1. Introduction
One of the most widely adopted and disseminated tobacco control interventions is the telephone tobacco quit line (QL) [1,2,3]. All 50 U.S. states have QLs funded through state governments and trusts with additional funding from the Centers for Disease Control and Prevention [4]. Although initially disseminated before determining their efficacy [5], QLs have been shown to be effective for increasing long-term smoking abstinence rates [6]. QLs are more effective if they are proactive (i.e., calls initiated by the counselor) rather than reactive (i.e., participant-initiated), offer free nicotine replacement therapy (NRT), and provide at least three treatment sessions [1,7]. In a randomized clinical trial, we observed that a military-tailored proactive intervention increased smoking abstinence rates more than a reactive QL at one year (OR = 1.8, P < 0.0001) among active duty (Army, Navy, Air Force, Marines and Coast Guard), retirees and dependents [8]. However, significant relapse back to smoking after end-of-treatment was observed with 49% achieving smoking cessation at 8 weeks relapsing back to smoking at one year [1]. This is particularly important given that the military has much higher rates of tobacco use than civilians. In a recent report published by the Department of Defense in 2013, 24.5% of military personnel reported past 30-day cigarette smoking and 12.8% reported past 30-day smokeless tobacco use [9].
Opportunities exist to re-engage smokers who relapse to smoking after treatment. Unfortunately, limited research has been conducted on strategies to re-engage smokers following a failed quit attempt. Pro-actively reaching out to smokers has shown promise in re-engaging smokers after failed quit attempts. [10–13] In our military QL study [8], all participants who initially called the military QL were motivated to quit, but one year later half of these formerly motivated participants were smoking. Previous research has found that these smokers feel greater levels of disappointment and lower self-efficacy following a failed quit attempt which may prevent them from actively seeking treatment [13]. State QLs encourage one of two methods for re-engaging individuals who have relapsed or failed to quit on their target quit date and call back for help: (1) repeating the intervention (i.e., Recycle); or (2) reducing the number of cigarettes per day (i.e., Rate Reduction) with the ultimate goal of quitting [14]. However, many relapsed smokers have limited knowledge of interventions available for additional quit attempts [11]. Additionally, no evidence exists supporting the effectiveness of these interventions.
We designed a study to address the following research questions: 1) Does re-engaging a participant after a failed quit attempt enhance their chances of tobacco abstinence at a one-year follow-up? 2) What method of treatment is most effective, Recycle or Rate Reduction, for re-engaging participants following a failed quit attempt?
In the current paper we describe the design of the “Freedom Quit Line,” an ongoing randomized controlled trial for active duty military, retired military, and their families, to treat cigarette smokers who relapse or fail to quit through a standard smoking cessation intervention by a proactive QL. The study compares smoking abstinence rates across three conditions: (1) repeating the standard smoking cessation intervention (Recycle); (2) receiving a rate reduction intervention (Rate Reduction); or (3) participant choice between Recycle or Rate Reduction (Choice).
2. Materials and Methods
2.1. Study Design
This study is funded by the National Heart, Lung and Blood Institute (NHLBI; HL123978) and conducted in the setting of an established military tobacco QL. Every eligible cigarette smoker who calls requesting assistance with smoking cessation receives a non-randomized, baseline standard smoking cessation program using our proactive QL (see Fig. 1) [8]. In addition, all participants receive 8 weeks of NRT in the form of a patch. Smokers who fail to quit or relapse with this initial QL intervention are randomized to one of three conditions at 3 months: Recycle, Rate Reduction, or Choice conditions. The primary outcome is prolonged smoking abstinence at the one-year follow-up defined as self-reported continuous abstinence with a grace period of 2 weeks after the target quit date. NRT in the form of nicotine patch is distributed to those in the Recycle condition and nicotine gum is distributed to those randomized to the Rate Reduction condition. This study was approved by the Institutional Review Boards Wilford Hall Ambulatory Surgical Center and the University of Tennessee Health Science Center.
Figure. 1.
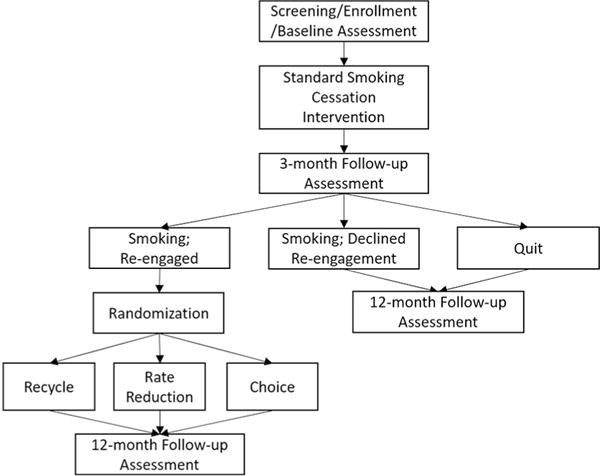
Study design.
2.2. Participants
Recruitment for the study began in May 2015 and will continue through June 2019. Study participants are Department of Defense (DoD) TRICARE beneficiaries, which includes active duty and retired (Army, Navy, Air Force, Marines and Coast Guard) and their dependents. TRICARE is a health care program of the DoD Military Health System. To be eligible for enrollment, potential participants must be at least 18 years of age and have smoked five or more cigarettes a day for at least one year. Participants must be located within the continental United States, Hawaii, Alaska, and bases worldwide with adequate medical infrastructure and an address at a military installation where NRT can be mailed. Participants must have the ability to understand informed consent procedures and have access to a telephone for participation. Participants are excluded from enrollment if they are pregnant women, breastfeeding or planning to become pregnant during the next 12 months. Participants with a contraindication to NRT or have an unstable heart condition are eligible for the behavioral intervention only and are not provided NRT.
2.3. Recruitment, Screening and Randomization
Participants are being recruited with flyers, business cards, and posters distributed on military bases, medical and dental facilities, as well as through various forms of electronic media. Recruitment efforts focus on: (1) disseminating information directly to potential participants; and (2) developing alliances with other military family-centric and health organizations. We have strong partnerships with military health promotion and medical officers throughout the DoD. We work with these military partners to feature the QL on the following websites: TRICARE.mil, Military.com, thewingmantoolkit.org, Health Net Federal Services (hnfs.org), soldierforlife.com, 79mdw.af.mil and UCANQUIT2.org.
At the screening visit, informed consent is obtained and eligibility is determined. If eligible for participation, study staff review the consent document with the participant and answer any questions. Once verbal consent is obtained, the participant completes the baseline questionnaires. At this point, the participant is enrolled in a non-randomized baseline standard smoking cessation program using our validated military proactive QL [8] and begins the nicotine patch, which is mailed to the participant. Part of the initial consent process is to explain that individuals who are still smoking at the 3-month follow-up will receive the opportunity to re-engage. Participants who choose to re-engage at 3 months are centrally randomized into the Recycle, Rate Reduction or Choice conditions via computerized block design. All participants will receive a follow-up questionnaire one year after enrollment. This includes those who: 1) declined re-engagement, 2) accepted re-engagement, and 3) were quit at 3-month follow-up. (Fig. 1).
2.4. Intervention Approach and Theory
All of the interventions in the Freedom Quit Line study utilize a proactive (i.e., counselor initiated telephone call) approach. The counseling strategies and tactics used in the interventions are derived from previous multi-component interventions with smokers based on Social Learning Theory [24,25] and utilize Motivational Interviewing [15]. Motivational Interviewing (MI) is a collaborative conversation style for strengthening a person’s own motivation and commitment to change [15]. The basic components have frequently been applied to tobacco cessation research and are consistent with a number of current protocols including the California Quit line and Alere Wellbeing. The materials have been tailored for a military population focusing on key elements of motivation, coping skills, and social support.
MI attempts to increase internal motivation and to guide participants toward making choices that will help with their desired goal of tobacco cessation. MI is useful for aiding in the treatment of lifestyle problems and disease. Four general processes that underlie MI: (1) engaging by establishing a helpful connection with the participant; (2) focusing to develop and maintain a specific direction in the conversation about change; (3) evoking through an elicitation of a participants own motivation for change; and (4) planning through conversations about what actions might best serve the desired behavior change [15]. A meta-analysis of MI for smoking cessation observed that MI significantly increases smoking abstinence compared to control conditions [21].
2.5. Interventionist Training
A critical part of any intervention conducted in the military is tailoring the intervention to fit the unique needs of our military population. In our military collaborative projects, we accomplish this in several ways. First, most of our QL counselors are former military members and are able to enhance connections with active duty personnel, as they are able to relate to the experiences of the study participants. The military is a culture of its own. As a result, we have found it is easier and more effective to hire and train retired military members or those associated with the military rather than selecting skilled research interventionists and attempt to teach them the military culture. All our staff in addition to having a robust knowledge of the military also received 100 hours of research training and formal motivational interviewing training that is ongoing.
A member of our research team clinically trained in MI and a member of the Motivational Interviewing Network of Trainers is responsible for training and supervision of intervention staff [1]. QL counselors have undergone a two-day in person training and ongoing one-on-one tape reviews based on the Motivational Interviewing Treatment Integrity 3.1 [16] to meet the minimum requirements for proficiency. They participate in weekly training to maintain skills covering a variety of topics including increasing change talk, reflections, open questions, and goal setting.
2.6. Non-randomized Intervention
2.6.1. Standard Smoking Cessation program
All participants receive a four-session standard smoking cessation intervention using our validated military proactive QL [8], designed to be delivered weekly over the course of four weeks telephonically. The goal of this intervention is to help the participant set a quit date and quit smoking by the end of the four treatment sessions (Table 1). Session 1 begins with a discussion to strengthen the participant’s motivation and commitment to quit smoking. The participant is primed to set a quit date and learns skills to prepare them for reaching their target quit date (e.g., identifying triggers, reducing tobacco intake). In Session 2, a specific quit date is selected if the participant is able and steps involved in preparing to quit are discussed, including the 5A’s for staying tobacco-free (e.g., Avoid, Alter, Alternatives, Anticipate, and Active), making a public commitment, removal of triggers, and proper use of the nicotine patch. In Session 3, counselors discuss ways to manage withdrawal symptoms and begin development of an individualized relapse prevention plan. In Session 4, participants review progress to date, continue to discuss ways to manage withdrawal symptoms and complete development of an individualized relapse prevention plan.
Table 1.
Intervention treatment condition.
| Condition | Intervention | NRT | Intervention goal | Intervention sessions |
|---|---|---|---|---|
| Non-randomized intervention: | ||||
| Baseline Intervention | Standard Smoking Cessation Intervention | Patch | Quit smoking | Session 1: Increase motivation to quit; learn skills to prepare for quit date |
| Session 2: Prepare to quit; set quit date | ||||
| Session 3: Develop short-term relapse prevention plan | ||||
| Session 4: Develop long-term relapse prevention plan | ||||
| Randomized Interventions: | ||||
| Recycle | Standard Smoking Cessation Intervention | Patch | Quit smoking | Same as Standard Smoking Cessation Intervention |
| Rate Reduction | Rate Reduction Intervention | Gum | Reduce rate of smoking | Session 1: Increase motivation to quit; learn rate reduction techniques |
| Session 2: Discuss triggers and strategies to avoid smoking | ||||
| Session 3: Reduce smoking and prepare to quit when ready | ||||
| Choice | Participant chooses between Standard Smoking Cessation Intervention or Rate Reduction Intervention | Patch or gum | Quit smoking or reduce rate of smoking | Either Standard Smoking Cessation Intervention or Rate Reduction Intervention sessions |
Abbreviations: NRT = Nicotine Replacement Therapy.
2.6.2. Randomized Interventions
Participants who are still smoking at the three-month follow-up and agree to be re-engaged are randomized to one of the following conditions. Participants who are still smoking at the three-month follow-up and do not agree to be randomized will be contacted for the one year follow-up.
2.6.3. Recycle
Participants randomized to the Recycle condition repeat the same four-session standard smoking cessation program using our validated military proactive QL [8]. We explain that most smokers need to make several quit attempts before they are successfully abstinent and many smokers learn valuable lessons about their previous failed quit attempt.
2.6.4. Rate reduction
In this arm, participants receive our validated three-session Rate Reduction intervention over the course of three weeks (Table 1). There is evidence that systematic reductions in smoking behavior can lead to long-term smoking cessation [17,18]. The Rate Reduction intervention has demonstrated long-term efficacy in multiple settings [19–25]. The behavioral intervention focuses on making three systematic reductions in smoking behavior. Session 1 strengthens motivation and commitment to quit smoking. Participants learn how to self-monitor their smoking habit and to keep a smoking diary. They receive instruction on how to change their brand of cigarettes and about how to use the nicotine gum to reduce cigarette smoking. During Session 2, participants continue to develop self-monitoring skills relating to situational (e.g., smoking in the car), temporal (e.g., time of day) and access (e.g., cigarettes in their pocket) triggers to smoking. Session 3 incorporates instructions on how to smoke less of each cigarette. The rest of the session is spent on how to make a quit attempt when they are ready. Key components of quitting smoking (e.g., withdrawal symptoms, lapses, and setting a quit date) are also discussed. Throughout the sessions, participants work toward a 75% reduction in baseline smoking rates with approximately a 25% reduction relative to baseline in Session 1 and a 50% reduction relative to baseline in Session 2.
2.6.5. Choice
In this condition, participants are informed that the Recycle or Rate Reduction interventions are available to them. Participants are provided the rationale for both methods and collaboratively discuss each method and which method would be best suited for them. Following this discussion, participants choose which intervention they want to receive.
2.6.6. Nicotine Replacement Therapy (NRT)
All participants in the nonrandomized intervention receive eight weeks of the nicotine patch (Table 2). In addition, participants in the Recycle condition and those in the Choice condition who choose Recycle receive another eight weeks of the patch. Because rate reduction is a gradual process that happens over time, participants in the Rate Reduction condition and those in the Choice condition who choose Rate Reduction receive up to 16 weeks of nicotine gum to facilitate rate reduction. Although participants randomized to Rate Reduction and Recycle will receive different forms of NRT, we do not anticipate this confounding our results. A recent network meta-analysis showed no significant difference between nicotine gum and nicotine patch (OR 0.88, 95% CI 0.75–1.03) [26]. The dosing of the NRT is determined for the non-randomized intervention based on reported cigarettes per day at baseline. For the Recycle and Rate Reduction interventions, NRT is based on reported cigarettes per day at the 3-month follow-up.
Table 2.
Nicotine replacement therapy dosing and distribution by condition.
| Condition | Cigarettes per day | NRT | Tapering regimen
|
|||
|---|---|---|---|---|---|---|
| Weeks 1–4 | Weeks 5–8 | Weeks 9–12 | Weeks 13–16 | |||
| Baseline intervention | ≥20 | Patch | 21 mg | 14 mg 2 weeks/7 mg 2 weeks | – | – |
| 10–19 | 14 mg | 7 mg | – | – | ||
| 5–9 | 7 mg | 7 mg | – | – | ||
| Recycle | ≥20 | Patch | 21 mg | 14 mg 2 weeks/7 mg 2 weeks | – | – |
| 10–19 | 14 mg | 7 mg | – | – | ||
| 5–9 | 7 mg | 7 mg | – | – | ||
| Rate reduction | ≥25 | Gum | 300 pieces/4 mg | 300 pieces/4 mg | 126 pieces/4 mg | 84 pieces/4 mg |
| <25 | 300 pieces/2 mg | 300 pieces/2 mg | 126 pieces/2 mg | 84 pieces/2 mg | ||
2.6.7. Data Collection and Assessments
All measures are completed telephonically at baseline, 3-month, and one-year follow-ups. Follow-up is calculated based on enrollment date.
2.6.8. Assessments at Baseline and 3-month Follow-up
At baseline, we collect demographics (e.g., gender, age, ethnicity, marital status, race, education, military status), TRICARE DoD beneficiary status, and service branch. At baseline and 3-month follow-up, we assess nicotine dependence with the Fagerström Test for Nicotine Dependence (FTND). We also measure the number of cigarettes smoked per day, whether they are using other tobacco products on a regular basis, years smoked, prior quit attempts, self-efficacy to quit smoking [27], stages of change [28], reasons for quitting [29], perceived harm of cigarettes [30], and motivation to quit smoking [31].
2.6.9. Assessment at One-year Follow-up
Our primary outcome is the self-reported prolonged smoking abstinence at the one-year follow-up [32]. Prolonged smoking abstinence is defined as self-reported continuous abstinence with a grace period of 2 weeks after the target quit date. A secondary outcome is 7-day point prevalence abstinence, defined as any smoking (“even a puff”) in the past seven days [32]. We also assess military variables thought to be related to smoking relapse, such as deployment.
2.6.10. Process Measures
One major predictor of success is adherence to behavioral (i.e., number of sessions completed) and pharmacologic (i.e., how much NRT used) interventions. This is theorized to directly predict both short-term and long-term success. For instance, if the Choice condition has superior efficacy, it will be important to determine the causal pathway in the intervention by assessing degree of adherence in this condition relative to Recycling and Rate Reduction. One could postulate that adherence will be better in a condition in which the smoker gets to choose which re-engagement strategy to use.
2.6.11. Program Evaluation
At the one-year follow-up, we assess program satisfaction by asking participants how helpful they found the QL, how much they enjoyed participating in the phone sessions and if they would recommend the program to a friend (5-point Likert scale; 1 = not at all to 5 = extremely) [33]. Additionally, participants are asked to select what they liked most about the study.
2.6.12. Treatment Fidelity
We assess fidelity of implementation focusing on adherence to both the intervention materials and the spirit of MI by randomly reviewing 15% of all sessions. Selected sessions are audio-recorded and a standard form based on the Motivational Interviewing Treatment Integrity 3.1 is used to score the sessions [16]. Feedback is provided to counselors on reviewed sessions as part of ongoing training.
3. Statistical Analyses and power Calculations
3.1.1. Power Calculations
Our primary outcome is the self-reported prolonged abstinence at the one-year follow-up [32]. We calculated the sample size based on minimal detectable odds ratio of 1.8 using PASS12 software [34]. The minimal detectable odds ratio is based on results from our previous evaluation of a proactive versus reactive QL in the military [8]. Given that in the current study all conditions are proactive, it is possible that the odds ratio for the current study will be different, however this is the closest justifiable assumption for our design. We proposed to recruit 1500 smokers approximately 66% of whom we hypothesized would relapse (N = 990) based on our previous military QL [8] and enter the randomization phase of the study, equating to 330 per condition. Accounting for 15% loss to follow up, approximately 280 completes per group (280 × 3 = 840 total completers) achieve 87% power to detect a hypothesized OR of 1.8 between any two group proportions, such as Rate Reduction and Recycle (approximately 11% effect or increase in abstinence between groups assuming the null proportion of 22%) [8]. In a case that loss to follow-up is lower than 15%, or that actual detected effect is larger than 1.8 we will have more than 80% power to detect differences. In addition, several analytical approaches allow us to evaluate all participants regardless of loss to follow-up. Using intent-to-treat principles, we can assume no change in the outcome measure for those that were incomplete (conservative toward null), or perform multiple imputations using model based Markov chain Monte Carlo approach for arbitrary missing data mechanism with more realistic variance estimates. Both methods allow all 990 initially randomized participants to be incorporated analytically while minimizing the loss of power.
In our previous clinical trial assessing a tobacco QL in the military, the proactive condition had a 22% abstinent rate at the one-year follow-up [8]. As such, we anticipate that individuals that were smokers at the 3-month follow-up but chose to not re-engage will have a 22% abstinent rate at the one-year follow-up.
3.2. Statistical Analyses
All analyses will be carried out using SAS/STATv14.1 [35]. Due to the nature of data capture and inbuilt quality control measures erroneous values are expected to be minimal; however, all data will be inspected for outliers and out-of-range values and any inconsistencies will be corrected using data queries. Examination of distributions may prompt transformations, where they are justifiable and are a component of the best available analysis strategy. Our follow-up rates at a one-year follow-up are around 80%, so we will perform analyses to determine whether differential attrition between conditions occurred by subject baseline characteristics.
As mentioned above, analyses will follow the intent-to-treat principle. All randomized subjects will be included in the analyses. To deal with missing data we are going to carry out multiple imputations based on the logistic regression predictive model via Markov Chain Monte Carlo (MCMC) algorithm assuming missing at random and missing not at random data mechanisms. Imputed data sets will be analyzed and results combined to produce appropriate variance estimates. Results between two assumed missing data mechanisms are going to be compared before deciding which one provides a better fit for the underlying data. Sensitivity analysis will be conducted to look at completers only, in comparison to multiple imputations results under two assumed missing data mechanisms.
Given that our outcomes and treatment assignment are of discrete nature, we are going to use categorical analytical methods; however, we will describe baseline characteristics using proportions, means with standard deviations, and medians with interquartile ranges as appropriate and apply comparisons using chi-square test for categorical variables (Fishers exact test where chi-square assumptions are not met), analysis of variance (ANOVA) for normally distributed continuous variables, and appropriate non-parametric equivalent tests such as Kruskal–Wallis test for non-normally distributed continuous variables. We will also compare those lost to follow-up with the study completers to examine any potential non-random patterns. Our primary outcome is prolonged smoking abstinence and our secondary outcome is point prevalence at the one-year follow-up. We will use the same analytical methods for both outcomes. Since those who successfully quit may be more likely to be successful at long-term cessation than those who fail to even initially quit we will use the stratification factor of “did not quit in treatment” versus “quit during treatment but relapsed” in final analyses. We will explore the heterogeneity of treatment effects by demographics and number of cigarettes smoked per day strata, by adding and testing the mentioned two-way interaction in the model. We will also explore differences in both the primary and secondary outcomes across individuals who quit smoking during the non-randomized intervention and those who were re-engaged into one of the three treatment conditions. We will use the same analytical methods described above. All associations will be considered significant at the alpha level of 0.05.
4. Conclusions
The current study is an effort stemming from a 20-year collaboration between the U.S. Air Force and our Military Population Health research team [36] and is a logical extension of our previous research findings. We have demonstrated that a Proactive QL conducted in the military significantly increased smoking abstinence rates compared to a Reactive condition at long-term follow-up (one-year) [8]. In that study, however, we observed significant smoking relapse following completion of the QL intervention. Specifically, among 344 participants achieving prolonged smoking abstinence at 8 weeks, 170 (49%) relapsed to smoking by 12 months. Our telephone survey of state-funded tobacco QLs revealed that QLs use both Recycling and Rate Reduction to re-engage smokers [14]. The Freedom Quit Line study assesses which strategy is the more effective for re-engaging smokers who relapse to smoking or fail to achieve abstinence.
Rates of smoking in the military are much higher than civilian populations. Almost a quarter (24.5%) of active duty military personnel report past 30-day cigarette smoking [37]. However, many smokers in the military are interested in quitting; 24% of current smokers are planning to quit in the next 30 days and 38% say they are intending to quit in the next six months [38]. Given that the DoD is the world’s largest employer with 3.2 million employees [9], half of which are active duty military, this is a sizeable number of smokers in need of effective treatment. Each year the DoD spends on average $1.6 billion treating tobacco-related morbidity among active duty military personnel (e.g., medical care, hospitalizations, lost work days). As such, the dissemination of our findings from the Freedom Quit Line study could have a significant public health impact.
Significant challenges to conducting research in the US military exist. Current challenges include severely restricted access to military installations and military personnel, all researchers must have a secret security clearance, access to military records has become severely limited, and it is vital that the US military sees our project as worthwhile and relevant to the mission of maintaining a healthy and fit force. As such we feel that a telephone QL is the perfect intervention modality for reaching the 9.4 million TRICARE beneficiaries worldwide [39]. Tobacco QLs have the ability to reach the beneficiaries dispersed throughout the world and these interventions can be disseminated while maintaining a high level of treatment fidelity thereby ensuring intervention efficacy.
Despite this uniqueness of the military population, the findings of the current study can be applied not only to military, but also to every QL in the US. Our study addresses a critical paucity of data on strategies already disseminated in QLs. The literature indicates that civilian tobacco programs often fail to work in military settings [40–43]. This may be related to uniqueness of the military experience (e.g., training and deployment). Military service members are at high risk for adverse health behaviors such as tobacco use given the demographics (e.g., young, pre-dominantly male, low income, high prevalence of minorities [44]) and psychological risk factors (e.g. risk takers, high prevalence of alcohol abuse, stress of deployment). Therefore, we suggest that if Recycling, Rate Reduction, or Choice increases smoking abstinence in this high-risk population living in this unique setting, the findings will be applicable to civilian quit lines.
It is possible that our results will be subject to selection bias given that only participants interested in quitting will enroll in the study. However, that is true for the majority of tobacco QL intervention research [45], which limit participation to individuals interested in making a quit attempt in the next 30 days. Given that the majority of smokers are not ready to quit in the next 30 days [46], in combination with the dearth of research focusing on this populations [47], future research should focus on testing these same re-engagement strategies with smokers not ready to quit. It should be noted that participants are randomized to conditions, so comparisons between re-engagement interventions will not be subject to selection bias.
QLs reach up to 6% of smokers each year in the U.S. [48] Advancing our understanding of the best approach for engaging relapsed smokers or those that fail to quit with a standard QL intervention holds the potential for significantly impacting public health.
Acknowledgments
The authors gratefully acknowledge the support of Wilford Hall Ambulatory Surgical Center, Joint Base San Antonio, Lackland Air Force Base. The opinions expressed on this document are solely those of the authors and do not represent an endorsement by or the views of the United States Air Force, the Department of Defense, or the United States Government.
Funding
This work was supported by a grant from National Heart, Lung, and Blood Institute [R01 HL123978, 2014]. This study is a collaborative endeavor between the United States Air Force and the University of Tennessee Health Science Center via a Cooperative Research and Development Agreement [CRADA # 10-217-WHMC-CRADA01-MOD01].
References
- 1.Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update, Clinical Practice Guideline, U.S Department of Health and Human Services. Public Health Service; Rockville, MD: May, 2008. [Google Scholar]
- 2.Lichtenstein E, Zhu SH, Tedeschi GJ. Smoking cessation quitlines: an underrecognized intervention success story. Am Psychol. 2010;65(4):252–261. doi: 10.1037/a0018598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhu SH, Anderson CM, Tedeschi GJ, et al. Evidence of real-world effectiveness of a telephone quitline for smokers. N Engl J Med. 2002;347(14):1087–1093. doi: 10.1056/NEJMsa020660. [DOI] [PubMed] [Google Scholar]
- 4.North American Quitline Consortium. Tobacco Cessation Quitlines. NAQC Issue Paper. 2009 Available at: http://www.naquitline.org/resource/resmgr/docs/naqc_issuepaper_tobaccocessa.pdf>.
- 5.Anderson C, Zhu S-H. Tobacco quit lines: Looking forward and looking back. Tob Control. 2007;16(S-H1–S-H8) [Google Scholar]
- 6.Stead LF, Hartmann-Boyce J, Perera R, Lancaster T. Telephone counselling for smoking cessation. Cochrane Database Syst Rev. 2013;8:Cd002850. doi: 10.1002/14651858.CD002850.pub3. [DOI] [PubMed] [Google Scholar]
- 7.Stead LF, Perera R, Lancaster T. Telephone counselling for smoking cessation. Cochrane Database Syst Rev. 2006;3:CD002850. doi: 10.1002/14651858.CD002850.pub2. [DOI] [PubMed] [Google Scholar]
- 8.Klesges R, Ebbert JO, Talcott GW, et al. Efficacy of a Tobacco Quitline in Active Duty Military and TRICARE Beneficiaries: a Randomized Controlled Trial. Military medicine. 2015;180(8):917–925. doi: 10.7205/MILMED-D-14-00513. [DOI] [PubMed] [Google Scholar]
- 9.Barlas FM, Higgins WB, Pflieger JC, Diecker K. Department of Defense TMA, Defense Health Cost Assessment, Program Evaluation (DHCAPE) In: the United States Coast Guard, editor. 2011 Health Related Behaviors Survey of Active Duty Military personnel. Fairfax, Virginia: 2013. [Google Scholar]
- 10.Carlini B, Miles L, Doyle S, Celestino P, Koutsky J. Using Diverse Communication strategies to Re-Engage Relapsed Tobacco quitline Users in Treatment, New York State, 2014. Preventing Chronic Disease. 2015;12:E179. doi: 10.5888/pcd12.150191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Carlini BH, McDaniel AM, Weaver MT, et al. Reaching out, inviting back: Using Interactive voice response (IVR) technology to recycle relapsed smokers back to Quitline treatment–a randomized controlled trial. BMC Public Health. 2012;12:507. doi: 10.1186/1471-2458-12-507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gonzales D, Hajek P, Pliamm L, et al. Retreatment with varenicline for smoking cessation in smokers who have previously taken varenicline: a randomized, placebo-controlled trial. Clinical pharmacology and therapeutics. 2014;96(3):390–396. doi: 10.1038/clpt.2014.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lando HA, Pirie PL, Roski J, McGovern PG, Schmid LA. Promoting abstinence among relapsed chronic smokers: the effect of telephone support. Am J Public Health. 1996;86(12):1786–1790. doi: 10.2105/ajph.86.12.1786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Asfar T, Ebbert JO, Klesges RC, Klosky JL. Use of smoking reduction strategies among U.S. tobacco quitlines. Addict Behav. 2012;37(4):583–586. doi: 10.1016/j.addbeh.2012.01.018. [DOI] [PubMed] [Google Scholar]
- 15.Miller W, Rollnick S. Motivational interviewing: Preparing People for change. Guilford Press; New York, NY: 2012. [Google Scholar]
- 16.Moyers T, Martin T, Manuel J, M WR, Ernst D. Revised Global Scales: Motivational Interviewing Treatment Integrity 3.1.1 (MITI 3.1.1) 2010 [Google Scholar]
- 17.Hughes JR, Carpenter MJ. Does smoking reduction increase future cessation and decrease disease risk? A qualitative review. Nicotine Tob Res. 2006;8(6):739–749. doi: 10.1080/14622200600789726. [DOI] [PubMed] [Google Scholar]
- 18.Stead LF, Lancaster T. Interventions to reduce harm from continued tobacco use. Cochrane Database Syst Rev. 2007;(3):Cd005231. doi: 10.1002/14651858.CD005231.pub2. [DOI] [PubMed] [Google Scholar]
- 19.Brown JM, Williams J, Bray RM, Hourani L. Postdeployment alcohol use, aggression, and post-traumatic stress disorder. Military medicine. 2012;177(10):1184–1190. doi: 10.7205/milmed-d-11-00119. [DOI] [PubMed] [Google Scholar]
- 20.Glasgow RE, Gaglio B, Estabrooks PA, et al. Long-term results of a smoking reduction program. Medical care. 2009;47(1):115–120. doi: 10.1097/MLR.0b013e31817e18d1. [DOI] [PubMed] [Google Scholar]
- 21.Glasgow RE, Klesges RC, Godding PR, Gegelman R. Controlled smoking, with or without carbon monoxide feedback, as an alternative for chronic smokers. Behavior Therapy. 1983;14(3):386–397. [Google Scholar]
- 22.Glasgow RE, Klesges RC, Klesges LM, Vasey MW, Gunnarson DF. Long-term effects of a controlled smoking program: A 2 1/2 year follow-up. Behavior Therapy. 1985;16(3):303–307. [Google Scholar]
- 23.Glasgow RE, Klesges RC, Mizes JS, Pechacek TF. Quitting smoking: strategies used and variables associated with success in a stop-smoking contest. Journal of consulting and clinical psychology. 1985;53(6):905–912. doi: 10.1037//0022-006x.53.6.905. [DOI] [PubMed] [Google Scholar]
- 24.Glasgow RE, Klesges RC, O’Neill HK. Programming social support for smoking modification: an extension and replication. Addict Behav. 1986;11(4):453–457. doi: 10.1016/0306-4603(86)90027-4. [DOI] [PubMed] [Google Scholar]
- 25.Glasgow RE, Klesges RC, Vasey MW. Controlled smoking for chronic smokers: an extension and replication. Addict Behav. 1983;8(2):143–150. doi: 10.1016/0306-4603(83)90008-4. [DOI] [PubMed] [Google Scholar]
- 26.Cahill K, Stevens S, Perera R, Lancaster T. Pharmacological interventions for smoking cessation: an overview and network meta-analysis. Cochrane Database Syst Rev. 2013;5:Cd009329. doi: 10.1002/14651858.CD009329.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Etter JF, Bergman MM, Humair JP, Perneger TV. Development and validation of a scale measuring self-efficacy of current and former smokers. Addiction. 2000;95(6):901–913. doi: 10.1046/j.1360-0443.2000.9569017.x. [DOI] [PubMed] [Google Scholar]
- 28.DiClemente CC, Prochaska JO, Fairhurst SK, Velicer WF, Velasquez MM, Rossi JS. The process of smoking cessation: an analysis of precontemplation, contemplation, and preparation stages of change. Journal of consulting and clinical psychology. 1991;59(2):295–304. doi: 10.1037//0022-006x.59.2.295. [DOI] [PubMed] [Google Scholar]
- 29.Zvolensky MJ, Vujanovic AA, Miller MO, et al. Incremental validity of anxiety sensitivity in terms of motivation to quit, reasons for quitting, and barriers to quitting among community-recruited daily smokers. Nicotine Tob Res. 2007;9(9):965–975. doi: 10.1080/14622200701540812. [DOI] [PubMed] [Google Scholar]
- 30.Hyland A, Borland R, Li Q, et al. Individual-level predictors of cessation behaviours among participants in the International Tobacco Control (ITC) Four Country Survey. Tob Control. 2006;15(Suppl 3):iii83–iii94. doi: 10.1136/tc.2005.013516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Herzog TA, Blagg CO. Are most precontemplators contemplating smoking cessation? Assessing the validity of the stages of change. Health Psychol. 2007;26(2):222–231. doi: 10.1037/0278-6133.26.2.222. [DOI] [PubMed] [Google Scholar]
- 32.Hughes JR, Keely JP, Niaura RS, Ossip-Klein DJ, Richmond RL, Swan GE. Measures of abstinence in clinical trials: issues and recommendations. Nicotine & Tobacco Research. 2003;5(1):13–25. [PubMed] [Google Scholar]
- 33.Jeong BY, Lim MK, Yun EH, et al. User Satisfaction as a Tool for Assessment and Improvement of Quitline in the Republic of Korea. Nicotine Tob Res. 2011:ntr290. doi: 10.1093/ntr/ntr290. [DOI] [PubMed] [Google Scholar]
- 34.Hintze J. PSASS 12. NCSS, LLC; Kaysville, Utah: 2013. [Google Scholar]
- 35.SAS Institute Inc. SASv9.3. Cary, NC: [Google Scholar]
- 36.Talcott GW, Ebbert JO, Klesges RC, et al. Tobacco Research in the Military: reflections on 20 Years of Research in the United States Air Force. Military medicine. 2015;180(8):848–850. doi: 10.7205/MILMED-D-14-00501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Defense Do. Department of Defense TMADHCA. In: Program E, the United States Coast G, editor. 2011 Health Related Behaviors Survey of Active Duty Military Personnel. Fairfax, Virginia: 2013. [Google Scholar]
- 38.Bray RM, Pemberton MR, Hourani LL, et al. Department of Defense Survey of Health Related Behaviors Among Active Duty Military Personnel(DTIC Document) 2009 [Google Scholar]
- 39.TRICARE, Number of Beneficiaries, http://www.tricare.mil/About/Facts/BeneNumbers.aspx 2016 (Accessed 11/18, 2016).
- 40.Bushnell FK, Forbes B, Goffaux J, Dietrich M, Wells N. Smoking cessation in military personnel. Mil Med. 1997;162(11):715–719. [PubMed] [Google Scholar]
- 41.Swanson NA, Burroughs CC, Long MA, Lee RW. Controlled trial for smoking cessation in a Navy shipboard population using nicotine patch, sustained-release buproprion, or both. Mil Med. 2003;168(10):830–834. [PubMed] [Google Scholar]
- 42.Conway TL, Woodruff SI, Edwards CC, Elder JP, Hurtado SL, Hervig LK. Operation Stay Quit: evaluation of two smoking relapse prevention strategies for women after involuntary cessation during US Navy recruit training. Mil Med. 2004;169(3):236–242. doi: 10.7205/milmed.169.3.236. [DOI] [PubMed] [Google Scholar]
- 43.Cronan TA, Conway TL, Hervig LK. Evaluation of smoking interventions in recruit training. Mil Med. 1989;154(7):371–375. [PubMed] [Google Scholar]
- 44.Anderson CM. Which is the World’s Largest Employer. 2012 http://www.bbc.co.uk/news/magazine-17429786.
- 45.Fiore MC. Treating tobacco use and dependence: 2008 update: Clinical Practice guideline. DIANE Publishing; 2008. [Google Scholar]
- 46.Wewers ME, Stillman FA, Hartman AM. DR Shopland, Distribution of daily smokers by stage of change: current population Survey results. Prev Med. 2003;36(6):710–720. doi: 10.1016/s0091-7435(03)00044-6. [DOI] [PubMed] [Google Scholar]
- 47.Asfar T, Ebbert JO, Klesges RC, Relyea GE. Do smoking reduction interventions promote cessation in smokers not ready to quit? Addict Behav. 2011;36(7):764–768. doi: 10.1016/j.addbeh.2011.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Woods SS, Haskins AE. Increasing reach of quitline services in a US state with comprehensive tobacco treatment. Tob Control. 2007;16(Suppl 1):i33–i36. doi: 10.1136/tc.2007.019935. [DOI] [PMC free article] [PubMed] [Google Scholar]


