Abstract
Objective
This study aimed to evaluate the effects of the combination of curcumin and piperine supplementation on Fasting Plasma Glucose (FPG), Homeostatic Model of Insulin Resistance (HOMA-IR), and Body Mass Index (BMI) in patients with prediabetes and type 2 Diabetes Mellitus (T2DM). This review was done to identify potential herbal remedies that may help improve glycemic parameters, leading to better health outcomes in combination with current antidiabetic treatment.
Methodology
This systematic review was based on Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). It was conducted in 2023 with sources and databases from MEDLINE, EBSCO-Host, ScienceDirect and ProQuest. This paper included randomized-controlled trials exploring the effects of the combination of curcumin and piperine on patients with prediabetes and T2DM. Systematic reviews, observational studies, case reports, case series, conference abstracts, book sections, commentaries/editorials, non-human studies and articles with unavailable full-text and written in non-English language, were excluded. The key terms for the literature search were “curcumin,” “piperine,” “prediabetes” and “Type 2 Diabetes Mellitus.” We use Cochrane Risk of Bias (RoB) 2 for quality assessment of the included studies and Review Manager (RevMan) 5.4 to do the meta-analysis.
Results
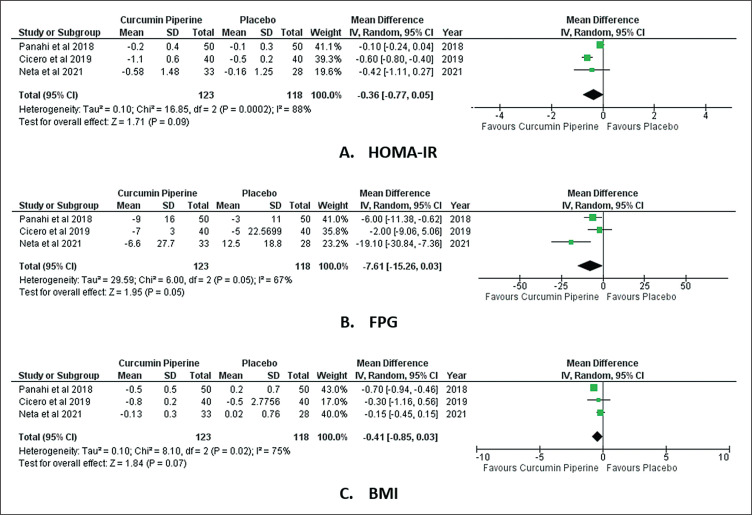
A total of three studies were included in this systematic review. Two studies from Neta et al., and Cicero et al., showed no significant difference in HOMA-IR, BMI and FPG levels between the curcumin, piperine and placebo groups. One study from Panahi et al. demonstrated a significant difference in BMI levels between the curcumin and piperine and placebo groups (p <0.01). The meta-analysis showed that FPG levels, HOMA-IR and BMI improved among patients with diabetes given in curcumin and piperine with reported mean differences (MD) of = -7.61, 95% CI [-15.26, 0.03], p = 0.05, MD = -0.36, 95% CI [-0.77 to 0.05], p = 0.09, and MD = -0.41, 95% CI [-0.85 to 0.03], p = 0.07, respectively).
Conclusions
The supplementation of curcumin and piperine showed a numerical reduction in FPG, HOMA-IR and BMI, but were not statistically significant. Further research is needed as there is a paucity of studies included in the review.
Keywords: glycemic profile, curcumin, piperine, prediabetes, type 2 diabetes mellitus
INTRODUCTION
Type 2 Diabetes mellitus (T2DM) is a chronic condition characterized by hyperglycemia. The term prediabetes is used to describe a state between normality and diabetes.1 The latest survey conducted in 2021 by the International Diabetes Federation (IDF) showed that 537 million adults worldwide and about 90 million in Southeast (SE) Asia are living with diabetes. These numbers affected the overall health and well-being of patients. Diabetes causes serious complications, resulting in a high mortality rate of 6.7 million deaths worldwide (around 747,000 in SE Asia) in 2021.2 Moreover, it contaminates the quality of life (QoL) with a disability-adjusted life year (DALY) sum of 66.3 million in 2019.3 Total health expenditures reached up to 996 billion USD.2
Chronic hyperglycemia contributes to the production of Advanced Glycation End products (AGEs) and Reactive Oxidative Species (ROS), which increase the risk of microvascular and macrovascular complications. Various medications to combat hyperglycemia and its complications have been developed. These medications include insulin analogs and oral medications that improve insulin sensitivity, increase insulin production, inhibit glucose absorption and more.4 However, these benefits have disadvantages, such as high costs and some adverse effects.5 Alternatives, such as herbal remedies, were created to minimize these drawbacks without compromising the benefits of modern drugs. Furthermore, these remedies provide anti-inflammatory and antioxidant effects that could potentially prevent complications.6
Curcumin, an active compound of turmeric, is among the most well-studied herbal remedies. It has antidiabetic properties as it can reduce hepatic glucose production by activating AMP kinase and inhibit both glucose-6-phosphatase and phosphoenolpyruvate carboxykinase activity; therefore it may potentially prevent T2DM complications by reducing oxidative stress.6 It may also play a role in improving fasting plasma glucose (FPG), HbA1c, body mass index (BMI), lipid profile and insulin sensitivity.7,8
Another herbal remedy of interest as a diabetic medication is piperine, a chemical compound found in black pepper. Its therapeutic benefits in diabetes are similar to curcumin. Giving piperine in combination with curcumin was found to have a synergistic effect. Piperine increases curcumin bioavailability, either through increased absorption or reduced metabolism.10 Individually, these herbal remedies were extensively studied, however, they are yet to be reviewed in combination. Therefore, this systematic review and meta-analysis aimed to evaluate the effects of combined curcumin and piperine supplementation on the glycemic profile of patients with prediabetes and T2DM.
METHODOLOGY
This systematic review was designed and conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) 2020 statement guideline.11
Eligibility criteria
Types of studies
This systematic review included all published and unpublished randomized-controlled trials that investigated the effects of the combination of curcumin and piperine supplementation on patients with prediabetes and T2DM. Conversely, reviews, cross-sectional studies, cohort studies, case reports, case series, conference abstracts, book sections, commentaries/editorials and non-human studies were excluded. Articles not in English and without available fulltext manuscripts were also excluded.
Participants
All patients aged ≥18 with prediabetes and T2DM were included in this study. Diagnosis of prediabetes and type 2 diabetes mellitus was based on the American Diabetes Association criteria.12 Patients were diagnosed with T2DM if glycated hemoglobin (HbA1c) was ≥6.5% or fasting plasma glucose (FPG) was ≥126 mg/dL. Prediabetes was diagnosed at an HbA1c level of 5.7 to 6.4% or FPG of 100 to 125 mg/dL. There were no limitations for sex and race. Patients with current glucocorticoid use, cardiovascular disease, kidney failure, inflammatory diseases, acute infections and who were pregnant or breastfeeding were excluded from the study.
Variable of interest
Our study aimed to evaluate the effect of combined curcumin and piperine supplementation on glycemic profile in patients with prediabetes and T2DM.
Outcome of interest
Outcomes of interest in this study were the changes in fasting plasma glucose (FPG), homeostatic model assessment for insulin resistance (HOMA-IR), and body mass index (BMI) before and after intervention with combined curcumin/piperine. Other glycemic and metabolic parameters, such as hormonal and lipid parameters, inflammatory biomarkers and oxidative molecules, were considered additional outcomes.
Search strategy and study selection
We used MEDLINE, EBSCO-Host, Science Direct and ProQuest electronic databases to search for eligible studies as of 2023. EBSCO-Host and ProQuest databases were also screened for grey literature to identify unpublished studies with suitable PICO criteria. Five independent authors identified eligible studies by using the following keywords: (Type 2 Diabetes Mellitus or Diabetes Mellitus, Noninsulin-Dependent or NIDDM or Maturity-Onset Diabetes Mellitus or Diabetes Mellitus, Type 2 or Prediabetes or Prediabetic State OR Prediabetic States) and (curcumin or curcuma or curcuminoid) and (Piperidines OR Piperine).
All obtained studies were exported into the Mendeley reference manager software. The authors independently reviewed and screened these studies for duplicates. Studies with titles and abstracts unbefitting for this paper’s objectives were excluded. Full texts of the selected studies were thoroughly assessed using the eligibility criteria described above, and those eligible were subsequently included in this review. The review team resolved any disagreements.
Data collection process
All studies were analyzed and the following data were extracted: first author, country of origin, study design, sample sizes, age, sex, prediabetes and diabetes criteria, curcumin and piperine administration protocol, adjusted confounding factors/population matching and the outcome of interest.
Summary measures
HOMA-IR, FPG and BMI for patients in the curcumin plus piperine group and placebo group were measured and reported as numerical (continuous) data. The data were presented in mean ± standard deviation for normally distributed data or median (interquartile range) for nonnormally distributed data. The p-value and confidence interval were also included for each item to determine significance.
Assessment of risk of bias/ Quality assessment
Each study was assessed using the Cochrane Risk of Bias Tool 2.0 (RoB 2) for randomized controlled trials.13 The tool consists of seven main domains: (a) Random Sequence Generation; (b) Allocation Concealment; (c) Blinding of Participants and Personnel; (d) Blinding of Outcome Assessment; (e) Incomplete Outcome Data; (f) Selective Reporting; and (g) Other Source of Bias. From each domain, the risk of bias was considered as low, high, and moderate bias. Each trial's overall quality was divided into three groups based on the degree of bias present: (1) low risk of bias (low risk of bias across all domains); (2) high risk of bias (high risk of bias across multiple domains); and (3) some concerns some concerns across at least one domain). Two reviewers evaluated each article separately, and disagreements were discussed among the whole review team until agreement was obtained.
Synthesis of results and statistical analysis
For all continuous outcomes, we calculated the standardized mean differences (SMDs) and 95% Confidence Intervals (CIs) based on the mean changes from baseline to the end of the study from each group. Statistical analyses were done for the between-group comparison. Missing data, such as Standard Deviation (SD), were calculated from the Standard Error (SE). SE of a Mean Difference (MD) was calculated from a p-value by finding the associated t-value. Having calculated the SE of the MD, the SD can be calculated from the SE. Because some studies reported primary outcomes using different evaluation or calculation methods, the meta-analyses were conducted with a random effects model. This model presupposes that the treatment impact will be distributed over certain populations, giving each study an equal weighting. The combined effect measures of the direct comparisons from individual interventions were compared by using the inverse variance method.
Heterogeneity across trials was assessed using the I2 statistic. An I2 value of less than 25% is considered low heterogeneity, between 25% and 50% indicates moderate to substantial heterogeneity, and more than 50% is considered high heterogeneity.14 When heterogeneity was present, possible causes were investigated via sensitivity analyses. A p-value <0.05 was considered significant. A continuous outcome was analyzed as a weighted mean difference. Differences across studies were calculated based on population sample sizes. In addition, publication bias was evaluated visually using a funnel plot, in which the effect of each trial was plotted by the inverse of its SE. All analyses were conducted using RevMan software version 5.4.15
Quality assessment in the cumulative evidence
Grading of Recommendation, Assessment, Development, and Evaluation (GRADE) was described to determine the confidence in cumulative evidence. The decision was made by considering several aspects of the studies, such as study limitations, consistency, directness, precision and reporting bias. The evidence results were graded as very low, low, moderate and high.
RESULT
PRISMA
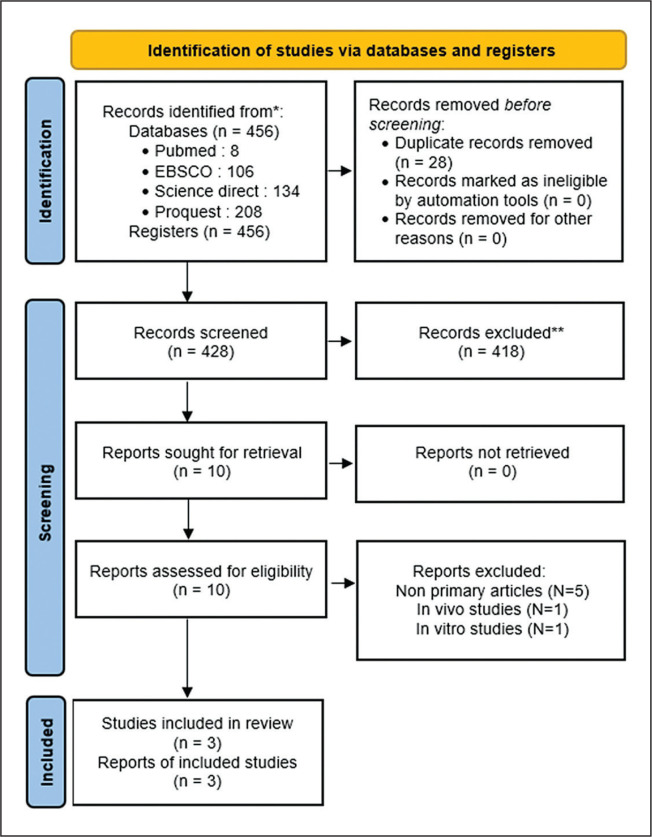
A flow chart of the research selection process and its results are summarized in Figure 1. The search strategy yielded 456 potentially relevant studies. According to the selection criteria, ten studies were identified for further full-text assessment, of which five articles were review articles (nonprimary articles), one was an in-vitro study, and one was an in-vivo study. No unpublished studies were included, minimizing publication bias qualitatively. Finally, three studies were included in the systematic review and three studies eligible for data extraction were included in the meta-analysis. All studies were published between 2003 and 2023.
Figure 1.
PRISMA 2020 Flow Diagram of Included Studies.
Quality assessment
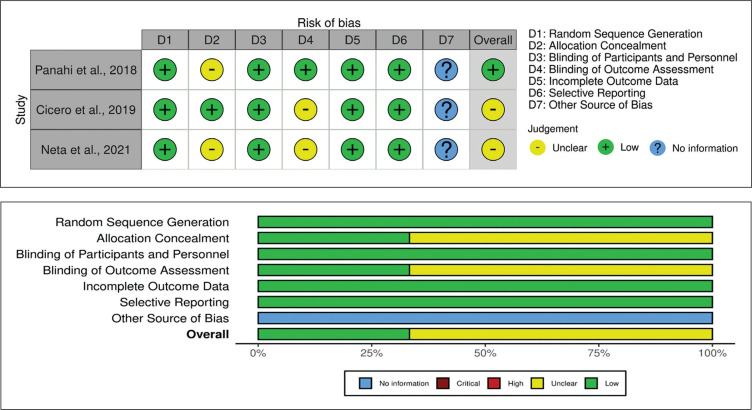
Quality assessment for obtained studies was performed using the Cochrane Risk of Bias Tool 2.0 (RoB 2) for randomized-controlled trials (Figure 2). We obtained one low-bias study16 and two unclear bias studies.17,18 Across all parameters of bias, two studies16,18 reported an unclear bias of the allocation concealment, and two studies17,18 reported an unclear bias in the blinding of outcome assessment.
Figure 2.
Results of Study Quality Assessment.
Characteristics of the included studies
Three randomized-controlled studies met the inclusion criteria. These studies included 123 patients receiving a combination of curcumin/piperine and 118 patients in the placebo groups. The characteristics of the included studies, including the number of participants (N), age (years), prediabetes and diabetes diagnosis, curcumin and piperine administration protocol, adjusted confounding factors/population matching, and the outcome of interest were extracted from each study and reported in Table 1. The mean ages of the intervention groups compared with the placebo groups ranged from 43 ± 8 to 63.1 ± 11.1 years and 41 ± 7 to 61.9 ± 11.0 years, respectively. One study16 did population matching of baseline characteristics at the beginning of the study, and the other two studies17,18 matched the population in terms of diet and physical activity. Out of the three studies, one study18 was conducted in Brazil and the other two studies16,17 were conducted in Iran. The inclusion criteria, dosages and frequency of taking curcumin varied across studies. The participants of each study were adults, either male or female, diagnosed with T2DM, except in Cicero et al.,17 who used subjects with no T2DM, but with prediabetes.
Table 1.
Study characteristics
| No. | Author, publication year, country | Types of Study Curcumin Piperine | Population | Curcumin piperine and placebo administration protocol | Prediabetes and diabetes criteria | Adjusted confounding factors/population matching | Outcome of interest | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Total (N) | Age (years) | |||||||||
| Curcumin piperine | Placebo | Curcumin piperine | Placebo | |||||||
| 1 | Panahi et al., 2018, Iran16 | RCT | Male: 25 Female: 25 | Male: 26 Female: 24 | 43 ± 8 | 41 ± 7 | - Intervention Groups: Curcuminoids (Curcumin C3 Complex®, Sami Labs LTD, Bangalore, India; 500mg/day). The curcuminoids preparation (C3 Complex®) contained curcumin, demethoxycurcumin and bisdemethoxycurcumin in a patented ratio. Each curcuminoids capsule also contained 5 mg piperine (Bioperine®; Sami Labs LTD, Bangalore, India). - Control Groups: Placebo (not further explained). - Duration of treatment: 3 months |
T2D based on fasting plasma glucose ≥126 mg/dL, glycated hemoglobinA1c (HbA1c) ≥6.5 %, or the use of standard anti-diabetic treatments. | Patients' characteristics were considered similar through randomization, but there is no further information regarding the adjusted confounding factors. | Fasting serum concentrations of insulin, glucose, HbA1c, C-peptide, aspartate aminotransferase (AST), alanine aminotransferase (ALT) and high-sensitivity C-reactive protein (hs-CRP), homeostatic model assessments (HOMA) of insulin resistance (HOMA-IR) and beta-cell function (HOMA-β) |
| 2 | Cicero et al., 2019, Iran.17 | RCT | Male: 18 Female: 22 | Male: 19 Female: 21 | 54 ± 3 | 53 ± 5 | - Intervention Groups: Tablets containing 800 mg phytosomal curcumin (Curserin®: 200 mg curcumin, 120 mg phosphatidylserine, 480 mg phosphatidylcholine, associated to 8 mg piperine from Piper nigrum L. dry extract) taken 2 tablets per day after dinner. - Control Groups: Placebo (not further explained). - Duration of treatment: 3 months |
FPG levels between 100 and 125 mg/dL | During the study, subjects were encouraged to follow basic guidelines for a Mediterranean diet, refrain from consuming too much dairy and food derived from red meat, and keep generally stable eating patterns. Additionally, people were urged to boost their physical activity by cycling or walking briskly for 20-30 mins, 3-5x each week. | Total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), triglycerides (TG), LDL-Cholesterol (LDL-C), fasting plasma glucose (FPG), glutamic-oxaloacetic transaminase (GOT), glutamic-pyruvic transaminase (GPT), and gamma-glutamyl transferase (gamma-GT) |
| 3 | Neta et al., 2021, Brazil.18 | RCT | Male: 10 Female:23 | Male 4 Female: 24 | 63.1 ± 11.1 | 61.9 ± 11.0 | - Intervention Groups: 500 mg of Curcuma longa L. to be ingested once a day on an empty stomach. 5 mg piperine was added to every capsule - Control Groups: Placebo (500 mg of carboxymethyl cellulose once a day on an empty stomach for 120 days - Duration of treatments: 3 months |
Not stated | Participants were advised not to make any special changes in their diet and physical activity, as well as to report any changes in medications during the intervention period. | (LDL-C), fasting plasma glucose (FPG), glutamic-oxaloacetic |
Final results
All three studies included in this systematic review showed insignificant differences in Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) between the intervention and placebo groups, as shown in Table 2. Insignificant differences were also shown in all studies for the Fasting Plasma Glucose (FPG) levels, as shown in Table 3. A randomized-controlled study by Panahi et al.,16 showed a significant difference in body mass index (BMI) between the two groups, while the rest showed otherwise (Table 4). There were no reports of attrition with regard to treatment during the follow-up period. Furthermore, all included studies reported no adverse effects during the treatment duration.
Table 2.
Changes in Homeostatic Model Assessment For Insulin Resistance (HOMA-IR) before and after intervention
| No | Author, Year Curcumin + Piperine | HOMA-IR |
|||
|---|---|---|---|---|---|
| Curcumin + Piperine | Placebo | Mean difference (95% CI) | p* | ||
| 1 | Panahi et al., 201816 | − 0.2 ± 0.4 | − 0.1 ± 0.3 | -0.10 (-0.24, 0.04) | 0.511 |
| 2 | Cicero et al., 201917 | − 1.1 ± 0.6 | -0.5 ± 0.2 | -0.60 (-0.80, -0.40) | 0.013 |
| 3 | Neta et al., 202118 | -0.58 ± 1.48 | -0.16 ± 1.25 | -0.42 (-1.11, 0.27) | 0.458 |
Table 3.
Changes in Fasting Plasma Glucose (FPG) before and after intervention
| No | Author, Year Curcumin + Piperine | FPG |
|||
|---|---|---|---|---|---|
| Curcumin + Piperine | Placebo | Mean difference (95% CI) | p* | ||
| 1 | Panahi et al., 201815 | − 9 ± 16 | − 3 ± 11 | -6.00 (11.38, -0.62) | 0.048 |
| 2 | Cicero et al., 201917 | − 7 ± 3 | -5 ± 22.56 | -2.00 (-9.06, 5.06) | 0.317 |
| 3 | Neta et al., 202116 | -6.6 ± 27.7 | 12.5 ± 18.8 | -19.10 (-30.84, -7.36) | 0.630 |
Table 4.
Changes in Body Mass Index (BMI) before and after intervention
| No | Author, Year Curcumin + Piperine | BMI |
|||
|---|---|---|---|---|---|
| Curcumin + Piperine | Placebo | Mean difference (95% CI) | p-value* | ||
| 1 | Panahi, et al. 201816 | − 0.5 ± 0.5 | 0.2 ± 0.7 | -0.70 (-0.94, -0.46) | <0.001 |
| 2 | Cicero et al., 201915 | − 0.8 ± 0.2 | -0.5 ± 2.7756 | -0.30 (-1.16, 0.56) | 0.286 |
| 3 | Neta et al., 202117 | -0.13 ± 0.3 | 0.02 ± 0.76 | -0.15 (-0.45, 0.15) | 0.387 |
significant if p-value ≤0.05
Meta-analysis results
The results of quantitative synthesis for each outcome (FPG, HOMA-IR and BMI) are reported in continuous data as mean differences (MD), as shown in Figures 1, 2 and 3, respectively. In one study, fasting plasma glucose is proven to be lower in curcumin-piperine groups compared to placebo groups (p=0.05), with a mean difference of -7.61, but analyzed together, the results are not significant due to the overall estimates of its forest plot intersecting with the line of no effect (95% CI [-15.26, 0.03]). The most significant differences in FPG between the two groups before and after the intervention are observed in Neta et al., with a mean difference of -19.10 (95% CI [-30.84, -7.36])18 and the smallest is shown in Cicero et al., with the mean difference of -2.00 (95% CI [-9.06, 5.06]).17 HOMA-IR and BMI are found to be numerically lower in curcumin-piperine groups but not statistically significant, with both overall estimates crossing the line of no effect (p=0.09 and p=0.07, mean differences of -0.36, 95% CI [-0.77, 0.05] and -0.41, 95% CI [-0.85, 0.03], respectively).
Figure 3.
(A) Meta-analysis results [Forest plot] for Fasting Plasma Glucose (FPG); (B) Meta-analysis results [Forest plot] for Homeostatic Model Assessment for Insulin Resistance (HOMA-IR); and (C) Meta-analysis results [Forest plot] for Body Mass Index (BMI) in T2DM patients before and after supplementation of curcumin piperine capsule compared to placebo groups.
DISCUSSION
Population characteristics, such as ethnicity, play a significant role in treatment response. It is found that ethnicity influences insulin sensitivity, thereby improving glycemic control.19 Moreover, it also greatly affects the pharmacokinetic properties of therapeutic substances.20 A study conducted by Viana et al., in Brazil,21 which is similar to Neta et al.,18 found that DM patients have poor glycemic control despite treatment.21 Meanwhile, Panahi et al.,16 and Cicero et al.,17 discovered that the treatment has a crucial effect on a person's glycemic control.21 There is also a difference in the spectrum of hyperglycemia (prediabetes and DM) among the three studies. Diabetes mellitus is the progression of prediabetes, which is characterized by worsening glycemic control and insulin resistance necessitating treatment intensification.22 Furthermore, differences were noted regarding the use of anti-diabetic drugs between the studies. Although the efficacy of these drugs is clearly stated,4 it can be disregarded since each study stated the baseline parameters and no changes were made in anti-diabetic drugs used. All these dissimilarities could potentially alter the overall results of the studies.
Baseline parameters are primarily similar across the three studies. The average BMIs are classified as overweight-obese in all studies. Factors leading to a higher BMI include a sedentary lifestyle, alcohol consumption, low socioeconomic status, poor diet, age, sex and genetics.23 A study by Kim et al. showed that women are more likely to become obese when compared to men.24 Mosha et al. found that individuals aged 35 years and older were at the highest risk (69%) of becoming overweight and obese.25 These findings are consistent with the characteristics observed in the study by Neta et al.,18 which included a predominantly female and retired population who are known to be more sedentary. It also aligns with the average age reported in all three studies, ranging between 40 and 50 years. The HOMA-IR scores across the three studies are also relatively similar, showing significant insulin resistance. Among numerous factors that may contribute to the worsening of this parameter, BMI has the greatest impact.26 The difference was observed solely in the FPG values, particularly in the study conducted by Neta et al.,18 where it exhibited higher levels in the experimental group than in the control group. This variation occurred regardless of randomization protocol and despite a homogenous sociodemographic profile between the two groups. The FPG values are also distinct in the study by Cicero et al.,17 since the prediabetes population was used. The inconsistencies identified may have an impact on the findings of the studies.
Curcumin is a hydrophobic polyphenol, an extract from the rhizome of Curcuma longa L. (turmeric), a species belonging to the Curcuma genus (Zingiberaceae family). The main constituents of curcumin are curcumin I (1,7-bis(4-hydroxy-3-methoxyphenyl) -1,6-heptadiene-3,5-dione), curcumin II (demethoxycurcumin) and curcumin III (bisdemethoxycurcumin).27 The three types of curcumin constituents differ in their aromatic rings. These three have been shown to exert anti-inflammatory and antioxidant effects, amongst other beneficial biological activities, including antitumoral, neuroprotective, antimicrobial, hepatoprotective and antirheumatic activities.27
T2DM patients with poor glycemic control can be characterized by their constant hyperglycemic state. The constant and chronic hyperglycemic state contributes to the production of Advanced Glycation End products (AGEs) and Reactive Oxidative Species (ROS).28 These products will result in lipid peroxidation, an increase in oxidative stress (reinforced by the upregulation of MAPK, NADPHOX, NFKB, and TGFB), inflammation (caused by the upregulation of NFKB, TNF-a, and IL-6), VEGF, ICAM-1, VCAM-1, endothelial dysfunction and cellular apoptosis. These processes increase the risk of microvascular and macrovascular complications.8
In T2DM, hyperglycemia and hyperinsulinemia occur secondary to disruptions in beta cell function and insulin resistance. The increase in lipid peroxidation, hyperglycemia and increased hepatic glucose production promotes lipotoxicity, glucotoxicity and glucolipotoxicity, respectively, leading to the activation of pro-apoptotic signals to the pancreatic beta-cells.28 This event amplifies the insulin-resistant state of the multiple organs caused by the mitochondrial dysfunction. As the cells continuously become more resistant to insulin, a decrease in beta-cell function can also be expected. These processes lead to the progression of T2DM.
The antidiabetic properties of curcumin can be attributed to the reduction of hepatic glucose production by activating AMP kinase and inhibiting both glucose-6-phosphatase and phosphoenolpyruvate carboxykinase activity.16 This leads to the reduction in glucolipotoxicity, therefore reducing oxidative stress. Not only that, but curcumin also exerts unique biological properties. The active compound of Curcuma produces significant immunosuppressants that inhibit the production of IL-2 and IL-12.It also inhibits iNOS (inducible nitric oxide synthase), COX-2 (cyclooxygenase-2), lipoxygenase-5 and other pro-inflammatory cytokines, such as TNF-α, IL-1, IL-6, and IL-8.8
The anti-apoptotic characteristic of curcuminoids can be seen, as neurotoxic factors in macrophages and alveolar monocytes stimulated by lipopolysaccharides were also suppressed. It inhibits phosphorylation and degradation of the nuclear factor of kappa light polypeptide gene enhancer in B cells and activates the gamma receptor mechanism activated by the peroxisome proliferator.8 This, in turn, reduces overall inflammation induced by the NF-kB pathway. The anti-inflammatory and anti-apoptotic effects will assist in the preservation of the beta-cell functions, together with the reduction of glycemic parameters and improvement in the HOMA-IR, therefore reducing the progression of T2DM.8
Researchers have yet to find an effective dose of curcumin.35 However, many have hypothesized that it has a dose-dependent effect, producing greater benefits at a higher dosage.8,36 In animal and human studies, curcumin has been shown to have good tolerability and safety, even at high doses (up to 12 g/day orally). Curcumin is to be taken with precaution and is not recommended for pregnant or lactating women, children, or those with anemia or liver disease.29 Despite the wide range of beneficial effects, curcumin presents with the downside of having low solubility (in water), degradation in alkaline environments, crystallization in acidic environments and rapid metabolism (glucuronidation and sulfation in the liver and plasma, converting it into water-soluble metabolites to be excreted in the urine). These characteristics decrease its absorption in the gastrointestinal tract, translating to low bioavailability.8
Piperine is an alkaloid mainly found in P. nigrum (Black Pepper). The chemical structure of piperine is N-piperoylpiperidin; (E, E)-1-(5-[1,3-benzodioxol-5-yl] -1-oxo-2,4-pentadienyl)- piperidine. Many studies have shown that piperine increases the bioavailability of many drugs by promoting rapid absorption of drugs and nutrients or by inhibiting several cytochrome P450 enzymes and phase II reactions.30 Piperine has been shown to increase solubility, plasma levels, improve the pharmacokinetic profile and cellular absorption of curcumin.31 Shiba et al., showed that the administration of 2 g of curcumin together with 20 mg of piperine to healthy individuals increased the bioavailability of curcumin by 2000 % with no adverse effects.32 The study by Panahi et al., showed a significant reduction in serum levels of C-peptide, HbA1c and glucose in the curcumin-piperine-treated group versus the placebo group.16 These studies showed the synergistic effect of curcumin and piperine, especially on glycaemic, inflammatory and hepatic markers in T2DM patients.29
Meta-analysis was conducted and the outcome was presented as mean and standard deviation. The meta-analysis showed no significant difference (p = 0.05) in FPG between the curcumin-piperine and placebo groups, with a mean difference of 7.61, favoring the curcumin-piperine group. An in vivo study done by Kaur et al., on diabetic rats showed significantly decreased plasma glucose levels (p<0.001) treated with curcumin with piperine and quercetin (extracted from Allium cepa) for four weeks.33
The meta-analysis for HOMA-IR and BMI also showed insignificant differences (p = 0.07 and p=0.09, respectively) for the curcumin/piperine group compared to the placebo group. These results are in accordance with another study performed in Iran by Hodaei et al.,34 which found that oral supplementation of curcumin could reduce the levels of inflammatory biomarkers, but not insulin resistance markers such as HOMA-IR. The author hypothesized that it may be due to the short duration of intervention in both groups. In this review, we also found that duration of interventions is varied between studies, with Neta et al.,18 being the longest (120 days) and Cicero et al., as the shortest (60 days);17 the other is Panahi et al (90 days).16 Regarding the BMI results, the lack of association may be due to the failure to adjust for body weight at the beginning of the study, therefore the baseline values were not similar. The limited number and high heterogeneity of these studies could have caused the lack of significant results for this meta-analysis.
Strength and limitation of the study
This study provided a comprehensive systematic review and meta-analysis of the effect of the combination of curcumin and piperine on the glycemic profile in patients with prediabetes and T2DM. However, this systematic review still has several limitations. Two studies from Panahi et al.,16 and Neta et al.,18 included T2DM patients who take standard anti-diabetic medications as their study participants. Neta et al., excluded patients who consume insulin as antidiabetic treatment.18 Cicero et al., administered a higher dosage of the interventions compared to the other two studies.17 Furthermore, they included prediabetic patients and excluded patients who are on oral or injectable medications for diabetes. The lack of studies was a significant limitation that may have affected the results. Language bias was inevitable as these studies were written in languages other than English, leading to other types of bias, as the number of studies did not fulfill the requirement for a quantitative publication bias analysis using a funnel plot. Further studies, particularly those with clinical significance, must be conducted.
CONCLUSION
In summary, the supplementation of curcumin and piperine showed a trend toward reducing FPG, HOMA-IR and BMI, but the results were not statistically significant. Further research is needed as there is a paucity of studies included in the review.
Acknowledgments
The authors would like to express their gratitude to all their colleagues from Atma Jaya Catholic University of Indonesia for all their support.
Funding Statement
Funding Source None.
Statement of Authorship
All authors certified fulfillment of ICMJE authorship criteria.
CRediT Author Statement
NDW: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing – original draft preparation, Writing – review and editing, Visualization, Project administration; ET: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing – original draft preparation, Writing – review and editing, Project administration; LFJJ: Conceptualization, Methodology, Validation, Investigation, Resources, Writing – original draft preparation, Writing – review and editing; SA: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Resources, Data Curation, Writing – original draft preparation, Writing – review and editing, Project administration; ESA: Conceptualization, Methodology, Validation, Investigation, Resources, Writing – original draft preparation, Writing – review and editing; MRI: Conceptualization, Methodology, Investigation, Resources, Writing – original draft preparation, Writing – review and editing, Supervision.
Author Disclosure
The authors declared no conflict of interest.
References
- 1.World Health Organization . Diabetes. Accessed June 10, 2023. https://www.who.int/news-room/fact-sheets/detail/diabetes
- 2.IDF Diabetes Atlas, 10th ed. Accessed June 10, 2023. https://diabetesatlas.org/.
- 3.Safiri S, Karamzad N, Kaufman JS, et al. Prevalence, deaths and disability-adjusted-life-years (dalys) due to type 2 diabetes and its attributable risk factors in 204 countries and territories, 1990-2019: Results from the global burden of disease study 2019. Front Endocrinol. 2022; 13:838027. PMID: 35282442. PMCID: . 10.3389/fendo.2022.838027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Get a Handle on Diabetes Medication. American Diabetes Association. Accessed June 10, 2023. https://diabetes.org/healthy-living/medication-treatments [Google Scholar]
- 5.Kumar S, Mittal A, Babu D, Mittal A. Herbal medicines for diabetes management and its secondary complications. Curr Diabetes Rev. 2021;17(4):437–56. PMID: 33143632. 10.2174/1573399816666201103143225. [DOI] [PubMed] [Google Scholar]
- 6.Pang GM, Li FX, Yan Y, et al. Herbal medicine in the treatment of patients with type 2 diabetes mellitus. Chin Med J (Engl). 2019;132(1):78–85. PMID: 30628962. PMCID: . 10.1097/CM9.0000000000000006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhang DW, Fu M, Gao SH, Liu JL. Curcumin and diabetes: A systematic review. Evid-Based Complement Alternat Med. 2013;2013: 636053. PMID: 24348712. PMCID: . 10.1155/2013/636053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marton LT, Pescinini-E-Salzedas LM, Camargo MEC, et al. The effects of curcumin on diabetes mellitus: A systematic review. Front Endocrinol (Lausanne). 2021;12: 669448. PMID: 34012421. PMCID: . 10.3389/fendo.2021.669448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nogara L, Naber N, Pate E, Canton M, Reggiani C, Cooke R. Piperine’s mitigation of obesity and diabetes can be explained by its up-regulation of the metabolic rate of resting muscle. Proc Natl Acad Sci. 2016;113(46):13009–14. PMID: 27799519. PMCID: . 10.1073/pnas.1607536113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Heidari H, Bagherniya M, Majeed M, Sathyapalan T, Jamialahmadi T, Sahebkar A. Curcumin-piperine co-supplementation and human health: A comprehensive review of preclinical and clinical studies. Phytother Res. 2023;37(4):1462–87. PMID: 36720711. 10.1002/ptr.7737. [DOI] [PubMed] [Google Scholar]
- 11.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Syst Rev. 2021;10(1):89. PMID: 33781348. PMCID: . 10.1186/s13643-021-01626-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2010;33(Suppl 1):S62–9. PMID: 20042775. PMCID: . 10.2337/dc10-S062. Erratum in Diabetes Care. 2010;33(4):e57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sterne JAC, Savović J, Page MJ, et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. PMID: 31462531. 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 14.Fletcher J. What is heterogeneity and is it important? BMJ. 2007;334(7584):94–6. PMID: 17218716. PMCID: . 10.1136/bmj.39057.406644.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tantry TP, Karanth H, Shetty PK, Kadam D. Self-learning software tools for data analysis in meta-analysis. Korean J Anesthesiol. 2021;74(5):459–61. PMID: 33677944. PMCID: . 10.4097/kja.21080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Panahi Y, Hosseini MS, Khalili N, et al. Effects of curcumin on serum cytokine concentrations in subjects with metabolic syndrome: A post-hoc analysis of a randomized controlled trial. Biomed Pharmacother. 2016;82:578–82. PMID: 27470399. 10.1016/j.biopha.2016.05.037. [DOI] [PubMed] [Google Scholar]
- 17.Cicero AFG, Sahebkar A, Fogacci F, Bove M, Giovannini M, Borghi C. Effects of phytosomal curcumin on anthropometric parameters, insulin resistance, cortisolemia and non-alcoholic fatty liver disease indices: A double-blind, placebo-controlled clinical trial. Eur J Nutr. 2020;59(2):477–83. PMID: 30796508. PMCID: . 10.1007/s00394-019-01916-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.de Figueiredo Neta JFVeras VS, de Sousa DF, et al. Effectiveness of the piperine-supplemented Curcuma longa L. in metabolic control of patients with type 2 diabetes: A randomised double-blind placebo-controlled clinical trial. Int J Food Sci Nutr. 2021;72(7):968–77. PMID: 33586583. 10.1080/09637486.2021.1885015. [DOI] [PubMed] [Google Scholar]
- 19.Spanakis EK, Golden SH. Race/ethnic difference in diabetes and diabetic complications. Curr Diab Rep. 2013;13(6):814-23. PMID: 24037313. PMCID: . 10.1007/s11892-013-0421-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shah RR, Gaedigk A. Precision medicine: does ethnicity information complement genotype-based prescribing decisions? Ther Adv Drug Saf. 2018;9(1):45–62. PMID: 29318005. PMCID: . 10.1177/2042098617743393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Viana LV, Leitão CB, Kramer CK, et al. Poor glycaemic control in Brazilian patients with type 2 diabetes attending the public healthcare system: A cross-sectional study. BMJ Open. 2013;3(9):e003336. PMID: 24052610. PMCID: . 10.1136/bmjopen-2013-003336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Khan RMM, Chua ZJY, Tan JC, Yang Y, Liao Z, Zhao Y. From pre-diabetes to diabetes: Diagnosis, treatments and translational research. Medicina (Mex). 2019;55(9):546. PMID: 31470636. PMCID: DOI: 10.3390/medicina55090546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Flores-Dorantes MT, Díaz-López YE, Gutiérrez-Aguilar R. Environment and gene association with obesity and their impact on neurodegenerative and neurodevelopmental diseases. Front Neurosci. 2020;14:863. PMID: 32982666. PMCID: . 10.3389/fnins.2020.00863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim D, Hou W, Wang F, Arcan C. Factors affecting obesity and waist circumference among US adults. Prev Chronic Dis. 2019;16:E02. PMID: 30605422. PMCID: . 10.5888/pcd16.180220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mosha D, Paulo HA, Mwanyika-Sando M, et al. Risk factors for overweight and obesity among women of reproductive age in Dar es Salaam, Tanzania. BMC Nutr. 2021;7(1):37. PMID: 34266482. PMCID: . 10.1186/s40795-021-00445-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jhorawat R, Bansal N, Beniwal P, Agarwal D, Malhotra V. Factors Affecting insulin resistance and its relation to vitamin d status and clinical nutritional parameters in dialysis patients: A single-center Indian study. Indian J Nephrol. 2018;28(1):41–5. PMID: 29515300. PMCID: . 10.4103/ijn.IJN_361_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Servida S, Panzeri E, Tomaino L, et al. Overview of curcumin and piperine effects on glucose metabolism: The case of an insulinoma patient’s loss of consciousness. Int J Mol Sci. 2023;24(7):6621. PMID: 37047589. PMCID: . 10.3390/ijms24076621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Galicia-Garcia U, Benito-Vicente A, Jebari S, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21(17):6275. PMID: 32872570. PMCID: . 10.3390/ijms21176275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pivari F, Mingione A, Brasacchio C, Soldati L. Curcumin and type 2 diabetes mellitus: Prevention and treatment. Nutrients. 2019;11(8):1837. PMID: 31398884. PMCID: . 10.3390/nu11081837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Atal S, Atal S, Vyas S, Phadnis P. Bio-enhancing effect of piperine with metformin on lowering blood glucose level in alloxan induced diabetic mice. Pharmacogn Res. 2016;8(1):56–60. PMID: 26941537. PMCID: . 10.4103/0974-8490.171096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Meghwal M, Goswami TK. Piper nigrum and piperine: An update. Phytother Res PTR. 2013;27(8):1121–30. PMID: 23625885. 10.1002/ptr.4972. [DOI] [PubMed] [Google Scholar]
- 32.Shoba G, Joy D, Joseph T, Majeed M, Rajendran R, Srinivas PS. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998;64(4):353–6. PMID: 9619120. 10.1055/s-2006-957450. [DOI] [PubMed] [Google Scholar]
- 33.Kaur G, Meena C. Amelioration of obesity, glucose intolerance, and oxidative stress in high-fat diet and low-dose streptozotocin-induced diabetic rats by combination consisting of “curcumin with piperine and quercetin.” ISRN Pharmacol. 2012;2012:957283. PMID: 22474599. PMCID: . 10.5402/2012/957283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hodaei H, Adibian M, Nikpayam O, Hedayati M, Sohrab G. The effect of curcumin supplementation on anthropometric indices, insulin resistance and oxidative stress in patients with type 2 diabetes: a randomized, double-blind clinical trial. Diabetol Metab Syndr. 2019;11:41. PMID: 31149032. PMCID: . 10.1186/s13098-019-0437-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Den Hartogh DJ, Gabriel A, Tsiani E. Antidiabetic properties of curcumin II: Evidence from in vivo studies. Nutrients. 2019;12(1):58. PMID: 31881654. PMCID: . 10.3390/nu12010058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Den Hartogh DJ, Gabriel A, Tsiani E. Antidiabetic properties of curcumin I: Evidence from in vitro studies. Nutrients. 2020;12(1):118. PMID: 31906278. PMCID: . 10.3390/nu12010118. [DOI] [PMC free article] [PubMed] [Google Scholar]