Abstract
Emerging research on severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) shows that it is spreading to multiple organs in addition to the respiratory system. Though the SARS-CoV2 enters the human body by binding to ACE2 receptors on pulmonary alveolar cells, recent studies indicate that it is spreading to the central nervous system, cardiac and skeletal muscles leading to various pathological conditions in these organs. In particular, the effects of SARS-CoV-2 on triggering the cytokine storm and its consequential effects on skeletal muscles has generated a lot of discussion. The effects of this virus on muscular function especially in susceptible elderly populations is still being explored. However, its effects on diaphragm, a respiratory muscle which plays an important role in determining lung capacity are not completely explored. Currently, as new evidence on using lung ultrasounds to confirm COVID-19 diagnosis is gaining traction, it is necessary to explore the role of diaphragm in treating COVID-19 patients. This article will review the effects of cytokine storm triggered by the SARS-CoV-2 and its resultant effects on skeletal muscle with a specific focus on the diaphragm in order to identify knowledge gaps in effectively treating COVID-19 patients, especially those who are on a mechanical ventilator.
Keywords: COVID-19, Cytokine storm, Skeletal muscle, Diaphragm
Graphical abstract
1. Introduction
Coronavirus disease-2019 (COVID-19) which began in December 2019 in China has caused an unprecedented global pandemic posing the most serious threat to global health. Though not much was known about COVID-19 in the beginning, the past year provided critical insights in understanding the specific nature of this SARS-CoV-2 virus. According to the World Health Organization (WHO), this virus has affected 119.6 million people that resulted in more than 2.64 million deaths around the world by March 15th, 2021 (Coronavirus disease (, 2019). It is now established that SARS-CoV-2 uses the angiotensin converting enzyme 2 (ACE2) as a functional receptor for its entry into pulmonary alveolar cells and induces acute respiratory distress syndrome (ARDS), a life-threatening condition characterized by poor oxygenation and pulmonary infiltration (Gheblawi et al., 2020). As a membrane protein ACE2 is widely distributed in multiple organs including the skeletal muscles (Chappell, 2021). Several studies on mouse limb muscle and human diaphragm have clearly shown the ACE2 expression on their cell membranes (Giordani et al., 2019; Shi et al., 2020). As the largest organ of the human body, skeletal muscle has the ability to produce and release a number of cytokines known as myokines. When the SARS-CoV-2 enters the circulatory system and reaches skeletal muscle cells, it interacts with ACE2 receptors on their cell membranes. Numerous studies have shown that myokines like TNFα, IL-6, IL-8, IL-10, and IFNγ are released in response to diverse physiological and pathophysiological conditions (Görgens et al., 2015; Hallberg et al., 2010; Peake et al., 2015). These myokines then act in endocrine, paracrine or autocrine manner on multiple organs which may in turn initiate immune response (Pedersen and Febbraio, 2008; Pedersen, 1985).
Skeletal muscles are rich in proteins and mitochondria and therefore more prone to undergo loss of their mass or function in response to either metabolic stress or pathological conditions. Diaphragm, one of the essential skeletal muscles of the respiratory system, plays a key role in maintaining lung capacity and any decline in its function can lead to decreased respiratory efficiency. With increasing age, sarcopenia is more evident with changes in morphological features that make these muscles less elastic with a lower shortening speed. Diaphragm also undergoes this age-related decline in its structure and function. Data from animal models and clinical studies show that the diaphragm is highly sensitive to shock. For example, sepsis like condition has shown to reduce the lung's ability to produce inspiratory pressure (Demoule et al., 2013). Clinical studies demonstrate that patients with sepsis conditions often need mechanical ventilation for their survival. Available literature also illustrates that mechanical ventilation acts as a two-edged sword associated with progressive development of diaphragm atrophy and ventilator-induced diaphragmatic dysfunction (VIDD) while providing the much-needed mechanical ventilation (Maes et al., 2014; Levine et al., 2008). During the ARDS condition, the preserved diaphragmatic activity is associated with a reduced risk of VIDD. Thus, assisted spontaneous breathing is considered the most appropriate physiologic method of respiratory support (Russotto et al., 2018).
Surprisingly, a time-dependent decrease of diaphragm strength after initiation of mechanical ventilation, has gained much research interest while diaphragm dysfunction at the onset of critical illness did not receive due attention (Petrof et al., 2010). Though more than 3000 articles on cytokine storm and the role of skeletal muscles in COVID-19 are published by March 15th, 2021, only 24 articles investigated its effects on diaphragm. Therefore, this paper will provide a research update on the role of cytokine storm and skeletal muscles in COVID-19 disease and then provide some insights into the knowledge gaps specifically related to diaphragm that needs to be explored in order to optimize care for elderly COVID-19 patients in need of a mechanical ventilator.
2. Cytokine storm and the diaphragm
Cytokine storm is a cascade of auto-amplifying cytokine production due to unregulated immune response of the host to different triggers like infection or malignancy. In COVID-19 patients, this uncontrolled abnormal immune response results in attacking its own cells and tissues instead of the virus particles leading to multiorgan failure and ultimately death. Recent studies have already shown that patients with COVID-19 have higher concentration of these proinflammatory cytokines (Ragab et al., 2020). In their review of current evidence and treatment strategies for COVID-19, Tang et al., summarizes other studies that indicate poor prognosis in patients with higher levels of these cytokines (Tang et al., 2020). As most COVID-19 patients report skeletal muscle weakness and fatigue coupled with the fact that ACE2 receptors are located on these muscle membranes, it is plausible that skeletal muscles especially diaphragm plays a critical role in patient prognosis.
Further, when COVID-19 mortality risk is calculated across the age groups, about 70% of all COVID-19 deaths in USA are from the age group of 70 years and above (Goldstein and Lee, 2020). Therefore, primary sarcopenia which is an age-related decrease in skeletal muscle function and muscle mass is a critical factor in understanding cytokine storm in older COVID-19 patients. Primary sarcopenia is generally associated with a decline in motor activity, cognitive function, respiratory function and immune senescence in older adults and cancer patients. Based on these characteristics, a number of studies have speculated that patients with sarcopenia and COVID-19 will have greater respiratory disease severity, and elevated mortality rates (Welch et al., 2020; Morley et al., 2020; Wang et al., 2021). However, so far there were no direct studies that investigated the relationship between sarcopenia and COVID-19 in older patients. The only direct evidence connecting weight loss, increase in inflammatory cytokines and COVID-19 comes from two animal studies conducted on Syrian hamsters and mice (Lau et al., 2020; Chan et al., 2020; Sheahan et al., 2020).
Previous mechanistic studies conducted on healthy volunteers demonstrated that sepsis induced changes in the diaphragm are due to excessive production of proinflammatory cytokines, free-radical generation, and activation of the diverse proteolytic systems (Vassilakopoulos et al., 2002). Studies show that oxidative stress is a major stimulus for the cytokines production under inspiratory resistive breathing conditions where monocytes do not play any role (Vassilakopoulos et al., 2002). This is supported by another study conducted on rats which documented gene expression of different cytokines in diaphragm and showed that strenuous sensitive breathing induces plasma cytokines. Data from this study on normal rats also show that IL-6, IL-1β, TNFα, IL-10, IFNγ, IL-4 cytokines mRNA levels were high in diaphragm but not in gastrocnemius muscle (Vassilakopoulos et al., 2004). Increased levels of IL-6 in diaphragm was further confirmed by immunoblotting as well as immunostaining (Vassilakopoulos et al., 2004). These studies demonstrate the role of diaphragm in producing cytokines during strenuous breathing. Similar to animal studies, increases in the pro-inflammatory cytokine expression (TNFα, IL-1β, and IL-6) were observed in the external intercostal muscles of COPD patients as well (Casadevall et al., 2007).
In contrast to primary sarcopenia, Duchenne muscular dystrophy (DMD) is a severe, progressive, muscle-wasting genetic disorder of the younger patients. Studies conducted on DMD patients and dystrophic mice highlighted the relationship between cytokine (i.e. IL-6) and redox balance in diaphragm muscle of both DMD patients and dystrophic mice (Petrillo et al., 2017). More specifically, Petrillo et al., has demonstrated that IL-6 overexpression in transgenic mouse model parallels the antioxidant expression profile and the severity of dystrophic muscle observed in DMD patients (Petrillo et al., 2017). Data from dystrophic mice show that deregulated levels of IL-6 alters the local and systemic redox signaling markers. In addition, IL-6 also acts as a central player in disturbing the redox balance in the diaphragm (Pelosi et al., 2017). In support of these findings, another study demonstrated that overexpression of circulating IL-6 enhances the generation and accumulation of free radicals in the diaphragm that leads to skeletal muscle atrophy in adult transgenic mice (Forcina et al., 2019).
Overall, these studies indicate that oxidative stress, inflammation and cytokines are closely related to each other and coexist in a muscle environment under diverse clinical settings. For example, a study conducted in rats demonstrated that during resistive breathing the production of nitric oxide decreased inside the diaphragm which in turn contributed to intra-diaphragmatic up-regulation of cytokines (IL-6, IL-10, IL-2, TNFα, and IL-1β). However, when nitric oxide is supplemented this response is downregulated. Available data also demonstrate that cytokines are differentially regulated by different signaling pathways i.e. IL-10, IL-2, TNFα, and IL-1β are regulated NF-κB dependent pathways (muscle atrophy-related transcription factor) while IL-6 is regulated by a pathway independent of NF-κB (Sigala et al., 1985).
3. Skeletal muscle function in COVID-19
From the available COVID-19 related literature, we found two ideas that need further investigation. The first idea requires exploring the cytokine storm to identify potential targets for intervention. For example, IL-6 blockade has been reported to be in use in China for COVID-19 patients as an increase in the level of IL-6 has also been reported in these patients, suggesting its crucial role in cytokine storm (Ruan et al., 2020; Liu et al., 2020). Another approach is to target the oxidative stress pathway. An extracellular superoxide dismutase, an antioxidant enzyme released by muscle due to regular exercise, may reduce the risk of acute respiratory distress syndrome (ARDS) in COVID-19 patients (Yan and Spaulding, 2020). Further, a new clinical trial to use antioxidant vitamin c against oxidative stress induced tissue damage for COVID-19 patients has begun in Wuhan, China (Carr, 2020). A recent study on COVID-19 patients has confirmed skeletal muscle injury in these patients that correlated with an increase in creatinine kinase levels in their blood (Mao et al., 2020). Therefore, specific studies on using creatinine kinase levels in blood as an early biomarker for COVID-19 could be very helpful in long term care for the patients.
The second idea requires optimizing the conditions for using mechanical ventilation. Prolonged intensive care unit (ICU) stay is required for stabilization of COVID-19 patients and that may itself directly impact loss of skeletal muscle mass and function which may further add on to human morbidity (Barazzoni et al., 2020). Available clinical reports clearly illustrate that prolonged mechanical ventilation is associated with gradual development of diaphragm atrophy and ventilator-induced diaphragmatic dysfunction (VIDD) (Vassilakopoulos and Petrof, 2004). To prevent or reverse VIDD, United States FDA approved lung pacer diaphragm pacing therapy system for COVID-19 patients in April 2020. Recent studies on COVID-19 ICU patients have provided clear histological evidence for ACE-2 expression and SARS-CoV-2 infiltration in to the diaphragm that ultimately leads to development of fibrosis (Shi et al., 2021). In addition, diagnostic tests like lung ultrasound are currently used to assess diaphragm function in COVID-19 patients and manage their weaning process (Guarracino et al., 2021).
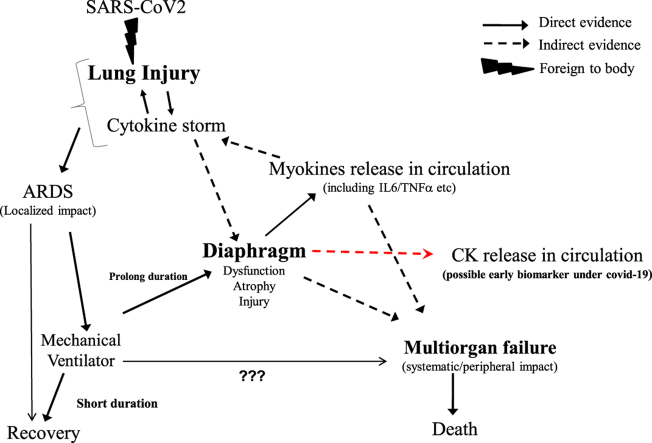
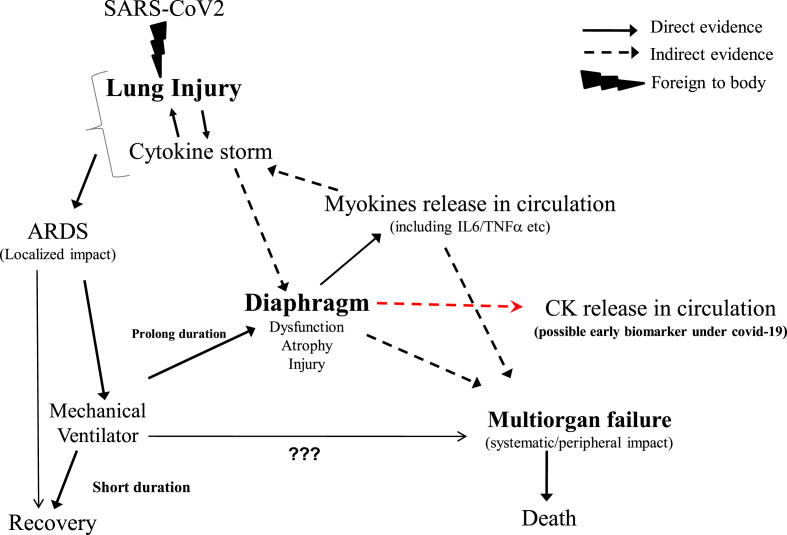
These observations strongly emphasize the significance of skeletal muscles in the treatment of ARDS associated with COVID-19. The SARS-CoV-2 enters the body through the respiratory system and then gradually enters the circulatory system. As ACE2 receptors are widely distributed in all the tissues, the virus enters other organs especially like skeletal muscles that are likely to trigger the cytokine storm [Fig. 1]. Based on the current knowledge of SARS-CoV-2 pathology and its clinical manifestations, the search for specific antivirals along with immune-modulating drugs must include supplemental drugs that act on the skeletal muscles especially the diaphragm. This two-pronged approach will not only minimize the multiorgan failure by controlling the cytokine storm but also enhance the recovery of patients on mechanical ventilators by improving diaphragm function.
Fig. 1.
Graphical abstract showing the interaction between SARS-CoV2, cytokine storm and the diaphragm.
4. Conclusion
The current literature and clinical data on the role of diaphragm in COVID-19 patients indicate a complex interplay between SARS-CoV-2 mediated lung injury, cytokines storm, ARDS, ventilator, and aging which eventually becomes fatal to the susceptible patient. Diaphragm not only is in the center of such correlation but is critically important during ICU where the patients are on ventilators. Based on our review of available COVID-19 literature, we identified the following knowledge gaps that require further investigation:
-
●
Direct studies in COVID-19 patients investigating the effects of cytokine storm on diaphragm function are lacking. Specific studies on identifying signaling pathways of the cytokines in diaphragm and mechanisms of their regulation are also not known. These studies are critical as they will help in identifying potential drug targets by differentiating between NF-κB dependent and NF-κB independent pathways.
-
●
Re-evaluate the optimal conditions of the mechanical ventilators by considering effects of sarcopenia when using them in elderly COVID-19 patients. As multi organ failure is common in these patients, studies on chest radiography in addition to cardiac and vascular ultrasounds must be explored to have a comprehensive understanding on clinical management of COVID-19 patients on ventilators. Though the effects of nitric oxide on cytokines levels in rat diaphragm were studied before the COVID-19 pandemic, further studies exploring their role in treating COVID-19 patients especially those on ventilator are warranted.
Completion of the above-mentioned studies will provide a foundational basis to identify therapeutic targets and develop a potential treatment plans for COVID-19 patients. This is necessary as the currently available COVID-19 vaccines will protect people from the effects of SARS-CoV-2 but not helpful in treating COVID-19 patients.
CRediT authorship contribution statement
Ashwani Mittal: Conceptualization, Writing – original draft. Anita Dua: Data curation, Visualization. Sanjeev Gupta: Resources, Project administration. Elisha Injeti: Investigation, Conceptualization, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work is supported by Govt. of India UGC-MRP grant (F. 41–1266/2012 (SR) to Ashwani Mittal.
References
- Barazzoni R., Bischoff S.C., Breda J., et al. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin. Nutr. 2020;39(6):1631–1638. doi: 10.1016/j.clnu.2020.03.022. doi: S0261-5614(20)30140-0 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carr A.C. A new clinical trial to test high-dose vitamin C in patients with COVID-19. Crit. Care. 2020;24(1):133–134. doi: 10.1186/s13054-020-02851-4. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casadevall C., Coronell C., Ramírez-Sarmiento A.L., et al. Upregulation of pro-inflammatory cytokines in the intercostal muscles of COPD patients. Eur. Respir. J. 2007;30(4):701–707. doi: 10.1183/09031936.00152005. doi: 09031936.00152005 [pii] [DOI] [PubMed] [Google Scholar]
- Chan J.F., Zhang A.J., Yuan S., et al. Simulation of the clinical and pathological manifestations of coronavirus disease 2019 (COVID-19) in a golden syrian hamster model: implications for disease pathogenesis and transmissibility. Clin. Infect. Dis. 2020;71(9):2428–2446. doi: 10.1093/cid/ciaa325. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chappell M.C. Commentary for "endocrine significance of SARS-CoV-2's reliance on ACE2". Endocrinology. 2021;162(4) doi: 10.1210/endocr/bqaa222. bqaa222. doi: 10.1210/endocr/bqaa222. doi: 10.1210/endocr/bqaa222 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coronavirus disease (COVID-19) pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019 Web site.
- Demoule A., Jung B., Prodanovic H., et al. Diaphragm dysfunction on admission to the intensive care unit. prevalence, risk factors, and prognostic impact-a prospective study. Am. J. Respir. Crit. Care Med. 2013;188(2):213–219. doi: 10.1164/rccm.201209-1668OC. [doi] [DOI] [PubMed] [Google Scholar]
- Forcina L., Miano C., Scicchitano B.M., et al. Increased circulating levels of interleukin-6 affect the redox balance in skeletal muscle. Oxid Med Cell Longev. 2019;2019:3018584. doi: 10.1155/2019/3018584. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gheblawi M., Wang K., Viveiros A., et al. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020;126(10):1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giordani L., He G.J., Negroni E., et al. High-dimensional single-cell cartography reveals novel skeletal muscle-resident cell populations. Mol. Cell. 2019;74(3):609–621. doi: 10.1016/j.molcel.2019.02.026. e6. doi: S1097-2765(19)30137-6 [pii] [DOI] [PubMed] [Google Scholar]
- Goldstein J.R., Lee R.D. Demographic perspectives on the mortality of COVID-19 and other epidemics. Proc. Natl. Acad. Sci. U.S.A. 2020;117(36):22035–22041. doi: 10.1073/pnas.2006392117. http://www.pnas.org/content/117/36/22035.abstract doi: 10.1073/pnas.2006392117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Görgens S.W., Eckardt K., Jensen J., Drevon C.A., Eckel J. Exercise and regulation of adipokine and myokine production. Prog Mol Biol Transl Sci. 2015;135:313–336. doi: 10.1016/bs.pmbts.2015.07.002. doi: S1877-1173(15)00134-9 [pii] [DOI] [PubMed] [Google Scholar]
- Guarracino F., Vetrugno L., Forfori F., et al. Lung, heart, vascular, and diaphragm ultrasound examination of COVID-19 patients: a comprehensive approach. J. Cardiothorac. Vasc. Anesth. 2021;35(6):1866–1874. doi: 10.1053/j.jvca.2020.06.013. https://www.sciencedirect.com/science/article/pii/S105307702030519X [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallberg L., Janelidze S., Engstrom G., Wisén A.G., Westrin A., Brundin L. Exercise-induced release of cytokines in patients with major depressive disorder. J. Affect. Disord. 2010;126(1–2):262–267. doi: 10.1016/j.jad.2010.02.133. ([doi]) [DOI] [PubMed] [Google Scholar]
- Lau S.Y., Wang P., Mok B.W., et al. Attenuated SARS-CoV-2 variants with deletions at the S1/S2 junction. Emerg. Microb. Infect. 2020;9(1):837–842. doi: 10.1080/22221751.2020.1756700. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine S., Nguyen T., Taylor N., et al. Rapid disuse atrophy of diaphragm fibers in mechanically ventilated humans. N. Engl. J. Med. 2008;358(13):1327–1335. doi: 10.1056/NEJMoa070447. ([doi]) [DOI] [PubMed] [Google Scholar]
- Liu B., Li M., Zhou Z., Guan X., Xiang Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J. Autoimmun. 2020;111:102452. doi: 10.1016/j.jaut.2020.102452. doi: S0896-8411(20)30067-6 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maes K., Stamiris A., Thomas D., et al. Effects of controlled mechanical ventilation on sepsis-induced diaphragm dysfunction in rats. Crit. Care Med. 2014;42(12):772. doi: 10.1097/CCM.0000000000000685. ([doi]) [DOI] [PubMed] [Google Scholar]
- Mao L., Jin H., Wang M., et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, China. JAMA Neurol. 2020;77(6):683–690. doi: 10.1001/jamaneurol.2020.1127. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morley J.E., Kalantar-Zadeh K., Anker S.D. COVID-19: a major cause of cachexia and sarcopenia? J Cachexia Sarcopenia Muscle. 2020;11(4):863–865. doi: 10.1002/jcsm.12589. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peake J.M., Della Gatta P., Suzuki K., Nieman D.C. Cytokine expression and secretion by skeletal muscle cells: regulatory mechanisms and exercise effects. Exerc. Immunol. Rev. 2015;21:8–25. [PubMed] [Google Scholar]
- Pedersen B.K. Edward F. adolph distinguished lecture: muscle as an endocrine organ: IL-6 and other myokines. J. Appl. Physiol. 1985;107(4):1006–1014. doi: 10.1152/japplphysiol.00734.2009. 2009. ([doi]) [DOI] [PubMed] [Google Scholar]
- Pedersen B.K., Febbraio M.A. Muscle as an endocrine organ: focus on muscle-derived interleukin-6. Physiol. Rev. 2008;88(4):1379–1406. doi: 10.1152/physrev.90100.2007. [doi] [DOI] [PubMed] [Google Scholar]
- Pelosi L., Forcina L., Nicoletti C., Scicchitano B.M., Musarò A. Increased circulating levels of interleukin-6 induce perturbation in redox-regulated signaling cascades in muscle of dystrophic mice. Oxid Med Cell Longev. 2017;2017:1987218. doi: 10.1155/2017/1987218. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrillo S., Pelosi L., Piemonte F., et al. Oxidative stress in duchenne muscular dystrophy: focus on the NRF2 redox pathway. Hum. Mol. Genet. 2017;26(14):2781–2790. doi: 10.1093/hmg/ddx173. [doi] [DOI] [PubMed] [Google Scholar]
- Petrof B.J., Jaber S., Matecki S. Ventilator-induced diaphragmatic dysfunction. Curr. Opin. Crit. Care. 2010;16(1):19–25. doi: 10.1097/MCC.0b013e328334b166. ([doi]) [DOI] [PubMed] [Google Scholar]
- Ragab D., Salah Eldin H., Taeimah M., Khattab R., Salem R. The COVID-19 cytokine storm; what we know so far. Front. Immunol. 2020;11:1446. doi: 10.3389/fimmu.2020.01446. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from wuhan, China. Intensive Care Med. 2020;46(5):846–848. doi: 10.1007/s00134-020-05991-x. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russotto V., Bellani G., Foti G. Respiratory mechanics in patients with acute respiratory distress syndrome. Ann. Transl. Med. 2018;6(19):382. doi: 10.21037/atm.2018.08.32. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheahan T.P., Sims A.C., Zhou S., et al. An orally bioavailable broad-spectrum antiviral inhibits SARS-CoV-2 in human airway epithelial cell cultures and multiple coronaviruses in mice. Sci. Transl. Med. 2020;12(541) doi: 10.1126/scitranslmed.abb5883. Epub 2020 Apr 6. doi: 10.1126/scitranslmed.abb5883 [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Z., de Vries H.J., Vlaar A.P.J., et al. Diaphragm pathology in critically ill patients with COVID-19 and postmortem findings from 3 medical centers. JAMA internal medicine. 2020 doi: 10.1001/jamainternmed.2020.6278. doi: 10.1001/jamainternmed.2020.6278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Z., de Vries H.J., Vlaar A.P.J., et al. Diaphragm pathology in critically ill patients with COVID-19 and postmortem findings from 3 medical centers. JAMA Intern Med. 2021;181(1):122–124. doi: 10.1001/jamainternmed.2020.6278. Accessed 3/18/2021. doi: 10.1001/jamainternmed.2020.6278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sigala I., Zacharatos P., Boulia S., et al. Nitric oxide regulates cytokine induction in the diaphragm in response to inspiratory resistive breathing. J. Appl. Physiol. 1985;113(10):1594–1603. doi: 10.1152/japplphysiol.00233.2012. 2012. ([doi]) [DOI] [PubMed] [Google Scholar]
- Tang Y., Liu J., Zhang D., Xu Z., Ji J., Wen C. Cytokine storm in COVID-19: the current evidence and treatment strategies. Front. Immunol. 2020;11:1708. doi: 10.3389/fimmu.2020.01708. [doi] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vassilakopoulos T., Petrof B.J. Ventilator-induced diaphragmatic dysfunction. Am. J. Respir. Crit. Care Med. 2004;169(3):336–341. doi: 10.1164/rccm.200304-489CP. doi: 169/3/336 [pii] [DOI] [PubMed] [Google Scholar]
- Vassilakopoulos T., Katsaounou P., Karatza M.H., Kollintza A., Zakynthinos S., Roussos C. Strenuous resistive breathing induces plasma cytokines: role of antioxidants and monocytes. Am. J. Respir. Crit. Care Med. 2002;166(12 Pt 1):1572–1578. doi: 10.1164/rccm.200203-177OC. doi: 200203-177OC [pii] [DOI] [PubMed] [Google Scholar]
- Vassilakopoulos T., Divangahi M., Rallis G., et al. Differential cytokine gene expression in the diaphragm in response to strenuous resistive breathing. Am. J. Respir. Crit. Care Med. 2004;170(2):154–161. doi: 10.1164/rccm.200308-1071OC. doi: 200308-1071OC [pii] [DOI] [PubMed] [Google Scholar]
- Wang P., Li Y., Wang Q. Sarcopenia: an underlying treatment target during the COVID-19 pandemic. Nutrition. 2021;84:111104. doi: 10.1016/j.nut.2020.111104. https://www.sciencedirect.com/science/article/pii/S0899900720303877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welch Carly, Greig Carolyn, Masud Tahir, Wilson Daisy, Jackson Thomas A. COVID-19 and acute sarcopenia. Aging and disease. 2020;11(6):1345. doi: 10.14336/AD.2020.1014. http://www.aginganddisease.org/EN/article/article_148018.shtml doi: 10.14336/AD.2020.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan Z., Spaulding H.R. Extracellular superoxide dismutase, a molecular transducer of health benefits of exercise. Redox Biol. 2020;32:101508. doi: 10.1016/j.redox.2020.101508. doi: S2213-2317(20)30319-0 [pii] [DOI] [PMC free article] [PubMed] [Google Scholar]