Abstract
Hyperthermia, or extreme elevations in body temperature, can be life-threatening and may be caused by prescription drugs or illegal substances acting at a number of different levels of the neuraxis. Several psychotropic drug classes and combinations have been associated with a classic clinical syndrome of hyperthermia, skeletal muscle hypermetabolism, rigidity or rhabdomyolysis, autonomic dysfunction and altered mental status ranging from catatonic stupor to coma. It is critical for clinicians to have a high index of suspicion for these relatively uncommon drug-induced adverse effects and to become familiar with their management to prevent serious morbidity and mortality. Although these syndromes look alike, they are triggered by quite different mechanisms, and apart from the need to withdraw or restore potential triggering drugs and provide intensive medical care, specific treatments may vary. Clinical similarities have led to theoretical speculations about common mechanisms and shared genetic predispositions underlying these syndromes, suggesting that there may be a common “thermic stress syndrome” triggered in humans and animal models by a variety of pharmacological or environmental challenges.
Keywords: Induced hyperthermia, Malignant hyperthermia, Neuroleptic malignant syndrome, Serotonin syndrome, Antipsychotic agents, Substance use disorders.
INTRODUCTION
Maintenance of normal body temperature, or thermoregulation, is essential to health [1]. Body temperature is one of the most tightly regulated vital functions. Thermo-regulation is a complex physiologic process involving multiple levels of the nervous system as well as other body organs and systems. As a result, there are many steps that can be affected by drugs or toxins which impair thermoregulation and lead to hyperthermia.
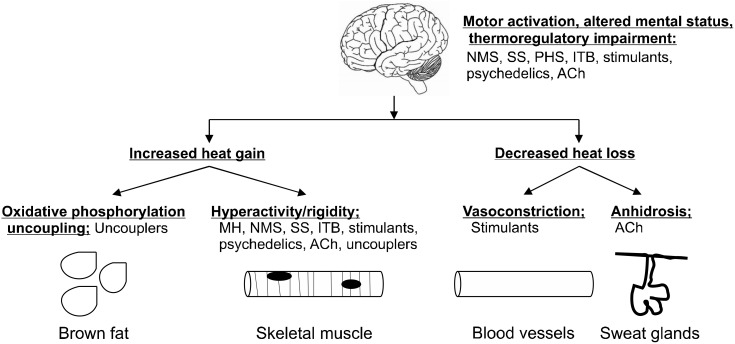
While it is important for clinicians to be familiar with all drugs that interfere with thermoregulation, one category consists of drugs that cause hyperthermia at least in part by increasing internal heat production in the absence of a hot environment (Table 1, Fig. 1) [2]. These syndromes, characterized by contraction and hypermetabolism of skeletal muscle, manifest elevated temperatures, muscle rigidity, rhabdomyolysis, elevated vital signs, and changes in mental status that closely resemble malignant hyperthermia of anesthesia (MH) [3 -6]. This clinical picture can be a medical emergency with risk of serious complications and death (Table 2).
Table 1.
Drug-induced hyperthermic syndromes
| Clinical Features | Malignant Hyperthermia | Neuroleptic Malignant Syndrome | Serotonin Syndrome | Parkinsonism Hyperpyrexia Syndrome | Baclofen Withdrawal Syndrome | Adrenergic Stimulation/ Psychedelics | Oxidative Phosphorylation Uncouplers | Anticholinergic Syndrome |
|---|---|---|---|---|---|---|---|---|
| Drug triggers | Inhalational anesthetics, succinylcho-line | Dopamine antagonists | Serotonergic agents; SSRI, SNRI or MAOI antidepressants, some opioids, triptans, antibiotics (linezolid), tryptophan, St. John’s wort, MDMA, methylene blue | Withdrawal or failure of dopaminergic drugs, loss of DBS stimulation | Withdrawal of baclofen, failure of intrathecal pump | Amphetamine, MDMA, cocaine, cathinones/ ketamine, phencyclidine, LSD, psilocybin, mescaline | Salicylates, 2,4-dintrophenol | Antiparkinsonian, antipsychotic, antidepressant, antihistaminic, or ocular drugs, over-the-counter and herbal products |
| Clinical signs | Hyperthemia, rigidity, autonomic dysfunction, tachypnea, rhabdomyoly-sis | Hyperthermia rigidity,tremor autonomic dysfunction, rhabdomyoly-sis, sweating, altered mental status | Mild: Agitated delirium Severe: Hyperthermia rigidity, clonus, autonomic dysfunction, sweating, altered mental status, GI symptoms |
Hyperthermia, rigidity, tremor, autonomic dysfunction, altered mental status | Hyperthermia, rigidity, tremor, autonomic dysfunction, altered mental status, seizures | Mild: Agitation, tachycardia, confusion, dilated and reactive pupils/ ataxia, hypertension, rigidity or agitation, sweating, catatonia Severe: Hyper- or hypothermia, rigidity, autonomic dysfunction, rhabdomyoly-sis, coma, seizures |
Mild: Hyperventilation, tinnitus, GI symptoms Severe: Hyperther-mia rigidity, autonomic dysfunction, rhabdomyoly-sis, altered mental status |
Mild: dry mouth, anhidrosis, dilated nonreactive pupils, tachycardia, hypertension, urinary retention, absent bowel sounds Severe: Hyperthermia, ataxia, agitation, hallucinations, delirium, seizures, coma |
| Laboratory studies | Hypercarbia, acidosis, hyperkalemia, elevated CK, in vitro contracture test, genetic testing (RYR1) | Acidosis, hypoxia, elevated CK, leukocytosis, EEG slowing | Acidosis, elevated CK | Elevated CK, leukocytosis | Elevated CK | Blood salicylate levels | Tricyclic antidepressant levels, EKG findings | |
| Frequency | 1:10,000 to 1: 250,000 anesthetics | 0.02−0.11% patients receiving antipsychotics | 0.09−0.23% patients receiving SSRI antidepressants | 0.3−3.6% PD patients receiving and withdrawn from levodopa | ||||
| Mortality | 1−5% | 5−20% | 5% | 15% | ||||
| Risk factors | Family history, prior episodes, certain myopathies, history of exertional heatstroke or rhabdomyloy-sis | Dehydration, exhaustion, agitation, catatonia, prior episodes, high doses or high potency antipsychotics | Older adults, overdose, drug combinations, (MDMA: “rave” dance parties) | Advanced PD, high daily levodopa doses, motor fluctuations, psychosis, dehydration, prior episodes | Intrathecal infusion pump, high level spinal cord injury, cerebral spasticity or dystonia | Substance use disorders, ambient temperature (MDMA: “rave” dance parties) | Overdose, weight loss regimens | Older adults, overdose, drug combinations |
| Treatment | Cessation of anesthesia, intensive care, intravenous dantrolene | Cessation of dopamine antagonists, intensive care, dopaminergic drugs, benzodiazepines, dantrolene, ECT | Cessation of serotonergic drugs, intensive care, cyproheptadine, benzodiazepines, dantrolene | Re-store dopaminergic drugs, intensive care, re-establish DBS stimulation | Re-store baclofen, intensive care, re-establish pump function, benzodiazepines | Cessation of adrenergic stimulants and psychedelics, intensive care | Cessation of oxidative phosporylation uncoupling drugs, intensive care, (salicylates: gastric lavage, alkalinize blood and urine, hemodialysis) | Cessation of anticholinergic drugs, physostigmine for severe cases (risk of cholinergic crisis, seizures, heart block in tricyclic antidepressant overdose) |
SSRI, selective serotonin reuptake inhibitor; SNRI, serotonin norepinephrine reuptake inhibitor; MAOI, monoamine oxidase inhibitor; MDMA, 3,4-methylenedioxymethamphetamine; DBS, deep brain stimulation; LSD, lysergic acid diethylamide; GI, gastrointestinal; CK, creatine kinase; EEG, electroencephalogram; EKG, electrocardiogram; ECT, electroconvulsive therapy; PD, Parkinson’s disease; RYR1, ryanodine receptor-1.
Fig. 1.
Pathophysiology underlying drug-induced hyperthermic syndromes. MH, malignant hyperthermia; NMS, neuroleptic malignant syndrome; SS, serotonin syndrome; PHS, parkinsonism-hyperpyrexia syndrome; ITB, intrathecal baclofen withdrawal syndrome; ACh, anticholinergic syndrome; Uncouplers, drugs that uncouple oxidative phosphorylation.
Table 2.
Practice points for managing drug-induced hyperthermic syndromes
| 1.Drug-induced hyperthermic syndromes should be considered in the differential diagnosis of all patients presenting with elevated temperatures 2.It is important to rule out other infectious, metabolic, neuropsychiatric, or environmental causes of elevated temperature 3.Increased heat production indicated by the four cardinal signs of extreme hyperthermia, muscle rigidity, autonomic changes and altered mental status often suggests drug-induced hyperthermia 4.Decreased heat loss indicated by signs of delirium, dry mouth and skin, urinary retention, and decreased bowel sounds, suggests the anticholinergic syndrome 5.Withdrawing or restoring the implicated triggering drugs and intensive supportive measures are the mainstay of treatment for all drug-induced hyperthermic syndromes 6.Specific treatments for several drug-induced hyperthermic syndromes have been studied (MH, dantrolene; NMS, benzodiazepines, dopamine agonists, dantrolene, ECT; SS, cyproheptadine; ACh, physostigmine) |
MH, malignant hyperthermia; NMS, neuroleptic malignant syndrome; ECT, electroconvulsive therapy; SS, serotonin syndrome; ACh, anticholinergic syndrome.
While these syndromes frequently present in emergency units, they also may be encountered and should be considered during consultations in psychiatric and medical hospital settings. For example, polypharmacy is common, especially among the elderly, such that patients receiving neuropsychiatric treatment who develop hyperthermia often have been taking multiple potential triggering drugs. The association of several drugs (e.g., certain antiemetics, antibiotics, herbal remedies) with risk of hyperthermic reactions is not well recognized. Finally, since the management of these syndromes may vary in preventing serious adverse outcomes, the differential diagnosis is important.
This overview describes a selection of drug-induced hyperthermic syndromes that consulting psychiatrists may encounter in daily practice. While these syndromes primarily reflect extreme hyperthermia due to drug-induced increases in internal heat production, the anticholinergic syndrome which results in hyperthermia mostly by inhibiting peripheral heat loss is also covered for completeness. Other medical disorders in the extensive differential diagnosis of extreme hyperthermia (e.g., exertional heatstroke, encephalitis, sepsis, etc.) that are not drug-induced are equally important but beyond the scope of this review. In addition, there are a number of theoretical speculations about common mechanisms and shared genetic predispositions underlying these syndromes, which suggest that there may be a common “thermic stress syndrome” that can be triggered in humans and animal models by a variety of pharmacological or environmental challenges (Fig. 1) [7 -9]. Although this concept is intriguing in predicting risk and understanding the pathophysiology of these disorders, it is also beyond the objective of this review which focuses instead on the practical clinical recognition and management of these drug-induced syndromes.
HYPERTHERMIC SYNDROMES
Malignant Hyperthermia of Anesthesia
MH is a pharmacogenetic disorder of skeletal muscle that presents as a hypermetabolic response to potent inhalational volatile agents and the depolarizing muscle relaxant succinylcholine [10,11]. While it is encountered primarily during anesthesia, MH-susceptibility is increasingly recognized as predisposing to hyperthermic episodes due to heat or other stresses outside the operating theater. As such, the MH syndrome serves as the prototype of acute drug-induced hyperthermia and has received the most advanced research on the underlying pathophysiology and genetic mechanisms. Practically, in post-operative and critical care settings, psychiatrists may be consulted to differentiate MH from other drug-induced syndromes discussed below and therefore should be familiar with its clinical features.
Standardized diagnostic criteria of MH include hyperthermia, tachycardia, tachypnea, increased carbon dioxide production, increased oxygen consumption, mixed metabolic and respiratory acidosis, hyperkalemia, muscle rigidity, and rhabdomyolysis [12]. The estimated inci-dence of MH reactions ranges from 1:10,000 to 1: 250,000 anesthetics. The diagnosis can be confirmed by an in vitro skeletal muscle contracture test. In humans, MH-susceptibility is inherited in an autosomal dominant pattern and has been mapped in up to 50−70% or more of cases to variants in the ryanodine receptor-1 gene (RYR1) on chromosome 19q13.1 affecting calcium sequestration in the sarcoplasmic reticulum.
Treatment of MH episodes with intravenous dantrolene to inhibit calcium release into muscle cytoplasm has resulted in reduction of the expected mortality due to MH from > 70−80% to as low as 1.4−5% in North America [11].
Neuroleptic Malignant Syndrome
Estimates of the incidence of Neuroleptic Malignant Syndrome (NMS) range from 0.02−0.11% of patients treated with dopamine blocking agents, primarily antipsychotics [13 -15]. Haloperidol, used frequently for sedation in critical care, is associated with NMS in nearly half of reported cases. Although the incidence and mortality of NMS may have diminished with the introduction of newer less potent antipsychotics, it remains potentially lethal if neglected. NMS also results from treatment with dopamine blocking drugs used to prevent nausea (metoclopramide, prochlorperazine) [16], and rarely dopamine depleting drugs (tetrabenazine, valbenazine, deutetrabenazine) [17].
Risk factors for NMS include dehydration, exhaustion, agitation, catatonia, previous NMS episodes, and rapidly increasing doses of high potency antipsychotics [18]. Recent case reports suggest that patients with underlying encephalitis may be at risk for NMS when treated with antipsychotics [19,20]. NMS may develop within hours, but usually evolves over a few days. Most cases occur within 1−2 weeks after drugs are started. Standardized diagnostic criteria have been validated and include elevated temperatures, rigidity, tremors, altered consciousness, and changes in vital signs [21,22]. Laboratory abnormalities, including muscle enzyme elevations (creatine kinase), acidosis and hypoxia, are common. No single test confirms the diagnosis, but thorough evaluation is essential to exclude other medical conditions. While elevated serum creatine kinase elevations are observed with antipsychotics and in cases of NMS, they are non-specific and not pathognomonic [23,24]. While antipsychotics and other psychotropics may also contribute to heatstroke in hot environments by inhibiting central and peripheral heat loss mechanisms [25], NMS can be distinguished from heatstroke by the presence of rigidity and stupor as opposed to flaccidity and delirium. In addition, hyperthermia in NMS is derived from an internally generated heat load and occurs independent of ambient temperatures.
Evidence strongly implicates drug-induced blockade of brain dopamine receptors as the primary triggering mechanism as opposed to MH which is triggered primarily in skeletal muscle (Fig. 1). Other evidence indicates that NMS and MH are different disorders; muscle contracture tests in patients with NMS have been negative for MH-susceptibility; case reports of RYR1 pathogenic variants in NMS patients are inconclusive; NMS patients and family members have had anesthesia without adverse effects; NMS but not MH symptoms are reversed by peripheral-acting skeletal muscle relaxants; NMS has never been reported to occur intraoperatively [26].
Once antipsychotics are withdrawn, most NMS cases resolve within 1−2 weeks, although patients may have prolonged symptoms if long-acting antipsychotics were received. A few patients develop a prolonged residual state that may respond to electroconvulsive therapy (ECT). The primary treatment of NMS consists of early diagnosis, discontinuing neuroleptics and supportive medical care. In the absence of randomized controlled trials, benzodiazepines, dopamine agonists, dantrolene and ECT have been used empirically with varying success.
For additional information, the Neuroleptic Malignant Syndrome Information Service, a subsidiary of the Malignant Hyperthermia Association of the United States, offers additional resources (www.nmsis.org).
Parkinsonism-Hyperpyrexia Syndrome
Patients with Parkinson’s disease (PD) and related disorders (e,g., Lewy body dementia,) are at risk for severe fluctuations of motor symptoms. Although rigidity, tremors and bradykinesia are effectively controlled by dopaminergic drugs, patients are sensitive to dosing changes. In particular, patients may develop a Parkinsonism- Hyperpyrexia Syndrome (PHS) indistinguishable from NMS and MH following abrupt decreases in dopaminergic medication [27]. Dopamine depletion correlates with risk of developing PHS based on findings of lower homovanillic acid levels in spinal fluid and reduced striatal dopamine transporter binding on single-photon emission computerized tomography imaging in susceptible patients [27 -29].
PHS is reported in the context of non-compliance with dopaminergic medications, during severe “off” periods, concurrent illness, dysphagia or changes in diet, and withholding of medications prior to medical or surgical procedures [30,31]. For example, PHS may be observed after surgery in patients with PD instructed to stop dopamine medications prior to surgery and not restarted promptly post-operatively. PHS may also emerge in PD patients after implantation of electrodes for deep brain stimulation (DBS), due to reduction of dopaminergic drugs or sudden loss of DBS stimulation (battery failure, explantation) [32,33]. Reports suggest PHS may occur in 0.3−3.6% of PD patients treated with and withdrawn from levodopa, and several deaths have been reported. Symptoms develop from several hours to a few days after drug discontinuation.
Apart from intensive medical and nursing care, restarting dopamine treatment is vital. However, prevention is most important and abrupt cessation of dopaminergic medication should be avoided if possible.
Serotonin Syndrome
It is essential that clinicians obtain an accurate drug history in assessing risk for Serotonin Syndrome (SS). Serotonergic drugs are frequently administered to patients with other serotonergic drugs already in their system raising the risk of toxicity [4,34]. SS generally results from drug combinations, but also occurs following overdoses and during single drug exposure [35,36]. The drugs most often associated with SS include antidepressants inhibiting serotonin reuptake [35]. Antidepressants that act as monoamine oxidase inhibitors (MAOIs) are particularly dangerous when used in drug combinations. Other drugs and herbal products are less recognized as MAOIs (methylene blue, linezolid, St. John’s wort) but are equally high risk for SS [37,38]. Opioid analgesics implicated in SS include fentanyl, tramadol, meperidine, oxycodone, dextromethorphan, and methadone [34]. While an US Food and Drug Administration advisory against co-prescribing triptan antimigraine drugs with antidepressants has been challenged, their risk of SS with MAOIs is unclear [39]. Although the incidence of SS is unknown, the risk among patients on serotonin reuptake inhibitor antidepressants has been estimated in the range of 0.09−0.23%, and up to 14−16% after overdose [40].
The onset of symptoms is usually abrupt within hours [40]. Standardized symptom criteria for serotonin toxicity are dose-related and include altered mental status (agitation, delirium), neuromuscular excitation (rigidity, tremor, hyperreflexia, clonus), and autonomic dysfunction (tachycardia, hypo/hypertension, and hyperthermia) [4,35,40 -44]. Gastrointestinal symptoms may occur. Most often, patients with SS display agitation and confusion with elevated vital signs and gastrointestinal symptoms, but severe cases appear as a hyperthermic syndrome identical to NMS and MH.
Although the mechanisms are obscured by the complexity of thermoregulation, evidence implicates the serotonin 5HT-2a receptor as a primary target [4,40]. Most affected patients recover within 24 hours after drug discontinuation and initiation of supportive therapy [4,40]. Some cases benefit from cyproheptadine, a 5HT-2a blocker, while other drugs (benzodiazepines, dantrolene) may provide symptomatic relief (4,40,45). Although antipsychotics have been suggested as treatment, the risk of NMS and propensity to compromise thermoregulation and autonomic function confound any benefit.
Baclofen Withdrawal Syndrome
Baclofen is a gamma-aminobutyric acid (GABAb) agonist used orally or intrathecally to treat severe muscle spasm in patients with spinal cord injury, cerebral palsy, traumatic brain injury, and multiple sclerosis. Chronic spinal infusion of baclofen downregulates the inhibitory effects of GABAb receptor activity, such that abrupt withdrawal results in rebound excitation within 1−3 days and an intrathecal baclofen (ITB) withdrawal syndrome that is indistinguishable from MH and NMS [4,46]. Episodes consisting of hyperthermia, muscle rigidity, mental status and autonomic changes, respiratory distress, muscle breakdown and coagulopathies have been reported in patients withdrawn from either oral or intrathecal baclofen. ITB withdrawal may occur after mechanical malfunction of baclofen pumps or catheters, delayed refill or after removal.
Besides aggressive supportive care, definitive treatment is the restoration of baclofen administration by the same route and dosage. If enteral or intrathecal administration cannot be promptly restored, GABAα-mediated neuronal activation by parenteral benzodiazepines may be lifesaving [46].
Adrenergic Stimulation Syndrome
Drugs with adrenergic stimulating properties including amphetamines, cocaine (“crack”), 3,4-methylenedioxymethamphetamine (MDMA or “ecstasy”), other methamphetamine analogs (“ice”) and cathinones (“bath salts”) continue to be synthesized, sold and abused [3]. The clinical presentation of sympathomimetic intoxication consists of sweating, hypertension, tachycardia, dilated and reactive pupils, agitation, delirium, psychosis, tremors, rhabdomyolysis and hyperthermia which can progress to arrhythmias, shock, renal failure, myocardial infarction, stroke, coagulation disorders and death [47]. Hyperthermia and rhabdomyolysis result from impaired heat dissipation and ischemia due to vasoconstriction, compounded by increased heat production from agitation or seizures. While agitation and confusion are typical, some patients develop NMS or MH-like syndromes with muscle rigidity and stupor. Hyperthermia and agitation are treated with benzodiazepines, physical cooling measures, and paralysis if needed.
While MDMA has only one tenth the adrenergic stimulant effect of amphetamine, toxicity has also been attributed to serotonin toxicity leading to SS, as MDMA releases and inhibits reuptake of serotonin [47]. Hyperthermic deaths attributable to MDMA used as a “club” drug have been associated with hot settings during “rave” dance parties, implying that stimulant-driven exertional heatstroke may also play a role in its toxicity [48]. Although treatment is generally supportive, dantrolene may improve survival in some patients with extreme hyperthermia from MDMA consistent with preliminary evidence linking it to MH-susceptibility [49 -52].
Psychedelic Drug Toxicity
Synthetic analogs of the anesthetic ketamine (“special K”), including phencyclidine (PCP, “angel dust”), continue to be abused with serious depressant, stimulant, hallucinogenic, and analgesic toxicity. Signs of intoxication are dose-related and may resemble NMS and MH. At low doses, patients may show ataxia, nystagmus, slurred speech, numbness of the extremities, hypertension, sweating, muscle rigidity, agitation or signs of catatonia or a “disso-ciative state”. At high doses, anesthesia, agitation, stupor, or coma may appear, accompanied by tachycardia, hypertension, sialorrhea, sweating, hyper- or hyporthermia, rigidity, and convulsions 72 to 96 hours after drug ingestion [53 -55]. Rhabdomyolysis progressing to renal failure is a common and serious complication of PCP intoxication [56].
Lysergic acid diethylamide (LSD) and other synthetic “designer” psychedelic drugs with serotonergic properties (phenethylamine analogs or “N-Bombs”, mescaline), may produce hyperthermia along with other effects on the autonomic nervous system [57 -60]. Patients typically experience hallucinations, agitation and tachycardia, but occasionally may develop a more severe syndrome of psychosis, catatonic stupor, hyperactivity or rigidity, rhabdomyolysis, and sympathetic activation leading to hyperthermia, coagulopathies, respiratory arrest, and coma. Although hyperthermia in these cases may reflect extreme exertion, it also underscores the potential significance of serotoninergic mechanisms, specifically 5HT-2a receptor activation, underlying adverse effects [61].
Oxidative Phosphorylation Uncoupling Agents
The process of oxidative phosphorylation in the mitochondria is essential for the production of energy-rich ATP. When this process is uncoupled from ATP production, energy is released as heat instead, causing potentially extreme increases in body temperature [3]. Salicylates, which are included in a number of marketed analgesics and anti-inflammatory products, remain a common cause of poisoning, and produce hyperthermia by uncoupling mitochondrial oxidative phosphorylation. Although patients with salicylate poisoning usually present initially with hyperventilation, tinnitus and gastrointestinal irritation, severe cases may resemble MH with hyperthermia, rhabdomyolysis, abnormal vital signs, respiratory alkalosis, metabolic acidosis, tachypnea, altered mental status, and abdominal complaints. Mild hyperthermia is common, but severe hyperthermia has been reported [62]. This can lead to pulmonary and cerebral edema, hypotension, seizures, hypoxia, hypoglycemia, and cardiopulmonary arrest. The diagnosis can be confirmed by the level of salicylates in the blood. Treatment consists of intensive care, gastric lavage, alkalinization of the serum and urine, and hemodialysis if necessary to remove remaining salicylates.
Another toxin that causes hyperthermia by uncoupling mitochondrial oxidative phosphorylation is 2,4-dinitro-phenol. Dinitrophenol has been used in chemical manufacturing but became popular for weight loss in the 1930s. Although recognized as causing serious toxicity and banned from distribution, it has again become available through unregulated mail-order websites with resulting increases in reported cases of poisoning. Dintrophenol can cause an MH-like syndrome with severe hyperthermia, rigidity, rhabdomyolysis, acidosis, and abnormal vital signs leading to multiple organ failure and death [63,64]. In addition to intensive care, dantrolene has been recommended as a treatment though evidence is conflicting [65,66].
Anticholinergic Syndrome
Although the previous syndromes share in common hyperthermia caused by induction of internal heat production in muscles from hyperactivity, agitation, seizures or rigidity, drugs with anticholinergic activity facilitate hyperthermia primarily as a result of peripheral blockade of acetylcholine-mediated muscarinic receptors in sweat glands producing anhidrosis and impaired heat dissipation (Fig. 1) [1,2,6,67]. Restlessness, agitation, and seizures may also occur and further increase temperatures. Implicated drugs include antipsychotics, antidepressants, antiparkinsonian drugs, antispasmodics, ocular medications and antihistaminic drugs often prescribed in combinations, although drugs vary considerably in anticholinergic potency within classes. Numerous other plants and mushrooms consumed as herbal products may also have anticholinergic properties. Therapeutic doses of drugs with anticholinergic properties are not likely to significantly alter temperature, at least in healthy subjects in thermally neutral environments, but hyperthermia may occur following overdose. Once sweating ceases due to even therapeutic doses of anticholinergic drugs, body temperature may rise precipitously in a hot environment [68].
Intoxication with anticholinergic drugs results in a well-described syndrome of atropinic poisoning characterized by central and peripheral signs (e.g., dry mouth, flushed dry skin, dilated and nonreactive pupils, blurred vision, tachycardia, hypertension, urinary retention, and intestinal paralysis), which may progress to ataxia, hyperactivity, agitation, increased muscle tone, hallucinations, delirium, seizures and coma [3,69,70]. Hyperthermia due to anticholinergic intoxication is usually mild and unlikely to be mistaken for NMS because of the pronounced signs of anticholinergic toxicity in the periphery and the uncommon appearance of severe temperature elevations, rhabdomyolysis, or rigidity.
Apart from supportive care and sedation, physostig-mine, a reversible inhibitor of acetylcholinesterase that counteracts the effects of anticholinergics, has been studied as a specific antidote [3]. However, physostigmine should be reserved for severe agitation, tachycardia with hemodynamic instability, and extreme hyperthermia with impaired sweating because it may overcompensate for anticholinergic blockade and cause a paradoxical cholinergic crisis, including seizures and heart block, especially following tricyclic antidepressant overdose.
CONCLUSIONS
Although the differential diagnosis of hyperthermia is extensive, several drugs can induce a malignant hyperthermia-like syndrome. It is important for clinicians to recognize these syndromes and institute treatment quickly to prevent morbidity and mortality. Although these syndromes often look alike, they are triggered by quite different mechanisms, and apart from the need to withdraw or restore potential triggering drugs and provide intensive care, specific treatments may vary. It is important to know the toxicity of these drugs, especially when used in combinations, or during exertion in hot environments. Clinicians should be also be aware of dangers associated with the use of illegal and herbal substances obtained over the internet, which is unfortunately increasingly common.
Stanley N. Caroff served as consultant to Neurocrine Biosciences, Teva Pharmaceuticals, and received research grants from Neurocrine Biosciences, Osmotica Pharmaceuticals and Eagle Pharmaceuticals. Charles B. Watson and Henry Rosenberg report no conflicts or disclosures.
Conceptualization: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg. Data acquisition: Stanley N. Caroff, Charles B, Henry Rosenberg. Watson. Formal analysis: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg. Supervision: Stanley N. Caroff. Writing—original draft: Stanley N. Caroff. Writing—review & editing: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg.
Footnotes
Conflicts of Interest
Stanley N. Caroff served as consultant to Neurocrine Biosciences, Teva Pharmaceuticals, and received research grants from Neurocrine Biosciences, Osmotica Pharmaceuticals and Eagle Pharmaceuticals. Charles B. Watson and Henry Rosenberg report no conflicts or disclosures.
Author Contributions
Conceptualization: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg. Data acquisition: Stanley N. Caroff, Charles B, Henry Rosenberg. Watson. Formal analysis: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg. Supervision: Stanley N. Caroff. Writing—original draft: Stanley N. Caroff. Writing—review & editing: Stanley N. Caroff, Charles B. Watson, Henry Rosenberg.
References
- 1.Knochel JP, Reed G. Disorders of heat regulation. In: Narins RG, editor. Clinical disorders of fluid and electrolyte metabolism. 5th ed. McGraw-Hill; New York: 1994. [Google Scholar]
- 2.Clark WG, Lipton JM. Drug-related heatstroke. Pharmacol Ther. 1984;26:345–388. doi: 10.1016/0163-7258(84)90040-8. [DOI] [PubMed] [Google Scholar]
- 3.Hayes BD, Martinez JP, Barrueto F., Jr Drug-induced hyperthermic syndromes: part I. Hyperthermia in overdose. Emerg Med Clin North Am. 2013;31:1019–1033. doi: 10.1016/j.emc.2013.07.004. [DOI] [PubMed] [Google Scholar]
- 4.McAllen KJ, Schwartz DR. Adverse drug reactions resulting in hyperthermia in the intensive care unit. Crit Care Med. 2010;38(6 Suppl):S244–S252. doi: 10.1097/CCM.0b013e3181dda0d4. [DOI] [PubMed] [Google Scholar]
- 5.Rusyniak DE, Sprague JE. Toxin-induced hyperthermic synd-romes. Med Clin North Am. 2005;89:1277–1296. doi: 10.1016/j.mcna.2005.06.002. [DOI] [PubMed] [Google Scholar]
- 6.Caroff SN. Hyperthermia associated with other neuropsychiatric drugs. In: Mann SC, Caroff SN, Keck SPEN, Lazarus A, editors. Neuroleptic malignant syndrome and related conditions. 2nd ed. American Psychiatric Publishing, Inc.; Washington, D.C.: 2003. pp. 93–120. [Google Scholar]
- 7.Zhao X, Song Q, Gao Y. Hypothesis: exertional heat stroke-induced myopathy and genetically inherited malignant hyperthermia represent the same disorder, the human stress syndrome. Cell Biochem Biophys. 2014;70:1325–1329. doi: 10.1007/s12013-014-0059-5. [DOI] [PubMed] [Google Scholar]
- 8.Meyers EF, Meyers RW. Thermic stress syndrome. JAMA. 1982;247:2098–2099. [PubMed] [Google Scholar]
- 9.Breed D, Meyer LCR, Steyl JCA, Goddard A, Burroughs R, Kohn TA. Conserving wildlife in a changing world: understanding capture myopathy-a malignant outcome of stress during capture and translocation. Conserv Physiol. 2019;7:coz027. doi: 10.1093/conphys/coz027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Riazi S, Kraeva N, Hopkins PM. Updated guide for the management of malignant hyperthermia. Can J Anaesth. 2018;65:709–721. doi: 10.1007/s12630-018-1108-0. [DOI] [PubMed] [Google Scholar]
- 11.Rosenberg H, Pollock N, Schiemann A, Bulger T, Stowell K. Malignant hyperthermia: a review. Orphanet J Rare Dis. 2015;10:93. doi: 10.1186/s13023-015-0310-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Larach MG, Localio AR, Allen GC, Denborough MA, Ellis FR, Gronert GA, et al. A clinical grading scale to predict malignant hyperthermia susceptibility. Anesthesiology. 1994;80:771–779. doi: 10.1097/00000542-199404000-00008. [DOI] [PubMed] [Google Scholar]
- 13.Caroff SN, Mann SC. Neuroleptic malignant syndrome. Med Clin North Am. 1993;77:185–202. doi: 10.1016/s0025-7125(16)30278-4. [DOI] [PubMed] [Google Scholar]
- 14.Strawn JR, Keck PE, Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164:870–876. doi: 10.1176/ajp.2007.164.6.870. [DOI] [PubMed] [Google Scholar]
- 15.Lao KSJ, Zhao J, Blais JE, Lam L, Wong ICK, Besag FMC, et al. Antipsychotics and risk of neuroleptic malignant syndrome: a population-based cohort and case-crossover study. CNS Drugs. 2020;34:1165–1175. doi: 10.1007/s40263-020-00767-9. [DOI] [PubMed] [Google Scholar]
- 16.Caroff SN, Rosenberg H, Mann SC, Campbell EC, Gliatto MF, Sullivan KA. Neuroleptic malignant syndrome in the perioperative setting. Am J Anesthesiol. 2001;28:387–393. [Google Scholar]
- 17.Caroff SN. Risk of neuroleptic malignant syndrome with vesicular monoamine transporter inhibitors. Clin Psychopharmacol Neurosci. 2020;18:322–326. doi: 10.9758/cpn.2020.18.2.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Su YP, Chang CK, Hayes RD, Harrison S, Lee W, Broadbent M, et al. Retrospective chart review on exposure to psychotropic medications associated with neuroleptic malignant syndrome. Acta Psychiatr Scand. 2014;130:52–60. doi: 10.1111/acps.12222. [DOI] [PubMed] [Google Scholar]
- 19.Schumacher LT, Mann AP, MacKenzie JG. Agitation management in pediatric males with anti-N-methyl-D-aspartate receptor encephalitis. J Child Adolesc Psychopharmacol. 2016;26:939–943. doi: 10.1089/cap.2016.0102. [DOI] [PubMed] [Google Scholar]
- 20.Caroff SN. Phenomenology and management of encephalitis. J Neuropsychiatry Clin Neurosci. 2019;31:399. doi: 10.1176/appi.neuropsych.18120392. [DOI] [PubMed] [Google Scholar]
- 21.Gurrera RJ, Caroff SN, Cohen A, Carroll BT, DeRoos F, Francis A, et al. An international consensus study of neuroleptic malignant syndrome diagnostic criteria using the Delphi method. J Clin Psychiatry. 2011;72:1222–1228. doi: 10.4088/JCP.10m06438. [DOI] [PubMed] [Google Scholar]
- 22.Gurrera RJ, Mortillaro G, Velamoor V, Caroff SN. A validation study of the international consensus diagnostic criteria for neuroleptic malignant syndrome. J Clin Psychopharmacol. 2017;37:67–71. doi: 10.1097/JCP.0000000000000640. [DOI] [PubMed] [Google Scholar]
- 23.Nisijima K, Shioda K. Temporal changes in serum creatine kinase concentration and degree of muscle rigidity in 24 patients with neuroleptic malignant syndrome. Neuropsychiatr Dis Treat. 2013;9:853–859. doi: 10.2147/NDT.S45084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Masi G, Milone A, Viglione V, Mancini A, Pisano S. Massive asymptomatic creatine kinase elevation in youth during antipsychotic drug treatment: case reports and critical review of the literature. J Child Adolesc Psychopharmacol. 2014;24:536–542. doi: 10.1089/cap.2014.0047. [DOI] [PubMed] [Google Scholar]
- 25.Mann SC. Thermoregulatory mechanisms and antipsychotic drug-related heatstroke. In: Mann SC, Caroff SN, Keck PE Jr., Lazarus A, editors. Neuroleptic Malignant Syndrome and Related Conditions. Second Edition ed. American Psychiatric Publishing, Inc.; Washington, D.C.: 2003. pp. 45–74. [Google Scholar]
- 26.Caroff SN. Neuroleptic malignant syndrome. In: Mann SC, Caroff SN, Keck PE Jr., Lazarus A, editors. Neuroleptic malignant syndrome and related conditions. 2nd ed. American Psychiatric Publishing, Inc.; Washington, D.C.: 2003. pp. 1–44. [Google Scholar]
- 27.Huddleston DE, Factor SA. Parkinsonism-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht SJ, editor. Movement disorder emergencies: diagnosis and treatment. 2nd ed. Humana Press; New York: 2013. pp. 29–42. [Google Scholar]
- 28.Martino G, Capasso M, Nasuti M, Bonanni L, Onofrj M, Thomas A. Dopamine transporter single-photon emission computerized tomography supports diagnosis of akinetic crisis of Parkinsonism and of neuroleptic malignant syndrome. Medicine (Baltimore) 2015;94:e649. doi: 10.1097/MD.0000000000000649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kaasinen V, Joutsa J, Noponen T, Päivärinta M. Akinetic crisis in Parkinson’s disease is associated with a severe loss of striatal dopamine transporter function: a report of two cases. Case Rep Neurol. 2014;6:275–280. doi: 10.1159/000369448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fujioka S, Fukae J, Ogura H, Mishima T, Yanamoto S, Higuchi MA, et al. Hospital-based study on emergency admission of patients with Parkinson’s disease. eNeurologicalSci. 2016;4:19–21. doi: 10.1016/j.ensci.2016.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alty J, Robson J, Duggan-Carter P, Jamieson S. What to do when people with Parkinson’s disease cannot take their usual oral medications. Pract Neurol. 2016;16:122–128. doi: 10.1136/practneurol-2015-001267. [DOI] [PubMed] [Google Scholar]
- 32.Artusi CA, Merola A, Espay AJ, Zibetti M, Romagnolo A, Lanotte M, et al. Parkinsonism-hyperpyrexia syndrome and deep brain stimulation. J Neurol. 2015;262:2780–2782. doi: 10.1007/s00415-015-7956-4. [DOI] [PubMed] [Google Scholar]
- 33.Caroff SN. Parkinsonism-hyperthermia syndrome and deep brain stimulation. Can J Anaesth. 2017;64:675–676. doi: 10.1007/s12630-017-0837-9. [DOI] [PubMed] [Google Scholar]
- 34.Baldo BA, Rose MA. The anaesthetist, opioid analgesic drugs, and serotonin toxicity: a mechanistic and clinical review. Br J Anaesth. 2020;124:44–62. doi: 10.1016/j.bja.2019.08.010. [DOI] [PubMed] [Google Scholar]
- 35.Abadie D, Rousseau V, Logerot S, Cottin J, Montastruc JL, Montastruc F. Serotonin syndrome: analysis of cases registered in the French pharmacovigilance database. J Clin Psychopharmacol. 2015;35:382–388. doi: 10.1097/JCP.0000000000000344. [DOI] [PubMed] [Google Scholar]
- 36.Direk MÇ, Yıldırım V, Güneş S, Bozlu G, Okuyaz Ç. Serotonin syndrome after clomipramine overdose in a child. Clin Psychopharmacol Neurosci. 2016;14:388–390. doi: 10.9758/cpn.2016.14.4.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Basta MN. Postoperative serotonin syndrome following methylene blue administration for vasoplegia after cardiac surgery: a case report and review of the literature. Semin Cardiothorac Vasc Anesth. 2020:10. doi: 10.1177/1089253220960255. doi: 10.1177/1089253220960255. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 38.Gatti M, Raschi E, De Ponti F. Serotonin syndrome by drug interactions with linezolid: clues from pharmacovigilancepharmacokinetic/pharmacodynamic analysis. Eur J Clin Pharmacol. 2020 doi: 10.1007/s00228-020-02990-1. doi: 10.1007/s00228-020-02990-1. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Orlova Y, Rizzoli P, Loder E. Association of coprescription of triptan antimigraine drugs and selective serotonin reuptake inhibitor or selective norepinephrine reuptake inhibitor antidepressants with serotonin syndrome. JAMA Neurol. 2018;75:566–572. doi: 10.1001/jamaneurol.2017.5144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Francescangeli J, Karamchandani K, Powell M, Bonavia A. The serotonin syndrome: from molecular mechanisms to clinical practice. Int J Mol Sci. 2019;20:2288. doi: 10.3390/ijms20092288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Dunkley EJ, Isbister GK, Sibbritt D, Dawson AH, Whyte IM. The hunter serotonin toxicity criteria: simple and accurate diagnostic decision rules for serotonin toxicity. QJM. 2003;96:635–642. doi: 10.1093/qjmed/hcg109. [DOI] [PubMed] [Google Scholar]
- 42.Radomski JW, Dursun SM, Reveley MA, Kutcher SP. An exploratory approach to the serotonin syndrome: an update of clinical phenomenology and revised diagnostic criteria. Med Hypotheses. 2000;55:218–224. doi: 10.1054/mehy.2000.1047. [DOI] [PubMed] [Google Scholar]
- 43.Sternbach H. The serotonin syndrome. Am J Psychiatry. 1991;148:705–713. doi: 10.1176/ajp.148.6.705. [DOI] [PubMed] [Google Scholar]
- 44.Boyer EW, Shannon M. The serotonin syndrome. N Engl J Med. 2005;352:1112–1120. doi: 10.1056/NEJMra041867. [DOI] [PubMed] [Google Scholar]
- 45.Nguyen H, Pan A, Smollin C, Cantrell LF, Kearney T. An 11-year retrospective review of cyproheptadine use in serotonin syndrome cases reported to the California Poison Control System. J Clin Pharm Ther. 2019;44:327–334. doi: 10.1111/jcpt.12796. [DOI] [PubMed] [Google Scholar]
- 46.Coffey RJ, Edgar TS, Francisco GE, Graziani V, Meythaler JM, Ridgely PM, et al. Abrupt withdrawal from intrathecal baclofen: recognition and management of a potentially life-threatening syndrome. Arch Phys Med Rehabil. 2002;83:735–741. doi: 10.1053/apmr.2002.32820. [DOI] [PubMed] [Google Scholar]
- 47.Doyle AJ, Meyer J, Breen K, Hunt BJ. N-methyl-3,4-methylendioxymethamphetamine (MDMA)-related coagulopathy and rhabdomyolysis: a case series and literature review. Res Pract Thromb Haemost. 2020;4:829–834. doi: 10.1002/rth2.12360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Armenian P, Mamantov TM, Tsutaoka BT, Gerona RR, Silman EF, Wu AH, et al. Multiple MDMA (Ecstasy) overdoses at a rave event: a case series. J Intensive Care Med. 2013;28:252–258. doi: 10.1177/0885066612445982. [DOI] [PubMed] [Google Scholar]
- 49.Grunau BE, Wiens MO, Brubacher JR. Dantrolene in the treatment of MDMA-related hyperpyrexia: a systematic review. CJEM. 2010;12:435–442. doi: 10.1017/s1481803500012598. [DOI] [PubMed] [Google Scholar]
- 50.Russell T, Riazi S, Kraeva N, Steel AC, Hawryluck LA. Ecstacy- induced delayed rhabdomyolysis and neuroleptic malignant syndrome in a patient with a novel variant in the ryanodine receptor type 1 gene. Anaesthesia. 2012;67:1021–1024. doi: 10.1111/j.1365-2044.2012.07226.x. [DOI] [PubMed] [Google Scholar]
- 51.Fiege M, Wappler F, Weisshorn R, Gerbershagen MU, Menge M, Schulte Am Esch J. Induction of malignant hyperthermia in susceptible swine by 3,4-methylenedioxymethamphetamine (“ecstasy”) Anesthesiology. 2003;99:1132–1136. doi: 10.1097/00000542-200311000-00020. [DOI] [PubMed] [Google Scholar]
- 52.Watson JD, Ferguson C, Hinds CJ, Skinner R, Coakley JH. Exertional heat stroke induced by amphetamine analogues. Does dantrolene have a place? Anaesthesia. 1993;48:1057–1060. doi: 10.1111/j.1365-2044.1993.tb07526.x. [DOI] [PubMed] [Google Scholar]
- 53.Baldridge EB, Bessen HA. Phencyclidine. Emerg Med Clin North Am. 1990;8:541–550. [PubMed] [Google Scholar]
- 54.Armen R, Kanel G, Reynolds T. Phencyclidine-induced malignant hyperthermia causing submassive liver necrosis. Am J Med. 1984;77:167–172. doi: 10.1016/0002-9343(84)90455-8. [DOI] [PubMed] [Google Scholar]
- 55.Smith KM, Larive LL, Romanelli F. Club drugs: methylenedioxymethamphetamine, flunitrazepam, ketamine hydrochloride, and gamma-hydroxybutyrate. Am J Health Syst Pharm. 2002;59:1067–1076. doi: 10.1093/ajhp/59.11.1067. [DOI] [PubMed] [Google Scholar]
- 56.Akmal M, Valdin JR, McCarron MM, Massry SG. Rhabdomyolysis with and without acute renal failure in patients with phencyclidine intoxication. Am J Nephrol. 1981;1:91–96. doi: 10.1159/000166498. [DOI] [PubMed] [Google Scholar]
- 57.Leonard JB, Anderson B, Klein-Schwartz W. Does getting high hurt? Characterization of cases of LSD and psilocybin-containing mushroom exposures to national poison centers between 2000 and 2016. J Psychopharmacol. 2018;32:1286–1294. doi: 10.1177/0269881118793086. [DOI] [PubMed] [Google Scholar]
- 58.Rosenberg J, Pentel P, Pond S, Benowitz N, Olson K. Hyper-thermia associated with drug intoxication. Crit Care Med. 1986;14:964–969. doi: 10.1097/00003246-198611000-00011. [DOI] [PubMed] [Google Scholar]
- 59.Zawilska JB, Kacela M, Adamowicz P. NBOMes-highly potent and toxic alternatives of LSD. Front Neurosci. 2020;14:78. doi: 10.3389/fnins.2020.00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Katarzyna K, Paweł Ś, Kamilla M. 25I-NBOME (2-(4-iodo-2,5-dimethoxyphenyl)-N-[(2-methoxyphenyl)methyl]ethanamine)-a harmful hallucinogen review. J Anal Toxicol. 2020:10. doi: 10.1093/jat/bkaa022. doi: 10.1093/jat/bkaa022. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 61.López-Giménez JF, González-Maeso J. Hallucinogens and serotonin 5-HT2A receptor-mediated signaling pathways. Curr Top Behav Neurosci. 2018;36:45–73. doi: 10.1007/7854_2017_478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Candy JM, Morrison C, Paton RD, Logan RW, Lawson R. Salicylate toxicity masquerading as malignant hyperthermia. Paediatr Anaesth. 1998;8:421–423. doi: 10.1046/j.1460-9592.1998.00209.x. [DOI] [PubMed] [Google Scholar]
- 63.Kamour A, George N, Gwynnette D, Cooper G, Lupton D, Eddleston M, et al. Increasing frequency of severe clinical toxicity after use of 2,4-dinitrophenol in the UK: a report from the National Poisons Information Service. Emerg Med J. 2015;32:383–386. doi: 10.1136/emermed-2013-203335. [DOI] [PubMed] [Google Scholar]
- 64.Sousa D, Carmo H, Roque Bravo R, Carvalho F, Bastos ML, Guedes de Pinho P, et al. Diet aid or aid to die: an update on 2,4-dinitrophenol (2,4-DNP) use as a weight-loss product. Arch Toxicol. 2020;94:1071–1083. doi: 10.1007/s00204-020-02675-9. [DOI] [PubMed] [Google Scholar]
- 65.Kopec KT, Kim T, Mowry J, Aks S, Kao L. Role of dantrolene in dinitrophenol (DNP) overdose: a continuing question? Am J Emerg Med. 2019;37:1216.e1–1216.e2. doi: 10.1016/j.ajem.2019.03.035. [DOI] [PubMed] [Google Scholar]
- 66.Van Schoor J, Khanderia E, Thorniley A. Dantrolene is not the answer to 2,4-dinitrophenol poisoning: more heated debate. BMJ Case Rep. 2018;11:e225323. doi: 10.1136/bcr-2018-225323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Torline RL. Extreme hyperpyrexia associated with central anticholinergic syndrome. Anesthesiology. 1992;76:470–471. doi: 10.1097/00000542-199203000-00023. [DOI] [PubMed] [Google Scholar]
- 68.Mann SC, Boger WP. Psychotropic drugs, summer heat and humidity, and hyperpyrexia: a danger restated. Am J Psychiatry. 1978;135:1097–1100. doi: 10.1176/ajp.135.9.1097. [DOI] [PubMed] [Google Scholar]
- 69.Shader RI, Greenblatt DJ. Uses and toxicity of belladonna alkaloids and synthetic anticholinergics. Semin Psychiatry. 1971;3:449–476. [PubMed] [Google Scholar]
- 70.Greenblatt DJ, Shader RI. Drug therapy. Anticholinergics. N Engl J Med. 1973;288:1215–1219. doi: 10.1056/NEJM197306072882306. [DOI] [PubMed] [Google Scholar]