ABSTRACT
Background
The effects of revascularization extent (RE) on the long‐term prognosis of patients with stable angina pectoris and 3‐vessel disease who underwent percutaneous coronary intervention were unknown.
Hypothesis
The study was aimed at evaluating whether there was an effect of RE on patients presenting with stable angina pectoris and 3‐vessel disease.
Methods
RE, which was calculated by baseline SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score minus residue SYNTAX score divided by baseline SYNTAX score, was initially used in our study. Five hundred fifty‐eight patients presenting with stable angina pectoris and 3‐vessel disease were assigned to and compared among tertiles according to RE and clinical outcomes. The primary end point was the major adverse cardiovascular event (MACE), a composite of cardiac death, nonfatal myocardial infarction (MI), and any repeat revascularization.
Results
The median follow‐up period was 56.9 months (interquartile range, 52.1–63.6). The incidence of MACE increased significantly as RE increased (13.3%, 31.4%, and 44.1%, log‐rank P < 0.001). The same tendency was observed in occurrences of target‐vessel failure (TVF) (a composite of cardiac death, MI, or target‐vessel revascularization) (8.8%, 20.3%, and 28.4%, log‐rank P < 0.001), repeat revascularization (11.8%, 26.2%, and 35.6%, log‐rank P < 0.001), and MI (1.1%, 2.9%, and 12.6%, log‐rank P < 0.001). Multivariate analysis confirmed the tendencies mentioned above.
Conclusions
For patients presenting with stable angina pectoris and 3‐vessel disease, the increasing extent of revascularization resulted in a less favorable prognosis.
Introduction
The prognostic impact of incomplete revascularization (IR) after percutaneous coronary intervention (PCI) in patients presenting with multivessel disease has been inconsistent between studies.1, 2, 3, 4, 5, 6, 7 Substantial baseline variation between different studies and varying definitions of complete revascularization were 2 main contributing factors that have fuelled this debate.
Most previous studies focusing on the impact of IR after PCI have not discriminated the indications for PCI,1, 2, 3, 4, 5, 6 which was an important consideration when the decision for PCI strategy was made. Mixed patients presenting with stable angina pectoris (SAP) and acute coronary syndromes (ACS) can confound the assessment of IR. The clinical significance of IR after PCI with drug‐eluting stents (DESs) in patients with SAP and multivessel disease remains uncertain.
Definitions of complete revascularization in prior studies varying according to the degree of coronary stenosis (eg, ≥50% vs ≥70%) or vessel size diameter (eg, ≥1.5 – ≥ 2.5 mm) also confounded these reports.3, 4, 7 However, in these studies, the incompleteness of coronary revascularization was established as a binary outcome, and patients with large areas of ischemic, nonrevascularized myocardium or merely small, untreated branches were pooled together. Compared to the binary form previously used, the quantification of the extent of revascularization could better depict the patients' characterization. Recently, the so‐called residue SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) score, which could be calculated after PCI, has been proposed to quantify residual atherosclerosis after multivessel PCI.8, 9 The revascularization extent (RE), which is calculated by the baseline SYNTAX score minus the residue SYNTAX score divided by the baseline SYNTAX score, was first used in our study. Whether this approach has a prognostic value on multivessel PCI is unknown.
Therefore, this study was aimed at evaluating the effect of RE on patients presenting with SAP and 3‐vessel disease who underwent PCI.
Methods
Patient Population
All of the patients provided written informed consent for both the procedure and the subsequent data collection and analysis for research purposes. The study was conducted in accordance with the Declaration of Helsinki and had been approved by the local ethics committee of Beijing An Zhen Hospital, Capital Medical University. The local ethics committee approved the use of the data for this study. The inclusion criteria were as follows: (1) SAP, (2) 3‐vessel coronary artery disease (CAD), (3) de novo lesions, (4) implanted a with DES, and (5) written informed consent. The exclusion criteria were designated as follows: (1) acute coronary syndrome; (2) left main disease, or single or 2‐vessel disease; (3) prior PCI or coronary artery bypass grafting (CABG); (4) life expectancy <2 years; (5) unable to sign the informed consent. According to the designated inclusive and exclusive criteria, 10 332 patients who underwent PCI with a DES between January 2007 and December 2008 in Beijing An Zhen Hospital were prospectively screened, and 558 patients were enrolled in our study (Figure 1). This study protocol was originally designed to assess the long‐term performance of a DES in patients with SAP and 3‐vessel disease. Therefore, this is a post hoc analysis of the initial single‐center registry, which was not dedicated to the present question. There was no industry involvement in the design, conduct, financial support, or analysis of the study.
Figure 1.
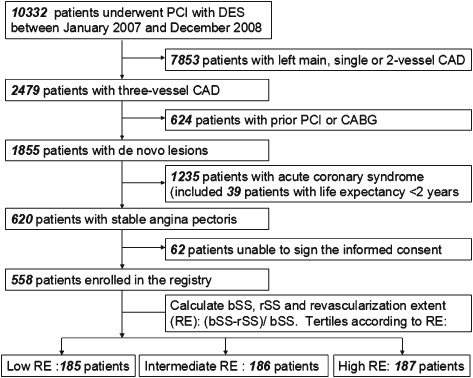
Patients' flow chart. Abbreviations: bSS, baseline SYNTAX score; CABG, coronary artery bypass grafting; CAD, coronary artery disesase; DES, drug‐eluting stent; PCI, percutaneous coronary intervention; RE, revascularization extent; rSS, residue SYNTAX score; SYNTAX, Synergy Between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery.
Procedures
Before the PCI surgery, patients presenting with atypical chest pain were suggested to undergo an exercise test, radionuclide scintigraphy, or stress echocardiography,10 whereas the patients with typical angina pectoris did not undergo such assessments for diagnosis. The reasons underlying the PCI for the patients with SAP enrolled in our study were as follows: (1) patients with unacceptable angina despite optimal medical therapy, and (2) patients with high‐risk criteria on noninvasive testing regardless of angina severity. All of the procedures were performed using standard interventional techniques. Before stent implantation, all of the patients received aspirin according to their physicians' normal procedures and either clopidogrel (75 mg/d) for 3 days prior to the procedure or a preprocedural loading dose of clopidogrel ≥300 mg. The patients continued the clopidogrel regimen for at least 1 year (75 mg/d) and aspirin indefinitely (100 mg/d) after the procedure. The use of DES types and glycoprotein IIb/IIIa inhibitors were left at the discretion of the surgeons.
Calculation of Revascularization Extent
The SYNTAX score was developed to characterize the coronary vasculature with respect to the number of lesions and their functional impact, location, and complexity.11 Higher SYNTAX scores, indicative of more complex disease, represent a bigger therapeutic challenge and have a potentially worse prognosis. The baseline SYNTAX score (bSS) and residue SYNTAX score (rSS) were obtained prior to and after the procedure, respectively. In case of staged PCI, rSS was assessed when the final planned procedure was completed. The bSS and rSS were retrospectively calculated by 2 of 3 experienced interventional cardiologists who had been trained for SYNTAX score assessment and who were blinded to the baseline data and the clinical outcome. In case of a disagreement, the opinion of the third observer was obtained, and the final decision was made by consensus. RE was defined as (bSS − rSS)/bSS. The entire enrolled population was assigned to tertiles according to RE, and the clinical outcomes were compared among tertiles.
End Point, Definition, and Follow‐up
The primary end points included a major adverse cardiovascular event (MACE), composite of cardiac death, nonfatal myocardial infarction (MI), or any repeat revascularization including PCI or CABG. Secondary end points included all‐cause mortality, nonfatal acute MI, stroke, repeat revascularization, or target‐vessel failure (TVF) (a composite of cardiac death, MI, or target‐vessel revascularization). All deaths were considered cardiac unless an unequivocal noncardiac cause could be established. We defined MI in accordance with the Joint European Society of Cardiology, the American College of Cardiology Foundation, the American Heart Association, and the World Heart Federation Task Force for the Redefinition of Myocardial Infarction.12
Information about in‐hospital clinical outcomes was obtained from the electronic centralized database of Beijing An Zhen Hospital. After discharge, follow‐up was performed at 1, 6, and 12 months, and additional clinical follow‐up occurred at yearly intervals. All follow‐up data were prospectively collected by angiographic follow‐up and outpatient or telephone interview. In the case of staged PCI, the final planned procedure was considered as the entry point for this study. Clinical events were adjudicated by an independent clinical events committee that was blinded to treatment assignment and procedural outcomes.
Statistical Analysis
Patient characteristics and outcome measures were stratified according to RE tertiles among all of the enrolled patients. Continuous variables were presented as mean values ± standard deviation or medians (interquartile range [IQR]) when appropriate. Comparisons were performed using 1‐way analysis of variance for continuous variables following a normal distribution. The normality assumption for continuous variables was evaluated using the Kolmogorov‐Smirnov test. Categorical variables were reported as counts and percentages, and differences among the 3 groups were assessed by χ2 test. The Spearman test was used to assess the correlation between rSS or bSS and RE.
Time to the primary end point was evaluated using the Kaplan‐Meier method, and the log‐rank test was applied to evaluate differences among groups. Crude and adjusted risks for adverse outcomes were compared by univariate and multivariate Cox proportional hazards regression analyses. Variables with P values <0.05 and clinically relevant covariates irrespective of their statistical relevance in univariate analyses were candidates for inclusion in multivariate Cox proportional hazards models. Univariate analyses included RE, age, gender, type 2 diabetes mellitus, hypertension, dyslipidemia, smoking history, family history, glomerular filtration rate, glycoprotein IIb/IIIa, low‐molecular‐weight heparin (LMWH), β‐blocker, angiotensin‐converting enzyme inhibitor/angiotensin receptor blocker (ACEI/ARB), statin, bSS, rSS, total occlusion, bifurcation lesions, small vessels, overlap stents, lengths of stents, number of stents, targeted left anterior descending (LAD) artery, targeted left circumflex artery, and targeted right coronary artery (RCA). Variables with P values <0.05 and clinically relevant covariates, irrespective of their statistical relevance in univariate analyses, were candidates for inclusion in the multivariate Cox proportional hazards models. After the significant, independent, baseline predictors were identified, the Cox model was rebuilt to include the RE variable using the enter methods. The number of covariates, in addition to RE, was carefully chosen to avoid overfitting. Crude and adjusted hazard ratios and corresponding 95% confidence intervals were reported. A propensity score analysis was conducted, and the results are shown in the Appendix. All of the analyses were performed using the SPSS 17.0 system (SPSS Inc., Chicago, IL). A P value <0.05 was considered statistically significant.
Results
Five hundred fifty‐eight patients with SAP and 3‐vessel disease were analyzed. As shown in Figure 1, 185, 186, and 187 patients were stratified into the low, intermediate, and high RE group, respectively, according to the RE tertiles. The median follow‐up period was 56.9 months (IQR, 52.1–63.6). Fifteen patients were lost during follow‐up, which accounted for 2.7% of the overall patient population.
Baseline clinical and angiographic characteristics of the patients stratified by RE are shown in Table 1. Low, intermediate, and high RE were defined as values of 0.04 to 0.37, 0.38 to 0.69, and 0.70 to 1, respectively. With decreasing RE, the patients tended to be older and exhibited significantly increased prevalence of hypertension and prior MI. However, the higher RE group suffered increased rates of family history. There were significant differences among the 3 groups in terms of the incidence rate of glycoprotein IIb/IIIa, LMWH, and ACEI/ARB. In terms of anatomical factors, although significant imbalance was found in the ratio of bifurcation lesions among the 3 groups, the baseline SYNTAX score, a widely used angiographic tool for grading the anatomical complexity of CAD, was balanced among the 3 groups. As a result of the RE tertile grouping, there were more stents implanted and targeted vessels treated, the higher the prevalence of overlap stents and the lower the residue SYNTAX score in the high RE group. The Spearman test showed that RE correlated with rSS (P < 0.001) but did not correlate with bSS (P = 0.249).
Table 1.
Baseline Clinical and Angiographic Characteristics of Patients Stratified by Revascularization Extent
| Low RE, n = 185 | Intermediate RE, n = 186 | High RE, n = 187 | P Value | |
|---|---|---|---|---|
| Revascularization extent | 0.21 ± 0.10 | 0.54 ± 0.09 | 0.87 ± 0.10 | <0.001 |
| Age, y | 63.68 ± 10.96 | 61.39 ± 10.05 | 58.26 ± 9.51 | <0.001 |
| Male, n (%) | 140 (75.7%) | 138 (74.2%) | 137 (73.3%) | 0.866 |
| Type 2 diabetes mellitus, n (%) | 69 (37.3%) | 64 (34.4%) | 50 (26.7%) | 0.081 |
| Prior stroke, n (%) | 21 (11.4%) | 23 (12.4%) | 10 (5.3%) | 0.046 |
| PVD, n (%) | 6 (3.2%) | 6 (3.2%) | 3 (1.6%) | 0.532 |
| Prior MI, n (%) | 33 (17.8%) | 23 (12.4%) | 15 (8.0%) | 0.017 |
| Hypertension, n (%) | 138 (74.6%) | 134 (72.0%) | 115 (61.5%) | 0.015 |
| Dyslipidemia, n (%) | 57 (30.8%) | 55 (29.6%) | 65 (34.8%) | 0.531 |
| Smoking history, n (%) | 72 (38.9%) | 74 (39.8%) | 81 (43.3%) | 0.658 |
| Family history, n (%) | 18 (9.7%) | 20 (10.8%) | 36 (19.3%) | 0.012 |
| LVEF (%) | 63.97 ± 7.88 | 62.43 ± 7.89 | 63.37 ± 8.68 | 0.188 |
| GFR | 82.58 ± 27.95 | 85.53 ± 26.37 | 87.39 ± 24.02 | 0.205 |
| Glycoprotein IIb/IIIa, n (%) | 23 (12.4%) | 43 (23.1%) | 42 (22.5%) | 0.014 |
| LMWH, n (%) | 140 (75.7%) | 129 (69.4%) | 154 (82.4%) | 0.013 |
| β‐Blocker, n (%) | 146 (78.9%) | 144 (77.4%) | 149 (79.7%) | 0.864 |
| ACEI/ARB, n (%) | 105 (56.8%) | 120 (64.5%) | 93 (49.7%) | 0.015 |
| Statin, n (%) | 180 (97.3%) | 175 (94.1%) | 180 (96.3%) | 0.283 |
| bSS | 28.21 ± 12.73 | 27.74 ± 11.55 | 26.51 ± 9.56 | 0.330 |
| 0 < bSS ≤ 22 | 65 | 70 | 63 | 0.723 |
| 23 ≤ bSS ≤ 32 | 63 | 47 | 72 | 0.021 |
| bSS ≥ 33 | 57 (30.8%) | 69 (37.1%) | 52 (27.8%) | 0.146 |
| bSS ≥ 23 | 120 | 116 | 124 | 0.723 |
| Residue SYNTAX score | 22.58 ± 11.29 | 12.69 ± 5.88 | 3.51 ± 3.30 | <0.001 |
| Total occlusion, n (%) | 66 (35.7%) | 48 (25.8%) | 51 (27.3%) | 0.080 |
| Bifurcation lesions, n (%) | 147 (79.5%) | 141 (75.8%) | 161 (86.1%) | 0.040 |
| Small vessels, n (%) | 99 (53.5%) | 96 (51.6%) | 98 (52.4%) | 0.935 |
| Overlap stents, n (%) | 53 (28.6%) | 75 (40.3%) | 106 (56.7%) | <0.001 |
| Total length of stents, mm | 39.92 ± 23.12 | 55.68 ± 32.87 | 69.75 ± 37.25 | <0.001 |
| No. of stents | 1.71 ± 0.80 | 2.25 ± 1.24 | 2.91 ± 1.54 | <0.001 |
| Targeted LAD, n (%) | 71 (38.4%) | 141 (75.8%) | 157 (84%) | <0.001 |
| Targeted LCX, n (%) | 75 (40.5%) | 63 (33.9%) | 85 (45.5%) | 0.072 |
| Targeted RCA, n (%) | 78 (42.2%) | 76 (40.9%) | 104 (55.6%) | 0.007 |
Abbreviations: ACEI, angiotensin‐converting enzyme inhibitor; ARB, angiotensin receptor blocker; bSS, baseline SYNTAX score; GFR, glomerular filtration rate; LAD, left anterior descending; LCX, left circumflex; LMWH, low‐molecular‐weight heparin; LVEF, left ventricular eject fraction; MI, myocardial infarction; PVD, peripheral vascular disease; RCA, right coronary artery; RE, revascularization extent; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery.
The clinical outcomes according to the RE tertiles are shown in Table 2 and Figure 2. The incidence of MACE significantly increased as RE increased (13.3%, 31.4%, and 44.1%, log‐rank P < 0.001). The same trend was observed for incidence of target vessel failure (TVF) (8.8%, 20.3%, and 28.4%, log‐rank P < 0.001), repeat revascularization (11.8%, 26.2%, and 35.6%, log‐rank P < 0.001), and MI (1.1%, 2.9%, and 12.6%, log‐rank P < 0.001). Both univariate and multivariate analyses confirmed RE as the independent risk factor for MACE, TVF, repeat revascularization, and MI. No significant differences were observed in the incidence of all‐cause death and cardiac death among the 3 groups.
Table 2.
Clinical Outcomes According to Different Revascularization Extent
| Clinical Outcomes | Crude | Multivariate Adjusted | |||
|---|---|---|---|---|---|
| HR (95% CI) | P Value | HR (95% CI) | P Value | ||
| MACE | a: 21 (13.3%) | ||||
| b: 52 (31.4%) | 2.721 (1.639‐4.516) | <0.001 | 2.928 (1.753‐4.892) | <0.001a | |
| c: 68 (44.1%) | 3.586 (2.197‐5.852) | <0.001 | 3.864 (2.331‐6.404) | <0.001a | |
| TVF | a: 12 (8.8%) | ||||
| b: 34 (20.3%) | 3.007 (1.557‐5.808) | 0.001 | 3.426 (1.754‐6.691) | <0.001b | |
| c: 48 (28.4%) | 4.354 (2.313‐8.198) | <0.001 | 4.890 (2.538‐9.423) | <0.001b | |
| RR | a: 18 (11.8%) | ||||
| b: 44 (26.2%) | 2.672 (1.544‐4.626) | <0.001 | 2.330 (1.310‐4.145) | 0.004c | |
| c: 51 (35.6%) | 3.048 (1.780‐5.217) | <0.001 | 2.175 (1.199‐3.948) | 0.011c | |
| Cardiac death | a: 3 (1.6%) | ||||
| b: 8 (5.6%) | 2.617 (0.694‐9.864) | 0.155 | 2.799 (0.734‐10.675) | 0.132d | |
| c: 8 (4.3%) | 2.690 (0.714‐10.140) | 0.144 | 2.990 (0.759‐11.780) | 0.117d | |
| All‐cause death | a: 6 (3.2%) | ||||
| b: 13 (8.4%) | 2.134 (0.811‐5.615) | 0.125 | 2.118 (0.778‐5.766) | 0.142e | |
| c: 10 (5.4%) | 1.673 (0.608‐4.604) | 0.319 | 1.594 (0.545‐4.664) | 0.394e | |
| MI | a: 2 (1.1%) | ||||
| b: 5 (2.9%) | 2.491 (0.483‐12.838) | 0.275 | 2.569 (0.495‐13.333) | 0.261f | |
| c: 23 (12.6%) | 11.809 (2.784‐50.088) | 0.001 | 13.744 (3.140‐60.162) | 0.001f | |
| Cardiac death or MI | a: 5 (2.7%) | ||||
| b: 13 (8.5%) | 2.584 (0.921‐7.249) | 0.071 | 2.703 (0.959‐7.624) | 0.060g | |
| c: 23 (12.6%) | 4.761 (1.810‐12.524) | 0.002 | 5.258 (1.943‐14.230) | 0.001g | |
Abbreviations: ACEI, angiotensin‐converting enzyme inhibitor; ARB, angiotensin receptor blocker; CI, confidence interval; HR, hazard ratio; LVEF, left ventricular eject fraction; MACE, major adverse cardiac event; MI, myocardial infarction; RR, repeat revascularization; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery; TVF, target vessel failure.
a: low revascularization extent (reference); b: intermediate revascularization extent; c: high revascularization extent.
Adjusted factors are specifically described in Table 3.
Adjusted for age, gender, LVEF, hypertension, diabetes mellitus, prior MI, SYNTAX score, and ACEI/ARB.
Adjusted for age, gender, hypertension, diabetes mellitus, dyslipidemia, smoking history, prior MI, total length of stents, SYNTAX score and statin.
Adjusted for age, hypertension, and LVEF.
Adjusted for age, hypertension, diabetes mellitus, and prior MI.
Adjusted for age, hypertension, diabetes mellitus, and SYNTAX score.
Adjusted for age, hypertension, diabetes mellitus, and SYNTAX score.
Figure 2.
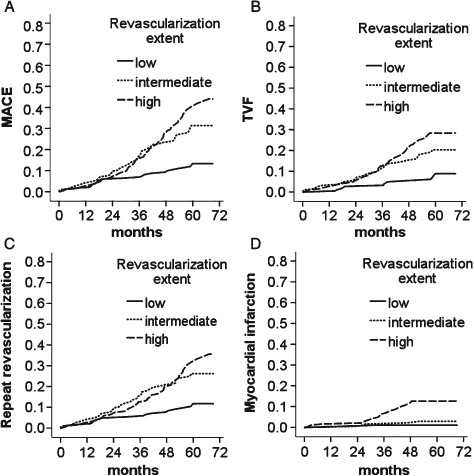
Cumulative incidences of outcomes according to tertiles of revascularization extent. Kaplan‐Meier incidence curves of clinical outcomes in patients with stable angina pectoris and 3‐vessel disease. (A) MACE, Kaplan‐Meier P < 0.001. (B) TVF, Kaplan‐Meier P < 0.001. (C) Repeat revascularization, Kaplan‐Meier P < 0.001. (D) Myocardial infarction, Kaplan‐Meier P < 0.001. Abbreviations: MACE, major adverse cardiovascular event; TVF, target vessel failure.
Table 3 displays univariate and multivariate analyses of predictors of MACE. Univariate analyses of predictors of MACE showed revascularization extent, age, male gender family history, residue SYNTAX score, bifurcation lesions, small vessels, length of stents, number of stents, and targeted LAD and RCA are independent risk factors of MACE. Multivariate analyses demonstrated revascularization extent, age, male gender, baseline SYNTAX score, and hypertension are independent risk factors of MACE. Both univariate and multivariate analyses showed use of ACEI/ARB and the increasing of ejection fraction were protective factors that prevented MACE.
Table 3.
Univariate and Multivariate Analyses of Predictors of Major Adverse Cardiac Events
| Variable | P Value | HR | 95% CI |
|---|---|---|---|
| Univariate analysis | |||
| Revascularization extent | <0.001 | 1.762 | 1.421‐2.184 |
| Age | 0.020 | 0.982 | 0.966‐0.9947 |
| Male gender | 0.011 | 1.757 | 1.140‐2.709 |
| EF | <0.001 | 0.956 | 0.934‐0.977 |
| Type 2 diabetes mellitus | 0.533 | 0.892 | 0.623‐1.277 |
| Hypertension | 0.126 | 1.344 | 0.920‐1.962 |
| Hyperlipidemia | 0.126 | 1.308 | 0.927‐1.845 |
| Smoking history | 0.096 | 1.325 | 0.952‐1.846 |
| Family history | 0.029 | 1.608 | 1.050‐2.464 |
| GFR | 0.348 | 0.997 | 0.991‐1.003 |
| Glycoprotein IIb/IIIa | 0.853 | 0.961 | 0.632‐1.462 |
| LMWH | 0.900 | 1.026 | 0.692‐1.521 |
| β‐Blocker | 0.239 | 0.795 | 0.542‐1.165 |
| ACEI/ARB | 0.004 | 0.613 | 0.440‐0.853 |
| Statin | 0.565 | 0.787 | 0.347‐1.783 |
| Baseline SYNTAX score | 0.056 | 1.014 | 1.000‐1.028 |
| Residue SYNTAX score | 0.009 | 0.976 | 0.958‐0.994 |
| Total occlusion | 0.297 | 1.208 | 0.847‐1.721 |
| Bifurcation lesions | <0.001 | 2.081 | 1.460‐2.966 |
| Small vessels | <0.001 | 2.401 | 1.673‐3.445 |
| Overlap stents | 0.249 | 1.215 | 0.872‐1.692 |
| Length of stents | <0.001 | 1.008 | 1.004‐1.013 |
| No. of stents | <0.001 | 1.275 | 1.144‐1.421 |
| Targeted LAD | 0.004 | 1.809 | 1.208‐2.708 |
| Targeted LCX | 0.251 | 0.818 | 0.581‐1.153 |
| Targeted RCA | 0.002 | 1.688 | 1.208‐2.359 |
| Multivariate analysis | |||
| Revascularization extent | <0.001 | 1.822 | 1.456‐2.278 |
| Age | 0.323 | 0.991 | 0.973‐1.009 |
| Male gender | 0.003 | 2.036 | 1.284‐3.230 |
| EF | <0.001 | 0.950 | 0.928‐0.974 |
| Baseline SYNTAX score | 0.001 | 1.027 | 1.011‐1.043 |
| Type 2 diabetes mellitus | 0.651 | 0.915 | 0.625‐1.342 |
| Hypertension | 0.001 | 2.014 | 1.327‐3.057 |
| Dyslipidemia | 0.491 | 1.142 | 0.783‐1.665 |
| Family history | 0.907 | 0.971 | 0.595‐1.585 |
| ACEI/ARB | 0.003 | 0.588 | 0.412‐0.837 |
Abbreviations: ACEI, angiotensin‐converting enzyme inhibitor; ARB, angiotensin receptor blocker; CI, confidence interval; EF, ejection fraction; GFR, glomerular filtration rate; HR, hazard ratio; LAD, left anterior descending; LCX, left circumflex; LMWH, low‐molecular‐weight heparin; RCA, right coronary artery; SYNTAX, Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery.
The propensity score analysis showed similar outcomes as the results demonstrated above (Appendix).
Discussion
To our knowledge, this is the first study to investigate the impact of IR after PCI with a DES on patients with SAP and 3‐vessel disease. Moreover, RE was first used to quantify the degree of revascularization. The main findings of our study were as follows: (1) RE is an independent risk factor for MACE, TVF, repeat revascularization and MI in patients presenting with SAP and 3‐vessel disease who underwent PCI; (2) With increasing RE, patients presenting with SAP and 3‐vessel disease suffered more MACE, TVF, and repeat revascularization and MI.
The prognostic implications of complete revascularization in patients with multivessel disease have been widely reported in PCI series with mixed results.1, 2, 3, 4, 5, 6 However, the above studies did not discriminate the indications for PCI. As a result, SAP and ACS were pooled together.13 The PCI strategy differs between SAP and ACS. The culprit lesion was always given the priority for treatment in patients with ACS, whereas in stable patients, PCI is dictated by anatomical factors. Recently, Rosner et al7 reported that in patients with ACS after PCI, IR was strongly associated with 1‐year MI, ischemia‐driven unplanned revascularization, and MACEs. However, this result applies only to patients with non–ST‐segment elevation ACS, and it is unknown whether the impact of incomplete revascularization would be similar in patients with SAP. Furthermore, as IR was defined as a binary outcome, we cannot directly assess the extent to which residue ischemia explains the relationship between incomplete revascularization and subsequent adverse outcomes. Therefore, Généreux et al8 used the residue SYNTAX score to quantify and to risk‐stratify the degree and complexity of residual stenosis after PCI, showing that rSS >8.0 after PCI in patients with moderate‐ and high‐risk ACS is associated with poor prognosis.
As mentioned above, the impact of IR on patients with SAP is uncertain. Although the 2 patients with bSS 30 and 10, both with rSS values of 8 after PCI, were pooled together due their identical rSS values, they exhibited distinct anatomical characteristics. We argue that the rSS was not sufficiently thorough to reflect the extent of revascularization. The algorithm to assess RE was defined as (bSS − rSS)/bSS and appeared more appropriate in terms of reflecting the extent of revascularization. Therefore, our study was aimed at evaluating the effect of RE on patients with SAP and 3‐vessel disease after PCI.
The results were not anticipated. With increasing RE, the incidence of adverse outcomes also increased. Previous studies have reported that the effect of incomplete revascularization is either detrimental or neutral, but no studies have reported results that are similar to ours. Thus, the results should be interpreted with caution. Several reasons might account for the results. First, special subsets of patients might contribute to this result. The COURAGE (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation) randomized controlled trial randomized 2287 patients with stable coronary artery disease to optimal medical therapy (OMT) alone or to OMT + PCI.14 At a median follow‐up period of 4.6 years, there was no significant difference in the incidences of death, MI, or a composite of death, MI, stroke, or hospitalization for unstable angina between the 2 groups. The result of the COURAGE study implied that patients with stable coronary artery disease benefit little from PCI compared to OMT. FAME (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation), another milestone study, randomly assigned 1005 patients with multivessel CAD to undergo PCI with implantation of DESs as guided by angiography alone or guided by fractional flow reserve (FFR) measurements in addition to angiography.15 The 1‐year results indicated that routine measurement of FFR significantly reduces the rate of the composite end point of death, nonfatal MI, and repeat revascularization. The measurement of FFR reminded us that stenosis does not necessarily indicate ischemia. The PCI of a stenotic, ischemia‐noninducing lesion might increase the chance of an adverse event that is attributed to the risk of thrombosis and restenosis associated with the placement of the stent. Based on the results of COURAGE and FAME, we speculate that for SAP, the expected benefit of aggressive and extensive revascularization is limited, and that such limited benefit might be outweighed by disadvantages of PCI, particularly when stents are placed in non–ischemia‐producing lesions. Previous studies mixed with SAP and ACS patients can confound the association of the relationship between the degree of revascularization and clinical outcome. Second, the adverse prognoses of patients with IR in prior studies might in part be attributed to more extensive diffuse coronary atherosclerosis. In our study, however, the baseline SYNTAX score was balanced among the 3 groups, increasing the comparability. Third, the initial use of RE in our study might also be responsible for the results. The quantification of the degree of revascularization other than the binary variety used in prior studies to reclassify many patients might more favorably represent the extent of revascularization. Fourth, the longer follow‐up period allows (56.9 months; IQR, 52.1–63.6 months) for the observation of significantly more information. Late adverse events, such as very late ST and late target lesion revascularization, are continuous hazards that can last at a minimum up to 5 years after the implantation of DESs.16 Most of previous studies are limited by short follow‐up periods. As shown in our study (Figure 2), the incidences of adverse outcomes were similar among different tertiles at the beginning of the observation period, whereas differences started to appear at 24 months. Increasing late adverse events associated with more stents in the higher RE group might be observed in longer follow‐up periods (>2 years).
Our results have demonstrated that extensive revascularization by PCI for the treatment of SAP offered no benefit, but rather, could be detrimental. The measurement of FFR to guide the PCI strategy in patients with stable ischemic heart disease has been previously shown to be warranted.17 As shown in Table 3, the use of ACEI/ARB significantly reduced the occurrence of MACE, emphasizing the crucial role of optimal medicinal therapy in patients with SAP, consistent with the latest guidelines.18
There were several limitations in our study. First, the results should be considered as hypothesis generating. Although multivariable analyses were conducted to adjust for potential confounders, residue confounding might still exist. Second, results might vary if bSS and rSS were assessed by core lab technicians using quantitative coronary angiography. Third, we did not prospectively collect the surgeon's intention for the extent of revascularization. Although exercise tests, radionuclide scintigraphy, or stress echocardiography might provide information to guide the surgeon's decision regarding PCI strategy, exactly which lesion should be treated was unclear due to lack of FFR measurement.19 Fourth, we did not assess the angina relief using some quantitative measurement such as the Seattle Angina Questionnaire. Angina relief should have been chosen as a end point when we studied patients with stable angina who underwent PCI.
Conclusion
For patients with SAP and 3‐vessel coronary artery disease, an increased extent of revascularization resulted in a less favorable prognosis.
Table A1.
Baseline Clinical and Angiographic Characteristics of Patients Stratified by Revascularization Extent
| Low RE, n = 128 | Intermediate RE, n = 128 | High RE, n = 128 | P Value | |
|---|---|---|---|---|
| Revascularization extent | 0.21 ± 0.10 | 0.55 ± 0.09 | 0.88 ± 0.10 | <0.001 |
| Age, y | 60.46 ± 10.66 | 58.47 ± 9.29 | 58.88 ± 9.83 | 0.240 |
| Male, n (%) | 96 (75.0%) | 104 (81.3%) | 90 (70.3%) | 0.124 |
| Type 2 diabetes mellitus, n (%) | 51 (39.8%) | 53 (41.4%) | 38 (29.7%) | 0.108 |
| Prior stroke, n (%) | 17 (13.3%) | 15 (11.7%) | 10 (7.8%) | 0.353 |
| PVD, n (%) | 6 (4.7%) | 6 (4.7%) | 3 (2.3%) | 0.536 |
| Prior MI, n (%) | 15 (11.7%) | 11 (8.6%) | 6 (4.7%) | 0.125 |
| Hypertension, n (%) | 90 (70.3%) | 87 (68.0%) | 90 (70.3%) | 0.895 |
| Dyslipidemia, n (%) | 39 (30.5%) | 35 (27.3%) | 44 (34.4%) | 0.474 |
| Smoking history, n (%) | 56 (43.8%) | 61 (47.7%) | 54 (42.2%) | 0.663 |
| Family history, n (%) | 15 (11.7%) | 17 (13.3%) | 19 (14.8%) | 0.762 |
| LVEF (%) | 63.85 ± 7.05 | 63.50 ± 7.68 | 63.10 ± 8.95 | 0.751 |
| GFR | 83.58 ± 30.67 | 86.70 ± 28.64 | 86.56 ± 25.64 | 0.610 |
| Glycoprotein IIb/IIIa, n (%) | 16 (12.5%) | 37 (28.9%) | 25 (19.5%) | 0.005 |
| LMWH, n (%) | 91 (71.1%) | 88 (68.8%) | 103 (80.5%) | 0.080 |
| β‐Blocker, n (%) | 97 (75.8%) | 97 (75.8%) | 102 (79.7%) | 0.692 |
| ACEI/ARB, n (%) | 82 (64.1%) | 73 (57.0%) | 75 (58.6%) | 0.484 |
| Statin, n (%) | 124 (96.9%) | 119 (93.0%) | 121 (94.5%) | 0.367 |
| Baseline SYNTAX score | 26.92 ± 10.80 | 27.83 ± 11.48 | 26.93 ± 9.08 | 0.728 |
| Residue SYNTAX score | 21.33 ± 9.35 | 12.56 ± 5.76 | 3.36 ± 3.01 | <0.001 |
| Total occlusion, n (%) | 45 (35.2%) | 25 (19.5%) | 35 (27.3%) | 0.020 |
| Bifurcation lesions, n (%) | 98 (76.6%) | 109 (85.2%) | 108 (84.4%) | 0.141 |
| Small vessels, n (%) | 67 (52.3%) | 71 (55.5%) | 74 (57.8%) | 0.677 |
| Overlap stents, n (%) | 39 (30.5%) | 51 (39.8%) | 77 (60.2%) | <0.001 |
| Total length of stents, mm | 36.90 ± 21.14 | 55.28 ± 33.92 | 73.23 ± 39.16 | <0.001 |
| No. of stents | 1.62 ± 0.74 | 2.25 ± 1.30 | 2.98 ± 1.57 | <0.001 |
| Targeted LAD, n (%) | 51 (39.8%) | 98 (76.6%) | 109 (85.2%) | <0.001 |
| Targeted LCX, n (%) | 54 (42.2%) | 43 (33.6%) | 61 (47.7%) | 0.070 |
| Targeted RCA, n (%) | 46 (35.9%) | 55 (43.0%) | 71 (55.5%) | 0.006 |
Abbreviations: ACEI, angiotensin‐converting enzyme inhibitor; ARB, angiotensin receptor blocker; GFR, glomerular filtration rate; LAD, left anterior descending; LCX, left circumflex; LMWH, low‐molecular‐weight heparin; LVEF, left ventricular eject fraction; MI, myocardial infarction; PVD, peripheral vascular disease; RCA, right coronary artery; RE, revascularization extent; SYNTAX, Synergy Between PCI with Taxus and Cardiac Surgery.
Table A2.
Clinical Outcomes According to Different Revascularisation Extents
| Clinical Outcomes | Tertiles According to RE | HR (95% CI) | P |
|---|---|---|---|
| MACE | Low RE (Ref) | ||
| Intermediate RE | 5.344 (2.677‐10.667) | <0.001 | |
| High RE | 5.477 (2.749‐10.914) | <0.001 | |
| TVF | Low RE (Ref) | ||
| Intermediate RE | 6.523 (2.272‐18.731) | <0.001 | |
| High RE | 8.541 (3.007‐24.256) | <0.001 | |
| RR | Low RE (Ref) | ||
| Intermediate RE | 4.762 (2.247‐10.089) | <0.001 | |
| High RE | 3.359 (1.512‐7.462) | 0.003 | |
| Cardiac death | Low RE (Ref) | ||
| Intermediate RE | 6.307 (0.754‐52.732) | 0.089 | |
| High RE | 8.642 (1.074‐69.509) | 0.043 | |
| All‐cause death | Low RE (Ref) | ||
| Intermediate RE | 4.430 (0.951‐20.625) | 0.058 | |
| High RE | 4.490 (0.973‐20.715) | 0.054 | |
| MI | Low RE (Ref) | ||
| Intermediate RE | 1.478 (0.244‐8.957) | 0.671 | |
| High RE | 7.651 (1.699‐34.444) | 0.008 | |
Abbreviations: CI, confidence interval; HR, hazards ratio; MACE, major adverse cardiac event; MI, myocardial infarction; RE, revascularization extent; RR, repeat revascularization; TVF, target vessel failure.
Adjusted factors were the same as the multivariable analysis in the Cox proportional hazards models in Table 2.
Drs Quan Li and Xianpeng Yu are joint first authors and contributed equally to the writing of this article.
The authors have no funding, financial relationships, or conflicts of interest to disclose.
References
- 1. Rogers WJ, Bourassa MG, Andrews TC, et al. Asymptomatic Cardiac Ischemia Pilot (ACIP) study: outcome at 1 year for patients with asymptomatic cardiac ischemia randomized to medical therapy or revascularization. The ACIP Investigators. J Am Coll Cardiol. 1995;26:594–605. [DOI] [PubMed] [Google Scholar]
- 2. Hannan EL, Racz M, Holmes DR, et al. Impact of completeness of percutaneous coronary intervention revascularization on long‐term outcomes in the stent era. Circulation. 2006;113:2406–2412. [DOI] [PubMed] [Google Scholar]
- 3. Hannan EL, Wu C, Walford G, et al. Incomplete revascularization in the era of drug‐eluting stents: impact on adverse outcomes. JACC Cardiovasc Interv. 2009;2:17–25. [DOI] [PubMed] [Google Scholar]
- 4. Kim YH, Park DW, Lee JY, et al. Impact of angiographic complete revascularization after drug‐eluting stent implantation or coronary artery bypass graft surgery for multivessel coronary artery disease. Circulation. 2011;123:2373–2381. [DOI] [PubMed] [Google Scholar]
- 5. van den Brand MJ, Rensing BJ, Morel MA, et al. The effect of completeness of revascularization on event‐free survival at one year in the ARTS trial. J Am Coll Cardiol. 2002;39:559–564. [DOI] [PubMed] [Google Scholar]
- 6. Shaw LJ, Berman DS, Maron DJ, et al; COURAGE Investigators . Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: results from the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial nuclear substudy. Circulation. 2008;117:1283–1291. [DOI] [PubMed] [Google Scholar]
- 7. Rosner GF, Kirtane AJ, Genereux P, et al. Impact of the presence and extent of incomplete angiographic revascularization after percutaneous coronary intervention in acute coronary syndromes: the Acute Catheterization and Urgent Intervention Triage Strategy (ACUITY) trial. Circulation. 2012;125:2613–2620. [DOI] [PubMed] [Google Scholar]
- 8. Généreux P, Palmerini T, Caixeta A, et al. Quantification and impact of untreated coronary artery disease after percutaneous coronary intervention: the residual SYNTAX (Synergy Between PCI with Taxus and Cardiac Surgery) score. J Am Coll Cardiol. 2012;59:2165–2174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Capodanno D, Chisari A, Giacoppo D, et al. Objectifying the impact of incomplete revascularization by repeat angiographic risk assessment with the residual SYNTAX score after left main coronary artery percutaneous coronary intervention. Catheter Cardiovasc Interv. 2013;82:333–340. [DOI] [PubMed] [Google Scholar]
- 10. Celik A, Karayakali M, Erkorkmaz U, et al. Presence of angina pectoris is related to extensive coronary artery disease in diabetic patients. Clin Cardiol. 2013;36:475–479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sianos G, Morel MA, Kappetein AP, et al. The SYNTAX score: an angiographic tool grading the complexity of coronary artery disease. EuroIntervention. 2005;1:219–227. [PubMed] [Google Scholar]
- 12. Thygesen K, Alpert JS, White HD; on behalf of the Joint ESC/ACCF/AHA/WHF Task Force for the Redefinition of Myocardial Infarction. Universal definition of myocardial infarction. Eur Heart J. 2007;28:2525–2538. [DOI] [PubMed] [Google Scholar]
- 13. Gossl M, Faxon DP, Bell MR, et al. Complete versus incomplete revascularization with coronary artery bypass graft or percutaneous intervention in stable coronary artery disease, Circ Cardiovasc Interv. 2012;5:597–604. [DOI] [PubMed] [Google Scholar]
- 14. Boden WE, O'Rourke RA, Teo KK, et al; COURAGE Trial Research Group . Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007;356:1503–1516. [DOI] [PubMed] [Google Scholar]
- 15. Tonino PA, De Bruyne B, Pijls NH, et al; FAME Study Investigators . Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360:213–224. [DOI] [PubMed] [Google Scholar]
- 16. Kimura T, Morimoto T, Nakagawa Y, et al. Very late stent thrombosis and late target lesion revascularization after sirolimus‐eluting stent implantation: five‐year outcome of the j‐Cypher Registry. Circulation. 2012;125:584–591. [DOI] [PubMed] [Google Scholar]
- 17. De Bruyne B, Pijls NH, Kalesan B, et al; FAME 2 Trial Investigators . Fractional flow reserve‐guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012;367:991–1001. [DOI] [PubMed] [Google Scholar]
- 18. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012;60:e44–e164. [DOI] [PubMed] [Google Scholar]
- 19. Iguchi T, Hasegawa T, Nishimura S, et al. Impact of lesion length on functional significance in intermediate coronary lesions. Clin Cardiol. 2013;36:172–177. [DOI] [PMC free article] [PubMed] [Google Scholar]


