Abstract
Background
Rates of trichinellosis have declined significantly in the United States due to improved pork production practices and public awareness of the danger of eating raw or undercooked pork. In April 2011, the Minnesota Department of Health received a report of presumptive trichinellosis in a 50-year-old man with a history of wild boar consumption. A public health investigation was initiated.
Methods
Medical record reviews and patient and family interviews were conducted. Trichinella species serology was performed on patient and family serum samples, and larval identification was attempted on clinical specimens and meat samples.
Results
The index patient harvested a wild boar from an Iowa game farm; he processed the meat after returning home and developed gastrointestinal symptoms 2 days later. Four days after his illness onset, all 5 family members consumed a roast from the boar. The index patient sought healthcare 4 times after illness onset before being definitively diagnosed with trichinellosis. Following initiation of albendazole therapy, the index patient developed atrial fibrillation. One additional family member who processed the raw meat was diagnosed with trichinellosis. Trichinella spiralis larvae were identified in wild boar meat samples.
Conclusions
Trichinellosis has long been recognized as a potential hazard of consuming undercooked wild carnivore meat, and historically has been associated with consumption of pork from domestic swine, but may be unfamiliar to practicing clinicians in the United States. Education of hunters and the broader population on the potential for trichinellosis and the importance of proper handling and cooking meat from wild or free-range animals needs to be reinforced.
Keywords: human trichinellosis, outbreak, Trichinella spiralis, pork, wild boar
Trichinellosis is a parasitic disease caused by ingestion of raw or undercooked meat containing encysted larvae of the genus Trichinella. Historically, cases in the United States have been associated with consumption of Trichinella-contaminated pork [1]. Reported cases declined dramatically during the second half of the 20th century following a ban on feeding uncooked garbage to commercial swine and movement of the US swine herd to modern production systems that virtually eliminate swine exposure to rodents and other wildlife [1–3]. Most cases in recent years have been associated with wild animal meat consumption including bear and feral swine [1, 4–6].
The prompt diagnosis of trichinellosis in humans can be difficult because most patients present with nonspecific signs and symptoms. Due to the decline in case numbers, most clinicians are unfamiliar with the disease, and infected patients may make repeated visits to medical facilities before being correctly diagnosed, leading to delayed treatment [2].
On 19 April 2011, the Minnesota Department of Health received a report from an infection preventionist at a local hospital of presumptive trichinellosis in a 50-year-old man with a history of wild boar consumption. A public health investigation was initiated to confirm the diagnosis, identify any additional cases, determine the source of infection, and implement public health control measures at the game farm of origin for the wild boar.
METHODS
Clinical and Epidemiologic Investigation
The index patient was interviewed with a standard questionnaire regarding illness signs and symptoms, meat preparation and consumption, and travel history in the previous 2 months. Additional ill persons identified by the index patient were also interviewed. Medical records from all ill individuals were reviewed.
Laboratory Testing
Laboratory testing of patient serum collected during routine patient care and the public health investigation was conducted at the Centers for Disease Control and Prevention (CDC) using an enzyme-linked immunosorbent assay (ELISA) (Scimedx, Inc, Denville, New Jersey) that detects Trichinella-specific antibodies to excretory/secretory antigens of T. spiralis [7].
Parasitological studies were performed on implicated meat products at the CDC, the United States Department of Agriculture (USDA) Agricultural Research Service, and the University of Minnesota. A wet mount was made from meat and examined microscopically for Trichinella larvae. Confirmation to the species level was performed using a multiplex polymerase chain reaction (PCR) based on amplification of regions of the internal transcribed spacers 1 and 2 and the expansion segment V region of the ribosomal RNA gene [8]. The larval concentration in several meat products was determined by counting the larvae found in 1-g samples of partially digested muscle [6]. Larvae infectivity was determined by orally inoculating larvae recovered from each tissue digestion into 5 Swiss-Webster mice. After 45 days, the mice were euthanized and examined for the presence of larvae by muscle tissue digestion.
RESULTS
Clinical and Epidemiologic Investigation
On 4 April 2011, the index patient presented to an urgent care clinic with a history of severe retro-orbital pain refractory to acetaminophen and ibuprofen therapy, and leg weakness lasting >1 week (Figure 1). Results of his physical exam, a complete blood count (CBC) without differential, and basic chemistry panel were unremarkable. He was diagnosed with viral syndrome, and treatment with over-the-counter decongestants was recommended. Two days later, he again presented to urgent care with worsening symptoms, burning sensations in his arms and legs, and joint and muscle pain. A rapid antigen detection test for group A Streptococcus was negative, and serum was submitted for Lyme disease serology due to recent tick exposure. He began doxycycline treatment for the presumptive diagnosis of Lyme disease.
Figure 1.

Timeline of events preceding and during an investigation of a trichinellosis outbreak associated with wild boar consumption. Abbreviations: ER, emergency room; GI, gastrointestinal; MDH, Minnesota Department of Health.
Four days later, he was hospitalized with 2 weeks of persistent retro-orbital headache, discomfort on movement of his eyes, fever, chills, and burning pain in his muscles, especially with flexing and extending his ankles or bending his knees. At that time, an infectious disease physician was consulted by telephone. Upon further questioning, the patient revealed he recently consumed wild boar meat. Physical examination demonstrated muscle tenderness, including pain in the masseter muscles and in the thighs. His pulse was slightly elevated at 100 beats per minute, and his temperature was 37.2°C (99°F). Initial laboratory testing results were notable for a normal peripheral total white blood cell (WBC) count (9200 cells/μL; expected range, 4500–11 000 cells/μL) with an eosinophilia (1400 cells/μL; expected range, 200–600 cells/μL), elevated creatine phosphokinase (CK) (887 IU/L; expected range, 49–397 IU/L), and elevated aspartate aminotransferase (AST) (101 IU/L; expected range, 15–41 IU/L), alkaline phosphatase (ALP) (194 IU/L; expected range, 38–126 IU/L), and alanine transaminase (ALT) (194 IU/L; expected range, 17–63 IU/L). Computed tomographic imaging of the head and abdomen and ultrasound of the liver were initially considered unremarkable. Antibody titers for Lyme disease, hepatitis A, B, and C viruses, mononucleosis, and Trichinella species and PCR for influenza A and B were negative. The index patient was empirically treated with methylprednisolone and a tapered prednisone regimen; his symptoms and clinical laboratory values began to improve within 24 hours of initiating therapy. He was discharged with a diagnosis of febrile illness and eosinophilia after a 3-day hospital stay.
The following day, the index patient returned to the emergency room with severe muscle pain and fever despite prednisone therapy. Physical exam again demonstrated generalized and persistent muscle tenderness. Laboratory testing results were notable for a normal total WBC count (9600 cells/μL), with a marked eosinophilia (3900 cells/μL), elevated myoglobin levels (612 ng/mL; expected range, 17.4–106 ng/mL), elevated CK level (792 IU/L), and elevated AST (115 IU/L), ALP (156 IU/L), and ALT (154 IU/L) levels. He was diagnosed with myositis and his prednisone dosage was increased.
Due to the patient’s concern about dermatomyositis, a condition a sibling had, an infectious disease consultant examined the patient the following day. A left sartorius muscle biopsy was performed and many Trichinella species larvae were observed when examined microscopically (Figure 2). Repeat examination of the head computed tomographic imaging demonstrated rectus eye muscle edema (Figure 3).
Figure 2.
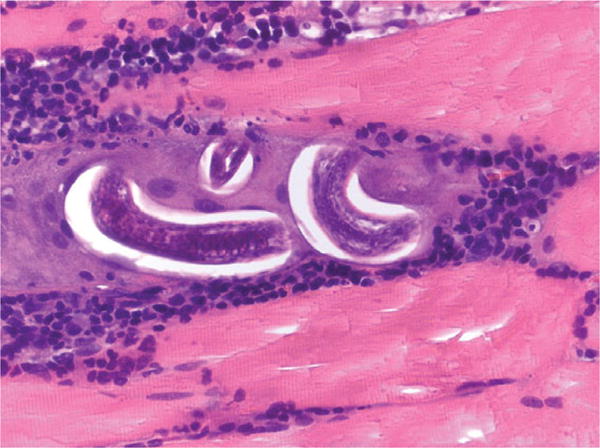
Trichinella larvae encysted in sartorius muscle of index patient (hematoxylin and eosin stain, magnification 40×).
Figure 3.
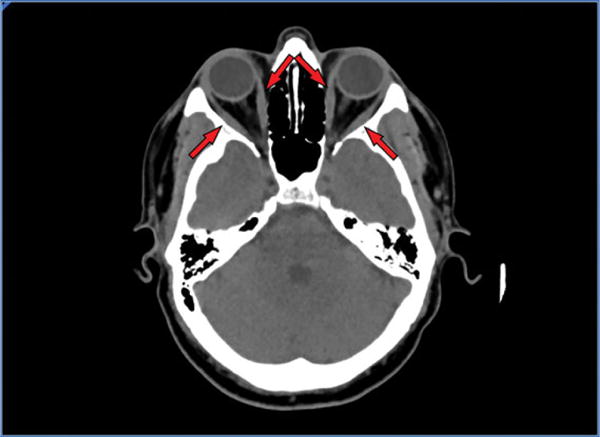
Computed tomographic imaging of the index patient’s head demonstrating swollen rectus muscles of the eye (as indicated by arrows).
Following the trichinellosis diagnosis, the index patient began albendazole treatment in addition to the prednisone therapy that preceded the diagnosis. The following day, he developed atrial fibrillation with a heart rate of 130 beats per minute. An initial dose of diltiazem failed to convert the atrial fibrillation to a normal sinus rhythm. Normal sinus rhythm was achieved by electrical cardioversion.
Patient interviews revealed that the index patient and his 10-year-old son had traveled to a wild game farm in Iowa in mid-March (Figure 1). At the farm, the index patient had killed a wild boar, which was then field-dressed by the farm staff, cut into quarters, and packed in coolers for transport. The day after returning home, he and 2 of his sons, including the son who went hunting, processed the meat into meal-sized portions. The index patient and his son who went hunting did not wear gloves, whereas his older son wore rubber gloves when cutting the raw meat. Both the patient and his younger son reported consuming food and drink while processing the meat. All of the meat was placed in a home freezer. The index patient reported developing gastrointestinal symptoms, including nausea and abdominal pain, 2 days following the meat processing. Four days after the index patient developed gastrointestinal symptoms, his wife prepared 2 wild boar roasts for the family. All 6 family members, including the index patient, consumed the roasts. The family reported the roasts were “well done” and “dry” but did not use a meat thermometer to record the internal cooking temperature. The index patient’s gastrointestinal symptoms continued. He then developed a headache 10 days later and sought the above-mentioned medical care 15 days following the meal.
The index-patient’s son who went on the hunting trip developed influenza-like symptoms including fever, headache, cough, and generalized malaise 2 days after the index patient first sought medical care (Figure 1). The son was seen in an outpatient clinic; diagnosed with influenza, pharyngitis, and bronchitis; and prescribed amoxicillin. In response to the outbreak investigation, the son was reexamined by the infectious disease specialist and found to have moderate calf tenderness. Laboratory results revealed a normal WBC count (9020 cells/μL; expected range, 4500–13 500 cells/μL) with eosinophilia (2600 cells/μL; expected range, 0–1000 cells/μL) and an elevated CK level (798 IU/L). He was treated with albendazole and a tapering prednisone regimen with no ill effects.
None of the remaining family members reported any signs or symptoms consistent with trichinellosis. All were examined concurrently to the younger son, and none had any remarkable exam or laboratory findings.
Laboratory Testing
All 6 family members consented to blood collection for Trichinella serology testing 1 month after wild boar consumption. The index patient and his younger son who went on the hunting trip, both of whom handled raw meat with their hands, were both positive for Trichinella species immunoglobulin G (IgG) antibody, despite the index patient having an initial negative test result. The remaining family members were seronegative and refused convalescent sample collection.
Tissue from the index patient’s muscle biopsy, along with a roast, breakfast sausage, and bratwurst from the wild boar, were submitted to the CDC for Trichinella identification. Encapsulated Trichinella larvae were visualized in the index patient’s muscle biopsy (Figure 2) and in a wet mount of the roast (Figure 4). The index patient’s biopsy specimen and samples of breakfast sausage and bratwurst were positive for T. spiralis by multiplex PCR. Tissue digestion was performed at both the USDA Agricultural Research Service and the University of Minnesota on 1-g samples of ham roast, breakfast sausage, and bratwurst. Larvae were identified from all samples, and digestion results varied from 12 to 36 larvae/g, with the highest concentration found in the bratwurst. Mouse inoculation results demonstrated that the larvae were not infective; however, the meat products had been frozen in the family freezer for 38 days at the time of sample collection, which may have killed the larvae.
Figure 4.
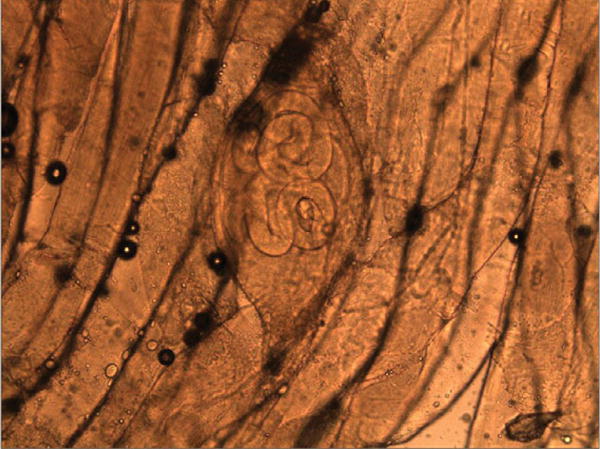
Trichinella larvae encysted in muscle tissue from wild boar ham roast (wet mount, magnification 40×).
Site Investigation
In May 2011, Iowa Meat and Poultry Bureau compliance officers visited the game farm and interviewed the owner about the hunting and carcass-handling processes. The farm was licensed by the Iowa Department of Natural Resources as a game preserve and offered guided hunts for elk, deer, Sitka deer, buffalo, wild rams, wild goats, and wild boars. Approximately 100 Russian wild boars were raised on 11 acres and fed a diet of soybean meal and corn. However, the animals could be cannibalistic if one of the wild boars died and the owner did not remove the carcass immediately. In addition, the boars had opportunities to consume other wildlife. The owner confirmed that farm staff bled out, eviscerated, and removed the hide after the animals were killed by the hunters. Additional meat processing was available on request, in violation of Iowa regulations. Once notified of this violation, the owner agreed to discontinue the further processing of wild boars and to warn future hunters about the risks of acquiring Trichinella infection when consuming wild boar by offering hunters trichinellosis fact sheets [9].
DISCUSSION
We report a human trichinellosis outbreak associated with wild boar meat harvested at a game farm. Although most recent reported US outbreaks have been associated with consumption of bear meat [1], this outbreak highlights the continued risk of trichinellosis from other types of game meat or any susceptible species raised outdoors where exposure to reservoir species can occur, including wild boar or domestic swine raised in a pasture setting. In addition, this outbreak suggests that cross-contamination during processing of the wild boar meat rather than consumption of undercooked meat was the exposure that led to the outbreak.
The clinical characteristics of this outbreak highlight the difficulty clinicians can have in recognizing patients infected with Trichinella so that appropriate treatment can be initiated. Patients will often have 2 phases of illness in acute infections. First, gastrointestinal symptoms including diarrhea and abdominal pain will present within 2 days following ingestion of larvae as the larvae migrate from the stomach to the small intestine where they mature and reproduce. The second phase of the illness corresponds with the migratory and muscular stages of the parasite, in which the newborn larvae migrate from the intestines to the striated skeletal muscles where they encyst and can remain viable for years to decades [2, 10]. In this phase, patients experience nonspecific influenza-like signs and symptoms including fever, headache, myalgias, and cough in addition to eyelid or facial edema [2]. Both the index patient and his younger son presented with a nonspecific illness that was initially misdiagnosed and resulted in unnecessary antimicrobial therapy and delayed appropriate treatment.
Clinical laboratory diagnostic testing can be useful in deciphering the nonspecific presentation of patients infected with Trichinella species. Eosinophilia appears early and in almost every case of trichinellosis, highlighting the importance of including a differential when ordering a CBC [11]. The index patient was evaluated twice before an eosinophil count was obtained while he was hospitalized. In addition, 75%–90% of patients will have elevated levels of muscle enzymes such as CK within 2–5 weeks postinfection [12]. The presence of both eosinophilia and elevated CK along with nonspecific influenza-like illness with or without gastrointestinal signs is highly suspicious of trichinellosis.
Serologic tests for Trichinella species are helpful to clinicians but are not sufficient to diagnose trichinellosis alone. IgG antibodies can be found within 12–60 days postinfection but the antibody response is dependent on the number of ingested larvae, the species involved, and the individual immune response [2, 13, 14]. Trichinella species IgG was not detected when the index patient initially was tested using a commercially available ELISA with reported sensitivity and specificity of 96.3% and 93.8%, respectively. A serum sample that was drawn 11 days later (35 days after symptom onset) was positive. Timing of serologic testing is critical to interpreting test results. One report based on an outbreak in Thailand demonstrated that T. spiralis IgG ELISA sensitivity reached 100% on the 50th day of infection [15], highlighting the importance of obtaining a second specimen if the first is negative but there remains a clinical suspicion of trichinellosis. Prior to seroconversion, compatible clinical signs and symptoms, exposure history, and, in severe cases, identification of Trichinella larvae in a muscle biopsy sample can provide a timely diagnosis.
Cardiac involvement is a rare but sometimes fatal complication of trichinellosis [16–18]. In a study of 154 patients diagnosed with trichinellosis, 18% had abnormalities of heart rate and rhythm. Sinus tachycardia was the most frequently identified abnormality, and 1 patient experienced atrial fibrillation [18]. Most patients appear to have rapid improvement of cardiac problems following the administration of antihelminthics and corticosteroids [17]. However, despite initial corticosteroid therapy, the index patient developed atrial fibrillation that required electrical cardioversion. The patient had no further cardiac episodes in the year following illness onset.
The patient’s atrial fibrillation began 1 day after albendazole administration for treatment of trichinellosis. A literature review of trichinellosis treatment demonstrated that although the usefulness of antihelminthics in later stages may be debatable [2, 19], there have been no reports of adverse events at any stage of infection when using these medications. Although it is impossible to establish a cause and effect relationship between the 2 events, further exploration into this possible complication may be warranted.
The exact exposure of the index patient and his son is unknown. Family member interviews revealed that the wild boar was processed into meal-sized packages at the index patient’s home 2 days after the animal was harvested from the Iowa game farm. Neither the index patient nor his ill son wore gloves during this process. Possible exposure during meat processing has been implicated in prior outbreaks. In a 1990 Virginia outbreak, a meat handler who developed trichinellosis worked at a pork processing plant where implicated pork sausage was produced but denied consuming the pork sausage [20]. Cross-contamination of other food products from contaminated meat has also been suggested as a possible route of exposure [6]. In the current outbreak, the family also consumed 2 roasts that were frozen for 6 days following processing. Freezing can kill encysted T. spiralis larvae in pork, including wild boar meat; however, meat cuts up to 15 cm in thickness need to be frozen at − 15°C (5°F) for a minimum of 3 weeks [2]. Because some Trichinella species are freeze-resistant given certain time/temperature/host animal combinations, cooking is the most reliable way to kill encysted larvae [21–23]. The USDA recommends that consumers cook wild game meat to an internal temperature of 71.1°C (160°F) [1, 2, 9]. The family reported cooking the roasts thoroughly; however, no internal temperature of the meat was taken. Reported attack rates in wild boar-associated trichinellosis outbreaks vary from 78% to 100% [13, 24, 25]. Based on the relatively low attack rate of 33% at the family meal and the fact that the index patient experienced gastrointestinal symptoms prior to the meal, we believe the more likely exposure was during the boar meat processing.
Outbreaks of trichinellosis following the consumption of feral swine or wild boar meat are not uncommon, and wild boar has been implicated as the source of sporadic Trichinella infections [1, 5, 13, 25–29]. There are an estimated 4 million feral swine, wild boar, or hybrids of the 2 types in the United States, and the popularity of hunting these animals is growing as their territory is stretching northward [30]. In addition, the popularity of wild boar and free-range pork products is growing. Meat products from these animals are increasingly being made available to consumers; however, pasture-based rearing practices increase the likelihood that these animals come into contact with wildlife or wildlife carcasses infected with Trichinella, compared with pigs raised in confinement settings with vector control management [1–3]. Farms that raise swine in pasture settings have been shown to be at increased risk for the introduction of infections from reservoir species [31].
CONCLUSIONS
Trichinellosis has long been recognized as a potential hazard of consuming undercooked pork and wild carnivore meat. Modern pork production practices have decreased the number of infections in the United States to the point where clinicians may not recognize the classic syndrome even in the face of suggestive exposure. In addition, growing interest in consuming wild boar and free-range pork has the potential to expose more individuals to this disease. Education of hunters, game farm owners, and the broader population on the potential for trichinellosis and the importance of proper processing, handling, and cooking of meat derived from wild boar, feral pigs, and commercially reared free-range pork needs to be reinforced.
Footnotes
Disclaimer. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
Potential conflicts of interest. All authors: No potential conflicts of interest.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Kennedy ED, Hall RL, Montgomery SP, Pyburn DG, Jones JL. Trichinellosis surveillance—United States, 2002–2007. MMWR Surveill Summ. 2009;58:1–7. [PubMed] [Google Scholar]
- 2.Gottstein B, Pozio E, Nockler K. Epidemiology, diagnosis, treatment, and control of trichinellosis. Clin Microbiol Rev. 2009;22:127–45. doi: 10.1128/CMR.00026-08. table of contents. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Davies PR. Intensive swine production and pork safety. Foodborne Pathog Dis. 2011;8:189–201. doi: 10.1089/fpd.2010.0717. [DOI] [PubMed] [Google Scholar]
- 4.Hill DE, Gamble HR, Zarlenga DS, Coss C, Finnigan J. Trichinella nativa in a black bear from Plymouth, New Hampshire. Vet Parasitol. 2005;132:143–6. doi: 10.1016/j.vetpar.2005.05.043. [DOI] [PubMed] [Google Scholar]
- 5.Roy SL, Lopez AS, Schantz PM. Trichinellosis surveillance—United States, 1997–2001. MMWR Surveill Summ. 2003;52:1–8. [PubMed] [Google Scholar]
- 6.Hall RL, Lindsay A, Hammond C, et al. Outbreak of human trichinellosis in northern California caused by Trichinella murrelli. Am J Trop Med Hyg. 2012;87:297–302. doi: 10.4269/ajtmh.2012.12-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wilson M, Schantz P, Nutman T. Molecular and immunological approaches to the diagnosis of parasitic infection. In: Detrick B, Hamilton R, Folds JD, editors. Manual of molecular and clinical laboratory immunology. Washington, DC: American Society for Microbiology; 2006. pp. 557–68. [Google Scholar]
- 8.Pozio E, La Rosa G. Trichinella. In: Liu D, editor. Molecular detection of human parasitic pathogens. Boca Raton, FL: CRC Press; 2012. pp. 691–703. [Google Scholar]
- 9.US Department of Agriculture. Game from farm to table. Available at: http://www.fsis.usda.gov/wps/portal/fsis/topics/food-safety-education/get-answers/food-safety-fact-sheets/meat-preparation/game-from-farm-to-table/CT_Index. Accessed 8 August 2013.
- 10.Froscher W, Gullotta F, Saathoff M, Tackmann W. Chronic trichinosis. Clinical, bioptic, serological and electromyographic observations. Eur Neurol. 1988;28:221–6. doi: 10.1159/000116271. [DOI] [PubMed] [Google Scholar]
- 11.Dupouy-Camet J, Kociecka W, Bruschi F, Bolas-Fernandez F, Pozio E. Opinion on the diagnosis and treatment of human trichinellosis. Expert Opin Pharmacother. 2002;3:1117–30. doi: 10.1517/14656566.3.8.1117. [DOI] [PubMed] [Google Scholar]
- 12.Capo VA, Despommier DD, Polvere RI. Trichinella spiralis: vascular endothelial growth factor is up-regulated within the nurse cell during the early phase of its formation. J Parasitol. 1998;84:209–14. [PubMed] [Google Scholar]
- 13.Pozio E, Varese P, Morales MA, Croppo GP, Pelliccia D, Bruschi F. Comparison of human trichinellosis caused by Trichinella spiralis and by Trichinella britovi. Am J Trop Med Hyg. 1993;48:568–75. doi: 10.4269/ajtmh.1993.48.568. [DOI] [PubMed] [Google Scholar]
- 14.Bruschi F, Tassi C, Pozio E. Parasite-specific antibody response in Trichinella sp. 3 human infection: a one year follow-up. Am J Trop Med Hyg. 1990;43:186–93. doi: 10.4269/ajtmh.1990.43.186. [DOI] [PubMed] [Google Scholar]
- 15.Morakote N, Sukhavat K, Khamboonruang C, Siriprasert V, Suphawitayanukul S, Thamasonthi W. Persistence of IgG, IgM, and IgE antibodies in human trichinosis. Trop Med Parasitol. 1992;43:167–9. [PubMed] [Google Scholar]
- 16.Hidron A, Vogenthaler N, Santos-Preciado JI, Rodriguez-Morales AJ, Franco-Paredes C, Rassi A., Jr Cardiac involvement with parasitic infections. Clin Microbiol Rev. 2010;23:324–49. doi: 10.1128/CMR.00054-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lazarevic AM, Neskovic AN, Goronja M, et al. Low incidence of cardiac abnormalities in treated trichinosis: a prospective study of 62 patients from a single-source outbreak. Am J Med. 1999;107:18–23. doi: 10.1016/s0002-9343(99)00161-8. [DOI] [PubMed] [Google Scholar]
- 18.Puljiz I, Beus A, Kuzman I, Seiwerth S. Electrocardiographic changes and myocarditis in trichinellosis: a retrospective study of 154 patients. Ann Trop Med Parasitol. 2005;99:403–11. doi: 10.1179/136485905X36307. [DOI] [PubMed] [Google Scholar]
- 19.Pozio E, Sacchini D, Sacchi L, Tamburrini A, Alberici F. Failure of mebendazole in the treatment of humans with Trichinella spiralis infection at the stage of encapsulating larvae. Clin Infect Dis. 2001;32:638–42. doi: 10.1086/318707. [DOI] [PubMed] [Google Scholar]
- 20.Trichinella spiralis infection—United States, 1990. MMWR Morb Mortal Wkly Rep. 1991;40:57–60. [PubMed] [Google Scholar]
- 21.Lacour SA, Heckmann A, Mace P, et al. Freeze-tolerance of Trichinella muscle larvae in experimentally infected wild boars. Vet Parasitol. 2013;194:175–8. doi: 10.1016/j.vetpar.2013.01.049. [DOI] [PubMed] [Google Scholar]
- 22.Kapel CM, Webster P, Malakauskas A, Hurnikova Z, Gamble HR. Freeze tolerance of nine Trichinella genotypes in muscle tissue of experimentally infected pigs, horses, wild boars, mice, cats, and foxes. XIth International Conference on Trichinellosis; 8–12 August 2004; San Diego, CA. [Google Scholar]
- 23.Pozio E, La Rosa G, Amati M. Factors influencing the resistance of Trichinella muscle larvae to freezing. In: Campbell CW, Pozio E, Bruschi F, editors. Trichinellosis. Rome, Italy: ISS Press; 1994. pp. 173–8. [Google Scholar]
- 24.Kim G, Choi MH, Kim JH, et al. An outbreak of trichinellosis with detection of Trichinella larvae in leftover wild boar meat. J Korean Med Sci. 2011;26:1630–3. doi: 10.3346/jkms.2011.26.12.1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rodriguez-Osorio M, Abad JM, de Haro T, Villa-Real R, Gomez-Garcia V. Human trichinellosis in southern Spain: serologic and epidemiologic study. Am J Trop Med Hyg. 1999;61:834–7. doi: 10.4269/ajtmh.1999.61.834. [DOI] [PubMed] [Google Scholar]
- 26.Meng XJ, Lindsay DS, Sriranganathan N. Wild boars as sources for infectious diseases in livestock and humans. Philos Trans R Soc Lond B Biol Sci. 2009;364:2697–707. doi: 10.1098/rstb.2009.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pozio E, Rinaldi L, Marucci G, et al. Hosts and habitats of Trichinella spiralis and Trichinella britovi in Europe. Int J Parasitol. 2009;39:71–9. doi: 10.1016/j.ijpara.2008.06.006. [DOI] [PubMed] [Google Scholar]
- 28.Murrell KD, Pozio E. Worldwide occurrence and impact of human trichinellosis, 1986–2009. Emerg Infect Dis. 2011;17:2194–202. doi: 10.3201/eid1712.110896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Greene Y, Padovani T, Rudroff JA, Hall R, Austin C, Vernon M, Centers for Disease Control and Prevention Trichinellosis caused by consumption of wild boar meat—Illinois, 2013. MMWR Morb Mortal Wkly Rep. 2014;63:451. [PMC free article] [PubMed] [Google Scholar]
- 30.US Department of Agriculture, Animal and Plant Health Inspection Service. (Agriculture Information Bulletin. No 799).Feral/wild pigs: potential problems for farmers and hunters. 2005 Available at: http://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5313597.pdf. Accessed 8 August 2013.
- 31.Burke R, Masuoka P, Murrell KD. Swine trichinella infection and geographic information system tools. Emerg Infect Dis. 2008;14:1109–11. doi: 10.3201/eid1407.071538. [DOI] [PMC free article] [PubMed] [Google Scholar]


