Abstract
In an effort to enhance welfare, behavioural management continually refines methods of non-human primate (NHP) care. Chimpanzees (Pan troglodytes) are one of the most cognitively complex captive NHPs and they have been observed to self-medicate in the wild. The population of captive chimpanzees in the US is aged (due to a breeding moratorium instituted in 1998) and will progressively require more medical care as they get older. To functionally simulate natural self-medication behaviour, provide chimpanzees with the opportunity to voluntarily participate in their own healthcare, and open new avenues of communication between caregivers and chimpanzees, we used a medication choice paradigm that allowed chimpanzees to choose their daily arthritis medication. We provided four arthritic, mobility-impaired chimpanzees with meloxicam or ibuprofen in blue or green Gatorade® to establish associations between the coloured drinks and the effects of the medications. We subsequently gave each chimpanzee a choice between the two medications. Behaviour was recorded using 15-min focal animal observations. Mobility was assessed using interactive mobility tests and a caregiver-rating system. One chimpanzee showed a medication preference (ibuprofen over meloxicam). The chimpanzees exhibited no significant behavioural or mobility differences over time, suggesting that ibuprofen and meloxicam may not differ significantly in their ability to alleviate arthritic symptoms. Whether or not the chimpanzees show a medication preference, the opportunity to make meaningful choices and the functional simulation of a complex behaviour, self-medication, is present when using this medication choice technique. Furthermore, the paradigm itself could have potential applications for additional medication options and treatment regimens.
Keywords: animal welfare, behavioural management, captivity, chimpanzees, choice, voluntary participation
Introduction
Behavioural management strategies aim to refine and improve captive non-human primate (NHP) care and enhance welfare (National Institutes of Health 2013; Schapiro 2017). As behavioural management evolves closer toward the ultimate goal of optimal care, there is an increased focus on providing animals with opportunities to make meaningful choices in captive settings. Several decades of research have outlined the welfare benefits of providing captive animals with choice in environments and/or situations, including improved physiological responses, such as reduced reactions to pain and stress (Hanson et al 1976; Mineka et al 1986; Friend 1991; Lambeth et al 2006; Leotti et al 2010; Behringer et al 2014), and improved psychological responses, such as decreased aggression, fewer abnormal behaviours, and increased social contact and play (Hanson et al 1976; Perlmuter & Monty 1977; Sambrook & Buchanan-Smith 1997; Lambeth et al 2001; Owen et al 2005; Ross 2006).
Conversely, a lack of control over the environment has been linked to negative outcomes, such as helplessness and the lack of adaptive coping strategies (Tennessen 1989; Morgan & Tromborg 2007). Current research, theory, and practice are aimed at providing captive NHPs with an increased number of choices (Baker et al 2017; Schapiro et al 2017). This is reflected in the recent incorporation of the provision of opportunities for environmental choice into welfare regulations provided by the National Institutes of Health (NIH 2013), and welfare aims and objectives put forth by international animal welfare groups (Mellor 2016).
The importance of choice is also reflected in the increased implementation of positive reinforcement training (PRT) techniques to train non-human primates to voluntarily participate in health- and management-related procedures (Reinhardt 1997; Laule & Whittaker 2002, 2007; Baker et al 2007; Magden et al 2013; Reamer et al 2014; Baker 2016; Graham 2017; Magden 2017). Allowing NHPs to choose whether or not to participate in procedures has beneficial effects on welfare. For example, Lambeth et al (2006) found that chimpanzees that voluntarily presented a body part for an injection of anaesthetic had lower white blood cell counts (WBC), segmented neutrophils, and glucose levels compared to chimpanzees that were injected nonvoluntarily. Furthermore, within subjects, chimpanzees showed lower WBC and GLU values when they voluntarily presented for the anaesthetic injection compared to when they were non-voluntarily injected. This suggests that this type of choice, as expressed by the voluntary participation of chimpanzees in selected healthcare procedures, is associated with diminished stress and increased overall wellbeing. Voluntary participation is now a relatively common practice in many health-related procedures, such as venipuncture (Reinhardt 2003; Videan et al 2005; Lambeth et al 2006; Coleman et al 2008), acupuncture and laser therapy (Magden et al 2013, 2016; Magden 2017), blood glucose and urine sampling (Laule et al 1996; Reamer et al 2014; Graham 2017), and body part presentation (Pomerantz & Terkel 2009), as well as husbandry-related procedures (Bloomsmith et al 1994, 1998; Desmond &Laule 1994; Schapiro et al 2001; McKinley et al 2003).
Chimpanzees, among the most cognitively complex NHP species, are ideal for assessments of the efficacy of sophisticated refinements in behavioural management techniques. Due to the breeding moratorium for captive chimpanzees put in place by the NIH in 1998, the population of research chimpanzees in the United States has a skewed age distribution (Hopkins & Latzman 2017). This ageing population of chimpanzees is increasingly experiencing maladies that ageing humans also face, including arthritis, high blood pressure, diabetes, and heart conditions (Nunamaker et al 2012;Magden et al 2013). As the population continues to get older, the animals will require ever greater amounts of medical care and specific ageing-related treatment regimens (Association of Zoos and Aquariums [AZA] 2010; NIH 2013; Hopkins & Latzman 2017; Magden 2017). Therefore, an important refinement in captive chimpanzee care is to promote the voluntary participation of the animals in their own healthcare.
Part of the evolution of behavioural management is innovation in functional simulations of natural conditions and behaviours. One natural behaviour with the potential for functional simulation that has yet to be investigated in behavioural management programmes is self-medication. Wild chimpanzees self-medicate by chewing and/or ingesting plants with low nutritional, but high pharmacological (anti-tumour, anti-bacterial, anti-diarrheal, and antiparasitic) effects (Huffman 1997; Fowler et al 2007; Huffman et al 2010; Masi et al 2012; Obbo et al 2013). The self-medication process requires complex cognitive processing, including the chimpanzee recognising that it is not feeling well, understanding that ingestion of a specific external substance can alleviate symptoms, and remembering the therapeutic effects of the substance the next time the illness is experienced. Behavioural management practices can functionally simulate aspects of this cognitive and behavioural chain by providing captive chimpanzees with opportunities to choose medications for health-related conditions, such as arthritis. In addition to functional simulations and increased choice, voluntary participation via self-medication opens new channels for human-chimpanzee communication by giving chimpanzees the opportunity to communicate their preference, rather than caregivers making assumptions about preferences.
Meloxicam and ibuprofen are two non-steroidal anti-inflammatory drugs (NSAIDs) commonly used for pain relief for captive chimpanzees (Popilskis et al 2008). The two medications have similar effects and mechanisms of action, but do differ in certain respects, including: i) the number of times the medications must be administered each day (ie ibuprofen must be administered 3× per day and meloxicam 1× per day); ii) the presence of possible side-effects (ibuprofen can cause gastrointestinal upset with long-term use [Boelsterli et al 2013]) that the chimpanzee may experience, but typically cannot communicate to caregivers; and iii) the cost of the medications. Given these differences and keeping in mind the ageing population of captive chimpanzees that is increasingly facing health-related situations, including arthritis, it is valuable to determine whether individual chimpanzees prefer one of these common types of medications over the other.
In a previous initiative to functionally simulate natural behaviours and increase choice within the captive environment, we conducted a pilot study in 2014 in which four arthritic chimpanzees were given crushed ibuprofen or crushed meloxicam pills in blue or green Gatorade® during alternating two-month phases using an ABBA design (Schapiro et al 2014). They were then given a choice between the two medications/coloured drinks for two months. All four chimpanzees significantly preferred meloxicam to ibuprofen. Furthermore, when chimpanzees were given a choice between medications, they tended to exhibit higher levels of species-typical behaviour than they did during previous phases of the study when they had no choice. However, it should be noted that the crushed ibuprofen pills were ‘granulated’ and could be seen and tasted in the Gatorade® mixture, whereas this was not the case with the meloxicam. Therefore, it is possible that the chimpanzees’ observed preference for meloxicam was actually an aversion to the granulated ibuprofen, rather than a preference for the effects of meloxicam.
The present study was part of the National Center for Chimpanzee Care’s (NCCC’s) systematic effort to assess the effects of management strategies and environment on the welfare of captive chimpanzees (Neal Webb et al 2018, in prep). Specifically, the current study served as an initial framework for the establishment of a paradigm or approach that aims to provide arthritic, mobility-impaired chimpanzees with opportunities to ameliorate their symptoms through a functional simulation of self-medication behaviour, and to communicate to us (through their choices), which of the medications they preferred. Four chimpanzees voluntarily participated in the present study, which included additional methodological controls compared to the initial pilot medication choice study (Schapiro et al 2014): i) additional control procedures were utilised, specifically liquid forms of the medications were compounded in raspberry flavouring, rather than the previous technique of crushing pills into Gatorade®; ii) the observer was blind to the experimental condition in the current study; and iii) a number of dependent measures were added to the protocol, including techniques for empirically assessing mobility. We hypothesised that chimpanzees would exhibit a significant preference for one medication over the other, and that they would exhibit increased mobility and locomotion and, thus, decreased inactivity, while consuming their preferred medication. Similarly, because choice in captive environments has been shown to enhance welfare in captive NHPs, we hypothesized that behavioural indices would be indicative of enhanced well-being during the choice phase, specifically in terms of increased levels of species-typical behaviour, social play, affiliative behaviour, and time spent in proximity to social partners.
Materials and methods
Subjects and study site
Four captive chimpanzees were included in the study based on: i) veterinarian-diagnosed arthritis; ii) mobility impairments, including difficulties associated with climbing, brachiating, limb placement and function, speed of movement, and stiffness; and iii) prescriptions for the daily administration of analgesics. The subjects were three females and one male, ranging from 44 to 51 years of age (mean age = 49 years). Two of the subjects lived in the same social group (PE and PR). All animals were housed at the NCCC of the Michale E Keeling Center for Comparative Medicine and Research of The University of Texas MD Anderson Cancer Center in Bastrop, Texas. The Keeling Center has been continuously accredited by AAALAC-I since 1979. These chimpanzees were born in the wild but had lived at the NCCC for at least 30 years. Five non-mobility-impaired chimpanzees served as control subjects for mobility measurements. These subjects were two males and three females, ranging from 20 to 26 years of age (mean = 23.6 years), and all were born in captivity. All chimpanzees lived in compatible social groups ranging in size from three to nine individuals. Groups were housed in indoor-outdoor corrals or Primadomes™ (Figures 1, 2 and 3), with access to all areas at all times (except during cleaning). One chimpanzee (MA) in the current study had also participated in the pilot medication choice study (Schapiro et al 2014).
Figure 1.
Corral housing. Several ramps serve as easy access climbing paths for mobility-impaired chimpanzees.
Figure 2.
Dome housing (see also Figure 3). Several ramps serve as easy access climbing paths for mobility-impaired chimpanzees.
Figure 3.
Materials
We used clear G2 Mixed Berry Gatorade® combined with a small quantity (22 drops) of green or blue food colouring to create green- and blue-coloured Gatorade®. Colour preference tests performed for the pilot study showed that chimpanzees do not exhibit a preference for either green over blue or vice versa. The colour of Gatorade® was counterbalanced across types of medications and order of medication was counterbalanced across groups one and two (see Procedure below). A compounding pharmacy created liquid, colourless, raspberry-flavoured versions of meloxicam and ibuprofen. Depending on the phase of the study and counterbalanced order of medications for each chimpanzee (Table 1), the animals either received 400 mg (a 2-ml dose) of ibuprofen three times daily (at 0800, 1400 and 2000h), or 7.5 mg (a 1-ml dose) of meloxicam once daily (at 0800h), plus two placebo doses at 1400 and 2000h. The placebo doses consisted of 1 ml of the concentrated colourless raspberry flavouring that was used to compound the meloxicam and ibuprofen in the appropriately coloured Gatorade®. Medications were mixed with 177.44 ml of the appropriate colour of Gatorade® in transparent ‘squirt bottles’ for each chimpanzee each day during all phases of the study. Thus, all chimpanzees received three Gatorade® drinks, with either ibuprofen (in all three) or meloxicam (in only one), each day.
Table 1.
Study schedule per group (Medication and Gatorade® colour).
| Chimpanzee | Baseline Phase 1 | A Phase 2 | B B Phases 3 and 4 | A Phase 5 | Choice Phase 6 | |
|---|---|---|---|---|---|---|
| Group One | CO | Meloxicam | Ibuprofen BLUE | Meloxicam GREEN | Ibuprofen BLUE | Ibuprofen BLUE OR Meloxicam GREEN |
| PR | ||||||
| Group Two | PE | Meloxicam | Meloxicam BLUE | Ibuprofen GREEN | Meloxicam BLUE | Ibuprofen GREEN OR Meloxicam BLUE |
| MA |
Procedure
Chimpanzees were randomly assigned to either Group one or Group two, which determined both the order of medications each chimpanzee received and the colour of Gatorade® with which each medication was mixed (Table 1). There were six phases of the study, including a baseline at the beginning of the study, an ABBA design corresponding to phases two, three, four, and five, and a choice phase at the end of the study (Baseline, A, B, B, A, Choice). Each phase lasted approximately four weeks, for a total of approximately six months for the entire study (September 2016–April 2017). The length of these phases was sufficient to control for any lasting effects of the drugs administered during previous phases, as both ibuprofen and meloxicam show plasma concentrations near zero 72 h following administration (Albert & Gernaat 1984; Bae et al 2007). As mentioned above, phase one served as the baseline for behavioural observations. As part of their routine care prior to the beginning of this study, all arthritic chimpanzees were receiving crushed meloxicam pills mixed with orange juice in a paper cup from a veterinary technician for treatment of their symptoms. This medication delivery method continued throughout the Baseline phase, as we did not want to withhold analgesic treatment from these arthritic chimpanzees for the purposes of this study.
During each of phases two through five, chimpanzees received one of the two medications as described in Table 1. During phase six (the Choice phase), an experimenter presented individual chimpanzees each morning with both medications in the appropriate colour of Gatorade® (one in each hand, counterbalanced across left and right). Chimpanzees could then choose the one they wanted by initiating movement toward one medication. If the chimpanzee chose meloxicam in the morning, he or she received two placebo doses later in the day as described above in Materials. If the chimpanzee chose ibuprofen in the morning, he or she received two more doses of ibuprofen as described above. During the Choice phase, the medication initially chosen by each chimpanzee was recorded daily (to determine preference), in addition to the behavioural data described below.
All behavioural observations were collected by one observer on a laptop computer using The Observer XT (Version 10.0, Noldus, Leesburg, VA, USA). Focal animal sampling techniques (Altmann 1974) were used for behavioural observations throughout each phase of the study. The observer, blind to experimental condition, recorded behaviour for 15 min while focal chimpanzees were living in their social group. Categories of behaviour recorded included locomotive, aggressive, submissive, sexual, self-directed, affiliative, abnormal, object manipulation, and other. Proximity of the focal animal to other animals within the group was also recorded (see Appendix 1 in the supplementary material to papers published in Animal Welfare: https://www.ufaw.org.uk/the-ufawjournal/supplementary-material). Behavioural observations were conducted for each chimpanzee once per day (Monday–Friday) throughout each phase of the experiment, yielding a total of approximately 120 15-min focal observations (30 h of focal data) per chimpanzee.
Mobility scores
We used a mobility scoring system to rate ease of movement (Table 2) twice per week, once in the morning, after the chimpanzees had received their first dose of either ibuprofen or meloxicam at 0800h, and once in the afternoon, after chimpanzees had received their dose of ibuprofen or placebo at 1400h. Six categories of mobility (rising from resting position, climbs/brachiates, bears weight on all limbs, limb placement and function, speed of movement, discomfort and stiffness) were scored independently and reliably by veterinary technicians, caregivers, and the observer (intraclass correlation = 0.93). Mobility scores ranged from 1 (lowest level of impairment) to 5 (highest level of impairment) for each category. Thus, higher scores were indicative of more highly impaired mobility.
Table 2.
Mobility scoring system.
| Behaviour | Rating 1–5 (descriptions of selected ratings) |
|---|---|
| Rising from resting position | 1 (no difficulty)–5 (cannot sit up) |
| Climbs/brachiates | 1 (no difficulty)–5 (cannot climb/brachiate) |
| Bears weight on all limbs | 1 (no impairment)–5 (impairment of all limbs) |
| Limb placement and function | 1 (no impairment)–5 (no control) |
| Speed of movement | 1 (moves with ease)–5 (cannot move) |
| Discomfort and stiffness | 1 (none observed)–5 (continuous severe discomfort) |
Mobility test
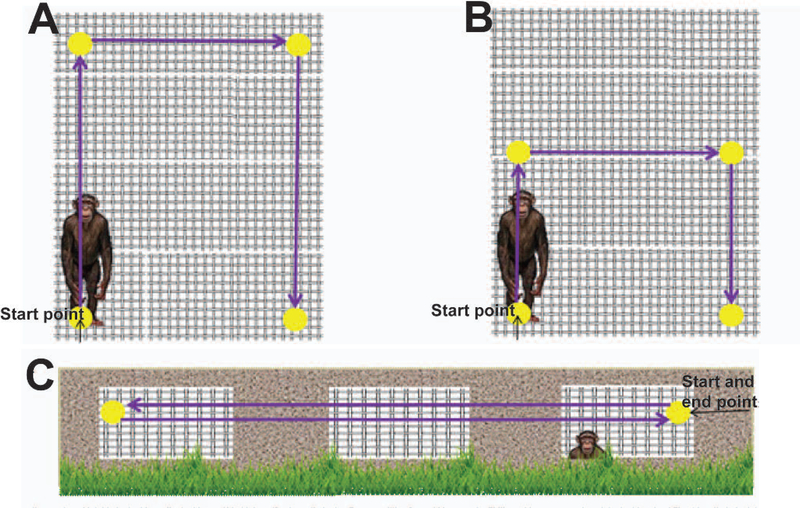
As an additional, empirical test of mobility, chimpanzees also participated in a sequence of structured climbing and walking movements within the enclosure. Chimpanzees were trained to ‘target’, a series of behaviours that included moving to, and touching, a target designated by the experimenter. For two of the subjects, the experimenter sequentially positioned a series of four targets across the indoor portion of the enclosure (Figure 4). Each of these movements was approximately 2 m in length, covering a total distance of 6 m of combined horizontal and vertical movement. Due to mobility constraints from a pre-existing medical condition, one of the other two chimpanzees completed a half-height version of the course, which covered a total distance of 4 m of combined horizontal and vertical movement. Additionally, due to the low dominance rank of the fourth chimpanzee, he was tested on an outdoor course, in which he moved back and forth between two targets, comprising an approximate total of 7 m of horizontal-only locomotion. Chimpanzees participated in this test once per week during Ibuprofen and Meloxicam phases (eight times total during Ibuprofen phases, eight times total during Meloxicam phases) and one–two times per week during the Choice phase (six to eight times total). Five nonmobility-impaired chimpanzees served as control subjects and were also tested on each version of the mobility test at least once per month throughout the course of the study.
Figure 4.
Mobility test schematic (not to scale). Yellow circles = targets, arrows = direction of movement. A) Two chimpanzees moved to and touched four targets one at a time as directed by the experimenter. Each individual arrow represents approximately 2 m of distance. B) One chimpanzee moved to and touched four targets one at a time as directed by the experimenter. Vertical arrows represent approximately 1 m of distance and the horizontal arrow represents approximately 2 m of distance. C) One chimpanzee moved to and touched two targets as directed by the experimenter. Each arrow represents approximately 3 m of distance.
Dependent measures for this mobility test included latency to begin the course (defined as the number of seconds between the time at which the experimenter presented the target to the chimpanzee and when the chimpanzee physically touched the first target of the mobility test), and speed (m s−1) to complete the course, calculated by dividing the total distance by the total duration to complete the course.
Analysis
To examine chimpanzee medication preference, we counted the total number of times each chimpanzee chose ibuprofen or meloxicam throughout the Choice phase. Medication preferences for each chimpanzee were analysed using exact binomial tests (Heilbronner et al 2008; McDonald 2014).
In order to create an ‘ibuprofen phase’ and a ‘meloxicam phase’ for analyses, we averaged total durations of each behaviour across phases 2 and 5, and across phases 3 and 4 (see Table 1), as well as within the Baseline and Choice phase for each subject. Durations were then converted into percentages (Percent time = [Duration (s)/900 s] × 100), representing the average percentage of time spent engaged in each behaviour. Due to small sample size and non-normal distributions of (positively skewed) behavioural data, we used non-parametric Friedman’s Rank tests for within-subjects comparisons across phases, with Wilcoxon post hoc tests for significant differences. Mobility test and mobility score data were normally distributed. Therefore, repeated measures ANOVAs were used for within-subjects comparisons for mobility test speeds and mobility scores across phases. Bootstrapped independent samples t-tests were used for between-subjects (control vs study subjects) mobility test and mobility score comparisons.
Lastly, given the relatively novel use of an empirical assessment of mobility (ie the mobility test) and subjective nature of the mobility scoring system, we aimed to determine if mobility test speeds were correlated with scores of the mobility scoring system, which are easier to obtain. We examined the convergent validity (ie the degree to which the two similar constructs are related) among both mobility measures and the observed time spent locomoting across all phases of the study using Pearson’s correlation coefficients. Mobility scores and mobility test speeds were matched by subject and date as closely as possible. For example, the mobility test for chimpanzee CO on February 7th, 2017 was matched with his mobility score from February 8th, 2017.
All analyses were performed in SPSS Statistics 22 (IBM Corporation, Chicago, IL, USA) to assess differences in behaviour and mobility across phases of the study.
Results
Choice of medication
Figure 5 shows the number of times chimpanzees chose ibuprofen and meloxicam during the 29–36 days of the Choice phase of the study. Arthritic chimpanzee MA was given a choice for 29 out of 36 possible days for veterinary reasons. Similarly, chimpanzees CO and PR were given a choice on 35 out of 36 days due to scheduled annual physical examinations during the Choice phase. An exact binomial test showed that, as a group, arthritic chimpanzees did not exhibit a significant preference for ibuprofen or meloxicam. The observed proportions of ibuprofen choices (0.56) and meloxicam choices (0.44) did not differ significantly from the expected proportion (0.50; P = 0.23). In examining potential preferences of individual chimpanzees, only one subject showed a preference for one medication over the other; chimpanzee PR chose ibuprofen more than twice as often as meloxicam (69% of the time during the Choice phase). An exact binomial test confirmed that the observed proportion of PR’s choices for ibuprofen (0.69) was significantly higher than the expected proportion (0.50; P = 0.02).
Figure 5.
Chimpanzee medication choices. Total number of times each chimpanzee chose meloxicam or ibuprofen over the course of the approximately one-month Choice phase.
Behavioural measures
Friedman’s Rank tests for two behaviours identified significant omnibus differences across the Baseline, Ibuprofen, Meloxicam, and Choice phases. Average percentage of time spent rough-scratching was significantly different across phases, χ23 = 9.0; P = 0.026 (Figure 6). Wilcoxon post hoc tests showed trending differences in rough-scratching between the Choice phase (median = 0.19%), Baseline phase (median = 0.33%), Ibuprofen phase (median = 0.40%), and Meloxicam phase (median = 0.52%; z = −1.83; P = 0.068). Percentage of time spent locomoting differed significantly across phases, χ23 = 9.90; P = 0.019 (Figure 7). Wilcoxon post hoc tests showed trending differences between the Baseline phase (median = 4.07%), Ibuprofen phase (median = 6.86%), Meloxicam phase (median = 6.04%), and Choice phase (median = 7.01%; z = −1.83; P = 0.068). Analyses of the rest of the behavioural measures, including social proximity, revealed no significant differences across phases.
Figure 6.
Percentage of time spent rough-scratching. Median percentage of time chimpanzees spent rough-scratching within each study phase.
Figure 7.
Percentage of time spent locomoting. Median percentage of time chimpanzees spent locomoting within each study phase
Mobility measures
Mobility scores
We recorded chimpanzees’ mobility ratings twice per week and created an average within each phase. Recall that higher scores are indicative of greater mobility impairment. Scores could theoretically range from 6 (least mobility impaired) to 30 (extremely mobility impaired) but observed scores for medication choice subjects only ranged from 6–17, while mobility scores were consistently 6–7 for control (nonmobility-impaired) subjects. A bootstrapped independent samples t-test confirmed that there was a significant difference between control chimpanzees’ mean (± SEM) mobility scores (6.02 [± 0.02]) and arthritic chimpanzees’ mobility scores (10.19 [± 1.02]; P = 0.007). Figure 8 shows that each chimpanzee’s mobility scores were consistent across phases of the study, and a repeated measures ANOVA confirmed that there were no significant differences in each chimpanzee’s (mean [± SEM]) mobility scores across phases (Baseline phase = 9.59 [± 1.46]; Ibuprofen phase = 9.82 [± 1.60]; Meloxicam phase = 9.77 [± 1.47]; or Choice phase = 9.52 [± 1.59]; P = 0.73)
Figure 8.
Mobility scores across phase. Observer ratings of each chimpanzee’s overall mobility on a scale of 6–30. Higher scores indicate higher impairment in mobility (6 = least impaired). Intraclass correlation = 0.931. Error bars represent standard error of the mean.
Mobility test
We were unable to obtain a sufficient quantity of mobility test data during the Baseline phase due to issues in training. Therefore, mobility test data for only the Ibuprofen, Meloxicam, and Choice phases are presented. Mobility test completion times were converted into speed (m s−1) and averaged within each phase for each chimpanzee chimpanzee. First, we examined whether arthritic chimpanzees’ mobility test speeds (m s−1) were indeed slower than control chimpanzees’ (ie non-mobility-impaired). An independent samples t-test confirmed that the mean speeds (± SEM) of arthritic subjects’ mobility tests were significantly slower (0.34 [± 0.06] m s−1) than control subjects (0.55 [± 0.03] m s−1); P = 0.03 (Figure 9). We then examined differences in arthritic chimpanzees’ mobility test speeds across phases; a repeated measures ANOVA revealed no significant differences in mean speeds (± SEM) among the Ibuprofen (0.34 [± 0.05] m s−1), Meloxicam (0.33 [± 0.07] m s−1), or Choice phases (0.34 [± 0.08] m s−1; P = 0.92; Figure 9).
Figure 9.
Mobility test speed. Chimpanzee speed (m s−1) to complete the mobility test (see Figure 4 for Mobility test schematic) across study phase. Error bars represent standard error of the mean.
Average latency to begin the mobility test was also calculated for each phase. We first compared arthritic chimpanzees’ mean (± SEM) latencies (0.88 [± 0.11 s]) with those of control chimpanzees (0.80 [± 0.29 s]) using an independent-samples t-test and found no significant difference; P = 0.78. We then compared arthritic chimpanzees’ latencies across the study phases; a repeated measures ANOVA revealed no significant (mean [± SEM]) differences (Ibuprofen phase = 0.95 [± 0.14]; Meloxicam phase = 1.03 [± 0.19]; Choice phase = 0.65 [± 0.22]; P = 0.51; Figure 10).
Figure 10.
Mobility test latency. Chimpanzee latency (s) to begin the mobility test (ie time to touch first target of mobility test) across study phase. Error bars represent standard error of the mean.
Convergent validity
Mobility test speeds were significantly negatively correlated with mobility scores (r = −0.64; P < 0.001); higher mobility scores (indicative of higher mobility impairment) were related to slower speeds to complete the mobility test, indicating high convergent validity between the easy to use mobility scoring system and the mobility test. To assess convergent validity with locomotion, average mobility test speeds, average mobility scores, and average percentage of time spent in locomotion were matched on phase of study for each subject. The speed (m s−1) at which the subjects performed the mobility test was significantly positively correlated with percentage of time spent locomoting (r = 0.83; P < 0.01); faster speeds to complete the mobility test were related to more time spent locomoting, indicating high convergent validity between locomotion and mobility test speeds. Mobility scores were not significantly correlated with time spent locomoting (P = 0.21).
Discussion
Chimpanzees are one of the most cognitively complex NHPs and are known to self-medicate in the wild (Huffman 1997). Important refinements to captive chimpanzee care involve providing chimpanzees with innovative functional simulations of natural conditions and opportunities to exercise their cognitive capacities in captivity. With this in mind, we aimed to refine captive chimpanzee care using a medication choice procedure. This particular study served as an initial endeavour to establish a paradigm that could be used in future studies offering various types of healthcare-related choices (see below). Contrary to our hypotheses, three of the four chimpanzees showed no significant preferences for one medication over the other, which may be due to the similarity in mechanism of action between the two medications. It is noteworthy that PR, the chimpanzee that exhibited the highest level of mobility impairment, showed a preference for ibuprofen over meloxicam. However, with the exception of a decrease in rough-scratching, her behaviour and mobility indices remained relatively constant throughout the study. This makes it difficult to interpret the practical significance of her choice on welfare and behaviour. Furthermore, with the exception of roughscratching, chimpanzee behaviour did not differ significantly across phases, which may be due to the small sample size. The finding that rough-scratching, an indicator of tension and anxiety in chimpanzees (Baker & Aureli 1997), was lowest during the Choice phase compared to all others (each of which did not involve a choice) may be described as a positive change. One might interpret this difference in rough-scratching as lowered tension or anxiety resulting from increased choice. However, this result may have limited practical significance, given that rough-scratching made up less than 1% of all activity throughout the study.
Due to the overall lack of significant medication preferences and behavioural changes, we cannot explicitly state that the welfare of these chimpanzees was enhanced as a result of this particular medication choice study. Nonetheless, we believe the paradigm could be a useful approach in future studies as applied to an array of medication options, treatment regimens, and health conditions. Using this technique, chimpanzees could be given a choice between more dissimilar types of medications (eg an NSAID vs a narcotic, such as tramadol) for painful conditions, including arthritis. The choice procedure could also be used to help titrate doses for individuals and determine preferences for medications for other chronic health issues experienced by captive chimpanzees, including hypertension and diabetes. Lastly, this method could be used to allow chimpanzees to choose between different types of treatments or treatment regimens. For example, chimpanzees could choose between acupuncture (Magden et al 2013) or laser therapy for arthritis (Magden et al 2016), or between sugar paste and laser therapy for wound treatment. In addition to examining medication and treatment efficacy (assuming that the animals prefer the more efficacious medication or treatment) in this manner, the implementation of multiple studies that employ the choice procedure would provide insight into the value of voluntary participation and increased opportunities to make meaningful choices on overall well-being. Lastly, it would be valuable to replicate and/or refine this choice paradigm with a larger number of chimpanzees, and to expand this technique to additional species of NHPs that are known to self-medicate in the wild (baboons [Papio anubis]; de Roode et al 2013; and bonobos [Pan paniscus]; Fruth et al 2014; for a review, see Huffman 1997).
Given these applications to future studies (see above), the paradigm could have the potential to offer several welfare benefits to captive chimpanzees, including the abilities to: i) make meaningful choices within the captive environment; ii) perform a cognitively complex functional simulation of natural behaviour; and iii) directly participate in their own care, by communicating their preferences to humans. Although the findings were not as we had hypothesised (ie chimpanzees did not exhibit a medication preference or major changes in behaviour as a result of having a choice), the important aspect, and purpose of this study, is that animals were provided with the opportunity to exercise choice concerning their treatment. Whether this particular choice between two similar types of medications was meaningful from the chimpanzees’ perspective is questionable given the lack of preference shown by three of the four chimpanzees. Nonetheless, we believe that the application of this paradigm to more dissimilar types of choices (eg opioid vs NSAID, acupuncture vs laser therapy) may prove to be a more meaningful choice for chimpanzees. As the current chimpanzee population ages and continues to require more and more medical care, and as behavioural management continues to evolve toward the goal of optimal care, examining treatment preferences and the efficacy of those treatments will be an increasingly important component of captive care. Therefore, medication choice paradigms should become progressively more valuable tools in chimpanzee behavioural management programmes.
The finding that only one of the four chimpanzees chose one medication (ibuprofen) more often than the other (meloxicam) contrasts with findings from our pilot study, in which chimpanzees significantly preferred meloxicam over ibuprofen (Schapiro et al 2014). This seems to lend support to the idea that the preference for meloxicam in the previous study was likely to be an aversion to the visibly granulated, unpleasant tasting ibuprofen. The fact that three of the four chimpanzees in the current study exhibited no preference for either ibuprofen or meloxicam suggests that the animals may not have been able to distinguish differences in the effects of these two compounds with respect to ameliorating their arthritis-related symptoms. A lack of preference would be an important finding, given that ibuprofen and meloxicam are two commonly used NSAID pain-relief agents that differ in cost, administration frequency, and potential side-effects. If chimpanzees indeed do not have a preference for one over the other, veterinarians making decisions about analgesic treatment options may prefer using the easier to administer meloxicam over ibuprofen.
Impaired mobility is an important observable indicator of the effects of arthritis in chimpanzees (Magden et al 2013). As such, examining the efficacy of arthritis treatments requires assessments of multiple measures of mobility. In the current study, we measured mobility in a number of ways, including changes in average percentage of time spent locomoting, speed (m s−1) when completing a mobility test, and mobility scores using a caregiver-rating system. Chimpanzees exhibited similar percentages of time in locomotion during the ibuprofen and meloxicam phases, again suggesting that these two medications may be similarly effective from the animals’ perspective. Chimpanzees did spend 3–4% more of their time locomoting during the Ibuprofen, Meloxicam, and Choice phases than they did during the Baseline phase. This is approximately equivalent to an additional 21–28 min of locomotion per 12-h day and can be seen as a positive behavioural change as a result of participation in the study phases. However, it should be noted that the chimpanzees received their daily medication during the baseline phase in the form of crushed meloxicam pills mixed in a paper cup with orange juice administered by a veterinary technician. The change in administration techniques during the study (coloured Gatorade® containing medication [or placebo] presented three times a day by the experimenter from transparent squirt bottles) likely resulted in an increase in animal interaction with the experimenter. Therefore, it is possible that the increase in locomotion was due to aspects of the change in medication administration technique, rather than the medications themselves or the opportunity to choose daily medication. A similar effect of decreased inactivity as a result of increased human interaction has been reported in chimpanzees (Baker 2004) and cynomolgus macaques (Macaca fascicularis) (Tasker & Buchanan-Smith 2016).
Chimpanzees exhibited consistency across phases in mobility test speeds (m s−1) and mobility scores, and we established the validity of these mobility measures. Again, when examining the efficacy of therapies or treatments for arthritis, it is essential to use empirical assessments of mobility. The mobility test serves this purpose, as changes in speed can provide insights into the effectiveness of therapies and/or progression of symptoms. Furthermore, the fact that the mobility test speeds were significantly correlated with subjective ratings of mobility (from caregivers, veterinary technicians, and the observer) confirms the validity and enhances the value of the mobility scoring system, which requires virtually no staff or animal training. Using these empirical mobility assessments, we can identify specific aspects of mobility (eg climbing and brachiating) that require improvement for specific individuals. Personalised mobility enhancement strategies can then be developed to increase locomotion as an exercise regimen and/or as physical therapy (Bridges et al 2015). In turn, these mobility measures can be used again to assess the efficacy of the therapy.
Voluntary participation of chimpanzees in their own care via self-medication serves as an initial step toward developing ‘personalised’ medicine for captive NHPs. Understanding individual differences among animals, in this case in terms of each chimpanzee’s medication or treatment preferences, should allow identification of optimal treatments specific to individual animals and improve the overall care that each animal receives. We know that individual NHPs respond differently to similar medications and procedures, and that different treatments may be equally effective from the animals’ perspective. As such, it will become increasingly important to develop personalised treatments (Capitanio 2017).
Animal welfare implications and conclusion
The medication choice paradigm as applied to a variety of other healthcare-related options could be a useful approach for addressing chimpanzee health issues, as the animals choose their treatment, thereby voluntarily participating in their healthcare. We believe that the modest number of significant findings from the current study (which may be due to the similarity between the two medications), does not preclude the utility of the paradigm and methodology itself, which could be applied to a variety of other treatment options, regimens, and medications. The medication choice paradigm represents a behavioural management refinement that: i) increases the animals’ choice in the environment; ii) opens an additional channel of communication between chimpanzees and humans; and iii) functionally simulates some of the complex cognitive and behavioural aspects of self-medication-related behaviours that chimpanzees exhibit in the wild. This type of refinement is vital for both the behavioural and veterinary management of an ageing captive chimpanzee population that requires increasing medical care.
Supplementary Material
Acknowledgements
We would like to thank Dr Stephanie Buchl, Dr Elizabeth Magden, Mary Catherine Mareno, Lisa Reamer, Jeff Haller, Amanda Ott, Rachel Haller, Raquel Herrera, Heather Webb, Susan (Lambeth) Pavonetti, and the caregivers at the National Center for Chimpanzee Care for medication administration and assistance in solving logistical issues. This work was supported by NIH U42-OD 011197 and the University of Copenhagen. We have no conflicts of interest to declare.
References
- Albert KS and Gernaat CM 1984. Pharmacokinetics of ibuprofen. American Journal of Medicine 77: 40–46. 10.1016/S0002-9343(84)80017-0 [DOI] [PubMed] [Google Scholar]
- Altmann J 1974. Observational study of behavior: Sampling methods. Behaviour 49: 227–267. 10.1163/156853974X00534 [DOI] [PubMed] [Google Scholar]
- Association of Zoos and Aquariums (AZA) Ape TAG 2010. Chimpanzee (Pan troglodytes) Care Manual. Association of Zoos and Aquariums: Silver Spring, Maryland, USA [Google Scholar]
- Bae JW, Kim MJ, Jang CG and Lee SY 2007. Determination of meloxicam in human plasma using a HPLC method with UV detection and its application to a pharmacokinetic study. Journal of Chromatography, B, Analytical Technologies in the Biomedical and Life Sciences 859: 69–73. 10.1016/j.jchromb.2007.09.004 [DOI] [PubMed] [Google Scholar]
- Baker KC 2004. Benefits of positive human interaction for socially-housed chimpanzees. Animal Welfare 13: 239–245. [PMC free article] [PubMed] [Google Scholar]
- Baker KC 2016. Survey of 2014 behavioral management programs for laboratory primates in the United States. American Journal of Primatology 78: 780–796. 10.1002/ajp.22543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker KC and Aureli F 1997. Behavioural indicators of anxiety: An empirical test in chimpanzees. Behaviour 134: 1031–1050. 10.1163/156853997X00386 [DOI] [Google Scholar]
- Baker KC, Bloomsmith MA, Coleman K, Crockett CM, Worlein J, Lutz CK, McCowan B, Pierre P and Weed J 2017. The behavioral management consortium: A partnership for promoting consensus and best practices In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 9–23. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-3 [DOI] [Google Scholar]
- Baker KC, Weed JL, Crockett CM and Bloomsmith MA 2007. Survey of environmental enhancement programs for laboratory primates. American Journal of Primatology 69: 377–394. [DOI] [PubMed] [Google Scholar]
- Behringer V, Stevens JMG, Hohmann G, Möstl E, Selzer D and Deschner T 2014. Testing the effect of medical positive reinforcement training on salivary cortisol levels in bonobos and orangutans. PLoS ONE 9: 1–9. 10.1371/journal.pone.0108664 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloomsmith MA, Laule GE, Alford PL and Thurston RH 1994. Using training to moderate chimpanzee aggression during feeding. Zoo Biology 13: 557–566. 10.1002/zoo.1430130605 [DOI] [Google Scholar]
- Bloomsmith MA, Stone AM and Laule GE 1998. Positive reinforcement training to enhance the voluntary movement of grouphoused chimpanzees within their enclosures. Zoo Biology 17: 333–341. [DOI] [Google Scholar]
- Boelsterli UA, Redinbo MR and Saitta KS 2013. Multiple NSAID-induced hits injure the small intestine: underlying mechanisms and novel strategies. Toxicological Sciences 131: 654–667. 10.1093/toxsci/kfs310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bridges JP, Haller RL, Buchl SJ, Magden ER, Lambeth SP and Schapiro SJ 2015. Establishing a behavioral management program for geriatric chimpanzees. American Journal of Primatology 77: 111 [Google Scholar]
- Capitanio JP 2017. Variation in biobehavioral organization In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 55–73. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-6 [DOI] [Google Scholar]
- Coleman K, Pranger L, Maier A, Lambeth SP, Perlman JE, Thiele E and Schapiro SJ 2008. Training rhesus macaques for venipuncture using positive reinforcement techniques: A comparison with chimpanzees. Journal of the American Association for Laboratory Animal Science 47: 37–41 [PMC free article] [PubMed] [Google Scholar]
- de Roode JC, Lefèvre T and Hunter MD 2013. Self-medication in animals. Science, 340: 150–151. 10.1126/science.1235824 [DOI] [PubMed] [Google Scholar]
- Desmond T and Laule G 1994. Use of positive reinforcement training in the management of species. Zoo Biology 13: 471–477. 10.1002/zoo.1430130509 [DOI] [Google Scholar]
- Fowler A, Koutsioni Y and Sommer V 2007. Leaf-swallowing in Nigerian chimpanzees: Evidence for assumed self-medication. Primates 48: 73–76. 10.1007/s10329-006-0001-6 [DOI] [PubMed] [Google Scholar]
- Friend TH 1991. Symposium: Response of animals to stress. Journal of Dairy Science 74: 292–303. 10.3168/jds.S0022-0302(91)78173-3 [DOI] [PubMed] [Google Scholar]
- Fruth B, Ikombe NB, Matshimba GK, Metzger S, Muganza DM, Mundry R and Fowler A 2014. New evidence for self-medication in bonobos: Manniophyton fulvum leaf- and stemstrip-swallowing from LuiKotale, Salonga National Park, DR Congo. American Journal of Primatology 76: 146–158. 10.1002/ajp.22217 [DOI] [PubMed] [Google Scholar]
- Graham ML 2017. Positive reinforcement training and research In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 187–200. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-13 [DOI] [Google Scholar]
- Hanson JD, Larson ME and Snowdon CT 1976. The effects of control over high intensity noise on plasma cortisol levels in rhesus monkeys. Behavioral Biology 16: 333–340. 10.1016/S0091-6773(76)91460-7 [DOI] [PubMed] [Google Scholar]
- Heilbronner SR, Rosati AG, Stevens JR, Hare B and Hauser MD 2008. A fruit in the hand or two in the bush? Divergent risk preferences in chimpanzees and bonobos. Biology Letters 4: 246–249. 10.1098/rsbl.2008.0081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hopkins WD and Latzman RD 2017. Future research with captive chimpanzees in the United States: Integrating scientific programs with behavioral management In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 139–155. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-11 [DOI] [Google Scholar]
- Hopper LM, Freeman HD and Ross SR 2016. Reconsidering coprophagy as an indicator of negative welfare for captive chimpanzees. Applied Animal Behaviour Science 176: 112–119. 10.1016/j.applanim.2016.01.002 [DOI] [Google Scholar]
- Huffman MA 1997. Current evidence for self-medication in primates: A multidisciplinary perspective. American Journal of Physical Anthropology 104: 171–200. [DOI] [Google Scholar]
- Huffman MA, Spiezio C, Sgaravatti A and Leca JB 2010. Leaf swallowing behavior in chimpanzees (Pan troglodytes): Biased learning and the emergence of group level cultural differences. Animal Cognition 13: 871–880. 10.1007/s10071-010-0335-8 [DOI] [PubMed] [Google Scholar]
- Lambeth S, Bloomsmith M, Baker K, Perlman JE, Hook M and Schapiro SJ 2001. Control over videotape enrichment for socially housed chimpanzees: Subsequent challenge tests. American Journal of Primatology 54: 62–63 [Google Scholar]
- Lambeth SP, Hau J, Perlman JE, Martino M and Schapiro SJ 2006. Positive reinforcement training affects hematologic and serum chemistry values in captive chimpanzees (Pan troglodytes). American Journal of Primatology 68: 245–256. 10.1002/ajp.20148 [DOI] [PubMed] [Google Scholar]
- Laule G, Thurston RH, Alford PL and Bloomsmith MA 1996. Training to reliably obtain blood and urine samples from a diabetic chimpanzee (Pan troglodytes). Zoo Biology 15: 587–591. [DOI] [Google Scholar]
- Laule G and Whittaker M 2002. The use of positive reinforcement techniques with chimpanzees for enhanced care and welfare In: Brent L (ed) Special Topics in Primatology, Volume 2: Care and Management of Captive Chimpanzees pp 242–265. American Society of Primatologists: San Antonio, Texas, USA [Google Scholar]
- Laule G and Whittaker M 2007. Enhancing nonhuman primate care and welfare through the use of positive reinforcement training. Journal of Applied Animal Welfare Science 10: 31–38. 10.1080/10888700701277311 [DOI] [PubMed] [Google Scholar]
- Leotti LA, Iyengar SS and Ochsner KN 2010. Born to choose: The origins and value of the need for control. Trends in Cognitive Sciences 14: 457–463. 10.1016/j.tics.2010.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magden ER 2017. Positive reinforcement training and health care In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 201–215. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-14 [DOI] [Google Scholar]
- Magden ER, Haller RL, Thiele EJ, Buchl SJ, Lambeth SP and Schapiro SJ 2013. Acupuncture as an adjunct therapy for osteoarthritis in chimpanzees (Pan troglodytes). Journal of the American Association for Laboratory Animal Science 52: 475–480 [PMC free article] [PubMed] [Google Scholar]
- Magden ER, Sleeper MM, Buchl SJ, Jones RA, Thiele EJ and Wilkerson GK 2016. Use of an implantable loop recorder in a chimpanzee (Pan troglodytes) to monitor cardiac arrhythmias and assess the effects of acupuncture and laser therapy. Comparative Medicine 66: 52–58 [PMC free article] [PubMed] [Google Scholar]
- Masi S, Gustafsson E, Saint Jalme M, Narat V, Todd A, Bomsel MC and Krief S 2012. Unusual feeding behavior in wild great apes, a window to understand origins of self-medication in humans: Role of sociality and physiology on learning process. Physiology and Behavior 105: 337–349. 10.1016/j.physbeh.2011.08.012 [DOI] [PubMed] [Google Scholar]
- McDonald JH 2014. Handbook of Biological Statistics, Third Edition pp 29–39. Sparky House Publishing: Baltimore, Maryland, USA [Google Scholar]
- McKinley J, Buchanan-Smith H, Bassett L and Morris K 2003. Training common marmosets (Callithrix jacchus) to cooperate during routine laboratory procedures: Ease of training and time investment. Journal of Applied Animal Welfare Science 6: 209–220. 10.1207/S15327604JAWS0603_06 [DOI] [PubMed] [Google Scholar]
- Mellor D 2016. Moving beyond the ‘Five Freedoms’ by updating the ‘Five Provisions’ and introducing aligned ‘Animal Welfare Aims’. Animals 6: 59–79. 10.3390/ani6100059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mineka S, Gunnar M and Champoux M 1986. Control and early socioemotional development: Infant rhesus monkeys reared in controllable versus uncontrollable environments. Child Development 57: 1241–1256. 10.2307/1130447 [DOI] [Google Scholar]
- Morgan KN and Tromborg CT 2007. Sources of stress in captivity. Applied Animal Behaviour Science 102: 262–302. 10.1016/j.applanim.2006.05.032 [DOI] [Google Scholar]
- National Institutes of Health 2013. Council of councils working group on the use of chimpanzees in NIH-supported research report. https://dpcpsi.nih.gov/council/chimpanzee_research
- Neal Webb SJ, Hau J and Schapiro SJ 2018. Captive chimpanzee (Pan troglodytes) behavior as a function of space per animal and enclosure type. American Journal of Primatology 80: e22749 10.1002/ajp.22749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neal Webb SJ, Hau J and Schapiro SJ Does (group) size matter? Captive chimpanzee (Pan troglodytes) behavior as a function of group size and composition, in prep [DOI] [PMC free article] [PubMed]
- Nunamaker EA, Rick Lee D and Lammey ML 2012. Chronic diseases in captive geriatric female chimpanzees (Pan Troglodytes). Comparative Medicine 62: 131–136 [PMC free article] [PubMed] [Google Scholar]
- Obbo CJD, Makanga B, Mulholland DA, Coombes PH and Brun R 2013. Antiprotozoal activity of Khaya anthotheca, (Welv) CDC a plant used by chimpanzees for self-medication. Journal of Ethnopharmacology 147: 220–223. 10.1016/j.jep.2013.03.007 [DOI] [PubMed] [Google Scholar]
- Owen MA, Swaisgood RR, Czekala NM and Lindburg DG 2005. Enclosure choice and well-being in giant pandas: Is it all about control? Zoo Biology 24: 475–481. 10.1002/zoo.20064 [DOI] [Google Scholar]
- Perlmuter LC and Monty RA 1977. The importance of perceived control: Fact or fantasy? Experiments with both humans and animals indicate that the mere illusion of control significantly improves performance in a variety of situations. American Scientist 65: 759–765 [PubMed] [Google Scholar]
- Pomerantz O and Terkel J 2009. Effects of positive reinforcement training techniques on the psychological welfare of zoo-housed chimpanzees (Pan troglodytes). American Journal of Primatology 71: 687–695. 10.1002/ajp.20703 [DOI] [PubMed] [Google Scholar]
- Popilskis SJ, Lee DR and Elmore DB 2008. Anesthesia and analgesia in nonhuman primates In: Anesthesia and Analgesia in Laboratory Animals, Second Edition pp 335–363. Elsevier: Amsterdam, The Netherlands: 10.1016/B978-012373898-1.50016-4 [DOI] [Google Scholar]
- Reamer LA, Haller RL, Thiele EJ, Freeman HD, Lambeth SP and Schapiro SJ 2014. Factors affecting initial training success of blood glucose testing in captive chimpanzees (Pan troglodytes): Chimpanzee diabetic treatment training. Zoo Biology 33: 212–220. 10.1002/zoo.21123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinhardt V 1997. Training nonhuman primates to cooperate during handling procedures: A review. Animal Technology 48: 55–74 [Google Scholar]
- Reinhardt V 2003. Working with rather than against macaques during blood collection. Journal of Applied Animal Welfare Science 6: 189–197. 10.1207/S15327604JAWS0603_04 [DOI] [PubMed] [Google Scholar]
- Ross SR 2006. Issues of choice and control in the behaviour of a pair of captive polar bears (Ursus maritimus). Behavioural Processes 73: 117–120. 10.1016/j.beproc.2006.04.003 [DOI] [PubMed] [Google Scholar]
- Sambrook TD and Buchanan-Smith HM 1997. Control and complexity in novel object enrichment. Animal Welfare 6: 207–216 [Google Scholar]
- Schapiro SJ 2017. Handbook of Primate Behavioral Management. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652 [DOI] [Google Scholar]
- Schapiro SJ, Brosnan SF, Hopkins WD, Whiten A, Kendal R, Sherwood CC and Lambeth SP 2017. Collaborative research and behavioral management In: Schapiro SJ (ed) Handbook of Primate Behavioral Management pp 243–254. CRC Press: Boca Raton, Florida, USA: 10.1201/9781315120652-17 [DOI] [Google Scholar]
- Schapiro SJ, Perlman JE and Boudreau BA 2001. Manipulating the affiliative interactions of group-housed rhesus macaques using positive reinforcement training techniques. American Journal of Primatology 55: 137–149. 10.1002/ajp.1047 [DOI] [PubMed] [Google Scholar]
- Schapiro SJ, Reamer LA, Mareno MC and Lambeth SP 2014. Providing chimpanzees with opportunities to voluntarily participate in their own care: Choice of medications. American Journal of Primatology 76: 73 (Abstract) [Google Scholar]
- Tasker L and Buchanan-Smith HM 2016. Linking welfare and quality of scientific output through refinement of enhanced socialization with carestaff in cynomolgus macaques (Macaca fascicularis) used for regulatory toxicology. Joint Meeting of the International Primatological Society and the American Society of Primatologists 21–26 August 2016, Chicago, Illinois, USA [Google Scholar]
- Tennessen T 1989. Coping with confinement: features of the environment that influence animals’ ability to adapt. Applied Animal Behaviour Science 22: 139–149. 10.1016/0168-1591(89)90050-6 [DOI] [Google Scholar]
- Videan EN, Fritz J, Schwandt ML, Smith HF and Howell S 2005. Controllability in environmental enrichment for captive chimpanzees (Pan troglodytes). Journal of Applied Animal Welfare Science 8: 117–130. 10.1207/s15327604jaws0802_4 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.