Abstract
Background
The spread of antibiotic-resistant bacteria may be driven by human–animal–environment interactions, especially in regions with limited restrictions on antibiotic use, widespread food animal production, and free-roaming domestic animals. In this study, we aimed to identify risk factors related to commercial food animal production, small-scale or “backyard” food animal production, domestic animal ownership, and practices related to animal handling, waste disposal, and antibiotic use in Ecuadorian communities.
Methods and findings
We conducted a repeated measures study from 2018 to 2021 in 7 semirural parishes of Quito, Ecuador to identify determinants of third-generation cephalosporin-resistant E. coli (3GCR-EC) and extended-spectrum beta-lactamase E. coli (ESBL-EC) in children. We collected 1,699 fecal samples from 600 children and 1,871 domestic animal fecal samples from 376 of the same households at up to 5 time points per household over the 3-year study period. We used multivariable log-binomial regression models to estimate relative risks (RR) of 3GCR-EC and ESBL-EC carriage, adjusting for child sex and age, caregiver education, household wealth, and recent child antibiotic use. Risk factors for 3GCR-EC included living within 5 km of more than 5 commercial food animal operations (RR: 1.26; 95% confidence interval (CI): 1.10, 1.45; p-value: 0.001), household pig ownership (RR: 1.23; 95% CI: 1.02, 1.48; p-value: 0.030) and child pet contact (RR: 1.23; 95% CI: 1.09, 1.39; p-value: 0.001). Risk factors for ESBL-EC were dog ownership (RR: 1.35; 95% CI: 1.00, 1.83; p-value: 0.053), child pet contact (RR: 1.54; 95% CI: 1.10, 2.16; p-value: 0.012), and placing animal feces on household land/crops (RR: 1.63; 95% CI: 1.09, 2.46; p-value: 0.019). The primary limitations of this study are the use of proxy and self-reported exposure measures and the use of a single beta-lactamase drug (ceftazidime with clavulanic acid) in combination disk diffusion tests for ESBL confirmation, potentially underestimating phenotypic ESBL production among cephalosporin-resistant E. coli isolates. To improve ESBL determination, it is recommended to use 2 combination disk diffusion tests (ceftazidime with clavulanic acid and cefotaxime with clavulanic acid) for ESBL confirmatory testing. Future studies should also characterize transmission pathways by assessing antibiotic resistance in commercial food animals and environmental reservoirs.
Conclusions
In this study, we observed an increase in enteric colonization of antibiotic-resistant bacteria among children with exposures to domestic animals and their waste in the household environment and children living in areas with a higher density of commercial food animal production operations.
In a repeated-measures study conducted between 2018-2021 in 7 semi-rural parishes of Quito, Ecuador, Heather Kathleen Amato and colleagues aim to identify determinants of antibiotic resistant E.coli infection in children.
Author summary
Why was this study done?
An estimated 1.27 million deaths in 2019 were attributable to bacterial antibiotic-resistant infections, 89% of which occurred in low- and middle-income countries (LMICs).
Small-scale and commercial-scale food animal production is expanding rapidly in middle-income countries like Ecuador and antibiotic use in food animals is increasing at the fastest rate in these settings, fueling the emergence and selection of multidrug-resistant bacteria.
Evidence is needed to inform action against the spread of antibiotic resistance in communities with varying degrees of both small-scale or “backyard” and commercial food animal production, especially in South America where research on community-acquired antibiotic resistance is lacking.
What did the researchers do and find?
We conducted a repeated measures study in a food animal-producing region in Ecuador to compare the risk of antibiotic-resistant bacterial (ARB) carriage among children with varying degrees of exposure to backyard and commercial food animals.
We used multivariable log-binomial regression models to estimate relative risks (RR) of ARB carriage, adjusting for child sex and age, caregiver education, household wealth, and recent child antibiotic use.
Living within 5 km of >5 commercial food animal operations, household pig or dog ownership, child contact with pets, and placing animal feces on household crops were identified as significant risk factors for ARB carriage in children.
What do these findings mean?
This study underscores the need for improved waste management and surveillance of antibiotic resistance in LMICs with widespread food animal production.
A significant challenge remains: There is a lack of available data on antibiotic usage and resistance in commercial food animal operations and their effluent, in Ecuador and globally, due to limited oversight and surveillance.
In future studies, researchers should work with local and national governments to monitor antibiotic use and resistance in food animals, food animal production waste, nearby environmental reservoirs, and food animal products in order to more accurately characterize sources of exposure and transmission routes of community-acquired ARB.
The primary limitations of this study are the use of proxy and self-reported exposure measures and the use of a single beta-lactamase drug (ceftazidime with clavulanic acid) in combination disk diffusion tests for ESBL confirmation, potentially underestimating phenotypic ESBL production among cephalosporin-resistant E. coli isolates.
Introduction
Environmental fecal contamination from food animal production is increasingly recognized as an important contributor to the global antibiotic resistance crisis. Globally, large quantities of clinically important antibiotics are administered to food animals (poultry and livestock raised for meat and dairy products) to promote growth and prevent infection [1]. An estimated 1.27 million deaths in 2019 were attributable to bacterial antibiotic-resistant infections, 89% of which occurred in low- and middle-income countries (LMICs) [2]. Antibiotic-resistant bacteria, including extended-spectrum beta-lactamase producing Enterobacterales (ESBL-E), found in humans have been linked to food animals [3,4]. ESBL-E—deemed a serious threat to global public health by the World Health Organization and the US Centers for Disease Control and Prevention—confer resistance to a broad spectrum of beta-lactam antibiotics including penicillins and cephalosporins, the most commonly used treatments for bacterial infections [5,6].
With the rapid expansion of food animal production in LMICs, antibiotic use in these settings is projected to increase by upwards of 200% from 2010 to 2030, fueling the emergence and selection of these multidrug-resistant bacteria [1]. Estimating the risks of ESBL-E colonization in LMIC communities with exposures to food animal production is a crucial step towards developing strategies to combat the global spread of antibiotic-resistant bacteria. With limited treatment options, cephalosporin-resistant and ESBL-producing bacterial infections result in longer and more costly hospital stays, increased severity of illness, and increased risk of mortality [7–10]. Asymptomatic carriage of ESBL-E in commensal gut bacteria may still pose a threat to health; ESBLs are frequently encoded by plasmids that facilitate horizontal transfer of resistance genes, allowing commensal bacteria to share ESBL-encoded genes with pathogenic bacteria [11–14]. Horizontal gene transfer rapidly propagates phenotypic resistance among diverse bacteria in animals, the environment, and humans [11,15]. Globally, the prevalence of ESBL-E is increasing; as of 2018, an estimated 20% of healthy individuals harbor ESBL-producing Escherichia coli in their guts [16].
In upper middle-income countries like Ecuador, both commercial and small-scale or “backyard” food animal production are increasing as population growth and increasing wealth drive consumption of animal products [17]. Antibiotics are largely unregulated in Ecuador and other LMICs; medically important antibiotics are routinely used in both large-scale and small-scale food animal operations at subtherapeutic doses for growth promotion and disease prevention [18,19]. In Ecuador, an estimated 84% of rural households and 29% of urban households own livestock, and small-scale poultry farmers have reported regularly administering a range of 6 different classes of antibiotics [20,21]. Contact with animal waste is elevated in LMICs, increasing the potential for exposure to ESBL-E and other antibiotic-resistant bacteria. Domestic animals and backyard food animals commonly defecate in the household environment [22,23], and young children are frequently exposed to high doses of poultry, livestock, and domestic animal feces through the consumption of soil and hand-to-mouth behaviors [24,25]. Domestic animals and fecal contamination of household soil, food, and drinking water are common sources of resistant bacteria in LMICs [26–29]. Exposure to animal feces can also increase the risk of diarrhea [30–32]. Epidemiological studies in LMICs have focused on exposures to feces or fecal pathogens, broadly; few studies have assessed exposures and risk factors for antibiotic-resistant and ESBL-E infections among children in LMICs [33–36].
Given the anticipated growth in unrestricted antibiotic use for food animals in LMICs, there is an urgent need to quantify the risks of antibiotic-resistant and ESBL-E carriage and infections among children living near small-scale and/or commercial food animal production in these settings. This study aimed to estimate the risk of cephalosporin-resistant and ESBL-producing E. coli carriage among children with varying degrees of exposure to small-scale and/or commercial food animal production in semirural parishes of Quito, Ecuador. We hypothesized a priori that (1) household-level exposures to small-scale food animal production are associated with an increased risk of third-generation cephalosporin-resistant E. coli (3GCR-EC) and ESBL-producing E. coli (ESBL-EC); (2) exposures to commercial food animal production operations in the community are associated with an increased risk of 3GCR-EC and ESBL-EC; and (3) combined exposures to both small-scale and commercial food animal production are associated with a greater increased risk of 3GCR-EC and ESBL-EC in children than small-scale or commercial food animal exposures, alone.
Methods
Ethics statement
This study was approved by the Office for Protection of Human Subjects at the University of California, Berkeley (IRB# 2019-02-11803), the Bioethics Committee at the Universidad San Francisco de Quito (#2017-178M), and the Ecuadorian Health Ministry (#MSPCURI000243-3). Formal written consent was obtained from each primary caregiver of children enrolled in the study prior to participation.
Study site
This study was carried out in semirural communities east of Quito, Ecuador by researchers at the Instituto de Microbiología at the Universidad San Francisco de Quito (USFQ) and the University of California, Berkeley School of Public Health. The study area was approximately 320 km2 and included commercial food animal operations and small-scale or “backyard” production of food animals for both subsistence farming and trade. Irrigation canals fed by nearby rivers flow throughout communities in the study area and are used for subsistence farming and small-scale agricultural production.
Study design
This repeated measures observational study aimed to enroll 360 households through stratified random sampling across 7 semirural parishes. For power calculations and sample size determination, please see SI (Sample Size and Power Calculations in S1 Files). Random sampling was stratified at the neighborhood level (within parishes) based on the following strata, as determined by community-based fieldworkers: (1) backyard food animal production present in neighborhood; (2) backyard food animal production present and commercial food animal production within 1 km of neighborhood; and (3) no backyard food animal production and no commercial production within 1 km. Households were enrolled if they met the following inclusion criteria at the time of enrollment: (1) a primary child caretaker who was over 18 years of age was present; and (2) a child between the ages of 6 months and 5 years old was present in the household. Informed written consent was provided by the child’s caretaker prior to participation in the study. If there was more than 1 child at a given household, the youngest child was selected for participation. Due to high rates of migration out of the study site and loss to follow-up after the onset of the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) pandemic, we enrolled new households each cycle. Households were visited up to 5 times between August 2018 and August 2021 by trained field staff who conducted household surveys and collected household GPS coordinates and biological samples at each visit. This study is reported as per the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline (Supporting information (SI), Checklist in S1 Files). Analysis plans for this manuscript were developed beginning in May 2020 and were last revised in March 2021 after sequencing results became available; we amended our analysis plan to include descriptive results of antibiotic resistance genes (ARGs) identified from sequenced E. coli isolates. This research was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (NIH) under Award Number R01AI135118.
Exposure assessment
The primary exposures of interest were exposures to commercial food animal production in the community and household-level exposures to small-scale food animal production. Exposures and other household characteristics and practices were assessed at each household visit to capture time-varying exposures. Exposure to commercial food animal production was assessed in 3 ways: (1) distance to the nearest commercial food animal production facility; (2) density of commercial food animal production facilities; and (3) proximity to drainage paths of commercial food animal production facilities. Commercial poultry production facilities—vertically integrated operations marked by long barns with a metal roof that typically held approximately 20,000 birds or more—were located using satellite imagery in Google Maps and confirmed as active operations through site visits and ground-truthing. Other types of food animal production operations were identified through local knowledge and ground-truthing.
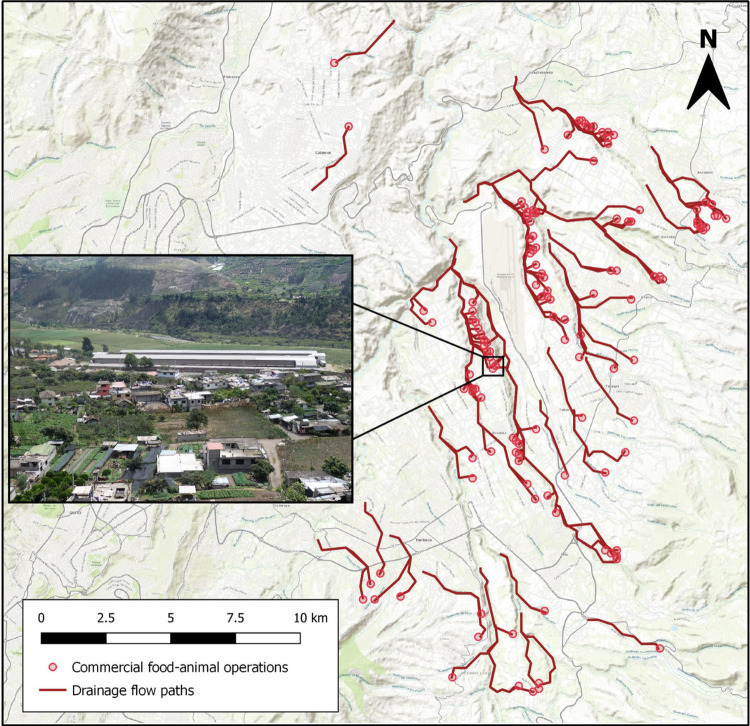
For the first measure of commercial food animal production exposure, Euclidean distance between each household and the nearest active commercial food animal operation was measured at each time point. For the second measure of exposure, the density of commercial food animal operations was assessed by summing the number of operations within a five-kilometer buffer of each household at each time point. We used the sf package in R to create these first 2 exposure variables [37,38]. While proximity may serve as a proxy for the likelihood of environmental contamination from nearby commercial operations, density may serve as a proxy for the extent of environmental contamination from nearby operations. Density of commercial poultry operations has been associated with cephalosporin-resistant E. coli in nearby stream water and sediment in the United States [39]. Finally, for the third exposure measure, drainage paths from each commercial food animal operation were identified using ArcGIS Online Trace Downstream tool, which uses a digital elevation model to identify downstream flow paths from elevation surfaces, drainage directions, river networks, and watershed boundaries obtained from the HydroSHEDS 90 m database [40]. We then created buffers around drainage paths, which ranged from 2.9 to 3.1 km in length, and spatially joined study households to buffer layers in QGIS to identify which households were within 100 or 500 meters of commercial operation drainage paths [41] (Fig 1). Proximity to these drainage paths may capture exposures to antibiotic-resistant bacteria transported from commercial food animal operations through waterways, even when households are located further from the food animal operation, itself.
Fig 1. Map of active commercial food animal production operations (n = 130) and their drainage flow paths in the study area, east of Quito, Ecuador.
The inset is an aerial photograph of commercial poultry production facilities in the study area (image credit: Jay P. Graham). Map created in QGIS; contains information from OpenStreetMap and OpenStreetMap Foundation, which is made available under the Open Database License.
Household-level exposure to small-scale food animal production was defined as household ownership of any food animals (i.e., chickens, pigs, cattle, sheep, goats, ducks, guinea pigs, rabbits, or quail). Household surveys captured information on caregiver-reported household ownership of food animals and other domestic animals, as well as household characteristics, caregiver and child demographics, health status, and other potential risk factors. Surveys were created using Open Data Kit (ODK) Build and trained enumerators used ODK Collect on Android devices for mobile data collection [42]. Encrypted survey forms were sent to ODK Aggregate on a secure server at USFQ upon completion and were subsequently downloaded for de-identification and analysis.
Outcome assessment
A single stool sample from the youngest eligible child per household was collected at each visit to determine enteric carriage of 3GCR-EC and ESBL production based on phenotypic testing, described below. Caregivers were given supplies to collect a child stool sample, which were double-bagged and stored in a fridge or on ice (4°C), and the enumerator returned to collect the sample the next day. In the case that a child had not defecated before the enumerator returned, the enumerator came back to the household the following day to collect the sample. If domestic or food animals were present at the household at the time of data collection, a single stool sample per animal species was collected from the environment where the animals defecate. Fecal samples were placed in sterile containers and stored on ice (4°C) during transportation to the microbiology lab at USFQ. Samples were processed at USFQ within 5 h of collection to identify 3GCR-EC and conduct antibiotic susceptibility testing.
Microbiological methods
To screen for 3GCR-EC and improve sensitivity for detecting ESBL production among both dominant and nondominant strains of E. coli, fecal samples were plated on MacConkey agar (Difco, Sparks, Maryland) with 2 mg/L of ceftriaxone and incubated for 18 h at 37°C [43]. Up to 5 ceftriaxone-resistant isolates phenotypically matching E. coli were selected from each fecal sample and preserved at −80°C in Trypticase Soy Broth medium (Difco, Sparks, Maryland) with 20% glycerol. 3GCR-EC isolates for each fecal sample were thawed and regrown on MacConkey agar at 37°C for 18 to 24 h for evaluation of antibiotic susceptibility by the disk diffusion method (Kirby Bauer test) on Mueller–Hinton agar (Difco, Sparks, Maryland). To confirm presumptive E. coli isolates, colonies were inoculated onto Chromocult coliform agar (Merck, Darmstadt, Germany).
Antibiotic susceptibility testing
Antibiotic susceptibility testing of all 3GCR-EC isolates was conducted for 10 antibiotics: ampicillin (AM; 10 μg), ceftazidime (CAZ; 30 μg), ciprofloxacin (CIP; 5 μg), cefotaxime (CTX; 30 μg), cefazolin (CZ; 30 μg), cefepime (FEP; 30 μg), gentamicin (GM; 10 μg), imipenem (IPM; 10 μg), trimethoprim/sulfamethoxazole (SXT; 1.25 per 23.75 μg), and tetracycline (TE; 30 μg). Isolates were identified as either susceptible or resistant to each antibiotic according to the resistance or susceptibility interpretation criteria from Clinical and Laboratory Standards Institute (CLSI) guidelines [44]. E. coli ATCC 25922 was used as the quality control strain. Multidrug resistance (resistant to 3 or more classes) was determined based on the number of macro-classes to which each isolate was resistant. Macro-classes were defined as cephalosporin/beta-lactamase inhibitors, penicillins, aminoglycosides, carbapenems, fluoroquinolones, tetracyclines, and folate pathway inhibitors.
For phenotypic confirmation of ESBL production, the combination disk diffusion test was used with CAZ and CAZ/CLA (ceftazidime with clavulanic acid) as outlined in the CLSI guidelines [44]. In the first 4 cycles of data collection, up to 5 E. coli isolates per sample were selected and preserved for analysis. 3GCR-EC isolates from the same fecal sample with identical phenotypic resistance profiles were considered duplicates and were de-duplicated prior to analyses. Due to limited laboratory resources after the SARS-CoV-2 pandemic began in 2020 and the high rate of clonal relationships between E. coli isolated from the same sample (based on preliminary sequencing), only 1 E. coli isolate per sample was selected and preserved for analysis during the fifth cycle of data collection.
DNA sequencing and analysis
Genomic DNA was extracted from the isolates using Wizard Genomic DNA Purification (Promega) kits and QIAGEN DNEasy Blood & Tissue Kits according to the manufacturer’s instructions. We conducted quality control of extracted DNA prior to library creation at the University of Minnesota Genomics Center, including PicoGreen quantification, quantitative capillary electrophoretic sizing (Agilent), and functional quantification (KAPA Biosystems qPCR). Whole-genome sequencing was carried out at the University of Minnesota. In brief, we sequenced whole-genome E. coli isolates using either Illumina MiSeq or NovaSeq with Nextera XT libraries. Following sequencing, raw reads were quality-trimmed and adapter-trimmed using trimmomatic [45]. Assemblies of reads was performed using SPAdes [46], then ARGs were identified using ABRicate (version 0.8.13) and a curated version of the ResFinder database [47]. We also performed in silico multilocus sequence typing (MLST) based on 7 housekeeping genes (adk, fumC, gyrB, icd, mdh, purA, and recA), an additional 8 housekeeping genes (dinB, icdA, pabB, polB, putP, trpA, trpB, and uidA), and core genome (cgMLST) using MLST 2.0 [48] and cgMLSTFinder 1.1 [49]. Detailed methods are previously described [50]. Raw reads from isolates sequenced in this study are available at the NCBI Short Read Archive (SRA) under BioProject accession no. PRJNA861272.
Statistical analyses
Multivariable log-binomial regression models were used to estimate unadjusted and adjusted relative risks (RRs) for the associations between commercial or household food animal exposures and 3GCR-EC and ESBL-EC carriage (i.e., main effects). To control for confounding and strong predictors of the outcomes variables, all adjusted models included prespecified covariates (child age, child sex, child antibiotic use in the past 3 months, household asset score as a proxy for socioeconomic status, and caregiver education level) identified using a directed acyclic graph and existing literature (Fig A in S1 Files) [51]. Next, we included an interaction term in adjusted models, comparing combined effects of household and commercial food animal exposures to a single referent group (unexposed to both commercial and household food animals). We also assessed effect measure modification by estimating the effect of each measure of exposure to commercial food animals among those with versus without food animals (i.e., stratum-specific effects). Estimates and P-values from interaction models were used to determine multiplicative interaction based on an alpha level of <0.10. Finally, we ran additional log-binomial regression models to estimated associations between secondary risk factors of interest related to household animal ownership, animal contact and feces management, and presence of 3GCR-/ESBL-EC in animal stool collected at households. Exposures were treated as binary and categorical using cut-points selected based on the data distribution and interpretability. The outcomes, 3GCR-EC and ESBL-EC carriage, were treated as binary and modelled at the isolate level, adjusting for clustering at the individual/household level and estimating robust standard errors with generalized estimating equations. In a sensitivity analysis, we reproduced this main analysis using only the first E. coli isolate per fecal sample to assess the potential for bias due to the change in number of isolates and probability of detecting the outcomes at each time point. Individual observations with missing exposure, outcome, or covariate data were removed for this complete case analysis. Statistical analyses and visualizations were completed in R version 3.6.1 [38] using the dplyr [52], tableone [53], ggplot2 [54], geepack [55], multcomp [56], and car [57] packages. Exposure and outcome data used in epidemiological analyses are published and publicly available on Dryad (https://doi.org/10.5061/dryad.41ns1rnm7).
Results
A total of 605 households across 7 semirural parishes east of Quito, Ecuador were enrolled throughout the study period between July 2018 and September 2021. We enrolled 374 households in the initial cycle of data collection and recruited new households (using the same enrollment criteria) to enroll in subsequent data collection cycles to account for loss to follow-up. During the fourth cycle, data collection was halted in March 2020 due to lockdown restrictions during the SARS-CoV-2 pandemic, resulting in significant loss to follow-up (39.7%, 151/380 lost to follow-up). However, in the fifth cycle in 2021, we re-enrolled 63.4% (241/380) of participants from cycle 3 and 78.8% (186/236) of participants from cycle 4 (Fig B in S1 Files). A total of 1,739 child fecal samples were collected throughout the study period, from which 920 distinct 3GCR-EC colonies were isolated. Two percent (40/1,739) of child fecal samples were missing corresponding survey data, resulting in a total of 910 3GCR-EC isolates from 1,699 child fecal samples across 600 children from different households. The median number of household visits (i.e., fecal samples) for each child was 3; 23% of the 600 households were visited 5 times, 18% 4 times, 11% 3 times, 11% 2 times, and 37% were visited once. Overall, 1.8% (11/605) of all enrolled households were missing either exposure, outcome, or covariate data and were not included in statistical analyses. After removing children with missing data, 904 3GCR-EC isolates from 1,677 child fecal samples from 594 children remained. This resulted in a total of 1,940 observations (including multiple isolates per fecal sample) in the final dataset for the primary statistical analysis.
Household and child characteristics
In a majority of the 594 included households, most primary caregivers had at least a high school or college level education, ranging from 67.1% to 79.5% across the data collection cycles (Table 1). The average age of children participating in the study was 1.8 years during the first cycle of data collection (Table 2). Access to drinking water and sanitation was high; 98.8% of households had a flush toilet (to sewer or septic tank), 92.2% had piped drinking water inside their home, and 98.6% had 24-h access to drinking water (Table M in S1 Files). Over 93% of households had household handwashing stations with the presence of both soap and water, confirmed by observation (Table 1). However, caregiver-reported child handwashing frequency suggested that most children rarely (44.4% to 69.6%) washed their hands after contact with animals (Table 2). Over the course of the entire study period, 36.7% of caregivers reported that their child played near animal feces in the last 3 weeks, 36.0% reported that their child had contact with livestock at least once per week in the last 3 months, and 67.9% reported child contact with pets at least once per week in the last 3 months. Seven-day caregiver-reported child diarrhea prevalence was 20.3% at the beginning of the study period in 2018 and declined to 4.2% at the final data collection cycle in 2021 during the SARS-CoV-2 pandemic (Table 2). Similarly, treatment for infection in the last 3 months declined from 31.1% at the first cycle to 8.1% at the fifth cycle; child antibiotic use in the last 3 months also declined from 26.2% at cycle 1 to 5.9% at cycle 5 (Table 2).
Table 1. Characteristics of study households in semirural Quito, Ecuador at each cycle of data collection between July 2018 and September 2021.
| Data collection cycle | |||||
|---|---|---|---|---|---|
| 1 n (%) |
2 n (%) |
3 n (%) |
4 n (%) |
5 n (%) |
|
| Total | 370 (100) | 358 (100) | 365 (100) | 225 (100) | 356 (100) |
| Parish | |||||
| El Quinche | 20 (5.4) | 17 (4.7) | 30 (8.2) | 16 (7.1) | 26 (7.3) |
| Puembo | 20 (5.4) | 21 (5.9) | 37 (10.1) | 16 (7.1) | 23 (6.5) |
| Pifo | 86 (23.2) | 77 (21.5) | 75 (20.5) | 55 (24.4) | 75 (21.1) |
| Tababela | 12 (3.2) | 9 (2.5) | 8 (2.2) | 6 (2.7) | 13 (3.7) |
| Tumbaco | 23 (6.2) | 35 (9.8) | 24 (6.6) | 17 (7.6) | 22 (6.2) |
| Yaruqui | 146 (39.5) | 146 (40.8) | 141 (38.6) | 80 (35.6) | 141 (39.6) |
| Missing | 4 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Caregiver education level | |||||
| High school or college | 276 (74.6) | 263 (73.5) | 256 (70.1) | 151 (67.1) | 283 (79.5) |
| Elementary | 93 (25.1) | 95 (26.5) | 108 (29.6) | 68 (30.2) | 69 (19.4) |
| Missing | 1 (0.3) | 0 (0.0) | 1 (0.3) | 6 (2.7) | 4 (1.1) |
| Household size (mean (SD)) | 4.5 (1.4) | 4.6 (1.5) | 4.6 (1.4) | 4.4 (1.2) | 4.4 (1.5) |
| Household water treatment | |||||
| No treatment | 194 (52.4) | 170 (47.5) | 223 (61.1) | 138 (61.3) | 229 (64.3) |
| Boil | 159 (43.0) | 154 (43.0) | 119 (32.6) | 68 (30.2) | 109 (30.6) |
| Chlorinate or filter | 3 (0.8) | 5 (1.4) | 5 (1.3) | 3 (1.3) | 2 (0.6) |
| Other | 14 (3.8) | 29 (8.1) | 18 (4.9) | 16 (7.1) | 16 (4.5) |
| Household handwashing station | |||||
| Soap and water | 354 (95.7) | 344 (96.1) | 347 (95.1) | 211 (93.8) | 340 (95.5) |
| Soap or water, only | 9 (2.4) | 14 (4.0) | 17 (4.7) | 14 (6.2) | 16 (4.5) |
| Neither | 6 (1.6) | 0 (0.0) | 1 (0.3) | 0 (0.0) | 0 (0.0) |
| Missing | 1 (0.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Caregiver worked with animals in past 6 months a | 105 (28.4) | 79 (22.1) | 80 (21.9) | 73 (32.4) | 113 (31.7) |
| Household owns animals | 233 (63.0) | 241 (67.3) | 244 (66.8) | 157 (69.8) | 236 (66.3) |
| Missing | 3 (0.8) | 1 (0.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Household owns food animals | 110 (29.7) | 117 (32.7) | 107 (29.3) | 63 (28.0) | 92 (25.8) |
| Missing | 2 (0.5) | 1 (0.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Household food animals (mean (SD)) | 6.9 (19.3) | 5.7 (14.1) | 6.9 (19.1) | 9.2 (39.4) | 5.9 (21.3) |
| Antibiotic use for household food animals | 29 (26.4) | 22 (18.8) | 11 (10.3) | 6 (9.5) | 0 (0.0) |
| Missing | 7 (6.4) | 4 (3.4) | 1 (0.9) | 0 (0.0) | 9 (9.8) |
| Distance to nearest commercial food animal operation | |||||
| ≥1.5 km | 155 (41.9) | 162 (45.3) | 154 (42.2) | 97 (43.1) | 170 (47.8) |
| 1–1.49 km | 90 (24.3) | 86 (24.0) | 87 (23.8) | 52 (23.1) | 70 (19.7) |
| 0.5–0.9 km | 60 (16.2) | 54 (15.1) | 73 (20.0) | 45 (20.0) | 71 (19.9) |
| <0.5 km | 61 (16.5) | 56 (15.6) | 51 (14.0) | 31 (13.8) | 45 (12.6) |
| Missing | 4 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Commercial food animal operations within 5 km radius | |||||
| ≤5 | 115 (31.1) | 118 (33.0) | 113 (31.0) | 71 (31.6) | 203 (57.0) |
| 6–10 | 93 (25.1) | 89 (24.9) | 84 (23.0) | 52 (23.1) | 44 (12.4) |
| 11–20 | 88 (23.8) | 90 (25.1) | 76 (20.8) | 51 (22.7) | 47 (13.2) |
| >20 | 70 (18.9) | 61 (17.0) | 92 (25.2) | 51 (22.7) | 62 (17.4) |
| Missing | 4 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Distance to drainage path of commercial food animal operation | |||||
| >500 m | 154 (41.6) | 161 (45.0) | 150 (41.1) | 99 (44.0) | 167 (46.9) |
| 101–500 m | 158 (42.7) | 133 (37.2) | 162 (44.4) | 92 (40.9) | 138 (38.8) |
| ≤100 m | 60 (16.2) | 67 (18.7) | 59 (16.2) | 39 (17.3) | 56 (15.7) |
| Missing | 4 (1.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
a Including working with live animals, with animal feces, or in meat processing.
Table 2. Characteristics and behaviors of children in study households at each cycle of data collection between July 2018 and September 2021.
| Data collection cycle | |||||
|---|---|---|---|---|---|
| 1 n (%) |
2 n (%) |
3 n (%) |
4 n (%) |
5 n (%) |
|
| Total | 370 (100) | 358 (100) | 365 (100) | 225 (100) | 356 (100) |
| Child age in years (mean (SD)) | 1.8 (1.3) | 2.0 (1.3) | 2.4 (1.4) | 3.0 (1.3) | 3.8 (1.6) |
| Child sex | |||||
| Female | 171 (46.2) | 162 (45.3) | 154 (42.2) | 96 (42.7) | 158 (44.4) |
| Male | 199 (53.8) | 196 (54.7) | 211 (57.8) | 129 (57.3) | 198 (55.6) |
| Child contact with livestock in last 3 months | |||||
| Never | 248 (67.0) | 254 (70.9) | 238 (65.2) | 126 (56.0) | 205 (57.6) |
| <1 time per week | 42 (11.4) | 26 (7.3) | 54 (14.8) | 14 (6.2) | 25 (7.0) |
| 1–2 times per week | 31 (8.4) | 32 (8.9) | 39 (10.7) | 50 (22.2) | 50 (14.0) |
| 3+ times per week | 49 (13.2) | 46 (12.8) | 34 (9.3) | 35 (15.6) | 75 (21.1) |
| Missing | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (0.3) |
| Child contact with pets in last 3 months | |||||
| Never | 132 (35.7) | 122 (34.1) | 108 (29.6) | 64 (28.4) | 112 (31.5) |
| <1 time per week | 54 (14.6) | 34 (9.5) | 65 (17.8) | 12 (5.3) | 23 (6.5) |
| 1–2 times per week | 52 (14.1) | 50 (14.0) | 73 (20.0) | 59 (26.2) | 50 (14.0) |
| 3+ times per week | 132 (35.7) | 152 (42.5) | 119 (32.6) | 90 (40.0) | 171 (48.0) |
| Child played near animal feces in last 3 weeks | 115 (31.1) | 99 (27.7) | 148 (40.5) | 77 (34.2) | 175 (49.2) |
| Missing | 2 (0.5) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 3 (0.8) |
| Child handwashing after contact with animals | |||||
| Never | 43 (11.6) | 13 (3.6) | 4 (1.1) | 3 (1.3) | 3 (0.8) |
| Rarely | 185 (50.0) | 249 (69.6) | 245 (67.1) | 129 (57.3) | 158 (44.4) |
| Sometimes | 83 (22.4) | 68 (19.0) | 109 (29.9) | 87 (38.7) | 159 (44.7) |
| Always | 18 (4.9) | 9 (2.5) | 7 (1.9) | 6 (2.7) | 36 (10.1) |
| Do not know/does not apply | 41 (11.1) | 19 (5.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Child had diarrhea in last 7 days | 75 (20.3) | 77 (21.5) | 43 (11.8) | 36 (16.0) | 15 (4.2) |
| Missing | 2 (0.5) | 1 (0.3) | 1 (0.3) | 0 (0.0) | 2 (0.6) |
| Child treated for infection in last 3 months | 115 (31.1) | 98 (27.4) | 84 (23.0) | 54 (24.0) | 29 (8.1) |
| Missing | 3 (0.8) | 1 (0.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Child took antibiotics in last 3 months | 97 (26.2) | 67 (18.7) | 63 (17.3) | 26 (11.6) | 21 (5.9) |
| Missing | 0 (0.0) | 2 (0.6) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
Food animal production and domestic animals
We identified 130 active commercial food animal operations in our study site including 122 poultry facilities, 5 hog facilities, 2 horse facilities, and 1 milk production facility. Across the 7 parishes in the study site, 4 parishes (Tababela, Tumbaco, Checa (Chilpa), and Pifo) had low-intensity commercial production with fewer than 10 commercial food animal operations, each. Three parishes (El Quinche, Puembo, and Yaruqui) had high-intensity production, with more than 30 commercial food animal operations, each (Table F in S1 Files). More than half of study households were located <1.5 km from a commercial food animal operation (55.7%) or were within 500 m of a commercial operation’s drainage flow path (56.1%) throughout the study period. During the first 4 cycles of data collection, at least 67% of households were within 5 km of 6 or more commercial food animal operations; this dropped to 43% in the fifth cycle (Table 1).
We collected 1,871 animal fecal samples from 376 matched households and identified 1,060 3GCR-EC isolates to assess as a potential risk factor. Throughout the study period, 66.4% of caregivers reported owning any type of animal, with 29.2% owning food animals and an average of 5.7 to 9.2 food animals at each data collection cycle (Table 1). Among households that owned food animals, reported antibiotic use in food animals was low, declining from 26.4% at the beginning of the study period to 0% at the end of the study (Table 1). Thirty-two percent of households owned backyard chickens, 17% owned guinea pigs, 11% owned pigs, 7% owned cattle, and 3% owned goats or sheep. The average flock size for backyard chickens was 13.7 (standard deviation (SD): 29.9). There was an average of 16 guinea pigs (SD: 19.6), 4.3 pigs (SD: 6.6), 2.8 cows (SD: 2.6), 2.9 goats (SD: 3.2), and 4.0 sheep (SD: 6.3). Eighty percent of households owned dogs, with an average of 2.3 dogs per household (SD: 1.6).
Characterization of 3GCR-EC isolates
Sixty-one percent of children (n = 365/600) were carriers of 3GCR-EC and 25% (n = 149/600) were carriers of ESBL-EC at least once throughout the study period. 3GCR-EC were detected in 38% of child samples (n = 652/1,699) and 51% of animal samples (n = 959/1,871) from matched households. Of 910 3GCR-EC isolated from child samples, 36% were resistant to fourth-generation cephalosporin, cefepime, in phenotypic susceptibility testing (Table B in S1 Files). Eighty-six percent of child 3GCR-EC isolates were multidrug-resistant (3 or more classes), 37% were extensively drug-resistant (5 or more classes), and 22% were phenotypic ESBL producers (Table B in S1 Files). Notably, 5 children (<1%) from different households had 1 E. coli isolate that was phenotypically resistant to the carbapenem drug, imipenem (Table B in S1 Files). All of these imipenem-resistant isolates were multidrug-resistant to 4 or more drug classes, with each isolate also expressing resistance to ampicillin and cephalosporin antibiotics. Phenotypic resistance to antibiotics among 3GCR-EC isolates from animal fecal samples are described in Table C in S1 Files.
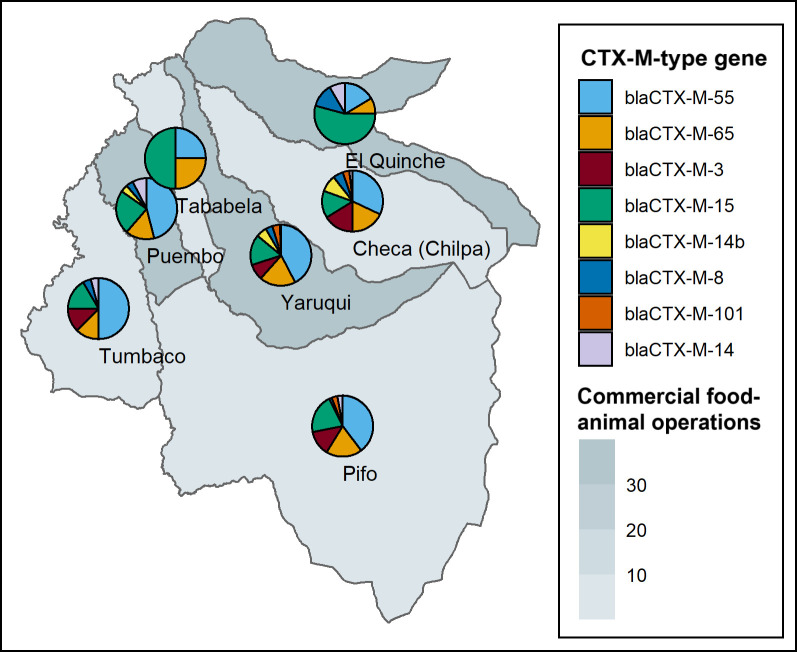
We analyzed whole-genome sequencing data for a subset of 571 3GCR-EC isolated from fecal samples of the 365 children that were 3GCR-EC carriers. We first selected 1 isolate per child for sequencing, then aimed to randomly select an additional 200 isolates to include more ESBL-EC in sequencing analyses. The most prevalent sequence type (ST) among these isolates was ST 10 (7%, 40/571), while other clinically important STs like ST 131 and ST 117 accounted for about 3% of sequenced isolates (Table D in S1 Files). 3GCR-EC isolates had a mean of 9.7 total ARGs (SD: 4.3). Overall, the proportion of 3GCR-EC isolates with bla genes was 65%, 52%, 8%, 5%, and 5% for blaCTX-M-encoding, blaTEM, blaCMY, blaOXA, and blaSHV genes, respectively. Ten (2%) 3GCR-EC isolates had an mcr-1 gene, indicating resistance to the last-line antibiotic colistin. Eighty-two percent (n = 119/145) of phenotypic ESBL-EC and 77% (n = 330/426) of non-ESBL-producing 3GCR-EC carried at least 1 bla gene. Among phenotypic ESBL-EC, the most prevalent bla genes were blaCTX-M-55, blaTEM-141, blaTEM-1B, blaCTX-M-15, and blaOXA-1 (Table E in S1 Files). Among the most prevalent CTX-M-encoding genes, the most prevalent genes in parishes with high-intensity commercial food animal production were blaCTX-M-55, blaCTX-M-65, blaCTX-M-15, and blaCTX-M-14 (Fig 2 and Table H in S1 Files). Parishes with lower intensity commercial food animal production had a higher prevalence of blaCTX-M-3, though blaCTX-M-55, blaCTX-M-65, and blaCTX-M-15 were also detected in these parishes (Fig 2 and Table H in S1 Files).
Fig 2. Map of distribution of CTX-M-type genes from a subset of 571 3GCR-EC isolated from children in 7 parishes east of Quito, Ecuador.
Pie chart sections represent the proportion of isolates with a specific CTX-M-type gene detected, among those represented in the plot legend. Shaded areas represent parishes, where darker shading indicates a higher number of commercial food animal operations in a given parish. Map created in RStudio; Ecuador parish boundary shapefiles are made publicly available by the INEC and the United Nations OCHA. Data and license information are available at https://data.humdata.org/dataset/cod-ab-ecu?. 3GCR-EC, third-generation cephalosporin-resistant E. coli; INEC, Instituto Nacional de Estadística y Censos; OCHA, Office for the Coordination of Humanitarian Affairs.
Risk factors for 3GCR-EC
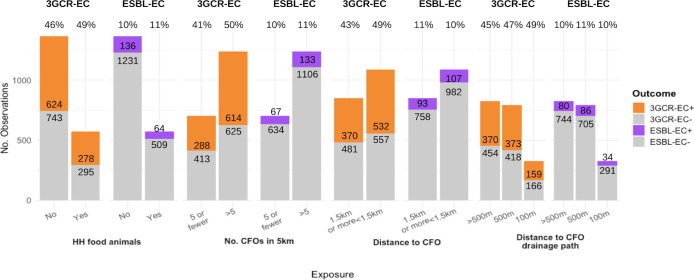
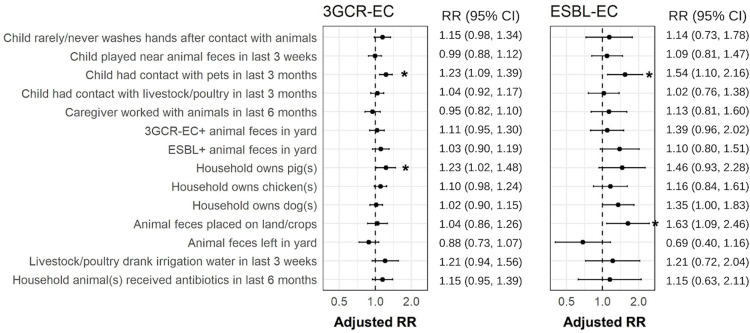
The prevalence of 3GCR-EC was higher in all food animal exposure groups compared to the unexposed, where the denominators include all fecal samples and isolates within each exposure group (Fig 3). In models assessing main effects of household and commercial food animal exposures, the density of commercial operations (>5 in 5 km radius) was the only exposure associated with an increased risk of 3GCR-EC (RR: 1.27; 95% CI: 1.11, 1.46) (Table 3). In models adjusting for interaction between household and commercial food animal exposures, children with >5 commercial food animal operations in a 5-km radius of their household had 1.36 times the risk (95% confidence interval (CI): 1.16, 1.59) of 3GCR-EC carriage than those with ≤5 operations within 5 km when controlling for household food animal ownership (Table 4). However, among those with household food animals, there was no effect of commercial operation density on 3GCR-EC carriage (RR: 1.05; 95% CI: 0.83, 1.33) (Table 4). There was not an excess risk of being exposed to both household food animals and >5 commercial operations (relative excess risk due to interaction (RERI): −0.29, 95% CI: −0.64, 0.06) (Table 4). However, the combination of owning household food animals and living <1.5 km from the nearest commercial operation was associated—with borderline significance—with an increased risk in 3GCR-EC carriage (RR: 1.15; 95% CI: 0.98, 1.36) compared to those without food animals who lived further from commercial food animal operations (Table 4). Proximity to a drainage flow path from a commercial food animal operation was not associated with 3GCR-EC carriage, regardless of household food animal ownership (Table 4). These results were largely robust to sensitivity analyses (Table I in S1 Files). Other risk factors for 3GCR-EC among children included child pet contact in the last 3 months (RR: 1.23; 95% CI: 1.09, 1.39) and pig ownership (RR: 1.23; 95% CI: 1.02, 1.48) (Fig 4) (Table J in S1 Files).
Fig 3. Bar plot of distribution of 3GCR-EC and ESBL-EC carriage stratified by exposures to HH food animals and CFOs.
Number of observations includes fecal samples that were negative for 3GCR-EC (light gray bars for 3GCR-EC), isolates from fecal samples that were 3GCR-EC (orange bars), or ESBL-EC (purple bars) based on phenotypic susceptibility testing, and isolates plus fecal samples negative for both 3GCR-EC and ESBL-EC (light gray bars for ESBL-EC). Percent of observations positive for each outcome are listed above each bar. 3GCR-EC, third-generation cephalosporin-resistant E. coli; CFO, commercial food animal operation; ESBL-EC, extended-spectrum beta-lactamase E. coli; HH, household.
Table 3. Unadjusted and adjusted RRs of 3GCR-EC and ESBL-EC for main effects of household food animal ownership and exposures to commercial food animal production.
| 3GCR-EC | ESBL-EC | |||||||
|---|---|---|---|---|---|---|---|---|
| Unadjusted RR (95% CI) | P-Value | Adjusted RR (95% CI) | P-Value | Unadjusted RR (95% CI) | P-Value | Adjusted RR (95% CI) | P-Value | |
| Household owns food animals (ref = no)a | 1.06 (0.94, 1.19) | 0.388 | 1.04 (0.92, 1.17) | 0.521 | 1.12 (0.82, 1.52) | 0.472 | 1.10 (0.81, 1.51) | 0.541 |
| > 5 CFO in 5 km radius (ref = ≤5)b | 1.26 (1.10, 1.45) | 0.001 | 1.27 (1.11, 1.46) | 0.001 | 1.12 (0.84, 1.51) | 0.444 | 1.14 (0.84, 1.53) | 0.404 |
| < 1.5 km to nearest CFO (ref = ≥ 1.5 km)b | 1.21 (0.84, 1.75) | 0.305 | 1.10 (0.98, 1.25) | 0.118 | 1.24 (0.60, 2.60) | 0.561 | 0.91 (0.68, 1.22) | 0.520 |
| 101–500 m to CFO drainage path (ref = >500 m)b | 1.05 (0.91, 1.21) | 0.486 | 1.04 (0.91, 1.20) | 0.563 | 1.14 (0.83, 1.57) | 0.426 | 1.15 (0.83, 1.59) | 0.392 |
| 0–100 m to CFO drainage path (ref = >500 m)b | 1.13 (0.96, 1.33) | 0.132 | 1.08 (0.92, 1.27) | 0.331 | 1.15 (0.77, 1.71) | 0.510 | 1.15 (0.77, 1.72) | 0.499 |
All RRs estimated with log-binomial regression models using generalized estimating equations to adjust for repeated measures and estimate robust 95% CI. Adjusted RR included the following covariates: caregiver education, asset score, child age and sex, and child antibiotic use in the last 3 months. N = 1,940 observations across 1,677 child fecal samples (including 910 total 3GCR-EC isolates) for 594 children. CFO = commercial food animal operation. RR: relative risk. CI: confidence interval. 3GCR-EC: third-generation cephalosporin-resistant E. coli. ESBL-EC: extended-spectrum beta-lactamase producing E. coli.
a Adjusted models also controlled for number of commercial food animal productions in 5km radius.
b Adjusted models also controlled for household food animal ownership.
Table 4. Adjusted RRs of 3GCR-EC carriage among children given combined exposures to commercial food animal production and effect measure modification by household food animal ownership.
| Adjusted RR for 3GCR-EC (95% CI) | Interaction P-value | RERI (95% CI) | |||
|---|---|---|---|---|---|
| No household food animals | Household food animals | ||||
| No. CFOs in 5 km radius | ≤5 | 1.00 (ref) | 1.22 (0.95, 1.55) | - | - |
| >5 | 1.36 (1.16, 1.59) | 1.28 (1.06, 1.54) | 0.077 | −0.29 (−0.64, 0.06) | |
| >5 within strata of household food animals | 1.36 (1.16, 1.59) | 1.05 (0.83, 1.33) | - | - | |
| Distance to nearest CFO | ≥1.5 km | 1.00 (ref) | 1.02 (0.83, 1.26) | - | - |
| <1.5 km | 1.09 (0.95, 1.27) | 1.15 (0.98, 1.36) | 0.827 | 0.03 (−0.23, 0.30) | |
| <1.5 km within strata of household food animals | 1.09 (0.95, 1.27) | 1.13 (0.91, 1.39) | - | - | |
| Distance to nearest CFO drainage path | >500 m | 1.00 (ref) | 0.98 (0.80, 1.21) | - | - |
| 101–500 m | 1.00 (0.85, 1.18) | 1.16 (0.90, 1.49) | 0.327 | 0.16 (−0.14, 0.45) | |
| ≤100 m | 1.10 (0.91, 1.34) | 1.07 (0.82, 1.38) | 0.818 | −0.04 (−0.36, 0.28) | |
| 101–500 m within strata of household food animals | 1.00 (0.85, 1.18) | 1.14 (0.93, 1.39) | - | - | |
| ≤100 m within strata of household food animals | 1.10 (0.91, 1.34) | 1.05 (0.84, 1.31) | - | - | |
Log-binomial regression models with generalized estimating equations included interaction terms between commercial and household food animal exposure variables, and included the following covariates: caregiver education, asset score, child age and sex, and child antibiotic use in the last 3 months. N = 1,940 observations across 1,677 child fecal samples (including 910 total 3GCR-EC isolates) for 594 children. CFO = commercial food animal operation. RR: relative risk. CI: confidence interval. 3GCR-EC: third-generation cephalosporin-resistant E. coli. RERI: relative excess risk due to interaction.
Fig 4. Additional risk factors for 3GCR-EC and ESBL-EC carriage among children in semirural parishes of Quito, Ecuador.
Points are adjusted RR and error bars are 95% CIs; asterisks (*) indicate significance given alpha = 0.05 (corresponding data including sample sizes and P-values in Tables K and J in S1 Files). RR are adjusted for repeated measures and controlled for the following covariates: caregiver education, asset score, child age and sex, and child antibiotic use in the last 3 months. 3GCR-EC, third-generation cephalosporin-resistant E. coli; CI, confidence interval; ESBL-EC, extended-spectrum beta-lactamase E. coli; RR, relative risk.
Risk factors for ESBL-EC
We did not detect significant associations or interactions between the density of or proximity to commercial food animal operations, household food animal ownership, and ESBL-EC carriage. In models adjusting for interactions between exposures, household food animal ownership and increased proximity to a drainage path were not significantly associated with an increased risk of ESBL-EC carriage among children. However, the effect sizes were larger for combined and increasing intensity of exposures. When controlling for household food animal ownership, the RR for ESBL-EC was 1.10 (95% CI: 0.76, 1.60) for those living 101 to 500 m from a drainage path and was 1.32 (95% CI: 0.71, 2.46) for those 101 to 500 m from a drainage path compared to those >500 m (Table 5). The RR further increased to 1.80 (95% CI: 0.94, 3.45) for those living within 100 m of a drainage path and with household food animals compared to those >500 m from a drainage path and without household food animals; this interaction was near-significant with an interaction P-value of 0.108 (RERI: 0.85; 95% CI: −0.10, 1.81) (Table 5). These findings were also robust to sensitivity analyses (Table I in S1 Files). Additional risk factors of ESBL-EC carriage among children in our study site included placing animal feces on household land/crops (RR: 1.63; 95% CI: 1.09, 2.46), household dog ownership (RR: 1.35; 95% CI: 1.00, 1.83), and child pet contact in the last 3 months (RR: 1.54; 95% CI: 1.10, 2.16) (Fig 4 and Table K in S1 Files).
Table 5. Adjusted RRs of ESBL-EC carriage among children given combined exposures to commercial food animal production and household food animals, including both interaction effects (effects of individual and combined exposures vs. no exposures) and stratum-specific effects (effects of commercial food animal exposures within strata of household food animal ownership vs. no exposures).
| Adjusted RR for ESBL-EC (95% CI) | Interaction P-value | RERI (95% CI) | |||
|---|---|---|---|---|---|
| No household food animals | Household food animals | ||||
| No. CFOs in 5 km radius | ≤5 | 1.00 (ref) | 1.18 (0.69, 2.01) | - | - |
| >5 | 1.17 (0.82, 1.68) | 1.24 (0.82, 1.86) | 0.737 | −0.12 (−0.89, 0.66) | |
| >5 within strata of household food animals | 1.17 (0.82, 1.68) | 1.05 (0.61, 1.81) | - | - | |
| Distance to nearest CFO | ≥1.5 km | 1.00 (ref) | 0.91 (0.57, 1.46) | - | - |
| <1.5 km | 0.81 (0.57, 1.16) | 1.05 (0.69, 1.59) | 0.268 | 0.32 (−0.23, 0.88) | |
| <1.5 km within strata of household food animals | 0.81 (0.57, 1.16) | 1.15 (0.70, 1.91) | - | - | |
| Distance to nearest CFO drainage path | >500 m | 1.00 (ref) | 0.88 (0.50, 1.53) | - | - |
| 101–500 m | 1.10 (0.76, 1.60) | 1.32 (0.71, 2.46) | 0.619 | 0.18 (−0.53, 0.89) | |
| ≤100 m | 0.85 (0.46, 1.57) | 1.80 (0.94, 3.45) | 0.108 | 0.85 (−0.10, 1.81) | |
| 101–500 m within strata of household food animals | 1.10 (0.76, 1.60) | 1.15 (0.73, 1.83) | - | - | |
| ≤100 m within strata of household food animals | 0.85 (0.46, 1.57) | 1.24 (0.82, 1.86) | - | - | |
RRs and robust 95% CIs estimated using log-binomial regression models with generalized estimating equations included interaction terms between commercial and household food animal exposure variables and included the following covariates: caregiver education, asset score, child age and sex, and child antibiotic use in the last 3 months. N = 1,940 observations across 1,677 child fecal samples (including 910 total 3GCR-EC isolates) for 594 children. CFO = commercial food animal operation. RR: relative risk. CI: confidence interval. 3GCR-EC: third-generation cephalosporin-resistant E. coli. RERI: relative excess risk due to interaction.
Discussion
In this study of 7 semirural parishes in Ecuador, we found that increased density of commercial food animal production facilities, household food animal ownership, child pet contact, and rarely/never washing hands after contact with animals were risk factors for 3GCR-EC carriage. The combination of owning household food animals and living within 100 m of a drainage flow path from a commercial food animal operation may have increased the risk of ESBL-EC carriage among children. Other risk factors for ESBL-EC carriage were household dog ownership, child pet contact, and placing animal feces on household land/crops. Clinically relevant STs such as ST 10, ST 131, ST 38, and ST 117 associated with extraintestinal pathogenic E. coli (ExPEC) were detected in child fecal samples, highlighting the public health significance of community-acquired ESBL-producing E. coli carriage [58]. The results of this study emphasize the need for a One Health approach in the control and prevention of antibiotic-resistant bacterial (ARB) infections, particularly in the context of globally expanding commercial food animal production.
Epidemiologic studies have established a clear link between commercial food animal production and ARB carriage among commercial animal farm workers, their household contacts, and community members [19,59–63]. Epidemiological research on associations between small-scale food animal production and community-acquired resistance in humans, however, has been limited. Previous studies have used samples that are not spatiotemporally matched with household-level exposures, cross-sectional study designs, small sample sizes, or descriptive statistics, only [64–67]. These methodological limitations prevent the reliable estimation of associations between small-scale food animal exposures and community-acquired antibiotic-resistant infections. This study attempted to address these gaps by leveraging repeated measures data from 600 households in which household-level exposure data—including data on ARB carriage in household animals—are spatiotemporally matched with outcome data. With our robust longitudinal One Health study design, we were able to estimate the impacts of food animal exposures, domestic animal exposures, and hygiene practices on antibiotic-resistant E. coli carriage in children.
Prevalence estimates in the literature suggest that community-acquired ESBL-EC carriage in healthy populations is increasing globally, with a recent pooled estimate of 21% in 2015 to 2018 [16]. However, there are few studies estimating community-acquired ESBL-producing infections in South America, and estimates are variable. Bezabih and colleagues (2021) estimated a pooled ESBL-EC prevalence of <10% for the Americas, while a 2015 review of ESBL-E estimated a prevalence of 2% for the Americas [68]; both reviews included estimates from studies from the United States in pooled estimates and did not include any studies from Ecuador. Of note, studies in these reviews used selective media to screen for 3GCR-EC or ESBL-EC prior to ESBL confirmatory testing, comparable to the methods used in the present study. A 2008 multicountry study in South and Central America (not including Ecuador) detected ESBL-producing bacteria in 31% of community-acquired intra-abdominal infections [69]. The high prevalence of 3GCR-EC, ESBL-EC, and MDR E. coli carriage among healthy children in our study site is concerning. Over 60% of children were carriers of 3GCR-EC with frequent detection of blaCTX-M genes and 25% were carriers of ESBL-EC at least once throughout the study period, with 86% of all 3GCR-EC being MDR. Even among non-ESBL-producing E. coli isolates based on phenotypic testing, bla genes encoding for extended-spectrum beta-lactam resistance were detected in over 75% of 3GCR-EC isolates. CTX-M-encoding genes blaCTX-M-55, blaCTX-M-65, and blaCTX-M-15 were the most frequently detected bla genes in this food animal-producing region of Ecuador. blaCTX-M-55, blaCTX-M-65, and blaCTX-M-15 are dominant in food animals such as chickens, pigs, and cattle, as well as meat products in China, South Korea, Hong Kong, Canada, Portugal, and elsewhere [70–74]. The distribution of CTX-M-encoding genes in our study site suggests that food animal production plays a critical role in driving the community spread of 3GCR-EC and ESBL-EC in this region of Ecuador.
Other analyses of data collected for this cohort study have previously provided evidence for both horizontal gene transfer and clonal spread of 3GCR-EC in children and animals within and between households in the study site [50,75]. Animal waste management and handling practices were poor in a majority of households with clonal relationships between 3GCR-EC in children and animals [50]. Though evidence of resistant bacteria transmission between backyard chickens, dogs, and humans has been documented in this study site and study period through previous analyses [50,75], it is unclear whether antibiotic use in household food animals and domestic animals or human antibiotic use is driving selection of resistant bacteria in these communities. In fact, reported antibiotic use in children and household animals was low in this study site and participants had limited knowledge about antibiotics and antibiotic stewardship [34]. A recent qualitative study in the same communities found that small-scale poultry and livestock producers typically rely on low-cost traditional veterinary practices rather than administering antibiotics [76]. Notably, antibiotic use in children and domestic animals declined throughout the study period. One possible explanation is that public health measures such as the SARS-CoV-2 pandemic lockdowns may have curbed infectious disease transmission in humans, reducing the need for antibiotic treatment [77]. This hypothesis is supported by the apparent reduction in reported child illness from cycle 3 (before pandemic lockdowns) to cycle 5 (after lockdowns began) in our study. These observed secular trends appear to be nondifferential by household food animal ownership (Table L in S1 Files).
While antibiotic use in household food animals may not be a primary driver of resistance in this area, commercial-scale production operations administering high volumes of antibiotics may be a key driver of emergence and selection of resistant bacteria [1,19]. Results from our study suggest that household food animals and domestic animals still play an important role in determining risk of antibiotic-resistant E. coli carriage among community members. Household animals may act as a vector for transmission of resistant bacteria between environmental reservoirs contaminated by commercial food animal waste and humans with whom they come into contact. GPS-tracked movement patterns of free-range poultry in northwestern Ecuador confirmed that backyard chickens (not given antibiotics) travel an average of 17 m from their household and that this range overlapped with small-scale farms of broiler chickens (given antibiotics) [78]. Free-roaming dogs are also common in urban and rural Quito [79] and have been shown to roam up to 28 km from their homes on average in rural Southern Chile [80]. While some backyard chickens in our study site were kept in coops, some free-range chickens and free-roaming dogs may have been exposed to ARB in environmental contamination from nearby commercial operations. For example, animals may be exposed to high levels of fecal contamination in irrigation canals [81], though household animals drinking irrigation water was not identified as a significant risk factor for human ARB carriage in our study. Household members may be exposed to ARB through contact with companion animals, food animals, or their feces. In fact, children with recent contact with pets, from households with ESBL+ animal feces in the yard, or from households that applied animal waste to household food crops had a slightly greater risk of ESBL-EC colonization in our study. We also reported increasing RRs of ESBL-EC for children in households that both owned food animals and were close to a commercial food animal operation drainage path, which often feed into irrigation canals. Furthermore, the irrigation canals run throughout the study site, which may, in part, drive the geographic spread of beta-lactamase genes we detected in parishes with both high and low numbers of commercial production facilities.
A limitation of the present study is the change in the number of E. coli isolated per sample midway through the study. This may impact the probability of detecting the outcome; for example, phenotypic ESBL-EC would be more likely to be detected for a child with multiple isolates tested for ESBL production compared to a child with only 1 isolate tested. We attempted to address this by analyzing data at the isolate level and adjusting for imbalanced data in our statistical approach rather than aggregating the binary outcome at the individual level. We also included a sensitivity analysis using only the first isolate per sample. Despite some slight differences in point estimates, the overall findings were robust to sensitivity analyses, suggesting a low risk of bias in our outcome assessment methods. Another limitation is that we only isolated 3GCR-EC in order to improve detection of ESBL-EC, limiting our findings to this specific type of ARB species. We did not identify other ESBL-producing Enterobacterales, such as Klebsiella pneumoniae, which also have clinical importance given the high mortality rates associated with ESBL-producing K. pneumoniae infections [82]. Finally, the CLSI guidelines indicate the use of CAZ and CAZ/CLA as well as CTX and CTX/CLA for confirmatory ESBL testing; if a ≥5-mm difference in at least one of these combinations is identified, it is correct to consider the isolate tested as an ESBL-producing isolate. For this study, we evaluated ESBL-production based on the CAZ and CAZ/CLA combination disk diffusion test alone. Therefore, some ESBL-negative isolates reported in the manuscript had the potential to be ESBL-positive if we also had evaluated CTX and CTX/CLA. Based on CLSI guidelines and methods used in this study, all of the ESBL-positive isolates detected and reported in our manuscript are ESBL-positive. The difference between the number of isolates with ESBL genes and isolates with ESBL expression (phenotypically resistant) may be due to the methodology used (only using CAZ and CAZ/CLA and not CTX and CTX/CLA).
Additionally, several risk factors were based on caregiver-reported information and are subject to recall bias. We attempted to reduce bias by asking caregivers to recall weekly frequencies of events (e.g., child contact with animals) over longer periods of time (e.g., 3 months), rather than asking for “yes/no” responses. Finally, this study was halted in the middle of the fourth cycle of data collection due to SARS-CoV-2 lockdowns in March 2020 and did not resume until April 2021, resulting in significant participant dropout. Prior to the pandemic, there was some dropout to do migration out of the study site. This loss to follow-up may have induced some selection bias, though we aimed to address the imbalanced nature of the data in our statistical approach using semiparametric generalized estimating equations with an exchangeable working correlation.
A significant challenge in identifying the source of ARB remains: There is a lack of available data on antibiotic usage and resistance in intensive, commercial food animal operations and their effluent in Ecuador and globally due to limited oversight and surveillance. To accurately characterize the extent to which antibiotic use in commercial production drives community-acquired antibiotic-resistant infections, policies should require that commercial food animal operations monitor and report antibiotic use and conduct routine surveillance for antibiotic-resistant bacteria in food animals, food animal production waste, nearby environmental reservoirs, and food animal products. Future studies should attempt to collect this data to characterize ARB in commercial food animal production settings in Quito. Local and national policies requiring improved waste management practices in large-scale commercial food animal production operations are also lacking, especially in LMICs [83]. Strategies may include containing and/or diverting animal waste, moving animal grazing areas away from waterways, transporting excess waste to more remote areas with sufficient non-food crop land to apply waste as fertilizer, and monitoring levels of nutrients and antibiotic-resistant bacteria in soil and waterways near discharge points. Governments could implement a permitting process that requires commercial operations to submit a waste management plan for approval by agriculture and environmental protection agencies. For example, in the United States, the Environmental Protection Agency established the National Pollution Discharge Elimination System, which regulates the discharge of pollution from point sources at large animal feeding operations and other industrial sites to bodies of water. Agencies could provide subsidies to support producers with initial costs of improving waste management. Policies and regulations should be tailored to farm size; some practices may be cost-prohibitive for small-scale producers, so appropriate financial incentives should be used to promote best management practices among small food animal farms [84]. Finally, national policies should follow global recommendations to restrict the use of clinically important antibiotics like third-generation cephalosporins in food animals [85]. Restricting antibiotic use in cattle farms has been shown to reduce detection of blaCTX-M genes in cattle [86]. To improve the effectiveness of such policies, antibiotic stewardship training programs from a One Health lens could be offered to physicians, veterinarians, and food animal producers with an emphasis on the growing antibiotic resistance crisis.
Our study underscores the need for increased monitoring of waste management practices and improved surveillance of antibiotic use and community-acquired antibiotic resistance in LMICs with widespread food animal production. Increased contact with domestic animals, household food animal ownership, and proximity to large-scale food animal production operations—especially in high-density areas—were associated with an increased risk of antibiotic-resistant bacteria carriage in young children. With zoonotic infectious disease risks and hygiene-related prevention strategies at the forefront of public health messaging due to the SARS-CoV-2 pandemic, national governments should prioritize policies and communication strategies that promote improved food animal waste management and safe hygiene practices to reduce the prevalence of ARB carriage and infections.
Supporting information
Checklist: Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist of items that should be included in reports of cohort studies. Sample Size and Power Calculations. Table A. Prevalence of third-generation cephalosporin-resistant, extended-spectrum beta-lactamase, multidrug-resistant, and extensively drug-resistant E. coli among children. Table B. Proportion of third-generation cephalosporin-resistant E. coli (3GCR-EC) isolates resistant to individual antibiotics in phenotypic susceptibility testing by data collection cycle. Table C. Antibiotic resistance of 3GCR-EC isolates from animal fecal samples (1 colony isolated per fecal sample) collected at the same households as child fecal samples, stratified by animal species. Table D. Prevalence of clinically important sequence types (ST) among sequenced 3GCR-EC isolates (N = 571) from child fecal samples. Table E. Proportion of 3GCR-EC isolates with beta-lactamase genes (among 15 most prevalent) detected in whole-genome sequences, stratified by phenotypic ESBL production. Table F. Prevalence of beta-lactamase resistance genes among sequenced 3GCR-EC isolates from children, stratified by parishes and intensity of commercial food animal production. Table G. Average number of total antibiotic resistance genes (ARGs) per third-generation cephalosporin-resistant E. coli isolate from children, stratified by parish. Table H. Prevalence of CTX-M-type genes among sequenced 3GCR-EC isolates from children, stratified by parish and intensity of commercial food animal production. Table I. Sensitivity analysis results for main analysis associations between combined food animal exposures and 3GCR-EC and ESBL-EC including only 1 isolate per child fecal sample. Table J. Associations between secondary risk factors and 3GCR-EC carriage among children. Table K. Associations between secondary risk factors and ESBL-EC carriage among children. Table L. Secular trends in caregiver-reported child illness and antibiotic use stratified by household food animal ownership. Table M. Access to water and sanitation at households included in the main analysis (N = 594). Fig A. Directed acyclic graph of causal relationship between exposures to commercial and household food animal production and ESBL-E. coli carriage in children. SES: socioeconomic status. ESBL: extended-spectrum beta-lactamase E. coli. Fig B. Flow chart of enrollment and follow-up by data collection cycle. Households were included in the final analysis if they had the necessary exposure, outcome, and covariate data. Fig C. Prevalence of beta-lactamase genes (top 15 most prevalent) among sequenced third-generation cephalosporin-resistant E. coli isolates from children, stratified by phenotypic extended-spectrum beta-lactamase (ESBL) production. Fig D. Prevalence of beta-lactamase genes by type among third-generation cephalosporin-resistant E. coli (3GCR-EC) isolated from children, stratified by parish.
(DOCX)
Acknowledgments
We sincerely thank the data collection team and community partners in Ecuador for their hard work, time, and dedication to this research, especially through the SARS-CoV-2 pandemic. Thanks to Kathleen Kurowski and Rachel Marusinec for their contributions to laboratory work during the initial year of this study. Thanks to Professor Lisa Barcellos, Professor Ayesha Mahmud, and Professor Ellen Eisen for their invaluable input on the methodological approach in the early phases of this analysis. We are deeply saddened by the loss of our co-author, Dr. Lee Riley, who was a kind mentor, outstanding scientist, physician, researcher, and good friend. Finally, we are extremely grateful for the generosity and commitment of our study participants in Quito, without whom this work would not be possible.
Abbreviations
- 3GCR-EC
third-generation cephalosporin-resistant E. coli
- ARG
antibiotic resistance gene
- CFO
commercial food animal operation
- CI
confidence interval
- CLSI
Clinical and Laboratory Standards Institute
- ESBL
extended-spectrum beta-lactamase
- ESBL-E
extended-spectrum beta-lactamase producing Enterobacterales
- ESBL-EC
extended-spectrum beta-lactamase E. coli
- ExPEC
extraintestinal pathogenic E. coli
- LMIC
low- and middle-income country
- MLST
multilocus sequence typing
- NIH
National Institutes of Health
- ODK
Open Data Kit
- RERI
relative excess risk due to interaction
- RR
relative risk
- SARS-CoV-2
Severe Acute Respiratory Syndrome Coronavirus 2
- SD
standard deviation
- ST
sequence type
Data Availability
Raw reads from isolates sequenced in this study are available at the NCBI Short Read Archive (SRA) under BioProject accession no. PRJNA861272. Exposure and outcome data used in epidemiological analyses are published and publicly available on Dryad (https://doi.org/10.5061/dryad.41ns1rnm7).
Funding Statement
This research was supported by the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (NIH) under Award Number R01AI135118, awarded to JG. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Van Boeckel TP, Brower C, Gilbert M, Grenfell BT, Levin SA, Robinson TP, et al. Global trends in antimicrobial use in food animals. Proc Natl Acad Sci U S A. 2015;112:5649–5654. doi: 10.1073/pnas.1503141112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Murray CJL, Ikuta KS, Sharara F, et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022. doi: 10.1016/S0140-6736(21)02724-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Seiffert SN, Hilty M, Perreten V, Endimiani A. Extended-spectrum cephalosporin-resistant Gram-negative organisms in livestock: an emerging problem for human health? Drug Resist Updat. 2013;16:22–45. doi: 10.1016/j.drup.2012.12.001 [DOI] [PubMed] [Google Scholar]
- 4.Ma F, Xu S, Tang Z, Li Z, Zhang L. Use of antimicrobials in food animals and impact of transmission of antimicrobial resistance on humans. Biosafety and Health. 2021;3:32–38. [Google Scholar]
- 5.Centers for Disease Control and Prevention (U.S.) Antibiotic Resistance Threats in the United States, 2019. U.S. Department of Health and Human Services, Centres for Disease Control and Prevention. 2019 [Google Scholar]
- 6.WHO publishes list of bacteria for which new antibiotics are urgently needed. Available from: https://www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed. Accessed 2022 Apr 25.
- 7.Flokas ME, Karanika S, Alevizakos M, Mylonakis E. Prevalence of ESBL-Producing Enterobacteriaceae in Pediatric Bloodstream Infections: A Systematic Review and Meta-Analysis. PLoS ONE. 2017;12:e0171216. doi: 10.1371/journal.pone.0171216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.MacVane SH, Tuttle LO, Nicolau DP. Impact of extended-spectrum β-lactamase-producing organisms on clinical and economic outcomes in patients with urinary tract infection. J Hosp Med. 2014;9:232–238. [DOI] [PubMed] [Google Scholar]
- 9.Ray S, Anand D, Purwar S, Samanta A, Upadhye KV, Gupta P, et al. Association of high mortality with extended–spectrum β-lactamase (ESBL) positive cultures in community acquired infections. J Crit Care. 2018;44:255–260. [DOI] [PubMed] [Google Scholar]
- 10.Cosgrove SE, Kaye KS, Eliopoulous GM, Carmeli Y. Health and economic outcomes of the emergence of third-generation cephalosporin resistance in Enterobacter species. Arch Intern Med. 2002;162:185–190. doi: 10.1001/archinte.162.2.185 [DOI] [PubMed] [Google Scholar]
- 11.Furuya EY, Lowy FD. Antimicrobial-resistant bacteria in the community setting. Nat Rev Microbiol. 2006;4:36–45. doi: 10.1038/nrmicro1325 [DOI] [PubMed] [Google Scholar]
- 12.Pallecchi L, Bartoloni A, Fiorelli C, Mantella A, Di Maggio T, Gamboa H, et al. Rapid dissemination and diversity of CTX-M extended-spectrum beta-lactamase genes in commensal Escherichia coli isolates from healthy children from low-resource settings in Latin America. Antimicrob Agents Chemother. 2007;51:2720–2725. doi: 10.1128/AAC.00026-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Giamarellou H. Multidrug resistance in Gram-negative bacteria that produce extended-spectrum β-lactamases (ESBLs). Clin Microbiol Infect. 2005;11:1–16. [DOI] [PubMed] [Google Scholar]
- 14.Hargreaves ML, Shaw KM, Dobbins G. Clonal Dissemination of Enterobacter cloacae Harboring blaKPC-3 in the Upper Midwestern United States. Antimicrob Agents Chemother. 2015;59:7723–7734. doi: 10.1128/AAC.01291-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.von Wintersdorff CJH, Penders J, van Niekerk JM, Mills ND, Majumder S, van Alphen LB, et al. Dissemination of Antimicrobial Resistance in Microbial Ecosystems through Horizontal Gene Transfer. Front Microbiol. 2016. doi: 10.3389/fmicb.2016.00173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bezabih YM, Sabiiti W, Alamneh E, Bezabih A, Peterson GM, Bezabhe WM, et al. The global prevalence and trend of human intestinal carriage of ESBL-producing Escherichia coli in the community. J Antimicrob Chemother. 2021;76:22–29. doi: 10.1093/jac/dkaa399 [DOI] [PubMed] [Google Scholar]
- 17.Gilbert M, Conchedda G, Van Boeckel TP. Income Disparities and the Global Distribution of Intensively Farmed Chicken and Pigs. PLoS ONE. 2015;10:e0133381. doi: 10.1371/journal.pone.0133381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lowenstein C, Waters WF, Roess A, Leibler JH, Graham JP. Animal Husbandry Practices and Perceptions of Zoonotic Infectious Disease Risks Among Livestock Keepers in a Rural Parish of Quito, Ecuador. Am J Trop Med Hyg. 2016;95:1450–1458. doi: 10.4269/ajtmh.16-0485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Silbergeld EK, Graham J, Price LB. Industrial Food Animal Production, Antimicrobial Resistance, and Human Health. Annu Rev Public Health. 2008;29:151–169. doi: 10.1146/annurev.publhealth.29.020907.090904 [DOI] [PubMed] [Google Scholar]
- 20.Livestock production in Latin America and the Caribbean. Available from: http://www.fao.org/americas/prioridades/produccion-pecuaria/en/. Accessed 2022 Apr 25.
- 21.Braykov NP, Eisenberg JNS, Grossman M. Antibiotic Resistance in Animal and Environmental Samples Associated with Small-Scale Poultry Farming in Northwestern Ecuador. mSphere. 2016. doi: 10.1128/mSphere.00021-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Barnes AN, Anderson JD, Mumma J, Mahmud ZH, Cumming O. The association between domestic animal presence and ownership and household drinking water contamination among peri-urban communities of Kisumu, Kenya. PLoS ONE. 2018;13:e0197587. doi: 10.1371/journal.pone.0197587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ngure F, Gelli A, Becquey E. Exposure to livestock feces and water quality, sanitation, and hygiene (WASH) conditions among caregivers and young children: Formative research in rural Burkina Faso. Am J Trop Med Hyg. 2019;100:998–1004. doi: 10.4269/ajtmh.18-0333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kwong LH, Ercumen A, Pickering AJ, Arsenault JE, Islam M, Parvez SM, et al. Ingestion of fecal bacteria along multiple pathways by young children in rural Bangladesh participating in a cluster-randomized trial of water, sanitation, and hygiene interventions (WASH benefits). Environ Sci Technol. 2020;54:13828–13838. doi: 10.1021/acs.est.0c02606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ngure FM, Humphrey JH, Mbuya MNN, Majo F, Mutasa K, Govha M, et al. Formative research on hygiene behaviors and geophagy among infants and young children and implications of exposure to fecal bacteria. Am J Trop Med Hyg. 2013;89:709–716. doi: 10.4269/ajtmh.12-0568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lateef A, Oloke JK, Gueguimkana EB. The prevalence of bacterial resistance in clinical, food, water and some environmental samples in Southwest Nigeria. Environ Monit Assess. 2005;100:59–69. doi: 10.1007/s10661-005-7062-7 [DOI] [PubMed] [Google Scholar]
- 27.Kalter HD, Gilman RH, Moulton LH, Cullotta AR, Cabrera L, Velapatiño B. Risk factors for antibiotic-resistant Escherichia coli carriage in young children in Peru: community-based cross-sectional prevalence study. Am J Trop Med Hyg. 2010;82:879–888. doi: 10.4269/ajtmh.2010.09-0143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bartley PS, Moretto VT, Silva LK, Cordeiro SM, Reis MG, Blanton RE, et al. THE ASSOCIATION BETWEEN FECAL CONTAMINATION AND ENVIRONMENTAL ANTIBIOTIC RESISTANCE IN RURAL BRAZIL. In: AMERICAN JOURNAL OF TROPICAL MEDICINE AND HYGIENE. AMER SOC TROP MED & HYGIENE 8000 WESTPARK DR, STE 130, MCLEAN, VA 22101 USA, p 26–27. 2017. [Google Scholar]
- 29.Purohit MR, Chandran S, Shah H, Diwan V, Tamhankar AJ, Stålsby Lundborg C. Antibiotic resistance in an Indian rural community: A “one-health” observational study on commensal coliform from humans, animals, and water. Int J Environ Res Public Health. 2017;14:386. doi: 10.3390/ijerph14040386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zambrano LD, Levy K, Menezes NP, Freeman MC. Human diarrhea infections associated with domestic animal husbandry: a systematic review and meta-analysis. Trans R Soc Trop Med Hyg. 2014;108:313–325. doi: 10.1093/trstmh/tru056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bauza V, Byrne DM, Trimmer JT, Lardizabal A, Atiim P, Asigbee MAK, et al. Child soil ingestion in rural Ghana–frequency, caregiver perceptions, relationship with household floor material and associations with child diarrhoea. Trop Med Int Health. 2018;23:558–569. doi: 10.1111/tmi.13050 [DOI] [PubMed] [Google Scholar]
- 32.Penakalapati G, Swarthout J, Delahoy MJ, McAliley L, Wodnik B, Levy K, et al. Exposure to Animal Feces and Human Health: A Systematic Review and Proposed Research Priorities. Environ Sci Technol. 2017;51:11537–11552. doi: 10.1021/acs.est.7b02811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kurowski KM, Marusinec R, Amato HK, Saraiva-Garcia C, Loayza F, Salinas L, et al. Social and Environmental Determinants of Community-Acquired Antimicrobial-Resistant Escherichia coli in Children Living in Semirural Communities of Quito, Ecuador. Am J Trop Med Hyg. 2021;105:600–610. doi: 10.4269/ajtmh.20-0532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Marusinec R, Kurowski KM, Amato HK, Saraiva-Garcia C, Loayza F, Salinas L, et al. Caretaker knowledge, attitudes, and practices (KAP) and carriage of extended-spectrum beta-lactamase-producing E. coli (ESBL-EC) in children in Quito, Ecuador. Antimicrobial Resistance & Infection Control. 2021. doi: 10.1186/s13756-020-00867-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hijazi SM, Fawzi MA, Ali FM, Abd El Galil KH. Multidrug-resistant ESBL-producing Enterobacteriaceae and associated risk factors in community infants in Lebanon. J Infect Dev Ctries. 2016;10:947–955. doi: 10.3855/jidc.7593 [DOI] [PubMed] [Google Scholar]
- 36.Herindrainy P, Randrianirina F, Ratovoson R, Ratsima Hariniana E, Buisson Y, Genel N, et al. Rectal carriage of extended-spectrum beta-lactamase-producing gram-negative bacilli in community settings in Madagascar. PLoS ONE. 2011;6:e22738. doi: 10.1371/journal.pone.0022738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pebesma E, Bivand R, Racine E. sf: Simple Features for R. 2021. [Google Scholar]
- 38.R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria; 2020. [Google Scholar]
- 39.Amato HK, Wong NM, Pelc C, Taylor K, Price LB, Altabet M, et al. Effects of concentrated poultry operations and cropland manure application on antibiotic resistant Escherichia coli and nutrient pollution in Chesapeake Bay watersheds. Sci Total Environ. 2020;735:139401. doi: 10.1016/j.scitotenv.2020.139401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lehner B, Verdin K, Jarvis A. New global hydrography derived from spaceborne elevation data. Eos. 2008;89:93. [Google Scholar]
- 41.QGIS Association. Qgis.org. In: QGIS Geographic Information System. Available from: http://www.qgis.org. 2022.
- 42.Hartung C, Lerer A, Anokwa Y, Tseng C, Brunette W, Borriello G. Open data kit: tools to build information services for developing regions. In: Proceedings of the 4th ACM/IEEE International Conference on Information and Communication Technologies and Development. New York, NY, USA: Association for Computing Machinery; 2010. p 1–12. [Google Scholar]
- 43.Lautenbach E, Bilker WB, Tolomeo P, Maslow JN. Impact of diversity of colonizing strains on strategies for sampling Escherichia coli from fecal specimens. J Clin Microbiol. 2008;46:3094–3096. doi: 10.1128/JCM.00945-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.(2018) Performance standards for antimicrobial susceptibility testing; 27th informational supplement. M100-S28. Wayne, PA. [Google Scholar]
- 45.Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics. 2014;30:2114–2120. doi: 10.1093/bioinformatics/btu170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bankevich A, Nurk S, Antipov D. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zankari E, Hasman H, Cosentino S, Vestergaard M, Rasmussen S, Lund O, et al. Identification of acquired antimicrobial resistance genes. J Antimicrob Chemother. 2012;67:2640–2644. doi: 10.1093/jac/dks261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Larsen MV, Cosentino S, Rasmussen S. Multilocus sequence typing of total-genome-sequenced bacteria. J Clin Microbiol. 2012;50:1355–1361. doi: 10.1128/JCM.06094-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alikhan N-F, Zhou Z, Sergeant MJ, Achtman M. A genomic overview of the population structure of Salmonella. PLoS Genet. 2018;14:e1007261. doi: 10.1371/journal.pgen.1007261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Salinas L, Loayza F, Cárdenas P, Saraiva C, Johnson TJ, Amato H, et al. Environmental spread of extended spectrum beta-lactamase (ESBL) producing Escherichia coli and ESBL genes among children and domestic animals in Ecuador. Environ Health Perspect. 2021;129:27007. doi: 10.1289/EHP7729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lee DS, Choe H-S, Kim HY. Role of age and sex in determining antibiotic resistance in febrile urinary tract infections. Int J Infect Dis. 2016;51:89–96. doi: 10.1016/j.ijid.2016.08.015 [DOI] [PubMed] [Google Scholar]
- 52.Wickham H, François R, Henry L, Müller K, RStudio (2021) dplyr: A Grammar of Data Manipulation. [Google Scholar]
- 53.Yoshida K, Bartel A, Chipman JJ, Bohn J, McGowan LD, Barrett M, et al. gbouzill (2020) tableone: Create “Table 1” to Describe Baseline Characteristics with or without Propensity Score Weights. [Google Scholar]
- 54.Wickham H, Chang W, Henry L, Pedersen TL, Takahashi K, Wilke C, et al. RStudio (2021) ggplot2: Create Elegant Data Visualisations Using the Grammar of Graphics. [Google Scholar]
- 55.Halekoh U, Højsgaard S, Yan J. The R Package geepack for Generalized Estimating Equations. J Stat Softw. 2006;15(2):1–11. [Google Scholar]
- 56.Hothorn T, Bretz F, Westfall P. Simultaneous Inference in General Parametric Models. Biom J. 2008;50:346–363. doi: 10.1002/bimj.200810425 [DOI] [PubMed] [Google Scholar]
- 57.Fox J, Weisberg S. An R Companion to Applied Regression. 2019. [Google Scholar]
- 58.Manges AR, Geum HM, Guo A, Edens TJ, Fibke CD, Pitout JDD. Global Extraintestinal Pathogenic Escherichia coli (ExPEC) Lineages. Clin Microbiol Rev. 2019. doi: 10.1128/CMR.00135-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nadimpalli M, Rinsky JL, Wing S. Persistence of livestock-associated antibiotic-resistant Staphylococcus aureus among industrial hog operation workers in North Carolina over 14 days. Occup Environ Med. 2015;72:90–99. doi: 10.1136/oemed-2014-102095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Nadimpalli M, Stewart JR, Pierce E. Livestock-Associated, Antibiotic-Resistant Staphylococcus aureus Nasal Carriage and Recent Skin and Soft Tissue Infection among Industrial Hog Operation Workers. PLoS ONE. 2016;11:e0165713. doi: 10.1371/journal.pone.0165713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Nadimpalli ML, Stewart JR, Pierce E. Face mask use and persistence of livestock-associated Staphylococcus aureus nasal carriage among industrial hog operation workers and household contacts, USA. Environ Health Perspect. 2018;126:127005. doi: 10.1289/EHP3453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.van den Bogaard AE, Stobberingh EE. Epidemiology of resistance to antibiotics. Links between animals and humans. Int J Antimicrob Agents. 2000;14:327–335. doi: 10.1016/s0924-8579(00)00145-x [DOI] [PubMed] [Google Scholar]
- 63.Dorado-García A, Dohmen W, Bos MEH, Verstappen KM, Houben M, Wagenaar JA, et al. Dose-response relationship between antimicrobial drugs and livestock-associated MRSA in pig farming. Emerg Infect Dis. 2015;21:950–959. doi: 10.3201/eid2106.140706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Li J, Bi Z, Ma S. Inter-host Transmission of Carbapenemase-Producing Escherichia coli among Humans and Backyard Animals. Environ Health Perspect. 2019;127:107009. doi: 10.1289/EHP5251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hedman HD, Eisenberg JNS, Trueba G, Rivera DLV, Herrera RAZ, Barrazueta JV, et al. Impacts of small-scale chicken farming activity on antimicrobial-resistant Escherichia coli carriage in backyard chickens and children in rural Ecuador. One Health. 2019;8:100112. doi: 10.1016/j.onehlt.2019.100112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Addis Z, Kebede N, Worku Z, Gezahegn H, Yirsaw A, Kassa T. Prevalence and antimicrobial resistance of Salmonella isolated from lactating cows and in contact humans in dairy farms of Addis Ababa: a cross sectional study. BMC Infect Dis. 2011;11:222. doi: 10.1186/1471-2334-11-222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Riccobono E, Pallecchi L, Mantella A, Bartalesi F, Zeballos IC, Trigoso C, et al. Carriage of antibiotic-resistant Escherichia coli among healthy children and home-raised chickens: a household study in a resource-limited setting. Microb Drug Resist. 2012;18:83–87. doi: 10.1089/mdr.2011.0003 [DOI] [PubMed] [Google Scholar]
- 68.Karanika K. Arvanitis Fecal Colonization With Extended-spectrum Beta-lactamase–Producing Enterobacteriaceae and Risk Factors Among Healthy Individuals: A Systematic Review and …. Rev. Infect. Dis. [DOI] [PubMed] [Google Scholar]
- 69.Villegas MV, Blanco MG, Sifuentes-Osornio J, Rossi F. Increasing prevalence of extended-spectrum-betalactamase among Gram-negative bacilli in Latin America: 2008 update from the Study for Monitoring Antimicrobial Resistance Trends (SMART). Braz J Infect Dis. 2011;15:34–39. [PubMed] [Google Scholar]
- 70.Leão C, Clemente L, Moura L, Seyfarth AM, Hansen IM, Hendriksen RS, et al. Emergence and Clonal Spread of CTX-M-65-Producing Escherichia coli From Retail Meat in Portugal. Front Microbiol. 2021;12:653595. doi: 10.3389/fmicb.2021.653595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zheng H, Zeng Z, Chen S. Prevalence and characterisation of CTX-M β-lactamases amongst Escherichia coli isolates from healthy food animals in China. Int J Antimicrob Agents. 2012;39:305–310. [DOI] [PubMed] [Google Scholar]
- 72.Park H, Kim J, Ryu S, Jeon B. Predominance of blaCTX-M-65 and blaCTX-M-55 in extended-spectrum β-lactamase-producing Escherichia coli from raw retail chicken in South Korea. J Glob Antimicrob Resist. 2019;17:216–220. [DOI] [PubMed] [Google Scholar]
- 73.Ho C, Lai L. Extensive dissemination of CTX-M-producing Escherichia coli with multidrug resistance to “critically important” antibiotics among food animals in Hong Kong, 2008. –10. J At Mol Phys. [DOI] [PubMed] [Google Scholar]
- 74.Cormier A, Zhang PLC, Chalmers G, Weese JS, Deckert A, Mulvey M, et al. Diversity of CTX-M-positive Escherichia coli recovered from animals in Canada. Vet Microbiol. 2019;231:71–75. doi: 10.1016/j.vetmic.2019.02.031 [DOI] [PubMed] [Google Scholar]
- 75.Salinas L, Cárdenas P, Johnson TJ, Vasco K, Graham J, Trueba G. Diverse Commensal Escherichia coli Clones and Plasmids Disseminate Antimicrobial Resistance Genes in Domestic Animals and Children in a Semirural Community in Ecuador. mSphere. 2019. doi: 10.1128/mSphere.00316-19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Waters WF, Baca M, Graham JP, Butzin-Dozier Z, Vinueza L. Antibiotic use by backyard food animal producers in Ecuador: a qualitative study. BMC Public Health. 2022;22:685. doi: 10.1186/s12889-022-13073-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Brueggemann R. Shaw Changes in the incidence of invasive disease due to Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis during the COVID-19. Lancet Digit. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hedman HD, Zhang L, Trueba G. Spatial Exposure of Agricultural Antimicrobial Resistance in Relation to Free-Ranging Domestic Chicken Movement Patterns among Agricultural Communities in Ecuador. Am J Trop Med Hyg. 2020;103:1803–1809. doi: 10.4269/ajtmh.20-0076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cárdenas M, Grijalva CJ, de la Torre S. Free-Roaming Dog Surveys in Quito, Ecuador: Experiences, Lessons Learned, and Future Work. Front Vet Sci. 2021;8:766348. doi: 10.3389/fvets.2021.766348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Saavedra-Aracena L, Grimm-Seyfarth A, Schüttler E. Do dog-human bonds influence movements of free-ranging dogs in wilderness? Appl Anim Behav Sci. 2021;241:105358. [Google Scholar]
- 81.Montero L, Irazabal J, Cardenas P, Graham JP, Trueba G (2021) Extended-spectrum beta-lactamase producing-Escherichia coli isolated from irrigation waters and produce in Ecuador. Front Microbiol doi: 10.3389/fmicb.2021.709418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Gupta A, Ampofo K, Rubenstein D, Saiman L. Extended Spectrum β Lactamase-producing Klebsiella pneumoniae Infections: a Review of the Literature. J Perinatol. 2003;23:439–443. [DOI] [PubMed] [Google Scholar]
- 83.Lam Y, Fry JP, Nachman KE (2019) Applying an environmental public health lens to the industrialization of food animal production in ten low- and middle-income countries. Globalization and Health 10.1186/s12992-019-0479-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Leet JK, Volz DC. Improving waste management strategies for small livestock farms. Environ Sci Technol. 2013;47:11940–11941. doi: 10.1021/es404078b [DOI] [PubMed] [Google Scholar]
- 85.World Health Organization WHO guidelines on use of medically important antimicrobials in food-producing animals. World Health Organization. 2017 [PubMed] [Google Scholar]
- 86.Nobrega DB, Tang KL, Caffrey NP, De Buck J, Cork SC, Ronksley PE et al. Prevalence of antimicrobial resistance genes and its association with restricted antimicrobial use in food-producing animals: a systematic review and meta-analysis. J Antimicrob Chemother. 2021;76:561–575. doi: 10.1093/jac/dkaa443 [DOI] [PubMed] [Google Scholar]