Abstract
Background
Little is known on the vertical transmission of human papillomavirus (HPV) and on the dynamics of HPV among children. Our objective was to determine the risk of HPV recurrence, persistence, and incidence over 2 years of age among children born to HPV-positive mothers.
Methods
We conducted the HERITAGE study among pregnant women recruited between 2010 and 2016 in Canada. HPV DNA testing was done on vaginal samples collected during the first and third trimesters of pregnancy, and on conjunctival, oral, pharyngeal, and genital samples collected in children from birth and at every 3–6 months up to 2 years. We estimated the probability of HPV vertical transmission, and of HPV recurrence, persistence, and incidence among children during follow-up. Time to clear HPV among children was estimated using Kaplan-Meier technique.
Results
Among the 422 women with HPV during pregnancy, 390 carried pregnancy to term, and 395 children were born alive including twins/triplets. HPV vertical transmission was estimated at 7.3% (95% confidence interval [CI], 5.0%–10.4%) with a genotype concordance of 85.2%. During the entire follow-up, we observed 91 HPV detections (among 51 children) including 2 recurrent and 1 persistent. Incident genotypes occurred in 26 of the 270 (9.6%) children with valid HPV testing during follow-up. Most HPV infections detected in children cleared with a mean time of 3.9 months (95% CI, 3.6–4.2 months).
Conclusions
HPV vertical transmission and incident HPV occasionally occur during infancy, but the risk of persistence or recurrence is overall very low.
Keywords: human papillomavirus (HPV), pregnancy, children, persistence, recurrence
Vertical human papillomavirus (HPV) transmission and HPV incidence during early childhood are not rare, although most infections in children typically resolve quickly, with few cases of persistence and recurrence.
Graphical Abstract
Graphical Abstract.
This graphical abstract is also available at Tidbit: https://tidbitapp.io/tidbits/hpv-persistence-and-incidence-in-early-childhood-2daa26b9-c42d-47f2-af90-ba5e3d1d2653
Human papillomavirus (HPV) infection is the most widespread sexually transmitted infection in the world and is a necessary cause of cervical cancer [1]. Many studies have been carried out to help us understand the harmful effects of HPV. However, these studies have been extensively focused on adults, whereas relatively little is known about the impact of vertical transmission of HPV and the risk of persistence and recurrence of HPV in children. Since the prevalence of HPV is highest among young people, the most fertile age group, pregnant women are at high risk of HPV infection [2]. Vertical and horizontal HPV transmissions are known to occur [3]. Vertical transmission may occur during conception, in utero, or during childbirth [4–6]. Horizontal transmission through nonsexual means, such as auto-inoculation, hetero-inoculation, or contact with contaminated objects, is also possible [7]. Although HPV infection in children is mostly asymptomatic, it can lead to various types of lesions, such as anogenital warts [8] and recurrent respiratory papillomatosis, a rare but highly morbid disease in children [9]. Previous studies estimated the risk of vertical transmission from an HPV-positive mother to a child at around 10% [2, 10, 11]. In adults, most HPV infections do not persist more than 1–2 years, but little is known about the natural history of HPV in children [2, 4]. A study suggests that HPV can persist for several years in children [12]. Further study on the risk of vertical transmission of HPV and on the dynamic of HPV among children is needed. Our objective was to determine the risk of HPV recurrence, persistence, and incidence over 2 years of age among children born to HPV-positive mothers.
MATERIALS AND METHODS
The HERITAGE (Human Papillomavirus perinatal transmission and risk of HPV persistence among children) study, a multicenter cohort, was set up to study different outcomes related to HPV infection during pregnancy. The design, methods, and some results have previously been published [11, 13–18]. In brief, 1051 pregnant women of at least 18 years of age were enrolled between 2010 and 2016 during the first trimester of their pregnancies (between 6 and 14 weeks of gestation) from 3 academic hospitals in Montreal, Canada (Centre hospitalier universitaire Sainte-Justine, Centre hospitalier universitaire de l’Université de Montréal, and Saint-Mary's Hospital) or affiliated clinics. Women were included if they had no human immunodeficiency virus infection, planned to deliver at the participating hospitals, and spoke French or English. The institutional ethics and research review boards of the participating hospitals approved the study protocol, and all participants signed an informed consent form. This analysis includes the 422 pregnant women with HPV infection at baseline. Supplementary Figure 1 provides the flow diagram of participants into this analysis.
At first (baseline) and third (between 32 and 35 weeks of gestation) trimesters of pregnancy, women provided self-collected vaginal samples using a dry Dacron swab (Copan Italia S.p.A.) for HPV DNA testing. After birth, placental biopsies and swabs were also taken on both the maternal and fetal sides of the placenta for HPV DNA testing as previously described [13]. Children born to women with HPV at baseline were followed every 3–6 months from birth until 2 years of age. Samples were collected in children at every visit in eyes (conjunctival sample using FLOQSwabs, Copan Flock Technologies), mouth (oral sample, Dacron swab), throat (pharyngeal sample, Dacron swab), and genital surface (labia or prepuce and scrotum and anal region sample, Dacron swab). Children born to the first 75 women with HPV recruited in the cohort were also followed with additional visits at 3, 4 and 5 years old. Data on lifestyle, medical, and sociodemographic information were collected by means of questionnaires filled out by the mothers during the recruitment and follow-up visits and by a review of the medical records.
Sample Processing and HPV Testing
Swabs collected from mothers and children were directly rinsed into liquid-based cytology vial (PerservCyt) and kept at room temperature pending transfer to laboratory for HPV DNA testing. The biopsies were stored in cryogenic tubes at −70°C until HPV DNA testing. Genotyping of the different HPV genotypes was carried out using the Roche Diagnostic Linear Array Test that detected 36 genotypes comprising high-risk HPV (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68, 73, and 82) and low-risk HPV (6, 11, 26, 34, 40, 42, 44, 53, 54, 61, 62, 67, 69, 70, 71, 72, 81, 83, 84, and 89) [19].
Statistical Analysis
Characteristics of the mothers and children were described using proportions, mean (standard deviation [SD]), and median (interquartile range [IQR]). Detection of HPV in children was described for all sites (conjunctival, oral, pharyngeal, and genital samples), for all visits from birth to 24 months. The probability of transmission of HPV among children born to mothers with HPV was estimated as the detection of HPV found in children at birth or at 3 months divided by the total number of mothers with HPV for individual sites and all sites combined. The proportion of concordance between the genotypes found in the mother (during pregnancy or in the placenta) and those found in the children was estimated.
We also estimated the risk of recurrence, persistence, and incidence of new genotypes in children during follow-up and their 95% confidence intervals (CIs). Recurrent HPV was defined as the redetection of a same genotype that was detected at least at 1 visit (between birth and 24 months), cleared for at least 1 visit, and then was redetected at a subsequent visit. Persistent HPV was defined as a specific HPV genotype that was detected in children for at least 2 consecutive visits (between birth and 24 months). Incident HPV in children was defined as the detection of a new genotype during the follow-up (until 24 months) that was not detected at birth and/or at the 3-month visit. The median (IQR) and mean (95% CI) time to clear genotype-specific HPV episodes in children was estimated using the Kaplan-Meier technique.
RESULTS
Among the 422 women with HPV during pregnancy, 390 carried their pregnancy to term, 2 children were stillbirths, and 395 children were born alive including twin and triplet pregnancies (Supplementary Figure 1). Table 1 summarizes the characteristics of the mothers and their children in the cohort. The mean age of the mothers was 30.7 years (SD, 4.7 years). Most of the children were born vaginally (71.8%) and 28.2% were born by cesarean delivery. Of all mothers, 10.9% were single and 89.1% had a regular partner at baseline. Globally, we tested 7301 samples for HPV DNA among mothers and their children.
Table 1.
Characteristics of the 422 Pregnant Women With Human Papillomavirus Infection in the HERITAGE Study
| Characteristic | Variable | Value |
|---|---|---|
| At baseline | ||
| Age, y | Mean ± SD | 30.7 ± 4.7 |
| Median (IQR) | 30 (27–34) | |
| Range | 19–47 | |
| No. of years of schooling | Mean ± SD | 15.8 ± 0.16 |
| Median (IQR) | 16 (14–18) | |
| Range | 7–28 | |
| Relationship status | Single | 46 (10.9) |
| Regular partner | 376 (89.1) | |
| No. of lifetime sexual partners | <2 | 26 (7.0) |
| 2–4 | 73 (19.6) | |
| 5–9 | 171 (45.8) | |
| ≥10 | 103 (27.6) | |
| Missing | 49 | |
| No. of new sexual partners during the last year | 0 | 401 (95.2) |
| 1 | 17.0 (4.0) | |
| ≥2 | 3 (0.8) | |
| Missing | 1 | |
| Received at least 1 dose of an HPV vaccine | No | 350 (88.8) |
| Yes | 44 (11.2) | |
| Missing | 28 | |
| Ethnicity | White | 329 (77.9) |
| Native African | 37 (8.7) | |
| African American | 3 (0.7) | |
| East Asian | 4 (0.9) | |
| Arab/West Asian | 18 (4.3) | |
| Latin American | 25 (5.9) | |
| Native/Aboriginal People | 1 (0.2) | |
| Othera | 5 (1.2) | |
| Current smoking | No | 357 (84.6) |
| Yes | 65 (15.4) | |
| Cytology result | Normal | 376 (90.2) |
| ASCUS | 26 (6.2) | |
| LSIL | 7 (1.7) | |
| HSIL | 8 (1.9) | |
| Missing | 5 | |
| HPV infection during pregnancy | HR-HPV (at least 1 genotype) | 304 (72.0) |
| LR-HPV (at least 1 genotype) | 292 (69.2) | |
| During pregnancy | ||
| Pregnancy-induced hypertensive disorders | No | 407 (96.7) |
| Yes | 14 (3.3) | |
| Missing | 1 | |
| Gestational diabetes | No | 339 (87.8) |
| Yes | 47 (12.2) | |
| Missing | 36 | |
| Urinary or genital infection | No | 399 (97.3) |
| Yes | 11 (2.7) | |
| Missing | 12 | |
| Pregnancy type | Single pregnancy | 417 (98.8) |
| Multiple pregnancy, twins | 3 (0.7) | |
| Multiple pregnancy, triplets | 2 (0.5) | |
| Pregnancy outcome | Live birthb | 395 (95.2) |
| Spontaneous abortions | 9 (2.2) | |
| Molar pregnancy | 2 (0.5) | |
| Pregnancy termination | 7 (1.7) | |
| Stillbirth | 2 (0.5) | |
| Missing | 12 | |
| Gestational age at delivery, wk (n = 390) | Mean ± SD | 39.1 ± 2.3 |
| Median (IQR) | 39.4 (38.4–40.4) | |
| Range | 20–42 | |
| <28 wk | 2 (0.5) | |
| 28–31 wk | 3 (0.8) | |
| 32–36 wk | 26 (6.7) | |
| ≥37 wk | 357 (92.0) | |
| Loss to follow-up | 2 | |
| Delivery mode (n = 390) | Vaginal | 277 (71.9) |
| Cesarean delivery | 108 (28.1) | |
| Missing | 5 | |
| Children (n = 395b) | ||
| Birth weight, g |
Mean ± SD | 3298.9 ± 539.2 |
| Median (IQR) | 3298.9 (2685–3310) | |
| Range | 415–4640 | |
| <2500 g | 25 (8.5) | |
| ≥2500 g | 369 (91.5) | |
| Missing | 1 | |
| Child sex | Male | 197 (50) |
| Female | 197 (50) | |
| Missing | 1 | |
Data are presented as No. (%) unless otherwise indicated.
Abbreviations: ASCUS, atypical squamous cells of undetermined significance; HPV, human papillomavirus; HR-HPV, high-risk human papillomavirus; HSIL, high-grade squamous intraepithelial lesion; IQR, interquartile range; LR-HPV, low-risk human papillomavirus; LSIL, low-grade squamous intraepithelial lesion; Missing, missing data; SD, standard deviation.
aOther ethnicity includes 4 individuals from Haiti and 1 from the French Caribbean.
bn = 395 live births, including 3 twins and 1 triplet out of 390 pregnancies.
Table 2 presents the detection of genotype-specific HPV in children during follow-up. Overall, HPV was detected in 51 of the 373 (13.7%) children with at least 1 sample tested for HPV during the entire follow-up. Multiple genotypes were detected in 17 of these children. A total of 91 HPV detections was observed in all samples and visits combined in children. Most of the genotypes detected in children were high-risk HPV. The most prevalent genotype found was HPV-66 (3.2% [95% CI, 1.8%–5.5%]), followed by HPV-51 (2.4% [95% CI, 1.3%–4.5%]) and HPV-62 (2.1%, [95% CI, 1.1%–4.2%]). No HPV-11 was detected in the children, and the detection of HPV-6, HPV-16, and HPV-18 was 0.5% (95% CI, .1%–1.9%), 0.8% (95% CI, .3%–2.3%), and 0.8% (95% CI, .3%–2.3%), respectively.
Table 2.
Detection of Human Papillomavirus in Children (n = 373a) During Follow-up in All Sites Combined
| Genotype | No. (%) | 95% CI |
|---|---|---|
| HPV-6 | 2 (0.5) | .1–1.9 |
| HPV-11 | 0 (0) | NA |
| HPV-16 | 3 (0.8) | .3–2.3 |
| HPV-18 | 3 (0.8) | .3–2.3 |
| HPV-26 | 0 (0) | NA |
| HPV-31 | 5 (1.3) | .6–3.1 |
| HPV-33 | 3 (0.8) | .3–2.3 |
| HPV-34 | 0 (0) | NA |
| HPV-35 | 2 (0.5) | .1–1.9 |
| HPV-39 | 6 (1.6) | .7–3.5 |
| HPV-40 | 1 (0.3) | .1–1.5 |
| HPV-42 | 3 (0.8) | .3–2.3 |
| HPV-44 | 2 (0.5) | .1–1.9 |
| HPV-45 | 0 (0) | NA |
| HPV-51 | 9 (2.4) | 1.3–4.5 |
| HPV-52 | 3 (0.8) | .3–2.3 |
| HPV-53 | 2 (0.5) | .1–1.9 |
| HPV-54 | 0 (0) | NA |
| HPV-56 | 0 (0) | NA |
| HPV-58 | 3 (0.8) | .3–2.3 |
| HPV-59 | 0 (0) | NA |
| HPV-61 | 1 (0.3) | .1–1.5 |
| HPV-62 | 8 (2.1) | 1.1–4.2 |
| HPV-66 | 12 (3.2) | 1.8–5.5 |
| HPV-67 | 0 (0) | NA |
| HPV-68 | 0 (0) | NA |
| HPV-69 | 0 (0) | NA |
| HPV-70 | 0 (0) | NA |
| HPV-71 | 0 (0) | NA |
| HPV-72 | 5 (1.3) | .6–3.1 |
| HPV-73 | 1 (0.3) | .1–1.5 |
| HPV-81 | 0 (0) | NA |
| HPV-82 | 4 (1.1) | .4–2.7 |
| HPV-83 | 1 (0.3) | .1–1.5 |
| HPV-84 | 7 (1.9) | .9–3.8 |
| HPV-89 | 5 (1.3) | .6–3.1 |
| No. of children with single HPV genotype | 34 (9.1) | 6.6–12.5 |
| No. of children with multiple HPV genotype | 17 (4.6) | 2.8–7.2 |
| No. of children with HPV detection | 51 (13.7) | 10.5–17.6 |
| Total HR-HPV detected | 54 (14.5) | 11.3–18.4 |
| Total LR-HPV | 37 (9.9) | 7.3–13.4 |
| Total HPV detected | 91 (24.4) | 20.3–29.0 |
Abbreviations: CI, confidence interval; HPV, human papillomavirus; HR-HPV, high-risk human papillomavirus; LR-HPV, low-risk human papillomavirus; NA, not applicable.
an = 373 children with at least 1 valid HPV test during follow-up.
Table 3 displays data on HPV detection in children according to individual sites and visits during follow-up. HPV was detected among 27 of 372 children with valid HPV testing at birth and/or 3-month visit. Based on these data, the probability of transmission of HPV for all sites combined was 7.3% (95% CI, 5.0%–10.4%). If we consider data on HPV detected only at birth (not including that of the 3-month visit), the probability of vertical transmission at all sites combined was 6% (95% CI, 4.0%–8.9%). The conjunctiva was the most important site of HPV detection in children with 13 cases at birth and/or 3-month visit (3.5% [95% CI, 2.0%–5.9%]). The percentage of HPV detection in oral, pharyngeal, and genital sites was 3.0% (95% CI, 1.6%–5.3%), 0.8% (95% CI, .3%–2.5%), and 2.7% (95% CI, 1.4%–4.9%), respectively. Among the children followed until 5 years of age (4 children tested at 3 years, 11 at 4 years, and 5 at 5 years of follow-up), no HPV was detected at any sites.
Table 3.
Number of Children With Detection of Human Papillomavirus, by Site, During Follow-up
| Visit | Conjunctival | Oral | Pharyngeal | Genital | All Sites Combined |
|---|---|---|---|---|---|
| Birth (n = 366) | 11 (3) | 6 (1.6) | 3 (0.8) | 9 (2.5) | 22 (6.0) |
| 1.7–5.3 | .8–3.5 | .3–2.4 | 1.3–4.7 | 4.0–8.9 | |
| 3 mo (n = 218) | 3 (1.4) | 5 (2.3) | 0 (0) | 1 (0.5) | 6 (2.7) |
| .4–4.2 | .9–5.4 | NA | .07–3.2 | 1.2–6.0 | |
| Birth and/or 3-mo visit combined (n = 372a) | 13 (3.5) | 11 (3.0) | 3 (0.8) | 10 (2.7) | 27 (7.3) |
| 2.0–5.9 | 1.6–5.3 | .3–2.5 | 1.4–4.9 | 5.0–10.4 | |
| 6 mo (n = 198) | 3 (1.5) | 3 (1.5) | 0 (0) | 3 (1.5) | 8 (4.0) |
| .5–4.6 | .5–4.6 | NA | .5–4.6 | 2.0–7.9 | |
| 9 mo (n = 210) | 1 (0.5) | 0 (0) | 1 (0.5) | 2 (1.0) | 3 (1.4) |
| .1–3.3 | NA | .1–3.3 | .2–3.8 | .5–4.4 | |
| 12 mo (n = 222) | 5 (2.3) | 2 (0.9) | 2 (0.9) | 1 (0.5) | 6 (2.7) |
| .9–5.3 | .2–3.2 | .2–3.2 | .1–3.2 | 1.2–5.9 | |
| 18 mo (n = 211) | 2 (0.9) | 4 (1.9) | 1 (0.5) | 2 (0.9) | 8 (3.8) |
| .2–3.7 | .7–5.0 | .1–3.3 | .2–3.7 | 1.9–7.4 | |
| 24 mo (n = 200) | 4 (2.0) | 0 (0) | 1 (0.5) | 1 (0.5) | 6 (3.0) |
| .7–5.2 | NA | .1–3.5 | .1–3.5 | 1.3–6.4 |
Data are presented as No. (%) and 95% confidence interval.
Abbreviations: HPV, human papillomavirus; NA, not applicable.
an = 372: total number of children include those with valid HPV testing at birth and/or at 3 months (includes 366 children with HPV testing at birth and 6 children with only HPV testing at the 3-month visit).
Data on recurrence, persistence, and incidence of HPV in children are presented in Table 4. Among the 44 children tested at least for 3 consecutive visits, 2 (4.5% [95% CI, 1.3%–15.1%]) had recurrent HPV (genotype detected that cleared and was redetected in a subsequent visit). The first child was infected with HPV-72 in the conjunctiva and the other child with HPV-31 in the mouth (oral site). The risk of persistence was also very low. Among the 48 children tested at least with 2 consecutive visits, 1 child (2.1% [95% CI, .4%–10.4%]) had a persistent HPV with genotype 72 at the conjunctival site. Among the 270 children with valid HPV testing at birth and/or at 3-month visit and with at least 1 test between 6 and 24 months, 26 (9.6%) children had an incident genotype during follow-up (at least 1 genotype not detected at birth or 3-month visit that was newly detected after that) for a total of 43 incident detections accounting for multiple HPV. The site with most new HPV detections was the conjunctiva, with 16 (5.9%) incident cases, follow by oral (4.1%), genital (3.7%), and pharyngeal (2.2%) sites.
Table 4.
Recurrent, Persistent, and Incident Human Papillomavirus Detection in Children
| Detection | Conjunctival | Oral | Pharyngeal | Genital | All Sites Combined |
|---|---|---|---|---|---|
| Recurrent HPV detection (n = 44a) | |||||
| No. (%) | 1 (2.3) | 1 (2.3) | 0 (0) | 0 (0) | 2 (4.5) |
| 95% CI | .4–11.8 | .4–11.8 | NA | NA | 1.3–15.1 |
| Persistent HPV detection (n = 48b) | |||||
| No. (%) | 1 (2.1) | 0 (0) | 0 (0) | 0 (0) | 1 (2.1) |
| 95% CI | .4–10.4 | NA | NA | NA | .4–10.4 |
| Incident HPV detection (n = 270c) | |||||
| No. (%) | 16 (5.9) | 11 (4.1) | 6 (2.2) | 10 (3.7) | 43 (15.9) |
| 95% CI | 3.7–9.4 | 2.3–7.1 | 1.0–4.8 | 2.0–6.7 | 12.4–20.8 |
Abbreviations: CI, confidence interval; HPV, human papillomavirus; NA, not applicable.
aAnalysis based on 44 children who had at least 3 consecutive visits with valid HPV testing (among the 51 children detected with HPV); 2 recurrent detections (numbers 9 and 47 in Supplementary Table 1).
bAnalysis based on 48 children who had at least 2 consecutive visits with valid HPV testing (among the 51 children detected with HPV); 1 case of persistent detection (number 47 in Supplementary Table 1).
cAnalysis based on 270 children with at least 1 visit with valid HPV testing between 6 months and 24 months and who were HPV negative for a specific genotype at birth and/or 3-month visit; we observed 43 incident HPV detection considering multiple detection among 26 children (numbers 25–51 in Supplementary Table 1, except for number 43 because of invalid HPV testing at birth and/or 3-month visit).
Supplementary Table 1 describes the detection of genotype-specific HPV among women and their children by sites and visits for all children in whom HPV was detected at some point during follow-up. Among the 27 children with HPV detection at birth and/or 3-month visit, 23 had similar HPV genotypes to their mother. Therefore, the proportion of concordance between mother during pregnancy and children at birth and/or 3-month visit was 85.2% (95% CI, 65.4%–94.6%). However, concordance decreases to 33.3% (95% CI, 17.8%–53.6%) when considering genotypes detected between 6 and 24 months, with only 9 of 27 children who had genotype concordance with their mother. One child (number 43 in Supplementary Table 1) had missing data at birth and the 3-month visit; this child showed HPV-53 at 18 months at the genital site, and the genotype was not concordant with the mother. The concordance was 62.7% (95% CI, 48.5%–75.1%) when considering all HPV detected during the entire follow-up (32/51 children).
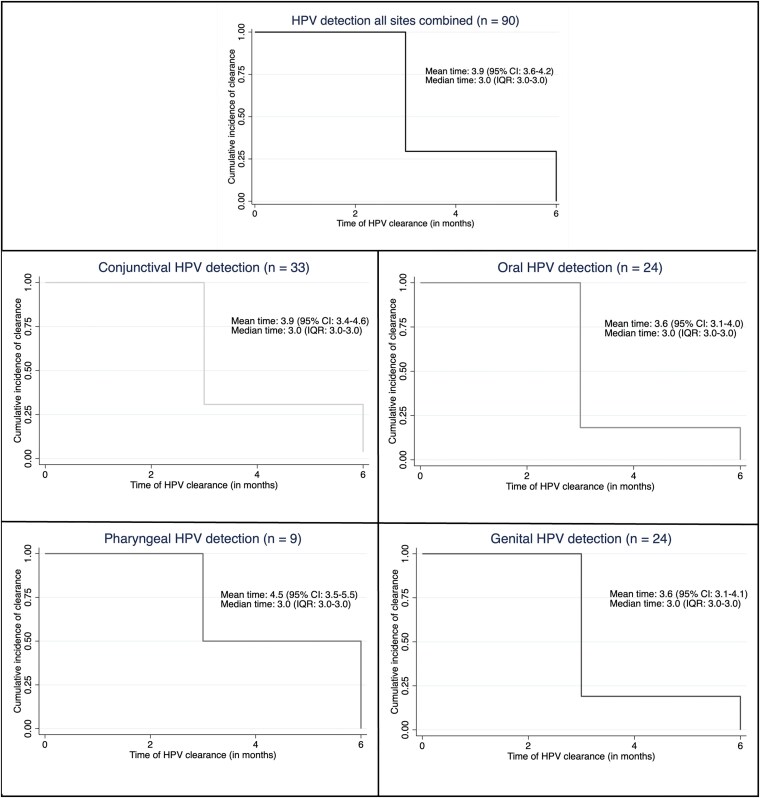
Figure 1 presents data on HPV clearance for any HPV detected in children during the entire follow-up. The Kaplan-Meier curves rely on 90 HPV episodes detected on all sites (90 transient HPVs and 1 persistent HPV). The mean and median clearance times for any HPV episodes for all sites combined were 3.9 months (95% CI, 3.6–4.2 months) and 3.0 months (95% CI, 3.0–3.0 months), respectively. Time to clearance was similar when individual sites were considered with a mean time to clear in conjunctiva of 3.9 months (95% CI, 3.4–4.6 months), in mouth of 3.6 months (95% CI, 3.1–4.0 months), in pharynx of 4.5 months (95% CI, 3.5–5.5 months), and in genitals of 3.6 months (95% CI, 3.1–4.1 months).
Figure 1.
Clearance of genotype-specific human papillomavirus (HPV) episodes detected during follow-up. Data are based on the 90 genotype-specific HPV episodes (out of the 91 HPV detections, only 1 episode was persistent at the conjunctiva; the total number of episodes was therefore 90). Recurrent detections were all considered independently as unique infections that have cleared after a negative visit. Abbreviations: CI, confidence interval; HPV, human papillomavirus; IQR, interquartile range.
DISCUSSION
The probability of vertical transmission of HPV considering all mucosal sites combined was 7.3% (95% CI, 5.0%–10.4%). Of the genotypes detected in children at birth and/or at the 3-month visit, 85.2% were concordant with genotypes found in mothers during pregnancy. These data are consistent with previous studies on HPV vertical transmission [10, 20–23]. A meta-analysis published in 2005 estimated the probability of HPV vertical transmission at 6.5% (95% CI, 5.0%–8.0%) [2]. Other studies such as Smith et al showed a probability of transmission of 5.1% (5/99) considering both oral and genital sites in children, with a concordance between mothers and newborns of 71% [24]. Castellsagué et al reported a probability of HPV vertical transmission of 9.4% (5/53) with oral samples in children, although only 1 child (1/5) had a concordant genotype with their mother [10]. However, Park et al and Hahn et al, who tested children at the oral site, have reported a higher percentage of vertical transmission (18.2% and 20.8%, respectively), with a concordance of 100% in both studies [23, 25]. These percentages were similar in the Finnish Family HPV Study where 22.8% of the 331 children had an HPV-positive oral sample at birth [12]. Differences in methodology such as the sites tested, HPV DNA detection method, or the duration of follow-up might explain differences between studies. For example, most of the studies have tested 1 or 2 sites in children (oral and genital samples). Only 2 studies describe the presence of HPV in the conjunctiva. In our study, we found that 3.2% of conjunctival samples at birth or the 3-month visit were positive for HPV. This is consistent with Balbi et al, who followed 71 mothers with HPV during pregnancy and their children over a 2-year period and showed that 3.2% of conjunctival samples of children were positive [26]. In our study, we calculated a probability of vertical transmission by combining birth and 3-month visits to consider the fact that some children did not have valid tests at birth and to consider a delay of a few weeks to allow HPV detection. With this definition, we estimated a probability of transmission across all sites of 7.3% (95% CI, 5.0%–10.4%) instead of 6.0% (95% CI, 4.0%–8.9%) when considering HPV detected at birth only. However, despite some variations in definition and methodology between studies, all of the results suggest that vertical transmission of HPV to several mucosa (eye, mouth, throat, genitals) does happen.
The occurrence of new genotypes detected during follow-up (not detected at birth or 3 months) was also not rare in our study. Among the 270 children with a valid HPV testing at birth and/or 3-month visit and with a valid HPV test between 6 and 24 months, 26 children (9.6%) acquired at least 1 new genotype during follow-up, totaling 43 new genotypes among these children accounting for multiple genotypes. We observed a decrease in the probability of concordance (33.3%) when considering new detections between 6 and 24 months of age while the concordance was 85.2% for HPV detected in the perinatal period (birth to 3 months). This may suggest that incident HPV detected between 6 and 24 months might be more likely to occur from horizontal transmission from the mother, other family members, or caregivers through, for example, hand contact. However, it is also possible that we missed genotypes during pregnancy because we only collected vaginal and placental samples and not oral and anal samples. It is also possible that HPV comes from the father, as some studies have shown [6, 27]. Only a very few pediatric studies described HPV detection from birth to later in life. In our study, among the subsample of children with 5 years follow-up, incident HPV was not detected after 2 years of life, suggesting a low risk of HPV incidence during childhood. However, few children were followed and tested beyond 2 years of age in our study. It is therefore difficult to draw a clear picture of the long-term dynamics of HPV. Another study has shown a higher risk of HPV during childhood such as an increase in oral HPV detection in children at 6 years of age [12]. All these results suggest that the incidence of HPV during childhood is not rare. One question that remained is whether HPV can persist or reactivate in children.
Among all HPV detections in children during the follow-up, only 2 were recurrent (cleared and reappeared at subsequent visit) and 1 was persistent (detected for at least 2 consecutive visits). It is important to highlight that in all cases of recurrence or persistence, HPV genotypes of both mothers and children were concordant. This may suggest that HPV was vertically transmitted, was causing a low-copy-number infection, and reactivated at multiple sites in children at subsequent visits. Although the definitions of recurrence and persistence are challenging because it is difficult to really distinguish between new detection from reactivation/recurrence or whether HPV is really cleared, the results of this study suggest that most HPV infections detected in children do not persist. This adds to most studies that show that HPV detected during the perinatal period generally does not persist into the first months of life [23, 25, 28]. In the study of Watts et al including 151 infants tested for HPV at birth and at the ages of 6 weeks and 6, 12, 18, 24, and 36 months, an overall prevalence of HPV of 1.5% was found in genital samples, 1.2% in anal samples, and 0% in oral or nasopharyngeal samples, with all positive samples preceded or followed by a negative HPV sample, suggesting absence of persistence [28]. Tenti et al also found similar results after an analysis of 711 mother–newborn pairs. These authors detected HPV in the oropharyngeal mucosa of 11 newborns from 37 women who had HPV-positive vaginal samples at delivery (vertical transmission estimated at 30%), but no HPV was detected in these 11 infants tested 5 weeks after delivery [29]. The study of Park et al, involving 291 pregnant women, showed that although 3.4% (10/291) of newborns had HPV DNA detected in oral samples at birth, none showed HPV positivity 6 months after delivery [23]. In the study by Hahn et al of 469 pregnant women, 15 newborns (3.2%) had HPV DNA detected in oral samples at birth without detection at 2 months postpartum [25]. However, some HPV persistence in children has been observed. In the Finnish cohort study, a total of 48 of the 323 (14.9%) children followed until 6 years of age had persistent oral HPV infection. Interestingly, 4 children positive for HPV-6 at birth still had this genotype detected at their 6-year follow-up visit [12]. Other studies have also shown a certain HPV persistency in children but mainly concern mothers with history of abnormal smears or genital warts, which may cause a problem of generalization [22, 30]. It is therefore important to note that although perinatally acquired HPV persistence is possible, current evidence suggests that it is rare. It is unclear, however, whether HPV infections no longer detected in newborns are really cleared or if some HPV remained undetected in basal cells or are present at low copy number and could reactivate later in childhood. Large cohort studies with a long follow-up from birth until adulthood are needed to answer these questions.
Our study estimated time to HPV clearance among infants. Overall, most children in whom HPV was detected had transient HPV and cleared rapidly. Most genotypes detected in children were cleared with a mean time of 3.9 months (95% CI, 3.6–4.2 months). This estimate is, however, influenced by the time between follow-up visits. It may represent an overestimate because most of the HPV detections observed in our study were transient (occurred in only 1 visit), while their elimination could have occurred several weeks before the subsequent visit where the negative test was observed. These estimates could also be underestimates because we considered the 2 recurrent detections to be independent infections that were eliminated, whereas they could be the same infections that persisted.
Our study has strengths and limitations. HERITAGE is a large cohort study with HPV detection among pregnant mothers and longitudinal follow-up of children with multiple testing to at least 2 years of age. Samples for HPV testing were collected twice during pregnancy and in the placenta as well as in 4 different sites in children (conjunctival, oral, pharyngeal, and genital). HPV DNA was tested using the Roche Linear Array genotyping assay, a well-established method known for its robustness, specificity, and sensitivity in identifying 36 individual HPV genotypes. However, our research also has limitations. Some participants were lost to follow-up or had missing data. This has reduced precision of our estimates. Additionally, only vaginal samples were tested on mothers during pregnancy without follow-up HPV testing after birth, and fathers and other members of the family were not tested. For example, recent studies suggest that paternal HPV infection may contribute to HPV transmission to children [6, 27]. Finally, although follow-up of children in our cohort was at least 2 years, a longer follow-up is important to study recurrence and persistence in children.
CONCLUSIONS
The findings of our study suggest that vertical transmission of HPV to different mucosa (conjunctival, oral, pharyngeal, and genital) of newborns occurs, but rarely results in persistent infection. Considering that HPV testing is increasingly used for cervical cancer screening, it is expected that an increasing number of pregnant women will be aware that they are infected with HPV and be worried about the implication for their child. Our results showing that the risk of HPV recurrence or persistence is very low in children should be reassuring.
Supplementary Material
Contributor Information
Eméra Alice Bénard, Department of Social and Preventive Medicine, Université de Montréal, Montreal, Québec, Canada; Centre de Recherche du Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
Ana Maria Carceller, Centre de Recherche du Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada; Department of Pediatrics, Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
Marie-Hélène Mayrand, Centre de Recherche du Centre Hospitalier de l’Université de Montréal, Montreal, Québec, Canada; Department of Obstetrics and Gynecology, Université de Montréal, Montreal, Québec, Canada.
Jacques Lacroix, Department of Pediatrics, Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
Joseph Niyibizi, Department of Social and Preventive Medicine, Université de Montréal, Montreal, Québec, Canada; Centre de Recherche du Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
Louise Laporte, Centre de Recherche du Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
François Audibert, Department of Obstetrics and Gynecology, Université de Montréal, Montreal, Québec, Canada; Department of Obstetrics and Gynecology, Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
François Coutlée, Centre de Recherche du Centre Hospitalier de l’Université de Montréal, Montreal, Québec, Canada; Départements clinique de Médecine de laboratoire et de Médecine, Services de biologie moléculaire et d’infectiologie, Québec, Canada; Centre Hospitalier de l’Université de Montréal, Montréal, Québec, Canada; Département de Microbiologie, Infectiologie et Immunologie, Université de Montréal, Montreal, Québec, Canada.
Helen Trottier, Department of Social and Preventive Medicine, Université de Montréal, Montreal, Québec, Canada; Centre de Recherche du Centre Hospitalier Universitaire Sainte-Justine, Université de Montréal, Montreal, Québec, Canada.
for the HERITAGE Study Group:
Marie-Hélène Mayrand, François Coutlée, Patricia Monnier, Louise Laporte, Joseph Niyibizi, Monica Zahreddine, Ana Maria Carceller, Paul Brassard, Jacques Lacroix, Diane Francoeur, Marie-Josée Bédard, Isabelle Girard, François Audibert, William Fraser, Helen Trottier, Hasna Meddour, Myra Geoffrion, Kathleen Auclair, Véronique Prévost, Fabiola Correa Botello, Sophie Perreault, Lise-Angela Ouellet, Sylvie Daigle, Sophie Leblanc, Mélanie Robinson, Siham Aboulfadi, Josée Poirier, Audrée Janelle-Montcalm, Isabelle Krauss, Cindy Rousseau, François Beaudoin, Patricia Monnier, Julie Guenoun, Émilie Comète, and Pierre Forest
Supplementary Data
Supplementary materials are available at The Journal of Infectious Diseases online (http://jid.oxfordjournals.org/). Supplementary materials consist of data provided by the author that are published to benefit the reader. The posted materials are not copyedited. The contents of all supplementary data are the sole responsibility of the authors. Questions or messages regarding errors should be addressed to the author.
Notes
Acknowledgments . The HERITAGE study group is composed of Marie-Hélène Mayrand, François Coutlée, Patricia Monnier, Louise Laporte, Joseph Niyibizi, Monica Zahreddine, Ana Maria Carceller, Paul Brassard, Jacques Lacroix, Diane Francoeur, Marie-Josée Bédard, Isabelle Girard, François Audibert, William Fraser, and Helen Trottier. The authors express their gratitude to all study participants and acknowledge the dedicated efforts of the research staff who collaborated with patients and managed specimens across all sites, as well as for their help with the recruitment of patients: Hasna Meddour, Myra Geoffrion, Kathleen Auclair, Véronique Prévost, Fabiola Correa Botello, Sophie Perreault, and Lise-Angela Ouellet (Centre hospitalier universitaire [CHU] Sainte-Justine); Sylvie Daigle, Sophie Leblanc, and Mélanie Robinson (Centre hospitalier universitaire de l’Université de Montréal [CHUM]); Siham Aboulfadi (St Mary's Hospital); Josée Poirier, Audrée Janelle-Montcalm, Isabelle Krauss, and Cindy Rousseau (CHU Sainte-Justine); and François Beaudoin (in memoriam) and Patricia Monnier (in memoriam) (CHU Sainte-Justine). We also express our gratitude to Julie Guenoun, Émilie Comète, and Pierre Forest (CHUM) for their contributions to the DNA extraction and HPV testing procedures.
Author contributions. H. T., M.-H. M., F. C., F. A., A. M. C., and J. L. directly contributed to the conception and design of the study. J. N., L. L., H. T., M.-H. M., and F. C. participated in the acquisition of data. E. A. B., H. T., M.-H. M., and F. C. carried out the data analysis and interpretation of the results. E. A. B., H. T., and A. M. C. wrote the first draft of the manuscript. All authors subsequently read, critically reviewed, revised, and approved the submitted version. H. T. is responsible for the overall content as the guarantor and takes full responsibility for the work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Disclaimer. The Canadian Institutes of Health Research was not involved in the study design; the collection, analysis and interpretation of the data; the writing of the report; or the decision to submit this article for publication.
Financial support . This work was supported by a grant from the Canadian Institutes of Health Research (CIHR) (grant numbers MOP-93564 and MOP-136833 to H. T.). H. T. held a salary award (chercheur-boursier) for the duration of this study from the Fonds de recherche du Québec - Santé (FRQS), and from CIHR (new investigator salary award). M.-H. M. held a salary award (chercheur-boursier clinicien) from the FRQS until 2022. Funding for quality control of HPV testing was provided in part by the AIDS and Infectious Disease Network of FRQS to F. C.
References
- 1. Doorbar J, Egawa N, Griffin H, Kranjec C, Murakami I. Human papillomavirus molecular biology and disease association. Rev Med Virol 2015; 25(Suppl 1):2–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Medeiros LR, Ethur AB, Hilgert JB, et al. Vertical transmission of the human papillomavirus: a systematic quantitative review. Cad Saude Publica 2005; 21:1006–15. [DOI] [PubMed] [Google Scholar]
- 3. Syrjänen S. Current concepts on human papillomavirus infections in children. APMIS 2010; 118:494–509. [DOI] [PubMed] [Google Scholar]
- 4. Ardekani A, Taherifard E, Mollalo A, et al. Human papillomavirus infection during pregnancy and childhood: a comprehensive review. Microorganisms 2022; 10:1932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Weinberg M, Sar-Shalom Nahshon C, Feferkorn I, Bornstein J. Evaluation of human papilloma virus in semen as a risk factor for low sperm quality and poor in vitro fertilization outcomes: a systematic review and meta-analysis. Fertil Steril 2020; 113:955–69.e4. [DOI] [PubMed] [Google Scholar]
- 6. Skoczyński M, Goździcka-Józefiak A, Kwaśniewska A. Co-occurrence of human papillomavirus (HPV) in newborns and their parents. BMC Infect Dis 2019; 19:930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Syrjänen S, Puranen M. Human papillomavirus infections in children: the potential role of maternal transmission. Crit Rev Oral Biol Med 2000; 11:259–74. [DOI] [PubMed] [Google Scholar]
- 8. Richart RM, Wright TC Jr. Human papillomavirus. Curr Opin Obstet Gynecol 1992; 4:662–9. [PubMed] [Google Scholar]
- 9. Niyibizi J, Rodier C, Wassef M, Trottier H. Risk factors for the development and severity of juvenile-onset recurrent respiratory papillomatosis: a systematic review. Int J Pediatr Otorhinolaryngol 2014; 78:186–97. [DOI] [PubMed] [Google Scholar]
- 10. Castellsagué X, Drudis T, Canadas MP, et al. Human papillomavirus (HPV) infection in pregnant women and mother-to-child transmission of genital HPV genotypes: a prospective study in Spain. BMC Infect Dis 2009; 9:74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Khayargoli P, Niyibizi J, Mayrand M-H, et al. Human papillomavirus transmission and persistence in pregnant women and neonates. JAMA Pediatr 2023; 177:684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Syrjänen S, Rintala M, Sarkola M, et al. Oral human papillomavirus infection in children during the first 6 years of life, Finland. Emerg Infect Dis 2021; 27:759–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Trottier H, Mayrand MH, Coutlée F, et al. Human papillomavirus (HPV) perinatal transmission and risk of HPV persistence among children: design, methods and preliminary results of the HERITAGE study. Papillomavirus Res 2016; 2:145–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Zahreddine M, Mayrand MH, Therrien C, et al. Antibodies to human papillomavirus types 6, 11, 16 and 18: vertical transmission and clearance in children up to two years of age. EClinicalMedicine 2020; 21:100334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Niyibizi J, Mayrand MH, Audibert F, et al. Association between human papillomavirus infection among pregnant women and preterm birth. JAMA Netw Open 2021; 4:e2125308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Niyibizi J, Mayrand MH, Audibert F, et al. Risk factors for placental human papillomavirus infection. Sex Transm Infect 2022; 98:575–81. [DOI] [PubMed] [Google Scholar]
- 17. Khayargoli P, Mayrand MH, Niyibizi J, et al. Association between human papillomavirus 16 viral load in pregnancy and preterm birth. Viruses 2024; 16:298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Nantel É, Mayrand MH, Audibert F, et al. Association between the mode of delivery and vertical transmission of human papillomavirus. Viruses 2024; 16:303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Muñoz N, Bosch FX, de Sanjosé S, et al. Epidemiologic classification of human papillomavirus types associated with cervical cancer. N Engl J Med 2003; 348:518–27. [DOI] [PubMed] [Google Scholar]
- 20. Pakarian F, Kaye J, Cason J, et al. Cancer associated human papillomaviruses: perinatal transmission and persistence. Br J Obstet Gynaecol 1994; 101:514–7. [DOI] [PubMed] [Google Scholar]
- 21. Puranen M, Yliskoski M, Saarikoski S, Syrjänen K, Syrjänen S. Vertical transmission of human papillomavirus from infected mothers to their newborn babies and persistence of the virus in childhood. Am J Obstet Gynecol 1996; 174:694–9. [DOI] [PubMed] [Google Scholar]
- 22. Rombaldi RL, Serafini EP, Mandelli J, Zimmermann E, Losquiavo KP. Perinatal transmission of human papillomavirus DNA. Virol J 2009; 6:83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Park H, Lee SW, Lee IH, et al. Rate of vertical transmission of human papillomavirus from mothers to infants: relationship between infection rate and mode of delivery. Virol J 2012; 9:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Smith EM, Parker MA, Rubenstein LM, Haugen TH, Hamsikova E, Turek LP. Evidence for vertical transmission of HPV from mothers to infants. Infect Dis Obstet Gynecol 2010; 2010:326369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hahn HS, Kee MK, Kim HJ, et al. Distribution of maternal and infant human papillomavirus: risk factors associated with vertical transmission. Eur J Obstet Gynecol Reprod Biol 2013; 169:202–6. [DOI] [PubMed] [Google Scholar]
- 26. Balbi G, Schiattarella A, Fasulo D, et al. Vertical transmission of human papillomavirus: experience from a center of southern Italy. Minerva Obstetr Gynecol 2023; 75:45–54. [DOI] [PubMed] [Google Scholar]
- 27. Suominen NT, Luukkaala TH, Laprise C, et al. Human papillomavirus concordance between parents and their newborn offspring: results from the Finnish Family Human Papillomavirus Study. J Infect Dis 2024; 229:448–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Watts DH, Koutsky LA, Holmes KK, et al. Low risk of perinatal transmission of human papillomavirus: results from a prospective cohort study. Am J Obstet Gynecol 1998; 178:365–73. [DOI] [PubMed] [Google Scholar]
- 29. Tenti P, Zappatore R, Migliora P, Spinillo A, Belloni C, Carnevali L. Perinatal transmission of human papillomavirus from gravidas with latent infections. Obstet Gynecol 1999; 93:475–9. [DOI] [PubMed] [Google Scholar]
- 30. Cason J, Kaye JN, Jewers RJ, et al. Perinatal infection and persistence of human papillomavirus types 16 and 18 in infants. J Med Virol 1995; 47:209–18. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.