ABSTRACT
Background:
Herpes simplex encephalitis (HSE), caused by herpes simplex virus (HSV) is the most common cause of sporadic encephalitis that often presents as an emergency case of acute or sub-acute nature associated with poor prognosis. Early suspicion and prompt diagnostic testing with adequate antiviral therapy can only reduce morbidity and mortality associated with the disease. This study aims to evaluate the role of serological and molecular diagnosis of encephalitis caused by HSV 1 and 2 for timely detection of the disease.
Methods:
This cross-sectional study was conducted at the Department of Microbiology, Rajendra Institute of Medical Science, Ranchi, from April 2021 to September 2022. Cerebrospinal fluid (CSF) samples were collected and processed for the detection of HSV DNA by polymerase chain reaction (PCR) as well as serum samples were collected from those patients and were processed for the detection of IgM by ELISA.
Results:
A total of 188 CSF samples were collected, out of which 4 HSV-1 and 1 HSV-2 samples were found to be positive by conventional PCR. 36 samples were positive for HSV-1 and 2 IgM ELISA. 26.06% of cases belonged to patients between 0 and 10 years of age followed by 17.06% in 51-60 years age groups.
Conclusion:
PCR is the optimal diagnostic test, but the sample must be collected in the early stages of the disease. Diagnosis by PCR assay of CSF samples targeting HSV DNA is the best confirmatory test at hand for early detection and also helps in estimating the incidence of herpes simplex virus encephalitis.
Keywords: CSF, herpes simplex encephalitis, HSV-1, HSV-2, IgM ELISA, PCR
Introduction
Herpes simplex virus (HSV) is a human herpes virus that causes HSV encephalitis (HSE), which is one of the most common sporadic fatal encephalitis in humans. Despite the availability of antiviral treatment, HSE in the pediatric age group is associated with a high death rate. HSE presents as an acute or subacute disease that can manifest as focal or global cerebral dysfunction caused by either HSV-1 (type 1) or HSV-2 (type 2). Signs and symptoms, such as an altered mental state, fever, vomiting, and neurological deficits, should be investigated for a possible diagnosis of HSE.[1] HSV-1 is responsible for major number of herpes simplex encephalitis cases whereas HSV-2 accounts for less than 10%. HSV-1 is also the most common cause of herpes encephalitis during the post-neonatal period, leading to fatal encephalitis, causing sporadic and non-seasonal patterns. On the other hand, HSV-2 is more common in neonates and immunocompromised patients.[2]
In India, data specific to HSV encephalitis are limited, but studies suggest that HSV accounts for 10-20% of viral encephalitis cases in tertiary care centers. The infection affects individuals of all ages, though it is more prevalent in infants and the elderly. Immunocompromised individuals and those with limited access to healthcare are at higher risk. Unlike some infections, HSV encephalitis does not exhibit a significant seasonal trend. India faces significant diagnostic challenges due to limited access to advanced tests like polymerase chain reaction (PCR) for HSV DNA in cerebrospinal fluid (CSF), leading to underdiagnosis. The untreated mortality rate for HSV encephalitis is up to 70%, which can be reduced to 20-30% with timely antiviral treatment. Public health efforts in India should focus on improving diagnostic capabilities, raising awareness among healthcare providers, enhancing surveillance systems, and potentially developing vaccination programs to reduce the disease burden.
DNA detection by PCR of CSF is the test of choice for confirming the diagnosis of HSV.[3] Diagnosis can be confirmed by the HSV PCR test or rarely via the brain biopsy method. Usually, CSF glucose level is normal or slightly low and in 5-10% of patients, especially in children, other profiles could also initially be normal. CSF analysis is often normal in the early phase of the disease in immunocompromised patients.[4] HSV genes like UL23, UL30, and UL5 are increasingly of great interest because these encode for thymidine kinase, DNA polymerase, and helicase respectively and also help in identifying organism through PCR.[5] PCR for HSV-1 and HSV-2 has overshadowed other detection methods as the test of choice, provided CSF should be used which has high sensitivity (96%) and specificity (99%).[6] False-negative PCR occurs early in the illness and if the clinical suspicion is still high, empirical treatment could be started, and repeat CSF HSV PCR should be obtained within 3–7 days.[7]
Immunological assays have proven to be an excellent marker for HSV infection. Enzyme-linked immunosorbent assays (ELISA) is preferred for effective detection of antigens (HSV glycoproteins) or antibodies specific to HSV glycol-proteins. ELISA is easier to perform, generates fast results and is compatible with every kind of laboratory setup, which makes it the standard serological method. HSV-1 and HSV-2-specific IgG antibodies are detected by ELISA.[8,9] However, ELISA is unable to detect HSV-2 antibodies in HSV-1 seropositive patients, which can turn the course of the disease towards a worse outcome.[10] Our study was performed to analyze the diagnostic ability of serological and molecular tests.
Methods
This was a cross sectional study done at the Department of Microbiology, Rajendra Institute of Medical Science, Ranchi, from April 2021 to September 2022. Serum and CSF samples were collected from 188 clinically suspected cases of encephalitis, presenting to our hospital. Biochemical analysis of CSF for encephalitis was also performed. Clinically suspected cases with HSV 1 and 2 were detected by IgM ELISA kit (Calbiotech, USA) from serum samples.[11] Following this molecular detection of HSV 1 and 2 were done by molecular method from CSF specimen of all patients. DNA was extracted by using QI Amp DNA Mini Kit (QIAGEN, Germany). Both forward and reverse primers[12] for HSV 1 and 2 used were as follows: HSV-1F: ATGGTGAACATCGACATGTACGG. HSV-1R: CCTCGCGTTCGTCCTCGTCCTCC, (UL-30, 469bp); HSV-2F: ATGGTGAACATCGACATGTACGG, HSV-2R: CCTCCTTGTCGAGGCCCCAAAC (UL-30, 391bp), Blood and CSF biochemical analysis (Glucose, protein) for HSV (1 and 2) were also performed. CSF was analyzed for mild to moderately elevated protein level, glucose level, pleocytosis and mononuclear infiltrate, which are important markers of viral encephalitis. The DNA fragments of HSV-1 and 2 for UL-30 were amplified by PCR thermocycler. Amplicon of 469bp for UL-30 gene of HSV-1 and amplicon of 391bp for UL-30 gene for HSV-2 was considered positive.
Results
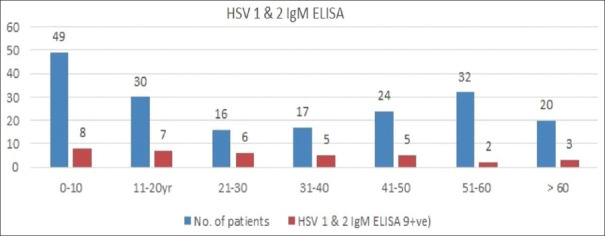
In our study, out of 188 total patients, 26.06% were of age group 0-10 years that were most commonly affected by encephalitis. Maximum numbers of HSV cases were seen in the age group below 20 years and above 50 years [Figure 1]. Male-to-female ratio was 1.4:1, with male 109 (57.97%) and 79 (42.03%) females. The number of patients from urban areas was 64 (34.05%) as compared to 124 (65.95%) belonging to rural areas. Encephalitis cases with an acute onset (119 patients; 63.30%) were more commonly noted than sub-acute presentation (69 patients; 36.70%) in this study. Clinical manifestation of encephalitis was shown in Table 1. One hundred and fifty-five patients (82.44%) were having normal CSF glucose level as compared to 17.56% were having raised glucose values. Also, 63.29% were having normal CSF protein values whereas 36.71% were having abnormally higher protein values.
Figure 1.
Sero-prevalence of HSV 1 and 2 based on age distribution
Table 1.
Clinical manifestation of encephalitis in suspected cases
| Clinical features | Percentage | P | Degree of freedom | Chi-squared |
|---|---|---|---|---|
| Fever | 87.76 | <0.05 | 11 | 1657.776 |
| Headache | 78.19 | <0.05 | 14 | 1705.133 |
| Seizure | 64.36 | <0.05 | 15 | 1588.447 |
| Altered sensorium | 50.53 | <0.05 | 17 | 1538.838 |
| Irritability | 45.21 | <0.05 | 17 | 1516.169 |
| Blister | 09.57 | <0.05 | 17 | 1308.515 |
| Neck stiffness | 7.23 | <0.05 | 18 | 1522.394 |
| Vomiting | 17.55 | <0.05 | 18 | 1318.271 |
| Hypotension | 31.91 | <0.05 | 18 | 1459.145 |
Table 2 shows comparison between PCR (HSV-1, UL-30 gene) and HSV 1 and 2 IgM ELISA.
Table 2.
Comparison between PCR (HSV-1, UL-30 gene) and IgM ELISA (HSV 1&2)
| HSV 1&2 IgM ELISA | PCR (HSV-1, UL-30 gene) | SEN (%) | SPE. (%) | PPV (%) | NPV (%) | Accuracy (%) | P | ||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| + | - | Total | |||||||
| + | 4 | 32 | 36 | 100 | 82.60 | 11.11 | 100 | 82.97 | 0.001 |
| - | 0 | 152 | 152 | ||||||
| Total | 4 | 184 | 188 | ||||||
From this study, when comparing HSV-1 (UL-30 gene) with results of HSV-1 and 2 (IgM ELISA), the relative risk was 37.216 and 95% CI was 2.0485-676.13 and P value was 0.0145 and odd ratio was 42.2308.
Out of 188 patients, HSV genes were found in 2.65% by molecular method, out of which HSV1 was found in 4 (80%) and 1 (20%) HSV2.
Table 3 compares HSV-2 (UL-30 gene) with the result of HSV-1 and 2 (IgM ELISA). The relative risk was 12.4054 and 95% CI was 0.5156-298.4472, the P value was 0.039, and the odds ratio was 12.8873.
Table 3.
Comparison between PCR (HSV-2, UL-30 gene) and HSV 1&2, IgM ELISA
| HSV 1&2 IgM ELISA | PCR (HSV-2, UL-30 gene) | SEN. (%) | SPE. (%) | PPV (%) | NPV (%) | Accuracy (%) | P | ||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| + | - | Total | |||||||
| + | 1 | 35 | 36 | 100 | 81.28 | 2.77 | 100 | 81.38 | 0.039 |
| - | 0 | 152 | 152 | ||||||
| Total | 1 | 187 | 188 | ||||||
Discussion
HSE is a potentially fatal disease with wide array of clinical scenario from asymptomatic cases to severe necrotising encephalitis. Diagnosis of HSV has proven to be a huge obstacle for clinical physicians leading to high mortality as the patient is deprived of correct antiviral treatment. Our study was carried out with the intention of analysing the performance of PCR assay, the gold standard and confirmatory test targeting HBV DNA, in diagnosis of suspected HSE cases.
In our study, the majority of the patients presenting were of extremes of age with below 20 years accounting for 42% and above 50 years, accounting for 27%, which is comparable to other study by Whitley RJ.[13] The male-to-female ratio was found to be 1.4:1. Although the disease was present throughout the year, the maximum cases were reported during the months of summer. A male preponderance with a similar seasonal pattern has been reported in a study from North West India by Panagariya et al.[14]
The majority of the patients had acute onset of the disease, that is, 63.30%. A report by Suraj Singh et al.[15] mentions that subacute and chronic onset is extremely rare.
In our study, fever was the predominant clinical feature, which accounted for 87.76%. Other signs and symptoms suggestive of HSE that are noted in our study were presence of fever, headache, behavioral changes, confusion, focal neurologic findings, and abnormal cerebrospinal. A similar pattern of clinical presentation has been reported by Sili et al.[16] in a 10-year-long retrospective and another published by Bradshaw et al.[17]
The percentage of patients having normal glucose levels was 82.44%.CSF parameters were used to screen specimens before PCR analysis for HSV DNA detection as indicated in a study by Tang et al.[18]
IgM ELISA for HSV-1 and 2 was found to be positive in sera of 36 patients. There is a lack of data regarding serological assays performed on the sera of HSE patients; therefore, we could not strongly correlate our findings with other studies instead we continued with our novel findings. Although, one review article by Bradshaw et al reports the seropositivity of HSV-1 to be 60–90%. By the time a serological diagnosis can be made, the patient’s condition would be critical already.
HSV DNA was detected in five patients by conventional PCR. Out of the 5 PCR positive CSF samples, HSV-1 gene was detected in 4 samples and HSV-2 gene in 1 sample. Shalchi et al.[19] reports HSV-1 to be the cause in 90% of cases of HSE. PCR positivity hugely relies on early sample collection and processing. The golden period for diagnosis of PCR is the initial phase of infection but as the infection remains asymptomatic in the early stages, patients do not seek diagnosis. Suboptimal handling of the precious CSF sample further reduces the chances of DNA recovery. This scenario contributes to low sensitivity.
HSE is a critical condition requiring prompt diagnosis and treatment to reduce mortality and morbidity. Family physicians play a crucial role in early recognition, referral, and management of HSE. In Jharkhand, where advanced medical facilities may be limited, they bridge the gap by providing holistic care, educating patients about HSV, and advocating for improved diagnostic facilities. Managing comorbidities and ensuring long-term follow-up and rehabilitation in primary care settings are also vital, enhancing patient outcomes and public health.
Conclusion
PCR is the optimal diagnostic test, but the sample must be collected in the early stages of the disease. Prompt work-up and management are required in suspected HSE as delay in treatment is the single most significant factor for high mortality and morbidity. Routine laboratory parameters are insufficient in arriving at timely detection. The diagnosis of HSV is demanding and often missed on initial examination. PCR has proven to be tremendously beneficial in reaching swift and accurate diagnosis. HSV encephalitis still remains a dilemma for clinicians and microbiologists. Our study has found that PCR of CSF samples for the detection of HSV DNA in suspected cases of herpes simplex encephalitis will substantially help in making the correct diagnosis.
Ethical approval
Ethical approval had been obtained with IEC 363- dated on 15-09-2021.
Limitation
This study has several limitations. The sample size of 188 patients may not fully represent the broader population, limiting generalizability. Conducted at a single tertiary care center, the study may introduce selection bias. PCR and ELISA, while sensitive and specific, might miss early-phase cases due to low viral loads. Lack of long-term follow-up prevents insights into chronic impacts and treatment effectiveness. Additionally, insufficient demographic data, including socio-economic status and detailed medical histories, limit control for potential confounding factors. Future studies should address these issues by including larger, multi-center cohorts, longitudinal follow-up, and more comprehensive diagnostic and demographic data.
Conflicts of interest
There are no conflicts of interest.
Funding Statement
Nil
References
- 1.Tang YW, Mitchell PS, Espy MJ, Smith TF, Persing DH. Molecular diagnosis of herpes simplex virus infections in the central nervous system. J Clin Microbiol. 1999;37:2127–36. doi: 10.1128/jcm.37.7.2127-2136.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Le Doare K, Menson E, Patel D, Lim M, Lyall H, Herberg J. Fifteen minute consultation: Managing neonatal and childhood herpes encephalitis. Arch Dis Child Educ Pract Ed. 2015;100:58–63. doi: 10.1136/archdischild-2014-306321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Venkatesan A, Tunkel AR, Bloch KC, Lauring AS, Sejvar J, Bitnun A, et al. Case definitions, diagnostic algorithms, and priorities in encephalitis: Consensus statement of the international encephalitis consortium. Clin Infect Dis. 2013;57:1114–28. doi: 10.1093/cid/cit458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mook-Kanamori B, van de Beek D, Wijdicks EF. Herpes simplex encephalitis with normal initial cerebrospinal fluid examination. J Am Geriatr Soc. 2009;57:1514–5. doi: 10.1111/j.1532-5415.2009.02356.x. [DOI] [PubMed] [Google Scholar]
- 5.Lakeman FD, Whitley RJ. Diagnosis of herpes simplex encephalitis: Application of polymerase chain reaction to cerebrospinal fluid from brain-biopsied patients and correlation with disease. J Infect Dis. 1995;171:857–63. doi: 10.1093/infdis/171.4.857. [DOI] [PubMed] [Google Scholar]
- 6.Steiner I, Schmutzhard E, Sellner J, Chaudhuri A, Kennedy PG, European Federation of Neurological Sciences et al. EFNS-ENS guidelines for the use of PCR technology for the diagnosis of infections of the nervous system. Eur J Neurol. 2012;19:1278–91. doi: 10.1111/j.1468-1331.2012.03808.x. [DOI] [PubMed] [Google Scholar]
- 7.Tunkel AR, Glaser CA, Bloch KC, Sejvar JJ, Marra CM, Roos KL, et al. The management of encephalitis: Clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2008;47:303–27. doi: 10.1086/589747. [DOI] [PubMed] [Google Scholar]
- 8.Katz D, Hilliard JK, Mirkovic RR, Word RA. ELISA for detection of IgG and IgM antibodies to HSV-1 and HSV-2 in human sera. J Virol Methods. 1986;14:43–55. doi: 10.1016/0166-0934(86)90006-6. [DOI] [PubMed] [Google Scholar]
- 9.Kimmel N, Friedman MG, Sarov I. Enzyme-linked immunosorbent assay (ELISA) for detection of herpes simplex virus-specific IgM antibodies. J Virol Methods. 1982;4:219–27. doi: 10.1016/0166-0934(82)90068-4. [DOI] [PubMed] [Google Scholar]
- 10.Ashley RL. Sorting out the new HSV type specific antibody tests. Sex Transm Infect. 2001;77:232–7. doi: 10.1136/sti.77.4.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Langeland N, Haarr L, Mhalu F. Prevalence of HSV-2 antibodies among STD clinic patients in Tanzania. Int J STD AIDS. 1998;9:104–7. doi: 10.1258/0956462981921765. [DOI] [PubMed] [Google Scholar]
- 12.Dhull D, Sharma V, Sharma Y, Kaushik S. Applicability of molecular assays for detection and typing of herpes simplex viruses in encephalitis cases. Virusdisease. 2019;30:504–10. doi: 10.1007/s13337-019-00558-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Whitley RJ, Gnann JW. Viral encephalitis: Familiar infections andemerging pathogens. Lancet. 2002;359:507–14. doi: 10.1016/S0140-6736(02)07681-X. [DOI] [PubMed] [Google Scholar]
- 14.Panagariya A, Jain RS, Gupta S, Garg A, Sureka RK, Mathur V. Herpes simplex encephalitis in North West India. Neurol India. 2001;49:360–5. [PubMed] [Google Scholar]
- 15.Suraj Singh TH, Singh K, Wairokpam T. Subacute presentation of herpes simplex virus-1 encephalitis: A rare case report. J Med Soc. 2018;32:144–6. [Google Scholar]
- 16.Sili U, Kaya A, Mert A HSV Encephalitis Study Group. Herpes simplex virus encephalitis: Clinical manifestations, diagnosis and outcome in 106 adult patients. J Clin Virol. 2014;60:112–8. doi: 10.1016/j.jcv.2014.03.010. [DOI] [PubMed] [Google Scholar]
- 17.Bradshaw MJ, Venkatesan A. Herpes simplex virus-1 encephalitis in adults: Pathophysiology, diagnosis, and management. Neurotherapeutics. 2016;13:493–508. doi: 10.1007/s13311-016-0433-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tang YW, Hibbs JR, Tau KR, Qian Q, Skarhus HA, Smith TF, et al. Effective use of polymerase chain reaction for diagnosis of central nervous system infections. Clin Infect Dis. 1999;29:803–6. doi: 10.1086/520439. [DOI] [PubMed] [Google Scholar]
- 19.Shalchi Z, Bennett A, Hargroves D, Nash J. Diagnostic delay in a case of herpes simplex encephalitis. BMJ Case Rep. 2009;2009 doi: 10.1136/bcr.12.2008.1350. bcr12.2008.1350. [DOI] [PMC free article] [PubMed] [Google Scholar]