Abstract
Introduction
Regular physical activity and dietary variety are modifiable and influential factors of health outcomes. However, the cumulative effects of these behaviors are not well understood. Metabolomics may have a promising research potential to extend our knowledge and use it in the attempts to find a long-term and sustainable personalized approach in exercise and diet recommendations.
Objective
The main aim was to investigate the effect of the 12 week very low carbohydrate high fat (VLCHF) diet and high-intensity interval training (HIIT) on lipidomic and metabolomic profiles in individuals with overweight and obesity.
Methods
The participants (N = 91) were randomly allocated to HIIT (N = 22), VLCHF (N = 25), VLCHF + HIIT (N = 25) or control (N = 19) groups for 12 weeks. Fasting plasma samples were collected before the intervention and after 4, 8 and 12 weeks. The samples were then subjected to untargeted lipidomic and metabolomic analyses using reversed phase ultra-high-performance liquid chromatography coupled to high-resolution mass spectrometry.
Results
The VLCHF diet affected plasma lipids considerably while the effect of HIIT was unremarkable. Already after 4 weeks of intervention substantial changes of plasma lipids were found in both VLCHF diet groups. The changes persisted throughout the entire 12 weeks of the VLCHF diet. Specifically, acyl carnitines, plasmalogens, fatty acyl esters of hydroxy fatty acid, sphingomyelin, ceramides, cholesterol esters, fatty acids and 4-hydroxybutyric were identified as lipid families that increased in the VLCHF diet groups whereas lipid families of triglycerides and glycerophospholipids decreased. Additionally, metabolomic analysis showed a decrease of theobromine.
Conclusions
This study deciphers the specific responses to a VLCHF diet, HIIT and their combination by analysing untargeted lipidomic and metabolomic profile. VLCHF diet caused divergent changes of plasma lipids and other metabolites when compared to the exercise and control group which may contribute to a better understanding of metabolic changes and the appraisal of VLCHF diet benefits and harms.
Clinical Trial Registry number
NCT03934476, registered 1st May 2019 https://clinicaltrials.gov/ct2/show/NCT03934476?term=NCT03934476&draw=2&rank=1.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11306-023-02071-1.
Keywords: Carbohydrates, Lipids, Exercise, Diet, Lipidomics, Metabolomics
Introduction
The traditional and usually applied “one size fits all” approach for exercise and diet interventions may not be suitable for a long-term and sustainable effective way in primary and secondary prevention of various chronic noncommunicable diseases such as obesity, diabetes, neurodegenerative diseases, cardiovascular diseases or cancer (Agostoni et al., 2021). Therefore, in the context of nutrition science, the research focus has turned to a personalized or precision nutrition (PN) approach. This is a complex effort to understand the health effects of the relationships among genetics, microbiome, antibiotic and probiotic use, metabolism, food environment and physical activity, as well as economic, social, and other behavioral characteristics (Rodgers & Collins, 2020). Since metabolism plays a central role in nutrition and the rapid development in molecular analysis, metabolomics have been established as an important analytical tool in human nutritional studies (Ulaszewska et al., 2019) and attempts for PN.
Metabolomics, along with genomics, transcriptomics, and proteomics, is a part of novel omics technologies that have shown a promising research potential (Sébédio, 2017). Metabolomics identifies and quantifies low molecular weight molecules in biofluids, tissues or cells (Müller & Bosy-Westphal, 2020). The metabolomics analysis results in lists of molecules (the metabolome) describing the metabolic phenotype of individuals (metabotype) (Sébédio, 2017). However, metabolic profiling approaches to identify markers of dietary exposure and the interpretation of metabolomic data are still in the early developmental phase (Müller & Bosy-Westphal, 2020; Ulaszewska et al., 2019). Also, metabolomic “fingerprinting” should not be confused with a mechanistic understanding (Müller & Bosy-Westphal, 2020).
Nutrition, physical activity and exercise play a crucial role in prevention and treatment of multiple chronic diseases (Pedersen & Saltin, 2015). Systemic and metabolic diseases such as type 2 diabetes, high blood pressure, obesity or metabolic syndrome, have risen globally to an epidemic burden, all of them with typical different pathophysiological patterns. These disorders increase the risk for many other chronic diseases, including cardiovascular diseases and cancer. A large body of evidence showed that avoiding unhealthy eating patterns, particularly high amount of processed or poor quality foods (Ludwig et al., 2023) and regular physical activity support people achieving and maintaining health, and reduce the risk of chronic diseases (Ehrman et al. 2019; Riebe, 2018). In order to treat such diseases, targeted individual exercise and diet programs need to be developed to overcome the actual “One-size fits all” strategy.
The optimal macronutrient proportion in humans’ nutrition has never been established in detail. Thus, it is still unclear whether a high or low carbohydrate intake is the best for human health in modern times. Contrary to usually “beliefs” of a dominant carbohydrate intake to be beneficial, a carbohydrate restricted diet was recently shown to represent an alternative promising dietary strategy to prevent various pathological conditions involving inflammatory and autoimmune pathways (Lennerz et al., 2021; Miller et al., 2020; Youm et al., 2015). A significant decrease of waist circumference was presented in individuals with overweight and obesity who participated in a combined exercise and low carbohydrate ketogenic diet intervention (Lee & Lee, 2021; Miller et al., 2020). This effect was observed also despite maintaining the participants daily intake of energy and levels of physical activity (Gram‐Kampmann et al., 2022). The possible explanation of a body mass reducing effect of such a carbohydrate restricted diet was suggested an increased total energy expenditure after approximately 2.5 week of lower-carbohydrate intake (Ludwig et al., 2020). However, a satiety effect of a carbohydrate restricted high fat diet, which may cause a total energy decrease, must be considered (Ludwig et al., 2023).
High-intensity interval training (HIIT) is a training alternative to the traditional moderate-intensity continuous endurance training. HIIT is defined as periods of intense activity separated by low-intensity breaks. HIIT can improve the fitness level to a greater extent than continuous exercise despite a reduced training volume (Milanović et al., 2015). Such a superior effect of HIIT has been shown also to improve metabolic health (Cassidy et al., 2017; Jelleyman et al., 2015), vascular function, cardiovascular risk factors (Grace et al., 2018; Sawyer et al., 2016), oxidative stress, inflammation, and insulin sensitivity (Ramos et al., 2015). However, similar responses to HIIT and moderate-intensity continuous exercise were presented for cardiometabolic adaptation (De Nardi et al., 2018; Gillen et al., 2016). When an acute effect of HIIT and moderate-intensity continuous exercise matched for strain were analyzed from the metabolomic perspective, a comparable metabolic and hormonal response was shown despite substantial differences in work-bout intensities (Moser et al., 2015; Zafeiridis et al., 2016).
Regular physical activity and dietary variety are modifiable and influential factors of health outcomes. However, the cumulative effects of these behaviors are not well understood. We aimed to investigate the effect of a 12 week very low carbohydrate high fat (VLCHF) diet and HIIT on lipidomic and metabolomic profiles in individuals with overweight and obesity. This work extends our earlier findings, which focused primarily on body composition, exercise performance and standard biochemical outcomes (Cipryan et al., 2021, 2022).
Methods
Parent study
It was a randomized, controlled, four-arm, parallel exercise and/or dietary intervention study (ClinicalTrials.gov: NCT03934476), with the primary aim of examining the VLCHF and HIIT effects on body composition and cardiorespiratory fitness (Cipryan et al., 2021). Data were collected in Ostrava, Czech Republic. There were 91 participants included into four study groups and they completed a 12 week experimental period. Participants were randomly allocated to one of the four study groups: (1) HIIT and habitual diet, (2) very low-carbohydrate, high-fat diet (VLCHF) and habitual physical activity (no regular exercise training), (3) VLCHF diet and HIIT and (4) control (habitual diet and physical activity, no regular exercise training). Dual-energy X-ray absorptiometry (DXA) and graded exercise tests to volitional exhaustion were used for body composition and cardiorespiratory fitness (CRF) assessments, respectively.
To obtain measures of no intervention, a control group was utilized. Participants in the control group were advised not to change their habitual diet and physical activity regime. Therefore, no diet advice was provided.
We utilized the infrastructure of this trial to conduct a pre-planned ancillary study focused on lipidomic and metabolomic analysis. We analysed blood samples following a 3 h fast before the experimental period (T0) and after 4, 8 and 12 weeks (T1, T2 and T3, respectively). The participant set used for the primary and secondary analyses was identical with the participant set presented in this metabolomics study (intention-to-treat analysis).
Participants
Participants were randomly allocated to four study groups: (1) HIIT and habitual diet, (2) very low-carbohydrate, high-fat diet (VLCHF) and habitual physical activity (no regular exercise training), (3) VLCHF diet and HIIT, (4) control (habitual diet and physical activity, no regular exercise training) (Cipryan et al., 2021, 2022). Inclusion criteria were age 20–59 years, non-smokers, overweight/obesity (BMI 25.00–40.00 kg/m2), no specific sports training or regular exercise (low physical activity), no excessive alcohol intake, willingness to accept random assignment, no evidence of liver, renal, metabolic and cardiopulmonary disease and diseases contraindicating physical activity, no cancer, no psychiatric illness, no pregnancy or breast-feeding, not on any specific diet, PAR-Q pass, body weight stable for the last 2 months and not on a weight loss plan, no hypoglycaemic, lipid lowering, antihypertensive or psychiatric medications as well as medications known to affect body weight or energy expenditure. Participants had no previous experience with the VLCHF diet and HIIT. All study participants provided written informed consent. The study design was approved by the local University Ethics Committee.
High-intensity interval training (HIIT)
Before the start of the intervention, the participants in both HIIT and VLCHF + HIIT groups received a detailed instruction on the HIIT program. Participants were told to complete 3 sessions per week. Two HIIT sessions were completed during weeks 4, 8 and 12 when the participants visited laboratory, one session was home based. Each HIIT session started and finished with 5 min of slow walking. HIIT was composed of series of 3 min of a high intensity walking (Borg´s scale RPE 18–19) followed by 3 min of low intensity walking (RPE 9–11). There were 4, 6 and 8 high intensity intervals set up for the first, second and third 4 week period, respectively. Therefore, the total session time increased from 31 to 43 min and finally 55 min during each 4 week period. Training intensity was monitored with a Polar M430 watch (Polar Electro, Oy, Finland). Training data were uploaded to Polar Flow and regularly analysed by an experienced researcher.
Dietary intervention
Participants in the HIIT only and the control group were asked to maintain their habitual dietary intake without restriction. The VLCHF diet was defined as allowing no more than 50 g of CHO per day (Feinman et al., 2015). The diet included no specific caloric goal. However, the participants in the VLCHF groups were advised to compensate for the total energy decrease caused by CHO intake restriction by increasing their natural non-trans fat intake (e.g., cream, butter and olive and coconut oil). The target for protein intake was recommended 1.5 g/kg lean body mass and, unlike the strict CHO restriction, participants were asked just to get as close as possible. The use of all sweetened and grain-based products was strongly limited. The recommended food included whole food sources such as meat, vegetables, non-sweetened products, full-fat dairy items, nuts and seeds. Detailed dietary advice was provided by a dietitian before and during the study (on request or at least once a month). A handbook was given to participants containing food lists, guidelines for estimating macronutrient amounts, and sample recipes. All foods and quantities consumed were recorded daily in all study groups beginning from seven days before the intervention period (www.kaloricketabulky.cz). Alcoholic beverages were restricted during the intervention period and dietary supplements were not permitted 1 month prior to and during the intervention, while caffeinated beverages were restricted only before the laboratory sessions.
Chemicals and materials
Methanol and 2-propanol were supplied by Merck (Darmstadt, Germany). Ammonium acetate, ammonium formate, formic acid and acetic acid were obtained from Sigma-Aldrich (Prague, Czech Republic). Click Fit 2 mL Eppendorf tubes were purchased from TreffLab (Degersheim, Germany); 2 mL cryovials and autosampler vials were purchased from Labicom (Olomouc, Czech Republic).
Sample preparation
The samples were prepared by mixing 50 μL of plasma with 150 μL MeOH. After vortexing, 500 μL methyl tert-butyl ether (MTBE) was added, and the mixture was shaken for 15 min. Subsequently, 150 μL of deionized water was added to form a two-phase system. After vortexing for 30 s, the mixture was deproteinized and centrifugated at 10,000 rpm (10,621×g) for 10 min at room temperature. The resultant supernatants were lyophilized and stored in an − 80 °C freezer if needed for later use. The freeze-dried lipid residues were resuspended in 2-propanol/methanol/deionized water (65:30:5, v/v/v) and used for subsequent analysis. For the metabolomic experiment 50 uL of plasma were mixed with 150 uL acetonitrile to precipitate proteins, the mixture was then centrifuged at 10,000 rpm and the supernatant collected in a new vial and evaporated to dryness on a SpeedVac (Labconco) instrument. Before analysis, the dried extract was resuspended in H2O:acetonitrile (1:1).
Instrumental conditions
Both lipidomic and metabolomic analysis used an Infinity 1290 (Agilent) UHPLC coupled to the 6560 Ion Mobility Q-TOF LC/MS (Agilent) with an Agilent Jet Stream (AJS) electrospray (ESI) source. The details of the lipidomic method are described elsewhere (10.3390/metabo12020124), briefly an Acquity BEH C18 column (1.7 μm, 2.1 mm × 150 mm; Waters, USA) was used for chromatographic separation. Lipids were separated by a gradient of mobile phases A: 10 mM ammonium formate and 0.1% formic acid in acetonitrile:water (60:40, v/v); mobile phase B was 10 mM ammonium formate and 0.1% formic acid in 2-propanol:acetonitrile (90:10, v/v) in ESI + mode, for ESI- mode formic acid and ammonium formate were replaced by acetic acid and ammonium acetate.
The details of metabolomic method are described in the supplement. Briefly, metabolites were separated on an Acquity HSS T3 column (1.7 μm, 2.1 mm × 100 mm; Waters, USA) using a gradient of mobile phases A: 5 mM ammonium formate and 0.1% formic acid in water:MeOH (95:5, v/v) and mobile phase B: 5 mM ammonium formate and 0.1% formic acid in MeOH in ESI + mode, for ESI- mode formic acid and ammonium formate were replaced by acetic acid and ammonium acetate. The mass spectrometer acquired data in Full MS and autoMS/MS modes in the m/z range of 50–1200.
Processing and statistical analysis of data generated by fingerprinting experiments
Regarding to the lipidomics data analysis, data were processed by the LipidMatch suite [26], which uses MZmine 2 for feature extraction and an R script for lipid identification. Lipids were identified based on fragmentation spectra and accurate mass in silico libraries, which are part of the LipidMatch suite. Fragmentation spectra of the significant compounds from metabolomics were also compared to those present in METLIN and LIPIDMAPS databases, and their identities were confirmed. In case of metabolomics data, the data were processed by Profinder software (Agilent Technologies, Santa Clara, CA, USA) and MZmine 2.
The data matrices obtained from lipidomics and metabolomics data processing were imported to SIMCA for multivariate analysis and Mass Profiler Professional (MPP, Agilent technologies for univariate analysis and metabolite profiling. Logarithmic transformation and pareto scaling were used to pre-process the data. Two-way ANOVA (corrected p-value cut-off: 0.05; p-value computation: Asymptotic; Multiple Testing Correction: Benjamini-Hochberg) analysis was applied to the data matrix to filter significant entities affected by diet group and sampling point factors.
Sample size estimation
An a priori power analysis using GPOWER (Faul et al., 2007) with power set at 0.80 and significance level set at 0.05 was calculated retrospectively. The power analysis indicated that a total sample of 76 people would be needed to detect large effects (f = 0.40) for this study with 4 groups. A total sample of 180 people would be needed to detect medium effects (f = 0.25) Thus, the sample size was sufficient to reveal that a large effect could not be interpreted as non-significant.
Results
Parent study
Results for the primary outcome have been previously reported, showing that the VLCHF diet, either in isolation or in combination with HIIT, caused a significant reduction in body mass, visceral adipose tissue (VAT) mass and body composition variables. HIIT alone did not induce such effects on body composition, but improved CRF (Cipryan et al., 2021). We followed with a secondary analysis, which focused on cardiometabolic risk factors. The VLCHF diet was effective for favorable changes in the homeostasis model assessment of insulin resistance (HOMA-IR), Adiponectin/Leptin ratio and diastolic blood pressure. HIIT, or HIIT combined with the VLCHF diet, had no additional benefits for the analysed variables (Cipryan et al., 2022). Of note, total energy intake significantly decreased in the HIIT, VLCHF and VLCHF + HIIT groups. Carbohydrate intake significantly decreased and fat intake increased in the VLCHF and VLCHF + HIIT groups. Protein intake did not significantly change in any of the study groups (Cipryan et al., 2021).
Body composition
VAT mass and body composition changes after 12 weeks significantly differed between study groups. VAT mass only decreased in the VLCHF and VLCHF + HIIT groups (p < 0.001, median [95% CI]: VLCHF: − 142.0 [− 187.0; − 109.5] g; VLCHF + HIIT: − 104.0 [− 135.0; − 71.0] g). Similarly, changes in body mass, total body fat, trunk fat mass, waist and hip circumferences were distinctly decreased in the VLCHF and VLCHF + HIIT groups, when compared to HIIT and Control groups. However, total lean mass significantly decreased in the VLCHF and VLCHF + HIIT groups (− 2.1 [− 3.0; − 1.6] kg and − 2.5 [− 3.6; − 1.8] kg, respectively), but not in HIIT and control groups, as presented in the parent study (Cipryan et al., 2021).
Serum lipidomic analysis
Multivariate model analysis
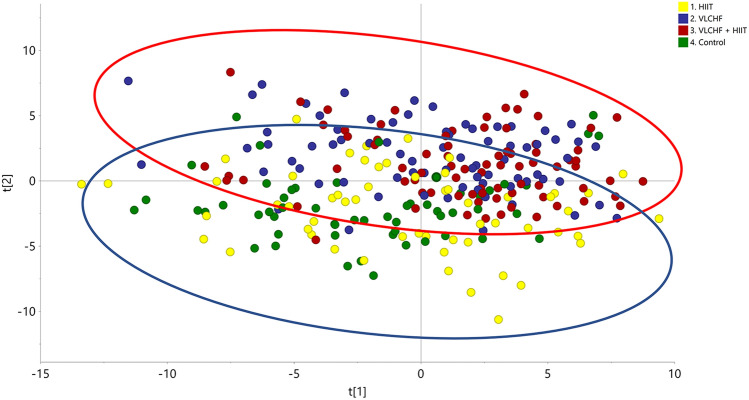
The pre-processing operations provided a data matrix based on 409 entities from the full data set. The data matrix was imported to SIMCA software for data processing including data log transformation and pareto scaling (van der Berg et al., 2006). A PCA model of the final data matrix was created by SIMCA software to describe the total variance of the full data set (Fig. 1). The calculated PCA model based on 267 samples described 84.9% of the variation in the data matrix (R2 X = 0.849) according to the cross-validation prediction of Q2 = 0.651. The PCA showed a global trend of the data variation describing a clear sample grouping affected by the VLCHF diet (VLCHF and VLCHF + HIIT groups; red circle, Fig. 1) and non-VLCHF diet (HIIT and Control groups; blue circle, Fig. 1). The results showed similar distributions of lipid compounds in both the VLCHF and VLCHF + HIIT groups and similar distributions of lipid compounds in both the HIIT only and the Control group.
Fig. 1.
PCA plot of diet groups intervention including samples after 4, 8, and 12 weeks (T1, T2, T3). Control, green label: control group; HIIT, yellow label: HIIT group; VLCHF, blue label: VLCHF diet group; VLCHF + HIIT, red label: VLCHF diet and HIIT group. Red ellipse: Samples grouped by VLCHF diet group; Blue ellipse: Samples grouped by non-VLCHF diet group
Lipids identification and kinetic
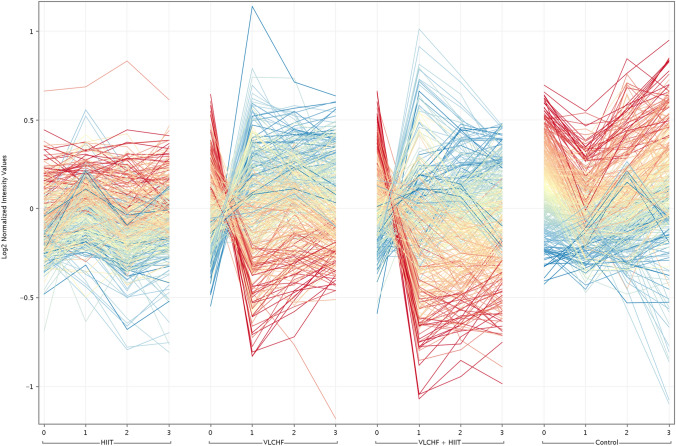
Two-way ANOVA (corrected p-value cut-off: 0.05) was applied to the data matrix in order to filter significant entities affected by diet groups and sampling point factors. The 146 entities were significant in both the VLCHF and VLCHF + HIIT groups (Fig. 2). The final list of entities was filtered using the correlation between the acquired values and parameter values (sampling point) in order to identify entities correlated with the increasing and decreasing trends along the study. This approach was carried out using Euclidean distance and Pearson correlation (correlation coefficient cut-off: 0.95).
Fig. 2.
Profile plot of significantly changed lipids. Colored by changes respect to diet group at T1, T2, T3. Red lines: Highly correlated with decreasing trend; Blue lines: Highly correlated with increasing trend; Yellow lines: low fold change according the trend
The identified lipids were classified in 10 different families including plasmalogens (plasmanyl and plasmenyl), fatty acyl esters of hydroxy fatty acid (FAHFA), phosphatidylcholines (PC), phosphatidylethanolamines (PE), phosphatidylinositols (PI), Lysophsphatidylcholines (LPC), Lysophosphatidyletanolamines (LPE), dimethylphosphatidylethanolamines (DMPE), triglycerides (TG), fatty acids (FA), acetyl-L-carnitines (AcCar), cholesterol esters (CE) sphingomyelins (SM), ceramides (CER) and sulfated glycosphingolipids (sulfatide). The differences in trends of each lipid class could be clearly evaluated along the intervention groups as the members of the class almost uniformly followed the same pattern. Only in a minority of cases, different trends were observed for individual lipids compared to the other members of the class. One such example was Plasmenyl PS(P-20:1/20:4), which was downregulated in both dietary intervention groups but the rest of the lipid class family exhibited upregulation. The AcCars, plasmalogens, FAHFAs, SMs, CER, CE, FAs and sulfatide were mostly identified as increased markers in plasma after both the dietary and the training intervention. TGs and glycerophospholipids (PC, PE, PI and DMPEs) were mostly decreased lipids in serum after the VLCHF and VLCHF + HIIT interventions. In addition, AcCar, SMs, CER, CE, FAs and sulfatide were exclusively identified as families increased in the VLCHF and VLCHF + HIIT groups. PE, PI, LPE, LPC and DMPEs were exclusively identified as family decreased during the diet intervention.
Seventy-eight entities out of 146 were significantly unique for the VLCHF and VLCHF + HIIT groups (Fig. 2). No lipid compounds specifically influenced by exercise were identified (HIIT and Control groups).
Thirty-nine lipids increased in the VLCHF and VLCHF + HIIT groups (Table 1). Plasmanyl-PC(O-16:0/20:5), Plasmanyl-PC(O-18:1/20:4) and SM(36:2) were the identified lipids with a highest increase in the VLCHF group after the 12 week intervention (T3). FAHFA(18:2/20:4), FAHFA(18:1/20:3) and SM(36:2) showed the highest variation after the first intervention month (T1) in the VLCHF group. Conversely, a total of 38 lipids with a decreasing trend were found in the VLCHF and VLCHF + HIIT groups and with stable trend in control and HIIT groups being principally triglycerides and glycerophospholipids (Table 2). FAHFA(14:0/22:3), PC(14:0/16:1) and PC(18:0/20:3) were the lipids with the greatest decrease identified in the VLCHF group after the 12 week intervention (T3). In addition to this, FAHFA(14:0/22:3) were the unique lipids that decreased during the entire 12 week study. PC(14:0/16:1), PC(14:0/18:2) and PC(18:0/20:3) showed the largest decrease after the first month of intervention (T1) in both VLCHF and VLCHT + HIIT groups.
Table 1.
Serum lipids identified as significantly UP regulated biomarkers in the VLCHF and VLCHF + HIIT groups
| ID | Compound | Family | Mass | Rt |
|---|---|---|---|---|
| 1 | AcCar(18:1) | Acetyl-L-carnitines* | 426.3582 | 2.74 |
| 2 | AcCar(18:0) | Acetyl-L-carnitines | 428.3743 | 3.72 |
| 3 | AcCar(16:0) | Acetyl-L-carnitines | 400.3425 | 2.63 |
| 4 | Plasmenyl-PE(P-16:0/20:4) | Plasmalogens | 722.5112 | 9.44 |
| 5 | Plasmenyl-PE(P-18:1/20:4) | Plasmalogens | 748.5268 | 9.48 |
| 6 | Plasmenyl-PE(P-18:0/20:4) | Plasmalogens | 750.5397 | 9.28 |
| 7 | Plasmanyl-PE(O-16:0/22:5) | Plasmalogens | 750.5407 | 9.67 |
| 8 | Plasmenyl-PE(P-18:0/20:4) | Plasmalogens | 750.5423 | 10.35 |
| 9 | Plasmanyl-PE(O-18:0/20:4) | Plasmalogens | 752.5572 | 10.54 |
| 10 | Plasmenyl-PE(P-18:0/20:3) | Plasmalogens | 752.5577 | 10.91 |
| 11 | Plasmenyl-PE(P-18:0/22:6) | Plasmalogens | 774.5422 | 10.02 |
| 12 | Plasmenyl-PE(P-18:1/22:5) | Plasmalogens | 774.5429 | 9.43 |
| 13 | Plasmenyl-PE(P-18:0/22:5) | Plasmalogens | 776.5592 | 10.31 |
| 14 | Plasmanyl-PE(O-18:0/22:5) | Plasmalogens | 778.5755 | 10.5 |
| 15 | Plasmenyl-PE(P-20:0/20:4) | Plasmalogens | 778.5747 | 11.16 |
| 16 | Plasmenyl-PE(P-20:0/22:6) | Plasmalogens | 802.5748 | 10.91 |
| 17 | Plasmanyl-PC(O-16:0/18:2) | Plasmalogens | 744.589 | 9.21 |
| 18 | Plasmanyl-PC(O-16:0/20:5) | Plasmalogens | 766.5751 | 8.91 |
| 19 | Plasmanyl-PC(O-16:0/20:4) | Plasmalogens | 768.5895 | 9.05 |
| 20 | Plasmenyl-PC(P-16:0/22:6) | Plasmalogens | 790.573 | 8.65 |
| 21 | Plasmenyl-PC(P-18:1/20:4) | Plasmalogens | 792.5887 | 8.93 |
| 22 | Plasmanyl-PC(O-18:1/20:4) | Plasmalogens | 794.6042 | 9.64 |
| 23 | Plasmanyl-PC(O-20:1/18:3) | Plasmalogens | 796.6189 | 9.57 |
| 24 | Plasmanyl-PC(O-18:0/20:4) | Plasmalogens | 796.6202 | 9.77 |
| 25 | Plasmanyl-PC(O-18:1/22:6) | Plasmalogens | 818.6037 | 8.81 |
| 26 | Plasmanyl-PC(O-20:1/20:4) | Plasmalogens | 822.6356 | 9.74 |
| 27 | FAHFA(18:2/20:4) | Fatty acyl esters of hydroxy fatty acid | 581.4536 | 3.94 |
| 28 | FAHFA(18:1/20:3) | Fatty acyl esters of hydroxy fatty acid | 585.4851 | 5.06 |
| 29 | FAHFA(18:1/22:3) | Fatty acyl esters of hydroxy fatty acid | 613.5216 | 5.06 |
| 30 | SM(d17:1/16:0) | Sphingomyelin* | 689.5589 | 8.15 |
| 31 | SM(d18:0/16:0) | Sphingomyelin | 705.5893 | 8.87 |
| 32 | SM(d17:1/18:0) | Sphingomyelin | 717.5896 | 8.98 |
| 33 | SM(36:2) | Sphingomyelin | 787.5947 | 8.88 |
| 34 | SM(d18:1/18:0) | Sphingomyelin | 731.6059 | 9.38 |
| 35 | SM(36:0) | Sphingomyelin | 791.6243 | 10.10 |
| 36 | HexCer-NS(d-18:1/23:0) | Ceramides* | 796.6645 | 12.14 |
| 37 | CE(20:4) | Cholesterol ester* | 695.5738 | 13.52 |
| 38 | FA(18:1) | Fatty acids* | 281.2488 | 5.06 |
| 39 | Sulfatide(39:5) | Sulfatide | 840.5304 | 8.54 |
*Family exclusively identified in the group of lipids that increase
Table 2.
Serum lipids identified as significantly DOWN regulated biomarkers in the VLCHF and VLCHF + HIIT groups
| ID | Compound Name | Family | Mass | Rt |
|---|---|---|---|---|
| 1 | Plasmenyl-PS(P-20:1/20:4) | Plasmalogens | 820.546 | 9.19 |
| 2 | FAHFA(14:0/22:3) | Fatty acyl esters of hydroxy fatty acid | 559.4694 | 8.49 |
| 3 | TG(12:0/16:0/18:1) | Triglycerides | 794.7225 | 13.27 |
| 4 | TG(12:0/18:2/18:2) | Triglycerides | 816.7069 | 12.49 |
| 5 | TG(14:1/16:0/18:3) | Triglycerides | 816.707 | 12.60 |
| 6 | TG(16:1/16:1/16:1) | Triglycerides | 823.6784 | 12.87 |
| 7 | TG(14:0/16:1/18:1) | Triglycerides | 820.7392 | 13.27 |
| 8 | TG(14:0/16:0/18:1) | Triglycerides | 822.7542 | 13.65 |
| 9 | TG(14:0/18:2/18:3) | Triglycerides | 842.7226 | 12.62 |
| 10 | TG(14:0/18:2/18:2) | Triglycerides | 849.6939 | 13.09 |
| 11 | TG(16:0/16:0/18:4) | Triglycerides | 849.694 | 12.91 |
| 12 | TG(16:0/16:1/18:3) | Triglycerides | 844.7387 | 12.92 |
| 13 | TG(14:0/16:0/20:4) | Triglycerides | 844.7389 | 13.13 |
| 14 | TG(14:0/18:1/18:2) | Triglycerides | 846.7545 | 13.28 |
| 15 | TG(16:0/16:0/18:3) | Triglycerides | 851.7099 | 13.26 |
| 16 | TG(16:1/18:2/18:3) | Triglycerides | 868.7381 | 12.74 |
| 17 | TG(16:0/18:3/18:3) | Triglycerides | 873.6943 | 12.73 |
| 18 | TG(16:0/18:2/18:3) | Triglycerides | 870.7541 | 12.97 |
| 19 | PC(28:0) | Glycerophospholipids | 700.4884 | 7.73 |
| 20 | PC(14:0/16:1) | Glycerophospholipids | 762.5306 | 7.91 |
| 21 | PC(14:0/16:0) | Glycerophospholipids | 706.5383 | 8.53 |
| 22 | PC(14:0/18:2) | Glycerophospholipids | 730.538 | 7.95 |
| 23 | PC(16:0/16:1) | Glycerophospholipids | 732.553 | 8.61 |
| 24 | PC(14:0/20:4) | Glycerophospholipids | 754.5367 | 7.79 |
| 25 | PC(34:3) | Glycerophospholipids | 778.536 | 8.31 |
| 26 | PC(17:1/18:2) | Glycerophospholipids | 770.5686 | 8.59 |
| 27 | PC(14:0/22:6) | Glycerophospholipids | 836.5411 | 7.62 |
| 28 | PC(18:2/18:3) | Glycerophospholipids | 780.5529 | 7.58 |
| 29 | PC(16:0/22:4) | Glycerophospholipids | 810.5981 | 8.98 |
| 30 | PC(18:0/20:3) | Glycerophospholipids | 812.6163 | 9.64 |
| 31 | PC(18:0/22:5) | Glycerophospholipids | 836.6115 | 9.61 |
| 32 | PE(18:0/20:3) | Glycerophospholipids | 768.5526 | 10.31 |
| 33 | LPC(14:0)* | Glycerophospholipids | 468.3088 | 2.06 |
| 34 | LPC(16:1) | Glycerophospholipids | 494.3241 | 2.16 |
| 35 | LPC(20:3) | Glycerophospholipids | 546.3549 | 2.50 |
| 36 | DMPE(16:0/16:1) | Glycerophospholipids | 716.5225 | 8.83 |
| 37 | DMPE(16:0/20:3) | Glycerophospholipids | 768.5523 | 9.20 |
| 38 | DMPE(18:0/20:3) | Glycerophospholipids | 796.5873 | 10.13 |
*PE, LPC and DMPE were exclusively identified in the group of lipids that decrease
Serum metabolomic analysis
Multivariate model analysis
The multivariate model analysis procedures were performed under the same criteria as lipidomics. The pre-processing operations gave in case of metabolomics analysis a data matrix based on 5618 entities from full data set.
These results showed similar intervention group distributions as lipidomics. The metabolites in plasma samples showed a clear sample grouping, i.e. (VLCHF and VLCHF + HIIT groups) vs. (HIIT and Control groups).
Metabolites identification and kinetic
Two-way ANOVA (corrected p-value cut-off: 0.05; p-value computation: Asymptotic; Multiple Testing Correction: Benjamini-Hochberg) statistics analysis was applied to the data matrix in a similar manner as in lipidomics. A total of 164 of these metabolites satisfying the filtration criteria for up and down regulation were phospholipids already identified and described in the previous lipidomics analysis. A total of 8 entities were exclusively identified in the metabolomics analysis and these entities significantly increased in the VLCHF and VLCHF + HIIT groups after 12 weeks (T3).
The metabolites identified tentatively were classified in 4 families including glycerophospholipids, xanthine, acyl-carnitine and alpha hydroxy acids. The glycerophospholipids and xanthine were identified as metabolites that decrease in the VLCHF and VLCHF + HIIT groups. The acyl-carnitines and alpha hydroxyl acids were identified as metabolites that increased in the VLCHF group during the 12 week study (T3).
Regarding to the individual kinetics of the metabolites identified, a total of 4 metabolites were found with an increasing trend in the VLCHF and VLCHF + HIIT groups along the 12 week study and without a defined trend in the HIIT and the Control group (Table 3). Decanoylcarnitine, 2-Decenoylcarnitine, Octanoylcarnitine and 4-Hydroxybutyric acid were found according with an increased trend in both VLCHF and VLCHF + HIIT groups. In the opposite, a total of 4 metabolites including three isomers of PE-NMe (18:1/18:3) and theobromine were found according with a decreased trend in the VLCHF and VLCHF + HIIT groups. Acyl-carnitines, hydroxybutiric acid and xanthine, tentatively identified by untargeted metabolomics, were confirmed by MS/MS spectra fragmentation identification therefore increase their identification accuracy to level 2 (Table 3) (Sumner et al., 2007).
Table 3.
Significantly changed plasma metabolites in the VLCHF and VLCHF + HIIT groups identified by untargeted metabolomics
| ID | Compound name | Regulation | MS/MS fragment | Mass | Rt |
|---|---|---|---|---|---|
| 1 | Decanoylcarnitine1 | UP | 316.2465,257.1732,155.1413,144.1031 | 315.2405 | 13.50 |
| 2 | 2-Decenoylcarnitine1 | UP | 314.231,85.0279,255.1586,297.1568* | 313.2249 | 12.66 |
| 3 | Octanoylcarnitine1 | UP | 288.2163,229.1436,85.0286,127.1131* | 287.209 | 11.23 |
| 4 | 4-Hydroxybutyric1 | UP | 103.0398, 57.0332 | 104.0468 | 1.12 |
| 5 | PE-NMe(18:1/18:3)2 | DOWN | – | 753.5298 | 20.01 |
| 6 | PE-NMe(18:1/18:3)2 | DOWN | – | 753.5309 | 20.01 |
| 7 | PE-NMe(18:1/18:3)2 | DOWN | – | 753.5299 | 20.01 |
| 8 | Theobromine1 | DOWN | 181.0714,138.0656,110.0712,163.0630 | 180.0636 | 4.42 |
Discussion
This is the first randomized control trial which investigated the effects of a 12 week VLCHF diet and HIIT interventions, as well as their combination, on changes of plasma lipidomic and metabolomic profiles in individuals with overweight and obesity. We revealed that the VLCHF diet, alone or combined with HIIT, caused a substantial increase in serum plasmalogens, FAHFAs, FAs, AcCar, CE, SMs, CER, and sulfatides. Glycerophospholipids and TGs substantially decreased in both the VLCHF and VLCHF + HIIT groups. The highest lipid changes occurred already after 4 weeks and then their plasma levels remained stable or showed a countermovement trend until the end of the intervention after 12 weeks (Fig. 2). Similar to lipidomic, the metabolomic analysis revealed differences between the groups including the diet (i.e. VLCHF and VLCHF + HIIT) versus the HIIT and Control group. Four metabolites were significantly upregulated and four were downregulated in the VLCHF and VLCHF + HIIT groups. HIIT alone had no substantial changing effect on lipidomic and metabolomic markers investigated although data were more stable than compared to the control group which showed contrary trends to both diet groups. This may indicate a stabilizing effect of exercise on lipid and metabolic markers.
Plasmalogens
We revealed a significant increase in plasmalogens (Table 1). Plasmalogens are a class of glycerophospholipids, reflect the functional activity of peroxisomes and have been reduced (or deficient) in chronic diseases related to oxidative stress, chronic inflammation and aging, such as Parkinson disease, Alzheimer disease, metabolic syndrome and type 2 diabetes mellitus (Bozelli et al., 2021; Braverman & Moser, 2012; Kytikova et al., 2020) and their levels are altered also in various types of cancers (Messias et al., 2018). Despite the biological mechanism of action for plasmalogens remains unclear, it seems that plasmalogens play a crucial role as endogenous antioxidants, protecting other phospholipids, lipids and lipoprotein particles from oxidative stress, as well as participate in PUFA cell metabolism and play an important role as neuroprotectors and modulators of the signalling mechanisms realized in cell membrane (Hossain et al., 2016; Kytikova et al., 2020).
It has been shown that a plasmalogen replacement therapy can be a successful anti-inflammatory strategy, which ameliorates several pathological hallmarks of neurodegenerative diseases, atherosclerosis, insulin resistance and hepatic steatosis (Bozelli et al., 2021; Paul et al., 2019), alleviates negative mood states and sleep problems and enhance mental concentration (Fujino et al., 2022). Some plasmalogens subtypes levels (ethanolamine) are decreased in the Alzheimer´s disease patients and this lower level correlates with cognition deficit and severity of the disease (Su et al., 2019). Therefore, a therapeutic modulation of plasmalogens can be a promising tool in prevention and treatment of several chronic diseases. For example, supplementation with alkylglycerol, e.g. abundant in shark liver oil, bypass the peroxisome because alkylglycerol can be incorporated directly into the phospholipid pathway, which increases plasmalogen biosynthesis, similarly as a consumption other marine foodstuff (particularly blue mussel and ascidian). This plasmalogen enrichment may provide protection against obesity-related dyslipidaemia and inflammation (Paul et al., 2019, 2021). We showed for the first time that plasmalogens can be increased by a VLCHF diet, which can serve as a potential mechanistic co-explanation of some beneficial effects of such a VLCHF diet on chronic diseases (Silverii et al., 2022).
FAHFAS
Fatty acid esters of hydroxyl-fatty acids (FAHFAs) are another extensive group of lipids, which increased after the 12 week VLCHF diet. Since Yore et al. (Yore et al., 2014) reported in 2014 the discovery of the FAHFAs, there is still very few human data available and their biological effects are not well understood. FAHFAs have been found in different foods. Animal products contain more FAHFAs compared to plant products (Yore et al., 2014) and it seems that nutrition has a predominant impact on FAHFA level in serum (Kellerer et al., 2021). As the VLCHF diet is typically abundant in animal food sources, it is not surprising that we have found a FAHFA serum increase.
FAHFAs can also be incorporated in triacylglycerols (TG) as one of the three fatty acids. FAHFA-TGs are major reservoir of FAHFAs in cells and tissues (Tan et al., 2019) and, therefore, the nutrition impact on the FAHFA level in serum may not be exclusive. Also, the FAHFA levels are increased when lipolysis is induced (Tan et al., 2019) which is the elementary underpinning of the VLCHF diet (i.e. shifting from carbohydrate to predominant lipid metabolism).
There is mounting evidence that type 2 diabetes can be reversed through carbohydrate restricted diets (Goldenberg et al., 2021; Gram‐Kampmann et al., 2022; Joseph et al., 2022; Skow & Jha, 2022; Volek et al., 2021), however, the exact mechanism on a cellular level is not clear. Since FAHFAs show anti-inflammatory and anti-diabetic effects (Kellerer et al., 2021; Riecan et al., 2022; Tan et al., 2019; Yore et al., 2014), our finding of increased FAHFA levels after a 12 week VLCHF diet may contribute to this explanation.
Acetyl-L-Carnitines
Another upregulated lipid group after the 12 week VLCHF diet was Acetyl-L-Carnitines (ALC). ALC is a natural derivate of carnitine and can be found in various mammalian tissues, particularly in liver and skeletal muscles. Carnitine and its derivates play an essential role in the intermediary metabolism of glucose and β-oxidation of long-chain fatty acids. As a widely diffused dietary supplement, ALC may have a potential for the prevention of the oxidative stress (Lopez-Maldonado et al., 2021).
It has been shown in animal models that ALC inhibits the development of atherosclerosis by regulating blood lipids and inhibiting the gene expression of inflammatory factors and oxidative stress (Wang et al., 2020). This ALC anti-inflammatory and anti-angiogenic properties probably cause also the capability of ALC to down-modulate growth, adhesion, migration and invasion of prostate cancer cells (Baci et al., 2019). Bene et al. reviewed that ALC improves neurophysiological parameters, reduces pain and vascular-related symptoms in diabetic patients (Bene et al., 2018). ALC crosses the blood–brain barrier and improves neuronal energetic and repair mechanisms. It is therefore of great interest for its wide clinical application in various neurological disorders (e.g. Alzheimer’s dementia or depression) as well as HIV infection, chronic fatigue syndrome, peripheral neuropathies, ischemia and reperfusion of brain (Malaguarnera, 2012). Animal models showed that ALC supplementation ameliorates cognitive disabilities and combats oxidative stress-induced neuroinflammation (Verma et al., 2021). Beside such promising effects, the potential use of ALC supplementation in the pharmacotherapy of Alzheimer disease is still under debate (Pennisi et al., 2020). In contrast to these possible benefits, there are also some indices that L-carnitine-related metabolites can increase the risk for cardio-metabolic disease (Bene et al., 2018).
Oleic acid
We showed substantial increase of serum oleic acid (OA) (18:1)) which is a monounsaturated fatty acid and major fatty acid in dietary fat and in plasma triglycerides. OA-rich diet is associated with improved body mass and body composition. Particularly, abdominal fat and central obesity can be reduced following consumption of high-OA-containing meals (Tutunchi et al., 2020). OA is the most present fatty acid in olive oil, where it accounts for 58.5–83.2% of all methyl esters (Boskou et al., 2006). Olive oil is an important part of the Mediterranean diet, which is considered a healthy dietary pattern (US Department of Agriculture, 2020). Since we recommended to increase the fat intake including olive oil within the VLCHF diet in this study, it might be an explanation of the increased OA concentration found in serum of the diet group subjects. However, an alternative explanation can be based just on an increased lipolysis in adipose tissue and releasing OA into the blood, since the OA is the most abundant fatty acid in adipose tissue (Berry et al., 1986; Hellmuth et al., 2013).
The consumption of olive oil has been associated with several health benefits. However, it is unclear whether these beneficial effects are related to its polyphenol contents, OA or to the combination of them. It seems that consumption of olive oil or OA is as good as other strategies for Metabolic Syndrome management (Pastor et al., 2021). OA has also a potential role in controlling insulin resistance and type 2 diabetes mellitus, improving mitochondrial function, endothelial function, β-cell function, glucolipotoxicity, inflammation, oxidative stress, apoptosis, etc. (Rehman et al., 2020). However, studies of OA have reported contradictory results. For example, it has been reviewed that OA supplementation causes a significant reduction in blood concentrations of CRP in individuals with higher baseline level but has no significant effect on other inflammatory markers (Wang et al., 2022).
Sphingomyelin and ceramide
Sphingomyelin and ceramide were other upregulated lipids in the VLCHF and VLCHF + HIIT groups. Sphingolipids are a large class of lipids with diverse functions in many (patho-) physiological processes. Sphingomyelin (SM) is the most abundant sphingolipid in the cell. It is one of the major components of the plasma membrane. The hydrolysis of SM increases the concentration of ceramide (Cer), a bioactive molecule, which is involved in cellular proliferation, growth and apoptosis. SM is essential for brain development and cognitive abilities and it is altered in neurodegeneration. Disturbances of enzymes involved in the SM cycle (i.e. ceramide/SM balance system) were observed in many CNS pathologies, including ischemia, Alzheimer disease, Parkinson disease, depression or schizophrenia (Bienias et al., 2016; Signorelli et al., 2021). It appears that increasing ceramide/SM ratio is important in the suppression of tumour growth (Taniguchi & Okazaki, 2021). Nevertheless, the specific role of the diverse SM species in plasma membrane are not well understood. It has been shown that an increased serum SM is associated with renal and coronary heart disease in type 1 diabetes (Pongrac Barlovic et al., 2020) and palmitoyl SM is significantly associated with cardiovascular risk in a Chinese population with type 2 diabetes (Chen et al., 2022). Conversely, an inverse association between SM (32:1) and incident ischemic stroke independent of established cardiovascular risk factors was identified in Swedish cohorts (Lind et al., 2020). SM from food, particularly milk and dairy products, are increasingly recognized as bioactive lipids but there is a lack of clinical trials data about role of SM intake (Jiang et al., 2022).
Cer is a central molecule among the various sphingolipids because it is involved in synthesizing complex structural sphingolipids (sphingomyelin and ganglioside) and as a precursor of sphingosine-1-phosphate (S1P). Cer has been recognized a cause of selective insulin resistance and dyslipidaemia. Increased serum Cer levels are strongly associated with adverse CV risks and events. Therefore, decreasing ceramide levels through pharmacology and gene modification of the enzymes involved in the synthesis and degradation of ceramide can be a potential strategy to combat CVDs (Shu et al., 2022; Tippetts et al., 2021). Plasma Cers are elevated in humans with early coronary atherosclerosis and coronary endothelial dysfunction (Akhiyat et al., 2022). De novo synthesis pathway of Cers in hepatocytes has been shown to exacerbate non-alcoholic fatty liver disease (Yu & Wang, 2022).
The increased serum Cer level in our study might be related to the marked increase in fat intake in the VLCHF diet groups. When FA availability exceeds the cell’s storage and energetic capacities, Cers and other intermediates in these biosynthetic pathways start to accrue. Further, Cers initiate actions to enable storage or utilization of FAs to help the cell adapt to the surplus of FAs and protect membrane bilayers (Tippetts et al., 2021). Recent evidence has shown that lipids, particularly saturated FA (SFA), can interfere with de novo Cer synthesis and hypothalamic regulatory disturbance. However, this usually happens in response to excessive consumption of SFA associated with dietary simple sugars. While Cers are associated with hypothalamic dysfunction in energy balance and may play a role in obesity development, S1P seems to be a central satiety factor in the hypothalamus (Reginato et al., 2021), which can contribute to an explanation of a typical higher satiety in the VLCHF diet.
We showed also a significant increase of sulphated glycosphingolipids (GSLs). However, there is still limited knowledge about their biological function. The sulphated GSLs play an important role in cell–cell communication (Honke et al., 2004) and it seems that they enhance apoptotic cell clearance and modulate macrophage activity within tumours (Popovic et al., 2007).
Metabolomic outcomes
Decanoylcarnitine and octanoylcarnitine are members of the class of acylcarnitines with a general role to transport acyl-groups from the cytoplasm into the mitochondria for β-oxidation. Increased or decreased levels of these medium-chain fatty acids are associated with many diseases but without clear mechanistic explanations (Dambrova et al., 2022). However, we attribute the presented decanoylcarnitine and octanoylcarnitine, as well as short-chain fatty acid 4-hydroxybutyric, upregulations to increased fatty acids metabolism in the VLCHF and VLCHF + HIIT groups.
The downregulation of glycerophospholipid PE-NMe (18:1/18:3) (Table 3) and other triglycerides and glycerophospholipids within the lipidomic analysis (Table 2) is an expected response which may occur in carbohydrate intake restriction (Gjuladin-Hellon et al., 2019). This effect is explained by increased dependence on fat as a metabolic substrate which causes its increased peripheral uptake (Norwitz et al., 2022).
We showed a decrease of theobromine, a methylxanthine alkaloid derivate which is highly presented in cocoa in the VLCHF and VLCHF + HIIT groups. An animal study shows a theobromine beneficial role in body mass gain and effects on lipid and glucose metabolism (Camps-Bossacoma et al., 2019). There is an isolated evidence that theobromine may improve non-alcoholic fatty liver disease by inhibiting lipogenesis and fatty acid uptake and promoting fatty acid oxidation in the liver and hepatocytes (Wei et al., 2021). However, the theobromine role in human metabolism remains vague and do not allow to make a strong evidence-based conclusion about the presented outcomes.
Strength and limitations
We consider the main strength of this study showing another research perspective for the VLCHF and HIIT issue which can contribute to a better understanding the effects of such a diet with and without exercise. The lipidomic and metabolomic results complement the performance, body composition and biochemical data which have been already presented elsewhere (Cipryan et al., 2021, 2022). Plasma lipidomic profiles may contribute to a better prediction of cardiovascular (CV) risk and events. Sphingolipids, phospholipids (including lyso- and ether- species), cholesteryl esters and glycerolipids were associated with future CV events and CV deaths in elderly with type 2 diabetes mellitus and more than one additional CV risk factors (Alshehry et al., 2016).
The limitations of the study design have been discussed previously (Cipryan et al., 2021, 2022). Specifically to these results, plasmalogen levels change as a function of human age (Bozelli et al., 2021). We have to consider this fact since the participant’s age was substantially heterogenous in all four study groups. Additionally, the long-term dietary intervention studies are likely to cause modulation of the gut microbiota. The modulations of the microbiota would modify the final pool of secondary bile acids by altering for example the bacterial Bile salt Hydrolase (BSH) enzyme activity and producing an alteration of the presence of total free bile acids. This modification might affect the interaction with the specific intestinal receptors as Farnesoid X receptor (FXR) and affect to the regulation of the lipids metabolism (Garcia et al., 2022; Lei et al., 2022; Porez et al., 2012). Also, we need to consider a possible interaction of the presented lipidomic and metabolomic outcomes with body composition changes after 12 weeks.
We have demonstrated the metabolomic and lipidomic differences between the VLCHF and VLCHF + HIIT groups versus HIIT and control groups along with a significant total energy decrease in all intervention groups (except the control group) (Cipryan et al., 2021). Therefore, we presume that the presented changes are not the total energy decrease related.
Conclusions
In addition to a significant loss of body fat and body mass, the VLCHF diet affected plasma lipids considerably while the effect of HIIT was insignificant, although HIIT alone obviously stabilized the profiles compared to all other groups including controls. We showed substantial changes of plasma lipids already after 4 weeks of intervention in the participants in the VLCHF and VLCHF-HIIT groups which might correspond to the enhancement of metabolic flexibility to use lipid compounds as a substrate for energy metabolism. These changes persisted throughout the entire 12 week of both VLCHF diet interventions. We did not observe any synergistic effect of the VLCHF diet and HIIT on lipidomic and metabolomic profiles. Specifically, acyl carnitines, plasmalogens, fatty acyl esters of hydroxy fatty acid, sphingomyelin, ceramides, cholesterol esters, fatty acids, and 4-hydroxybutyric were identified as lipid families that increased in the VLCHF diet groups whereas lipid families of triglycerides and glycerophospholipids decreased. Additionally, metabolomic analysis showed a decrease of theobromine.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
We are grateful to all participants in the trial.
Author contributions
LC designed the study; collected, interpreted the data; and drafted, revised, and submitted the manuscript. TD collected, drafted and revised the manuscript. VK, CJG, KB, JH analyzed and interpreted the data; drafted and revised the manuscript, PH designed the study, interpreted the data, and drafted and revised the manuscript. All authors approved the final version of the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Funding
Open access publishing supported by the National Technical Library in Prague. This work was supported by the Czech Science Foundation (Grant 18-08358S). The laboratory facilities used for this research were bought with the support of the Healthy Aging in Industrial Environment project (registration number CZ.02.1.01/0.0/0.0/16_019/0000798, founded by the European Union via the Ministry of Education, Youth and Sports of the Czech Republic).
Data availability
Data described in the manuscript, code book, and analytic code will be made available upon request pending approval by authors.
Declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article. The authors declare they have no other financial interests
Ethical approval
The study design was approved by the local University Ethics Committee in advance (nr. /2018) and is in accordance with the principles of the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Agostoni C, Boccia S, Banni S, Mannucci PM, Astrup A. Sustainable and personalized nutrition: From earth health to public health. European Journal of Internal Medicine. 2021;86:12–16. doi: 10.1016/j.ejim.2021.02.012. [DOI] [PubMed] [Google Scholar]
- Akhiyat N, Vasile V, Ahmad A, Sara JD, Nardi V, Lerman LO, Allan J, Amir L. Plasma ceramide levels are elevated in patients with early coronary atherosclerosis and endothelial dysfunction. Journal of the American Heart Association. 2022 doi: 10.1161/JAHA.121.022852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alshehry ZH, Mundra PA, Barlow CK, Mellett NA, Wong G, McConville MJ, Simes J, Tonkin AM, Sullivan DR, Barnes EH, Nestel PJ, Kingwell BA, Marre M, Neal B, Poulter NR, Rodgers A, Williams B, Zoungas S, Meikle PJ. Plasma lipidomic profiles improve on traditional risk factors for the prediction of cardiovascular events in type 2 diabetes mellitus. Circulation. 2016;134(21):1637–1650. doi: 10.1161/CIRCULATIONAHA.116.023233. [DOI] [PubMed] [Google Scholar]
- Baci D, Bruno A, Cascini C, Gallazzi M, Mortara L, Sessa F, Pelosi G, Albini A, Noonan DM. Acetyl-L-carnitine downregulates invasion (CXCR4/CXCL12, MMP-9) and angiogenesis (VEGF, CXCL8) pathways in prostate cancer cells: Rationale for prevention and interception strategies. Journal of Experimental & Clinical Cancer Research. 2019;38(1):464. doi: 10.1186/s13046-019-1461-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bene J, Hadzsiev K, Melegh B. Role of carnitine and its derivatives in the development and management of type 2 diabetes. Nutrition & Diabetes. 2018;8(1):8. doi: 10.1038/s41387-018-0017-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berry EM, Zimmerman J, Peser M, Ligumsky M. Dietary fat, adipose tissue composition, and the development of carcinoma of the colon. Journal of the National Cancer Institute. 1986;77(1):93–97. doi: 10.1093/jnci/77.1.93. [DOI] [PubMed] [Google Scholar]
- Bienias K, Fiedorowicz A, Sadowska A, Prokopiuk S, Car H. Regulation of sphingomyelin metabolism. Pharmacological Reports. 2016;68(3):570–581. doi: 10.1016/j.pharep.2015.12.008. [DOI] [PubMed] [Google Scholar]
- Boskou D, Blekas G, Tsimidou M. 4—olive oil composition. In: Boskou D, editor. Olive oil. 2. AOCS Press; 2006. pp. 41–72. [Google Scholar]
- Bozelli JC, Azher S, Epand RM. Plasmalogens and chronic inflammatory diseases. Frontiers in Physiology. 2021;12:1–19. doi: 10.3389/fphys.2021.730829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braverman NE, Moser AB. Functions of plasmalogen lipids in health and disease. Biochimica Et Biophysica Acta (BBA)-Molecular Basis of Disease. 2012;1822(9):1442–1452. doi: 10.1016/j.bbadis.2012.05.008. [DOI] [PubMed] [Google Scholar]
- Camps-Bossacoma M, Garcia-Aloy M, Saldaña-Ruiz S, Cambras T, González-Domínguez R, Franch À, Pérez-Cano FJ, Andres-Lacueva C, Castell M. Role of Theobromine in Cocoa’s metabolic properties in healthy rats. Journal of Agricultural and Food Chemistry. 2019;67(13):3605–3614. doi: 10.1021/acs.jafc.8b07248. [DOI] [PubMed] [Google Scholar]
- Cassidy S, Thoma C, Houghton D, Trenell MI. High-intensity interval training: A review of its impact on glucose control and cardiometabolic health. Diabetologia. 2017;60(1):7–23. doi: 10.1007/s00125-016-4106-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y, Jia H, Qian X, Wang J, Yu M, Gong Q, An Y, Li H, Li S, Shi N, Zou Z, Li G. Circulating palmitoyl sphingomyelin is associated with cardiovascular disease in individuals with type 2 diabetes: findings from the China Da Qing diabetes study. Diabetes Care. 2022;45(3):666–673. doi: 10.2337/dc21-1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cipryan L, Dostal T, Litschmannova M, Hofmann P, Maffetone PB, Laursen PB. Effects of a very low-carbohydrate high-fat diet and high-intensity interval training on visceral fat deposition and cardiorespiratory fitness in overfat individuals: A randomized controlled clinical trial. Frontiers in Nutrition. 2021;8:1–13. doi: 10.3389/fnut.2021.785694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cipryan L, Litschmannova M, Maffetone PB, Plews DJ, Dostal T, Hofmann P, Laursen PB. Very low-carbohydrate high-fat diet improves risk markers for cardiometabolic health more than exercise in men and women with overfat constitution: Secondary analysis of a randomized controlled clinical trial. Frontiers in Nutrition. 2022;9:1–13. doi: 10.3389/fnut.2022.867690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dambrova M, Makrecka-Kuka M, Kuka J, Vilskersts R, Nordberg D, Attwood MM, Smesny S, Sen ZD, Guo AC, Oler E, Tian S, Zheng J, Wishart DS, Liepinsh E, Schiöth HB. Acylcarnitines: Nomenclature, biomarkers, therapeutic potential, drug targets, and clinical trials. Pharmacological Reviews. 2022;74(3):506–551. doi: 10.1124/pharmrev.121.000408. [DOI] [PubMed] [Google Scholar]
- De Nardi AT, Tolves T, Lenzi TL, Signori LU, da Silva AMV. High-intensity interval training versus continuous training on physiological and metabolic variables in prediabetes and type 2 diabetes: A meta-analysis. Diabetes Research and Clinical Practice. 2018;137:149–159. doi: 10.1016/j.diabres.2017.12.017. [DOI] [PubMed] [Google Scholar]
- Ehrman JK, Gordon PM, Visich PS, Keteyian SJ, editors. Clinical exercise physiology. Champaign: Human Kinetics; 2019. [Google Scholar]
- Faul F, Erdfelder E, Lang AG, Buchner A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behavior Research Methods. 2007;39(2):175–191. doi: 10.3758/BF03193146. [DOI] [PubMed] [Google Scholar]
- Feinman RD, Pogozelski WK, Astrup A, Bernstein RK, Fine EJ, Westman EC, Accurso A, Frassetto L, Gower BA, McFarlane SI, Nielsen JV, Krarup T, Saslow L, Roth KS, Vernon MC, Volek JS, Wilshire GB, Dahlqvist A, Worm N. Dietary carbohydrate restriction as the first approach in diabetes management: Critical review and evidence base. Nutrition. 2015;31(1):1–13. doi: 10.1016/j.nut.2014.06.011. [DOI] [PubMed] [Google Scholar]
- Fujino M, Fukuda J, Isogai H, Ogaki T, Mawatari S, Takaki A, Wakana C, Fujino T. Orally administered plasmalogens alleviate negative mood states and enhance mental concentration: A randomized, double-blind, placebo-controlled trial. Frontiers in Cell and Developmental Biology. 2022;10:1–11. doi: 10.3389/fcell.2022.894734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia CJ, Kosek V, Beltrán D, Tomás-Barberán FA, Hajslova J. Production of new microbially conjugated bile acids by human gut microbiota. Biomolecules. 2022;12(5):687. doi: 10.3390/biom12050687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillen JB, Martin BJ, MacInnis MJ, Skelly LE, Tarnopolsky MA, Gibala MJ. Twelve weeks of sprint interval training improves indices of cardiometabolic health similar to traditional endurance training despite a five-fold lower exercise volume and time commitment. PLoS ONE. 2016;11(4):e0154075. doi: 10.1371/journal.pone.0154075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gjuladin-Hellon T, Davies IG, Penson P, Amiri Baghbadorani R. Effects of carbohydrate-restricted diets on low-density lipoprotein cholesterol levels in overweight and obese adults: A systematic review and meta-analysis. Nutrition Reviews. 2019;77(3):161–180. doi: 10.1093/nutrit/nuy049. [DOI] [PubMed] [Google Scholar]
- Goldenberg JZ, Day A, Brinkworth GD, Sato J, Yamada S, Jönsson T, Beardsley J, Johnson JA, Thabane L, Johnston BC. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ. 2021;372:m4743. doi: 10.1136/bmj.m4743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grace F, Herbert P, Elliott AD, Richards J, Beaumont A, Sculthorpe NF. High intensity interval training (HIIT) improves resting blood pressure, metabolic (MET) capacity and heart rate reserve without compromising cardiac function in sedentary aging men. Experimental Gerontology. 2018;109:75–81. doi: 10.1016/j.exger.2017.05.010. [DOI] [PubMed] [Google Scholar]
- Gram-Kampmann EM, Hansen CD, Hugger MB, Jensen JM, Brønd JC, Hermann AP, Krag A, Olsen MH, Nielsen HB, Højlund K. Effects of a six-month low-carbohydrate diet on glycemic control, body composition and cardiovascular risk factors in patients with type 2 diabetes: an open-label RCT. Diabetes, Obesity and Metabolism. 2022 doi: 10.1111/dom.14633. [DOI] [PubMed] [Google Scholar]
- Hellmuth C, Demmelmair H, Schmitt I, Peissner W, Blüher M, Koletzko B. Association between plasma nonesterified fatty acids species and adipose tissue fatty acid composition. PLoS ONE. 2013;8(10):e74927. doi: 10.1371/journal.pone.0074927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honke K, Zhang Y, Cheng X, Kotani N, Taniguchi N. Biological roles of sulfoglycolipids and pathophysiology of their deficiency. Glycoconjugate Journal. 2004;21(1–2):59–62. doi: 10.1023/B:GLYC.0000043749.06556.3d. [DOI] [PubMed] [Google Scholar]
- Hossain MS, Mineno K, Katafuchi T. Neuronal orphan G-protein coupled receptor proteins mediate plasmalogens-induced activation of ERK and Akt signaling. PLoS ONE. 2016;11(3):e0150846. doi: 10.1371/journal.pone.0150846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelleyman C, Yates T, O’Donovan G, Gray LJ, King JA, Khunti K, Davies MJ. The effects of high-intensity interval training on glucose regulation and insulin resistance: A meta-analysis. Obesity Reviews. 2015;16(11):942–961. doi: 10.1111/obr.12317. [DOI] [PubMed] [Google Scholar]
- Jiang C, Cheong LZ, Zhang X, Ali AH, Jin Q, Wei W, Wang X. Dietary sphingomyelin metabolism and roles in gut health and cognitive development. Advances in Nutrition. 2022;13(2):474–491. doi: 10.1093/advances/nmab117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joseph JJ, Deedwania P, Acharya T, Aguilar D, Bhatt DL, Chyun DA, Di Palo KE, Golden SH, Sperling LS. Comprehensive management of cardiovascular risk factors for adults with type 2 diabetes: A scientific statement from the American Heart Association. Circulation. 2022 doi: 10.1161/CIR.0000000000001040. [DOI] [PubMed] [Google Scholar]
- Kellerer T, Kleigrewe K, Brandl B, Hofmann T, Hauner H, Skurk T. Fatty acid esters of hydroxy fatty acids (FAHFAs) are associated with diet, BMI and Age. Frontiers in Nutrition. 2021;8:1–11. doi: 10.3389/fnut.2021.691401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kytikova OY, Novgorodtseva TP, Antonyuk MV, Gvozdenko TA. Plasmalogens in the pathophysiology and therapy of age-specific diseases. Advances in Gerontology. 2020;10(3):272–281. doi: 10.1134/S207905702003011X. [DOI] [PubMed] [Google Scholar]
- Lee HS, Lee J. Effects of combined exercise and low carbohydrate ketogenic diet interventions on waist circumference and triglycerides in overweight and obese individuals: A systematic review and meta-analysis. International Journal of Environmental Research and Public Health. 2021;18(2):828. doi: 10.3390/ijerph18020828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei L, Zhao N, Zhang L, Chen J, Liu X, Piao S. Gut microbiota is a potential goalkeeper of dyslipidemia. Frontiers in Endocrinology. 2022;13:1–10. doi: 10.3389/fendo.2022.950826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lennerz BS, Koutnik AP, Azova S, Wolfsdorf JI, Ludwig DS. Carbohydrate restriction for diabetes: rediscovering centuries-old wisdom. Journal of Clinical Investigation. 2021 doi: 10.1172/JCI142246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lind L, Salihovic S, Ganna A, Sundström J, Broeckling CD, Magnusson PK, Pedersen NL, Siegbahn A, Prenni J, Fall T, Ingelsson E. A multi-cohort metabolomics analysis discloses sphingomyelin (32:1) levels to be inversely related to incident ischemic stroke. Journal of Stroke and Cerebrovascular Diseases. 2020;29(2):104476. doi: 10.1016/j.jstrokecerebrovasdis.2019.104476. [DOI] [PubMed] [Google Scholar]
- Lopez-Maldonado A, Pastoriza S, Rufián-Henares JÁ. Assessing the antioxidant and metabolic effect of an alpha-lipoic acid and acetyl-L-carnitine nutraceutical. Current Research in Food Science. 2021;4:336–344. doi: 10.1016/j.crfs.2021.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludwig DS, Dickinson SL, Henschel B, Ebbeling CB, Allison DB. Do lower-carbohydrate diets increase total energy expenditure? An updated and reanalyzed meta-analysis of 29 controlled-feeding studies. The Journal of Nutrition. 2020 doi: 10.1093/jn/nxaa350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludwig DS, Hu FB, Lichtenstein AH, Willett WC. Low-fat diet Redux at WHO. The American Journal of Clinical Nutrition. 2023;118(5):849–851. doi: 10.1016/j.ajcnut.2023.09.006. [DOI] [PubMed] [Google Scholar]
- Malaguarnera M. Carnitine derivatives: Clinical usefulness. Current Opinion in Gastroenterology. 2012;28(2):166–176. doi: 10.1097/MOG.0b013e3283505a3b. [DOI] [PubMed] [Google Scholar]
- Messias MCF, Mecatti GC, Priolli DG, de Oliveira Carvalho P. Plasmalogen lipids: Functional mechanism and their involvement in gastrointestinal cancer. Lipids in Health and Disease. 2018;17(1):41. doi: 10.1186/s12944-018-0685-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milanović Z, Sporiš G, Weston M. Effectiveness of high-intensity interval training (HIT) and continuous endurance training for VO2max improvements: A systematic review and meta-analysis of controlled trials. Sports Medicine. 2015;45(10):1469–1481. doi: 10.1007/s40279-015-0365-0. [DOI] [PubMed] [Google Scholar]
- Miller VJ, LaFountain RA, Barnhart E, Sapper TS, Short J, Arnold WD, Hyde PN, Crabtree CD, Kackley ML, Kraemer WJ, Villamena FA, Volek JS. A ketogenic diet combined with exercise alters mitochondrial function in human skeletal muscle while improving metabolic health. American Journal of Physiology-Endocrinology and Metabolism. 2020;319(6):E995–E1007. doi: 10.1152/ajpendo.00305.2020. [DOI] [PubMed] [Google Scholar]
- Moser O, Tschakert G, Mueller A, Groeschl W, Pieber TR, Obermayer-Pietsch B, Koehler G, Hofmann P. Effects of high-intensity interval exercise versus moderate continuous exercise on glucose homeostasis and hormone response in patients with type 1 diabetes mellitus using novel ultra-long-acting insulin. PLoS ONE. 2015;10(8):e0136489. doi: 10.1371/journal.pone.0136489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller MJ, Bosy-Westphal A. From a “Metabolomics fashion” to a sound application of metabolomics in research on human nutrition. European Journal of Clinical Nutrition. 2020;74(12):1619–1629. doi: 10.1038/s41430-020-00781-6. [DOI] [PubMed] [Google Scholar]
- Norwitz NG, Soto-Mota A, Kaplan B, Ludwig DS, Budoff M, Kontush A, Feldman D. The lipid energy model: reimagining lipoprotein function in the context of carbohydrate-restricted diets. Metabolites. 2022;12(5):460. doi: 10.3390/metabo12050460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pastor R, Bouzas C, Tur JA. Beneficial effects of dietary supplementation with olive oil, oleic acid, or hydroxytyrosol in metabolic syndrome: Systematic review and meta-analysis. Free Radical Biology and Medicine. 2021;172:372–385. doi: 10.1016/j.freeradbiomed.2021.06.017. [DOI] [PubMed] [Google Scholar]
- Paul S, Lancaster GI, Meikle PJ. Plasmalogens: A potential therapeutic target for neurodegenerative and cardiometabolic disease. Progress in Lipid Research. 2019;74:186–195. doi: 10.1016/j.plipres.2019.04.003. [DOI] [PubMed] [Google Scholar]
- Paul S, Smith AAT, Culham K, Gunawan KA, Weir JM, Cinel MA, Jayawardana KS, Mellett NA, Lee MK, Murphy AJ, Lancaster GI. Shark liver oil supplementation enriches endogenous plasmalogens and reduces markers of dyslipidemia and inflammation. Journal of Lipid Research. 2021;62:100092. doi: 10.1016/j.jlr.2021.100092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen BK, Saltin B. Exercise as medicine—evidence for prescribing exercise as therapy in 26 different chronic diseases. Scandinavian Journal of Medicine & Science in Sports. 2015;25(3):1–72. doi: 10.1111/sms.12581. [DOI] [PubMed] [Google Scholar]
- Pennisi M, Lanza G, Cantone M, D’Amico E, Fisicaro F, Puglisi V, Vinciguerra L, Bella R, Vicari E, Malaguarnera G. Acetyl-L-carnitine in dementia and other cognitive disorders: A critical update. Nutrients. 2020;12(5):1389. doi: 10.3390/nu12051389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pongrac Barlovic D, Harjutsalo V, Sandholm N, Forsblom C, Groop PH. Sphingomyelin and progression of renal and coronary heart disease in individuals with type 1 diabetes. Diabetologia. 2020;63(9):1847–1856. doi: 10.1007/s00125-020-05201-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Popovic ZV, Sandhoff R, Sijmonsma TP, Kaden S, Jennemann R, Kiss E, Tone E, Autschbach F, Platt N, Malle E, Hermann-Josef G. Sulfated glycosphingolipid as mediator of phagocytosis: SM4s enhances apoptotic cell clearance and modulates macrophage activity. The Journal of Immunology. 2007;179(10):6770–6782. doi: 10.4049/jimmunol.179.10.6770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porez G, Prawitt J, Gross B, Staels B. Bile acid receptors as targets for the treatment of dyslipidemia and cardiovascular disease. Journal of Lipid Research. 2012;53(9):1723–1737. doi: 10.1194/jlr.R024794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramos JS, Dalleck LC, Tjonna AE, Beetham KS, Coombes JS. The impact of high-intensity interval training versus moderate-intensity continuous training on vascular function: a systematic review and meta-analysis. Sports Medicine. 2015;45(5):679–692. doi: 10.1007/s40279-015-0321-z. [DOI] [PubMed] [Google Scholar]
- Reginato A, Veras ACC, da Nóbrega Baqueiro M, Panzarin C, Siqueira BP, Milanski M, Lisboa PC, Torsoni AS. The role of fatty acids in ceramide pathways and their influence on hypothalamic regulation of energy balance: a systematic review. International Journal of Molecular Sciences. 2021;22(10):5357. doi: 10.3390/ijms22105357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehman K, Haider K, Jabeen K, Akash MSH. Current perspectives of oleic acid: Regulation of molecular pathways in mitochondrial and endothelial functioning against insulin resistance and diabetes. Reviews in Endocrine and Metabolic Disorders. 2020;21(4):631–643. doi: 10.1007/s11154-020-09549-6. [DOI] [PubMed] [Google Scholar]
- Riebe D, editor. Guidelines for exercise testing and prescription (ACSM) 10. Wolters Kluwer Health, Lippincott Williams & Wilkins; 2018. [Google Scholar]
- Riecan M, Paluchova V, Lopes M, Brejchova K, Kuda O. Branched and linear fatty acid esters of hydroxy fatty acids (FAHFA) relevant to human health. Pharmacology & Therapeutics. 2022;231:107972. doi: 10.1016/j.pharmthera.2021.107972. [DOI] [PubMed] [Google Scholar]
- Rodgers GP, Collins FS. Precision nutrition—the answer to “What to Eat to Stay Healthy”. JAMA. 2020;324(8):735. doi: 10.1001/jama.2020.13601. [DOI] [PubMed] [Google Scholar]
- Sawyer BJ, Tucker WJ, Bhammar DM, Ryder JR, Sweazea KL, Gaesser GA. Effects of high-intensity interval training and moderate-intensity continuous training on endothelial function and cardiometabolic risk markers in obese adults. Journal of Applied Physiology. 2016;121(1):279–288. doi: 10.1152/japplphysiol.00024.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sébédio JL. Advances in food and nutrition research. Elsevier; 2017. Metabolomics, nutrition, and potential biomarkers of food quality, intake and health status; pp. 83–116. [DOI] [PubMed] [Google Scholar]
- Shu H, Peng Y, Hang W, Li N, Zhou N, Wang DW. Emerging roles of ceramide in cardiovascular diseases. Aging and Disease. 2022;13(1):232. doi: 10.14336/AD.2021.0710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Signorelli P, Conte C, Albi E. The multiple roles of sphingomyelin in Parkinson’s disease. Biomolecules. 2021;11(9):1311. doi: 10.3390/biom11091311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverii GA, Cosentino C, Santagiuliana F, Rotella F, Benvenuti F, Mannucci E, Cresci B. Effectiveness of low-carbohydrate diets for long-term weight loss in obese individuals: A meta-analysis of randomized controlled trials. Diabetes, Obesity and Metabolism. 2022;24(8):1458–1468. doi: 10.1111/dom.14709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skow SL, Jha RK. A ketogenic diet is effective in improving insulin sensitivity in individuals with type 2 diabetes. Current Diabetes Reviews. 2022 doi: 10.2174/1573399818666220425093535. [DOI] [PubMed] [Google Scholar]
- Su XQ, Wang J, Sinclair AJ. Plasmalogens and Alzheimer’s disease: A review. Lipids in Health and Disease. 2019;18(1):100. doi: 10.1186/s12944-019-1044-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sumner LW, Amberg A, Barrett D, Beale MH, Beger R, Daykin CA, Fan TWM, Fiehn O, Goodacre R, Griffin JL, Hankemeier T, Hardy N, Harnly J, Higashi R, Kopka J, Lane AN, Lindon JC, Viant MR. Proposed minimum reporting standards for chemical analysis. Metabolomics. 2007;3(3):211–221. doi: 10.1007/s11306-007-0082-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan D, Ertunc ME, Konduri S, Zhang J, Pinto AM, Chu Q, Kahn BB, Siegel D, Saghatelian A. Discovery of FAHFA-containing triacylglycerols and their metabolic regulation. Journal of the American Chemical Society. 2019;141(22):8798–8806. doi: 10.1021/jacs.9b00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi M, Okazaki T. Role of ceramide/sphingomyelin (SM) balance regulated through “SM cycle” in cancer. Cellular Signalling. 2021;87:110119. doi: 10.1016/j.cellsig.2021.110119. [DOI] [PubMed] [Google Scholar]
- Tippetts TS, Holland WL, Summers SA. Cholesterol—the devil you know; ceramide—the devil you don’t. Trends in Pharmacological Sciences. 2021;42(12):1082–1095. doi: 10.1016/j.tips.2021.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tutunchi H, Ostadrahimi A, Saghafi-Asl M. The effects of diets enriched in monounsaturated oleic acid on the management and prevention of obesity: A systematic review of human intervention studies. Advances in Nutrition. 2020;11(4):864–877. doi: 10.1093/advances/nmaa013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulaszewska MM, Weinert CH, Trimigno A, Portmann R, Andres Lacueva C, Badertscher R, Brennan L, Brunius C, Bub A, Capozzi F, Rosso MC, Cordero CE, Daniel H, Durand S, Egert B, Ferrario PG, Feskens EJM, Franceschi P, Vergères G. Nutrimetabolomics: An integrative action for metabolomic analyses in human nutritional studies. Molecular Nutrition & Food Research. 2019;63(1):1800384. doi: 10.1002/mnfr.201800384. [DOI] [PubMed] [Google Scholar]
- US Department of Agriculture, US Department of Health and Human Services. (2020). Dietary Guidelines for Americans, 2020–2025.
- van den Berg, R. A., Hoefsloot, H. C., Westerhuis, J. A., Smilde, A. K., & van der Werf, M. J. (2006). Centering, scaling, and transformations: improving the biological information content of metabolomics data. BMC genomics, 7(1), 142. 10.1186/1471-2164-7-142 [DOI] [PMC free article] [PubMed]
- Verma N, Gupta JK, Varshney KK, Srivastava R. Ameliorative effect of Acetyl L-carnitine in Alzheimer’s disease via downregulating of homocysteine levels in hyperhomocysteinemia induced cognitive deficit in mouse model. Drug Metabolism Letters. 2021;14(3):219–231. doi: 10.2174/1872312814666211209102136. [DOI] [PubMed] [Google Scholar]
- Volek JS, Phinney SD, Krauss RM, Johnson RJ, Saslow LR, Gower B, Yancy WS, King JC, Hecht FM, Teicholz N, Bistrian BR, Hamdy O. Alternative dietary patterns for americans: low-carbohydrate diets. Nutrients. 2021;13(10):3299. doi: 10.3390/nu13103299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q, Liu R, Chang M, Zhang H, Jin Q, Wang X. Dietary oleic acid supplementation and blood inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Critical Reviews in Food Science and Nutrition. 2022;62(9):2508–2525. doi: 10.1080/10408398.2020.1854673. [DOI] [PubMed] [Google Scholar]
- Wang S, Xu J, Zheng J, Zhang X, Shao J, Zhao L, Hao J. Anti-inflammatory and antioxidant effects of acetyl-L-carnitine on atherosclerotic rats. Medical Science Monitor. 2020;26:1–11. doi: 10.12659/MSM.920250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei D, Wu S, Liu J, Zhang X, Guan X, Gao L, Xu Z. Theobromine ameliorates nonalcoholic fatty liver disease by regulating hepatic lipid metabolism via mTOR signaling pathway in vivo and in vitro. Canadian Journal of Physiology and Pharmacology. 2021;99(8):775–785. doi: 10.1139/cjpp-2020-0259. [DOI] [PubMed] [Google Scholar]
- Yore MM, Syed I, Moraes-Vieira PM, Zhang T, Herman MA, Homan EA, Patel RT, Lee J, Chen S, Peroni OD, Dhaneshwar AS, Hammarstedt A, Smith U, McGraw TE, Saghatelian A, Kahn BB. Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects. Cell. 2014;159(2):318–332. doi: 10.1016/j.cell.2014.09.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youm YH, Nguyen KY, Grant RW, Goldberg EL, Bodogai M, Kim D, Planavsky N, Lupfer C, Kanneganti TD, Kang S, Horvath TL, Fahmy TM, Crawford PA, Biragyn A, Alnemri E, Dixit VD. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nature Medicine. 2015;21(3):263–269. doi: 10.1038/nm.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu XD, Wang JW. Ceramide de novo synthesis in non-alcoholic fatty liver disease: Pathogenic mechanisms and therapeutic perspectives. Biochemical Pharmacology. 2022;202:115157. doi: 10.1016/j.bcp.2022.115157. [DOI] [PubMed] [Google Scholar]
- Zafeiridis A, Chatziioannou AC, Sarivasiliou H, Kyparos A, Nikolaidis MG, Vrabas IS, Pechlivanis A, Zoumpoulakis P, Baskakis C, Dipla K, Theodoridis GA. Global metabolic stress of isoeffort continuous and high intensity interval aerobic exercise: A comparative 1H NMR metabonomic study. Journal of Proteome Research. 2016;15(12):4452–4463. doi: 10.1021/acs.jproteome.6b00545. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data described in the manuscript, code book, and analytic code will be made available upon request pending approval by authors.