Abstract
Background
Amyloid deposition is a potential contributor to postoperative cognitive dysfunction. We hypothesized that 6-week global cortical amyloid burden, determined by 18F-florbetapir positron emission tomography, would be greater in those patients manifesting cognitive dysfunction at 6 weeks postoperatively.
Methods
Amyloid deposition was evaluated in cardiac surgical patients at 6 weeks (n=40) and 1 year (n=12); neurocognitive function was assessed at baseline (n=40), 6 weeks (n=37), 1 year (n=13), and 3 years (n=9). The association of 6-week amyloid deposition with cognitive dysfunction was assessed by multivariable regression, accounting for age, years of education, and baseline cognition. Differences between the surgical cohort with cognitive deficit and the Alzheimer’s Disease Neuroimaging Initiative cohorts (normal and early/late mild cognitive impairment) was assessed, adjusting for age, education, and APOE4 genotype.
Results
We found that 6-week abnormal global cortical amyloid deposition was not associated with cognitive dysfunction (13/37, 35%) at 6 weeks postoperatively (median standard uptake value ratio [IQR]: cognitive dysfunction 0.92 [0.89–1.07] vs. 0.98 [0.93–1.05]; p=0.455). In post-hoc analyses, global cortical amyloid was also not associated with cognitive dysfunction at 1 or 3 years postoperatively. Amyloid deposition at 6 weeks in the surgical cohort was not different from that in normal Alzheimer’s Disease Neuroimaging Initiative subjects but increased over 1 year in many areas at a rate greater than in controls.
Conclusion
In this study, postoperative cognitive dysfunction was not associated with 6-week cortical amyloid deposition. The relationship between cognitive dysfunction and regional amyloid burden and the rate of postoperative amyloid deposition merit further investigation.
Introduction
Up to 50% of patients undergoing cardiac surgery may experience postoperative cognitive dysfunction at the time of hospital discharge.1 While there appears to be an initial improvement in the months after surgery, cognitive dysfunction persists in up to 42% at 5 years after surgery1, resulting in a diminished quality of life and loss of functional independence.2
The mechanisms underlying postoperative cognitive dysfunction remain elusive, although several contributing factors have been proposed: preoperative cognitive impairment, genetic predisposition, transcerebral platelet activation, cerebral microembolism or hypoperfusion during cardiopulmonary bypass (CPB), central nervous and systemic inflammatory responses to surgery, hemodilution, hyperglycemia, hyperthermia, and the unmasking of Alzheimer’s disease and acceleration of amyloid deposition associated with inhalational anesthetics (reviewed elsewhere)3. Because cardiac surgery generally takes place in older adults, the possibility exists that cognitive dysfunction after cardiac surgery represents a form of mild cognitive impairment, which also affects older adults. Mild cognitive impairment has been suggested to be a transitional phase between normal aging and dementia and a precursor to Alzheimer’s disease4. Both mild cognitive impairment and Alzheimer’s disease involve the accumulation of β-amyloid in the central nervous system. Laboratory studies have shown that inhalational anesthetics increase β-amyloid generation5 and promote β-amyloid oligomerization in cultured cells.6 Thus, anesthesia itself may influence β-amyloid processing and play a role in the evolution of cognitive dysfunction in the aging, in common with mild cognitive impairment/Alzheimer’s disease. However, human studies have provided conflicting results about whether cerebrospinal fluid (CSF) β-amyloid levels rise, fall, or remain unchanged after anesthesia and surgery.7,8 Thus, we attempted to directly measure amyloid deposition in the brain following surgery using the positron emission tomography tracer 18F-florbetapir.
Positron emission tomography agents have shown great promise in mapping fibrillar amyloid deposition in the brain. 18F-florbetapir [(E)-4-(2-(6-(2-(2-(2-18F-fluoroethoxy)ethoxy)ethoxy)pyridin-3-yl)vinyl)-N-methylbenzamine] is a novel imaging agent that binds with high affinity (Kd 3.1 nM+0.7) to β-amyloid peptide fibrils in brain amyloid plaques.9,10 In a multicenter study, 18F-florbetapir was shown to have the highest cortical retention in Alzheimer’s subjects, the lowest in cognitively normal subjects, and intermediate retention in those with mild cognitive impairment.11 18F-florbetapir has been used in an increasing number of investigations of Alzheimer’s disease and has demonstrated comparable or better sensitivity and specificity for diagnosing Alzheimer’s disease compared to clinical criteria.12 Furthermore, 18F-florbetapir imaging is the modality used in the longitudinal Alzheimer’s Disease Neuroimaging Initiative – a multicenter investigation of subjects with normal cognition, varying degrees of mild cognitive impairment, and Alzheimer’s disease.13
In this study, we utilized 18F-florbetapir imaging to assess the relationship between global cortical and regional amyloid deposition and cognitive dysfunction in patients at 6 weeks after cardiac surgery with CPB. We also conducted follow-up cognitive testing and imaging at 1 and 3 years post-surgery for post-hoc analyses. We hypothesized that 6-week 18F-florbetapir cortical amyloid burden would be greater in those patients manifesting postoperative cognitive dysfunction at 6 weeks and that the amyloid deposition pattern in patients with cognitive dysfunction would be similar to that seen in individuals from the Alzheimer’s Disease Neuroimaging Initiative cohort with mild cognitive impairment.
Methods
Study Population
Following approval by the Duke University Health Systems Institutional Review Board and informed consent, 40 patients aged ≥60 years and undergoing cardiac surgery [coronary artery bypass grafting (CABG), CABG+valve, or valve only] with CPB were prospectively enrolled between July 2011 and November 2013. Patients were excluded if they had a history of symptomatic cerebrovascular disease (e.g., prior stroke) with residual deficits, alcoholism (>2 drinks/day), psychiatric illness (any clinical diagnoses requiring therapy), drug abuse (any illicit drug use in the preceding 3 months before surgery), hepatic insufficiency (liver function tests >1.5 times the upper limit of normal), severe pulmonary insufficiency (requiring home oxygen), or renal failure (serum creatinine >2.0 mg/dl). Pregnant or premenopausal women and patients who were unable to read and thus complete the cognitive testing or who scored <24 on a baseline Mini Mental State examination or >27 on the baseline Center for Epidemiological Studies Depression scale were similarly excluded. Patients who received any anti-amyloid therapies or had any radiopharmaceutical imaging in the 7 days prior to the surgery were also excluded.
Elderly control patients and patients with early mild cognitive impairment and late mild cognitive impairment (early vs. late defined by specific cut-offs on the Logical Memory II subscale of the Wechsler Memory Scale – Revised, as defined by ADNI2)14, who had been previously enrolled and imaged with 18F-florbetapir positron emission tomography through the Alzheimer’s Disease Neuroimaging Initiative (adni.loni.usc.edu)15,16 were utilized to compare regional patterns of amyloid deposition to our surgical cohort. The Alzheimer’s Disease Neuroimaging Initiative was launched in 2003 as a public-private partnership, led by Principal Investigator Michael W. Weiner, MD. The primary goal of the Alzheimer’s Disease Neuroimaging Initiative has been to assess whether serial magnetic resonance imaging, positron emission tomography, biological markers, and clinical and neuropsychological assessment can be combined to measure the progression of mild cognitive impairment and early Alzheimer’s disease. 18F-florbetapir imaging was included in the Alzheimer’s Disease Neuroimaging Initiative-GO and Alzheimer’s Disease Neuroimaging Initiative-2 protocols. All participants gave written informed consent that was approved by the institutional review board of each participating institution.
Surgical Patient Management
Anesthesia was induced with propofol, midazolam, fentanyl, and neuromuscular blocking agents, and isoflurane was used for maintenance. All patients underwent nonpulsatile, hypothermic (30°C - 32°C) CPB with a membrane oxygenator and arterial line filter by a pump primed with crystalloid. Serial hematocrit levels were maintained at ≥0.21. Before initiating CPB, heparinization (300 - 400 U/kg) was performed to a target activated coagulation time of >480 s. Perfusion was maintained at flow rates of 2 – 2.4 L/min/m2 throughout CPB to maintain a mean arterial pressure of 50 – 80 mmHg. Arterial blood gases were measured every 15 – 30 min to maintain the PaCO2 at 35 – 40 mmHg, unadjusted for temperature (α-stat) and the PaO2 at 150 – 250 mmHg.
Neuroimaging
Cardiac surgical study participants underwent 18F-florbetapir positron emission tomography/computerize tomography imaging at the Duke Positron Emission Tomography Center at 6 weeks after surgery. Given funding constraints, imaging was performed at 6 weeks after surgery, since amyloid burden is not expected to change significantly over a 6-week period.17 Approximately at the midpoint of the study, imaging was added at the 1-year postoperative time point to provide pilot data on the change in amyloid burden over this time interval. A 10 mCi (370 MBq) dose of 18F-florbetapir (Avid Radiopharmaceuticals, Philadelphia, PA, USA) was assayed with a dose calibrator and administered via bolus injection through a peripheral vein. Once 50 min had elapsed after 18F-florbetapir injection, patients underwent 10 min of continuous brain positron emission tomography imaging. A low-dose computerized tomography scan was also performed for attenuation-correction of the positron emission tomography images. Positron emission tomography images were immediately reconstructed after the scan, and if any motion was detected another 10-min continuous scan was performed.
For quantitative evaluation, 18F-florbetapir images were spatially normalized to the stereotactic Montreal Neurologic Institute brain atlas space.18 A standard uptake value ratio was calculated using an average of six target regions (medial orbital frontal, anterior cingulate, parietal, posterior cingulate, precuneus and lateral temporal) with respect to the whole cerebellum as a reference region. 18F-florbetapir signal was also measured in the hippocampus, pons, centrum, putamen, and caudate, and the standard uptake value ratio for each region was calculated with respect to the cerebellum. Amyloid burden, as previously described19, was identified based on standard uptake value ratio values (≥ 1.10 is β-amyloid-positive [abnormal amyloid deposition] and < 1.10 is β-amyloid-negative).
Neurocognitive Testing
Neurocognitive testing was performed at baseline (preoperatively) and at 6 weeks. Post-hoc, 1-year and 3-year follow-up points were added to provide pilot data on the relationship between baseline amyloid burden and long term neurocognitive function. In accordance with the consensus statement on assessment of neurobehavioral outcomes after cardiac surgery20, the following tests were included in the assessment battery: 1) Hopkins Verbal Learning Test21, 2) Randt Short Story Memory Test22, 3) Modified Visual Reproduction Test from the Wechsler Memory Scale23, 4) Digit Span and Digit Symbol and Vocabulary subtests from the Wechsler Adult Intelligence Scale-Revised23, and 5) Trail Making Test, Parts A and B24.
Blood Sample and APOE Genotyping
One 10-mL sample of peripheral blood was obtained from each patient and stored at 4°C. Genomic DNA were extracted for each sample and stored at the Duke Molecular Physiology Institute at −20°C. Genotyping for apolipoprotein E was performed at the Molecular Genetics Core at the Duke Molecular Physiology Institute following previously described protocols.25
Statistical Analyses
To characterize cognitive function over time while minimizing potential redundancy in the cognitive measures, a factor analysis with oblique rotation (a linear transformation of the data, which allows for correlated factors) was performed on the 14 cognitive test scores from baseline. Scoring coefficients (weights) of each test on each factor were determined using the rotated factor solution from the factor analysis conducted on 508 eligible cardiac patients in our ongoing prospective post-CABG cognitive testing database. Factors of each subject in our cohort were computed for all time points using the same scoring coefficients, so that the cognitive domain structure remained consistent and comparable over time. Factor analysis suggested a 5-factor solution, which accounts for 80% of the variability in the original test scores, and represents 5 cognitive domains: 1) structured verbal memory (i.e., the ability to recall from a list), 2) unstructured verbal memory (i.e., the ability to remember from a narrative), 3) visual memory, 4) executive function, and 5) attention and concentration. Two outcome measures were calculated to represent postoperative cognitive dysfunction: 1) continuous outcome: the change in cognitive score calculated by subtracting the baseline cognitive index (the 5-domain mean) from the follow-up cognitive index (a change score of 0 indicates no change from baseline while a negative score indicates cognitive decline and a positive score indicates cognitive improvement); 2) binary outcome (cognitive deficit): defined as a decline of >1 standard deviation in at least 1 domain.
The relationship between 6-week global cortical amyloid burden (standard uptake value ratio ≥ 1.1) and cognition at 6 weeks after surgery was pre-specified as the primary outcome. Secondary outcomes included the relationship between regional amyloid burden and cognition at 6 weeks postoperatively and the relationship between global amyloid burden and cognition at 1 year. We used the χ2 test, Fisher exact test, or Wilcoxon rank-sum test, as appropriate, to examine differences between patients with and without cognitive deficit. We then computed Pearson’s correlation coefficients of amyloid burden with age, years of education, baseline cognitive score, 6-week cognitive score, and change in cognitive score. Finally, multivariable regression was used to test the association of cognitive deficit and mean 6-week cognitive score change with abnormal amyloid deposition (standard uptake value ratio ≥ 1.1), accounting for age, years of education, and baseline cognition.
Subject demographics and amyloid burden in our surgical cohort was compared to age- and sex-matched normal, early mild cognitive impairment, and late cognitive impairment subsets in the Alzheimer’s Disease Neuroimaging Initiative database using the 2-sample t-test, Wilcoxon rank-sum test, χ2 test, or Fisher exact test, as appropriate.
Apolipoprotein E4 genotype was categorized by the presence (homozygous or heterozygous) or absence of the apolipoprotein E-ε4 allele. The association of apolipoprotein E4 status with amyloid burden at 6 weeks was assessed using the two-sample t-test, Wilcoxon rank-sum test, χ2 test or Fisher exact test, as appropriate. An analysis of covariance (ANCOVA) model was then used to test differences among four cognitive categories: cognitive deficit at 6 weeks in our surgical cohort and normal, early mild cognitive impairment, and late cognitive impairment in the Alzheimer’s Disease Neuroimaging Initiative cohort, adjusting for age, years of education, and apolipoprotein E4 genotype.
In the absence of any published data on amyloid deposition in surgical patients, we relied upon preliminary data from a study conducted by a co-investigator (PMD) evaluating amyloid deposition in healthy, mild cognitive impairment, and Alzheimer’s disease subjects, where the estimated mean and standard deviation (SD) of the healthy and mild cognitive impairment groups were used for power calculation. Based on these data, we assessed the statistical power for detecting the correlation between cognitive score changes and amyloid burden. Under a linear regression model with the standard deviation of amyloid burden at 0.25 from preliminary data, we estimated that 40 patients in the cardiac surgical group would provide 80% power to detect a correlation between cognitive score changes and amyloid burden at an R-square of 0.171.
All analyses were performed with SAS version 9.4 (SAS Institute Inc.; Cary, NC). P<0.05 was considered significant. Post-hoc analyses of regional amyloid deposition were adjusted for multiple comparisons by computing a false discovery rate.
Results
Neurocognitive outcomes
Of the 40 patients initially enrolled, 37 had complete baseline and 6-week cognitive and neuroimaging data; at 1 year after surgery, 28 patients had complete baseline and 1-year cognitive data and 12 had neuroimaging; and at 3 years after surgery 18 patients completed cognitive testing. The mean (SD) cognitive change score (from baseline) was 0.10 (0.29) at 6 weeks, 0.13 (0.31) at 1 year after surgery, and 0.08 (0.51) at 3 years after surgery. Cognitive deficit, defined as ≥1 SD decline in at least 1 cognitive domain, was present in 35% (13/37) of the cardiac surgical patients at 6 weeks after surgery, 57% (16/28) at 1 year, and 44% (8/18) at 3 years. Interestingly, several patients without deficit at 6 weeks went on to develop deficit at 1 year postoperatively, while others recovered. (Supplemental Figure 1, Supplemental Table 1) Table 1 lists the demographic and surgical characteristics of the enrolled patients.
Table 1.
Characteristics of the Cardiac Surgical Cohort
| Variable | Baseline (n=38)* | 6 weeks (n=40)** | 1 year (n = 12)** | 3 years (n = 18)* |
|---|---|---|---|---|
| Age, y | 69 (6) | 69.4 (6) | 71 (5) | 71 (6) |
| Sex, % female | 9 (23%) | 8 (22%) | 2 (17%) | 4 (22%) |
| Race, % white | 34 (85%) | 32 (86%) | 11 (92%) | 15 (86%) |
| Weight, kg | 85 (17) | 86 (18) | 83 (17) | 85 (16) |
| History of hypertension | 33 (83%) | 31 (84%) | 9 (75%) | 15 (84%) |
| Diabetes mellitus | 22 (55%) | 20 (54%) | 4 (33%) | 9 (54%) |
| Previous MI | 13 (33%) | 13 (35%) | 3 (25%) | 4 (35%) |
| Ejection fraction (%) | 51 (9) | 51 (9) | 52 (8) | 54 (4) |
| Years of education | 14 (4) | 14 (3) | 16 (5) | 15 (4) |
| Preoperative statins, % | 28 (80%) | 27 (82%) | 9 (90%) | 14 (82%) |
| Preoperative platelet inhibitors, % | 30 (86%) | 29 (88%) | 7 (70%) | 16 (88%) |
| Surgical procedure, % | ||||
| CABG | 24 (60%) | 23 (62%) | 8 (67%) | 11 (62%) |
| CABG+valve | 8 (20%) | 7 (19%) | 2 (17%) | 3 (19%) |
| Valve only | 8 (20%) | 7 (19%) | 2 (17%) | 4 (19%) |
| No. of grafts, %*** | ||||
| 1 | 6 (19%) | 6 (20%) | 2 (20%) | 3 (21%) |
| 2 | 5 (16%) | 4 (13%) | 1 (10%) | 2 (14%) |
| 3 | 10 (31%) | 10 (33%) | 5 (50%) | 5 (36%) |
| >3 | 11 (34%) | 10 (33%) | 2 (20%) | 4 (29%) |
| Cross-clamp time, min | 90 (34) | 87 (33) | 90 (30) | 84 (34) |
| CPB time, min | 145 (49) | 141 (44) | 136 (35) | 142 (46) |
| Baseline cognitive score | −0.17 (0.57) | −0.15 (0.56) | −0.19 (0.48) | −0.17 (0.64) |
| 6-week cognitive score | −0.07 (0.61) | −0.05 (0.62) | −0.06 (0.64) | −0.09 (0.69) |
Values are mean (SD) unless otherwise indicated. CABG, coronary artery bypass grafting; CPB, cardiopulmonary bypass.
N represents the number of patients with cognitive testing data at these time points.
N represents the number of patients with imaging at these time points.
Only includes patients undergoing CABG or CABG+valve procedures. Differences in demographics and comorbities between baseline/6-week and the 1- and 3-year time points are due to patient loss to follow-up.
Global cortical amyloid deposition and postoperative cognitive dysfunction at 6 weeks and 1 year
Representative images from our study cohort of normal (A) and abnormal (B) amyloid deposition as measured by 18F-florbetapir imaging are shown in Figure 1. Global cortical amyloid deposition was measured as standard uptake value ratio 1.03 (0.17) in the 40 patients imaged at 6 weeks and 1.04 (0.20) in the 12 patients imaged at 1 year. Cortical amyloid deposition was considered abnormal (standard uptake value ratio ≥ 1.1) in 7 patients (17.5%) imaged at 6 weeks and in 2 patients (16.7%) imaged at 1 year. The cognitive change score at 6 weeks and 1 year after surgery in patients with and without 6-week abnormal global cortical amyloid deposition was 0.108 (0.186) vs. 0.095 (0.311); p=0.92; and 0.193 (0.266) vs. −0.121 (0.325); p=0.62, respectively.
Figure 1.
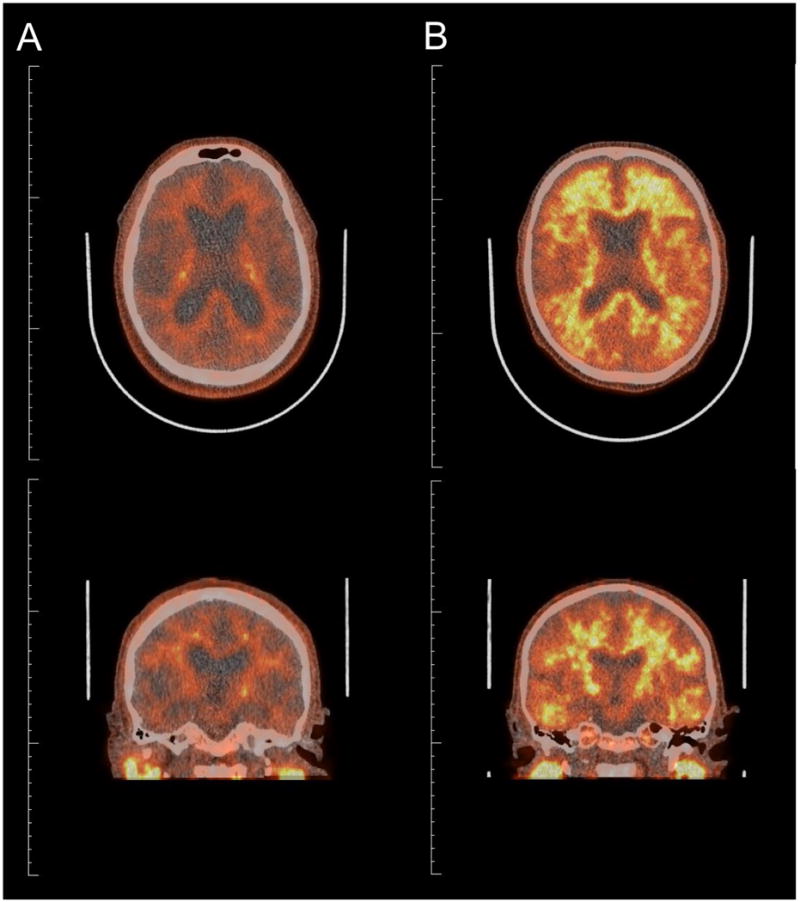
Images of patients with normal (A) and abnormal (B) amyloid deposition by 18F-florbetapir PET imaging. The brighter orange to yellow colors indicate greater amyloid deposition.
With regard to our primary outcome, 6-week global cortical amyloid deposition was not different in patients with and without cognitive deficit at 6 weeks (median standard uptake value ratio [interquartile range, IQR]: 0.92 [0.89–1.07] vs. 0.98 [0.93–1.05]; p=0.455; Table 2), nor was there a difference in the proportion of patients with and without postoperative cognitive dysfunction who had abnormal amyloid deposition (proportion difference: 0.106). Similar patterns were seen at 1 year after surgery (median standard uptake value ratio [IQR]: 0.96 [0.89–1.02] vs. 1.02 [0.97–1.06]). Abnormal 6-week amyloid deposition was seen in 3 patients with cognitive deficit and in 3 patients without deficit at 6 weeks postoperatively (p=0.644; Table 3). Similarly, 1 patient with cognitive deficit at 1 year had abnormal global cortical amyloid deposition at 1 year while 1 patient without deficit had abnormal deposition. There were no significant correlations of global cortical amyloid deposition at 6 weeks with baseline, 6-week, 1-year, or 3-year cognitive scores or change scores. In multivariable regression analyses, we found no significant association between 6-week abnormal global cortical amyloid burden and cognitive change scores (beta: 0.09, model R2: 0.12) or with the occurrence of postoperative cognitive dysfunction at 6 weeks (odds ratio [OR] 0.47, 95% confidence interval [CI] 0.07–3.43, model R2 0.09) when controlling for age, years of education, and baseline cognition. There were also no significant associations of 6-week amyloid burden with cognitive outcomes at 1 or 3 years after surgery.
Table 2.
Global cortical and regional SUVr values in patients with and without cognitive deficit at 6 weeks
| Brain Region | No Deficit (n = 24) SUVr (median[IQR]) |
Deficit (n = 13) SUVr (median[IQR]) |
P Value | FDR | Mean Difference [95% CI] |
|---|---|---|---|---|---|
| Global cortical | 0.98 [0.93–1.05] | 0.92 [0.89–1.07] | 0.455 | 0.754 | −0.06 [−0.23–0.10] |
| Posterior cingulate | 0.98 [0.88–1.04] | 0.87 [0.84–1.08] | 0.514 | 0.754 | −0.04 [−0.21–0.13] |
| Precuneus | 1.09 [1.02–1.15] | 0.99 [0.94–1.20] | 0.417 | 0.754 | −0.05 [−0.24–0.15] |
| Frontal medial orbital | 0.89 [0.87–0.93] | 0.88 [0.85–0.92] | 0.691 | 0.754 | −0.06 [−0.18–0.07] |
| Parietal | 0.90 [0.82–1.03] | 0.89 [0.81–1.09] | 0.622 | 0.754 | −0.09 [−0.26–0.09] |
| Temporal | 1.07 [1.01–1.13] | 1.06 [1.00–1.16] | 0.787 | 0.787 | −0.07 [−0.22–0.07] |
| Anterior cingulate | 1.01 [0.95–1.10] | 0.96 [0.93–1.07] | 0.417 | 0.754 | −0.08 [−0.29–0.12] |
| Hippocampus | 1.05 [1.01–1.09] | 1.10 [1.02–1.12] | 0.417 | 0.754 | 0 [−0.06–0.06] |
| Centrum | 1.67 [1.59–1.78] | 1.62 [1.56–1.71] | 0.301* | 0.754 | 0.04 [−0.05–0.13] |
| Occipital | 1.00 [0.86–1.04] | 0.94 [0.90–1.08] | 0.763* | 0.754 | −0.02 [−0.14–0.09] |
| Putamen | 1.19 [1.15–1.30] | 1.18 [1.13–1.24] | 0.417 | 0.754 | 0.01 [−0.08–0.09] |
| Caudate | 0.81 [0.70–0.94] | 0.96 [0.85–1.03] | 0.047 | 0.561 | −0.12 [−0.25–0] |
SUVr, standard uptake value relative to cerebellum; IQR, interquartile range; FDR, false discovery rate; CI, confidence interval.
P-value determined by t-test. All other P-values determined by Wilcoxon rank sum test.
Table 3.
Frequency of abnormal global cortical and regional amyloid deposition at 6 weeks in patients with and without cognitive deficit
| Brain Region | No Deficit (n = 24) SUVr ≥1.1 (%) |
Deficit (n = 13) SUVr ≥1.1 (%) |
P Value†‡ | FDR |
|---|---|---|---|---|
| Global cortical | 3 (12.5) | 3 (23.1) | 0.644† | 0.787 |
| Posterior cingulate | 3 (12.5) | 3 (23.1) | 0.644† | 0.787 |
| Precuneus | 9 (37.5) | 5 (38.5) | 0.954‡ | 1 |
| Frontal medial orbital | 0 (0) | 2 (15.4) | 0.117† | 0.429 |
| Parietal | 2 (8.3) | 2 (15.4) | 0.602† | 0.787 |
| Temporal | 7 (29.2) | 6 (46.2) | 0.302‡ | 0.787 |
| Anterior cingulate | 5 (25.0) | 6 (46.3) | 0.108† | 1 |
| Hippocampus | 5 (20.8) | 7 (54.8) | 0.041‡ | 0.429 |
| Occipital | 3 (12.5) | 3 (23.1) | 0.644† | 0.787 |
| Putamen | 23 (95.8) | 10 (76.9) | 0.115† | 0.429 |
| Caudate | 2 (8.3) | 0 (0) | 0.532† | 0.787 |
SUVr, standard uptake value relative to cerebellum; FDR, false discovery rate. P-value determined by
Fischer exact test or
Chi-square test.
Regional amyloid deposition and postoperative cognitive dysfunction
In post-hoc analyses we found that the frequency of abnormal amyloid deposition (standard uptake value ratio ≥ 1.1) in the hippocampus was significantly different between patients with and without postoperative cognitive dysfunction at 6 weeks postoperatively, although this difference was no longer significant after adjustment for multiple comparisons (p=0.041, false discovery rate=0.429; Table 3). Patients who had abnormal 6-week amyloid in the hippocampus showed significantly greater decline in the structured verbal memory domain (median change score [IQR]: −1.08 [−2.05 to −0.69] in patients with standard uptake value ratio ≥1.1 vs. −0.075 [−0.82 to 0.46] for patients with standard uptake value ratio <1.1; p=0.019). Total hippocampal standard uptake value ratio (continuous variable), although higher, was not statistically different between patients with and without deficit (Table 2). The caudate standard uptake value ratio was also greater in patients who had a cognitive deficit at 6 weeks, but this difference was no longer significant after adjustment for multiple comparisons (median [IQR]: Deficit 0.96 [0.85–1.03] vs. No Deficit 0.81 [0.70–0.94]; p=0.047, false discovery rate=0.561). Furthermore, the standard uptake value ratios in the caudate failed to meet the ≥1.1 threshold for defining abnormal amyloid deposition.
Trajectory of amyloid deposition
While cognitive deficit and the cognitive change score was not associated with abnormal global cortical amyloid deposition in the smaller cohort of 12 patients with 1-year neuroimaging, amyloid deposition increased in many brain regions over time. In these 12 patients, global cortical amyloid deposition increased significantly from 6 weeks to 1 year (mean standard uptake value ratio change: 0.02±0.02; p=0.011). Statistically significant increases in standard value uptake ration from 6 weeks to 1 year postoperatively were observed in the hippocampus, posterior cingulate, caudate, and occipital regions (Figure 2).
Figure 2.

Box plots showing median (IQR) change in SUVr in each imaged brain region from 6 weeks to 1 year postoperatively. Positive values represent increase in SUVr, while negative values represent decrease in SUVr. N=12.
Comparison to Alzheimer’s Disease Neuroimaging Initiative cohort
Overall, our cardiac surgical cohort was more similar to the Alzheimer’s Disease Neuroimaging Initiative subjects with normal cognition than the age- and sex-matched early or late mild cognitive impairment cohorts, with regard to education and apolipoprotein E4 carrier status (Supplemental Table 2) and global and regional amyloid deposition (Supplemental Table 3 and Supplemental Figure 2).
Abnormal amyloid deposition and cognitive deficit in apolipoprotein E4 carriers
Apolipoprotein E genotype was available for 37 patients in our cohort. Eight patients were found to be apolipoprotein E4 carriers (7 heterozygous, 1 homozygous), and 29 were non-carriers. Of the 8 apolipoprotein E4 carriers, 7 had complete 6-week cognitive testing data; of the 29 non-carriers, 27 had complete 6-week cognitive testing data. Of the 7 apolipoprotein E4 carriers with complete 6-week cognitive testing, 4 (57%) demonstrated cognitive deficit at 6 weeks. This was not statistically different from the deficit rate in the non-apolipoprotein E4 carriers (7/27, 26%; p=0.178). Apolipoprotein E4 genotype was, however, significantly associated with worse baseline cognitive score [mean (SD): −0.631 (0.382) vs. 0.001 (0.555) in non-carriers; p=0.005), but there was no difference at 6 weeks or in the change score from baseline to 6 weeks between apolipoprotein E4 carriers and non-carriers. Global and regional standard uptake value ratios in apolipoprotein E4 carriers versus non-carriers are shown in Supplemental Table 4. ANCOVA model incorporating the Alzheimer’s Disease Neuroimaging Initiative cohort revealed a significant association of global cortical amyloid deposition at 6 weeks with apolipoprotein E4 genotype (p<0.001) and age (p=0.001) and that the surgical cohort with cognitive deficit at 6 weeks had smaller global cortical amyloid deposition at 6 weeks than the late mild cognitive impairment subjects (p=0.001) in the Alzheimer’s Disease Neuroimaging Initiative cohort, but not the normal (p=0.68) or early mild cognitive impairment patients (p=0.07).
When evaluating regional standard uptake value ratio ≥ 1.1 in apolipoprotein E4 carriers, only the parietal region was different between the carriers and non-carriers before adjustment for multiple comparisons (38% (3/8) in carriers vs. 3% (1/29) in non-carriers; p=0.02, false discovery rate=0.282).
Discussion
We did not find an association between 6-week global cortical amyloid burden and cognitive dysfunction at 6 weeks following cardiac surgery.
Cognitive dysfunction after cardiac surgery remains a significant problem without a clear etiology. Given that cardiac surgery predominantly takes place in the aged, the possibility exists that the cognitive decline seen in some patients is similar to mild cognitive impairment, which in many will eventually progress to Alzheimer’s disease. Both diseases are believed to involve the accumulation of β-amyloid and tau proteins in the brain. While the role of β-amyloid oligomers in the pathogenesis of Alzheimer’s disease remains controversial26, there is evidence that β-amyloid can lead to synaptic dysfunction and memory deficits in animals27. CPB is known to disrupt the blood-brain barrier28, and blood-brain barrier dysfunction is associated with increased entry of amyloid into the brain.29 Alzheimer’s-type neurodegeneration is accelerated by neuroinflammation30, raising the possibility that perioperative inflammation could stimulate/accelerate β-amyloid-mediated neurologic degeneration, which could contribute to postoperative cognitive dysfunction. Finally, cardiac surgical patients share many of the risk factors for Alzheimer’s disease31, and there is a known intersection between Alzheimer’s and cardiovascular/cerebrovascular disease.32 While there is no evidence that cardiovascular disease severity directly affects amyloid burden33, it is plausible that some component of postoperative cognitive dysfunction may be related to cardiovascular risk factors that accelerate the progression towards Alzheimer’s-type cognitive decline.
To investigate the relationship between postoperative cognitive dysfunction and β-amyloid protein deposition in patients undergoing cardiac surgery, we utilized the novel positron emission tomography tracer, 18F-florbetapir, which binds with high affinity to β-amyloid fibrils and has been shown to differentiate cerebral β-amyloid deposition between cognitively normal and deficient subjects.34 With regard to our primary outcome, we did not find a significant association between 6-week global cortical amyloid burden and cognitive dysfunction at 6 weeks. 6-week amyloid burden was also not associated with cognitive dysfunction at 1 or 3 years postoperatively, although our sample size at these time points was very small.
In post-hoc analyses of regional amyloid deposition, we found an increased proportion of patients with cognitive dysfunction at 6 weeks with abnormal 6-week amyloid deposition in the hippocampus, which was associated with a verbal memory deficit. While these results were not significant after adjustment for multiple comparisons, the unadjusted findings may point to regions that deserve closer scrutiny in future studies. The hippocampus is an intriguing region because it plays an important role in the acquisition and storage of episodic memories35 – those related to unique personal experiences – and has been linked with postsurgical cognitive changes in animals36. Hippocampal synapse loss occurs early in Alzheimer’s and to a greater extent than in other brain regions in advanced Alzheimer’s. Furthermore, hippocampal damage correlates better with cognitive impairment in Alzheimer’s disease than the presence/quantity of β-amyloid plaques or neurofibrillary tangles.37 Finally, Badgaiya et al. previously showed significant decreases in memory-related regional cerebral blood flow within the hippocampus and parahippocampus after cardiac surgery,38 which may indicate regions of the brain that are more vulnerable to ischemic blood-brain barrier dysfunction and consequent cerebral deposition of circulating amyloid after cardiac surgery.29
We also observed that amyloid deposition significantly increased from 6 weeks to 1 year after surgery in many regions of the brain and that this rate of increase was greater than that reported elsewhere. In a study by Palmqvist et al.39 the mean global standard uptake value ratio change/year in non-demented subjects with normal positron emission tomography scans was 0.0024 (95% CI: 0.0010–0.0039), while that for subjects with abnormal CSF and scan results was 0.011 (95% CI: 0.0083–0.013). Similarly, in the longitudinal Alzheimer’s Disease Neuroimaging Initiative cohort, the mean standard uptake value ratio change in 154 control subjects with a normal baseline scan was 0.0027 (0.0100) but was 0.0160 (0.0161) in 61 controls with abnormal baseline scans (Susan Landau, PhD, personal communication). In comparison, the mean (SD) global cortical standard uptake value ratio change in our surgical patients was 0.02 (0.02), nearly 10-fold that seen in scan-negative non-surgical patients and just slightly higher than that in scan-positive non-surgical patients. When we removed patients with abnormal 6-week amyloid deposition (n=2) from our analyses, the rate of change in our surgical cohort remained higher at 0.014 (0.018). Percent standard uptake value ratio change, which discounts variation in reference regions, may be more informative; in the Palmqvist study39, the percent global standard uptake value ratio change/year was 0.35% (95% CI: 0.14%–0.56%) compared to 1.9 ± 2.0% in our surgical cohort. However, our study sample is limited, and changes over the course of a year are small.
This trajectory of amyloid at 1 year after surgery raises the question of how surgery and/or anesthesia may impact cerebral amyloid deposition and longer term cognitive outcomes. Several in vitro and animal studies have established a link between anesthetic agents and enhanced β-amyloid formation, aggregation, and β-amyloid-induced cytotoxicity.5,6 Human studies have demonstrated that low preoperative ratios of β-amyloid/Tau proteins in the CSF are associated with postoperative delirium and cognitive decline, although this is thought to predict a predisposition to cognitive dysfunction due to preclinical Alzheimer’s disease rather than a direct anesthetic effect.40 Surgery may also have an independent effect on the risk of Alzheimer’s development in the postoperative setting.41 Tang et al.7 and Berger et al.8 both demonstrated a CSF change in the ratio of β-amyloid/Tau in patients undergoing surgery consistent with that seen in Alzheimer’s and correlated with perioperative neuroinflammatory mediator release. The association of perioperative inflammatory changes and Alzheimer’s-like cognitive decline may be particularly relevant in the highly inflammatory milieu of cardiac surgery. Cardiac surgery with CPB has been show to produce an intense cerebral inflammatory response in conjunction with Alzheimer’s-like changes in CSF β-amyloid.42 Vascular dysfunction and inflammation, both hallmarks of cardiac surgery, have also been associated with amyloid deposition43. Thus, one concern has been whether cardiac surgery itself could increase the rate of amyloid deposition as a consequence of blood-brain barrier disruption28.
Our finding that some patients experienced cognitive decline over time while others improved also merits further study and correlation with observed changes in the trajectory of global and regional cortical amyloid deposition. Cognitive improvement over time after surgery is certainly a recognized phenomenon and can been seen either globally or in select cognitive domains44. However, no mechanistic explanation has yet been uncovered as to why some patients improve while others continue to decline/recover more slowly.
It is important to interpret our findings in the context of our sample size limitations. Without any prior studies on amyloid burden in surgical patients, our initial sample size estimation was based on the hypothesis that patients with postoperative cognitive dysfunction would have amyloid deposition to individuals with mild cognitive impairment. For our primary outcome, comparison of the binary variables of abnormal amyloid deposition and postoperative cognitive dysfunction demonstrated a proportion difference of 0.106; we are only powered to detect a proportion difference of 0.5, thus we cannot conclusively exclude an association between 6-week abnormal amyloid deposition and a clinically meaningful decline in cognitive function after cardiac surgery. Furthermore, we estimate that we have 80% power to detect a mean standard uptake value ratio difference of 0.16 between patients with and without postoperative cognitive at 6 weeks postoperatively; thus, our detected standard uptake value ratio difference of 0.06 falls below this threshold. Based on these data, we estimate that 117 patients would be needed to achieve 80% power in a future study (see Supplement I).
The lack of baseline imaging and longer term (> 3 years) follow-up are further limitations of our study. While the existing literature indicates a longer time course for change in amyloid17, we cannot say with certainty that surgery does not produce changes in amyloid deposition in the immediate postoperative period. Future studies should include a baseline assessment of brain amyloid prior to surgery as well as longer duration of follow-up. Based on the existing literature in mild cognitive impairment subjects, the time course of clinically significant β-amyloid deposition needed to produce cognitive decline may be significantly longer.45,46 Finally, we are limited by the relatively younger and male-dominated nature of our surgical cohort. Older age has been shown to increase the risk for postoperative cognitive dysfunction47 and Alzheimer’s disease, and multiple studies have indicated that females have a higher prevalence and incidence of Alzheimer’s disease48 and mild cognitive impairment progression over time than men49. Future studies should include an older surgical group that more closely matches the Alzheimer’s Disease Neuroimaging Initiative mild cognitive impairment cohort.
In conclusion, this study employed amyloid imaging using 18F-florbetapir to investigate 6-week and evolving brain amyloid burden in patients undergoing cardiac surgery with CPB. We observed that postoperative cognitive dysfunction was not associated with 6-week global cortical amyloid deposition, but the rate of amyloid deposition following surgery was greater than what has been reported in normal elderly subjects. The findings from this study support further investigation of 1) the relationship between hippocampal amyloid deposition and early postoperative cognitive dysfunction and 2) the significance of amyloid deposition increases within 1 year of cardiac surgery.
Supplementary Material
Brief Summary Statement.
Cerebral amyloid deposition as measured by 18F-florbetapir PET/CT was not associated with changes in cognitive performance in patients after cardiac surgery.
Acknowledgments
We thank Abhinay Joshi from Avid Radiopharmaceuticals for assistance with image analytics in a blinded fashion. We also thank Avid Radiopharmaceuticals for their support of this study through the provision of 18F-florbetapir.
Regarding the ADNI data used in this study: data collection and sharing for this project was funded by the Alzheimer’s Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (www.fnih.org). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Funding. This study is supported in part by grants # HL108280, HL096978, and HL109971 from the National Institutes of Health.18F-florbetapir is provided courtesy of Avid Radiopharmaceuticals, but Avid had no input into the clinical study design or decision to publish this report.
Conflicts of Interest/Disclosures
PMD has received research grants (through Duke University) from Avid, Lilly, Neuronetrix, Avanir, Alzheimer’s Drug Discovery Foundation, Forum, and has received speaking or advisory fees from Anthrotronix, Cognoptix, Takeda, Genomind, Sonexa, Targacept, Neurocog Trials, Forum, T3D Therapeutics, Alzheimer’s Association, Hintsa, MindLink, Global Alzheimer’s Platform, and University of Miami. PMD owns shares in Maxwell Health, Muses Labs, Anthrotronix, Evidation Health, Turtle Shell Technologies, and Advera Health Analytics. PMD is a co-inventor on patents relating to dementia biomarkers that are unlicensed.
Appendix I
*Alzheimer’s Disease Neuroimaging Initiative (ADNI)
I. ADNI I, GO and II
Part A: Leadership and Infrastructure
Principal Investigator
| Michael W. Weiner, MD | UC San Francisco |
ADCS PI and Director of Coordinating Center Clinical Core
| Paul Aisen, MD | University of Southern California |
Executive Committee
| Michael Weiner, MD | UC San Francisco |
| Paul Aisen, MD | University of Southern California |
| Ronald Petersen, MD, PhD | Mayo Clinic, Rochester |
| Clifford R. Jack, Jr., MD | Mayo Clinic, Rochester |
| William Jagust, MD | UC Berkeley |
| John Q. Trojanowki, MD, PhD | U Pennsylvania |
| Arthur W. Toga, PhD | USC |
| Laurel Beckett, PhD | UC Davis |
| Robert C. Green, MD, MPH | Brigham and Women’s Hospital/Harvard Medical School |
| Andrew J. Saykin, PsyD | Indiana University |
| John Morris, MD | Washington University St. Louis |
| Leslie M. Shaw | University of Pennsylvania |
ADNI External Advisory Board (ESAB)
| Zaven Khachaturian, PhD | Prevent Alzheimer’s Disease 2020 (Chair) |
| Greg Sorensen, MD | Siemens |
| Maria Carrillo, PhD | Alzheimer’s Association |
| Lew Kuller, MD | University of Pittsburgh |
| Marc Raichle, MD | Washington University St. Louis |
| Steven Paul, MD | Cornell University |
| Peter Davies, MD | Albert Einstein College of Medicine of Yeshiva University |
| Howard Fillit, MD | AD Drug Discovery Foundation |
| Franz Hefti, PhD | Acumen Pharmaceuticals |
| David Holtzman, MD | Washington University St. Louis |
| M. Marcel Mesulam, MD | Northwestern University |
| William Potter, MD | National Institute of Mental Health |
| Peter Snyder, PhD | Brown University |
ADNI 2 Private Partner Scientific Board (PPSB)
| Adam Schwartz, MD | Eli Lilly (Chair) |
Data and Publications Committee
| Robert C. Green, MD, MPH | BWH/HMS (Chair) |
Resource Allocation Review Committee
| Tom Montine, MD, PhD | University of Washington (Chair) |
Clinical Core Leaders
| Ronald Petersen, MD, PhD | Mayo Clinic, Rochester (Core PI) |
| Paul Aisen, MD | University of Southern California |
Clinical Informatics and Operations
| Ronald G. Thomas, PhD | UC San Diego |
| Michael Donohue, PhD | UC San Diego |
| Sarah Walter, MSc | UC San Diego |
| Devon Gessert | UC San Diego |
| Tamie Sather, MA | UC San Diego |
| Gus Jiminez, MBS | UC San Diego |
| Archana B. Balasubramanian, PhD | UC San Diego |
| Jennifer Mason, MPH | UC San Diego |
| Iris Sim | UC San Diego |
Biostatistics Core Leaders and Key Personnel
| Laurel Beckett, PhD | UC Davis (Core PI) |
| Danielle Harvey, PhD | UC Davis |
| Michael Donohue, PhD | UC San Diego |
MRI Core Leaders and Key Personnel
| Clifford R. Jack, Jr., MD | Mayo Clinic, Rochester (Core PI) |
| Matthew Bernstein, PhD | Mayo Clinic, Rochester |
| Nick Fox, MD | University of London |
| Paul Thompson, PhD | UCLA School of Medicine |
| Norbert Schuff, PhD | UCSF MRI |
| Charles DeCArli, MD | UC Davis |
| Bret Borowski, RT | Mayo Clinic |
| Jeff Gunter, PhD | Mayo Clinic |
| Matt Senjem, MS | Mayo Clinic |
| Prashanthi Vemuri, PhD | Mayo Clinic |
| David Jones, MD | Mayo Clinic |
| Kejal Kantarci | Mayo Clinic |
| Chad Ward | Mayo Clinic |
PET Core Leaders and Key Personnel
| William Jagust, MD | UC Berkeley (Core PI) |
| Robert A. Koeppe, PhD | University of Michigan |
| Norm Foster, MD | University of Utah |
| Eric M. Reiman, MD | Banner Alzheimer’s Institute |
| Kewei Chen, PhD | Banner Alzheimer’s Institute |
| Susan Landau, PhD | UC Berkeley |
Neuropathology Core Leaders
| John C. Morris, MD | Washington University St. |
| Louis Nigel J. Cairns, PhD, FRCPath | Washington University St. Louis |
| Erin Franklin, MS, CCRP | Washington University St. Louis |
| Lisa Taylor-Reinwald, BA, HTL | Washington University St. Louis (ASCP) – Past Investigator |
Biomarkers Core Leaders and Key Personnel
| Leslie M. Shaw, PhD | UPenn School of Medicine |
| John Q. Trojanowki, MD, PhD | UPenn School of Medicine |
| Virginia Lee, PhD, MBA | UPenn School of Medicine |
| Magdalena Korecka, PhD | UPenn School of Medicine |
| Michal Figurski, PhD | UPenn School of Medicine |
Informatics Core Leaders and Key Personnel
| Arthur W. Toga, PhD | USC (Core PI) |
| Karen Crawford | USC |
| Scott Neu, PhD | USC |
Genetics Core Leaders and Key Personnel
| Andrew J. Saykin, PsyD | Indiana University |
| Tatiana M. Foroud, PhD | Indiana University |
| Steven Potkin, MD UC | UC Irvine |
| Li Shen, PhD | Indiana University |
| Kelley Faber, MS, CCRC | Indiana University |
| Sungeun Kim, PhD | Indiana University |
| Kwangsik Nho, PhD | Indiana University |
Initial Concept Planning & Development
| Michael W. Weiner, MD | UC San Francisco |
| Lean Thal, MD | UC San Diego |
| Zaven Khachaturian, PhD | Prevent Alzheimer’s Disease 2020 |
Early Project Proposal Development
| Leon Thal, MD | UC San Diego |
| Neil Buckholtz | National Institute on Aging |
| Michael W. Weiner, MD | UC San Francisco |
| Peter J. Snyder, PhD | Brown University |
| William Potter, MD | National Institute of Mental Health |
| Steven Paul, MD | Cornell University |
| Marilyn Albert, PhD | Johns Hopkins University |
| Richard Frank, MD, PhD | Richard Frank Consulting |
| Zaven Khachaturian, PhD | Prevent Alzheimer’s Disease 2020 |
NIA
| John Hsiao, MD | National Institute on Aging |
Part B: Investigators By Site
Oregon Health & Science University:
Jeffrey Kaye, MD
Joseph Quinn, MD
Lisa Silbert, MD
Betty Lind, BS
Raina Carter, BA – Past Investigator
Sara Dolen, BS – Past Investigator
University of Southern California:
Lon S. Schneider, MD
Sonia Pawluczyk, MD
Mauricio Becerra, BS
Liberty Teodoro, RN
Bryan M. Spann, DO, PhD – Past Investigator
University of California – San Diego:
James Brewer, MD, PhD
Helen Vanderswag, RN
Adam Fleisher, MD – Past Investigator
University of Michigan:
Judith L. Heidebrink, MD, MS
Joanne L. Lord, LPN, BA, CCRC – Past
Investigator
Mayo Clinic, Rochester:
Ronald Petersen, MD, PhD Sara S. Mason, RN
Colleen S. Albers, RN David Knopman, MD
Kris Johnson, RN – Past Investigator
Baylor College of Medicine:
Rachelle S. Doody, MD, PhD
Javier Villanueva-Meyer, MD
Valory Pavlik, PhD
Victoria Shibley, MS
Munir Chowdhury, MBBS, MS – Past Investigator
Susan Rountree, MD – Past Investigator
Mimi Dang, MD – Past Investigator
Columbia University Medical Center:
Yaakov Stern, PhD
Lawrence S. Honig, MD, PhD
Karen L. Bell, MD
Washington University, St. Louis:
Beau Ances, MD
John C. Morris, MD
Maria Carroll, RN, MSN
Mary L. Creech, RN, MSW
Erin Franklin, MS, CCRP
Mark A. Mintun, MD – Past Investigator
Stacy Schneider, APRN, BC, GNP – Past Investigator
Angela Oliver, RN, BSN, MSG – Past Investigator
University of Alabama - Birmingham:
Daniel Marson, JD, PhD
David Geldmacher, MD
Marissa Natelson Love, MD
Randall Griffith, PhD, ABPP – Past Investigator
David Clark, MD – Past Investigator
John Brockington, MD – Past Investigator
Erik Roberson, MD – Past Investigator
Mount Sinai School of Medicine:
Hillel Grossman, MD
Effie Mitsis, PhD
Rush University Medical Center:
Raj C. Shah, MD
Leyla deToledo-Morrell, PhD – Past Investigator
Wien Center:
Ranjan Duara, MD
Maria T. Greig-Custo, MD
Warren Barker, MA, MS
Johns Hopkins University:
Marilyn Albert, PhD Chiadi Onyike, MD Daniel D’Agostino II, BS
Stephanie Kielb, BS – Past Investigator
New York University: Martin Sadowski, MD, PhD Mohammed O. Sheikh, MD Anaztasia Ulysse
Mrunalini Gaikwad
Duke University Medical Center:
P. Murali Doraiswamy, MBBS, FRCP
Jeffrey R. Petrella, MD
Salvador Borges-Neto, MD
Terence Z. Wong, MD – Past Investigator
Edward Coleman – Past Investigator
University of Pennsylvania:
Steven E. Arnold, MD
Jason H. Karlawish, MD
David A. Wolk, MD
Christopher M. Clark, MD
University of Kentucky:
Charles D. Smith, MD
Greg Jicha, MD
Peter Hardy, PhD
Partha Sinha, PhD
Elizabeth Oates, MD
Gary Conrad, MD
University of Pittsburgh:
Oscar L. Lopez, MD
MaryAnn Oakley, MA
Donna M. Simpson, CRNP, MPH
University of Rochester Medical Center:
Anton P. Porsteinsson, MD
Bonnie S. Goldstein, MS, NP
Kim Martin, RN
Kelly M. Makino, BS – Past Investigator
M. Saleem Ismail, MD – Past Investigator
Connie Brand, RN – Past Investigator
University of California, Irvine:
Steven G. Potkin, MD
Adrian Preda, MD Dana Nguyen, PhD
University of Texas Southwestern Medical School:
Kyle Womack, MD
Dana Mathews, MD, PhD
Mary Quiceno, MD
Emory University:
Allan I. Levey, MD, PhD
James J. Lah, MD, PhD
Janet S. Cellar, DNP, PMHCNS-BC
University of Kansas, Medical Center:
Jeffrey M. Burns, MD
Russell H. Swerdlow, MD
William M. Brooks, PhD
University of California, Los Angeles:
Liana Apostolova, MD
Kathleen Tingus, PhD
Ellen Woo, PhD
Daniel H.S. Silverman, MD, PhD
Po H. Lu, PsyD – Past Investigator
George Bartzokis, MD – Past Investigator
Mayo Clinic, Jacksonville:
Neill R Graff-Radford, MBBCH, FRCP (London)
Francine Parfitt, MSH, CCRC Kim Poki-Walker, BA
Indiana University:
Martin R. Farlow, MD
Ann Marie Hake, MD
Brandy R. Matthews, MD – Past Investigator
Jared R. Brosch, MD
Scott Herring, RN, CCRC
Yale University School of Medicine:
Christopher H. van Dyck, MD
Richard E. Carson, PhD
Martha G. MacAvoy, PhD
Pradeep Varma, MD
McGill Univ., Montreal-Jewish General Hospital:
Howard Chertkow, MD
Howard Bergman, MD
Chris Hosein, MEd
Sunnybrook Health Sciences, Ontario:
Sandra Black, MD, FRCPC
Bojana Stefanovic, PhD
Curtis Caldwell, PhD
U.B.C. Clinic for AD & Related Disorders:
Ging-Yuek
Robin Hsiung, MD, MHSc, FRCPC
Benita Mudge, BS
Vesna Sossi, PhD
Howard Feldman, MD, FRCPC – Past Investigator
Michele Assaly, MA – Past Investigator
Cognitive Neurology - St. Joseph’s, Ontario:
Elizabeth Finger, MD
Stephen Pasternack, MD, PhD
Irina Rachisky, MD
Dick Trost, PhD – Past Investigator
Andrew Kertesz, MD – Past Investigator
Cleveland Clinic Lou Ruvo Center for Brain Health:
Charles Bernick, MD, MPH
Donna Munic, PhD
Northwestern University:
Marek-Marsel Mesulam, MD
Emily Rogalski, PhD
Kristine Lipowski, MA
Sandra Weintraub, PhD
Borna Bonakdarpour, MD
Diana Kerwin, MD – Past Investigator
Chuang-Kuo Wu, MD, PhD – Past Investigator
Nancy Johnson, PhD – Past Investigator
Premiere Research Inst (Palm Beach Neurology):
Carl Sadowsky, MD
Teresa Villena, MD
Georgetown University Medical Center:
Raymond Scott Turner, MD, PhD
Kathleen Johnson, NP
Brigid Reynolds, NP
Brigham and Women’s Hospital:
Reisa A. Sperling, MD
Keith A. Johnson, MD
Gad Marshall, MD
Stanford University:
Jerome Yesavage, MD
Joy L. Taylor, PhD
Barton Lane, MD
Allyson Rosen, PhD – Past Investigator
Jared Tinklenberg, MD – Past Investigator
Banner Sun Health Research Institute:
Marwan N. Sabbagh, MD
Christine M. Belden, PsyD
Sandra A. Jacobson, MD
Sherye A. Sirrel, CCRC
Boston University:
Neil Kowall, MD
Ronald Killiany, PhD
Andrew E. Budson, MD
Alexander Norbash, MD – Past Investigator
Patricia Lynn Johnson, BA – Past Investigator
Howard University:
Thomas O. Obisesan, MD, MPH
Saba Wolday, MSc
Joanne Allard, PhD
Case Western Reserve University:
Alan Lerner, MD
Paula Ogrocki, PhD
Curtis Tatsuoka, PhD
Parianne Fatica, BA, CCRC
University of California, Davis – Sacramento:
Evan Fletcher, PhD
Pauline Maillard, PhD
John Olichney, MD
Charles DeCarli, MD – Past Investigator
Owen Carmichael, PhD – Past Investigator
Neurological Care of CNY:
Smita Kittur, MD – Past Investigator
Parkwood Hospital:
Michael Borrie, MB ChB
T-Y Lee, PhD
Dr Rob Bartha, PhD
University of Wisconsin:
Sterling Johnson, PhD
Sanjay Asthana, MD
Cynthia M. Carlsson, MD, MS
University of California, Irvine - BIC:
Steven G. Potkin, MD Adrian Preda, MD Dana Nguyen, PhD
Banner Alzheimer’s Institute:
Pierre Tariot, MD Anna Burke, MD
Ann Marie Milliken, NMD
Nadira Trncic, MD, PhD, CCRC – Past Investigator
Adam Fleisher, MD – Past Investigator
Stephanie Reeder, BA – Past Investigator
Dent Neurologic Institute:
Vernice Bates, MD Horacio Capote, MD
Michelle Rainka, PharmD, CCRP
Ohio State University:
Douglas W. Scharre, MD Maria Kataki, MD, PhD Brendan Kelley, MD
Albany Medical College: Earl A. Zimmerman, MD Dzintra Celmins, MD Alice D. Brown, FNP
Hartford Hospital, Olin Neuropsychiatry
Research Center: Godfrey D. Pearlson, MD Karen Blank, MD
Karen Anderson, RN
Dartmouth-Hitchcock Medical Center:
Laura A. Flashman, PhD
Marc Seltzer, MD
Mary L. Hynes, RN, MPH
Robert B. Santulli, MD – Past Investigator
Wake Forest University Health Sciences:
Kaycee M. Sink, MD, MAS
Leslie Gordineer
Jeff D. Williamson, MD, MHS – Past Investigator
Pradeep Garg, PhD – Past Investigator
Franklin Watkins, MD – Past Investigator
Rhode Island Hospital:
Brian R. Ott, MD
Geoffrey Tremont, PhD
Lori A. Daiello, Pharm.D, ScM
Butler Hospital:
Stephen Salloway, MD, MS
Paul Malloy, PhD
Stephen Correia, PhD
UC San Francisco:
Howard J. Rosen, MD
Bruce L. Miller, MD
David Perry, MD
Medical University South Carolina:
Jacobo Mintzer, MD, MBA
Kenneth Spicer, MD, PhD
David Bachman, MD
St. Joseph’s Health Care:
Elizabeth Finger, MD
Stephen Pasternak, MD
Irina Rachinsky, MD
John Rogers, MD
Andrew Kertesz, MD – Past Investigator
Dick Drost, MD – Past Investigator
Nathan Kline Institute:
Nunzio Pomara, MD
Raymundo Hernando, MD
Antero Sarrael, MD
University of Iowa College of Medicine:
Susan K. Schultz, MD
Karen Ekstam Smith, RN
Hristina Koleva, MD
Ki Won Nam, MD
Hyungsub Shim, MD– Past Investigator
Cornell University:
Norman Relkin, MD, PhD
Gloria Chiang, MD
Michael Lin, MD
Lisa Ravdin, PhD
University of South Florida: USF Health ByrdAlzheimer’s Institute
Amanda Smith, MD
Balebail Ashok Raj, MD
Kristin Fargher, MD– Past Investigator
DOD ADNI
Part A: Leadership and Infrastructure
Principal Investigator
| Michael W. Weiner, MD | University of California, San Francisco |
ADCS PI and Director of Coordinating Center Clinical Core
| Paul Aisen, MD | University of Southern California |
Executive Committee
| Michael Weiner, MD | UC San Francisco |
| Paul Aisen, MD | University of Southern California |
| Ronald Petersen, MD, PhD | Mayo Clinic, Rochester |
| Robert C. Green, MD, MPH | Brigham and Women’s Hospital/Harvard Medical School |
| Danielle Harvey, PhD | UC Davis |
| Clifford R. Jack, Jr., MD | Mayo Clinic, Rochester |
| William Jagust, MD | UC Berkeley |
| John C. Morris, MD | Washington University St. Louis |
| Andrew J. Saykin, PsyD | Indiana University |
| Leslie M. Shaw, PhD | Perelman School of Medicine, UPenn |
| Arthur W. Toga, PhD | USC |
| John Q. Trojanowki, MD, PhD | Perelman School of Medicine, University of Pennsylvania |
Psychological Evaluation/PTSD Core
| Thomas Neylan, MD | UC San Francisco |
Traumatic Brain Injury/TBI Core
| Jordan Grafman, PhD | |
| Jordan Grafman, PhD | Rehabilitation Institute of Chicago, Feinberg School of Medicine, Northwestern University |
Data and Publication Committee (DPC)
| Robert C. Green, MD, MPH | BWH/HMS (Chair) |
Resource Allocation Review Committee
| Tom Montine, MD, PhD | University of Washington (Chair) |
Clinical Core Leaders
| Michael Weiner MD | Core PI |
| Ronald Petersen, MD, PhD | Mayo Clinic, Rochester (Core PI) |
| Paul Aisen, MD | University of Southern California |
Clinical Informatics and Operations
| Ronald G. Thomas, PhD | UC San Diego |
| Michael Donohue, PhD | UC San Diego |
| Devon Gessert | UC San Diego |
| Tamie Sather, MA | UC San Diego |
| Melissa Davis | UC San Diego |
| Rosemary Morrison, MPH | UC San Diego |
| Gus Jiminez, MBS | UC San Diego |
| Thomas Neylan, MD | UC San Francisco Jacqueline |
| Hayes | UC San Francisco Shannon |
| Finley | UC San Francisco |
Biostatistics Core Leaders and Key Personnel
| Danielle Harvey, PhD | UC Davis (Core PI) |
| Michael Donohue, PhD | UC San Diego |
MRI Core Leaders and Key Personnel
| Clifford R. Jack, Jr., MD | Mayo Clinic, Rochester (Core PI) |
| Matthew Bernstein, PhD | Mayo Clinic, Rochester |
| Bret Borowski, RT | Mayo Clinic |
| Jeff Gunter, PhD | Mayo Clinic |
| Matt Senjem, MS | Mayo Clinic Kejal |
| Kantarci | Mayo Clinic Chad |
| Ward | Mayo Clinic |
PET Core Leaders and Key Personnel
| William Jagust, MD | UC Berkeley (Core PI) |
| Robert A. Koeppe, PhD | University of Michigan |
| Norm Foster, MD | University of Utah |
| Eric M. Reiman, MD | Banner Alzheimer’s Institute |
| Kewei Chen, PhD | Banner Alzheimer’s Institute |
| Chet Mathis, MD | University of Pittsburgh |
| Susan Landau, PhD | UC Berkeley |
Neuropathology Core Leaders
| John C. Morris, MD | Washington University St. Louis |
| Nigel J. Cairns, PhD, FRCPath | Washington University St. Louis |
| Erin Householder, MS | Washington University St. Louis |
Biomarkers Core Leaders and Key Personnel
| Leslie M. Shaw, PhD | Perelman School of Medicine, UPenn |
| John Q. Trojanowki, MD, PhD | Perelman School of Medicine, UPenn |
| Virginia Lee, PhD, MBA | Perelman School of Medicine, UPenn |
| Magdalena Korecka, PhD | Perelman School of Medicine, UPenn |
| Michal Figurski, PhD | Perelman School of Medicine, UPenn |
Informatics Core Leaders and Key Personnel
| Arthur W. Toga, PhD | USC (Core PI) |
| Karen Crawford | USC |
| Scott Neu, PhD | USC |
Genetics Core Leaders and Key Personnel
| Andrew J. Saykin, PsyD | Indiana University |
| Tatiana M. Foroud, PhD | Indiana University |
| Steven Potkin, MD UC | UC Irvine |
| Li Shen, PhD | Indiana University |
| Kelley Faber, MS, CCRC | Indiana University |
| Sungeun Kim, PhD | Indiana University |
| Kwangsik Nho, PhD | Indiana University |
Initial Concept Planning & Development
| Michael W. Weiner, MD | UC San Francisco |
| Karl Friedl | Department of Defense (retired) |
Part B: Investigators By Site
University of Southern California:
Lon S. Schneider, MD, MS
Sonia Pawluczyk, MD
Mauricio Becerra
University of California, San Diego:
James Brewer, MD, PhD
Helen Vanderswag, RN
Columbia University Medical Center:
Yaakov Stern, PhD
Lawrence S. Honig, MD, PhD
Karen L. Bell, MD
Rush University Medical Center:
Debra Fleischman, Ph.D.
Konstantinos Arfanakis, Ph.D.
Raj C. Shah, M.D.
Wien Center:
Dr. Ranjan Duara MD PI
Dr. Daniel Varon MD Co-PI
Maria T Greig HP Coordinator
Duke University Medical Center:
P. Murali Doraiswamy, MBBS
Jeffrey R. Petrella, MD
Olga James, MD
University of Rochester Medical Center:
Anton P. Porsteinsson, MD (director)
Bonnie Goldstein, MS, NP (coordinator)
Kimberly S. Martin, RN
University of California, Irvine:
Steven G. Potkin, MD
Adrian Preda, MD
Dana Nguyen, PhD
Medical University South Carolina:
Jacobo Mintzer, MD, MBA
Dino Massoglia, MD, PhD
Olga Brawman-Mintzer, MD
Premiere Research Inst (Palm Beach Neurology):
Carl Sadowsky, MD
Walter Martinez, MD Teresa Villena, MD
University of California, San Francisco:
William Jagust MD
Susan Landau PhD
Howard Rosen, MD
David Perry
Georgetown University Medical Center:
Raymond Scott Turner, MD, PhD
Kelly Behan
Brigid Reynolds, NP
Brigham and Women’s Hospital:
Reisa A. Sperling, MD
Keith A. Johnson, MD
Gad Marshall, MD
Banner Sun Health Research Institute:
Marwan N. Sabbagh, MD
Sandra A. Jacobson, MD
Sherye A. Sirrel, MS, CCRC
Howard University:
Thomas O. Obisesan, MD, MPH
Saba Wolday, MSc
Joanne Allard, PhD
University of Wisconsin:
Sterling C. Johnson, Ph.D.
J. Jay Fruehling, M.A.
Sandra Harding, M.S.
University of Washington:
Elaine R. Peskind, MD
Eric C. Petrie, MD, MS
Gail Li, MD, PhD
Stanford University:
Jerome A. Yesavage, MD
Joy L. Taylor, PhD
Ansgar J. Furst, PhD
Steven Chao, M.D.
Cornell University:
Norman Relkin, MD, PhD
Gloria Chiang, MD
Lisa Ravdin, PhD
Appendix II
**Neurologic Outcome Research Group (NORG)
Director: Joseph P. Mathew, M.D., Co-Director: James A. Blumenthal, Ph.D. Anesthesiology: Miles Berger, M.D., Ph.D., Jorn A. Karhausen, M.D., Miklos D. Kertai, M.D., Rebecca Y. Klinger, M.D., M.S., Yi-Ju Li, Ph.D., Joseph P. Mathew, M.D., Mark F. Newman, M.D, Mihai V. Podgoreanu, M.D., Mark Stafford-Smith, M.D., Madhav Swaminathan, M.D., Niccolo Terrando, Ph.D., David S. Warner, M.D., Bonita L. Funk, R.N., CCRP, Narai Balajonda, M.D., Rachele Brassard, BSW, Tiffany Bisanar, R.N., B.S.N., Mary Cooter, MS, Yanne Toulgoat-Dubois, B.A., Peter Waweru, CCRP.
Behavioral Medicine: Michael A. Babyak, Ph.D., James A. Blumenthal, Ph.D., Jeffrey N. Browndyke, Ph.D., Kathleen A. Welsh-Bohmer, Ph.D.
Cardiology: Michael H. Sketch, Jr., M.D.
Neurology: Ellen R. Bennett, Ph.D., Carmelo Graffagnino, M.D., Daniel T. Laskowitz, M.D., Warren J. Strittmatter, M.D.
Perfusion Services: Kevin Collins, B.S., C.C.P., Greg Smigla, B.S., C.C.P., Ian Shearer, B.S., C.C.P.
Surgery: Thomas A. D’Amico, M.D., Mani A. Daneshmand, M.D., R. Jeffrey G. Gaca, M.D., Donald D. Glower, M.D., Jack Haney, M.D., R. David Harpole, M.D., Mathew G. Hartwig, M.D., G. Chad Hughes, M.D., Jacob A. Klapper, M.D., Shu S. Lin, M.D., Andrew J. Lodge, M.D., Carmelo A. Milano, M.D., Ryan P. Plichta, M.D, Jacob N. Schroeder, M.D., Peter K. Smith, M.D., Betty C. Tong, M.D.
References
- 1.Newman MF, Kirchner JL, Phillips-Bute B, Gaver V, Grocott H, Jones RH, Mark DB, Reves JG, Blumenthal JA, Neurological Outcome Research G, the Cardiothoracic Anesthesiology Research Endeavors I Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N Engl J Med. 2001;344:395–402. doi: 10.1056/NEJM200102083440601. [DOI] [PubMed] [Google Scholar]
- 2.Phillips-Bute B, Mathew JP, Blumenthal JA, Grocott HP, Laskowitz DT, Jones RH, Mark DB, Newman MF. Association of neurocognitive function and quality of life 1 year after coronary artery bypass graft (CABG) surgery. Psychosom Med. 2006;68:369–75. doi: 10.1097/01.psy.0000221272.77984.e2. [DOI] [PubMed] [Google Scholar]
- 3.Berger M, Burke J, Eckenhoff R, Mathew J. Alzheimer’ s disease, anesthesia, and surgery: a clinically focused review. J Cardiothorac Vasc Anesth. 2014;28:1609–23. doi: 10.1053/j.jvca.2014.04.014. [DOI] [PubMed] [Google Scholar]
- 4.Grundman M, Petersen RC, Ferris SH, Thomas RG, Aisen PS, Bennett DA, Foster NL, Jack CR, Jr, Galasko DR, Doody R, Kaye J, Sano M, Mohs R, Gauthier S, Kim HT, Jin S, Schultz AN, Schafer K, Mulnard R, van Dyck CH, Mintzer J, Zamrini EY, Cahn-Weiner D, Thal LJ. Alzheimer’ s Disease Cooperative S: Mild cognitive impairment can be distinguished from Alzheimer disease and normal aging for clinical trials. Arch Neurol. 2004;61:59–66. doi: 10.1001/archneur.61.1.59. [DOI] [PubMed] [Google Scholar]
- 5.Xie Z, Culley DJ, Dong Y, Zhang G, Zhang B, Moir RD, Frosch MP, Crosby G, Tanzi RE. The common inhalation anesthetic isoflurane induces caspase activation and increases amyloid beta-protein level in vivo. Ann Neurol. 2008;64:618–27. doi: 10.1002/ana.21548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Eckenhoff RG, Johansson JS, Wei H, Carnini A, Kang B, Wei W, Pidikiti R, Keller JM, Eckenhoff MF. Inhaled anesthetic enhancement of amyloid-beta oligomerization and cytotoxicity. Anesthesiology. 2004;101:703–9. doi: 10.1097/00000542-200409000-00019. [DOI] [PubMed] [Google Scholar]
- 7.Tang JX, Baranov D, Hammond M, Shaw LM, Eckenhoff MF, Eckenhoff RG. Human Alzheimer and inflammation biomarkers after anesthesia and surgery. Anesthesiology. 2011;115:727–32. doi: 10.1097/ALN.0b013e31822e9306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Berger M, Nadler JW, Friedman A, McDonagh DL, Bennett ER, Cooter M, Qi W, Laskowitz DT, Ponnusamy V, Newman MF, Shaw LM, Warner DS, Mathew JP, James ML. team M-Pt: The Effect of Propofol Versus Isoflurane Anesthesia on Human Cerebrospinal Fluid Markers of Alzheimer’s Disease: Results of a Randomized Trial. J Alzheimers Dis. 2016;52:1299–310. doi: 10.3233/JAD-151190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carpenter AP, Jr, Pontecorvo MJ, Hefti FF, Skovronsky DM. The use of the exploratory IND in the evaluation and development of 18F-PET radiopharmaceuticals for amyloid imaging in the brain: a review of one company’ s experience. Q J Nucl Med Mol Imaging. 2009;53:387–93. [PubMed] [Google Scholar]
- 10.Choi SR, Schneider JA, Bennett DA, Beach TG, Bedell BJ, Zehntner SP, Krautkramer MJ, Kung HF, Skovronsky DM, Hefti F, Clark CM. Correlation of amyloid PET ligand florbetapir F 18 binding with Abeta aggregation and neuritic plaque deposition in postmortem brain tissue. Alzheimer Dis Assoc Disord. 2012;26:8–16. doi: 10.1097/WAD.0b013e31821300bc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Johnson KA, Sperling RA, Gidicsin CM, Carmasin JS, Maye JE, Coleman RE, Reiman EM, Sabbagh MN, Sadowsky CH, Fleisher AS, Murali Doraiswamy P, Carpenter AP, Clark CM, Joshi AD, Lu M, Grundman M, Mintun MA, Pontecorvo MJ, Skovronsky DM, group AAs Florbetapir (F18-AV-45) PET to assess amyloid burden in Alzheimer’ s disease dementia, mild cognitive impairment, and normal aging. Alzheimers Dement. 2013;9:S72–83. doi: 10.1016/j.jalz.2012.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yeo JM, Waddell B, Khan Z, Pal S. A systematic review and meta-analysis of (18)F-labeled amyloid imaging in Alzheimer’ s disease. Alzheimers Dement (Amst) 2015;1:5–13. doi: 10.1016/j.dadm.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jagust WJ, Landau SM, Koeppe RA, Reiman EM, Chen K, Mathis CA, Price JC, Foster NL, Wang AY. The Alzheimer’ s Disease Neuroimaging Initiative 2 PET Core: 2015. Alzheimers Dement. 2015;11:757–71. doi: 10.1016/j.jalz.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aisen PS, Petersen RC, Donohue MC, Gamst A, Raman R, Thomas RG, Walter S, Trojanowski JQ, Shaw LM, Beckett LA, Jack CR, Jr, Jagust W, Toga AW, Saykin AJ, Morris JC, Green RC, Weiner MW. Clinical core of the Alzheimer’ s Disease Neuroimaging Initiative: progress and plans. Alzheimers Dement. 2010;6:239–46. doi: 10.1016/j.jalz.2010.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Weiner MW, Aisen PS, Jack CR, Jr, Jagust WJ, Trojanowski JQ, Shaw L, Saykin AJ, Morris JC, Cairns N, Beckett LA, Toga A, Green R, Walter S, Soares H, Snyder P, Siemers E, Potter W, Cole PE, Schmidt M. Alzheimer’ s Disease Neuroimaging I: The Alzheimer’ s disease neuroimaging initiative: progress report and future plans. Alzheimers Dement. 2010;6:202–11 e7. doi: 10.1016/j.jalz.2010.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Murphy KR, Landau SM, Choudhury KR, Hostage CA, Shpanskaya KS, Sair HI, Petrella JR, Wong TZ, Doraiswamy PM. Alzheimer’ s Disease Neuroimaging I: Mapping the effects of ApoE4, age and cognitive status on 18F-florbetapir PET measured regional cortical patterns of beta-amyloid density and growth. Neuroimage. 2013;78:474–80. doi: 10.1016/j.neuroimage.2013.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Landau SM, Fero A, Baker SL, Koeppe R, Mintun M, Chen K, Reiman EM, Jagust WJ. Measurement of longitudinal beta-amyloid change with 18F-florbetapir PET and standardized uptake value ratios. J Nucl Med. 2015;56:567–74. doi: 10.2967/jnumed.114.148981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Joshi AD, Pontecorvo MJ, Lu M, Skovronsky DM, Mintun MA, Devous MD., Sr A Semiautomated Method for Quantification of F 18 Florbetapir PET Images. J Nucl Med. 2015;56:1736–41. doi: 10.2967/jnumed.114.153494. [DOI] [PubMed] [Google Scholar]
- 19.Joshi AD, Pontecorvo MJ, Clark CM, Carpenter AP, Jennings DL, Sadowsky CH, Adler LP, Kovnat KD, Seibyl JP, Arora A, Saha K, Burns JD, Lowrey MJ, Mintun MA, Skovronsky DM, Florbetapir FSI. Performance characteristics of amyloid PET with florbetapir F 18 in patients with alzheimer’ s disease and cognitively normal subjects. J Nucl Med. 2012;53:378–84. doi: 10.2967/jnumed.111.090340. [DOI] [PubMed] [Google Scholar]
- 20.Murkin JM, Newman SP, Stump DA, Blumenthal JA. Statement of consensus on assessment of neurobehavioral outcomes after cardiac surgery. Ann Thorac Surg. 1995;59:1289–95. doi: 10.1016/0003-4975(95)00106-u. [DOI] [PubMed] [Google Scholar]
- 21.Rasmusson DX, Bylsma FW, Brandt J. Stability of performance on the Hopkins Verbal Learning Test. Arch Clin Neuropsychol. 1995;10:21–6. [PubMed] [Google Scholar]
- 22.Randt C, Brown E. Adminstration manual: Randt memory test. New York: Life Sciences Associates; 1983. [Google Scholar]
- 23.Wechsler D. The Wechsler Adult Intelligence Scale-Revised (Manual) Psychological Corporation; 1981. [Google Scholar]
- 24.Reitan R. Validity of the trail making test as an indicator of organic brain damage. Percept Mot Skills. 1958;8:271–6. [Google Scholar]
- 25.Tupler LA, Krishnan KR, Greenberg DL, Marcovina SM, Payne ME, MacFall JR, Charles HC, Doraiswamy PM. Predicting memory decline in normal elderly: genetics, MRI, and cognitive reserve. Neurobiol Aging. 2007;28:1644–56. doi: 10.1016/j.neurobiolaging.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 26.Morris GP, Clark IA, Vissel B. Inconsistencies and controversies surrounding the amyloid hypothesis of Alzheimer’ s disease. Acta Neuropathol Commun. 2014;2:135. doi: 10.1186/s40478-014-0135-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shankar GM, Li S, Mehta TH, Garcia-Munoz A, Shepardson NE, Smith I, Brett FM, Farrell MA, Rowan MJ, Lemere CA, Regan CM, Walsh DM, Sabatini BL, Selkoe DJ. Amyloid-beta protein dimers isolated directly from Alzheimer’ s brains impair synaptic plasticity and memory. Nat Med. 2008;14:837–42. doi: 10.1038/nm1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Merino JG, Latour LL, Tso A, Lee KY, Kang DW, Davis LA, Lazar RM, Horvath KA, Corso PJ, Warach S. Blood-brain barrier disruption after cardiac surgery. AJNR Am J Neuroradiol. 2013;34:518–23. doi: 10.3174/ajnr.A3251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pluta R, Amek MU. Brain ischemia and ischemic blood-brain barrier as etiological factors in sporadic Alzheimer’ s disease. Neuropsychiatr Dis Treat. 2008;4:855–64. doi: 10.2147/ndt.s3739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Agostinho P, Cunha RA, Oliveira C. Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer’ s disease. Curr Pharm Des. 2010;16:2766–78. doi: 10.2174/138161210793176572. [DOI] [PubMed] [Google Scholar]
- 31.Gorelick PB, Scuteri A, Black SE, Decarli C, Greenberg SM, Iadecola C, Launer LJ, Laurent S, Lopez OL, Nyenhuis D, Petersen RC, Schneider JA, Tzourio C, Arnett DK, Bennett DA, Chui HC, Higashida RT, Lindquist R, Nilsson PM, Roman GC, Sellke FW, Seshadri S, American Heart Association Stroke Council CoE, Prevention CoCNCoCR, Intervention Council on Cardiovascular S, Anesthesia Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the american heart association/american stroke association. Stroke. 2011;42:2672–713. doi: 10.1161/STR.0b013e3182299496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Santos CY, Snyder PJ, Wu WC, Zhang M, Echeverria A, Alber J. Pathophysiologic relationship between Alzheimer’ s disease, cerebrovascular disease, and cardiovascular risk: A review and synthesis. Alzheimers Dement (Amst) 2017;7:69–87. doi: 10.1016/j.dadm.2017.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Irina A, Seppo H, Arto M, Paavo R, Sr, Hilkka S. beta-amyloid load is not influenced by the severity of cardiovascular disease in aged and demented patients. Stroke. 1999;30:613–8. doi: 10.1161/01.str.30.3.613. [DOI] [PubMed] [Google Scholar]
- 34.Wong DF, Rosenberg PB, Zhou Y, Kumar A, Raymont V, Ravert HT, Dannals RF, Nandi A, Brasic JR, Ye W, Hilton J, Lyketsos C, Kung HF, Joshi AD, Skovronsky DM, Pontecorvo MJ. In vivo imaging of amyloid deposition in Alzheimer disease using the radioligand 18F-AV-45 (florbetapir [corrected] F 18) J Nucl Med. 2010;51:913–20. doi: 10.2967/jnumed.109.069088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hornberger M, Piguet O. Episodic memory in frontotemporal dementia: a critical review. Brain. 2012;135:678–92. doi: 10.1093/brain/aws011. [DOI] [PubMed] [Google Scholar]
- 36.Zhang MD, Barde S, Yang T, Lei B, Eriksson LI, Mathew JP, Andreska T, Akassoglou K, Harkany T, Hokfelt TG, Terrando N. Orthopedic surgery modulates neuropeptides and BDNF expression at the spinal and hippocampal levels. Proc Natl Acad Sci U S A. 2016;113:E6686–E6695. doi: 10.1073/pnas.1614017113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Terry RD, Masliah E, Salmon DP, Butters N, DeTeresa R, Hill R, Hansen LA, Katzman R. Physical basis of cognitive alterations in Alzheimer’ s disease: synapse loss is the major correlate of cognitive impairment. Ann Neurol. 1991;30:572–80. doi: 10.1002/ana.410300410. [DOI] [PubMed] [Google Scholar]
- 38.Badgaiyan RD, Weise S, Wack DS, Vidal Melo MF. Attenuation of regional cerebral blood flow during memory processing after coronary artery bypass surgery. Anesth Analg. 2014;119:550–3. doi: 10.1213/ANE.0000000000000334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Palmqvist S, Mattsson N, Hansson O. Alzheimer’ s Disease Neuroimaging I: Cerebrospinal fluid analysis detects cerebral amyloid-beta accumulation earlier than positron emission tomography. Brain. 2016;139:1226–36. doi: 10.1093/brain/aww015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Evered L, Silbert B, Scott DA, Ames D, Maruff P, Blennow K. Cerebrospinal Fluid Biomarker for Alzheimer Disease Predicts Postoperative Cognitive Dysfunction. Anesthesiology. 2016;124:353–61. doi: 10.1097/ALN.0000000000000953. [DOI] [PubMed] [Google Scholar]
- 41.Wan Y, Xu J, Meng F, Bao Y, Ge Y, Lobo N, Vizcaychipi MP, Zhang D, Gentleman SM, Maze M, Ma D. Cognitive decline following major surgery is associated with gliosis, beta-amyloid accumulation, and tau phosphorylation in old mice. Crit Care Med. 2010;38:2190–8. doi: 10.1097/CCM.0b013e3181f17bcb. [DOI] [PubMed] [Google Scholar]
- 42.Lee TA, Wolozin B, Weiss KB, Bednar MM. Assessment of the emergence of Alzheimer’ s disease following coronary artery bypass graft surgery or percutaneous transluminal coronary angioplasty. J Alzheimers Dis. 2005;7:319–24. doi: 10.3233/jad-2005-7408. [DOI] [PubMed] [Google Scholar]
- 43.Salminen A, Kauppinen A, Kaarniranta K. Hypoxia/ischemia activate processing of Amyloid Precursor Protein: impact of vascular dysfunction in the pathogenesis of Alzheimer’ s disease. J Neurochem. 2017;140:536–549. doi: 10.1111/jnc.13932. [DOI] [PubMed] [Google Scholar]
- 44.Berger M, Nadler JW, Browndyke J, Terrando N, Ponnusamy V, Cohen HJ, Whitson HE, Mathew JP. Postoperative Cognitive Dysfunction: Minding the Gaps in Our Knowledge of a Common Postoperative Complication in the Elderly. Anesthesiol Clin. 2015;33:517–50. doi: 10.1016/j.anclin.2015.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Condello C, Schain A, Grutzendler J. Multicolor time-stamp reveals the dynamics and toxicity of amyloid deposition. Sci Rep. 2011;1:19. doi: 10.1038/srep00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Klunk WE, Mathis CA, Price JC, Lopresti BJ, DeKosky ST. Two-year follow-up of amyloid deposition in patients with Alzheimer’ s disease. Brain. 2006;129:2805–7. doi: 10.1093/brain/awl281. [DOI] [PubMed] [Google Scholar]
- 47.Moller JT, Cluitmans P, Rasmussen LS, Houx P, Rasmussen H, Canet J, Rabbitt P, Jolles J, Larsen K, Hanning CD, Langeron O, Johnson T, Lauven PM, Kristensen PA, Biedler A, van Beem H, Fraidakis O, Silverstein JH, Beneken JE, Gravenstein JS. Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet. 1998;351:857–61. doi: 10.1016/s0140-6736(97)07382-0. [DOI] [PubMed] [Google Scholar]
- 48.Ruitenberg A, Ott A, van Swieten JC, Hofman A, Breteler MM. Incidence of dementia: does gender make a difference? Neurobiol Aging. 2001;22:575–80. doi: 10.1016/s0197-4580(01)00231-7. [DOI] [PubMed] [Google Scholar]
- 49.Lin KA, Choudhury KR, Rathakrishnan BG, Marks DM, Petrella JR, Doraiswamy PM. Alzheimer’ s Disease Neuroimaging I: Marked gender differences in progression of mild cognitive impairment over 8 years. Alzheimers Dement (N Y) 2015;1:103–110. doi: 10.1016/j.trci.2015.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


