Abstract
BACKGROUND:
Sedentary behavior (SB) is observationally associated with cardiovascular disease risk. However, randomized clinical trials testing causation are limited. We hypothesized that reducing SB would decrease blood pressure (BP) and pulse wave velocity (PWV) in sedentary adults.
METHODS:
This parallel-arm, 3-month randomized clinical trial recruited desk workers, age 18 to 65 years, with systolic BP 120 to 159 or diastolic BP (DBP) 80 to 99 mm Hg, off antihypertensive medications, and reporting <150 min/wk of moderate to vigorous intensity physical activity. Participants were randomized to a SB reduction intervention or a no-contact control group. The intervention sought to replace 2 to 4 h/d of SB with standing and stepping through coaching, a wrist-worn activity prompter, and a sit–stand desk. SB and physical activity were measured with a thigh-worn accelerometer and quantified during all waking hours and separately during work and nonwork times. Clinic-based resting systolic BP (primary outcome) and DBP, 24-hour ambulatory BP, and PWV were assessed by blinded technicians at baseline and 3 months.
RESULTS:
Participants (n=271) had a mean age of 45 years and systolic BP/DBP 129/83 mm Hg. Compared with controls, intervention participants reduced SB (−1.15±0.17 h/d), increased standing (0.94±0.14 h/d), and increased stepping (5.4±2.4 min/d; all P<0.05). SB and activity changes mainly occurred during work time and were below the goal. The intervention did not reduce BP or PWV in the intervention group compared with controls. Between-group differences in resting systolic BP and DBP changes were −0.22±0.90 (P=0.808) and 0.13±0.61 mm Hg (P=0.827), respectively. The findings were similarly null for ambulatory BP and PWV. Decreases in work-time SB were associated with favorable reductions in resting DBP (r=0.15, P=0.017). Contrary to our hypotheses, reductions in work-time SB (r=−0.19, P=0.006) and increases in work-time standing (r=0.17, P=0.011) were associated with unfavorable increases in carotid-femoral PWV. As expected, increases in nonwork-time standing were favorably associated with carotid-femoral PWV (r=−0.14, P=0.038).
CONCLUSIONS:
A 3-month intervention that decreased SB and increased standing by ≈1 hour during the work day was not effective for reducing BP. Future directions include examining effects of interventions reducing SB through activity other than work-time standing and clarifying association between standing and PWV in opposite directions for work and nonwork time.
REGISTRATION:
URL: https://www.clinicaltrials.gov; Unique identifier: NCT03307343.
Keywords: behavior, blood pressure, randomized controlled trial, vascular stiffness, work
Clinical Perspective.
What Is New?
An intervention that replaced ≈1 h/d of sedentary behavior with mostly standing at work did not improve blood pressure (BP) or arterial stiffness (measured by pulse wave velocity) in desk workers with untreated elevated or high BP.
Greater reductions in sedentary behavior during work time were associated with small benefits in diastolic BP.
An unexpected effect was observed where less sedentary behavior and more standing at work were associated with increases in central arterial stiffness, and increases in standing outside of work were associated with less arterial stiffness.
What Are the Clinical Implications?
Using a sit–stand desk to replace sitting with standing for ≈1 h/d during work should not be prescribed as an effective lifestyle treatment for patients with elevated or high BP.
Further research is needed in different populations (eg, older age, higher BP, lower activity levels) and to test the cardiovascular effects of greater reductions in sedentary behavior and replacing sedentary behavior with behaviors other than work-time standing at a sit–stand desk.
High blood pressure (BP) is a leading and modifiable cause of cardiovascular disease (CVD) and death.1 First-line treatments for high BP are nonpharmacologic interventions, such as increasing moderate to vigorous intensity physical activity (MVPA), improving diet, losing weight, and reducing excessive alcohol intake.2 The prevalence of optimal BP <120/80 mm Hg has declined to <1 in 5 among US adults on the basis of the most recent national data.3
Sedentary behavior (SB) is waking behavior of low intensity that is performed in seated, reclined, or lying posture.4 SB is increasingly recognized as behaviorally and physiologically distinct from inactivity, defined as a lack of MVPA.5 For example, a desk worker with high SB can also have high MVPA if they engage in daily exercise after work. Cohort studies consistently find that high SB is associated with higher risk of CVD and death, and that such risks are even higher when SB is accumulated in prolonged bouts and with low levels of MVPA.6–8 Short-term laboratory studies in humans find a range of negative cardiovascular effects from SB lasting 3 to 8 hours, including lower-extremity blood pooling, reduced vascular reactivity, increased BP, and arterial stiffening as measured by pulse wave velocity (PWV).9,10 These acute effects are mitigated if the SB is interrupted by light-intensity physical activity breaks, such as walking, resistance exercises, or standing.9–11
The direct relationship between prolonged SB and CVD risk in randomized crossover laboratory studies and observational cohorts has led to the hypothesis that the acute negative cardiovascular effects of prolonged SB may accumulate, resulting in high BP and, eventually, CVD.10 However, results of SB-reduction randomized clinical trials (RCTs) are inconclusive, because most have not specifically studied individuals with high BP or measured BP comprehensively.12–14
Thus, we conducted the RESET-BP study (Effect of Reducing Sedentary Behavior on Blood Pressure), an RCT testing the efficacy of a multicomponent SB-reduction intervention to reduce resting BP, 24-hour ambulatory BP, and PWV. Enrollment was restricted to adult desk workers with high BP measured in the clinic, off antihypertensive medications, and reporting <150 min/wk of MVPA (which is below current guidelines15). Our primary hypothesis was that the SB-reduction intervention would reduce BP and PWV to a greater extent compared with the control condition. Secondary hypotheses were that individuals with greater decreases in device-measured SB and greater increases in light-intensity standing and stepping activity would have greater improvements in BP and PWV.
METHODS
Study Design and Sample
RESET-BP, conducted in Pittsburgh, PA, was a parallel RCT with 2 arms: a 3-month, multicomponent SB-reduction intervention and a no-contact (usual behavior) control group. The trial was registered on October 11, 2017 (URL: https://www.clinicaltrials.gov; Unique identifier: NCT03307343); enrolled participants between January 3, 2018, and August 4, 2022; and completed follow-up assessments in November 2022. All research procedures were approved by the University of Pittsburgh Human Research Protection Office and all participants provided informed consent. A detailed protocol is published elsewhere, including modifications made due to the COVID-19 pandemic.16 Data sharing is available upon written request, approval of the study investigators, and execution of a data sharing agreement.
Participant inclusion criteria were as follows: age 21 to 65 years; average clinic-based resting systolic BP (SBP) 120 to 159 mm Hg or diastolic BP (DBP) 80 to 99 mm Hg; employed for ≥20 h/wk in sedentary work at a desk; office location within ~25 miles or our research center; stable employment in current job for ≥3 months and expected to stay in current position for the following 3 months; able to obtain supervisor approval to have a sit–stand desk or attachment installed; and a mobile phone able to receive text messages. Participants were excluded if they had average clinic-based resting SBP ≥160 or DBP ≥100 mm Hg; were using antihypertensive or glucose-lowering medications; had a history of CVD; were unable to obtain written consent to participate from a medical provider; were pregnant in the previous 3 months or had plans to become pregnant in the following 3 months; were planning to be away from their work desk for an extended period during follow-up; were using a sit–stand desk, standing desk, or wearable prompter to interrupt SB; or were currently enrolled in a lifestyle intervention.
Several strategies were used for recruitment, including electronic or paper mailings to targeted zip codes and local employers, advertisement on the University of Pittsburgh research registry, and partnering with the University of Pittsburgh Medical Center primary care practices. Interested potential participants provided informed consent during an orientation, and then completed screening procedures to further evaluate eligibility, including clinic-based resting BP and completion of baseline assessments.
Randomization
A stratified randomization scheme was used with 1:1 randomization to either intervention or control, in random block sizes of 4 and 6, and within strata of sex (male/female) and BP stage (elevated/stage 1 or stage 2).2
Intervention
The 3-month RESET-BP intervention targeted reducing SB, especially SB accumulated in prolonged bouts, by replacing it with standing and other light-intensity physical activity.16 The multicomponent intervention used behavioral strategies, including self-monitoring, goal setting, problem solving,17 environmental modification with a study-provided sit–stand desk or attachment,18 proximal reminders to interrupt sitting with a wrist-worn prompter that vibrated during participant-selected daytime hours if the participant did not achieve 250 steps during the hour (Fitbit Flex 2), and twice-weekly text messaging.19 Behavioral counseling included monthly lessons (initially conducted in person, but modified to videoconference due to the COVID-19 pandemic) alternating with shorter monthly phone or videoconference check-ins. During desk work, participants were instructed to replace sitting with standing for 15 to 30 min/h at their sit–stand workstation (thereby accumulating 2–4 h/d of standing20) and to reduce periods of prolonged SB (ie, ≥60 minutes) by adding 4 to 8 light-intensity movement breaks (eg, a 2- to 3-minute walk) across the seated work day. Participants were also counseled to generally reduce SB during nonworking hours, although goals were more individualized and less structured. Further details on the rationale, design, and COVID-19–related adaptations to the intervention are available in our published protocol.16
Measurements
Resting BP was measured at baseline and 3-month follow-up. Clinic-based resting SBP (primary outcome) and DBP were calculated as the average of measurements obtained on 2 separate occasions during baseline and follow-up using a standardized protocol16 developed using guidelines.2,21 During the first visit, arm circumferences were measured to choose appropriately sized cuffs, BP was measured on both arms, and the arm with the higher SBP was selected for use in all subsequent measurements. BP measurements occurred between 6:00 and 11:00 am after verbal confirmation of 8-hour abstention from food, caffeine, and nicotine, and 24-hour abstention from MVPA and alcohol; after bladder emptying and a 10-minute quiet rest with arm supported at heart level and feet and back supported; and using a validated oscillometric device (HEM-907XL; Omron Healthcare).22 During every assessment, 2 measurements were taken, with a 1-minute rest between them, and averaged. If SBP differed by ≥10 mm Hg or DBP differed by ≥6 mm Hg, a third measurement was obtained and included in the average.
Twenty-four–hour ambulatory BP was assessed at baseline and 3 months using an Oscar 2 ambulatory BP monitor (Suntech Medical) and a standard protocol.23 An appropriately sized cuff was placed on the nondominant arm and participants were instructed to wear the monitor continuously for 25 hours. At follow-up, participants were instructed to continue their typical behaviors (intervention or control). The monitor was programmed to obtain a reading approximately every 30 and 60 minutes during typical wake and sleep times, respectively. Ambulatory BP data were downloaded using AccuWin Pro software, and artifact or erratic oscillotertic signals identified by the software were discarded. Valid readings were classified as daytime or nocturnal on the basis of a participant wear log. Then, daytime BP (average of all daytime readings), nocturnal BP (average of all nocturnal readings), and 24-hour BP (average of all readings) were calculated. To account for the influence of posture and movement during the preceding time period on ambulatory BP readings, which was expected to differ by group at follow-up, we developed a novel method reported in detail elsewhere.24 In this method, daytime BP averages were synchronized with device-measured posture and activity and separated into daytime readings that occurred after 5 minutes of continuous sitting (seated BP) or with any standing or stepping in the previous 5 minutes (nonseated BP). Seated BP is not influenced by recent activity, improving comparability across randomized groups.24
PWV—the gold standard noninvasive measure of arterial stiffness25—was assessed at baseline and 3 months using tonometry and following published recommendations.25 Arterial path lengths were estimated using the subtractive method for the carotid-femoral path and direct distance for the carotid-radial path. After measurement of resting BP, participants rested supine for 10 minutes. Then, sensors were applied to the carotid, radial, and femoral sites to capture 3 scans of 10 high-quality pulse pressure waveforms. Sensor output was processed by the Complior Analyse (ALAM Medical) and averaged across scans to estimate central (carotid-femoral PWV [cfPWV]) and peripheral (carotid-radial PWV [crPWV]) arterial stiffness. Using this protocol, our laboratory has excellent inter- and intratechnician reliability, with intraclass correlation coefficients ≥0.90.
Height was assessed at baseline as the average of 2 measures within 0.5 cm from a wall-mounted stadiometer. Weight was assessed at baseline and 3-month follow-up as the average of 2 measures within 0.1 kg using a calibrated Tanita digital scale.
Intervention fidelity was measured at each intervention contact by assessing provision of the intervention components (delivery); self-report that the sit–stand desk, wrist-worn prompter, and text messages were working properly, as well as goal setting during lessons (receipt); and self-report of self-monitoring and goal achievement (enactment).26
SB and physical activity were assessed at baseline and 3 months using best practice methodology for field measurement of SB (24-hour wear protocol; thigh-worn activPAL3 micro; PALTechnologies) and MVPA (waking wear protocol; waist-worn GT3X; ActiGraph).27,28 Participants were instructed to wear both devices for 9 days during a typical working schedule. Only wear days 3 through 9 (7 full days) were used to calculate averages. The first 2 days of wear were not included, because they were concurrent with ambulatory BP monitoring, which could have affected activity patterns.
activPAL data were exported as event files using PALTechnologies software. Using participant diaries that recorded work, sleep, and nonwear time during monitoring, event files were processed using standard procedures to calculate daily durations of SB, SB accumulated in prolonged bouts of ≥30 and ≥60 minutes, standing time, stepping time, steps per day, and sit–stand transitions.28,29 SB and physical activity were quantified across all waking hours and then separately during work and nonwork times. GT3X accelerometer data were exported as 60-second epochs using ActiLife software and nonwear periods were removed using the Choi algorithm.30 Daily MVPA was quantified using Freedson vector magnitude cut points.28 activPAL and GT3X data were considered valid if ≥4 days with ≥10 hours of waking wear time were captured.31 Valid days were aggregated to generate weekly MVPA (GT3X) and daily averages of SB and other physical activity variables (activPAL). activPAL variables were also averaged separately for working and nonworking times.
Self-reported leisure-time MVPA was assessed using the Paffenbarger Physical Activity Questionnaire.32
Medical History, Medication Changes, and Adverse Events
A detailed medical history including current medications was obtained during baseline screening. At 3-month follow-up, a standardized interval medical history interview systematically assessed any changes in medical history or medications since randomization. New or worsening medical conditions triggered the completion of an adverse event form by the blinded study coordinator, including an evaluation of severity and plausible relationship to assessment procedures or the intervention, which was then reviewed by the principal investigator. Adverse events were also passively collected by study personnel on the basis of voluntary reporting during any study-related contact.
Sample Size and Statistical Analyses
Assuming SBP (our primary outcome) had a baseline SD of 10 mm Hg and a change in SD of 11 mm Hg,33,34 80% statistical power, and a 2-sided α=0.05, we estimated a required sample size of 240 (120 per group) completers to detect a between-arm difference in SBP as small as 4 mm Hg (Cohen d=0.4; PASS 2012 software; Number Cruncher Statistical Systems).33,34 Although we proposed to recruit 300 participants, allowing for 20% attrition, we stopped recruitment at 271 participants with approval from our Data Safety and Monitoring Board due to the lower than expected attrition rate of 3%.
We compared the baseline characteristics between intervention and control groups using independent samples t, Wilcoxon rank sum, χ2, or Fisher exact tests. Intervention fidelity and adverse events were summarized descriptively.
We used an intention-to-treat approach to evaluate the efficacy of the intervention for yielding greater improvements in SB, activity, and study outcomes (BP, PWV, and weight), compared with control, with multiple imputation using Markov chain Monte Carlo method to account for missing data.35 We fit a series of analysis of covariance models with baseline to 3-month change in outcomes as the dependent variable, baseline value of the outcome as a covariate, and intervention arm as the effect of interest. For device-based SB and physical activity, time point–specific accelerometer wear time was also included as a covariate in follow-up analyses using linear mixed models. Statistical significance (at α=0.05) of the between-arm comparisons was considered the formal test of hypothesis. To evaluate the robustness of our findings on outcomes, sensitivity analyses were conducted excluding participants without follow-up data; carrying the last observation forward (although not generally recommended); using subgroup analyses by sex, age (<50 versus ≥50 years), BP stage (elevated or stage 1 versus stage 2), and body mass index (<30 versus ≥30 kg/m2); excluding participants in whom follow-up BP was not measured per protocol (eg, outside of window, measured only once, measured outside of the laboratory); excluding participants who reported substantial changes in their work environment; and using a linear mixed model with both baseline and follow-up measures as dependent variable, group and time (with interaction term) as independent factors, and a participant random effect.
We examined how changes in SB and physical activity were associated with changes in outcomes among all participants and separately by randomized group with correlation coefficients (r). We also examined partial correlation coefficients adjusting for baseline values of outcomes, SB, and physical activity.
SAS software version 9.4 (SAS Institute, Inc.) was used for statistical analysis.
RESULTS
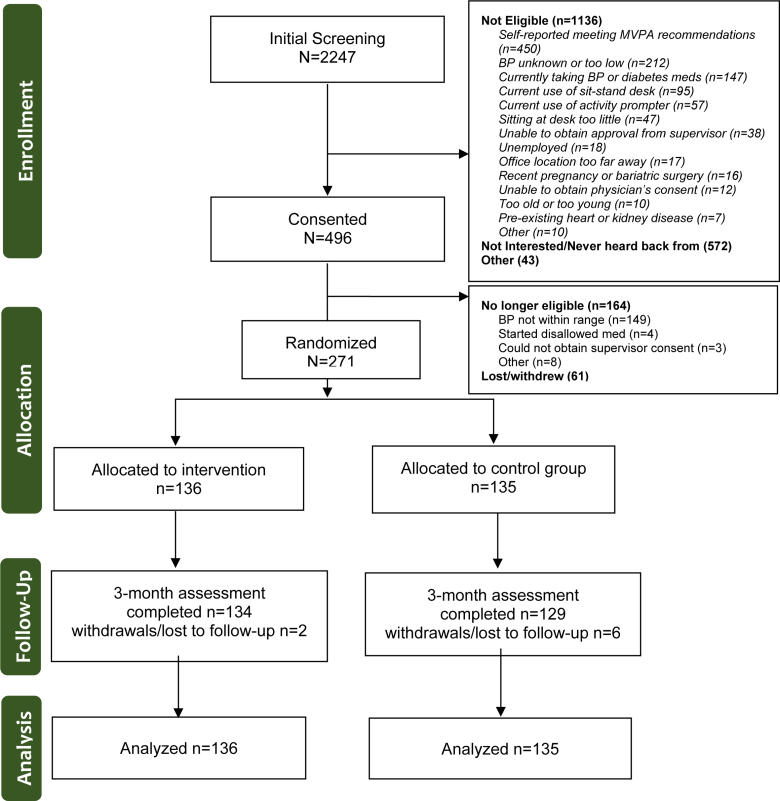
Enrollment, randomization, and retention are summarized in the CONSORT (Consolidated Standards of Reporting Trials) diagram (Figure). Primary reasons for ineligibility during initial screening were self-reported MVPA >150 min/wk or lack of interest. Of 496 participants who provided informed consent, 271 (55%) were randomized to intervention (n=136) or control (n=135). Individuals typically did not progress to randomization due to an ineligible screening BP or lack of interest. The primary outcome measure (resting SBP) was obtained in 134 intervention (99%) and 129 (96%) control participants at follow-up, corresponding to 97% retention. Two participants were lost to follow-up, 2 withdrew due to time constraints, 2 became pregnant, 1 withdrew because of medical reasons, and another withdrew because of COVID-19 exposure concerns. No participant initiated antihypertensive medication use during follow-up.
Figure.
CONSORT diagram. BP indicates blood pressure; CONSORT, Consolidated Standards of Reporting Trials; and MVPA, moderate to vigorous intensity physical activity.
Participant characteristics by randomized group are reported in Table 1. Participants in the intervention versus control group were slightly older (age 47.2 versus 43.2 years; P=0.0042) and had higher cfPWV (8.2 versus 7.7 m/s; P=0.0025). All other demographic and clinical characteristics were similar across groups.
Table 1.
Participant Baseline Characteristics by Randomized Group

Intervention Fidelity and Effects on SB and Physical Activity
More than 95% of intervention lessons were delivered over the intervention period (Table S1). Mean±SD lesson durations were 74±19 minutes (lesson 1) and 42±7 minutes (lessons 2 and 3). Monthly check-ins averaged 14 to 15 minutes. Delivery of intervention components was successful (sit–stand desk working properly, >95%; wrist-worn prompter working properly, >94%; text messages sent, >88%). Intervention receipt success was high, with participants reporting wearing the wrist-worn prompter (>94%) and receiving text messages (>86%) across the intervention. Participant report of self-monitoring standing and movement breaks was ≥83% throughout follow-up.
The intervention had significant effects on SB as compared with the control group (Table 2). Considering waking time (all day), intervention as compared with control participants had decreased SB by 1.15 h/d (P<0.0001). When segmented into working and nonworking hours, SB reductions in the intervention compared with the control group were observed during work times (−1.14 h/d; P<0.0001) but not during nonwork times (−0.20 h/d; P=0.1881). Similar patterns were observed for SB accumulated in prolonged bouts ≥30 or ≥60 minutes. SB appears to have been replaced primarily with standing; all-day standing increased by 0.94 h/d (P<0.0001) and work-time standing increased by 1.25 h/d (P<0.0001). Time spent stepping also increased significantly, although the magnitude of effects was small (≤6 min/d; P<0.05). Daily steps only increased significantly during work time (420 steps/d; P=0.0043). No greater increases for intervention participants compared with controls were observed for sit–stand transitions, MVPA, or any non–work-time behaviors. Adjusting for wear time had minimal effects on results (Table S2).
Table 2.
Sedentary Time and Physical Activity by Randomized Group
Effects of on BP and PWV
The effect of the intervention on our primary outcome (3-month change in resting SBP) and secondary outcomes (3-month changes in resting DBP, ambulatory SBP and DBP, cfPWV, and crPWV) are reported in Table 3 and Figure S1. Although resting SBP decreased in both groups, there was no difference in 3-month changes between groups (−0.22±0.90 mm Hg; P=0.8084). A similar pattern was observed in resting DBP (0.13±0.61 mm Hg; P=0.8265). Between-group changes in ambulatory BP were similar (eg, 24-hour SBP: 0.31±0.87 mm Hg; P=0.7267; 24-hour DBP: 0.85±0.61 mm Hg; P=0.1670), as were intervention effects on cfPWV (0.10±0.11 m/s; P=0.3553) and crPWV (0.03±0.12 m/s; P=0.7850). Changes in weight (−0.19±0.32 kg; P=0.5438) were also similar between randomized groups.
Table 3.
Blood Pressure, Pulse Wave Velocity, and Weight by Randomized Group
Associations of SB and Physical Activity With BP and PWV
Correlations between changes in SB and activity with changes in BP and PWV are presented in Table 4 (unadjusted) and Table S3 (partial correlations adjusted for baseline values). Few and inconsistent associations were observed across analyses (ie, overall or group-specific, unadjusted or adjusted), between changes in SB or activity with changes in SBP or changes in crPWV.
Table 4.
Correlations Between Sedentary Behavior and Physical Activity Changes and Blood Pressure/Pulse Wave Velocity Changes Among All Participants
Consistent with our hypotheses, among all participants, decreases in work-time SB were associated with decreases in DBP (r=0.15; P=0.0165). Similar associations were observed with changes in SB accumulated in bouts of ≥30 minutes during work time (r=0.14; P=0.0291). Although not observed in the intervention group, several associations in the hypothesized direction were observed between SB, standing, and steps or stepping and DBP in the control group (|r|=0.21–0.27; Table 4). Partial correlations had similar results in the expected direction among the full sample, with associations being apparent in the control but not the intervention group (Table S3).
SB and standing changes during work time were also associated with cfPWV change but in an unexpected direction. Reductions in work-time SB were associated with increases in cfPWV (r=−0.19; P=0.0059). Similar associations were observed with work-time SB accumulated in ≥30-minute bouts (r=−0.15; P=0.0322; Table 4). Increases in work-time standing were associated with increases in cfPWV (r=0.17; P=0.0111); however, increases in standing during nonwork time were associated with decreases in cfPWV (r=−0.14; P=0.0379), a finding in the expected direction (Table 4). These findings persisted in partial correlations: work-time SB r=−0.23 (P=0.0005), work-time standing r=0.22 (P=0.0010), and non–work-time standing r=−0.14 (P=0.0352), respectively (Table S3). Within randomized groups, associations with PWV largely remained similar even if not always statistically significant due to halving of the sample size (Table 4 and Table S3).
Adverse Events
Table S4 details adverse events that were reported by 32 (23.5%) intervention participants (42 events) and 24 (17.8%) control participants (30 events). Most events were unrelated to the study (61.9% intervention, 46.7% control). Serious adverse events were rare (4 in each arm) and none was related to the study. Some events (12 intervention, 15 control) were related to study assessment procedures, such as bruising during ambulatory BP monitoring or skin irritation from the activPAL. No events were definitely related to the intervention. Five (4 intervention, 1 control) were possibly related to the intervention, and included lower-extremity musculoskeletal pain (3), a torn knee ligament (1), and sciatica (1).
Sensitivity Analyses
Sensitivity analyses produced results not materially different from the main analyses. Repeating analyses with an alternative mixed modeling strategy, ignoring missing data, or using a last-observation-carried-forward approach resulted in nonsignificant intervention effects of similar magnitudes (Table S5). Subgroup analyses after stratification by age, sex, hypertension status, and body mass index generally revealed some variability in estimates, although all remained nonsignificant (Table S6). Intervention effects remained similar after excluding participants with protocol violations to the 3-month outcome assessment or who reported a change in their work environment or schedule (ie, due to COVID-19; Table S7).
DISCUSSION
The RESET-BP study reduced SB and increased standing by about 1 hour during the work day, but did not reduce BP or arterial stiffness among desk workers with untreated elevated or moderately high BP. Despite the lack of efficacy for improving BP or PWV, it is imperative that we document our findings from a rigorously conducted trial to inform future directions and facilitate meta-analyses without publication bias. We did confirm a secondary hypothesis whereby greater decreases in SB during work were favorably associated with decreases in resting DBP, although the strength of the association was mild, and similar favorable associations were not observed with resting SBP, cfPWV, or crPWV. Also, as hypothesized, increased standing during nonwork time was mildly associated with decreases in cfPWV. However, we identified unexpected associations where decreases in work-time SB and increases in work-time standing were associated with unfavorable increases in PWV. Taken together, our findings indicate that reducing SB by ≈1 hour each day and primarily through standing during work is not an effective nonpharmacologic strategy for reducing BP and improving arterial stiffness.
Strengths of the RESET-BP study include the RCT design and rigorous, published protocol,16 a sample indicated to receive lifestyle treatment for high BP, device-based quantification of SB and physical activity overall and during work and nonwork times, comprehensive measurements of resting and ambulatory BP along with PWV, high fidelity and retention, and robustness of findings across sensitivity analyses. Although our study was ongoing during the COVID-19 pandemic, our conclusions are strengthened by the randomized design, rigorous methods to maintain the integrity of our intervention and assessments during restrictions,16 and consistent findings across sensitivity analyses considering COVID-19–related protocol deviations.
Limitations of the study include external validity concerns, because the participants were all desk workers, ≤65 years old, and free of CVD; were not using medication to treat high BP or diabetes; reported low leisure-time MVPA at baseline; were mostly non-Hispanic White; and had a high education level. The average BP in our sample was only 129/83 mm Hg, which may have resulted in a floor effect, but sensitivity analyses restricted to those with BP ≥140/90 mm Hg did not suggest a statistically or clinically significant intervention effect. Furthermore, despite our inclusion criterion of low self-reported leisure-time MVPA, accelerometer-measured MVPA at baseline and follow-up was ~250 min/wk, which exceeds current recommendations.15 Although this remained stable over follow-up, this higher MVPA level may have blunted intervention effects.5 Device-based SB and activity monitoring only occurred at baseline and 3-month follow-up, and we did not institute minimum wear requirements for work and nonwork time estimates. Also, this report only includes the effect of the RESET-BP intervention on BP and PWV; SB-reduction interventions have been shown to improve other health outcomes, such as musculoskeletal health, mental health, adiposity, and glycemic control, in other trials.12–14,36
A limitation that merits elaboration is the intervention group not achieving the targeted increase in standing time of 2 to 4 h/d and 4 to 8 movement breaks per day. The targeted increases were based on the best available data at the time the intervention was designed: an expert statement20 that extrapolated to recommend desk-based workers accumulate at least 2 and up to 4 hours of standing and light activity during the work day. Notably, the intervention and control groups had 2.0 h/d of standing and stepping during work at baseline; at 3-month follow-up, the control group did not change, whereas the intervention group had increased time spent standing and stepping to 3.5 h/d during work. Therefore, intervention participants engaged in between 2 and 4 hours of standing and light activity during work, but did not increase by the targeted amount. This could be the reason that the intervention did not improve BP. On the basis of our follow-up analyses, the nonsignificant or mild associations between increases in standing and stepping and BP suggest that decreasing SB or increasing standing by 4 h/d would equate to only a small effect on BP (<2 mm Hg). Thus, although we cannot rule out that greater increases in standing and stepping may have reduced BP, we surmise that it is unlikely that achievement of our targeted SB reduction and standing increases in our population would have improved BP in a clinically meaningful way.
To our knowledge, RESET-BP is the first RCT to test an SB-reduction intervention in a clinical population with elevated or high BP and with comprehensive measurements of resting BP, ambulatory BP, and PWV. Nonetheless, several SB-reduction intervention trials in different participant groups have reported BP findings. A 2021 systematic review12 of 18 randomized trials summarized the effects of SB-reduction interventions on SB among various populations (eg, with overweight or obesity, diabetes, or CVD). Most studies were of 3-month duration, similar to RESET-BP. The pooled estimate of SB reduction was similar, at −1.07 h/d (P<0.001), but these studies averaged greater increases in time spent stepping (0.45 h/d; P<0.001) and lesser increases in standing (0.47 h/d; P=0.06). Across studies reporting effects on SBP (n=10) and DBP (n=9), the pooled estimates of changes were comparable to our findings, at −0.5 mm Hg (P=0.55) for SBP and −0.8 mm Hg (P=0.21) for DBP.
Also relevant for comparison are 2 cluster-randomized SB reduction trials in desk workers published while RESET-BP was being conducted. The Stand and Move trial13 (United States; n=630) and the SMART Work and Life trial14 (United Kingdom; n=756) recruited general office populations and studied a variety of outcomes over 12 months. Similar to RESET-BP, both studies achieved significant SB reductions (≈1 h/d) along with significant increases in standing among those who received sit–stand desks. Furthermore, although baseline BP was lower, because these studies did not require elevated or high BP for enrollment, null effects on BP were observed between intervention groups. Along with our findings within a sample required to have higher BP, designed with resting SBP as the primary outcome, and uniquely measuring ambulatory BP and PWV, we can reasonably conclude that reducing SB though primarily standing during work time is not an effective lifestyle treatment strategy for reducing BP.
Findings from our secondary aim were more varied. Among all participants, as hypothesized, reductions in work-time SB were mildly associated with reductions in resting DBP. Also consistent with our expectations, increasing light-intensity activity in the form of non–work-time standing was mildly associated with benefits to cfPWV. Our secondary aim also identified that changes in work-time behaviors (reduced SB and increased standing) were associated with unfavorable changes in PWV in the opposite of the hypothesized direction. This later finding may be explained by the postulated phenomenon whereby exposure to occupational standing (which is typically prolonged and static) may result in blood pooling and a similar cascade of negative cardiovascular effects that occur with prolonged sitting.37,38 Our recently published review summarized the mostly beneficial associations of all-day standing with mortality rates from general population cohorts in contrast to the sometimes harmful associations of occupational standing with subclinical cardiovascular outcomes, and postulated physiologic mechanisms that may underlie this paradox.37 Because decreasing SB by increasing standing has been recommended in some guidelines,39 and sit–stand desks are recommended and becoming more common in workplaces,20 careful investigations of different patterns and domains of standing as well as the use of sit–stand desks as they affect cardiovascular risk and other health outcomes is an area ripe for further research.38
RESET-BP has implications for the treatment of BP. Individuals seeking to decrease elevated or moderately high BP levels through lifestyle approaches should be prescribed established nonpharmacologic strategies, including increasing physical activity, improving diet, weight loss, and alcohol moderation40; however, replacing SB with mostly standing at work for ≈1 h/d was ineffective. Future research is needed to clarify whether interventions that elicit greater SB reductions and replace SB with activity other than work-time standing at a sit–stand desk are more beneficial to BP and cardiovascular health. Still, large SB decreases may be necessary to realize clinically meaningful benefits. At the same time, the possible negative effects of replacing sitting with high amounts of standing during work time on arterial stiffness should be rigorously evaluated to clarify workplace health and general physical activity guidelines.
CONCLUSION
The 3-month RESET-BP intervention decreased SB and increased standing by ≈1 hour during the work day but was not effective for reducing BP or arterial stiffness.
ARTICLE INFORMATION
Acknowledgments
The authors thank the participants, staff, and students who contributed to the RESET-BP study (Effect of Reducing Sedentary Behavior on Blood Pressure); and the RESET-BP Data Safety and Monitoring Board members Drs Patrick Sparto (chair), Sarah Stahl, Evan Ray, and Robert Boudreau.
Sources of Funding
This research was conducted with funding from the National Institutes of Health (grant R01 HL134809). Support for recruitment and other aspects of study administration was provided by the National Institutes of Health Clinical and Translational Science Award program (grant UL1 TR001857).
Disclosures
Dr Jakicic reports participation on a Scientific Advisory Board for Wondr Health, Inc; ownership of Healthy Lifestyles Consulting, LLC; and a role as principal investigator of the clinical site for a research study awarded to the University of Kansas Medical Center by Epitomee Medical, Inc. The other authors report no relevant conflicts of interest.
Supplemental Material
Tables S1–S7
Figure S1
Supplementary Material
Nonstandard Abbreviations and Acronyms
- BP
- blood pressure
- cfPWV
- carotid-femoral pulse wave velocity
- CONSORT
- Consolidated Standards of Reporting Trials
- crPWV
- carotid-radial pulse wave velocity
- CVD
- cardiovascular disease
- DBP
- diastolic blood pressure
- MVPA
- moderate to vigorous intensity physical activity
- PWV
- pulse wave velocity
- RCT
- randomized clinical trial
- RESET-BP
- Effect of Reducing Sedentary Behavior on Blood Pressure
- SB
- sedentary behavior
- SBP
- systolic blood pressure
Circulation is available at www.ahajournals.org/journal/circ
This article is part of the Null Hypothesis Collection, a collaborative effort among the CBMRT, AHA Journals, and Wolters Kluwer, and has been made freely available through funds provided by the CBMRT. For more information, visit https://www.ahajournals.org/null-hypothesis.
Supplemental Material is available at https://www.ahajournals.org/doi/suppl/10.1161/CIRCULATIONAHA.123.068564.
Continuing medical education (CME) credit is available for this article. Go to http://cme.ahajournals.org to take the quiz.
For Sources of Funding and Disclosures, see page 1426.
Contributor Information
Subashan Perera, Email: ksp9@pitt.edu.
Kimberly A. Huber, Email: kah31@pitt.edu.
Molly B. Conroy, Email: Molly.Conroy@hsc.utah.edu.
John M. Jakicic, Email: jjakicic@kumc.edu.
Matthew F. Muldoon, Email: mfm10@pitt.edu.
REFERENCES
- 1.Zhou B, Perel P, Mensah GA, Ezzati M. Global epidemiology, health burden and effective interventions for elevated blood pressure and hypertension. Nat Rev Cardiol. 2021;18:785–802. doi: 10.1038/s41569-021-00559-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Whelton PK, Carey RM, Aronow WS, Casey DE, Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Hypertension. 2018;71:e13–e115. doi: 10.1161/HYP.0000000000000065 [DOI] [PubMed] [Google Scholar]
- 3.Muntner P, Miles MA, Jaeger BC, Hannon L, III, Hardy ST, Ostchega Y, Wozniak G, Schwartz JE. Blood pressure control among US adults, 2009 to 2012 through 2017 to 2020. Hypertension. 2022;79:1971–1980. doi: 10.1161/HYPERTENSIONAHA.122.19222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tremblay MS, Aubert S, Barnes JD, Saunders TJ, Carson V, Latimer-Cheung AE, Chastin SFM, Altenburg TM, Chinapaw MJM; SBRN Terminology Consensus Project Participants. Sedentary Behavior Research Network (SBRN)–terminology consensus project process and outcome. Int J Behav Nutr Phys Act. 2017;14:75. doi: 10.1186/s12966-017-0525-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Katzmarzyk PT, Powell KE, Jakicic JM, Troiano RP, Piercy K, Tennant B; 2018 Physical Activity Guidelines Advisory Committee. Sedentary behavior and health: update from the 2018 physical activity guidelines advisory committee. Med Sci Sports Exerc. 2019;51:1227–1241. doi: 10.1249/MSS.0000000000001935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ekelund U, Tarp J, Fagerland MW, Johannessen JS, Hansen BH, Jefferis BJ, Whincup PH, Diaz KM, Hooker S, Howard VJ, et al. Joint associations of accelerometer-measured physical activity and sedentary time with all-cause mortality: a harmonised meta-analysis in more than 44 000 middle-aged and older individuals. Br J Sports Med. 2020;54:1499–1506. doi: 10.1136/bjsports-2020-103270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Diaz KM, Howard VJ, Hutto B, Colabianchi N, Vena JE, Safford MM, Blair SN, Hooker SP. Patterns of sedentary behavior and mortality in U.S. middle-aged and older adults. Ann Intern Med. 2017;167:465–475. doi: 10.7326/M17-0212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bellettiere J, LaMonte MJ, Evenson KR, Rillamas-Sun E, Kerr J, Lee I-M, Di C, Rosenberg DE, Stefanick ML, Buchner DM, et al. Sedentary behavior and cardiovascular disease in older women. Circulation. 2019;139:1036–1046. doi: 10.1161/CIRCULATIONAHA.118.035312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barone Gibbs B, Kowalsky RJ, Perdomo SJ, Taormina JM, Balzer JR, Jakicic JM. Effect of alternating standing and sitting on blood pressure and pulse wave velocity during a simulated workday in adults with overweight/obesity. J Hypertens. 2017;35:2411–2418. doi: 10.1097/HJH.0000000000001463 [DOI] [PubMed] [Google Scholar]
- 10.Dempsey PC, Larsen RN, Dunstan DW, Owen N, Kingwell BA. Sitting less and moving more: implications for hypertension. Hypertension. 2018;72:1037–1046. doi: 10.1161/HYPERTENSIONAHA.118.11190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Paterson C, Fryer S, Zieff G, Stone K, Credeur DP, Barone Gibbs B, Padilla J, Parker JK, Stoner L. The effects of acute exposure to prolonged sitting, with and without interruption, on vascular function among adults: a meta-analysis. Sports Med. 2020;50:1929–1942. doi: 10.1007/s40279-020-01325-5 [DOI] [PubMed] [Google Scholar]
- 12.Nieste I, Franssen WMA, Spaas J, Bruckers L, Savelberg HHCM, Eijnde BO. Lifestyle interventions to reduce sedentary behaviour in clinical populations: a systematic review and meta-analysis of different strategies and effects on cardiometabolic health. Prev Med. 2021;148:106593. doi: 10.1016/j.ypmed.2021.106593 [DOI] [PubMed] [Google Scholar]
- 13.Pereira MA, Mullane SL, Toledo MJL, Larouche ML, Rydell SA, Vuong B, Feltes LH, Mitchell NR, de Brito JN, Hasanaj K, et al. Efficacy of the ‘Stand and Move at Work’ multicomponent workplace intervention to reduce sedentary time and improve cardiometabolic risk: a group randomized clinical trial. Int J Behav Nutr Phys Act. 2020;17:133. doi: 10.1186/s12966-020-01033-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Edwardson CL, Biddle SJH, Clemes SA, Davies MJ, Dunstan DW, Eborall H, Granat MH, Gray LJ, Healy GN, Jaicim NB, et al. Effectiveness of an intervention for reducing sitting time and improving health in office workers: three arm cluster randomised controlled trial. BMJ. 2022;378:e069288. doi: 10.1136/bmj-2021-069288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, George SM, Olson RD. The physical activity guidelines for Americans. JAMA. 2018;320:2020–2028. doi: 10.1001/jama.2018.14854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Barone Gibbs B, Conroy MB, Huber K, Muldoon MF, Perera S, Jakicic JM. Effect of Reducing Sedentary Behavior on Blood Pressure (RESET-BP): rationale, design, and methods. Contemp Clin Trials. 2021;106:106428. doi: 10.1016/j.cct.2021.106428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bandura A. The primacy of self-regulation in health promotion. Appl Psychol. 2005;54:245–254. doi: 10.1111/j.1464-0597.2005.00208.x [Google Scholar]
- 18.Owen N, Sugiyama T, Eakin EE, Gardiner PA, Tremblay MS, Sallis JF. Adults’ sedentary behavior: determinants and interventions. Am J Prev Med. 2011;41:189–196. doi: 10.1016/j.amepre.2011.05.013 [DOI] [PubMed] [Google Scholar]
- 19.Ryan RM, Deci EL. Self-determination theory and the facilitation of intrinsic motivation, social development, and well-being. Am Psychol. 2000;55:68–78. doi: 10.1037//0003-066x.55.1.68 [DOI] [PubMed] [Google Scholar]
- 20.Buckley JP, Hedge A, Yates T, Copeland RJ, Loosemore M, Hamer M, Bradley G, Dunstan DW. The sedentary office: an expert statement on the growing case for change towards better health and productivity. Br J Sports Med. 2015;49:1357–1362. doi: 10.1136/bjsports-2015-094618 [DOI] [PubMed] [Google Scholar]
- 21.Muntner P, Shimbo D, Carey RM, Charleston JB, Gaillard T, Misra S, Myers MG, Ogedegbe G, Schwartz JE, Townsend RR, et al. Measurement of blood pressure in humans: a scientific statement from the American Heart Association. Hypertension. 2019;73:e35–e66. doi: 10.1161/HYP.0000000000000087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.El Assaad MA, Topouchian JA, Darné BM, Asmar RG. Validation of the Omron HEM-907 device for blood pressure measurement. Blood Press Monit. 2002;7:237–241. doi: 10.1097/00126097-200208000-00006 [DOI] [PubMed] [Google Scholar]
- 23.O’Brien E, Asmar R, Beilin L, Imai Y, Mallion J-M, Mancia G, Mengden T, Myers M, Padfield P, Palatini P, et al. European Society of Hypertension recommendations for conventional, ambulatory and home blood pressure measurement. J Hypertens. 2003;21:821–848. doi: 10.1097/00004872-200305000-00001 [DOI] [PubMed] [Google Scholar]
- 24.Barone Gibbs B, Muldoon MF, Conroy MB, Paley JL, Shimbo D, Perera S. Influence of recent standing, moving, or sitting on daytime ambulatory blood pressure. J Am Heart Assoc. 2023;12:e029999. doi: 10.1161/JAHA.123.029999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D, Pannier B, Vlachopoulos C, Wilkinson I, Struijker-Boudier H; European Network for Non-invasive Investigation of Large Arteries. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J. 2006;27:2588–2605. doi: 10.1093/eurheartj/ehl254 [DOI] [PubMed] [Google Scholar]
- 26.Gitlin LN, Parisi JM. Are treatment effects real? The role of fidelity. In: Gitlin LN, Czaja SJ, eds. Behavioral Intervention Research: Designing, Evaluating and Implementing. Springer: 2016;213–239. [Google Scholar]
- 27.Edwardson CL, Winkler EAH, Bodicoat DH, Yates T, Davies MJ, Dunstan DW, Healy GN. Considerations when using the activPAL monitor in field-based research with adult populations. J Sport Health Sci. 2017;6:162–178. doi: 10.1016/j.jshs.2016.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sasaki JE, John D, Freedson PS. Validation and comparison of ActiGraph activity monitors. J Sci Med Sport. 2011;14:411–416. doi: 10.1016/j.jsams.2011.04.003 [DOI] [PubMed] [Google Scholar]
- 29.Barone Gibbs B, Kline CE. When does sedentary behavior become sleep? A proposed framework for classifying activity during sleep-wake transitions. Int J Behav Nutr Phys Act. 2018;15:81. doi: 10.1186/s12966-018-0712-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Choi L, Liu Z, Matthews CE, Buchowski MS. Validation of accelerometer wear and nonwear time classification algorithm. Med Sci Sports Exerc. 2011;43:357–364. doi: 10.1249/MSS.0b013e3181ed61a3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Matthews CE, Hagströmer M, Pober DM, Bowles HR. Best practices for using physical activity monitors in population-based research. Med Sci Sports Exerc. 2012;44:S68–S76. doi: 10.1249/MSS.0b013e3182399e5b [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paffenbarger RS, Wing AL, Hyde RT. Physical activity as an index of heart attack risk in college alumni. Am J Epidemiol. 1978;108:161–175. doi: 10.1093/oxfordjournals.aje.a112608 [DOI] [PubMed] [Google Scholar]
- 33.Barone Gibbs B, Brach JS, Byard T, Creasy S, Davis KK, McCoy S, Peluso A, Rogers RJ, Rupp K, Jakicic JM. Reducing sedentary behavior versus increasing moderate-to-vigorous intensity physical activity in older adults. J Aging Health. 2017;29:247–267. doi: 10.1177/0898264316635564 [DOI] [PubMed] [Google Scholar]
- 34.Kozey Keadle S, Lyden K, Staudenmayer J, Hickey A, Viskochil R, Braun B, Freedson PS. The independent and combined effects of exercise training and reducing sedentary behavior on cardiometabolic risk factors. Appl Physiol Nutr Metab. 2014;39:770–780. doi: 10.1139/apnm-2013-0379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Little RJ, D’Agostino R, Cohen ML, Dickersin K, Emerson SS, Farrar JT, Frangakis C, Hogan JW, Molenberghs G, Murphy SA, et al. The prevention and treatment of missing data in clinical trials. N Engl J Med. 2012;367:1355–1360. doi: 10.1056/NEJMsr1203730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Barone Gibbs B, Hergenroeder AL, Perdomo SJ, Kowalsky RJ, Delitto A, Jakicic JM. Reducing sedentary behaviour to decrease chronic low back pain: the stand back randomised trial. Occup Environ Med. 2018;75:321–327. doi: 10.1136/oemed-2017-104732 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gibbs BB, Diaz KM, Kowalsky RJ, Smith PM, Stoner L. Association of standing with cardiovascular disease and mortality in adults. Curr Epidemiol Rep. 2021;8:200–211. doi: 10.1007/s40471-021-00276-3 [Google Scholar]
- 38.Kowalsky RJ, Stoner L, Faghy MA, Barone Gibbs B. A call to clarify the intensity and classification of standing behavior. Int J Environ Res Public Health. 2021;18:8460. doi: 10.3390/ijerph18168460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ross R, Chaput J-P, Giangregorio LM, Janssen I, Saunders TJ, Kho ME, Poitras VJ, Tomasone JR, El-Kotob R, McLaughlin EC, et al. Canadian 24-hour movement guidelines for adults aged 18–64 years and adults aged 65 years or older: an integration of physical activity, sedentary behaviour, and sleep. Appl Physiol Nutr Metab. 2020;45:S57–S102. doi: 10.1139/apnm-2020-0467 [DOI] [PubMed] [Google Scholar]
- 40.Barone Gibbs B, Hivert M-F, Jerome GJ, Kraus WE, Rosenkranz SK, Schorr EN, Spartano NL, Lobelo F; American Heart Association Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular and Stroke Nursing; and Council on Clinical Cardiology. Physical activity as a critical component of first-line treatment for elevated blood pressure or cholesterol: who, what, and how? A scientific statement from the American Heart Association. Hypertension. 2021;78:e26–e37. doi: 10.1161/HYP.0000000000000196 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.