Abstract
Aging is accompanied by considerable changes in the gut microbiome, yet the molecular mechanisms driving aging and the role of the microbiome remain unclear. Here we combined metagenomics, transcriptomics and metabolomics from aging mice with metabolic modelling to characterize host–microbiome interactions during aging. Reconstructing integrated metabolic models of host and 181 mouse gut microorganisms, we show a complex dependency of host metabolism on known and previously undescribed microbial interactions. We observed a pronounced reduction in metabolic activity within the aging microbiome accompanied by reduced beneficial interactions between bacterial species. These changes coincided with increased systemic inflammation and the downregulation of essential host pathways, particularly in nucleotide metabolism, predicted to rely on the microbiota and critical for preserving intestinal barrier function, cellular replication and homeostasis. Our results elucidate microbiome–host interactions that potentially influence host aging processes. These pathways could serve as future targets for the development of microbiome-based anti-aging therapies.
Subject terms: Computer modelling, Metabolomics, Metagenomics, Gene expression, Symbiosis
A multi-omics approach reveals host–microbiome interactions in aging in mice.
Main
Aging and aging-related diseases are central contributors to morbidity and mortality in Western societies1. Although research has identified specific hallmarks of aging2 and revealed the conservation of aging-associated changes across species and tissues3, the primary causative factors of aging remain elusive2. The microbiome, comprising a diverse bacterial community that resides within and on host organisms, is gaining recognition for its interplay with host aging processes. It is implicated in many aging-associated physiological processes4, showing notable shifts in its composition as the host ages and strong correlations with aging-related phenotypes5. Microbiome transfer experiments revealed that introducing young microbiota to old hosts extends their lifespan6,7 and reverses specific aspects of aging in animal models8. However, some studies have also shown beneficial effects of aged microbiota9 or signatures specific to healthy aging in centenarians6 that indicate that some aging-associated changes in the microbiota might also be compensatory by counteracting aging-associated changes in the host7. Pathological changes in the host’s gastroenteric system, such as obstipation, constipation and barrier dysfunction, are comorbidities of many aging-related diseases and often precede the manifestation of these diseases by many years8. Moreover, the aging-associated loss of intestinal barrier function, which facilitates the translocation of living bacteria and their products into the bloodstream, is implicated as a driver of systemic inflammaging, a hallmark of aging characterized by a constant low-grade inflammation even without the presence of a detectable pathogen10,11.
However, it remains unclear which microbiome changes are causes of aging in the host and which are consequences12. The primary reasons for this uncertainty are the high plasticity and complexity of the microbiota, which comprise dozens to hundreds of species13, the low species-level conservation of microorganisms across human cohorts14 and the myriad of metabolites through which the microbiota and host can interact15. One approach to overcome this complexity is constraint-based metabolic modelling16. This method builds on in silico representations of the metabolic networks of individual species—so-called genome-scale metabolic networks—and allows the prediction of metabolic fluxes in individual species or entire communities17. This approach enables the integration of different types of omics datasets to derive context-specific metabolic networks (that is, networks representing the metabolic state of particular tissues or cells)18. Therefore, several studies have used constraint-based metabolic modelling to investigate changes in microbiome–host interactions in various diseases19 and identify specific microbial processes linked to therapeutic response17,20.
In this study, we used tissue transcriptomic, metagenomic and metabolomic data to elucidate the metabolic mechanisms through which the gut microbiota could contribute to host aging. We extensively characterized microbiome–host interactions at the level of global associations between host transcript levels and microbiome functions and then focused on metabolic interactions using an integrated metabolic model of the host and the microbiota. Our results revealed many known interactions between the host and the microbiota and postulated numerous hitherto unknown ones. Next we investigated how these interactions change in the context of aging. We observed a considerable reduction in microbiome metabolic activity with age, which seemed to be driven by substantial changes in within-microbiota ecological interactions. We subsequently connected aging-related changes in the host with alterations in the microbiota and discovered that aging-regulated gene networks were significantly enriched for both microbiome-dependent genes and microbiota-dependent host functions, as predicted by our models. These functions showed a marked decline with age. Our findings indicate that the microbiome is a major contributor to aging-associated metabolic decline, which we also observe at the metabolome level and thereby pinpoints metabolic pathways through which the microbiome may influence aging in the host.
Results
Taxonomic and functional description of the mouse microbiome
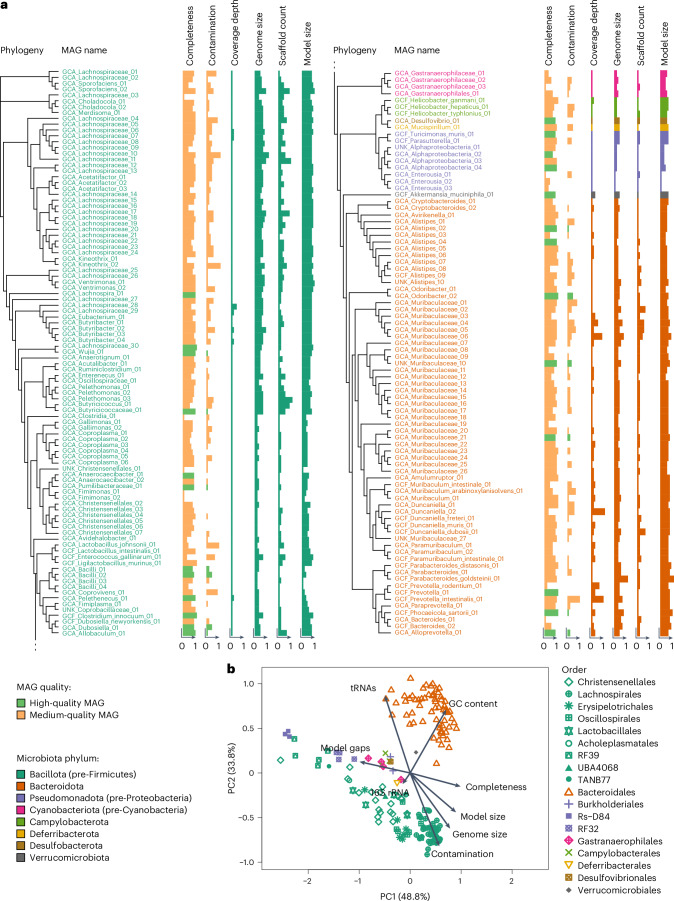
We studied the effects of aging in 52 male wild-type C57BL/6J/Ukj mice, separated into 5 age groups between 2 months and 30 months old, representing early adulthood until late age with ~5% survival21. We obtained transcriptome sequencing data for the colon, liver and brain, as well as shotgun (167 Gbp) and long-read sequencing data (13.7 Gbp) for faecal samples, which we used to reconstruct 181 metagenome-assembled genomes (MAGs; total 367 Mbp) of bacteria comprising their gut microbiome (Fig. 1a and Extended Data Fig. 1a–e). Taxonomic classification with the Genome Taxonomy Database Toolkit (GTDB-Tk)22 assigned 175 MAGs to known taxa (with the prefix ‘GCA_’ or ‘GCF_’), whereas 6 MAGs did not have a matching genome (prefixed ‘UNK_’). Of those 181 MAGs, 25 were considered high-quality drafts according to established criteria23, and the rest were considered medium-quality drafts. Notably, we used more stringent cut-offs (≥80% completeness and ≤10% contamination) than those suggested in ref. 23 for medium-quality MAGs to require less gap filling and thus obtain more reliable metabolic models for downstream analysis (Fig. 1a). Most of the MAGs were attributed to the phyla Bacillota (previously Firmicutes; n = 97) and Bacteroidota (n = 65). The reconstructed genomes from rarer phyla included Pseudomonadota (previously Proteobacteria; n = 9), Cyanobacteriota (previously Cyanobacteria; n = 4), Campylobacterota (n = 3), Deferribacterota (n = 1), Desulfobacterota (n = 1) and Verrucomicrobiota (n = 1). Regarding overall abundance, the most abundant MAGs, with a coverage depth >1%, belonged to Bacteroidota in the families Bacteroidaceae (n = 5) and Muribaculaceae (n = 12). The genome sizes of the MAGs ranged from 0.9 Mbp to 6.7 Mbp (Fig. 1a).
Fig. 1. Mouse microbiome and metabolic model characterization.
a, Phylogenetic tree of the 181 MAGs. See Supplementary Table 1.2 for detailed metadata information. b, Principal component analysis of the metabolic models of the mouse microbiota. Metadata associations to PCs are overlaid as arrows; the shapes denote the taxonomic rank order; the colouring of the symbols are according to a.
Extended Data Fig. 1. Metagenomic processing steps and results.
a Metagenomics workflow. b, c Abundance of metagenomics reads derived from b microbes or c mouse DNA. d Alpha diversity derived from metagenomic reads mapped against MAGs. b-d Dunn’s test FDR, all not significant; sample size: 30mo.: n = 12, all others n = 10. e Abundance profile of the 60 most abundant MAGs in the full cohort. Bacteroidota predominated in the cohort. Boxplot elements: center line, median; box limits, 25%–75% quantiles; whiskers, 1.5x IQR; points, outliers.
To functionally annotate the assembled MAGs, we used gapseq24 to reconstruct their corresponding genome-scale metabolic networks. In a principal component analysis of the networks (Fig. 1b), principal component (PC) 1 mainly separated models by the completeness score (R2 = 0.15) of the underlying MAGs and the taxonomic rank ‘order’ (R2 = 0.87). The completeness of the MAGs significantly impacted the prevalence of pathway gaps within the models. Consequently, the occurrence of such gaps (R2 = 0.55) and the sizes of the models (R2 = 0.84) or genomes (R2 = 0.58) partially accounted for the observed differentiation along the first two PCs. PC2 separated the metabolic models by the phylum, GC content (R2 = 0.29) and contamination score (R2 = 0.06).
Microbiome functions correlate with host transcripts
After reconstructing the metabolic models of the bacterial species of the mice, we first determined host functions associated with microbiome functions independent of age. Filtering by association strength and a false discovery rate (FDR)-adjusted P ≤ 0.1, we identified 12,732 correlated microbiome reactions and host genes for the colon, 3,425 for the liver and 2,499 for the brain. Enriching these features with gene ontology (GO)25 biological processes (host genes), and metabolic subsystems (microbiome reactions), we obtained 1,377 pairs of host–microbiome-associated processes for the colon, 283 for the liver and 167 for the brain; we further summarized these with level 2 GO biological processes and MetaCyc26 superpathways (Fig. 2a–c, Extended Data Fig. 2a and Supplementary Tables 2.1–2.4).
Fig. 2. Correlation-derived host–microbiome interactions.
a–c, Interactions between host biological processes and microbiome metabolic subsystems for the colon (a), liver (b) and brain (c). Only processes with the most significant associations (colon FDR ≤ 1 × 10−10, liver FDR ≤ 1 × 10−4 and brain FDR ≤ 1 × 10−3) with at least two interactions are shown. For complete data and full pathway names, see Supplementary Tables 2.1–2.4. d, Enrichment of microbiome-associated host genes among microbiome-colonization-responsive genes. FDR-corrected P values from upper-tailed (one-sided) hypergeometric tests of the overlap plotted above each bar.
Extended Data Fig. 2. Correlation-derived host–microbiome interactions.

a High-level pathway overview of host–microbiome associations. For complete data and full pathway names, see Supplementary Table 2.4. b HUMAnN3 predicted microbial functions (MetaCyc) correlated to host colon transcription (GO biological processes) using Biobakery 3. Bacterial pyruvate fermentation and co-factor biosynthesis were negatively correlated with host side neuron survival, while microbial fatty acids were found positively associated with colon immune processes (MHC class I) and negatively with transcription regulation. These HUMAnN3-based results were generally in concordance, although much less detailed, to our MAG and metabolic model derived host-microbiome correlations (cf. Fig. 2a).
The most strongly correlated host functions for the colon involved innate and adaptive immune processes and protein processing (Fig. 2a, Extended Data Fig. 2a and Supplementary Table 2.1). These included a negative correlation between host immune system processes and microbial galactose and arabinose degradation pathways. Moreover, we observed strong positive correlations between microbial purine metabolism and mitochondrial respiration in the host. Furthermore, we found that microbial pathways involved in lipid metabolism were correlated with host processes involved in tissue homeostasis, such as DNA damage responses and cell death. By directly inferring functions from quality-controlled metagenomic read data (HUMAnN3)27, we found fewer, yet comparable, host–microbiome associations (Extended Data Fig. 2b and Supplementary Table 2.7). For the liver, we detected associations between central metabolic pathways of the microbiota and chromatin organization in the host as well as between T cell proliferation and microbial branched-chain amino acid metabolism (Fig. 2b, Extended Data Fig. 2a and Supplementary Table 2.2). For the brain, we found strong correlations between protein catabolic processes and microbial nucleotide metabolism (Fig. 2c, Extended Data Fig. 2a and Supplementary Table 2.3). To independently validate host genes associated with microbiome functions, we determined their regulation in response to microbial colonization. To this end, we generated transcriptomic data from five tissues (colon, liver, brain, gonadal white adipose tissue and quadriceps) of three groups of mice: conventionally raised wild-type mice (CONVR), germ-free (GF) mice and mice conventionalized with faecal material from WT mice (CONVD; n = 8 per group). Comparing genes responsive to microbial colonization (comparison CONVR versus GF and CONVD versus GF; Supplementary Tables 2.8–2.12) and those associated with microbiome function, we found a highly significant overlap in colon and liver, but not in brain (Fig. 2d). The lack of significant overlap with microbiome-responsive genes in the brain might be partially due to the relatively small number of microbiome-responsive genes in this tissue (Supplementary Table 2.12).
Host–microbiome interactions in the metaorganism model
We next aimed to gain a more mechanistic understanding of the underlying metabolic pathways mediating host–microbiome associations with an integrated metabolic metamodel of the host and the microbiome. In this metamodel, the host is represented by three different tissues (colon, liver and brain) connected through the bloodstream and interacting with the microbiome through the gut lumen (Fig. 3a). Each host tissue is represented by a unique instance of the human metabolic reconstruction Recon 2.2 (ref. 28), whereas the microbiome is represented by a combined model including all the metabolic reactions occurring in at least one bacterial metabolic model reconstructed from the MAGs (Fig. 3a). Subsequently, context-specific metabolic metamodels representing the metabolic state of each mouse were built based on tissue transcriptomic and metagenomic data using fastcore29 (Supplementary Table 3.1).
Fig. 3. Model-predicted host–microbiome interactions.
a, Structure of the metamodel. The solid borders indicate compartments of the metamodel. The black arrows indicate metabolite exchanges between compartments. The dashed borders indicate compartments represented only by exchange reactions. The white arrows indicate the direction of metabolic exchanges along the bloodstream. BBB, blood–brain barrier. b, Frequency of microbiome dependence of metabolite import (positive) and export (negative) across organs. Metabolites with the highest frequency of exchange across 52 models are shown (Supplementary Table 3.2). For metabolite abbreviations, see Supplementary Table 3.2. c, Microbiome dependency of microbiome-responsive host genes in a cohort of GF, conventionalized and conventionally raised mice (n = 8 each). The y axis indicates sets of genes differentially regulated in tissues and contrasts; the x-axis shows the microbiome dependency of corresponding reactions. ‘Shared’ indicates genes regulated in at least three tissues. FDR-corrected P values of Dunn’s tests following a group-level Kruskal–Wallis test are shown next to the bar plots of means with error bars representing the standard deviation. Only comparisons with a Kruskal–Wallis test P < 0.05 are shown. *P < 0.05; **P < 0.01; ***P < 0.001. Exact P values are provided in Supplementary Table 3.11. d–f Subsystem enrichment of model-predicted interactions between host and microbiome reactions for subsystems connected with at least two host subsystems and an FDR-corrected enrichment P < 10−4 (one-sided Fisher’s exact test; Supplementary Tables 3.3–3.5). For pathway abbreviations, please see Supplementary Tables 3.3–3.5.
To explore the extent to which the metamodel could reconstitute known host–microbiome interactions, we used it to predict metabolites exchanged between host and microbiota (Fig. 3b and Supplementary Table 3.2). In the colon, we observed many known interactions including a provision of the microbiome with bile acids as well as fucose, a part of mucins30, by the host and a microbial production of short-chain fatty acids. Moreover, we observed that the microbiota produced many nucleotides, including nucleotide derivatives such as NAD and coenzyme A. For the liver, we observed a provision of primary bile acids to the microbiota and microbial production of nucleotides and short-chain fatty acids. For the brain, we observed that the host was provided with the microbial fermentation product ethanol and several pyrimidines. The brain and colon provided the pyrimidine precursor orotate and the nucleotide degradation product uracil to the microbiota, while the microbiota provided uridine and deoxycytidine in return. Overall, we found that among the predicted interactions shown in Fig. 3b, 42 (51%) were already supported by previous experimental evidence across all three organs (Supplementary Table 3.9), thereby strongly supporting the ability of the metamodel to capture metabolic microbiome–host interactions.
To elucidate the underlying metabolic pathways connecting the host and microbiota that might mediate the extensive associations we have observed, we sampled elementary flux modes (EFMs)31 in the metamodel with the EFMSampler32. Each host reaction was defined as an indicator reaction through which EFMs were sampled. By recording the frequency at which microbial reactions occurred in the EFMs of a host indicator reaction, we obtained an interaction matrix of the frequency at which microbiome reactions occurred in the pathways sampled for individual host indicator reactions. Using these interaction matrices, we found that correlated host gene–microbiome reaction pairs (see Fig. 2) had a higher frequency of model-predicted interactions compared with randomly sampled pairs for liver and colon, but not for brain (Extended Data Fig. 3a). These findings suggest a coupling of host metabolic transcription and microbiome metabolic functionality, even though this might be biased by the utilization of the same data basis for determining host–microbiome correlations and reconstructing the metamodel.
Extended Data Fig. 3. Metamodel validation.
a Maximal interaction scores for pairs of host–microbiome-associated processes versus 100 randomly selected pairs of host genes and microbiome reactions for each tissue (see Methods). P-values indicate the significance of the differences according to a two-sided Wilcoxon rank-sum test. b Randomization of germfree mouse analysis (cf. Fig. 3c). The identification of significant differences in microbiome dependence between significantly up-regulated, down-regulated as well as unregulated genes was repeated 1000 times after randomization of gene assignments and the number of significant associations was counted. The dashed red line indicates the number of significant associations in the original analysis. c Association between model-predicted microbiome dependency of metabolites in blood and explained variation of serum concentrations of the corresponding metabolite by microbiome composition in a human cohort (each dot is one metabolite). The association between microbiome dependency and explained variance is significant using Spearman’s correlation (rho=0.43, p-value = 1.5×10-3). Boxplot elements: center line, median; box limits, 25%–75% quantiles; whiskers, 1.5x IQR; points, outliers.
To validate the metamodel’s ability to identify microbiome–host interactions, we examined how microbial colonization influenced predicted microbiome-dependent host reactions using gene expression data from our GF mouse cohort. In addition to the colon, liver and brain, we analysed gonadal white adipose tissue and quadriceps to assess whether microbiome dependency in one tissue could inform predictions for others. For each tissue, we identified upregulated, downregulated and unregulated genes, mapped them to reactions and evaluated their predicted microbiome dependency. Upregulated reactions showed significantly higher microbiome dependency than unregulated reactions in seven comparisons, while downregulated reactions showed higher dependency in three (Fig. 3c). Only two instances showed lower dependency in regulated reactions, strongly supporting the ability of the metamodel to capture functional host–microbiome interactions. To show that these results were not due to modelling-inherent biases, we repeated the analysis with gene labels randomized and did not find a single case with a higher number of significant associations across 1,000 randomized repetitions (Extended Data Fig. 3b). We further validated the metamodel by showing a strong correlation between model-predicted microbiome dependence of serum metabolites and microbiome-driven variance of those metabolites in an independent human metabolomics cohort33 (Spearman’s ρ = 0.43, P = 1.5 × 10−3; Extended Data Fig. 3c and Supplementary Table 3.10).
To further functionally characterize the host–microbiome-interaction matrix, we performed enrichments for host and microbial metabolic subsystems (Fig. 3d–f). In the colon, we found host pathways associated with energy metabolism, nucleotide metabolism, vitamin metabolism and amino acid metabolism (Fig. 3d) depending on fermentation products, nucleotide metabolism and vitamin biosynthesis pathways of the microbiota. In the liver, energy-producing pathways and bile acid synthesis were prominent on the host side and fermentation pathways on the microbiome side (Fig. 3e). In the brain, microbiome-dependent host reactions were enriched in nucleotide metabolism, folate metabolism and the metabolism of neurotransmitter precursors, such as tryptophan and tyrosine (Fig. 3f). Although most host–microorganism interactions were relatively generic, relying on basic microbial metabolic functions (such as glycolysis and fermentation), we also identified specific interactions, such as colonic nucleotide interconversion dependent on microbial ATP synthesis and colonic coenzyme A catabolism reliant on microbial production of phosphopantothenate, a coenzyme A precursor.
Aging is linked to reduced microbiome metabolic activity
We next explored functional and taxonomic changes in the aging microbiome. Consistent with previous reports in mice34,35, we observed that age was associated with a decrease in the abundance of Bacillota and an increase in Bacteroidota (Fig. 4a), also when inferring taxonomic changes from metagenomic data directly (Extended Data Fig. 4a and Supplementary Table 4.15). To obtain a better functional understanding of these species-level changes, we used community flux balance analysis (FBA)17 to predict microbial metabolic activities (Supplementary Methods). In contrast to the metaorganism modelling approach used in the previous section, which does not differentiate between individual microbial species owing to computational limitations, community FBA models each microbial species individually. Summarizing age-associated reactions on the pathway level, we mainly observed negative associations (Fig. 4b) involving many biosynthetic pathways essential for bacterial replication, such as synthesis of amino acids, nucleotides, vitamins and cell wall components. Similarly, for metabolic interactions between the microbiota and the host as well as within the microbiota, we mainly observed strong reductions in both the consumption and production of metabolites (Fig. 4c and Extended Data Fig. 4b), including the production of the short-chain fatty acid butyrate, and increased production of few metabolites, including pro-inflammatory succinate36. Consistent with a generally reduced microbial metabolism, we also found that model-predicted and metagenomics-derived microbial growth rates decreased considerably with age (Fig. 4d) and were strongly correlated (Extended Data Fig. 4c). Furthermore, we evaluated the change of FBA-predicted community growth upon removal of single bacterial members, and observed that MAGs suppressed in aging had a beneficial effect on community productivity and growth, while MAGs which were enriched in old mice showed a negative impact (Extended Data Fig. 4d,e).
Fig. 4. Microbiome alterations associated with host age.
a, Aging-associated changes in MAG abundance. b, Subsystem-level aging-associated changes in microbiome internal reaction fluxes. c, Aging-associated changes of host–microbiota metabolic exchange. d, Comparison of microbiome community growth rates derived from FBA or the PTR (30 months: n = 12; all others, n = 10; FDR-corrected P values from Dunn’s test following Kruskal–Wallis test). e, Aging-associated changes in model-predicted ecological interactions in the microbiota. Linear-model-derived regression with 95% confidence intervals (30 months: n = 12; all others, n = 10). f, Aging-associated changes in faecal metabolite concentrations in mice. All age-associated metabolites are shown (FDR-adjusted P ≤ 0.1 from Spearman correlations; log2(fold change (FC)) of 3 to 28 months; 3 months: n = 15; 9 months: n = 16; 15 months: n = 15; 24 months: n = 17; 28 months: n = 18; Supplementary Table 4.12). The origin of bile acids is indicated. ‘Both’ refers to bile acids produced by the host but regulated by the microbiota. Metabolites with the prefix ‘BA_Feature’ have not been fully resolved. incr., increase; decr., decrease; CA-7S, cholic acid-7-sulfate; CDCA-7S, chenodeoxycholic acid-7-sulfate; MCA, muricholic acid; TMCA, tauromuricholic acid. Box plot elements: centre line, median; box limits, 25–75% quantiles; whiskers, 1.5× interquartile range (IQR); points, outliers.
Extended Data Fig. 4. Age-dependent microbiome changes.
a MetaPhlAn4 derived species with significant changes in abundance with host age. The MetaPhlAn-based approach identified 335 bacteria at the species level of which 69 were found differentially abundant with age. The species Lactobacillus johnsonii matched the MAG based analysis (Fig. 4a). However, due to different taxonomic databases and methods of annotation the resulting species names might vary. On phylum level, the overall aging pattern of both MetaPhlAn and MAG-based taxonomic analysis yielded comparable results, which was a general decrease of Bacillota spec. and an increase of Bacteroidota spec. with host age. b Age-associated changes in metabolic fluxes within the community. c Comparison of community growth rates derived from FBA or PTR. d, e Change of microbiome community growth rate upon removal of a single MAG from the community. The y-axis shows the difference of community growth rate compared to the full community while the x-axis names the MAG that was removed in d or the age group of the host in e (p-values via FDR corrected Wilcoxon’s rank sum test). f Relative abundance of universal adaptive strategies in microbiome communities by age (p-values via FDR corrected Dunn’s test). Sample size for a-f: 30mo.: n = 12, all others n = 10. g Metabolomics derived D-galactose concentration in mouse feces increases with age (Spearman’s ρ = 0.22, unadjusted p = 0.04; Pairwise p-values via Dunn’s test with FDR correction; 3mo.: n = 15, 9mo.: n = 16, 15mo.: n = 15, 24mo.: n = 17, 28mo.: n = 18). Boxplot elements: center line, median; box limits, 25%–75% quantiles; whiskers, 1.5x IQR; points, outliers.
To gain insight into the potential microbiome-intrinsic causes of the observed aging-associated suppression of metabolism, we used community FBA17 to predict the frequencies of ecological interactions (Methods). We observed a significant decrease in amensal, commensal and neutral interactions at the expense of increased competitive interactions (Fig. 4e). These shifts in community interactions were also observed at the level of individual microbial ecological strategies derived from the universal adaptive strategies theory framework37,38, indicating a shift in the community towards the dominance of ruderals, which are first colonizers of niches and poor interaction partners owing to reduced catabolic diversity37 (Extended Data Fig. 4f).
To further explore the predicted loss of microbiome metabolic activity and metabolic cooperativity with age, we performed an untargeted metabolomics analysis of faecal samples from an independent cohort of 82 mice across all age groups. We determined the correlation between the abundance of identified metabolomic features and age and found that 374 of 561 features (67%) showed significant downregulation (FDR-adjusted P ≤ 0.1; Fig. 4f). Within this dataset, we specifically annotated bile acids using reference standards because of their previously documented role in host aging39. Consistent with the reduction in microbiome metabolic activity with age, we found that the concentrations of host-regulated bile acids were significantly increased (four out of six features). By contrast, the concentrations of microbiome-regulated bile acids were mostly reduced (seven out of eight features). Intriguingly, metabolomic features annotated as cholic acid-7-sulfate, which is produced by the host but regulated by the microbiota40, were exclusively downregulated with age. Also, further microbiome-regulated metabolites, including valine, betaine, nicotinamide, enterolactone and 3-hydroxykynurenine, were downregulated with age (Supplementary Table 4.12). Moreover, we found an increase in the pro-inflammatory microbial metabolite d-galactose, for which we observed a strong association with host immune processes in the colon (Fig. 2b), although only significant before FDR correction (Extended Data Fig. 4g).
Aging decline of microbial metabolism impacts host functions
Next, we investigated how the aging-associated loss of microbiome metabolic function potentially impacted host functions. To this end, we used differential gene expression analysis and GO term enrichment to identify aging-regulated genes and processes. Consistent with our previous work, we found a considerable conservation of aging-regulated genes across tissues3 including 157 transcripts that were consistently downregulated and 526 genes that were consistently upregulated. Upregulated genes were mostly enriched for immune-associated processes and downregulated genes in cellular maintenance and tissue regeneration processes (Fig. 5, Extended Data Fig. 5a–d and Supplementary Tables 5.1‒5.9). Exploring connections between aging and the microbiome, we found a highly significant enrichment of microbiome-correlated transcripts among aging-regulated genes across all tissues (Fig. 6a). Along with the loss of microbiome metabolic function with age (see Fig. 4), we also found a pronounced loss of host–microbiome associations with age (Extended Data Fig. 6a–e). We found a higher number of GO biological processes for both aging and microbiome-associated genes in the colon than in the liver and brain (Fig. 6b, Extended Data Fig. 6f,g and Supplementary Tables 6.1–6.4). Notably, tissue homeostasis and organ regeneration processes were downregulated in the colon with age but positively correlated with microbial metabolic pathways. Conversely, aging-induced processes, primarily defence, inflammatory and immune responses, were negatively associated with microbial metabolism (Fig. 6b). Brain development was negatively correlated with microbial metabolism and downregulated with aging (Extended Data Fig. 6g). On the microbial side, glycolysis, nucleotide synthesis and d-galactose degradation were suppressed with age and mostly negatively correlated with host gene expression (Fig. 6c). In connection with the aging-associated increase in microbial production of the pro-inflammatory metabolite succinate, observed in community modelling (see Fig. 4c), we also identified many aging-associated changes in succinate-metabolizing microbial pathways correlated with host gene expression (for example, oxalate and itaconate degradation).
Fig. 5. Aging-associated transcriptomic changes across host tissues.

Enriched GO biological processes, shared by at least two organs, are shown as the average expression of all associated features, stratified by age group and organ (hypergeometric test FDR cutoff for displayed terms: colon, 10−4; liver, 10−6, brain 10−6; 30 months: n = 12; all others n = 10). For complete data and full pathway names, see Supplementary Tables 5.4‒5.6.
Extended Data Fig. 5. Aging-associated transcriptomic changes across host tissues.
a Each row represents a gene that shows common expression changes across all tissues studied (see Supplementary Table 5.7‒5.9). b–d Enriched GO biological processes are shown as the average expression of all associated features, stratified by age group and organ. (Hypergeometric test FDR cutoff for displayed terms: colon, 10−4; liver, 10−6, brain 10−6; 30 months: n = 12; all others n = 10). For complete data and full pathway names, see Supplementary Tables 5.4‒5.6.
Fig. 6. Aging-associated changes in host–microbiome interactions.
a, Overlap between aging-regulated and microbiome-regulated host genes (P values via upper-tailed hypergeometric test). b, Colon-specific gene expression changes with age in processes correlated with microbiome metabolic functions (legend shared with c). c, Aging-dependent changes in microbiome processes correlated with host gene expression. For complete data and full pathway names, see Supplementary Tables 6.1 and 6.4. d, Frequency of microbiome dependence of aging-regulated metabolic modules across host tissues (P values via one-sided Fisher’s exact test). e, Subsystem-level enrichment of indicator reactions of aging-regulated metabolic modules (one-sided Fisher’s exact test). The x axis represents enriched host subsystems; the y axis represents aging-regulated gene sets. f, Aging association of brain metabolites predicted to be exchanged between the microbiome and host. Data from ref. 41 (Supplementary Table 6.9). P values obtained by two-sided Wilcoxon rank-sum test. g, Aging-associated metabolome changes for selected model-predicted microbiota-produced and microbiota-consumed metabolites (P values via Kruskal–Wallis test). Data from ref. 41 (n = 64 mice). Box plot elements: centre line, median; box limits, 25–75% quantiles; whiskers, 1.5× IQR; points, outliers. Significance: *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001. Exact P values are provided in Supplementary Table 6.9.
Extended Data Fig. 6. Host-Microbiome associations in aging.
a–e Strong host–microbiome correlations stratified by age group, compared with Yates’s chi-squared test (one-sided). a Frequency of microbiome–colon correlations stratified by age group. Significance was tested via Yates’ Chi-squared test with Bonferroni multiple testing correction. b Liver transcripts correlated with microbiome reactions. c Liver transcripts partially correlated with microbiome reactions, corrected for sequencing batch. d Brain transcripts correlated with microbiome reactions. e Brain transcripts partially correlated with microbiome reactions, corrected for sequencing batch. f–g Organ-specific gene expression changes with age in GO biological processes that were also correlated with microbiome metabolic functions (F = liver, G = brain). For complete data and full pathway names, see Supplementary Tables 6.2, 6.3.
Given the observed reduction in microbiome–host associations, we next aimed to identify the underlying metabolic pathways potentially mediating those changes. To achieve this, we defined aging-regulated metabolic modules among the metamodel’s metabolic reactions using the host–microbiome-interaction matrix obtained from EFM sampling. Metabolic modules were determined according to sampled EFMs (compare with Fig. 3d–f), selecting reactions present in at least 20% of the EFMs for each indicator reaction (Methods). Aging regulation of those modules was then inferred from the over-representation of aging-regulated reactions in each module. In the colon, liver and brain, we identified aging-induced (51, 88 and 99, respectively) and aging-repressed (2,509, 1,702 and 524, respectively) metabolic modules (Supplementary Tables 6.5–6.7), with aging-repressed modules being significantly more dependent on the microbiome across all tissues (Fig. 6d). These modules revealed downregulation of colon metabolic pathways linked to fatty acid oxidation, N-glycan synthesis and sphingolipid metabolism, which are central to cellular homeostasis. In the liver, downregulated modules were enriched in bile acid synthesis and triacylglycerol synthesis, aligning with age-related shifts in bile acid profiles (see Fig. 4f). In the brain, aging-regulated modules were enriched in nucleotide as well as sphingolipid metabolism and transport pathways (Fig. 6e and Supplementary Table 6.8). Given the strong effect of aging-suppressed microbiome metabolism on the brain (Fig. 6d,e), we analysed its impact on the brain metabolome using a public mouse dataset41. Correlations between metabolite concentrations and mouse age revealed that metabolites provided from host to microbiota accumulated with age, while microbiota-derived metabolites were depleted (Fig. 6f). This included accumulation of nucleotide precursors such as orotate and uracil, and depletion of salvage pathway products such as adenosine, 2-deoxycytidine and uridine (Fig. 6g).
Discussion
In this study, we performed a model-based analysis of aging-associated alterations in host–microbiota interactions in mice. We reconstructed 181 MAGs using shotgun and long-read sequencing, converting them into constraint-based metabolic networks. Our investigation revealed extensive associations between microbiome functions and the host’s colon, liver and brain transcriptome. Many correlations involved host immune processes, mitochondrial function and chromatin modification, alongside microbiome-derived metabolites such as d-galactose, known to promote neurodegeneration and inflammation42, and leucine, a regulator of T cell function43. Associations also highlighted microbial fermentation and nucleotide metabolism, consistent with the roles of microorganism-produced short-chain fatty acids and nucleotides in colonic energy balance44 and intestinal barrier function45 (Extended Data Fig. 7).
Extended Data Fig. 7. Summary of study highlights.

Multi-omics analysis and metabolic modeling of aging mice revealed a decline in microbiome metabolic activity, reducing beneficial host-microbe and microbe-microbe interactions. Aging-related microbiome changes led to the accumulation of pro-aging metabolites (D-galactose, succinate) and a decline in beneficial metabolites (nucleotides, butyrate). These shifts correlated with increased systemic inflammation and downregulation of essential host pathways, like energy and nucleotide metabolism, which are involved in intestinal barrier function and cellular homeostasis.
A key aspect of our analysis was the reconstruction of metabolic metaorganism models, adapted from whole-body metabolic models for humans46. These models successfully recovered well-documented microbiome–host interactions involving short-chain fatty acids, bile acids and other microbial metabolites. Notably, 51% of the predicted high-confidence interactions were corroborated by existing literature. Furthermore, genes found to be regulated by microbial colonization also showed a higher microbiome dependence in the metamodels, underscoring the metamodel’s ability to infer host–microbiome interactions accurately.
Examining reaction-level dependencies via EFM analysis revealed that the host most often depended on central metabolic reactions of the microbiome. This strategy may reduce reliance on specific bacterial species, broadening the potential pool of interaction partners47, consistent with conserved gut microbiome functions across human cohorts48. However, the focus on central pathways might also reflect biases in the model’s representation of bacterial metabolism.
Another interesting aspect of the predicted microbiome–host exchanges are metabolites that the host can produce itself, such as nucleotides. The reasons for the existence of such exchanges could include advantages from a division of labour, as frequently observed within microbial communities49, a reliance of the host on the microbiota as a metabolic backup system to increase phenotypic plasticity50, or evolutionary addiction, whereby mutual dependencies develop owing to the constant exposure of the host to microbially produced metabolites51.
Aging-associated changes of the microbiome communities revealed increases in Bacteroidota and decreases in Bacillota species, reflecting human studies linking Bacteroidota persistence to poorer health and Bacillota enrichment to healthier aging5. Furthermore, we observed reduced microbiome growth and metabolic activity, specifically for the production of butyrate as key changes in aging. This aligns with findings of decreased serum butyrate levels in aged mice and humans35,52. By contrast, metabolic modelling predicted increased production of the pro-inflammatory metabolite succinate53, a known indicator of a dysbiotic gut environment54, which we found to be associated with key processes deregulated in aging on the host side including, as previously reported, DNA damage response55 and protein homeostasis56. Reduced microbial growth could underlie increased constipation risk57 and longer colonic transit times58 observed during aging. Reduced microbial growth and a decreased capacity to turn nutrients into biomass have previously also been observed as a direct effect of increased transit times in a bioreactor setup mimicking the colon59.
Our analysis suggested that age-related changes in gut ecology involved increased competition and decreased cross-feeding, reducing dietary resource utilization efficiency. These trends were reflected in faecal metabolomics, in which most metabolic features decreased with age. Host-regulated bile acids increased, while microbiome-regulated bile acids declined. Anti-inflammatory metabolites such as valine, betaine and 3-hydroxykynurenine60–62 decreased, while pro-inflammatory metabolites such as d-galactose increased63.
Aging-associated inflammation and suppressed cellular replication across host tissues were consistent with our previous findings on conserved aging signatures3. Colon-specific changes in the transcriptome, supported by previous studies, included altered gut motility64, reduced colonic barrier function65 and proliferation66, while the liver showed a decrease in mitochondrial biogenesis, as previously observed67.
Aging also considerably affected host–microbiome interactions. Aging-regulated host genes were enriched for those correlated with microbiome functions, particularly in downregulated metabolic modules central to cellular homeostasis, such as fatty acid oxidation and nucleotide uptake. These findings align with our correlation analyses showing positive associations between microbial metabolism and host homeostasis and negative associations with inflammation. We previously observed a similar loss of microbiome–host interactions as a key component of pathology in inflammatory bowel disease20. The observed loss of host–microbiome interactions across all organs indicates that the microbiome might contribute to crucial aspects of the systemic aging process, such as metabolic decline68,69 and the loss of cellular proliferation, along with stem cell exhaustion2.
Microbially produced nucleotides emerged as a key metabolic exchange, with model predictions indicating host provision of precursors (for example, orotate) and degradation products (for example, uracil) to the microbiota, which provided nucleotides in return. This exchange aligns with our observation of widespread correlations across all tissues between host gene expression and microbial nucleotide metabolism. Despite host capability for de novo nucleotide synthesis, recent studies emphasize the microbiota’s contributing role, particularly in the colon45,70. Bacterial species such as Escherichia coli and Bacteroides spp. actively excrete ATP, non-lyticly, during growth71,72, and bacterial ATP contributes to intestinal barrier function45 as well as immune modulation via purinergic receptors70. An aging-associated decline in nucleotide co-metabolism could underlie diminished intestinal barrier integrity45, linked to age-related diseases73,74, reduced systemic proliferative capacity75 and impaired mitochondrial function76. Furthermore, microbiome involvement in brain nucleotide salvage, crucial for DNA repair and cellular homeostasis77, might relate to neurodegeneration78.
In summary, we identified pronounced aging-associated changes in microbiome–host interactions, largely driven by reduced microbial metabolic activity. Although limited by its reliance on modelling, our study validated many interactions through independent analysis and literature, offering insights into the systemic aging process. Notably, while metabolic metamodels identify specific metabolite exchanges, transporter promiscuity and modelling limitations may imply the exchange of structurally related compounds in vivo. A further limitation of our study was the exclusive use of male mice, as the logistical challenges of establishing a separate aging cohort for females precluded the inclusion of both sexes. Focusing initially on males ensured consistency by avoiding sex-specific variability. Consequently, sex-specific changes were not investigated in this study but will be considered in future research. Besides chronological age, future work should also incorporate epigenetic clocks and biological age markers such as frailty, loss of motor function and cognitive decline79. Finally, our identification of a loss of microbiome metabolic activity indicates a potentially crucial aging-associated change that could contribute to many aging-associated pathologies in the host. Therefore, microbiome metabolic activity could be a target for future microbiome-based therapies. Our modelling approach could play a crucial role in designing targeted interventions aimed at mitigating microbiome-driven aspects of aging.
Methods
Mouse strains
Main study and metabolomics cohorts
The mice used for the aging study were an in-house strain derived from the C57BL/6J strain (The Jackson Laboratory). These C57BL/6J/Ukj mice lack two common mutations found in the C57BL/6J strain: the DIP686 mutation in the crumbs family member 1 (Crb1) gene, which is vital for eyesight in aging mice, and a mutation in the nicotinamide nucleotide transhydrogenase (Nnt) gene, which encodes mitochondrial NAD(P) transhydrogenase, protecting against oxidative stress. Preserving both these genes is advantageous for metabolic and aging studies in mice.
GF mice cohort
The GF mice used for the analysis of host responses to microbial colonization were rederived axenic conventional Jackson Laboratory C57BL/6J, strain 000664 mice (The Jackson Laboratory). The mice were housed in the Experimental Biomedicine facility at the University of Gothenburg, Sweden.
Animal handling
Main study cohort
Male C57BL/6J/Ukj mice were bred in the Central Experimental Animal Facility at Jena University Hospital (Jena, Germany). The mice were housed at 22 ± 2 °C with a 14:10 h day–night cycle and a relative humidity of 55% ± 10%. They were co-housed according to their birth cohort (similar ages) in standard cages (GM500, Type III; Tecniplast Deutschland), and a maximum of two mice from the same cage were used for experiments. The mice had unlimited access to water and food (mouse V1534-300, ssniff Spezialdiäten). Next-generation RNA sequencing of host tissues and metagenomics of faecal samples were conducted in 52 mice of different ages spanning the mouse’s adult lifespan (2–3 months (mean = 2.5 months), 9–10 months (mean = 9.8 months), 15–17 months (mean = 15.9 months), 24–25 months (mean = 24.8 months) and 28–31 months (mean = 29.1 months)). For simplicity, the five age groups are referred to as 2 months (n = 10), 9 months (n = 10), 15 months (n = 10), 24 months (n = 10) and 30 months (n = 12) throughout the paper (Supplementary Table 1.1). In our study, we focused exclusively on male mice for two primary reasons. First, we aimed to minimize potential confounding factors arising from fluctuations in sex hormones in female mice, which are known to influence metabolic processes across tissues during aging80. Second, addressing sex differences in aging would have required a fully stratified experimental design3 and, consequently, a separate cohort of female mice. Given that only 10–15% of animals typically reach the age of 30 months, achieving comparable sample sizes and statistical power for the oldest age group alone would have necessitated approximately 100 female mice.
Metabolomics cohort
An independent mouse cohort was used for the metabolomics analysis of faecal samples. This cohort comprised 83 male mice in five age groups: 3 months (n = 16), 9 months (n = 16), 15 months (n = 16), 24 months (n = 17) and 28 months (n = 18) (Supplementary Table 4.11). These mice were bred and housed in the same mouse facility under the same conditions.
GF mice cohort
For the analysis of host responses to microbial colonization, tissues of female C57BL/6J mice (n = 24) were obtained from the Experimental Biomedicine facility at the University of Gothenburg, Sweden. Throughout the experiment, the mice had ad libitum access to chow and water and were exposed to a 12:12 h light–dark cycle. The mice were divided into three treatment groups: GF (n = 8), conventionally raised (CONVR, n = 8) and conventionalized (CONVD, n = 8). The mice within each group were not all littermates. A CONVR mouse, which was not part of the sampled CONVR group and was ~10 weeks old, served as the donor for the conventionalization process. GF mice were orally gavaged with gut microbiome at 10 weeks of age on average (Supplementary Table 1.1). The gut microbiome used for conventionalization was extracted from the caecum and mixed with reduced phosphate-buffered saline to obtain a final volume of 200 μl.
Sample collection
Main study and metabolomics cohort
The mice were sacrificed by cervical dislocation in three cohorts (randomized by age) on three consecutive mornings. The left hemisphere of the brain was prepped on ice, transferred to liquid nitrogen for storage and used later for RNA extraction. Faeces were collected from the colon by squeezing the colon contents towards the distal end and snap-freezing one pellet in liquid nitrogen; the pellets were used later for metagenomic sequencing (for the first cohort) or metabolite measurement by hydrophilic interaction liquid chromatography ultrahigh-performance liquid chromatography–tandem mass spectrometry (for the second cohort). The colons were rinsed with sterile phosphate-buffered saline and cut longitudinally; a piece measuring the length of one-eighth of the left half of the mid colon was frozen in liquid nitrogen for later use in RNA extraction. A piece with a length of approximately 1 cm was cut from the end of the left lateral lobe of the liver and snap-frozen in liquid nitrogen for later use in RNA extraction. RNA was extracted from tissue samples of the liver, colon and left brain hemisphere using the phenol–chloroform extraction method with 1 ml of Qiazol Lysis Reagent (Qiagen)81.
All studies were performed in strict compliance with the recommendations of the European Commission for the protection of animals used for scientific purposes and with the approval of the local government (Thüringer Landesamt für Verbraucherschutz, Germany; license: 02-024/15; TWZ-000-2017). Experiments were performed according to the ARRIVE guidelines82.
GF mouse cohort
At ~12 weeks of age, all mice were sacrificed for the extraction of brain, colon, liver, gonadal white adipose tissue (gWAT) and quadriceps tissues. RNA was isolated from the brain, colon, liver and quadriceps via the ‘RNeasy mini kit’ (Qiagen) according to the manufacturer’s protocol, while RNA from gonadal white adipose tissue was isolated using the TRIZOL method83. Briefly, 1 ml TRIzol was added to 50–75 mg pestle-homogenized tissue followed by vortexing, a 5-min incubation at room temperature and addition of 200 μl chloroform. After mixing, further incubation at room temperature for 2–3 min and centrifugation (12,000 g) at 4 °C for 5 min, the clear supernatant was mixed with 500 μl isopropanol followed by incubation at room temperature for 10 min. After further centrifugation (12,000 g) at 4 °C for 10 min, the supernatant was discarded and the pellet washed with 1 ml cold 75% EtOH followed by vortexing and centrifugation (7,500 g, 4 °C, 5 min). The pellet was dried and dissolved in RNase-free water. All animal protocols were approved by the Gothenburg Animal Ethics Committee (vote #2652-19).
Metagenomic sequencing
Microbial DNA was extracted from colon contents with the DNeasy PowerSoil Kit (Qiagen) following the manufacturer’s protocol. Next, the DNA was prepared at the Max Planck Institute for Evolutionary Biology (Plön, Germany) with the Illumina NexteraXT Library Kit. All 52 samples were pooled and sequenced for 2 × 150 cycles in paired-end mode on all four lanes of an Illumina NextSeq 500 machine. Demultiplexing was performed with one mismatch allowed in barcodes. The raw read data were merged sample-wise and subjected to quality control for adaptor contamination and base call qualities. Adaptor sequences with an overlap of ≥3 bp and base calls with a Phred+33 quality score of <30 were trimmed from the 3′ ends of reads using Cutadapt (version 1.12). Illumina’s Nextera transposon sequence and the reverse complement of TruSeq primer sequences were used as adaptor sequences.
Subsequently, reads were subjected to quality control using Prinseq lite (version 0.20.4) with a sliding window approach that applied a step size of 5 bp, a window size of 10 bp, a mean base quality of <30 and a minimum-length filter that discarded any reads shorter than 50 bp after all other quality control steps. To filter out host sequences, the remaining sequences were mapped to the mouse reference genome (GRCm38.99) with Bowtie (version 2.2.5). The remaining unmapped reads were then used for MAG assembly.
No significant differences were detected in the total microbial read depth or host contamination between age groups (Kruskal–Wallis test with post hoc Dunn’s test and Benjamini–Hochberg multiple-testing correction conducted with the DunnTest function in the DescTools R package (version 0.99.50); Extended Data Fig. 1b,c).
Long-read sequencing was performed at the next-generation sequencing (NGS) core facility of the FLI Leibniz Institute on Aging (Jena, Germany). The DNA quality was assessed with an Agilent Bioanalyzer 2100 with a DNA 12000 Kit (Agilent Technologies) and quantified with an Invitrogen Quant-iT PicoGreen dsDNA Assay (Thermo Fisher Scientific). The sequencing library was prepared according to the Pacific Biosystems’ manual ‘Procedure & Checklist - 20 kb Template PreparationUsingBluePippin Size-SelectionSystem’ (version 10, January 2018) with the SMRTbell Template Prep Kit 1.0 (Pacific Biosciences). Specifically, DNA from age-matched samples was pooled, fragmented (75 kb) by a Megaruptor (Diagenode) and size selected for >6-kbp fragments with a BluePippin and 0.75% Gel Cassette (programme: 0.75% DF Marker S1 High-Pass 6–10 kb vs3; Sage Science). Each pool was loaded onto a SMRTcell and sequenced on a Pacific Biosystems RSII machine with DNA-Sequencing Kit 4.0 v2, MagBeadBuffer Kit v2, MagBead Binding Buffer Kit v2 and DNA Polymerase Kit P6v2. The sequence output of these eight runs had an average read length of 7.8–9.7 kb with a minimum yield of 750 kbp per SMRTcell. The raw read data were subjected to quality control, processed into circular consensus sequences and subreads, and exported as FASTQ files via the SMRTportal (provided by Pacific Biosciences).
MAG assembly and annotation
MAGs were constructed as follows (outlined in Extended Data Fig. 1a). Pacific Biosystems circular consensus sequences and subreads were used as is, while Illumina shotgun reads were filtered for low read quality, adaptors and host contamination (Metagenomic Sequencing). A full cohort assembly was done in metaSPAdes (SPAdes version 3.13.1) in hybrid mode with k-mer sizes of 21, 33, 55 and 77. Concatenated, quality-controlled, forward and reverse Illumina short read files of all samples were used as input. In addition, the assembly software was informed with the eight Pacific Biosystems long read banks (hybrid mode) in the form of filtered subreads and circular consensus sequences.
The resulting scaffolds were filtered for a minimum length of 1,000 bp and coverage ≥7.7815. The cut-offs were determined by scatter plotting coverage versus length, as described in ref. 84. The quality-controlled metagenomic reads were mapped back to the filtered scaffolds with Bowtie (version 2.2.5); the insert size was 0–1,000 bp in the very sensitive, non-deterministic, ‘fr’ stranded mode with end-to-end alignment. Non-unique mappings and unaligned reads were discarded. The scaffold coverage depth was determined with the jgi_summarize_bam_contig_depths script from MetaBAT (version 2.12.1). This coverage depth information was then used to sort the remaining scaffolds into bins, each representing single bacterial genomes, with the binning tools MetaBAT (version 2.12.1), CONCOCT (version 1.1.0) and MaxBin (version 2.2.4). For CONCOCT, the scaffolds were broken up into 10-kbp chunks. Bin refinement was conducted with the combined results of all three binners (252 bins) with DASTool (version 1.1.2); subsequently, quality metrics were calculated by CheckM (version 1.1.2). Bins with a quality estimate of >80% and a contamination estimate of <10% were considered for further analysis and are henceforth referred to as MAGs. In our reporting of medium- and high-quality MAG drafts, we referred to the standards and metrics laid out by The Genome Standards Consortium23. Accordingly, a MAG will be considered high quality with a completion >90%, contamination <5% and whether genes for 23S, 16S and 5S rRNA and at least 18 tRNAs are recovered. While 133 of our 181 MAGs fulfil these very strict completion and contamination cut-offs, only 25 of those 133 could be considered true high-quality MAGs only due to some missing rRNA or tRNA genes. The lack of those genes, however, does not impact the quality of our MAG-derived metabolic models. Only 18 of our MAGs showed a contamination score greater than 5%, ranging from 5.1% to 9.8% with a mean of 6.7%, and only 2 of those 18 had a completeness score <90% (Supplementary Table 1.2). We used slightly less strict cut-offs for contamination and completeness to include a larger variety of MAGs in our study. While the metabolic model construction from MAGs can partially compensate for lack of completeness via gap filling and for contamination by pathway-completeness checks, our more loose contamination cut-offs might reduce the accuracy of taxonomic assignments.
The 181 final MAGs were taxonomically annotated with GTDB-Tk (version 2.1.1) and database version r214. The tRNA genes were characterized using tRNAscan-SE (version 2.0.9). The 16S rRNA genes were detected by barrnap (version 0.9) in the ‘kingdom bacteria’ mode. A phylogenetic tree of the 181 MAGs (Fig. 1a) was created from a multiple sequence alignment created by GTDB-Tk (align/gtdbtk.bac120.user_msa.fasta.gz) with the European Bioinformatics Institute’s online Simple Phylogeny tool (ClustalW version 2.1) and visualized with R statistical software. The complete characterization of the MAGs is provided in Supplementary Table 1.2.
For association with age, MAG abundances were calculated via the mean of the scaffold coverage depths across all scaffolds belonging to a MAG, normalized by total sample abundance and then correlated with age in linear models for each MAG across all samples. The P values were corrected for multiple testing with the Benjamini–Hochberg FDR method. Significant age-associated MAGs (FDR-adjusted P value ≤ 0.05) were plotted (Fig. 4a).
Microbiome metabolic model construction
Metabolic models were constructed for each of the 181 mouse gut bacteria inferred from our MAGs in samples from the 52 mice. The reconstruction was performed in gapseq (version 1.2) with default settings including gap filling of models (git commit: 159ad378; sequence DB md5sum: bf8ba98)24. Gap filled reactions for each model are indicated in the reconstructed models in the Zenodo archive. The nutritional input for the computational models was designed according to the fortified rat and mouse diet (V1534-300; ssniff Spezialdiäten). The diet was reconstructed according to the vendor’s information on its molecular constituents translated into the corresponding metabolites in the models, following the protocol described in ref. 85. We assumed an average daily uptake of 3.5 g of food based on reference values86. This amount was used to transform the percentages into grams and then millimoles (millimoles per day). Limited information was reported on fibre in the mouse diet; therefore, their values were imputed from the consumed quantities of cereal and grain products of a German human cohort17. Because the simulations depicted the intestinal setting, the absorption in the small intestine was considered when calculating the dietary input (see Supplementary Tables 1.3–1.11 for the respective calculations and references).
Growth rate prediction from metagenomic data
To further validate the model growth rates, CoPTR87 was used to estimate growth rates from the MAGs in each sample. This method uses the peak-to-trough ratio (PTR) (that is, the ratio of sequencing coverage near the replication origin and the replication terminus) to estimate the growth of a MAG in a sample88. We first indexed the MAGs with the command ‘coptr index –bt2-threads’. Next, using this index, we mapped our quality-controlled metagenomic reads against our 181 MAGs with the command ‘coptr map –threads 4 –paired’. Then, read positions were extracted with the command ‘coptr extract’, and the PTR was estimated with the command ‘coptr estimate’. Default parameters were used for all commands except ‘coptr index’ and ‘coptr map’ for which the number of threads was specified. In addition, ‘–paired’ was set for the ‘coptr map’ command to inform the software about the use of paired-end reads. Community growth was determined for each mouse’s microbiome community by calculating the median growth rate across all MAGs in its sample. We did not weight growth rate predictions by individual species’ abundances to obtain a community-level growth rate. Thereby, we avoided spurious correlations with community growth rates predicted using community FBA as community FBA explicitly incorporates abundance information. Please note that while a previous study found little correlation between PTR estimates and experimentally measured growth rates89, this study did not include CoPTR in the benchmark and CoPTR itself was explicitly validated on MAGs.
Modelling of ecological relationships within the microbiome
The ecological relationships for each pair of bacteria across all species were predicted. To this end, the growth achieved by a single bacterium was compared with that achieved when each bacterium was co-grown with other bacteria. The relationships were characterized using the ecological relationships described in a previous study (Fig. 1 in ref. 90) as a reference. Growth was estimated by FBA for single growth and community FBA for combined growth. To achieve this, we used the R packages sybil91 and MicrobiomeGS2 (www.github.com/Waschina/MicrobiomeGS2) and the linear programming solver IBM ILOG CPLEX 22.10. The six types of ecological relationships and their frequencies among each microbial community were inferred with the R EcoGS package (https://github.com/maringos/EcoGS). To this end, we considered for each microbiome each potential pair of species. The type of ecological interaction between the pair was determined by comparing individual growth rates with growth rates when both species were combined. Summing pairwise frequencies for each type of inferred interaction, we then obtained the frequency of an interaction in each community. To obtain relative frequencies, the abundance of ecological relations was normalized sample-wise to a sum of 1. Next, a linear model analysis of each ecological interaction type with age was conducted and P values were adjusted for multiple testing using the Benjamini–Hochberg FDR method.
Host–microbiome partial correlations
The transcriptomic data were normalized separately for each organ (colon, liver and brain) using variance-stabilizing transformation informed with age and sequencing batch (blind = FALSE) implemented in the R package DESeq2 (version 1.40.2)92. A near-zero variance filter was also applied using the nearZeroVar function of the R package caret (version 6.0-94). The active reactions of each mouse’s microbiome community were predicted as described in ‘Estimation of Functional Capacity of Microbiomes’. The host transcript abundances were correlated pairwise with microbiome active reactions (each transcript with each reaction), correcting for age and sequencing batch (only for the liver and brain), with Spearman’s partial correlations (implemented in the R package ppcor (version 1.0)93). To balance stringent false discovery cut-offs with reasonable result counts, strong correlations with a Benjamini–Hochberg FDR-corrected94 P ≤ 0.1 and Spearman’s ρ ≥ 0.55 were considered for downstream analysis. Correlated feature pairs were obtained for the colon (n = 12,732), liver (n = 3,425) and brain (n = 2,499). They consisted of n unique features for the colon (microbiome, n = 1,606; host, n = 2,815), liver (microbiome, n = 1,359; host, n = 1,277) and brain (microbiome, n = 1,236; host, n = 926). The strong correlations were stratified into either positive or negative correlations according to their correlation values and then annotated with GO biological processes25 (host transcripts) or MetaCyc Pathways26 (microbiome reactions) using hypergeometric over-representation tests with the phyper function of the R stats package (version 4.3.2; x = ‘correlated features enriched for the term’ − 1, m = ‘the total of all correlated features,’ n = ‘all features’ – ‘correlated features’ and k = ‘the total of the features in the term’). Enriched terms (pathways and processes) with at least three features and an FDR-corrected over-representation P ≤ 0.05 were reported (Supplementary Tables 2.1–2.3 and Fig. 2). After enrichment, we obtained n process pairs for the colon (n = 1,377), liver (n = 283) and brain (n = 167), as shown in Fig. 2 and Supplementary Tables 2.1–2.3. The negative decadic logarithm of the over-representation FDR P values was calculated and reported as is for positive correlations and multiplied by −1 for negative correlations. Only process pairs that were associated with at least two other pathways were plotted (Fig. 2) and filtered to highlight the most significant enrichments with FDR P value cut-offs of ≤1 × 10−10 for the colon (Fig. 2a), ≤1 × 10−4 for the liver (Fig. 2b) and ≤1 × 10−3 for the brain (Fig. 2c).
For a broader overview of host–microbiome associations, the GO biological processes were grouped by their higher-ranking level 2 GO biological process, and the MetaCyc pathways were grouped by their respective highest-level superpathways (see Supplementary Tables 2.5 and 2.6 for the process and pathway groups). The level 2 GO biological process groups were cellular process, metabolic process, biological regulation, localization, developmental process, response to stimulus, immune system process, multicellular organismal process, viral process, reproduction, homeostatic process and growth. The MetaCyc microbial superpathways were lipids, carbohydrates, utilization, energy metabolism, nucleotides, secondary metabolites, amino acids, other, signalling, carboxylates, cofactors, carriers, metabolic regulators, c1 compounds, electron transfer, noncarbon nutrients, cell structure, biosynthesis, detoxification, interconversion, glycans, tRNA and bioluminescence. The −log10(FDR-corrected P values) were summed for each level 2 GO and MetaCyc superpathway pair. The values are plotted in Extended Data Fig. 2a and listed in Supplementary Table 2.4.
The pairwise correlations between all host features and all microbiome features were repeated, stratified by organ and age group. Thus, the ratio of significant host–microbiome correlations to all tested pairs was obtained for each organ and age group. These ratios were compared using Pearson’s chi-squared test with Yates’ continuity correction and Bonferroni’s multiple testing correction to identify significant differences between consecutive age groups (Extended Data Fig. 6a–e).
The overlaps of aging-regulated and microbiome-associated transcripts between the three studied organs were determined to identify shared aging-regulated and microbiome-regulated host transcripts. The overlap between microbiome-associated and aging-associated transcripts was statistically evaluated using hypergeometric over-representation tests separately for each organ. The numbers of shared transcripts were plotted (Fig. 6a) for each possible combination for the colon (age associated, n = 4,715; microbiome associated, n = 2,815; shared, n = 589; hypergeometric P = 4.9 × 10−15), liver (age associated, n = 8,285; microbiome associated, n = 1,277; shared, n = 485; hypergeometric P = 1.4 × 10−6) and brain (age associated, n = 6,505; microbiome associated, n = 926; shared, n = 236; hypergeometric P = 3.7 × 10−5). Reported P values from hypergeometric tests were corrected for multiple testing via Benjamini and Hochberg’s method.
Validation of microbiome-associated host genes
Differentially expressed genes and transcripts were derived from the GF validation cohort as described in the Supplementary Methods section ‘Differential Gene Expression Analysis’. We identified the overlaps of microbiome-associated transcripts, identified from our main study cohort via partial correlations (see Methods section ‘Host–Microbiome Partial Correlations’), with the differentially expressed transcripts from our GF validation cohort in both treatment contrasts (GF versus CONVR and GF versus CONVD). The number of shared microbiome-associated transcripts between both cohorts was statistically evaluated using hypergeometric over-representation tests stratified by organ and treatment contrast. The numbers of shared transcripts were plotted (Fig. 2d) for each possible combination, for the colon (main cohort, n = 2,815; GF cohort versus CONVR, n = 2,703; shared, n = 438; hypergeometric P = 1.1 × 10−32 and GF cohort versus CONVD, n = 7,637; shared, n = 1,207; hypergeometric P = 7.2 × 10−102), for the liver (main cohort, n = 1,277; GF cohort versus CONVR, n = 3,281; shared, n = 223; hypergeometric P = 2.7 × 10−7 and GF cohort versus CONVD, n = 351; shared, n = 24; hypergeometric P = 7.9 × 10−2) and for the brain (main cohort, n = 926; GF cohort versus CONVR, n = 0; and GF cohort versus CONVD, n = 554; shared, n = 20; hypergeometric P = 1.7 × 10−1). Reported P values from hypergeometric tests were corrected for multiple testing via Benjamini and Hochberg’s method.
Reconstruction of the generic metamodel
A two-step procedure was followed to obtain a metamodel for each mouse. In the first step, a generic metamodel representing the individual organs and the microbiome was assembled. In the second step, a specific metamodel of each mouse was derived by integrating expression and metagenomic data.
In the first step, we joined three times the human metabolic reconstruction Recon 2.2 (ref. 28) representing the individual organs with a microbiome metabolic model according to their physiological interactions (Fig. 3a). A mouse-specific metabolic reconstruction was not used, as the human reconstructions are by far the best curated and there is a high overlap in metabolic content between mice and humans95. All compartments interfaced with each other via common exchange environments, such as the gut lumen (microbiome and colon) and the bloodstream (colon, brain and liver). Some exchanges along the bloodstream were defined as directional, following the physiological interactions of the organs (see Fig. 3a). Metabolite uptake into the brain was restricted to metabolites known to cross the blood–brain barrier (see Supplementary Table 3.7 for a list). To compile this list of compounds, literature resources46,96,97 were used; in addition, we selected the compounds in Recon 2.2 (ref. 28), along with those identified on the Virtual Metabolic Human website (www.vmh.life) whose physicochemical properties would allow them to cross the blood–brain barrier98. For the microbiome metabolic model, all individually reconstructed MAGs were merged into a single model by combining all microbial reactions of the individual bacterial cellular compartments into a single reaction space. This merged microbiome model could then interact with the human metabolic models via the lumen exchange environment. We decided to use a merged microbiome model instead of species-level metabolic reconstructions to maintain computational tractability of the metamodel for comprehensive downstream analysis (for example, flux variability analysis and EFM sampling).
To better account for organ- and microbiome-specific uptake and secretion of metabolites, exchange reactions of the individual compartments were split into irreversible forwards and backwards directions. To model the dietary uptake of the mice, the molar concentrations of all metabolites in their diet were derived and represented in the model following an established protocol85. In addition, information on the absorption of dietary metabolites before entry into the colon was obtained to differentiate between ileal and colonic uptake. The diet was integrated into the model by a direct inflow of absorbed compounds to the bloodstream and unabsorbed compounds to the colonic lumen for microbiome and colonic use.
Following the merging of the human and bacterial metabolic models, several energy-generating cycles (that is, sets of metabolic reactions that can form ATP from ADP without the consumption of other metabolites) were identified and resolved by correcting potential problems in the reversibility of participating reactions (Supplementary Table 3.6). The metamodel can be found under accession MODEL2310020001 in the EBI BioModels database (https://www.ebi.ac.uk/biomodels/)99.
Reconstruction of mouse context-specific metamodels
In the second step, we built a context-specific metamodel for each mouse. To achieve this, StanDep100 was applied to the transcriptomic and metagenomic data to derive the core reactions for each tissue required to reconstruct context-specific models using fastcore29. Transcriptomic data were preprocessed by transforming counts into fragments per kilobase of transcript per million mapped reads (FPKMs). After removing genes with at least one sample with zero detected expression or a mean FPKM < 0.1 and log2 transformation, FPKM values were normalized using Combat101 and then transformed back to their original scale. To identify core reactions, mouse genes were mapped to their corresponding human orthologues using Ensembl Biomart102. The gene expression data of all tissues were combined into a single matrix, and tissue and age groups were used as separating categories for StanDep. StanDep was applied with ‘chi2dist’ as the distance method and ‘complete’ as the linkage method. After screening optimal cluster numbers, predicted core reactions remained stable when using 39 clusters (that is, the Jaccard distance of derived core reactions for StanDep runs with increasing cluster numbers was below 0.05 using at least 39 clusters).
For metagenomic data, we obtained a reaction abundance matrix for each microbiome sample. To this end, reads were mapped to MAGs to derive species-level counts. These were then multiplied with a reaction contribution matrix indicating for each reaction in which species they are present (normalized to a sum of one for each species) and normalizing to a sum of one across all reactions in a sample. Reaction abundances were used as input to StanDep with the age group of the sample as the separating factor, ‘chi2dist’ as the distance method and ‘complete’ as the linkage method. Following the same procedure as for the gene activity data, 15 clusters were identified as optimal for reaction abundance data.
In addition, metabolic exchanges between individual organs and the bloodstream previously measured in pigs were included103 by mapping IDs of exchanged metabolites to the corresponding metabolite identifiers in Recon 2.2. If an organ took up or secreted a metabolite, the corresponding uptake or secretion reactions were added to the core reactions. If the kidney took up a metabolite, the corresponding outflow reaction from the blood was added to the core reactions because the kidney was not modelled explicitly. Subsequently, the core reactions for each sample and the generic metamodel were used as input for fastcore to derive a context-specific metamodel for each mouse. To run fastcore, CORPSE (https://github.com/Porthmeus/CORPSE) was used as an interface to the corresponding functions of the TROPPO toolbox104.
Host and microbiome dependence of reactions
To determine microbiome- or host-dependent functions, flux variability analysis with and without the microbiota were conducted. FVA was performed105 by maximizing and minimizing flux through each reaction using the ‘flux_variability_analysis()’ function of CobraPy106 without optimization of growth (fraction_of_optimum = 0) as for most tissues in mammals there is only negligible cellular replication107. Because internal exchange reactions were split into irreversible forwards and backwards steps, they were treated separately by always blocking the corresponding opposing direction. FVA results were summarized by determining admissible flux ranges, by subtracting minimal from maximal flux. Microbiome-dependent exchange reactions in the host were identified by repeating the FVA but blocking each microbiome reaction. An exchange reaction was deemed microbiome dependent if its flux range was reduced to less than 10% when blocking microbiome reactions. To elucidate the metabolites exchanged between the host and the microbiota, the microbiota-dependent uptake and secretion reactions of metabolites for a given organ were counted. If the number of cases of microbiome-dependent secretion subtracted from the frequency of microbiome-dependent uptake was larger than 10, a metabolite was classified as being provided by microbiota to the host or vice versa (20% of samples). For plotting (Fig. 3b), only metabolites with a difference of at least 35 for colon and a difference of at least 12 for brain and liver were shown.
Identification of reaction-level host–microbiome dependencies
To determine the dependencies of individual host reactions on individual microbial reactions, EFMSampler32 was used to sample EFMs with each host and microbial reaction as indicator reaction for sampling. The indicator reaction is used to define the specific reaction in a model through which EFMs should be determined. For each target reaction, EFMSampler was run in eight parallel threads using flux minimization as objective until either 10,000 EFMs were sampled or >200 s had elapsed and the average frequency of occurrence of reactions in EFMs was recorded. Subsequently, occurrence frequencies were averaged across all 52 mice. This yielded a matrix in which each column corresponded to a target reaction and each row indicated the frequency at which all other reactions occurred in EFMs containing that reaction. Thus, a value of ‘1’ indicates microbiome reactions that always co-occur in EFMs of the target reaction and a value of ‘0’ no co-occurrence
To compare EFM-predicted interactions to host–microbiome correlations, scores in the interaction matrix were compared between genes and microbiome reactions with significant associations. To this end, for each significant host gene–microbiome reaction association (FDR-adjusted P ≤ 0.1), the maximum interaction score in the sub-matrix containing reactions associated with the host gene and the microbiome reaction were determined and collected across all significant host gene–microbiome reaction associations in a tissue to derive a set of ‘true’ maximal interaction scores. The same analysis was performed 100 times for randomly drawn genes associated with reactions present in the tissue and randomly selected microbiome reactions to obtain ‘random’ maximal interaction scores. Then, true and randomly generated maximal interaction scores were compared using the Wilcoxon rank-sum test.
To analyse the most strongly interacting metabolic processes between the host and microbiome, an interaction was assumed if the microbiome reaction occurred in at least 50% of the EFMs sampled from that host reaction across all metamodels. Then, for each host–microbiome reaction pair, we determined which metabolic subsystems they were associated with and counted each corresponding host–microbiome subsystem pair across all such pairs in the interaction matrix. The enrichment of pairs was then tested using Fisher’s exact test comparing for each pair the number of mutual interactions of reactions belonging to the host and microbiome subsystems to the frequency of interactions across the entire interaction matrix. An enrichment was assumed with an FDR-corrected P ≤ 0.05, calculated using the p.adjust function in R.
Identification of aging-regulated metabolic modules
To identify aging-regulated metabolic modules, we defined sets of reactions associated with each indicator reaction used for EFM sampling. A reaction was assumed to belong to the metabolic modules of an indicator reaction if it occurred in at least 20% of the EFMs sampled for that indicator reaction. Unlike in the analysis of reaction-level dependencies between host and microbiota, we considered both the host and microbiome components of the EFMs; thus, metabolic modules contained both host and microbiome reactions. A metabolic module was considered dependent on the microbiome if it contained at least 20 microbial reactions. To identify aging-regulated metabolic modules, aging-induced and aging-repressed genes (Supplementary Tables 5.1–5.3) were translated into the reactions with which they were associated in the metabolic model. Then, for each metabolic module, we tested whether the corresponding set of reactions was enriched for aging-induced or aging-repressed metabolic reactions using Fisher’s exact test, assuming the entire set of reactions occurring in a tissue as background. Subsequently, we determined host pathways in which indicator reactions of aging-regulated modules dependent on the microbiota were enriched. To this end, we filtered microbiome-dependent aging-regulated modules with an enrichment of aging-regulated reactions with P ≤ 0.01. For each module, we determined the corresponding indicator reactions and performed a pathway enrichment based on the subsystem annotation of the Recon 2.2 model using Fisher’s exact test. For transport reactions, we also added a subsystem annotation for the transport of nucleotides (encompassing deoxyribonucleic and ribonucleic acids) and amino acids. All reactions of a tissue occurring in at least one metamodel were used as background or universe in the Fisher’s exact test of that tissue.
Materials availability
This study did not generate new unique reagents.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Supplementary information
Supplementary results, methods and references.
All data tables related to the results and figures in the main text combined into one Excel file.
Acknowledgements
We acknowledge funding by the German Research Foundation to C.K. within the scope of CRC1182 (project A1.5), the Research Group miTarget (FOR5042) and the Cluster of Excellence ‘Precision Medicine in Chronic Inflammation’ (EXC2167). We also acknowledge funding from the German Research Foundation (DFG, project number: 416 418087534) to C.K. and C.F., project SO1141/10-1 to F.S., intramural grants of the medical faculty of Kiel University (grants nos K126408 and K126493) to F.S., the Carl Zeiss Foundation IMPULS programme (project number: P2019-01-006) to C.F. and the European Union’s Horizon 2020 Research and Innovation Programme (under the Marie Sklodowska-Curie grant: agreement number 859890 (SmartAge)) to O.W.W., C.K. and C.F. P.R. was supported by BMBF iTREAT, DFG SFB1182 (C2), RU miTARGET (P3) and EXC 2167 RTFVI and TI1. This work was supported by the DFG Research Infrastructure NGS_CC (project number: 407495230) as part of the Next Generation Sequencing Competence Network (project number: 423957469). Short-read sequencing was conducted at the Competence Centre for Genomic Analysis (Kiel, Germany). We thank K. Cloppenborg-Schmidt for the excellent technical assistance in preparing the samples for metagenomic sequencing. We thank L. Mannerås Holm and F. Bäckhed (University of Gothenburg) for providing tissue samples from wild-type, GF and conventionalized C57BL/6J mice.
Extended data
Author contributions
Conceptualization: C.F., C.K., D.E., L.B., M.H., O.W.W. and S.S. Methodology: A.S.K., A.W., C.F., C.K., D.E., G.M., J.F.B., J.Z., L.B., M.G., M.H., F.S., P.S.-K., R.H., R.S., S. Flor, A.M.G., S.K., S.W. and T.D. Software: A.S.K., C.K., G.M., J.T., J.Z., L.B., S. Flor, S.W. and T.D. Investigation: A.W., C.F., C.K., D.E., L.B., M.G., M.H., P.S.-K., R.H., R.S., S. Franzenburg, F.S. and S.K. Formal analysis: A.S.K., A.W., C.K., G.M., J.Z., L.B., S. Flor, A.M.G., S.W. and T.D. Writing, original draft: A.S.K., A.W., C.K., G.M., J.Z., L.B., S. Flor and T.D. Writing, review and editing: C.F., J.F.B., J.T and F.S. Visualization: C.K. and L.B. Supervision: C.F., C.K., O.W.W., P.S.-K. and S.W. Funding acquisition: C.F., C.K., J.F.B., P.R., F.S. and O.W.W. Project administration: C.F., C.K. and L.B. The order of those authors who contributed equally was determined alphabetically. To accurately represent their equal contributions in any type of professional or academic documentation, they are permitted to rearrange the order of their names among the shared equally contributed positions at their discretion.
Peer review
Peer review information
Nature Microbiology thanks Sean Gibbons and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Funding
Open access funding provided by Christian-Albrechts-Universität zu Kiel.
Data availability
Metagenomic raw read and MAG assembly data were deposited in the European Nucleotide Archive (ENA) under BioProject PRJEB73981 (ebi.ac.uk/ena/browser/view/PRJEB73981). Individual accession numbers for each MAG are listed in Supplementary Table 1.2. Gene expression data were published in the GEO database under record GSE262290 (ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE262290) and record GSE278548 (ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE278548). Metabolomics data have been made available at the MassIVE database (massive.ucsd.edu) with identifiers MSV000094409, MSV000094410 and MSV000094436. The metamodel can be found under accession MODEL2310020001 in the BioModels database (ebi.ac.uk/biomodels/MODEL2310020001)99. Detailed sample metadata, the microbial metabolic models and supplementary resources are available via Zenodo at 10.5281/zenodo.10844502 (ref. 108).
Code availability
The source code used for data analysis is available via GitHub at github.com/sciwitch/MouseMicrobiomeAging.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Thomas Dost, Daniela Esser, Stefano Flor, Andy Mercado Gamarra, Madlen Haase, A. Samer Kadibalban, Georgios Marinos, Alesia Walker, Johannes Zimmermann.
These authors jointly supervised this work: Christiane Frahm, Christoph Kaleta.
Extended data
is available for this paper at 10.1038/s41564-025-01959-z.
Supplementary information
The online version contains supplementary material available at 10.1038/s41564-025-01959-z.
References
- 1.Lopez, A. D., Mathers, C. D., Ezzati, M., Jamison, D. T. & Murray, C. J. L. Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data. Lancet367, 1747–1757 (2006). [DOI] [PubMed] [Google Scholar]
- 2.López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell186, 243–278 (2023). [DOI] [PubMed] [Google Scholar]
- 3.Aramillo Irizar, P. et al. Transcriptomic alterations during ageing reflect the shift from cancer to degenerative diseases in the elderly. Nat. Commun.9, 327 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhou, Y., Hu, G. & Wang, M. C. Host and microbiota metabolic signals in aging and longevity. Nat. Chem. Biol.17, 1027–1036 (2021). [DOI] [PubMed] [Google Scholar]
- 5.Wilmanski, T. et al. Gut microbiome pattern reflects healthy ageing and predicts survival in humans. Nat. Metab.3, 274–286 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kong, F. et al. Gut microbiota signatures of longevity. Curr. Biol.26, R832–R833 (2016). [DOI] [PubMed] [Google Scholar]
- 7.Wilmanski, T., Gibbons, S. M. & Price, N. D. Healthy aging and the human gut microbiome: why we cannot just turn back the clock. Nat. Aging2, 869–871 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bhutto, A. & Morley, J. E. The clinical significance of gastrointestinal changes with aging. Curr. Opin. Clin. Nutr. Metab. Care11, 651–660 (2008). [DOI] [PubMed] [Google Scholar]
- 9.Kundu, P. et al. Neurogenesis and prolongevity signaling in young germ-free mice transplanted with the gut microbiota of old mice. Sci. Transl. Med.11, eaau4760 (2019). [DOI] [PubMed] [Google Scholar]
- 10.Thevaranjan, N. et al. Age-associated microbial dysbiosis promotes intestinal permeability, systemic inflammation, and macrophage dysfunction. Cell Host Microbe21, 455–466.e4 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guedj, A. et al. Gut microbiota shape ‘inflamm-ageing’ cytokines and account for age-dependent decline in DNA damage repair. Gut69, 1064–1075 (2020). [DOI] [PubMed] [Google Scholar]
- 12.Nagpal, R. et al. Gut microbiome and aging: physiological and mechanistic insights. Nutr. Healthy Aging4, 267–285 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gilbert, J. A. et al. Current understanding of the human microbiome. Nat. Med.24, 392–400 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Falony, G. et al. Population-level analysis of gut microbiome variation. Science352, 560–564 (2016). [DOI] [PubMed] [Google Scholar]
- 15.Krautkramer, K. A., Fan, J. & Bäckhed, F. Gut microbial metabolites as multi-kingdom intermediates. Nat. Rev. Microbiol.19, 77–94 (2021). [DOI] [PubMed] [Google Scholar]
- 16.Heinken, A., Basile, A. & Thiele, I. Advances in constraint-based modelling of microbial communities. Curr. Opin. Syst. Biol.27, 100346 (2021). [Google Scholar]
- 17.Pryor, R. et al. Host–microbe–drug–nutrient screen identifies bacterial effectors of metformin therapy. Cell178, 1299–1312.e29 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Passi, A. et al. Genome-scale metabolic modeling enables in-depth understanding of big data. Metabolites12, 14 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hertel, J. et al. Integrated analyses of microbiome and longitudinal metabolome data reveal microbial–host interactions on sulfur metabolism in Parkinson’s disease. Cell Rep.29, 1767–1777.e8 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Aden, K. et al. Metabolic functions of gut microbes associate with efficacy of tumor necrosis factor antagonists in patients with inflammatory bowel diseases. Gastroenterology157, 1279–1292.e11 (2019). [DOI] [PubMed] [Google Scholar]
- 21.Frahm, C. et al. Transcriptional profiling reveals protective mechanisms in brains of long-lived mice. Neurobiol. Aging52, 23–31 (2017). [DOI] [PubMed] [Google Scholar]
- 22.Chaumeil, P.-A., Mussig, A. J., Hugenholtz, P. & Parks, D. H. GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics36, 1925–1927 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bowers, R. M. et al. Minimum information about a single amplified genome (MISAG) and a metagenome-assembled genome (MIMAG) of bacteria and archaea. Nat. Biotechnol.35, 725–731 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zimmermann, J., Kaleta, C. & Waschina, S. gapseq: informed prediction of bacterial metabolic pathways and reconstruction of accurate metabolic models. Genome Biol.22, 81 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.The Gene Ontology Consortium. et al. The Gene Ontology knowledgebase in 2023. Genetics224, iyad031 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caspi, R. et al. The MetaCyc database of metabolic pathways and enzymes—a 2019 update. Nucleic Acids Res.48, D445–D453 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Beghini, F. et al. Integrating taxonomic, functional, and strain-level profiling of diverse microbial communities with bioBakery 3. eLife10, e65088 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Swainston, N. et al. Recon 2.2: from reconstruction to model of human metabolism. Metabolomics12, 109 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pacheco, M. P. & Sauter, T. The FASTCORE family: for the fast reconstruction of compact context-specific metabolic networks models. Methods Mol. Biol.1716, 101–110 (2018). [DOI] [PubMed] [Google Scholar]
- 30.Leal, J., Smyth, H. D. C. & Ghosh, D. Physicochemical properties of mucus and their impact on transmucosal drug delivery. Int. J. Pharm.532, 555–572 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schuster, S., Dandekar, T. & Fell, D. A. Detection of elementary flux modes in biochemical networks: a promising tool for pathway analysis and metabolic engineering. Trends Biotechnol.17, 53–60 (1999). [DOI] [PubMed] [Google Scholar]
- 32.Bohl, K. et al. CASOP GS: computing intervention strategies targeted at production improvement in genome-scale metabolic networks. In Lecture Notes in Informatics (eds Schomburg, D. & Grote, A.) 71–80 (German Conference on Bioinformatics, 2010).
- 33.N, B. et al. A reference map of potential determinants for the human serum metabolome. Nature588, 135–140 (2020). [DOI] [PubMed] [Google Scholar]
- 34.Wu, C.-S. et al. Age-dependent remodeling of gut microbiome and host serum metabolome in mice. Aging13, 6330–6345 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lee, J. et al. Gut microbiota-derived short-chain fatty acids promote poststroke recovery in aged mice. Circ. Res.127, 453–465 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Macias-Ceja, D. C. et al. Succinate receptor mediates intestinal inflammation and fibrosis. Mucosal Immunol.12, 178–187 (2019). [DOI] [PubMed] [Google Scholar]
- 37.Fierer, N. Embracing the unknown: disentangling the complexities of the soil microbiome. Nat. Rev. Microbiol.15, 579–590 (2017). [DOI] [PubMed] [Google Scholar]
- 38.Zimmermann, J. et al. The functional repertoire contained within the native microbiota of the model nematode Caenorhabditis elegans. ISME J.14, 26–38 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Perino, A., Demagny, H., Velazquez-Villegas, L. & Schoonjans, K. Molecular physiology of bile acid signaling in health, disease, and aging. Physiol. Rev.101, 683–731 (2021). [DOI] [PubMed] [Google Scholar]
- 40.Chaudhari, S. N. et al. A microbial metabolite remodels the gut–liver axis following bariatric surgery. Cell Host Microbe29, 408–424.e7 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ding, J. et al. A metabolome atlas of the aging mouse brain. Nat. Commun.12, 6021 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ho, S.-C., Liu, J.-H. & Wu, R.-Y. Establishment of the mimetic aging effect in mice caused by d-galactose. Biogerontology4, 15–18 (2003). [DOI] [PubMed] [Google Scholar]
- 43.Ananieva, E. A., Powell, J. D. & Hutson, S. M. Leucine metabolism in T cell activation: mTOR signaling and beyond. Adv. Nutr.7, 798S–805S (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Donohoe, D. R. et al. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab.13, 517–526 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lee, J. S. et al. Microbiota-sourced purines support wound healing and mucous barrier function. iScience23, 101226 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thiele, I. et al. Personalized whole-body models integrate metabolism, physiology, and the gut microbiome. Mol. Syst. Biol.16, e8982 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vieira-Silva, S. et al. Species–function relationships shape ecological properties of the human gut microbiome. Nat. Microbiol.1, 16088 (2016). [DOI] [PubMed] [Google Scholar]
- 48.Tian, L. et al. Deciphering functional redundancy in the human microbiome. Nat. Commun.11, 6217 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tsoi, R. et al. Metabolic division of labor in microbial systems. Proc. Natl Acad. Sci. USA115, 2526–2531 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kolodny, O. & Schulenburg, H. Microbiome-mediated plasticity directs host evolution along several distinct time scales. Philos. Trans. R. Soc. Lond. B375, 20190589 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hammer, T. J. Why do hosts malfunction without microbes? Missing benefits versus evolutionary addiction. Trends Microbiol.10.1016/j.tim.2023.07.012 (2023). [DOI] [PubMed]
- 52.Kirschner, S. K. et al. Short-chain fatty acid production in accessible and inaccessible body pools as assessed by novel stable tracer pulse approach is reduced by aging independent of presence of COPD. Metabolism141, 155399 (2023). [DOI] [PubMed] [Google Scholar]
- 53.Connors, J., Dawe, N. & Van Limbergen, J. The role of succinate in the regulation of intestinal inflammation. Nutrients11, 25 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Serena, C. et al. Elevated circulating levels of succinate in human obesity are linked to specific gut microbiota. ISME J.12, 1642–1657 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Schumacher, B., Pothof, J., Vijg, J. & Hoeijmakers, J. H. J. The central role of DNA damage in the ageing process. Nature592, 695–703 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.van de Ven, R. A. H., Santos, D. & Haigis, M. C. Mitochondrial sirtuins and molecular mechanisms of aging. Trends Mol. Med.23, 320–331 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gallegos-Orozco, J. F., Foxx-Orenstein, A. E., Sterler, S. M. & Stoa, J. M. Chronic constipation in the elderly. Am. J. Gastroenterol.107, 18–25 (2012). [DOI] [PubMed] [Google Scholar]
- 58.Madsen, J. L. Effects of gender, age, and body mass index on gastrointestinal transit times. Dig. Dis. Sci.37, 1548–1553 (1992). [DOI] [PubMed] [Google Scholar]
- 59.Minnebo, Y. et al. Gut microbiota response to in vitro transit time variation is mediated by microbial growth rates, nutrient use efficiency and adaptation to in vivo transit time. Microbiome11, 240 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jian, H. et al. Dietary valine ameliorated gut health and accelerated the development of nonalcoholic fatty liver disease of laying hens. Oxid. Med. Cell. Longev.2021, 4704771 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 61.Zhao, G. et al. Betaine in inflammation: mechanistic aspects and applications. Front. Immunol.9, 1070 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Krause, D. et al. The tryptophan metabolite 3-hydroxyanthranilic acid plays anti-inflammatory and neuroprotective roles during inflammation: role of hemeoxygenase-1. Am. J. Pathol.179, 1360–1372 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Derrien, M. et al. Modulation of mucosal immune response, tolerance, and proliferation in mice colonized by the mucin-degrader Akkermansia muciniphila. Front. Microbiol.2, 166 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Patel, B. A. et al. Impaired colonic motility and reduction in tachykinin signalling in the aged mouse. Exp. Gerontol.53, 24–30 (2014). [DOI] [PubMed] [Google Scholar]
- 65.Branca, J. J. V., Gulisano, M. & Nicoletti, C. Intestinal epithelial barrier functions in ageing. Ageing Res. Rev.54, 100938 (2019). [DOI] [PubMed] [Google Scholar]
- 66.Funk, M. C., Zhou, J. & Boutros, M. Ageing, metabolism and the intestine. EMBO Rep.21, e50047 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nahata, M. et al. Decline in liver mitochondria metabolic function is restored by hochuekkito through sirtuin 1 in aged mice with malnutrition. Front. Physiol.13, 848960 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Häsler, R. et al. Genetic interplay between human longevity and metabolic pathways—a large-scale eQTL study. Aging Cell16, 716–725 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Palmer, A. K. & Jensen, M. D. Metabolic changes in aging humans: current evidence and therapeutic strategies. J. Clin. Invest.132, e158451 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Perruzza, L. et al. T follicular helper cells promote a beneficial gut ecosystem for host metabolic homeostasis by sensing microbiota-derived extracellular ATP. Cell Rep.18, 2566–2575 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mempin, R. et al. Release of extracellular ATP by bacteria during growth. BMC Microbiol.13, 301 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ihssen, J., Jovanovic, N., Sirec, T. & Spitz, U. Real-time monitoring of extracellular ATP in bacterial cultures using thermostable luciferase. PLoS ONE16, e0244200 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lewis, C. V. & Taylor, W. R. Intestinal barrier dysfunction as a therapeutic target for cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol.319, H1227–H1233 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Pellegrini, C. et al. The intestinal barrier in disorders of the central nervous system. Lancet Gastroenterol. Hepatol.8, 66–80 (2023). [DOI] [PubMed] [Google Scholar]
- 75.Pentinmikko, N. et al. Notum produced by Paneth cells attenuates regeneration of aged intestinal epithelium. Nature571, 398–402 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chistiakov, D. A., Sobenin, I. A., Revin, V. V., Orekhov, A. N. & Bobryshev, Y. V. Mitochondrial aging and age-related dysfunction of mitochondria. Biomed Res. Int.2014, 238463 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Rolig, R. L. & McKinnon, P. J. Linking DNA damage and neurodegeneration. Trends Neurosci.23, 417–424 (2000). [DOI] [PubMed] [Google Scholar]
- 78.Feng, M. et al. Gut microbiota may be involved in Alzheimer’s disease pathology by dysregulating pyrimidine metabolism in APP/PS1 mice. Front. Aging Neurosci.14, 967747 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Olecka, M. et al. Nonlinear DNA methylation trajectories in aging male mice. Nat. Commun.15, 3074 (2024). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bresilla, D. et al. The sex-specific metabolic signature of C57BL/6NRj mice during aging. Sci. Rep.12, 21050 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ederer, M.-L. et al. Voluntary wheel running in old C57BL/6 mice reduces age-related inflammation in the colon but not in the brain. Cells11, 566 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Percie du Sert, N. et al. The ARRIVE guidelines 2.0: updated guidelines for reporting animal research. PLoS Biol.18, e3000410 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pan, W.-H. et al. Exposure to the gut microbiota drives distinct methylome and transcriptome changes in intestinal epithelial cells during postnatal development. Genome Med.10, 27 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Douglass, A. P. et al. Coverage-versus-length plots, a simple quality control step for de novo yeast genome sequence assemblies. G39, 879–887 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Marinos, G., Kaleta, C. & Waschina, S. Defining the nutritional input for genome-scale metabolic models: a roadmap. PLoS ONE15, e0236890 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Starr, M. E. & Saito, H. Age-related increase in food spilling by laboratory mice may lead to significant overestimation of actual food consumption: implications for studies on dietary restriction, metabolism, and dose calculations. J. Gerontol. A67, 1043–1048 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Joseph, T. A., Chlenski, P., Litman, A., Korem, T. & Pe’er, I. Accurate and robust inference of microbial growth dynamics from metagenomic sequencing reveals personalized growth rates. Genome Res.32, 558–568 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Korem, T. et al. Growth dynamics of gut microbiota in health and disease inferred from single metagenomic samples. Science349, 1101–1106 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Long, A. M., Hou, S., Ignacio-Espinoza, J. C. & Fuhrman, J. A. Benchmarking microbial growth rate predictions from metagenomes. ISME J.15, 183–195 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zélé, F., Magalhães, S., Kéfi, S. & Duncan, A. B. Ecology and evolution of facilitation among symbionts. Nat. Commun.9, 4869 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gelius-Dietrich, G., Desouki, A. A., Fritzemeier, C. J. & Lercher, M. J. Sybil—efficient constraint-based modelling in R. BMC Syst. Biol.7, 125 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol.15, 550 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kim, S. ppcor: an R package for a fast calculation to semi-partial correlation coefficients. Commun. Stat. Appl. Methods22, 665–674 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B57, 289–300 (1995). [Google Scholar]
- 95.Sigurdsson, M. I., Jamshidi, N., Steingrimsson, E., Thiele, I. & Palsson, B. Ø. A detailed genome-wide reconstruction of mouse metabolism based on human Recon 1. BMC Syst. Biol.4, 140 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Pardridge, W. M. The blood–brain barrier: bottleneck in brain drug development. NeuroRx2, 3–14 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Laffel, L. Ketone bodies: a review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab. Res. Rev.15, 412–426 (1999). [DOI] [PubMed] [Google Scholar]
- 98.Pajouhesh, H. & Lenz, G. R. Medicinal chemical properties of successful central nervous system drugs. NeuroRx2, 541–553 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Malik-Sheriff, R. S. et al. BioModels—15 years of sharing computational models in life science. Nucleic Acids Res.48, D407–D415 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Joshi, C. J. et al. StanDep: capturing transcriptomic variability improves context-specific metabolic models. PLoS Comput. Biol.16, e1007764 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Johnson, W. E., Li, C. & Rabinovic, A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics8, 118–127 (2007). [DOI] [PubMed] [Google Scholar]
- 102.Martin, F. J. et al. Ensembl 2023. Nucleic Acids Res.51, D933–D941 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Jang, C. et al. Metabolite exchange between mammalian organs quantified in pigs. Cell Metab.30, 594–606.e3 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Vieira, V., Ferreira, J. & Rocha, M. A pipeline for the reconstruction and evaluation of context-specific human metabolic models at a large-scale. PLoS Comput. Biol.18, e1009294 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Mahadevan, R. & Schilling, C. H. The effects of alternate optimal solutions in constraint-based genome-scale metabolic models. Metab. Eng.5, 264–276 (2003). [DOI] [PubMed] [Google Scholar]
- 106.Ebrahim, A., Lerman, J. A., Palsson, B. O. & Hyduke, D. R. COBRApy: COnstraints-Based Reconstruction and Analysis for Python. BMC Syst. Biol.7, 74 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Iismaa, S. E. et al. Comparative regenerative mechanisms across different mammalian tissues. NPJ Regen. Med.3, 6 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Best, L. Metabolic modeling reveals the aging-associated decline of host–microbiome metabolic interactions in mice. Zenodo10.5281/zenodo.13899808 (2024). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary results, methods and references.
All data tables related to the results and figures in the main text combined into one Excel file.
Data Availability Statement
Metagenomic raw read and MAG assembly data were deposited in the European Nucleotide Archive (ENA) under BioProject PRJEB73981 (ebi.ac.uk/ena/browser/view/PRJEB73981). Individual accession numbers for each MAG are listed in Supplementary Table 1.2. Gene expression data were published in the GEO database under record GSE262290 (ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE262290) and record GSE278548 (ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE278548). Metabolomics data have been made available at the MassIVE database (massive.ucsd.edu) with identifiers MSV000094409, MSV000094410 and MSV000094436. The metamodel can be found under accession MODEL2310020001 in the BioModels database (ebi.ac.uk/biomodels/MODEL2310020001)99. Detailed sample metadata, the microbial metabolic models and supplementary resources are available via Zenodo at 10.5281/zenodo.10844502 (ref. 108).
The source code used for data analysis is available via GitHub at github.com/sciwitch/MouseMicrobiomeAging.