Abstract
In-depth characterization of heart-brain communication in critically ill patients with severe acute respiratory failure is attracting significant interest in the COronaVIrus Disease 19 (COVID-19) pandemic era during intensive care unit (ICU) stay and after ICU or hospital discharge. Emerging research has provided new insights into pathogenic role of the deregulation of the heart-brain axis (HBA), a bidirectional flow of information, in leading to severe multiorgan disease syndrome (MODS) in patients with confirmed infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Noteworthy, HBA dysfunction may worsen the outcome of the COVID-19 patients. In this review, we discuss the critical role HBA plays in both promoting and limiting MODS in COVID-19. We also highlight the role of HBA as new target for novel therapeutic strategies in COVID-19 in order to open new translational frontiers of care. This is a translational perspective from the Italian Society of Cardiovascular Researches.
Keywords: Heart-brain axis, Multiorgan disease syndrome, COVID-19
Graphical Abstract
1. Introduction: cardiovascular and brain disease susceptibility during COVID-19 pandemic
Current concepts on the physiological and pathophysiological interdependence between the cardiovascular (CV) system and the brain under stress will be helpful in understanding adaptive response in COronaVIrus Disease 19 (COVID-19) patients, in discovering new pharmacological targets and in designing unexpected therapeutic strategies. Indeed, simultaneous injury of the heart and brain is emerging as a key risk factor for severe multiorgan disease in patients with confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pneumonia [1], [2], [3], [4]. Therefore, in-depth characterization of heart-brain communication in critically ill patients with severe acute respiratory failure following SARS-CoV-2 dissemination is attracting significant interest in the COVID-19 pandemic era during intensive care unit (ICU) stay or after ICU or hospital discharge (3).
The heart-brain axis (HBA) is characterized by bidirectional flow of information that may modulate susceptibility to injury both locally and systemically to other organs including the lungs [5]. The network of intramedullary neurons generating the sympathetic and parasympathetic outflow to the CV system is finely regulated by cortical neural control systems (i.e., medial prefrontal cortex, insular cortex, anterior cingulate cortex), the amygdala, the hypothalamus, and the hippocampus. These structures perceive and process an array of somato-visceral information, mainly CV afferent inputs, thereby initiating an integrated pattern of endocrine, autonomic, and behavioral responses to emotionally relevant or stressful stimuli [6], [7].
It is well known that psychosocial factors, such as social support, job loss, loneliness, social disruption, bereavement, social status, social distancing and lockdown, can contribute significantly to the pathogenesis of different non-communicable diseases, including acute myocardial infarction (AMI) and heart failure [8], [9], stroke, cognitive impairment [10], [11], major depressive disorder[12], [13], [14], chronic obstructive pulmonary disease [15], asthma [16], obesity [17], chronic kidney disease [18] and cancer [19]. In the context of the COVID-19 pandemic, frail subjects are afraid of leaving home, being infected and going to the hospital when they perceive symptoms or comorbidities worsen [20], [21], [22]. These stressful environmental conditions mainly contribute to the onset of both CV and neurological complications in infected individuals at greatest risk of developing severe forms of COVID-19 and among the patients who show the first symptoms of COVID-19. Several reports, indeed, have shown that CV [23] and neurological morbidities [24], [25], [26], [27] seriously worsen the outcome of COVID-19 patients, although the earlier clinical features remain fever, fatigue, decreased sense of smell and taste, and happy hypoxemia (extremely low blood oxygenation without sensation of dyspnea) [28].
Herein we conduct an in-depth appraisal of the literature in order to depict how SARS-CoV-2 could interfere with the HBA, thereby impairing overall HBA homeostasis and simultaneously worsening acute respiratory failure and leading to major multiorgan complications, such as arrhythmias, AMI, stress-induced cardiomyopathy, stroke and neuroinflammatory disorders ( Fig. 1). We further analyze first data on sex-related HBA alterations [29]. Finally, we describe how the modulation of signaling mediators of heart-brain crosstalk, including cytokines, neurotrophins and extracellular vesicles (EVs), could inspire different approaches in order to improve clinical outcome of COVID-19 patients.
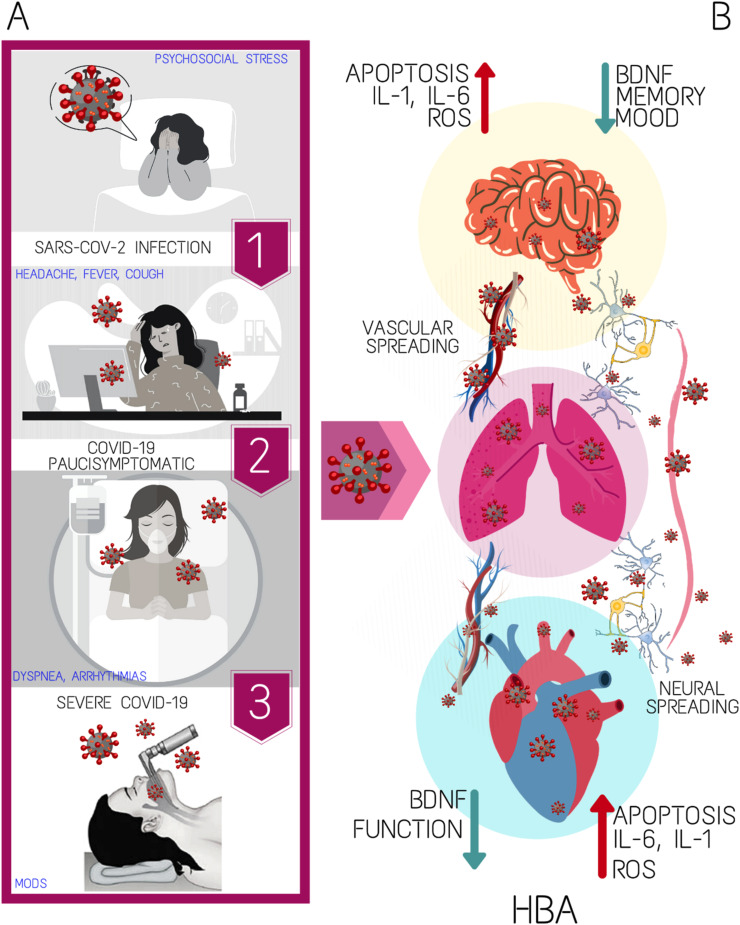
Fig. 1.
The scheme recapitulates the sequence of SARS-CoV-2 infectious disease evolution from high-risk subjects exposed to psychosocial stress towards patients with severe COVID-19 (A); (B) main features of heart-brain axis dysfunction in humans with interstitial pneumonia induced by SARS-CoV-2 following its vascular and transneuronal spreading. BDNF, brain-derived neurotrophic factor; HBA, heart-brain axis; IL, interleukin; MODS, multiorgan disease syndrome; ROS, radical oxygen species.
2. Neurotropism of SARS-CoV-2 short-circuits the neural cardiovascular control
The nucleus of the solitary tract (NTS), located in the dorsal medulla oblongata, represents the first synaptic relay of the visceral afferent fibers carrying sensory information from multiple structures, including arterial and cardiac baroreceptors, chemoreceptors, and lung stretch receptors. The different afferent information entering the NTS is conveyed both to brainstem and supramedullary sites (i.e., medial prefrontal cortex and insular cortex), which may in turn signal back to NTS neurons. This event produces the most appropriate autonomous response to adjust CV response to stimuli (i.e. tachycardia and vasoconstriction/vasodilation in the alerting response) [30]. While acute responses are adaptive, they can turn in excessive sympathetic outflow and maladaptive chronic CV diseases (CVDs) in response to prolonged impairment of NTS.
Hitherto unexpected SARS-CoV-2 neurotropism emerged [31], and the underlying mechanism has been object of intense investigation. From the very beginning of the pandemics, a variety of neurological symptoms, including loss of smell (hyposmia) and taste (hypogeusia), headache, nausea, vomiting, fatigue, have been reported in subjects affected by COVID-19 [24], [32], [33]. Additional neurological complications include stroke, impaired consciousness, seizures, and encephalopathy [24], [25]. Furthermore, the SARS-CoV-2 mRNA has been detected in the brain of 22 patients died by COVID-19 in Germany [34] and in the cerebral spinal fluid of a patient affected by encephalitis/meningitis even though its presence was not confirmed in the nasopharyngeal swab [35]. In agreement with these observations, angiotensin-converting enzyme 2 (ACE2), the main receptor that facilitates the entry of SARS-CoV-2 into host cells, is expressed in neurons from different brain regions [3], [36], [37]. The neurotropism even characterizes SARS-CoV [38], [39], which has been identified in the cytosol of hypothalamic and cortical neurons [40], [41], and Middle East Respiratory Syndrome Coronavirus (MERS-CoV) [42], which may infect thalamus and brainstem. Based on this similarity, SARS-CoV-2 could enter the central nervous system (SNS) through peripheral nerve terminals [1], [4], [31], [36], [43] or via the hematogenous/lymphatic route. The latter pathway is supported by the notion that brain microvascular endothelial cells express ACE2 [43], [44] and, therefore, are susceptible for SARS-CoV-2 entry. In agreement with this hypothesis, viral structural and nonstructural genes have been detected in the endothelial layer of the frontal lobe of COVID-19 patients [45], as well as the viral Spike protein has been detected in over 95% of human microvascular brain endothelial cells by another independent study [46]. This evidence led some authors to suggest that SARS-CoV-2 could infect brainstem neurons by crossing the blood-brain barrier (BBB) [31], [43], [47]. Alternately, SARS-CoV-2 could hijack macrophages, which also express ACE2 [48], to cross the BBB and to access the brain [47], [49], as demonstrated for human immunodeficiency virus (HIV) [50], or it could infect the choroid plexus [47], [51], thereby disrupting the blood-cerebrospinal fluid barrier.
An additional route for SARS-CoV-2 to gain access to the brainstem is represented by olfactory nerve terminals [1], [31]. Epithelial cells of the olfactory mucosa largely express ACE2 [52], whereas the olfactory nerve has long been known to provide a shortcut for several viruses, including influenza and West Nile virus, to enter CNS [53]. In accord with these observations, hyposmia is a common symptom among COVID-19 patients [31], [36]. It has, therefore, been proposed that SARS-CoV-2 infects the olfactory neuroepithelium, whose neurons project dendrites into the nasal cavity, and then exploits the axonal retrograde transport machinery to reach the olfactory bulb via the cribriform plate. Once entered the CNS, SARS-CoV-2 could then use this mode of trans-synaptic transfer to propagate to other brain regions connected to the olfactory bulb, including amygdala, hypothalamus, limbic and pre-frontal cortex, piriform cortex, and brainstem [31], [36], [54], which contains the main centers responsible for the neural control of the CV function. This hypothesis, which was supported by an early report on SARS-CoV neurotropism [38], was confirmed by a recent investigation, showing that SARS-CoV-2 primarily infects the olfactory mucosa and then impinges on the transport along the olfactory tract of the central nervous system to reach the olfactory bulb and medulla oblongata [55]. Herein, severe microthromboembolic events were also detected and associated to local endothelial cell damage [55] due to axonal spreading of virus. These local ischemic infarctions are predicted to exacerbate the damage imposed by viral infection to the brainstem. Moreover, SARS-CoV-2-induced neuronal cell death pauperizes ACE2 in the CV loci of NTS leading to chronic activation of the sympathetic system (with concomitant reduced vagal activity), which triggers different pathogenic mechanisms leading to CVDs (i.e., altered baroreflex, endothelial dysfunction, atherosclerotic plaques, hypertension, left ventricular hypertrophy and arrhythmia) [56], [57], [58], [59], beyond clinical evidence of brainstem lesions [60]. Noteworthy, Cantuti-Castelvetri et al. provided further evidence regarding the SARS-CoV-2 neurotropism [61]. Based on the presence of a cleavage site for the protease furin in the spike protein of SARS-CoV-2, the authors showed that the virus uses neuropilin-1 (NRP1), a receptor that binds furin-cleaved substrates, for its entry and infectivity [61]. Unlike ACE2, NRP1 is highly expressed in the respiratory and olfactory epithelium [62] ; therefore, this receptor could significantly potentiate SARS-CoV-2 infectivity by facilitating the interaction of the virus with ACE2.
Hence, SARS-CoV-2 may spread through either the hematogenous route [63] or the retrograde transport along cranial nerves [64]. Since ACE2 receptors are highly expressed within the lower medulla [65], this is a beta-coronaviruses-susceptible area [38]. A proof of this peculiar neuro-morbidity is that epileptic attacks attributed to autonomic dysfunction have emerged in COVID-19 patients [66]. Moreover, as noticed above, the size of brainstem lesions may be enhanced by combination of systemic inflammation, ischemic thrombotic/embolic lesions or ependymal capillary vasculitis within the brain ventricles [55], [67]. Notably, we cannot exclude the confinement of SARS-CoV-2 in cranial nerve ganglia during remission and re-invasion of the CNS, heart and lungs via the axonal retrograde transport, in accord with other neurotropic viruses [68].
2.1. Autonomic imbalance may lead to stress-induced CV diseases
Although coronaviruses can infect the autonomic center in the brainstem, potentially contributing to cardiovascular and respiratory failure, the direct effect of SARS-CoV2 on the autonomic nervous system is not well known yet. Actually, autonomic imbalance induced by physical and/or emotional stress is often associated with CVDs. The most remarkable example of emotional stress-induced cardiomyopathy is the Takotsubo syndrome (TTS) [69]. An excessive sympathetic response could significantly impact endothelial function, blood pressure, cardiac phenotype (left ventricular hypertrophy and arrhythmia) and the renin-angiotensin-aldosterone system (RAAS) [69], [70], thereby leading to hypertension, increased afterload and eventually congestive heart failure. It is conceivable that the abovementioned sequence of detrimental events may be triggered by the psychosocial stress imposed by COVID-19 pandemic [71], [72] and possibly worsened by CNS infection and injury.
Autonomic imbalance could seriously alter HBA in SARS-CoV-2-positive asymptomatic and paucisymptomatic patients, and in COVID-19 survivors, leading to multiple organ dysfunction syndromes (MODS) up to death. Post-traumatic stress disorder (PTSD), a common consequence of serious medical conditions [73] and major disasters, including the current COVID-19 pandemic [74], could lead to ischemic heart disease [75]. Indeed, PTSD is a significant risk factor in COVID-19 survivors for CVDs, neurological diseases, including thromboembolic stroke [76], [77], [78], and airflow limitations [79]. The influence of PTSD on the HBA is well supported by the onset of acute coronary syndromes and major adverse cardiac events (MACEs), likely contributing to worsening outcomes even in cerebrovascular events in COVID-19 patients [80], [81]. Moreover, PTSD was found to be an independent predictor of ischemic-appearing ECG changes on exercise treadmill tests in patients without known coronary artery disease (CAD), although PTSD patients are frequently exposed to traditional major CV risk factors (i.e., cigarette smoking, arterial hypertension, obesity) [82].
PTSD is indeed characterized by dysregulation of the hypothalamic-pituitary-adrenal axis, with increased cortisol release and sympathetic nervous system activation, similarly to patients with critical illnesses [83]. Elevated catecholamine levels enhance myocardial oxygen consumption, through an increase in heart rate, afterload and myocardial contractility, and negatively affect vascular endothelial function, plaque stability and platelet function. Furthermore, PTSD-induced autonomic imbalance increases plasma glucose levels and RAAS activity with consequent insulin resistance and hypertension [84]. Besides acute renal outcomes (Na+ and water reabsorption), brain RAAS overactivation might contribute to the development of hypertension and heart failure by jeopardizing neuroinflammation due to high ACE levels in the circumventricular organs (i.e., subfornical organ and area postrema) [85], [86] and lower, albeit significant, expression in the hypothalamus [87] and brainstem [88]. Since RAAS has a longer-term impact on inflammation, fibrosis and chronic oxidative stress [89], ACE inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs) counteract arterial hypertension and heart remodeling in PTDS patients. Despite above mentioned drugs may induce ACE2 expression [90], [91], [92], SARS-CoV-2 may down-regulate ACE2 activity, favoring ACE/angiotensin II arm of the RAAS (76). Therefore, the scientific community does not encourage ACEIs withdrawal in hypertensive COVID-19 patients [23], [92]. Moreover, we cannot ignore that emotional/physical stress associated with infection of cortical neurons may increase preload through water reabsorption by stimulating the hypothalamic-hypophyseal axis for antidiuretic hormone (ADH) release [93]. These detrimental effects of PTSD might be enhanced in patients with known CAD and hypertension, portending higher risk of MACEs during exposure to “cytokine storm”.
Indeed, PTSD has been associated to a systemic inflammation which can per se contribute to atherosclerotic disease [94], which may be further exacerbated by the inflammatory burst reported in COVID-19 [23], thereby leading to a vicious circle impairing HBA and providing a life-threatening pro-arrhythmic trigger. Nearly 50% arrhythmias in COVID-19 patients are due to direct myocardial damages caused by viral infection, hypoxia-induced apoptosis, and systemic inflammatory process [95]. Noteworthy, proarrhythmic potential of systemic inflammation may result also from indirect neuronal mechanisms controlling heart rate. Inflammatory cytokines increase cardiac sympathetic outflow via central and peripheral pathways, namely the inflammatory reflex involving the hypothalamus and the left stellate ganglia activation [96]. It serves as a feedback system to putatively dampen elevated cytokine production and immune-inflammatory activation mediated by β2-adrenoceptors in circulating lympho-monocytes. Nevertheless, sympathetic overactivation could favor ventricular arrhythmias even through activation of non-neuronal populations, such as astrocytes of the hypothalamus paraventricular nucleus [97]. It has been suggested correcting this sympathetic tone imbalance through vagus nerve stimulation [98], which also has an anti-inflammatory effect. Indeed, a “cholinergic anti-inflammatory pathway” has been described as able to modulate, by a neural-immune link, the systemic response of the organism in the presence of pathogens producing faster beneficial effects with respect to the classical anti-inflammatory mechanisms (83). In the context of COVID-19-induced stress, characterized by a typical “fight or flight” sympathetic activation, the vagus stimulation may represent an additional protective strategy also for the cardiovascular system. Thus, by exploiting the brain's ability to modulate systemic inflammatory responses to endotoxin, it would be possible to activate a rapid endogenous mechanism capable of suppressing the lethal effects of biological toxins during the “cytokine storm” induced by COVID syndrome. Alternatively, since α7 nicotinic acetylcholine receptors (α7-nAChRs) are potentially involved in modulating pro-inflammatory cytokine secretion and suppressing the “cytokine storm”, nicotinic cholinergic agonists should be examined as potential therapeutic options in the COVID syndrome [99], [100].
3. SARS-CoV-2 and the bidirectional neuronal-to-vascular communication
Due to the importance of vasculo-neuronal communication (VNC), here we consider how COVID-19 may affect this important cross-talk between neuronal and vascular components of the CNS. Indeed, neuronal activity (NA) may directly tune the diameter of microvessels within the neurovascular unit (NVU) [101], a network of neurons, astrocytes, endothelial cells (ECs) and pericytes responsible for the local control of cerebral blood flow (CBF). Therefore, higher NA raises CBF towards activated brain regions [101], [102]. Local changes in CBF may, in turn, signal back to adjacent neurons, modulating NA and synaptic plasticity according to VNC [103], [104]. Mounting evidence showed that neuronal control of blood vessels initiates at capillaries level, which are better positioned to detect NA as they are closer than arterioles to firing neurons (8–23 µm vs. 70–160 µm) [101]. Capillaries, thus, could dilate even before “feeding arterioles” because of relaxation of pericytes running along the endothelial tube [101], [105].
Somato-sensory stimulation may activate endothelial N-methyl-D-aspartate receptors (NMDARs) stimulating Ca2+-dependent nitric oxide (NO) release and triggering neurovascular coupling (NVC) [101], [106]. NA-induced local K+ accumulation could activate endothelial inward-rectifier K+ channels and elicit a propagating hyperpolarization that causes upstream arteriolar relaxation via myoendothelial gap junctions [107]. Endothelial-mediated detection of NA could then be sustained by metabotropic glutamate receptors [108], [109]. The following increase in CBF far exceeds the metabolic needs of the tissue, probably to subtly shape NA and synaptic activity [104] Indeed, an increase in CBF may indirectly inhibit pyramidal NA through the TRPV4-dependent activation of perisynaptic astrocytes [103] which stimulate local interneurons [103] and regulate K+ homeostasis [110]. Furthermore, hemodynamic signals could potentiate shear stress-induced NO release from capillary ECs [101], which support long-term potentiation in several brain regions [111], [112].
SARS-CoV-2 infected ECs in multiple vascular districts [113], including brain capillaries [45], thereby inducing endothelial dysfunction associated with apoptosis [55], [113], [114]. We cannot exclude that SARS-CoV-2 affects NVC by impairing EC ability to sense NA and release NO, an effect that could be exacerbated after COVID-19-induced pericyte loss, which could arise either indirectly (because of stress-induced glucocorticoid signaling) [115] or directly (because of ACE2-mediated SARS-CoV-2 entry in pericytes) [116]. The emerging role of capillary signaling in NVC is a potential explanation for the high incidence of stroke [23] and the severe neurological complications in COVID-19 patients [117]. Of note, also brain lymphatics are subjected to COVID-19-induced systemic endotheliitis [113] and could favor SARS-COV-2 entry into the brain [118]. Whether brain lymphatic, as major mediators of neuroinflammation and immune cell trafficking [119] are implicated in VNC derangement during SARS-COV-2 infection, remains to be determined. Thus, SARS-CoV-2 infection could worsen the HBA both systemically (by increasing the susceptibility to psychosocial factors) and locally (by interfering with the bidirectional neuronal-to-vascular communication). The altered VNC might increase the occurrence of headache, confusion, delirium, and poor outcomes, including mortality for stroke in COVID-19 patients.
4. Heart-brain axis in COVID-19: role of sex
COVID-19 similarly affects both sexes, but mortality in men is about two-fold than that of young women. Conversely, mortality is higher in both sexes in older subjects with pre-existing CV comorbidities [120], [121]. Among COVID-19 patients, fewer women than men presented with cardiac injury, but those women with cardiac injury had higher levels of systemic inflammatory markers, suggesting a male inflammatory phenotype [122]. Therefore, here we briefly consider sex-related HBA alterations and other sex/gender related issues, which may be also useful to develop personalized treatments.
Women are more susceptible to psychosocial stress-induced injury than men are [123], [124]. Besides social factors, this different susceptibility to multiorgan injury originates, in part, from processes initiated during fetal development. For instance, men respond to stress with an increase in vascular resistance, while women with an increase in heart rate. The different molecular pathways between sexes involve numerous mediators, such as growth factors, neurotransmitters and RAAS. These factors influence differently the neuronal and vascular development of hypothalamic-pituitary-adrenal-axis regions (i.e., the paraventricular nucleus) controlling blood pressure [123]. Stress affects RAAS in the brain and heart leading to an imbalance in the ACE/ACE2 ratio and autonomic outflow. Sex differences in RAAS blood pressure regulation and in the pathophysiology of hypertension have been shown in both clinical and animal models [125]. As mentioned above, beta-coronaviruses target ACE2 and MODS is characterized by an unbalanced ACE/ACE2 ratio [91]. Since ACE2 is an anti-inflammatory/estrogen-regulated enzyme whose gene is on the X-chromosome, gender-related differences in ACE/ACE2 ratio and byproducts exist [126]. Moreover, transmembrane protease serine 2 (TMPRSS2), an enzyme that primes spike S1 protein, is a testosterone regulated gene and may have a lower expression in women than in men [127]. Interestingly, aging alters activities of these enzymes besides hormones. Women have a longer life expectancy than men and this may explain why more women are infected than men over the age of 80 [23]. This is part of female-male health-survival paradox.
A cardiac disease with strong correlation between stress and HBA is TTS, which has preference for postmenopausal women. Estrogen deprivation plays a facilitator role in TTS, likely involving endothelial dysfunction [128]. Increased cortisol and catecholamine release, and RAAS dysregulation, may lead to higher cardiac workload and many signs of acute phase of TTS. The causal relationship between TTS and RAAS is anecdotally demonstrated by the efficacy of ACEIs and ARBs in reducing the likelihood of recurrent episodes of apical, midventricular, or basal left ventricular ballooning [129], [130]. Indeed, a Pubmed search for “Takotsubo and COVID-19” (Date accessed: 15/02/2021) provided 89 results, one of which is about an inverted (basal) TTS in a 50-year-old man [131]. More recently, a minireview on TTS and COVID-19 has been published in which a few more cases are described [132]. TTS in COVID-19 has probably been underestimated so far, yet the investigation on its underlying mechanisms might shed light on the gender-related different HBA alterations induced by COVID-19.
5. Potential therapeutic strategies: from drugs to functional foods
HBA offers hitherto unexpected targets to develop innovative approaches of multiorgan protection and care in COVID-19 patients.
5.1. Drug-based treatments
Currently, there are no regulatory agency-approved medications for COVID-19. However, a multitude of drugs approved for other indications are undergoing trials to repurpose their safety and efficacy (https://www.who.int/ictrp/en/). Most relevant compounds include: remdesivir (investigational antiviral) [133] and monoclonal antibodies disrupting the interaction of receptor binding domain of SARS-CoV-2 Spike S1 protein with ACE2 receptor [134]. Hyperinflammation has provided the rationale for testing anti-inflammatory drugs to prevent the fatal “cytokine storm” in COVID-19. These include corticosteroids that were commonly used during previous pneumonia outbreaks, like SARS, and are currently used in COVID-19 patients, despite controversial efficacy [135]. Corticosteroids, like dexamethasone, may be useful in the short-term as they restrain the “cytokine storm”, while being detrimental in the long-term due to the inhibition of protective T cells and the block of B cell antibody production. Indeed, dexamethasone seems to reduce mortality only in critical COVID-19 patients [136], and methylprednisolone is a valid alternative to dexamethasone [137]. A case report showed that co-administration of methylprednisolone (200 mg/day, 4 days) and human immunoglobulin (20 g/day, 4 days) may reverse myocardial injury and reduce cardiac troponin levels [138]. On the other hand, Ibuprofen, a commonly prescribed non-steroidal anti-inflammatory drug, reduced IL-6 levels in COVID-19 patients and several trials of anti-IL-6 therapies, like tocilizumab, are actively recruiting [139]. Finally, steroids and non-steroidal anti-inflammatory drugs even exert a dose-dependent inhibition of platelet aggregation by inhibiting intravascular microaggregation and pulmonary shunting as well. Since these drugs target pro-inflammatory processes and pro-thrombotic factors common to COVID-19, PTSD and HBA dysfunction, further studies are required to assess whether these drugs may attenuate the respiratory syndrome by exerting HBA protection. We cannot ignore that the potential therapies for COVID-19 discussed above have important toxicities that require caution, monitoring cardiac troponin levels (i.e., during chloroquine and hydroxychloroquine therapy) [95] and the onset of delirium due to drug interactions (i.e.: during co-medication with psychotropic drugs) [140].
5.2. Extracellular vesicles
Extracellular vesicles (EVs), nano-sized membrane-bound particles released by majority of the cells, are emerging as paracrine mediators between the brain and immune system in critically ill patients [141]. EVs can modulate target cells via their cargo, enriched by non-coding RNAs, such as microRNAs (miRNAs), proteins, lipids and other metabolites. Recently, plasma EVs have gained attention as new theranostic tool, given their protective and regenerative effects (such as in the case of mesenchymal progenitor stromal-EVs), as well as their possible diagnostic role even in older surgical patients [142]. Interestingly, endothelial and neural cells after brain injury can secrete EVs crossing the blood brain barrier, reaching peripheral organs and regulating systemic inflammation [143]. Notably, EVs-delivered miRNA-126 supports cardiac function and is modulated following stroke. In fact, EVs isolated from ECs of conditional miR-126 knock-out mice display lower paracrine protective effects on cultured cardiomyocytes exposed to hypertrophy-inducing stimuli [144]. Smallest EVs, also defined as exosomes, as isolated from several different sources of stem/progenitor cells, may exert neuro- and/or cardio-protective effects by quenching inflammation in several preclinical experimental models [145], [146], [147], [148], [149], [150], [151]. Thus, EVs may be involved in the HBA homeostasis under stress.
ECs-derived EVs (EEVs) could be released upon SARS-CoV-2-induced endothelial injury, thereby contributing to the pro-inflammatory and pro-coagulant milieu, which underlies the fatal outcome of COVID-19 [152], [153]. Therefore, EEVs have been put forward as potential prognostic peripheral blood markers in individuals potentially affected by COVID-19 [153]. Likewise, circulating EVs could be enriched with cardiac troponin and might represent another reliable prognostic marker of myocardial injury [154]. On the other hand, the therapeutic use of EVs isolated from Mesenchymal progenitor Stromal Cells (MSCs) in COVID-19 patients is actively debated. Allogenic MSCs display beneficial immunomodulatory activity in critically ill patients with acute respiratory distress syndrome (ARDS) [155], with their EVs reproducing most of the parental cell paracrine anti-inflammatory potential [156]. Therefore, MSC-EVs are currently under consideration as putative supportive treatment to possibly tackle the systemic “cytokine storm” and hyper-activation of the immune cell system [135], [157]. Despite some trials on the use of somatic or cardiac MSCs with or without additional treatment of MSC-EVs are ongoing in China, Iran and US [135], [157], the International Society of Extracellular Vesicles (ISEV) and the International Society for Cell and Gene Therapy (ISCT) have very recently released a statement expressing specific concerns about EVs-based COVID-19 therapy [158]. Otherwise, ticagrelor, an established anti-platelet drug, drives the release of antiapoptotic exosomes from human cardiac-derived progenitors [159], thereby suggesting an alternative approach to direct exosome transplantation in both vital organs beyond lowering the risk of thrombotic events.
5.3. Functional foods
Recent emphasis on the HBA protection has shifted to the epigenetic regulation of gene expression [160]. Enhancing the adaptive response of cardiac and brain cells without changing DNA sequence could improve outcome of critically ill patients [161]. Functional food components and their metabolites could prevent organ failure through the chemical modulation of chromatin structure and microRNAs release to pattern gene activity [162]. A recent study has highlighted the role of brain-derived neurotrophic factor (BDNF) as a crucial neurohormonal mediator within the HBA, as its levels are simultaneously decreased under oxidative stress [163]. Since failure of BDNF/TrkB signaling pathway is related to cardiac and hippocampal dysfunction, its decay may enhance the susceptibility to both cardiac and brain damage in SARS-CoV-2-positive patients [164]. Therefore, preventive use of functional diet or artificial nutrition in the critical care setting with epigenetically active dietary compounds may help to prevent/counteract organ failure or improve the outcome of critically ill patients. Preliminary pre-clinical results have demonstrated that regular intake of barley beta-glucan or saffron, natural inhibitors of class I histone deacetylases (HDACs), or curcumin, a natural inhibitor of histone acetyltransferases (HATs), may sustain the HBA through the upregulation of cardiac and hippocampal BDNF/TrkB pathway [160], [165]. Moreover, sulforaphane, a hydrolysis product of glucoraphanin present in Brassica vegetables, enhances neuronal BDNF expression by inhibiting class I HDACs [166]. Moreover, diet supplementation with eicosapentaenoic acid (EPA) can prevent BDNF downregulation during neuroinflammation [167] since it reduces the expression of class I DNA methyltransferases (DNMTs) [160]. Resveratrol, a well-known natural polyphenol compound with anti-inflammatory properties [168] that enriches grapes, red wine, berries and extra virgin olive oil (EVOO), has been proposed to counteract SARS-CoV-2 [169] as it significantly abated Middle East Respiratory Syndrome Coronavirus (MERs-CoV) replication in vitro [170]. Since resveratrol targets DNMTs, HDACs and lysine-specific demethylase [171], it upregulates BDNF gene expression after 10 days oral intake [172]. So far, one randomized proof-of-concept clinical trial (NCT04400890) aimed to evaluate the safety and the effectiveness of resveratrol in two hundred COVID-19 patients. Both groups (Resveratrol and placebo) will also receive vitamin D3 (100,000 IU) to augment the effects of resveratrol [173]. Interestingly, their combination has been also shown to act on HBA, by preventing cognitive decline [174] and ameliorating ischemia-reperfusion process after myocardial injury [175] in rodents. These results suggest that combination of different dietary compounds may lead to therapeutic food synergism, as aimed by ristoceutica, a new science announced in 2016 at Scuola Superiore Sant’Anna (Pisa, Italy) and directed to develop functional meal based on combination of functional foods [161], [176]. A specific smart diet is yet to be developed to prevent/attenuate the adverse effects of SARS-CoV-2 on the HBA. A better protection against viral infection is likely to be conferred by a varied dietary regimen [177]. Although the flavonoid quercetin is a reasonably potent inhibitor of the protease SARS-CoV-2 3CLpro (Ki ~ 7 μM) in vitro [178], foods rich of isoquercitrin (red onions, red raspberry, black grapes, broccoli, black tea), which is hydrolyzed to quercetin in the small intestine and rapidly absorbed, cannot replace the therapeutic treatment prescribed to severely ill COVID-19 patients. In accord with co-medications, association of different foods rich of calcium and/or vitamin D could be successfully suggested to reduce the side effects related to prolonged bed rest and steroid treatment (i.e. dairy products and salmon). Ristoceutical studies will be helpful to assess whether specific functional food compounds, i.e. barley beta-glucan, isoquercitrin, curcumin and resveratrol, may act synergistically with commercially available drugs to hamper the viral infection and cytokine storm, and to counteract associated complications related to their high dosage. Finally, Covid19 patients treated with steroids should avoid regular intake of foods that are high in salt and sugar in order to prevent the onset of metabolic disorders and arterial hypertension.
6. Conclusions
SARS-CoV-2-positive or COVID-19 patients developing heart and brain dysfunction is attracting growing interest. SARS-CoV-2 can interfere with HBA either through direct neurotropism towards the neural CV control centers and by interfering with the local bidirectional neuronal-to-vascular communication or by increasing the susceptibility to psychosocial factors resulting in emotional stress and/or anxiety, thereby potentially leading to cardiac hypertrophy, arrhythmia, AMI or TTS ( Fig. 2). Perturbance of the delicate balance of neurohumoral cues regulating CV function may in turn damage brain by resulting in stroke. It is conceivable that the virus can spread to brain microvessels leading to pericyte loss and/or endothelial dysfunction, which may alter CBF through impairment of NVC regulation and recurrent microthrombotic events. The brain damage will in turn reinforce the vicious circle initiated by SARS-CoV-2 infection triggering systemic inflammation that will further impact on cardiac function by enhancing oxidative stress, cardiomyocyte remodeling, collagen deposition and rhythm alterations as well as macrophage infiltration [145]. Resident microglia activated upon injury produce cytokines, primarily IL-6 and IL-1β, which pass through the damaged blood-brain barrier. Furthermore, dying brain cells release damage-associated proteins that enter the systemic circulation, activate Toll-like receptors (TLRs) and boost cytokine and chemokine production [69]. Notably, TLRs are also expressed in cardiomyocytes and have been implicated in the development of cardiac inflammation and heart failure [179]. Estrogens might protect the HBA both from the viral infection (because of gender-related differences in ACE/ACE2 ratio and in TMPRSS2 expression) and from the detrimental consequences of RAAS dysregulation (induced either by the emotional stress or by the direct infection of medulla oblongata). The above-mentioned complex scenario opens new avenues to identify unexpected targets and develop innovative non –pharmacological personalized approaches, such as smart drugs, EVs and functional foods. Finally, rehabilitation is important for COVID-19 patients and survivors [180]. Indeed, the protection of HBA in COVID-19 patients who have mild symptoms could benefit from physical exercise which positively impacts on both CV function and mental health, as recently reviewed in [181]. Understanding the signaling pathways of HBA engaged by physical exercise to stimulate neurocardiac fitness is likely to speed up the identification of an effective pharmacological strategy to rescue HBA upon SARS-CoV-2 infection even in the presence of functional motor limitations.
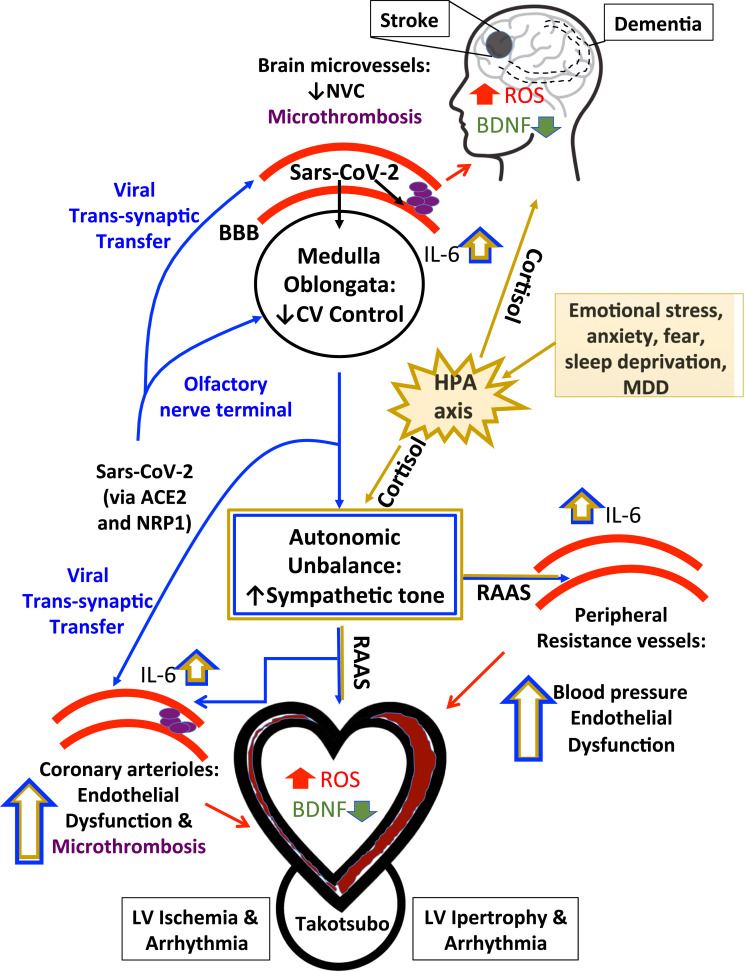
Fig. 2.
Overview of proposed routes of heart-brain crosstalk leading to simultaneous heart and brain injury in COVID19 patients. ACE2, angiotensin-converting enzyme 2; BBB, blood brain barrier; BDNF, brain-derived neurotrophic factor; CV, cardiovascular; HPA, hypothalamic-pituitary-adrenal (HPA) axis; IL-6, interleukin-6; LV, left ventricular; MDD, major depressive disorder; NVC, neurovascular coupling; NRP1, neuropilin 1; RAAS, renin–angiotensin–aldosterone system; ROS, radical oxygen species.
Funding
We thank the following grant that supported research conducted in Authors’ laboratories and discussed in the manuscript: ETHERNA Project (Prog. no. 161/16, Fondazione Pisa, Italy) and the EU Horizon 2020 FETPROACT-01-2018 (NeuHeart) (V.L.); FIL_2018_Miragoli (M.M.); COST Action BIONECA CA16122 (F.Q.); University of Genova, Italy, Curiosity Driven Programme (S.B.); Italian Ministry of Education, University and Research (MIUR): Dipartimento di Eccellenza Program (2018–2022) – Dept. of Biology and Biotechnology "L. Spallanzani", University of Pavia, Italy and the EU Horizon 2020 FETOPEN-2018–2020 Program under Grant Agreement No. 828984 (LION-HEARTED) (Francesco Moccia); Italian Ministry of Education (MIUR) (PRIN 2017PZY5K7) (Fabio Mangiacapra); POR Calabria FESR-FSE 2014/2020: Azione 10.5.12 – LineaB (European Union & Calabria Region Union & Calabria Region, Italy) (C.R.); MIUR (PRIN respectively); MIUR (PRIN 2017XZMBYX), Fondazione CR Firenze and University of Florence, Italy (R.C. and L.S.); DiSS_PSR2019_DIP_013 and PNRA18_00071 n. 1314, Italy (M.S.). Funding source had no such involvement in study design, in the collection, analysis, interpretation of data, in the writing of the report; and in the decision to submit the paper for publication. A.G. is co-founder and stakeholder of Kither Biotech, a pharmaceutical product company focused on respiratory medicine not in conflict with statements in this review.
CRediT authorship contribution statement
Vincenzo Lionetti designed and directed the project. Other authors contributed equally and are listed in alphabetical order. All authors discussed and commented on the manuscript, provided critical feedback and helped shape the final version of the manuscript.
Declaration of interest statement
None.
Acknowledgments
The Italian Society of Cardiovascular Researches is gratefully thankful to the Fondazione Cassa di Risparmio di Imola for continuous support to our scientific and dissemination activities.
References
- 1.Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;92(6):552–555. doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Steardo L., Steardo L., Jr., Zorec R., Verkhratsky A. Neuroinfection may contribute to pathophysiology and clinical manifestations of COVID-19. Acta Physiol. 2020;229(3) doi: 10.1111/apha.13473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yashavantha Rao H.C., Jayabaskaran C. The emergence of a novel coronavirus (SARS-CoV-2) disease and their neuroinvasive propensity may affect in COVID-19 patients. J. Med. Virol. 2020;92(7):786–790. doi: 10.1002/jmv.25918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chigr F., Merzouki M., Najimi M. Autonomic brain centers and pathophysiology of COVID-19. ACS Chem. Neurosci. 2020;11(11):1520–1522. doi: 10.1021/acschemneuro.0c00265. [DOI] [PubMed] [Google Scholar]
- 5.Mrozek S., Gobin J., Constantin J.M., Fourcade O., Geeraerts T. Crosstalk between brain, lung and heart in critical care. Anaesth. Crit. Care Pain Med. 2020 doi: 10.1016/j.accpm.2020.06.016. [DOI] [PubMed] [Google Scholar]
- 6.Tahsili-Fahadan P., Geocadin R.G. Heart-brain axis: effects of neurologic injury on cardiovascular function. Circ. Res. 2017;120(3):559–572. doi: 10.1161/CIRCRESAHA.116.308446. [DOI] [PubMed] [Google Scholar]
- 7.Palma J.A., Benarroch E.E. Neural control of the heart: recent concepts and clinical correlations. Neurology. 2014;83(3):261–271. doi: 10.1212/WNL.0000000000000605. [DOI] [PubMed] [Google Scholar]
- 8.Rozanski A., Blumenthal J.A., Kaplan J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation. 1999;99(16):2192–2217. doi: 10.1161/01.cir.99.16.2192. [DOI] [PubMed] [Google Scholar]
- 9.Chilunga F.P., Boateng D., Henneman P., Beune E., Requena-Mendez A., Meeks K., Smeeth L., Addo J., Bahendeka S., Danquah I., Schulze M.B., Klipstein-Grobusch K., Mannens M., Agyemang C. Perceived discrimination and stressful life events are associated with cardiovascular risk score in migrant and non-migrant populations: the RODAM study. Int. J. Cardiol. 2019;286:169–174. doi: 10.1016/j.ijcard.2018.12.056. [DOI] [PubMed] [Google Scholar]
- 10.Clemens V., Deschamps P., Fegert J.M., Anagnostopoulos D., Bailey S., Doyle M., Eliez S., Hansen A.S., Hebebrand J., Hillegers M., Jacobs B., Karwautz A., Kiss E., Kotsis K., Kumperscak H.G., Pejovic-Milovancevic M., Christensen A.M.R., Raynaud J.P., Westerinen H., Visnapuu-Bernadt P. Potential effects of “social” distancing measures and school lockdown on child and adolescent mental health. Eur. Child Adolesc. Psychiatry. 2020;29(6):739–742. doi: 10.1007/s00787-020-01549-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kuhlmann S., Piel M., Wolf O.T. Impaired memory retrieval after psychosocial stress in healthy young men. J. Neurosci. 2005;25(11):2977–2982. doi: 10.1523/JNEUROSCI.5139-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lara B., Carnes A., Dakterzada F., Benitez I., Pinol-Ripoll G. Neuropsychiatric symptoms and quality of life in Spanish patients with Alzheimer’s disease during the COVID‐19 lockdown. Eur. J. Neurol. 2020;27:1744–1747. doi: 10.1111/ene.14339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Smith L., Jacob L., Yakkundi A., McDermott D., Armstrong N.C., Barnett Y., Lopez-Sanchez G.F., Martin S., Butler L., Tully M.A. Correlates of symptoms of anxiety and depression and mental wellbeing associated with COVID-19: a cross-sectional study of UK-based respondents. Psychiatry Res. 2020;291 doi: 10.1016/j.psychres.2020.113138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ozamiz-Etxebarria N., Idoiaga Mondragon N., Dosil Santamaria M., Picaza Gorrotxategi M. Psychological symptoms during the two stages of lockdown in response to the COVID-19 outbreak: an investigation in a sample of citizens in Northern Spain. Front. Psychol. 2020;11:1491. doi: 10.3389/fpsyg.2020.01491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Clark A.J., Strandberg-Larsen K., Masters Pedersen J.L., Lange P., Prescott E., Rod N.H. Psychosocial risk factors for hospitalisation and death from chronic obstructive pulmonary disease: a prospective cohort study. COPD. 2015;12(2):190–198. doi: 10.3109/15412555.2014.922175. [DOI] [PubMed] [Google Scholar]
- 16.Landeo-Gutierrez J., Forno E., Miller G.E., Celedon J.C. Exposure to violence, psychosocial stress, and asthma. Am. J. Respir. Crit. Care Med. 2020;201(8):917–922. doi: 10.1164/rccm.201905-1073PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baratin C., Beune E., van Schalkwijk D., Meeks K., Smeeth L., Addo J., de-Graft Aikins A., Owusu-Dabo E., Bahendeka S., Mockenhaupt F.P., Danquah I., Schulze M.B., Spranger J., Boateng D., Klipstein-Grobusch K., Stronks K., Agyemang C. Differential associations between psychosocial stress and obesity among Ghanaians in Europe and in Ghana: findings from the RODAM study. Soc. Psychiatry Psychiatr. Epidemiol. 2020;55(1):45–56. doi: 10.1007/s00127-019-01682-1. [DOI] [PubMed] [Google Scholar]
- 18.Bruce M.A., Beech B.M., Sims M., Brown T.N., Wyatt S.B., Taylor H.A., Williams D.R., Crook E. Social environmental stressors, psychological factors, and kidney disease. J. Investig. Med. 2009;57(4):583–589. doi: 10.231/JIM.0b013e31819dbb91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chida Y., Hamer M., Wardle J., Steptoe A. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nat. Clin. Pract. Oncol. 2008;5(8):466–475. doi: 10.1038/ncponc1134. [DOI] [PubMed] [Google Scholar]
- 20.Chen A.T., Ge S., Cho S., Teng A.K., Chu F., Demiris G., Zaslavsky O. Reactions to COVID-19, information and technology use, and social connectedness among older adults with pre-frailty and frailty. Geriatr. Nurs. 2020 doi: 10.1016/j.gerinurse.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marroquin B., Vine V., Morgan R. Mental health during the COVID-19 pandemic: effects of stay-at-home policies, social distancing behavior, and social resources. Psychiatry Res. 2020;293 doi: 10.1016/j.psychres.2020.113419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Banerjee D., Viswanath B. Neuropsychiatric manifestations of COVID-19 and possible pathogenic mechanisms: insights from other coronaviruses. Asian J. Psychiatr. 2020;54 doi: 10.1016/j.ajp.2020.102350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Moccia F., Gerbino A., Lionetti V., Miragoli M., Munaron L.M., Pagliaro P., Pasqua T., Penna C., Rocca C., Samaja M., Angelone T. COVID-19-associated cardiovascular morbidity in older adults: a position paper from the Italian Society of Cardiovascular Researches. GeroScience. 2020;42:1021–1049. doi: 10.1007/s11357-020-00198-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan. JAMA Neurol. 2020;77:683. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Helms J., Kremer S., Merdji H., Clere-Jehl R., Schenck M., Kummerlen C., Collange O., Boulay C., Fafi-Kremer S., Ohana M., Anheim M., Meziani F. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020;382(23):2268–2270. doi: 10.1056/NEJMc2008597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu Y., Xu X., Chen Z., Duan J., Hashimoto K., Yang L., Liu C., Yang C. Nervous system involvement after infection with COVID-19 and other coronaviruses. Brain Behav. Immun. 2020;87:18–22. doi: 10.1016/j.bbi.2020.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ellul M.A., Benjamin L., Singh B., Lant S., Michael B.D., Easton A., Kneen R., Defres S., Sejvar J., Solomon T. Neurological associations of COVID-19. Lancet Neurol. 2020;19:767–783. doi: 10.1016/S1474-4422(20)30221-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Archer S.L., Sharp W.W., Weir E.K. Differentiating COVID-19 pneumonia from acute respiratory distress syndrome and high altitude pulmonary edema: therapeutic implications. Circulation. 2020;142(2):101–104. doi: 10.1161/CIRCULATIONAHA.120.047915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Baroni C., Lionetti V. The impact of sex and gender on heart–brain axis dysfunction: current concepts and novel perspectives. Can. J. Physiol. Pharmacol. 2021;99:151–160. doi: 10.1139/cjpp-2020-0391. [DOI] [PubMed] [Google Scholar]
- 30.Verberne A.J., Owens N.C. Cortical modulation of the cardiovascular system. Prog. Neurobiol. 1998;54(2):149–168. doi: 10.1016/s0301-0082(97)00056-7. [DOI] [PubMed] [Google Scholar]
- 31.Zhou Z., Kang H., Li S., Zhao X. Understanding the neurotropic characteristics of SARS-CoV-2: from neurological manifestations of COVID-19 to potential neurotropic mechanisms. J. Neurol. 2020;267(8):2179–2184. doi: 10.1007/s00415-020-09929-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhang M., Zhou L., Wang J., Wang K., Wang Y., Pan X., Ma A. The nervous system-a new territory being explored of SARS-CoV-2. J. Clin. Neurosci. 2020;82(Pt. A):87–92. doi: 10.1016/j.jocn.2020.10.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Puelles V.G., Lutgehetmann M., Lindenmeyer M.T., Sperhake J.P., Wong M.N., Allweiss L., Chilla S., Heinemann A., Wanner N., Liu S., Braun F., Lu S., Pfefferle S., Schroder A.S., Edler C., Gross O., Glatzel M., Wichmann D., Wiech T., Kluge S., Pueschel K., Aepfelbacher M., Huber T.B. Multiorgan and renal tropism of SARS-CoV-2. N. Engl. J. Med. 2020;383(6):590–592. doi: 10.1056/NEJMc2011400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moriguchi T., Harii N., Goto J., Harada D., Sugawara H., Takamino J., Ueno M., Sakata H., Kondo K., Myose N., Nakao A., Takeda M., Haro H., Inoue O., Suzuki-Inoue K., Kubokawa K., Ogihara S., Sasaki T., Kinouchi H., Kojin H., Ito M., Onishi H., Shimizu T., Sasaki Y., Enomoto N., Ishihara H., Furuya S., Yamamoto T., Shimada S. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fenrich M., Mrdenovic S., Balog M., Tomic S., Zjalic M., Roncevic A., Mandic D., Debeljak Z., Heffer M. SARS-CoV-2 dissemination through peripheral nerves explains multiple organ injury. Front. Cell. Neurosci. 2020;14:229. doi: 10.3389/fncel.2020.00229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Doobay M.F., Talman L.S., Obr T.D., Tian X., Davisson R.L., Lazartigues E. Differential expression of neuronal ACE2 in transgenic mice with overexpression of the brain renin-angiotensin system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007;292(1):R373–R381. doi: 10.1152/ajpregu.00292.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Netland J., Meyerholz D.K., Moore S., Cassell M., Perlman S. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J. Virol. 2008;82(15):7264–7275. doi: 10.1128/JVI.00737-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.McCray P.B., Jr., Pewe L., Wohlford-Lenane C., Hickey M., Manzel L., Shi L., Netland J., Jia H.P., Halabi C., Sigmund C.D., Meyerholz D.K., Kirby P., Look D.C., Perlman S. Lethal infection of K18-hACE2 mice infected with severe acute respiratory syndrome coronavirus. J. Virol. 2007;81(2):813–821. doi: 10.1128/JVI.02012-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z., Zhuang H., Wu B., Zhong H., Shao H., Fang W., Gao D., Pei F., Li X., He Z., Xu D., Shi X., Anderson V.M., Leong A.S. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005;202(3):415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xu J., Zhong S., Liu J., Li L., Li Y., Wu X., Li Z., Deng P., Zhang J., Zhong N., Ding Y., Jiang Y. Detection of severe acute respiratory syndrome coronavirus in the brain: potential role of the chemokine mig in pathogenesis. Clin. Infect. Dis. 2005;41(8):1089–1096. doi: 10.1086/444461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li K., Wohlford-Lenane C., Perlman S., Zhao J., Jewell A.K., Reznikov L.R., Gibson-Corley K.N., Meyerholz D.K., McCray P.B., Jr. Middle east respiratory syndrome coronavirus causes multiple organ damage and lethal disease in mice transgenic for human dipeptidyl peptidase 4. J. Infect. Dis. 2016;213(5):712–722. doi: 10.1093/infdis/jiv499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Alquisiras-Burgos I., Peralta-Arrieta I., Alonso-Palomares L.A., Zacapala-Gomez A.E., Salmeron-Barcenas E.G., Aguilera P. Neurological complications associated with the blood-brain barrier damage induced by the inflammatory response during SARS-CoV-2 infection. Mol. Neurobiol. 2020;58:520–535. doi: 10.1007/s12035-020-02134-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Baig A.M., Khaleeq A., Ali U., Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11(7):995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- 45.Paniz-Mondolfi A., Bryce C., Grimes Z., Gordon R.E., Reidy J., Lednicky J., Sordillo E.M., Fowkes M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) J. Med. Virol. 2020;92(7):699–702. doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nuovo G.J., Magro C., Shaffer T., Awad H., Suster D., Mikhail S., He B., Michaille J.J., Liechty B., Tili E. Endothelial cell damage is the central part of COVID-19 and a mouse model induced by injection of the S1 subunit of the spike protein. Ann. Diagn. Pathol. 2021;51 doi: 10.1016/j.anndiagpath.2020.151682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rodriguez M., Soler Y., Perry M., Reynolds J.L., El-Hage N. Impact of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in the nervous system: implications of COVID-19 in neurodegeneration. Front. Neurol. 2020;11 doi: 10.3389/fneur.2020.583459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sokolowska M., Lukasik Z.M., Agache I., Akdis C.A., Akdis D., Akdis M., Barcik W., Brough H.A., Eiwegger T., Eljaszewicz A., Eyerich S., Feleszko W., Gomez-Casado C., Hoffmann-Sommergruber K., Janda J., Jimenez-Saiz R., Jutel M., Knol E.F., Kortekaas Krohn I., Kothari A., Makowska J., Moniuszko M., Morita H., O’Mahony L., Nadeau K., Ozdemir C., Pali-Scholl I., Palomares O., Papaleo F., Prunicki M., Schmidt-Weber C.B., Sediva A., Schwarze J., Shamji M.H., Tramper-Stranders G.A., van de Veen W., Untersmayr E. Immunology of COVID-19: mechanisms, clinical outcome, diagnostics, and perspectives-a report of the European Academy of Allergy and Clinical Immunology (EAACI) Allergy. 2020;75(10):2445–2476. doi: 10.1111/all.14462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hu J., Jolkkonen J., Zhao C. Neurotropism of SARS-CoV-2 and its neuropathological alterations: similarities with other coronaviruses. Neurosci. Biobehav. Rev. 2020;119:184–193. doi: 10.1016/j.neubiorev.2020.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kim W.K., Corey S., Alvarez X., Williams K. Monocyte/macrophage traffic in HIV and SIV encephalitis. J. Leukoc. Biol. 2003;74(5):650–656. doi: 10.1189/jlb.0503207. [DOI] [PubMed] [Google Scholar]
- 51.Pellegrini L., Albecka A., Mallery D.L., Kellner M.J., Paul D., Carter A.P., James L.C., Lancaster M.A. SARS-CoV-2 infects the brain choroid plexus and disrupts the blood-CSF barrier in human brain organoids. Cell Stem Cell. 2020;27(6):951–961. doi: 10.1016/j.stem.2020.10.001. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Guo Y., Korteweg C., McNutt M.A., Gu J. Pathogenetic mechanisms of severe acute respiratory syndrome. Virus Res. 2008;133(1):4–12. doi: 10.1016/j.virusres.2007.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.van Riel D., Verdijk R., Kuiken T. The olfactory nerve: a shortcut for influenza and other viral diseases into the central nervous system. J. Pathol. 2015;235(2):277–287. doi: 10.1002/path.4461. [DOI] [PubMed] [Google Scholar]
- 54.Conde Cardona G., Quintana Pajaro L.D., Quintero Marzola I.D., Ramos Villegas Y., Moscote Salazar L.R. Neurotropism of SARS-CoV 2: mechanisms and manifestations. J. Neurol. Sci. 2020;412 doi: 10.1016/j.jns.2020.116824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Meinhardt J., Radke J., Dittmayer C., Franz J., Thomas C., Mothes R., Laue M., Schneider J., Brunink S., Greuel S., Lehmann M., Hassan O., Aschman T., Schumann E., Chua R.L., Conrad C., Eils R., Stenzel W., Windgassen M., Rossler L., Goebel H.H., Gelderblom H.R., Martin H., Nitsche A., Schulz-Schaeffer W.J., Hakroush S., Winkler M.S., Tampe B., Scheibe F., Kortvelyessy P., Reinhold D., Siegmund B., Kuhl A.A., Elezkurtaj S., Horst D., Oesterhelweg L., Tsokos M., Ingold-Heppner B., Stadelmann C., Drosten C., Corman V.M., Radbruch H., Heppner F.L. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat. Neurosci. 2020 doi: 10.1038/s41593-020-00758-5. [DOI] [PubMed] [Google Scholar]
- 56.Alenina N., Bader M. ACE2 in brain physiology and pathophysiology: evidence from transgenic animal models. Neurochem. Res. 2019;44(6):1323–1329. doi: 10.1007/s11064-018-2679-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Diz D.I., Garcia-Espinosa M.A., Gegick S., Tommasi E.N., Ferrario C.M., Ann Tallant E., Chappell M.C., Gallagher P.E. Injections of angiotensin-converting enzyme 2 inhibitor MLN4760 into nucleus tractus solitarii reduce baroreceptor reflex sensitivity for heart rate control in rats. Exp. Physiol. 2008;93(5):694–700. doi: 10.1113/expphysiol.2007.040261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Xu P., Sriramula S., Lazartigues E. ACE2/ANG-(1-7)/Mas pathway in the brain: the axis of good. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011;300(4):R804–R817. doi: 10.1152/ajpregu.00222.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.South A.M., Diz D.I., Chappell M.C. COVID-19, ACE2, and the cardiovascular consequences. Am. J. Physiol. Heart Circ. Physiol. 2020;318(5):H1084–H1090. doi: 10.1152/ajpheart.00217.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Manganelli F., Vargas M., Iovino A., Iacovazzo C., Santoro L., Servillo G. Brainstem involvement and respiratory failure in COVID-19. Neurol. Sci. 2020;41(7):1663–1665. doi: 10.1007/s10072-020-04487-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cantuti-Castelvetri L., Ojha R., Pedro L.D., Djannatian M., Franz J., Kuivanen S., van der Meer F., Kallio K., Kaya T., Anastasina M., Smura T., Levanov L., Szirovicza L., Tobi A., Kallio-Kokko H., Osterlund P., Joensuu M., Meunier F.A., Butcher S.J., Winkler M.S., Mollenhauer B., Helenius A., Gokce O., Teesalu T., Hepojoki J., Vapalahti O., Stadelmann C., Balistreri G., Simons M. Neuropilin-1 facilitates SARS-CoV-2 cell entry and infectivity. Science. 2020;370(6518):856–860. doi: 10.1126/science.abd2985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hikmet F., Mear L., Edvinsson A., Micke P., Uhlen M., Lindskog C. The protein expression profile of ACE2 in human tissues. Mol. Syst. Biol. 2020;16(7) doi: 10.15252/msb.20209610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sun T., Guan J. Novel coronavirus and the central nervous system. Eur. J. Neurol. 2020;27 doi: 10.1111/ene.14227. [DOI] [PubMed] [Google Scholar]
- 64.Tassorelli C., Mojoli F., Baldanti F., Bruno R., Benazzo M. COVID-19: what if the brain had a role in causing the deaths? Eur. J. Neurol. 2020;27 doi: 10.1111/ene.14275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xia H., Lazartigues E. Angiotensin-converting enzyme 2 in the brain: properties and future directions. J. Neurochem. 2008;107(6):1482–1494. doi: 10.1111/j.1471-4159.2008.05723.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Logmin K., Karam M., Schichel T., Harmel J., Wojtecki L. Non-epileptic seizures in autonomic dysfunction as the initial symptom of COVID-19. J. Neurol. 2020;267:2490–2491. doi: 10.1007/s00415-020-09904-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Benghanem S., Mazeraud A., Azabou E., Chhor V., Shinotsuka C.R., Claassen J., Rohaut B., Sharshar T. Brainstem dysfunction in critically ill patients. Crit. Care. 2020;24(1):5. doi: 10.1186/s13054-019-2718-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Abdelaziz O.S., Waffa Z. Neuropathogenic human coronaviruses: a review. Rev. Med. Virol. 2020;30 doi: 10.1002/rmv.2118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen Z., Venkat P., Seyfried D., Chopp M., Yan T., Chen J. Brain-heart interaction: cardiac complications after stroke. Circ. Res. 2017;121(4):451–468. doi: 10.1161/CIRCRESAHA.117.311170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Armando I., Seltzer A., Bregonzio C., Saavedra J.M. Stress and angiotensin II: novel therapeutic opportunities. Curr. Drug Targets CNS Neurol. Disord. 2003;2(6):413–419. doi: 10.2174/1568007033482661. [DOI] [PubMed] [Google Scholar]
- 71.Goldstein D.S. The extended autonomic system, dyshomeostasis, and COVID-19. Clin. Auton. Res. 2020;30(4):299–315. doi: 10.1007/s10286-020-00714-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Porzionato A., Emmi A., Barbon S., Boscolo-Berto R., Stecco C., Stocco E., Macchi V., De Caro R. Sympathetic activation: a potential link between comorbidities and COVID-19. FEBS J. 2020;287:3681–3688. doi: 10.1111/febs.15481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hans-Peter K. Posttraumatic stress disorder in survivors of acute respiratory distress syndrome (ARDS) and septic shock. Psychosom. Kons. 2008;2(4):220. doi: 10.1007/s11800-008-0129-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Rogers J.P., Chesney E., Oliver D., Pollak T.A., McGuire P., Fusar-Poli P., Zandi M.S., Lewis G., David A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7(7):611–627. doi: 10.1016/S2215-0366(20)30203-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Akosile W., Colquhoun D., Young R., Lawford B., Voisey J. The association between post-traumatic stress disorder and coronary artery disease: a meta-analysis. Australas. Psychiatry Bull. R. Aust. N. Z. Coll. Psychiatr. 2018;26(5):524–530. doi: 10.1177/1039856218789779. [DOI] [PubMed] [Google Scholar]
- 76.Scherrer J.F., Salas J., Cohen B.E., Schnurr P.P., Schneider F.D., Chard K.M., Tuerk P., Friedman M.J., Norman S.B., van den Berk-Clark C., Lustman P.J. Comorbid conditions explain the association between posttraumatic stress disorder and incident cardiovascular disease. J. Am. Heart Assoc. 2019;8(4) doi: 10.1161/JAHA.118.011133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Vaccarino V., Goldberg J., Rooks C., Shah A.J., Veledar E., Faber T.L., Votaw J.R., Forsberg C.W., Bremner J.D. Post-traumatic stress disorder and incidence of coronary heart disease: a twin study. J. Am. Coll. Cardiol. 2013;62(11):970–978. doi: 10.1016/j.jacc.2013.04.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Song H., Fang F., Arnberg F.K., Mataix-Cols D., Fernandez de la Cruz L., Almqvist C., Fall K., Lichtenstein P., Thorgeirsson G., Valdimarsdottir U.A. Stress related disorders and risk of cardiovascular disease: population based, sibling controlled cohort study. BMJ. 2019;365:l1255. doi: 10.1136/bmj.l1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Spitzer C., Koch B., Grabe H.J., Ewert R., Barnow S., Felix S.B., Ittermann T., Obst A., Volzke H., Glaser S., Schaper C. Association of airflow limitation with trauma exposure and post-traumatic stress disorder. Eur. Respir. J. 2011;37(5):1068–1075. doi: 10.1183/09031936.00028010. [DOI] [PubMed] [Google Scholar]
- 80.Edmondson D., von Kanel R. Post-traumatic stress disorder and cardiovascular disease. Lancet Psychiatry. 2017;4(4):320–329. doi: 10.1016/S2215-0366(16)30377-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Vickrey B.G., Williams L.S. Posttraumatic stress disorder after cerebrovascular events: broadening the landscape of psychological assessment in stroke and transient ischemic attack. Stroke. 2014;45(11):3182–3183. doi: 10.1161/STROKEAHA.114.006865. [DOI] [PubMed] [Google Scholar]
- 82.Turner J.H., Neylan T.C., Schiller N.B., Li Y., Cohen B.E. Objective evidence of myocardial ischemia in patients with posttraumatic stress disorder. Biol. Psychiatry. 2013;74(11):861–866. doi: 10.1016/j.biopsych.2013.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Mazzeo A.T., Guaraldi F., Filippini C., Tesio R., Settanni F., Lucchiari M., Mengozzi G., Grottoli S., Ghigo E., Mascia L. Activation of pituitary axis according to underlying critical illness and its effect on outcome. J. Crit. Care. 2019;54:22–29. doi: 10.1016/j.jcrc.2019.07.006. [DOI] [PubMed] [Google Scholar]
- 84.Dallman M.F., Pecoraro N., Akana S.F., La Fleur S.E., Gomez F., Houshyar H., Bell M.E., Bhatnagar S., Laugero K.D., Manalo S. Chronic stress and obesity: a new view of “comfort food”. Proc. Natl. Acad. Sci. USA. 2003;100(20):11696–11701. doi: 10.1073/pnas.1934666100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Saavedra J.M., Chevillard C. Angiotensin-converting enzyme is present in the subfornical organ and other circumventricular organs of the rat. Neurosci. Lett. 1982;29(2):123–127. doi: 10.1016/0304-3940(82)90340-8. [DOI] [PubMed] [Google Scholar]
- 86.Chevillard C., Saavedra J.M. Distribution of angiotensin-converting enzyme activity in specific areas of the rat brain stem. J. Neurochem. 1982;38(1):281–284. doi: 10.1111/j.1471-4159.1982.tb10883.x. [DOI] [PubMed] [Google Scholar]
- 87.Strittmatter S.M., Lo M.M., Javitch J.A., Snyder S.H. Autoradiographic visualization of angiotensin-converting enzyme in rat brain with [3H]captopril: localization to a striatonigral pathway. Proc. Natl. Acad. Sci. USA. 1984;81(5):1599–1603. doi: 10.1073/pnas.81.5.1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Correa F.M., Plunkett L.M., Saavedra J.M. Quantitative distribution of angiotensin-converting enzyme (kininase II) in discrete areas of the rat brain by autoradiography with computerized microdensitometry. Brain Res. 1986;375(2):259–266. doi: 10.1016/0006-8993(86)90746-8. [DOI] [PubMed] [Google Scholar]
- 89.Santos R.A.S., Oudit G.Y., Verano-Braga T., Canta G., Steckelings U.M., Bader M. The renin-angiotensin system: going beyond the classical paradigms. Am. J. Physiol. Heart Circ. Physiol. 2019;316(5):H958–H970. doi: 10.1152/ajpheart.00723.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Vaduganathan M., van Meijgaard J., Mehra M.R., Joseph J., O’Donnell C.J., Warraich H.J. Prescription fill patterns for commonly used drugs during the COVID-19 pandemic in the United States. JAMA. 2020;323:2524. doi: 10.1001/jama.2020.9184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pagliaro P., Penna C. ACE/ACE2 ratio: a key also in 2019 coronavirus disease (Covid-19)? Front. Med. 2020;7:335. doi: 10.3389/fmed.2020.00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Iaccarino G., Grassi G., Borghi C., Ferri C., Salvetti M., Volpe M., S.-R. Investigators Age and multimorbidity predict death among COVID-19 patients: results of the SARS-RAS study of the Italian Society of Hypertension. Hypertension. 2020;76(2):366–372. doi: 10.1161/HYPERTENSIONAHA.120.15324. [DOI] [PubMed] [Google Scholar]
- 93.Yousaf Z., Al-Shokri S.D., Al-Soub H., Mohamed M.F.H. COVID-19-associated SIADH: a clue in the times of pandemic! Am. J. Physiol. Endocrinol. Metab. 2020;318(6):E882–E885. doi: 10.1152/ajpendo.00178.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Hori H., Kim Y. Inflammation and post-traumatic stress disorder. Psychiatry Clin. Neurosci. 2019;73(4):143–153. doi: 10.1111/pcn.12820. [DOI] [PubMed] [Google Scholar]
- 95.Guzik T.J., Mohiddin S.A., Dimarco A., Patel V., Savvatis K., Marelli-Berg F.M., Madhur M.S., Tomaszewski M., Maffia P., D’Acquisto F., Nicklin S.A., Marian A.J., Nosalski R., Murray E.C., Guzik B., Berry C., Touyz R.M., Kreutz R., Wang D.W., Bhella D., Sagliocco O., Crea F., Thomson E.C., McInnes I.B. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020;116:1666–1687. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lazzerini P.E., Capecchi P.L., Laghi-Pasini F. Systemic inflammation and arrhythmic risk: lessons from rheumatoid arthritis. Eur. Heart J. 2017;38(22):1717–1727. doi: 10.1093/eurheartj/ehw208. [DOI] [PubMed] [Google Scholar]
- 97.Chen J., Yin D., He X., Gao M., Choi Y., Luo G., Wang H., Qu X. Modulation of activated astrocytes in the hypothalamus paraventricular nucleus to prevent ventricular arrhythmia complicating acute myocardial infarction. Int. J. Cardiol. 2020;308:33–41. doi: 10.1016/j.ijcard.2020.01.035. [DOI] [PubMed] [Google Scholar]
- 98.Borovikova L.V., Ivanova S., Zhang M., Yang H., Botchkina G.I., Watkins L.R., Wang H., Abumrad N., Eaton J.W., Tracey K.J. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000;405(6785):458–462. doi: 10.1038/35013070. [DOI] [PubMed] [Google Scholar]
- 99.Russo P., Bonassi S., Giacconi R., Malavolta M., Tomino C., Maggi F. COVID-19 and smoking: is nicotine the hidden link? Eur. Respir. J. 2020;55(6) doi: 10.1183/13993003.01116-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Farsalinos K., Angelopoulou A., Alexandris N., Poulas K. COVID-19 and the nicotinic cholinergic system. Eur. Respir. J. 2020;56(1) doi: 10.1183/13993003.01589-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Guerra G., Lucariello A., Perna A., Botta L., De Luca A., Moccia F. The role of endothelial Ca2+ signaling in neurovascular coupling: a view from the lumen. Int. J. Mol. Sci. 2018;19(4):938. doi: 10.3390/ijms19040938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mapelli L., Gagliano G., Soda T., Laforenza U., Moccia F., D’Angelo E.U. Granular layer neurons control cerebellar neurovascular coupling through an NMDA receptor/NO-dependent system. J. Neurosci. 2017;37(5):1340–1351. doi: 10.1523/JNEUROSCI.2025-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Kim K.J., Ramiro Diaz J., Iddings J.A., Filosa J.A. Vasculo-neuronal coupling: retrograde vascular communication to brain neurons. J. Neurosci. 2016;36(50):12624–12639. doi: 10.1523/JNEUROSCI.1300-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Moore C.I., Cao R. The hemo-neural hypothesis: on the role of blood flow in information processing. J. Neurophysiol. 2008;99(5):2035–2047. doi: 10.1152/jn.01366.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Hall C.N., Reynell C., Gesslein B., Hamilton N.B., Mishra A., Sutherland B.A., O’Farrell F.M., Buchan A.M., Lauritzen M., Attwell D. Capillary pericytes regulate cerebral blood flow in health and disease. Nature. 2014;508(7494):55–60. doi: 10.1038/nature13165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hogan-Cann A.D., Lu P., Anderson C.M. Endothelial NMDA receptors mediate activity-dependent brain hemodynamic responses in mice. Proc. Natl. Acad. Sci. USA. 2019;116:10229–10231. doi: 10.1073/pnas.1902647116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Longden T.A., Dabertrand F., Koide M., Gonzales A.L., Tykocki N.R., Brayden J.E., Hill-Eubanks D., Nelson M.T. Capillary K(+)-sensing initiates retrograde hyperpolarization to increase local cerebral blood flow. Nat. Neurosci. 2017;20(5):717–726. doi: 10.1038/nn.4533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Negri S., Faris P., Pellavio G., Botta L., Orgiu M., Forcaia G., Sancini G., Laforenza U., Moccia F. Group 1 metabotropic glutamate receptors trigger glutamate-induced intracellular Ca(2+) signals and nitric oxide release in human brain microvascular endothelial cells. Cell Mol. Life Sci. 2019 doi: 10.1007/s00018-019-03284-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zuccolo E., Kheder D.A., Lim D., Perna A., Nezza F.D., Botta L., Scarpellino G., Negri S., Martinotti S., Soda T., Forcaia G., Riboni L., Ranzato E., Sancini G., Ambrosone L., D’Angelo E., Guerra G., Moccia F. Glutamate triggers intracellular Ca(2+) oscillations and nitric oxide release by inducing NAADP- and InsP3-dependent Ca(2+) release in mouse brain endothelial cells. J. Cell. Physiol. 2019;234(4):3538–3554. doi: 10.1002/jcp.26953. [DOI] [PubMed] [Google Scholar]
- 110.Tapella L., Soda T., Mapelli L., Bortolotto V., Bondi H., Ruffinatti F.A., Dematteis G., Stevano A., Dionisi M., Ummarino S., Di Ruscio A., Distasi C., Grilli M., Genazzani A.A., D’Angelo E., Moccia F., Lim D. Deletion of calcineurin from GFAP-expressing astrocytes impairs excitability of cerebellar and hippocampal neurons through astroglial Na(+) /K(+) ATPase. Glia. 2020;68(3):543–560. doi: 10.1002/glia.23737. [DOI] [PubMed] [Google Scholar]
- 111.Hopper R.A., Garthwaite J. Tonic and phasic nitric oxide signals in hippocampal long-term potentiation. J. Neurosci. 2006;26(45):11513–11521. doi: 10.1523/JNEUROSCI.2259-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Haul S., Godecke A., Schrader J., Haas H.L., Luhmann H.J. Impairment of neocortical long-term potentiation in mice deficient of endothelial nitric oxide synthase. J. Neurophysiol. 1999;81(2):494–497. doi: 10.1152/jn.1999.81.2.494. [DOI] [PubMed] [Google Scholar]
- 113.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., Mehra M.R., Schuepbach R.A., Ruschitzka F., Moch H. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Yin X.X., Zheng X.R., Peng W., Wu M.L., Mao X.Y. Vascular endothelial growth factor (VEGF) as a vital target for brain inflammation during the COVID-19 outbreak. ACS Chem. Neurosci. 2020;11:1704–1705. doi: 10.1021/acschemneuro.0c00294. [DOI] [PubMed] [Google Scholar]
- 115.Katychev A., Wang X., Duffy A., Dore-Duffy P. Glucocorticoid-induced apoptosis in CNS microvascular pericytes. Dev. Neurosci. 2003;25(6):436–446. doi: 10.1159/000075669. [DOI] [PubMed] [Google Scholar]
- 116.Chen L., Li X., Chen M., Feng Y., Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovasc. Res. 2020;116(6):1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Romoli M., Jelcic I., Bernard-Valnet R., Garcia Azorin D., Mancinelli L., Akhvlediani T., Monaco S., Taba P., Sellner J. A systematic review of neurological manifestations of SARS‐CoV‐2 infection: the devil is hidden in the details. Eur. J. Neurol. 2020;27:1712–1726. doi: 10.1111/ene.14382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bostanciklioglu M. SARS-CoV2 entry and spread in the lymphatic drainage system of the brain. Brain Behav. Immun. 2020;87:122–123. doi: 10.1016/j.bbi.2020.04.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Louveau A., Herz J., Alme M.N., Salvador A.F., Dong M.Q., Viar K.E., Herod S.G., Knopp J., Setliff J.C., Lupi A.L., Da Mesquita S., Frost E.L., Gaultier A., Harris T.H., Cao R., Hu S., Lukens J.R., Smirnov I., Overall C.C., Oliver G., Kipnis J. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat. Neurosci. 2018;21(10):1380–1391. doi: 10.1038/s41593-018-0227-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.〈https://globalhealth5050.org/covid19/〉, Sex, Gender and Covid-19, 2020.
- 121.〈https://www.iss.it/coronavirus〉, Italian Institute of Health, 2020.
- 122.Shi S., Qin M., Shen B., Cai Y., Liu T., Yang F., Gong W., Liu X., Liang J., Zhao Q., Huang H., Yang B., Huang C. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Mayor E. Gender roles and traits in stress and health. Front. Psychol. 2015;6:779. doi: 10.3389/fpsyg.2015.00779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Goldstein J.M., Hale T., Foster S.L., Tobet S.A., Handa R.J. Sex differences in major depression and comorbidity of cardiometabolic disorders: impact of prenatal stress and immune exposures. Neuropsychopharmacology. 2019;44(1):59–70. doi: 10.1038/s41386-018-0146-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.de Morais S.D.B., Shanks J., Zucker I.H. Integrative physiological aspects of brain RAS in hypertension. Curr. Hypertens. Rep. 2018;20(2):10. doi: 10.1007/s11906-018-0810-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Penna C., Mercurio V., Tocchetti C.G., Pagliaro P. Sex-related differences in COVID-19 lethality. Br. J. Pharmacol. 2020 doi: 10.1111/bph.15207. bph.15207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Tomlins S.A., Rhodes D.R., Perner S., Dhanasekaran S.M., Mehra R., Sun X.W., Varambally S., Cao X., Tchinda J., Kuefer R., Lee C., Montie J.E., Shah R.B., Pienta K.J., Rubin M.A., Chinnaiyan A.M. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science. 2005;310(5748):644–648. doi: 10.1126/science.1117679. [DOI] [PubMed] [Google Scholar]
- 128.Pelliccia F., Kaski J.C., Crea F., Camici P.G. Pathophysiology of Takotsubo syndrome. Circulation. 2017;135(24):2426–2441. doi: 10.1161/CIRCULATIONAHA.116.027121. [DOI] [PubMed] [Google Scholar]
- 129.Boyd B., Solh T. Takotsubo cardiomyopathy: review of broken heart syndrome. JAAPA Off. J. Am. Acad. Physician Assist. 2020;33(3):24–29. doi: 10.1097/01.JAA.0000654368.35241.fc. [DOI] [PubMed] [Google Scholar]
- 130.Paur H., Wright P.T., Sikkel M.B., Tranter M.H., Mansfield C., O’Gara P., Stuckey D.J., Nikolaev V.O., Diakonov I., Pannell L., Gong H., Sun H., Peters N.S., Petrou M., Zheng Z., Gorelik J., Lyon A.R., Harding S.E. High levels of circulating epinephrine trigger apical cardiodepression in a beta2-adrenergic receptor/Gi-dependent manner: a new model of Takotsubo cardiomyopathy. Circulation. 2012;126(6):697–706. doi: 10.1161/CIRCULATIONAHA.112.111591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Sala S., Peretto G., Gramegna M., Palmisano A., Villatore A., Vignale D., De Cobelli F., Tresoldi M., Cappelletti A.M., Basso C., Godino C., Esposito A. Acute myocarditis presenting as a reverse Tako-Tsubo syndrome in a patient with SARS-CoV-2 respiratory infection. Eur. Heart J. 2020;41(19):1861–1862. doi: 10.1093/eurheartj/ehaa286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Okura H. Update of takotsubo syndrome in the era of COVID-19. J. Cardiol. 2020 doi: 10.1016/j.jjcc.2020.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C., Hohmann E., Chu H.Y., Luetkemeyer A., Kline S., Lopez de Castilla D., Finberg R.W., Dierberg K., Tapson V., Hsieh L., Patterson T.F., Paredes R., Sweeney D.A., Short W.R., Touloumi G., Lye D.C., Ohmagari N., Oh M.D., Ruiz-Palacios G.M., Benfield T., Fatkenheuer G., Kortepeter M.G., Atmar R.L., Creech C.B., Lundgren J., Babiker A.G., Pett S., Neaton J.D., Burgess T.H., Bonnett T., Green M., Makowski M., Osinusi A., Nayak S., Lane H.C., Members A.-S.G. Remdesivir for the treatment of Covid-19 - final report. N. Engl. J. Med. 2020;383(19):1813–1826. doi: 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Wang S., Peng Y., Wang R., Jiao S., Wang M., Huang W., Shan C., Jiang W., Li Z., Gu C., Chen B., Hu X., Yao Y., Min J., Zhang H., Chen Y., Gao G., Tang P., Li G., Wang A., Wang L., Zhang J., Chen S., Gui X., Yuan Z., Liu D. Characterization of neutralizing antibody with prophylactic and therapeutic efficacy against SARS-CoV-2 in rhesus monkeys. Nat. Commun. 2020;11(1):5752. doi: 10.1038/s41467-020-19568-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Akhmerov A., Marban E. COVID-19 and the heart. Circ. Res. 2020;126(10):1443–1455. doi: 10.1161/CIRCRESAHA.120.317055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Theoharides T.C., Conti P. Dexamethasone for COVID-19? not so fast. J. Biol. Regul. Homeost. Agents. 2020;34(3) doi: 10.23812/20-EDITORIAL_1-5. [DOI] [PubMed] [Google Scholar]
- 137.Salton F., Confalonieri P., Meduri G.U., Santus P., Harari S., Scala R., Lanini S., Vertui V., Oggionni T., Caminati A., Patruno V., Tamburrini M., Scartabellati A., Parati M., Villani M., Radovanovic D., Tomassetti S., Ravaglia C., Poletti V., Vianello A., Gaccione A.T., Guidelli L., Raccanelli R., Lucernoni P., Lacedonia D., Foschino Barbaro M.P., Centanni S., Mondoni M., Davi M., Fantin A., Cao X., Torelli L., Zucchetto M., Montico M., Casarin A., Romagnoli M., Gasparini S., Bonifazi M., D’Agaro P., Marcello A., Licastro D., Ruaro B., Volpe M.C., Umberger R., Confalonieri M. Prolonged low-dose methylprednisolone in patients with severe COVID-19 pneumonia. Open Forum Infect. Dis. 2020;7(10) doi: 10.1093/ofid/ofaa421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Hu H., Ma F., Wei X., Fang Y. Coronavirus fulminant myocarditis treated with glucocorticoid and human immunoglobulin. Eur. Heart J. 2021;42(2):206. doi: 10.1093/eurheartj/ehaa190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Scavone C., Brusco S., Bertini M., Sportiello L., Rafaniello C., Zoccoli A., Berrino L., Racagni G., Rossi F., Capuano A. Current pharmacological treatments for COVID-19: what’s next? Br. J. Pharmacol. 2020;177:4813–4824. doi: 10.1111/bph.15072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Mohebbi N., Talebi A., Moghadamnia M., Nazari Taloki Z., Shakiba A. Drug interactions of psychiatric and COVID-19 medications. Basic Clin. Neurosci. 2020;11(2):185–200. doi: 10.32598/bcn.11.covid19.2500.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Terrasini N., Lionetti V. Exosomes in critical illness. Crit. Care Med. 2017;45(6):1054–1060. doi: 10.1097/CCM.0000000000002328. [DOI] [PubMed] [Google Scholar]
- 142.Carrozzo A., Casieri V., Di Silvestre D., Brambilla F., De Nitto E., Sardaro N., Papini G., Storti S., Settanni G., Solinas M., Mauri P., Paparella D., Lionetti V. Plasma exosomes characterization reveals a perioperative protein signature in older patients undergoing different types of on-pump cardiac surgery. GeroScience. 2020 doi: 10.1007/s11357-020-00223-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Dickens A.M., Tovar Y.R.L.B., Yoo S.W., Trout A.L., Bae M., Kanmogne M., Megra B., Williams D.W., Witwer K.W., Gacias M., Tabatadze N., Cole R.N., Casaccia P., Berman J.W., Anthony D.C., Haughey N.J. Astrocyte-shed extracellular vesicles regulate the peripheral leukocyte response to inflammatory brain lesions. Sci. Signal. 2017;10(473) doi: 10.1126/scisignal.aai7696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Chen J., Cui C., Yang X., Xu J., Venkat P., Zacharek A., Yu P., Chopp M. MiR-126 affects brain-heart interaction after cerebral ischemic stroke. Transl. Stroke Res. 2017;8(4):374–385. doi: 10.1007/s12975-017-0520-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Venkat P., Chen J., Chopp M. Exosome-mediated amplification of endogenous brain repair mechanisms and brain and systemic organ interaction in modulating neurological outcome after stroke. J. Cereb. Blood Flow Metab. 2018;38(12):2165–2178. doi: 10.1177/0271678X18782789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Otero-Ortega L., Gomez de Frutos M.C., Laso-Garcia F., Rodriguez-Frutos B., Medina-Gutierrez E., Lopez J.A., Vazquez J., Diez-Tejedor E., Gutierrez-Fernandez M. Exosomes promote restoration after an experimental animal model of intracerebral hemorrhage. J. Cereb. Blood Flow Metab. 2018;38(5):767–779. doi: 10.1177/0271678X17708917. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 147.Barile L., Moccetti T., Marban E., Vassalli G. Roles of exosomes in cardioprotection. Eur. Heart J. 2017;38(18):1372–1379. doi: 10.1093/eurheartj/ehw304. [DOI] [PubMed] [Google Scholar]
- 148.Barile L., Cervio E., Lionetti V., Milano G., Ciullo A., Biemmi V., Bolis S., Altomare C., Matteucci M., Di Silvestre D., Brambilla F., Fertig T.E., Torre T., Demertzis S., Mauri P., Moccetti T., Vassalli G. Cardioprotection by cardiac progenitor cell-secreted exosomes: role of pregnancy-associated plasma protein-A. Cardiovasc. Res. 2018;114(7):992–1005. doi: 10.1093/cvr/cvy055. [DOI] [PubMed] [Google Scholar]
- 149.Davidson S.M., Andreadou I., Barile L., Birnbaum Y., Cabrera-Fuentes H.A., Cohen M.V., Downey J.M., Girao H., Pagliaro P., Penna C., Pernow J., Preissner K.T., Ferdinandy P. Circulating blood cells and extracellular vesicles in acute cardioprotection. Cardiovasc. Res. 2019;115(7):1156–1166. doi: 10.1093/cvr/cvy314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Barile L., Lionetti V., Cervio E., Matteucci M., Gherghiceanu M., Popescu L.M., Torre T., Siclari F., Moccetti T., Vassalli G. Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc. Res. 2014;103(4):530–541. doi: 10.1093/cvr/cvu167. [DOI] [PubMed] [Google Scholar]
- 151.Balbi C., Lodder K., Costa A., Moimas S., Moccia F., van Herwaarden T., Rosti V., Campagnoli F., Palmeri A., De Biasio P., Santini F., Giacca M., Goumans M.J., Barile L., Smits A.M., Bollini S. Reactivating endogenous mechanisms of cardiac regeneration via paracrine boosting using the human amniotic fluid stem cell secretome. Int. J. Cardiol. 2019;287:87–95. doi: 10.1016/j.ijcard.2019.04.011. [DOI] [PubMed] [Google Scholar]
- 152.Jung F., Kruger-Genge A., Franke R.P., Hufert F., Kupper J.H. COVID-19 and the endothelium. Clin. Hemorheol. Microcirc. 2020;75(1):7–11. doi: 10.3233/CH-209007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Inal J. Complement-mediated extracellular vesicle release as a measure of endothelial dysfunction and prognostic marker for COVID-19 in peripheral blood – letter to the editor. Clin. Hemorheol. Microcirc. 2020;75(4):383–386. doi: 10.3233/CH-200958. [DOI] [PubMed] [Google Scholar]
- 154.Wu A.H.B., Zhang Y., Webber R. Extracellular vesicles released in blood of COVID-19 patients: mechanism for detection of cardiac troponin after myocardial injury? Biomarkers. 2020;25:613–615. doi: 10.1080/1354750X.2020.1829055. [DOI] [PubMed] [Google Scholar]
- 155.Matthay M.A., Calfee C.S., Zhuo H., Thompson B.T., Wilson J.G., Levitt J.E., Rogers A.J., Gotts J.E., Wiener-Kronish J.P., Bajwa E.K., Donahoe M.P., McVerry B.J., Ortiz L.A., Exline M., Christman J.W., Abbott J., Delucchi K.L., Caballero L., McMillan M., McKenna D.H., Liu K.D. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial. Lancet Respir. Med. 2019;7(2):154–162. doi: 10.1016/S2213-2600(18)30418-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Tsuchiya A., Takeuchi S., Iwasawa T., Kumagai M., Sato T., Motegi S., Ishii Y., Koseki Y., Tomiyoshi K., Natsui K., Takeda N., Yoshida Y., Yamazaki F., Kojima Y., Watanabe Y., Kimura N., Tominaga K., Kamimura H., Takamura M., Terai S. Therapeutic potential of mesenchymal stem cells and their exosomes in severe novel coronavirus disease 2019 (COVID-19) cases. Inflamm. Regen. 2020;40:14. doi: 10.1186/s41232-020-00121-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.O’Driscoll L. Extracellular vesicles from mesenchymal stem cells as a Covid-19 treatment. Drug Discov. Today. 2020;25:1124–1125. doi: 10.1016/j.drudis.2020.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Borger V., Weiss D.J., Anderson J.D., Borras F.E., Bussolati B., Carter D.R.F., Dominici M., Falcon-Perez J.M., Gimona M., Hill A.F., Hoffman A.M., de Kleijn D., Levine B.L., Lim R., Lotvall J., Mitsialis S.A., Monguio-Tortajada M., Muraca M., Nieuwland R., Nowocin A., O’Driscoll L., Ortiz L.A., Phinney D.G., Reischl I., Rohde E., Sanzenbacher R., Thery C., Toh W.S., Witwer K.W., Lim S.K., Giebel B. International Society for Extracellular Vesicles and International Society for Cell and Gene Therapy statement on extracellular vesicles from mesenchymal stromal cells and other cells: considerations for potential therapeutic agents to suppress coronavirus disease-19. Cytotherapy. 2020;22:482–485. doi: 10.1016/j.jcyt.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Casieri V., Matteucci M., Pasanisi E.M., Papa A., Barile L., Fritsche-Danielson R., Lionetti V. Ticagrelor enhances release of anti-hypoxic cardiac progenitor cell-derived exosomes through increasing cell proliferation in vitro. Sci. Rep. 2020;10(1):2494. doi: 10.1038/s41598-020-59225-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Agrimi J., Baroni C., Anakor E., Lionetti V. Perioperative heart-brain axis protection in obese surgical patients: the nutrigenomic approach. Curr. Med. Chem. 2020;27(2):258–281. doi: 10.2174/0929867325666181015145225. [DOI] [PubMed] [Google Scholar]
- 161.Lionetti V., Barile L. Perioperative cardioprotection: back to bedside. Minerva Anestesiol. 2020;86(4):445–454. doi: 10.23736/S0375-9393.19.13848-5. [DOI] [PubMed] [Google Scholar]
- 162.Lionetti V., Tuana B.S., Casieri V., Parikh M., Pierce G.N. Importance of functional food compounds in cardioprotection through action on the epigenome. Eur. Heart J. 2019;40(7):575–582. doi: 10.1093/eurheartj/ehy597. [DOI] [PubMed] [Google Scholar]
- 163.Agrimi J., Spalletti C., Baroni C., Keceli G., Zhu G., Caragnano A., Matteucci M., Chelko S., Ramirez-Correa G.A., Bedja D., Casieri V., Di Lascio N., Scalco A., Beltrami A.P., Paolocci N., Caleo M., Lionetti V. Obese mice exposed to psychosocial stress display cardiac and hippocampal dysfunction associated with local brain-derived neurotrophic factor depletion. EBioMedicine. 2019;47:384–401. doi: 10.1016/j.ebiom.2019.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Hascup E.R., Hascup K.N. Does SARS-CoV-2 infection cause chronic neurological complications? GeroScience. 2020;42:1083–1087. doi: 10.1007/s11357-020-00207-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Agrimi J., Spalletti C., Matteucci M., Casieri V., Torelli M., Caleo M., Lionetti V. Long term intake of barley beta-D-glucan attenuates glucose intolerance, mood disorders and cognitive decline in high-fat diet-induced obese mice exposed to chronic psychosocial stress. FASEB J. 2017;31(1) Supplement 298.8. [Google Scholar]
- 166.Kim J., Lee S., Choi B.R., Yang H., Hwang Y., Park J.H., LaFerla F.M., Han J.S., Lee K.W., Kim J. Sulforaphane epigenetically enhances neuronal BDNF expression and TrkB signaling pathways. Mol. Nutr. Food Res. 2017;61(2) doi: 10.1002/mnfr.201600194. [DOI] [PubMed] [Google Scholar]
- 167.Dong Y., Xu M., Kalueff A.V., Song C. Dietary eicosapentaenoic acid normalizes hippocampal omega-3 and 6 polyunsaturated fatty acid profile, attenuates glial activation and regulates BDNF function in a rodent model of neuroinflammation induced by central interleukin-1beta administration. Eur. J. Nutr. 2018;57(5):1781–1791. doi: 10.1007/s00394-017-1462-7. [DOI] [PubMed] [Google Scholar]
- 168.Savi M., Bocchi L., Sala R., Frati C., Lagrasta C., Madeddu D., Falco A., Pollino S., Bresciani L., Miragoli M., Zaniboni M., Quaini F., Del Rio D., Stilli D. Parenchymal and stromal cells contribute to pro-inflammatory myocardial environment at early stages of diabetes: protective role of resveratrol. Nutrients. 2016;8(11):729. doi: 10.3390/nu8110729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Marinella M.A. Indomethacin and resveratrol as potential treatment adjuncts for SARS-CoV-2/COVID-19. Int. J. Clin. Pract. 2020;74 doi: 10.1111/ijcp.13535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Lin S.C., Ho C.T., Chuo W.H., Li S., Wang T.T., Lin C.C. Effective inhibition of MERS-CoV infection by resveratrol. BMC Infect. Dis. 2017;17(1):144. doi: 10.1186/s12879-017-2253-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Fernandes G.F.S., Silva G.D.B., Pavan A.R., Chiba D.E., Chin C.M., Dos Santos J.L. Epigenetic regulatory mechanisms induced by resveratrol. Nutrients. 2017;9(11):1201. doi: 10.3390/nu9111201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Rahvar M., Nikseresht M., Shafiee S.M., Naghibalhossaini F., Rasti M., Panjehshahin M.R., Owji A.A. Effect of oral resveratrol on the BDNF gene expression in the hippocampus of the rat brain. Neurochem. Res. 2011;36(5):761–765. doi: 10.1007/s11064-010-0396-8. [DOI] [PubMed] [Google Scholar]
- 173.Dampf Stone A., Batie S.F., Sabir M.S., Jacobs E.T., Lee J.H., Whitfield G.K., Haussler M.R., Jurutka P.W. Resveratrol potentiates vitamin D and nuclear receptor signaling. J. Cell. Biochem. 2015;116(6):1130–1143. doi: 10.1002/jcb.25070. [DOI] [PubMed] [Google Scholar]
- 174.Cheng J., Rui Y., Qin L., Xu J., Han S., Yuan L., Yin X., Wan Z. Vitamin D combined with resveratrol prevents cognitive decline in SAMP8 mice. Curr. Alzheimer Res. 2017;14(8):820–833. doi: 10.2174/1567205014666170207093455. [DOI] [PubMed] [Google Scholar]
- 175.Safari F., Zarei F., Shekarforoush S., Fekri A., Klishadi M.S., Hekmatimoghaddam S. Combined 1,25-dihydroxy-vitamin D and resveratrol: a novel therapeutic approach to ameliorate ischemia reperfusion-induced myocardial injury. Int. J. Vitam. Nutr. Res. 2015;85(3–4):174–184. doi: 10.1024/0300-9831/a000236. [DOI] [PubMed] [Google Scholar]
- 176.Clodoveo M.L., Di Lorenzo L., Sabba C., Moschetta A., Gesualdo L., Corbo F. The emerging discipline of precision cooking: a suitable tool for the precision nutrition. Int. J. Food Sci. Nutr. 2020;71(4):525–528. doi: 10.1080/09637486.2019.1679726. [DOI] [PubMed] [Google Scholar]
- 177.Alkhatib A. Antiviral functional foods and exercise lifestyle prevention of coronavirus. Nutrients. 2020;12(9):2633. doi: 10.3390/nu12092633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Abian O., Ortega-Alarcon D., Jimenez-Alesanco A., Ceballos-Laita L., Vega S., Reyburn H.T., Rizzuti B., Velazquez-Campoy A. Structural stability of SARS-CoV-2 3CLpro and identification of quercetin as an inhibitor by experimental screening. Int. J. Biol. Macromol. 2020;164:1693–1703. doi: 10.1016/j.ijbiomac.2020.07.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Yang Y., Lv J., Jiang S., Ma Z., Wang D., Hu W., Deng C., Fan C., Di S., Sun Y., Yi W. The emerging role of toll-like receptor 4 in myocardial inflammation. Cell Death Dis. 2016;7 doi: 10.1038/cddis.2016.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Demeco A., Marotta N., Barletta M., Pino I., Marinaro C., Petraroli A., Moggio L., Ammendolia A. Rehabilitation of patients post-COVID-19 infection: a literature review. J. Int. Med. Res. 2020;48(8) doi: 10.1177/0300060520948382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.De Sousa R.A.L., Improta-Caria A.C., Aras-Junior R., de Oliveira E.M., Soci U.P.R., Cassilhas R.C. Physical exercise effects on the brain during COVID-19 pandemic: links between mental and cardiovascular health. Neurol. Sci. 2021;42:1325–1334. doi: 10.1007/s10072-021-05082-9. [DOI] [PMC free article] [PubMed] [Google Scholar]