Abstract
Needle-based injections currently enable the administration of a wide range of biomacromolecule therapies across the body, including the gastrointestinal tract1–3, through recent developments in ingestible robotic devices4–7. However, needles generally require training, sharps management and disposal, and pose challenges for autonomous ingestible systems. Here, inspired by the jetting systems of cephalopods, we have developed and evaluated microjet delivery systems that can deliver jets in axial and radial directions into tissue, making them suitable for tubular and globular segments of the gastrointestinal tract. Furthermore, they are implemented in both tethered and ingestible formats, facilitating endoscopic applications or patient self-dosing. Our study identified suitable pressure and nozzle dimensions for different segments of the gastrointestinal tract and applied microjets in a variety of devices that support delivery across the various anatomic segments of the gastrointestinal tract. We characterized the ability of these systems to administer macromolecules, including insulin, a glucagon-like peptide-1 (GLP1) analogue and a small interfering RNA (siRNA) in large animal models, achieving exposure levels similar to those achieved with subcutaneous delivery. This research provides key insights into jetting design parameters for gastrointestinal administration, substantially broadening the possibilities for future endoscopic and ingestible drug delivery devices.
Subject terms: Biomedical engineering, Drug delivery
Tethered or ingestible delivery systems that deliver liquid microjets in axial and radial directions can be used to deliver macromolecules to different parts of the gastrointestinal tract with good bioavailability.
Main
The earliest use of hypodermic needles for drug delivery in a large mammal was documented in 1656 by Sir Christopher Wren, who performed intravenous injections of alcohol and opium in dogs8,9. Since then, hypodermic needles have become the mainstay for administration of therapeutic agents, including macromolecules such as insulin, vaccines and monoclonal antibodies. Endoscopic injections to gastrointestinal tract organs have also been widely adopted for various treatments1–3. However, needle-based drug administration results in the burden of sharps management, which is a challenge for both patients and medical professionals. Furthermore, a recent study found that recipients of injections and prescribing physicians prefer oral dosing for administration of macromolecules, including with increased frequency and larger-than-typical (000) capsules10. Recent developments in the field of drug delivery have demonstrated the potential to deliver a broad set of macromolecules through self-administrable, robotic ingestible devices, thereby optimizing the administration of therapies that are traditionally reliant on parenteral routes. These autonomous systems have included ones capable of self-orientation4, self-unfolding5, self-inflating6 and hypodermic needle retraction7, with demonstrated success in preclinical or early clinical studies. However, they have centred on the application of solid needle formats for delivery. By contrast, microjet injection represents a needle-free mode of administration in which a collimated, pressurized liquid stream is used to simultaneously penetrate tissue and deliver the drug. It has previously been applied and characterized for cutaneous delivery of macromolecules11, and has been applied to the buccal mucosa for vaccination in rabbits12.
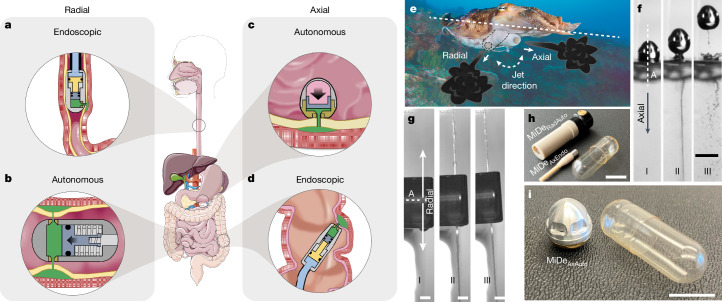
Drawing inspiration from the unique locomotion and ink-expulsion capabilities exhibited by cephalopods, we recognized the potential of directional jet delivery in the gastrointestinal tract. Cephalopods use pressure augmentation in their mantle to propel water and ink through a manoeuverable siphon, creating directional thrust or ink clouds to assist in the distraction and evasion of predators13 (Fig. 1e). We took inspiration from this natural phenomenon to design different microjet delivery (MiDe) systems, each with a unique, axial or radial jet orientation for precise targeting of diverse conditions within the gastrointestinal tract. There are two crucial high-level design features of MiDe systems for the gastrointestinal tract. First, there is the means of localization by which the jet is oriented with the mucosa. Axial systems target cavernous or globular organs such as the stomach or colon, in which it is critical to actively localize the axis of the jet, whereas radial systems are more suitable for narrower tubular organs such as the oesophagus or small intestine, in which localization of the jet is passively assured by its natural proximity during transit. Second, there is the level of autonomy of the system. Tethered systems are ideal for endoscopic applications in which the therapeutic delivery event is overseen and triggered at a precise location and moment by a gastroenterologist, whereas autonomous or ‘self-standing’ systems are compatible with oral self-administration. Here we draw from the principles of cephalopod locomotion to recognize the medical necessity for axial and radial jet delivery in various contexts within the gastrointestinal tract. We detail the prototyping, characterization and application of jet injection devices tailored for different gastrointestinal segments, including their deployment in the stomach and small intestine. Our objective was to establish both tethered and autonomous MiDe systems that are capable of supporting radial and axial delivery (Fig. 1). We initiated the development process by examining the dynamics of jetting in vitro, followed by ex vivo assessments in potential target tissues to ascertain the conditions for optimal intramucosal delivery. Subsequently, we conducted in vivo trials using prototype devices with macromolecular therapeutics in large animal models, including pigs and dogs, enabling us to measure systemic drug levels and assess effectiveness. This multifaceted approach, incorporating both preclinical and in vivo studies, presents a significant step toward expanding the potential for advanced, endoscopic and ingestible drug delivery jetting devices.
Fig. 1. Axial and radial MiDe jetting concepts for needle-free drug delivery to gastrointestinal organs.
a, Radial endoscopic concept for jet delivery in the oesophagus (MiDeRadEndo). b, Radial autonomous concept for jet delivery in the small intestine (MiDeRadAuto). c, Axial autonomous concept for jet delivery in the stomach (MiDeAxAuto). d, Axial endoscopic concept for jet delivery in the colon (MiDeAxEndo). In a–d, organ images are modified with permission from Servier Medical Art under CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/). e, Visualization of a cuttlefish (Sepia officinalis), which is capable of actively adjusting the jet direction relative to its axis of rotation (indicated by the dashed line) and comparison between two distinct funnel orientations of the cuttlefish in axial and radial directions (image used in modified form with permission from Aleksei Permiakov). f,g, Corresponding MiDe drug delivery devices with sequential frames capturing the jet dynamics from the axial MiDeAxAuto delivery system (f; scale bar, 1 cm) and the radial MiDeRadAuto delivery system (g; scale bars, 2 mm), before triggering (I), during jet expulsion (II) and after the expulsion ceases (III). A is the axis of rotation. h, Photograph of the radial autonomous (MiDeRadAuto) and axial endoscopic (MiDeAxEndo) drug delivery systems alongside a 000 capsule for size reference. Scale bar, 1 cm. i, Photograph showing the axial autonomous gastric delivery system (MiDeAxAuto) alongside a 000 capsule for size reference. Scale bar, 1 cm.
Ex vivo tissue jetting experiments
After fully characterizing the in vitro performance of our experimental jetting apparatus (for example, jetting period, force, power and pressure factor; Extended Data Figs. 1 and 2 and Supplementary Methods 1–3), we performed a set of ex vivo jetting studies using tissue samples from the cheek, oesophagus, stomach, jejunum, colon and rectum of female Yorkshire pigs to identify the threshold conditions for an effective delivery to the submucosa. We first fixed the diameter of the nozzle at 257 µm and varied the input pressure (corrected mean ampule pressure; Supplementary Methods 3, equation (E9)) between 2.9 and 55.4 bar, injecting each tissue type with a micro-computed tomography (micro-CT) contrast agent and green dye solution. We then performed additional jetting studies—varying both ampule pressure and nozzle size—on tissue from the jejunum and stomach, since the small intestines and stomach would ultimately serve as the target organs for our proof-of-concept prototype devices.
Extended Data Fig. 1. Setup for jetting performance study and physical characterisation of jet dynamics.
a, Diagram of an ideal jetting system. b, Diagram of our test-stand for the jetting mechanistic study. When the clamp is released, the hand-held jetting device can be removed so that the ampule can be refilled. c, Jetting impingement force vs. time profiles for four different ampule pressures (10.6 Bar, 14.1 Bar, 17.7 Bar and 21.2 Bar; n = 3 technical replicates per ampule pressure). For all experiments shown in this figure we used our pressure-regulated jetting test stand, a nozzle with an orifice diameter of 257 µm and deionized water (ρ = 1000 kg·m−3, ν = 1 cP) as the working fluid. d, Measured jetting period for each of the four ampule pressures. The columns represent experimental performance, while the dashed line represents theoretical performance per equation (E2) (see Supplementary Methods 1). e, Measured steady-state delivery force for the four ampule pressures. The theoretical curve was calculated using equation (E3). f, Measured delivery power for each of the four ampule pressures determined by inputting each of the force measurements into equation (E4). The theoretical curve was calculated using equation (E5). g, Experimental pressure factor from each of the four ampule pressures, calculated by inputting force measurements and corresponding theoretical values into equation (E8) (see Supplementary Methods 3). In (d-g), individual values are shown as black dots.
Extended Data Fig. 2. Summary of results for jetting performance study.
a, Summary of jetting period values as determined by calculating the elapsed time between the beginning and end of the jetting event. b, Summary of steady-state forces as measured directly by the force transducer. c, Summary of steady-state flow rate calculated from the force using the Bernoulli energy balance (Supplementary Methods 1). d, Summary of peak jetting forces as measured directly by the force transducer. e, Summary of percentage force overshoot values as calculated by equation (E6) (see Supplementary Methods 3). These values were used to generate the linear regressions in Supplementary Fig. 2 and Supplementary Table 2. f, Summary of steady-state power values as calculated by Equation E7. g, Summary of pressure factor values as calculated by Equation E8. These values were used to generate the linear regressions in Supplementary Fig. 1 and Supplementary Table 1. In (a-g), each different shade of the horisontal columns represents a certain nozzle diameter ranging from 167 µm (darkest) to 551 µm (lightest). Data are mean (reflected by the column length and written next to each column) and individual values (black dots) (n = 3 technical replicates). The pressure labels on each horisontal column in (a) are valid for columns in the same position in (b-g). h, Results from our evaluation of jetting performance at varying working-fluid viscosities with dj = 257 µm nozzle and pin = 14.1 Bar. Viscosity was varied by changing concentration of dissolved glycerol. The initial data point shows the performance of deionized water (DIW). The dotted line indicates ideal jetting performance. Data are mean (diamonds), 95% confidence intervals (error bars) and individual values (black dots) (n = 3 technical replicates). i, Comparison of output jetting forces at each of three ampule pressures between DIW and the contrast agent we used in subsequent ex vivo work (dj = 257 µm). j, Comparison of corresponding pressure factors at each of the three ampule pressures between DIW and the contrast agent (dj = 257 µm). In (i-j), data are mean and individual values (black dots) (n = 3 technical replicates).
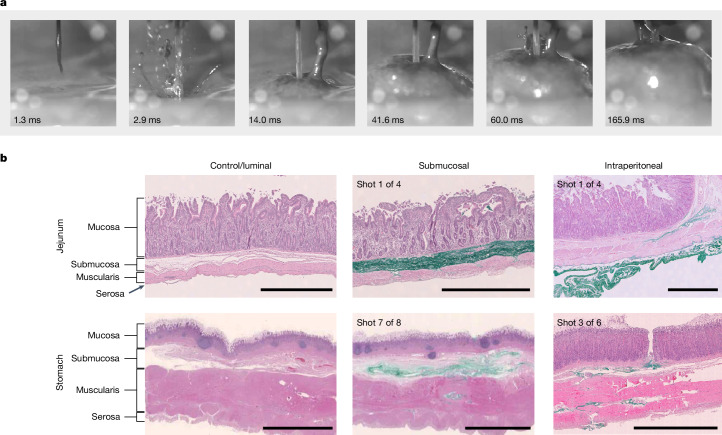
Figure 2a shows a depot forming in jejunal intestinal tissue, as captured by high-speed imaging. Exemplary histological images of resulting dye distribution in jejunal and stomach tissue samples are shown in Fig. 2b. ‘Luminal’ shots represent injections in which only superficial penetration was observed, ‘submucosal’ shots are those in which the fluid was present in the submucosal layer and ‘intraperitoneal’ shots are those in which the fluid penetrated all layers of the tissue. In the submucosal shots, dye was observed to be present primarily in the submucosal tissue for all tissue types. In the intraperitoneal shots, dye is distributed throughout the submucosa, muscularis and serosa (dye that went through the entire thickness of the tissue is not shown).
Fig. 2. Jet dynamics and histological characterization reveals bolus location past the mucosal barrier.
a, Frames from high-speed imaging showing a depot forming in porcine jejunal tissue ex vivo (see Supplementary Video 1). b, Representative haematoxylin and eosin (H&E)-stained histological tissue sections of porcine jejunum and stomach (antrum/lower corpus). Tissue layers are labelled for reference and typical depositions (green) are shown for targeting the submucosa or the intraperitoneal space. Number of independent replicates: n = 1 for jejunum and stomach control/luminal, n = 4 for jejunum submucosal and intraperitoneal, n = 8 for stomach submucosal, and n = 6 for stomach intraperitoneal (all with similar results as the representative sections shown). Scale bars: 1 mm (jejunum) and 5 mm (stomach).
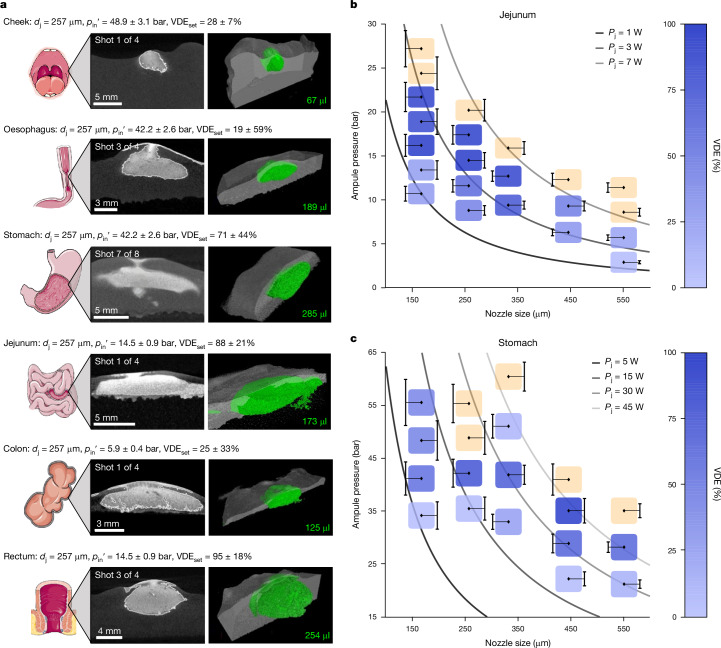
Micro-CT scans of the submucosal volume (before diffusion correction) of exemplary injections from our study with all six tissue types are shown in Fig. 3a. For a comprehensive quantification of successful delivery, we introduce the term volumetric delivery efficiency (VDE), which defines the ratio between the volume contained within the tissue and the volume expelled from the ampule. Compiled VDE results and histology for this study are shown in Extended Data Figs. 4a and 5, respectively. Figure 3b,c shows heat maps with results from our three-dimensional VDE studies in jejunal and stomach tissue. The VDE mean and uncertainty values (along with input parameters, qualitative notes and number of replicates) for every set of injections are tabulated in Supplementary Tables 4 and 5. Consistent with transdermal observations14, our data show that for any given nozzle diameter, there is a positive correlation between jetting power and VDE in gastrointestinal tissue.
Fig. 3. Ex vivo tissue characterization following liquid jet delivery to multiple gastrointestinal locations.
a, Exemplary submucosal depots from the cross-organ study with porcine tissue. For each organ, the image on the left is a single slice from the micro-CT scan. Bright regions represent dye, dark grey regions represent tissue and black indicates void. The image on the right is the corresponding 3D reconstruction, with segmented submucosal fluid displayed in green. For each example, the nozzle diameter (dj), corrected ampule pressure (pin′) and mean VDE for the associated set (VDEset) are included. Organ cartoons are included with permission from Servier Medical Art under CC BY 4.0 (https://creativecommons.org/licenses/by/4.0/). b,c, Heat maps capturing the VDE in jejunal (b) and stomach (antrum/lower corpus) (c) tissue. The sizes of the coloured boxes are arbitrary and the error bars next to each box represent 99% prediction intervals for corrected ampule pressure (per linear regressions of the output performance of the jetting apparatus). The shade of the box surrounding each experimental point indicates the VDE of that point. Light orange boxes indicate shots that were observed to have gone through the tissue (intraperitoneal shots). Lines of constant jetting power (Pj) are also shown for reference.
Extended Data Fig. 4. Organ comparison study results.
a, Each sub-plot in this figure corresponds to one of thirteen corrected ampule pressures marked at the bottom. The corrected ampule pressure interval between each sub-plot is not constant. The upper, middle and lower sections of each sub-plot represent the luminal, submucosal and intraperitoneal tissue region, respectively. Each coloured bar has a length corresponding to 100% of the expelled volume and its position on the vertical axis represents the distribution of expelled volume between the tissue regions at the respective ampule pressure. Error bars represent 95% confidence intervals for the position of the coloured bars. As an example, for the colon tissue bar (brown) in the second sub-plot, approximately 75% of the volume is in the lumen and 25% within the submucosal tissue (and this distribution is between approximately 100%/0% and 45%/55% with 95% confidence). Likewise, for the oesophagus tissue bar (dark blue) in the last sub-plot, approximately 45% of the volume is within the submucosal tissue and 55% in the peritoneum (and this distribution is between approximately 90%/10% and 0%/100% with 95% confidence). All experiments shown in this figure were performed with contrast agent at dj = 257 µm. Number of replicates (tissue samples): n = 2 for oesophagus (28.6 Bar) and rectum (8.8 and 42.2 Bar), n = 6 for oesophagus (48.9 Bar) and stomach (48.9 and 55.4 Bar), n = 8 for stomach (35.5 and 42.2 Bar) and n = 4 for all other organs and ampule pressures. Colon and oesophagus samples originate from one animal, intestine samples from four animals, rectum and cheek samples from two animals, and stomach (antrum/lower corpus) samples from five animals. Exact VDE and uncertainty values, along with qualitative notes can be found in Supplementary Table 3. b-e, Micro-CT scans of a top-, side- and 3D-view of two different shots from the same set of injections. The input parameters and VDE of the set are detailed in the title of each panel. Submucosal depots in jejunal and stomach tissue are shown in (b) and (c), respectively. In these shots, most of the fluid is inside of the tissue. Intraperitoneal shots in jejunal and stomach tissue are shown in (d) and (e), respectively. In these shots, most of the fluid is in the foam beneath the tissue with a small depot visible in the tissue.
Extended Data Fig. 5. Histology from organ comparison study.
Each row shows a histological section of an exemplary injection (shot) in each tissue type from the organ comparison study. A control section for each tissue type (without any injection) is also shown for reference. The tables on the left provide details about the set of injections from which each section was taken. For example, the first table indicates that the adjacent histological section was taken from one of four injections in cheek tissue with dj = 257 µm and pin′ = 35.5 ± 2.2 Bar, the VDE of this set of injections was 17 ± 18% and the typical depth of penetration for this set of injections was submucosal.
In addition to nozzle size and input pressure, we varied the distance between the orifice and tissue surface (standoff) and the angle between the axis of the jet and the tissue surface. From these experiments, we learned that at standoff distances of up to 5 mm, mean VDE changed by approximately −20% (Extended Data Fig. 3h); and at angles of incidence of up to 67.5° (with respect to the vertical), mean VDE changed by approximately −40% (Extended Data Fig. 3i). These results indicate that a jetting device does not need to be in direct contact with gastrointestinal tissue surface to produce the desired outcome. This result points to a potential advantage of jets over solid or hypodermic needles in the gastrointestinal tract, where intimate contact with the mucosa cannot always be guaranteed.
Extended Data Fig. 3. Ex vivo tissue testing, procedure for segmenting scanned volumes and ex vivo characterisation.
a, First, the tissue is placed on a block of foam in a petri dish filled with buffer solution. Next, the hand-held jetting device is positioned above the sample with an adjustable linkage, and the device is triggered. In between trials, the jetting device is removed so that its ampule can be refilled with the payload fluid (a mixture of a iodine-based contrast agent and green tissue-staining dye). After injection, the tissue is transported to an imaging facility where it is scanned with a micro-CT machine. After scanning, the tissue is stored in formalin and sent to the histology facility while the 3D data from each micro-CT scan are then segmented (see f). b, Bulk sections of rectal tissue approximately 15 cm in length. c, The hand-held jetting device clamped to the linkage and positioned above a tissue sample. d, Close-up of the ampule positioned above a sample with the standoff collar fixture attached. e, Rectal tissue samples in histology storage cassettes after injection. f, During the procedure for segmenting scanned volumes, firstly, the micro-CT scan is reconstructed so that 2D slices can be extracted. Then, a starting plane is selected and 2D polygons are drawn to create segmented regions of interest (ROIs). Each 2D slice is then checked and, if necessary, the original ROIs are adjusted to maintain the desired segmentation. Finally, 2D ROIs on all slices are connected to create 3D ROIs so that all relevant voxels can be segmented into luminal, submucosal and intraperitoneal categories. g, Linear regression of volumetric diffusion of contrast agent in tissue after injection. Individual data points are shown as black dots (n = 4 tissue sections from the same animal for each 10 and 20 min delay). The solid line represents the regression line with the intercept set at 0, 0 and the shaded region represents the 95% confidence interval for the slope. h, Impact of standoff on VDE. The experimental replicates are referenced with respect to the mean of the control replicates at 0 mm standoff (n = 6 tissue samples for 0 and 5 mm standoff and n = 8 tissue samples for 2.5 mm standoff, all from the same animal). i, Impact of angle of incidence on VDE. The experimental replicates are referenced with respect to the mean of the control replicates at 0 degrees (n = 4 tissue samples per angle of incidence, all from the same animal). In (g-i), all replicates were taken at dj = 257 µm and pin′ = 11.6 ± 0.7 Bar. In (h-i), individual replicates are shown as black dots and means as horisontal lines.
Device implementation
We used results from the above experiments to guide the design of the four high-level system concepts previously described. We utilized two radial MiDe systems to target the small intestines: an autonomous small intestine MiDeRadAuto system (Figs. 1h and 4f) and a modified tethered version for endoscopic delivery (MiDeRadEndo; Extended Data Fig. 6b). We also implemented two axial systems: the endoscopic version, MiDeAxEndo (Fig. 1h), and the autonomous self-standing version targeting the stomach, MiDeAxAuto (Figs. 1i and 4i). The self-standing systems, MiDeRadAuto and MiDeAxAuto, are capable of supporting 200 µl and 80 µl payloads, respectively (Extended Data Figs. 6 and 7 and Methods).
Fig. 4. In vivo testing with proof-of-concept jetting devices.
a,b, Plasma concentration of insulin analogue after jet delivery (0.21 mg in 200 µl) at different jet output pressures to pig small intestine (a) and dog stomach antrum (b) using MiDeAxEndo (under anaesthesia with recovery directly after). a, n = 3 pigs for 3.5 and 5.0 bar and n = 7 pigs for 9.4 bar. b, n = 4 dogs for 11.3 bar and n = 7 dogs for 24.5 bar. c, Plasma concentration of long-acting inactive GLP1 analogue after jet delivery to pig small intestine using MiDeRadEndo (at 14 ± 1.5 bar) (n = 6 pigs) or oral dosing of tablets with a chemical enhancer formulation to dog stomach (n = 8 dogs). Data are normalized by dose and body weight. d, Serum concentration of siRNA (20.4 mg in 120 µl) delivered to pig small intestine using MiDeAxEndo at 9.5 bar (n = 7 pigs) and subcutaneous control (SC; n = 4 pigs). e, Insulin bioavailability for each MiDe device. n represents number of animals. c.v., coefficient of variation. f, Section diagram of the autonomous intestinal device, MiDeRadAuto. g, Plasma concentration of insulin analogue from in vivo deployments (0.21 mg in 200 µl) with MiDeRadAuto (inserted into pig jejunum under anaesthesia, and activated and injected in fully awake animals 60 to 90 min post insertion). Results shown for device deployments (n = 7 pigs), subcutaneous controls (n = 6 pigs) and negative controls (NC; an intraluminal injection to the small intestine; n = 8 pigs). h, X-ray images from in vivo deployment of MiDeRadAuto in pigs. i, Section diagram of the autonomous gastric device, MiDeAxAuto. j, Plasma concentration of human insulin from in vivo deployments (0.25 U kg−1 in 80 µl) with MiDeAxAuto in pigs. Results shown for device deployments, subcutaneous controls, intragastric controls (IG) and negative controls (n = 3 pigs per group). k, Endoscopic image from in vivo deployment of MiDeAxAuto in pigs. Data in a–d,g,j are mean ± s.d.
Extended Data Fig. 6. Section diagrams of axial and radial endoscopic MiDes together with assembly steps of the radial autonomous MiDe.
a, System description of the axial endoscopic MiDe (MiDeAxEndo) including a section diagram. b, Section diagram of the radial endoscopic MiDe (MiDeRadEndo). Proximity bag not shown. c, Exploded view of MiDeRadAuto with parts labeled (pre-assembly). d, Trigger fabrication steps in which the polymer pellet is made by injection molding PVA. e, The filling steps in which the payload fluid is added to the device’s ampule. f, The load setting steps in which the spring is compressed and fixed in place. g, The final assembly step in which the ampule and spring sub-assemblies are joined. h, Views of MiDeRadAuto after the device has been filled (post-assembly).
Extended Data Fig. 7. Assembly of axial autonomous device for gastric delivery (MiDeAxAuto).
a, Exploded view of MiDeAxAuto with parts labeled (pre-assembly). b, Trigger fabrication steps in which the sugar plug is potted in the setscrew. c, The filling steps in which the payload fluid is added to the device. d, The pressurisation steps in which dry ice is added to the device and the top is joined to the bottom. e, Views of MiDeAxAuto after the device has been filled and pressurised (post-assembly).
The radial devices (MiDeRadEndo and MiDeRadAuto) use the same nozzle dimensions, with the MiDeRadAuto using a dissolvable ‘pellet’ coupled to support triggering of the system (Extended Data Fig. 6c–h) and using a spring to pressurize the payload. Furthermore, the MiDeRadAuto device is distinctively designed with two nozzles situated diametrically opposite to each other. This configuration effectively counteracts the recoil ordinarily associated with a single jet stream and thereby provides stability during the injection phase (see images showing minimal recoil in Fig. 1g). By contrast, the MiDeAxEndo maintains stability during therapeutic delivery by mechanical fixation within the endoscope and its payload is pressurized by activation of a valve connected to an external pressure source (Extended Data Fig. 6a). The payload of MiDeAxAuto is pre-pressurized to approximately 60 bar (temperature dependent) by an onboard CO2 chamber separated from the payload by a piston, which can deploy into the tissue following self-orientation as previously described4. Its triggering mechanism is composed of a dissolvable ‘sugar plug’ and burst membrane in front of the jetting orifice. When the sugar plug dissolves, the pre-pressurized fluid ruptures the membrane and can escape and form a jet (as shown in Extended Data Fig. 9a) which penetrates the tissue beneath the device. As a result of the exchange of momentum, the device recoils in the opposite direction of the jet at velocities of up to an estimated 4 m s−1 (see image of recoil in Fig. 1f and high-speed imaging in Supplementary Videos 2 and 3). Although all of the payload fluid is expelled from the device before it recoils more than 1 cm, the recoil could have an adverse effect on VDE and should be addressed in future device iterations. Additional details about the operation, design, assembly, triggering, force profile and CO2 chamber of MiDeAxAuto can be found in Extended Data Figs. 7 and 8c–f and Methods.
Extended Data Fig. 9. Design and testing of gastric device.
a, Our sugar-plug and burst-membrane based triggering mechanism. The steps involved in triggering are as follows: (I) The device is placed in a solvent, (II) The solvent begins to dissolve, (III) As the dissolution nears completion, the burst-membrane becomes unsupported and begins to deflect, (IV) When the sugar is completely dissolved, the burst membrane ruptures, releasing the jet. b, Setup for imaging the triggering event in which the device is placed on a film with a hole over a beaker, then triggered with a droplet of water. Further details are provided in Supplementary Methods 9. c, Qualitative results from the triggering tests showing proportion of successes to the two observed failure modes. d, Mass loss vs. time for five different MiDeAxAuto devices. e, Gas leakage rate for five different MiDeAxAuto devices. Data are mean±95% confidence intervals as calculated with linear regression and intercept set at 0, 0. Further details are provided in Supplementary Methods 10.
Extended Data Fig. 8. Testing of proof-of-concept jetting devices with force transducer.
a, Test setup for MiDeRadAuto-X in which the device is fixed above the force transducer and triggered by manual removal of a steel rod. b, Resulting force profiles from a single MiDeRadAuto (n = 4 technical replicates). c, Test setup for MiDeAxAuto in which the device is placed on a film over the force transducer and triggered with a droplet of water. d-f, Resulting force profiles from three different MiDeAxAutos (n = 2, n = 2 and n = 3 technical replicates, respectively). Only MiDeAxAuto-A3 and MiDeAxAuto-A4 were used in in vivo experiments. In (b) and (d-f), dashed horisontal lines are estimates of thresholds for VDE performance based on linear interpolation of previous ex vivo data.
In vivo studies
We used each of our MiDe systems to administer insulin to large animal models. In the first set of experiments the axial endoscopic device, MiDeAxEndo, was utilized to investigate the pressure dependency on the pharmacological performance. Pressure ranges were chosen according to ex vivo findings. A fixed dose of 30 nmol of an insulin analogue was used in all studies. Injection to pig small intestine at 3.5, 5 and 9.4 bar resulted in bioavailabilities (versus subcutaneous injection) of 1% (s.d. 1), 38% (s.d. 18) and 69% (s.d. 22), respectively (Fig. 4a,e). In dog stomach, injections at 11.3 and 24.5 bar with a nozzle diameter of 257 µm resulted in bioavailabilities of 26% (s.d. 37) and 90% (s.d. 24), respectively (Fig. 4b,e). Injections to the small intestine and the stomach revealed a positive correlation between pressure increase and systemic exposure (bioavailability). These in vivo studies refine our understanding of pressure requirements for high VDE, complementing our ex vivo tissue experiments. The integration of heat map data with pressure scaling studies provides a more accurate representation of physiological conditions, underscoring the value of validating findings across different experimental models.
In addition to insulin, a fixed dose of 68 nmol of a long-acting inactive GLP1 analogue was delivered to pig small intestine using the endoscopic MiDeRadEndo, which resulted in bioavailability of 67% (s.d. 43). MiDeRadEndo dosings were compared to the oral formulation of the long-acting inactive GLP1 analogue, which achieved modest oral bioavailability15 of approximately 1% (Fig. 4c). In our exploration of larger molecule absorption, MiDeAxEndo was used to administer a 21-kDa siRNA, resulting in bioavailability of 82% (s.d. 37) (Fig. 4d).
On the basis of the pressure escalation study using the endoscopic MiDe systems, our autonomous devices were adjusted to deliver at pressures with high exposure values. For MiDeRadAuto, a fixed dose of 30 nmol of an insulin analogue was used in all studies. Intestinal devices were placed directly into the jejunum with an endoscope. Triggering was confirmed by inspection of the spring with X-ray imaging (Fig. 4h). In the case of MiDeAxAuto, a weight-based dose of 0.25 U kg−1 was used in all studies. An image of MiDeAxAuto in the stomach is shown in Fig. 4k. The in vivo triggering event is shown in Supplementary Video 4.
Upon triggering, blood samples were taken at regular intervals for up to 8 h. In total, seven injections with MiDeRadAuto and three injections with MiDeAxAuto were successfully executed. Pharmacokinetic insulin exposure profiles for the two devices are shown in Fig. 4g,j. For MiDeAxAuto, the pharmacokinetic profiles of three intragastric control trials, in which the mucosa of the gastric antrum was injected with the same dose via Carr–Locke needle, and three negative control trials, in which devices were deployed with the same dose, but drastically reduced pressurization (<5 bar), were also plotted. The bioavailabilities in the MiDeRadAuto and MiDeAxAuto studies were 31% (s.d. 21) and 23% (s.d. 4), respectively (Fig. 4e). To evaluate the safety profile of device transit throughout the gastrointestinal tract, devices were administered to Yorkshire pigs and safe passage was documented for all devices (Extended Data Fig. 10b–f).
Extended Data Fig. 10. Passage, inflammation and gastric transit.
a, Endoscopic image of three MiDeAxAuto dummies in the stomach on day zero of the study. b-f, X-ray images of the right flank of the pig showing the progress of device passage from day 0 to day 25. Further details are provided in Supplementary Methods 12. g, Sections from the in vivo inflammation study employing gastric devices. No major reactions were observed (though one minor acute inflammatory focus was located in the submucosa of Set A, Slice 1). Further details are provided in Supplementary Methods 13. h-i, Jet penetration wounds from ex vivo studies (for comparison). j, Example X-ray images (dog 2) following co-administration of scaled-MiDeRadAuto devices (to a 00-el capsule size) and apple juice (pH 3.3) and a contrast agent in a Beagle dog model. Apple juice was used to ensure a human-like acidic pH environment in the stomach, and an enteric capsule was placed over the activation module of the device. X-ray imaging was performed at 15 min intervals to assess the device location and activation time. k, All six devices transited the stomach within 2 h and activated between 60 and 75 min. Four of six devices activated in the small intestine (as desired), and four of six devices were passed by the dog in 24 h. Further details are provided in Supplementary Methods 11.
Building on the pharmacokinetic studies of MiDeRadAuto, a proof-of-concept study to validate the successful targeting of the small intestine and subsequent exit from the gastrointestinal tract was performed, incorporating a pH-sensitive capsule around the activation pellet. In a peroral dosing using a scaled version of MiDeRadAuto, each device transited the gastrointestinal tract without any adverse events, and in four out of the six animals, activation at the intended target location within the proximal small intestine was detected. Verification with C-arm fluoroscopy and a contrast agent accurately traced the passage and activation of the devices along the gastrointestinal tract (Extended Data Fig. 10j,k). These results not only reinforce the safety and precision of MiDeRadAuto, but also demonstrate the effectiveness of our pH-sensitive capsule in ensuring precise targeting, further solidifying the promising therapeutic application of the device concept.
Discussion
Here we present the development and characterization of a family of MiDe systems for axial and radial delivery of macromolecules in the gastrointestinal tract by needle-free means. These devices achieved systemic double-digit bioavailability of macromolecule drugs by jetting, representing an order of magnitude improvement over currently prescribed oral biologic dosing methods15, on par with other ingestible robotic devices4–7. Future development will involve optimization of the jetting expulsion event through adjustment of ampule pressure, nozzle dimensions and ejection volume. Additionally, further optimization of the localization mechanisms, particularly in relation to the effect of concomitant food intake, is necessary for the development of MiDe systems.
The optimized MiDeAxEndo (9.4 bar) and MiDeAxEndo (24.5 bar) injection conditions for an insulin analogue had bioavailability c.v. values of 31% and 27%, respectively, which are comparable to both the variation seen in our ex vivo VDE assessments (Fig. 3b,c) and the c.v. for traditional needle-based subcutaneous pen injection in humans for basal insulin, reported to be 28% for NPH (neutral protamine hagedorn) insulin and 33% for insulin glargine16. Given that subcutaneous pen injectors operate under tight control and our endoscopic jet delivery devices are optimized, biological variability emerges as the key driver of variation, minimizing the effect of relative tissue differences. It is also worth noting that there is recognized variability in absorption in the gastrointestinal tract in clinical settings, owing to factors such as patient size, age, health and food intake17. Thus, the oral route may be initially best suited for therapeutics with a broad therapeutic window and long half-life (such as semaglutide). As our understanding of effective human translation expands, this will inform future ideal active pharmaceutical ingredients that could be included in the family of devices described here.
Bioavailbility c.v. values of 68% and 16% were observed for autonomous delivery in the MiDeRadAuto and MiDeAxAuto studies, respectively. As shown in the ex vivo evaluation (Extended Data Fig. 3h,i), the VDE decreases by up to 40% when the jet–tissue angle is 45°. The self-orientating feature of the MiDeAxAuto device autonomously aligned the device on the stomach floor and tests were conducted in anaesthetized animals, which is likely to explain the low variability. By contrast, the MiDeRadAuto device migrates in an awake animal for an hour before injection, and the jet position and orientation to the tissue are not controlled beyond maintaining co-axial position in the intestine (owing to the aspect ratio of the device), resulting in an increased variability.
In light of the presented investigations, our study supports effective absorption of large molecules (insulin, GLP1 analogue and siRNA) using jet injection technology, with comparable pharmacokinetics to standard subcutaneous injection. Furthermore, a study involving the same injection location as used here for MiDeAxAuto has demonstrated good absorption of monoclonal antibodies, providing a strong indication for effective absorption of larger proteins using our jet injection method7. Future work could expand on this to investigate a molecular size-to-absorption dependency for effective delivery via this method. The introduction of approaches that enable high systemic bioavailability have the potential to enable the oral delivery of therapies that are currently largely limited to parenteral routes and reduce the cost of goods by decreasing the total mass of active pharmaceutical ingredients required, as the MiDe platform essentially approximates parenteral dosing, in contrast to enhancer-mediated absorption15,18.
Further development will involve additional miniaturization to minimize the risk of obstruction. Of our devices, only MiDeAxAuto currently falls within the footprint of ingestible devices previously approved by the US Food and Drug Administration for regular dosing19. Decreasing payload volume (and if necessary, increasing the active pharmaceutical ingredient concentration) is the most direct way to reduce the overall footprint. Doing so would not only save space, but could also reduce the spring force and stroke length requirements, enabling the use of smaller springs and thinner walls. Furthermore, if a single dose was divided over multiple, smaller devices, the failure of a single device to achieve high VDE would have less consequence on the bioavailability of the overall dose. Further optimization is essential to ensure reliable activation timing. The simplicity and flexibility of ingestible jetting devices allow for cost-effective improvements, and compatibility with high-throughput manufacturing and expanding biodegradable materials support scalability and environmental considerations. Key factors such as patient anatomy (for example, Roux-en-Y gastric bypass and strictures), co-morbidities (such as achlohydria) and feeding status must be considered when deploying these devices. Stability during storage and protection from premature exposure to moisture are also critical for commercial viability. Future development will include safety testing to ensure readiness for human clinical trials.
In conclusion, the penetration of mucosal tissue in the gastrointestinal tract using jets has shown promise for the oral and endoscopic delivery of biological agents. Consistent delivery of liquid payloads across multiple gastrointestinal tissue types was achieved using proof-of-concept prototype devices, which demonstrated bioavailability of greater than 10% for insulin, GLP1 analogue and siRNA. Beyond the optimizations addressed above, future work should include broader in vivo testing with additional therapeutic agents, along with ex vivo testing using porcine and human organs to better understand biological variability.
Methods
Jet physics and theoretical model
To study the influence of jetting parameters (ampule pressure, jet diameter and jet velocity) on tissue deposition, a theoretical model was derived (Extended Data Fig. 1a and Supplementary Methods 1). The jetting apparatus used to investigate jet delivery into ex vivo tissue is described and characterized in Extended Data Fig. 1b–g. The main components of this apparatus were a handheld jetting device for generating jets, and a piezoelectric force transducer (9215 A with 5165A4KH10 LabAmp amplifier; Kistler Instrument) and software (Network Setup Wizard; Kistler Instrument) for measuring jetting impingement force (at 200 kHz sampling frequency). Based on previous literature and theory, variation of nozzle diameter and ampule pressure has the highest impact on penetration in soft tissue20–22. A subset of 4 exemplary jetting force profiles with a 257 µm nozzle is shown in Extended Data Fig. 1c. From these force profiles the experimental period of the jetting event, T (Extended Data Fig. 1d), the experimental steady-state jetting force, Fj,exp (Extended Data Fig. 1e), and the experimental steady-state jetting power, Pj (Extended Data Fig. 1f) were extracted. An expanded set of results, including all nozzle–pressure permutations, is shown in Extended Data Fig. 2a–g and the procedure is described in Supplementary Methods 2 and 3.
To examine the effect of fluid viscosity on jet performance the ampule pressure and diameter were fixed and viscosity was measured23–25 between 1 and 219 cPa. The system pressure factor dropped significantly—from a mean of 0.82 to a mean of 0.40 (Extended Data Fig. 2h), while no significant changes were observed for the CT contrast fluid (Extended Data Fig. 2i,j).
Porcine tissue excision and processing methods
Female Yorkshire pigs, 3–5 months old and weighing 35–70 kg were used. After euthanasia (procedure described below), a midline incision allowed access to the abdominal cavity. Stainless steel pean forceps were placed at the oral and aboral part of the organ of interest. The organ was then separated from the omentum and removed from the carcass with a scalpel. Precautions were taken to ensure that the tunica serosa and other important tissue layers were kept intact. After excision, the tissue was placed in a plastic bag kept on ice and shipped to the laboratory where ex vivo injections were performed on the same day.
Bulk sections of tissue ranging from 10 to 35 cm in length (depending on the organ) were extracted from the organs with Mayo scissors and placed into beakers filled with phosphate buffer solution (PBS), pH 7.4. For both the cheek and oesophagus, tissue from the entire organ was used. For the stomach, only tissue from the antrum/lower corpus was used. For the small intestine, only jejunal tissue at least 20 cm from the pylorus was used. For the colon and rectum, distal sections were used. An image of a bulk section of rectal tissue is shown in Extended Data Fig. 3b.
Smaller sections of tissue (approximately 4 × 4 cm, the final sample size) were extracted from the bulk with scissors. These samples were further cleaned in a separate wash beaker with PBS to remove any remaining chyme or debris. Finally, immediately before injection, the tissue samples were placed on a piece of foam in a 6.5 × 4.7 cm plastic dish. In total, the time between euthanasia of the animal and injection experiments was 2–3 h.
Ex vivo jet injection study with porcine tissue
Prior to each set of ex vivo experiments, the polycarbonate ampules were lubricated to improve sliding of the plunger. To accomplish this, the ampules were dipped in a mixture of 5% hexamethyldisiloxane, 95% deionized water solution, then left to dry overnight. Before each injection, the ampule was filled with 232 µl of an aqueous mixture containing 30% micro-CT contrast agent (Iomeron 350 mg ml−1; Bracco), 1% green tissue dye (WAK-HM-G-1/60, WAK-Chemie Medical) and 69% deionized water. The ampule was then mounted onto the handheld jetting device and the input pressure of the device was adjusted to the desired level with a digital pressure regulator. An example image of the device positioned for injection is depicted in Extended Data Fig. 3c,d. Finally, the triggering sleeve was actuated, causing the jetting injection event. Between two and eight replicates were performed for each pair of pressure-diameter inputs, with certain sets of shots performed on the same tissue sample (space permitting). All experimental points can be found in Supplementary Table 3.
Computed tomography scanning (Phoenix Nanotom M micro-CT; GE Inspection Technologies) was used to evaluate each sample. The stage and detector were positioned at travel distances of 130 mm and 300 mm from the X-ray source, respectively. Background detector calibrations were performed before each scan series. Scans were performed at a tube voltage of 100 kV, radiation intensity of 100 μA and a target scan time of 6 min.
All scans were performed 10–15 min after injection. After each scan, the tissue samples were placed in plastic histology cartridges (Extended Data Fig. 3e) and stored in 10% formalin solution. Although it was not possible to maintain the original structure of the depot with this storage method due to diffusion and dissolution, the use of green tissue dye in the payload fluid dye enabled us to later identify the tissue layer(s) in which the depot resided. The entire injection and scanning procedure is depicted in Extended Data Fig. 3a.
Volumetric diffusion, standoff distance and jetting angle ex vivo studies
Using the methods described above, we also performed a set of studies to determine the impact of volumetric diffusion within tissue, standoff between nozzle and tissue, and angle of incidence between the jet and tissue on delivery characteristics.
To evaluate the effect of diffusion on the apparent volume of fluid in tissue after injection we performed a set of calibration experiments and found that the apparent volume of fluid in tissue increased at a rate of 0.5 ± 0.1% per min (95% confidence interval). Results from this diffusion study are shown in Extended Data Fig. 3g. GraphPad Prism (GraphPad Software) was used to calculate the linear rate of change of volume with 95% confidence (the intercept was set at 0, 0). Furthermore, the upper error boundary from this analysis was used to make conservative corrections to VDE estimates in subsequent analyses. Photos and diagrams showing the injection, scanning and segmentation processes are shown in Extended Data Fig. 3b–f. Additional scans from jejunal and stomach tissue are shown in Extended Data Fig. 4b–e. Next, each set of injections was semantically categorized as luminal, submucosal or intraperitoneal based on where most of the micro-CT scanned fluid resided on average.
The results of the nozzle-to-tissue standoff and angle study are shown in Extended Data Fig. 3h,i. For the angle study, a constant standoff of 5 mm was chosen.
Histological imaging of samples from ex vivo injection studies
Tissue samples were fixed in 10% formalin solution for a minimum of 24 h. They were then processed in an ASP300S fully enclosed tissue processor (Leica Biosystems), embedded in paraffin wax and cut on a microtome in 4 µm thick sections. From there, sections were mounted on glass slides and stained with haematoxylin and eosin. Finally, the slides were scanned on a NanoZoomer S60 Digital Slide scanner (Hamamatsu Photonics) at 40×. Results from this process can be seen in Fig. 2b and Extended Data Fig. 5.
Processing of volumetric data from ex vivo injection studies
Acquisition, processing and reconstruction of images were performed with Phoenix Datos|x (GE Measurement & Control). Examples of processed scans are shown in Fig. 3a and Extended Data Fig. 4b–e. Once the scanning and reconstruction was complete, a segmentation of regions of the contrast fluid that were on top of, beneath and contained inside the tissue was performed. A simplified representation of the segmentation process is shown in Extended Data Fig. 3f. A standard Student’s t-distribution was used to determine 95% confidence intervals for the submucosal volume and VDE. Statistical analysis was applied only after all other quantitative analyses—including segmentation and diffusion correction—were complete. Further details on the processing of volumetric data are provided in Supplementary Methods 4.
Axial endoscopic device prototype MiDeAxEndo
The MiDeAxEndo prototype can deploy a therapeutic dose via the working channel of an endoscope. A schematic of all its components is shown in Extended Data Fig. 6a. The MiDeAxEndo system consists of a nitrogen pressure tank, a pressure controller, an air-tight 18 ml polycarbonate drug reservoir with an internal plunger separating pressurized gas from the drug product, a high-speed valve operated by a microcontroller and 2.8 m of polyether-ether-ketone (PEEK) tubing capped with a computer numerical control (CNC) machined PEEK adapter and stainless steel nozzle. Polytetrafluoroethylene (PTFE) sealing tape was used at all junctions to ensure air-tight connections. The stainless steel nozzle was assessed to have a 254 µm diameter with scanning electron microscopy. Jetting is conducted by loading the reservoir and tubing with the drug product, applying the desired pressure, and then opening the high-speed valve to initiate jetting. Operating valve times were characterized for each jetting pressure such that 200 µl was ejected. In Fig. 4a,b, valve times were 120 ms, 97 ms, 77 ms, 75 ms and 50 ms for the 3.5 bar, 5.0 bar, 9.4 bar, 11.3 bar and 24.5 bar jetting pressures, respectively.
Radial endoscopic device prototype MiDeRadEndo
In this study, a spring-loaded drug delivery device with a radially oriented jetting nozzle was utilized (Extended Data Fig. 6b). Activation of the device was achieved through a pneumatic tube, which also served as a tether for holding the device in place. The pneumatic tube was threaded through the endoscopic working channel for simultaneous delivery and positioning. The device, capable of ejecting a drug volume of 188 µl, was designed with an inflatable bag, featuring a diameter of 32 mm, to ensure optimal device-tissue proximity. Further details on the operation, design and assembly of MiDeRadEndo are provided in Supplementary Methods 5.
Autonomous intestinal device prototype MiDeRadAuto
The devices were used with the minor assistance from an endoscope. It is triggered via a dissolvable ‘polymer pellet’ that holds a detent pin captive. When the polymer pellet dissolves, the detent pin is released, allowing the spring to pressurize the ampule. The device is placed directly into the duodenum with an endoscope and allowed to trigger and pass without further assistance.
The nominal ampule volume and diameter for MiDeRadAuto are 200 µl and 6 mm, respectively. The stainless steel spring used in the device has an initial force of 70 N, resulting in corrected ampule pressures of 14 ± 1.5 bar (n = 48). The nozzles are oriented radially, and its diameter is 240 ± 10 µm (95% confidence, n = 8). The overall footprint of the device is depicted in Extended Data Fig. 6c–h. The device is made of machined polyoxymethylene (POM), PEEK and common metals. The piston seals are made from NBR-70. A photo of this device is shown in Fig. 1h and a section view is shown in Fig. 4f. Diagrams of MiDeRadAuto before and after assembly are shown in Extended Data Fig. 6c–h, and each of the assembly steps are described in Supplementary Methods 6.
Autonomous gastric device prototype MiDeAxAuto
The autonomous axial prototype (MiDeAxAuto) can deploy a therapeutic dose to the stomach via direct oral ingestion. The device relies on a sugar plug-based triggering mechanism which passively degrades in the stomach. The sugar plug is located directly in front of the nozzle orifice, so the payload fluid—which is constantly pressurized—cannot escape from the chamber. To prevent the payload fluid from degrading the sugar plug, the plug is separated from the orifice exit by a thin polymeric burst membrane. Thus, when the sugar plug dissolves enough so that it can no longer support the pressure exerted by the payload fluid, the burst membrane ruptures and the payload exits the device as a columnar jet. A diagram of the triggering mechanism is shown in Extended Data Fig. 9a.
As the stomach is a cavernous organ, we needed to implement an axial localization mechanism to align the jet. To achieve this, we drew inspiration from the methods used by Abramson et al.4 in their self-orientating system. MiDeAxAuto is 10.8 mm in diameter and 11.8 mm in height, with a centre of mass 3.5 mm above the bottom face. The diameter of the nozzle orifice is 298 ± 10 µm (95% confidence, n = 3). The nominal ampule volume and diameter are 80 µl and 7.9 mm, respectively. These ampule dimensions, combined with the pressure requirements for delivery in the stomach, mandated use of a relatively strong spring. We found that any sufficiently strong coil or disk spring’s mass interfered with the device’s self-orientation properties, so we decided to use compressed CO2 at its saturation pressure of 60 bar instead. Assuming a pressure factor of 70%, we deemed the aforementioned nozzle diameter to be most appropriate per our heat map results in Fig. 3c.
In order to both maintain self-orientation and safely contain the gas pressure, we chose to machine the bottom piece of the MiDeAxAuto from brass and its top piece from 7075 aluminium. The piston is made from POM, the seals from silicone rubber and the burst-film from 25 µm thick fluorinated ethylene propylene (FEP). A photo of this device is shown in Fig. 1i and a section view is shown in Fig. 4i. Diagrams of MiDeAxAuto before and after assembly are shown in Extended Data Fig. 7, and each of the assembly steps are described in Supplementary Methods 7.
Force profile studies with MiDeRadAuto and MiDeAxAuto
The aim of the force profile studies was to determine whether the devices we had fabricated produced sufficiently strong jets to deliver therapeutics. A Kistler 9215 A piezoelectric force transducer was again used to measure jetting force. Because MiDeAxEndo and MiDeRadAuto have identical ampules, springs and nozzles, we decided that it was only necessary to test the latter of the two devices. Diagrams of setups for force profile testing of MiDeRadAuto and MiDeAxAuto devices and their associated results are shown in Extended Data Fig. 8. Further details are provided in Supplementary Methods 8.
Formulation of therapeutic payloads
For the MiDeAxEndo and MiDeRadAuto studies—including controls—we used an insulin payload solution (pH 7.4) with the following concentrations: 244.2 µM insulin analogue (Novo Nordisk), 8.05 mM sodium phosphate dibasic, 1.96 mM potassium dihydrogen phosphate and 140 mM sodium chloride. This solution (200 µl) was added to the device, resulting in an insulin dose of 0.28 mg (8 U). For the siRNA, a payload solution with a concentration of 170 mg ml−1 in 10 mM phosphate buffer at pH 7.4 was used. The solutions were stored at 4 °C until they were used.
For the MiDeAxAuto studies—including controls—we used an insulin payload solution (7 < pH < 8) which was tailored based on the animal’s weight. To make this solution, between 10 and 20 mg of human insulin powder (Novo Nordisk) was weighed, and the exact mass noted. The insulin was then added into a 2 ml vial, followed (successively) by the following excipients: 600 µl 0.1 M sodium hydroxide, 0.5 mg PF68 (Sigma-Aldrich), 12.6 mg HEPES (Sigma-Aldrich), 300 µl 0.1 M hydrochloric acid and 100 µl deionized water. This solution was then diluted with deionized water in a separate vial based on the weight of the animal (35–80 kg) and payload volume of the device (80 µl) to achieve a dose of 0.25 U kg−1.
In vivo pharmacokinetic exposure studies
All in vivo pharmacokinetic studies were performed either at MIT or Novo Nordisk’s animal facilities by trained veterinary technicians and complied with relevant ethical regulations on animal research. Our procedures were reviewed and approved by review boards at each respective site (Committee on Animal Care at MIT and the Animal Experiment Inspectorate, Ministry of Justice, Denmark). For MiDeAxAuto, MiDeRadAuto and MiDeAxEndo (exclusively small intestine delivery) studies, female LYD (crossbred Landrace, Yorkshire and Duroc) pigs (body weight 50–70 kg; Novo Nordisk) and female Yorkshire pigs (body weight 35–80 kg; Tufts University, USA) were used, and for the gastric MiDeAxEndo study, female Beagle dogs, from 10 months old and weighing 8–13 kg were used. Pigs were placed on a liquid diet up to two days before each study and fasted overnight with the aim of reducing the amount of chyme and food debris in the gastrointestinal tract.
On the day of the study, anaesthesia was induced either with propofol intravenously (5 ml and supplemented as needed) or with a mixture of Telazol (tiletamine/zolazepam; 4–6 mg kg−1) and xylazine (2–4 mg kg−1) intramuscularly. Immediately after sedation, animals were transferred to an operating room where they were intubated and immediately provided with isoflurane (1.5–3% mixture with oxygen). The isoflurane was used throughout the entire duration of the study to maintain anaesthetization. During the study, vital signs were continuously monitored and noted at least every 15 min. Vital signs included breathing rate, end tidal CO2, oxygen saturation (SpO2) level in blood and pulse rate. The maximum period of anaesthetization was 4 h for non-terminal procedures and 8 h for terminal procedures. Whether a study was terminal was decided in advance and depended on external factors such as the age and weight of the animal.
During studies in which a device was deployed, a 100 cm over-tube (McMaster-Carr Tygon PVC tubing, 5/8 inch internal diameter, 13/16 inch outer diameter) was inserted into the oesophagus. This over-tube made it easier to transfer devices to and from the target location. The over-tube was removed immediately after device deployment was completed. For subcutaneous control studies, we used a 1 ml syringe with a hypodermic needle to administer the same dose as with the jet device to the subcutis on the animal’s belly or neck. For intraluminal or intragastric control studies, we used a Carr–Locke needle and endoscope to administer the same dose to the target organ.
Blood sampling was performed via a central line placed either in the ear (for non-terminal studies) or the femoral vein (for terminal studies). Though sampling frequency and duration varied depending on the test site and type of study, in general, samples were extracted at least every 15 min for the first 2 h and then at least every hour for up to 8 h. In the case of non-terminal studies, blood samples were drawn from the ear-catheter after the animal was recovered. Each blood sample was extracted from the catheter with a 3 ml syringe, then stored on ice in tubes pre-coated with EDTA. After collection, samples were centrifuged for 10 min at 1,500g. From there, 500–600 µl of plasma from each sample was extracted with pipettes and deposited into 750 µl Micronic tubes (Micronic). Plasma from each time point was stored in up to three separate aliquots at −80 °C until bioanalysis was performed.
In the case of terminal studies, pigs were euthanised with 80–100 mg kg−1 pentobarbital sodium intravenously or via an intra-cardiac injection. In all cases—before euthanasia—full anaesthetization was verified by the absence of pain-responsive reflexes (for example through a limb withdrawal test).
For the inactive GLP1 analogue tablet control study, 8 healthy male Beagle dogs (2.8–3.5 years old, 9.9–14.5 kg) were fasted overnight for ≥18 h, before receiving a single tablet orally the next morning along with 10 ml of tap water. Tablets contained 2.65 mg active pharmaceutical ingredient, 101 mg sodium N-[8-(2-hydroxybenzoyl) aminocaprylate] (SNAC), 66.7 mg nicotinamide, and 0.8 mg magnesium stearate. To reduce inter-individual variance among dogs, all dogs received a subcutaneous glucagon injection (3.2 nmol kg−1) 10 min prior to the tablet administration. Approximately 0.8 ml of whole blood was drawn into EDTA-coated tubes at the time points 5, 10, 15, 20, 30, 45 min, and 1, 1.5, 2, 4, 7, and 10 h, respectively, including one baseline sample immediately before dosing. 100 μl plasma from each sample was transferred into Micronic tubes and subsequently centrifuged at 4,000 rpm for 4 min at 4 °C.
In vivo deployment of MiDeAxEndo, MiDeRadEndo and MiDeRadAuto
All three devices were assembled and filled with the therapeutic payload within 10 min of deployment. The MiDeAxEndo device was then threaded through the endoscope’s channel and inserted into the over-tube. To reach the small intestine, the endoscope was further inserted into the pylorus and advanced 10–20 cm into the small intestines. During in vivo endoscopic operations, the jetting nozzle of the MiDeAxEndo device was positioned orthogonal to the tissue and secured in place by activating the elevator on the endoscope (to prevent MiDeAxEndo from recoil during the jetting event). At this point, the device was triggered and blood sample collection was initiated. After jetting, MiDeAxEndo was held in place for 10 s, then removed to allow for optical observation of the delivery site. The device was then withdrawn from the animal, disassembled, cleaned with isopropyl alcohol or soapy water and stored for future use. Results from MiDeAxEndo studies can be found in Fig. 4a,b for small intestine and stomach depositions, respectively.
The MiDeRadEndo was attached to the front of the endoscope via a rigid pneumatic tube which was fed through the working channel of the endoscope and allowed the operator to extend and retract the device as needed. As with MiDeAxEndo, an over-tube was used to reach the small intestine. To secure the device in place and establish its proximity to the tissue, the attached bag was inflated via a pneumatic tube threaded through the second working channel. A pressure of 25 mbar was applied for inflation. After optical confirmation of successful inflation, the device was activated to deliver the intended drug dosage, followed by deflation of the bag using negative pressure and retraction of the device.
The MiDeRadAuto device was gripped with endoscopic forceps and inserted into the intestine directly. The MiDeRadAuto device was then released from the endoscope, the endoscope retracted from the animal and the animal went through emergence from anaesthesia. Once the animal was fully awake the device actuated autonomously. X-rays were recorded 4 h after dosing to inspect the device’s spring and thereby determine whether it had triggered. All devices (n = 7) successfully activated in the small intestine. The above study was executed successfully seven times with MiDeRadAuto and an insulin analogue payload. Results from the MiDeRadAuto studies can be found in Fig. 4g.
To establish that there is no systemic exposure when peptides are delivered to the lumen of the small intestine (no disruption to the mucosal barrier) a negative control was performed. Eight female LYD pigs weighing 50–70 kg were anaesthetized according to the same anaesthetic protocol listed above. Once under deep and stable anaesthesia, an endoscope was navigated to the proximal small intestine of each animal. When in location, a primed tubing was fed through the working channel of the endoscope and 50 nmol (200 µl of 250 µM solution) of an insulin analogue was delivered to the lumen via a syringe attached to the primed tubing. The tubing was primed with the liquid insulin solution for 30 min before the procedure to mitigate any leeching of insulin into the tubing material during dosing. Thereafter the tubing was flushed with fresh insulin before each dosing. After dosing, the animals were recovered, and blood samples were taken for the next 4 h. The samples confirmed that there was no plasma exposure for each of the eight animals.
In vivo deployment of MiDeAxAuto
MiDeAxAuto devices were filled and pressurized within 10 min of deployment. The device was then dropped into the over-tube and advanced into the stomach with an endoscope. Once inside the stomach, the endoscope was used to monitor the device. An endoscopic image of a MiDeAxAuto in the stomach is shown in Fig. 4k. The device was continuously monitored until triggering occurred, at which point the time was marked and blood sampling was initiated. When triggering occurs, the device visibly jumps (because of recoil). After triggering, the device was retrieved with a Roth Net Standard Retriever (Steris). The device was then disassembled, and actuation of the piston was confirmed through inspection. Finally, the device parts were cleaned with isopropyl alcohol and stored for future use. The above study was executed successfully three times with MiDeAxAuto. Results from the MiDeAxAuto studies can be found in Fig. 4j.
For negative control studies with the MiDeAxAuto, we repeated the original procedure and dosage, except instead of using CO2 for pressurization, we used a weak spring (which generated pressures no more than 0.5 bar). All the above control studies were successfully executed three times.
Bioanalysis of blood samples
Blood samples from animal experiments were analysed for human insulin or insulin analogue in plasma using an AlphaLISA assay (Perkin Elmer), for the GLP1 analogue in plasma using LC-MS and for siRNA in plasma using the Meso Scale Discovery platform. Further details are provided in Supplementary Methods 14 for the human insulin and insulin analogue assay and in Supplementary Methods 15 for the siRNA quantification.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Online content
Any methods, additional references, Nature Portfolio reporting summaries, source data, extended data, supplementary information, acknowledgements, peer review information; details of author contributions and competing interests; and statements of data and code availability are available at 10.1038/s41586-024-08202-5.
Supplementary information
Linear regressions of pressure factor results.
Linear regressions of overshoot results.
Analysis of linear regressions of pressure factor results.
Analysis of linear regressions of overshoot results.
Results from organ comparison study.
Results from heat map study of jejunal tissue.
Results from heat map study of antral tissue.
Dynamics of depot formation in ex vivo jejunal tissue. This high-speed video captures the dynamic process of depot formation within excised porcine jejunum tissue. The jetting apparatus, positioned at a nozzle-to-tissue distance of 5 mm (dj=257 µm, pin=14.5 bar), is thoroughly described and characterized in Extended data fig. 1b-g.
Recoil and jetting dynamics of four MiDeAxAuto devices. This video presents simultaneous recordings of four MiDeAxAuto devices, demonstrating their triggering, jetting, and recoil behaviours. The devices are placed on a plastic film with a hole, through which they jet freely into a glass beaker, showcasing the reproducibility of the recoil effect.
Triggering and injection in ex vivo gastric antrum. This video illustrates the triggering and jetting process of a MiDeAxAuto device into excised porcine gastric antrum, providing insight into its injection dynamics.
In vivo triggering event of MiDeAxAuto in a porcine stomach. This video depicts the in vivo positioning and triggering event of the MiDeAxAuto device within the stomach of an anaesthetized porcine, offering a real-time observation of its functionality.
Acknowledgements
The authors thank R. Langer, A. Abramson, J. Coffey and M. Bochenek for consulting on high-level research scope; K. Cormier for histological imaging; J. Cheah for bioanalysis; T. Hua, L. H. Olesen, T. S. Hansen and D. N. Sørensen for support on fluid and structural mechanics; H. H. F. Refsgaard for pharmacokinetic modelling; L. Hovgaard for formulation support; I. B. Pfander, M. R. Huertas, H. Ljunggreen and B. Jensen for mechanical design guidance, manufacturing and device operation. We acknowledge C. Roepstorff for providing medical writing support funded by Novo Nordisk. J.L. was supported by The Natural Sciences and Engineering Research Council of Canada (NSERC). This study was funded by Novo Nordisk A/S. Additionally, G.T. and his laboratory were supported in part by Karl van Tassel (1925), Career Development Professorship, the Department of Mechanical Engineering, Massachusetts Institute of Technology (MIT) and the Division of Gastroenterology, Brigham and Women’s Hospital, the Advanced Research Projects Agency for Health (ARPA-H) under award number D24AC00040-00. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Advanced Research Projects Agency for Health.
Extended data figures and tables
Author contributions
G.A., D. Sticker, A.G. and G.T. developed the overall concept, shape and devices, and drafted the manuscript with contributions from Y.L., D.G. and S.M. Y.L. planned and conducted ex vivo and in vivo studies including data analysis. H.D.P., J.C., N.E.E., S.T., C.C., K.I., A.H.U., J.K. and A.H. conducted in vivo studies. T.D., D.G., J.W., J.P.H.J., T.S.L. and M. Pereverzina made mechanical designs, conducted ex vivo studies and supported in vivo studies. M. Poulsen, C.A. and E.M.D.A. conducted ex vivo CT scans and data analysis. D. Schultz and A.B. formulated and tested activation pellets. K.H., S.B.G. and I.P. planned and conducted bioanalysis. R.K.K. and C.M.D. conducted histology. S.M. executed in vitro studies including data analysis, in vivo studies and drafted and revised the manuscript. P.K., S.Y., A.A., J.L., N.R., P.H., M.R.F. and U.L.R. contributed with high-level consultation and drafted and revised the manuscript. J.J.W., S.T.B. and G.T. provided oversight and leadership responsibility for the research activity planning, contributed to discussions and drafted and revised the manuscript. All authors discussed the results, reviewed the draft manuscript and approved the final manuscript.
Peer review
Peer review information
Nature thanks Claus-Michael Lehr, Eoin O’Cearbhaill and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer review reports are available.
Data availability
All data that support the findings of this study are available within this paper, its supplementary information and the source data that are provided with this paper. Any additional information is available upon request.
Competing interests
D. Sticker, A.G., T.D., B.M., J.P.H.J., D. Schultz, E.M.D.A., M. Poulsen, C.A., H.D.P., N.E.E., C.C., A.H.U., M. Pereverzina, R.K.K., C.M.D., S.B.G., I.P., A.B., A.A., M.R.F., P.H., U.L.R., J.J.W. and S.T.B. are employees and shareholders of Novo Nordisk. G.T. has received consulting fees and grant funding from Novo Nordisk. B.M., J.P.H.J. and M.R.F. are inventors of the following patent: ‘Jensen, B. et al. Medical Device with Actuation Mechanism. WO2020157324 (6 August 2020)’. G.A., D. Sticker, A.G., Y.L., D.G., B.M., J.W., J.P.H.J., T.S.L., C.C., S.Y., A.B., N.R., J.J.W. and G.T. are inventors of the following patent: ‘Arrick, G. et al. Drug Delivery Device. WO2022034041 (17 February 2022)’. G.A., D. Sticker, A.G., D.G., J.W., J.P.H.J., M. Poulsen, C.C., J.J.W. and G.T. are inventors of the following patent: ‘Traverso, G. et al. Ingestible Drug Delivery Device. WO2022035750 (17 February 2022)’. The other authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: G. Arrick, D. Sticker, A. Ghazal
Contributor Information
S. T. Buckley, Email: spby@novonordisk.com
G. Traverso, Email: cgt20@mit.edu
Extended data
is available for this paper at 10.1038/s41586-024-08202-5.
Supplementary information
The online version contains supplementary material available at 10.1038/s41586-024-08202-5.
References
- 1.Smeets, F. G. M. et al. Endoscopic botulinum toxin for gastroparesis: results of a retrospective series. Gastrointest. Disord.1, 164–175 (2019). [Google Scholar]
- 2.Santucci, N. R. et al. Outcomes of combined pyloric botulinum toxin injection and balloon dilation in dyspepsia with and without delayed gastric emptying. Saudi J. Gastroenterol.28, 268–275 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nelson, D. B. et al. ASGE technology status evaluation report. Injection needles. February 1999. Gastrointest. Endosc.50, 928–931 (1999). [DOI] [PubMed] [Google Scholar]
- 4.Abramson, A. et al. An ingestible self-orienting system for oral delivery of macromolecules. Science363, 611–615 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Abramson, A. et al. A luminal unfolding microneedle injector for oral delivery of macromolecules. Nat. Med.25, 1512–1518 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dhalla, A. K. et al. A robotic pill for oral delivery of biotherapeutics: safety, tolerability, and performance in healthy subjects. Drug Deliv. Transl. Res.12, 294–305 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Abramson, A. et al. Oral delivery of systemic monoclonal antibodies, peptides and small molecules using gastric auto-injectors. Nat. Biotechnol.40, 103–109 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Norn, S., Kruse, P. R. & Kruse, E. On the history of injection. Dan. Medicinhist. Arbog34, 104–113 (2006). [PubMed] [Google Scholar]
- 9.Lawrence, G. The hypodermic syringe. Lancet359, 1074 (2002). [Google Scholar]
- 10.Myers, J. T. et al. Preference for a novel oral alternative to parenterally administered medications. Patient Prefer. Adherence18, 1547–1562 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mitragotri, S. Current status and future prospects of needle-free liquid jet injectors. Nat. Rev. Drug Discov.5, 543–548 (2006). [DOI] [PubMed] [Google Scholar]
- 12.Aran, K. et al. An oral microjet vaccination system elicits antibody production in rabbits. Sci. Transl. Med.9, eaaf6413 (2017). [DOI] [PubMed] [Google Scholar]
- 13.Hikidi, Y. et al. An elaborate behavioural sequence reinforces the decoy effect of ink during predatory attacks on squid. J. Ethol.38, 155–160 (2020). [Google Scholar]
- 14.Schramm-Baxter, J. & Mitragotri, S. Needle-free jet injections: dependence of jet penetration and dispersion in the skin on jet power. J. Control. Release97, 527–535 (2004). [DOI] [PubMed] [Google Scholar]
- 15.Buckley, S. T. et al. Transcellular stomach absorption of a derivatized glucagon-like peptide-1 receptor agonist. Sci. Transl. Med.10, eaar7047 (2018). [DOI] [PubMed] [Google Scholar]
- 16.Heise, T. et al. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes53, 1614–1620 (2004). [DOI] [PubMed] [Google Scholar]
- 17.Vinarov, Z. et al. Impact of gastrointestinal tract variability on oral drug absorption and pharmacokinetics: an UNGAP review. Eur. J. Pharm. Biopharm.162, 11–23 (2021). [DOI] [PubMed] [Google Scholar]
- 18.Abramson, A. et al. Quantifying the value of orally delivered biologic therapies: a cost-effectiveness analysis of oral semaglutide. J. Pharm. Sci.108, 3138–3145 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bass, D. M., Prevo, M. & Waxman, D. S. Gastrointestinal safety of an extended-release, nondeformable, oral dosage form (OROS): a retrospective study. Drug Saf.25, 1021–1033 (2002). [DOI] [PubMed] [Google Scholar]
- 20.Shergold, O. A., Fleck, N. A. & King, T. S. The penetration of a soft solid by a liquid jet, with application to the administration of a needle-free injection. J. Biomech.39, 2593–2602 (2006). [DOI] [PubMed] [Google Scholar]
- 21.Symons, D. D. Inertial liquid loading on the nozzle of a needle-free injection device. Proc. Inst. Mech. Eng. C218, 233–240 (2004). [Google Scholar]
- 22.Wendell, D. M., Hemond, B. D., Hogan, N. C., Taberner, A. J. & Hunter, I. W. The effect of jet parameters on jet injection. Conf. Proc. IEEE Eng. Med. Biol. Soc.2006, 5005–5008 (2006). [DOI] [PubMed] [Google Scholar]
- 23.Portal Instruments. Needle-free: solving challenges with viscous biologic self-injections. biopharmadealmakers (Advertising Feature) https://www.nature.com/articles/d43747-020-00305-y (2017).
- 24.Borwankar, A. U. et al. Viscosity reduction of a concentrated monoclonal antibody with arginine·HCl and arginine·glutamate. Ind. Eng. Chem. Res.55, 11225–11234 (2016). [Google Scholar]
- 25.Segur, J. B. & Oberstar, H. E. Viscosity of glycerol and its aqueous solutions. Ind. Eng. Chem.43, 2117–2120 (1951). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Linear regressions of pressure factor results.
Linear regressions of overshoot results.
Analysis of linear regressions of pressure factor results.
Analysis of linear regressions of overshoot results.
Results from organ comparison study.
Results from heat map study of jejunal tissue.
Results from heat map study of antral tissue.
Dynamics of depot formation in ex vivo jejunal tissue. This high-speed video captures the dynamic process of depot formation within excised porcine jejunum tissue. The jetting apparatus, positioned at a nozzle-to-tissue distance of 5 mm (dj=257 µm, pin=14.5 bar), is thoroughly described and characterized in Extended data fig. 1b-g.
Recoil and jetting dynamics of four MiDeAxAuto devices. This video presents simultaneous recordings of four MiDeAxAuto devices, demonstrating their triggering, jetting, and recoil behaviours. The devices are placed on a plastic film with a hole, through which they jet freely into a glass beaker, showcasing the reproducibility of the recoil effect.
Triggering and injection in ex vivo gastric antrum. This video illustrates the triggering and jetting process of a MiDeAxAuto device into excised porcine gastric antrum, providing insight into its injection dynamics.
In vivo triggering event of MiDeAxAuto in a porcine stomach. This video depicts the in vivo positioning and triggering event of the MiDeAxAuto device within the stomach of an anaesthetized porcine, offering a real-time observation of its functionality.
Data Availability Statement
All data that support the findings of this study are available within this paper, its supplementary information and the source data that are provided with this paper. Any additional information is available upon request.