Abstract
Human milk, the gold standard for infant nutrition, precisely captures infant needs. This study analyzed the particle size distribution, fatty acid (FA) profile, and milk fat globule membrane (MFGM) proteome of milk fat globules (MFGs) in human milk and eight types of animal milks. Donkey and horse milks showed a greater abundance of smaller MFGs (0–5 μm). Human milk was richer in monounsaturated FAs, such as oleic acid and nervonic acid, whereas donkey had the most polyunsaturated FAs like linoleic acid and cis-11,14-eicosadienoic acid. Among the identified 1253 MFGM proteins, human milk showed significantly higher abundance of several novel immune enhancers including adiponectin B and vitronectin, antioxidases, and lipid metabolism regulators. Meanwhile, sheep milk and yak milk displayed greater abundance of zinc-α-2-glycoprotein and selenoprotein F, respectively. This study revealed physicochemical differences in MFGs between humans and eight types of animals, offering insights for improving animal-milk-based formulas for infants' nutritional needs.
Keywords: Human milk, Dairy animal milks, Fat globules, Proteomics, Fatty acid
Highlights
-
•
Studied the composition differences in milk fat globules of human and animals.
-
•
Smaller milk fat globules were observed in the milk of donkey and horse.
-
•
Human milk was rich in monounsaturated fatty acids and donkey in polyunsaturated.
-
•
Human milk notably contained immune enhancers, antioxidases and lipid-regulator.
1. Introduction
The composition of human milk is formed by natural selection acting on infant and mother, which meets the nutritional needs of the infant's growth and development to the greatest extent. It is thus recognized as a normative standard for infant feeding (Ballard & Morrow, 2013). On the other hand, the majority of dairy products in the world are based on bovine milk, such as formula for non-breastfeeding infants. Nevertheless, special milks, including those from goat, camel, horse, and donkey, are gaining attention in dietary nutrition because of their lower allergenicity and enhanced immunological activity compared with bovine milk (Muthukumaran, Mudgil, Baba, Ayoub, & Maqsood, 2023; Salimei & Fantuz, 2012). The comprehensive elucidation of distinctions between human and animal milks holds the potential to enhance the exploitation of diverse milk sources as nutritional resources, particularly in the formulation of infant food.
Milk lipids are important nutrients and energy sources, accounting for about 4 %–5 % of milk, and they are mostly present as fat globules, which are packaged and released by the mammary epithelial cells (Singh & Gallier, 2017). The triglyceride core of milk fat globules (MFGs) is surrounded by a three-layer membrane structure composed of phospholipids or cholesterol and protein. This complex membrane system, which plays the role of an emulsifier, is called milk fat globule membrane (MFGM) (Liao, Alvarado, Phinney, & Lönnerdal, 2011). The important functions and nutritional characteristics of MFGs in early infant development cannot be ignored. Lipids, which are the primary macromolecules in milk, play a pivotal role in supplying essential fatty acids (FAs), phospholipids, and fat-soluble vitamins. Additionally, certain FAs mediate the secretion of hormones involved in growth and fundamental metabolic functions (Hadley, Ryan, Forsyth, Gautier, & Salem, 2016). Interestingly, although MFGM proteins account for only 1 %–4 % of the total protein content of milk, they exhibit a higher degree of complexity compared to other milk fractions and are involved in a wide range of biological functions, including membrane/protein transport, developmental promotion, and antibacterial effect (Liao et al., 2011). Previous studies have revealed the anticancer activity of the MFGM: it can reduce the proliferation of cancer cells and promote their apoptosis by activating the effector caspase-3 (Ji, Xu, Cui, Ma, & Zhou, 2019; Zanabria, Griffiths, & Corredig, 2020).
MFGs are intricately linked to the stability and technical characteristics of milk, especially for infant fat digestion (Michalski, Briard, Michel, Tasson, & Poulain, 2005). Previous studies have shown that compared with human MFGs, buffalo MFGs are larger, while bovine and infant formula with added emulsified vegetable oils are smaller. Compared with human colostrum, the particle size of MFGs in human mature milk is smaller (Ménard et al., 2010; Michalski et al., 2005). Triglycerides are the primary component of milk fat (98.3 %), and the FAs that constitute triglycerides are significantly influenced by species. The distinctive feature of human milk lies in its elevated concentrations of polyunsaturated fatty acids (PUFAs), including docosahexaenoic acid and arachidonic acid (Wang et al., 2022), which confer numerous health benefits, especially in helping the healthy development of the neonatal brain and nervous system (Zhang et al., 2018). Ruminant milk is characterized by its richness in saturated fatty acids (SFAs) including caproic acid (C6:0), undecanoic acid (C11:0), palmitic acid (C16:0), and trans-octadecenoic acids, which are present in smaller amounts in human milk (Wang et al., 2022; Zou et al., 2013). It is essential to underscore that existing research on MFGs in the context of comparative analyses between human and dairy animals remains notably limited and lacks comprehensive coverage. This is particularly evident in the scarcity of fully comparative studies focusing on the FA composition and particle size of MFGs of human and non-ruminant species.
Proteomics has emerged as a reliable method for the comprehensive characterization of milk proteins. Some major proteins in MFGM of human and dairy animals, including xanthine dehydrogenase/oxidase (XDH), lactadherin, and butyrophilin subfamily 1 member A1 (BTN1A1), have been widely identified (Manoni, Di Lorenzo, Ottoboni, Tretola, & Pinotti, 2020). An early study characterized the dynamic changes of the human MFGM proteome during the 12-month lactation period, thus accurately clarifying the nutritional needs of infants (Liao et al., 2011). Subsequent study systematically characterized the MFGM proteomes of human, ruminants, camel, and horse, showing that the MFGM glycoproteins were closely related to the stimulus response (Yang et al., 2016). Other similar studies identified the MFGM proteomes of human, goat, cow, and yak and illustrated a greater presence of immunity-related proteins and lipid metabolism enzymes in human MFGM, including carboxyl ester lipase and hormone-sensitive lipase (Lu et al., 2016; Yang et al., 2015). Recent studies have analyzed MFGM proteomes of bovine, buffalo, yak, goat, and camel on the basis of nontarget proteomics. Proteins such as α-S1-casein, XDH, and lactoferrin were used for the taxonomic identification of different species (Chen et al., 2023; Han et al., 2022). Although related work has been performed previously, we have observed dispersion and incompleteness in current studies pertaining to the MFGM. Specifically, current research lacks a systematic multispecies comparison of the MFGM proteome and its biological activities, particularly between human milk and milk from dairy species such as sheep and donkey.
This study meticulously collected milk from various species and thoroughly compared the MFGs of eight different species, namely, Holstein cattle, buffalo, yak, goat, sheep, donkey, and horse, with those in human milk. The results of this study will contribute to a comprehensive understanding of the microstructure, FA composition, and proteomic characteristics of MFGs in human and dairy animal milks, providing valuable insights and data support for the future optimization of formula ingredients with respect to MFGs.
2. Materials and methods
2.1. Sample collection
Human milk samples were collected from 21 healthy, non-smoking, and non-drinking volunteer mothers residing in Linyi, Shandong Province, China. The mothers were 25–35 years old and 80–120 days postpartum. The collectors were all trained, and prior to collection, the hands of the collectors as well as the areola and nipple of the volunteer mother were cleaned. Samples were hygienically and thoroughly collected from both breasts in the morning and afternoon by using an automatic breast pump. The collection process was completed within a single day. Written informed consent was obtained from all participants. The milk samples were transported to the laboratory in dry ice and then stored at −80 °C.
Before the dairy animal milk was collected, all animals to be sampled on the farms were ensured to be healthy and in their peak lactation period. Tank milk from Holstein cattle and goat was collected from large farms near Qingdao, Shandong. Tank milk from buffalo was collected from a large farm near Nanning, Guangxi. Tank milks from yak and donkey were collected from farms near Xining, Qinghai, and Liaocheng, Shandong. These milk samples were collected using automatic milking machines in the morning and afternoon to ensure the representativeness of the milk composition.
Milk samples from camel and horse were collected from farms near Urumqi, Xinjiang, and milk samples from sheep were collected from a farm near Hefei, Anhui. Prior to sampling of each animal, the udder was thoroughly cleaned. Trained milkers then immediately collected the milk samples using automatic milking machines. After collection, the milk from individual animals was pooled to ensure that the samples were representative of each species. Milk was collected twice daily, in the morning and afternoon. All animal milk samples were transported to the laboratory in dry ice and then stored at −80 °C.
This study ensured that the work described has been carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki). All experiments involving animals were conducted according to the ethical policies and procedures approved by the Institutional Animal Care and Use Committee at the Institute of Animal Sciences of the Chinese Academy of Agricultural Sciences (approval no. IAS2023–136).
2.2. Chemicals
Methanol (HPLC–MS grade), acetone (HPLC–MS grade), ethylene diamine tetraacetic acid (EDTA), phosphate buffer saline (PBS), Tris, sodium dodecyl sulfate (SDS), hydrochloric acid (guaranteed reagent), dithiothreitol, iodoacetamide, ammonium bicarbonate, and formic acid (HPLC–MS grade) were obtained from Sinopharm Chemical Reagent Co., Ltd. Trypsin (tosyl phenylalanyl chloromethyl ketone -treated) was obtained from Thermo Fisher Scientific (Rockford, IL, USA). The pure water needed in the experiment was ultrapure water prepared at 18.2 MΩ∙cm and 25 °C.
2.3. Determination of MFG particle size distributions
According to the method described by Ménard et al. (2010), the particle size of MFGs in human and dairy animal milks was assessed using a Mastersizer 2000 dynamic light-scattering particle size analyzer (Malvern Instruments, Malvern, UK), which was equipped with a He/Ne laser (λ = 633 nm) and an electroluminescent diode (λ = 466 nm). The milk sample with added EDTA was loaded into the instrument to chelate the calcium ions in the milk, dissociate the casein micelles, and prevent the aggregation of MFGs. Three biological replicates were prepared for each sample.
2.4. Milk FA analysis
The FA in milk from humans and eight dairy animals were determined based on methods described in previous research (Fan et al., 2023). During sample preparation, 2 mL milk samples were mixed with n-hexane/isopropanol and Na2SO4, followed by centrifugation. NaOH (dissolved in CH3OH) was then added to the supernatant, heated at 50 °C, and subsequently acidified with HCl. After additional heating and cooling steps, the samples were extracted three times with n-hexane, diluted to 10 mL with n-hexane, and filtered through a 0.22 μm nylon membrane (Navigator Lab Instrument Co., Ltd., Tianjin, China). The methylated milk samples were placed at 4 °C for further detection.
The composition of FAs in milk was analyzed by gas chromatography (GC) (Agilent 7890 A, USA) equipped with flame ionization detector (FID) and HP-88 column (100 m × 0.25 mm × 0.2 μm; Agilent Technologies). The sample injection volume was 1 μL. The test environment for GC was set as follows: 270 °C injection temperature, 280 °C detector temperature, 25:1 shunt ratio, and 32 psi pressure. The oven temperature profile was as follows: the initial-stage temperature was 100 °C for 5 min, which was then raised to 170 °C at a rate of 10 °C/min and then kept at 170 °C for 8 min. Next, we raised the temperature to 200 °C at a rate of 1 °C/min and held it there for 20 min. Finally, the temperature was raised to 230 °C at a rate of 8 °C/min and kept for 10 min. According to the normalization method described in GB5009.168–2016, a standard mixture of 37 fatty acid methyl esters (FAMEs) (Sigma-Aldrich Chemical Co., St. Louis, MO, USA) was used for the qualitative analysis of FA in the samples, and the chromatographic peaks of the samples were normalized for relative quantification. The chromatogram for the 37 FAME standards is presented in Fig. S1, and their detailed information is listed in Table S1. Six biological replicates were prepared for each sample, with each biological replicate undergoing two technical replicates during GC-FID analysis.
2.5. Separation of MFGM proteins
Milk samples were centrifuged at 4000 ×g for 30 min at 4 °C to collect the upper milk fat. Milk fat was washed with 1 × PBS three times and finally washed with ultrapure water. Next, the milk fat was mixed with 2–3 volumes of 4 % SDS and 100 mM Tris/HCl (pH = 7.6), followed by freeze–thaw cycles to lyse the proteins. The samples were then heated in a 95 °C water bath for 10 min, cooled to 25 °C, and centrifuged at 12,000 ×g for 10 min to collect the proteins. Finally, the protein samples were mixed with 4–5 volumes of precooled acetone and stored at −18 °C for 24 h to precipitate proteins, followed by centrifugation at 4 °C and 9000 ×g for 15 min to collect protein precipitates. The concentrations of MFGM proteins were quantified through a bicinchoninic acid assay using bovine serum albumin as the standard reference. Three biological replicates were prepared for each sample.
2.6. Protein digestion
Following the procedure described by Ji et al. (2024), we extracted the peptide mixture using the filter-aided sample preparation method. In brief, 30 μg of MFGM proteins was reduced with 100 mM dithiothreitol solution and then washed with UT buffer (8 M urea, 100 mM Tris-HCl, pH 8.5). Next, the samples were alkylated with iodoacetamide and digested with trypsin buffer (1 μg of sequencing-grade trypsin in 50 mM NH4HCO3). Finally, the samples were washed with NH4HCO3, desalted, and vacuum-dried to obtain a peptide mixture.
2.7. Proteomic analysis
The peptide mixture was reconstituted with 0.1 % formic acid and analyzed using an Easy nLC 1000 chromatography system coupled with an Orbitrap Fusion Lumos apparatus (Thermo Fisher Scientific, CA, USA). The samples were loaded onto an Aqua C18 trapping column (100 μm × 2 cm, 5 μm; Thermo Fisher Scientific) with solution A (0.1 % FA) at a flow rate of 300 nL/min and separated using an Aqua C18 analytical column (100 mm × 75 μm, 3 μm; Thermo Fisher Scientific). A gradient elution strategy was implemented by increasing the percentage of mobile phase B from 0 % to 10 % over 3 min, from 10 % to 35 % over 45 min, from 35 % to 80 % over 26 min, reaching 100 % within 1 min. A final holding period at 100 % was maintained for the last 15 min.
The proteomic analysis was conducted in positive ion mode, employing a parent ion scanning range from 300 to 1800 mass/charge (m/z) ratio with automatic switching between MS and MS/MS acquisition modes. The MS parameters were configured as follows: resolution set at 60,000, AGC target set to 400,000, maximum injection time set to 50 ms, and exclusion duration set to 40 s. For MS/MS analysis, the top 20 precursor ions with multiple charges were selected, and high-energy collision dissociation was applied (normalized collision energy: 27 eV). The resolution power of the MS/MS scans was 15,000, automated gain control target was 50,000, maximum injection time was 50 ms, dynamic exclusion was 40 s, number of microscans was 1, and RF lens was 30 %.
2.8. Protein characterization
The raw data were acquired using Xcalibur software (Thermo Fisher Scientific) and subsequently processed with MaxQuant (version 2.3.1.0) for protein identification utilizing a label-free quantification (LFQ) workflow. The database for Homo sapiens, Bubalus bubalis, Bos grunniens, Bos taurus, Camelus bactrianus, Capra hircus, Ovis aries, Equus caballus, and Equus asinus were downloaded from UniProt (https://www.uniprot.org/). In the context of protein identification, carbamidomethylation of cysteine was established as a fixed modification, while oxidation of methionine and N-terminal acetylation of proteins were designated as variable modifications. The search parameters included a precursor mass tolerance of 10 ppm, a fragment mass tolerance of 0.1 Da, and allowance for two missed cleavages. Default settings were applied for other conditions. It was imperative to highlight that proteins identified with a minimum of two peptides were deemed acceptable.
2.9. Statistical analysis and bioinformatics
For the MFG particle size, the XY diagram was used to construct the particle size distribution of MFGs. Analysis of variance (ANOVA) of FA and MFG particle sizes was performed using SPSS software package (SPSS, Chicago, IL, USA), with P < 0.05 considered to indicate a significant difference. Differentially abundant proteins were determined according to fold-change (FC) of ≥2 and P < 0.05. Gene Ontology (GO) enrichment and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis of differentially abundant proteins were performed using DAVID Bioinformatics Resources (david.ncifcrf.gov/summary.jsp). For the visualization after data analysis, principal component analysis (PCA) was performed using Perseus software (www.maxquant.org/perseus/), and orthogonal partial-least-squares discriminant analysis (OPLS-DA) of FAs was conducted using Simca 16.1 (Umetrics, Umeå, Sweden). Additionally, visualizations of other analyses were carried out using the R package ggplot2 (v3.4.0).
3. Results
3.1. Particle size distribution of MFGs in human and dairy animal milks
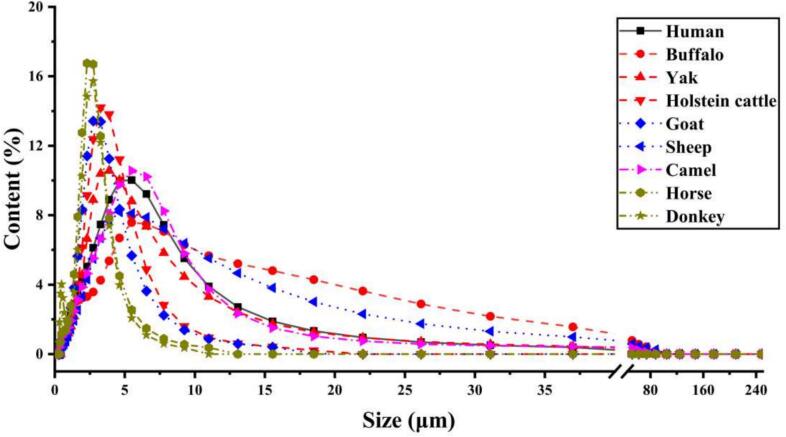
The MFG particle sizes across samples from humans and eight dairy animal species (Table S2) reveal significant interspecies differences in the surface-area-weighted average particle size (D [3,2]). Among them, buffalo and sheep milk exhibited the largest MFGs, followed by human and yak, camel, Holstein cattle, and goat, while donkey milk had the smallest MFGs (1.54 ± 0.01 μm). The volume-weighted average particle size (D [4,3]) showed a slightly different trend, with buffalo milk having the largest MFGs (11.33 ± 0.41 μm), followed by sheep, camel, human, and yak milks, while donkey milk had the smallest (2.61 ± 0.01 μm). Furthermore, Fig. 1 illustrates the comparative analysis of MFG particle size distribution among the species. MFGs in both human and animal milks were broadly distributed between 1 and 10 μm, with donkey and horse milk exhibiting a higher concentration of MFGs within the 1 and 5 μm range, whereas buffalo and sheep milk had a greater proportion of MFGs in the 10–100 μm range. These findings emphasize the substantial interspecies variation in MFG size, particularly the characteristic larger MFGs in buffalo and sheep milk and smaller MFGs in equine milk.
Fig. 1.
Milk fat globule (MFG) particle size distribution in milks from humans and from eight species of dairy animals.
3.2. FA profile
Table S3 presents the ANOVA results of FA analysis of milks from humans and from the eight species of animals. The most significant differences between human and ruminant milk were observed in SFAs and unsaturated fatty acids (UFAs), with human milk exhibiting significantly higher levels (P < 0.05) of oleic acid (C18:1n9c) and palmitoleic acid (C16:1) among the monounsaturated fatty acids (MUFAs), as well as linoleic acid (C18:2n6c) and γ-linolenic acid (C18:3n6) among the PUFAs. Conversely, ruminant milk was characterized by significantly higher levels (P < 0.05) of short chain SFAs and long-chain SFAs, particularly butyric acid (C4:0), C6:0, C16:0, and stearic acid (C18:0), further highlighting the distinct FA composition between human and ruminant milk. Furthermore, we observed a significantly higher level of the FAs myristic acid (C14:0) and C16:0, specifically to buffalo milk as compared to those in human and other animal samples, as well as myristoleic acid (C14:1) and cis-11,14,17-eicosatrienoic acid (C20:3n3) in Holstein cattle milk, tricosanoic acid (C23:0) and cis-13,16-docosadienoic acid (C22:2) in yak milk, and heptadecanoic acid (C17:0) in sheep milk.
The FA composition of non-ruminant horse and donkey milks exhibited characteristics distinct from those of human milk, with the most significant differences observed in LC-FAs and medium chain fatty acids (MC-FAs). Human milk contained significantly higher levels (P < 0.05) of LC-FAs, including C18:1n9c and erucic acid (C22:1n9), compared to horse and donkey milk. Additionally, the MUFA content in horse milk (34.83 %) was comparable to that in human milk (37.30 %); however, horse milk contained significantly higher levels (P < 0.05) of MC-FAs, including caprylic acid (C8:0) and capric acid (C10:0), as well as characteristic fatty acids such as C16:1 and α-linolenic acid (C18:3n3), compared to human milk. In donkey milk, both MC-FAs and PUFAs were significantly higher (P < 0.05) compared to those in human milk. Specifically, C8:0, lauric acid (C12:0), C18:2n6c, and cis-11,14-eicosadienoic acid (C20:2) were identified as characteristic FAs in donkey milk, exhibiting significantly higher levels (P < 0.05) than in human and other animal milks.
The most significant differences between human and camel milk were observed in PUFAs and SFAs. Compared to camel milk, human milk exhibited significantly higher levels (P < 0.05) of PUFAs, including C18:2n6c and C18:3n3. In contrast, SFAs were significantly more abundant in camel milk than in human milk, with C18:0 and arachidic acid (C20:0) being particularly enriched in camel milk, showing significantly higher levels (P < 0.05) than those in human and other animal milks.
Overall, MUFAs were a characteristic FA group in human milk, with significantly higher levels (P < 0.05) than those in animal milks, particularly C18:1n9c, which was markedly enriched in human milk. In contrast, animal milks were distinguished by a higher content of SFAs, including C14:0, C16:0, and C18:0. Additionally, sheep, yak and donkey milks were notably rich in certain odd-chain saturated FAs, further differentiating their FA profiles from human milk.
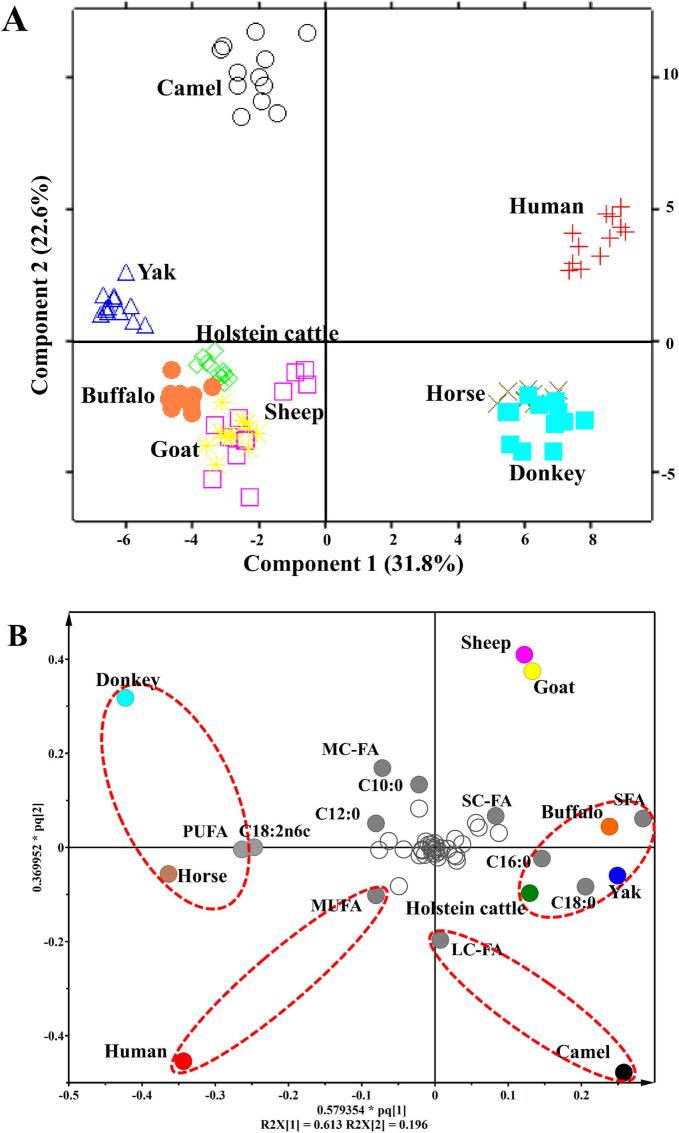
PCA was applied to analyze the FA profile of human and dairy animal milks, as shown in Fig. 2A. According to PCA score plots, ruminants (buffalo, yak, and Holstein cattle as large ruminants; and goat and sheep as small ruminants) exhibited similar FA profiles among themselves; a similar pattern was observed among equine species (horse and donkey). In contrast, human and camel milk were distinctly separated from the others, highlighting their unique FA compositions. Several individual FAs and FA groups served as distinct characteristic traits among different milk types. Specifically, MUFAs in human milk; PUFAs in horse and donkey milks; SFAs (including C16:0 and C18:0) in buffalo, yak, and Holstein cattle; and LC-FAs in camel milk were identified as key differentiating factors. These characteristic FAs were highlighted in the OPLS-DA loading plots (Fig. 2B). The quality of the OPLS-DA model was assessed by the goodness of fit (R2X) and predictive ability (Q2). The model yielded R2(X), R2(Y), and Q2 values of 0.988, 0.935, and 0.854, respectively. Additionally, 200 random permutation tests (Fig. S2) showed a positive slope, with intercept values of 0.052 for Q2 and 0.203 for R2. These results confirm that the model was not overfitted and demonstrated strong adaptability and predictive capability. The PCA results further emphasize significant interspecies differences in FA composition between human, camel, equine species, and ruminant milks. Moreover, the FA profiles identified through OPLS-DA are consistent with the ANOVA results, reinforcing the observed distinctions among the milk types.
Fig. 2.
Principal component analysis (PCA) (A) performed on the fatty acids (FAs) in milks of humans and eight species of dairy animals using gas chromatography with flame ionization (GC-FID). Loading plots of orthogonal partial-least-squares discriminant analysis (OPLS-DA) (B) of the FAs in milks of humans and of eight species of dairy animals using GC.
3.3. Variations of MFGM proteins between humans and animals
A total of 1253 proteins were quantified in milk MFGM samples from humans and from the eight dairy animal species (Table S4). Key MFGM proteins, including BTN1A1, XDH, perilipin, and lactadherin, were identified in high abundance, demonstrating the robustness of the methodology. Additionally, subgroups of whey proteins and casein micelle proteins, such as β-lactoglobulin and caseins, were identified in the MFGM fraction of each species.
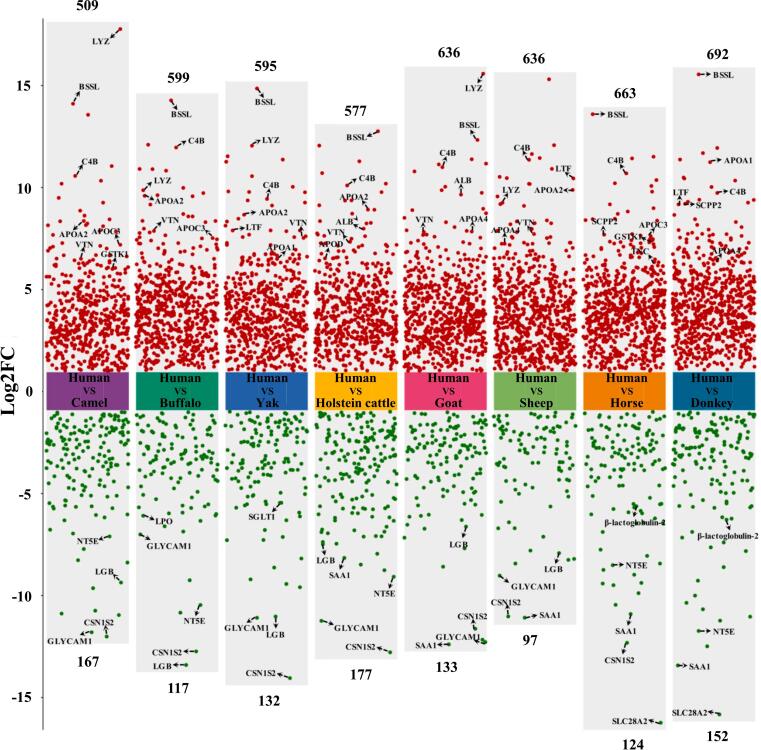
The ANOVA results reveal significant differences in the MFGM proteome between species, identifying a total of 1170 differentially expressed proteins (Table S5). To visually represent the observed interspecies variations, we generated multigroup differential scatter plots based on P values and FC, illustrating the comparisons between human milk and the milks of Holstein cattle, buffalo, yak, camel, goat, sheep, horse, and donkey (Fig. 3).
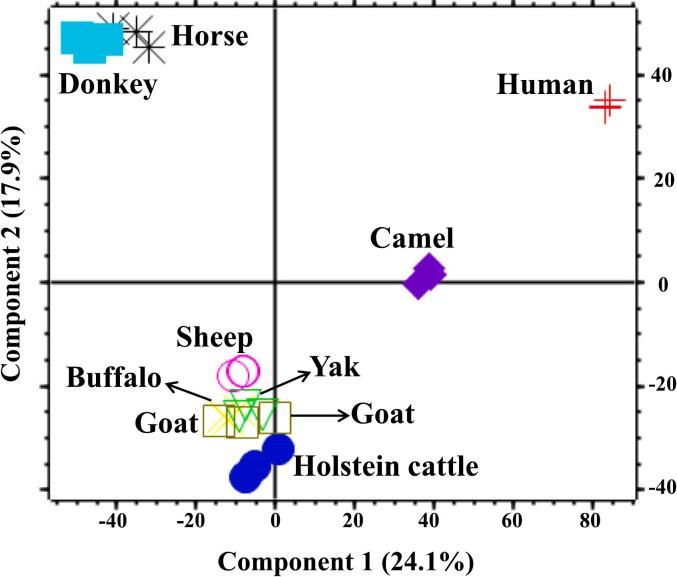
Fig. 3.
Principal component analysis (PCA) performed on the milk fat globule membrane (MFGM) proteome in milks of humans and of eight species of dairy animals using proteomic methods.
The differential MFGM proteins between human and large ruminants, including buffalo, yak, and Holstein cattle, were similar. Compared to the MFGM proteins of large ruminants, human MFGM exhibited significantly higher levels (P < 0.05) of adiponectin B, glutathione-S-transferase κ (GSTK1), glutathione peroxidase 4, retinol dehydrogenase 11 (RDH11), perilipin-3, apolipoproteins including apolipoprotein A-II (APOA2), apolipoprotein A-I (APOA1), and apolipoprotein A-IV (APOA4), selenoprotein P (SELENOP), and lactotransferrin, whereas selenoprotein F, Na+/glucose cotransporter (SGLT1), and 5′-nucleotidase (NT5E) were significantly lower in human MFGM (P < 0.05). Furthermore, compared with human MFGM, buffalo MFGM had significantly higher levels (P < 0.05) of angiotensinogen, semaphorin-7 A, and secretoglobin family 1D member; yak MFGM had significantly higher levels (P < 0.05) of monocyte differentiation antigen and transketolase; and Holstein cattle MFGM exhibited significantly higher levels (P < 0.05) of haptoglobin, hornerin, and cystatin-A.
The differential MFGM proteins between human and small ruminants, including goat and sheep, were similar. Compared to the MFGM proteins of small ruminants, human MFGM exhibited significantly higher levels (P < 0.05) of vitronectin, tenascin, GSTK1, RDH11, sterol carrier protein 2 (SCP-2), apolipoproteins A, and SELENOP, while SGLT1, NT5E, serum amyloid A protein (SAA1), lipocalin 2, and glycosylation-dependent cell adhesion molecule 1 (GLYCAM1) were significantly lower in human MFGM (P < 0.05). Additionally, compared to human MFGM, goat MFGM showed significantly higher levels (P < 0.05) of copper transport protein, selenium-binding protein 1, and elastase, while sheep MFGM exhibited significantly higher levels (P < 0.05) of zinc-α-2-glycoprotein, vitamin D binding protein, glycoprotein 2, and profilaggrin.
The MFGM proteomes of horse and donkey were similar. Compared to the MFGM proteins in horse and donkey, human MFGM exhibited significantly higher levels (P < 0.05) of several proteins, including adiponectin B, GSTK1, RDH11, SCP-2, perilipin-3, and SELENOP. Conversely, SGLT1, NT5E, amine oxidase (AOC1), aldehyde dehydrogenase, and BTN1A1 were significantly lower in human MFGM (P < 0.05). Additionally, compared to human MFGM, horse MFGM showed significantly higher levels (P < 0.05) of glyceraldehyde-3-phosphate dehydrogenase, α-1B-glycoprotein, and lipopolysaccharide-binding protein, while donkey MFGM exhibited significant elevation (P < 0.05) of bactericidal-permeability-increasing protein and platelet glycoprotein 4.
Compared to camel MFGM, human MFGM exhibited significantly higher levels (P < 0.05) of adiponectin B, GSTK1, APOA2, SCP-2, SELENOP, and lactotransferrin, while SGLT1, AOC1, GLYCAM1, lactoperoxidase, sodium/nucleoside cotransporter 2, and whey acidic protein were significantly lower (P < 0.05) in human MFGM.
In summary, we observed that several classes of proteins with specific biological functions exhibited significantly higher abundance in human MFGM compared to animal MFGM. Specifically, novel immunoenhancing molecules (FC = 9.2–294.1, P < 0.05), including adiponectin B, vitronectin, and tenascin; antioxidant enzymes (FC = 57.2–12,876.3, P < 0.05), including GSTK1, RDH11, and catalase; lipid regulatory proteins (FC = 3.2–477.7, P < 0.05) including SCP-2; and trace-element-transport-related proteins (FC = 5.7–1351.2, P < 0.05), including SELENOP, iron-responsive element-binding protein 2, and lactotransferrin, were significantly more abundant in human MFGM. Conversely, proteins such as NT5E, SGLT1, XDH, GLYCAM1, and α-caseins showed high abundance in dairy-animal MFGM. Additionally, proteins involved in FA synthesis, such as long-chain fatty acid-CoA ligase, fatty acid synthase, and acetyl-CoA carboxylase 2, were observed to be significantly more abundant in human MFGM than in animal MFGM.
3.4. PCA and hierarchical clustering analysis of human and animal MFGM proteomes
The corresponding scores are shown in Fig. 4. In the PC1 direction (accounting for 24.1 % of the variance in proteins) and PC2 direction (accounting for 17.9 % of the variance in proteins), the MFGM proteins of human, ruminants, equine species, and camel milk were distinctly separate, further emphasizing the substantial interspecies differences in the MFGM proteome.
Fig. 4.
Multigroup differential scatter plots of differentially abundant milk fat globule membrane (MFGM) proteins from humans and eight species of dairy animals using the proteomic approach. FC = fold change.
The results of the hierarchical clustering analysis applied to the identified MFGM proteome from both humans and various animal species are depicted in Fig. S3. This analytical approach revealed a distinct clustering pattern characterized by two major clusters. Specifically, the MFGM proteins from human milk and animals formed separate primary clusters. Within the animal subgroup, the MFGM proteomes of sheep, buffalo, and yak exhibited similar proteomic patterns, while Holstein cattle and goat milk shared a comparable MFGM profile. Notably, the MFGM proteomes of these ruminant species collectively formed a larger cluster. Horse and donkey MFGM showed similar proteomic patterns, forming a large cluster that, along with the ruminants, constituted an even larger group. Camel milk formed a separate cluster, but it also formed a larger group when combined with other animal MFGM samples. The pattern of clustering was similar to the result of PCA.
3.5. Functional analysis of differentially abundant proteins
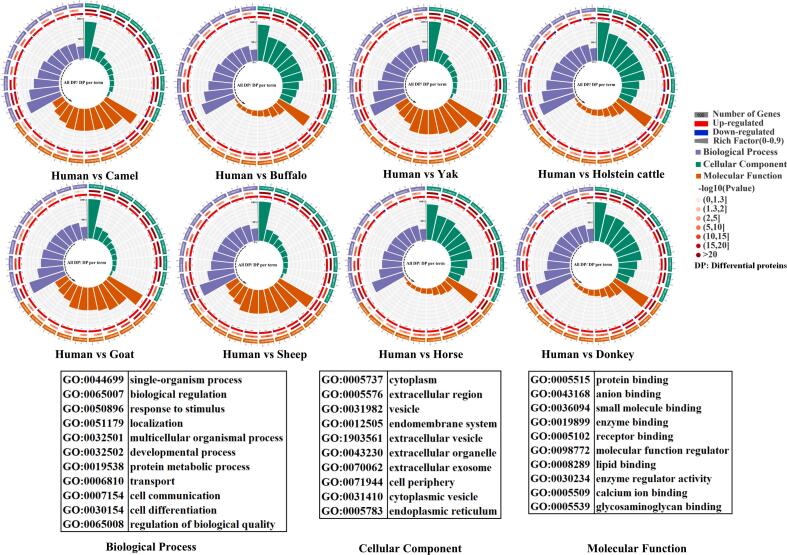
According to the protein annotations, the differentially abundant MFGM proteins identified between human and animal milks were classified based on biological processes (BP), cellular components (CC), and molecular functions (MF) (Fig. 5). The most common BP included single-organism process, biological regulation, responsed to stimulus, and multicellular organismal process. Regarding CC, the differential MFGM proteins were mainly located in the cytoplasm, extracellular region, vesicle, endomembrane system, and extracellular organelle. The predominant MFs of differentially expressed MFGM proteins encompassed protein binding, catalytic activity, ion binding, organic-cyclic-compound binding, small-molecule binding, carbohydrate derivative binding, and hydrolase activity. These findings highlight the similarities in the biological functions of the differentially abundant MFGM proteins between human and animal milks.
Fig. 5.
Gene ontology (GO) analysis of different abundant milk fat globule membrane (MFGM) proteins in human milk versus milk from eight species of dairy animals. Biological process (A), cellular component (B), and molecular function (C).
KEGG pathway predictions were performed for the differentially abundant MFGM proteins in the comparison between human and animal milks, as shown in Table S6. The results reveal that the differentially abundant proteins were primarily associated with ribosome, protein processing in the endoplasmic reticulum, phagosome, endocytosis, complement and coagulation cascades, and carbon metabolism. These findings further underscore the crucial roles of the differential proteins between human and animal milk in maintaining cellular function and immune regulation.
3.6. Correlation analysis
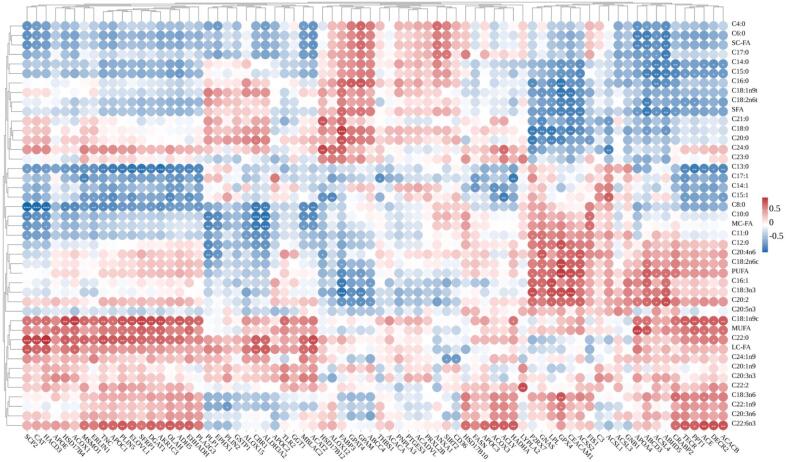
BP related to FA metabolism, such as the FA biosynthetic process, UFA metabolic process, and FA oxidation, were identified, as shown in Table 1. Subsequently, differentially abundant proteins involved in FA metabolism were further analyzed, and their correlations with FAs were visually represented through a correlation heatmap (Fig. 6). Fig. 6 reveals an extremely strong positive correlation (|r| > 0.9) between SCP-2 and both C22:0 and LC-FAs, while we can observe an extremely strong negative correlation (|r| > 0.9) between SCP-2 and C8:0. APOA4 shows an extremely strong positive correlation (|r| > 0.9) with MUFA while exhibiting a significant negative correlation (|r| > 0.8) with SC-FA and C6:0. In addition, several proteins, including elongation of very-long-chain fatty acids protein 1, secreted frizzled-related protein 1, diacylglycerol-O-acyltransferase 1, AKR1C3, and ELOVL1, exhibited a significant positive correlation (|r| > 0.8) with C18:1n9c and a significant negative correlation (|r| > 0.8) with C13:0. In summary, these results underscore the strong correlations between specific proteins and FAs involved in FA metabolism, with key proteins such as SCP-2 and APOA4 exhibiting extremely strong associations with distinct FAs, emphasizing their pivotal roles in FA regulation.
Table 1.
Biological processes related to fatty acid metabolism enriched by milk fat globule membrane proteins.
| Term | Count | Percentage | P Value | Fold Enrichment |
|---|---|---|---|---|
| Fatty acid metabolic process | 61 | 5.81 | 4.47E-12 | 2.69 |
| Fatty acid biosynthetic process | 33 | 3.14 | 3.54E-09 | 3.34 |
| Unsaturated fatty acid metabolic process | 18 | 1.71 | 1.85E-03 | 2.33 |
| Fatty acid oxidation | 17 | 1.62 | 1.42E-04 | 3.02 |
| Long-chain fatty acid metabolic process | 16 | 1.52 | 8.33E-04 | 2.69 |
| Fatty acid transport | 16 | 1.52 | 2.52E-03 | 2.42 |
| Fatty acid catabolic process | 15 | 1.43 | 1.84E-03 | 2.59 |
| Fatty acid derivative metabolic process | 15 | 1.43 | 1.02E-02 | 2.15 |
| Fatty acid beta-oxidation | 14 | 1.33 | 9.77E-05 | 3.65 |
| Regulation of fatty acid metabolic process | 13 | 1.24 | 1.11E-02 | 2.29 |
| Unsaturated fatty acid biosynthetic process | 12 | 1.14 | 2.44E-03 | 2.93 |
| Response to fatty acid | 12 | 1.14 | 6.26E-03 | 2.59 |
| Long-chain fatty acid transport | 11 | 1.05 | 2.47E-03 | 3.12 |
| Fatty-acyl-coa metabolic process | 10 | 0.95 | 1.49E-04 | 4.88 |
| Regulation of fatty acid biosynthetic process | 10 | 0.95 | 2.10E-03 | 3.46 |
| Fatty acid derivative biosynthetic process | 9 | 0.86 | 2.00E-02 | 2.63 |
| Fatty-acyl-coa biosynthetic process | 8 | 0.76 | 2.94E-04 | 5.85 |
| Long-chain fatty-acyl-coa metabolic process | 7 | 0.67 | 1.54E-03 | 5.33 |
| Very long-chain fatty acid metabolic process | 7 | 0.67 | 1.04E-02 | 3.7 |
| Negative regulation of fatty acid metabolic process | 7 | 0.67 | 4.22E-02 | 2.72 |
| Cellular response to fatty acid | 7 | 0.67 | 7.23E-02 | 2.38 |
| Long-chain fatty-acyl-coa biosynthetic process | 6 | 0.57 | 2.48E-03 | 6.01 |
| Regulation of fatty acid transport | 6 | 0.57 | 2.76E-02 | 3.46 |
| Positive regulation of fatty acid metabolic process | 6 | 0.57 | 6.21E-02 | 2.78 |
| Fatty acid beta-oxidation using acyl-coa oxidase | 5 | 0.48 | 2.66E-03 | 7.93 |
| Positive regulation of fatty acid biosynthetic process | 5 | 0.48 | 3.47E-02 | 3.96 |
| Negative regulation of fatty acid biosynthetic process | 5 | 0.48 | 4.50E-02 | 3.66 |
| Long-chain fatty acid biosynthetic process | 5 | 0.48 | 6.32E-02 | 3.28 |
| Fatty acid elongation | 4 | 0.38 | 3.40E-02 | 5.44 |
| Negative regulation of fatty acid transport | 3 | 0.29 | 7.76E-02 | 6.34 |
| Long-chain fatty acid import | 3 | 0.29 | 9.37E-02 | 5.71 |
| Plasma membrane long-chain fatty acid transport | 3 | 0.29 | 9.37E-02 | 5.71 |
Fig. 6.
Correlation heatmap analysis of differential proteins involved in fatty acid (FA) metabolism related to biological processes (BPs) and FAs. The correlation coefficient r is depicted through color gradation. Red indicates a positive correlation (r > 0), while blue signifies a negative correlation (r < 0). |r| > 0.7, |r| > 0.8, and |r| > 0.9 indicate strong correlation, significant correlation, and extremely strong correlation, respectively, and they are represented by *, **, and ***, respectively. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
4. Discussion
4.1. Difference in particle size and FAs of MFGs
Although MFGs have similar distribution pattern of particle size, we noted that buffalo and sheep have the highest MFG particle sizes, followed by human and yak, while horse and donkey have the lowest MFG particle sizes. Several previous studies have extensively summarized the particle sizes of MFGs in milks from human and different animals, and they have revealed that the MFG particle size of donkey was the smallest, with an average particle size of 1.92 μm, while the MFG particle sizes of camel, human, and ruminants were close to each other, about 1.5–2 times that of horse and donkey (Attaie & Richter, 2000; Martini, Altomonte, Licitra, & Salari, 2018; Michalski et al., 2005). Our results confirm these findings. Additionally, a study reported a higher amount of large MFGs (>8 μm) for buffalo compared with cow, and it revealed that the particle size of MFGs was positively correlated with milk fat (Ménard et al., 2010). The particle size of MFGs is significant for infant fat digestion and dairy processing. The presence of smaller MFGs in donkey and horse milk may offer an expanded surface area conducive to the effective action of lipid-digesting enzymes, thus potentially enhancing the hydrolysis and subsequent absorption of fat (Roy, Ye, Moughan, & Singh, 2020). Thus, the smaller MFG particle size in equine milk reflects favorable fat globule characteristics, and we suggest incorporating it into formula as a lipid digestion enhancer more suitable for infants.
Regarding FAs, human milk had lower short-chain SFAs including C4:0 and C6:0, as well as long-chain SFAs including C16:0 and C18:0, compared to ruminant milk. The obvious difference in FA composition between human and ruminant milk has been widely proven in previous studies, and the observed results are consistent with this study (Fan et al., 2023, Wang et al., 2022). Research has indicated that FAs with distinctive nutritional characteristics, such as pentadecanoic acid (C15:0) and 17:0, are more frequently synthesized during rumen microbial fermentation than within mammary glands (Liu, Rochfort, & Cocks, 2018).
The difference in phylogeny leads to the completely different FA composition of equine milk and ruminant milk. In the current study, horse milk had significantly higher C14:1 and cis-10-heptadecenoic acid (C17:1) compared with human and other animals. It has been reported that the proportion of MUFAs in horse milk was 26.8 %–36.2 %, similar to that in human milk and higher than the proportion in donkey milk (Salimei & Fantuz, 2012), similar to our study. Here, donkey milk was characterized by a significantly higher abundance MC-FAs and PUFAs including C18:2n6c and cis-5,8,11,14,17-eicosapentaenoic acid (C20:5n3). Consistent with the results of this study, a previous review has demonstrated that, compared to human milk, donkey milk contains a comparable amount of UFAs, along with higher levels of C18:3n3 (Martini et al., 2018). These FAs are crucial for the development of the infant brain and nervous system. Conversely, another study reported lower concentrations of PUFAs in donkey milk compared to human milk, which slightly diverges from our findings (Wang et al., 2022). This discrepancy may be attributed to the different feeding practices resulting from the lack of systematic farming of dairy donkey. In short, compared to conventional bovine milk, the distinctively higher PUFA content of donkey milk may indicate its potential as a candidate lipid source for human-milk-based formulations.
Compared to other studied samples, camel milk was characterized by significantly higher levels of LC-FAs, mainly SFAs such as C18:0 and C20:0. We also observed elevated levels of trans-MUFA∙PUFA, including C18:1n9t and linolelaidic acid (C18:2n6t). A previous study reported elevated levels of LC-FAs, such as C17:1, in camel milk (Wang et al., 2022). A GC-FID analysis revealed that camel milk has higher levels of trans-MUFA/PUFA, specifically, trans-octadecenoic acid (trans double bonds at C9 and C10) and trans-octadecadienoic acid (trans double bonds at C9 and C12), compared to human and ruminant milk (Teng, Wang, Yang, Ma, & Day, 2017). Notably, while a high intake of industrial trans fats increases cardiovascular risk, the link between naturally occurring trans-fats in milk and cardiovascular disease has not been firmly established (Chardigny et al., 2008). Another important point to emphasize is that the composition of milk from non-ruminant animals (such as camel and horse) is known to be influenced by feeding practices (Baloš, Pelić, Jakšić, & Lazić, 2023). However, in some nomadic regions, these dairy animals are still raised using traditional grazing or mixed-grazing-stall feeding system. To optimize the use of these milks in infant food, standardization of feeding practices may be necessary.
4.2. Difference analysis of MFGM proteins
4.2.1. Analysis of novel immunoenhancing factors
The immunoenhancing effects have consistently served as an advantage of human milk, distinguishing it from traditional animal milks. Proteins involved in immune system modulation, such as immunoglobulins, lactotransferrin, lysozyme, and complement system proteins, showed significant differences between human MFGM and that of ruminants and non-ruminants (Yang et al., 2016; Zhang et al., 2021). This study expanded its scope to examine the prevalence of other novel immunity-enhancing molecules in MFGM, particularly adiponectin B, vitronectin, and tenascin, which were observed to be more abundant in the MFGM of human milk compared to that of animal-derived milk. According to their annotations, these proteins were closely related to host defense mechanisms as well. The adiponectin concentration in human milk has been reported to be higher than that in bovine and donkey milk (Trinchese et al., 2018). Notably, adiponectin was found in significantly higher abundance in human whey compared to those of large ruminants, equine species, and camel (Ji et al., 2024). Additionally, the adiponectin content of human milk decreases by about 5 % to 6 % every month with lactation (Newburg, Woo, & Morrow, 2010), indicating the importance of adiponectin in early infant development. Adiponectin plays a key role in appetite regulation and has a pivotal anti-inflammatory function, including the down-modulation of innate immune cells and cytokines, as well as the promotion of regulatory T-cell activity (Grases-Pintó et al., 2021). It is noteworthy that adiponectin is synthesized in adipose tissue, which may contribute to its preferential accumulation within the MFGM (Newburg et al., 2010). Vitronectin and tenascin were highly expressed in human MFGM, while they were hardly identified in ruminants. The abundance of vitronectin in human MFGM was observed to be significantly higher than that in ruminants, camel, and horse based on relative quantitative proteomics (Lu et al., 2016; Yang et al., 2015). In addition, vitronectin was identified in human whey, while it was hardly identified in ruminants (Ji et al., 2024). The results of these studies are consistent with our findings. Vitronectin regulates complement activity during membrane attack complex formation and is involved in immune processes, inflammation regulation, and tissue repair (Li et al., 2023; Singh, Su, & Riesbeck, 2010). Notably, vitronectin is an extracellular matrix adhesion factor protein, which can form selective cell-matrix adhesion (Sen & Ta, 2023). Thus, we speculated that this might be contribute to the binding of vitronectin to MFGM and enhance immune processes. It has been reported that tenascin in human MFGM was significantly higher than that in ruminants, camel, horse, and donkey based on LC–MS/MS proteomics (Lu et al., 2016; Yang et al., 2016; Zhang et al., 2021). Additionally, we noticed that tenascin has been identified in human whey and MFGM across different studies (Froehlich et al., 2010; Wang et al., 2023), but not in MFGM of buffalo, bovine, goat, yak, and donkey in other studies (Chen et al., 2023; Li et al., 2019). These findings align with our results. Notably, tenascin was detected in high abundance in human colostrum whey but not in mature milk, suggesting a specific requirement for tenascin in early infancy (Froehlich et al., 2010). Tenascin is a high-molecular-weight polymer protein that holds a pivotal role in the developmental processes of the fetal brain and wound healing. In particular, it has been reported that tenascin can prevent infectious viruses from combining with mucosal epithelial cells, and that its efficacy is higher than that of lactoferrin and albumin (Fouda et al., 2013). In summary, these findings emphasize the importance of incorporating immunoenhancing factors into infant formula to better replicate the immunological benefits of human milk, rather than solely focusing on proteins of high abundance.
4.2.2. The necessity of antioxidant enzyme enhancement
MFGM is rich in functional enzymes, which catalyze many BP. In this study, several antioxidant enzymes, such as GSTK1, RDH11 and catalase, were significantly higher in human MFGM than in other studied samples. Multiple studies have quantitatively analyzed the MFGM proteome of human and various dairy animals and have revealed significantly higher levels of GSTK1, glutathione peroxidase, catalase, and other antioxidant proteins in human MFGM compared to ruminant and non-ruminant MFGM, consistent with our findings (Lu et al., 2016; Yang et al., 2015; Zhang et al., 2021). A previous study has reported significantly higher levels of enzymatic antioxidants, such as superoxide dismutase, glutathione peroxidase, and catalase, in human milk compared to cow and goat milk (Khan et al., 2019). Similarly, our recent study found that the catalase of human whey was significantly higher than that of ruminants, camel, and equine species (Ji et al., 2024). A study using animal models found a significant increase in serum glutathione-S-transferase levels in mice fed human milk compared to those fed bovine milk, potentially indicating a superior capacity of human milk to enhance antioxidant defense mechanisms in neonates (Trinchese et al., 2015). Another study has shown that human milk has a greater ability to mitigate oxidative stress and reduce oxidative DNA damage in newborns compared to infant formula. This suggests that human milk contains a unique defense mechanism that is absent in commercial infant formula or bovine milk (Shoji et al., 2004). It was widely reported that GSTK1, RDH11, and catalase catalyze the scavenging of superoxide radicals (O2−) and are involved in the biological function of response to toxic substances and in cellular oxidant detoxification (Khan et al., 2019). These findings underscore the need to fortify infant formula with antioxidant enzymes to enhance oxidative stress recovery in early neonatal development.
4.2.3. Analysis of lipid regulatory proteins
Several proteins associated with lipid regulation such as SCP-2 and some apolipoproteins in human MFGM were at significantly higher amounts than that in dairy animals. Sterol carrier protein was not identified in the MFGM of ruminants and donkey (Chen et al., 2023; Li et al., 2019). However, there is no information on the quantitative difference in SCP-2 between human and animals. Here, SCP-2 was first identified in MFGMs of different species, especially humans. A previous study found that human milk contains low level of cholesterol, which is possibly linked to the higher expression of SCP-2 compared to ruminants (Pietrzak-Fiećko & Kamelska-Sadowska, 2020). Cholesterol is essential for both the structural integrity and functional processes within the biological system, and it is the precursor of various metabolically active compounds. Notably, SCP-2 is a high-affinity binding protein for LC-FA and long-chain acyl-CoA, which enhances the formation and transport of cholesteryl esters, thereby participating in the dynamic homeostasis and metabolic processes of cholesterol (Burgardt, Gianotti, Ferreyra, & Ermácora, 2017; Jolly, Chao, Kier, Billheimer, & Schroeder, 2000). Particularly in this study, SCP-2 was observed to exhibit a significant correlation with LC-FA (|r| = 0.82). Thus, we hypothesize that SCP-2 may have a crucial role in the metabolic regulation of lipids in milk, including LC-FA. Camel MFGM, because of its high SCP-2 content, should be considered as a formula ingredient for enhancing lipid composition.
APOA1, APOA2, and APOA4 were at significantly higher proportions in human MFGM than in ruminants, camel, and donkey in this study. It has been reported that the abundance of APOA2 in human MFGM was similar to that in horse and that it was significantly higher than that in ruminants and camel (Yang et al., 2015). Additionally, the relative abundance of APOA4 in the MFGM of human milk was significantly higher compared to that in ruminants (Lu et al., 2016). These results are similar to those of our study. Additionally, in this study, apolipoprotein A and other regulatory factors, such as perilipin-3, were observed to be enriched in biological functions related to lipid regulation such as lipid storage and localization. APOA1 and APOA2 are the two principal subtypes of apolipoprotein A. Compared to other types of apolipoproteins, their unique roles in reverse cholesterol transport, cholesterol metabolism regulation, and lipoprotein structure stabilization make them crucial for maintaining cardiovascular health (Walldius & Jungner, 2004). Thus, considering the crucial role of apolipoprotein A in lipid regulation and metabolism, as well as strategically incorporating MFGM rich in apolipoprotein A from horse milk into infant formula, may be an effective approach to enhancing its humanization.
4.2.4. Specific expression of trace-element-transport-related proteins
In milk, lipids serve as the “natural carriers” of mineral elements. Here, a complex variability in the expression of several trace-element-transport-related proteins between human and animal milks has been observed. Among them, SELENOP, iron-responsive-element-binding protein 2, and lactotransferrin in human MFGM were significantly more abundant than those in animals, while selenoprotein F and zinc-α-2-glycoprotein with high abundance were identified in ruminant MFGM, especially in yak and sheep. The abundance of SELENOP and lactotransferrin in human MFGM has been reported to be significantly higher than in ruminants (Lu et al., 2016, Yang et al., 2016). Other studies have found that the abundance of zinc-α-2-glycoprotein in human MFGM was significantly higher than in donkey, and that the content of selenoprotein F and zinc-α-2-glycoprotein in Holstein cattle exceeded those in camel, goat, and human (Han, Zhang, & Zhou, 2022; Yang et al., 2016; Zhang et al., 2021). Recently, we observed significant disparities in the abundance of selenoprotein F and zinc-α-2-glycoprotein between ruminant and human milk whey, with higher levels of selenoprotein M and lactotransferrin in human milk (Ji et al., 2024). These results are similar to our findings. Mineral elements play important roles in the early development of infants, and SELENOP and lactotransferrin are the main transporters for transferring maternal selenium and iron, respectively, to the infant (Hill, Motley, Winfrey, & Burk, 2014). The differential expression of trace element transport-related proteins in MFGM across species reveals unique advantages or compensatory nutritional mechanisms inherent to distinct milk sources, thereby providing valuable insights for the humanized development of infant formula.
5. Conclusion
In this study, differences in MFG particle size, FAs, and MFGM proteome between humans and eight dairy animal species were revealed. The particle sizes of MFGs in horse and donkey milk primarily ranged between 0 and 5 μm, exhibiting a smaller average particle size compared to human milk and other animal milks. Furthermore, compared to other animal milks, horse and donkey milks were rich in MUFAs and PUFAs, respectively. Regarding the MFGM proteome between humans and animals, human MFGM contained more immunity-related lipid regulatory proteins and antioxidases, including GSTK1, RDH11, and catalase, while animal milks were rich in SGLT1, NT5E, and GLYCAM1. The findings of this research offer new insights into the physicochemical characteristics of MFGs in human and animal milks. Currently, the feasibility of supplementing MFGM to bridge the gap between human milk and infant formula has been recognized and has already reached commercial-scale application. As a result, the use of these components as infant formula supplements is becoming increasingly practical, necessitating a deeper understanding of the specific functions of bioactive substances in human MFGM. This, in turn, will guide the development and optimization of methods for isolating MFGM compounds from milk-based sources on a commercial scale. Thus, highlighting the subtle differences between animal milks and human milk may provide valuable insights for refining the composition of current infant foods. In future research, absolute quantification of proteins may be necessary to further validate our findings.
CRediT authorship contribution statement
Zhongyuan Ji: Writing – original draft, Visualization, Investigation, Formal analysis, Data curation. Lu Meng: Validation, Supervision, Software. Xueheng Sun: Investigation, Formal analysis. Rongwei Han: Methodology, Conceptualization. Yongxin Yang: Supervision, Resources. Jiaqi Wang: Funding acquisition. Nan Zheng: Writing – review & editing, Methodology, Funding acquisition.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by The National Key R&D Program of China (2022YFD1600104), The earmarked fund for CARS (CARS-36), The Agricultural Science and Technology Innovation Program (ASTIP-IAS12), Central Public-interest Scientific Institution Basal Research Fund (2023-YWF-ZX-06).
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.fochx.2025.102431.
Appendix A. Supplementary data
Supplementary material 1
Supplementary material 2
Supplementary material 3
Supplementary material 4
Supplementary material 5
Supplementary material 6
Supplementary material 7
Data availability
No data was used for the research described in the article.
References
- Attaie R., Richter R. Size distribution of fat globules in goat milk. Journal of Dairy Science. 2000;83(5):940–944. doi: 10.3168/jds.S0022-0302(00)74957-5. [DOI] [PubMed] [Google Scholar]
- Ballard O., Morrow A.L. Human milk composition: Nutrients and bioactive factors. Pediatric Clinics. 2013;60(1):49–74. doi: 10.1016/j.pcl.2012.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baloš M.Ž., Pelić D.L., Jakšić S., Lazić S. Donkey milk: An overview of its chemical composition and main nutritional properties or human health benefit properties. Journal of Equine Veterinary Science. 2023;121:104225. doi: 10.1016/j.jevs.2023.104225. [DOI] [PubMed] [Google Scholar]
- Burgardt N.I., Gianotti A.R., Ferreyra R.G., Ermácora M.R. A structural appraisal of sterol carrier protein 2. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics. 2017;1865(5):565–577. doi: 10.1016/j.bbapap.2017.03.002. [DOI] [PubMed] [Google Scholar]
- Chardigny J.-M., Destaillats F., Malpuech-Brugère C., Moulin J., Bauman D.E., Lock A.L.…Chaumont P. Do trans fatty acids from industrially produced sources and from natural sources have the same effect on cardiovascular disease risk factors in healthy subjects? Results of the trans fatty acids collaboration (TRANSFACT) study. The American Journal of Clinical Nutrition. 2008;87(3):558–566. doi: 10.1093/ajcn/87.3.558. [DOI] [PubMed] [Google Scholar]
- Chen L., Hong T., Li Z., Shen G., Gu Y., Han J. A comparison of milk fat globule membranes and whey proteomes: New insight into variation nutrient differences between Buffalo, cow, goat, and yak. Food Chemistry. 2023;429:136845. doi: 10.1016/j.foodchem.2023.136845. [DOI] [PubMed] [Google Scholar]
- Fan R., Shi R., Ji Z., Du Q., Wang J., Jiang H.…Yang Y. Effects of homogenization and heat treatment on fatty acids in milk from five dairy species. Food Quality and Safety. 2023;7:fyac069. doi: 10.1093/fqsafe/fyac069. [DOI] [Google Scholar]
- Fouda G.G., Jaeger F.H., Amos J.D., Ho C., Kunz E.L., Anasti K.…Ohashi T. Tenascin-C is an innate broad-spectrum, HIV-1–neutralizing protein in breast milk. Proceedings of the National Academy of Sciences. 2013;110(45):18220–18225. doi: 10.1073/pnas.1307336110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Froehlich J.W., Dodds E.D., Barboza M., McJimpsey E.L., Seipert R.R., Francis J.…Lebrilla C.B. Glycoprotein expression in human milk during lactation. Journal of Agricultural and Food Chemistry. 2010;58(10):6440–6448. doi: 10.1021/jf100112x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grases-Pintó B., Abril-Gil M., Torres-Castro P., Castell M., Rodríguez-Lagunas M.J., Pérez-Cano F.J., Franch À. Rat milk and plasma immunological profile throughout lactation. Nutrients. 2021;13(4):1257. doi: 10.3390/nu13041257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadley K.B., Ryan A.S., Forsyth S., Gautier S., Salem N., Jr. The essentiality of arachidonic acid in infant development. Nutrients. 2016;8(4):216. doi: 10.3390/nu8040216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han B., Zhang L., Zhou P. Comparison of milk fat globule membrane protein profile among bovine, goat and camel milk based on label free proteomic techniques. Food Research International. 2022;162:112097. doi: 10.1016/j.foodres.2022.112097. [DOI] [PubMed] [Google Scholar]
- Hill K.E., Motley A.K., Winfrey V.P., Burk R.F. Selenoprotein P is the major selenium transport protein in mouse milk. PLoS One. 2014;9(7) doi: 10.1371/journal.pone.0103486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji X., Xu W., Cui J., Ma Y., Zhou S. Goat and buffalo milk fat globule membranes exhibit better effects at inducing apoptosis and reduction the viability of HT-29 cells. Scientific Reports. 2019;9(1):2577. doi: 10.1038/s41598-019-39546-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji Z., Dong R., Du Q., Jiang H., Fan R., Bu D.…Yang Y. Insight into differences in whey proteome from human and eight dairy animal species for formula humanization. Food Chemistry. 2024;430:137076. doi: 10.1016/j.foodchem.2023.137076. [DOI] [PubMed] [Google Scholar]
- Jolly, C.A., Chao, H., Kier, A.B., Billheimer, J.T., & Schroeder, F. (2000). Sterol carrier protein-2 suppresses microsomal acyl-CoA hydrolysis. Molecular and Cellular Biochemistry, 205, 83–90. 10.1023/A:1007001614939. [DOI] [PubMed]
- Khan I.T., Nadeem M., Imran M., Ullah R., Ajmal M., Jaspal M.H. Antioxidant properties of Milk and dairy products: A comprehensive review of the current knowledge. Lipids in Health and Disease. 2019;18:1–13. doi: 10.1186/s12944-019-0969-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S., Wang Y., Yang R., Zhu X., Bai H., Deng X.…Li Z. Outer membrane protein OMP76 of Riemerella anatipestifer contributes to complement evasion and virulence by binding to duck complement factor vitronectin. Virulence. 2023;14(1):2223060. doi: 10.1080/21505594.2023.2223060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W., Li M., Cao X., Yang M., Han H., Kong F., Yue X. Quantitative proteomic analysis of milk fat globule membrane (MFGM) proteins from donkey colostrum and mature milk. Food & Function. 2019;10(7):4256–4268. doi: 10.1039/C9FO00386J. [DOI] [PubMed] [Google Scholar]
- Liao Y., Alvarado R., Phinney B., Lönnerdal B. Proteomic characterization of human milk fat globule membrane proteins during a 12 month lactation period. Journal of Proteome Research. 2011;10(8):3530–3541. doi: 10.1021/pr200149t. [DOI] [PubMed] [Google Scholar]
- Liu Z., Rochfort S., Cocks B. Milk lipidomics: What we know and what we don’t. Progress in Lipid Research. 2018;71:70–85. doi: 10.1016/j.plipres.2018.06.002. [DOI] [PubMed] [Google Scholar]
- Lu J., Wang X., Zhang W., Liu L., Pang X., Zhang S., Lv J. Comparative proteomics of milk fat globule membrane in different species reveals variations in lactation and nutrition. Food Chemistry. 2016;196:665–672. doi: 10.1016/j.foodchem.2015.10.005. [DOI] [PubMed] [Google Scholar]
- Manoni M., Di Lorenzo C., Ottoboni M., Tretola M., Pinotti L. Comparative proteomics of milk fat globule membrane (MFGM) proteome across species and lactation stages and the potentials of MFGM fractions in infant formula preparation. Foods. 2020;9(9):1251. doi: 10.3390/foods9091251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martini M., Altomonte I., Licitra R., Salari F. Nutritional and nutraceutical quality of donkey milk. Journal of Equine Veterinary Science. 2018;65:33–37. doi: 10.1016/j.jevs.2017.10.020. [DOI] [Google Scholar]
- Ménard O., Ahmad S., Rousseau F., Briard-Bion V., Gaucheron F., Lopez C. Buffalo vs. cow milk fat globules: Size distribution, zeta-potential, compositions in total fatty acids and in polar lipids from the milk fat globule membrane. Food Chemistry. 2010;120(2):544–551. doi: 10.1016/j.foodchem.2009.10.053. [DOI] [Google Scholar]
- Michalski M.-C., Briard V., Michel F., Tasson F., Poulain P. Size distribution of fat globules in human colostrum, breast milk, and infant formula. Journal of Dairy Science. 2005;88(6):1927–1940. doi: 10.3168/jds.S0022-0302(05)72868-X. [DOI] [PubMed] [Google Scholar]
- Muthukumaran M.S., Mudgil P., Baba W.N., Ayoub M.A., Maqsood S. A comprehensive review on health benefits, nutritional composition and processed products of camel milk. Food Reviews International. 2023;39(6):3080–3116. doi: 10.1080/87559129.2021.2008953. [DOI] [Google Scholar]
- Newburg D.S., Woo J.G., Morrow A.L. Characteristics and potential functions of human milk adiponectin. The Journal of Pediatrics. 2010;156(2):S41–S46. doi: 10.1016/j.jpeds.2009.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrzak-Fiećko R., Kamelska-Sadowska A.M. The comparison of nutritional value of human milk with other mammals’ milk. Nutrients. 2020;12(5):1404. doi: 10.3390/nu12051404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy D., Ye A., Moughan P.J., Singh H. Composition, structure, and digestive dynamics of milk from different species—A review. Frontiers in Nutrition. 2020;7:577759. doi: 10.3389/fnut.2020.577759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salimei E., Fantuz F. Equid milk for human consumption. International Dairy Journal. 2012;24(2):130–142. doi: 10.1016/j.idairyj.2011.11.008. [DOI] [Google Scholar]
- Sen A., Ta M. Vitronectin acts as a key regulator of adhesion and migration in human umbilical cord-derived MSCs under different stress conditions. Experimental Cell Research. 2023;423(2):113467. doi: 10.1016/j.yexcr.2023.113467. [DOI] [PubMed] [Google Scholar]
- Shoji H., Shimizu T., Shinohara K., Oguchi S., Shiga S., Yamashiro Y. Suppressive effects of breast milk on oxidative DNA damage in very low birthweight infants. Archives of Disease in Childhood-Fetal and Neonatal Edition. 2004;89(2):F136–F138. doi: 10.1136/adc.2002.018390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh B., Su Y.C., Riesbeck K. Vitronectin in bacterial pathogenesis: A host protein used in complement escape and cellular invasion. Molecular Microbiology. 2010;78(3):545–560. doi: 10.1111/j.1365-2958.2010.07373.x. [DOI] [PubMed] [Google Scholar]
- Singh H., Gallier S. Nature’s complex emulsion: The fat globules of milk. Food Hydrocolloids. 2017;68:81–89. doi: 10.1016/j.foodhyd.2016.10.011. [DOI] [Google Scholar]
- Teng F., Wang P., Yang L., Ma Y., Day L. Quantification of fatty acids in human, cow, buffalo, goat, yak, and camel milk using an improved one-step GC-FID method. Food Analytical Methods. 2017;10:2881–2891. doi: 10.1007/s12161-017-0852-z. [DOI] [Google Scholar]
- Trinchese G., Cavaliere G., Canani R.B., Matamoros S., Bergamo P., De Filippo C.…Negri R. Human, donkey and cow milk differently affects energy efficiency and inflammatory state by modulating mitochondrial function and gut microbiota. The Journal of Nutritional Biochemistry. 2015;26(11):1136–1146. doi: 10.1016/j.jnutbio.2015.05.003. [DOI] [PubMed] [Google Scholar]
- Trinchese G., Cavaliere G., De Filippo C., Aceto S., Prisco M., Chun J.T.…Demurtas A. Human milk and donkey milk, compared to cow milk, reduce inflammatory mediators and modulate glucose and lipid metabolism, acting on mitochondrial function and oleylethanolamide levels in rat skeletal muscle. Frontiers in Physiology. 2018;9:32. doi: 10.3389/fphys.2018.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walldius G., Jungner I. Apolipoprotein B and apolipoprotein A-I: Risk indicators of coronary heart disease and targets for lipid-modifying therapy. Journal of Internal Medicine. 2004;255(2):188–205. doi: 10.1046/j.1365-2796.2003.01276.x. [DOI] [PubMed] [Google Scholar]
- Wang C., Zhao R., Fu W., Li S., Cheng J., Jiang S., Guo M. Insights from 4D label-free proteomic analysis into variation of milk fat globule membrane proteins of human milk associated with infant’s gender. Journal of Agricultural and Food Chemistry. 2023;71(31):12116–12128. doi: 10.1021/acs.jafc.3c01257. [DOI] [PubMed] [Google Scholar]
- Wang F., Chen M., Luo R., Huang G., Wu X., Zheng N.…Wang J. Fatty acid profiles of milk from Holstein cows, Jersey cows, buffalos, yaks, humans, goats, camels, and donkeys based on gas chromatography–mass spectrometry. Journal of Dairy Science. 2022;105(2):1687–1700. doi: 10.3168/jds.2021-20750. [DOI] [PubMed] [Google Scholar]
- Wang X., Zhu H., Zhang W., Zhang Y., Zhao P., Zhang S.…Lv J. Triglyceride and fatty acid composition of ruminants milk, human milk, and infant formulae. Journal of Food Composition and Analysis. 2022;106:104327. doi: 10.1016/j.jfca.2021.104327. [DOI] [Google Scholar]
- Yang Y., Zheng N., Wang W., Zhao X., Zhang Y., Han R.…Guo T. N-glycosylation proteomic characterization and cross-species comparison of milk fat globule membrane proteins from mammals. Proteomics. 2016;16(21):2792–2800. doi: 10.1002/pmic.201500361. [DOI] [PubMed] [Google Scholar]
- Yang Y., Zheng N., Zhao X., Zhang Y., Han R., Ma L.…Wang J. Proteomic characterization and comparison of mammalian milk fat globule proteomes by iTRAQ analysis. Journal of Proteomics. 2015;116:34–43. doi: 10.1016/j.jprot.2014.12.017. [DOI] [PubMed] [Google Scholar]
- Zanabria R., Griffiths M.W., Corredig M. Does structure affect biological function? Modifications to the protein and phospholipids fraction of the milk fat globule membrane after extraction affect the antiproliferative activity of colon cancer cells. Journal of Food Biochemistry. 2020;44(2) doi: 10.1111/jfbc.13104. [DOI] [PubMed] [Google Scholar]
- Zhang X., Jiang B., Ji C., Li H., Yang L., Jiang G.…Min L. Quantitative label-free proteomic analysis of milk fat globule membrane in donkey and human milk. Frontiers in Nutrition. 2021;8:670099. doi: 10.3389/fnut.2021.670099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Wang X., Xie D., Zou S., Jin Q., Wang X. Synthesis and concentration of 2-monoacylglycerols rich in polyunsaturated fatty acids. Food Chemistry. 2018;250:60–66. doi: 10.1016/j.foodchem.2018.01.027. [DOI] [PubMed] [Google Scholar]
- Zou X., Huang J., Jin Q., Guo Z., Liu Y., Cheong L.…Wang X. Lipid composition analysis of milk fats from different mammalian species: Potential for use as human milk fat substitutes. Journal of Agricultural and Food Chemistry. 2013;61(29):7070–7080. doi: 10.1021/jf401452y. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material 1
Supplementary material 2
Supplementary material 3
Supplementary material 4
Supplementary material 5
Supplementary material 6
Supplementary material 7
Data Availability Statement
No data was used for the research described in the article.