Summary
This case report examines a premenopausal woman in her 30s with double heterozygosity (DH) for BRCA1 and BRCA2. The patient’s DH led her parents to undergo genetic testing as well, and new carriers were found, highlighting the importance of genetic counselling and surveillance for the entire family. Due to the rarity of DH, cancer risk and optimal surveillance strategies are not addressed in current guidelines or other sources. This report summarises existing evidence and highlights the need for further research to better understand cancer risks and to develop effective management strategies for individuals with this unique genetic profile.
Keywords: Genetic screening / counselling, Breast surgery, Surgical oncology, Heterozygote
Background
Breast cancer is a significant malignancy affecting women globally. Approximately 6–10% of patients carry pathogenic variants indicative of hereditary breast cancer, most frequently in the BRCA1 and BRCA2 genes.1 2 These genetic alterations significantly increase the risk of breast cancer and other malignancies. Double heterozygosity (DH) in BRCA1 and BRCA2 is rare, and cancer risks and surveillance strategies for patients with DH remain poorly understood.3 4 This case report presents a patient with breast cancer who was found to have DH in BRCA1 and BRCA2 and includes a review of the literature on cancer risks and surveillance strategies.
Case presentation
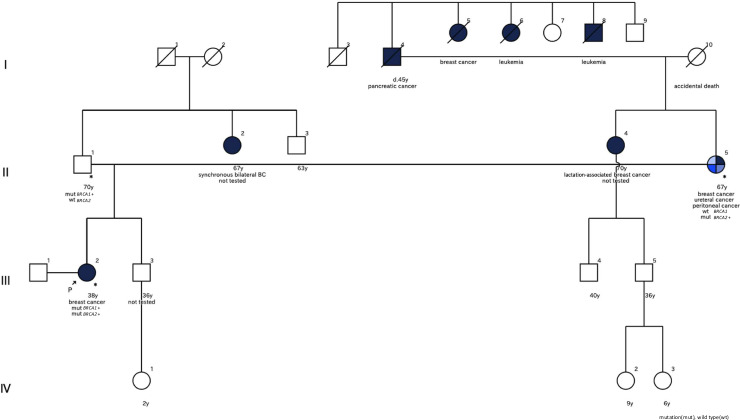
A premenopausal woman in her 30s presented with a palpable mass in her right breast. She had Hashimoto’s disease but no history of malignancies. She had a partner, no history of pregnancy or childbirth, and was considering infertility treatment. Physical examination revealed a palpable induration in the right breast. Her family history included multiple malignancies (figure 1). The patient’s mother had breast cancer and was undergoing treatment for ureteral and peritoneal cancer. Her maternal aunt had lactation-associated breast cancer, her paternal aunt had bilateral breast cancer, and her maternal grandfather died of pancreatic cancer. Additionally, several members of her maternal family had been diagnosed with leukaemia.
Figure 1. Pedigree chart showing the patient’s family history of hereditary breast cancer with BRCA test results.
Investigations
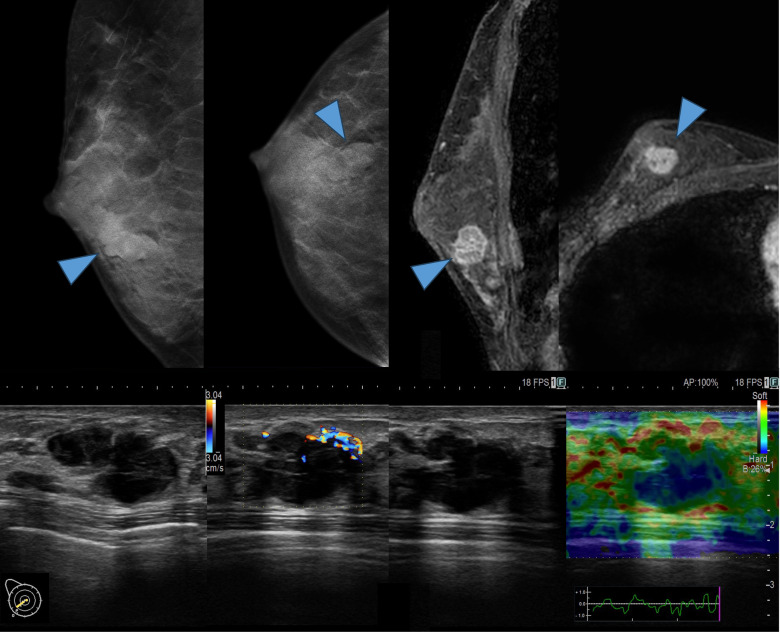
Mammography identified a high-density, irregular mass in the right breast. Ultrasound revealed a lobulated, low-echoic, hypervascular mass in the right lower outer quadrant of the breast. Strain elastography showed decreased strain in this mass. MRI showed a mass with an irregular shape, indistinct margins and heterogeneity with rapid washout kinetics (figure 2). Based on these findings, the imaging results suggested breast cancer. CT and bone scintigraphy revealed no distant metastasis. Vacuum-assisted biopsy (VAB) confirmed the presence of an invasive ductal carcinoma. The preoperative diagnosis was right breast cancer, staged as T1cN0M0, Stage IA, according to the Union for International Cancer Control tumour–node–metastasis classification (eighth edition).
Figure 2. Imaging tests: Top left—mammography, top right—contrast-enhanced MRI, bottom left—ultrasound blood flow evaluation and bottom right—ultrasound strain evaluation.
Differential diagnosis
In this case, the diagnosis of breast cancer was established through standard clinical procedures, ultimately leading to surgical intervention. A mass was detected in the right breast, and based on mammography and breast ultrasonography findings, the differential diagnoses included fibroadenoma, phyllodes tumour and breast cancer. Malignancy was suspected based on the history of breast cancer in the patient’s family. Contrast-enhanced breast MRI findings strongly suggested malignancy, and the diagnosis of breast cancer was confirmed through VAB.
Treatment
The patient underwent a right mastectomy and sentinel lymph node biopsy; all sentinel nodes were negative for metastasis. Postoperative pathological results showed an invasive diameter of 20 mm, with the tumour exhibiting nuclear grade 3, histological grade 3, lymphatic invasion and vascular invasion, an Allred score of 5+3 for ER, 5+2 for PGR, HER2 status of 1+ and a Ki-67 proliferation index of 34%. Additionally, the patient underwent a multigene assay, which indicated a benefit from additional cytotoxic chemotherapy (Oncotype DX Breast Recurrence Score: recurrence score of 35). Due to her young age and family history of breast cancer, BRCA testing was recommended and preoperative BRCA testing revealed double heterozygous mutations in BRCA1 (c.2389_2390del, p. Glu797Thfs*3) and BRCA2 (c.1888dupA, p. Thr630Asnfs*6).
Outcome and follow-up
The patient prioritised chemotherapy over infertility treatment. She received dose-dense epirubicin and cyclophosphamide followed by dose-dense paclitaxel and subsequently started tamoxifen with leuprorelin. During chemotherapy, she underwent genetic counselling and informed her parents of the results.
BRCA analysis revealed that her mother carried the same BRCA2 mutation. Her mother had completed postoperative chemotherapy for ureteral cancer and was planning risk-reducing salpingo-oophorectomy (RRSO); however, a CT scan revealed peritoneal cancer, and she is currently being treated with olaparib.
Given the mother’s results, we informed the patient’s father of the possibility of a BRCA1 mutation, and he requested a single-site test. This test revealed that he carried the same BRCA1 mutation. In addition to regular medical check-ups, we planned to conduct surveillance for prostate cancer using prostate-specific antigen according to the 2021 guidelines for the Diagnosis and Treatment of Japanese Hereditary Breast and Ovarian Cancer (JOHBOC).5 It was not possible to contact the relatives who had a history of with cancer.
The patient has remained recurrence-free for 17 months. Due to the rarity of DH cases, appropriate cancer risk and surveillance strategies remain unclear. We proposed standard surveillance for hereditary breast and ovarian cancer according to the JOHBOC guidelines, which includes annual mammography, breast ultrasound, breast MRI, whole-body contrast-enhanced CT and regular gynaecological examinations.6 7 Future considerations include referrals to another institution for pancreatic surveillance, contralateral risk-reducing mastectomy (CRRM) or RRSO.
Discussion
This study emphasised two crucial points. First, identifying DH has led to specialised genetic counselling, enhancing the understanding of genetic risks for patients and their families. Second, summarising the characteristics of individuals with DH for BRCA1 and BRCA2, compared with a single BRCA pathogenic variant, is crucial from an educational perspective.
The clinical diagnosis of BRCA pathogenic variants enables tailored medical care based on genetic test results, thus facilitating medical management for patients and their families. Genetic testing based on patient autonomy is essential, and healthcare providers must offer timely information. BRCA testing significantly influences treatment strategies, particularly preoperatively, by providing valuable information on surgical options and preventive measures such as CRRM and RRSO. Data show that 88% of patients with preoperative BRCA mutations opt for bilateral mastectomy, emphasising the need for swift and appropriate responses.8
In this case, the incidental discovery of DH reaffirmed the importance of genetic counselling for both the patient and her parents. Typically, a single BRCA gene mutation means a 50% risk of being passed to the offspring. However, with DH, the risk that one or both pathogenic variants of BRCA genes will be inherited by the offspring increases to 75%, necessitating communication with the patient and the provision of more precise risk information.9
Additionally, BRCA1/2 DH may sporadically occur in rare cases, but the majority are inherited from one or both parents. Compared with a single mutation, DH poses a higher likelihood of affecting a greater number of family members.10 Therefore, repeated genetic counselling sessions are essential to emphasise the importance of appropriate surveillance and early detection strategies. In this case, the patient’s father initially hesitated to undergo BRCA testing. However, following the mother’s diagnosis of a BRCA2 mutation, he recognised the increased probability of carrying the mutation himself and agreed to testing. This highlights the critical role of family communication in genetic risk management. Furthermore, ongoing genetic counselling significantly reduces the psychological burden on patients and their families by facilitating early diagnosis and implementing preventive measures.
The identification of BRCA1/2 DH is relatively recent, with BRCA1 and BRCA2 genes discovered in the 1990s followed by subsequent reports of DH.11,13 Initially, DH was more frequently observed among Ashkenazi Jews, with only a few cases reported in Japan since 2015.14,17 According to Rebbeck et al, the prevalence of DH cases is approximately 0.3% among BRCA mutation carriers.4 In the OlympiAD and OlympiA trials, DH was found in four of 302 (1.3%) and seven of 1836 (0.3%) BRCA mutation carriers, respectively.18 19 Although DH is rare, it occurs worldwide, although in small numbers.
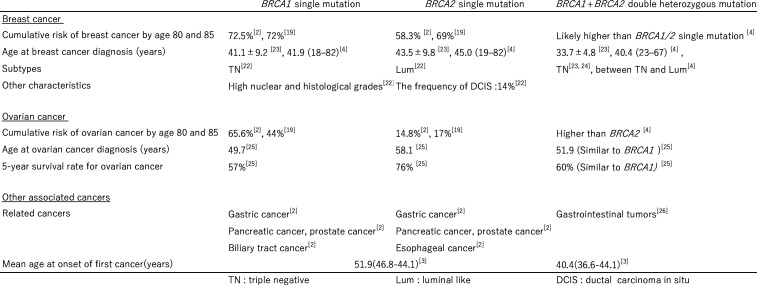
In Japan, the cumulative cancer risk for single BRCA variant carriers has been reported to be 72.5% for breast cancer with BRCA1 and 58.3% with BRCA2. For ovarian cancer, the risk is 65.6% with BRCA1 and 14.8% with BRCA2, with some regional variations.2 20 Notably, the age of onset for breast cancer is significantly lower in BRCA mutation carriers than in non-carriers.21 Breast cancer subtypes differ between BRCA1 and BRCA2 mutations. BRCA1 is linked to triple-negative (TN) breast cancer, an aggressive subtype that is negative for hormone receptors and HER2. In contrast, BRCA2 is associated with hormone receptor-positive, HER2-negative (Luminal) breast cancer.22 In addition to pancreatic and prostate cancers, BRCA1 is associated with bile duct cancer, BRCA2 with esophageal cancer and both BRCA1 and BRCA2 with gastric cancer.2 Thus, the risk of onset, age of onset, tumour characteristics and cancer types differ between BRCA1 and BRCA2.
While BRCA1 and BRCA2 mutations each confer distinct risks and clinical characteristics, individuals carrying DH exhibit unique features that differ from those seen in single-mutation carriers (figure 3). Rebbeck et al reported that DH carriers of BRCA1/2 mutations have a higher risk of developing breast cancer compared with single BRCA mutation carriers. Furthermore, DH carriers tend to develop breast cancer at an earlier age than BRCA2 carriers, with an onset age comparable to that of BRCA1 carriers. Additionally, DH carriers exhibit characteristics of an intermediate subtype between BRCA1 and BRCA2.4 Conversely, some Asian studies have indicated that DH carriers not only present with breast cancer at an earlier age than single BRCA mutation carriers but also tend to exhibit a TN subtype, which is associated with poorer prognosis.23 24 In the context of ovarian cancer, DH carriers are diagnosed at a median age of 51.9 years, which is significantly younger than the 58.1 years for BRCA2 carriers and comparable to the 49.7 years for BRCA1 carriers.25 The 5 year survival rate for DH carriers is 60%, which is markedly lower than the 76% for BRCA2 carriers and similar to the 57% for BRCA1 carriers.25 DH carriers are also associated with gastrointestinal tumours.26 Based on these findings, the presence of loss-of-function mutations in two distinct tumour suppressor genes may potentially result in synergistic effects.
Figure 3. Comparison of the characteristics and risks of breast, ovarian and other cancers associated with BRCA1 single mutation, BRCA2 single mutation and BRCA1+BRCA2 double heterozygosity.
This discussion focuses on the necessity of surveillance for BRCA1/2 DH carriers. Several reviews of DH cases support the potential synergistic interactions between genes. It has been reported that non-cancerous DH states exhibit more severe phenotypes compared with single-gene mutations.27 28 Furthermore, even in DH carriers of BRCA1/2, where one allele is the same as that of a single BRCA mutation carrier, it is suggested that the onset of primary cancer may occur earlier and with greater severity.3 29 This indicates that, compared with single BRCA mutations, DH carriers may benefit from more intensive follow-up, monitoring and preventive surgeries from a younger age.
While there are opinions suggesting no significant differences in age of onset or cumulative lifetime risk,30 31 it is a fact that this case represents the earliest incidence of breast cancer among relatives. Although other relatives did not experience recurrence of breast cancer, the patient in question had a high risk of recurrence, highlighting the importance of more frequent follow-up. In addition to intensive surveillance, it is crucial to continue providing information regarding preventive surgeries such as CRRM and RRSO.
Moreover, according to research by Raffaele et al, 75.0% of BRCA1/2 DH carriers develop primary cancers, with 33.3% of them developing multiple cancers.32 DH carriers are suggested to have an earlier age of cancer onset compared with single mutations and a higher likelihood of developing severe diseases.29 These findings indicate the need for a more cautious approach to cancer information among relatives, not only concerning known associated cancers such as breast and ovarian cancer.
In this case, in addition to the family history of BRCA-related breast cancer, pancreatic cancer and peritoneal cancer, there were also multiple cases of leukaemia and ureteral cancer. Bi-allelic mutations in BRCA2 are associated with Fanconi anaemia, a rare chromosomal instability syndrome characterised by childhood aplastic anaemia, leukaemia and cancer susceptibility.33 However, there are currently no reports linking BRCA to age-related leukaemia or ureteral cancer, and it is necessary to focus on this potential relationship in the future.34 4 Since monitoring strategies tailored to specific mutation sites in DH patients have been proven effective, it may be possible to achieve secondary cancer prevention by meticulously updating the pedigree chart rather than relying solely on single mutation carriers. This could lead to more effective risk management and early detection.
In conclusion, BRCA1/2 DH carriers exhibit characteristics of both mutations and have mutations linked to a higher risk of early-onset and more severe cancer. Therefore, rather than relying only on single BRCA mutation carriers, more intensive and dual-focused surveillance strategies are needed. Considering the risks of multiple cancers and other BRCA-related cancers, it is essential to continuously provide individualised optimal counselling and surveillance while updating the pedigree chart. DH cases remain relatively rare, and further research in this field is warranted. Future studies are expected to clarify the unique risks and management strategies for DH carriers.
Patient’s perspective.
Thank you very much for this opportunity. Your help with my mother was also very much appreciated until the very end.
Learning points.
Timely genetic counselling, family screening and genetic testing in hereditary breast cancer influence treatment and prevention strategies.
These interventions provide significant benefits to patients and their families through ongoing management and support.
Patients with double heterozygosity for BRCA1 and BRCA2 exhibit characteristics of both mutations, resulting in an increased risk of early-onset and severe cancers, which necessitates more intensive and dual-focused surveillance strategies.
Acknowledgements
This paper has been supported by the Comprehensive Support Project for Oncological Research of Breast Cancer (CSPOR-BC).
Footnotes
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Case reports provide a valuable learning resource for the scientific community and can indicate areas of interest for future research. They should not be used in isolation to guide treatment choices or public health policy.
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Consent obtained directly from patient(s).
References
- 1.Tung N, Lin NU, Kidd J, et al. Frequency of Germline Mutations in 25 Cancer Susceptibility Genes in a Sequential Series of Patients With Breast Cancer. J Clin Oncol. 2016;34:1460–8. doi: 10.1200/JCO.2015.65.0747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Momozawa Y, Iwasaki Y, Parsons MT, et al. Germline pathogenic variants of 11 breast cancer genes in 7,051 Japanese patients and 11,241 controls. Nat Commun. 2018;9:4083. doi: 10.1038/s41467-018-06581-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Heidemann S, Fischer C, Engel C, et al. Double heterozygosity for mutations in BRCA1 and BRCA2 in German breast cancer patients: implications on test strategies and clinical management. Breast Cancer Res Treat. 2012;134:1229–39. doi: 10.1007/s10549-012-2050-4. [DOI] [PubMed] [Google Scholar]
- 4.Rebbeck TR, Friebel TM, Mitra N, et al. Inheritance of deleterious mutations at both BRCA1 and BRCA2 in an international sample of 32,295 women. Breast Cancer Res. 2016;18:112. doi: 10.1186/s13058-016-0768-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hereditary breast and ovarian cancer (HBOC) clinical practice guidelines 2021. Tokyo: Kanehara & Co., Ltd; 2021. Japan hereditary breast and ovarian cancer comprehensive medical care system organization. [Google Scholar]
- 6.National Comprehensive Cancer Network Genetic/familial high-risk assessment: breast, ovarian, and pancreatic. NCCN clinical practice guidelines in oncology (NCCN guidelines) 2021. https://www2.tri-kobe.org/nccn/guideline/gynecological/english/genetic_familial.pdf Available. [DOI] [PubMed]
- 7.Sessa C, Balmaña J, Bober SL, et al. Risk reduction and screening of cancer in hereditary breast-ovarian cancer syndromes: ESMO Clinical Practice Guideline. Ann Oncol. 2023;34:33–47. doi: 10.1016/j.annonc.2022.10.004. [DOI] [PubMed] [Google Scholar]
- 8.Ain Q, Richardson C, Mutebi M, et al. Does mainstream BRCA testing affect surgical decision-making in newly-diagnosed breast cancer patients? Breast. 2023;67:30–5. doi: 10.1016/j.breast.2022.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Madar L, Majoros V, Szűcs Z, et al. Double Heterozygosity for Rare Deleterious Variants in the BRCA1 and BRCA2 Genes in a Hungarian Patient with Breast Cancer. Int J Mol Sci. 2023;24:15334. doi: 10.3390/ijms242015334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Garcia-Casado Z, Romero I, Fernández-Serra A, et al. A de novo complete BRCA1 gene deletion identified in a Spanish woman with early bilateral breast cancer. BMC Med Genet. 2011;12:134. doi: 10.1186/1471-2350-12-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miki Y, Swensen J, Shattuck-Eidens D, et al. A strong candidate for the breast and ovarian cancer susceptibility gene BRCA1. Science. 1994;266:66–71. doi: 10.1126/science.7545954. [DOI] [PubMed] [Google Scholar]
- 12.Wooster R, Neuhausen SL, Mangion J, et al. Localization of a breast cancer susceptibility gene, BRCA2, to chromosome 13q12-13. Science. 1994;265:2088–90. doi: 10.1126/science.8091231. [DOI] [PubMed] [Google Scholar]
- 13.Ramus SJ, Friedman LS, Gayther SA, et al. A breast/ovarian cancer patient with germline mutations in both BRCA1 and BRCA2. Nat Genet. 1997;15:14–5. doi: 10.1038/ng0197-14. [DOI] [PubMed] [Google Scholar]
- 14.Lavie O, Narod S, Lejbkowicz F, et al. Double heterozygosity in the BRCA1 and BRCA2 genes in the Jewish population. Ann Oncol. 2011;22:964–6. doi: 10.1093/annonc/mdq460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nomizu T, Matsuzaki M, Katagata N, et al. A case of familial breast cancer with double heterozygosity for BRCA1 and BRCA2 genes. Breast Cancer. 2015;22:557–61. doi: 10.1007/s12282-012-0432-4. [DOI] [PubMed] [Google Scholar]
- 16.Kitahara M, Hozumi Y, Machinaga M, et al. A case of triple-negative breast cancer with germline pathogenic variants in both BRCA1 and BRCA2. Case Rep Oncol. 2021;14:1645–51. doi: 10.1159/000520148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nakamura S, Takahashi M, Tozaki M, et al. Prevalence and differentiation of hereditary breast and ovarian cancers in Japan. Breast Cancer. 2015;22:462–8. doi: 10.1007/s12282-013-0503-1. [DOI] [PubMed] [Google Scholar]
- 18.Robson M, Im SA, Senkus E, et al. Olaparib for Metastatic Breast Cancer in Patients with a Germline BRCA Mutation. N Engl J Med. 2017;377:523–33. doi: 10.1056/NEJMoa1706450. [DOI] [PubMed] [Google Scholar]
- 19.Tutt ANJ, Garber JE, Kaufman B, et al. Adjuvant Olaparib for patients with BRCA1- or BRCA2-mutated breast cancer. N Engl J Med. 2021;384:2394–405. doi: 10.1056/NEJMoa2105215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA. 2017;317:2402–16. doi: 10.1001/jama.2017.7112. [DOI] [PubMed] [Google Scholar]
- 21.Arai M, Yokoyama S, Watanabe C, et al. Correction: Genetic and clinical characteristics in Japanese hereditary breast and ovarian cancer: first report after establishment of HBOC registration system in Japan. J Hum Genet. 2018;63:541–2. doi: 10.1038/s10038-017-0395-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krammer J, Pinker-Domenig K, Robson ME, et al. Breast cancer detection and tumor characteristics in BRCA1 and BRCA2 mutation carriers. Breast Cancer Res Treat. 2017;163:565–71. doi: 10.1007/s10549-017-4198-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bang YJ, Kwon WK, Nam SJ, et al. Clinicopathological Characterization of Double Heterozygosity for BRCA1 and BRCA2 Variants in Korean Breast Cancer Patients. Cancer Res Treat. 2022;54:827–33. doi: 10.4143/crt.2021.791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Noh JM, Choi DH, Nam SJ, et al. Characteristics of double heterozygosity for BRCA1 and BRCA2 germline mutations in Korean breast cancer patients. Breast Cancer Res Treat. 2012;131:217–22. doi: 10.1007/s10549-011-1718-5. [DOI] [PubMed] [Google Scholar]
- 25.Le Page C, Rahimi K, Rodrigues M, et al. Clinicopathological features of women with epithelial ovarian cancer and double heterozygosity for BRCA1 and BRCA2: A systematic review and case report analysis. Gynecol Oncol. 2020;156:377–86. doi: 10.1016/j.ygyno.2019.11.019. [DOI] [PubMed] [Google Scholar]
- 26.Zuradelli M, Peissel B, Manoukian S, et al. Four new cases of double heterozygosity for BRCA1 and BRCA2 gene mutations: clinical, pathological, and family characteristics. Breast Cancer Res Treat. 2010;124:251–8. doi: 10.1007/s10549-010-0853-8. [DOI] [PubMed] [Google Scholar]
- 27.Richard P, Isnard R, Carrier L, et al. Double heterozygosity for mutations in the beta-myosin heavy chain and in the cardiac myosin binding protein C genes in a family with hypertrophic cardiomyopathy. J Med Genet. 1999;36:542–5. [PMC free article] [PubMed] [Google Scholar]
- 28.Brüning JC, Winnay J, Bonner-Weir S, et al. Development of a novel polygenic model of NIDDM in mice heterozygous for IR and IRS-1 null alleles. Cell. 1997;88:561–72. doi: 10.1016/s0092-8674(00)81896-6. [DOI] [PubMed] [Google Scholar]
- 29.Sokolenko AP, Bogdanova N, Kluzniak W, et al. Double heterozygotes among breast cancer patients analyzed for BRCA1, CHEK2, ATM, NBN/NBS1, and BLM germ-line mutations. Breast Cancer Res Treat. 2014;145:553–62. doi: 10.1007/s10549-014-2971-1. [DOI] [PubMed] [Google Scholar]
- 30.Leegte B, van der Hout AH, Deffenbaugh AM, et al. Phenotypic expression of double heterozygosity for BRCA1 and BRCA2 germline mutations. J Med Genet. 2005;42:e20. doi: 10.1136/jmg.2004.027243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Laish I, Friedman E, Levi-Reznick G, et al. Double heterozygotes of BRCA1/BRCA2 and mismatch repair gene pathogenic variants: case series and clinical implications. Breast Cancer Res Treat. 2021;188:685–94. doi: 10.1007/s10549-021-06258-9. [DOI] [PubMed] [Google Scholar]
- 32.Palmirotta R, Lovero D, Stucci LS, et al. Double Heterozygosity for BRCA1 Pathogenic Variant and BRCA2 Polymorphic Stop Codon K3326X: A Case Report in a Southern Italian Family. Int J Mol Sci. 2018;19:285. doi: 10.3390/ijms19010285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fradet-Turcotte A, Sitz J, Grapton D, et al. BRCA2 functions: from DNA repair to replication fork stabilization. Endocr Relat Cancer. 2016;23:T1–17. doi: 10.1530/ERC-16-0297. [DOI] [PubMed] [Google Scholar]
- 34.Nassar AH, Abou Alaiwi S, AlDubayan SH, et al. Prevalence of pathogenic germline cancer risk variants in high-risk urothelial carcinoma. Genet Med. 2020;22:709–18. doi: 10.1038/s41436-019-0720-x. [DOI] [PMC free article] [PubMed] [Google Scholar]