Abstract
Background
Early goal-directed therapy (EGDT) is used to reduce mortality from septic shock and could be used in early fluid resuscitation of acute respiratory distress syndrome (ARDS). The aim of the present study was to assess the effects of restrictive (RFR) and nonrestrictive fluid resuscitation (NRFR) on hemodynamics, oxygenation, pulmonary function, tissue perfusion, and inflammation in piglets with pulmonary or extrapulmonary ARDS (ARDSp and ARDSexp).
Material/Methods
Chinese miniature piglets (6–8 weeks; 15±1 kg) were randomly divided into 2 groups (n=12/group) for establishing ARDSp and ARDSexp models, and were further divided into 2 subgroups (n=6/subgroup) for performing RFR and NRFR. Piglets were anesthetized and hemodynamic, pulmonary, and oxygenation indicators were collected at different time points for 6 hours. The goal of EGDT was set for PiCCO parameters (mean arterial pressure (MAP), urine output and cardiac index (CI), and central venous oxygen saturation (ScvO2).
Results
Piglets under RFR had lower urine output compared with NRFR, as well as lower total fluid volume (P<0.05). EVLW was decreased in ARDSp+RFR and NRFR, as well as in ARDSexp+RFR, but EVLW increased in ARDSexp+NRFR (P<0.05). PaO2/FiO2 decreased in ARDSp using both methods, but was higher with RFR (P<0.05), and was increased in ARDSexp+RFR. Other pulmonary indicators were comparable. The anti-inflammatory cytokines IL-10 and LXA4 were increased in ARDSexp after RFR (P<0.05), but not in the other groups.
Conclusions
RFR led to better oxygenation in ARDSp and ARDSexp compared with NRFR, but fluid restriction improved oxygenation in ARDSexp only.
Keywords: Climatotherapy; Pulmonary Ventilation; Respiratory Distress Syndrome, Adult; Resuscitation
Background
Acute respiratory distress syndrome (ARDS) is an acute inflammatory response that compromises the alveolar-capillary membrane integrity [1] and is characterized by bilateral pulmonary infiltrates and severe hypoxemia in the absence of evidence of cardiogenic pulmonary edema [2]. ARDS is associated with significant mortality (26–58%) [3–5]. ARDS may be classified based on pulmonary (ARDSp) or extrapulmonary causes (ARDSexp). The etiology, pathophysiology, treatments, and outcomes of ARDSp and ARDSexp are different [6–8]. Since ARDSp mainly show lung consolidation and because ARDSexp predominantly show interstitial edema and alveolar collapse [6], the 2 diseases might not respond in the same manner to fluid resuscitation, and appropriate fluid management is critical [9].
Whether restrictive fluid resuscitation (RFR) could be used in the treatment of ARDS is still controversial [9,10]. RFR could improve pulmonary function in ARDSexp by reducing capillary hydrostatic pressure and relieving high permeability-induced interstitial edema [10]. However, because ARDSp is manifested by alveolar exudate, low lung compliance and low lung capacity, RFR could cause a reduction in effective circulation and further aggravate the ventilation-perfusion imbalance [10].
Early goal-directed therapy (EGDT) can correct hemodynamic abnormalities and alleviate tissue hypoxia during the early phases of the disease [11]. EGDT could also be used in early fluid resuscitation of ARDS [12]. According to the strategy proposed by ARDSnet [13], using mean arterial pressure (MAP), central venous oxygen saturation (ScvO2) and urine output as targets of EGDT resuscitation would better distinguish and guide the use of RFR and NRFR.
sICAM-1 overexpression is a major factor contributing to capillary endothelial cell injury, increasing vascular permeability and blood barrier damage [14]. SP-A reflects the status of alveolar epithelial barrier injury [15]. The intact alveolar-capillary barrier could inhibit SP-A from entering the blood, thus only a trace amount of SP-A could be detected in the serum. In turn, the normal alveolar-capillary barrier can inhibit serum sICAM-1 from entering the pulmonary interstitium and alveoli, but damaged alveolar-capillary barrier may result in bidirectional leak of both SP-A and intravascular substances.
Therefore, a hypothesis was raised: RFR and NRFR lead to different oxygenation parameters in ARDSp and ARDSexp. Therefore, the aim of the present study was to assess the effects of RFR and NRFR on hemodynamics, oxygenation, pulmonary function, tissue perfusion and inflammation in piglet models of ARDSp or ARDSexp. This study may help to suggest new criteria for clinical selection of individualized early fluid therapy for ARDS.
Material and Methods
Experimental animals and grouping
Twenty-four Chinese miniature piglets (6–8 weeks; 15±1 kg; Huangpi experimental station of the national technology system of pig industry, Hubei, China) were randomized into 2 groups (n=12/group) for establishing ARDSp (group A) and ARDSexp (group B) models. Pigs in both groups were further randomized into 2 subgroups (n=6/subgroup) for performing RFR (groups A1 and B1) or NRFR (groups A2 and B2). All randomization processes were performed using random number tables. All procedures and animal experiments were approved by the Animal Care and Use Committee of the Tongji Medical College, Huazhong University of Science and Technology (Hubei, China).
Establishment of ARDS models
Anesthesia was induced by injection of ketamine IM (4 mg/kg), midazolam IV (0.3 mg/kg) and propofol IV (2 mg/kg), and maintained by continuous infusion of 2% propofol (0.3–0.4 mg/kg/h) and fentanyl (8 μg/kg/h). Vecuronium (0.1 mg/kg) was injected to make sure that the animals were not breathing on their own. The trachea was orally intubated and the animals were spontaneously breathing during the instrumentation period. After the instrumentation period, the animals were not spontaneously breathing.
A Healthcare 16448 Ventilator (Viasis Healthcare, USA) was used to control breathing, and the initial respiratory parameters were set as tidal volume of 8–10 mL/kg, respiratory rate of 25 bpm, FiO2 40% and positive end-respiratory pressure (PEEP) of 0 cmH2O, and partial pressure of carbon dioxide (PaCO2) was maintained within 35–45 mmHg. During induction of the lung injury, the ventilator settings were adjusted to: tidal volume 6 mL/kg, FiO2 1.0 and PEEP 5 cmH2O. After induction of the lung injury, PEEP was increased to 10 cmH2O, which was indicative of ARDS [16–18]. Oxygenation was also verified and had to be representative of ARDS [16–18].
Since the pig is a superior mammal that shares many physiological similarities with humans, the use of the PiCCO system in pigs might be warranted. The PiCCO system has been used in pigs in a number of studies [19,20]. The right femoral artery was isolated. A PiCCO (PV2013L07; Pulsion Medical Systems, Fedkirchen, Germany) was placed and connected to an IntelliVue MP60 monitor (Philips, Best, The Netherlands) for monitoring heart rate (HR), MAP, and cardiac index (CI). Ice-cold normal saline (15–20 ml) was injected into the central venous catheter rapidly for measuring cardiac output (CO) and extravascular lung water (EVLW). Partial arterial oxygen pressure (PaO2), arterial oxygen saturation (SaO2), venous oxygen saturation (SvO2) and hemoglobin (Hb) were assessed to evaluate oxygen delivery (DO2), oxygen uptake (VO2), and oxygen extraction ratio (O2ER). Each data were tested thrice and the mean value was used.
The right internal jugular vein was isolated. A catheter (CS-14502, Arrow International Inc., Asheboro, NC, USA) was placed for monitoring CVP. A PiCCO probe was connected. A tonometry catheter (16G; Datex Ohmeda, Helsinki, Finland) was introduced via the mouth through the esophagus into the stomach, and was connected to a tonometer (Datex Ohmeda, Helsinki, Finland) for detecting gastric intramural pH (pHi). pHi has been shown to reflect gastric perfusion [21,22]. In the present study, pHi was used to reflect the effect of fluid resuscitation [22]. Cystostomy was performed to monitor hourly urine output.
The ARDSp model was established by a single intratracheal injection of hydrochloric acid (Chemical Reagent Factory of Xinyang, Henan, China) at a low dose (0.1 mol/L, 3.5 mL/kg) [1]. The ARDSexp model was established by the IV injection of oleic acid (101064923; Sigma, St Louis, MO, USA) at a dose of 0.05 mL/kg [1]. Modeling was completed within 30 min. After lung modeling, piglets were observed for 90 min and their arterial blood gas was sampled every 15 min. Piglets showing a PaO2/FiO2 <200 mmHg twice within 30 min were considered as successful models.
Fluid resuscitation
Piglets received Ringer’s lactate solution IV before fluid resuscitation. The dose of Ringer’s lactate solution was calculated as: 4 ml/kg/h for the first 10kg of body weight; 2ml/kg/h for the second 10kg; and 1ml/kg/h for the remaining kg.
After successful model establishment, RFR piglets were infused with fluid at the initial rate if their CVP met the criteria (CVP=3 cmH2O). If CVP exceeded the criteria, the infusion rate was decreased by 50%, or furosemide was used for diuresis when urine output was <0.5 ml/kg/h. NRFR piglets were infused with fluid at the initial rate if their CVP met the criteria (CVP=10 cmH2O). If piglets’ CVP was lower than this value, the infusion rate was increased by 50% or above until the CVP reached the criteria.
EGDT
CVP was maintained at 3 cmH2O in the RFR groups (A1 and B1), and at 10 cmH2O in the NRFR groups (A2 and B2). The subsequent goal of fluid resuscitation was set as MAP ≥50 mmHg, urine output ≥0.5 mL/kg/h, CI ≥2.5 L/min/m2 and ScvO2 ≥70%. Piglets’ MAP, urine output and CI were continuously monitored, and their ScvO2 was detected every 30 min. Norepinephrine (0.5–1 μg/kg/min) was used if the MAP was lower than 50 mmHg. Dobutamine (2.5–5 μg/kg/min) was used if the CI was too low. Furosemide was used for diuresis when urine output was <0.5ml/kg/h. Increasing PEEP or vasoactive drugs was used to increase ScvO2.
Experimental data collection
The following indicators were recorded before lung injury (T0), after lung injury but before resuscitation (T1), and at 2 h (T2), 4 h (T3), and 6 h (T4) after resuscitation. Hemodynamic parameters (Table 1), pulmonary indicators (Table 2), tissue perfusion parameters (Table 2) and hepatic and kidney function (Table 3) were collected.
Table 1.
Hemodynamic changes before and after resuscitation in piglets with ARDSp or ARDSexp.
| Indicators | Group | Time point | ||||
|---|---|---|---|---|---|---|
| T0 | T1 | T2 | T3 | T4 | ||
| HR (bpm) | A1 | 87.00±5.76 | 109.00±5.48e | 113.00±5.08ae | 115.00±7.59ace | 122.00±5.12acef |
| A2 | 91.17±5.04 | 127.07±30.05e | 132.53±29.55e | 144.50±23.33e | 165.50±33.06ef | |
| B1 | 93.50±5.68 | 125.60±7.86e | 150.67±37.09e | 148.07±20.62e | 151.53±18.91ebf | |
| B2 | 86.80±10.67 | 133.20±9.70e | 121.77±8.75e | 123.60±17.04e | 130.00±14.87e | |
| MAP (mmHg) | A1 | 55.60±4.22 | 46.20±7.26 | 56.60±7.53f | 55.90±5.76a | 68.20±4.45a |
| A2 | 59.20± 7.93 | 49.80±8.93 | 64.60±1.02 | 62.20±11.67e | 69.40±11.46 | |
| B1 | 62.40±8.01 | 50.17±4.63e | 55.40±5.68 | 63.40±2.33e | 64.40±7.63e | |
| B2 | 54.73±5.11 | 51.53±8.03 | 62.80±11.90 | 63.20±29.96f | 62.60±8.78 | |
| CO (L/min) | A1 | 2.07±0.36c | 3.27±0.68e | 2.49±0.64a | 2.90±1.00 | 3.29±0.63e |
| A2 | 2.62±0.45 | 2.55±0.86 | 2.71±0.32 | 3.02±0.91 | 2.64±0.85 | |
| B1 | 2.30±0.40 | 4.09±1.29e | 3.65±1.22eb | 2.97±0.87f | 2.97±0.54f | |
| B2 | 2.37±0.53 | 1.86±0.47e | 1.77±0.35e | 2.07±0.18 | 2.40±0.26f | |
| CI (L/min/m2) | A1 | 5.43±1.29 | 5.94±1.40 | 4.61±1.18a | 5.36±1.86 | 5.29±1.81 |
| A2 | 4.95±0.93 | 4.72±1.59 | 5.02±0.60 | 5.12±1.75 | 4.52±1.61 | |
| B1 | 5.87±0.45 | 7.45±2.47 | 6.58±2.30bf | 5.29±1.56 | 5.51±1.01 | |
| B2 | 4.52±1.11 | 3.56±0.87e | 3.27±0.64e | 3.83±0.34 | 40.45±0.47f | |
| SV (ml/beat) | A1 | 28.20±3.07 | 29.67±4.62c | 23.32±3.60f | 24.74±6.42c | 21.84±3.48e |
| A2 | 27.45±1.96 | 20.20±4.50e | 21.18±2.30e | 18.06±6.36e | 17.62±3.80e | |
| B1 | 26.33±3.10 | 32.86±12.18 | 25.66±9.77 | 19.78±4.99f | 19.68±2.16f | |
| B2 | 26.74±2.86 | 16.02±6.14e | 15.72±5.24e | 17.00±1.31e | 23.08±7.54f | |
| SVV (%) | A1 | 13.50±2.59 | 8.60±2.87ec | 9.80±1.72a | 13.20±4.71f | 16.20±5.19f |
| A2 | 11.20±1.83 | 17.60±6.05e | 10.40±2.42f | 13.00±1.79f | 16.40±4.32e | |
| B1 | 16.40±4.96 | 13.00±4.56b | 18.00±5.76 | 15.00±1.26 | 17.40±6.53 | |
| B2 | 14.00±1.41 | 6.60±1.62e | 9.00±1.79e | 14.20±4.35f | 21.60±5.43ef | |
| SVR (dyn·s·cm−2) | A1 | 3893.72±314.70 | 2232.40±275.40e | 3076.30±236.84 | 2241.40±304.49 | 2376.90±340.78 |
| A2 | 3022.90±305.90 | 2737.30±286.30 | 2752.80±238.90 | 2258.30±324.10ef | 2628.80±237.90f | |
| B1 | 4017.39±320.60 | 1960.10±325.10e | 2339.70±301.10 | 3144.80±314.60 | 3178.50±290.80 | |
| B2 | 2984.39±291.30 | 3308.10±310.60 | 4113.00±287.90 | 4019.30±298.70ef | 2208.30±307.50f | |
| ITBV (ml) | A1 | 382.00±74.04 | 523.60±101.97ae | 440.20±68.40a | 493.40±124.14e | 427.00±89.80a |
| A2 | 348.20±34.98 | 421.00±56.73e | 470.20±69.32e | 396.40±69.86 | 396.00±49.29 | |
| B1 | 446.33±152.14 | 708.40±268.45e | 644.60±311.69e | 497.00±133.69 | 523.00±53.75e | |
| B2 | 399.60±68.90 | 540.60±60.69 | 341.60±72.44 | 372.20±67.16 | 396.40±44.41 | |
| GEDV (ml) | A1 | 622.40±21.47 | 441.60±104.30e | 374.88±77.44 | 440.10±122.03 | 397.06±94.56 |
| A2 | 696.96±10.14 | 359.52±68.10e | 398.88±78.18 | 531.88±78.61 | 566.15±62.15 | |
| B1 | 674.18±44.12 | 589.44±97.48e | 538.40±82.07 | 446.15±79.67 | 476.35±65.72 | |
| B2 | 319.68±19.98 | 455.20±71.27e | 296.00±80.67 | 320.48±76.45 | 399.84±58.25 | |
| PBV (ml) | A1 | 111.42±15.30 | 151.84±29.57 | 127.66±19.84 | 143.09±36.00 | 123.83±26.04 |
| A2 | 101.56±14.80 | 172.09±16.45 | 136.36±20.10 | 114.96±20.26 | 114.84±14.29 | |
| B1 | 130.18±25.90 | 205.44±77.85e | 186.93±90.39 | 144.13±38.77 | 151.67±15.59 | |
| B2 | 116.55±19.60 | 128.77±17.60 | 99.06±21.01 | 107.94±19.48 | 114.96±12.88 | |
| PVPI | A1 | 2.19±0.86 | 3.06±1.02 | 2.69±0.58 | 2.55±0.53 | 2.97±0.73 |
| A2 | 1.95±0.67 | 2.64±0.78 | 2.92±0.76 | 3.28±0.96 | 3.46±0.85 | |
| B1 | 1.72±0.63 | 2.23±0.54 | 2.09±0.44 | 2.64±0.65 | 1.92±0.36 | |
| B2 | 2.03±0.87 | 3.42±0.91 | 4.53±1.03 | 3.87±0.89 | 3.38±0.74 | |
| CFI (L·min·ml−1) | A1 | 3.33±0.49 | 7.41±0.93e | 6.64±0.59e | 6.59±0.86e | 8.29±1.03e |
| A2 | 3.76±0.58 | 7.09±0.78e | 6.79±0.76e | 5.68±0.63 | 4.66±0.73 | |
| B1 | 3.41±0.54 | 6.94±0.68e | 6.78±0.88e | 6.66±0.64e | 6.24±0.92e | |
| B2 | 7.41±0.81 | 4.09±0.37e | 5.98±0.73 | 6.46±0.72 | 6.00±0.81 | |
| GEF (%) | A1 | 18.12±3.72 | 26.88±5.46e | 24.88±5.02 | 22.49±4.26 | 22.00±4.81 |
| A2 | 15.75±3.63 | 22.47±3.87e | 21.24±4.72 | 13.58±3.77 | 12.45±3.99 | |
| B1 | 15.62±3.68 | 22.30±3.92e | 19.06±5.08 | 17.73±5.03 | 16.53±4.29 | |
| B2 | 33.46±2.87 | 14.08±4.56e | 21.24±5.33 | 21.22±5.21 | 23.09±5.31 | |
Data are shown as mean ±SD. A1 (n=6), B1 (n=5), A2 (n=5), B2 (n=6). Before modeling (T0), after modeling and before resuscitation (T1), 2 h (T2), 4 h (T3) and 6 h (T4) after resuscitation.
P<0.05 A1 vs. A2;
P<0.05 B1 vs. B2;
P<0.05 A1 vs. B1;
P<0.05 A2 vs. B2;
P<0.05 vs. T0 within the same group;
P<0.05 vs. T1 within the same group.
HR – heart rate; MAP – mean arterial pressure; CO – cardiac output; CI – Cardic Index; SV – stroke volume; SVV – stroke volume variation; SVR – systemic vascular resistance; ITBV – intrathoracic blood volume; GEDV – global end-diastolic end volume; PBV – pulmonary blood volume; PVPI – pulmonary vascular permeability index; CFI – cardiac function index; GEF – global ejection fraction.
Table 2.
Changes in pulmonary indicators and tissue perfusion parameters in piglets with ARDSp or ARDSexp during resuscitation.
| Indicators | EVLW | PVPI | PIP | Pplat | Lung compliance | DO2 (ml/min) | VO2 (ml/min) | O2ER | Lac (mmol/L) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| ARDSp | A1 | T1 | 464.00 ±91.30 | 3.33 ±0.57 | 28.60 ±2.42 | 398.27 ±65.94 | 113.49 ±20.49 | 0.30 ±0.04 | 1.94 ±0.33 | 14.40 ±4.18 | 7.80 ±0.40 |
| T2 | 343.60 ±71.66e | 3.35 ±0.63 | 30.20 ±2.40 | 382.20 ±61.87 | 115.62 ±17.55 | 0.27 ±0.09 | 1.84 ±0.22 | 16.20 ±2.63 | 8.00 ±0.63 | ||
| T3 | 365.40 ±54.58e | 3.80 ±2.40 | 32.90 ±1.02e | 406.00 ±35.50 | 96.14 ±11.31 | 0.30 ±0.03 | 1.96 ±0.15 | 15.80 ±2.40 | 7.80 ±2.40 | ||
| T4 | 368.20 ±83.98e | 3.76 ±0.78 | 33.60 ±2.32e | 403.23 ±102.03 | 102.29 ±15.56 | 0.29 ±0.01 | 1.77 ±0.23 | 15.00 ±0.80 | 6.60 ±0.80 | ||
| A2 | T1 | 453.80 ±93.34 | 3.74 ±0.23 | 27.30 ±2.64 | 412.88 ±66.22 | 109.80 ±53.99 | 0.32 ±0.16a | 1.16 ±0.30 | 13.80 ±2.30 | 7.00 ±1.79 | |
| T2 | 398.20 ±62.90e | 3.45 ±2.06 | 29.60 ±1.50 | 385.80 ±69.40 | 109.20 ±39.18 | 0.33 ±0.15 | 1.84 ±0.69 | 15.40 ±2.06 | 6.40 ±0.49a | ||
| T3 | 377.00 ±70.03e | 3.90 ±1.33 | 34.80 ±0.40e | 380.80 ±62.36a | 99.63 ±20.79 | 0.29 ±0.21 | 1.76 ±0.04 | 14.80 ±1.33 | 5.80 ±0.40a | ||
| T4 | 397.80 ±51.35 | 4.00 ±1.80 | 35.40 ±0.80e | 404.62 ±106.36 | 110.58 ±37.12 | 0.30 ±0.12 | 1.88 ±0.90 | 14.70 ±0.80 | 5.80 ±0.50 | ||
| ARDSexp | B1 | T1 | 457.20 ±84.93 | 3.89 ±1.12 | 29.60 ±1.36 | 448.49 ±68.04 | 167.45 ±62.31 | 0.35 ±0.14 | 1.95 ±0.17 | 12.60 ±1.36 | 7.00 ±0.00 |
| T2 | 391.20 ±58.27e | 3.81 ±0.63 | 32.80 ±2.79 | 390.88 ±38.91 | 188.47 ±38.27 | 0.29 ±0.07 | 1.98 ±0.27 | 15.83 ±1.63 | 11.40 ±2.06ec | ||
| T3 | 380.60 ±66.28ec | 4.15 ±2.40 | 34.40 ±3.72 | 423.81 ±25.39 | 163.86 ±23.37 | 0.32 ±0.13 | 1.08 ±0.26 | 16.18 ±2.40 | 9.80 ±1.33ec | ||
| T4 | 271.20 ±46.56ec | 3.76 ±0.80 | 35.20 ±2.40e | 430.60 ±62.03 | 138.20 ±199.52 | 0.49 ±0.27 | 1.92 ±0.24 | 16.69 ±0.80 | 7.60 ±0.80 | ||
| B2 | T1 | 439.80 ±58.33 | 3.74 ±1.56 | 29.20 ±2.40 | 382.47 ±16.75 | 176.60 ±21.24 | 0.27 ±0.08 | 1.52 ±0.47 | 12.90 ± 0.12 | 7.00 ±1.26 | |
| T2 | 448.40 ±76.88e | 3.84 ±2.06 | 33.40 ±1.02 | 419.83 ±29.76 | 170.21 ±15.16 | 0.32 ±0.06 | 1.98 ±0.27 | 16.40 ±2.06 | 7.60 ±1.36b | ||
| T3 | 418.20 ±124.29eb | 4.29 ±1.33e | 35.60 ±4.22e | 420.84 ±57.41 | 163.93 ±28.27 | 0.29 ±0.15 | 1.08 ±0.26 | 15.80 ±1.33 | 6.60 ±1.36b | ||
| T4 | 388.40 ±63.85eb | 4.16 ±0.80eb | 36.90 ±2.71e | 395.40 ±28.42 | 154.40 ±52.75 | 0.23 ±0.19 | 1.84 ±0.02 | 16.80 ±0.80 | 6.40 ±1.02 | ||
Data are shown as mean ±SD. A1 (n=6), B1 (n=5), A2 (n=5), B2 (n=6). Before resuscitation (T1), 2 h (T2), 4 h (T3) and 6 h (T4) after resuscitation.
P<0.05 A1 vs. A2;
P<0.05 B1 vs. B2;
P<0.05 A1 vs. B1;
P<0.05 A2 vs. B2;
P<0.05 vs. T1 within the same group.
EVLW – extravascular lung water; PVPI – pulmonary vascular permeability index; PIP – peak inspiratory pressure; Pplat – inspiratory plateau pressure; DO2 – oxygen delivery; VO2 – oxygen uptake; O2ER – oxygen extraction rate; Lac – lactic acid.
Table 3.
Changes in tissue perfusion indicators before and after resuscitation in piglets with ARDSp or ARDSexp.
| Indicators | TBIL (mmol/L) | DBIL (mmol/L) | ALT (U/L) | AST (U/L) | BUN (mmol/L) | Cr (μmol/L) | ||
|---|---|---|---|---|---|---|---|---|
| ARDSp | A1 | T1 | 2.2±0.9 | 0.8±0.5 | 41.6±9.3 | 33.3±6.2 | 5.2±0.2 | 44.2±10.1 |
| T4 | 2.5±0.9 | 1.5±0.3 | 45.3±10.1 | 36.0±8.2 | 4.8±2.7 | 43.1±2.0 | ||
| A2 | T1 | 2.6±0.5 | 1.1±0.5 | 50.4±4.5 | 30.3±4.8 | 5.0±0.3 | 39.4±11.8 | |
| T4 | 2.4±0.7 | 1.0±0.2 | 44.2±9.8 | 33.2±6.0 | 5.1±0.6 | 42.5±8.4 | ||
| ARDSexp | B1 | T1 | 1.8±0.9 | 1.1±0.5 | 51.8±12.3 | 38.3±9.2 | 4.3±0.6 | 34.2±12.0 |
| T4 | 2.1±0.9 | 1.0±0.2 | 51.4±10.1 | 36.9±12.0 | 4.8±2.4 | 33.1±2.0 | ||
| B2 | T1 | 1.6±0.5 | 0.9±0.1 | 47.2±9.5 | 29.5±7.2 | 5.8±0.2 | 42.4±16.8 | |
| T4 | 1.8±0.7 | 1.2±0.6 | 42.8±9.9 | 35.5±6.3 | 5.6±0.9 | 32.5±8.4 | ||
Data are shown as mean ±SD. A1 (n=6), B1 (n=5), A2 (n=5), B2 (n=6). Before resuscitation (T1), 2 h (T2), 4 h (T3) and 6 h (T4) after resuscitation. TBIL – total bilirubin; DBIL – direct bilirubin; ALT – alanine transminase; AST – aspartate transminase; BUN – blood urea nitrogen; Cr – creatinine.
At T1 and T4, piglets’ serum and bronchoalveolar lavage fluid (BALF) were sampled for detecting tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), interleukin-6 (IL-6), interleukin-10 (IL-10), lipoxin A4 (LXA4), soluble intercellular adhesion molecule-1 (sICAM-1), and surfactant protein A (SP-A) using a double antibody sandwich enzyme-linked immunosorbent assay (ELISA; Mai Bio Co., Ltd., Shanghai, China).
Statistical analysis
This was an observational study, and no power analysis was initially conducted because of the lack of data in piglets to base a power analysis on. Statistical analysis was performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA). All continuous data were tested for normality, and all data were normally distributed. Data are presented as mean ± standard deviation (SD). Analysis of variance (ANOVA) with the least significant difference (LSD) post hoc test was used for intergroup comparisons at the same time point, while repeated measures ANOVA was used for intragroup comparisons at different time points. Independent samples t-tests were used for intergroup comparisons, while paired t-tests were used for intragroup comparisons. Statistical significance was defined as P<0.05.
Results
Model success
During experiment, one piglet died in groups ARDSp+NRFR and ARDSexp+RFR. Piglets’ MAP, CI and urine output reached the criteria in the early phase of EGDT, but ScvO2 required longer time. One piglet in groups ARDSp+NRFR and ARDSexp+RFR failed to meet the criteria for ScvO2 at the end of EGDT. One piglet in groups ARDSp+RFR and ARDSexp+RFR had to receive furosemide; 1 piglet in groups ARDSp+NRFR and ARDSexp+RFR had to received dobutamine; however, both piglets died at the end.
Table 4 shows that RFR piglets had significantly lower urine output compared with NRFR (ARDSp: −15.4%; ARDSexp: −17.4%; P<0.05), as well as lower total fluid volume (ARDSp: −16.2%; ARDSexp: −16.2%; P<0.05).
Table 4.
Comparison of urine output, total fluid volume and time to achieve EGDT in ARDS piglets receiving RFR or NRFR.
| Group | n | Urine output (ml) | Time to achieve EGDT (h) | Total fluid volume (ml) |
|---|---|---|---|---|
| A1 | 6 | 445.50±17.78* | 4.53±1.63 | 559.67±25.80* |
| B1 | 5 | 433.00±15.03# | 4.90±1.10 | 556.50±29.17# |
| A2 | 5 | 527.00±9.24 | 4.87±1.51 | 667.50±20.92 |
| B2 | 6 | 524.00±9.45 | 5.10±1.10 | 663.83±23.27 |
Data are shown as means ± standard deviation (SD). Pulmonary acute respiratory distress syndrome (ARDSp) (group A) and extrapulmonary acute respiratory distress syndrome (ARDSexp) (group B) models (n=12/group). Restrictive fluid resuscitation (RFR) (group A1 and B1) and nonrestrictive fluid resuscitation (NRFR) (group A2 and B2) (n=6/subgroup). One piglet died in groups A2 and B1.
P<0.05 A1 vs. A2;
P<0.05 B1 vs. B2.
EGDT – early goal-directed therapy.
According to the EGDT settings, diuresis was strictly completed in all groups. PEEP was ≥10 cmH2O in all piglets, indicating successful modeling and ARDS diagnosis [16–18].
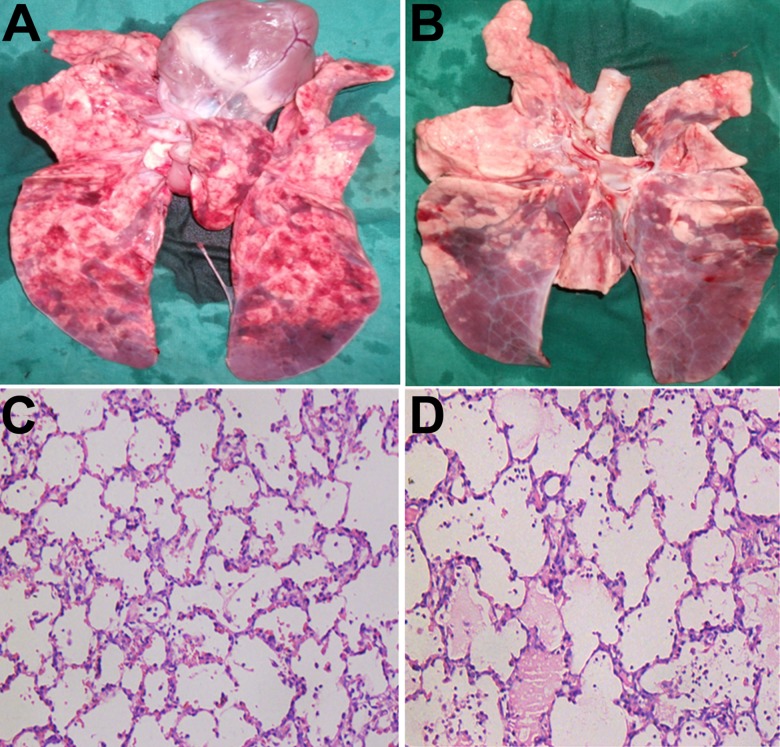
Figure 1 presents the postmortem pathological examination of representative piglets. In ARDSexp (Figure 1A), lungs showed diffuse pulmonary congestion and edema, as well as flower porphyritic changes. In ARDSp (Figure 1B), large red edema was observed. Histopathological examination of ARDSexp lung tissues showed relatively complete alveolar damage with septal oozing, few hemorrhage and a significant accumulation of neutrophiles (Figure 1C). ARDSp lung tissues showed severe alveolar damage, alveolar fusion, alveolar exudate, severe hemorrhage, an important recruitment of inflammatory cells and focal atelectasis (Figure 1D).
Figure 1.
Postmortem pathological examination of representative piglets. (A) Lungs from ARDSexp models showed diffuse pulmonary congestion and edema, as well as flower porphyritic changes. (B) Lungs from ARDSp models showed large red edema. (C) Histopathological examination of ARDSexp lung tissues showed relatively complete alveolar damage with septal oozing, few hemorrhage and a significant accumulation of neutrophiles. (D) Histopathological examination of ARDSp lung tissues showed severe alveolar damage, alveolar fusion, alveolar exudate, severe hemorrhage, an important recruitment of inflammatory cells and focal atelectasis. (HE staining, C,D ×200).
These results suggest that the piglet model was successful, and that the pathological changes were similar to those observed in the corresponding human conditions.
Hemodynamic changes
Table 1 shows that differences in heart rate changes were observed between groups. In the ARDSp groups, both methods increased heart rate from T1 to T4 (ARDSp+RFR: 109±6 to 122±5 bpm, P<0.05; ARDSp+NRFR: 127±30 to 166±33 bpm, P<0.05), but not mean arterial pressure. In ARDSexp piglets, heart rate was different only in the RFR group (ARDSexp+RFR: 126±8 to 152±19 bpm, P<0.05). At T4, heart rate was higher in ARDSp+NRFR compared with ARDSp+RFR (P<0.05), as well as in ARDSexp+RFR compared with ARDSexp+NRFR (P<0.05).
NRFR decreased systemic vascular resistance (SVR) in ARDSp and ARDSexp from T1 to T4 (−4.0% and −33.0%; P<0.05). Stroke volume variation (SVV) was increased from T1 to T4 in the ARDSp+RFR (+88.4%) and ARDSexp+NRFR (+227.3%) groups (P<0.05).
Stroke volume (SV) was decreased from T1 to T2 in group ARDSp+RFR (−21.5%, P<0.05). SV was decreased by −40.1% (P<0.05) from T1 to T4 in the ARDSexp+RFR group, but increased by 44.4% (P<0.05) from T1 to T4 in the ARDSexp+NRFR group (Table 1).
These results suggest that the 2 different fluid resuscitation methods achieve their effects through different means in the 2 disease models.
Pulmonary indicators
Table 2 shows that extravascular lung water in ARDSp and ARDSexp piglets was significantly increased (P<0.05). From T1 to T2, extravascular lung water was decreased in the ARDSp+RFR (−26.1%), ARDSp+NRFR (−12.3%) and ARDSexp+RFR (−14.4%) groups, but increased in ARDSexp+NRFR until T4 (+2.1%) (all P<0.05).
Peak inspiratory pressure (PIP) was increased in the ARDSp+RFR (+17.5%), ARDSp+NRFR (+29.7%), ARDSexp+RFR (+18.9%), and ARDSexp+NRFR (+26.4%) groups (P<0.05) during the whole experiment.
Lung compliance after establishment of the ARDSp and ARDSexp models was significantly decreased (P<0.05). Lung compliance improved in the ARDSexp+RFR group at T2 and T3, but decreased again by T4 (Table 2).
These results suggest that lung edema could be decreased by RFR in both diseases, but only in ARDSexp when using RFR.
Oxygenation
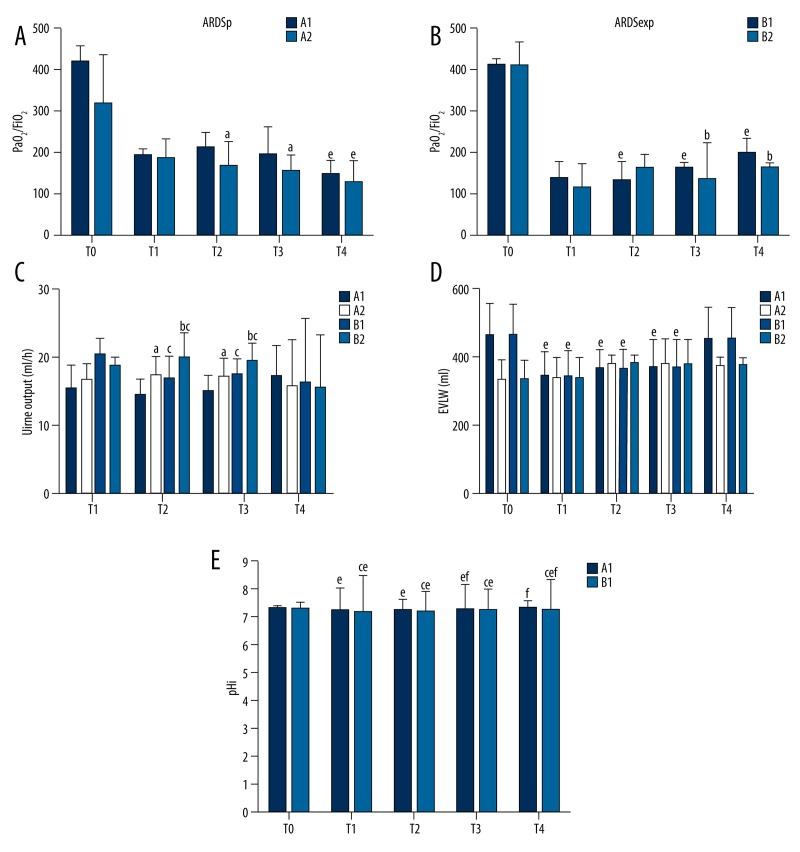
There were no changes in oxygenation parameters (P>0.05) (Table 2). PaO2/FiO2 was decreased from T1 to T4 in the ARDSp+RFR (−25.0%) and ARDSp+NRFR (−32.1%) groups, but increased in ARDSexp+RFR (+44.1%) (P<0.05) (Figure 2A, 2B). PaO2/FiO2 was lower in ARDSp+NRFR compared with ARDSp+RFR at T2 and T3 (P<0.05) (Figure 2A, 2B).
Figure 2.
Changes in oxygenation index, urine output, extravascular lung water (EVLW), and pHi during fluid resuscitation in ARDSp and ARDSexp piglets. (A) Pulmonary acute respiratory distress syndrome (ARDSp) (group A) and (B) extrapulmonary acute respiratory distress syndrome (ARDSexp) (group B) models (n=12/group). Restrictive fluid resuscitation (RFR) (group A1 and B1) and nonrestrictive fluid resuscitation (NRFR) (group A2 and B2) (n=6/subgroup). (C) Changes of urine output. (D) Changes of EVLW. (E) Changes in pHi. One piglet died in groups A2 and B1. Before modeling (T0), after modeling and before resuscitation (T1), at 2 h (T2), 4 h (T3) and 6 h (T4) after resuscitation. Data are shown as mean±standard deviation (SD). a P<0.05 A1 vs. A2; b P<0.05 B1 vs. B2; c P<0.05 A1 group vs. B1 group; e P<0.05 vs. T0 within the same group. f P<0.05 vs. T1 within the same group.
These results may suggest that RFR could improve oxygenation in ARDSexp.
Tissue perfusion indices
There were no differences in biochemistry (Table 3). To achieve EGDT, 1 piglet in each of groups ARDSp+RFR and ARDSexp+RFR received furosemide. Hourly urine output was increased with NRFR compared with RFR at T2 and T3 (all P<0.05) (Figure 2C). RFR decreased EVLW at T2 and T3 compared with T1 (all P<0.05). NRFR had no effect on EVLW (Figure 2D).
pHi was significantly decreased (P<0.05), indicating decreased tissue perfusion. During fluid resuscitation, pHi gradually recovered, and the increase in RFR was higher than in NRFR (P<0.05). Changes in pHi were more important in ARDSexp+RFR compared with ARDSp+RFR (Figure 2E).
Serum and BALF cytokines
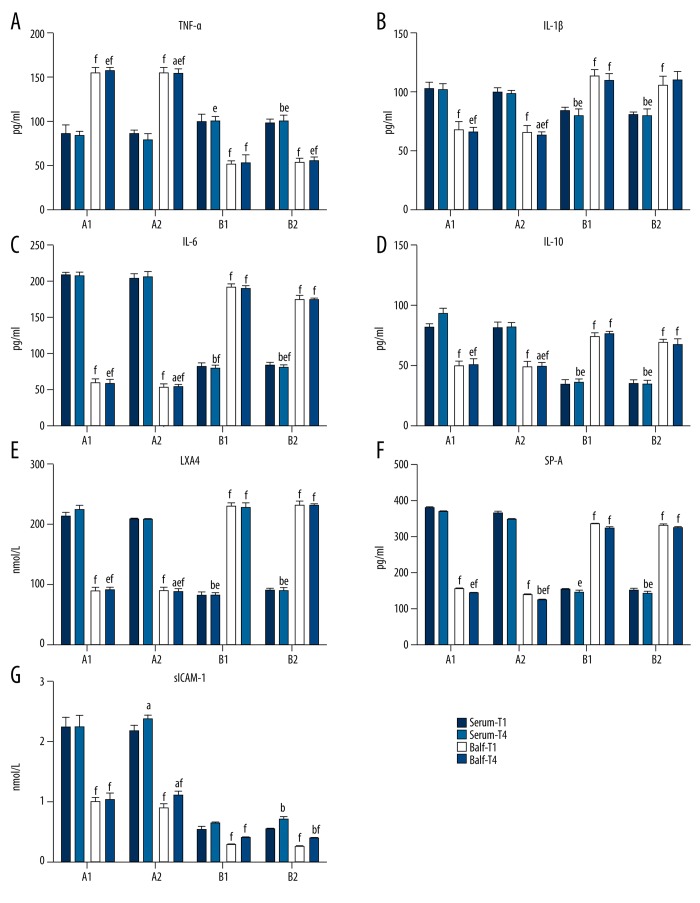
In ARDSp, all inflammatory parameters were higher in the serum compared with BALF (all P<0.05). On the other hand, BALF levels of these cytokines in ARDSexp were all higher compared with serum levels (all P<0.05) (Figure 3A–3F).
Figure 3.
Effects of early goal-directed therapy (EGDT)-guided fluid resuscitation on inflammatory cytokines in serum and bronchoalveolar lavage fluid (BALF) in piglets with ARDSp or ARDSexp. Inflammatory cytokines levels were determined by ELISA. (A) Tumor necrosis factor-α (TNF-α) levels. (B) Interleukin-1β (IL-1β) levels. (C) Interleukin-6 (IL-6) levels. (D) Interleukin-10 (IL-10) levels. (E) Lipoxin A4 (LXA4) levels. (F) Surfactant protein A (SP-A) levels. (G) Soluble intercellular adhesion molecule-1 (sICAM-1) levels. Data are shown as mean ±SD. A1 (n=6), B1 (n=5), A2 (n=5), B2 (n=6). a P<0.05 A1 vs. A2; b P<0.05 B1 vs. B2; e P<0.05 vs. T1 within the same group; f P<0.05 BALF vs. serum.
At T4, serum levels of TNF-α, IL-1β, IL-6, IL-10, LXA4 and SP-A in groups ARDSexp+RFR and ARDSexp+NRFR were significantly higher (P<0.05) than in groups ARDSp+RFR and ARDSp+NRFR. BALF levels of these cytokines in groups ARDSp+RFR and ARDSp+NRFR were significantly higher (P<0.05) than in ARDSexp+RFR and ARDSexp+NRFR (Figure 3A–3F).
Compared with T1, the serum and BALF levels of sICAM-1 at T4 were increased (P<0.05) in groups ARDSexp+NRFR, ARDSp+RFR, and ARDSp+NRFR (Figure 3G).
Discussion
Since ARDSp and ARDSexp are 2 distinct forms of the disease with distinct pathophysiology and treatments, it was necessary to compare the effects of RFR and NRFR between the 2 diseases. Piglets in the RFR groups had lower urine output compared with NRFR, as well as lower total fluid volume. EVLW was decreased in ARDSp under RFR and NRFR, as well as in ARDSexp under RFR, but NRFR increased EVLW in ARDSexp. PaO2/FiO2 decreased in ARDSp using both methods, but was higher with RFR and was increased in ARDSexp under RFR. Other pulmonary indicators were comparable. The anti-inflammatory cytokines IL-10 and LXA4 were increased in ARDSexp after RFR, but not in the other groups. Pathological changes were typical of ARDSexp and ARDSp [23,24].
Comparisons of the present study with previous studies might be difficult because of the vast heterogeneity in goals to achieve [25–31]. Furthermore, some of them examined the early outcomes [11,32], while other performed resuscitation later after ARDS onset [13]. In the present study, RFR and NRFR were performed in the early stage. Indeed, a previous study showed that early treatment of ARDS using fluids was very meaningful for prognosis [33].
In the present study, RFR did not significantly increase the incidence of shock and hypotension, which was consistent with a previous study [34]. Continuous monitoring of ITBV and EVLW could more accurately and timely reflect the changes in body fluid [35]. Monitoring ITBV and GEDV eliminates the interference of intrathoracic pressure and myocardial compliance, and would not be affected by vasoactive drugs. It is suggested that RFR would affect systemic capacity [35]. Therefore, changes in fluid distribution, decrease in peripheral vascular resistance, and insufficient tissue perfusion would be more obvious in piglets with ARDSexp. On the other hand, NRFR actively expanded blood volume, improving tissue perfusion.
In the present study, SV in RFR showed a decreasing trend. SVV is used to reflect the adequacy of fluid resuscitation and to show if less fluid should be provided [36]. Hemodynamic stability is an advantage of restrictive fluid resuscitation [37]. The RFR groups showed increased SVV at T4 compared with T1. As expected, RFR reduced the systemic blood volume as well as the venous blood return. However, NRFR increased the systemic blood volume, but may also result in excessive blood volume and increase the burden of systemic capacity and SVV. Moreover, these cardiac function indicators might be affected by diuretic and vasoactive drugs. After successful induction of ARDS, decrease in tissue oxygenation led to an increased HR as a compensatory mechanism to increase oxygen supply to the tissues and as a response to systemic stress via sympathetic response [38]. However, since no sophisticated cardiac monitoring was performed in the present study, further studies are necessary to address this issue.
We observed that although the CVP was strictly controlled to achieve EGDT, RFR could still possibly reduce cardiac preload and increase afterload. During EGDT, both MAP and CI reached the criteria at the early phase of resuscitation, while ScvO2 required a longer time. This might be attributed to the combined effects of pathological changes during ARDS, as well as to the use of diuretic and vasoactive drugs. Hence, results of the present study suggest that carrying out fluid management based on ARDS’s CVP may be inaccurate, and indices reflecting fluid response would be superior indicators in fluid resuscitation [39].
During RFR, the oxygenation indices in ARDSp gradually decreased, but EVLW did not vary significantly. Since the clearance of ARDSp-induced alveolar edema mainly depends on the active sodium-water transport system located on the alveolar epithelia [40], it was suggested that the failure of alveolar edema fluid clearance was primarily due to the ARDS-induced hypoxia causing alveolar damage [41]. Therefore, though fluid infusion was restricted, alveolar edema and subsequent pulmonary shunt and hypoxemia would not be alleviated. ARDSexp piglets receiving RFR showed decreased EVLW while they showed increased oxygenation indices. This may be due to the amount of infused fluid and the diuresis treatment that may reduce pulmonary vascular pressure.
Some authors showed that the use of RFR in patients with septic shock led to renal injury [32]. In the present study, the indicators of hepatic and renal function and urine output were assessed to evaluate the effects of RFR on the liver and kidneys. Results showed that piglets receiving RFR and NRFR displayed no significant differences in their hepatic and renal function indicators. This may be because resuscitation was terminated at a very early phase (6 h). The hourly urine output can sensitively reflect the status of renal perfusion. As EGDT was taken as a goal in resuscitation, piglets with urine output <0.5 mL/kg/h were treated with furosemide for diuresis. Two piglets in the RFR group were treated with furosemide, indicating that piglets may have had insufficient renal perfusion. However, in the present study, no markers of early kidney injury were measured, and the drugs used in the study could have affected kidney function. Therefore, further studies are necessary to address the effect of fluid resuscitation on the kidney function in ARDS.
pHi rapidly and accurately reflects the status of gastrointestinal perfusion [21,22]. During RFR, ARDSexp piglets showed significantly decreased pHi, which was not fully recovered at 6 h after resuscitation. In ARDSp piglets, pHi also decreased in the early phase of RFR, but pHi recovered within the normal range after resuscitation. Decreased pHi could be due to the hypoperfusion from relative hypovolemia. However, in the present study, adequate volume resuscitation was achieved in all pigs, leading to an adequate blood supply to the gastrointestinal tract. Therefore, pHi could adequately represent tissue oxygenation. It is speculated that the failure of pHi recovery in ARDSexp piglets may be due to insufficient gastrointestinal perfusion caused by systemic inflammation-induced vasoconstriction [42,43]. Early RFR in ARDSexp could improve systemic oxygenation, but it may also induce risks such as recessive compensatory shock or insufficient tissue perfusion.
The increase in EVLW is an important pathological change during ARDS, and is a primary factor causing decreased survival rate among patients with ARDS. Patients with ARDS with higher EVLW had increased mortality, and EVLW was positively associated with the severity of lung damage and negatively associated with lung compliance [44]. EVLW was also associated with the severity of ARDS, the number of days of mechanical ventilation, the number of days in intensive care unit, and the mortality of patients with ARDS [45]. Similarly to ARDS in humans, both ARDSp and ARDSexp piglets showed increased EVLW, PVPI, PIP, and Pplat, and they had decreased lung compliance. Such changes might be associated with the highly permeable alveolar or interstitial edema, which were caused by inhalation of hydrochloric acid or injection of oleic acid during modeling [46]. The alveolar edema-induced ventilation-perfusion imbalance and pulmonary shunt, and the interstitial edema-induced gas diffusion impairment, respectively, explained the decrease in oxygenation and the incidence of hypoxemia in both ARDSp and ARDSexp piglets [40,41].
In the ARDSexp group, cytokines levels were higher in the serum compared with BALF, while in the ARDSp group, cytokines levels were higher in BALF compared with serum. In ARDSp, the involvement of alveolar epithelia subsequently lead to alveolar exudate, reduced production and release of pulmonary surfactant, alveolar collapse and finally to lung consolidation [7]. In ARDSexp, it is caused by extrapulmonary factor-induced inflammatory mediator release, and it is the pulmonary response to systemic inflammation. Thus, ARDSexp is mainly manifested by increased endothelial cell permeability, interstitial pulmonary edema, and destruction of alveolar-capillary barrier [8]. The cytokine results observed in the present study are in accordance with these mechanisms. Indeed, in ARDSp, inflammatory parameters were higher in serum compared with BALF, while in ARDSexp these parameters were all higher in BALF compared with serum. Fluid resuscitation did not seem to have a beneficial effect on these parameters. However, the present study performed a short-term observation and the animals suffered from a serious disease. Therefore, it would be doubtful to observe beneficial effects of the treatments.
The present study suffers from some limitations. First, the number of animals in each group was relatively small. A retroactive power analysis showed that the power of comparing groups A1 and A2 was 11.4%, while power was 95.3% for comparing B1 and B2. Therefore, the results of the present preliminary study could be applied for ARDSexp, but further study in larger samples is necessary for ARDSp. Second, ARDS modeling using oleic acid or HCl have been shown to induce adequate models of ARDSp and ARDSexp [1], but they are not perfect. Nevertheless, PEEP was 10 cmH2O after modeling, which could be diagnosed as ARDS. Third, the present study was performed in the acute phase only, and long-term data are needed. Finally, even if some studies suggest that CVP should not be used to govern fluid resuscitation [47], CVP is still much used in China. Nevertheless, the PiCCO system was used in the present study, providing some data that could be used elsewhere.
Conclusions
In conclusion, ARDSexp and ARDSp might show better oxygenation response to RFR compared with NRFR, but oxygenation might be increased in ARSexp only. Results suggest that EVLW should not be used as an indication for RFR or NRFR. Few studies address the issue of fluid management in ARDS [9], but the present study provides some clues toward a better management of ARDS patients.
Footnotes
Source of support: This study was supported by the B. Braun Medical (Shanghai) International Trading Co., Ltd. research fund in 2011
Conflict of interest
The authors declare that they have no conflicts of interest.
References
- 1.Ballard-Croft C, Wang D, Sumpter LR, et al. Large-animal models of acute respiratory distress syndrome. Ann Thorac Surg. 2012;93:1331–39. doi: 10.1016/j.athoracsur.2011.06.107. [DOI] [PubMed] [Google Scholar]
- 2.Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–24. doi: 10.1164/ajrccm.149.3.7509706. [DOI] [PubMed] [Google Scholar]
- 3.Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–93. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- 4.Montgomery AB, Stager MA, Carrico CJ, Hudson LD. Causes of mortality in patients with the adult respiratory distress syndrome. Am Rev Respir Dis. 1985;132:485–89. doi: 10.1164/arrd.1985.132.3.485. [DOI] [PubMed] [Google Scholar]
- 5.Estenssoro E, Dubin A, Laffaire E, et al. Incidence, clinical course, and outcome in 217 patients with acute respiratory distress syndrome. Crit Care Med. 2002;30:2450–56. doi: 10.1097/00003246-200211000-00008. [DOI] [PubMed] [Google Scholar]
- 6.Gattinoni L, Pelosi P, Suter PM, et al. Acute respiratory distress syndrome caused by pulmonary and extrapulmonary disease. Different syndromes? Am J Respir Crit Care Med. 1998;158:3–11. doi: 10.1164/ajrccm.158.1.9708031. [DOI] [PubMed] [Google Scholar]
- 7.Modelska K, Pittet JF, Folkesson HG, et al. Acid-induced lung injury. Protective effect of anti-interleukin-8 pretreatment on alveolar epithelial barrier function in rabbits. Am J Respir Crit Care Med. 1999;160:1450–56. doi: 10.1164/ajrccm.160.5.9901096. [DOI] [PubMed] [Google Scholar]
- 8.Orfanos SE, Mavrommati I, Korovesi I, Roussos C. Pulmonary endothelium in acute lung injury: from basic science to the critically ill. Intensive Care Med. 2004;30:1702–14. doi: 10.1007/s00134-004-2370-x. [DOI] [PubMed] [Google Scholar]
- 9.Silva PL, Pelosi P, Rocco PR. Fluids in acute respiratory distress syndrome: pros and cons. Curr Opin Crit Care. 2014;20:104–12. doi: 10.1097/MCC.0000000000000049. [DOI] [PubMed] [Google Scholar]
- 10.Roch A, Guervilly C, Papazian L. Fluid management in acute lung injury and ards. Ann Intensive Care. 2011;1:16. doi: 10.1186/2110-5820-1-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med. 2001;345:1368–77. doi: 10.1056/NEJMoa010307. [DOI] [PubMed] [Google Scholar]
- 12.Dellinger RP, Levy MM, Carlet JM, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Intensive Care Med. 2008;34:17–60. doi: 10.1007/s00134-007-0934-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.National Heart L, Blood Institute Acute Respiratory Distress Syndrome Clinical Trials N. Wiedemann HP, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354:2564–75. doi: 10.1056/NEJMoa062200. [DOI] [PubMed] [Google Scholar]
- 14.Conner ER, Ware LB, Modin G, Matthay MA. Elevated pulmonary edema fluid concentrations of soluble intercellular adhesion molecule-1 in patients with acute lung injury: biological and clinical significance. Chest. 1999;116:83S–84S. doi: 10.1378/chest.116.suppl_1.83s. [DOI] [PubMed] [Google Scholar]
- 15.Zhang J. The effect of lung recruitment on extravascular lung water index and serum lung surfactant protein A in patients with ARDS. Chin J Crit Care Med. 2005;25:588–90. [Google Scholar]
- 16.Taylor MM. ARDS diagnosis and management: implications for the critical care nurse. Dimens Crit Care Nurs. 2005;24:197–207. doi: 10.1097/00003465-200509000-00001. quiz 208–9. [DOI] [PubMed] [Google Scholar]
- 17.Force ADT, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–33. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 18.Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med. 2012;38:1573–82. doi: 10.1007/s00134-012-2682-1. [DOI] [PubMed] [Google Scholar]
- 19.Imahase H, Inoue S, Sakamoto Y, et al. Comparison of PiCCO and VolumeView: simultaneous measurement in sepsis pig models. Crit Care. 2014;18:P143. [Google Scholar]
- 20.Liu XL, Tao YK, Yan ST, et al. Correlation of inferior vena cava respiratory variability index with central venous pressure and hemodynamic parameters in ventilated pigs with septic shock. Biomed Environ Sci. 2013;26:500–3. doi: 10.3967/0895-3988.2013.06.012. [DOI] [PubMed] [Google Scholar]
- 21.van Haren FM, Sleigh JW, Pickkers P, Van der Hoeven JG. Gastrointestinal perfusion in septic shock. Anaesth Intensive Care. 2007;35:679–94. doi: 10.1177/0310057X0703500505. [DOI] [PubMed] [Google Scholar]
- 22.Jakob SM, Parviainen I, Ruokonen E, et al. Tonometry revisited: perfusion-related, metabolic, and respiratory components of gastric mucosal acidosis in acute cardiorespiratory failure. Shock. 2008;29:543–48. doi: 10.1097/shk.0b013e31815d0c10. [DOI] [PubMed] [Google Scholar]
- 23.Tomashefski JF., Jr Pulmonary pathology of acute respiratory distress syndrome. Clin Chest Med. 2000;21:435–66. doi: 10.1016/s0272-5231(05)70158-1. [DOI] [PubMed] [Google Scholar]
- 24.Pelosi P, D’Onofrio D, Chiumello D, et al. Pulmonary and extrapulmonary acute respiratory distress syndrome are different. Eur Respir J Suppl. 2003;42:48s–56s. doi: 10.1183/09031936.03.00420803. [DOI] [PubMed] [Google Scholar]
- 25.Shoemaker WC, Appel PL, Kram HB, et al. Prospective trial of supranormal values of survivors as therapeutic goals in high-risk surgical patients. Chest. 1988;94:1176–86. doi: 10.1378/chest.94.6.1176. [DOI] [PubMed] [Google Scholar]
- 26.Martin C, Saux P, Eon B, et al. Septic shock: a goal-directed therapy using volume loading, dobutamine and/or norepinephrine. Acta Anaesthesiol Scand. 1990;34:413–17. doi: 10.1111/j.1399-6576.1990.tb03114.x. [DOI] [PubMed] [Google Scholar]
- 27.Fleming A, Bishop M, Shoemaker W, et al. Prospective trial of supranormal values as goals of resuscitation in severe trauma. Arch Surg. 1992;127:1175–79. doi: 10.1001/archsurg.1992.01420100033006. discussion 1179–81. [DOI] [PubMed] [Google Scholar]
- 28.Tuchschmidt J, Fried J, Astiz M, Rackow E. Elevation of cardiac output and oxygen delivery improves outcome in septic shock. Chest. 1992;102:216–20. doi: 10.1378/chest.102.1.216. [DOI] [PubMed] [Google Scholar]
- 29.Yu M, Levy MM, Smith P, et al. Effect of maximizing oxygen delivery on morbidity and mortality rates in critically ill patients: a prospective, randomized, controlled study. Crit Care Med. 1993;21:830–38. doi: 10.1097/00003246-199306000-00009. [DOI] [PubMed] [Google Scholar]
- 30.Gattinoni L, Brazzi L, Pelosi P, et al. A trial of goal-oriented hemodynamic therapy in critically ill patients. SvO2 Collaborative Group. N Engl J Med. 1995;333:1025–32. doi: 10.1056/NEJM199510193331601. [DOI] [PubMed] [Google Scholar]
- 31.Hayes MA, Timmins AC, Yau EH, et al. Elevation of systemic oxygen delivery in the treatment of critically ill patients. N Engl J Med. 1994;330:1717–22. doi: 10.1056/NEJM199406163302404. [DOI] [PubMed] [Google Scholar]
- 32.Rivers EP. Fluid-management strategies in acute lung injury – liberal, conservative, or both? N Engl J Med. 2006;354:2598–600. doi: 10.1056/NEJMe068105. [DOI] [PubMed] [Google Scholar]
- 33.Gattinoni L, Cressoni M, Brazzi L. Fluids in ARDS: from onset through recovery. Curr Opin Crit Care. 2014;20:373–77. doi: 10.1097/MCC.0000000000000105. [DOI] [PubMed] [Google Scholar]
- 34.Luecke T, Roth H, Herrmann P, et al. Assessment of cardiac preload and left ventricular function under increasing levels of positive end-expiratory pressure. Intensive Care Med. 2004;30:119–26. doi: 10.1007/s00134-003-1993-7. [DOI] [PubMed] [Google Scholar]
- 35.Giuliano KK, Scott SS, Brown V, Olson M. Backrest angle and cardiac output measurement in critically ill patients. Nurs Res. 2003;52:242–48. doi: 10.1097/00006199-200307000-00006. [DOI] [PubMed] [Google Scholar]
- 36.Berkenstadt H, Margalit N, Hadani M, et al. Stroke volume variation as a predictor of fluid responsiveness in patients undergoing brain surgery. Anesth Analg. 2001;92:984–89. doi: 10.1097/00000539-200104000-00034. [DOI] [PubMed] [Google Scholar]
- 37.Durairaj L, Schmidt GA. Fluid therapy in resuscitated sepsis: less is more. Chest. 2008;133:252–63. doi: 10.1378/chest.07-1496. [DOI] [PubMed] [Google Scholar]
- 38.McLellan SA, Walsh TS. Oxygen delivery and haemoglobin. Contin Educ Anaesth Crit Care Pain. 2004;4:123–26. [Google Scholar]
- 39.Kumar A, Anel R, Bunnell E, et al. Pulmonary artery occlusion pressure and central venous pressure fail to predict ventricular filling volume, cardiac performance, or the response to volume infusion in normal subjects. Crit Care Med. 2004;32:691–99. doi: 10.1097/01.ccm.0000114996.68110.c9. [DOI] [PubMed] [Google Scholar]
- 40.Matthay MA, Clerici C, Saumon G. Invited review: Active fluid clearance from the distal air spaces of the lung. J Appl Physiol (1985) 2002;93:1533–41. doi: 10.1152/japplphysiol.01210.2001. [DOI] [PubMed] [Google Scholar]
- 41.Rafii B, Tanswell AK, Otulakowski G, et al. O2-induced ENaC expression is associated with NF-kappaB activation and blocked by superoxide scavenger. Am J Physiol. 1998;275:L764–70. doi: 10.1152/ajplung.1998.275.4.L764. [DOI] [PubMed] [Google Scholar]
- 42.Ackland G, Grocott MP, Mythen MG. Understanding gastrointestinal perfusion in critical care: so near, and yet so far. Crit Care. 2000;4:269–81. doi: 10.1186/cc709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Schaper J, Ahmed R, Perschel FH, et al. Thoracic epidural anesthesia attenuates endotoxin-induced impairment of gastrointestinal organ perfusion. Anesthesiology. 2010;113:126–33. doi: 10.1097/ALN.0b013e3181de0fdd. [DOI] [PubMed] [Google Scholar]
- 44.Berkowitz DM, Danai PA, Eaton S, et al. Accurate characterization of extravascular lung water in acute respiratory distress syndrome. Crit Care Med. 2008;36:1803–9. doi: 10.1097/CCM.0b013e3181743eeb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Martin GS, Eaton S, Mealer M, Moss M. Extravascular lung water in patients with severe sepsis: a prospective cohort study. Crit Care. 2005;9:R74–82. doi: 10.1186/cc3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Monnet X, Anguel N, Osman D, et al. Assessing pulmonary permeability by transpulmonary thermodilution allows differentiation of hydrostatic pulmonary edema from ALI/ARDS. Intensive Care Med. 2007;33:448–53. doi: 10.1007/s00134-006-0498-6. [DOI] [PubMed] [Google Scholar]
- 47.Marik PE, Cavallazzi R. Does the central venous pressure predict fluid responsiveness? An updated meta-analysis and a plea for some common sense. Crit Care Med. 2013;41:1774–81. doi: 10.1097/CCM.0b013e31828a25fd. [DOI] [PubMed] [Google Scholar]