. 2002; Fig. 6B). Anti-Ki-67 also labeled smaller numbers of nuclei that were more widely scattered in the P0 sensory epithelium just medial to the striola (Fig. 6B). Little to no Ki-67 labeling was observed in P4 and P80 maculae, but Ki-67 labeling persisted in the non-sensory epithelium (data not shown).
Postnatal proliferation gives rise to significant numbers of new hair cells
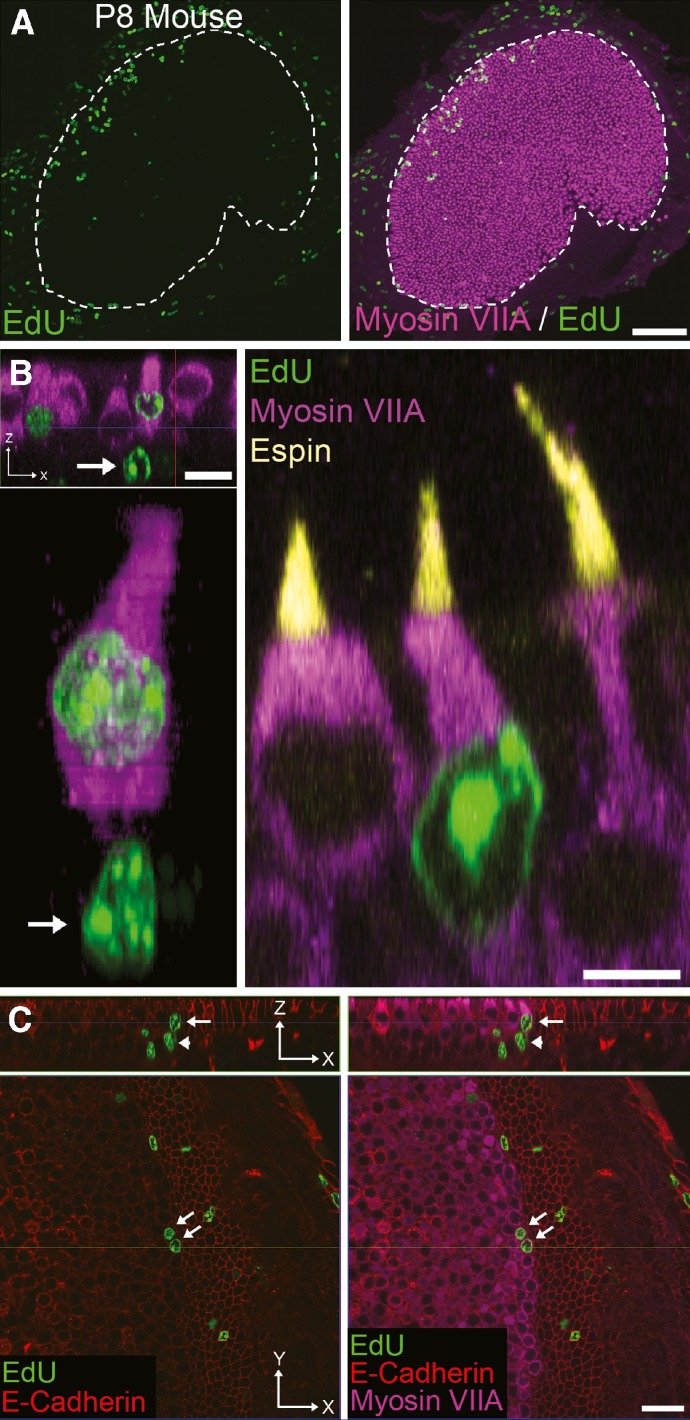
While utricular maculae from the P3 mice that were labeled by BrdU injections from P0 to P2 and fixed 12 h after the last BrdU administration each contained more than a hundred labeled cells, relatively few were myosin-VIIA-positive by that time point (BrdU-positive/myosin-VIIA-positive cells: mean = 3 ± 1). Utricles from P1 mice that received one injection of EdU at P0 also contained no EdU-positive/myosin-VIIA-positive cells. To test whether progeny produced from divisions of cells that replicated their DNA postnatally might require several more days to differentiate as new hair cells, we gave another group of mice one injection of EdU on P0 and fixed their utricles 5–18 days later. In the utricles fixed at P5 and P8, EdU-positive/myosin-VIIA-positive cells that had characteristic, flask-shaped hair cell bodies and espin-containing hair bundles comprised 16 ± 2 and 18 ± 1 % of the EdU-positive cells in the macula, respectively (n = 3–7 utricles per age; Figs. 7 and 8A). Between P8 and P12, the proportion of the EdU-positive cells that became hair cells increased significantly to 31 ± 1 %, and it rose to 35 ± 1 % in utricles fixed at P18 (p < 0.05, one-way ANOVA with Bonferonni’s multiple comparisons test; Fig. 8A). Many of the EdU-positive/myosin VIIA-positive hair cells appeared to be paired with myosin-VIIA-negative supporting cells or supporting-cell-like cells that contained basally positioned EdU-positive nuclei (arrows and arrowheads in Fig. 7B, C, respectively). The results show that cells that replicate their DNA and divide postnatally make a substantial contribution to the growth of the mouse utricle's hair cell population. They also demonstrate that cells produced from the progenitors that enter the S phase during postnatal life can take 3–12 days to differentiate as myosin VIIA-expressing hair cells.
FIG. 7.
Cells from neonatal mice that incorporate EdU in vivo differentiate into hair cells that develop recognizable hair bundles and express hair cell markers. A Confocal image of a utricle from a mouse that was injected once with EdU (green) at P0 and harvested at P8 showing that cells and their progeny survive for days after incorporating EdU. Hair cells are labeled with an antibody to myosin VIIA (purple). Scale bar, 100 μm. B Confocal images of utricles from P8 mice that received one EdU injection at P0. Arrows in these panels indicate two EdU-positive supporting cell nuclei paired with two of the EdU-positive/myosin-VIIA-positive hair cells. Top left x–z view parallel to the long axis of the hair cells. Scale bar, 10 μm. Bottom left Maximum-intensity projection of an EdU-positive hair cell/supporting cell pair. Right x–z view of two EdU-negative and one EdU-positive hair cells that all show myosin-VIIA-positive labeling of their cell bodies and espin labeling of their hair bundles (yellow). Scale bar, 5 μm. C Confocal images of the edge of the utricular macula from a P8 mouse that was injected with EdU once at P0. These panels show two EdU-positive/myosin-VIIA-positive hair cells in the outermost row at the edge of the macula. Non-sensory epithelial cells are labeled by an antibody to E-cadherin (red). Top An x–z view parallel to the long axis of the hair cells where the nucleus of an EdU-positive supporting cell (arrowhead) is paired with an EdU-positive/myosin-VIIA-positive hair cell (arrow). Bottom x–y view: cross-sectional view at the level of the hair cell nuclei. Two EdU-positive/myosin-VIIA-positive cells are visible (arrows). Scale bar, 20 μm.
Many of the EdU-positive/myosin-VIIA-positive cells were located in the outermost row of hair cells at the lateral boundary between the macula and the non-sensory epithelium (Fig. 7A, C). Plots of the positions of EdU-positive/myosin-VIIA-positive cells in P8 mouse utricles showed two clusters of EdU-labeled hair cells. One cluster, containing the majority of these cells, extended 10 μm inward from the macula’s lateral edge (typically within one row of hair cells). Scattered EdU-positive/myosin VIIA-positive cells were also found along other parts of the macula’s peripheral edge. The other somewhat scattered cluster of EdU-positive/myosin VIIA-positive cells spanned the medial–striolar region (Fig. 8B). Thus, new hair cells appear to arise in the PGZ and SGZ with distributions that closely mirror the distributions of BrdU-positive supporting cell nuclei that were labeled in the short-survival experiments (Fig. 6A).
Estimates of total postnatal cell production
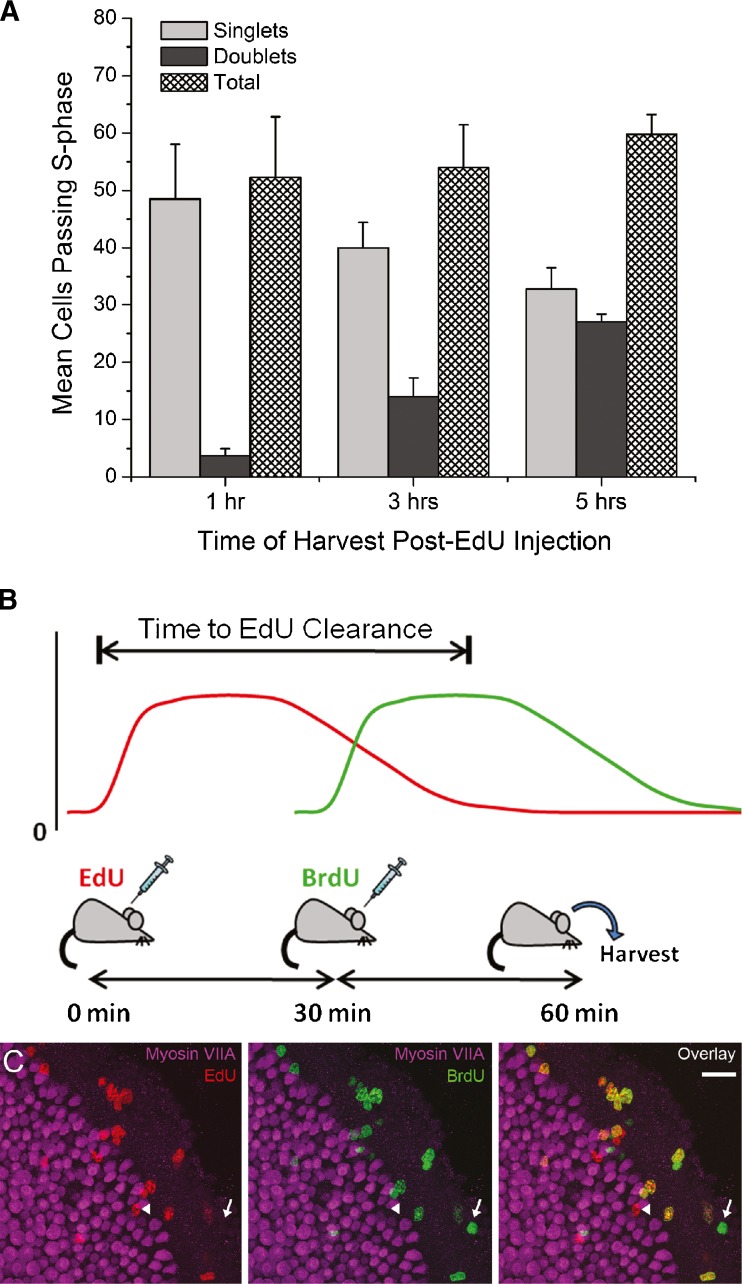
The rates of EdU clearance and incorporation can be used to estimate the total number of sensory epithelium cells that entered S phase and divided after birth. To determine clearance time, we injected EdU into a group of mice once at P0; fixed their utricles 1, 3, or 5 h later; and counted the number of EdU-positive nuclei in the macula, noting whether they occurred alone or in pairs. After 1 h, the number of S-phase-labeled cells did not change significantly, which is consistent with a clearance time of 1 h or less (p = 0.7782, one-way ANOVA with Bonferonni’s multiple comparisons test; n = 4; Fig. 9A). As an alternative measure of clearance time, we gave another group of P0 mice one injection of EdU and, 30 min later, followed that with one injection of BrdU. The utricles of those mice were fixed 30 min, 1 h, or 2 h after the BrdU injection. Other P0 mice were injected with EdU and BrdU contemporaneously, fixed 1 h later, and served as positive controls for the reactions used for EdU and BrdU detection. After processing to reveal both of the S phase DNA replication tracers, only EdU-positive/BrdU-positive nuclei were found in the utricles from co-injected control mice, but EdU-negative/BrdU-positive nuclei were detected in utricles from each of the experimental groups (Fig. 9B). The presence of EdU-negative/BrdU-positive nuclei in those utricles indicates that the EdU pulse, which had been injected just 30 min prior to the BrdU, was cleared in 30 min or less so that after that time only BrdU was available for incorporation into replicating DNA. These observations are consistent with a conservative estimate for EdU clearance time, which is ≤1 h (n = 3–4 utricles; Fig. 9A). The estimate also is consistent with a previous study which found that EdU has a serum half-life of 35 min after intravenous administration into mice (Cheraghali et al. 1995).
FIG. 9.
A Subcutaneous injection of EdU will only label utricular cells if they replicate DNA during the 1 h after the injection. A Graph of the numbers of singlet, doublet, and total EdU-positive nuclei labeled in the sensory epithelium of P0 mouse utricles that were harvested 1, 3, or 5 h after a single EdU injection. The total number of EdU-positive nuclei at the different harvest times changed little across that time span, consistent with the clearance of EdU occurring within 1 h. EdU singlets decreased from 1 to 5 h, while the EdU doublets increased as progenitor cells that labeled in the S phase completed mitosis and cytokinesis. The high incidence of singlets remaining at 5 h shows that cell cycle times vary in this population and can extend beyond 4 h (n = 4 utricles). Data are expressed as the mean ± SEM. BTop An illustration of the EdU/BrdU co-labeling protocol used to measure the EdU clearance time. P0 mice received one injection of EdU, followed 30 min later by one injection of BrdU. Some utricles were harvested 30 min after the BrdU injection and others after 1 h. The presence of EdU-negative/BrdU-positive nuclei shows that only BrdU was available for incorporation into those cells' replicating DNA and established that the EdU was already cleared at the time of the BrdU injection. Bottom Confocal image of a utricle that was harvested 30 min after the BrdU injection and 1 h after the EdU injection. EdU-positive/BrdU-negative (arrowhead) and EdU-negative/BrdU-positive nuclei (arrow) are present in the sensory and non-sensory epithelia. EdU-negative/BrdU-positive nuclei were detected in all utricles harvested 30 min and 1 h after the BrdU injection, indicating that EdU is cleared in ≤1 h. As a control (not shown in the illustration), EdU and BrdU were injected into P0 mice simultaneously and the utricles harvested 1 h later. In the utricles from the co-injected (control) mice, all the labeled nuclei were detectably EdU-positive/BrdU-positive (n = 4 utricles). Scale bar, 20 μm.
To calculate the hourly rate of EdU incorporation, we divided the dependent variable from Figure 5D by the maximum estimated EdU clearance time (1 h). We then plotted the rate of EdU incorporation versus time and fit a decaying exponential to the data. The equation with resultant parameter fits was
 |
where t is the time in hours (r2 = 1). Integrating this equation from 0 to 72 h (i.e., P0 to P3) yields 3,057 cells that would incorporate EdU if it were continuously available in the bloodstream. To obtain the data in Figure 5D, mice were injected once with EdU and their utricles fixed 24 h later, which would allow the cells sufficient time to divide after incorporating EdU. If all 3,057 the of cells were produced from divisions of EdU-labeled progenitors, then the number of sensory epithelium cells that entered S phase between P0 and P3 would be half, or 1,529.
To estimate the number of hair cells produced from the cell divisions that take place between P0 and P3, we multiplied 3,057 by the percentage of EdU-positive cells that differentiated into myosin VIIA-positive hair cells (16–35 %), which yielded a range of 489–1,070 new hair cells produced from progenitor cells that entered S phase postnatally. Since our counts showed that a mean of 1,888 hair cells are added to the murine utricle after birth, approximately 489–1,070 of the new hair cells would be expected to have arisen as progeny from postnatal cell divisions, accounting for 26–57 % of the postnatal increase in the utricle's hair cell population. We conclude that 43–74 % of the newly appearing hair cells arise postnatally through differentiation from cells whose progenitors last exited S phase of the cell cycle at a point prior to birth.
Discussion
Our results show that the macula of the mouse utricle adds over a thousand new hair cells as it expands in area during postnatal maturation. The peripheral edges of the macula, in particular its lateral edge, appear to be postnatal growth zones, with a lesser growth zone near the medial striola region. We have termed these growth zones the PGZ and the SGZ. Many of the new hair cells that arise in those zones differentiate as progeny produced from divisions of cells that last exited the S phase during embryonic development, but significant numbers of new hair cells arise as progeny from the divisions of progenitors that replicate DNA in the first days after birth. Proliferation in the macula declines sharply by the second day after birth, while the decline in proliferation occurs more slowly in the surrounding non-sensory epithelium, progressing through a ∼90 % decrease by the 12th day after birth. Lineage tracing shows that a substantial number of the hair cells that arise after birth originate from cells that express PLP, a marker protein that is expressed in a subset of supporting cells or supporting-cell-like progenitor cells. Thus, for a short period after birth, the sensory epithelium of the mouse utricle exhibits growth and hair cell production that are somewhat reminiscent of postembryonic growth and turnover processes that occur throughout life in the ears of many non-mammalian vertebrates.
Cell production contributes significantly to postnatal growth of the mouse utricle
As part of a classic examination of the embryonic ear's terminal mitoses, Ruben gave a group of mice 3H-thymidine on the day of birth and, after fixing the utricles in adulthood, counted <15 labeled cells in the utricular epithelium (Ruben 1967). Interpretation of those limited data appeared to lead to the conclusion that little or no mitotic production of hair cells occurs in mice after birth (Rusch et al. 1998; Kirkegaard and Nyengaard 2005).
Here, we first fixed utricles of mice at different ages and counted all of the hair cells. These complete counts show that postnatal addition accounts for over half of the hair cells in the adult utricle. Subsequent labeling with thymidine analogues showed that neonatal utricles contain substantially larger populations of dividing cells than might have been suspected, with our calculations suggesting that roughly 1,500 cells in the macula pass through S phase between P0 and P3. Differences between the inbred pigmented CBA-J strain investigated by Ruben (1967) and the outbred albino Swiss Webster strain investigated in our experiments may be responsible for differences in the results. Also, Ruben investigated terminal mitotic labeling by giving 3H-thymidine to mice as embryos or early neonates, then fixing their ears in adulthood, so the death of labeled cells and the potential for label dilution during subsequent cell cycling could account for some of the differences in the results. Labeling, detection, and the counting methods used differed between this and past studies as well.
In a more recent report, counts from representative sections of mouse utricles were used to estimate cell numbers in utricles of neonatal mice, leading to the conclusion that the utricle’s supporting cells decrease by 30 % between P1 and P16 (3,254 ± 281 cells at P1 to 2,186 ± 38 cells at P16; Kirkegaard and Nyengaard 2005). We did not count supporting cells, but our counts show a mean postnatal increase of >1,800 hair cells. Many of the postnatally added hair cells differentiate as progeny of progenitors that last passed through S phase prior to birth, but a significant number arise from progenitors that replicate DNA and divide after birth.
It seems likely that the decrease in supporting cell numbers that were reported previously may occur in part because such cells differentiate or convert into hair cells. Also, some supporting cells may be lost through apoptosis. In salamander lateral line organs, in avian utricles, and throughout development of the central and peripheral nervous systems, apoptotic cell death plays an important role in shaping mature tissues (Jones and Corwin 1996; Matsui et al. 2002; Nikolaev et al. 2009).
Lineage tracing in a novel mouse model showed that a significant portion of the new hair cells arose from PLP-expressing supporting cells, but the lineage data did not distinguish between cells that arose from supporting cell divisions and those that might have arisen through direct supporting cell to hair cell phenotype conversion (Fig. 2). eGFP-positive hair cells that arose in lineages from PLP-expressing supporting cells accounted for 22 % of the total new hair cells that are added to the utricle, as shown by the counts in Figure 1. This is consistent with expectations since most striolar supporting cells and a subset of extrastriolar supporting cells do not exhibit PLP promoter activity (Fig. 2). The lineage tracing results also suggest that utricular hair cells originate from more than one progenitor cell population, i.e., from PLP-expressing supporting cells and either PLP-negative supporting cells or undifferentiated yet-to-be-identified stem cells. The absence of PLP-expressing supporting cells from the striola and their presence as a subset of extrastriolar supporting cells could also be indications that type 1 and 2 hair cells originate from different lineages of supporting cells or supporting-cell-like progenitors.
Postnatal addition occurs in peripheral and striolar growth zones
BrdU- and EdU-positive supporting cells and hair cells are most common at the lateral edge of the neonatal macula and arise in lower numbers within the medial striola, pointing to the possibility that these regions may provide niches for supporting-cell-like stem cells (Figs. 4, 5, 6, 7A, and 8B). The edge-dominant proliferation in neonatal mouse maculae is reminiscent of the appositional growth zones at the outer edges of the continuously growing maculae in elasmobranchs and amphibians, where the presence of stem cells has long been suspected (Lewis and Li 1973; Li and Lewis 1979; Corwin 1981, 1983, 1985). Lower amounts of proliferation and interstitial addition of hair cells also occur in the central macula of those species, but the proportions of appositional and interstitial growth are known to vary among species and may vary during development (Popper and Hoxter 1984, 1990; Jorgensen and Mathiesen 1988; Roberson et al. 1992; Lombarte and Popper 1994; Lanford et al. 1996; Kil et al. 1997; Goodyear et al. 1999). It is notable that the highest densities of BrdU- and Ki-67-positive nuclei outside the macula’s edge in mice were just proximal to the sensory epithelium (Figs. 4, 5, and 6).
Qualitative impressions from BrdU and EdU labeling, the quantitative analysis of Ki-67-labeled nuclei, and the observation that immature hair cells are most common near the neonatal macula’s lateral edge indicate that it is the predominant site of postnatal growth (Figs. 3, 4, 5, 6, 7A, and 8B). That conclusion is also consistent with the doubling of the distance between the lateral edge and reversal line that occurs between E18.5 and maturity. In contrast, embryonic growth of the macula is most pronounced in the regions that extend anteriorly, medially, and posteriorly from the reversal line (Denman-Johnson and Forge 1999). Taken together, the evidence suggests that the macula expands along its anterior, medial, and posterior edges before birth and, after birth, expands more in the remaining quadrant, i.e., between the reversal line and the lateral edge.
The timing of postnatal cell cycle exit and hair cell differentiation
In the murine cochlea, a gradient of hair cell differentiation progresses in the opposite direction from the apex-to-base progression of cell cycle exit (Ruben 1967; Lee et al. 2006). There, cell cycle exit and the resulting zone of non-proliferation depend in part on the expression of the CDKI, p27kip1 (Lee et al. 2006). The base-to-apex gradient of hair cell differentiation in the embryonic cochlea is instead paralleled by a gradient of elevated retinoblastoma protein expression (Mantela et al. 2005). Such gradients have not been reported from the mouse utricle, where differentiating hair bundles are evenly distributed throughout a small sensory epithelium when they first appear at E13.5 (Mbiene et al. 1984; Denman-Johnson and Forge 1999). Yet, 3H-thymidine labeling in embryonic rats revealed a shallow gradient in the pattern of terminal mitoses, with cells near the striola exiting the cell cycle early and those nearer the periphery exiting later (Sans and Chat 1982). In our analysis, immature hair bundles were most common at the edges of the neonatal mouse utricles, but immature hair bundles were also found centrally in a scattered distribution (Fig. 3C, D), suggesting that the pattern in postnatal mice could reflect an extension of the embryonic growth pattern described in the rat.
The findings here indicate that the progeny from cell divisions that occur after birth take considerably longer to differentiate as hair cells than those that originate before birth (Figs. 8 and 9). The majority of the cells that were labeled by BrdU and EdU given to mice on P0 were localized to the utricle’s lateral edge, with cell cycle exit and hair cell differentiation occurring in similar spatiotemporal patterns. p27kip1 may have a role in cell cycle exit in the utricle at P0 as it appears to be most highly expressed just medial to the reversal line (Laine et al. 2010).
We have described circumferential F-actin belts at the apical junctions of supporting cells in the vestibular epithelia of mice and humans that become reinforced as mammals mature postnatally, while their counterparts in birds remain thin throughout life (Burns et al. 2008). The growth of the belts in rodents correlates inversely with a progressive decrease in the incidence of supporting cell proliferation in cultured explants of the vestibular epithelia from postnatal rodents (r = −0.98). Measurements of belt growth have shown that the most rapid thickening occurs between P0 and P2, coinciding with the time when the last cells in the macula exit the cell cycle in vivo (Fig. 5A, D; Ruben 1967). Rodent and human supporting cell–supporting cell junctions also differ from those in birds in that they express E-cadherin, with a sixfold increase in expression occurring as murine supporting cells mature postnatally (Collado et al. 2011). Yet, it remains to be determined whether the unique postnatal thickening that occurs in the circumferential F-actin belts or whether the expression of E-cadherin at the junctions between the mammalian utricle’s supporting cells has causal influences that might restrict cell cycle reentry.
Implications for regeneration
Our results suggest that for a brief period after birth, the mouse utricle exhibits signs of cellular plasticity in vivo. Similar signs of plasticity have been observed in utricular sensory epithelia isolated and cultured from neonatal mice, where exogenous growth factors promote rapid supporting cell spreading and proliferation (Montcouquiol and Corwin 2001a, b; Davies et al. 2007; Gu et al. 2007; Lu and Corwin 2008). Balance organs from non-mammals produce new hair cells and supporting cells in vivo. In culture, cells within those sensory epithelia show even more robust spreading and proliferation that remain undiminished from birth to adulthood (Burns et al. 2008), and they exhibit considerable regenerative capacities throughout life. In a separate article, we report that the utricles of mice that are beyond the age when proliferation occurs during normal postnatal growth of the macula retain the capacity for regenerating hair cells in vivo. However, that capacity is lost just a few days later (Burns et al. 2012).
The postnatal growth in utricular hair cell numbers and the continuation of proliferation that has been observed here in mice may be tied to the altricial nature of their offspring. In contrast, hair cell numbers in the balance organs of humans appear to reach steady levels after 14 weeks of gestation (Rosenhall 1972, 1973; Merchant et al. 2000). Rodents are often imperfect models for human conditions, but continuing investigations in neonatal and more mature mice hold the potential to reveal just how maturational changes in mammals restrict molecular circuitry and regenerative processes that are so effective in allowing recovery of balance and hearing after damage in non-mammals.
Acknowledgments
We thank B. Hinton (University of Virginia) for mT/mG mice; J. Johnson (University of Texas Southwestern Medical Center) for Math1/nGFP mice; T. Friedman (NIDCD), I. Belyantseva (NIDCD), and Z. Ahmed (Cincinnati Children’s Hospital Medical Center) for the protocadherin-15-CD2 antibody; and J. Bartles (Northwestern University) for the espin antibody. This work was supported by grants from the National Institutes of Health DC000200 (J.T.C) and DC010519 (J.C.B).
Conflict of interest
The authors declare that they have no conflict of interest.
Contributor Information
Joseph C. Burns, Email: jcb7j@virginia.edu
Doan On, Email: doanon@gmail.com.
Wendy Baker, Email: ws5z@virginia.edu.
M. Sol Collado, Email: mc4sr@virginia.edu.
Jeffrey T. Corwin, Phone: +1-434-9248364, FAX: +1-434-9823966, Email: jtc2k@virginia.edu
References
- Ahmed ZM, Goodyear R, Riazuddin S, Lagziel A, Legan PK, Behra M, Burgess SM, Lilley KS, Wilcox ER, Griffith AJ, Frolenkov GI, Belyantseva IA, Richardson GP, Friedman TB. The tip-link antigen, a protein associated with the transduction complex of sensory hair cells, is protocadherin-15. J Neurosci. 2006;26:7022–7034. doi: 10.1523/JNEUROSCI.1163-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bermingham NA, Hassan BA, Price SD, Vollrath MA, Ben-Arie N, Eatock RA, Bellen HJ, Lysakowski A, Zoghbi HY. Math1: an essential gene for the generation of inner ear hair cells. Science. 1999;284:1837–1841. doi: 10.1126/science.284.5421.1837. [DOI] [PubMed] [Google Scholar]
- Bertalanffy L. Untersuchungen Über die Gesetzlichkeit des Wachstums. I. Allgemeine Grundlagen der Theorie; mathematische und physiologische Gesetzlichkeiten des Wachstums bei Wassertieren. Arch Entwicklungsmech. 1934;131:613–652. doi: 10.1007/BF00650112. [DOI] [PubMed] [Google Scholar]
- Brigande JV, Heller S. Quo vadis, hair cell regeneration? Nat Neurosci. 2009;12:679–685. doi: 10.1038/nn.2311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burns JC, Christophel JJ, Collado MS, Magnus C, Carfrae M, Corwin JT. Reinforcement of cell junctions correlates with the absence of hair cell regeneration in mammals and its occurrence in birds. J Comp Neurol. 2008;511:396–414. doi: 10.1002/cne.21849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burns JC, Cox BC, Thiede BR, Zuo J, Corwin JT. In vivo proliferative regeneration of balance hair cells in newborn mice. J Neurosci. 2012;32:6570–6577. doi: 10.1523/JNEUROSCI.6274-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheraghali AM, Kumar R, Knaus EE, Wiebe LI. Pharmacokinetics and bioavailability of 5-ethyl-2′-deoxyuridine and its novel (5R,6R)-5-bromo-6-ethoxy-5,6-dihydro prodrugs in mice. Drug Metab Dispos. 1995;23:223–226. [PubMed] [Google Scholar]
- Collado MS, Thiede BR, Baker W, Askew C, Igbani LM, Corwin JT. The postnatal accumulation of junctional E-cadherin is inversely correlated with the capacity for supporting cells to convert directly into sensory hair cells in mammalian balance organs. J Neurosci. 2011;31:11855–11866. doi: 10.1523/JNEUROSCI.2525-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corwin JT. Postembryonic production and aging of inner ear hair cells in sharks. J Comp Neurol. 1981;201:541–553. doi: 10.1002/cne.902010406. [DOI] [PubMed] [Google Scholar]
- Corwin JT. Postembryonic growth of the macula neglecta auditory detector in the ray, Raja clavata: continual increases in hair cell number, neural convergence, and physiological sensitivity. J Comp Neurol. 1983;217:345–356. doi: 10.1002/cne.902170309. [DOI] [PubMed] [Google Scholar]
- Corwin JT. Perpetual production of hair cells and maturational changes in hair cell ultrastructure accompany postembryonic growth in an amphibian ear. Proc Natl Acad Sci U S A. 1985;82:3911–3915. doi: 10.1073/pnas.82.11.3911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corwin JT, Cotanche DA. Regeneration of sensory hair cells after acoustic trauma. Science. 1988;240:1772–1774. doi: 10.1126/science.3381100. [DOI] [PubMed] [Google Scholar]
- Davies D, Magnus C, Corwin JT. Developmental changes in cell–extracellular matrix interactions limit proliferation in the mammalian inner ear. Eur J Neurosci. 2007;25:985–998. doi: 10.1111/j.1460-9568.2007.05355.x. [DOI] [PubMed] [Google Scholar]
- Deans MR, Antic D, Suyama K, Scott MP, Axelrod JD, Goodrich LV. Asymmetric distribution of prickle-like 2 reveals an early underlying polarization of vestibular sensory epithelia in the inner ear. J Neurosci. 2007;27:3139–3147. doi: 10.1523/JNEUROSCI.5151-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denman-Johnson K, Forge A. Establishment of hair bundle polarity and orientation in the developing vestibular system of the mouse. J Neurocytol. 1999;28:821–835. doi: 10.1023/A:1007061819934. [DOI] [PubMed] [Google Scholar]
- Desai SS, Zeh C, Lysakowski A. Comparative morphology of rodent vestibular periphery. I. Saccular and utricular maculae. J Neurophysiol. 2005;93:251–266. doi: 10.1152/jn.00746.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doerflinger NH, Macklin WB, Popko B. Inducible site-specific recombination in myelinating cells. Genesis. 2003;35:63–72. doi: 10.1002/gene.10154. [DOI] [PubMed] [Google Scholar]
- Forge A, Li L, Corwin JT, Nevill G. Ultrastructural evidence for hair cell regeneration in the mammalian inner ear. Science. 1993;259:1616–1619. doi: 10.1126/science.8456284. [DOI] [PubMed] [Google Scholar]
- Gomez-Casati ME, Murtie J, Taylor B, Corfas G. Cell-specific inducible gene recombination in postnatal inner ear supporting cells and glia. J Assoc Res Otolaryngol JARO. 2010;11:19–26. doi: 10.1007/s10162-009-0191-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodyear RJ, Gates R, Lukashkin AN, Richardson GP. Hair-cell numbers continue to increase in the utricular macula of the early posthatch chick. J Neurocytol. 1999;28:851–861. doi: 10.1023/A:1007070121751. [DOI] [PubMed] [Google Scholar]
- Gu R, Montcouquiol M, Marchionni M, Corwin JT. Proliferative responses to growth factors decline rapidly during postnatal maturation of mammalian hair cell epithelia. Eur J Neurosci. 2007;25:1363–1372. doi: 10.1111/j.1460-9568.2007.05414.x. [DOI] [PubMed] [Google Scholar]
- Hasson T, Gillespie PG, Garcia JA, MacDonald RB, Zhao Y, Yee AG, Mooseker MS, Corey DP. Unconventional myosins in inner-ear sensory epithelia. J Cell Biol. 1997;137:1287–1307. doi: 10.1083/jcb.137.6.1287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen-Smith H, Hallworth R. Lateral wall protein content mediates alterations in cochlear outer hair cell mechanics before and after hearing onset. Cell Motil Cytoskeleton. 2007;64:705–717. doi: 10.1002/cm.20217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones JE, Corwin JT. Regeneration of sensory cells after laser ablation in the lateral line system: hair cell lineage and macrophage behavior revealed by time-lapse video microscopy. J Neurosci. 1996;16:649–662. doi: 10.1523/JNEUROSCI.16-02-00649.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorgensen JM, Mathiesen C. The avian inner ear. Continuous production of hair cells in vestibular sensory organs, but not in the auditory papilla. Naturwissenschaften. 1988;75:319–320. doi: 10.1007/BF00367330. [DOI] [PubMed] [Google Scholar]
- Kawamoto K, Izumikawa M, Beyer LA, Atkin GM, Raphael Y. Spontaneous hair cell regeneration in the mouse utricle following gentamicin ototoxicity. Hear Res. 2009;247:17–26. doi: 10.1016/j.heares.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kee N, Sivalingam S, Boonstra R, Wojtowicz JM. The utility of Ki-67 and BrdU as proliferative markers of adult neurogenesis. J Neurosci Methods. 2002;115:97–105. doi: 10.1016/S0165-0270(02)00007-9. [DOI] [PubMed] [Google Scholar]
- Kil J, Warchol ME, Corwin JT. Cell death, cell proliferation, and estimates of hair cell life spans in the vestibular organs of chicks. Hear Res. 1997;114:117–126. doi: 10.1016/S0378-5955(97)00166-4. [DOI] [PubMed] [Google Scholar]
- Kirkegaard M, Nyengaard JR. Stereological study of postnatal development in the mouse utricular macula. J Comp Neurol. 2005;492:132–144. doi: 10.1002/cne.20736. [DOI] [PubMed] [Google Scholar]
- Laine H, Sulg M, Kirjavainen A, Pirvola U. Cell cycle regulation in the inner ear sensory epithelia: role of cyclin D1 and cyclin-dependent kinase inhibitors. Dev Biol. 2010;337:134–146. doi: 10.1016/j.ydbio.2009.10.027. [DOI] [PubMed] [Google Scholar]
- Lanford PJ, Presson JC, Popper AN. Cell proliferation and hair cell addition in the ear of the goldfish, Carassius auratus. Hear Res. 1996;100:1–9. doi: 10.1016/0378-5955(96)00110-4. [DOI] [PubMed] [Google Scholar]
- Lee YS, Liu F, Segil N. A morphogenetic wave of p27Kip1 transcription directs cell cycle exit during organ of Corti development. Development. 2006;133:2817–2826. doi: 10.1242/dev.02453. [DOI] [PubMed] [Google Scholar]
- Lewis ER, Li CW. Evidence concerning the morphogenesis of saccular receptors in the bullfrog (Rana catesbeiana) J Morphol. 1973;139:351–361. doi: 10.1002/jmor.1051390305. [DOI] [PubMed] [Google Scholar]
- Li CW, Lewis ER. Structure and development of vestibular hair cells in the larval bullfrog. Ann Otol Rhinol Laryngol. 1979;88:427–437. doi: 10.1177/000348947908800323. [DOI] [PubMed] [Google Scholar]
- Li A, Xue J, Peterson EH. Architecture of the mouse utricle: macular organization and hair bundle heights. J Neurophysiol. 2008;99:718–733. doi: 10.1152/jn.00831.2007. [DOI] [PubMed] [Google Scholar]
- Lombarte A, Popper AN. Quantitative analyses of postembryonic hair cell addition in the otolithic endorgans of the inner ear of the European hake, Merluccius merluccius (Gadiformes, Teleostei) J Comp Neurol. 1994;345:419–428. doi: 10.1002/cne.903450308. [DOI] [PubMed] [Google Scholar]
- Lu Z, Corwin JT. The influence of glycogen synthase kinase 3 in limiting cell addition in the mammalian ear. Dev Neurobiol. 2008;68:1059–1075. doi: 10.1002/dneu.20635. [DOI] [PubMed] [Google Scholar]
- Lumpkin EA, Collisson T, Parab P, Omer-Abdalla A, Haeberle H, Chen P, Doetzlhofer A, White P, Groves A, Segil N, Johnson JE. Math1-driven GFP expression in the developing nervous system of transgenic mice. Gene Expr Patterns. 2003;3:389–395. doi: 10.1016/S1567-133X(03)00089-9. [DOI] [PubMed] [Google Scholar]
- Mantela J, Jiang Z, Ylikoski J, Fritzsch B, Zacksenhaus E, Pirvola U. The retinoblastoma gene pathway regulates the postmitotic state of hair cells of the mouse inner ear. Development. 2005;132:2377–2388. doi: 10.1242/dev.01834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsui JI, Ogilvie JM, Warchol ME. Inhibition of caspases prevents ototoxic and ongoing hair cell death. J Neurosci. 2002;22:1218–1227. doi: 10.1523/JNEUROSCI.22-04-01218.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mbiene JP, Favre D, Sans A. The pattern of ciliary development in fetal mouse vestibular receptors. A qualitative and quantitative SEM study. Anat Embryol (Berl) 1984;170:229–238. doi: 10.1007/BF00318726. [DOI] [PubMed] [Google Scholar]
- Merchant SN, Velazquez-Villasenor L, Tsuji K, Glynn RJ, Wall C, 3rd, Rauch SD. Temporal bone studies of the human peripheral vestibular system. Normative vestibular hair cell data. Ann Otol Rhinol Laryngol Suppl. 2000;181:3–13. doi: 10.1177/00034894001090s502. [DOI] [PubMed] [Google Scholar]
- Montcouquiol M, Corwin JT. Brief treatments with forskolin enhance S-phase entry in balance epithelia from the ears of rats. J Neurosci. 2001;21:974–982. doi: 10.1523/JNEUROSCI.21-03-00974.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montcouquiol M, Corwin JT. Intracellular signals that control cell proliferation in mammalian balance epithelia: key roles for phosphatidylinositol-3 kinase, mammalian target of rapamycin, and S6 kinases in preference to calcium, protein kinase C, and mitogen-activated protein kinase. J Neurosci. 2001;21:570–580. doi: 10.1523/JNEUROSCI.21-02-00570.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L. A global double-fluorescent Cre reporter mouse. Genesis. 2007;45:593–605. doi: 10.1002/dvg.20335. [DOI] [PubMed] [Google Scholar]
- Nikolaev A, McLaughlin T, O'Leary DD, Tessier-Lavigne M. APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature. 2009;457:981–989. doi: 10.1038/nature07767. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Popper AN, Hoxter B. Growth of a fish ear: I. Quantitative analysis of hair cell and ganglion cell proliferation. Hear Res. 1984;15:133–142. doi: 10.1016/0378-5955(84)90044-3. [DOI] [PubMed] [Google Scholar]
- Popper AN, Hoxter B. Growth of a fish ear. II. Locations of newly proliferated sensory hair cells in the saccular epithelium of Astronotus ocellatus. Hear Res. 1990;45:33–40. doi: 10.1016/0378-5955(90)90180-W. [DOI] [PubMed] [Google Scholar]
- Roberson DF, Weisleder P, Bohrer PS, Rubel EW. Ongoing production of sensory cells in the vestibular epithelium of the chick. Hear Res. 1992;57:166–174. doi: 10.1016/0378-5955(92)90149-H. [DOI] [PubMed] [Google Scholar]
- Rosenhall U. Mapping of the cristae ampullares in man. Ann Otol Rhinol Laryngol. 1972;81:882–889. doi: 10.1177/000348947208100622. [DOI] [PubMed] [Google Scholar]
- Rosenhall U. Degenerative patterns in the aging human vestibular neuro-epithelia. Acta Otolaryngol. 1973;76:208–220. doi: 10.3109/00016487309121501. [DOI] [PubMed] [Google Scholar]
- Ruben RJ. Development of the inner ear of the mouse: a radioautographic study of terminal mitoses. Acta Otolaryngol. 1967;Suppl 220:221–244. [PubMed] [Google Scholar]
- Rusch A, Lysakowski A, Eatock RA. Postnatal development of type I and type II hair cells in the mouse utricle: acquisition of voltage-gated conductances and differentiated morphology. J Neurosci. 1998;18:7487–7501. doi: 10.1523/JNEUROSCI.18-18-07487.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryals BM, Rubel EW. Hair cell regeneration after acoustic trauma in adult Coturnix quail. Science. 1988;240:1774–1776. doi: 10.1126/science.3381101. [DOI] [PubMed] [Google Scholar]
- Salic A, Mitchison TJ. A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proc Natl Acad Sci U S A. 2008;105:2415–2420. doi: 10.1073/pnas.0712168105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sans A, Chat M. Analysis of temporal and spatial patterns of rat vestibular hair cell differentiation by tritiated thymidine radioautography. J Comp Neurol. 1982;206:1–8. doi: 10.1002/cne.902060102. [DOI] [PubMed] [Google Scholar]
- Warchol ME. Sensory regeneration in the vertebrate inner ear: differences at the levels of cells and species. Hear Res. 2011;273:72–79. doi: 10.1016/j.heares.2010.05.004. [DOI] [PubMed] [Google Scholar]
- Warchol ME, Lambert PR, Goldstein BJ, Forge A, Corwin JT. Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science. 1993;259:1619–1622. doi: 10.1126/science.8456285. [DOI] [PubMed] [Google Scholar]
- Zheng JL, Gao WQ. Overexpression of Math1 induces robust production of extra hair cells in postnatal rat inner ears. Nat Neurosci. 2000;3:580–586. doi: 10.1038/75753. [DOI] [PubMed] [Google Scholar]