Abstract
Extracellular vesicles (EVs) facilitate the transfer of biological materials between cells throughout the body. Mitochondria, membrane-bound organelles present in the cytoplasm of nearly all eukaryotic cells, are vital for energy production and cellular homeostasis. Recent studies highlight the critical role of the transport of diverse mitochondrial content, such as mitochondrial DNA (mt-DNA), mitochondrial RNA (mt-RNA), mitochondrial proteins (mt-Prots), and intact mitochondria by small EVs (<200 nm) and large EVs (>200 nm) to recipient cells, where these cargos contribute to cellular and mitochondrial homeostasis. The interplay between EVs and mitochondrial components has significant implications for health, metabolic regulation, and potential as biomarkers. Despite advancements, the mechanisms governing EV-mitochondria crosstalk and the regulatory effect of mitochondrial EVs remain poorly understood. This review explores the roles of EVs and their mitochondrial cargos in health and disease, examines potential mechanisms underlying their interactions, and emphasizes the therapeutic potential of EVs for neurological and systemic conditions associated with mitochondrial dysfunction.
Keywords: Extracellular vesicles, mitochondria, energy metabolism, mitochondrial components
Introduction
Mitochondria play primary roles in energy production and cell function by regulating homeostatic cellular metabolism, particularly for high-energy-demanding neurons.1–3 Dysfunctional mitochondria are hallmarks of various diseases, including neurodegenerative diseases.2,4,5 Mitochondrial dysfunction often precedes and contributes to disease progression, largely via altered mitochondrial membrane potential, generation of reactive oxygen species (ROS), and decreased production of adenosine triphosphate (ATP).5,6 Mitochondria can be transferred between cells under physiological and pathophysiological conditions.1,7,8
Mitochondrial transfer refers to the transfer of intact mitochondria, mitochondrial components such as DNA, RNA, proteins, and fragmented mitochondria.9–12 Transfer can occur via direct mechanisms, including tunneling nanotubes and gap junctions, or via EV-mediated pathways.13,14 Extracellular vesicles (EVs) released by nearly all living cells mediate intercellular communication by transferring their cargo to recipient cells.7,15,16 Emerging studies show that EV-mediated intercellular mitochondrial transfer by delivery of encapsulated mitochondria and its components to recipient cells has a profound impact on cellular function and disease progression.7,17
EV-mediated mitochondrial transfer in the nervous system is a highly cell-type-specific process, serving as an important cellular communication mechanism both within the nervous system and between the nervous system and peripheral organs.1,18,19 This transfer undergoes significant alterations under pathological conditions, reflecting its critical role in disease progression and intercellular signaling.
This review highlights recent advances in EV-mediated mitochondrial transfer and its underlying mechanisms in disease progression and repair, with a primary focus on neurological disorders. The ability of EVs to deliver mitochondrial cargo across cellular barriers and over long distances underscores their pivotal role in maintaining cellular homeostasis, modulating metabolic function, and orchestrating intercellular communication. These processes are particularly critical in the nervous system, where precise and efficient signaling is essential for both normal function and recovery from injury.
Search strategy
The literature search was predominantly conducted through the PubMed database and Google Scholar, encompassing articles published up to October 2024. To enhance search comprehensiveness and accuracy, various text combinations were employed, including terms such as “extracellular vesicles,” “exosomes,” “microvesicles,” and “mitochondria.” The search strategy involved filtering articles pertaining to extracellular vesicles and mitochondria based on their abstracts and results. No restrictions were placed on publication time or language. In this review, we included only studies that adhere to the latest EV research guidelines, the minimal information for studies of extracellular vesicles (MISEV) 2018 and 202317,20 to ensure accuracy and avoid the potential impact of ambiguous research.
Mitochondria and mitochondrial transfer
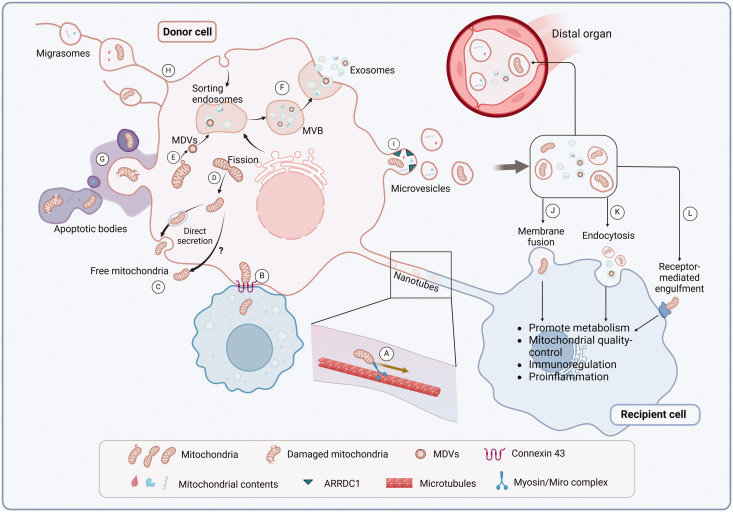
Mitochondria are double-membraned multifunctional organelles that are essential for cellular metabolism across nearly all eukaryotic cells. They are thought to have originated through an ancient endosymbiotic event in which proteobacteria were incorporated into host cells, giving rise to eukaryotic life forms ranging from yeast to plants to animals.3,7,21 While mitochondria are typically inherited vertically during cell division, cells can also export mitochondria to other cells, facilitating intercellular mitochondrial transfer.1,22 This transfer occurs among cells via contact-dependent mechanisms, such as tunneling nanotubes, connexin 43 (Cx43)-mediated gap junction channels, and contact-independent mechanisms, including free mitochondria transfer and EV-mediated transfer.7,13,14 Consequently, mitochondrial transfer can be broadly categorized into direct and EV-mediated mechanisms (Figure 1).
Figure 1.
Mechanisms of mitochondrial transfer. (a) Tunnel-nanotubes transfer mitochondria mediated by microtubules and Myosin/Miro1 complex. (b) Mitochondrial Gap-junction-channel transfer mediated by Connexin 43. (c) Free mitochondria are released directly through the secretory autophagy pathway or an unclarified mechanism. (d) Mitochondrial fission promoted by PGC-1α signaling has been suggested to mediate extracellular mitochondrial secretion. (e) MDVs’ generation is mediated by the PINK1/Parkin, also regulated by Snx9 and Rab9. MDVs are delivered to endosomes, leading to the formation of MDV-containing MVBs (f). (g) Various pathological stimuli, such as hypoxia, induce mitochondria-containing apoptotic bodies releasing. (h) Kif5b and Myosin19 induce migrasome-mediated mitochondria transfer. (i) ARRDC1 mediates the transport of mitochondria and mitochondrial contents into microvesicles. (j–l) Mitochondria secreted extracellularly can be taken up by recipient cells through membrane fusion (j) or endocytosis (k). Extracellular free mitochondria may also interact with the recipient cell surface for uptake (l). Then these internalized mitochondria can elicit biological effects on recipient cells. (Created in BioRender.).
Mitochondrial transfer is closely linked to the metabolic state of donor cells, particularly their dependence on oxidative phosphorylation (OXPHOS) for energy production.1,7,8 Cells that rely less on mitochondrial respiration, such as glycolytic tumor cells and stem cells, are more likely to engage in mitochondrial transfer.23,24 This suggests that altered metabolic states in donor cells, often observed under pathological conditions, influence mitochondrial transfer patterns.
The concept of direct mitochondrial transfer was first demonstrated in 2006 when ρ0 cells, which lack mitochondrial DNA (mt-DNA), regained aerobic respiration by acquiring mitochondria from neighboring cells in co-culture. 25 Subsequent in vivo studies in 2012 revealed that bone marrow-derived stromal cells could transfer intact mitochondria to damaged epithelial cells, alleviating acute lung injury (ALI). 26 These pioneering studies established direct mitochondrial transfer as a critical mechanism for intercellular metabolic regulation and cellular repair.
EVs and mitochondrial transfer
EVs are membrane-bound particles released by cells that carry a diverse array of bioactive molecules, including proteins, lipids, and nucleic acids.27,28 EVs naturally facilitate intercellular communication by delivering cargo biomaterials to nearby cells and transporting these materials via the circulatory system to distant organs.1,27,28 Their role in mitochondrial transfer has garnered significant attention due to their potential in regulating cellular processes, promoting repair, and influencing disease progression.
The first evidence of EV-mediated mitochondrial transfer, specifically involving mt-DNA and proteins, was observed in skeletal muscle cells. 29 In 2013, EVs (1–8 μm) released from cultured astrocytes were shown to transfer mitochondria and ATP, thereby activating neighboring cells. 22 Later, in vivo evidence demonstrated that astrocytes transfer mitochondria via EVs (>300 nm) to stroke-damaged neurons, thereby supporting neuronal survival after ischemic injury. 1 Additionally, mitochondrial-associated RNAs were first identified in EVs released by monocytes in 2019, where they regulated inflammatory responses in endothelial cells. 30
Mechanisms of mitochondrial sorting into EVs
While EVs are often classified based on their biogenesis and function—exosomes, ectosomes, apoptotic bodies, primary cilia protrusions, migrasomes, etc.—this review follows the MISEV 2018 and 2023 guidelines,17,20 broadly classifying EVs into two main categories: small EVs (<200 nm), typically exosomes, and large EVs (>200 nm), primarily microvesicles. 17
The biogenesis of EVs is closely tied to their mitochondrial cargo.31,32 Small EVs, mainly released by multivesicular bodies (MVBs), are enriched in mitochondrial fragments and smaller mitochondrial molecules, such as proteins, DNA, and RNA. In contrast, large EVs, which primarily bud from the plasma membrane, are more likely to carry structurally intact and functional mitochondria (Figure 1). These differences in biogenesis impart distinct roles to small and large EVs, influencing their specific contributions to disease pathogenesis and cellular repair mechanisms.17,18,31
Importantly, various pathological stimuli, including oxidative stress, inflammation, and cell damage, can influence the selective sorting of mitochondria into EVs19,33,34 (Figure 1). In placental cells exposed to antiphospholipid antibodies, mitochondrial disruption suggests that fragmented mitochondria may be incorporated into EVs. 35 Recent studies propose that mitochondrial-derived vesicles (MDVs) or mitovesicles facilitate the packaging of mitochondria and their components into EVs, regulated by sorting nexin 9 (SNX9) and optic atrophy 1 (OPA1). 36 Instead of undergoing lysosomal degradation, MDVs are released extracellularly under the influence of parkin and phosphatase and tensin homolog (PTEN)-induced kinase 1 (PINK1), 37 ultimately forming exosomes that selectively carry mitochondrial content or small mitochondria.38,39 Additionally, mitocytosis involves the transport of mitochondria to the plasma membrane via kinesin family member 5B (Kif5b), where Myosin19 anchors them to cortical actin before their encapsulation into migrasomes. 40 These findings suggest a complex interplay between EV-mediated mitochondrial transfer and disease pathogenesis.
Targeted delivery of mitochondrial EVs
EVs carry surface markers from their cell-of-origin, enabling targeted delivery to recipient cells.41,42 Macrophages (Mφs) transfer mitochondria via EVs to dorsal root ganglia (DRG) neurons, mitigating inflammatory pain responses. This process critically depends on the interaction between the CD200 receptor (CD200R) on Mφ-derived EVs and the intestinal secretory cell protein 1 (iSEC1) ligand on neurons, 43 highlighting the importance of targeted delivery in EV-mediated mitochondrial transfer. Similarly, astrocyte-derived mitochondrial EVs have been shown to target hypoxic neurons following ischemic stroke through a calcium-dependent mechanism mediated by the CD38/circulating ADP ribose (cADPR) signaling pathway, supporting neuronal survival and mitochondrial function. 1 Adipocyte-derived mitochondrial EVs circulate and target cardiomyocytes after ischemic injury organ-specifically, contributing to cardioprotection. 44 These targeting properties underscore the therapeutic potential of EV-mediated mitochondrial transfer in various disease contexts.
Scope of this review
With their unique structural and functional advantages, EVs enable enhanced stability, long-distance transport, targeted delivery, and amplified activity in recipient cells. To provide an overview of the advancements of EV-based mitochondrial transfer, this review focuses on various mitochondrial cargos and their biological implications. We begin with the EV-mediated transfer of intact mitochondria, emphasizing its therapeutic potential (Table 1). Then, we review the transfer of mitochondrial RNA and proteins, highlighting their localized regulatory effects in recipient cells, such as in axonal regulation (Table 2). Finally, we discuss the EV-mediated transfer of mt-DNA, emphasizing its utility as a biomarker for disease progression (Table 2). For each mitochondrial cargo type, we explore its specific packaging mechanisms, functional significance in recipient cells, and implications in both disease and health contexts.
Table 1.
Mitochondria in EVs.
| Key findings | EVs type (Size) | Pathogenic factors | Donor cells/organs | Recipient cells/organs | References |
|---|---|---|---|---|---|
| Promote metabolism/OXPHOS capacity | EVs | ALI | BMDSCs | Alveolar epithelial cells | 26 |
| EVs (100–1000 nm) | Wound healing | MSCs | Articular chondrocytes | 45 | |
| EVs (100–300 nm) | Renal artery stenosis | IRSTCs | Tubular epithelial cells | 119 | |
| EVs | COPD | MSCs | ASMCs | 120 | |
| Microvesicles | Sepsis | MSCs | Intestinal epithelial cells | 121 | |
| EVs (100–1000 nm) | ARDS | MSCs | Airway epithelial and endothelial cell | 122 | |
| EVs (>220 nm) | Heart failure | Cardiomyocytes | Hypoxia-injured cardiomyocytes | 123 | |
| Extracellular mitochondrial particles (300–1100 nm) | Cerebral IS | Astrocytes | Neurons | 1 | |
| Mitochondria-containing vesicles | Inflammatory pain | M2-like Mφs | Sensory neurons | 43 | |
| MVs (>150 nm) | Cerebral IS | BECs | Recipient BECs and neurons | 19,57 | |
| EVs | GBM | TASCs | ρ0 GBM cells | 13 | |
| Microparticles | Breast Cancer | Platelet | Breast cancer cell | 124 | |
| EVs (∼442 nm) | IS | BECs | BECs | 9,19 | |
| EVs (100–1000 nm) | Mitochondrial diseases | Platelet | Neutrophils and monocytes | 34 | |
| MVs (>500 nm) | Acute pancreatitis | MSCs | Pancreatic acinar cells | 125 | |
| Large CD133+ EVs (2–6 μm) | Melanoma | Melanoma cells | TME | 54 | |
| Mitochondrial quality‐control process | Axonal Protrusions/Evulsions | Neuronal dysfunction | RGC | Astrocytes | 56 |
| Exophers (∼3.5 μm) | Heart biology | Cardiomyocytes | Cardiac-resident Mφs | 126 | |
| Large EVs (115–550 nm) | Danon disease | Heart | Mφs | 50 | |
| Neutrophil immunoregulation | MVs (<1 μm) | ALI | Damaged alveolar epithelial cells | Neutrophils | 127 |
| T cell immunoregulation | EVs (>100 nm) | Asthma | MDRCs | T cell | 39 |
| Small EVs | Asthma | MDRCs | CD4+ T cells | 128 | |
| Macrophage immunoregulation | EVs (100–1,000 nm) | ARDS | MSCs | MDMs | 129 |
| EVs (>300 nm) | Skin wound | MSCs | Mφs | 130 | |
| Exosomes | ALI | Adipocytes | Alveolar Mφs | 131 | |
| EVs | Corneal epithelial defects | Human BMSCs | Mφs | 132 | |
| Proinflammatory response | Microparticles (100–1,000 nm) | RA | Platelet | Neutrophils/leukocyte | 52,53 |
| MVs (∼206.6 nm) | Inflammation | Activated Monocytes | Endothelial Cells | 30 | |
| Trigger a burst of ROS | Small EV (30–500 nm) | Cardiac ischemia/reperfusion injury | Adipocytes | Cardiomyocytes | 44 |
| Lead to target cell failure and apoptosis | EVs (50–150 nm) | Acute pancreatitis | M1 macrophages | Pancreatic beta cells | 133 |
| Induce mitochondrial dysfunction | MVs (100–1000 nm) | Sepsis | Macrophage | Neutrophils | 33 |
| Rescue the excessive accumulation of ROS | Microvesicles (∼141 nm) | Intervertebral disc degeneration | MSCs | Nucleus pulposus cells | 51 |
| Microvesicles (0.1–1 μm) | ALI | Alveolar epithelial cells | Neutrophils | 127 | |
| Induce cancer chemoresistant | EVs (100–1000 nm) | TNBS | Chemoresistant TNBC cells | Sensitive TNBC cells | 55 |
| Induce cancer growth | EVs | GBM | TASC | GBM cells | 13 |
| Augmented mitochondrial biogenesis and function | Large EVs (>200 nm) | Anthracycline-induced cardiomyopathy | iPSC | Cardiomyocytes | 134 |
| Optimizes the thermogenic program | MVs (∼350 nm) | Obesity | Brown adipocytes | Mφs | 135 |
ALI: acute lung injury; COPD: chronic obstructive pulmonary disease; ARDS: acute respiratory distress syndrome; IS: ischemic stroke; GBM: glioblastoma; RA: rheumatoid arthritis; TNBC: triple-negative breast cancer; BMDSCs: bone marrow-derived stromal cells; MSCs: mesenchymal stem cells; IRSTCs: intrinsic renal scattered tubular cells; ASMCs: airway smooth muscle cells; Mφs: macrophages; BECs: brain endothelial cells; TASCs: tumor-activated stromal cells; TME: tumor microenvironment; RGC: retinal ganglion cell; MDMs: monocyte–derived macrophages; MDRCs: myeloid-derived regulatory cells; TASC: tumor-activated stromal cells.
Table 2.
Mitochondrial contents in EVs.
| Content | Key findings | EVs type (Size) | Pathogenic factors | Donor cells/ organs | Recipient cells/ organs | References |
|---|---|---|---|---|---|---|
| mt-DNA | Biomarker | Exosomes | Epithelial ovarian cancer | Plasma | —— | 93 |
| Exosomes (50–400 nm) | Renal cell carcinoma | Plasma | —— | 94 | ||
| Exosomes | Non-small cell lung cancer | Plasma | —— | 91 | ||
| Exosomes (30–130 nm) | GBM | GBM cells, neural stem cells | —— | 98 | ||
| Proinflammatory response | Exosomes | Autism | Serum | Microglia | 106 | |
| Microparticles (50–1000 nm) | Excessive alcohol use | Liver | Neutrophil | 103,107 | ||
| EVs | BS | Monocytes | Circulation | 108 | ||
| EVs (∼100 nm) | On-Pump Heart Surgery | Plasma | Circulation | 136 | ||
| Enhance mitochondrial respiration/tumor progression | EVs (<200 nm) | Colon cancer | Colon cancer cell | Colonic epithelial cells | 11 | |
| Promote metabolism/OXPHOS capacity | Exosomes | Breast cancer | CAF | HTR cancer cells | 101 | |
| Activate endothelial cells | Exosomes | Preeclampsia | Placental explants | Endothelial cells | 35 | |
| Induce hepatocyte damage and fibrosis | Exosomes | Liver fibrosis | Damaged-hepatocytes | Mouse primary hepatocytes | 105 | |
| Attenuate mtDNA damage and inflammation | EVs (∼160 nm) | ARI | MSCs | HK-2 cells | 68 | |
| mt-Prots | Biomarker | Exosomes (40–300 nm) | Melanoma | Melanoma tissue | —— | 79 |
| EVs (70–118 nm) | AD | Neurons | Plasma | 73 | ||
| Small EVs | PD | Serum | —— | 80 | ||
| Small EVs (<100 nm) | PF&S | Serum | —— | 78 | ||
| Exosomes (30–100 nm) | Metabolic disorders | Brown adipose tissue (BAT) | Plasma | 76 | ||
| Promote metabolism/OXPHOS capacity | Engineered exosomes | Neurodegeneration | Tom40 overexpressing cells | Brain tissue | 74 | |
| EVs | IS | BECs | Brain tissue | 19 | ||
| Enhance mitochondrial respiration/ promote tumor survive | EVs (50–400 nm) | Prostate cancer | PC3 cells | PC3 cells | 83 | |
| Mitochondrial RNA-binding protein | Small EVs (100–200 nm) | IS | Neurons | Neurons | 64 | |
| Exosomes (30–150 nm) | —— | —— | —— | 65 | ||
| Induce mitochondrial dysfunction | Mitovesicles | DS | Brain tissue | Hippocampal slices | 12 | |
| T cell immunoregulation | EVs (∼154.5 nm) | Autoimmune hepatitis | MSCs | CD4+ T-cells | 82 | |
| mt-RNA | Biomarker | EVs (<600 nm) | AD | Plasma | —— | 10 |
| Microvesicles (30–200 nm) | CAD | Monocytes | Plasma | 137 | ||
| Rescue the excessive accumulation of ROS | EVs | ARI | MSCs | Kidney | 62 | |
| Proinflammatory response | Exosomes | Alcoholic liver disease | Hepatocytes | Kupffer cells | 66 | |
| Promote OXPHOS capacity/promote the osteogenesis | Exosomes | Bone defect | MSCs | DPSCs | 61 |
BS: Behçet’s syndrome; ARI: acute renal injury; CAD: coronary artery disease; AD: Alzheimer’s disease; PD: Parkinson’s disease; PF&S: physical frailty and sarcopenia; DS: Down syndrome; CAF: cancer-associated fibroblast; DPSCs: dental pulp stem cells.
EV-mediated transfer of intact mitochondria
Functional mitochondria are encapsulated in EVs derived from cells under physiological and pathophysiological conditions.1,18,22,31 The transfer of intact mitochondria via EVs is a direct form of intercellular regulation of metabolic responses with emerging and important implications in disease pathology.1,18 Notably, functional mitochondria can also be encapsulated in EVs released by healthy cells,45–47 offering promising therapeutic potential for repairing mitochondrial dysfunction in recipient cells (Table 1).
A wide range of cells can produce mitochondria-containing EVs, though production levels and functionality vary with cell type, metabolic state, and external stimuli. 7 Cells with surplus mitochondria or low mitochondrial demands, such as brain endothelial cells (BECs), are more likely to release mitochondrial-containing EVs. For example, under cerebral ischemic conditions, BECs with high mitochondrial loads release mitochondria-enriched EVs to support recipient cells, such as neurons, thereby boosting ATP levels and improving neurological outcomes. 19 Endogenous factors like resveratrol-activated peroxisome proliferator-activated receptor gamma coactivator-1-alpha (PGC-1α) can increase mitochondrial biogenesis in BECs, promoting the secretion of mitochondria-enriched EVs and supporting recipient cell metabolism under stress conditions. 48
Environmental factors and pathological changes also influence mitochondrial EV production. Mφs under lipopolysaccharide (LPS)-induced necroptosis or pyroptosis can release large EVs containing mitochondria.33,49 Molecules mediating intracellular transport, such as the actions of ras-related protein Rab7, Rab22a, and Kif5b, aid in transporting mitochondria to the plasma membrane for EV packaging.50,51 These findings support the strong therapeutic potential of EV-mediated mitochondrial transfer, especially under conditions involving energy deficits and mitochondrial dysfunction.
Pathological roles of EV-mediated mitochondrial transfer in disease
EV-mediated mitochondrial transfer can contribute to disease progression. For example, activated platelets release EVs containing mitochondria, which can lead to inflammatory responses, contributing to neuroinflammation in Alzheimer’s disease (AD) and Parkinson’s disease (PD).52,53 In cancer, mitochondrial transfer via EVs provides energetic support to tumor cells, meeting the demands of rapid cell division and enabling tumor growth. Glioblastoma multiforme (GBM) cells receive mitochondria-containing EVs from astrocytes, enhancing their tumorigenicity and resistance to therapies such as radiotherapy and chemotherapy. 13 In breast cancer, EV-mediated mitochondrial transfer from stromal cells contributes to chemoresistance and metastasis.54,55 Similarly, stress-induced EVs from adipocytes carry oxidatively damaged but respiration-competent mitochondria to cardiomyocytes, affecting ROS production and heart tissue resilience to oxidative stress. 44
Endogenous repair and protective mechanisms via EV-mediated mitochondrial transfer
EV-mediated mitochondrial transfer maintains tissue homeostasis and promotes cell survival and recovery. For example, retinal ganglion cells (RGCs) package damaged axonal mitochondria into large EVs for transfer to adjacent astrocytes, facilitating the clearance of dysfunctional organelles. 56 This mechanism also plays a key role in tissue repair and protection. Following stroke, astrocytes release large EVs containing functional mitochondria that can be taken up by hypoxic neurons, supporting neuronal mitochondrial function and survival. 1 BEC-derived EVs containing mitochondrial proteins, such as ATP synthase F1 subunit alpha (ATP5A), contribute to ATP production in recipient cells, enhancing metabolic recovery in ischemic models. 19 In mouse models of ischemic stroke, intravenous administration of mitochondria-containing EVs from BECs significantly reduce infarct volume and improve neurological function, demonstrating therapeutic promise.9,57
Neural stem cells release EVs with intact mitochondria that modulate recipient immune cells, such as Mφs, by restoring OXPHOS and maintaining mitochondrial function, which helps regulate inflammatory responses.24,39 In pain modulation, Mφ-derived EVs transfer mitochondria to DRG neurons, aiding in the resolution of inflammation and reducing inflammatory pain transmission to the brain. 43
In neurodegenerative disease models, mesenchymal stem cell-derived EVs (MSC-EVs) carrying intact mitochondria can attenuate oxidative stress, inhibit apoptosis, and improve mitochondrial function.45,58 In AD models, MSC-EVs enhance cellular metabolism and reduce cell death by delivering functional mitochondria to recipient cells. 58 These findings underscore the therapeutic potential of EV-mediated mitochondrial transfer for neuroprotection and metabolic support.
Given the role of EV-mediated mitochondrial transfer in energy homeostasis, encapsulating functional mitochondria in EVs has become an area of interest for therapeutic applications in neurological and systemic diseases.1,48,51 Elucidating the mechanisms of EV-mediated mitochondrial transfer may, therefore, lead to the development of targeted therapies in conditions marked by mitochondrial dysfunction.
EV-mediated transfer of mitochondrial RNA (mt-RNA)
EVs surpass synthetic delivery systems in RNA transport efficiency and can encapsulate and deliver diverse types of mt-RNA, including messenger RNA (mRNA), microRNA (miRNA), and long noncoding RNA (lncRNA).59,60 mt-RNA, sourced from both the mitochondrial and nuclear genomes, is abundant in small and large EVs,34,61 enabling targeted delivery to recipient cells. 62 This EV-mediated mt-RNA transfer plays multiple roles in disease progression; it can act as a pathogenic factor, contributing to cellular dysfunction and inflammation, and support cellular repair mechanisms by stabilizing mitochondrial function and protecting cells from damage (Table 2).
During the transport of coding and noncoding RNAs, RNA-binding proteins (RBPs) assist in packaging RNA into small EVs and facilitate RNA transport via RNA-ribonucleoprotein (RNP) complexes.63,64 They also contribute to mRNA localization at synapses, enabling neurons to respond swiftly to stimuli by promoting rapid protein synthesis.63,64 Key RBPs, e.g., fused in sarcoma (FUS), selectively bind specific mitochondrial mRNAs, packaging them into small EVs and delivering them to recipient cells. For example, hypoxic neuron-derived EVs (HypEVs) containing FUS transport mitochondrial mRNA to dendrites in recipient neurons, where they promote translation on endosomes, compensating for the loss of mitochondrial proteins. In stroke models, the knockdown of FUS significantly reduces the neuroprotective effect of HypEVs. 64 Another RBP, Y-box binding protein 1 (YBX1), selectively binds mitochondrial microRNA miR-223 through a specific sequence motif (UCAGU), promoting its sorting into small EVs. 65
EV-mt-RNA transfer as a pathogenic factor in disease
mt-RNA transported via EVs can promote disease pathology by triggering inflammation, immune activation, and cellular dysfunction in recipient cells. Under conditions of mitochondrial dysfunction, excessive ROS generation can damage mt-RNA, leading to the release of fragmented mt-RNAs through EVs.66,67 These damaged mitochondrial RNAs act as damage-associated molecular patterns (DAMPs), activating inflammatory responses and contributing to autoimmune reactions. For example, EVs containing mitochondrial mRNAs such as mt-Nd5 and mt-Nd6 can activate toll-like receptor 3 (TLR3) in recipient cells, initiating self-RNA-mediated autoimmunity. 66 Additionally, conditions such as metabolic syndrome can alter the miRNA cargo of EVs, disrupting the regulation of mitochondrial genes in recipient cells. This can impair the therapeutic efficacy of stem cell-derived EVs and exacerbate disease progression. 67 In ischemic and injured cells, mt-RNA and other mitochondrial components, like electron transport chain (ETC) proteins, are released through EVs, inducing pro-inflammatory cytokines, such as interleukin (IL)-8 and type I interferons (IFNs). 30 The robust inflammatory response triggered by mt-RNA-carrying EVs underscores the role of these vesicles as potential pathogenic factors that can intensify disease progression in inflammatory and autoimmune disorders. 66
EV-mt-RNA transfer as an endogenous repair mechanism
Conversely, mt-RNA transferred by EVs can support cellular repair and enhance mitochondrial function in recipient cells, particularly under conditions of stress or injury.61,62,64 EVs from healthy cells, including stem cells and neurons, transport mt-RNA to assist damaged cells in restoring mitochondrial function. For example, stem cell-derived small EVs deliver mitochondrial transcription factor A (TFAM) mRNA, which upregulates TFAM expression and regulates OXPHOS by enhancing glutamate dehydrogenase (GDH) activity, restoring mitochondrial function, and exerting anti-inflammatory effects. 61 Notably, the EV-mediated TFAM mRNA transfer to recipient cells is unaffected by transcriptional inhibition, highlighting the robust mechanism of EV-mediated mRNA delivery. 68 Neuronal small EVs generated under hypoxic conditions deliver mitochondrial mRNA (e.g., Cox7a1, mt-Nd6, mt-Co1) to neurons, promoting cell survival by preserving neuronal morphology and function after ischemic injury. 64 Similarly, in ischemia-reperfusion-induced acute kidney injury, MSC-EVs enriched with superoxide dismutase 2 (SOD2) mRNA reduce oxidative stress in recipient cells, protecting against ROS damage. 62
In neurodegenerative diseases, mt-RNA delivered to axonal termini supports mitochondrial function, essential for axon elongation, regeneration, and synapse stability. 69 We and others have demonstrated that EVs can deliver cargo miRNA to distal axons and facilitate localized protein regulation.15,16,70,71 The localized RNA regulatory pathogenesis plays a pivotal role in several neurodegenerative diseases, including amyotrophic lateral sclerosis/frontotemporal dementia, spinal muscular atrophy, AD, and fragile X syndrome (FXS). 72 This suggests that the mt-RNA EV transfer contributes to the rapid and steady supply of mitochondrial proteins to damaged or regenerating axons, potentially aiding recovery, and therefore, holds promise as an intrinsic mechanism for mitochondrial maintenance and neuroprotection in various disease states.
EV-mediated transfer of mitochondrial proteins (mt-prots)
The encapsulation and transfer of mitochondrial component proteins via EVs play significant roles in cellular communication, which can serve as biomarkers, metabolism regulators, and mt-RNA carriers63,64,73,74 (Table 2). Certain pathways may contribute to the sorting of mt-Prots into EVs. The deletion of arrestin domain-containing protein 1 (ARRDC1) in wild-type mouse embryonic fibroblasts led to an enrichment of mt-Prots in EVs, suggesting that ARRDC1 plays a role in regulating the incorporation of mt-Prots into microvesicles. 75
EV-mt-Prots transfer as a pathogenic factor in disease
The presence and altered levels of mitochondrial proteins in EVs have been associated with various diseases, particularly in neurodegenerative disorders, cancers, and metabolic diseases.12,76,77 Pathological conditions affect mitochondrial protein sorting mechanisms, leading to the selective packaging of disease-related mitochondrial proteins into EVs. 77
Mitochondrial proteins carried by small EVs are largely related to mitochondrial respiratory function, particularly in the nervous system.9,73 Neuronal-derived EVs (NDEVs) from the plasma of AD patients contain mitochondrial proteins, including electron transport chain (ETC) components, ATP synthase, and superoxide dismutase 1 (SOD1). 73 These proteins are present in differing levels between AD patients and controls, with reduced catalytic activity of ATP synthase and OXPHOS complex IV observed in NDEVs from AD patients. Similarly, Down Syndrome (DS) and AD models secrete EVs with elevated levels of monoamine oxidase B (MAO-B), a mitochondrial protein linked to impaired long-term potentiation (LTP) and cognitive decline, highlighting a common pathway in neurodegenerative disease progression. 12 In addition to neurological disease, small EVs from patients with Sarcopenia show reduced levels of key mitochondrial proteins, specifically ATP5A complex V, ubiquinone oxidoreductase subunit S3 (NDUFS3) complex I, and succinate dehydrogenase complex iron sulfur subunit B (SDHB) complex II, compared to controls. 78 This altered mitochondrial protein profile may contribute to reduced cellular energy and increased vulnerability to muscle degeneration in Sarcopenia. When brown adipose tissue (BAT) is activated, a mitochondrial tetrahydrofuran synthesis enzyme, methylenetetrahydrofolate dehydrogenase (NADP+ dependent) 1-like (MTHFD1L), is enriched in EVs, suggesting its potential as a plasma biomarker for BAT activity and metabolic health. 76
In addition to mitochondrial respiratory proteins, cancer-derived EVs have been shown to contain mitochondrial membrane proteins such as mitochondrially encoded cytochrome c oxidase II (MT-CO2) and cytochrome c oxidase subunit VIc (COX6c), which are associated with cancer-related mitochondrial functions and can be detected in blood, making them as potential biomarkers for cancer. 79 Additionally, mitochondrial proteins like ATP5A are present in EVs from murine cancer-associated fibroblasts (CAFs) and in serum from adults with PD, suggesting that these proteins may serve as indicators of metabolic dysregulation in various pathological states. 80
EV-mt-Prots transfer as an endogenous repair mechanism
EV-mediated transfer of mitochondrial proteins also plays protective and reparative roles in injured or metabolically stressed cells, particularly in the nervous system and immune systems, where mitochondrial protein transfer supports cellular recovery, metabolic reprogramming, and survival.9,81–83
EVs from BECs contain mitochondrial structural and functional proteins, such as translocase of outer mitochondrial membrane 20 (TOMM20) and ATP5A, which help maintain ATP production in ischemic stroke models. 19 In addition, combining mitochondrial protein transfer with mitochondria-containing EV complexes can further increase intracellular ATP levels and enhance cell viability in damaged endothelial cells, supporting neuroprotection and brain repair in ischemic conditions. 57 Treating BECs with heat shock protein 27 (HSP27), in conjunction with mitochondria-containing EVs, further elevates ATP levels and mitochondrial respiration in ischemic BECs, potentially enhancing recovery post-stroke. 9
EV-mediated mitochondrial protein transfer has also been shown to regulate immune cell function, notably in CD4+ T cells, by inducing metabolic reprogramming. 82 The transfer of mitochondrial antioxidant proteins such as peroxiredoxin 3 (PRDX3) and glutathione peroxidase 4 (GPX4) via EVs can increase the survival of recipient cells, as demonstrated in PC3 cell models, by reducing oxidative stress and protecting against apoptosis. 83
EV-mediated transfer of mt-DNA
EVs have been shown to carry DNA fragments, with the first report in 1990 of DNA within EVs emerging from human epithelial prostate cells. 84 More recently, DNA fragments representing entire genomes, including mutations, have been identified in EVs derived from cancer cell lines and plasma from patients with pancreatic cancer.85,86 Mitochondria contain their own genome encoding 13 essential polypeptides involved in the ETC and ATP synthase, which are critical for OXPHOS. 87 Genetic mutations and changes in mt-DNA copy number are associated with cell survival, aging, and developing severe diseases, such as cancer. 88 The first evidence that EVs from astrocytes and GBM cells carry mt-DNA was reported in 2010. 89 Subsequent studies have shown that small EVs secreted by C2C12 myoblasts contain both mt-DNA and mt-DNA-associated proteins. Notably, proteins such as insulin-like growth factor binding protein 5 (IGFBP-5) in micro-vesicles, associated with mt-DNA, enhance stability and facilitate long-range transfer of mt-DNA. 29
EV-mt-DNA transfer as a pathogenic factor in disease
Due to the absence of histone protection and direct exposure to ROS, mt-DNA is more prone to mutations than nuclear DNA.90,91 EVs carrying mutated or aberrant mt-DNA serve as potential biomarkers, as mt-DNA abnormalities often emerge in early disease stages and persist throughout disease progression.11,27,92 Bronchoalveolar lavage (BAL) fluid from asthmatic patients contains significantly higher mt-DNA levels than healthy controls. 39 In ovarian cancer patients, mt-DNA copy numbers in plasma EVs indicate cancer progression. 93 Similarly, high mt-DNA content in small EVs has been suggested as a promising biomarker for renal cell carcinoma. 94 In non-small cell lung cancer (NSCLC), EV-encapsulated mt-DNA strongly correlates with aggressive tumor features, such as larger tumor size, advanced tumor stage, lymph node involvement, and metastasis, offering improved sensitivity and specificity for diagnosis. 91
mt-DNA abnormalities and mutations have also been associated with neurodegenerative diseases and cancers, including GBM.95,96 In GBM patients, higher mt-DNA copy numbers in plasma EVs correlate with disease state. 97 In addition, aberrations in mt-DNA are detected in small EVs from GBM cells but not from neural stem cells. 98 Since brain tissue is difficult to access, EV analysis provides a noninvasive approach to detect disease and cancer in the CNS. EVs also carry surface markers that reflect their cellular origins.42,99 Combined detection of EV surface proteins and mt-DNA anomalies may enhance diagnostic accuracy, potentially transforming mt-DNA-containing EVs into powerful biomarkers for disease prediction and monitoring.
Moreover, EV-encapsulated mt-DNA contains the entire mitochondrial genome, allowing it to exert functional effects in recipient cells.11,100 EVs derived from CAFs transfer the complete mitochondrial genome to breast cancer stem-like cells, enhancing their metabolism and promoting tumor growth through OXPHOS upregulation. 101 Similarly, small EVs carrying the full mitochondrial genome facilitated metabolic crosstalk between colorectal cancer (CRC) cells and adjacent stromal cells (ASCs), impacting cellular respiration and ROS production to support tumor progression. 11
Interestingly, research has shown that mt-DNA levels of plasma EVs decrease with age, potentially affecting mitochondrial energetics in an age-dependent manner. 102 Although there is no current evidence of EV-mediated mt-DNA transfer altering the metabolic state in CNS-related diseases, these findings suggest that mt-DNA transfer could be therapeutically relevant for CNS disorders such as stroke.
EV-mt-DNA transfer and the inflammatory response
In addition to its biomarker role, mt-DNA functions as DAMPs when released from mitochondria, initiating inflammatory signaling by activating pathways such as TLR9, cyclic GMP-AMP synthase- stimulator of interferon genes (cGAS-STING), and ataxia-telangiectasia mutated- checkpoint kinase 2 (ATM-Chk2). 35 , 103 , 104 EV-encapsulated mt-DNA may trigger a stronger immune response than free mt-DNA due to its protection within the EV lipid bilayer. For example, ethanol-induced endoplasmic reticulum (ER) stress prompts hepatocytes to release mt-DNA-rich EVs, promoting neutrophil infiltration and liver damage. 103 Inhibition of mt-DNA transfer via tetramethylpyrazine (TMP) has shown promise in reducing EV-mediated inflammation in liver injury. 105 Similarly, breast cancer cells package mt-DNA into EVs through a PINK1-dependent mechanism, triggering invasive responses by activating TLR9 in recipient cells. 37
In the CNS, mt-DNA-containing small EVs from children with autism spectrum disorder have been shown to stimulate microglial IL-1β secretion, contributing to neuroinflammation. 106 Additionally, long-term alcohol exposure induces the release of mt-DNA-enriched EVs, promoting liver inflammation via apoptosis signal-regulating kinase 1 (ASK1) and p38 signaling. 107 Caspase-1 activation in hepatocytes can also promote mt-DNA packaging into EVs, resulting in sterile inflammation and exacerbating inflammatory diseases. 108
Although there is no direct evidence yet, increased secretion of mt-DNA-containing EVs following traumatic brain injury (TBI) or stroke could potentially lead to sterile neuroinflammation. Suppressing mt-DNA release or blocking its downstream effectors may thus provide a therapeutic approach for mitigating inflammation following trauma or surgery.
Discussion and prospective in neurological disorders
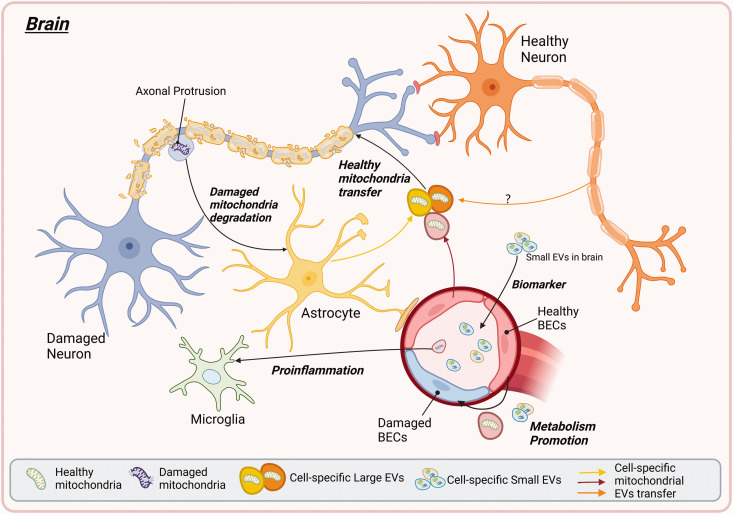
Emerging studies on EV-mediated mitochondria and their component transfer have revealed new aspects of intercellular communication, with intact mitochondria and mitochondrial components (mt-RNA, mt-Prot, and mt-DNA) playing distinct yet complementary roles in cellular maintenance and disease progression. As EV-based research advances, the therapeutic potential of mitochondrial transfer holds promise to combat various diseases, particularly within the CNS where localized, and for cell-specific regulation and communication between the CNS and peripheral organs 109–111 (Figure 2).
Figure 2.
The EV-mediated cell-type-specific mitochondrial transfer and its potential role in neurological disease. EVs, carrying cell-specific surface markers, transfer mitochondrial components such as DNA, RNA, proteins, and intact mitochondria between neurons, astrocytes, and microglia (color indicated), supporting axonal maintenance, localized metabolic regulation in EV-targeting cells, and brain remodeling. These EVs can also cross the BBB and deliver their mitochondrial cargo to distant organs, contributing to the pathological biomarker and systemic metabolic homeostasis factor. (Created in BioRender.).
The transfer of functional mitochondria via EVs could support neural regeneration, enhance cellular resilience, and stabilize metabolic processes across neural networks and distal organs by promoting ATP production, balancing ROS, and maintaining overall metabolic function.1,18 Given the scalability and ability to cross the blood-brain barrier (BBB),24,48,112 EV-mediated mitochondrial transfer provides an opportunity to develop therapies for neurological dysfunction.
The transfer of mt-RNA and mt-Prots by EVs facilitates rapid, localized regulation, particularly at axonal terminals, where such interventions could preserve or restore neural integrity (Figure 2). This feature enables treating neurological disorders with compromised axonal health, such as neurodegenerative diseases and CNS injuries. Mitochondria-specific miRNAs have been identified as key regulators of mitochondrial function, 113 and their EV-mediated transfer is suggested by miRNA motifs that facilitate sorting into EVs. 114 Furthermore, during the transport of coding and noncoding mt-RNA, RBPs in mitochondria play a crucial role in packaging mt-RNA into EVs and aiding mt-RNA transport via RNP complexes.64,65 EV-encapsulated mt-DNA offers both a biomarker for disease progression and a means to modulate mitochondrial function in recipient cells. Future research is warranted to investigate the therapeutic potential of controlled mt-DNA delivery to manage cellular energetics and mitigate metabolic dysregulation. Additionally, further studies could clarify the diagnostic potential of mt-DNA in EVs, providing non-invasive, cell-type-specific biomarkers for neurodegenerative conditions and other diseases.
Small and large EVs transport distinct mitochondrial components, but their interactions in mitochondrial transfer remain unclear. For instance, treating BECs with HSP27 and mitochondria-containing large EVs significantly enhances ATP production in ischemic cells. 9 Similarly, ATP5A-enriched large EVs correlate with improved ATP levels in BECs. 57 As small EVs primarily deliver RNAs and proteins, it is worth exploring whether their localized transfer primes cells for the uptake of intact mitochondria from large EVs, resulting in a synergistic metabolic response.
While pathways mediating mitochondrial encapsulation into EVs are well-documented,31,32 the molecular mechanisms governing their regulation remain poorly understood and warrant further investigation. For example, oxidative stress, inflammation, and cellular damage can influence mitochondrial sorting into EVs,33,34 but whether mitochondria enter different EV subtypes through shared or distinct mechanisms remains an open question.
Additionally, the intracellular destination of EV components within recipient cells plays a pivotal role in their functional impact.15,32,115 For example, mitochondrial RNAs delivered via EVs may facilitate communication between the nucleus and mitochondria. 50 EV-mediated transfer of mitochondrial components can increase cellular bioenergetics, survival, mt-DNA copy number, and modulate anti-inflammatory responses of recipient cells.18,32,97 However, further research is needed to clarify how EVs carrying intact mitochondria or specific mitochondrial components are routed within recipient cells to affect recipient cell metabolic function. The ER, through its direct interaction with mitochondria via mitochondria-associated membranes (MAMs), may act as a critical hub for coordinating these regulatory functions.116–118 Elucidating these pathways could advance the therapeutic potential of EV-mediated mitochondrial transfer.
The future of EV-based therapies, in-part, lies in improving delivery methods, increasing target specificity, and combining diverse mitochondrial components to treat multifactorial diseases. With ongoing research, EV-mediated mitochondrial transfer has the potential to become a powerful platform for diagnosing and treating conditions where mitochondrial health is critical for cellular resilience and function.
Footnotes
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH grants R01 NS111801 (ZGZ).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
ORCID iDs: Mingjin Wang https://orcid.org/0009-0006-6951-1206
Michael Chopp https://orcid.org/0000-0002-1948-4493
References
- 1.Hayakawa K, Esposito E, Wang X, et al. Transfer of mitochondria from astrocytes to neurons after stroke. Nature 2016; 535: 551–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rangaraju V, Lewis TL, Hirabayashi Y, et al. Pleiotropic mitochondria: the influence of mitochondria on neuronal development and disease. J Neurosci 2019; 39: 8200–8208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Trigo D, Avelar C, Fernandes M, et al. Mitochondria, energy, and metabolism in neuronal health and disease. FEBS Lett 2022; 596: 1095–1110. [DOI] [PubMed] [Google Scholar]
- 4.Islam MT. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol Res 2017; 39: 73–82. [DOI] [PubMed] [Google Scholar]
- 5.Wang Y, Xu E, Musich PR, et al. Mitochondrial dysfunction in neurodegenerative diseases and the potential countermeasure. CNS Neurosci Ther 2019; 25: 816–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang Z, Huang Q, Zhao D, et al. The impact of oxidative stress-induced mitochondrial dysfunction on diabetic microvascular complications. Front Endocrinol (Lausanne) 2023; 14: 1112363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Borcherding N, Brestoff JR. The power and potential of mitochondria transfer. Nature 2023; 623: 283–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu D, Gao Y, Liu J, et al. Intercellular mitochondrial transfer as a means of tissue revitalization. Signal Transduct Target Ther 2021; 6: 65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dave KM, Stolz DB, Venna VR, et al. Mitochondria-containing extracellular vesicles (EV) reduce mouse brain infarct sizes and EV/HSP27 protect ischemic brain endothelial cultures. J Control Release 2023; 354: 368–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim KM, Meng Q, Perez de Acha O, et al. Mitochondrial RNA in alzheimer’s disease circulating extracellular vesicles. Front Cell Dev Biol 2020; 8: 581882. doi:DOI:10.3389/fcell.2020.581882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Guan B, Liu Y, Xie B, et al. Mitochondrial genome transfer drives metabolic reprogramming in adjacent colonic epithelial cells promoting TGFβ1-mediated tumor progression. Nat Commun 2024; 15: 3653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.D’Acunzo P, Argyrousi EK, Ungania JM, et al. Mitovesicles secreted into the extracellular space of brains with mitochondrial dysfunction impair synaptic plasticity. Mol Neurodegener 2024; 19: 34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Salaud C, Alvarez-Arenas A, Geraldo F, et al. Mitochondria transfer from tumor-activated stromal cells (TASC) to primary glioblastoma cells. Biochem Biophys Res Commun 2020; 533: 139–147. [DOI] [PubMed] [Google Scholar]
- 14.Yang J, Liu L, Oda Y, et al. Extracellular vesicles and Cx43-Gap junction channels are the main routes for mitochondrial transfer from Ultra-Purified mesenchymal stem cells, RECs. Int J Mol Sci 2023; 24: 10294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang Y, Li C, Qin Y, et al. Small extracellular vesicles ameliorate peripheral neuropathy and enhance chemotherapy of oxaliplatin on ovarian cancer. J Extracell Vesicles 2021; 10: e12073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Y, Qin Y, Chopp M, et al. Ischemic cerebral endothelial cell-derived exosomes promote axonal growth. Stroke 2020; 51: 3701–3712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Welsh JA, Goberdhan DCI, O'Driscoll L, MISEV Consortium et al. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J Extracell Vesicles 2024; 13: e12404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fairley LH, Grimm A, Eckert A. Mitochondria transfer in brain injury and disease. Cells 2022; 11: 3603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dave KM, Venna VR, Rao KS, et al. Mitochondria-containing extracellular vesicles from mouse vs. human brain endothelial cells for ischemic stroke therapy. J Control Release 2024; 373: 803–822. [DOI] [PubMed] [Google Scholar]
- 20.Théry C, Witwer KW, Aikawa E, et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the international society for extracellular vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles 2018; 7: 1535750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Roger AJ, Muñoz-Gómez SA, Kamikawa R. The origin and diversification of mitochondria. Curr Biol CB 2017; 27: R1177–R1192. [DOI] [PubMed] [Google Scholar]
- 22.Falchi AM, Sogos V, Saba F, et al. Astrocytes shed large membrane vesicles that contain mitochondria, lipid droplets and ATP. Histochem Cell Biol 2013; 139: 221–231. [DOI] [PubMed] [Google Scholar]
- 23.Plotnikov EY, Khryapenkova TG, Vasileva AK, et al. Cell-to-cell cross-talk between mesenchymal stem cells and cardiomyocytes in co-culture. J Cell Mol Med 2008; 12: 1622–1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Peruzzotti-Jametti L, Bernstock JD, Willis CM, et al. Neural stem cells traffic functional mitochondria via extracellular vesicles. PLOS Biol 2021; 19: e3001166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spees JL, Olson SD, Whitney MJ, et al. Mitochondrial transfer between cells can rescue aerobic respiration. Proc Natl Acad Sci U S A 2006; 103: 1283–1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Islam MN, Das SR, Emin MT, et al. Mitochondrial transfer from bone-marrow–derived stromal cells to pulmonary alveoli protects against acute lung injury. Nat Med 2012; 18: 759–765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li J, Wang T, Hou X, et al. Extracellular vesicles: opening up a new perspective for the diagnosis and treatment of mitochondrial dysfunction. J Nanobiotechnology 2024; 22: 487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kumar MA, Baba SK, Sadida HQ, et al. Extracellular vesicles as tools and targets in therapy for diseases. Signal Transduct Target Ther 2024; 9: 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Guescini M, Guidolin D, Vallorani L, et al. C2C12 myoblasts release micro-vesicles containing mtDNA and proteins involved in signal transduction. Exp Cell Res 2010; 316: 1977–1984. [DOI] [PubMed] [Google Scholar]
- 30.Puhm F, Afonyushkin T, Resch U, et al. Mitochondria are a subset of extracellular vesicles released by activated monocytes and induce type I IFN and TNF responses in endothelial cells. Circ Res 2019; 125: 43–52. [DOI] [PubMed] [Google Scholar]
- 31.Zhou X, Liu S, Lu Y, et al. MitoEVs: a new player in multiple disease pathology and treatment. J Extracell Vesicles 2023; 12: e12320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dave KM, Stolz DB, Manickam DS. Delivery of mitochondria-containing extracellular vesicles to the BBB for ischemic stroke therapy. Expert Opin Drug Deliv 2023; 20: 1769–1788. [DOI] [PubMed] [Google Scholar]
- 33.Kuang L, Wu Y, Shu J, et al. Pyroptotic macrophage-derived microvesicles accelerate formation of neutrophil extracellular traps via GSDMD-N-expressing mitochondrial transfer during sepsis. Int J Biol Sci 2024; 20: 733–750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pelletier M, Breton Y, Allaeys I, et al. Platelet extracellular vesicles and their mitochondrial content improve the mitochondrial bioenergetics of cellular immune recipients. Transfusion (Paris) 2023; 63: 1983–1996. [DOI] [PubMed] [Google Scholar]
- 35.Tong M, Johansson C, Xiao F, et al. Antiphospholipid antibodies increase the levels of mitochondrial DNA in placental extracellular vesicles: Alarmin-g for preeclampsia. Sci Rep 2017; 7: 16556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Todkar K, Chikhi L, Desjardins V, et al. Selective packaging of mitochondrial proteins into extracellular vesicles prevents the release of mitochondrial DAMPs. Nat Commun 2021; 12: 1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rabas N, Palmer S, Mitchell L, et al. PINK1 drives production of mtDNA-containing extracellular vesicles to promote invasiveness. J Cell Biol 2021; 220: e202006049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shanmughapriya S, Langford D, Natarajaseenivasan K. Inter and intracellular mitochondrial trafficking in health and disease. Ageing Res Rev 2020; 62: 101128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hough KP, Trevor JL, Strenkowski JG, et al. Exosomal transfer of mitochondria from airway myeloid-derived regulatory cells to T cells. Redox Biol 2018; 18: 54–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jiao H, Jiang D, Hu X, et al. Mitocytosis, a migrasome-mediated mitochondrial quality-control process. Cell 2021; 184: 2896–2910.e13. [DOI] [PubMed] [Google Scholar]
- 41.Pitt JM, Kroemer G, Zitvogel L. Extracellular vesicles: masters of intercellular communication and potential clinical interventions. J Clin Invest 2016; 126: 1139–1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tkach M, Théry C. Communication by extracellular vesicles: Where We are and where We need to go. Cell 2016; 164: 1226–1232. [DOI] [PubMed] [Google Scholar]
- 43.van der Vlist M, Raoof R, Willemen HLDM, et al. Macrophages transfer mitochondria to sensory neurons to resolve inflammatory pain. Neuron 2022; 110: 613–626.e9. [DOI] [PubMed] [Google Scholar]
- 44.Crewe C, Funcke J-B, Li S, et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab 2021; 33: 1853–1868.e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thomas MA, Fahey MJ, Pugliese BR, et al. Human mesenchymal stromal cells release functional mitochondria in extracellular vesicles. Front Bioeng Biotechnol 2022; 10: 870193. doi:DOI:10.3389/fbioe.2022.870193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zorova LD, Kovalchuk SI, Popkov VA, et al. Do extracellular vesicles derived from mesenchymal stem cells contain functional mitochondria? Int J Mol Sci 2022; 23: 7408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nguyen A, Turko IV. Isolation protocols and mitochondrial content for plasma extracellular vesicles. Anal Bioanal Chem 2023; 415: 1299–1304. [DOI] [PubMed] [Google Scholar]
- 48.Dave KM, Dobbins DX, Farinelli MN, et al. Engineering extracellular vesicles to modulate their innate mitochondrial load. Cell Mol Bioeng 2022; 15: 367–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Spencer DM, Dye JR, Piantadosi CA, et al. The release of microparticles and mitochondria from RAW 264.7 murine macrophage cells undergoing necroptotic cell death in vitro. Exp Cell Res 2018; 363: 151–159. [DOI] [PubMed] [Google Scholar]
- 50.Liang W, Sagar S, Ravindran R, et al. Mitochondria are secreted in extracellular vesicles when lysosomal function is impaired. Nat Commun 2023; 14: 5031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shi P, Gao H, Cheng Z, et al. Static magnetic field-modulated mesenchymal stem cell-derived mitochondria-containing microvesicles for enhanced intervertebral disc degeneration therapy. J Nanobiotechnology 2024; 22: 457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Boudreau LH, Duchez A-C, Cloutier N, et al. Platelets release mitochondria serving as substrate for bactericidal group IIA-secreted phospholipase A2 to promote inflammation. Blood 2014; 124: 2173–2183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Duchez A-C, Boudreau LH, Naika GS, et al. Platelet microparticles are internalized in neutrophils via the concerted activity of 12-lipoxygenase and secreted phospholipase A2-IIA. Proc Natl Acad Sci U S A 2015; 112: E3564–E3573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Karbanová J, Deniz IA, Wilsch-Bräuninger M, et al. Extracellular lipidosomes containing lipid droplets and mitochondria are released during melanoma cell division. Cell Commun Signal 2024; 22: 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Abad E, Lyakhovich A. Movement of mitochondria with mutant DNA through extracellular vesicles helps cancer cells acquire chemoresistance. ChemMedChem 2022; 17: e202100642. [DOI] [PubMed] [Google Scholar]
- 56.Davis C-h. O, Kim K-Y, Bushong EA, et al. Transcellular degradation of axonal mitochondria. Proc Natl Acad Sci U S A 2014; 111: 9633–9638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.D’Souza A, Burch A, Dave KM, et al. Microvesicles transfer mitochondria and increase mitochondrial function in brain endothelial cells. J Controlled Release 2021; 338: 505–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang Z, Sheng H, Liao L, et al. Mesenchymal stem Cell-Conditioned medium improves mitochondrial dysfunction and suppresses apoptosis in okadaic Acid-Treated SH-SY5Y cells by extracellular vesicle mitochondrial transfer. J Alzheimers Dis 2020; 78: 1161–1176. [DOI] [PubMed] [Google Scholar]
- 59.Jusic A, Erpapazoglou Z, Dalgaard LT, et al. Guidelines for mitochondrial RNA analysis. Mol Ther Nucleic Acids 2024; 35: 102262. doi:DOI:10.1016/j.omtn.2024.102262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Murphy DE, De Jong OG, Evers MJW, et al. Natural or synthetic RNA delivery: a stoichiometric comparison of extracellular vesicles and synthetic nanoparticles. Nano Lett 2021; 21: 1888–1895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Guo J, Zhou F, Liu Z, et al. Exosome-shuttled mitochondrial transcription factor a mRNA promotes the osteogenesis of dental pulp stem cells through mitochondrial oxidative phosphorylation activation. Cell Prolif 2022; 55: e13324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hou Y, Lin S, Xia J, et al. Alleviation of ischemia-reperfusion induced renal injury by chemically modified SOD2 mRNA delivered via lipid nanoparticles. Mol Ther Nucleic Acids 2023; 34: 102067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Dalla Costa I, Buchanan CN, Zdradzinski MD, et al. The functional organization of axonal mRNA transport and translation. Nat Rev Neurosci 2021; 22: 77–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wu Y, Huang X, Tan Z, et al. FUS-mediated HypEVs: Neuroprotective effects against ischemic stroke. Bioact Mater 2023; 29: 196–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ma L, Singh J, Schekman R. Two RNA-binding proteins mediate the sorting of miR223 from mitochondria into exosomes. eLife 2023; 12: e85878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lee J-H, Shim Y-R, Seo W, et al. Mitochondrial double‐stranded RNA in exosome promotes interleukin‐17 production through toll‐like receptor 3 in alcohol‐associated liver injury. Hepatology 2020; 72: 609–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Farahani RA, Zhu X-Y, Tang H, et al. Metabolic syndrome alters the cargo of Mitochondria-Related microRNAs in swine mesenchymal stem Cell-Derived extracellular vesicles, impairing their capacity to repair the stenotic kidney. Stem Cells Int 2020; 2020: 8845635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhao M, Liu S, Wang C, et al. Mesenchymal stem cell-derived extracellular vesicles attenuate mitochondrial damage and inflammation by stabilizing mitochondrial DNA. ACS Nano 2021; 15: 1519–1538. [DOI] [PubMed] [Google Scholar]
- 69.Nagano S, Araki T. Axonal transport and local translation of mRNA in neurodegenerative diseases. Front Mol Neurosci 2021; 14: 697973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhang Y, Liu Z, Chopp M, et al. Small extracellular vesicles derived from cerebral endothelial cells with elevated microRNA 27a promote ischemic stroke recovery. Neural Regen Res 2025; 20: 224–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Antoniou A, Auderset L, Kaurani L, et al. Neuronal extracellular vesicles and associated microRNAs induce circuit connectivity downstream BDNF. Cell Rep 2023; 42: 112063. [DOI] [PubMed] [Google Scholar]
- 72.Lu J-X, Wang Y, Zhang Y-J, et al. Axonal mRNA localization and local translation in neurodegenerative disease. Neural Regen Res 2021; 16: 1950–1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yao PJ, Eren E, Goetzl EJ, et al. Mitochondrial electron transport chain protein abnormalities detected in plasma extracellular vesicles in alzheimer’s disease. Biomedicines 2021; 9: 1587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sayeed N, Sugaya K. Exosome mediated Tom40 delivery protects against hydrogen peroxide-induced oxidative stress by regulating mitochondrial function. Plos One 2022; 17: e0272511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Anand S, Foot N, Ang C-S, et al. Arrestin-domain containing protein 1 (Arrdc1) regulates the protein cargo and release of extracellular vesicles. Proteomics 2018; 18: e1800266. [DOI] [PubMed] [Google Scholar]
- 76.Leow MK-S, Rengaraj A, Narasimhan K, et al. Activated brown adipose tissue releases exosomes containing mitochondrial methylene tetrahydrofolate dehydrogenase (NADP dependent) 1-like protein (MTHFD1L). Biosci Rep 2022; 42BSR20212543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cereceda L, Cardenas JC, Khoury M, et al. Impact of platelet-derived mitochondria transfer in the metabolic profiling and progression of metastatic MDA-MB-231 human triple-negative breast cancer cells. Front Cell Dev Biol 2023; 11: 1324158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Picca A, Beli R, Calvani R, et al. Older adults with physical frailty and sarcopenia show increased levels of circulating small extracellular vesicles with a specific mitochondrial signature. Cells 2020; 9: 973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Jang SC, Crescitelli R, Cvjetkovic A, et al. Mitochondrial protein enriched extracellular vesicles discovered in human melanoma tissues can be detected in patient plasma. J Extracell Vesicles 2019; 8: 1635420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Picca A, Guerra F, Calvani R, et al. Mitochondrial signatures in circulating extracellular vesicles of older adults with parkinson’s disease: Results from the EXosomes in PArkiNson’s disease (EXPAND) study. J Clin Med 2020; 9: 504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Liu G, Wang Y, Pan Y, et al. Hypertonicity induces mitochondrial extracellular vesicles (MEVs) that activate TNF-α and β-catenin signaling to promote adipocyte dedifferentiation. Stem Cell Res Ther 2023; 14: 333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Shen M, Zhou L, Fan X, et al. Metabolic reprogramming of CD4+ T cells by mesenchymal stem Cell-Derived extracellular vesicles attenuates autoimmune hepatitis through mitochondrial protein transfer. Int J Nanomedicine 2024; 19: 9799–9819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Miller CE, Xu F, Zhao Y, et al. Hydrogen peroxide promotes the production of radiation-derived EVs containing mitochondrial proteins. Antioxidants 2022; 11: 2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Olsson I, Ronquist G. Nucleic acid association to human prostasomes. Arch Androl 1990; 24: 1–10. [DOI] [PubMed] [Google Scholar]
- 85.Thakur BK, Zhang H, Becker A, et al. Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res 2014; 24: 766–769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kahlert C, Melo SA, Protopopov A, et al. Identification of double-stranded genomic DNA spanning all chromosomes with mutated KRAS and p53 DNA in the serum exosomes of patients with pancreatic cancer*. J Biol Chem 2014; 289: 3869–3875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Attardi G, Schatz G. Biogenesis of mitochondria. Annu Rev Cell Biol 1988; 4: 289–333. [DOI] [PubMed] [Google Scholar]
- 88.Hsu C-C, Tseng L-M, Lee H-C. Role of mitochondrial dysfunction in cancer progression. Exp Biol Med (Maywood) 2016; 241: 1281–1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Guescini M, Genedani S, Stocchi V, et al. Astrocytes and glioblastoma cells release exosomes carrying mtDNA. J Neural Transm (Vienna) 2010; 117: 1–4. [DOI] [PubMed] [Google Scholar]
- 90.Cha M-Y, Kim DK, Mook-Jung I. The role of mitochondrial DNA mutation on neurodegenerative diseases. Exp Mol Med 2015; 47: e150–e150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lou C, Ma X, Chen Z, et al. The mtDNA fragments within exosomes might be novel diagnostic biomarkers of non-small cell lung cancer. Pathol Res Pract 2023; 249: 154718. [DOI] [PubMed] [Google Scholar]
- 92.Ye J, Duan C, Han J, et al. Peripheral mitochondrial DNA as a neuroinflammatory biomarker for major depressive disorder. Neural Regen Res 2025; 20: 1541–1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Keserű JS, Soltész B, Lukács J, et al. Detection of cell-free, exosomal and whole blood mitochondrial DNA copy number in plasma or whole blood of patients with serous epithelial ovarian cancer. J Biotechnol 2019; 298: 76–81. [DOI] [PubMed] [Google Scholar]
- 94.Arance E, Ramírez V, Rubio-Roldan A, et al. Determination of exosome mitochondrial DNA as a biomarker of renal cancer aggressiveness. Cancers (Basel) 2021; 14: 199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Hoekstra JG, Hipp MJ, Montine TJ, et al. Mitochondrial DNA mutations increase in early stage alzheimer disease and are inconsistent with oxidative damage. Ann Neurol 2016; 80: 301–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yeung KY, Dickinson A, Donoghue JF, et al. The identification of mitochondrial DNA variants in glioblastoma multiforme. Acta Neuropathol Commun 2014; 2: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Soltész B, Pös O, Wlachovska Z, et al. Mitochondrial DNA copy number changes, heteroplasmy, and mutations in plasma-derived exosomes and brain tissue of glioblastoma patients. Mol Cell Probes 2022; 66: 101875. [DOI] [PubMed] [Google Scholar]
- 98.Vaidya M, Sreerama S, Gaviria M, et al. Exposure to a pathological condition may Be required for the cells to secrete exosomes containing mtDNA aberration. J Nucleic Acids 2022; 2022: 7960198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhang X, Hubal MJ, Kraus VB. Immune cell extracellular vesicles and their mitochondrial content decline with ageing. Immun Ageing 2020; 17: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Li Y, Guo X, Guo S, et al. Next generation sequencing‑based analysis of mitochondrial DNA characteristics in plasma extracellular vesicles of patients with hepatocellular carcinoma. Oncol Lett 2020; 20: 2820–2828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sansone P, Savini C, Kurelac I, et al. Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancer. Proc Natl Acad Sci U S A 2017; 114: E9066–E9075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lazo S, Noren Hooten N, Green J, et al. Mitochondrial DNA in extracellular vesicles declines with age. Aging Cell 2021; 20: e13283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Cai Y, Xu M-J, Koritzinsky EH, et al. Mitochondrial DNA–enriched microparticles promote acute-on-chronic alcoholic neutrophilia and hepatotoxicity. JCI Insight 2017; 2: 2. doi:DOI:10.1172/jci.insight.92634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Jeon H, Lee J, Lee S, et al. Extracellular vesicles from KSHV-Infected cells stimulate antiviral immune response through mitochondrial DNA. Front Immunol 2019; 10: 876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Li Y-J, Liu R-P, Ding M-N, et al. Tetramethylpyrazine prevents liver fibrotic injury in mice by targeting hepatocyte-derived and mitochondrial DNA-enriched extracellular vesicles. Acta Pharmacol Sin 2022; 43: 2026–2041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Tsilioni I, Theoharides TC. Extracellular vesicles are increased in the serum of children with autism spectrum disorder, contain mitochondrial DNA, and stimulate human microglia to secrete IL-1β. J Neuroinflammation 2018; 15: 239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ma J, Cao H, Rodrigues RM, et al. Chronic-plus-binge alcohol intake induces production of proinflammatory mtDNA-enriched extracellular vesicles and steatohepatitis via ASK1/p38MAPKα-dependent mechanisms. JCI Insight 2020; 5 doi:DOI:10.1172/jci.insight.136496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Konaka H, Kato Y, Hirano T, et al. Secretion of mitochondrial DNA via exosomes promotes inflammation in behçet’s syndrome. Embo J 2023; 42: e112573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Venkat P, Chopp M. Exosome treatment for stroke with diabetic comorbidity. Neural Regen Res 2022; 17: 315–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Venkat P, Cui C, Chen Z, et al. CD133+exosome treatment improves cardiac function after stroke in type 2 diabetic mice. Transl Stroke Res 2021; 12: 112–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Fan B, Chopp M, Zhang ZG, et al. Treatment of diabetic peripheral neuropathy with engineered mesenchymal stromal cell-derived exosomes enriched with microRNA-146a provide amplified therapeutic efficacy. Exp Neurol 2021; 341: 113694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Manickam DS. Delivery of mitochondria via extracellular vesicles - A new horizon in drug delivery. J Control Release Off J Control Release Soc 2022; 343: 400–407. [DOI] [PubMed] [Google Scholar]
- 113.Rivera J, Gangwani L, Kumar S. Mitochondria localized microRNAs: an unexplored miRNA niche in Alzheimer’s disease and aging. Cells 2023; 12: 742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Garcia-Martin R, Wang G, Brandão BB, et al. MicroRNA sequence codes for small extracellular vesicle release and cellular retention. Nature 2022; 601: 446–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ghoshal B, Bertrand E, Bhattacharyya SN. Non-canonical argonaute loading of extracellular vesicle-derived exogenous single-stranded miRNA in recipient cells. J Cell Sci 2021; 134: jcs253914. [DOI] [PubMed] [Google Scholar]
- 116.Sassano ML, van Vliet AR, Agostinis P. Mitochondria-associated membranes as networking platforms and regulators of cancer cell fate. Front Oncol 2017; 7: 174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Missiroli S, Patergnani S, Caroccia N, et al. Mitochondria-associated membranes (MAMs) and inflammation. Cell Death Dis 2018; 9: 329–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Yu H, Sun C, Gong Q, et al. Mitochondria-Associated endoplasmic reticulum membranes in breast cancer. Front Cell Dev Biol 2021; 9: 629669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zou X, Kwon SH, Jiang K, et al. Renal scattered tubular-like cells confer protective effects in the stenotic murine kidney mediated by release of extracellular vesicles. Sci Rep 2018; 8: 1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Frankenberg Garcia J, Rogers AV, Mak JCW, et al. Mitochondrial transfer regulates bioenergetics in healthy and chronic obstructive pulmonary disease airway smooth muscle. Am J Respir Cell Mol Biol 2022; 67: 471–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Zheng D, Zhou H, Wang H, et al. Mesenchymal stem cell-derived microvesicles improve intestinal barrier function by restoring mitochondrial dynamic balance in sepsis rats. Stem Cell Res Ther 2021; 12: 299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Dutra Silva J, Su Y, Calfee CS, et al. Mesenchymal stromal cell extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur Respir J 2021; 58 doi:DOI:10.1183/13993003.02978-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ikeda G, Santoso MR, Tada Y, et al. Mitochondria-Rich extracellular vesicles from autologous stem Cell-Derived cardiomyocytes restore energetics of ischemic myocardium. J Am Coll Cardiol 2021; 77: 1073–1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Veilleux V, Pichaud N, Boudreau LH, et al. Mitochondria transfer by Platelet-Derived microparticles regulates breast cancer bioenergetic states and malignant features. Mol Cancer Res MCR 2024; 22: 268–281. [DOI] [PubMed] [Google Scholar]
- 125.Hu Z, Wang D, Gong J, et al. MSCs deliver hypoxia-treated mitochondria reprogramming acinar metabolism to alleviate severe acute pancreatitis injury. Adv Sci 2023; 10: 2207691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Nicolás-Ávila JA, Lechuga-Vieco AV, Esteban-Martínez L, et al. A network of macrophages supports mitochondrial homeostasis in the heart. Cell 2020; 183: 94–109.e23. [DOI] [PubMed] [Google Scholar]
- 127.Letsiou E, Teixeira Alves LG, Fatykhova D, et al. Microvesicles released from pneumolysin-stimulated lung epithelial cells carry mitochondrial cargo and suppress neutrophil oxidative burst. Sci Rep 2021; 11: 9529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hough KP, Trevor JL, Chacko BK, et al. Small extracellular vesicle signaling and mitochondrial transfer reprograms T helper cell function in human asthma. BioRxiv Prepr Serv Biol 2024; 2024.04.30: 589227. [Google Scholar]
- 129.Morrison TJ, Jackson MV, Cunningham EK, et al. Mesenchymal stromal cells modulate macrophages in clinically relevant lung injury models by extracellular vesicle mitochondrial transfer. Am J Respir Crit Care Med 2017; 196: 1275–1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Yao W-D, Zhou J-N, Tang C, et al. Hydrogel microneedle patches loaded with stem cell mitochondria-enriched microvesicles boost the chronic wound healing. ACS Nano 2024; 18: 26733–26750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Xia L, Zhang C, Lv N, et al. AdMSC-derived exosomes alleviate acute lung injury via transferring mitochondrial component to improve homeostasis of alveolar macrophages. Theranostics 2022; 12: 2928–2947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Ko JH, Kim HJ, Jeong HJ, et al. Mesenchymal stem and stromal cells harness Macrophage-Derived amphiregulin to maintain tissue homeostasis. Cell Rep 2020; 30: 3806–3820.e6. [DOI] [PubMed] [Google Scholar]
- 133.Gao Y, Mi N, Wu W, et al. Transfer of inflammatory mitochondria via extracellular vesicles from M1 macrophages induces ferroptosis of pancreatic beta cells in acute pancreatitis. J Extracell Vesicles 2024; 13: e12410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.O'Brien CG, Ozen MO, Ikeda G, et al. Mitochondria-rich extracellular vesicles rescue patient-specific cardiomyocytes from doxorubicin injury: insights into the SENECA trial. JACC Cardiooncol 2021; 3: 428–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Rosina M, Ceci V, Turchi R, et al. Ejection of damaged mitochondria and their removal by macrophages ensure efficient thermogenesis in brown adipose tissue. Cell Metab 2022; 34: 533–548.e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Baysa A, Fedorov A, Kondratov K, et al. Release of mitochondrial and nuclear DNA during on-pump heart surgery: kinetics and relation to extracellular vesicles. J Cardiovasc Transl Res 2019; 12: 184–192. [DOI] [PubMed] [Google Scholar]
- 137.Holvoet P, Vanhaverbeke M, Bloch K, et al. Low MT‐CO1 in monocytes and microvesicles is associated with outcome in patients with coronary artery disease. J Am Heart Assoc 2016; 5: e004207. [DOI] [PMC free article] [PubMed] [Google Scholar]