Abstract
All mammals are to different degrees exposed to stressors being physical or social, which may affect health and well-being. Stressful and traumatic situations have direct effects on immune responses that may alter susceptibility to developing somatic illnesses. In animal research, different types of stressors have been investigated in studying the effect on bowel disorders, some stressors being more or less of environmental origin. We aimed, therefore, to explore whether a more natural stressor would differ from a stressor of more unnatural characteristics on dextran sulphate sodium (DSS) induced colitis in adult rats. Specifically, whether social stress within a single social defeat (SD) paradigm would be a more potent stressor than brief inescapable foot-shocks (IFS) in causing elevated faecal granulocyte marker protein (GMP), and crypt- and inflammation scores in colonic tissue. Three groups of male Wistar rats were used; socially defeated rats; inescapable foot-shock rats; and comparison rats. Main findings showed no difference between the groups on GMP levels. However, there was a significant difference on inflammation and crypt scores for the distal part of colon, detected through histology, where socially defeated rats were more susceptible. A single SD seems to be more adverse than inescapable foot-shock on DSS induced colitis, but further studies are recommended to validate a broader range of different outcomes comparing two such different rodent stress models.
Introduction
In humans, at least, the main sources of stress are social, where relationships and sense of belonging are perceived to be threatened. Exposure to social stress varies in magnitude and intensity, and there is a vast amount of evidence for an impact on health [1] for review). Studies have revealed associations between gastrointestinal (GI) diseases such as irritable bowel syndrome (IBS) and inflammatory bowel disease (IBD) and stress, demonstrating the importance of brain-gut interactions [2 for review, 3]. IBD is a collective term for ulcerative colitis (UC) and Crohn’s disease (CD) and differs from IBS, which is a functional GI disorder with a multifaceted pathophysiology. It is unclear whether stressful life events lead to the development of GI diseases, or if they are more strongly related to associated mood disorders. For example, it has been reported that patients with IBD have higher lifetime rates of anxiety and mood disorders, and the onset of these precedes the diagnosis of IBD [4].
In animal research, social defeat (SD) is considered a naturalistic model of social stress. The SD protocol utilizes the resident/intruder paradigm. The paradigm is based on the fact that an adult male rat, the resident, will establish a territory and attack an unfamiliar male, the intruder, when introduced into its home cage. This social conflict situation has numerous effects on behaviour and the neuroendocrine system [5], and a single SD experience can mimic an acute stressor that can provoke long-term fear responses [6]. Inescapable foot-shocks (IFS) are known as a model of acute stress but, unlike SD which relies upon animal-animal interactions, IFS is easily quantifiable and can be precisely controlled. Although it does not normally cause any physical damage, brief exposure to just a few IFS may cause long-lasting behavioural changes [6]. However, critiques of using the IFS method include the pain inflicted on the animal and its artificiality. Less is known of how the more naturalistic stressor (SD) compares to the more artificial one (IFS).
Clinical studies provide evidence for significant correlations between increased intestinal permeability and disease activity in ulcerative colitis [7]. Permeability across the colon wall layers allows absorption of nutrients from food and fluids, as well as elimination of waste materials. However, if larger proteins and microorganisms such as bacteria infiltrate these layers, there is a heightened risk of an inflamed colon. Concentrations of faecal calprotectin (FC) are found to be elevated in patients with IBD compared to those with functional GI disorders [8–10]. Calprotectin is a protein complex in humans, released extracellularly by activation of neutrophils, and intestinal inflammation causes increased concentrations in the colon [11]. The parallel to human calprotectin, granulocyte marker protein (GMP), can be analysed in rodent faeces [12] to detect a disease activity in a non-invasive manner. Here we report both histological data, and measures of GMP in DSS-treated animals after either a single SD or brief IFS.
Previous studies on murine DSS-induced colitis and social stress have utilized models of chronic SD [13]. The present study is to our knowledge the first to assess the outcome of a single social defeat on DSS-induced colitis, and in the same study compare single social defeat to IFS on subsequent chemically induced colon tissue damage in rats. The aim was to compare a single session of social defeat (SD) with those of brief exposure to inescapable foot shocks (IFS) in terms of faecal granulocyte marker protein (GMP) concentrations and colon tissue histology in adult male rats after inducing a colitis-like condition by dextran sulphate sodium (DSS). We hypothesized that being physically attacked and defeated would render animals more prone to chemically induced colon tissue damage than would IFS. Further, we explored whether the effects would be related to pre-stress levels of corticosterone, as reported earlier for IFS [14].
Methods and materials
Animals and housing
All the testing and procedures were approved by the Norwegian Animal Research Authority (permit number: 2006010B) and were registered by the Authority. Every effort was made to minimize suffering. On the day after arrival, male Wistar rats (9 weeks of age and 260–299g on arrival) from two separate batches (Taconic, Lille Skvensved, Denmark) were single housed in individually ventilated cages (polypropylene Euro-standard Type III H) with free access to food and water. Within the cages, air was exchanged 75 times per hour, there was an average ambient temperature of 22°C and an average relative humidity of 65%. The room had a 12:12h light/dark schedule with lights on at 07:00h and lights off at 19:00h (progressive increase in light at 06:00h and progressive dimming at 18:00h). All rats were allowed 5 days of acclimatization, then 5 days of daily handling for 1–2 min and habituated to being moved between facilities, prior to blood sampling and experimental procedures.
The resident male rats (Wistar, Taconic) were at least 5 months of age and weighed >450g. To stimulate territorial behaviour, they were housed in pairs together with ovariectomized females (Wistar, Taconic) in individually ventilated cages for at least 2 weeks, housed in a separate colony room, and habituated to being moved. The females were not present during habituation and the SD procedure but were briefly removed from their cages. For the males, time of transport from the colony room to the test room was approximately 5 min and followed by 1h rest before the introduction of the intruder rat. Bedding was not renewed for at least 2 days prior to a social conflict to preserve the residents’ scent [6].
Experimental design and procedures
An overview of the experimental design is shown in Fig 1. All male rats (n = 90) underwent blood sampling prior to the experimental procedures.
Fig 1. An overview of the experimental design for the three experimental groups.
On the basis of the initial corticosterone concentration, the rats were ranged from the highest to the lowest corticosterone level and then divided into three groups: high corticosterone (mean ± Standard Error of the Mean (SEM): 239.3 ± 15.8 ng/ml; n = 30), middle corticosterone (106.3 ± 7.8 ng/ml; n = 30) and low corticosterone (32.6 ± 5.1 ng/ml; n = 30). The middle corticosterone group was excluded from the experiment to maximize any differences between experimental groups.
From the high and low corticosterone group, rats were divided by stratified randomization into IFS, SD, and comparison (COMP) group (n’s = 20). Thus, each of the three experimental groups (IFS, SD, and COMP) comprised one high and one low corticosterone subgroup. The subgroups were: IFS-HIGH, IFS-LOW, SD-HIGH. SD-LOW, COMP-HIGH, COMP-LOW (n`s = 10).
On day 0 of the experiment each experimental animal underwent IFS, SD or COMP procedures. On all other days, the procedures were identical for all groups. For a separate behavioural experiment ending day 24 (previously reported and discussed in [6], rats were once exposed to an acoustic startle response test and once a week for three weeks exposed to sucrose preference tests, open field tests, elevated plus maze tests and body weights were measured.
For chemical induction of colitis, all rats were exposed to 4% DSS in their drinking water from day 29 for 7 days.
Faecal pellets for measurements of GMP were collected prior to the DSS exposure, and after 2 and 7 days of exposure to DSS. The experimental animals were euthanized for histology of colon tissue on day 36 after the stress procedures.
Corticosterone sampling
To minimize suffering following the blood sampling procedure, blood from the saphenous vein was chosen over collection from the jugular vein. One day prior to blood sampling, the skin area above the saphenous vein was shaved. Blood sampling for corticosterone measures were conducted between 09:00 and 12:00h as the natural circadian rhythm of corticosterone release in rats is low and stable during this period [15]. The home cage was moved to the sampling room and the rat placed in a sealed chamber (23×12×11.5 cm) for anesthesia (flow of 1 l/min O2, and 1 l/min N2O vaporized by 5% isoflurane (Isoba Vet., Schering-Plough, Ballerup, Denmark)). After clear muscle relaxation, the rat was placed in a ventral position, 2% isoflurane was given through a face mask, and the saphenous vein was punctured. 40–400 μl blood was collected in BD Microtainer tubes (Medinor, Oslo, Norway), all within < 3.5 min. Samples were centrifuged at 1600g for 10 min, the serum separated and then frozen at −20°C until analysis using Rat Corticosterone Enzyme Immunoassay Kit (DSL-10-81100, MedProbe, Oslo, Norway) with the aid of a plate reader, Wallac 1420 Multilabel counter (PerkinElmer, Oslo, Norway).
Inescapable foot-shock (IFS) procedures
IFS procedures were previously used in our laboratory [14], and they were performed during the light phase (14:00–18:00h). The shock apparatus consisted of a shock generator (Coulbourn Instruments, Lehigh Valley, PA, USA) and a shock chamber (26×30×30 cm, model H10-11R-TC) with grid flooring inside a sound attenuated cubicle (80×50×50 cm). Foot-shocks were delivered through the grids by a computerized shock system (Habitest system; Graphic State 3.0 software). Each rat was placed individually in the chamber, left undisturbed for 2 min before receiving a total of 10 foot-shocks of 1mA intensity, each of 5s duration, with an inter-shock interval from 24s to 244s (mean 90s). The apparatus was thoroughly cleaned with a 20% ethanol solution between animals.
Single social defeat (SD) procedures
The single SD procedures were adopted from Meerlo and colleagues [16], and were performed between 21:00 and 02:00h (the dark phase). To provide a clear view of the resident–intruder conflict, the room was illuminated by red light, the top lid of the resident’s cage was removed, and an empty cage was placed upside down on top of the cage. The residents had been trained to fight at least five times in confronting younger intruder males (Wistar rats, Taconic). Only residents that had defeated the intruder in < 2 min on the last training session without inflicting injury were selected to proceed. Each SD rat was transported to the experimental room and placed in the cage of the resident. When the SD rat was defeated and showed submissive behaviour (lying motionless on its back), it was moved to a small wire-mesh cage in the resident’s cage for a total of 1 hour, protected from repeated attacks and potential physical injuries. Immediately after the defeat session, SD rats were returned to their home cages and the colony room.
Comparison group procedure
Each rat in the comparison group was removed from its home cage in the colony room where it was housed, and gently handled for 1 min on one occasion during the light phase (14:00–18:00h).
Chemical induction of colitis
All rats were given a 4% solution of dextran sulphate sodium (DSS, powder dissolved in distilled water) (TdB Consultancy AB, Uppsala, Sweden) in place of their normal drinking water. The solution was available ad libitum for 7 days, and freshly made each day. Daily consumption was recorded.
Granulocyte marker protein (GMP)
Fresh faecal pellets were collected from the animal cages.
Pellets were stored at −30°C until analysis. 1g of the sample was diluted in 4 ml extraction buffer (TRIS 12.1g/l, CaCl2 1.47g/l, Mertiolat 0.1g/l dissolved in 0.9% NaCl, pH adjusted to 8.0), and thoroughly homogenized using an Ultra Turrax (2000 r.p.m.) for 20 s or until the material was dissolved. The upper halves of the supernatants were carefully harvested and quantified by GMP ELISA.
Histology
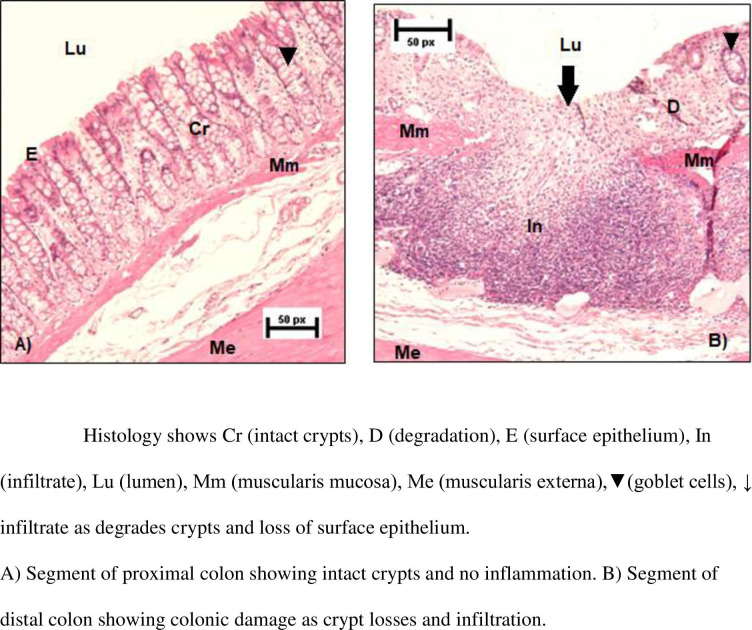
After 7 days of DSS exposure, the rats were euthanized by CO2 followed by dislocation of the cervical vertebrae. The abdominal cavity was opened longitudinally, the colon dislocated from the caecum and the small intestine, flushed with a phosphate buffer in a 10 ml syringe (NaCl, KH2PO4, Na2HPO4x2HH2O), and cut along the mesenteric line. Waste material was carefully removed using the syringe with phosphate buffer. A small segment of the upper and lower colon was discarded due to handling. The remaining distal and proximal segments were cut, gently rinsed, and pinned on a piece of polystyrene with the mucosal layer visible. Each segment was soaked in formalin (4%). Eight sections per segment were stained with hematoxylin and eosin (16 sections per rat), then blindly scored in a randomized manner. Validated scoring systems were used for crypts [17], and inflammation [18]: score 0-intact crypt; score 1-loss of the lower 1/3 of the crypt; score 2-loss of the lower 2/3 of the crypt; score 3-loss of the entire crypt, but intact surface epithelium; score 4-loss of the entire crypt and the surface epithelium. A score of 1 to 4 indicated % surface area affected (1 = 1–25%, 2 = 26–50%, 3 = 51–75%, and 4 = 76–100%). The total crypt score was a product of the crypt score and the affected area score. Inflammation: score 0-normal; score 1-focal inflammatory cell infiltration; score 2-inflammatory cell infiltration, “gland drop-outs” and crypt abscess; score 3-mucosal ulcerations. The score was a product of the inflammation score and the score for the affected area. See Fig 2A for an example of normal colon tissue. Fig 2B demonstrates loss of the entire crypt, loss of the surface epithelium and mucosal ulceration.
Fig 2.
A and B: Histology segments of intact and damaged colon respectfully.
The average scores of the eight sections for the proximal segment and for the distal segment on total crypt and total inflammation score were separately compared between the three groups. Interrater agreement was assessed for scoring of 271 histology sections by independent pairs of scorers amongst the five authors in mixed pairs gaining a scoring match of 269 of the sections (interrater agreement of 99.82%).
Statistics
Statistical analyses were performed using Statistica version 13.3 (TIBCO Software Inc. (2017)). All data are expressed as mean ± SEM. A p-level of <0.05 was considered statistically significant. For total DSS consumption, a factorial ANOVA was used (Group x CORT).
Repeated measures factorial ANOVA was used to analyse the faecal GMP data (group x day x CORT) followed by Bonferroni post-hoc analysis. Kruskal-Wallis tests were used to analyse the faecal GMP data between subgroups, due to a significant Levene`s test for (group x day x CORT) in the repeated measures ANOVA analysis. Due to the non-parametric nature of the histology data, Kruskal-Wallis tests were used to compare the three groups, and the six subgroups.
Results
Three rats were excluded from all analyses. One was excluded from the SD high corticosterone group due to no submissive behavior, one from the control high corticosterone group due to technical problems, and one from the IFS high corticosterone group due to overt signs of illness and marked loss of body weight before initiation of procedures for chemically induction of colitis. The included animals were: IFS-HIGH (n = 9), IFS-LOW (n = 10), SD-HIGH (n = 9), SD-LOW (n = 10), COMP-HIGH (n = 9), COMP-LOW (n = 10).
Behaviour in the social defeat confrontation
On average the SD rats received 1–2 attacks before they showed submissive supine posture within 2 minutes. All SD rats, except for one (see above), were eventually defeated before they were separated from the dominant resident rat.
One defeated animal in the HIGH group got a visible scratch on the head, where after three days there were no visible marks. This animal was not excluded from the analyses as this animal showed no colonic damage in the histological sections and GMP levels were not elevated when compared to rats in either group.
Consumption of dextran sulphate sodium (DSS) solution
Descriptive statistics of total DSS consumption for experimental groups and subgroups are shown in Table 1. Data are expressed as ml.
Table 1. Faecal GMP levels (mg/l) and total DSS consumption (ml).
| Group | Corticosterone | N | GMP levels Day 0 | GMP levels Day 2 | GMP levels Day 7 | Total DSS consumption |
|---|---|---|---|---|---|---|
| COMP | Low | 10 | 13.10± 2.86 | 49.90 ± 13.81 | 115.18 ± 17.92 | 188.35 ± 8.45 |
| COMP | High | 9 | 22.39 ± 9.48 | 36.00 ± 8.91 | 135.78 ± 21.50 | 197.17 ± 12.19 |
| COMP | All | 19 | 17.50 ± 4.72 | 43.32 ± 8.34* | 124.93 ± 13.70*** | 192.53 ± 7.15 |
| IFS | Low | 10 | 12.75 ± 2.23 | 53.48 ± 14.88 | 155.43 ± 40.77 | 188.40 ± 9.57 |
| IFS | High | 9 | 14.22± 4.15 | 19.00 ± 6.35 | 93.86 ± 15.82 | 178.33 ± 5.73 |
| IFS | All | 19 | 13.45 ± 2.23 | 37.14 ± 9.12* | 126.26 ± 23.29*** | 183.63 ± 5.69 |
| SD | Low | 10 | 19.73 ± 8.12 | 36.20 ± 9.49 | 142.80 ± 34.51 | 192.10 ± 10.48 |
| SD | High | 9 | 8.50 ± 1.34 | 66.31 ± 15.75 | 171.17 ± 40.17 | 186.39 ± 8.65 |
| SD | All | 19 | 14.41 ± 4.41 | 50.46 ± 9.41* | 156.24 ± 25.77*** | 189.39 ± 6.71 |
Descriptive statistics of faecal GMP levels (mg/l) for comparison group (COMP), inescapable foot-shock group (IFS) and single social defeat group (SD), and the groups divided in subgroups according to low and high initial levels of corticosterone (group mean ± SEM).
Faecal samples for GMP level analysis are collected prior to dextran sulphate sodium (DSS) exposure (DSS day 0), after 2 days and after 7 days exposure to DSS (DSS day 2 and 7, respectively). Total DSS consumption (ml) is the total consumption across the 7 days when DSS was given in place of the normal drinking water (group mean ± SEM).
Descriptive statistics of Histology scores from comparison group (COMP), inescapable foot-shock group (IFS) and social defeat group (SD), and the groups divided in subgroups according to low and high initial levels of corticosterone (group mean ± SEM).
Repeated measures factorial ANOVA (group x day x CORT) revealed a significant effect of day, (F(2, 102) = 72.51, p<0.001), but no significant effect of either group or CORT (F’s<0.1).
*p<0.05,
***p<0.001 change in faecal GMP level from day 0, all groups collectively (Bonferroni post-hoc analysis).
There was no group effect on total DSS consumption, no CORT effect, and no interaction between them (all p’s>0.1).
Faecal granulocyte marker protein (GMP)
Descriptive statistics of GMP levels (mg/l) across days are shown in Table 1.
Repeated measures factorial ANOVA (group x day x CORT) revealed a significant effect of day, (F(2, 102) = 72.51, p<0.001), but no significant effect of either group or CORT (F’s<0.1). None of the interaction effects were significant. Follow-up analysis showed higher levels of GMP after 2 and 7 days of DSS compared to the pre-DSS GMP levels (p = 0.02 and p<0.001, respectively). The Kruskal-Wallis test revealed no differences in GMP amongst subgroups on any for the days (all p’s>0.09).
Histology
Descriptive statistics of histology scores and inflammation score are shown in Table 2.
Table 2. An overview of the histology scores and inflammation score for each group.
| Group | Corticosterone | N | Proximal crypt score | Proximal inflammation score | Distal crypt score | Distal inflammation score |
|---|---|---|---|---|---|---|
| COMP | Low | 10 | 1.36 ± 0.57 | 0.99 ± 0.42 | 1.98 ± 0.57 | 1.66 ± 0.44 |
| COMP | High | 9 | 1.53 ± 0.53 | 1.18 ± 0.39 | 3.19 ± 0.77 | 2.13 ± 0.46 |
| COMP | All | 19 | 1.44 ± 0.38 | 1.08 ± 0.28 | 2.55 ± 0.48 | 1.88 ± 0.31 |
| IFS | Low | 10 | 1.34 ± 0.53 | 0.90 ± 0.38 | 1.39 ± 0.55 | 0.70± 0.37 |
| IFS | High | 9 | 0.42 ± 0.42 | 0.51 ± 0.35 | 1.00 ± 0.55 | 0.56 ± 0.38 |
| IFS | All | 19 | 0.90 ± 0.35 | 0.72 ± 0.26 | 1.20 ± 0.38# | 0.63 ± 0.26 ## † |
| SD | Low | 10 | 0.94 ± 0.48 | 0.81 ± 0.36 | 3.66 ± 0.95 | 2.45 ± 0.51 |
| SD | High | 9 | 1.57 ± 0.58 | 1.29 ± 0.43 | 2.50 ± 0.63 | 2.07 ± 0.52 |
| SD | All | 19 | 1.24 ± 0.37 | 1.04 ± 0.28 | 3.11 ± 0.59 | 2.27 ± 0.36 |
Descriptive statistics of histology scores from comparison group (COMP), inescapable foot-shock group (IFS) and single social defeat group (SD), and the groups divided in subgroups according to low and high initial levels of corticosterone (group mean ± SEM).
For the distal crypt scores, there was a significant difference between experimental groups (H(2, N = 57) = 7.63, p = 0.02).
For distal inflammation, there was a significant overall group effect (H (2, N = 57) = 12.87, p<0.002).
#p<0.05,
##p<0.01 difference between IFS group and SD group (Kruskal-Wallis tests).
† p<0.05, difference between and IFS group and COMP group (Kruskal-Wallis tests)
Proximal colon
For proximal crypt and inflammation scores, there were no significant differences between any of the groups or subgroups (all p’s>0.1) after a Kruskal-Wallis test.
Distal colon
For the distal crypt scores, a Kruskal-Wallis test showed significant difference between experimental groups (H(2, N = 57) = 7.63, p = 0.02), where the SD group had higher scores compared to the IFS group (p = 0.03). There was no significant difference between the IFS and COMP nor between the SD and COMP group, all p’s>0.1. Between the subgroups, a Kruskal-Wallis test showed no significant differences on distal crypt scores (p’s>0.1).
For distal inflammation, a Kruskal-Wallis test showed significant overall group effect (H(2, N = 57) = 12.87, p<0.002) with higher scores in the SD group compared to the IFS group (p = 0.003); furthermore, the COMP group had higher scores than the IFS group (p = 0.03), but there were no significant differences between the subgroups, all p’s>0.1.
Discussion
The aim of the current study was to examine whether a single social defeat exposure would differ from brief inescapable foot-shocks in its impact on experimentally induced colonic inflammation in rats. Specifically, whether a single SD would cause greater elevations of faecal GMP and render the colon tissue more susceptible to damage than brief IFS. We did not find a significant overall difference between groups on GMP levels. Exposure to single SD in general had a significant impact on histological measurements (crypt and inflammation scores) in the distal colon. Effects were not related to pre-stress levels of corticosterone. There are some additional findings that seem challenging to interpret, such as higher inflammation scores in animals not prior exposed to a stressor compared to animals exposed to foot-shocks. The results confirm previous findings that unrestricted oral ingestion of DSS is an effective method for inducing a colitis-like condition in rodents [19, 20].
It is however important to recognize that initial plasma corticosterone concentrations in the present study (mean ca. 125 ng/ml, range 30–240 ng/ml) are high, especially for those animals in the HIGH group. We therefore point to some arguments that plasma corticosterone concentrations are influenced by several factors including genetics, strain, suppliers, rearing conditions, method of sampling and analysis, housing, time of day and, not least, use of anaesthetics. The lowest levels are observed when plasma samples are obtained from rats habituated to handling and unanaesthetized (e.g. 9 ng/ml) [21]. Hence, we do not report that our levels are ‘basal’ or ‘resting’ levels, but rather levels that indicate when animals are exposed to the normal laboratory activities to which they are accustomed and are therefore designated as ‘pre-stressor’ levels.
Animal models of inflammatory bowel disease (IBD) are of value due to their wide range of options for investigating the various factors related to pathogenesis and to develop and evaluate medical treatment options. However, induced colitis models do not reproduce the complexity of the disease, and there are some limitations considering diagnostic subcategories that are rarely investigated in animals.
A valid model of human UC requires that animals develop a colitis-like condition with prominent neutrophils in the epithelium, cryptitis, crypt abscesses and erosions. High faecal levels of calprotectin indicate intestinal inflammation, and due to the simple and non-invasive sampling, numerous tests can be performed repeatedly on the same individual. We collected faeces from the rats thus enabling detection of the rodent granulocyte marker protein (GMP), the parallel to human faecal calprotectin, where high levels are strong indicators of colonic inflammation [22, 23]. Overall, longer exposure to DSS caused higher GMP levels. When analysing levels of GMP prior to the administration of DSS, after 48 hours and after 7 days on DSS, we were unable to find significant differences between rats exposed to SD, IFS, and those not prior exposed to an intended stressor. Thus, neither of the two stress procedures rendered the animals more sensitive to DSS as measured by GMP.
In the present study the natural social stressor was conducted in the dark/active phase [16] while the “unnatural” IFS procedure was conducted in the light/resting phase as previously done in our laboratory [14]. Social defeat is normally conducted during the rats’ active phase, and this was chosen also for the present study. Among others, a study on chronic mild stress (CMS) have shown that rats exposed to stress during the light/resting phase displayed signs of depressive- and anxiety-like behaviours, which was not seen for rats exposed to CMS during the dark/active phase [24]. For the present study, one may speculate that the effectiveness of social defeat was diminished compared to foot shock, and that there would be a difference between SD and foot shock in GMP had SD been administered during the light phase.
The histology data in the present study revealed that most of the erosions occurred in the distal colon and not in the proximal part of the colon. This result is in line with clinical findings where some patients with proctitis or left-sided colitis might also have a caecal patch of inflammation. Bloody diarrhoea is the characteristic symptom of the disease, but supportive findings are vital for establishing the diagnose [25 for update].
Importantly, the anatomy of rodent intestines is unlike human anatomy where the left-sided colitis is parallel to the subdivided distal part. In rats, the distal part lies on the right-hand side where the muscular layer is thicker than in the proximal left colon. Nonetheless, there is evidence for a similarity between the rat colon anatomy and the human colon anatomy [26]. In our study, rats exposed to single SD had significantly higher crypt scores and inflammation scores in the distal part of the colon compared to rats exposed to IFS, which may indicate differences in the consequences of being exposed to the more natural stressor of physical attack versus an “unnatural” stressor on immune function. Consequences on behaviour have been studied were for instance rats exposed to social defeat display significantly elevated startle responses compared to those previously exposed to brief foot-shocks [6].
Social defeat, as a result of territorial aggression is associated with emotional stress. It would be of interest to further study differences in aggressive behaviour in relation to susceptibility to induced intestinal inflammation. Studies have shown that animals with an aggressive and proactive coping style (fighting back during a SD confrontation) tend to have a higher sympathetic stress reactivity and lower corticosterone response than do reactive coping animals (non-aggressive submissive) which have a higher parasympathetic response and in general react with the highest corticosterone response [27]. Strategies of stress coping in humans are related to differences in immuno-stimulation [28]. Investigation of stress coping may therefore be of importance in animal studies on ulcerative colitis when using DSS after single SD or even other experimental stressors.
Surprisingly, rats that had not been exposed to either IFS or single SD had significantly higher inflammation scores in the distal colon compared to those who had received foot-shocks. We have no reason to underestimate the effects of IFS, but further studies should investigate the time course of recovery from various stressors. The effect of ten foot-shocks might have diminished to such an extent that these animals were more comparable to those left undisturbed. One limitation is lack of data on corticosterone measures immediately after stress procedures, which could have been informative, however, in the protocol we decided to include only one sampling procedure since there is a possibility of ceiling effects immediate after stress.
Most pre-clinical models on stress-related disorders have focused on male rats, despite women seeming to be more susceptible to stress-related symptoms in general. Here, we used only male rats, as the social defeat model was chosen as the social stressor, and the same gender should be used for subjects of both single SD and IFS. Studies on social defeat have traditionally been restricted to males since this model relies on territorial aggression between male rodents. This innate behavior is not normally seen in female rodents [29]. In addition, male rodents in general do not display aggressiveness towards females when introduced into their territory unless the male resident is a pathological aggressor [30]. In recent years, efforts have been made to establish social stress models using female rats [29]. Future studies on social stress models should consider using female rodents. In humans, the incidence of ulcerative colitis is similar in men and women before the age of 45, whilst above the age of 45, men have a higher risk than women [31]. It is therefore recommended to include both males and females if feasible when modelling human colitis.
We are aware that to strengthen the validity of the present study, several immune outcome measures should be included to better establish the presence of inflammation such as the concentration of pro-inflammatory cytokines in mucosal tissue. Another limitation in the experimental design is the time between stressing events, the number of behavioral tests, and the induction of DSS colitis which makes it difficult to draw direct correlation between stressors and the outcome measurements. We still want to highlight the fact that for human ulcerative colitis, being exposed to several stressors are highly associated with disease flare-ups and exacerbation of inflammatory symptoms. In conclusion, IFS leads to remarkably lower inflammation scores for both the proximal and distal colon, that is a less pronounced inflammation compared to controls and rats exposed to social defeat. It seems like being physically attacked renders the animals more susceptible to DSS-induced colitis. This was shown by more colonic damage in rats previously exposed to a single SD, where both groups had undergone similar experimentally inducement of colitis. The single SD in general had significantly more impact on histological measurements than brief IFS, shown by higher crypt and inflammation scores in the distal colon, but no differences in GMP measurements were found between the groups. The higher grade of inflammation and tissue damage in the distal part of the colon in socially defeated rats may indicate presentation of differences between the outcomes of ‘natural’ versus ‘unnatural’ stressors. A single social defeat seems to be more adverse for the animals in the present study, but further investigations are recommended to validate a broader range of outcomes comparing two such different rodent stress models in addition to the aspect of recovery.
Supporting information
(XLSX)
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This study was performed with support from the Faculty of Psychology, University of Bergen, Norway
References
- 1.Takahashi A, Flanigan ME, McEwen BS, Russo SJ. Aggression, social stress, and the immune system in humans and animal models. Frontiers in Behavioral Neuroscience. 2018;12, Article 56. 10.3389/fnbeh.2018.00056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Konturek PC, Brzozowski T, Konturek SJ. Stress And The Gut: Pathophysiology, Clinical Consequences, Diagnostic Approach And Treatment Options. Journal of Physiology and Pharmacology. 2011;62(6):591–599. [PubMed] [Google Scholar]
- 3.Takada M, Nishida K, Kataoka-Kato A, Gondo Y, Ishikawa H, Suda K, et al. Probiotic Lactobacillus casei strain Shirota relieves stress-associated symptoms by modulating the gut-brain interaction in human and animal models. Neurogastroenterology and Motility. 2016;28(7):1027–36. doi: 10.1111/nmo.12804 [DOI] [PubMed] [Google Scholar]
- 4.Bernstein CN, Zhang L, Lix LM, Graff LA, Walker JR, Fisk JD et al. The Validity and Reliability of Screening Measures for Depression and Anxiety Disorders in Inflammatory Bowel Disease. Inflammatory Bowel Diseases. 2018;24(9):1867–1875. doi: 10.1093/ibd/izy068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hammels C, Pishva E, De Vry J, van den Hove DLA, Prickaerts J, van Winkel R et al. Defeat stress in rodents: From behavior to molecules. Neuroscience and Biobehavioral Reviews. 2015;59:111–140. doi: 10.1016/j.neubiorev.2015.10.006 [DOI] [PubMed] [Google Scholar]
- 6.Kinn Rød AM, Milde AM, Grønli J, Jellestad FK, Sundberg H, Murison R. Long-term effects of footshock and social defeat on anxiety-like behaviours in rats: Relationships to pre-stressor plasma corticosterone concentration. Stress. 2012;15(6):658–670. doi: 10.3109/10253890.2012.663836 [DOI] [PubMed] [Google Scholar]
- 7.Gerova VA, Stoynov SG, Katsarov DS, Svinarov DA. Increased intestinal permeability in inflammatory bowel diseases assessed by iohexol test. World Journal of Gastroenterology. 2011;7(17):2211–5. doi: 10.3748/wjg.v17.i17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Berstad A, Arslan G, Folvik G. Relationship between Intestinal Permeability and Calprotectin Concentration in Gut Lavage Fluid. Scandinavian Journal of Gastroenterology. 2000;35:64–69. doi: 10.1080/003655200750024551 [DOI] [PubMed] [Google Scholar]
- 9.Manceau H, Chicha-Cattoir V, Puy H, Peoc’h K. Fecal calprotectin in inflammatory bowel diseases: update and perspectives. Clinical Chemistry and Laboratory Medicine (CCLM). 2017;55(4):473–483. doi: 10.1515/cclm-2016-0522 [DOI] [PubMed] [Google Scholar]
- 10.Mumolo MG, Bertani L, Ceccarelli L, Laino G, Di Fluri G, Albano E et al. From bench to bedside: Fecal calprotectin in inflammatory bowel diseases clinical setting. World Journal of Gastroenterology. 2018;7(24):3681–3694. doi: 10.3748/wjg.v24.i33.3681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lamb CA, Mansfield JC. Measurement of faecal calprotectin and lactoferrin in inflammatory bowel disease. Frontline Gastroenterology. 2011;2:13–18. doi: 10.1136/fg.2010.001362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Milde AM, Arslan G, Røseth A, Berstad A, Overmier JB, Murison R. Intestinal permeability and faecal granulocyte marker protein in dextran sulphate sodium—Induced colitis in rats. Scandinavian Journal of Laboratory Animal Science. 2003a;30(4):169–175. doi: 10.23675/sjlas.v30i4.51 [DOI] [Google Scholar]
- 13.Reber SO, Obermeier F, Straub HR, Falk W, Neumann ID. Chronic intermittent psychosocial stress (social defeat/overcrowding) in mice increases the severity of an acute DSS-induced colitis and impairs regeneration. Endocrinology. 2006;147(10):4968–4976. doi: 10.1210/en.2006-0347 [DOI] [PubMed] [Google Scholar]
- 14.Milde AM, Sundberg H, Roseth AG, Murison R. Proactive Sensitizing Effects of Acute Stress on Acoustic Startle Responses and Experimentally Induced Colitis in Rats: Relationship to Corticosterone. Stress. 2003b;649–57. doi: 10.1080/1025389031000075808 [DOI] [PubMed] [Google Scholar]
- 15.Allen-Rowlands CF, Allen JP, Greer MA, Wilson M. Circadian rhythmicity of ACTH and corticosterone in the rat. Journal of Endocrinological Investigation. 1980;3(4):371–377. doi: 10.1007/BF03349373 [DOI] [PubMed] [Google Scholar]
- 16.Meerlo P, Overkamp GJ, Benning MA, Koolhaas JM, Van den Hoofdakker RH. Long-term changes in open field behaviour following a single social defeat in rats can be reversed by sleep deprivation. Physiology and Behavior. 1996;60(1): 115–119. doi: 10.1016/0031-9384(95)02271-6 [DOI] [PubMed] [Google Scholar]
- 17.Murthy S, Cooper H, Shim H, Shah R, Ibrahim S, Sedergran D. Treatment of dextran sulfate sodium-induced murine colitis by intracolonic cyclosporin. Digestive Diseases and Sciences. 1993;38(9):1722–1734. doi: 10.1007/BF01303184 [DOI] [PubMed] [Google Scholar]
- 18.Onderdonk AB, Bartlett JG. Bacteriological studies of experimental ulcerative colitis. American Journal of Clinicial Nutrition. 1979;32(1):258–265. doi: 10.1093/ajcn/32.1.258 [DOI] [PubMed] [Google Scholar]
- 19.Martin JC, Bériou G, Josien R. Dextran Sulfate Sodium (DSS)-Induced Acute Colitis in the Rat. Methods in Molecular Biology. 2016;1371:197–203. doi: 10.1007/978-1-4939-3139-2_12 [DOI] [PubMed] [Google Scholar]
- 20.El-Salhy M, Umezawa K, Hatlebakk JG, Gilja OH. Abnormal differentiation of stem cells into enteroendocrine cells in rats with DSS-induced colitis. Molecular Medicine Reports. 2017;1(4):2106–2112. doi: 10.3892/mmr.2017.6266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Muñoz-Abellán C, Rabasa C, Daviu N, Nadal R, Armario A. Behavioral and endocrine consequences of simultaneous exposure to two different stressors in rats: Interaction or independence? Plos One, 2011;6(6):e21426. 10.1371/journal.pone.0021426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Derkacz A, Olczyk P, Komosinska-Vassev K. Diagnostic Markers for Nonspecific Inflammatory Bowel Diseases. Dis. Markers, 2018;11:7451946. doi: 10.1155/2018/7451946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Erichsen K, Milde AM, Arslan G, Helgeland L, Gudbrandsen OA, Ulvik RJ et al. Low-dose oral ferrous fumarate aggravated intestinal inflammation in rats with DSS-induced colitis. Inflammatory Bowel Diseases. 2005;11(8), 744–748. doi: 10.1097/01.mib.0000174374.83601.86 [DOI] [PubMed] [Google Scholar]
- 24.Aslani S., Harb M., Costa P., Almeida O., Sousa N., & Palha J. (2014). Day and night: diurnal phase influences the response to chronic mild stress. Frontiers in Behavioral Neuroscience, 8. Retrieved from https://www.frontiersin.org/article/10.3389/fnbeh.2014.00082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lynch WD, Hsu R. Ulcerative colitis. 2021;18. In: StatPearls [Internet]. Treasure Island (FL):StatPearls Publishing; 2022. Jan-. [Google Scholar]
- 26.Bartolí R, Boix J, Òdena G, De la Ossa ND, de Vega Moreno et al. Colonoscopy in rats: An endoscopic, histological and tomographic study. World Journal of Gastrointestinal Endoscopy. 2013;16(5):226–230. doi: 10.4253/wjge.v5.i5.226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Koolhaas JM. Coping style and immunity in animals: Making sense of individual variation. Brain Behav Immun, 2008;22:662–667. doi: 10.1016/j.bbi.2007.11.006 [DOI] [PubMed] [Google Scholar]
- 28.Zozulya AA, Gabaeva MV, Sokolov OY, Surkina ID, Kost NV. Personality, Coping Style, and Constitutional Neuroimmunology. Journal of Immunotoxicology. 2008;5(2):221–225. doi: 10.1080/15476910802131444 [DOI] [PubMed] [Google Scholar]
- 29.Takahashi A. Toward Understanding the Sex Differences in the Biological Mechanism of Social Stress in Mouse Models. Frontiers in Psychiatry. 2021;16. 10.3389/fpsyt.2021.644161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Koolhaas JM, Coppens CM, de Boer SF, Buwalda B, Meerlo P, Timmermans PJ. The resident-intruder paradigm: a standardized test for aggression, violence and social stress. Journal of Visual Experiments JoVE. 2013;77:e4367. 10.3791/4367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rustgi SD, Kayal M, Shah SC. Sex-based differences in inflammatory bowel diseases: a review. Therapeutic Advances in Gastroenterology. 2020;13:1–11. doi: 10.1177/1756284820915043 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.