Abstract
Escherichia coli is a pathogen commonly encountered in clinical laboratories, and is capable of causing a variety of diseases, both within the intestinal tract (intestinal pathogenic strains) and outside (extraintestinal pathogenic E. coli, or ExPEC). It is associated with urinary tract infections (UTIs), one of the most common infectious diseases in the world. This report represents the first comparative analysis of the draft genome sequences of 11 uropathogenic E. coli (UPEC) strains isolated from two tertiary hospitals located in Dhaka and Sylhet, Bangladesh, and is focused on comparing their genomic characteristics to each other and to other available UPEC strains. Multilocus sequence typing (MLST) confirmed the strains belong to ST59, ST131, ST219, ST361, ST410, ST448 and ST4204, with one of the isolates classified as a previously undocumented ST. De novo identification of the antibiotic resistance genes blaNDM-5, blaNDM-7, blaCTX-M-15 and blaOXA-1 was determined, and phenotypic-genotypic analysis of virulence revealed significant heterogeneity within UPEC phylogroups.
Subject terms: Infectious-disease diagnostics, Pathogens
Introduction
Urinary tract infections (UTIs) are the most common bacterial infections affecting approximately 11% of adult women each year globally, with approximately 60% of women experiencing UTI during their lifetime1, 2. Sporadic studies done on the prevalence of UTIs in Bangladesh and an investigation of 200 UTI patients, including men and women of various age groups, found females to be more susceptible to UTIs (80% positive) than males. In both genders, the prevalence rate was highest among those in the age group of 21–40 years (33%)3. The study also showed E. coli to be the predominant etiological agent, contributing to 57.38% of infections3.
Escherichia coli is an extremely diverse bacterial species which can be categorized into three major groups based on disease causing capability: commensal or nonpathogenic E. coli; intestinal pathogenic E. coli causing diarrhea; and extraintestinal pathogenic E. coli (ExPEC). The ExPEC term was described by Johnson et al. in 20004 and further subclassified as uropathogenic E. coli (UPEC), sepsis-associated E. coli (SEPEC), and neonatal meningitis-associated E. coli (MNEC)5. ExPECs are known to invade extraintestinal tissue and cause pathogenesis by harboring a variety of virulence factors, either present in the chromosome or carried in mobile genetic elements such as plasmids, thereby conferring greater diversity among ExPEC strains5,6.
Traditionally, E. coli phylogroups B2 and D have been understood to cause the majority of ExPEC infections, while phylogroups A and B1 were associated with comensal extraintestinal strains7. However, recent reports have revealed higher percentages of phylogroup A strains in UTI cases8. A strong association has also often been detected between a particular multilocus sequence type (MLST) with a pathology, such as the correlation of globally dominant E. coli ST131 and extraintestinal infections, especially in India9. Like ST131, many other successful clonal lineages of different sequence types (ST), including 410, 95 and 10 have disseminated globally due to their relatively higher virulence, fitness, and metabolic capabilities, along with acquisition of antibiotic resistance genes10–13.
Carbapenems are considered last-resort antibiotics, therefore resistance to this group of antibiotics is a greater health concern in treating infections caused by extended-spectrum-β-lactamase (ESBL)- or AmpC-producing bacteria14. New Delhi metallo-β-lactamase (NDM) is a relatively recent group of metallo-β-lactamase (MBL) that, over the last decade, has undergone rapid spread in the South-Asian continent15. While NDM producers have been found to be susceptible to a few antibiotics including colistin, several recent studies have reported that this treatment approach might not be sustainable and could become a very serious public health concern16,17. NDM genes are found both in plasmids and chromosomally integrated in various bacterial pathogens18,19. Reports of E. coli harboring NDM-1 and other ESBL genes such as CTX-M and OXA-48 have emerged from various parts of the world, including Japan, the Netherlands, South Korea, and Tanzania10–12,20–23. In addition, many such studies have detected the chromosomal integration of NDM genes in various ExPEC sequence types, such as ST38, ST410, ST131 and ST64824.
Association of different virulence factors, e.g., sat (secreted autotransporter toxin), iutA (aerobactin (siderophore) receptor), malX (pathogenicity island marker) and ompT (outer-membrane protease T), has also been reported, with specific sequence types such as ST38, ST131, ST405, and ST648 isolated25. In general, however, few genomic investigations have been done that could shed light on molecular mechanisms of pathogenesis and antibiotic resistance mechanisms and correlate those traits with the genotypes of local pathogens, especially in a developing country like Bangladesh.
To the best of our knowledge, there is no genomic information available on UPEC isolates circulating in Bangladesh. This study represents an initial effort to obtain genomic information on Bangladeshi UPEC isolates and to analyze genomic variations between Bangladeshi isolates and ExPECs from different parts of the world. Eleven strains representing different ExPEC phylogroups and antibiotic resistance were selected and their genomes determined using next-generation sequencing. Genotype–phenotype correlation analyses were also done on the isolates to determine virulence properties, e.g., biofilm formation, serum resistance, hemolysis, and antibiotic resistance.
Results
Antibiogram and phylogroup analysis
Presumptive identification using colony morphology revealed 47 of 74 (63.5%) bacterial isolates obtained from the Dhaka Central International Medical College and Hospital (DCIMCH) and 19 of 32 isolates (59.4%) from the Ibn Sina Hospital, Sylhet (ISH) were E. coli. All isolates from DCIMCH exhibited increased resistance to commonly used antimicrobials, including β-lactams (third- and fourth-generation cephalosporins), fluoroquinolones, and aminoglycosides (Table 1; Supplementary Table S1). Antibiotic sensitivity patterns differed between isolates collected from the two hospitals, with isolates from DCIMCH showing resistance to a larger number of antibiotics. For example, while most of the DCIMCH isolates were resistant to cefixime (85.1%), only 26.3% from ISH showed resistance to this antibiotic. DCIMCH isolates showed high frequency of resistance to second generation cephalosporin cefuroxime (83%), third generation cephalosporins ceftriaxone (80.9%) and ciprofloxacin (72.3%), and the monobactam, aztreonam (72.3%). ISH isolates, in contrast, showed resistance mainly to doxycycline (42.1%) and amoxicillin (36.8%). While ca. 17% (n = 8) of DCIMCH isolates conferred resistance to the carbapenem, imipenem, all of the ISH isolates were sensitive to carbapenems and all isolates included in this study were sensitive to colistin. Five isolates from DCIMCH were ESBL (Extended Spectrum β-lactamase) positive, but none from ISH were positive (Table S1).
Table 1.
Percentage of DCIMCH and ISH strains resistant to different antibiotics.
| DCIMCH | ISH | ||
|---|---|---|---|
| n = 47 [female = 25, male = 22] | n = 19 [female = 10, male = 9] | ||
| Colistin | 0% | Colistin | 0% |
| Polymixin B | 0% | Polymixin B | 0% |
| Cefotaxime | 80.90% | Ceftriaxone | 5.26% |
| Ceftriaxone | 80.90% | Ceftazidime | 0% |
| Ceftazidime | 46.80% | Cefixime | 26.30% |
| Cefixime | 85.10% | Cefoxitin | 26.32% |
| Cefuroxime | 80.90% | Imipenem | 0% |
| Cefepime | 55.30% | Meropenem | 0% |
| Aztreonam | 72.30% | Doxycycline | 42.10% |
| Imipenem | 17.00% | Gentamicin | 0% |
| Meropenem | 19.10% | Mecillinam | 31.60% |
| Ciprofloxacin | 72.30% | Amoxicillin | 36.80% |
| Gentamicin | 25.50% | Azithromycin | 31.60% |
| Co-trimoxazole | 51.10% | Trimethoprim | 26.32% |
| Levofloxacin | 63.80% | ||
| Nalidixic Acid | 80.90% | ||
| Netilmicin | 12.80% | ||
| Nitrofurantoin | 17.02% | ||
| Piperacillin/Tazobactum | 34.00% | ||
| Tigecycline | 2.13% | ||
| Amikacin | 14.90% | ||
| Amoxyclave | 55.30% | ||
Phylogroup determination based on PCR detection of chuA, yjaA and TspE4.C226, showed phylogroup B2 and phylogroup A to be most abundant, with B2 comprising 19 (40.4%) and A 14 (29.8%) of the 47 isolates (Table S2). A total of 11 strains (23.4%) were classified in phylogroup B1, while three (6.4%) were phylogroup D. Seven of 66 isolates harboured NDM-1 gene and all NDM positive strains were from DCIMCH. No association was observed between NDM and a particular phylogroup, with three strains from phylogroup A, two strains from B1, and one strain from B2 carrying the NDM gene.
Genomic features and strain characterization
Eleven isolates from the phylogroups were selected with the number of isolates from each phylogroup roughly proportional to prevalence of that phylogroup within the set of 47 isolates in this study. These were selected based on resistance patterns. Four isolates were selected from phylogroups A and B2, two from phylogroup B1 (NGE5 and NGCE100), and one from phylogroup D (NGE3). Combined length of contigs of the assembled genomes of each of the 11 strains ranged from ~ 4.3 to 5.4 Mbp, with N50 value (the minimum contig length required to cover 50% of the genome) ranging between 58,987 and 363,834 bp (Table 2). Size of the pangenome (i.e. total gene repertoire) was 16,797 genes and core genome 2,945 genes.
Table 2.
Genome assembly statistics of the 11 sequenced UPEC isolates.
| Sample name | Strain ID | Accession number | Genome size | Number of contigs (> 500 bp) | Largest contig size | N50 value |
|---|---|---|---|---|---|---|
| NGRI_A12 | NGE3 | QEXN00000000 | 5,104,547 | 147 | 319,826 | 167,222 |
| NGRI_A13 | NGE4 | QFAZ00000000 | 4,786,247 | 105 | 339,704 | 206,487 |
| NGRI_A14 | NGE5 | RCIF00000000 | 4,669,166 | 54 | 1,088,366 | 354,566 |
| NGRI_A15 | NGE6 | RCIE00000000 | 4,885,404 | 59 | 711,364 | 363,834 |
| NGRI_A16 | NGE7 | QFRN00000000 | 5,304,720 | 101 | 655,033 | 204,841 |
| NGRI_A18 | NGE9 | QFRT00000000 | 4,254,362 | 205 | 190,686 | 58,987 |
| NGRI_B10 | NGE16 | QFTM00000000 | 5,232,178 | 305 | 179,874 | 60,840 |
| NGRI_B29 | NGE22 | QFXA00000000 | 5,061,598 | 134 | 338,320 | 157,136 |
| NGRI_C17 | NGCE33 | RBWA00000000 | 4,805,154 | 99 | 553,030 | 204,552 |
| NGRI_C19 | NGCE94 | RAZR00000000 | 5,143,790 | 204 | 262,871 | 108,692 |
| NGRI_C20 | NGCE100 | RAZQ00000000 | 5,422,176 | 244 | 313,308 | 105,294 |
De novo analysis was used to confirm phylogroups of the assembled genomes and MLST analysis showed that, while strains belonging to a phylogroup were heterogenous in MLST types, there was direct correlation between serotype and MLST, with ST131 strains NGE7 and NGE16 both serotype O25:H4 (Table 3).
Table 3.
De novo prediction of phylogroups, MLST types and serotypes of the sequenced UPEC isolates.
| Strains | Hospital | Phylogroups | MLST type | Serotype |
|---|---|---|---|---|
| NGE3 | DCIMCH | D | ST-59 | O1:H7 |
| NGE4 | DCIMCH | A | ST-4204 | O6:H10 |
| NGE5 | DCIMCH | B1 | Unknown ST | O59:H20 |
| NGE6 | DCIMCH | B2 | ST-219 | O138:H48 |
| NGE7 | DCIMCH | B2 | ST-131 | O25:H4 |
| NGE9 | DCIMCH | B2 | ST-219 | O138:H48 |
| NGE16 | ISH | B2 | ST-131 | O25:H4 |
| NGE22 | ISH | A | ST-4204 | O6:H10 |
| NGCE33 | DCIMCH | A | ST-410 | O8:H9 |
| NGCE94 | DCIMCH | A | ST-361 | O9:H30 |
| NGCE100 | DCIMCH | B1 | ST-448 | Ounknown:H7 |
Phylogenetic and cluster dendrogram analysis
The 11 strains were compared to obtain the number of SNPs shared between any two strains. From the SNP matrix shown in Supplementary Table S3, isolates with the same ST shared a low SNP count, while isolates within the same phylogroup but different STs had high SNPs. For example, NGE7, NGE16, and reference strain (NA114) belong to ST131 and share a low SNP count of 411 bp, whereas ST219 strains (NGE9 and NGE6) and ST131 strains had high SNP count but were the same phylogroup.
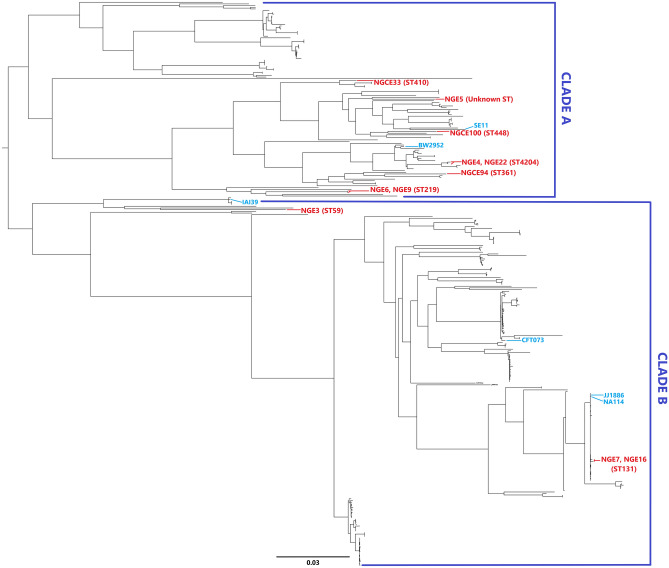
Core alignment using parSNP aligned 189 of 402 UPEC strains available online (list of strains is given in Supplementary Table S4). A total of the 60,815 SNPs identified was extracted and linked to construct a midpoint rooted phylogenetic tree (Fig. 1), showing two major clades, Clade A and Clade B. Clade A branched into subclades, with strains from phylogroup A and B1 in one subclade and ST219 strain of phylogroup B2 in another. NGCE33 is an ESBL-containing, highly virulent strain of ST410 which, despite belonging to phylogroup A, clustered distantly from the rest of phylogroup A strains (NGE22, NGE4 and NGCE94) and closer to phylogroup B1 (NGE5, NGCE100 and SE11). Clade B also branched phylogroup D and B2 away from each other. It was observed that isolates obtained from urine and blood samples interleaved, without significant clustering of infection type. However, NGE5 (proposed as a new ST) in clade A, NGE7, and NGE16 (ST131) in clade B joined strains isolated from blood. Strains belonging to the same MLST were placed together in the phylogenetic tree.
Figure 1.
Phylogenomic organization of publicly accessible UPEC isolates with sequenced Bangladeshi isolates in this study. Mid-point rooted SNPtree demonstrates the phylogenetic distribution of 11 UPEC genomes of Bangladeshi UPEC isolates amongst 5 UPEC reference genomes and 386 UPEC genomes (isolated from both urine and blood) available online. The well characterized reference genomes and UPEC isolates of this study have been labeled in blue and red respectively.
Hierarchical clustering of the 11 UPEC strain sequences represents similarity of shared accessory genome content, yielding three major clusters, C1, C2, and C3, respectively (Fig. 2). Phylogroup D out grouped, forming a distinct cluster, C1, while a combination of phylogroups A, B1, and B2 joined the remaining two clusters (C2 and C3). C2 further divided into two distinguishable clusters comprising strains of ST131 family in one group and four multidrug resistant, highly virulent strains of various MLSTs in another. These four isolates included one ST448 strain, one ST361 strain carrying both blaNDM and ESBL genes, one “high-risk” clone of ST410 lineage27, and one resistant strain of the ST4204 family. The clustering pattern of C2 suggests sharing of accessory genes between highly virulent strains, irrespective of phylogroups and ST. C3 also separated into two clusters, comprising two less virulent phylogroup B2 strains of the ST219 family in one clade, and the moderately virulent ST4204 strain of phylogroup A (NGE4) and the phylogroup B1 strain of unknown ST (NGE5) in the other.
Figure 2.
Dendrogram illustrating pan genome clustering of sequenced UPEC isolates. Dendrogram branches into three major clusters (C1, C2 and C3) based on the presence and absence of characterized accessory genes in the pan genome.
Accessory gene distribution responsible for this hierarchical clustering pattern is listed in Supplementary Table S5. Strains in cluster C2 share genes including OriC-binding nucleoid-associated protein (cnu), hemolysin expression-modulating protein (hha), suppressor of T4 td mutant (stpA) responsible for regulation of hemolysin (hly) gene expression and genes such as periplasmic inhibitor of g-type lysozyme (pliG) which provides lysozyme tolerance. Similarly, shared genomic content in cluster C3 involves genes RenD, ybcN and ybcK coding for uncharacterized prophage related proteins absent in C1 and C2 strains. Strains ST131 and ST219 belong to phylogroup B2 yet are located in two different clusters as ST131 strains contain 223 unique genes with significant enrichment in genes involved in biosynthetic and metabolic processes absent in ST219 strains, thus explaining the high numbers of SNPs between strains of the two STs.
De novo identification of antibiotic resistance markers
Genome annotation revealed that chromosomes of all strains sequenced had previously been reported to carry intrinsic antibiotic resistance genes, such as marA, gyrA, parC and parE28, as well as plasmid-mediated resistance genes belonging to AMR families, including β-lactamases, fluoroquinolones, aminoglycosides, macrolides, tetracyclines, trimethoprims, and sulfonamides. Genes associated with antibiotic resistance are shown in Fig. 3a.
Figure 3.
(a) Distribution of key antimicrobial resistance conferring genes within the 11 strains. (b) Distribution of key virulence factors within the 11 strains. Black: present, grey: absent.
Eleven isolates, irrespective of phylogroup and sequence type (ST), showed resistance to antibiotics according to presence of resistance genes. ESBL genotype blaCTX-M-15 is a predominant gene present in two NDM positive strains (NGCE100, NGCE94) and two ST131 strains (NGE16 and NGE7). NDM positive strains possessing blaNDM-5 and blaNDM-7 belong to phylogroup A and B1, and MLST groups ST361 and ST448, respectively. In addition, NGCE94 (ST361) contained β-lactamases blaTEM-1B and blaOXA-1, tetracycline-resistance tet(B), quinolone-resistance gene qepA, trimethoprim-resistance gene dfrA12 and chloramphenicol-resistance gene catA1. Variants of blaTEM and dfrA were also present in the highly resistant ST4204 strains (NGE4 and NGE22) belonging to the same phylogroup as ST361. Relatively less resistant ST219 strains (NGE6 and NGE9) of phylogroup B2 present a similar resistance pattern and contain resistance markers (tet(A) and qnrS1) common to phylogroup A rather than phylogroup B. Another member of phylogroup A (NGCE33) included highly resistant ST410 containing an array of β-lactamase genes including blaOXA-1B, blaCMY-2, blaDHA-1 and blaSHV-12. In addition, it contained a number of aminoglycosidase genes, fluoroquinolone-resistance gene qnrB4, macrolide gene mph(A) and trimethoprim-resistance genes drfA1 and drfA17. Genes strB and mph(A) were also shared by ST4204 isolates. Moreover, genes rmtB, aadA1, mph(A) and erythromycin-resistance gene erm(B) were harboured by NDM positive strains. Only a few resistance genes such as acc(6′)lb-cr, catB3 and blaOXA-1 were shared between the ST131 and ST410 strains.
Overall, the most resistant strain was NGCE33, based on genes coding for β-lactamase resistance, including the ESBL, blaCTX-M-15. This strain and NGCE100 were resistant to nitrofurantoin, a last resort nephrotoxic antibiotic that recently is more commonly used to treat carbapenem resistant UTIs.
Analysis of virulence profiles and genotype–phenotype correlation
UPEC pathogenesis encompasses a range of mechanisms including colonization of the urinary tract, protection against host defenses, and toxin production. Hemolysin production of 11 strains was tested, using blood agar and, while alpha hemolytic activity was observed only for ST131 strains NGE7 and NGE16, mild hemolysis was detected for NGE3, NGE4, and NGE22.
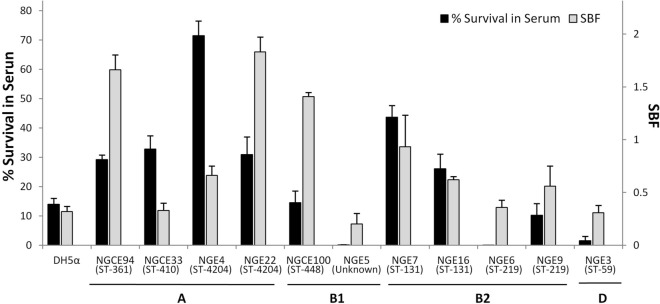
An important pathogenic determinant of UPEC is ability to form biofilm29. The biofilm formation assay results showed variation in biofilm formation both between and within the phylogroups (Fig. 4). Three strains (NGE22, NGCE94 and NGCE100) were classified as strong biofilm formers and four (NGE4, NGE7, NGE9 and NGE16) were moderate biofilm formers, based on specific biofilm formation value (SBF). Differences in biofilm forming ability were observed by sequence type, as exemplified by NGE7 and NGE16, both in the pandemic ST131 family, as well as ST219 strains (NGE6 and NGE9) and ST4204 strains (NGE4 and NGE22).
Figure 4.
Serum resistance and biofilm forming propensities of 11 E. coli isolates. Pooled NHS was inoculated with overnight LB culture in 1:10 ratio. Bacteria were enumerated at 0 and 3 h of incubation at 37 °C, and percentage survival in serum was calculated. For biofilm assay, bacteria were grown in M63 media and specific biofilm formation (SBF) was calculated. Error bars represent standard error (SE).
Bactericidal activity of serum complement acts as a major first-line defense against bacterial infection infiltrated into tissue30. In vitro serum resistance assay results showed all strains were susceptible to human serum bactericidal activity to varying degrees (Fig. 3). Except for NGE3, NGE5, and NGE6, they showed survival capacity equal to or greater than that of DH5α. While serum has a more pronounced bactericidal activity for strains belonging to phylogroup B1 and D, most strains of phylogroups A and B2 exhibited better survival in serum. Although little correlation was observed between degree of biofilm formation and serum resistance (r = 0.188) (Fig. 4), moderate/strong biofilm formers all showed greater survival in serum compared to weak biofilm formers.
Pan-genome analyses revealed virulomes of the 11 strains (Fig. 3b). In general, ST131 strains NGE7 and NGE16 carry an extensive repertoire of virulence genes. While the core genome encompasses gene families from different classes of virulence factors, not all are conserved, according to the SNP/bp ratio (Table 4).
Table 4.
SNP distribution of core virulence genes.
| Gene | Gene function | SNP count | Gene size (bp) | Alignment size (bp) | SNP/bp |
|---|---|---|---|---|---|
| ompA | Outer Membrane Protein A | 59 | 1,054 | 1,054 | 0.056 |
| uspA | Universal Stress Protein A | 79 | 438 | 435 | 0.181 |
| uspB | Universal Stress Protein B | 4 | 336 | 336 | 0.012 |
| uspC | Universal Stress Protein C | 12 | 430 | 430 | 0.028 |
| uspD | Universal Stress Protein D | 11 | 429 | 429 | 0.026 |
| uspE | Universal Stress Protein E | 29 | 951 | 951 | 0.03 |
| uspF | Universal Stress Protein F | 7 | 435 | 435 | 0.016 |
| uspG | Universal Stress Protein G | 34 | 429 | 429 | 0.079 |
| tolA | Tol-Pal System Protein A | 33 | 1,311 | 1,311 | 0.025 |
| tolB | Tol-Pal System Protein B | 30 | 1,293 | 1,293 | 0.023 |
| tolQ | Tol-Pal System Protein Q | 29 | 693 | 693 | 0.042 |
| entA | 2,3-Dihydro-2,3-dihydroxybenzoate dehydrogenase | 50 | 747 | 747 | 0.067 |
| entB | Enterobactin Synthase Component B | 37 | 858 | 858 | 0.043 |
| entC | Isochorismate Synthase | 58 | 1,176 | 1,176 | 0.049 |
| entD | Enterobactin Synthase Component D | 121 | 783 | 660 | 0.183 |
| entE | Enterobactin Synthase Component E | 116 | 1,611 | 1,611 | 0.072 |
| entF | Enterobactin Synthase Component F | 322 | 3,882 | 3,882 | 0.083 |
| entS | Enterobactin Synthase Component S | 117 | 1,251 | 1,251 | 0.094 |
| fimA | Type 1 fimbral protein, chain A | 218 | 567 | 567 | 0.384 |
| fimC | Chaperone Protein | 23 | 726 | 726 | 0.032 |
| fimD | Outer Membrane Usher Protein | 89 | 2,637 | 2,637 | 0.034 |
| fimF | Type 1 fimbral protein, chain F | 23 | 531 | 531 | 0.043 |
| fimG | Type 1 fimbral protein, chain G | 12 | 504 | 504 | 0.024 |
| fimH | Type 1 fimbrin d-mannose specific adhesin | 34 | 903 | 903 | 0.038 |
Comparison with VFDB indicated adhesion factors belonging to the ecp (Escherichia coli common pilus), csg (Curli fibers) and fim (type 1 fimbriae) gene families are part of the core genetic pool, while afaB/C (adherence fibrillar adhesion) and sfa (S fimbrial adhesion) are absent. Variation was noted in presence of the foc (F1C fimbriae) and pap (pyelonephritis associated pili) family genes. SNP distributions show that while other members of the fim genes are moderately conserved, fimA displays high genetic variability with SNP/bp of 0.383. Among the conserved usp (universal stress protein) genes that are involved in bacterial adhesion, uspA also indicated greater variation with SNP/bp of 0.181. Among toxicity conferring genes, hemolysin toxin hlyA was detected in five strains, including the two ST131 strains. Other genes, such as hlyB, hlyC, hlyD, cnf-1 and sat, were present exclusively in only ST131 strains.
All 11 strains carried the well-characterized serum resistance gene ompA, while traT and bor were missing from their genomes. Only ST131 strains harbored iss (increased serum survival) another important gene in the serum resistome of E. coli. The genomes were also analyzed for presence of 56 genes recently characterized as belonging to the serum resistome of EC985 by transposon-directed insertion site sequencing (TraDIS)31. Most of these genes, including tol(A,B,Q) and rfaH were detected in the core genome. However, some essential genes were either completely absent (hyxA and hyxR) or not identical to the reference strain from the VFDB database (waaP and waaG).
Analysis of pathogenicity islands (PAIs)
CFT073, a well characterized pyelonephritogenic strain, harbours PAIs that suggest strong virulence when present in UPEC isolates32,33. Many UTI associated strains, as well as commensal E. coli, carry PAIs that were first identified in strain 53634. To determine genetic composition of PAIs, genomes of the 11 UPEC isolates were analyzed for presence of the CFT073 and 536 associated PAI genes as retrieved from the PAthogenisity Island DataBase (https://www.paidb.re.kr/) (Supplementary Table S6).
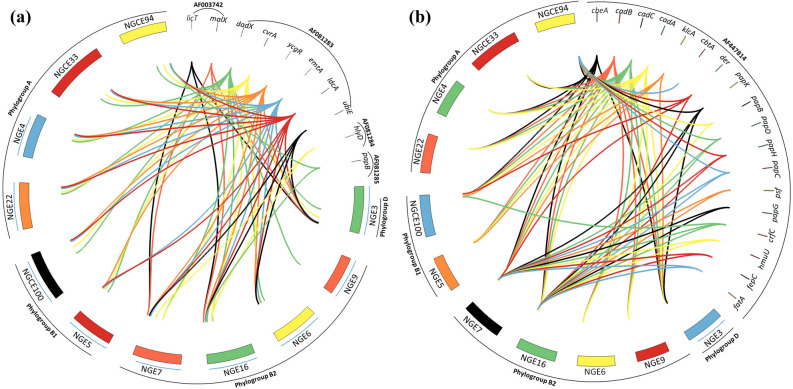
Analysis of PAI ICFT073 (Fig. 5a) showed that only two ST131 strains (NGE7 and NGE16) and NGE3 carried malX, a phosphotransferase system enzyme coding gene linked with occurrence of extraintestinal infections. The PAI ICFT073 gene dadX, however, was detected in all strains, while other genes, such as those coding for the motility regulating factors ycgR and emtA, a peptidogycan recycling enzyme ldcA (L, D-carboxypeptidase A) and cvrA (putative K+:H+ antiporter), were detected in all strains except NGE9. Apart from cad (BAC) which was present in all strains, most of the PAI IICFT073 associated genes (Fig. 5b) were found only in the ST131 isolates, NGE7 and NGE16. However, a phylogroup A strain NGCE94, was found to carry several genes of the pap (P fimbriae coding) operon indicating the possibility of acquisition of PAI IICFT073 from more virulent strains in the genitourinary microenvironment.
Figure 5.
Distribution of genes associated with a. PAI ICFT073 and b. PAI IICFT073 in 11 UPEC genomes. Relational diagrams were generated using CIRCOS software. Bands were created for each gene of CFT073 PAI-I and PAI-II along with the 11 sequenced strains to depict the presence of CFT073 associated PAI genes in the strains.
Analysis of Strain 536 associated genetic elements showed that PAI I536 (containing hemolysin genes hlyA, B, C, and D) as well as PAI II536 were exclusively found in NGE7 and NGE16. PAI III536 gene yciC (UPF0259 membrane protein) was only present in NGE3, NGE7 and NGE16. Furthermore, all strains apart from NGE3, NGE4 and NGE22 were enriched with genes from PAI V536. PAI IV536 genes on the contrary, had more diffuse distribution among the strains with only mtfA (mnemonic for Mlc titration factor A). That PAI IV536 is considered more stable than other PAIs of strain 53635 may explain these results.
Discussion
High-throughput sequencing technology development has led to a significant decrease in the cost of whole genome sequencing of bacterial pathogens so that sequencing is routine in developed countries. Infrastructure for next generation sequencing is now being developed in research centers located in developing countries including Bangladesh. The NSU Genome Research Institute (NGRI) at North South University, Bangladesh, has been established and aims to decipher whole genomes of bacterial pathogens of public health concern to Bangladesh. This initial study was carried out to determine genome characteristics of UPEC strains circulating in the country by sequencing 11 UPEC strains representing prevalent ExPEC phylogroups and antibiotic resistance profiles.
ST410 has been previously detected in Southeast Europe, Middle East and Greece28 and classified as a “high-risk clone” with essential regular monitoring due to its enhanced resistance mechanisms and moderate virulence36. The resistance and virulence patterns of ST410 strain NGCE33 validates this finding, and its very first identification in Bangladesh emphasizes the extent of its spread across continents. This study also reports an unknown MLST type i.e. NGE5, an observation that highlights the rapidly mutating nature of the UPEC genome and indicates the value of genomic characterization of local isolates.
The phylogenetic tree based on difference in SNPs (Fig. 1) showed phylogroups A and B1 branching separately from phylogroups B2 and D. This finding supports previous reports of phylogroup A and B1 belonging to sister lineages36,37. Phylogroup D and B2 can be concluded to have the same ancestral origin since most of the strains of these phylogroups are located in the same clade (Clade B) in SNP tree. A lack of clear phylogenomic separation of strains isolated from urine and blood coincides with results of previous related work28,38. The difference in SNP counts observed in this study further elucidates genomic relatedness since these sites account for the variation in nucleotide sequence. It was observed that ST219 and ST131 strains have an increased number of SNP difference despite belonging to the same phylogroup and thus cluster in separate clades in the phylogenetic tree. This may be due to difference in pathogenic capabilities and resistance potentials between STs, i.e., gain and loss of accessory genome in genomic diversity, with strains harbouring different fitness factors and MDR genotypes, irrespective of ST and phylogroup. Comparative genomic analysis of the sequenced UPEC strains showed that the phylogenetic analysis is congruous with serotype, sequence type, virulence and AMR pattern.
This is further supported by results of the genomic and phenotypic analyses (Fig. 4). The genetic architectures of strains included in this study are concordant with previous findings, with well known pandemic strains such as ST131 possessing many virulence genes and enriched PAIs9. However, as supported by the results of this study, although ST131 strains exhibit a notably dense virulome compared to other sequenced strains, a stark difference was not observed between the phenotypic virulence of ST131 and non ST131 strains (Fig. 4). Strains within the same phylogroup also displayed varied levels of virulence. While ST131 strains of phylogroup B2 (NGE7 and NGE16) displayed relatively strong phenotypic virulence, another strain NGE6, also classified under phylogroup B2 but of ST219 origin, displayed a much weaker pathogenic potential, such as weak biofilm formation.
Closer scrutiny of virulomes of the sequenced strains reveal certain virulence factors pertaining to resistance against serum bactericidal activity were either completely missing or present in degraded form within the bacterial genomes. These discrepancies explain that while most strains are capable of enduring serum bactericidal activity, they do not possess the robust serum resistome required to overcome complement action and proliferate in serum. The lack of any clear association between a particular gene and a given virulence phenotype suggests that likely there is a combinatorial effect of genes on pathogenic potential. E. coli possesses an open pan-genome by continuing gene acquisition, as found in other studies38,39. This characteristic may also explain phenotypic results as mentioned above and acquisition of yet-to-be characterized new genes may determine pathogenicity of strains. Several recent studies report an altered pathogenic potential of commensal E. coli40 and a similar observation can be made from this study. Emergence of highly virulent strains belonging to phylogroup A may be due to the open pan genome nature of E. coli, allowing it to acquire new virulence factors and resistance markers. SNP analyses of core virulence factors reveal that certain genes within a given gene family, such as fimA, are prominently more polymorphic compared with other members.
Several findings have indicated that septicemic/pyelonephritogenic strains carry certain virulent genes located in mobile genetic elements, called pathogenicity islands (PAI), usually absent in avirulent or less virulent strains41. Of all the strains sequenced in this study, ST131 strains NGE7 and NGE16 predictably possessed the most gene-dense CFT073 and 536 associated PAIs (Fig. 5). However, the presence of a large number of PAI IICFT073 mediated adhesion factors in the genome of NGCE94 indicates dissemination of virulence traits via horizontal transfer of PAIs from commonly virulent phylogroup B2 strains to less virulent phylogroup A strains. Transfer and evolution of genetic elements like PAIs contribute to fitness and pathogenic properties of UPEC.
Rapid emergence of multidrug resistant (MDR) strains of E. coli is a very serious concern, especially in a developing country like Bangladesh which experiences antibiotic misuse. Most resistance properties emerge via intra-species horizontal resistance gene transfer42. Clearly, however, excessive use of antibiotics in the global community creates evolutionary pressure towards enhanced resistance of UPEC28. Reported spread of the pan beta-lactam antibiotic resistance blaNDM family of genes is a major cause for concern, because of resistance conferred against penems, cephamycins, cephalosporins, and carbapenems, as well as horizontal transfer since the gene is located in a plasmid. This study reveals the presence of NDM gene varieties and a number of other ESBL genes in phylogroup A and B1 strains. In addition, the spread of virulence properties to strains like NGCE94, which is both NDM and ESBL positive, can have a profound effect on healthcare and spread of disease in Bangladesh.
In conclusion, this study presents useful insight into the genomes of Bangladeshi UPEC isolates, notably reporting for the first time an emerging pandemic clone ST410 in Bangladesh, contributing to the global distribution of this lineage. The study also demonstrates that strains belonging to phylogroup A exhibit virulence characteristics comparable to globally predominant known virulent ST131 (phylogroup B2) isolates, while other phylogroup B2 strains, such as ST219, display lower pathogenic potential. It also substantiates classification based on sequence type being an improved measure of genomic relatedness and pathogenicity. The risks posed by emerging pathogenic strains within different phylogroups need further assessment using comparative genomics and larger sample size.
Methods
Selection of isolates and antibiotic resistance profiling
A total of 106 bacterial isolates were cultured from urine of patients suffering from UTI and admitted to either the intensive care or emergency unit of two tertiary hospitals in Bangladesh from the period of June, 2017 and July, 2018. A total of 74 isolates were from inpatients admitted to Dhaka Central International Medical College and Hospital (DCIMCH), Dhaka and 32 isolates were collected from inpatients at Ibn Sina Hospital, Sylhet (ISH). E. coli colonies were presumptively identified by their colony morphology on MacConkey agar. Antibiotic susceptibility was carried out using the disc diffusion method and included 22 antibiotics for the 47 strains isolated from DCIMCH and 16 antibiotics for the 19 ISH strains (Table S1). Results were interpreted according to the 27th edition of Clinical and Laboratory Standards Institute (CLSI) guidelines. The isolates were transferred to North South University Genome Research Institute (NGRI), Dhaka, Bangladesh for further analysis.
PCR amplification and gel electrophoresis
The 66 E. coli isolates were inoculated into LB broth (HiMedia, India), grown at 37 °C overnight, and DNA was extracted using GeneJET Genomic DNA Purification Kit (Cat. No K0721) (ThermoFisher Scientific, USA) according to manufacturer’s protocol. Conventional singleplex PCR was carried out to detect the NDM gene43, and to classify strains into phylogroups using previously described protocols (Table S2)26. In brief, 12.5 µl of reaction volume was used containing 6.25 µl DreamTaq Green PCR Master Mix (ThermoFisher Scientific, USA), 1.0 µl 25 nmol of MgCl2, 20 pmol of forward and reverse primers and ca. 100 ng DNA. Amplification was carried out using GeneAtlas (Astec Co, Ltd), with the following assay conditions: denaturation at 94 °C for 5 min; 30 cycles of 30 s at 94 °C, 30 s at annealing temperature, 30 s at 72 °C, and final extension at 72 °C for 5 min. Agarose gel electrophoresis was used to visualize banding patterns of the strains.
Genome assembly and annotation
Library preparation and sequencing of the 11 selected strains were conducted at NGRI. Ca. 1 μg of high molecular weight E. coli genomic DNA was used to prepare Illumina libraries and employing Nextera DNA Library Preparation Kit (Cat. No. FC-121-1030) according to manufacturer’s guideline. De novo assembly of good quality paired-end Illumina reads (Q ≥ 30) was done by running genome assembly software SPAdes (v3.12)44 with filters to decrease the number of mismatches and short indels. Assembled contigs were annotated using PROKKA pipeline45 with contiglength < 500 bp filtered out. Possible genomic contaminations were assessed using the ContEst16S tool46. Pan and core genome size of the 11 isolates and reference genome NA11447 were identified using the GF (Gene Family) method of pan-genome analysis pipeline (PGAP) (v1.2.1)48. Further pan and core genome analyses were performed using Roary49. Hierarchical clustering based on presence and absence of accessory genes, was performed using PVclust: R package50, based on bootstrap resampling to generate p-values. The bootstrap value was set to n = 1,000, to cross-validate the clustering pattern. The functions of the accessory genes were analysed via STRING database (https://string-db.org/).
Evolutionary relationship and phylogenomic analysis
Single Nucleotide Polymorphism (SNP) matrix was generated using CSIphylogeny 1.4 (Conserved Signature Indels) (Table S3)51. The 11 sequenced UPEC isolates from this study, 386 publicly available published genomes12,38, and a few reference strains of diverse STs and phylogroups, including NA11447, CFT07352, IAI3953, SE1154 and BW295255 (Table S4), were aligned to generate a core alignment in order to derive whole genome SNP using Parsnp v1.2 from the Harvest suite56. SNP file was processed by SNPRelate: R package57 and phylogenomic tree was visualized using FigTree (https://tree.bio.ed.ac.uk/software/figtree/).
In silico analysis of UPEC genome sequences
Phylogroups were confirmed based on presence of marker genes arpA, chuA, yjaA and TspE4.C226 using local BLAST the scheme set by Clermont et al. in 201258. The ST of each annotated genome was extracted from the MLST 2.0 (Multi-locus sequence typing) database59, and serotypes were determined using SerotypeFinder 2.060. Similarly, antimicrobial resistance (AMR) genes and plasmids of each isolate was obtained from ResFinder 3.161 and PlasmidFinder 2.062 respectively. Furthermore, a virulence factor profile was generated by amalgamation of results obtained using BLASTp against the Virulence Factors of Bacterial Pathogens database (VFDB)63 that had been made available in 2016, and the tool VirulenceFinder 2.051. SNPs per gene were calculated using DnaSP v664. To study the genetic composition of possible PAIs, genomes of the 11 UPEC isolates were analysed for presence of CFT073 and 536 associated PAI genes retrieved from the PAthogenisity Island DataBase (https://www.paidb.re.kr/)65 and visualized using CIRCOS66.
Phenotypic virulence determination
Alpha and beta haemolytic reactions of the strains were demonstrated using blood agar (Oxoid, UK), prepared using sheep blood. A single isolated colony for each strain was streaked on a blood agar plate which was incubated overnight at 37 °C. Partially clear and completely clear zones around the colony were indicative of alpha and beta hemolytic activity respectively.
Biofilm formation assays were performed using previously described protocols with minor modifications13. Bacteria were grown overnight in M63 at 37 °C after which 2 µl aliquots were added to 198 µl fresh M63 medium in a sterile 96-well polystyrene microtitre plate with four replicate wells for each strain. M63 broth without inoculum served as negative control. The plates were incubated statically at 37 °C for 24 h and OD600 was measured both at the beginning of incubation (0 h) and end of incubation at 24 h (GloMax, Promega). The culture was then discarded and plates were gently washed twice with sterile saline and air dried. Ca. 250 µl 0.1% crystal violet was added to the wells and allowed to stain for 15 min. The plates were then washed thrice with distilled water and air dried. The stained bacterial cells were resolubilized in 200 µl of 33% glacial acetic acid and the plates were read at 560 nm to enumerate cells in the biofilm. Specific biofilm formation (SBF) was measured using the formula SBF = (AB – CW)/G, where AB is OD560 of stained cells, CW is OD560 of control wells, and G is bacterial cell growth calculated using the formula G = OD600nm(24 h) – OD600nm(0 h). The strains were classified as follows: SBF < 0.5 = weak biofilm former, 0.5 ≤ SBF < 1.0 = moderate biofilm former and SBF ≥ 1.0 = strong biofilm former67. The entire assay was performed at least twice for each strain.
Assay for serum resistance was performed using a slightly modified version of previously described protocols13. Ca. 5 µl from overnight cultures was added to 495 µl fresh LB broth (HiMedia, India) and inoculated statically for 2 h at 37 °C. The culture was then centrifuged at 5,000×g for 7 min and the pellet obtained was suspended in 500 µl of sterile saline. Ca. 20 µl aliquots from this mixture were transferred to 180 µl of normal human serum (NHS) in a sterile 96-well microtitre plate and incubated at 37 °C under static conditions for 3 h. Ca. 20 µl was removed from the culture at 0 h and after 3 h incubation and plated on LB plates after serial dilution. Bacteria were enumerated after the plates had incubated overnight at 37 °C. Resistance to serum was measured as percentage change in colony forming units (CFU) at the beginning and end of the incubation period. The entire experiment was run in duplicate.
Ethical statements
All isolates included in this study were collected for diagnostic purposes from two local tertiary hospitals where pathogens are isolated from clinical specimens as part of a routine diagnostic procedure and not for experimental purposes. All experiments and methods were carried out in accordance with relevant guidelines and regulations. All experimental protocols were approved by the North South University (NSU) Institutional Review Board (IRB) / Ethical Review Committee (ERC), protocol No. CTRG:NSU-RP-18-042. Clinical isolates used in this study were recovered for diagnostic purposes from local diagnostic centers or hospitals and were not experimental in nature. The clinical data were anonymized and unlinked and the requirement for informed consent was waived by the NSU IRB/ERC.
Supplementary information
Author contributions
M.H.: conceptualization, supervision, computational analysis, writing, reviewing, editing; T.T.: computational and data analysis, writing, editing; A.R.: laboratory experiments, data analysis, writing, editing: Ar.H.: data collection, data analysis, writing; T.A.: computational analysis, A.M.I.M.: computational analysis, writing; A.S.: data collection; M.S.: laboratory experiments; F.S.: supervision, reviewing; A.I.: reviewing, editing; A.K.: supervision; M.A.: supervision; An.H.: supervision; G.U.A.: supervision; R.R.C.: supervision, writing, reviewing, and editing. All authors read and reviewed the final manuscript.
Data access
The 11 uropathogenic E. coli genome sequences and analysis from this study have been submitted to GenBank database, with accession numbers that include QEXN00000000 (NGE3), QFAZ00000000 (NGE4), RCIF00000000 (NGE5), RCIE00000000 (NGE6), QFRN00000000 (NGE7), QFRT00000000 (NGE9), QFTM00000000 (NGE16), QFXA00000000 (NGE22), RBWA00000000 (NGCE33), RAZR00000000 (NGCE94) and RAZQ00000000 (NGCE100).
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Tahmina Tabassum and Aura Rahman.
Supplementary information
is available for this paper at 10.1038/s41598-020-71213-5.
References
- 1.Foxman B. Epidemiology of urinary tract infections: Incidence, morbidity, and economic costs. Dis. Mon. 2003;49:53–70. doi: 10.1067/mda.2003.7. [DOI] [PubMed] [Google Scholar]
- 2.Foxman B, Barlow R, D’Arcy H, Gillespie B, Sobel JD. Urinary tract infection: self-reported incidence and associated costs. Ann. Epidemiol. 2000;10:509–515. doi: 10.1016/s1047-2797(00)00072-7. [DOI] [PubMed] [Google Scholar]
- 3.Sanjee SA, et al. Prevalence and antibiogram of bacterial uropathogens of urinary tract infections from a tertiary care hospital of Bangladesh. J. Sci. Res. 2017;9:317. [Google Scholar]
- 4.Johnson JR, Stell AL. Extended virulence genotypes of Escherichia coli strains from patients with urosepsis in relation to phylogeny and host compromise. J. Infect. Dis. 2000;181:261–272. doi: 10.1086/315217. [DOI] [PubMed] [Google Scholar]
- 5.Horner C, et al. Escherichiacoli bacteraemia: 2 years of prospective regional surveillance (2010–12) J. Antimicrob. Chemother. 2014;69:59–66. doi: 10.1093/jac/dkt333. [DOI] [PubMed] [Google Scholar]
- 6.Wirth T, et al. Sex and virulence in Escherichiacoli: an evolutionary perspective. Mol. Microbiol. 2006;60:1136–1151. doi: 10.1111/j.1365-2958.2006.05172.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sarowska J, et al. Virulence factors, prevalence and potential transmission of extraintestinal pathogenic Escherichiacoli isolated from different sources: recent reports. Gut Pathog. 2019;11:1–16. doi: 10.1186/s13099-019-0290-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khairy RM, Mohamed ES, Ghany HMA, Abdelrahim SS. Phylogenic classification and virulence genes profiles of uropathogenic E. coli and diarrhegenic E. coli strains isolated from community acquired infections. PLoS ONE. 2019 doi: 10.1371/journal.pone.0222441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shaik S, et al. Comparative genomic analysis of globally dominant ST131 clone with other epidemiologically successful extraintestinal pathogenic Escherichiacoli (ExPEC) lineages. MBio. 2017;8:e01596–e1617. doi: 10.1128/mBio.01596-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lau HL, et al. Major uropathogenic Escherichiacoli strain isolated in the Northwest of England identified by multilocus sequence typing. J. Clin. Microbiol. 2008;46:1076–1081. doi: 10.1128/JCM.02065-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aibinu I, Odugbemi T, Koenig W, Ghebremedhin B. Sequence type ST131 and ST10 complex (ST617) predominant among CTX-M-15-producing Escherichia coli isolates from Nigeria. Clin. Microbiol. Infect. 2012;18:E49–E51. doi: 10.1111/j.1469-0691.2011.03730.x. [DOI] [PubMed] [Google Scholar]
- 12.Petty NK, et al. Global dissemination of a multidrug resistant Escherichiacoli clone. Proc. Natl. Acad. Sci. 2014;111:5694–5699. doi: 10.1073/pnas.1322678111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nandanwar N, et al. Extraintestinal pathogenic Escherichiacoli (ExPEC) of human and avian origin belonging to sequence type complex 95 (STC95) portray indistinguishable virulence features. Int. J. Med. Microbiol. 2014;304:835–842. doi: 10.1016/j.ijmm.2014.06.009. [DOI] [PubMed] [Google Scholar]
- 14.Fair RJ, Tor Y. Antibiotics and bacterial resistance in the 21st century. Perspect. Med. Chem. 2014;6:25–64. doi: 10.4137/PMC.S14459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Islam MA, et al. Environmental spread of New Delhi metallo-β-lactamase-1-producing multidrug-resistant bacteria in Dhaka, Bangladesh. Appl. Environ. Microbiol. 2017;83:e00793–e817. doi: 10.1128/AEM.00793-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Peirano G, Schreckenberger PC, Pitout JDD. Characteristics of NDM-1-producing Escherichiacoli isolates that belong to the successful and virulent clone ST131. Antimicrob. Agents Chemother. 2011;55:2986–2988. doi: 10.1128/AAC.01763-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tada T, et al. NDM-8 metallo-βlactamase in a multidrug-resistant Escherichiacoli strain isolated in Nepal. Antimicrob. Agents Chemother. 2013;57:2394–2396. doi: 10.1128/AAC.02553-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Poirel L, Dortet L, Bernabeu S, Nordmann P. Genetic features of blaNDM-1-positive Enterobacteriaceae. Antimicrob. Agents Chemother. 2011;55:5403–5407. doi: 10.1128/AAC.00585-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hu H, et al. Novel plasmid and its variant harboring both a bla NDM-1 gene and type IV secretion system in clinical isolates of Acinetobacterlwoffii. Antimicrob. Agents Chemother. 2012;56:1698–1702. doi: 10.1128/AAC.06199-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kumarasamy KK, et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect. Dis. 2010;10:597–602. doi: 10.1016/S1473-3099(10)70143-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Poirel L, Revathi G, Bernabeu S, Nordmann P. Detection of NDM-1-producing Klebsiellapneumoniae in Kenya. Antimicrob. Agents Chemother. 2011;55:934–936. doi: 10.1128/AAC.01247-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dortet L, Poirel L, Nordmann P. Worldwide dissemination of the NDM-type carbapenemases in Gram-negative bacteria. Biomed. Res. Int. 2014;2014:249856. doi: 10.1155/2014/249856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zou D, et al. A novel New Delhi metallo-β-lactamase variant, NDM-14, isolated in a Chinese hospital possesses increased enzymatic activity against carbapenems. Antimicrob. Agents Chemother. 2015;59:2450–2453. doi: 10.1128/AAC.05168-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Falgenhauer L, et al. Circulation of clonal populations of fluoroquinolone-resistant CTX-M-15-producing Escherichiacoli ST410 in humans and animals in Germany. Int. J. Antimicrob. Agents. 2016;47:457–465. doi: 10.1016/j.ijantimicag.2016.03.019. [DOI] [PubMed] [Google Scholar]
- 25.Pitout JDD. Extraintestinal pathogenic Escherichiacoli: a combination of virulence with antibiotic resistance. Front. Microbiol. 2012 doi: 10.3389/fmicb.2012.00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Clermont O, Bonacorsi S, Bingen E. Rapid and simple determination of the Escherichiacoli phylogenetic group. Appl. Environ. Microbiol. 2000;66:4555–4558. doi: 10.1128/aem.66.10.4555-4558.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roer L, et al. Escherichiacoli sequence type 410 is causing new international high-risk clones. mSphere. 2018;3:e00337-18. doi: 10.1128/mSphere.00337-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Abd El Ghany M, Sharaf H, Al-agamy MH, Shibl A, Hong P. Genomic characterization of NDM-1 and 5, and OXA-181 carbapenemases in uropathogenic Escherichiacoli isolates from Riyadh, Saudi Arabia. PLoS ONE. 2018;13:e0201613. doi: 10.1371/journal.pone.0201613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kostakioti M, Hadjifrangiskou M, Hultgren SJ. Bacterial biofilms: development, dispersal, and therapeutic strategies in the dawn of the postantibiotic era. Cold Spring. Harb. Perspect. Med. 2013;3:a010306. doi: 10.1101/cshperspect.a010306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Putrins M, et al. Phenotypic heterogeneity enables uropathogenic Escherichiacoli to evade killing by antibiotics and serum complement. Infect. Immun. 2015;83:1056–1067. doi: 10.1128/IAI.02725-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Phan MD, et al. The serum resistome of a globally disseminated multidrug resistant uropathogenic Escherichiacoli clone. PLoS Genet. 2013;9:e1003834. doi: 10.1371/journal.pgen.1003834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mobley HL, et al. Pyelonephritogenic Escherichiacoli and killing of cultured human renal proximal tubular epithelial cells: role of hemolysin in some strains. Infect. Immun. 1990;58:1281–1289. doi: 10.1128/iai.58.5.1281-1289.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kao JS, Stucker DM, Warren JW, Mobley HLT. Pathogenicity island sequences of pyelonephritogenic Escherichiacoli CFT073 are associated with virulent uropathogenic strains. Infect. Immun. 1997;65:2812–2820. doi: 10.1128/iai.65.7.2812-2820.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sabate M, Moreno T, Perez T, Andreu A, Prats G. Pathogenicity island markers in commensal and uropathogenic Escherichiacoli. Clin. Microbiol. Infect. Dis. 2006;12:880–886. doi: 10.1111/j.1469-0691.2006.01461.x. [DOI] [PubMed] [Google Scholar]
- 35.Middendorf B, Hochhut B, Leipold K, Dobrindt U, Blum-oehler G. Instability of pathogenicity islands in uropathogenic Escherichiacoli 536. J. Bacteriol. 2004;186:3086–3096. doi: 10.1128/JB.186.10.3086-3096.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gordon DM, Clermont O, Tolley H, Denamur E. Assigning Escherichiacoli strains to phylogenetic groups: multi-locus sequence typing versus the PCR triplex method. Environ. Microbiol. 2008;10:2484–2496. doi: 10.1111/j.1462-2920.2008.01669.x. [DOI] [PubMed] [Google Scholar]
- 37.Ciccozzi M, et al. Phylogenetic analysis of multidrug-resistant Escherichiacoli clones isolated from humans and poultry. New Microbiol. 2013;36:385–394. [PubMed] [Google Scholar]
- 38.Salipante SJ, et al. Large-scale genomic sequencing of extraintestinal pathogenic Escherichiacoli strains. Genome Res. 2015;25:119–128. doi: 10.1101/gr.180190.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rasko DA, et al. The pangenome structure of Escherichiacoli: comparative genomic analysis of E. coli commensal and pathogenic isolates. J. Bacteriol. 2008;190:6881–6893. doi: 10.1128/JB.00619-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Madoshi BP, et al. Characterisation of commensal Escherichiacoli isolated from apparently healthy cattle and their attendants in Tanzania. PLoS ONE. 2016;11:e0168160. doi: 10.1371/journal.pone.0168160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vejborg RM, Hancock V, Schembri MA, Klemm P. Comparative genomics of Escherichia coli strains causing urinary tract infections. Appl. Environ. Microbiol. 2011;77:3268–3278. doi: 10.1128/AEM.02970-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cordoni G, et al. Comparative genomics of European avian pathogenic E. coli (APEC) BMC Genomics. 2016;17:960. doi: 10.1186/s12864-016-3289-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liang Z, et al. Molecular basis of NDM-1, a new antibiotic resistance determinant. PLoS ONE. 2011;6:e23606. doi: 10.1371/journal.pone.0023606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bankevich A, et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Seemann T. Prokka: rapid prokaryotic genome annotation. Bioinformatics. 2014;30:2068–2069. doi: 10.1093/bioinformatics/btu153. [DOI] [PubMed] [Google Scholar]
- 46.Lee I, et al. ContEst16S: an algorithm that identifies contaminated prokaryotic genomes using 16S RNA gene sequences. Int. J. Syst. Evol. Microbiol. 2017;67:2053–2057. doi: 10.1099/ijsem.0.001872. [DOI] [PubMed] [Google Scholar]
- 47.Avasthi TS, et al. Genome of multidrug-resistant uropathogenic Escherichiacoli strain NA114 from India. J. Bacteriol. 2011;193:4272–4273. doi: 10.1128/JB.05413-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhao Y, et al. PGAP: pan-genomes analysis pipeline. Bioinformatics. 2012;28:416–418. doi: 10.1093/bioinformatics/btr655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Page AJ, et al. Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics. 2015;31:3691–3693. doi: 10.1093/bioinformatics/btv421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Suzuki R, Shimodaira H. Pvclust: an R package for assessing the uncertainty in hierarchical clustering. Bioinformatics. 2006;22:1540–1542. doi: 10.1093/bioinformatics/btl117. [DOI] [PubMed] [Google Scholar]
- 51.Joensen KG, et al. Real-time whole-genome sequencing for routine typing, surveillance, and outbreak detection of verotoxigenic Escherichiacoli. J. Clin. Microbiol. 2014;52:1501–1510. doi: 10.1128/JCM.03617-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Welch RA, et al. Extensive mosaic structure revealed by the complete genome sequence of uropathogenic Escherichia coli. PNAS. 2002;99:17020–17024. doi: 10.1073/pnas.252529799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Touchon M, et al. Organised genome dynamics in the Escherichiacoli species results in highly diverse adaptive paths. PLoS Genet. 2009;5:e1000344. doi: 10.1371/journal.pgen.1000344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Oshima K, et al. Complete genome sequence and comparative analysis of the wild-type commensal Escherichiacoli strain SE11 isolated from a healthy adult. DNA Res. 2008;15:375–386. doi: 10.1093/dnares/dsn026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ferenci T, et al. Genomic sequencing reveals regulatory mutations and recombinational events in the widely used MC4100 lineage of Escherichiacoli K-12. J. Bacteriol. 2009;191:4025–4029. doi: 10.1128/JB.00118-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Treangen TJ, Ondov BD, Koren S, Phillippy AM. The Harvest suite for rapid core-genome alignment and visualization of thousands of intraspecific microbial genomes. Genome Biol. 2014;15:524. doi: 10.1186/s13059-014-0524-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zheng X, et al. A high-performance computing toolset for relatedness and principal component analysis of SNP data. Bioinformatics. 2012;28:3326–3328. doi: 10.1093/bioinformatics/bts606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Clermont O, Christenson JK, Denamur E, Gordon DM. The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ. Microbiol. Rep. 2013;5:58–65. doi: 10.1111/1758-2229.12019. [DOI] [PubMed] [Google Scholar]
- 59.Larsen MV, et al. Multilocus sequence typing of total-genome-sequenced bacteria. J. Clin. Microbiol. 2012;50:1355–1361. doi: 10.1128/JCM.06094-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Joensen KG, Tetzschner AMM, Iguchi A, Aarestrup FM, Scheutz F. Rapid and easy in silico serotyping of Escherichiacoli isolates by use of whole-genome sequencing data. J. Clin. Microbiol. 2015;53:2410–2426. doi: 10.1128/JCM.00008-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zankari E, et al. Identification of acquired antimicrobial resistance genes. J. Antimicrob. Chemother. 2012;67:2640–2644. doi: 10.1093/jac/dks261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Carattoli A, et al. In silico detection and typing of plasmids using plasmidfinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 2014;58:3895–3903. doi: 10.1128/AAC.02412-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chen L, et al. VFDB: a reference database for bacterial virulence factors. Nucleic Acids Res. 2005;33:D325–D328. doi: 10.1093/nar/gki008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rozas J, et al. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 2017;34:3299–3302. doi: 10.1093/molbev/msx248. [DOI] [PubMed] [Google Scholar]
- 65.Yoon SH, Park YK, Kim JF. PAIDB v2.0: exploration and analysis of pathogenicity and resistance islands. Nucleic Acids Res. 2015;43:D624–D630. doi: 10.1093/nar/gku985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Krzywinski M, et al. Circos: an information aesthetic for comparative genomics. Genome Res. 2009;19:1639–1645. doi: 10.1101/gr.092759.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Martinez-Medina M, et al. Biofilm formation as a novel phenotypic feature of adherent-invasive Escherichiacoli (AIEC) BMC Microbiol. 2009;9:202. doi: 10.1186/1471-2180-9-202. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The 11 uropathogenic E. coli genome sequences and analysis from this study have been submitted to GenBank database, with accession numbers that include QEXN00000000 (NGE3), QFAZ00000000 (NGE4), RCIF00000000 (NGE5), RCIE00000000 (NGE6), QFRN00000000 (NGE7), QFRT00000000 (NGE9), QFTM00000000 (NGE16), QFXA00000000 (NGE22), RBWA00000000 (NGCE33), RAZR00000000 (NGCE94) and RAZQ00000000 (NGCE100).