Abstract
Gene expression of Plasmodium falciparum (Pf) liver-stage (LS) parasites has remained poorly characterized, although they are major vaccine and drug targets. Using a human liver-chimaeric mouse model and a fluorescent parasite line (PfNF54CSPGFP), we isolated PfLS and performed transcriptomics on key LS developmental phases. We linked clustered gene expression to ApiAP2, a major family of transcription factors that regulate the parasite life cycle. This provided insights into transcriptional regulation of LS infection and expression of essential LS metabolic and biosynthetic pathways. We observed expression of antigenically variant PfEMP1 proteins and the major Pf protein export machine PTEX and identified protein candidates that might be exported by LS parasites. Comparing Pf and P. vivax LS transcriptomes, we uncovered differences in their expression of sexual commitment factors. This data will aid LS research and vaccine and drug target identification for prevention of malaria infection.
Subject terms: Malaria, Parasite genomics, Transcriptomics
Transcriptomics of gene expression in Plasmodium falciparum liver-stage parasites reveals transcriptional regulation, metabolic pathways and antigen expression, facilitating vaccine and drug target identification.
Main
Plasmodium falciparum (Pf) parasites cause the most devastating form of human malaria and are responsible for the majority of clinical cases and deaths, followed by Plasmodium vivax (Pv) parasites in terms of health impact1. Extensive malaria control and treatment efforts have significantly reduced malaria morbidity and mortality in the past two decades. However, this decline has stagnated over the past 7 years2,3 and malaria is rising again with 241 million cases and 619,000 deaths annually in 20211. This necessitates the search for new interventions based on a better understanding of malaria parasite biology.
Plasmodium sporozoites, deposited into the skin of human hosts by Anopheles mosquito bites, invade hepatocytes and develop into exo-erythrocytic schizonts4, which release merozoites, initiating the symptomatic blood stage (BS) infection of the parasite life cycle. The asymptomatic LS infection is considered a promising target for malaria vaccine development and drug prophylaxis. Relatively low numbers of sporozoites are transmitted by mosquitoes and only a fraction of them successfully infect the liver and undergo complete LS development5. Furthermore, complete LS elimination prevents the onset of BS infection and onward sexual-stage parasite transmission6–8. The two licensed malaria vaccines RTS/S and R21 aim to prevent liver infection but do so imperfectly and thus show only moderate efficacy9. However, vaccination with live-attenuated malaria parasites has demonstrated that complete protection from infection is achievable10–14. Lastly, drugs targeting LS would be less prone to the development of drug resistance compared with drugs targeting the BS and would provide protection from infection15.
Yet, the gene expression landscape of PfLS remains largely uncharacterized due to technical challenges. Pf sporozoites have an almost exclusive tropism for primary human hepatocytes, infection rates are low in cultured hepatocytes, and abnormal LS development is common in human hepatoma cell lines16,17. Studies of Pf and PvLS biology advanced with improved in vitro primary human hepatocyte culture systems18–20 and the human liver-chimaeric mouse models21,22, the latter of which enable in vivo LS studies. One of these models are FRG NOD mice, transplanted with primary human hepatocytes (FRG huHep mice). These mice are highly susceptible to infection with Pf and Pv sporozoites and support full LS development and parasite transition to BS infection when they are additionally engrafted with human red blood cells (huRBCs)21,23–25.
Using FRG huHep mice in combination with a fluorescent PfNF54CSPGFP line to obtain LS infections, we herein conducted a comprehensive transcriptome analysis of PfLS development in vivo.
Results
PfGFP parasites enable isolation of infected hepatocytes
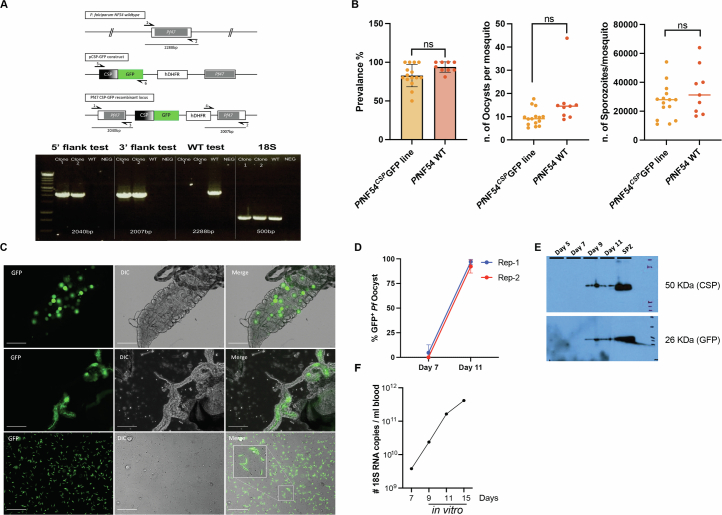
We created a fluorescent Pf parasite line (PfNF54CSPGFP) using the promoter region of the circumsporozoite protein gene (CSP) to drive strong expression of green fluorescent protein (GFP) (Extended Data Fig. 1a) in pre-erythrocytic parasite stages, including PfLS21,24. PfNF54CSPGFP parasites were characterized throughout the parasite life cycle and showed normal asexual BS growth and gametocyte maturation. Furthermore, parasite infection in mosquitoes was comparable to wildtype (WT) PfNF54 parasites (Extended Data Fig. 1b). CSP expression initiates during oocyst development26, and we confirmed that GFP expression occurred in the different mosquito stages by both western blot and live fluorescence microscopy (Extended Data Fig. 1c–e). We then assessed PfNF54CSPGFP parasite competence in successfully completing LS development. The PfNF54CSPGFP parasites, injected as sporozoites into FRG huHep mice repopulated with huRBCs, successfully transitioned to BS infection by day 7 post infection (PI) (Extended Data Fig. 1f).
Extended Data Fig. 1. PfNF54CSPGFP parasites display normal characteristics throughout life cycle.
A) Strategy to generate the PfNF54CSPGFP parasite line. Black arrows indicate PCR primer binding location used for the diagnostic PCR analysis. Gel shows PCR results with primer sets to amplify recombinant DNA template of pCSP-GFP construct and show 5’ integration (primers 1 and 3), 3’ integration (primers 4 and 2) and wild type DNA template (primers 1 and 2). Primers for Pf18S rRNA were used as loading DNA control. B) Comparison of the infection prevalence, number of oocysts per midgut and salivary glands sporozoites per mosquito, between PfNF54CSPGFP line (fifteen individual mosquitoes’ infections) and PfNF54 WT parasites (nine individual mosquitoes’ infections). Data are presented as means ± SD for the first graph and median values for the remaining graphs. C) Live imaging of PfNF54CSPGFP in the mosquito stages to assess GFP expression, top panel day 11 midguts, middle panel day 14 salivary glands, lower panel day 15 dissected sporozoites. Scale bar 50 μm. D) Percent oocysts expressing GFP under CSP promoter observed in the midguts under fluorescent microscope on Day 7 and 11 post PfNF54CSPGFP infected blood feed to the mosquitoes. In two biological replicates. Data are presented as means ± SD. E) Western blot analysis of CSP and GFP expression in the lysate prepared from 1 million sporozoites and ten PfNF54CSPGFP infected mosquito midguts of Day 5, 7, 9 and 11 post infected blood feed. F) Pf18S rRNA measured at different time point by qRT-PCR after the blood was collected from FRGhuHep mice infected with PfNF54CSPGFP on day 7 post sporozoite infection and then cultured in vitro.
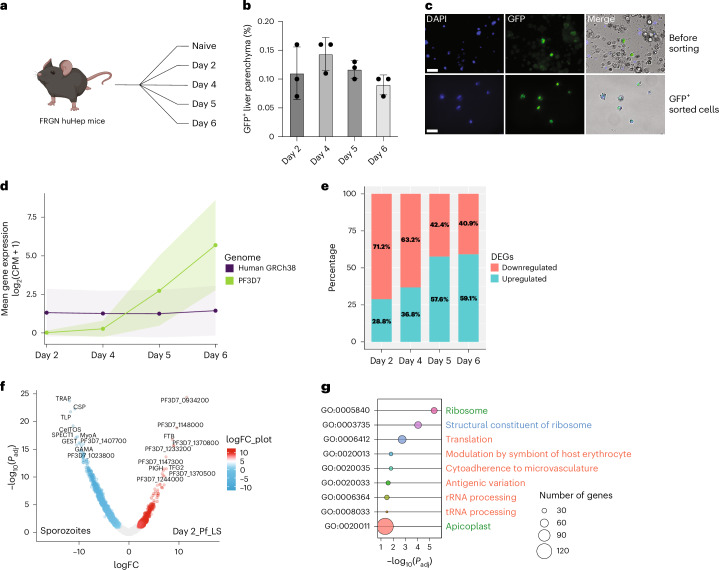
To isolate PfLS GFP+ parasite-infected hepatocytes (PfGFP+), FRG huHep mice infected with PfNF54CSPGFP sporozoites were euthanized on days 2, 4, 5 and 6 PI (Fig. 1a), and processed by fluorescence-activated cell sorting (FACS). A stringent gating strategy was applied during FACS to isolate pure PfGFP+ hepatocytes and minimize uninfected hepatocyte contamination (Extended Data Fig. 2a). A total of 2,500–3,000 PfGFP+ cells were isolated for each LS timepoint. Based on FACS analysis, infection rates averaged 0.1–0.15% (Fig. 1b). Purity of the PfGFP+ infected hepatocyte populations were confirmed by live fluorescence microscopy (Fig. 1c). Sorted cells were then processed for RNA-seq library creation and next generation sequencing (NGS).
Fig. 1. PfNF54CSPGFP parasites enable the isolation of PfLS-infected human hepatocytes from FRG huHep mice and transcriptome analysis.
a, Experimental design. FACS isolation of GFP+ Pf-infected human hepatocytes (PfGFP+) from FRG huHep mice on days 2, 4, 5 and 6 PI. Created with BioRender.com. b, Histogram showing percentage of PfGFP+ hepatocytes as compared to the total at each timepoint (n = 3) (refer to Extended Data Fig. 2a). Data are presented as mean ± s.d. c, Live imaging of PfGFP+ hepatocytes (green) before and after sorting. DNA stained with Hoechst dye (blue). Merge with DIC image. Scale bar, 50 μm. d, Ribbon graph showing the mean gene expression of H. sapiens (purple) and P. falciparum (green) transcriptomes over time. Ribbons represent the mean ± s.d. e, Stacked bar plot showing the number of DEGs for each PfLS developmental timepoint compared to sporozoites. f, Volcano plot showing DEGs between sporozoites and PfLS parasites at day 2 PI. g, Bubble plot with GO terms enriched (Padj < 0.05) in the upregulated genes at day 2 PI. Bubbles represent unique GO terms (y-axis) and bubble colours were assigned arbitrarily. The size of the bubbles illustrates the number of genes involved in each GO term, colour coded for biological process (red), cellular component (green) and molecular function (blue).
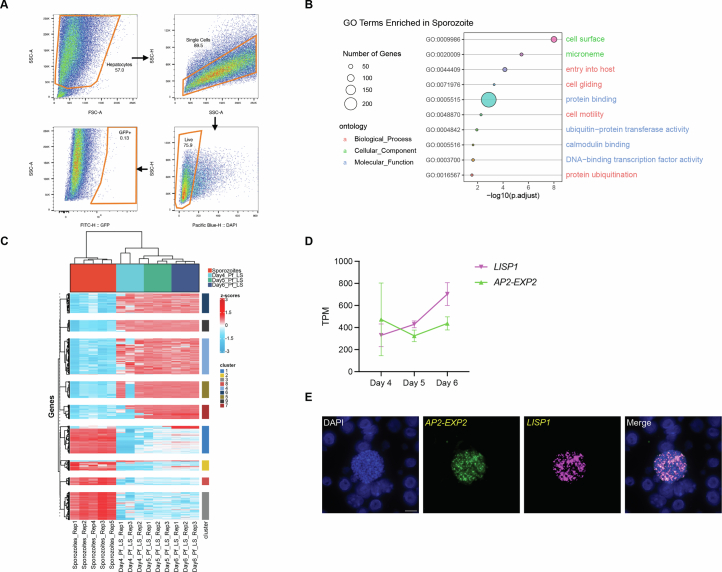
Extended Data Fig. 2. Liver stage transcriptome.
A) Representative FACS gating. i) Forward and side scatter gating based on general size to find hepatocytes. ii) Gating to distinguish single cells from doublets. iii) Live hepatocytes gating based on DAPI. iv) Gating of infected primary hepatocytes based on GFP content. B) Bubble plot showing GO term analysis of the enriched genes in Pf sporozoites (p < 0.06, adj. p < 0.23). Bubbles represent unique GO terms (y-axis) and bubble colours were assigned arbitrarily. The size of the bubbles display by different biological processes is positively correlated with the number of genes involved in each pathway. Color-coded (pink) biological process, (green) cellular component and (blue) molecular function. C) Heatmap showing time course cluster analysis of Pf sporozoites (red), Day 4 (light blue), Day 5 (green) and Day 6 (dark blue). Gene expression values are shown as Z-scores. D) Line-graph showing TPM expression of lisp1 and AP2-EXP2 during the progression of LS development. In three biological replicates. Data are presented as means ± SD. E) RNA-FISH analysis of AP2-EXP2 (green) and LISP1 (magenta) in PfLS schizont from FRGhuHep mice at Day 7 PI. DNA of the parasites is visualized by DAPI staining (blue). Scale bar: 10 μm.
The PfLS parasite transcriptomes
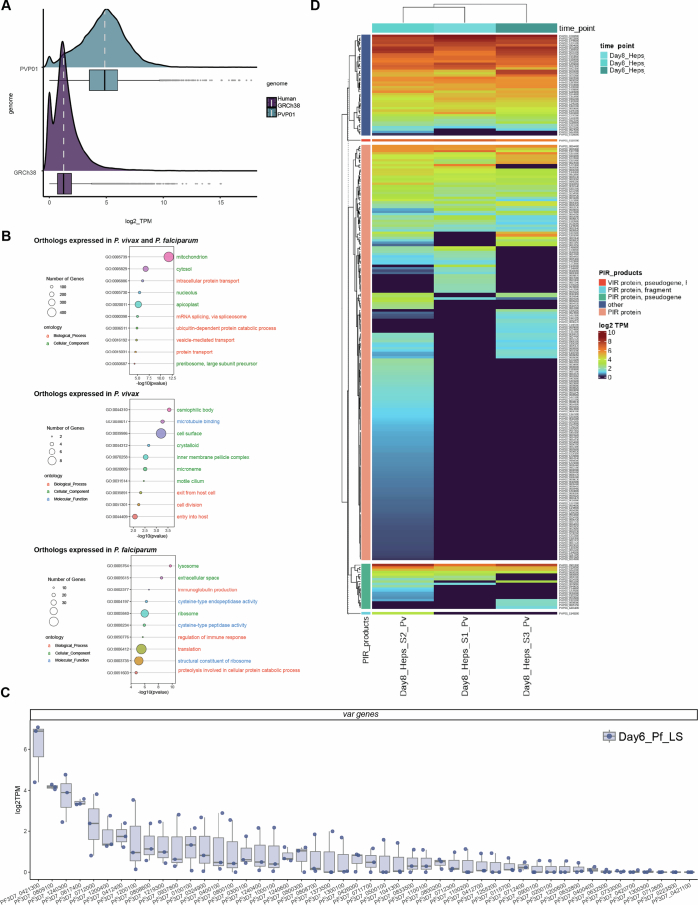
We sequenced a total of 23 PfGFP+ LS RNA-seq library samples and in addition, generated a PfNF54CSPGFP sporozoite transcriptome that was used alongside four previously generated Pf sporozoite transcriptome datasets27 to comparatively analyse steady-state RNA levels. The average normalized transcript abundance of LS-infected human hepatocytes showed detection of mostly human gene transcripts at day 2 PI, the early timepoint of infection. The proportion of the Pf transcript representation increased over time, reaching the highest value at day 6 PI (Fig. 1d and Supplementary Table 1). This was consistent with PfLS cell mass expansion and genome replication during LS schizogony28. Using a cut-off of ≥1 CPM (counts per million) in the three biological replicates, we detected over 4,000 Pf gene transcripts for each timepoint, except for the day 2 timepoint where we detected 1,525 genes passing the threshold in at least one replicate (Supplementary Table 1). Differential expression analysis during early (day 2) and mid (day 4) LS development revealed that most of the differentially expressed genes (DEGs) were less abundant (~70%), with only ~28–36% of transcripts showing higher transcript abundance when compared with sporozoites. Conversely, the proportion of more abundant transcripts during late LS (days 5 and 6) increased to ~57–59% (Fig. 1e and Supplementary Table 1).
To explore the gene expression patterns underlying LS parasite transformation into trophozoites, we investigated the DEGs of the PfLS day 2 dataset in comparison to sporozoites (Fig. 1f). Gene ontology enrichment analysis (GO term) of transcripts less abundant in LS trophozoites (false discovery rate (FDR) ≤ 0.5) revealed pathways involved in sporozoite motility (GO:0048870, GO:0071976) and cell invasion (GO:0044409, GO:0005515) (Extended Data Fig. 2b and Supplementary Table 1), including transcripts encoding MyoA, CSP, CelTOS, TLP, GAMA and TRAP (Fig. 1f and Supplementary Table 1). Conversely, transcripts that were significantly increased in LS trophozoites showed enrichment in translational regulation (GO:0006412) and ribosome biogenesis (GO:0005840, GO:0003735) pathways (Fig. 1g). At day 2 PI, genes associated with the modulation of host cells (GO:0020013) were upregulated in comparison to sporozoites. Genes enriched in this pathway included two classes of the clonally variant genes families (CVGs), 14 members of the rifin gene family and one member of the stevor gene family. Furthermore, 30 of the upregulated transcripts at day 2 encoded apicoplast-targeted proteins (GO:0020011) (Supplementary Table 1). These genes were further analysed by metabolic pathway enrichment analysis using KEGG and MetaCyc databases. This showed a preponderance of gene products mediating fatty acid biosynthesis and isoprenoid biosynthesis, which has previously been shown to be essential for rodent malaria parasite LS development29–31.
To further assess transcriptomes associated with PfLS development, we performed time-course gene expression analysis on the mid-LS timepoint (day 4) and the two later-LS timepoints (days 5 and 6), excluding the day 2 timepoint due to its relatively low parasite sequence read counts. Using sporozoites as a reference, we identified 1,510 genes significantly associated with temporal changes in LS development that were further categorized into 9 unique transcriptional clusters (Extended Data Fig. 2c and Supplementary Table 1). To identify key biological processes, GO term analysis found that ~25% of the LS-expressed genes are not annotated, resulting in low statistical significance for this analysis. To increase statistical power, co-expression clusters were categorized by expression trends in 5 major transcriptional profiles (I–V) (Fig. 2a and Supplementary Table 1). Profile I (Clusters 1 and 2) included genes downregulated throughout LS infection, including pathways mediating cell motility and adhesion (GO:0071976, GO:0048870 and GO:0098609) (Fig. 2b). Profile II (Clusters 3 and 8) included genes that were expressed at lower levels in comparison to sporozoites. The pathways enriched in this group were for genes that play a role in infection of the host (GO:0044409), regulation of transcription (GO:0006355) and lipid metabolic processes (GO:0006629) (Fig. 2b). Profile III (Clusters 4 and 6) included genes for which transcripts increased significantly (with a log2(fold change (FC)) ≥ 2) during LS development when compared with sporozoites. The enriched pathways represented key biological processes such as RNA binding (GO:0003723), ribosome biogenesis (GO:0022625, GO:0005840, GO:0003735 and GO:0022627) and translation (GO:0006412, GO:0005852 and GO:0002181). Profile IV (Clusters 5 and 9) included genes highly expressed in LS. Within this profile, genes were largely associated with mitochondrial processes (GO:0005739) and amino acid metabolism. Furthermore, we observed upregulation of oxidative stress responses (GO:0006979), indicating a parasite response to regulate redox homeostasis. Cluster 7 was the sole member of Profile V, characterized by genes upregulated during late LS development. Most of the genes fall into iron-sulfur cluster assembly (GO:0016226), indicating the importance of iron metabolism during LS maturation. These results summarize the core PfLS transcriptome during an in vivo infection and we validated two LS-expressed transcripts—liver specific protein 1 (LISP1) and the transcription factor (TF) AP2-EXP2—using RNA-fluorescence in situ hybridization (RNA-FISH) in day 7 LS schizonts (Extended Data Fig. 2d,e).
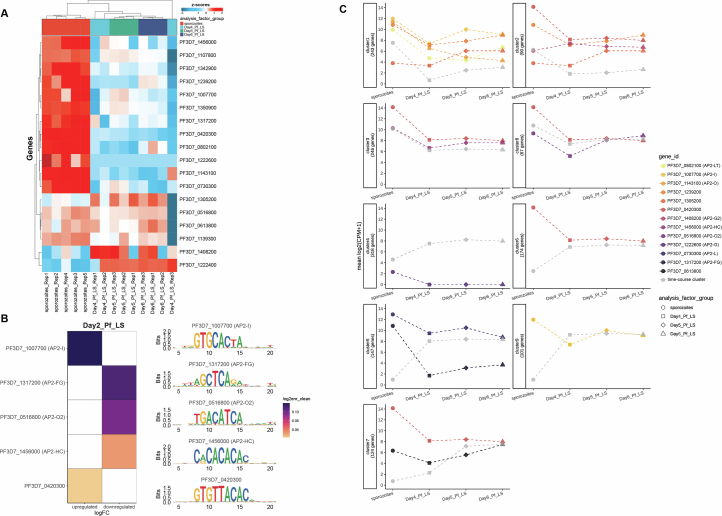
Fig. 2. Time-course cluster analysis of the PfLS transcriptome.
a, Boxplot showing the expression trend of the 9 clusters identified in Extended Data Fig. 2c. The expression was further grouped into 5 major transcriptional profiles (I–V). Boxplots display the median (50th percentile), 75th and 25th percentiles (upper and lower hinges), and the largest and smallest value within 1.5× interquartile range (IQR) from the hinge (whiskers). b, GO term analysis of the genes with significant temporal expression changes in the five profiles. Bubbles represent unique GO terms (y-axis) and bubble colours were assigned arbitrarily. The size of the bubble illustrates the number of genes involved in each GO term, colour coded for biological process (red), cellular component (green) and molecular function (blue) (P < 0.01, Padj < 0.22). GO term enrichment analysis was computed using the hypergeometric test for overrepresentation, and Benjamini–Hochberg adjusted P values were used to account for multiple comparisons. c, Maximum-weight connected subgraphs (MWCS) of each profile, identifying functional modules. Nodes (proteins) identified within a time-course cluster have larger point size, while their interaction partners have smaller point size; node colours show the mean log2FC based on the colour bars in panel d, across days 4, 5 and 6 versus sporozoites. Edge weights represent the random forest classifier score, where higher scores indicate higher-confidence interactions; weights and protein complex IDs are those reported in ref. 32. d, log2FC of genes identified in protein-protein interaction (PPI) subnetworks associated with time/temporal changes in expression for days 4, 5 and 6 liver-stage samples when compared to sporozoites.
We next integrated an interactome analysis of Pf protein–protein interaction (PPI) networks, generated from BS schizonts of three Plasmodium species32, with the LS gene expression data by utilizing the time-course cluster assignments and differential expression analysis to identify functional PPI modules (Fig. 2c and Supplementary Table 1). Time-course transcriptional profiles I and II comprised genes that were found to be downregulated, while profiles III–V were upregulated compared with sporozoites (Fig. 2d). Thus, maximally scored subgraphs for profiles I and II were restricted to the genes in that temporal cluster and the set of downregulated genes, where a gene must be downregulated in at least 2 of 3 LS timepoint datasets. Similarly, modules identified for profiles III–V were limited to genes in each time-course cluster and genes upregulated in at least 2 of 3 timepoints. Profile I modules identified downregulation of proteolysis proteins (ROM1/4) and inner pellicle membrane complexes (IMC1g and ELC), among others (Fig. 2c,d). Profile II functional networks revealed decreased expression of the AP2-EXP TF and chaperone complexes (HSP101, ClpY) (Fig. 2c,d). Subnetworks in Profile III were enriched in translation/ribosome and immunoproteasome-associated proteins (Fig. 2c,d). Transfer RNA (tRNA) ligase networks, including isoleucine, cysteine and alanine tRNA ligases, as well as messenger (m)RNA translation/ribosome, were among the highest-scored functional modules for Profile IV. Profile V identified an iron-sulfur cluster assembly protein and clustered-asparagine-rich protein (SufC/CARP) network, RPB8 RNA polymerase protein–protein interactors and proteins that may be involved in DNA protein kinase complexes (Fig. 2c,d). Furthermore, we noticed that only 20/124 gene IDs from Profile V were present in the PPI identified in the BS, confirming that this specific gene cluster should receive attention for additional investigation.
Transcriptional regulation during LS development
Regulation of Plasmodium gene expression involves complex interactions between TFs, DNA and various regulatory elements, including chromatin and epigenetic marks33,34. Plasmodium has a limited number of identified TFs, with the well-studied apicomplexan AP2 (ApiAP2) family members being the most prominent. These play critical roles in regulating asexual BS infection, sexual-stage development and mosquito infection35–37 but the roles of TFs in PfLS development remain mostly unknown.
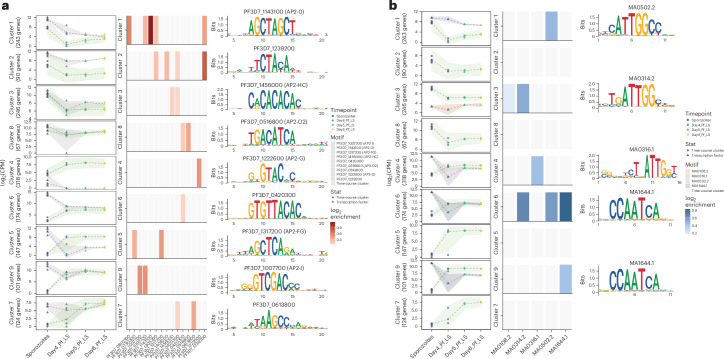
We therefore assessed ApiAP2 expression patterns throughout LS development (Extended Data Fig. 3a) and associated them with enriched DNA-target motifs of genes within co-expression clusters (Fig. 3a). Notably, on day 2 PI, motifs for ApiAP2 factors regulating RBC invasion, AP2-I and an uncharacterized ApiAP2 (PF3D7_0420300), were significantly enriched in the set of upregulated genes (DEGs, FC > 2.0, Padj < 0.05) when compared with sporozoites (Extended Data Fig. 3b)38. Conversely, ApiAP2 members with established roles in gametocyte and mosquito stages, such as the female-specific AP2-FG, AP2-O2 and the heterochromatin component AP2-HC, were overrepresented in the downregulated genes39–41. AP2-L exhibited a similar transcript abundance with Cluster 5, where its DNA motif was also enriched (Extended Data Fig. 3c and Supplementary Table 1). This finding suggests a role of PfAP2-L in regulating the expression of LS-specific genes, as previously shown for rodent malaria parasites42. Furthermore, uncharacterized ApiAP2s, including PF3D7_1305200, PF3D7_0613800 and the transcriptional repressor AP2-G5, were enriched during LS development based on motif enrichment analysis of the time-course transcriptional clusters43. One of the ApiAP2 TF motifs that exhibited significant enrichment during LS was for AP2-G2. In accordance with previous observation40,44,45, PfAP2-G2 might act as a repressor during PfLS development, resulting in decreased target gene expression (Fig. 3a, Cluster 2). In addition, an AP2 (PF3D7_1222400) which was highly upregulated in LS and highly correlated with multiple expression clusters (Supplementary Table 1) is restricted to malaria parasite species that infect primates (Extended Data Fig. 3a), suggesting a unique role in LS infection of primate hosts46.
Extended Data Fig. 3. AP2s expression in the liver stage.
A) Heatmap displaying the AP2 motifs that are enriched in upregulated and downregulated DEGs at the early time point (Day 2 PI) vs sporozoites, and on the right, it showcases the corresponding enriched DNA motifs identified. B) Heatmap showing the expression levels of all AP2 genes during the progression of LS development. C) Line plot depicting the gene expression trends of AP2 genes within each cluster exhibiting enriched motifs.
Fig. 3. Motif enrichment analysis.
a, ApiAP2s (red). b, Newly identified orthologous transcription factors (TFs) (blue). Motif enrichment analysis was carried out for each timepoint cluster. Left: line plot illustrating the gene expression (log2CPM, y axis) trend within each cluster at each timepoint (x axis); individual points represent the mean gene expression per biological replicate (green ribbon). Ribbon widths display the mean ± s.d. expression across all replicates at each timepoint. Expression of the TF whose motif is most significantly enriched within that cluster is highlighted in the grey and non-green ribbons; points represent expression of the TF for each biological replicate and ribbon width is defined as above. Middle: heat map representing the log2 positive enrichment values of all TF motifs overrepresented in the cluster. Motif enrichment analysis was carried out using the binomial test with background sequences randomly sampled from the Pf genome; enrichment values represent the log2-transformed ratio of the observed number of sequences containing the TF motif. Motif enrichment analysis P values and Benjamini–Hochberg Padj for each cluster are reported in Supplementary Table 1. Right: the most highly enriched DNA motif for each cluster.
We next employed OrthoMCL47 to identify novel binding motifs and TFs not belonging to the ApiAP2 family. We gathered a dataset (1,533,059 OrthoMCL accessions) corresponding to 772,570 UniProt accessions, encompassing 571 different species orthologue groups. Subsequently, we cross-referenced this dataset with 1,956 motifs sourced from the Jaspar 2022 Database. This extensive investigation yielded 12 motifs (Supplementary Table 1), neither identical nor reverse complements of each other, predicted to bind previously identified putative Pf TFs48. These TFs belong to transcription factor families, such as TATA-box-binding proteins and the initiation co-activator heteromeric CCAAT-binding factors NFYB (MA0502.2, UniProt P25208) and NFYC (MA1644.1, UniProt Q13952), which exhibited enrichment and displayed a negative correlation with the genes expressed in Clusters 1, 6 and 9 (Fig. 3b).
PfLS PTEX, PEXEL and PNEP expression
Protein export is well studied during PfBS infection but remains largely unstudied during PfLS development. Most exported proteins contain the plasmepsin V cleavable Plasmodium export element (PEXEL), characterized by a pentameric R/KxL/IxE/D/Q motif49–54. Pf also expresses PEXEL-negative exported proteins (PNEPs)55,56. Both PEXEL and PNEP proteins converge at the Plasmodium translocon of exported proteins (PTEX), which mediates their transport across the parasitophorous vacuole membrane (PVM) into the host cell57,58. PTEX comprises EXP2, HSP101 and PTEX150 and is flanked by the auxiliary proteins PTEX88 and TRX2 (ref. 57).
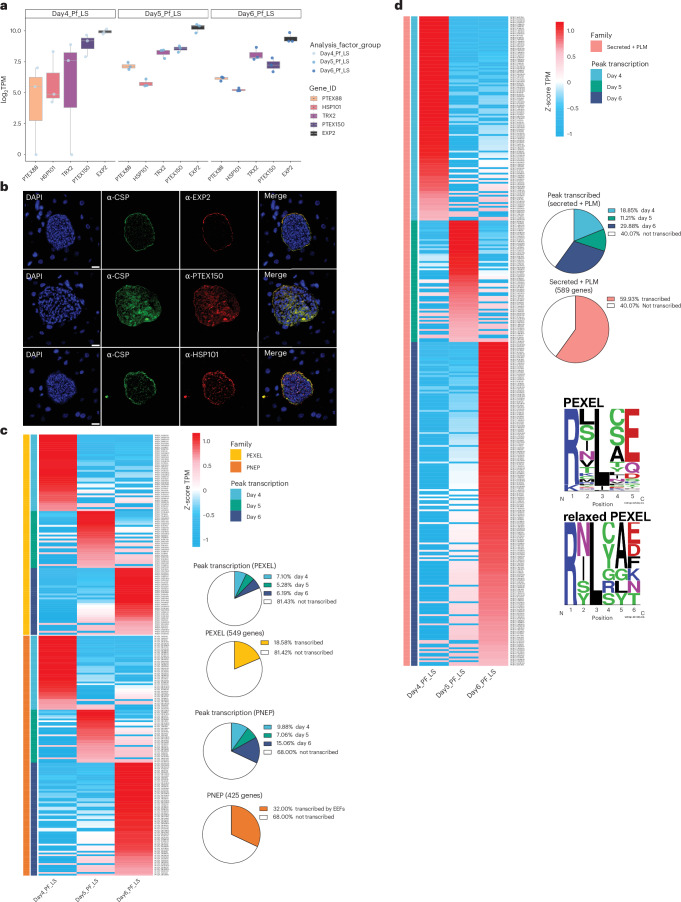
We first examined whether PfLS express the PTEX machinery. We found transcripts for each PTEX component on days 4, 5 and 6 PI (Fig. 4a), with EXP2 showing the most abundant transcripts, probably due to its dual role as a member of the PTEX complex and as a nutrient channel59,60. We confirmed the expression and localization of PTEX proteins at the PVM by immunofluorescence microscopy (Fig. 4b), including expression of HSP101, which performs ATP-dependent unfolding of substrates for translocation across the PVM61,62.
Fig. 4. Expression of the PTEX translocon, and PEXEL and PNEP-encoding genes during LS development.
a, Normalized median expression values (log2TPM) of genes encoding the PTEX translocon across the course of LS development in 3 biological replicates. Boxplots display the median (50th percentile), 75th and 25th percentiles of expression (upper hinge and lower hinge), and the largest and smallest values within 1.5× IQR from the hinge (whiskers). b, Immunofluorescence images showing expression of PTEX components in PfLS schizonts from FRG huHep mice on day 7 PI. Top to bottom: anti-EXP2, anti-PTEX150 and anti-HSP101 (red). The sections were counterstained with DAPI (blue) and anti-PfCSP (green). Scale bar, 10 μm. c, Heat map displaying the Z-score of the mean TPM from the 3 biological replicates for genes encoding PEXEL and PNEP proteins on days 4, 5 and 6 PI. The accompanying pie charts illustrate the percentage of genes with peak expression on each of days 4, 5 and 6 PI (top) and the percentage across the combined 3 timepoints (bottom). d, Heat map demonstrating the Z-score of the TPM averages from the 3 biological replicates for genes encoding ‘Secreted + PLM (PEXEL-like motif)’ proteins on days 4, 5 and 6 PI. The accompanying pie charts illustrate the percentage of genes with peak expression on each of days 4, 5 and 6 PI (top) and the percentage across the combined 3 timepoints (bottom). LOGOs representing PEXEL residues used to identify the Secreted + PLM proteins. Highly conserved amino acids such as the initial arginine (R) are represented as larger letters, and more interchangeable residues appear smaller in the motif (top: canonical PEXEL motif, bottom: relaxed PEXEL motif).
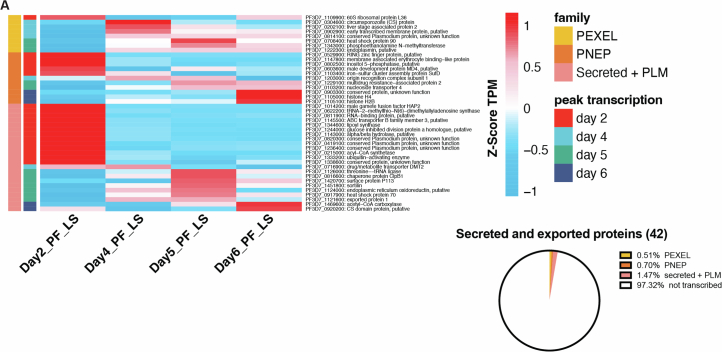
We next bioinformatically mined the PfLS transcriptomes for PEXEL-54,63,64 and PNEP-encoding transcripts55,56. Approximately 19% of genes encoding PEXEL proteins (PEXEL genes) and 32% of genes encoding PNEPs (PNEP genes) were transcribed during LS development. Subsets of PEXEL and PNEP genes peaked in expression on each day, indicating transcription dynamicity during PfLS development (Fig. 4c and Supplementary Table 2). PEXEL motifs are typically located 15–30 residues from the C terminal of the signal peptide cleavage site50,54. However, its position in LISP2 is greater than 300 residues from this position65,66. We therefore searched for additional genes to identify proteins similar to LISP2 and compiled a list of 589 genes encoding ‘Secreted + PEXEL-like motif’ (PLM) proteins of which ~60% showed transcripts during PfLS development (Fig. 4d and Supplementary Table 2). The PLM motifs comprised both canonical pentameric sequences and relaxed, 6-residue motifs54. In addition, we observed transcripts of genes encoding a potential exportome on day 2 PI, comprising 8 PEXEL proteins, 11 PNEPs and 23 Secreted + PLMs (Extended Data Fig. 4).
Extended Data Fig. 4. Analysis of PfLS exportome.
A) Heatmap displaying the Z-scores for TPM averages from the three biological replicates are displayed for genes encoding PEXEL, PNEP and ‘Secreted + PLM’ proteins on Day 2, 4, 5 and 6 PI. The accompanying pie chart illustrates the percentage of genes with peak expression on Day 2 PI.
PfLISP2 knockout parasite characterization in FRG huHep mice
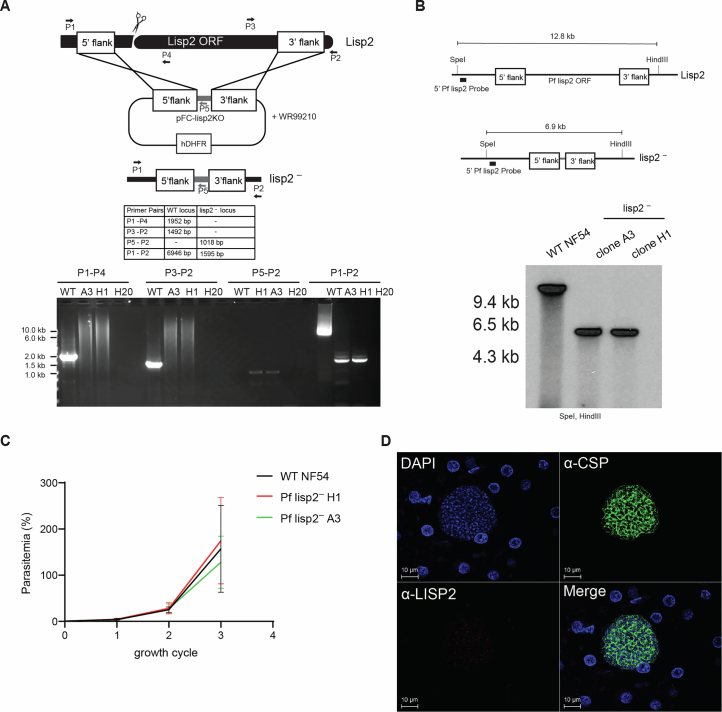
To further characterize an LS-specific protein, we analysed PfLISP2 expression and localization at different LS developmental timepoints. Immunofluorescence analysis using antibodies against PfLISP2 (ref. 66) and PfCSP showed protein expression in accordance with transcript expression (Fig. 5a,b). PfLISP2 protein was detectable in LS starting at day 2 PI, and robust expression of LISP2 was then observed at days 4 and 6 PI, with a localization similar to CSP (Fig. 5b). To investigate PfLISP2 function during LS, we deleted the lisp2 gene (Pflisp2−) from the Pf genome (Extended Data Fig. 5a,b) using established methodologies67. Pflisp2− parasites showed normal BS and mosquito stage development compared with WT PfNF54 (Fig. 5c and Extended Data Fig. 5c). To assess LS development, PfNF54 WT and Pflisp2− sporozoites were used to infect FRG huHep mice, and LS size was assessed comparatively. Interestingly, at day 6 PI, Pflisp2− LS were significantly smaller when compared with PfNF54 LS (Fig. 5d and Extended Data Fig. 5d), and this difference was more pronounced at day 7 PI (Fig. 5d,e), indicating a potential role for PfLISP2 in late LS growth. To further assess the impact of the growth defect, we tested LS-to-BS transition of Pflisp2− parasites (Fig. 5f)21. While 3 FRG huHep mice infected with PfNF54 WT sporozoites showed parasite transition to BS infection (Fig. 5g), 3/15 mice infected with Pflisp2− sporozoites remained BS negative and in vitro culture did not recover any parasites from these mice, further showing the negative impact of the LISP2 gene deletion on LS development.
Fig. 5. PfLISP2 localization and gene deletion.
a, TPM values of CSP and LISP2 at days 2, 4 and 6 PI in 3 biological replicates. Data are presented as mean ± s.e.m. b, PfLS schizonts from FRG huHep mice immunostained with DAPI (blue), anti-CSP (green) and anti-LISP2 (red) at days 2, 4 and 6 PI. Scale bars, 10 μm. c, Comparison of the number of salivary gland sporozoites per mosquito between the PfNF54 line and Pflisp2–. Each dot represents a biological replicate. Data are presented as mean ± s.e.m. d, Comparison of LS parasite size (based on area at the parasite’s largest circumference) between PfNF54 (3 mice) and Pflisp2– knockout parasites (15 mice) on days 6 and 7 PI. Data are presented as mean ± s.e.m. Statistical analysis was carried out using two-way analysis of variance (ANOVA) using a mixed-effects model (REML). ***P < 0.001, *P < 0.05, NSP > 0.05. e, Pflisp2− LS schizont from FRG huHep mice immunostained with DAPI (blue), anti-MSP1 (green) and anti-mTiP (red) at day 7 PI. Scale bar, 5 μm. f, Experimental design to analyse BS breakthrough of Pflisp2– on days 6 and 7 PI; huRBCs were injected intravenously to enable transition of LS parasites to BS infection. On day 7, mice were euthanized, blood was collected and used for RT–qPCR analysis to detect parasite 18S rRNA. The remaining blood was transferred to in vitro asexual parasite culture. Created with BioRender.com. g, Parasite BS densities in the blood of FRG huHep mice at the time of transition on day 7 PI, with 3 mice in the control group (PfNF54) and 15 mice in the Pflisp2− group. Data are presented as mean ± s.e.m.
Extended Data Fig. 5. Pf lisp2– parasites analysis.
A) The schematic depicts generation of Pflisp2– via CRISPR/Cas9-mediated gene editing. Primers used for confirming parasite genotypes are shown and the length of PCR amplicons are indicated in the table. The agarose gel electrophoresis indicates gene deletion of Pf lisp2– in two clones A3 and H1. Expected amplicon sizes for WT and PfLARC2 and the pFC plasmid are indicated in the schematic. B) The schematic and the corresponding Southern blot for the Pf NF54 and Pflisp2– locus. Genomic DNA from Pf NF54 and Pflisp2– clones A3 and H1 was digested with SpeI and HindIII and probed with a 5′ LISP2 probe to confirm gene deletion of Pf LISP2. C) Asexual BS parasitemia is compared between Pf NF54 (black line) and Pflisp2– clones A3 (green line) and H1 (red line) over three replication cycles. Data represent mean ± SD. n = 3 biological replicates. Statistical analysis was carried out using 2-way ANOVA. ns, not significant. P > 0.05 is taken as ns. Graphs comparing (D) Immunostaining of Pflisp2– schizont from FRGhuHep mice at Day 6 PI, immunostained with DAPI (blue), anti-CSP (green) and anti-LISP2 (red). Scale bar corresponds to 10 μm.
LS transcriptome conservation and differences between Pf and Pv
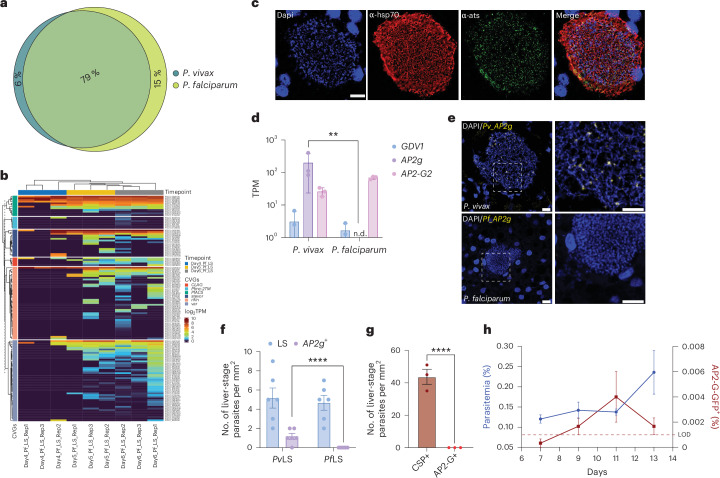
To assess conservation of pathways in LS of human malaria parasites, we next compared the transcriptomes of late Pf and Pv LS schizonts. Since the two species show differences in the duration of LS maturation, namely, 6.5 days for Pf and 9 days for Pv21,23, we selected Pf day 6 PI and Pv day 8 PI and confirmed LS maturity by the expression of MSP1 (Supplementary Table 1). Because PvLS cannot be isolated by FACS, we performed total RNA extraction of Pv-infected FRG huHep mouse livers at day 8 PI, followed by enrichment for Pv mRNA using magnetic pulldown with custom-made baits tiling the entire Pv genome19,68. Hybrid capture selection was followed by Illumina sequencing. All sample sequences were aligned to the H. sapiens, M. musculus and Pv P01 genomes as performed for the Pf dataset (Extended Data Fig. 6a). We first assessed the relatedness of Pf and Pv coding genomes and identified 4,485 Pf genes with Pv orthologues (78%) (Fig. 6a and Supplementary Table 1). We observed a substantial overlap of gene expression, with 79% of orthologous genes expressed at ≥1 TPM in all biological replicates of late LS for both species. Among the genes concordant in expression, we found gene products that play key roles during LS development, including LISP2 (Supplementary Table 1). GO term enrichment analysis for co-expressed genes showed enrichment in pathways important for parasite development, such as mitochondria-targeted gene products (GO:005739) and protein transport-associated gene products (GO:006886, GO:0015031) (Extended Data Fig. 6b).
Extended Data Fig. 6. P. falciparum and P. vivax expressed orthologues and pathways.
A) Density plot showing the mean gene expression (log2TPM) aligning against H. sapiens (purple) and P. vivax (blue). B) Bubble plot showing GO term analysis of the expressed genes in Pf and Pv (p < 0.06, adj. p < 0.23), GO terms enriched (p < 0.06, adj. p < 0.19) in genes expressed only in PvLS transcriptome and terms enriched (p < 0.03, adj. p < 0.21) in those expressed only in Pf. The size of the circles display by different biological processes is positively correlated with the number of genes involved in each pathway. Color-coded (pink) biological process, (green) cellular component and (blue) molecular function. C) RNA-seq shows var gene transcription during PfLS at day 6 PI. Data are presented as median value of three biological replicates. the box limits are the upper and lower quartiles. Transcripts corresponding to each var gene (shown on the x axis) are indicated as log2TPM, indicated on the y axis. Boxplots (box and whiskers plot) display the median (50th percentile), upper hinge as the 75th percentile, and lower hinge as the 25th percentile of expression; whiskers extend from the hinge to the largest or smallest value within 1.5 * IQR (interquartile range) from the hinge, respectively. D) Heatmap illustrating the expression patterns of P. vivax clonally variant gene families in the three biological replicates.
Fig. 6. Comparative transcriptome analysis of late Pf and Pv liver stages.
a, Venn diagram of orthologous genes expressed at ≥1 TPM in all 3 biological replicates of Pf or Pv. Overlapping section identifies genes detected in both Pf and Pv transcriptomes. b, Heat map illustrating the expression patterns of clonally variant gene families throughout the course of LS development. c, PfLS schizonts from FRG huHep mice immunostained with DAPI (blue), anti-PfHSP70 (red) and anti-PfATS (green) at day 7 PI. Scale bar, 10 μm. d, TPM values of a selection of gametocyte genes in the Pf and Pv datasets. Results are presented as mean ± s.d. of 3 independent experiments; **Padj = 0.007. e, RNA-FISH analysis of AP2-g (yellow) in PvLS schizont from FRG huHep mice at day 9 PI (top) and PfLS schizont from FRG huHep mice at day 7 PI (bottom). DNA of the parasites is visualized by DAPI staining (blue). Scale bar, 10 μm. f, Quantification of the number of PvLS and PfLS schizonts from FRG huHep mice at days 9 and 7 PI, respectively, expressing AP2-g by RNA-FISH. Results are presented as mean ± s.d. of 3 independent experiments; two-way ANOVA, ****P < 0.0001. g, Number of PfLS parasites positive for CSP and GFP recognizing PfNF54AP2GGFP parasites from FRG huHep mice at day 7 PI. Results are presented as mean ± s.e.m of 3 independent experiments; two-way ANOVA, ****P < 0.0001. h, Flow cytometry analysis of DNA dye+ (blue) and DNA dye+AP2-GFP+ (red) BS parasites following PfNF54AP2GGFP sporozoite infection in FRG huHep mice. The dotted line denotes the limit of detection (LOD), defined as the mean percentage of DNA dye+AP2-GFP+ events from the uninfected RBCs control. N = 3 mice. Results are combined and presented as mean ± s.e.m. of 2 independent experiments.
Among the PfLS-expressed genes without Pv orthologues, we found expression of multiple members of the CVG families (Fig. 6b). We focused on var genes (Extended Data Fig. 6c), encoding PfEMP1 proteins, which mediate infected red blood cell (iRBC) attachment to the vascular endothelium and are antigenically variant during PfBS infection69. Previous findings suggested that var gene members can be transcribed in gametocytes, ookinetes, oocysts and sporozoites27,70–72. We found transcripts for multiple var genes during PfLS development. Notably, there was consistent transcript detection of 10 var genes in all 3 biological replicates (Fig. 6b and Extended Data Fig. 6c), belonging to the UpsB and UpsC subtypes73. The PF3D7_0421300 and PF3D7_0809100 var genes, the latter of which is also expressed in sporozoites27, showed the highest var gene transcript abundance during LS development. We further investigated PfEMP1 protein expression using a pan antibody recognizing the PfEMP1 semi-conserved intracellular region (ATS) to examine expression in day 7 PfLS parasites. A punctate expression pattern was observed (Fig. 6c), confirming PfEMP1 expression in day 7 PfLS schizonts. Similarly, among the PvLS genes without Pf orthologues, we identified transcripts of multiple members of the pir and vir CVG families (Extended Data Fig. 6d).
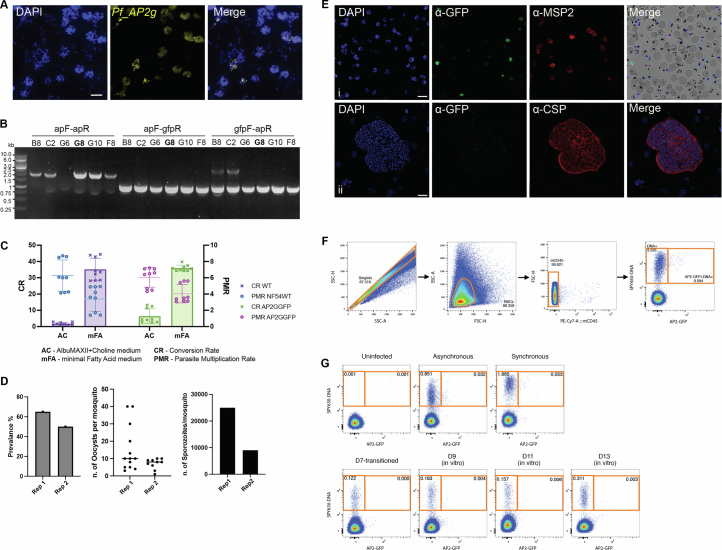
We next assessed whether Pf sexual commitment initiates during LS development, which might result in the release of exo-erythrocytic Pf merozoites ready to differentiate into gametocytes, as previously shown at the transcriptomic level for PvLS schizonts19,74–76. We did detect transcripts for the sexual-stage-specific proteins Pvs16 and Pvs25 and the AP2-g transcription factor in PvLS schizonts, the latter of which is the master regulator of sexual commitment during BS infection77–79 (Fig. 6d). In contrast, we did not detect the expression of AP2-g in the PfLS transcriptomes (Fig. 6d). To further evaluate whether mature PfLS schizonts exhibit sexual commitment, we performed RNA-FISH on day 7 Pf-infected livers and day 9 Pv-infected livers with PfAP2-g and PvAP2-g specific probes, respectively. RNA-FISH confirmed the absence of AP2-g transcripts in all PfLS schizonts examined (Fig. 6e,f and Extended Data Fig. 7a), while AP2-g RNA signal was detected in a subset of PvLS schizonts (Fig. 6e,f). To further investigate when Pf forms sexually committed parasites, we used an endogenously GFP-tagged AP2-G reporter line (PfNF54AP2GGFP) (Extended Data Fig. 7b,c)80. The PfNF54AP2GGFP line showed no transmission defect (Extended Data Fig. 7d), and sporozoites were generated and used to infect 3 FRG huHep mice. The infected mice were infused with huRBCs at day 7 PI and subsequently, liver tissue and infected blood samples were collected. The liver tissue sections were stained with anti-GFP antibodies previously validated on BS parasites (Extended Data Fig. 7e) to assess AP2-G protein expression in mature PfLS schizonts. The blood samples were further processed for flow cytometry to evaluate whether LS merozoites and/or early ring BS parasites expressed AP2-G. Similarly to the transcriptomes and RNA-FISH results, we did not detect any GFP-positive PfLS schizonts (Fig. 6g and Extended Data Fig. 7e). Flow cytometry analysis of the iRBCs showed no AP2G-GFP-positive BS parasites at day 7 PI (Fig. 6h and Extended Data Fig. 7f,g), confirming that PfLS parasites do not directly produce sexual-stage progeny from any egressing exo-erythrocytic merozoites. We further cultured the iRBCs in vitro and sampled cultures every 48 h to assess when GFP expression, and thus sexual commitment, occurred. AP2-G-GFP-positive BS parasites were first observed at 48 h post exo-erythrocytic merozoite egress (day 9 post sporozoite infection). The proportion of AP2-G-GFP-positive BS parasites then increased 96 h post exo-erythrocytic merozoite egress (Fig. 6h).
Extended Data Fig. 7. AP2s expression in Pf liver stage.
A) RNA-FISH analysis of AP2-g (yellow) in PfNF54 enriched schizonts to assess probe specificity. DNA of the parasites is visualized by DAPI staining (blue). Scale bar: 10 μm. B) The Agarose gel electrophoresis indicates six clones of the PfNF54AP2GGFP transgenic parasite line. Clone G8 (bold) was selected for further propagation and used in functional assays. C) Sexual conversion assay of the PfNF54AP2GGFP G8 clone, was performed to estimate the sexual conversion rate (CR) and the parasite multiplication rate (PMR) in comparison with NF54 WT. D) Analysis of the infection prevalence, number of oocysts per midgut and salivary glands sporozoites per mosquito, of PfNF54AP2GGFP line. In two biological replicates. E) Immunostaining of PfNF54AP2GGFP erythrocytic enriched schizonts (i) and PfLS schizont from FRGhuHep mice at Day 7 PI (ii), immunostained with DAPI (blue), anti-GFP (green) and anti-MSP2 or anti-CSP (red). Scale bar corresponds to 10 μm. F) Gating strategy. Single cells were gated based on side scatter (SSC-H vs SSC-A) followed by gating on the red blood cells. Any possible white blood cells were avoided from further analysis by gating on CD45 negative population. Finally, the BS parasites were gated based on positive DNA staining by SPY650-DNA dye, whereas AP2 expressing parasites were gated based on positive DNA staining and GFP content. G) Representing flow layouts. N=3 mice. Results are combined and presented as means ±SEM from two independent experiments. Source data are provided as a Source data file.
Discussion
PfLS infection is an attractive target for vaccine and drug development since LS elimination prevents the onset of symptomatic BS infection and onward parasite transmission. Yet, comprehensive PfLS gene expression profiles have not been reported. Here we successfully conducted a transcriptome analysis of PfLS throughout their intrahepatocytic development. Our analysis revealed active core genes and pathways driving PfLS development. The successful mosquito-to-human parasite transition of sporozoites requires rapid availability of gene products that mediate the successful establishment of the intrahepatocytic LS replication niche4. We observed downregulation of the sporozoite translational repression machinery at day 2 post hepatocyte infection, which is likely critical for sporozoite transformation into intracellular trophozoites and their establishment within hepatocytes33,81. These data extend previous findings indicating that Plasmodium parasites use translational repression during sporozoite transmission, storing transcripts in the sporozoite stage, which are then presumably translated after hepatocyte infection81. Furthermore, the LS transcriptome analysis delineated gene expression patterns and pathways that sustain the parasite’s drastic metamorphosis from sporozoite to trophozoite stage and its ability to then enter exo-erythrocytic schizogony. During LS development, fatty acid and isoprenoid biosynthetic metabolic pathways were enriched as early as day 2 PI44,82,83, and later in LS development, amino acid synthesis and numerous metabolic pathways were upregulated. In addition, gene products involved in redox homeostasis, probably used to counteract the oxidative stress generated in infected hepatocytes, were also upregulated44,84.
To analyse putative regulation of gene expression in LS, we examined TF DNA-binding motif enrichment in the upstream sequences of co-expressed genes. Our findings uncovered a link between the uncharacterized ApiAP2 TF genes PF3D7_0420300 and PF3D7_0613800 and Cluster 7, which was enriched for genes that are upregulated during late LS development. In addition, ApiAP2 PF3D7_1222400, exclusive to primate malaria parasites, was upregulated during LS development. This AP2 could potentially control expression of LS genes playing unique roles in primate hepatocyte infection.
Despite dramatic differences in host cellular environments between erythrocytes and hepatocytes85–87, LS might depend on the export of proteins to counteract hepatocyte intracellular defenses, obtain nutrients and thereby support LS survival and growth in the liver. We found that PfLS parasites expressed all the components of the PTEX machinery of protein export at the transcript and protein level, showing the presence of EXP2, PTEX150 and the unfoldase HSP101 (refs. 61,62). Our identification of HSP101 in LS confirms that all the members of the PTEX machinery are expressed in LS and this supports the notion that protein export might play a crucial role in intrahepatocytic parasite development. In agreement with this, a recent conditional knockout study of PfEXP2 and PfPTEX150 showed the important role of these PTEX components in LS development66. On the basis of the LS transcriptomes, we predicted an LS exportome consisting of 102 PEXEL, 136 PNEP and 353 Secreted + PLM proteins. Our further study of PfLISP2 by gene knockout revealed that this putative exported protein plays a role in LS growth. However, the analysis of LISP2 cellular localization found no evidence that this PEXEL-motif-containing protein is exported beyond the LS compartment.
We identified transcripts of members of the var gene family and observed PfEMP1 (ref. 69) protein expression in mature LS schizonts. These findings suggest that during parasite transmission and infection of new human hosts, resetting of var gene expression begins in the mosquito stages and is finalized in the LS, explaining the broad var gene repertoire observed in the first generation of BS parasites that emerge in human participants experimentally infected with Pf sporozoites88,89.
Our comparative transcriptome analysis of Pv and Pf late LS developmental stages revealed that although there is considerable conservation of LS gene expression across the core conserved genomes of these distantly related human malaria parasites90, unique features emerged. Importantly, we found that in contrast to late PvLS, late PfLS did not show any evidence of commitment to sexual-stage formation, hence the latter cannot generate a population of exo-erythrocytic merozoites committed to sexual-stage development. Our analysis of Pf parasites that had transitioned from LS to BS infection further ascertained this notion, showing that Pf must undergo at least one full asexual replication cycle in the blood for sexual commitment to occur. This finding might have implications for Pf transmission dynamics in malaria-endemic areas.
Finally, the datasets presented herein have limitations and future work is needed to further explore new research questions that have arisen from the data. We measured transcript abundance in pooled isolated LS parasites; single-cell studies will be crucial in uncovering any parasite heterogeneity during liver-stage development. In addition, the use of FRG huHep mice, which lack a functional immune system, limited our ability to assess the role of the human host responses in shaping LS parasite transcriptomic profiles. Furthermore, our findings on LS protein export underscore the need to experimentally validate protein export into the PfLS-infected hepatocyte for predicted exported proteins and to establish potential functional roles of PfEMP1 variant proteins in LS.
Methods
Creation of Pf parasite line expressing GFP under CSP promoter
The plasmid pEFGFP used to create 3D7HT-GFP was modified by replacing the EF1α promoter with a 1.2 kb DNA fragment from the 5′ UTR immediately upstream to the start codon of the PfCSP gene (CSP promoter)91. Plasmid integrity was confirmed by DNA sequencing and the confirmed plasmid was used for transfection of PfNF54. PfNF54 parasite culture was synchronized at ring stage with 5% sorbitol 2 days before transfection. On the following day, trophozoites were selected by incubation in 0.7% gelatin solution. Ring stages were transfected by electroporation at 0.31 kV and 950 µF with a BioRad Gene Pulser (BioRad)92. Cultures were put under drug pressure starting at 6 h post transfection using 5 nM WR99210 (Jacobus Pharmaceuticals). Integration was confirmed by PCR on parental population and clones obtained by limiting dilution as previously described using primers as follows: forward primer 5F 5′-TGAGCTTATTTCAATTGTTGTGT-3′; reverse primer 6R 5′-GGTACCGCTAATTTTCTCATCATGAATTGT-3′; forward primer 1F 5′-GGAGGGAAAAAATAATCGTACA-3′; reverse primer 3R 5′-TCAACCAAGTCATTCTGAGA-3′; forward primer 4F 5′ -GTAATTTATGGGATAGCGAT-3′; reverse primer 2R 5′-GTTTTTCATCGAAATGCGTA-3′.
Mosquito rearing and sporozoite production
Anopheles stephensi mosquitoes were reared and maintained following standard procedures outlined in Methods in Anopheles Research MR4 (https://www.beiresources.org/Publications/MethodsinAnophelesResearch.aspx). Mosquitoes were kept at 27 °C and 75% humidity in temperature- and humidity-controlled incubators on 12 h light/dark cycles within a secured ACL2 Facility. Cotton pads soaked in 8% dextrose and 0.05% PABA solution were placed daily on the top net of mosquito cages. PfNF54 WT, PfNF54CSPGFP, PfNF54lisp2− and PfNF54ap2gGFP asexual parasites were maintained by subculturing at 2% parasitemia in RPMI-1640 medium (25 mM HEPES, 2 mM l-glutamine medium containing 10% human serum and 50 μM hypoxanthine) and maintained at 37 °C in an atmosphere of 5% CO2, 5% O2 and 90% N2.
Gametocyte cultures were set up at 1% parasitemia and 5% haematocrit, and the asexual parent cultures had a parasitemia between 3 and 7%. The media of the gametocyte cultures were changed daily for 15 days while keeping the plates/flask on a slide warmer during media change to prevent dramatic temperature changes. Mature gametocyte cultures were spun at 800 g for 2 min at 37 °C in a temperature-controlled centrifuge. Parasitized red blood cell pellet was resuspended at 0.5% gametocytemia in 50:50 serum:blood mix and used for standard membrane feeding as previously described93. For Pf sporozoite production, 3–7-day-old female mosquitoes were used for every infectious blood meal. Mosquito infections were evaluated on day 7 by checking oocyst prevalence and oocyst number in 10–12 dissected mosquito midguts. Sporozoite numbers were determined by dissecting and grinding salivary glands on day 15 post feeding. These sporozoites were used for infecting FRGhuHep mice.
Mice
FRG huHep mice (female, >4 months of age) were purchased from Yecuris and were maintained in pathogen-free BSL2+ conditions with 12 h light/dark cycle, 72 °F temperature and 45% humidity at the Center for Global Infectious Disease Research, Seattle Children’s Research Institute (SCRI). All animal procedures were performed following the regulations of the SCRI’s Institutional Animal Care and Use Committee (IACUC). The animal procedures were approved by the IACUC under the 00480 protocol. Repopulation of human hepatocytes in FRG huHep mice was confirmed by measuring human serum albumin levels, and only animals with human serum albumin levels >4 mg ml−1 corresponding to 70% repopulation of human hepatocytes were used as previously described21. Animals were cycled on 8 mg l−1 of NTBC drug once a month for 4 days to maintain hepatocyte chimaerism. Mice were taken off NTBC before and during experimentation.
Analysis of PfNF54CSPGFP LS-to-BS transition in FRG huHep mice by quantitative PCR with reverse transcription (RT–qPCR)
FRG huHep mice were intravenously (i.v.) infected with 1 million PfNF54CSPGFP sporozoites. To support parasite transition from LS to BS, mice were injected with 400 μl of human RBCs i.v. at 70% haematocrit on days 6 and 7 post infection. The blood was then collected by cardiac puncture after exsanguinating mice on day 7. Fifty microlitres of blood were added to NucliSENS lysis buffer (bioM rieux) and frozen immediately at −80 °C, and the rest of the blood was transferred to in vitro culture. Fifty microlitres of blood samples were collected from in vitro culture on days 9, 11 and 15 post infection in mice (that is, days 2, 4 and 8 of in vitro culture), added to NucliSENS lysis buffer and frozen at −80 °C. All samples were processed and analysed for the presence of 18S rRNA as follows: The RT–qPCR reaction was performed using 35 µl SensiFAST Probe LoROX one-step kit (Bioline) and 15 µl of extracted eluate. Plasmodium 18S rRNA primers/probes (LCG BioSearch Technologies) were as follows: forward primer PanDDT1043F19 (0.2 µM): 5′-AAAGTTAAGGGAGTGAAGA-3′; reverse primer PanDDT1197R22 (0.2 µM): 5′-AAGACTTTGATTTCTCATAAGG-3′; probe (0.1 µM): 5′-[CAL Fluor Orange 560]- ACCGTCGTAATCTTAACCATAAACTA[T(Black Hole Quencher1)]GCCGACTAG-3′[Spacer C3]. Cycling conditions were reverse transcription (RT) (10 min) at 48 °C, denaturation (2 min) at 95 °C and 45 PCR cycles of 95 °C (5 s) and 50 °C (35 s).
Isolation of PfNF54CSPGFP LS-infected primary human hepatocytes
FRG huHep mice were intravenously injected with 2–3 million PfNF54CSPGFP sporozoites per mouse. The primary hepatocytes were collected by perfusing and digesting the livers on days 2, 4, 5 or 6 post sporozoite infection using modified protocol94. Briefly, the mice were deeply anaesthetized with ketamine (100 mg kg−1 body weight)/xylazine (10 mg kg−1 body weight) solution, and the livers were perfused and digested with perfusion buffers I (0.5 mM EGTA in 1× DPBS without Ca2+ and Mg2+) and II (50 μg ml−1 liberase TL with 800 μM CaCl2 in 1× DPBS), respectively. The hepatocytes were dispersed in 1× DMEM complete medium and centrifuged twice at low speed (50 g) for 2 min at 10 °C. The cell pellet was resuspended in the complete medium and viability was tested using trypan blue staining. The final cell concentration was adjusted to 2 × 106 per ml and further used for FACS. Approximately 2,500–3,000 GFP+ and GFP− cells were sorted directly into QIAzol lysis buffer.
RNA-seq library preparation
For Pf RNA-seq libraries preparation, total RNA was extracted from sorted infected primary human hepatocytes using miRNeasy micro kit (QIAGEN) according to manufacturer instructions, including on-column DNase digestion. Libraries were prepared using SMART-seq v.4 Ultra Low Input (Clontech) and were sequenced on the Illumina NextSeq 500 as 75-bp paired-end reads. The resulting data were demultiplexed using bcl2fastq2 (Illumina) to obtain fastq files for the downstream analysis. A minimum of 3 biological replicates were analysed; technical replicate libraries for each biological replicate were also sequenced. Additional raw sequence reads from Pf sporozoite RNA-seq samples (N = 4) were retrieved from the Sequence Read Archive (PRJNA344838)27.
For Pv RNA-seq library preparation, total RNA was extracted from the FRGhuHep mice livers infected with 1 million Pv sporozoites (field strain) using TRIzol (Thermo Fisher) and purified using RNeasy mini kit (Qiagen) according to manufacturer instructions. A SureSelect XT custom oligo library was designed with Agilent (Design ID: S0782852) to enrich for Pv-specific complementary (c)DNA among the pool of human, mouse and parasite cDNA obtained from the RNA extraction from the humanized mouse liver. A total of 85,000 probes of 120-bp size were tiled every 100 bp across the entire Pv Sal I genome. Sequences >30% similar to human sequences were excluded. Sequencing libraries were prepared according to the SureSelect XT RNA Target Enrichment for Illumina Multiplexed Sequencing protocol from Agilent (Ref: 5190-4393). Libraries were analysed using a BioAnalyzer and quantified using qPCR. Illumina libraries were sequenced on Mi-seq as 75-bp single-end reads.
Data analysis
Quality control of fastq files was performed using FastQC v.0.12.1 software; fastqs from paired biological and technical replicate liver-stage samples were concatenated to increase sequencing depth and coverage. Reads were aligned to a custom reference genome containing H. sapiens (GRCh38, Ensembl gene annotations (v.106)95), M. musculus (PRJNA20689, GRCm39, Ensembl gene annotations (v.103)96) and P. falciparum genome (PlasmoDB, PlasmoDB-58_Pfalciparum3D7 (ref. 97)) with STAR 2.7.9. The alignment was completed with default parameters with the addition of ‘–twopassMode Basic’ and ‘–quantMode GeneCounts’ to produce a gene count matrix. Gene-level counts were normalized to TPM. P. vivax samples were processed identically, except with the reference genome containing H. sapiens, M. musculus and the P. vivax genome (PRJEB14589, PlasmoDB-58_PvivaxP01 (ref. 68)).
Differential expression and clustering analysis
All analyses were conducted in the R v.4.1 statistical environment. The raw gene count matrix for liver-stage PfLS and sporozoite samples, which included the publicly available data from PRJNA344838, underwent batch correction using ComBat-seq (sva v.3.42.0) with default parameters98. The adjusted counts were used in differential gene expression analysis with Limma voom v.3.50.3; genes with absolute log2FC > 1 and FDR < 0.05 were retained99. Batch-corrected gene counts were normalized to the trimmed mean of M-values (TMM) and converted to log2 scale before differential expression analysis, unsupervised hierarchical clustering and time-course regression analysis100. Examination of changes in PfLS gene expression over time compared to sporozoites was conducted using maSigPro v.1.66.0 with a polynomial degree of 2 (quadratic)101. Comparison of Pf and Pv samples was carried out with TPM-normalized gene counts (unadjusted). For each species independently, genes were selected if expressed at ≥1 TPM in all 3 biological replicates; the expressed genes were converted into Pf orthologues using PlasmoDB v.58. Orthologues from Pf and Pv were overlapped and visualized using ggVennDiagram (v.1.2.2)102. Gene ontology analyses were performed for biological processes, cellular component and molecular function terms using ClusterProfiler (v.4.2.2)103, and were retained for further analysis if −log10(P) > 1.2 (P < 0.06). Available GO terms for Pf were downloaded from PlasmoDB v.58.
Protein–protein interaction network analysis
Protein–protein interaction (PPI) networks were retrieved from ref. 32; in brief, the PPIs were defined using quantitative mass spectrometry of protein isolates from 3 species of Plasmodium schizonts in the erythrocytic stage. The ref. 32 PPI graph contained 1,642 nodes and 23,544 edges by including only those proteins found to be expressed in liver-stage Pf RNA-seq data from this study. Maximally weighted connected subgraphs (MWCS) were utilized to identify a subset of nodes in the PPI graph that are most connected using the BioNet v.1.54.0 R package. For each time-course profile (5 total profiles), MWCS ‘modules’ were identified by fitting a beta-uniform mixture (BUM) model to a P-value distribution from the genes’ time-course regression analysis and differential expression analysis. The modules were scored using the fitted BUM model, with the FDR cut-off being the 5th or 10th percentile most-significant adjusted P values (FDR ≤ 6.14 × 10−10 for all profiles). MWCS for each profile were then selected using the fast Heinz algorithm v.2.0. The final MWCS modules for each profile contained the proteins whose gene expression was found to be associated with temporal changes via the time-course regression analysis as well as their interaction partners that exhibited differential expression.
Motif enrichment analysis
Motif enrichment analysis was completed with the monaLisa v.1.0.0 Bioconductor package104 with background=‘genome’, where query promoter sequences were compared to promoter region sequences randomly sampled from the Pf genome using default parameters, except genome.oversample=10 and binomial enrichment test. Motifs were retained if −log10(P) > 1.2 (P < 0.06). Genomic DNA sequences were derived from PlasmoDB-58_Pfalciparum3D7_Genome.fasta and promoter regions were defined as −1,000 bp from transcript start site using coordinates from PlasmoDB-58_Pfalciparum3D7.gff. PfAP2 transcription factor position weight matrices were downloaded from the supplementary materials of ref. 35. Pf transcription factor orthologues were identified using the OrthoMCL47 orthologue group IDs provided on PlasmoDB v.58. Stable gene identifiers from all available species belonging to orthologue groups from Pf PlasmoDB v.58 were retrieved from OrthoMCL DB v.6.14 (orthomcl.org). The stable gene identifiers were converted to species-specific Uniprot identifiers using UniProt.ws v.2.38.1 and used to retrieve the orthologues’ transcription factor motif position weight matrices from the JASPAR2022 v.0.99.7 Bioconductor annotation package.
PTEX, PEXEL, PNEP and Secreted + PLM analysis
The PfLS transcriptome was analysed to identify transcripts corresponding to published PfPEXEL and PNEP proteins (see Supplementary Table 2 for proteins and references). For transcriptional analysis of genes corresponding to proteins with a signal peptide + PLM, a list of 589 genes was first identified by searching PlasmoDB for proteins with a signal peptide as well as a consensus PfPEXEL sequence (normal PEXEL: [KR] [ACDEFGHIKLMNQRSTVWY] [FILV] [ACDEFGHIKLNPQRSTVY] [CDEFGHIKLNPQRSTVY]; relaxed PEXEL: [R] [INSY] [L] [CGRSY] [AGLY] [DEFKNT]) located anywhere in the C terminal of the signal sequence. The PLMs comprised both canonical pentameric sequences and relaxed, 6-residue motifs, generated by overlapping the known amino acid residues of the PEXEL motif of all known PfPEXEL proteins. The consensus motif was generated by aligning the PEXEL residues from all previously published PfPEXEL proteins to produce a LOGO using weblogo.berkeley.edu. We then searched for PfLS transcripts matching the 589 genes. Genes were selected if expressed at ≥1 TPM (unadjusted) in 2–3 biological replicates for each timepoint.
Generation of PfNF54lisp2- parasites
PfNF54lisp2− were generated in the PfNF54 WT via CRISPR/Cas9-mediated gene editing. Sorbitol (5%) synchronized ring-stage Pf strain NF54 parasites at 5–8% parasitemia were electroporated with 100 µg of pFC-LISP2 plasmid at 0.31 kV and 950 µF using a BioRad Gene Pulser (BioRad). Positive selection with 8 nM WR99210 (WR, Jacobus Pharmaceuticals) was administered 24 h after the transfection and kept for 5 days. Once drug-resistant parasites were visible on thin blood smears, these parasites were screened by PCR. PfNF54lisp2− parasites were cloned out of the mixed parasite pool by limiting dilution in 96-well flat-bottomed plates. Cultures were serially diluted and then plated at a density of 0.3 parasites per well in a 100-µl volume at 2% haematocrit. Cultures were fed once a week and fresh blood was added every 3–4 days at 0.3% haematocrit. The wells were screened for the presence of parasites using primers recognizing Pf18S RNA on day [18SF (5′ AACCTGGTTGATCCAGTAGTCATATG 3′) and 18SR (5′ CCAAAAATTGGCCTTGCATTGTTAT 3′)]. Positive wells were expanded and screened for recombination using the PCR strategies used above and using Southern blot analysis as previously described105. Pf lisp2– clones A3 and H1 were used for further phenotypic analysis. For evaluation of LS development, 750,000 Pf NF54 sporozoites and 750,000 PfNF54lisp2- sporozoites were injected intravenously (retro-orbital) into 4 and 5 FRG huHep mice per group, respectively. Livers were collected on days 6 and 7 and used for immunofluorescence assay (IFA) and analysis of liver burden by RT–qPCR. To evaluate for BS transition in FRG huHep mice, 750,000 PfNF54 sporozoites and 750,000 PfNF54lisp2- sporozoites were injected intravenously (retro-orbital) into 3 and 15 FRG huHep mice per group, respectively. On days 6 and 7, 400 µl of 70% RBCs were injected intravenously to enable transition of LS parasites to blood. Four hours after human RBC repopulation on day 7, mice were euthanized, blood was collected by cardiac puncture, 50 µl of blood from each mouse was used for RT–qPCR analysis to detect parasite 18S RNA and livers were collected for RNA extraction to quantify parasite liver load. Blood was washed three times in asexual media, a volume of human RBCs equal to the packed RBC volume was added and blood was transferred to in vitro culture. Fresh media were replaced daily and cultures were analysed every 2–3 days by thick smear for the presence of parasites for up to 6 weeks. Samples from in vitro culture were analysed for the presence of 18S rRNA by RT–qPCR after 7 days. For IFA for LS, livers were perfused with 1× PBS, fixed in 4% v/v paraformaldehyde (PFA) in 1× PBS and lobes were cut into 50-μm sections using a Vibratome apparatus (Ted Pella). For IFA, sections were permeabilized in 1× TBS containing 3% v/v H2O2 and 0.25% v/v Triton X-100 for 30 min at room temperature. Sections were then blocked in 1× TBS containing 5% v/v dried milk (TBS-M) for at least 1 h and incubated with primary antibody in TBS-M at 4 °C overnight. After washing in 1× TBS, fluorescent secondary antibodies with 1 μg ml−1 4′, 6-diamidino-2-phenylindole (DAPI) were added in TBS-M for 2 h at room temperature in a similar manner as above. After further washing, the section was incubated in 0.06% w/v KMnO4 for 2 min to quench background fluorescence and mounted with FluoroGuard anti-fade reagent (BioRad).
Primary antibodies used included anti-Pf CSP clone 2A10 (1:500, BEI Resources, BEI-MRA-183A, mouse monoclonal) and anti-Pf LISP2 (1:500, ref. 66). Secondary antibodies included donkey anti-mouse 488 (Invitrogen, A21202; RRID: AB_141607) and donkey anti-rabbit 594 (Invitrogen, A21207; RRID: AB_141637). All secondary antibodies were used at a dilution of 1:1,000.
PfNF54AP2GGFP transgenic parasite line and sexual conversion assay
The PfNF54AP2GGFP transgenic parasite line80 was cloned using a serial dilution method in a 96-well plate. Parasites that developed from as low as 0.25 parasites per well were selected for screening. Six clones were randomly selected, expanded and subjected to genotyping PCR using the same set of primers used in the original publication80. Clone G8 (Extended Data Fig. 7b) was selected for further propagation and used in downstream experiments. To determine the sexual conversion rate of the PfNF54AP2GGFP line, sexual commitment was induced using the nutrient deprivation induction method as previously described80. Briefly, parasites were tightly synchronized using Percoll (63% isotonic solution) density-gradient centrifugation to isolate mature-stage schizonts and allow invasion of naïve RBCs, followed by treatment with 5% sorbitol to kill the remaining schizonts in the culture, retaining only young rings (Day 0). Parasites were plated at 0.5% parasitemia and 2.5% haematocrit. To induce sexual commitment, the parasites were shifted to a minimal fatty acid (mFA) medium at 20–24 h post induction (day 1). mFA media were prepared by supplementing incomplete RPMI-1640 (complete medium without AlbuMAXII) with 0.39% fatty acid-free bovine serum albumin (BSA, Sigma-Aldrich), 30 µM oleic acid (Sigma-Aldrich) and 30 µM palmitic acid (Sigma-Aldrich). After 22–26 h post induction (day 2), media were replaced back to complete RPMI media. On day 3, parasitemia was quantified and the parasite multiplication rate was calculated using the formula: PMR = (day 3 parasitemia/day 1 parasitemia) × 100. At this point, 20 U ml−1 heparin was added to the culture until day 6. The medium was changed daily until quantification of gametocytemia on day 7. Sexual conversion rate was calculated using the formula: (day 7 gametocytemia/day 3 parasitemia) × 100.
Evaluation of PfNF54AP2GGFP expression during the LS-to-BS transition by flow cytometry
The FRG huHep mice were intravenously injected with 1.6 million PfNF54AP2GGFP sporozoites. On days 6 and 7, 400 μl of 70% human RBCs were intravenously injected to enable transition of LS parasites to blood as previously described67. Mice were euthanized at 4 h post human RBC repopulation on day 7, and blood was collected through cardiac puncture and further washed in RPMI containing 10% Albumax. A small aliquot of blood was taken for flow cytometry analysis, and the remaining blood was transferred to in vitro culture to reach 2% initial haematocrit. Fresh media with blood were replaced daily, gradually increasing the haematocrit to support parasite growth. On days 2, 4 and 6 of in vitro culture, 20-µl aliquots were sampled for flow cytometry analysis. The cells were resuspended in 1% FACS buffer (1× PBS with 1% fetal bovine serum) and incubated with mouse Fc block (Biolegend, 156604; 1:100 dilution) for 10 min at room temperature, followed by staining with anti-mouse CD45-PECy7 antibody (Biolegend, 103113; clone 30-F11; 1:100 dilution) for 10 min at room temperature on a shaker (70 r.p.m.). Cells were washed with 1% FACS buffer and then incubated with SPY650-DNA dye (Cytoskeleton, CY-SC501; 1:1,000 dilution) for 1 h on a shaker (70 r.p.m.) in a 37 °C incubator. Cells were washed twice, resuspended in 1% FACS buffer and immediately acquired in a BD FACS LSRII flow cytometer. The data were collected using BD FACSDiva software (v.8.0.1) and analysed using BD FlowJo software (v.10.10.0).
Live imaging, immunofluorescence assay and RNA-FISH
Live images were captured using a Keyence BZ-X700 fluorescence microscope, and Fiji software was used for the image analysis.
For the immunofluorescence assay, livers were collected from Pf-infected FRG huHep mice on day 7 PI and fixed in 4% v/v PFA (Alfa Aesar) in 1× PBS. Sections (50 µm) or cryosections (5 µm) of the liver were blocked in normal goat serum diluted at 1:500 for 2 h, washed twice in PBS and incubated overnight with anti-PfHSP70 antibody (1:1,000, rabbit polyclonal, Stressmarq PC-186), anti-PfATS antibody (1:500, rabbit polyclonal, Antibody Facility, Walter Eliza Hall Institute of Medical Research Biotechnology Centre, Melbourne, Australia), anti-PfCSP clone 2A10 (1:500, mouse monoclonal, BEI Resources, BEI-MRA-183A), anti-PfEXP2 (1:500, rabbit polyclonal), anti-PfPTEX150 (1:250, rabbit polyclonal), anti-PfHSP101 (1:250, rabbit polyclonal), anti-mTIP (1:500, rabbit polyclonal) and anti-MSP1 clone 12.1 (1:500, mouse monoclonal, European Malaria Reagent Repository, http://www.malariaresearch.eu), then washed twice in PBS, followed by incubation with secondary antibodies and DAPI for 2 h. All antibodies were diluted in PBS containing 1% BSA and 0.2% Triton X-100. All sections were washed twice in PBS before being mounted in anti-fading medium, and stored at 4 °C before analysis. Images were captured using the GE DeltaVision Elite optical/digital-sectioning fluorescence microscope, and Fiji software was used for the image analysis.
RNA-FISH was performed on BS schizonts enriched using magnetic-activated cell sorting (MACS) as well as LS schizonts obtained from cryosections of the liver of FRG huHep mice infected with P. falciparum (day 7 PI) and P. vivax (day 8 PI), using the RNAscope Multiplex Fluorescent Assay v.2 kit (Advanced Cell Diagnostics) essentially according to manufacturer instructions. Protease digestions were performed using pretreatment solution 3 from the kit at 1:10 dilution for 20 min at room temperature for BS and protease IV for LS parasites. Hybridizations were 2 h at 40 °C. Probes used were directed against PfLisp1 (PF3D7_1418100, 20ZZ pairs probe targeting regions 1,933–10,162), PfAP2-EXP2 (PF3D7_0611200, 7ZZ pairs probe targeting regions 38–833), PfAP2g (PF3D7_1222600, 20ZZ pairs probe targeting regions 2–6,287) and PvAP2g (PVP01_1440800, 20ZZ pairs probe targeting regions 161–1,645). After hybridization, tyramide signal amplification (TSA) steps with TSA Vivid Fluorophores were performed as described by the manufacturer. Following DAPI staining, slides were mounted, and images were acquired using the Stellaris 8 confocal microscope (Leica Microsystems) and processed using the Lightning software (Leica Microsystems).
Statistics and reproducibility
All experiments were performed with a minimum of three independent biological replicates to ensure statistical reliability and reproducibility. For each immunofluorescence experiment, consistent results were observed across replicates. Details on statistical tests, sample sizes and P-value thresholds are provided in the corresponding figure legends and in Methods.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Liver-stage transcriptome.
Liver stage exported proteins.
Sample manifest.
Acknowledgements
We thank the Seattle Children’s Research Institute Research Scientific Computing Infrastructure for bioinformatic support; S. Kumar for the initial culture and propagation of the PfNF54AP2GGFP parasite line; the vivarium staff at Seattle Children’s Research Institute for their support during animal studies and the insectary staff for mosquito rearing. This work was funded by NIH U01 AI155335 to S.H.I.K., NHMRC Investigator Grant 1176955 to J.A.B. and a BMGF grant (OPP1137694) to S.H.I.K. and S.A.M.
Extended data
Author contributions
G.Z., H.P., E.L.F., S.A.M., A.M.V. and S.H.I.K. conceptualized the project. G.Z., E.L.F., H.P., J.L.S., N.C., A.M.V. and S.H.I.K. developed the methodology. G.Z., H.P., J.L.S., N.C., Y.B., E.H., J.A.B., E.L.F., V.C., P.G., L.P., N.R., W.R., J.S., D.G., W.B., K.V., M.M., M.E.F., S.A.M., K.H., A.M.V. and S.H.I.K. conducted investigations. G.Z., J.L.S., H.P., N.C., M.M., K.V., E.H., J.A.B., A.M.V. and S.H.I.K. wrote and edited the manuscript. S.H.I.K. and S.A.M. acquired funding and resources.
Peer review
Peer review information
Nature Microbiology thanks Moriya Tsuji and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Data availability
The accession number for the RNA-seq fastq files generated in this study is GEO GSE220039. Please refer to Supplementary Table 3 for associated sample identifiers; biological and technical replicate samples are identified by matching ‘RNA_label’.
Competing interests
E.L.F., V.C. and M.E.F. are employees of Novartis Pharma AG. The remaining authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Gigliola Zanghí, Hardik Patel, Jenny L. Smith.
Contributor Information
Gigliola Zanghí, Email: gigliola.zanghi@seattlechildrens.org.
Ashley M. Vaughan, Email: ashley.vaughan@seattlechildrens.org
Stefan H. I. Kappe, Email: stefan.kappe@seattlechildrens.org
Extended data
is available for this paper at 10.1038/s41564-024-01905-5.
Supplementary information
The online version contains supplementary material available at 10.1038/s41564-024-01905-5.
References
- 1.World Malaria Report 2022. (World Health Organization, 2022); https://www.who.int/publications/i/item/9789240064898
- 2.Roux, A. T. et al. Chloroquine and sulfadoxine-pyrimethamine resistance in sub-Saharan Africa—a review. Front. Genet.12, 668574 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ashley, E. A. et al. Spread of artemisinin resistance in Plasmodium falciparum malaria. N. Engl. J. Med.371, 411–423 (2014). [DOI] [PMC free article] [PubMed]
- 4.Vaughan, A. M. & Kappe, S. H. I. Malaria parasite liver infection and exoerythrocytic biology. Cold Spring Harb. Perspect. Med.7, a025486 (2017). [DOI] [PMC free article] [PubMed]
- 5.Graumans, W., Jacobs, E., Bousema, T. & Sinnis, P. When is a Plasmodium-infected mosquito an infectious mosquito? Trends Parasitol.36, 705–716 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Itsara, L. S. et al. The development of whole sporozoite vaccines for Plasmodium falciparum malaria. Front. Immunol.9, 2748 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Richie, T. L. et al. Progress with Plasmodium falciparum sporozoite (PfSPZ)-based malaria vaccines. Vaccine33, 7452–7461 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hoffman, S. L. & Doolan, D. L. Malaria vaccines-targeting infected hepatocytes. Nat. Med.6, 1218–1219 (2000). [DOI] [PubMed] [Google Scholar]
- 9.Duffy, P. E., Gorres, J. P., Healy, S. A. & Fried, M. Malaria vaccines: a new era of prevention and control. Nat. Rev. Microbiol.10.1038/s41579-024-01065-7 (2024). [DOI] [PubMed] [Google Scholar]
- 10.Berenzon, D. et al. Protracted protection to Plasmodium berghei malaria is linked to functionally and phenotypically heterogeneous liver memory CD8+ T cells. J. Immunol.171, 2024–2034 (2003). [DOI] [PubMed] [Google Scholar]
- 11.Van Braeckel-Budimir, N. & Harty, J. T. CD8 T-cell-mediated protection against liver-stage malaria: lessons from a mouse model. Front. Microbiol.5, 272 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sahu, T. et al. Chloroquine neither eliminates liver stage parasites nor delays their development in a murine chemoprophylaxis vaccination model. Front. Microbiol.6, 283 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mwakingwe-Omari, A. et al. Two chemoattenuated PfSPZ malaria vaccines induce sterile hepatic immunity. Nature595, 289–294 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Butler, N. S. et al. Superior antimalarial immunity after vaccination with late liver stage-arresting genetically attenuated parasites. Cell Host Microbe9, 451–462 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mazier, D., Rénia, L. & Snounou, G. A pre-emptive strike against malaria’s stealthy hepatic forms. Nat. Rev. Drug Discov.8, 854–864 (2009). [DOI] [PubMed] [Google Scholar]
- 16.van der Boor, S. C. et al. Mid-liver stage arrest of Plasmodium falciparum schizonts in primary porcine hepatocytes. Front. Cell. Infect. Microbiol.12, 834850 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ng, S. et al. Human iPSC-derived hepatocyte-like cells support Plasmodium liver-stage infection in vitro. Stem Cell Rep.4, 348–359 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.March, S. et al. A microscale human liver platform that supports the hepatic stages of Plasmodium falciparum and vivax. Cell Host Microbe14, 104–115 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gural, N. et al. In vitro culture, drug sensitivity, and transcriptome of Plasmodium vivax hypnozoites. Cell Host Microbe23, 395–406.e4 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ruberto, A. A. et al. Single-cell RNA profiling of Plasmodium vivax-infected hepatocytes reveals parasite- and host-specific transcriptomic signatures and therapeutic targets. Front. Cell. Infect. Microbiol.12, 986314 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vaughan, A. M. et al. Complete Plasmodium falciparum liver-stage development in liver-chimeric mice. J. Clin. Invest.122, 3618–3628 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Soulard, V. et al. Plasmodium falciparum full life cycle and Plasmodium ovale liver stages in humanized mice. Nat. Commun.6, 7690 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mikolajczak, S. A. et al. Plasmodium vivax liver stage development and hypnozoite persistence in human liver-chimeric mice. Cell Host Microbe17, 526–535 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Goswami, D. et al. A replication-competent late liver stage-attenuated human malaria parasite. JCI Insight5, e135589 (2020). [DOI] [PMC free article] [PubMed]
- 25.Schäfer, C. et al. Partial protection against P. vivax infection diminishes hypnozoite burden and blood-stage relapses. Cell Host Microbe29, 752–756.e754 (2021). [DOI] [PubMed] [Google Scholar]
- 26.Kumar, S. et al. A slot blot immunoassay for quantitative detection of Plasmodium falciparum circumsporozoite protein in mosquito midgut oocyst. PLoS ONE9, e115807 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zanghi, G. et al. A specific PfEMP1 is expressed in P. falciparum sporozoites and plays a role in hepatocyte infection. Cell Rep22, 2951–2963 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goswami, D., Minkah, N. K. & Kappe, S. H. I. Malaria parasite liver stages. J. Hepatol.76, 735–737 (2022). [DOI] [PubMed] [Google Scholar]
- 29.Tarun, A. S., Vaughan, A. M. & Kappe, S. H. Redefining the role of de novo fatty acid synthesis in Plasmodium parasites. Trends Parasitol.25, 545–550 (2009). [DOI] [PubMed] [Google Scholar]
- 30.Shears, M. J. et al. Characterization of the Plasmodium falciparum and P. berghei glycerol 3-phosphate acyltransferase involved in FASII fatty acid utilization in the malaria parasite apicoplast. Cell. Microbiol.19, e12633 (2017). [DOI] [PMC free article] [PubMed]
- 31.Falkard, B. et al. A key role for lipoic acid synthesis during Plasmodium liver stage development. Cell. Microbiol.15, 1585–1604 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hillier, C. et al. Landscape of the Plasmodium interactome reveals both conserved and species-specific functionality. Cell Rep.28, 1635–1647.e5 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cui, L., Lindner, S. & Miao, J. Translational regulation during stage transitions in malaria parasites. Ann. N. Y. Acad. Sci.1342, 1–9 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Voss, T. S., Bozdech, Z. & Bártfai, R. Epigenetic memory takes center stage in the survival strategy of malaria parasites. Curr. Opin. Microbiol.20, 88–95 (2014). [DOI] [PubMed] [Google Scholar]
- 35.Campbell, T. L., De Silva, E. K., Olszewski, K. L., Elemento, O. & Llinás, M. Identification and genome-wide prediction of DNA binding specificities for the ApiAP2 family of regulators from the malaria parasite. PLoS Pathog.6, e1001165 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Balaji, S., Babu, M. M., Iyer, L. M. & Aravind, L. Discovery of the principal specific transcription factors of Apicomplexa and their implication for the evolution of the AP2-integrase DNA binding domains. Nucleic Acids Res.33, 3994–4006 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Josling, G. A., Williamson, K. C. & Llinás, M. Regulation of sexual commitment and gametocytogenesis in malaria parasites. Annu. Rev. Microbiol.72, 501–519 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Santos, J. M. et al. Red blood cell invasion by the malaria parasite is coordinated by the PfAP2-I transcription factor. Cell Host Microbe21, 731–741.e10 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Murata, Y., Nishi, T., Kaneko, I., Iwanaga, S. & Yuda, M. Coordinated regulation of gene expression in Plasmodium female gametocytes by two transcription factors. eLife12, RP88317 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Modrzynska, K. et al. A knockout screen of ApiAP2 genes reveals networks of interacting transcriptional regulators controlling the Plasmodium life cycle. Cell Host Microbe21, 11–22 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Carrington, E. et al. The ApiAP2 factor PfAP2-HC is an integral component of heterochromatin in the malaria parasite. iScience24, 102444 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Iwanaga, S., Kaneko, I., Kato, T. & Yuda, M. Identification of an AP2-family protein that is critical for malaria liver stage development. PLoS ONE7, e47557 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shang, X. et al. A cascade of transcriptional repression determines sexual commitment and development in Plasmodium falciparum. Nucleic Acids Res.49, 9264–9279 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Toro-Moreno, M., Sylvester, K., Srivastava, T., Posfai, D. & Derbyshire, E. R. RNA-seq analysis illuminates the early stages of Plasmodium liver infection. mBio11, e03234-19 (2020). [DOI] [PMC free article] [PubMed]
- 45.Yuda, M., Iwanaga, S., Kaneko, I. & Kato, T. Global transcriptional repression: an initial and essential step for Plasmodium sexual development. Proc. Natl Acad. Sci. USA112, 12824–12829 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Painter, H. J., Campbell, T. L. & Llinas, M. The apicomplexan AP2 family: integral factors regulating Plasmodium development. Mol. Biochem. Parasitol.176, 1–7 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Li, L., Stoeckert, C. J. & Roos, D. S. OrthoMCL: identification of ortholog groups for eukaryotic genomes. Genome Res.13, 2178–2189 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bischoff, E. & Vaquero, C. In silico and biological survey of transcription-associated proteins implicated in the transcriptional machinery during the erythrocytic development of Plasmodium falciparum. BMC Genomics11, 34 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Boddey, J. A., Moritz, R. L., Simpson, R. J. & Cowman, A. F. Role of the Plasmodium export element in trafficking parasite proteins to the infected erythrocyte. Traffic10, 285–299 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Marti, M., Good, R. T., Rug, M., Knuepfer, E. & Cowman, A. F. Targeting malaria virulence and remodeling proteins to the host erythrocyte. Science306, 1930–1933 (2004). [DOI] [PubMed] [Google Scholar]
- 51.Hiller, N. L. et al. A host-targeting signal in virulence proteins reveals a secretome in malarial infection. Science306, 1934–1937 (2004). [DOI] [PubMed] [Google Scholar]
- 52.Russo, I. et al. Plasmepsin V licenses Plasmodium proteins for export into the host erythrocyte. Nature463, 632–636 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Boddey, J. A. et al. An aspartyl protease directs malaria effector proteins to the host cell. Nature463, 627–631 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sargeant, T. J. et al. Lineage-specific expansion of proteins exported to erythrocytes in malaria parasites. Genome Biol.7, R12 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Spielmann, T. & Gilberger, T. W. Protein export in malaria parasites: do multiple export motifs add up to multiple export pathways? Trends Parasitol.26, 6–10 (2010). [DOI] [PubMed] [Google Scholar]
- 56.Heiber, A. et al. Identification of new PNEPs indicates a substantial non-PEXEL exportome and underpins common features in Plasmodium falciparum protein export. PLoS Pathog.9, e1003546 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.de Koning-Ward, T. F. et al. A newly discovered protein export machine in malaria parasites. Nature459, 945–949 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Beck, H. P. A traffic jam to reduce morbidity in malaria. Blood124, 3342–3343 (2014). [DOI] [PubMed] [Google Scholar]
- 59.Garten, M. et al. EXP2 is a nutrient-permeable channel in the vacuolar membrane of Plasmodium and is essential for protein export via PTEX. Nat. Microbiol.3, 1090–1098 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hussain, T. et al. The PTEX pore component EXP2 is important for intrahepatic development during the Plasmodium liver stage. mBio13, e0309622 (2022). [DOI] [PMC free article] [PubMed]
- 61.Beck, J. R., Muralidharan, V., Oksman, A. & Goldberg, D. E. PTEX component HSP101 mediates export of diverse malaria effectors into host erythrocytes. Nature511, 592–595 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Matthews, K. M., Kalanon, M. & de Koning-Ward, T. F. Uncoupling the threading and unfoldase actions of Plasmodium HSP101 reveals differences in export between soluble and insoluble proteins. mBio10, e01106-19 (2019). [DOI] [PMC free article] [PubMed]
- 63.Schulze, J. et al. The Plasmodium falciparum exportome contains non-canonical PEXEL/HT proteins. Mol. Microbiol.97, 301–314 (2015). [DOI] [PubMed] [Google Scholar]
- 64.Boddey, J. A. et al. Role of plasmepsin V in export of diverse protein families from the Plasmodium falciparum exportome. Traffic14, 532–550 (2013). [DOI] [PubMed] [Google Scholar]
- 65.Orito, Y. et al. Liver-specific protein 2: a Plasmodium protein exported to the hepatocyte cytoplasm and required for merozoite formation. Mol. Microbiol.87, 66–79 (2013). [DOI] [PubMed] [Google Scholar]
- 66.McConville, R. et al. Flp/FRT-mediated disruption of ptex150 and exp2 in Plasmodium falciparum sporozoites inhibits liver-stage development. Proc. Natl Acad. Sci. USA121, e2403442121 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Goswami, D. et al. A replication competent Plasmodium falciparum parasite completely attenuated by dual gene deletion. EMBO Mol. Med.16, 723–754 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Auburn, S. et al. A new Plasmodium vivax reference sequence with improved assembly of the subtelomeres reveals an abundance of pir genes. Wellcome Open Res.1, 4 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Smith, J. D. The role of PfEMP1 adhesion domain classification in Plasmodium falciparum pathogenesis research. Mol. Biochem. Parasitol.195, 82–87 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Real, E. et al. A single-cell atlas of Plasmodium falciparum transmission through the mosquito. Nat. Commun.12, 3196 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Reid, A. J. et al. Single-cell RNA-seq reveals hidden transcriptional variation in malaria parasites. Elife7, e33105 (2018). [DOI] [PMC free article] [PubMed]
- 72.Gómez-Díaz, E. et al. Epigenetic regulation of Plasmodium falciparum clonally variant gene expression during development in Anopheles gambiae. Sci. Rep.7, 40655 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lavstsen, T., Salanti, A., Jensen, A. T., Arnot, D. E. & Theander, T. G. Sub-grouping of Plasmodium falciparum 3D7 var genes based on sequence analysis of coding and non-coding regions. Malar. J.2, 27 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Roth, A. et al. A comprehensive model for assessment of liver stage therapies targeting Plasmodium vivax and Plasmodium falciparum. Nat. Commun.9, 1837 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Schafer, C. et al. A humanized mouse model for Plasmodium vivax to test interventions that block liver stage to blood stage transition and blood stage infection. iScience23, 101381 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Mancio-Silva, L. et al. A single-cell liver atlas of Plasmodium vivax infection. Cell Host Microbe30, 1048–1060.e5 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Brancucci, N. M. B. et al. Heterochromatin protein 1 secures survival and transmission of malaria parasites. Cell Host Microbe16, 165–176 (2014). [DOI] [PubMed] [Google Scholar]
- 78.Kafsack, B. F. et al. A transcriptional switch underlies commitment to sexual development in malaria parasites. Nature507, 248–252 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Gomes, A. R. et al. A transcriptional switch controls sex determination in Plasmodium falciparum. Nature612, 528–533 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Brancucci, N. M. B. et al. Lysophosphatidylcholine regulates sexual stage differentiation in the human malaria parasite Plasmodium falciparum. Cell171, 1532–1544.e15 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lindner, S. E. et al. Transcriptomics and proteomics reveal two waves of translational repression during the maturation of malaria parasite sporozoites. Nat. Commun.10, 4964 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Vaughan, A. M. et al. Type II fatty acid synthesis is essential only for malaria parasite late liver stage development. Cell. Microbiol.11, 506–520 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Guggisberg, A. M., Amthor, R. E. & Odom, A. R. Isoprenoid biosynthesis in Plasmodium falciparum. Eukaryot. Cell13, 1348–1359 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gomes, A. R. Q. et al. Oxidative stress in malaria: potential benefits of antioxidant therapy. Int. J. Mol. Sci. 23, 5949 (2022). [DOI] [PMC free article] [PubMed]
- 85.Singh, A. P. et al. Plasmodium circumsporozoite protein promotes the development of the liver stages of the parasite. Cell131, 492–504 (2007). [DOI] [PubMed] [Google Scholar]
- 86.Cockburn, I. A. et al. Dendritic cells and hepatocytes use distinct pathways to process protective antigen from plasmodium in vivo. PLoS Pathog.7, e1001318 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Montagna, G. N. et al. Antigen export during liver infection of the malaria parasite augments protective immunity. mBio5, e01321-14 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Bachmann, A. et al. Mosquito passage dramatically changes var gene expression in controlled human Plasmodium falciparum infections. PLoS Pathog.12, e1005538 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Pickford, A. K. et al. Expression patterns of Plasmodium falciparum clonally variant genes at the onset of a blood infection in malaria-naive humans. mBio12, e0163621 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Neafsey, D. E. et al. The malaria parasite Plasmodium vivax exhibits greater genetic diversity than Plasmodium falciparum. Nat. Genet.44, 1046–1050 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Talman, A. M., Blagborough, A. M. & Sinden, R. E. A Plasmodium falciparum strain expressing GFP throughout the parasite’s life-cycle. PLoS ONE5, e9156 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Crabb, B. S. et al. Transfection of the human malaria parasite Plasmodium falciparum. Methods Mol. Biol.270, 263–276 (2004). [DOI] [PubMed] [Google Scholar]
- 93.Ponnudurai, T. et al. Infectivity of cultured Plasmodium falciparum gametocytes to mosquitoes. Parasitology98, 165–173 (1989). [DOI] [PubMed] [Google Scholar]
- 94.Charni-Natan, M. & Goldstein, I. Protocol for primary mouse hepatocyte isolation. STAR Protoc.1, 100086 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pruitt, K. D. et al. RefSeq: an update on mammalian reference sequences. Nucleic Acids Res.42, D756–D763 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Church, D. M. et al. Modernizing reference genome assemblies. PLoS Biol.9, e1001091 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Gardner, M. J. et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature419, 498–511 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zhang, Y., Parmigiani, G. & Johnson, W. E. ComBat-seq: batch effect adjustment for RNA-seq count data. NAR Genom. Bioinform.2, lqaa078 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res.43, e47 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Robinson, M. D. & Oshlack, A. A scaling normalization method for differential expression analysis of RNA-seq data. Genome Biol.11, R25 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Nueda, M. J., Tarazona, S. & Conesa, A. Next maSigPro: updating maSigPro bioconductor package for RNA-seq time series. Bioinformatics30, 2598–2602 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Gao, C. H., Yu, G. & Cai, P. ggVennDiagram: an intuitive, easy-to-use, and highly customizable R package to generate Venn diagram. Front. Genet.12, 706907 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wu, T. et al. clusterProfiler 4.0: a universal enrichment tool for interpreting omics data. Innovation2, 100141 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Machlab, D. et al. monaLisa: an R/Bioconductor package for identifying regulatory motifs. Bioinformatics38, 2624–2625 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Mikolajczak, S. A. et al. A next-generation genetically attenuated Plasmodium falciparum parasite created by triple gene deletion. Mol. Ther.22, 1707–1715 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Liver-stage transcriptome.
Liver stage exported proteins.
Sample manifest.
Data Availability Statement
The accession number for the RNA-seq fastq files generated in this study is GEO GSE220039. Please refer to Supplementary Table 3 for associated sample identifiers; biological and technical replicate samples are identified by matching ‘RNA_label’.