INTRODUCTION
Although all types of diabetes result in hyperglycemia, the pathophysiology of each type of diabetes is different. These guidelines summarize available data specific to the comprehensive care of youth with type 2 diabetes. The objective is to enrich the recognition of type 2 diabetes in youth, its risk factors, its pathophysiology, its management, and the prevention of associated complications.
PATHOPHYSIOLOGY
Glucose homeostasis is maintained by a balance between insulin secretion from the pancreatic β-cells and sensitivity to insulin in skeletal muscle, adipose tissue, and liver (1). When insulin sensitivity declines, insulin secretion must increase to maintain glucose tolerance, and, in most youth, decreased insulin sensitivity due to puberty and/or obesity is compensated by increased insulin secretion. However, when β-cells cannot secrete sufficient insulin to compensate for insulin resistance, abnormalities in glucose homeostasis ensue, potentially progressing to prediabetes and type 2 diabetes as β-cell function deteriorates further (2–9). The relationship between β-cell function and insulin sensitivity in adults and youth has been demonstrated to be a hyperbolic function and can be described mathematically as the product of insulin sensitivity and β-cell function, called the disposition index (DI) (1). The DI essentially expresses the amount of insulin being secreted relative to the degree of insulin resistance and is a constant for a given degree of glucose tolerance in any one individual.
Overweight and obesity are major acquired contributors to the development of insulin resistance, particularly in the face of the physiologic insulin resistance characteristic of puberty. Robust pancreatic β-cell compensatory insulin secretion maintains normal glucose homeostasis. However, in adolescents with obesity who develop type 2 diabetes, there is severe peripheral and hepatic insulin resistance, with ∼50% lower peripheral insulin sensitivity than peers with obesity without diabetes, along with increased fasting hepatic glucose production and inadequate first- and second-phase insulin secretion, resulting in ∼85% lower DI (2). Additional abnormalities in youth with type 2 diabetes include impaired glucose sensitivity of insulin secretion, lower serum adiponectin concentrations, and reduced incretin effect (3,9–13). While upregulation of α-cell function with hyperglucagonemia has been implicated in the pathophysiology of type 2 diabetes in adults (14,15), there are limited data in youth with type 2 diabetes, with studies showing either hyperglucagonemia or no difference from control subjects without diabetes (3,11,16,17).
Cross-sectional and longitudinal studies in youth with obesity along the spectrum of glycemia from normoglycemia to prediabetes to type 2 diabetes show, as in adults, that β-cell failure with declining insulin secretion relative to insulin sensitivity results in prediabetes and type 2 diabetes in high-risk youth (5–9,18–21). Importantly, however, prior to reaching the American Diabetes Association (ADA)-defined fasting and oral glucose tolerance test (OGTT)-stimulated glycemic cut points for the diagnosis of prediabetes, youth, like adults, already demonstrate declining β-cell function relative to insulin sensitivity (6–8). Also, youth with A1C in the at-risk/prediabetes category (≥5.7 to <6.5%) demonstrate impaired β-cell function compared with those with A1C <5.7% (22). A combination of obesity, genetics, the hormonal milieu, incretins and/or their effect, and metabolic alterations, such as glucotoxicity and/or lipotoxicity, are likely to contribute to deteriorating β-cell function against the backdrop of insulin resistance, eventually culminating in prediabetes and type 2 diabetes in at-risk youth. Based on the baseline data from the Restoring Insulin Secretion (RISE) study (23,24), there appear to be important differences in insulin sensitivity and β-cell function between youth and adults with similar degrees of dysglycemia, including greater insulin resistance for any degree of adiposity and greater insulin secretion for any degree of insulin resistance in youth compared with adults.
RISK, SCREENING, AND DIAGNOSIS
Risk Factors
Nonmodifiable risk factors for youth-onset type 2 diabetes include genetics/epigenetics, manifested as a strong family history of type 2 diabetes in first- or second-degree relatives; being the offspring of a pregnancy complicated by gestational diabetes mellitus (GDM); minority race/ethnicity; and physiologic insulin resistance of puberty. Metabolic evidence of genetic susceptibility can be detected in the first decade of life, manifested as impaired insulin sensitivity and reduced insulin secretion in otherwise healthy youth with a family history of type 2 diabetes (25). This genetic susceptibility, when combined with environmental factors conducive to obesity and a sedentary lifestyle, may ultimately translate to type 2 diabetes. Indeed, in a study of youth with obesity, a genetic risk score for β-cell dysfunction from five single nucleotide polymorphisms was associated with a higher chance of prediabetes and type 2 diabetes (26). Dozens of specific genetic variants linked to type 2 diabetes have been identified in adults (27,28), but these only account for about 10% of its heritability (29,30). Particular genetic variants that predispose to diabetes in youth have been identified in Oji-Cree Native Canadians (31) and African American youth (32), but information in other populations is only now emerging.
Evidence from both animal and human studies suggests that maternal obesity and GDM contribute to obesity and type 2 diabetes in youth (33,34). In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, one-third were born after a pregnancy complicated by preexisting diabetes or GDM (35). In the SEARCH for Diabetes in Youth (SEARCH) study, a population-based study of the epidemiology of type 1 and type 2 diabetes in youth in the U.S., exposure to maternal GDM or pregestational diabetes and maternal obesity were independently associated with type 2 diabetes in adolescents, with intrauterine exposure to these two risk factors present in 47.2% of type 2 diabetes in the cohort (36). Age of onset of type 2 diabetes was also younger in those exposed to diabetes during gestation.
Incidence and prevalence of type 2 diabetes are highest among youth from a minority race/ethnicity (37), likely as a consequence of many factors, including genetics, metabolic characteristics, cultural/environmental influences, and quality of and access to health care. Several studies have demonstrated significant differences by race/ethnicity in insulin sensitivity and secretion that might heighten the risk of type 2 diabetes (38–42).
Type 2 diabetes typically occurs in adolescents at midpuberty (for example, the mean age of diagnosis was 14 years in the TODAY study) (43), most likely precipitated by the physiologic, but transient, pubertal insulin resistance aggravating the preexisting metabolic challenges of obesity. Cross-sectional and longitudinal studies show that insulin sensitivity declines by 25–30% as youth transition from prepuberty to puberty (44–46). In the presence of normally functioning β-cells, puberty-related insulin resistance is compensated by increased insulin secretion/hyperinsulinemia, such that DI remains normal. In youth who are predisposed to develop prediabetes and/or type 2 diabetes, β-cell compensation is inadequate with progressive decline in the DI, ultimately resulting in dysglycemia (46,47).
In youth-onset type 2 diabetes, the major modifiable risk factors are obesity and lifestyle habits of excess nutritional intake, low physical activity, and increased sedentary behaviors with decreased energy expenditure, resulting in the surplus of energy being stored as body fat. Other potentially modifiable risk factors for type 2 diabetes in adolescents and young adults include chronic stress and/or depressed mood (48,49) and sleep-related disorders (50–52).
Risk Assessment and Diagnostic Criteria
Recommendations
Risk-based screening for prediabetes and/or type 2 diabetes should be considered after the onset of puberty or after 10 years of age, whichever occurs earlier, in children and adolescents who are overweight (BMI ≥85th percentile) or obese (BMI ≥95th percentile) and who have one or more additional risk factors for diabetes (see Table 1 for evidence grading).
If tests are normal, repeat testing at a minimum of 3-year intervals E, or more frequently if BMI is increasing. C
Fasting plasma glucose, 2-h plasma glucose after 75-g OGTT, or A1C can be used to test for prediabetes or diabetes. B
Table 1.
Risk-based screening for type 2 diabetes or prediabetes in asymptomatic children and adolescents* in a clinical setting
| Criteria |
|---|
| Testing should be considered in youth* who are overweight (≥85%) or obese (≥95%) A and who have one or more additional risk factors based on the strength of their association with diabetes: |
| • Maternal history of diabetes or GDM during the child's gestation A |
| • Family history of type 2 diabetes in first- or second-degree relative A |
| • Race/ethnicity (Native American, African American, Latino, Asian American, Pacific Islander) A |
| • Signs of insulin resistance or conditions associated with insulin resistance (acanthosis nigricans, hypertension, dyslipidemia, polycystic ovary syndrome, or small-for-gestational-age birth weight) B |
After the onset of puberty or after 10 years of age, whichever occurs earlier.
Risk-based screening for prediabetes and/or type 2 diabetes is timed after the onset of puberty or after 10 years of age, whichever occurs earlier, because the majority of youth-onset type 2 diabetes occurs during puberty, as stated above, and rarely in prepubertal children. However, some youth with obesity may have earlier onset of puberty than usual, necessitating screening before 10 years of age. In addition, in North America almost all youth with type 2 diabetes are overweight/obese, hence the recommendation to screen youth with overweight/obesity. In other parts of the world where youth with type 2 diabetes are not necessarily overweight and/or obese, clinical judgment should guide whom to screen. Although there is no robust evidence-based rationale for the proposed frequency of testing, increasing BMI has been shown to be a predictor of deteriorating glycemia and progression to type 2 diabetes (21). Therefore, clinicians caring for youth with overweight/obesity with continued increase in their BMI should be aware of the need for more frequent screening.
The laboratory glycemia-based diagnostic criteria for diabetes and prediabetes are the same for youth and adults, regardless of type of diabetes (Table 2) (53). However, these criteria are extrapolated from adults, and the epidemiological studies that formed the basis for both glucose and A1C definitions of diabetes did not include pediatric populations. Therefore, the exact relevance of these definitions for pediatric populations remains unclear until more data become available.
Table 2.
Criteria for the diagnosis of prediabetes and diabetes
| Prediabetes |
|---|
| A1C 5.7% to <6.5% (39 to <48 mmol/mol). The test should be performed in a laboratory using a method that is NGSP certified and standardized to the DCCT assay. |
| IFG: fasting glucose ≥100 but <126 mg/dL (≥5.6 but <7.0 mmol/L). |
| IGT: 2-h plasma glucose ≥140 but <200 mg/dL (≥7.8 but <11.1 mmol/L) during an OGTT. The test should be performed as described by the World Health Organization, using a glucose load containing the equivalent of 1.75 mg/kg (max 75 g) anhydrous glucose dissolved in water.* |
| Diabetes |
| A1C ≥6.5% (≥48 mmol/mol). The test should be performed in a laboratory using a method that is NGSP certified and standardized to the DCCT assay.* |
| OR |
| FPG ≥126 mg/dL (7.0 mmol/L). Fasting is defined as no caloric intake for at least 8 h.* |
| OR |
| 2-h plasma glucose ≥200 mg/dL (11.1 mmol/L) during an OGTT. The test should be performed as described by the World Health Organization, using a glucose load containing the equivalent of 1.75 mg/kg (max 75 g) anhydrous glucose dissolved in water* |
| OR |
| In a patient with classic symptoms of hyperglycemia or hyperglycemic crisis, a random plasma glucose >200 mg/dL (11.1 mmol/L). |
FPG, fasting plasma glucose; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; max, maximum.
In the absence of unequivocal hyperglycemia, result should be confirmed by repeat testing.
The A1C test is universally available and can be performed any time of the day without need for fasting. However, several studies have questioned its validity in the pediatric population because of poor sensitivity for identifying children with dysglycemia and underestimation of the prevalence of prediabetes and diabetes (54–56). Fasting and OGTT criteria have not been validated in youth, either. Studies using continuous glucose monitoring (CGM) in youth with obesity demonstrated that A1C and OGTT are equally effective at identifying glycemic abnormalities on CGM, but the glycemic patterns differ (57); abnormal A1C was associated with higher overall and nighttime average glucose on CGM, while abnormal OGTT was associated with more time spent above the normal glucose range during the day. Institution of A1C screening in a large primary care network increased provider adherence to screening recommendations compared with OGTT screening while identifying the same prevalence of type 2 diabetes (58). Furthermore, in this cohort, the progression to clinically confirmed diabetes was substantially more likely for those with A1C >6% (18.4%) than for those with levels 5.7–6.0% (1.3%). Therefore, screening with fasting glucose, OGTT, or A1C is an acceptable approach but should be based on sound clinical judgment, recognition of the strengths and weaknesses of each test, and the facilities and resources available.
Confirming Diabetes Type
Recommendations
Children and adolescents with overweight/obesity in whom the diagnosis of type 2 diabetes is being considered should have a panel of pancreatic autoantibodies tested to exclude the possibility of autoimmune type 1 diabetes. B
Genetic evaluation to exclude monogenic diabetes should also be based on clinical characteristics and presentation. B
As stated above, youth with type 2 diabetes in the U.S. are characteristically overweight and/or obese, in mid- to late puberty, with overrepresentation of minority ethnic/racial groups and females (4,43,59). The clinical presentation varies widely from asymptomatic or minimally symptomatic, diagnosed incidentally during routine laboratory testing, to a severe presentation with symptomatic hyperglycemia, weight loss, metabolic decompensation, diabetic ketoacidosis (DKA), or hyperglycemic hyperosmolar nonketotic (HHNK) syndrome (4).
Obesity is a consistent feature of youth-onset type 2 diabetes in the U.S. However, because of the escalating rates of obesity in the general population, children with both type 1 diabetes and monogenic diabetes are also more likely to be overweight/obese than in the past (60), making the clinical distinction between type 2 diabetes and obese type 1 or monogenic diabetes difficult. This was illustrated in the TODAY study in which, of the 1,206 youth clinically diagnosed with type 2 diabetes and screened for circulating GAD65 and IA2 antibodies, 118 (9.8%) were antibody positive (Ab+) (61). Even though these Ab+ individuals had clinical characteristics that overlapped with the antibody-negative (Ab−) youth, they were less likely to be obese, have features of metabolic syndrome, have a family history of diabetes, be female, or be from a minority race/ethnicity, indicating a phenotype more similar to their peers with type 1 diabetes. Pathophysiologically, Ab− youth with obesity are more insulin resistant than Ab+ youth with obesity, while Ab+ youth have more severe insulin deficiency (61–64). Fasting and stimulated C-peptide are significantly lower in Ab+ youth with obesity and diabetes, though with appreciable overlap (63). Moreover, Ab− youth are more likely to exhibit features of the metabolic syndrome (elevated systolic blood pressure and ALT), while Ab+ youth have significantly more frequent ketonuria at initial presentation (61,64). The reported rates of positive pancreatic autoantibodies in youth clinically diagnosed with type 2 diabetes vary from 10% to 75% (4,62), likely depending on the ratio of type 1 and type 2 diabetes in the population. The clinical distinction between youth with type 2 diabetes and youth with obesity and type 1 or monogenic diabetes is further blurred because youth with type 2 diabetes often present with some degree of ketosis, including DKA (65).
The distinction between these forms of diabetes in youth with obesity has important implications for treatment (66), since Ab+ youth present more like individuals with type 1 diabetes, progressing to insulin requirement more rapidly (61), and are at risk for other autoimmune disorders. Therefore, measurement of pancreatic autoantibodies is recommended in all youth with clinical characteristics of type 2 diabetes. This testing should include GAD65 and IA2 antibodies, along with insulin autoantibody in individuals who have not yet been exposed to exogenous insulin. The benefit of measurement of ZnT8 antibody in individuals with phenotypic type 2 diabetes is not yet clear.
We further recommend that antibodies be measured in a laboratory aligned with the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) Pancreatic Autoantibody Standardization Program because currently available commercial assays may not be sufficiently sensitive or specific. However, in all cases, clinical judgment and the presence of other risk factors for type 1 diabetes or type 2 diabetes should be considered in making the diagnosis, and the health care team should remain open to reconsidering the initial diagnosis. Since 4.5–8.0% of youth with clinical features suggestive of type 2 diabetes have been found to have monogenic diabetes, genetic testing for monogenic forms of diabetes should be considered as well (67–69).
GLYCEMIC TARGETS
Recommendations
A1C should be measured every 3 months. E
A reasonable A1C goal for most children and adolescents with type 2 diabetes treated with oral agents alone is <7%. More stringent A1C goals (such as <6.5%) may be appropriate for selected individual patients if they can be achieved without significant hypoglycemia or other adverse effects of treatment. Appropriate patients might include those with short duration of diabetes and lesser degrees of β-cell dysfunction and patients treated with lifestyle or metformin only who achieve significant weight improvement. E
A1C targets for youth on insulin should be individualized, taking into account the relatively low rate of hypoglycemia in youth-onset type 2 diabetes. E
Home self-monitoring of blood glucose (SMBG) regimens should be individualized, taking into consideration the pharmacologic treatment of the patient. E
Previous target A1C guidelines by the ADA and the International Society for Pediatric and Adolescent Diabetes for youth with type 2 diabetes ranged from <6.5% to <7.0% (70,71) and <7.5% (72), mostly based on expert opinion and extrapolated from youth with type 1 diabetes and adults with type 2 diabetes. However, accumulating evidence provides support for more appropriate goals. The TODAY study showed that hypoglycemia is rare in adolescents with type 2 diabetes, even with insulin therapy (73), suggesting that more stringent A1C targets are acceptable. Also in TODAY, individuals with an A1C of >6.3% after 3 months of metformin or an increasing A1C, even in the nondiabetes range (74), had a substantially increased risk for loss of glycemic control, likely reflecting a greater degree of β-cell dysfunction (75,76). Furthermore, individuals with youth-onset type 2 diabetes have high rates of complications (77–79), many of which are associated with poor glycemic control, and rapid deterioration with increasing A1C. Finally, youth with type 2 diabetes can be expected to have long disease duration and, therefore, continued risk for accumulation of glycemia-related complications. Taken together, this evidence suggests that a more stringent A1C target can and should be attained in youth with type 2 diabetes.
The evidence is insufficient regarding the value of SMBG and how often testing should be performed by youth with type 2 diabetes not on insulin therapy. Until such data become available, the frequency of SMBG should be individualized, taking into account patient and family burden, the value of the information obtained and how it will be used to adjust therapy, and the associated hypoglycemia risk.
LIFESTYLE MANAGEMENT
Diabetes Education and Self-Management Skills
Recommendation
All youth with type 2 diabetes and their families should receive comprehensive diabetes self-management education/support that is specific to youth with type 2 diabetes and is culturally competent. B
It has been well established that diabetes education is necessary, but not sufficient, to enhance self-management in people with diabetes (80,81). The majority of these studies, however, focused on adults with type 2 diabetes and/or youth with type 1 diabetes. Since the population of youth with type 2 diabetes is more likely to be of minority ethnic/racial background than those with type 1 diabetes, and materials developed for adults may not address issues of development in youth, culturally appropriate programs specific to youth with type 2 diabetes and their families are necessary. Unfortunately, there are no randomized clinical trials of education and support programs for youth with type 2 diabetes. Nonetheless, descriptive reports suggest that programs that focus on building knowledge and skills appropriate to this population are important in ensuring adequate self-management.
In the TODAY trial (81), the diabetes education program included content about type 2 diabetes physiology and treatment, building skills of healthy eating habits, carbohydrate counting, portion sizes, reading food labels, glucose monitoring, and ketone testing, as well as problem solving, risk reduction, and living with diabetes. Full mastery of the program was achieved in an average of 5.5 90-min sessions. Factors associated with shorter time to mastery included more recent diagnosis and not having to use a translator, while sex, primary language of the youth and family, individual versus group sessions, or site of delivery were not. These program materials are available from the ADA as “Be Healthy Today” (82). Given the lack of clinical trials of various educational approaches, it is unclear that this program is superior to other approaches. Nonetheless, the program provides effective, engaging materials for youth with type 2 diabetes that were designed specifically for this population. Until comparative trials of various approaches are completed, diabetes education using these materials is appropriate (83).
Psychosocial Factors
Recommendations
Providers should assess social context, including potential food insecurity, housing stability, and financial barriers, and apply that information to treatment decisions. E
Use patient-appropriate standardized and validated tools to assess diabetes distress and mental/behavioral health in youth with type 2 diabetes, with attention to symptoms of depression and disordered eating behaviors, and refer to specialty care when indicated. B
When choosing glucose-lowering or other medications for youth with overweight/obesity and type 2 diabetes, consider medication adherence and treatment effects on weight. E
Starting at puberty, preconception counseling should be incorporated into routine diabetes clinic visits for all females of childbearing potential. A
Patients should be screened for smoking and alcohol use at diagnosis and regularly thereafter. C
The ADA position statement on the provision of psychosocial care for people living with diabetes recognizes the profound influence of psychosocial factors on health outcomes and well-being (84). The recommendations herein are consistent with those outlined in that position statement, an important resource for more detailed information about life-course issues and assessment of psychosocial comorbidities.
Most youth with type 2 diabetes come from racial and ethnic minority groups, have low socioeconomic status, and have a family history of diabetes (37,85,86). Families often experience multiple stressors including food insecurity, employment and housing instability, and difficulties with access to treatment; youth also may have been exposed to early adversity, which has been shown to affect health over time (87). Providers should personalize approaches to diabetes management to minimize barriers to care, enhance adherence, and maximize response to treatment by taking into consideration the sociocultural context of the patient and their family.
Youth with type 1 diabetes have high rates of diabetes distress and psychiatric symptoms and diagnoses (in particular, depression and disordered eating behaviors) necessitating ongoing surveillance of mental and behavioral health. Evidence about psychiatric disorders and symptoms in youth with type 2 diabetes is limited (88–92), but given the sociocultural context and the medical burden, as well as preexisting obesity-associated comorbidities together with type 2 diabetes, ongoing surveillance of mental health/behavioral health is also indicated in youth with type 2 diabetes.
Symptoms of depression and disordered eating are common in youth with type 2 diabetes and associated with poorer glycemic control (89). The prevalence of clinically significant symptoms of depression among youth with type 2 diabetes was reported to be 8.6% in the SEARCH cohort of youth with type 1 and type 2 diabetes (89) and 14.8% in the TODAY cohort of youth with type 2 diabetes (93). In addition, more than 25% of females and males in the SEARCH cohort of youth with type 2 diabetes reported symptoms of disordered eating behaviors, such as skipping insulin, vomiting, and using diet pills or laxatives, and these behaviors were associated with poorer glycemic control in females (89). Binge eating rates in the TODAY cohort were high (26%) and were associated with more severe obesity, psychological symptoms of disordered eating, and symptoms of depression (94).
More research is needed to evaluate rates of diagnosable psychiatric disorders, trauma, victimization, and psychotropic drug use in youth with type 2 diabetes. It also is important to elucidate the relationships among obesity, psychiatric disorders, and medication regimens because many of the drugs prescribed for diabetes and psychiatric disorders are associated with weight gain and increased concerns about eating, shape, and weight (95,96).
Finally, in accord with the ADA’s Standards of Medical Care in Diabetes—2018 (97), preconception counseling should be provided starting at puberty for all girls of childbearing potential in order to increase understanding of risk related to diabetes and improve health prior to conception. In the TODAY study (98), despite counseling on pregnancy reduction designed specifically for youth with type 2 diabetes, 10.2% of the females in the cohort became pregnant over an average of 3.8 years of study participation. Of note, 26.4% of pregnancies ended in a miscarriage, stillbirth, or intrauterine death, and 20.5% of the live-born infants had a major congenital anomaly. These data confirm the importance of educating young women with type 2 diabetes to time their pregnancies to reduce risks to themselves and their offspring. More research regarding pregnancy outcomes in youth with type 2 diabetes is needed.
Lifestyle Modification, Weight Management, Exercise, and Nutrition
Recommendations
Youth with overweight/obesity and type 2 diabetes and their families should be provided with developmentally and culturally appropriate comprehensive lifestyle programs that are integrated with diabetes management aiming to achieve 7–10% decrease in excess weight. C
Given the necessity of long-term weight control and lifestyle management for children and adolescents with type 2 diabetes, lifestyle intervention should be based on a chronic care model and offered in the context of diabetes care. E
Youth with diabetes, like all children, should be encouraged to participate in at least 30–60 min of moderate to vigorous physical activity at least 5 days per week (and strength training on at least 3 days per week) B and should be encouraged to decrease sedentary behavior. C
Nutrition for youth with type 2 diabetes, like all children, should focus on healthy eating patterns that emphasize consumption of nutrient-dense, high-quality foods and decrease consumption of calorie-dense, nutrient-poor foods, particularly sugar-added beverages. B
The utility of pharmacotherapy for weight reduction in youth with type 2 diabetes remains limited in the absence of approved, effective, and safe medications and the lack of clinical trials in youth with type 2 diabetes. B
Lifestyle modification programs that incorporate evidence-informed behavioral strategies to promote changes in diet and physical activity (99) are a cornerstone of treatment for adults with type 2 diabetes because the resulting reductions of 5–7% of initial body weight are associated with improvements in blood glucose levels and other risk parameters. Much less is known about the impact of lifestyle interventions in youth with type 2 diabetes, although 90% are overweight or obese. Family-based behavioral weight management programs in school-aged children without diabetes have a modest, but positive, impact on weight and cardiometabolic risk factors but are less effective in adolescents and children with more severe obesity (100–102). Intensive weight management, when compared with usual treatment, can have sustained benefits over a 2-year period for ethnically and racially diverse inner-city children and adolescents with an average BMI >35 (102,103). Although BMI changes in treated youth were modest (103), those who received usual care showed increases in BMI over the period of observation, while the intervention group had continued improvements in body composition and insulin resistance relative to those who did not receive weight management.
The most pertinent evidence regarding the impact of lifestyle interventions for youth with type 2 diabetes comes from the TODAY study (104), where the goal was to achieve 7–10% decrease in percent overweight. The addition of lifestyle intervention to metformin monotherapy was not associated with durable metabolic control beyond that of metformin alone. Youth receiving metformin plus lifestyle intervention showed short-term, but not sustained, weight loss and improvements in body composition relative to those in the two other intervention groups (105). While 31% of youth who received lifestyle intervention achieved the preplanned goal of a decrease of ≥7% in percent overweight through 24 months of intervention, this result did not differ significantly from that obtained with metformin monotherapy and no predictors of successful weight loss were identified. However, irrespective of treatment assignment, sustained weight losses ≥7% of excess body weight were associated with improvements in A1C, HDL, and C-peptide (105), indicating that obesity management remains a crucial goal.
Components of a comprehensive pediatric lifestyle intervention are well established (106,107), including those for youth with severe obesity (108). These include the involvement of family at a developmentally appropriate level and evidence-based behavioral strategies to facilitate enduring changes in nutrition and physical activity. Guidelines for physical activity and nutrition are based on those recommended by the American Academy of Pediatrics (2007) (107) and the Endocrine Society (2017) (106). Youth with type 2 diabetes frequently have severe obesity, and it is particularly important that behavior change goals for diet and activity incorporate stepwise, achievable targets developed in conjunction with the youth and family members, as appropriate.
Youth with type 2 diabetes will face increasing severity of obesity and diabetes complications as they age (109–111). An important first step is to integrate diabetes care and education, such as the approach used in the TODAY trial, with ongoing lifestyle intervention for obesity management (106) to maximize the impact of medical and lifestyle interventions over time. Comprehensive chronic care models have been recommended for youth with obesity and chronic illness (112,113).
With the exception of orlistat, weight loss medications are not approved for use in youth. The Endocrine Society guidelines for pediatric obesity (106) review the limited evidence for effectiveness of current weight-loss medications and recommends that their use be restricted to the research setting. More research into possible pharmacologic approaches to augment lifestyle interventions and their role in type 2 diabetes in youth is urgently needed.
Pharmacologic Approaches to Glycemic Management
Recommendations
Initiate pharmacologic therapy, in addition to lifestyle therapy, at diagnosis of type 2 diabetes. A
In incidentally diagnosed or metabolically stable patients (A1C <8.5% and asymptomatic), metformin is the initial pharmacologic treatment of choice if renal function is normal. A
Youth with marked hyperglycemia (blood glucose ≥250 mg/dL, A1C ≥8.5%) without acidosis at diagnosis who are symptomatic with polyuria, polydipsia, nocturia, and/or weight loss should be treated initially with basal insulin while metformin is initiated and titrated. B
In patients with ketosis/ketoacidosis, treatment with subcutaneous or intravenous insulin should be initiated to rapidly correct the hyperglycemia and the metabolic derangement. Once acidosis is resolved, metformin should be initiated while subcutaneous insulin therapy is continued. A
In individuals presenting with severe hyperglycemia (blood glucose ≥600 mg/dL), assess for HHNK syndrome. A
In patients initially treated with insulin and metformin who are meeting glucose targets based on home blood glucose monitoring, insulin can be tapered over 2–6 weeks by decreasing the insulin dose 10–30% every few days. B
If the glycemic target is no longer met using metformin alone, or if contraindications or intolerable side effects of metformin develop, basal insulin therapy should be initiated. B
If the combination of metformin plus basal insulin is ineffective at achieving or maintaining glycemic targets, more intensive approaches to insulin therapy may be initiated. E
The use of nonapproved medications in youth with type 2 diabetes is not recommended outside of research trials. B
In the clinical setting, only a minority of youth with type 2 diabetes are on lifestyle management alone (114,115) because it is often inadequate for achieving and maintaining the desired level of glycemic control and BMI improvement, with the percentage of patients remaining on lifestyle intervention alone declining further by 1 year (115). Therefore, in most cases, the addition of pharmacologic intervention early in the disease is warranted. As in adults, the pharmacologic intervention should be a stepped process. However, since only metformin and insulin are currently approved for the treatment of diabetes in patients under 18 years old, the approach in youth is more limited.
Initial Treatment
Initial treatment of youth-onset type 2 diabetes should include metformin and/or insulin alone or in combination, based on the metabolic status of the patient. Initial treatment of the youth with obesity and diabetes must take into account that diabetes type is often uncertain in the first few weeks of treatment owing to overlap in presentation and that a substantial percentage of youth with type 2 diabetes will present with clinically significant ketoacidosis (65). Therefore, immediate therapy should address the hyperglycemia and associated metabolic derangements irrespective of ultimate diabetes type, with adjustment of therapy once metabolic compensation has been established and subsequent information, such as antibody results, becomes available.
Figure 1 provides an approach to initial treatment.
Figure 1.
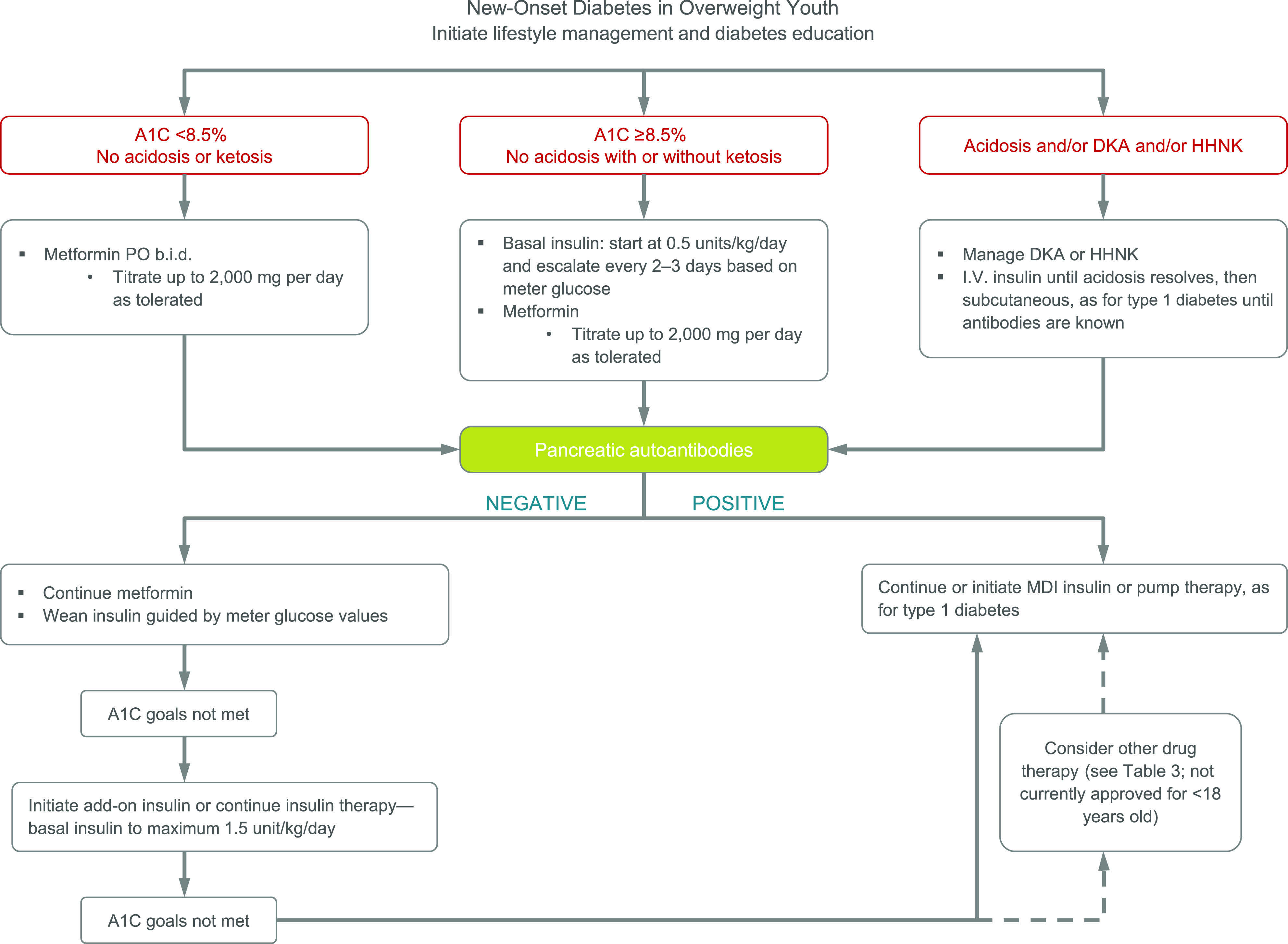
Management of new-onset diabetes in overweight youth suspected to have type 2 diabetes based on risk factors listed in Table 1. MDI, multiple daily injections.
Metformin
Metformin is the preferred drug for initial treatment of type 2 diabetes in adults and youth. In the TODAY study, 48.3% of youth with type 2 diabetes who were enrolled, with less than 2 years (median 8 months) of diabetes duration, maintained adequate glycemic control (A1C <8.0%) on metformin alone for up to 6 years (104). However, youth were more likely than adults to require additional pharmacologic treatment to meet glycemic targets, with the other 51.7% of youth on metformin requiring insulin by 4 years, with a median time to treatment failure of 11.8 months.
Asymptomatic youth with presumptive type 2 diabetes who present in a stable metabolic state and have A1C <8.5% should be started on metformin as initial therapy if renal function is normal. Asymptomatic patients with A1C ≥8.5% may also be given an initial trial of metformin monotherapy at the discretion of the health care provider, especially if the patient and family situation suggest the promise of excellent adherence to lifestyle change recommendations.
The recommended approach to metformin initiation is to start with a dose of 500–1,000 mg/day and gradually escalate it every 1–2 weeks, depending on patient tolerability, to the recommended therapeutic dose of 1,000 mg b.i.d. Slower dosage escalation may be needed if gastrointestinal side effects occur and, in some cases, the maximum dose may not be achievable. Extended-release metformin may have fewer gastrointestinal side effects and be more convenient for the patient, but there are no studies in youth comparing extended-release metformin to the standard metformin preparation.
Metformin Plus Insulin
Youth with marked hyperglycemia (blood glucose ≥250 mg/dL and/or A1C ≥8.5%) without acidosis at diagnosis but who are symptomatic with polyuria, polydipsia, nocturia, and/or weight loss should be treated initially with basal insulin while concurrently initiating and titrating metformin. In patients with ketosis/ketoacidosis at diagnosis, treatment with subcutaneous or intravenous insulin should be initiated to rapidly correct the hyperglycemia and the metabolic derangement. Once acidosis is resolved, metformin should be initiated while subcutaneous insulin therapy is continued (116). In individuals presenting with severe hyperglycemia (blood glucose ≥600 mg/dL), assess for HHNK syndrome.
Once glycemic stability is achieved, insulin may not be needed. Limited data suggest that adolescents with type 2 diabetes who present initially with DKA, ketosis, or symptomatic hyperglycemia can be managed successfully with metformin alone, at least initially after a short course of insulin therapy to establish glycemic stability (117). For example, in the TODAY study, more than 90% of the subjects screened for study participation were initially controlled adequately on metformin alone regardless of prior insulin therapy (117). However, these TODAY participants were frequently contacted and closely monitored by the research staff, a situation that may not be feasible in a clinical setting. Whether or not early treatment with insulin provides unique benefits in youth with type 2 diabetes remains questionable. The recently completed RISE Pediatric Medication Study in youth with obesity with impaired glucose tolerance or recent-onset type 2 diabetes did not demonstrate benefits of 3 months of basal insulin glargine followed by 9 months of metformin compared with metformin alone for 12 months in preserving or restoring β-cell function (118). It remains to be determined if longer periods of insulin treatment may prove beneficial in preserving β-cell function.
Ongoing Therapy
When the individualized glycemic target can no longer be met with metformin alone, or if metformin intolerance or renal insufficiency develops, insulin therapy should be initiated. This can be done alone or in combination with metformin, unless metformin is contraindicated. Because studies indicate that adherence with insulin therapy is a challenge in youth with type 2 diabetes (73,119), starting with a single daily dose of a long-acting insulin analog (glargine [Lantus, Basalglar, Toujeo], detemir [Levemir], or degludec [Tresiba]) may be preferred. Premixed insulins may be appropriate in some circumstances.
If the combination of metformin at the maximum tolerated dose (up to 1,000 mg b.i.d.) plus basal insulin at a maximum dose of 1.5 units/kg/day is ineffective at achieving the glycemic target, medication adherence should be actively addressed. When combined metformin and basal insulin therapy does not achieve targets, and in the absence of other approved drugs to treat diabetes in youth (<18 years old), higher doses of long-acting insulin or initiation of multiple daily injections of basal and premeal rapid-acting insulin should be considered, though adherence to the latter may be a barrier.
Because severe insulin resistance is characteristic of youth with type 2 diabetes, basal insulin doses above 1.5 units/kg/day may be required to achieve adequate glycemic control, particularly for those youth with elevated A1C and glucotoxicity and youth who are in mid- to late puberty. In these circumstances, it may be appropriate to use more concentrated insulin preparations (U-300 glargine [Toujeo], U-200 Tresiba, U-200 Humalog, U-500 regular) to avoid large-volume injections that may further diminish medication adherence.
The most significant adverse effect of insulin therapy in type 2 diabetes, as in type 1 diabetes, is hypoglycemia. Although the incidence of hypoglycemia in youth with type 2 diabetes is low, even with insulin therapy (73), patients treated with insulin should be educated about avoidance, recognition, and treatment of hypoglycemia and should be instructed on the use of glucagon for treatment of severe hypoglycemia. Also, since insulin may result in weight gain, involvement of a nutritionist in patient care and education is essential when insulin is initiated.
Other Therapies
Other than insulin and metformin, there are currently more than 25 medications in 10 general classes that are commercially available and FDA-approved for treatment of type 2 diabetes in adults in the U.S. (Table 3). It should be noted, however, that none of these are currently approved for use in youth (<18 years old), and while some of these agents have undergone or are currently undergoing pharmacokinetic, pharmacodynamics, and safety/tolerability testing in small pediatric studies, no efficacy or long-term safety results have yet been reported in youth.
Table 3.
Drugs for treating type 2 diabetes in adults (not including insulin or insulin analogs) but not yet approved in youth except for metformin
| Drug class | Available drugs in this class | Mechanism of action | Significant adverse effects | Approved in patients <18 years old |
|---|---|---|---|---|
| Biguanides |
|
Decreases insulin resistance; reduces hepatic glucose production; increases peripheral glucose uptake; decreases gastrointestinal absorption of glucose |
|
Yes |
| Sulfonylureas |
|
Stimulates secretion of insulin from the β-cell |
|
No |
| Meglitinides |
|
Stimulates glucose-dependent secretion of insulin from the β-cell |
|
No |
| α-Glucosidase inhibitors |
|
Delays absorption of glucose by intestines by inhibiting breakdown of complex sugars |
|
No |
| GLP-1 agonists |
|
Incretin effect; slows gastric emptying; enhances postprandial insulin biosynthesis; improves β-cell function; decreases appetite |
|
No |
| DPP-4 inhibitors |
|
Inhibits DPP-4 enzyme, reducing endogenous GLP-1 breakdown |
|
No |
| Amylin analog |
|
Inhibits postprandial glucagon secretion; delays gastric emptying; improves satiety |
|
No |
| Thiazolidinediones |
|
PPAR-γ inhibitor; increases insulin sensitivity in liver, muscle, and adipose tissue; decreases hepatic glucose output |
|
No |
| SGLT-2 inhibitors |
|
Allows more glucose to be excreted in the urine and hence lowers blood glucose |
|
No |
| Bile acid sequestrant |
|
Mechanism for glucose lowering is unknown |
|
No |
| Dopamine-2 agonist |
|
Modulates hypothalamic regulation of metabolism; increases insulin sensitivity |
|
No |
DPP-4, dipeptidyl peptidase 4; GLP-1, glucagon-like peptide 1; PPAR, peroxisome proliferator–activated receptor; SGLT2, sodium–glucose cotransporter 2; URI, upper respiratory infection; UTI, urinary tract infection.
Although the TODAY study demonstrated that the addition of rosiglitazone to metformin improved the durability of glycemic control (treatment failure rate 38.6% for metformin plus rosiglitazone vs. 51.7% for metformin alone) with no increased rate of adverse events over a 3–6 year period in youth with recent-onset type 2 diabetes, it is premature to recommend its widespread use in youth with type 2 diabetes, especially since its use is not approved in the pediatric population. Even though many of the newer agents approved in the adult population are promising and may have particular benefits in younger individuals with diabetes, we cannot recommend widespread use of these medications until additional studies are completed. Unfortunately, implementation and completion of such studies have been slow and many barriers have been identified (111). Therefore, we recommend that the use of these medications in youth with type 2 diabetes be avoided outside of research trials. However, collaboration among investigators, pharmaceutical sponsors, and governmental regulators is urgently needed to expand treatment options for this population of patients.
METABOLIC SURGERY
Recommendations
Metabolic surgery may be considered for the treatment of adolescents with type 2 diabetes who are markedly obese (BMI >35 kg/m2) and who have uncontrolled glycemia and/or serious comorbidities despite lifestyle and pharmacologic intervention. A
Metabolic surgery should be performed only by an experienced surgeon working as part of a well-organized and engaged multidisciplinary team including surgeon, endocrinologist, nutritionist, behavioral health specialist, and nurse. A
Bariatric or metabolic surgery, including Roux-en-Y gastric bypass, vertical sleeve gastrectomy, laparoscopic adjustable gastric banding, laparoscopic gastric plication, and biliopancreatic diversion, has been shown to significantly reduce weight, BMI (120), and cardiovascular comorbidities (121) in adults with obesity and is now considered a standard component of care for adults with morbid obesity. Metabolic surgery is also an effective strategy for prevention (122,123) and treatment of type 2 diabetes in obese and severely obese (BMI ≥30 kg/m2) adults (124–129) and is now endorsed as part of the algorithm for treating type 2 diabetes in adults (127).
Over the last decade, weight-loss surgery has been increasingly performed in adolescents with obesity, but the long-term experience remains limited. The current guidelines for metabolic surgery in adolescents generally include BMI >35 kg/m2 with significant comorbidities or BMI >40 kg/m2 with or without comorbidities (106,130–140). The Endocrine Society Clinical Practice Guideline on Pediatric Obesity discusses bariatric surgery for the management of pediatric obesity in detail, and interested readers can refer to it (106). Briefly, positive outcomes of metabolic surgery have included remission of type 2 diabetes, improvements in glucose homeostasis in youth without diabetes, improvement in surrogate markers of insulin sensitivity and secretion, resolution of sleep apnea, improvements in nonalcoholic fatty liver disease (NAFLD), and improvements in cardiovascular disease (CVD) risk factors, among others (106,134–141). Direct comparison between the medical management of youth with type 2 diabetes and bariatric surgery outcome, both short- and long-term, is very limited. A recent study compared youth with type 2 diabetes from the Teen-Longitudinal Assessment of Bariatric Surgery (Teen-LABS) cohort who had undergone a bariatric surgical procedure with youth with medically treated type 2 diabetes from the TODAY cohort. During 2 years, A1C decreased from 6.8% to 5.5% in Teen-LABS and increased from 6.4% to 7.8% in TODAY, BMI decreased by 29% in Teen-LABS and increased by 3.7% in TODAY, elevated blood pressure decreased from 45% to 20% of participants in Teen-LABS and increased from 22% to 41% in TODAY, and dyslipidemia decreased from 72% to 24% in Teen-LAB versus no appreciable change in TODAY (142).
Overall, studies in both adults and adolescents suggest that those who undergo bariatric surgery earlier in the course of diabetes (that is, at a younger age or with higher baseline β-cell function) have a higher remission rate despite similar weight loss (143). Initial diabetes remission rates in adults range between 40% and 70%, whereas in adolescents the reported initial rates are as high as 68–100% (144). The long-term durability of these remissions is unknown and will require longer follow-up.
Short-term and long-term complications of metabolic surgery need to be taken into consideration. In Teen-LABS, 13% of adolescents required a second operative procedure and another 13% required an endoscopic procedure because of a complication (145). In the recent Teen-LABS/TODAY comparison, 30% of the youth with diabetes undergoing surgical intervention required readmission and/or reoperation (142). Postoperative nutritional complications (vitamin B12, thiamine, and vitamin D deficiency) are also prevalent. Long-term follow-up and further research is required to better understand the mechanisms by which metabolic surgery improves type 2 diabetes and the short-term and long-term benefits and risks of this procedure in youth. Quality of life and economic (cost-benefit) analyses will also be important components of ongoing follow-up and research (146,147).
PREVENTION AND MANAGEMENT OF DIABETES COMPLICATIONS
Youth-onset type 2 diabetes is associated with significant microvascular and macrovascular risk burden and a substantial increase in the risk of cardiovascular morbidity and mortality at an earlier age than those individuals diagnosed later in life (148). The higher complication risk in earlier-onset type 2 diabetes is likely to be related to prolonged lifetime exposure to hyperglycemia and other atherogenic risk factors, including insulin resistance, dyslipidemia, hypertension, and chronic inflammation. These diabetes comorbidities also appear to be higher than in youth with type 1 diabetes despite shorter diabetes duration and lower A1C (149). In addition, the progression of vascular abnormalities appears to be more pronounced in type 2 diabetes diagnosed earlier in life compared with type 1 diabetes of similar duration, including ischemic heart disease and stroke (150,151).
Nephropathy
Recommendations
Blood pressure should be measured at every visit. A
Blood pressure should be optimized to reduce risk and/or slow the progression of diabetic kidney disease. A
If blood pressure is >95th percentile for age, sex, and height, increased emphasis should be placed on lifestyle management to promote weight loss. If blood pressure remains above the 95th percentile after 6 months, antihypertensive therapy should be initiated. C
Initial therapeutic options include ACE inhibitors or angiotensin receptor blockers. Other blood pressure–lowering agents may be added as needed. C
Protein intake should be at the recommended daily allowance of 0.8 g/kg/day. E
Urine albumin/creatinine ratio (UACR) should be obtained at the time of diagnosis and annually thereafter. An elevated UACR (>30 mg/g creatinine) should be confirmed on two of three samples. B
Estimated glomerular filtration rate (eGFR) should be determined at the time of diagnosis and annually thereafter. E
In nonpregnant youth with diabetes and hypertension, either an ACE inhibitor or an angiotensin receptor blocker is recommended for those with modestly elevated UACR (30–299 mg/g creatinine) D and strongly recommended for those with UACR >300 mg/g creatinine and/or eGFR <60 mL/min/1.73 m2. E
For those with nephropathy, continued monitoring (yearly UACR, eGFR, serum K) may aid in assessing adherence and detecting progression of disease. E
Referral to nephrology is recommended in case of uncertainty of etiology, worsening UACR, or decrease in eGFR. E
Diabetic kidney disease is diagnosed in the presence of elevated albumin excretion and decreased eGFR and is the leading cause of end-stage renal disease (152). Elevated UACR or albuminuria, defined as >30 mg/g creatinine, and hyperfiltration, defined as an eGFR between 120 mL/min/1.73 m2 and 150 mL/min/1.73 m2 (153,154), are early abnormalities that indicate increased risk of progression to diabetic kidney disease (154,155). Overt nephropathy is defined as persistent proteinuria ≥500 mg/day or UACR ≥300 mg/g creatinine and an eGFR <60 mL/min/1.73 m2.
Albuminuria and hyperfiltration detected early in youth with type 2 diabetes may occur because of obesity before the onset of diabetes (156) and can be related to early vascular dysfunction (157). In TODAY, the prevalence of microalbuminuria was 6.3% at randomization (mean 7.8 months since diagnosis of diabetes) and increased to 16.6% over 3 years (79). This coincided with progression of dyslipidemia (from 4.5% at baseline to 11%) and hypertension (from 11.6% at baseline up to 33%) (78,79) despite standardized therapy for these comorbidities. The main determining factor in albuminuria progression was A1C, with 17% higher risk of developing albuminuria per 1% increase in A1C (79), consistent with findings in other studies (149). Modifiable risk factors include obesity, dyslipidemia, hypertension, and glycemia (152). In some ethnic groups, particularly Pima Indians and First Nations people in Canada, the risk of nephropathy is much higher in youth with type 2 diabetes (158–161).
Spot UACR is generally recommended for screening of urinary albumin excretion, with an abnormal value confirmed on two of three consecutive tests obtained on different days within a 3- to 6-month period. Results can be affected by orthostatic proteinuria, marked hyperglycemia, exercise, menstruation, recent intercourse, and sample contamination. eGFR can be calculated from serum creatinine and the patient’s height using the Schwartz equation. However, recent studies suggest that this underestimates hyperfiltration, which is highly prevalent in youth with type 2 diabetes, and a combined estimation using serum creatinine and serum cystatin C is preferable when available (162).
In addition to optimizing glycemia, control of hypertension is important to prevent and slow the progression of nephropathy. Therapeutic options include the use of ACE inhibitors or angiotensin receptor blockers (152,163–165). If not tolerated, a calcium channel blocker or diuretic or combination therapy may be required if hypertension does not normalize on single-agent therapy.
Neuropathy
Recommendations
Youth with type 2 diabetes should be screened for the presence of neuropathy by foot examination at diagnosis and annually. The examination should include inspection, assessment of foot pulses, pinprick and 10-g monofilament sensation tests, testing of vibration sensation using a 128-Hz tuning fork, and ankle reflexes. C
Prevention should focus on achieving glycemic goals. C
Diabetic neuropathy can manifest as distal polyneuropathy (DPN), mononeuropathy, and/or autonomic neuropathy. Mononeuropathies are uncommon. DPN is usually the earliest clinically apparent manifestation of neuropathy in persons with diabetes and most commonly presents with paresthesia, numbness, or pain in the feet. DPN generally affects the small myelinated fibers first with burning or stabbing pain and reduced or absent thermal and pinprick sensation. It then progresses to larger myelinated fibers with numbness, tingling, and poor balance along with reduced or absent reflexes, vibration perception, and monofilament sensation. The “gold standard” for the diagnosis of DPN includes careful neurologic examination to rule out other potential causes of neuropathy and nerve conduction velocity studies. The Diabetes Control and Complications Trial (DCCT), which used a combination of examination by a neurologist, nerve conduction velocity testing, and autonomic neuropathy testing, clearly showed that improved glycemic control reduced the incidence of diabetic neuropathy, both DPN and autonomic, by 44–60%.
However, most large prospective studies have not been able to include the gold standard of nerve conduction studies and have utilized less invasive and less expensive approaches to the diagnosis of diabetic neuropathy. The most commonly used is the Michigan Neuropathy Screening Instrument (MNSI). The MNSI is a self-administered questionnaire (MNSIQ) and an examination (MNSIE) for foot abnormalities, distal vibration perception, and ankle reflexes; the MNSI has been validated in adults with type 1 diabetes as a screening tool for neuropathy (166–169). It should be noted, however, that the MNSIE does not include an assessment of small-fiber dysfunction.
Evidence of diabetic neuropathy using the MNSI was found in 26% of youth with type 2 diabetes in the SEARCH study (168) and in 21% of an Australian cohort using thermal (small fiber) and vibration (large fiber) threshold testing (149). In addition, more than half of the cohort had evidence of autonomic neuropathy (pupillary reactivity) after a median duration of diabetes of 1.3 years (149). In an Indian cohort of mean age 16 years at diagnosis, the prevalence of neuropathy increased from 3% in those with diabetes duration <5 years to 49% in those with duration >15 years (170). In the SEARCH study, the prevalence of abnormal MNSI was significantly higher in youth with type 2 diabetes compared with those with type 1 diabetes after adjustment for age and sex. This association was no longer significant after adjustment for the covariates of diabetes duration, waist circumference, blood pressure, HDL cholesterol, and microalbuminuria (168). In the Australian cohort, the prevalence of peripheral and autonomic neuropathy in adolescents with type 2 diabetes was similar to that of the type 1 diabetes cohort despite shorter diabetes duration (1.3 vs. 6.8 years) and lower A1C (7.3% vs. 8.5%) (149). In adolescents with type 1 diabetes, data from the DCCT support the importance of intensive glycemic therapy and reduction of A1C for the prevention of diabetic neuropathy (171,172). So far, such data do not exist in youth with type 2 diabetes.
The ADA recommends that assessment for symmetric DPN should include a careful history and assessment of either temperature or pinprick sensation (small-fiber function) and vibration sensation using a 128-Hz tuning fork (for large-fiber function). All patients should have annual 10-g monofilament testing to identify feet at risk for ulceration and amputation (173). Since it appears that youth with type 2 diabetes develop DPN at least as frequently as adults, youth with type 2 diabetes should be screened at the same frequency: at diagnosis and annually.
Retinopathy
Recommendations
Screening for retinopathy should be performed by dilated fundoscopy or retinal photography at or soon after diagnosis and annually thereafter. C
Optimizing glycemia is recommended to decrease the risk or slow the progression of retinopathy. B
Less frequent examination (every 2 years) may be considered if there is adequate glycemic control and a normal eye exam. C
Diabetic retinopathy refers to changes in the small vessels of the retina with the occurrence of hemorrhages, microaneurysms, exudates, or abnormal vessels. The prevalence of retinopathy in youth with type 2 diabetes is reported to be between 2% and 40%, depending on the methodology used, the age of the participants, and the duration of diabetes. The prevalence is higher with greater duration of the disease, although retinopathy has been reported at diagnosis (149,170,174). In the TODAY study, the prevalence of early retinopathy by digital fundus photography at a mean age of 18.1 years and mean duration of diabetes of 4.9 years was 13.7%, with no evidence of macular edema or proliferative retinopathy (77). Retinopathy was associated with older age (19.1 vs. 17.9 years), longer duration of diabetes (5.6 vs. 4.7 years), and higher A1C (8.3% vs. 6.9%). Moreover, the odds ratio for retinopathy increased with increasing A1C, age, and duration of diabetes (77). In the SEARCH study, the prevalence of retinopathy using retinal photography was 42% at a mean age of 21 years and mean duration of type 2 diabetes of 7.2 years (175). A1C and LDL cholesterol were significantly higher among those with retinopathy compared with those without. In Pima Indians, retinopathy was detected only after age 20 years and only after 5 years of diabetes duration (158). However, by 30 years of age, retinopathy had developed in 45% of this population (158).
NAFLD
Recommendations
Evaluation for NAFLD (by measuring ALT and AST) should be done at diagnosis of type 2 diabetes and annually thereafter. B
Referral to gastroenterology should be considered for persistently elevated or worsening transaminases. B
The prevalence of dysglycemia in youth with NAFLD is higher than in those without NAFLD (176). In a multicenter cohort of youth with NAFLD, primarily of Hispanic descent, a third of the children with NAFLD had abnormalities in glucose metabolism; 23.4% had prediabetes and 6.5% had type 2 diabetes (176). Moreover, type 2 diabetes in youth is associated with greater NAFLD histologic severity than in adults, which may imply a heightened risk of progression to fibrosis, cirrhosis, and hepatic failure (176,177). Therefore, it is particularly important to evaluate for NAFLD in youth with obesity and type 2 diabetes. For screening, transaminase levels are a clinical tool that is widely available and has a good sensitivity for the detection of more advanced stages of hepatitis or fibrotic changes, but these tests are not disease specific; therefore, other causes of chronic liver disease should be ruled out (178,179). Recently, population-based cutoffs lower than those used in clinical laboratories have been advocated to indicate abnormality (180). Among the noninvasive diagnostic tests, MRI/MRS are currently the preferred imaging modalities, though of limited clinical application (181). Liver ultrasound, though a widely available clinical tool, is operator dependent and detects liver fat >30% with sensitivity of 80%, but sensitivity is lower with lower degrees of fatty infiltration and the presence of morbid obesity. Hence, its value in the early diagnosis of NAFLD is limited (181). Among the noninvasive modalities, elastography is useful in evaluating advanced fibrosis and cirrhosis (181) and is gaining wider acceptance. Liver biopsy remains the gold standard for diagnosis and staging of liver disease and the only way to differentiate between nonalcoholic steatohepatitis and hepatic steatosis. Treatment options for NAFLD remain limited, with weight loss being most effective. Therapeutic agents tested in randomized clinical trials in youth include metformin, vitamin E, and cysteamine, with no clear established benefit on histologic outcomes or sustained reduction in ALT (182–184).
Obstructive Sleep Apnea
Recommendation
Screening for symptoms of obstructive sleep apnea (OSA) should be done at each visit, and referral to a pediatric sleep specialist for evaluation and a polysomnogram, if indicated, is recommended. OSA should be treated when documented. B
Sleep disturbance (insufficient or disrupted sleep, circadian rhythm dysregulation) and OSA are increasingly recognized as being associated with obesity (185–189), insulin resistance in adults and children (52,190–195), and type 2 diabetes in adults (196–199), as well as risk for future CVD (200). Experimental sleep restriction results in decreased glucose clearance and postprandial glucose elevation (193,201), decrease in glucose effectiveness, and variable decrease in insulin sensitivity (190,192,202). OSA may influence glycemic regulation in individuals with diabetes; in adults with type 2 diabetes, treatment of OSA with continuous positive airway pressure has been associated with improvement in the glycemic profile (203), decreased A1C, and improvement in insulin sensitivity indices (204) as well as inflammation (205) in some, but not all, studies. Further study is needed.
Polycystic Ovary Syndrome
Recommendations
Evaluate for polycystic ovary syndrome (PCOS) in female adolescents with type 2 diabetes, including laboratory studies when indicated. B
Oral contraceptives for treatment of PCOS are not contraindicated for girls with type 2 diabetes. C
Metformin in addition to lifestyle modification is likely to improve the menstrual cyclicity and hyperandrogenism in girls with type 2 diabetes. E
PCOS affects 5–10% of females in the reproductive age-group and is characterized by hyperandrogenism and amenorrhea or oligomenorrhea secondary to chronic anovulation (206,207). The prevalence of PCOS is significantly higher in adolescent girls with obesity compared with adolescent girls without overweight/obesity (208), but the prevalence in adolescent girls with type 2 diabetes is not well studied. Insulin resistance with compensatory hyperinsulinemia are metabolic features in both adult women with PCOS with and without overweight/obesity (209) and in adolescent girls with PCOS compared with control subjects of similar age, body composition, and abdominal adiposity (210). In adolescent girls with PCOS and obesity, this increased insulin resistance when combined with impaired β-cell function predisposes to prediabetes and type 2 diabetes (211), with higher prevalence of impaired glucose tolerance (30%) and type 2 diabetes (3.7%) (212). Therefore, it is important to obtain a menstrual history and evaluate female adolescents with type 2 diabetes for signs and symptoms of hyperandrogenism (irregular menses, hirsutism, acne) and to initiate appropriate diagnostic evaluation for PCOS if indicated (213,214). In the TODAY cohort, 21% of adolescent girls who were ≥1-year postmenarche had irregular menses. Those with irregular menses versus regular menses had higher total testosterone, free androgen index, BMI, and AST and lower sex hormone–binding globulin and estradiol (215). Treatment of PCOS in adolescents includes lifestyle changes (216–218), the use of oral contraceptive pills (OCPs), and insulin sensitizers, such as metformin (213). However, the use of some OCPs has been associated with unfavorable effects on indices of insulin sensitivity (219) and lipid profile (220). The use of metformin therapy for 3–12 months was associated with decrease in serum androgens, improvement in lipid profile, induction of ovulation, and improvement in glucose tolerance and insulin sensitivity (216,221,222). Therefore, in girls with type 2 diabetes and PCOS, treatment with metformin in addition to lifestyle modification is likely to improve the metabolic dysfunction associated with PCOS and may improve menstrual cyclicity and hyperandrogenism (213,218). However, for the girls in the TODAY study, all of whom received metformin, there was no treatment group (metformin alone, metformin plus lifestyle, and metformin plus rosiglitazone) effect on menses or sex steroids at 12 and 24 months and no association of sex steroids with surrogate estimates of insulin sensitivity or secretion (215). Despite the potential negative effects of OCPs, which may not be shared by all OCPs, on metabolic status and cardiovascular risk, hormonal contraceptive therapy is more effective at addressing the symptoms of hyperandrogenism and anovulation and is not contraindicated in female youth with type 2 diabetes (213).
CVD
Recommendation
Intensive lifestyle interventions focusing on weight loss, dyslipidemia, hypertension, and dysglycemia are important to prevent overt macrovascular disease in early adulthood. E
Dyslipidemia
Recommendations
Lipid testing should be performed when initial glycemic control has been achieved and annually thereafter. B
Optimal cholesterol goals are LDL <100 mg/dL (2.6 mmol/L), HDL >35 mg/dL (0.905 mmol/L), triglycerides <150 mg/dL (1.7 mmol/L). E
If LDL cholesterol is >130 mg/dL, blood glucose control should be maximized and dietary counseling should be provided using the American Heart Association Step 2 diet. E
If LDL cholesterol remains above goal after 6 months of dietary intervention, initiate therapy with statin, with goal of LDL <100 mg/dL. B
If triglycerides are >400 mg/dL (4.7 mmol/L) fasting or >1,000 mg/dL (11.6 mmol/L) nonfasting, optimize glycemia and begin fibrate, with a goal of <400 mg/dL (4.7 mmol/L) fasting (to reduce risk for pancreatitis). C
Although there have been no long-term studies of the outcome of cholesterol-lowering therapy in youth with type 2 diabetes, studies in youth with familial hypercholesterolemia have shown reduction in carotid intima-media thickness (IMT) with the use of statins (223,224), with similar efficacy and side effects as in adults. However, in a recent multicenter, multinational study of youth with type 1 diabetes, statin use did not have a significant effect on carotid IMT despite reductions in total LDL cholesterol and triglyceride concentrations (225). Although longitudinal, interventional data with statins in youth-onset type 2 diabetes are not yet available, statin therapy in youth with type 2 diabetes who do not meet LDL targets following lifestyle change intervention is considered a reasonable approach and aligned with overall recommendations for dyslipidemia (226), given that dyslipidemia in youth tracks into adulthood and is anticipated to confer increased cardiovascular risk. Similarly, though there have been no studies of the use of fibrates in youth with type 2 diabetes and hypertriglyceridemia to prevent pancreatitis, extrapolation from studies in adults supports the use of these agents for severe hypertriglyceridemia in adolescents. Adolescent girls treated with statins or fibrates should receive counseling on potential risk to the fetus and be encouraged to use effective birth control.
Cardiac Function Testing
Recommendation
Routine screening for heart disease with electrocardiogram, echocardiogram, or stress testing is not recommended in asymptomatic youth with type 2 diabetes. B
Macrovascular disease involves coronary, cerebral, and peripheral arterial disease. In adults, type 2 diabetes is associated with doubling of risk for CVD, including coronary heart disease and stroke as well as increased risk of heart failure, after adjusting for age, sex, smoking status, BMI, and systolic blood pressure (227). Diabetes duration is implicated as a major risk factor for CVD (228,229), though there may also be a worsened risk of CVD with early onset of type 2 diabetes (228,230). While overt cardiovascular events are not expected in youth with type 2 diabetes, epidemiological and clinical studies show that the atherosclerotic process starts during childhood (231), with strong relationships between childhood obesity, elevated blood pressure, low HDL cholesterol, and coronary artery disease in adulthood (232–235). Furthermore, studies of vascular function have demonstrated subclinical vascular disease in adolescents with obesity and type 2 diabetes, including elevated aortic pulse wave velocity, a marker of vascular stiffness (236), and increased carotid IMT, a structural measure of atherosclerosis, compared with normoglycemic youth with and without overweight/obesity (237). In the SEARCH study, youth with type 2 diabetes had worse arterial stiffness than those with type 1 diabetes, attributed to greater central adiposity and hypertension but not related to duration of diabetes or glycemic control (238). In studies of obese youth with and without type 2 diabetes, carotid IMT was significantly related to glycemia, while aortic pulse wave velocity was related to insulin resistance and inflammation (239). In addition, total body and abdominal adiposity were significant determinants of coronary artery calcifications in these youth (239). In TODAY, echocardiographic evaluation revealed a relationship of BMI and blood pressure with adverse cardiac measures (240), though there was a protective effect of cardiorespiratory fitness on functional measures of cardiac structure and function in this group of largely sedentary youth (241). Overall, studies to date indicate significant vascular dysfunction and greater risk of progression to overt CVD in youth with obesity and type 2 diabetes. The vascular dysfunction may begin prior to the diagnosis of type 2 diabetes as a result of obesity and insulin resistance.
In adults, type 2 diabetes is associated with an increased risk of mortality, with cardiac disease as a major cause of death (242); the excess mortality is related to worse glycemic control, impaired renal function, and younger age at diabetes diagnosis (243). Youth-onset type 2 diabetes appears to be associated with an earlier onset of complications and an increased mortality risk compared with type 1 diabetes (109,244,245). In a Swedish study, type 2 diabetes diagnosed between 15 and 34 years of age was associated with a higher standardized mortality ratio than type 1 diabetes (2.9 and 1.8, respectively), with an increased hazard ratio for males versus females (P = 0.0002) (244). Similarly, an epidemiological study from Australia reported a significant mortality excess over 15–30 years of follow-up in individuals diagnosed with type 2 diabetes between 15 and 30 years of age compared with type 1 diabetes, with a hazard ratio of 2.0 (95% CI 1.2–3.2), despite shorter average disease duration (26.9 vs. 36.5 years, P = 0.01) and similar glycemic control (109). The mortality excess was related to an excess of cardiovascular deaths in those with type 2 diabetes (50% vs. 30%, P < 0.05). In First Nations individuals, increased mortality with type 2 diabetes is reported in relation to end-stage renal disease (159) and is significantly higher than in individuals with youth-onset type 1 diabetes (245). In a large cohort of 354 patients with type 2 diabetes diagnosed between 15 and 30 years of age compared with a duration-matched cohort of 1,062 patients diagnosed between 40 and 50 years old, the negative effect of diabetes on morbidity (albuminuria and neuropathy scores) and mortality was greatest for those diagnosed at a young age. Standardized mortality adjusting for duration was highest, at any chronological age, for those diagnosed between 15 and 30 years of age (246). Taken together, these data raise significant concern regarding the long-term outcome of youth-onset type 2 diabetes and support the importance of aggressive management of glycemia and CVD risk factors in these youth.
Transitioning from Pediatric to Adult Care
Recommendation
Youth with type 2 diabetes should be transferred to an adult-oriented diabetes specialist when deemed appropriate by the patient and provider. E
The process of transferring the pediatric patient to an adult health care provider is a challenge that has only recently received attention in the literature but is now recognized to be “important and should begin well before patients are transferred” (247). Both the Society for Adolescent Medicine (248) and the American Academy of Pediatrics, along with other associations (249), have position statements related to transition of care for those with chronic diseases and special medical needs that emphasize the importance of a gradual and collaborative process starting a year or longer before the actual transition is to occur. Published literature on this subject recommends progressive implementation as eight developmentally linked steps (250,251).
The ADA, in partnership with the other organizations of the ADA Transitions Working Group (252), developed position statements in 2011 (252), in 2014 (253), and in 2018 (254). Even for youth with type 1 diabetes, deficiencies and gaps in the transition process have been demonstrated in observational cross-sectional research. These gaps are summarized in recent reviews (250,252,255) and include minimal empirical evidence about the best approaches, differences in the style and approach to health care delivery between pediatric and adult health care providers, lack of well-defined criteria of readiness for transition or tools to assess readiness, gaps in health insurance coverage, changing social structure as adolescents enter young adulthood, differences in learning styles of the patient and teaching styles of the provider, and lack of health care provider training related to transition of care.
Despite the prevailing evidence of the need for better transition of care, there are no controlled studies of the effectiveness of such programs in patients with youth-onset type 2 diabetes. Patients with type 1 diabetes have reduced dropout from medical care, increased number of visits, and reduced pregnancy loss, DKA, and severe hypoglycemia during the transition period using a “navigator” to assist young adults (18–30 years old) (256–258).
Since emerging adults with type 2 diabetes express similar concerns related to transition from pediatric to adult health care providers (259), the same principles discussed above and steps to facilitate transition that apply to those with type 1 diabetes should be considered in type 2 diabetes.
Conclusions
Even though our knowledge of youth-onset type 2 diabetes has increased tremendously over the last two decades, robust and evidence-based data are still limited regarding diagnostic and therapeutic approaches and prevention of complications. The current-day information indicates that there are fundamental differences in insulin sensitivity and β-cell function between youth and adults with prediabetes and type 2 diabetes, which could possibly explain why some youth develop type 2 diabetes decades earlier than adults (23,24,260,261). Youth are more insulin resistant and have β-cells that are hyperresponsive to stimulation compared with adults (23,24,260,261). Puberty-related physiologic insulin resistance, particularly in obese youth, may play a role in this heightened insulin resistance. It remains an enigma, though, why some individuals with youth-onset type 2 diabetes demonstrate durable control and others do not (74). Furthermore, type 2 diabetes appears to be more aggressive in youth than adults, with a faster rate of deterioration of β-cell function (76) and poorer response to glucose-lowering medications (104). Future research should probe the mechanisms responsible for this youth–adult contrast in the various aspects of type 2 diabetes. Lastly, complications in youth with type 2 diabetes appear early, resulting in higher rates of morbidity and mortality compared with type 1 diabetes. Preexisting obesity and its comorbidities might play a key role in amplifying the complications of youth-onset type 2 diabetes. Intervention/prevention strategies for type 2 diabetes should not be limited to youth with dysglycemia only, but youth with obesity at large.
In closing, the present guidelines are based on current data, experience, opinion, and gained “wisdom.” However, we anticipate that future guidelines will change as more scientific data emerge to support evidence-based recommendations.
Article Information
Duality of Interest. S.A. has served on a data monitoring committee for AstraZeneca, data safety monitoring board for Boehringer Ingelheim, and advisory boards for Eli Lilly, Novo Nordisk, and Sanofi-Aventis and has received research grants from Eli Lilly and Novo Nordisk through her institution. F.B. has received grant support through her institution from Johnson & Johnson and AstraZeneca. M.D.M. has served on the scientific advisory committee of WW International, Inc. P.Z. has served as a study design consultant for Daichii Sankyo, Merck, Boeringer Ingelheim, Janssen, and Eli Lilly (all contracts with the University of Colorado) and as a member of a data safety monitory board for Novo Nordisk. No other potential conflicts of interest relevant to this article were reported.
Footnotes
This position statement was reviewed and approved by the American Diabetes Assoication Professional Practice Committee and ratified by the American Diabetes Association Board of Directors in September 2018.
For further information on the ADA evidence-grading system and the levels of evidence, please see Table 1 in the American Diabetes Association’s Introduction section of the Standards of Medical Care in Diabetes—2018. Diabetes Care 2018;41(Suppl. 1):S2, https://doi.org/10.2337/dc18-SINT01.
The authors are solely responsible for the contents of this article, which do not necessarily represent the official views of the NIDDK.
References
- 1.Kahn SE, Prigeon RL, McCulloch DK, et al. Quantification of the relationship between insulin sensitivity and beta-cell function in human subjects. Evidence for a hyperbolic function. Diabetes 1993;42:1663–1672 [DOI] [PubMed] [Google Scholar]
- 2.Gungor N, Bacha F, Saad R, Janosky J, Arslanian S. Youth type 2 diabetes: insulin resistance, beta-cell failure, or both? Diabetes Care 2005;28:638–644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Michaliszyn SF, Mari A, Lee S, et al. β-Cell function, incretin effect, and incretin hormones in obese youth along the span of glucose tolerance from normal to prediabetes to type 2 diabetes. Diabetes 2014;63:3846–3855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Umpierrez GE. (Ed.). Therapy for Diabetes Mellitus and Related Disorders. 6th ed. Alexandria, VA, American Diabetes Association, 2014 [Google Scholar]
- 5.Bacha F, Lee S, Gungor N, Arslanian SA. From pre-diabetes to type 2 diabetes in obese youth: pathophysiological characteristics along the spectrum of glucose dysregulation. Diabetes Care 2010;33:2225–2231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tfayli H, Lee S, Arslanian S. Declining β-cell function relative to insulin sensitivity with increasing fasting glucose levels in the nondiabetic range in children. Diabetes Care 2010;33:2024–2030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Burns SF, Bacha F, Lee SJ, Tfayli H, Gungor N, Arslanian SA. Declining β-cell function relative to insulin sensitivity with escalating OGTT 2-h glucose concentrations in the nondiabetic through the diabetic range in overweight youth. Diabetes Care 2011;34:2033–2040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Giannini C, Weiss R, Cali A, et al. Evidence for early defects in insulin sensitivity and secretion before the onset of glucose dysregulation in obese youths: a longitudinal study. Diabetes 2012;61:606–614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Weiss R, Caprio S, Trombetta M, Taksali SE, Tamborlane WV, Bonadonna R. Beta-cell function across the spectrum of glucose tolerance in obese youth. Diabetes 2005;54:1735–1743 [DOI] [PubMed] [Google Scholar]
- 10.Kobayashi K, Amemiya S, Higashida K, et al. Pathogenic factors of glucose intolerance in obese Japanese adolescents with type 2 diabetes. Metabolism 2000;49:186–191 [DOI] [PubMed] [Google Scholar]
- 11.Elder DA, Prigeon RL, Wadwa RP, Dolan LM, D’Alessio DA. Beta-cell function, insulin sensitivity, and glucose tolerance in obese diabetic and nondiabetic adolescents and young adults. J Clin Endocrinol Metab 2006;91:185–191 [DOI] [PubMed] [Google Scholar]
- 12.Druet C, Tubiana-Rufi N, Chevenne D, Rigal O, Polak M, Levy-Marchal C. Characterization of insulin secretion and resistance in type 2 diabetes of adolescents. J Clin Endocrinol Metab 2006;91:401–404 [DOI] [PubMed] [Google Scholar]
- 13.Elder DA, Woo JG, D’Alessio DA. Impaired β-cell sensitivity to glucose and maximal insulin secretory capacity in adolescents with type 2 diabetes. Pediatr Diabetes 2010;11:314–321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Defronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 2009;58:773–795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tahrani AA, Bailey CJ, Del Prato S, Barnett AH. Management of type 2 diabetes: new and future developments in treatment. Lancet 2011;378:182–197 [DOI] [PubMed] [Google Scholar]
- 16.Umpaichitra V, Bastian W, Taha D, Banerji MA, AvRuskin TW, Castells S. C-peptide and glucagon profiles in minority children with type 2 diabetes mellitus. J Clin Endocrinol Metab 2001;86:1605–1609 [DOI] [PubMed] [Google Scholar]
- 17.Weiss R, D’Adamo E, Santoro N, Hershkop K, Caprio S. Basal α-cell up-regulation in obese insulin-resistant adolescents. J Clin Endocrinol Metab 2011;96:91–97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bacha F, Gungor N, Lee S, Arslanian SA. In vivo insulin sensitivity and secretion in obese youth: what are the differences between normal glucose tolerance, impaired glucose tolerance, and type 2 diabetes? Diabetes Care 2009;32:100–105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Goran MI, Bergman RN, Avila Q, et al. Impaired glucose tolerance and reduced β-cell function in overweight Latino children with a positive family history for type 2 diabetes. J Clin Endocrinol Metab 2004;89:207–212 [DOI] [PubMed] [Google Scholar]
- 20.Weigensberg MJ, Ball GDC, Shaibi GQ, Cruz ML, Goran MI. Decreased β-cell function in overweight Latino children with impaired fasting glucose. Diabetes Care 2005;28:2519–2524 [DOI] [PubMed] [Google Scholar]
- 21.Weiss R, Taksali SE, Tamborlane WV, Burgert TS, Savoye M, Caprio S. Predictors of changes in glucose tolerance status in obese youth. Diabetes Care 2005;28:902–909 [DOI] [PubMed] [Google Scholar]
- 22.Sjaarda LA, Michaliszyn SF, Lee S, et al. HbA1c diagnostic categories and β-cell function relative to insulin sensitivity in overweight/obese adolescents. Diabetes Care 2012;35:2559–2563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.RISE Consortium Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. observations using the hyperglycemic clamp. Diabetes Care 2018;41:1696–1706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.RISE Consortium Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. observations using the oral glucose tolerance test. Diabetes Care 2018;41:1707–1716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arslanian SA, Bacha F, Saad R, Gungor N. Family history of type 2 diabetes is associated with decreased insulin sensitivity and an impaired balance between insulin sensitivity and insulin secretion in white youth. Diabetes Care 2005;28:115–119 [DOI] [PubMed] [Google Scholar]
- 26.Giannini C, Dalla Man C, Groop L, et al. Co-occurrence of risk alleles in or near genes modulating insulin secretion predisposes obese youth to prediabetes. Diabetes Care 2014;37:475–482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sartorius T, Staiger H, Ketterer C, et al. Association of common genetic variants in the MAP4K4 locus with prediabetic traits in humans. PLoS One 2012;7:e47647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kwak SH, Park KS. Genetics of type 2 diabetes and potential clinical implications. Arch Pharm Res 2013;36:167–177 [DOI] [PubMed] [Google Scholar]
- 29.So H-C, Gui AHS, Cherny SS, Sham PC. Evaluating the heritability explained by known susceptibility variants: a survey of ten complex diseases. Genet Epidemiol 2011;35:310–317 [DOI] [PubMed] [Google Scholar]
- 30.Billings LK, Florez JC. The genetics of type 2 diabetes: what have we learned from GWAS? Ann N Y Acad Sci 2010;1212:59–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gill-Carey O, Hattersley AT. Genetics and type 2 diabetes in youth. Pediatr Diabetes 2007;8(Suppl. 9):42–47 [DOI] [PubMed] [Google Scholar]
- 32.Dabelea D, Dolan LM, D’Agostino R Jr, et al. Association testing of TCF7L2 polymorphisms with type 2 diabetes in multi-ethnic youth. Diabetologia 2011;54:535–539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dabelea D. The predisposition to obesity and diabetes in offspring of diabetic mothers. Diabetes Care 2007;30(Suppl. 2):S169–S174 [DOI] [PubMed] [Google Scholar]
- 34.McMillen IC, Rattanatray L, Duffield JA, et al. The early origins of later obesity: pathways and mechanisms. Adv Exp Med Biol 2009;646:71–81 [DOI] [PubMed] [Google Scholar]
- 35.Chernausek SD, Arslanian S, Caprio S, et al. Relationship between parental diabetes and presentation of metabolic and glycemic function in youth with type 2 diabetes: baseline findings from the TODAY trial. Diabetes Care 2016;39:110–117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dabelea D, Mayer-Davis EJ, Lamichhane AP, et al. Association of intrauterine exposure to maternal diabetes and obesity with type 2 diabetes in youth: the SEARCH Case-Control Study. Diabetes Care 2008;31:1422–1426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dabelea D, Mayer-Davis EJ, Saydah S, et al.; SEARCH for Diabetes in Youth Study . Prevalence of type 1 and type 2 diabetes among children and adolescents from 2001 to 2009. JAMA 2014;311:1778–1786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Arslanian SA. Metabolic differences between Caucasian and African-American children and the relationship to type 2 diabetes mellitus. J Pediatr Endocrinol Metab 2002;15(Suppl. 1):509–517 [PubMed] [Google Scholar]
- 39.Bacha F, Saad R, Gungor N, Janosky J, Arslanian SA. Obesity, regional fat distribution, and syndrome X in obese black versus white adolescents: race differential in diabetogenic and atherogenic risk factors. J Clin Endocrinol Metab 2003;88:2534–2540 [DOI] [PubMed] [Google Scholar]
- 40.Hasson RE, Adam TC, Davis JN, et al. Ethnic differences in insulin action in obese African-American and Latino adolescents. J Clin Endocrinol Metab 2010;95:4048–4051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bacha F, Gungor N, Lee S, Arslanian SA. Type 2 diabetes in youth: are there racial differences in β-cell responsiveness relative to insulin sensitivity? Pediatr Diabetes 2012;13:259–265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Weiss R, Dziura JD, Burgert TS, Taksali SE, Tamborlane WV, Caprio S. Ethnic differences in beta cell adaptation to insulin resistance in obese children and adolescents. Diabetologia 2006;49:571–579 [DOI] [PubMed] [Google Scholar]
- 43.Copeland KC, Zeitler P, Geffner M, et al.; TODAY Study Group . Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab 2011;96:159–167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes 2001;50:2444–2450 [DOI] [PubMed] [Google Scholar]
- 45.Hannon TS, Janosky J, Arslanian SA. Longitudinal study of physiologic insulin resistance and metabolic changes of puberty. Pediatr Res 2006;60:759–763 [DOI] [PubMed] [Google Scholar]
- 46.Ball GDC, Huang TT-K, Gower BA, et al. Longitudinal changes in insulin sensitivity, insulin secretion, and β-cell function during puberty. J Pediatr 2006;148:16–22 [DOI] [PubMed] [Google Scholar]
- 47.Kelly LA, Lane CJ, Weigensberg MJ, Toledo-Corral CM, Goran MI. Pubertal changes of insulin sensitivity, acute insulin response, and β-cell function in overweight Latino youth. J Pediatr 2011;158:442–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Carnethon MR, Kinder LS, Fair JM, Stafford RS, Fortmann SP. Symptoms of depression as a risk factor for incident diabetes: findings from the National Health and Nutrition Examination Epidemiologic Follow-up Study, 1971-1992. Am J Epidemiol 2003;158:416–423 [DOI] [PubMed] [Google Scholar]
- 49.Hannon TS, Rofey DL, Lee S, Arslanian SA. Depressive symptoms and metabolic markers of risk for type 2 diabetes in obese adolescents. Pediatr Diabetes 2013;14:497–503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cappuccio FP, D’Elia L, Strazzullo P, Miller MA. Quantity and quality of sleep and incidence of type 2 diabetes: a systematic review and meta-analysis. Diabetes Care 2010;33:414–420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hannon TS, Lee S, Chakravorty S, Lin Y, Arslanian SA. Sleep-disordered breathing in obese adolescents is associated with visceral adiposity and markers of insulin resistance. Int J Pediatr Obes 2011;6:157–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Koren D, Levitt Katz LE, Brar PC, Gallagher PR, Berkowitz RI, Brooks LJ. Sleep architecture and glucose and insulin homeostasis in obese adolescents. Diabetes Care 2011;34:2442–2447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.American Diabetes Association Classification and diagnosis of diabetes. Sec. 2. In Standard of Medical Care in Diabetes—2017. Diabetes Care 2017;40(Suppl. 1):S11–S24 [DOI] [PubMed] [Google Scholar]
- 54.Buse JB, Kaufman FR, Linder B, Hirst K, El Ghormli L, Willi S; HEALTHY Study Group . Diabetes screening with hemoglobin A1c versus fasting plasma glucose in a multiethnic middle-school cohort. Diabetes Care 2013;36:429–435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nowicka P, Santoro N, Liu H, et al. Utility of hemoglobin A1c for diagnosing prediabetes and diabetes in obese children and adolescents. Diabetes Care 2011;34:1306–1311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee JM, Wu E-L, Tarini B, Herman WH, Yoon E. Diagnosis of diabetes using hemoglobin A1c: should recommendations in adults be extrapolated to adolescents? J Pediatr 2011;158:947–952.e3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chan CL, Pyle L, Newnes L, Nadeau KJ, Zeitler PS, Kelsey MM. Continuous glucose monitoring and its relationship to hemoglobin A1c and oral glucose tolerance testing in obese and prediabetic youth. J Clin Endocrinol Metab 2015;100:902–910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Love-Osborne KA, Sheeder JL, Nadeau KJ, Zeitler P. Longitudinal follow up of dysglycemia in overweight and obese pediatric patients. Pediatr Diabetes 2018;19:199–204 [DOI] [PubMed] [Google Scholar]
- 59.Gungor N, Hannon T, Libman I, Bacha F, Arslanian S. Type 2 diabetes mellitus in youth: the complete picture to date. Pediatr Clin North Am 2005;52:1579–1609 [DOI] [PubMed] [Google Scholar]
- 60.DuBose SN, Hermann JM, Tamborlane WV, et al.; Type 1 Diabetes Exchange Clinic Network and Diabetes Prospective Follow-up Registry . Obesity in youth with type 1 diabetes in Germany, Austria, and the United States. J Pediatr 2015;167:627–632.e4 [DOI] [PubMed] [Google Scholar]
- 61.Klingensmith GJ, Pyle L, Arslanian S, et al.; TODAY Study Group . The presence of GAD and IA-2 antibodies in youth with a type 2 diabetes phenotype: results from the TODAY study. Diabetes Care 2010;33:1970–1975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hannon TS, Arslanian SA. The changing face of diabetes in youth: lessons learned from studies of type 2 diabetes. Ann N Y Acad Sci 2015;1353:113–137 [DOI] [PubMed] [Google Scholar]
- 63.Bacha F, Gungor N, Lee S, de las Heras J, Arslanian S. Indices of insulin secretion during a liquid mixed-meal test in obese youth with diabetes. J Pediatr 2013;162:924–929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tfayli H, Bacha F, Gungor N, Arslanian S. Phenotypic type 2 diabetes in obese youth: insulin sensitivity and secretion in islet cell antibody-negative versus -positive patients. Diabetes 2009;58:738–744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pinhas-Hamiel O, Dolan LM, Zeitler PS. Diabetic ketoacidosis among obese African-American adolescents with NIDDM. Diabetes Care 1997;20:484–486 [DOI] [PubMed] [Google Scholar]
- 66.Smith RJ, Nathan DM, Arslanian SA, Groop L, Rizza RA, Rotter JI. Individualizing therapies in type 2 diabetes mellitus based on patient characteristics: what we know and what we need to know. J Clin Endocrinol Metab 2010;95:1566–1574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kleinberger JW, Copeland KC, Gandica RG, et al. Monogenic diabetes in overweight and obese youth diagnosed with type 2 diabetes: the TODAY clinical trial. Genet Med 2018;20:583–590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Pihoker C, Gilliam LK, Ellard S, et al.; SEARCH for Diabetes in Youth Study Group . Prevalence, characteristics and clinical diagnosis of maturity onset diabetes of the young due to mutations in HNF1A, HNF4A, and glucokinase: results from the SEARCH for Diabetes in Youth. J Clin Endocrinol Metab 2013;98:4055–4062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hattersley AT, Greeley SAW, Polak M, et al. ISPAD 2018 Consensus Guidelines: the diagnosis and management of monogenic diabetes in children and adolescents. Pediatr Diabetes. 17 September 2018. [Epub before print]. DOI: 10.1111/pedi.12772 [DOI] [PubMed] [Google Scholar]
- 70.Zeitler P, Fu J, Tandon N, et al.; International Society for Pediatric and Adolescent Diabetes . ISPAD Clinical Practice Consensus Guidelines 2014. Type 2 diabetes in the child and adolescent. Pediatr Diabetes 2014;15(Suppl. 20):26–46 [DOI] [PubMed] [Google Scholar]
- 71.Zeitler P, Arslanian S, Fu J, et al. Type 2 diabetes mellitus (T2DM) in youth. Pediatr Diabetes. 12 July 2018 [Epub before print]. DOI: 10.1111/pedi.12719
- 72.American Diabetes Association 12. Children and adolescents: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018;41(Suppl. 1):S126–S136 [DOI] [PubMed] [Google Scholar]
- 73.TODAY Study Group Safety and tolerability of the treatment of youth-onset type 2 diabetes: the TODAY experience. Diabetes Care 2013;36:1765–1771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zeitler P, Hirst K, Copeland KC, et al.; TODAY Study Group . HbA1c after a short period of monotherapy with metformin identifies durable glycemic control among adolescents with type 2 diabetes. Diabetes Care 2015;38:2285–2292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Arslanian S, El Ghormli L, Bacha F, et al.; TODAY Study Group . Adiponectin, insulin sensitivity, β-cell function, and racial/ethnic disparity in treatment failure rates in TODAY. Diabetes Care 2017;40:85–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.TODAY Study Group Effects of metformin, metformin plus rosiglitazone, and metformin plus lifestyle on insulin sensitivity and β-cell function in TODAY. Diabetes Care 2013;36:1749–1757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.TODAY Study Group Retinopathy in youth with type 2 diabetes participating in the TODAY clinical trial. Diabetes Care 2013;36:1772–1774 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.TODAY Study Group Lipid and inflammatory cardiovascular risk worsens over 3 years in youth with type 2 diabetes: the TODAY clinical trial. Diabetes Care 2013;36:1758–1764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.TODAY Study Group Rapid rise in hypertension and nephropathy in youth with type 2 diabetes: the TODAY clinical trial. Diabetes Care 2013;36:1735–1741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Padgett D, Mumford E, Hynes M, Carter R. Meta-analysis of the effects of educational and psychosocial interventions on management of diabetes mellitus. J Clin Epidemiol 1988;41:1007–1030 [DOI] [PubMed] [Google Scholar]
- 81.Grey M, Schreiner B, Pyle L. Development of a diabetes education program for youth with type 2 diabetes. Diabetes Educ 2009;35:108–116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.American Diabetes Association Be Healthy Today; Be Healthy For Life [Internet]. Available from http://main.diabetes.org/dorg/PDFs/Type-2-Diabetes-in-Youth/Type-2-Diabetes-in-Youth.pdf. Accessed 12 September 2017
- 83.Atkinson A, Radjenovic D. Meeting quality standards for self-management education in pediatric type 2 diabetes. Diabetes Spectr 2007;20:40–46 [Google Scholar]
- 84.Young-Hyman D, de Groot M, Hill-Briggs F, Gonzalez JS, Hood K, Peyrot M. Psychosocial care for people with diabetes: a position statement of the American Diabetes Association. Diabetes Care 2016;39:2126–2140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Liu LL, Lawrence JM, Davis C, et al.; SEARCH for Diabetes in Youth Study Group . Prevalence of overweight and obesity in youth with diabetes in USA: the SEARCH for Diabetes in Youth study. Pediatr Diabetes 2010;11:4–11 [DOI] [PubMed] [Google Scholar]
- 86.Naughton MJ, Ruggiero AM, Lawrence JM, et al.; SEARCH for Diabetes in Youth Study Group . Health-related quality of life of children and adolescents with type 1 or type 2 diabetes mellitus: SEARCH for Diabetes in Youth Study. Arch Pediatr Adolesc Med 2008;162:649–657 [DOI] [PubMed] [Google Scholar]
- 87.Whalen DJ, Belden AC, Tillman R, Barch DM, Luby JL. Early adversity, psychopathology, and latent class profiles of global physical health from preschool through early adolescence. Psychosom Med 2016;78:1008–1018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Cefalu WT. “TODAY” reflects on the changing “faces” of type 2 diabetes. Diabetes Care 2013;36:1732–1734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lawrence JM, Standiford DA, Loots B, et al.; SEARCH for Diabetes in Youth Study . Prevalence and correlates of depressed mood among youth with diabetes: the SEARCH for Diabetes in Youth study. Pediatrics 2006;117:1348–1358 [DOI] [PubMed] [Google Scholar]
- 90.Levitt Katz LE, Swami S, Abraham M, et al. Neuropsychiatric disorders at the presentation of type 2 diabetes mellitus in children. Pediatr Diabetes 2005;6:84–89 [DOI] [PubMed] [Google Scholar]
- 91.Lewis-Fernández R, Rotheram-Borus MJ, Betts VT, et al. Rethinking funding priorities in mental health research. Br J Psychiatry 2016;208:507–509 [DOI] [PubMed] [Google Scholar]
- 92.Reinehr T. Type 2 diabetes mellitus in children and adolescents. World J Diabetes 2013;4:270–281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care 2011;34:2205–2207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wilfley D, Berkowitz R, Goebel-Fabbri A, et al.; TODAY Study Group . Binge eating, mood, and quality of life in youth with type 2 diabetes: baseline data from the today study. Diabetes Care 2011;34:858–860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Shelton RC. Depression, antidepressants, and weight gain in children. Obesity (Silver Spring) 2016;24:2450. [DOI] [PubMed] [Google Scholar]
- 96.Baeza I, Vigo L, de la Serna E, et al. The effects of antipsychotics on weight gain, weight-related hormones and homocysteine in children and adolescents: a 1-year follow-up study. Eur Child Adolesc Psychiatry 2017;26:35–46 [DOI] [PubMed] [Google Scholar]
- 97.American Diabetes Association 13. Management of diabetes in pregnancy: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018;41(Suppl. 1):S137–S143 [DOI] [PubMed] [Google Scholar]
- 98.Klingensmith GJ, Pyle L, Nadeau KJ, et al.; TODAY Study Group . Pregnancy outcomes in youth with type 2 diabetes: the TODAY study experience. Diabetes Care 2016:39:122–129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zylke JW, Bauchner H. The unrelenting challenge of obesity. JAMA 2016;315:2277–2278 [DOI] [PubMed] [Google Scholar]
- 100.Coles N, Birken C, Hamilton J. Emerging treatments for severe obesity in children and adolescents. BMJ 2016;354:i4116. [DOI] [PubMed] [Google Scholar]
- 101.Danielsson P, Kowalski J, Ekblom Ö, Marcus C. Response of severely obese children and adolescents to behavioral treatment. Arch Pediatr Adolesc Med 2012;166:1103–1108 [DOI] [PubMed] [Google Scholar]
- 102.Savoye M, Shaw M, Dziura J, et al. Effects of a weight management program on body composition and metabolic parameters in overweight children: a randomized controlled trial. JAMA 2007;297:2697–2704 [DOI] [PubMed] [Google Scholar]
- 103.Savoye M, Nowicka P, Shaw M, et al. Long-term results of an obesity program in an ethnically diverse pediatric population. Pediatrics 2011;127:402–410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.TODAY Study Group A clinical trial to maintain glycemic control in youth with type 2 diabetes. N Engl J Med 2012;366:2247–2256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Marcus MD, Wilfley DE, El Ghormli L, et al.; TODAY Study Group . Weight change in the management of youth-onset type 2 diabetes: the TODAY clinical trial experience. Pediatr Obes 2017;12:337–345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Styne DM, Arslanian SA, Connor EL, et al. Pediatric obesity—assessment, treatment, and prevention: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2017;102:709–757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Barlow SE; Expert Committee . Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: summary report. Pediatrics 2007;120(Suppl. 4):S164–S192 [DOI] [PubMed] [Google Scholar]
- 108.Kelly AS, Barlow SE, Rao G, et al.; American Heart Association Atherosclerosis, Hypertension, and Obesity in the Young Committee of the Council on Cardiovascular Disease in the Young, Council on Nutrition, Physical Activity and Metabolism, and Council on Clinical Cardiology . Severe obesity in children and adolescents: identification, associated health risks, and treatment approaches: a scientific statement from the American Heart Association. Circulation 2013;128:1689–1712 [DOI] [PubMed] [Google Scholar]
- 109.Constantino MI, Molyneaux L, Limacher-Gisler F, et al. Long-term complications and mortality in young-onset diabetes: type 2 diabetes is more hazardous and lethal than type 1 diabetes. Diabetes Care 2013;36:3863–3869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Imperatore G, Boyle JP, Thompson TJ, et al.; SEARCH for Diabetes in Youth Study Group . Projections of type 1 and type 2 diabetes burden in the U.S. population aged <20 years through 2050: dynamic modeling of incidence, mortality, and population growth. Diabetes Care 2012;35:2515–2520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Nadeau KJ, Anderson BJ, Berg EG, et al. Youth-onset type 2 diabetes consensus report: current status, challenges, and priorities. Diabetes Care 2016;39:1635–1642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Dietz WH, Solomon LS, Pronk N, et al. An integrated framework for the prevention and treatment of obesity and its related chronic diseases. Health Aff (Millwood) 2015;34:1456–1463 [DOI] [PubMed] [Google Scholar]
- 113.Wilfley DE, Staiano AE, Altman M, et al.; Improving Access and Systems of Care for Evidence-Based Childhood Obesity Treatment Conference Workgroup . Improving access and systems of care for evidence-based childhood obesity treatment: conference key findings and next steps. Obesity (Silver Spring) 2017;25:16–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Rothman RL, Mulvaney S, Elasy TA, et al. Self-management behaviors, racial disparities, and glycemic control among adolescents with type 2 diabetes. Pediatrics 2008;121:e912–e919 [DOI] [PubMed] [Google Scholar]
- 115.Shield JPH, Lynn R, Wan KC, Haines L, Barrett TG. Management and 1 year outcome for UK children with type 2 diabetes. Arch Dis Child 2009;94:206–209 [DOI] [PubMed] [Google Scholar]
- 116.Zeitler P, Haqq A, Rosenbloom A, Glaser N; Drugs and Therapeutics Committee of the Lawson Wilkins Pediatric Endocrine Society . Hyperglycemic hyperosmolar syndrome in children: pathophysiological considerations and suggested guidelines for treatment. J Pediatr 2011;158:9–14.e2. [DOI] [PubMed] [Google Scholar]
- 117.Kelsey MM, Geffner ME, Guandalini C, et al.; Treatment Options for Type 2 Diabetes in Adolescents and Youth Study Group . Presentation and effectiveness of early treatment of type 2 diabetes in youth: lessons from the TODAY study. Pediatr Diabetes 2016:17:212–221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.RISE Consortium Impact of insulin and metformin versus metformin alone on β-cell function in youth with impaired glucose tolerance or recently diagnosed type 2 diabetes. Diabetes Care 2018;41:1717–1725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Levitt Katz LE, Bacha F, Gidding SS, et al.; TODAY Study Group . Lipid profiles, inflammatory markers, and insulin therapy in youth with type 2 diabetes. J Pediatr 2018;196:208–216.e2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Blackburn GL, Hutter MM, Harvey AM, et al. Expert panel on weight loss surgery: executive report update. Obesity (Silver Spring) 2009;17:842–862 [DOI] [PubMed] [Google Scholar]
- 121.Adams TD, Arterburn DE, Nathan DM, Eckel RH. Clinical outcomes of metabolic surgery: microvascular and macrovascular complications. Diabetes Care 2016;39:912–923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Carlsson LMS, Peltonen M, Ahlin S, et al. Bariatric surgery and prevention of type 2 diabetes in Swedish obese subjects. N Engl J Med 2012;367:695–704 [DOI] [PubMed] [Google Scholar]
- 123.Plum L, Ahmed L, Febres G, et al. Comparison of glucostatic parameters after hypocaloric diet or bariatric surgery and equivalent weight loss. Obesity (Silver Spring) 2011;19:2149–2157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Schauer PR, Kashyap SR, Wolski K, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med 2012;366:1567–1576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Schauer PR, Bhatt DL, Kirwan JP, et al.; STAMPEDE Investigators . Bariatric surgery versus intensive medical therapy for diabetes—5-year outcomes. N Engl J Med 2017;376:641–651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet 2015;386:964–973 [DOI] [PubMed] [Google Scholar]
- 127.Rubino F, Nathan DM, Eckel RH, et al.; Delegates of the 2nd Diabetes Surgery Summit . Metabolic surgery in the treatment algorithm for type 2 diabetes: a joint statement by international diabetes organizations. Diabetes Care 2016;39:861–877 [DOI] [PubMed] [Google Scholar]
- 128.Cummings DE, Cohen RV. Bariatric/metabolic surgery to treat type 2 diabetes in patients with a BMI <35 kg/m2. Diabetes Care 2016;39:924–933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Schauer PR, Mingrone G, Ikramuddin S, Wolfe B. Clinical outcomes of metabolic surgery: efficacy of glycemic control, weight loss, and remission of diabetes. Diabetes Care 2016;39:902–911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Pratt JSA, Lenders CM, Dionne EA, et al. Best practice updates for pediatric/adolescent weight loss surgery. Obesity (Silver Spring) 2009;17:901–910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Dolan K, Creighton L, Hopkins G, Fielding G. Laparoscopic gastric banding in morbidly obese adolescents. Obes Surg 2003;13:101–104 [DOI] [PubMed] [Google Scholar]
- 132.Sugerman HJ, Sugerman EL, DeMaria EJ, et al. Bariatric surgery for severely obese adolescents. J Gastrointest Surg 2003;7:102–108 [DOI] [PubMed] [Google Scholar]
- 133.Inge TH, Garcia V, Daniels S, et al. A multidisciplinary approach to the adolescent bariatric surgical patient. J Pediatr Surg 2004;39:442–447; discussion 446–447 [DOI] [PubMed] [Google Scholar]
- 134.Lawson ML, Kirk S, Mitchell T, et al.; Pediatric Bariatric Study Group . One-year outcomes of Roux-en-Y gastric bypass for morbidly obese adolescents: a multicenter study from the Pediatric Bariatric Study Group. J Pediatr Surg 2006;41:137–143 [DOI] [PubMed] [Google Scholar]
- 135.Inge TH, Zeller M, Harmon C, et al. Teen-Longitudinal Assessment of Bariatric Surgery: methodological features of the first prospective multicenter study of adolescent bariatric surgery. J Pediatr Surg 2007;42:1969–1971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Ells LJ, Mead E, Atkinson G, et al. Surgery for the treatment of obesity in children and adolescents. Cochrane Database Syst Rev 2015;6:CD011740. [DOI] [PubMed] [Google Scholar]
- 137.Michalsky MP, Inge TH, Simmons M, et al.; Teen-LABS Consortium . Cardiovascular risk factors in severely obese adolescents: the Teen Longitudinal Assessment of Bariatric Surgery (Teen-LABS) Study. JAMA Pediatr 2015;169:438–444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zeinoddini A, Heidari R, Talebpour M. Laparoscopic gastric plication in morbidly obese adolescents: a prospective study. Surg Obes Relat Dis 2014;10:1135–1139 [DOI] [PubMed] [Google Scholar]
- 139.Göthberg G, Gronowitz E, Flodmark C-E, et al. Laparoscopic Roux-en-Y gastric bypass in adolescents with morbid obesity—surgical aspects and clinical outcome. Semin Pediatr Surg 2014;23:11–16 [DOI] [PubMed] [Google Scholar]
- 140.Inge TH, Prigeon RL, Elder DA, et al. Insulin sensitivity and β-cell function improve after gastric bypass in severely obese adolescents. J Pediatr 2015;167:1042–1048.e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Inge TH, Miyano G, Bean J, et al. Reversal of type 2 diabetes mellitus and improvements in cardiovascular risk factors after surgical weight loss in adolescents. Pediatrics 2009;123:214–222 [DOI] [PubMed] [Google Scholar]
- 142.Inge TH, Laffel LM, Jenkins TM, et al.; Teen–Longitudinal Assessment of Bariatric Surgery (Teen-LABS) and Treatment Options of Type 2 Diabetes in Adolescents and Youth (TODAY) Consortia . Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr 2018;172:452–460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Beamish AJ, D’Alessio DA, Inge TH. Controversial issues: when the drugs don’t work, can surgery provide a different outcome for diabetic adolescents? Surg Obes Relat Dis 2015;11:946–948 [DOI] [PubMed] [Google Scholar]
- 144.Shah AS, D’Alessio D, Ford-Adams ME, Desai AP, Inge TH. Bariatric surgery: a potential treatment for type 2 diabetes in youth. Diabetes Care 2016;39:934–940 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Inge TH, Jenkins TM, Xanthakos SA, et al. Long-term outcomes of bariatric surgery in adolescents with severe obesity (FABS-5+): a prospective follow-up analysis. Lancet Diabetes Endocrinol 2017;5:165–173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Rubin JK, Hinrichs-Krapels S, Hesketh R, Martin A, Herman WH, Rubino F. Identifying barriers to appropriate use of metabolic/bariatric surgery for type 2 diabetes treatment: Policy Lab results. Diabetes Care 2016;39:954–963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Morton JM. Ethnic considerations for metabolic surgery. Diabetes Care 2016;39:949–953 [DOI] [PubMed] [Google Scholar]
- 148.Song SH, Hardisty CA. Early onset type 2 diabetes mellitus: a harbinger for complications in later years—clinical observation from a secondary care cohort. QJM 2009;102:799–806 [DOI] [PubMed] [Google Scholar]
- 149.Eppens MC, Craig ME, Cusumano J, et al. Prevalence of diabetes complications in adolescents with type 2 compared with type 1 diabetes. Diabetes Care 2006;29:1300–1306 [DOI] [PubMed] [Google Scholar]
- 150.Song SH. Complication characteristics between young-onset type 2 versus type 1 diabetes in a UK population. BMJ Open Diabetes Res Care 2015;3:e000044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Dabelea D, Stafford JM, Mayer-Davis EJ, et al.; SEARCH for Diabetes in Youth Research Group . Association of type 1 diabetes vs type 2 diabetes diagnosed during childhood and adolescence with complications during teenage years and young adulthood. JAMA 2017;317:825–835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Tuttle KR, Bakris GL, Bilous RW, et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Diabetes Care 2014;37:2864–2883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Magee GM, Bilous RW, Cardwell CR, Hunter SJ, Kee F, Fogarty DG. Is hyperfiltration associated with the future risk of developing diabetic nephropathy? A meta-analysis. Diabetologia 2009;52:691–697 [DOI] [PubMed] [Google Scholar]
- 154.Ruggenenti P, Porrini EL, Gaspari F, et al.; GFR Study Investigators . Glomerular hyperfiltration and renal disease progression in type 2 diabetes. Diabetes Care 2012;35:2061–2068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Chida S, Fujita Y, Ogawa A, et al. Levels of albuminuria and risk of developing macroalbuminuria in type 2 diabetes: historical cohort study. Sci Rep 2016;6:26380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Burgert TS, Dziura J, Yeckel C, et al. Microalbuminuria in pediatric obesity: prevalence and relation to other cardiovascular risk factors. Int J Obes (Lond) 2006;30:273–280 [DOI] [PubMed] [Google Scholar]
- 157.Bartz SK, Caldas MC, Tomsa A, Krishnamurthy R, Bacha F. Urine albumin-to-creatinine ratio: a marker of early endothelial dysfunction in youth. J Clin Endocrinol Metab 2015;100:3393–3399 [DOI] [PubMed] [Google Scholar]
- 158.Krakoff J, Lindsay RS, Looker HC, Nelson RG, Hanson RL, Knowler WC. Incidence of retinopathy and nephropathy in youth-onset compared with adult-onset type 2 diabetes. Diabetes Care 2003;26:76–81 [DOI] [PubMed] [Google Scholar]
- 159.Dyck RF, Jiang Y, Osgood ND. The long-term risks of end stage renal disease and mortality among First Nations and non-First Nations people with youth-onset diabetes. Can J Diabetes 2014;38:237–243 [DOI] [PubMed] [Google Scholar]
- 160.Dart AB, Sellers EA, Martens PJ, Rigatto C, Brownell MD, Dean HJ. High burden of kidney disease in youth-onset type 2 diabetes. Diabetes Care 2012;35:1265–1271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Nelson RG, Newman JM, Knowler WC, et al. Incidence of end-stage renal disease in type 2 (non-insulin-dependent) diabetes mellitus in Pima Indians. Diabetologia 1988;31:730–736 [DOI] [PubMed] [Google Scholar]
- 162.Bjornstad P, Škrtić M, Lytvyn Y, Maahs DM, Johnson RJ, Cherney DZ. The Gomez’ equations and renal hemodynamic function in kidney disease research. Am J Physiol Renal Physiol 2016;311:F967–F975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Salardi S, Balsamo C, Zucchini S, et al. High rate of regression from micro-macroalbuminuria to normoalbuminuria in children and adolescents with type 1 diabetes treated or not with enalapril: the influence of HDL cholesterol. Diabetes Care 2011;34:424–429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Solis-Herrera C, Triplitt CL, Lynch JL. Nephropathy in youth and young adults with type 2 diabetes. Curr Diab Rep 2014;14:456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Heart Outcomes Prevention Evaluation Study Investigators Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet 2000;355:253–259 [PubMed] [Google Scholar]
- 166.Herman WH, Pop-Busui R, Braffett BH, et al.; DCCT/EDIC Research Group . Use of the Michigan Neuropathy Screening Instrument as a measure of distal symmetrical peripheral neuropathy in type 1 diabetes: results from the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications. Diabet Med 2012;29:937–944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Moghtaderi A, Bakhshipour A, Rashidi H. Validation of Michigan Neuropathy Screening Instrument for diabetic peripheral neuropathy. Clin Neurol Neurosurg 2006;108:477–481 [DOI] [PubMed] [Google Scholar]
- 168.Jaiswal M, Lauer A, Martin CL, et al.; SEARCH for Diabetes in Youth Study Group . Peripheral neuropathy in adolescents and young adults with type 1 and type 2 diabetes from the SEARCH for Diabetes in Youth follow-up cohort: a pilot study. Diabetes Care 2013;36:3903–3908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Mueller MJ. Identifying patients with diabetes mellitus who are at risk for lower-extremity complications: use of Semmes-Weinstein monofilaments. Phys Ther 1996;76:68–71 [DOI] [PubMed] [Google Scholar]
- 170.Amutha A, Datta M, Unnikrishnan R, Anjana RM, Mohan V. Clinical profile and complications of childhood- and adolescent-onset type 2 diabetes seen at a diabetes center in south India. Diabetes Technol Ther 2012;14:497–504 [DOI] [PubMed] [Google Scholar]
- 171.Diabetes Control and Complications Trial Research Group Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. J Pediatr 1994;125:177–188 [DOI] [PubMed] [Google Scholar]
- 172.White NH. Long-term outcomes in youths with diabetes mellitus. Pediatr Clin North Am 2015;62:889–909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.American Diabetes Association 10. Microvascular complications and foot care: Standards of Medical Care in Diabetes—2018. Diabetes Care 2018;41(Suppl. 1):S105–S118 [DOI] [PubMed] [Google Scholar]
- 174.Yokoyama H, Okudaira M, Otani T, et al. Existence of early-onset NIDDM Japanese demonstrating severe diabetic complications. Diabetes Care 1997;20:844–847 [DOI] [PubMed] [Google Scholar]
- 175.Mayer-Davis EJ, Davis C, Saadine J, et al.; SEARCH for Diabetes in Youth Study Group . Diabetic retinopathy in the SEARCH for Diabetes in Youth cohort: a pilot study. Diabet Med 2012;29:1148–1152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Newton KP, Hou J, Crimmins NA, et al.; Nonalcoholic Steatohepatitis Clinical Research Network . Prevalence of prediabetes and type 2 diabetes in children with nonalcoholic fatty liver disease. JAMA Pediatr 2016;170:e161971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Xanthakos SA, Jenkins TM, Kleiner DE, et al.; Teen-LABS Consortium . High prevalence of nonalcoholic fatty liver disease in adolescents undergoing bariatric surgery. Gastroenterology 2015;149:623–634.e8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Chan W-K, Sthaneshwar P, Nik Mustapha NR, Mahadeva S. Limited utility of plasma M30 in discriminating non-alcoholic steatohepatitis from steatosis—a comparison with routine biochemical markers. PLoS One 2014;9:e105903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Loomba R, Sirlin CB, Schwimmer JB, Lavine JE. Advances in pediatric nonalcoholic fatty liver disease. Hepatology 2009;50:1282–1293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Kohli R, Sunduram S, Mouzaki M, et al. Pediatric nonalcoholic fatty liver disease: a report from the Expert Committee on Nonalcoholic Fatty Liver Disease (ECON). J Pediatr 2016;172:9–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Tsai E, Lee T-P. Diagnosis and evaluation of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis, including noninvasive biomarkers and transient elastography. Clin Liver Dis 2018;22:73–92 [DOI] [PubMed] [Google Scholar]
- 182.Lavine JE, Schwimmer JB, Van Natta ML, et al.; Nonalcoholic Steatohepatitis Clinical Research Network . Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA 2011;305:1659–1668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Corey KE, Vuppalanchi R, Vos M, et al.; Nonalcoholic Steatohepatitis Clinical Research Network . Improvement in liver histology is associated with reduction in dyslipidemia in children with nonalcoholic fatty liver disease. J Pediatr Gastroenterol Nutr 2015;60:360–367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Schwimmer JB, Lavine JE, Wilson LA, et al.; NASH CRN . In children with nonalcoholic fatty liver disease, cysteamine bitartrate delayed release improves liver enzymes but does not reduce disease activity scores. Gastroenterology 2016;151:1141–1154.e9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Cappuccio FP, Taggart FM, Kandala N-B, et al. Meta-analysis of short sleep duration and obesity in children and adults. Sleep 2008;31:619–626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Broussard JL, Van Cauter E. Disturbances of sleep and circadian rhythms: novel risk factors for obesity. Curr Opin Endocrinol Diabetes Obes 2016;23:353–359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Reutrakul S, Van Cauter E. Interactions between sleep, circadian function, and glucose metabolism: implications for risk and severity of diabetes. Ann N Y Acad Sci 2014;1311:151–173 [DOI] [PubMed] [Google Scholar]
- 188.Chen X, Beydoun MA, Wang Y. Is sleep duration associated with childhood obesity? A systematic review and meta-analysis. Obesity (Silver Spring) 2008;16:265–274 [DOI] [PubMed] [Google Scholar]
- 189.Rutters F, Gerver WJ, Nieuwenhuizen AG, Verhoef SPM, Westerterp-Plantenga MS. Sleep duration and body-weight development during puberty in a Dutch children cohort. Int J Obes (Lond) 2010;34:1508–1514 [DOI] [PubMed] [Google Scholar]
- 190.Nedeltcheva AV, Kessler L, Imperial J, Penev PD. Exposure to recurrent sleep restriction in the setting of high caloric intake and physical inactivity results in increased insulin resistance and reduced glucose tolerance. J Clin Endocrinol Metab 2009;94:3242–3250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Buxton OM, Pavlova M, Reid EW, Wang W, Simonson DC, Adler GK. Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes 2010;59:2126–2133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Tasali E, Leproult R, Ehrmann DA, Van Cauter E. Slow-wave sleep and the risk of type 2 diabetes in humans. Proc Natl Acad Sci U S A 2008;105:1044–1049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. Lancet 1999;354:1435–1439 [DOI] [PubMed] [Google Scholar]
- 194.Javaheri S, Storfer-Isser A, Rosen CL, Redline S. Association of short and long sleep durations with insulin sensitivity in adolescents. J Pediatr 2011;158:617–623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Watson SE, Li Z, Tu W, et al. Obstructive sleep apnoea in obese adolescents and cardiometabolic risk markers. Pediatr Obes 2014;9:471–477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Ayas NT, White DP, Al-Delaimy WK, et al. A prospective study of self-reported sleep duration and incident diabetes in women. Diabetes Care 2003;26:380–384 [DOI] [PubMed] [Google Scholar]
- 197.Gangwisch JE, Heymsfield SB, Boden-Albala B, et al. Sleep duration as a risk factor for diabetes incidence in a large U.S. sample. Sleep 2007;30:1667–1673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Tasali E, Mokhlesi B, Van Cauter E. Obstructive sleep apnea and type 2 diabetes: interacting epidemics. Chest 2008;133:496–506 [DOI] [PubMed] [Google Scholar]
- 199.Chaput J-P, Després J-P, Bouchard C, Tremblay A. Association of sleep duration with type 2 diabetes and impaired glucose tolerance. Diabetologia 2007;50:2298–2304 [DOI] [PubMed] [Google Scholar]
- 200.Jean-Louis G, Zizi F, Clark LT, Brown CD, McFarlane SI. Obstructive sleep apnea and cardiovascular disease: role of the metabolic syndrome and its components. J Clin Sleep Med 2008;4:261–272 [PMC free article] [PubMed] [Google Scholar]
- 201.Buxton OM, Cain SW, O’Connor SP, et al. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci Transl Med 2012;4:129ra43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Shaw ND, McHill AW, Schiavon M, et al. Effect of slow wave sleep disruption on metabolic parameters in adolescents. Sleep (Basel) 2016;39:1591–1599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Mokhlesi B, Grimaldi D, Beccuti G, et al. Effect of one week of 8-hour nightly continuous positive airway pressure treatment of obstructive sleep apnea on glycemic control in type 2 diabetes: a proof-of-concept study. Am J Respir Crit Care Med 2016;194:516–519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Martínez-Cerón E, Barquiel B, Bezos A-M, et al. Effect of continuous positive airway pressure on glycemic control in patients with obstructive sleep apnea and type 2 diabetes. a randomized clinical trial. Am J Respir Crit Care Med 2016;194:476–485 [DOI] [PubMed] [Google Scholar]
- 205.Zychowski KE, Sanchez B, Pedrosa RP, et al. Serum from obstructive sleep apnea patients induces inflammatory responses in coronary artery endothelial cells. Atherosclerosis 2016;254:59–66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Ehrmann DA. Polycystic ovary syndrome. N Engl J Med 2005;352:1223–1236 [DOI] [PubMed] [Google Scholar]
- 207.Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab 2004;89:2745–2749 [DOI] [PubMed] [Google Scholar]
- 208.Christensen SB, Black MH, Smith N, et al. Prevalence of polycystic ovary syndrome in adolescents. Fertil Steril 2013;100:470–477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Dunaif A. Insulin action in the polycystic ovary syndrome. Endocrinol Metab Clin North Am 1999;28:341–359 [DOI] [PubMed] [Google Scholar]
- 210.Lewy VD, Danadian K, Witchel SF, Arslanian S. Early metabolic abnormalities in adolescent girls with polycystic ovarian syndrome. J Pediatr 2001;138:38–44 [DOI] [PubMed] [Google Scholar]
- 211.Arslanian SA, Lewy VD, Danadian K. Glucose intolerance in obese adolescents with polycystic ovary syndrome: roles of insulin resistance and β-cell dysfunction and risk of cardiovascular disease. J Clin Endocrinol Metab 2001;86:66–71 [DOI] [PubMed] [Google Scholar]
- 212.Palmert MR, Gordon CM, Kartashov AI, Legro RS, Emans SJ, Dunaif A. Screening for abnormal glucose tolerance in adolescents with polycystic ovary syndrome. J Clin Endocrinol Metab 2002;87:1017–1023 [DOI] [PubMed] [Google Scholar]
- 213.Legro RS, Arslanian SA, Ehrmann DA, et al.; Endocrine Society . Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2013;98:4565–4592 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Witchel SF, Oberfield S, Rosenfield RL, et al. The diagnosis of polycystic ovary syndrome during adolescence. Horm Res Paediatr 2015;83:376–389 [DOI] [PubMed] [Google Scholar]
- 215.Kelsey MM, Braffett BH, Geffner ME, et al.; TODAY Study Group . Menstrual dysfunction in girls From the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) study. J Clin Endocrinol Metab 2018;103:2309–2318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Hoeger K, Davidson K, Kochman L, Cherry T, Kopin L, Guzick DS. The impact of metformin, oral contraceptives, and lifestyle modification on polycystic ovary syndrome in obese adolescent women in two randomized, placebo-controlled clinical trials. J Clin Endocrinol Metab 2008;93:4299–4306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Ornstein RM, Copperman NM, Jacobson MS. Effect of weight loss on menstrual function in adolescents with polycystic ovary syndrome. J Pediatr Adolesc Gynecol 2011;24:161–165 [DOI] [PubMed] [Google Scholar]
- 218.Lass N, Kleber M, Winkel K, Wunsch R, Reinehr T. Effect of lifestyle intervention on features of polycystic ovarian syndrome, metabolic syndrome, and intima-media thickness in obese adolescent girls. J Clin Endocrinol Metab 2011;96:3533–3540 [DOI] [PubMed] [Google Scholar]
- 219.Mastorakos G, Koliopoulos C, Deligeoroglou E, Diamanti-Kandarakis E, Creatsas G. Effects of two forms of combined oral contraceptives on carbohydrate metabolism in adolescents with polycystic ovary syndrome. Fertil Steril 2006;85:420–427 [DOI] [PubMed] [Google Scholar]
- 220.Mastorakos G, Koliopoulos C, Creatsas G. Androgen and lipid profiles in adolescents with polycystic ovary syndrome who were treated with two forms of combined oral contraceptives. Fertil Steril 2002;77:919–927 [DOI] [PubMed] [Google Scholar]
- 221.Bridger T, MacDonald S, Baltzer F, Rodd C. Randomized placebo-controlled trial of metformin for adolescents with polycystic ovary syndrome. Arch Pediatr Adolesc Med 2006;160:241–246 [DOI] [PubMed] [Google Scholar]
- 222.Arslanian SA, Lewy V, Danadian K, Saad R. Metformin therapy in obese adolescents with polycystic ovary syndrome and impaired glucose tolerance: amelioration of exaggerated adrenal response to adrenocorticotropin with reduction of insulinemia/insulin resistance. J Clin Endocrinol Metab 2002;87:1555–1559 [DOI] [PubMed] [Google Scholar]
- 223.Vuorio A, Kuoppala J, Kovanen PT, et al. Statins for children with familial hypercholesterolemia. Cochrane Database Syst Rev 2017;7:CD006401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents: summary report. Pediatrics 2011;128(Suppl. 5):S213–S256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Marcovecchio ML, Chiesa ST, Bond S, et al.; AdDIT Study Group . ACE inhibitors and statins in adolescents with type 1 diabetes. N Engl J Med 2017;377:1733–1745 [DOI] [PubMed] [Google Scholar]
- 226.Kavey R-EW, Allada V, Daniels SR, et al.; American Heart Association Expert Panel on Population and Prevention Science; American Heart Association Council on Cardiovascular Disease in the Young; American Heart Association Council on Epidemiology and Prevention; American Heart Association Council on Nutrition, Physical Activity and Metabolism; American Heart Association Council on High Blood Pressure Research; American Heart Association Council on Cardiovascular Nursing; American Heart Association Council on the Kidney in Heart Disease; Interdisciplinary Working Group on Quality of Care and Outcomes Research . Cardiovascular risk reduction in high-risk pediatric patients: a scientific statement from the American Heart Association Expert Panel on Population and Prevention Science; the Councils on Cardiovascular Disease in the Young, Epidemiology and Prevention, Nutrition, Physical Activity and Metabolism, High Blood Pressure Research, Cardiovascular Nursing, and the Kidney in Heart Disease; and the Interdisciplinary Working Group on Quality of Care and Outcomes Research: endorsed by the American Academy of Pediatrics. Circulation 2006;114:2710–2738 [DOI] [PubMed] [Google Scholar]
- 227.Sarwar N, Gao P, Seshasai SR, et al.; Emerging Risk Factors Collaboration . Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies [published correction appears in Lancet 2010;376:958]. Lancet 2010;375:2215–2222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Huo X, Gao L, Guo L, et al. Risk of non-fatal cardiovascular diseases in early-onset versus late-onset type 2 diabetes in China: a cross-sectional study. Lancet Diabetes Endocrinol 2016;4:115–124 [DOI] [PubMed] [Google Scholar]
- 229.Yang W, Cai X, Han X, Ji L. Clinical characteristics of young type 2 diabetes patients with atherosclerosis. PLoS One 2016;11:e0159055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Hillier TA, Pedula KL. Complications in young adults with early-onset type 2 diabetes: losing the relative protection of youth. Diabetes Care 2003;26:2999–3005 [DOI] [PubMed] [Google Scholar]
- 231.McGill HC Jr, McMahan CA, Herderick EE, et al.; Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group . Obesity accelerates the progression of coronary atherosclerosis in young men. Circulation 2002;105:2712–2718 [DOI] [PubMed] [Google Scholar]
- 232.Baker JL, Olsen LW, Sørensen TIA. Childhood body-mass index and the risk of coronary heart disease in adulthood. N Engl J Med 2007;357:2329–2337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Tirosh A, Shai I, Afek A, et al. Adolescent BMI trajectory and risk of diabetes versus coronary disease. N Engl J Med 2011;364:1315–1325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Mahoney LT, Burns TL, Stanford W, et al. Coronary risk factors measured in childhood and young adult life are associated with coronary artery calcification in young adults: the Muscatine Study. J Am Coll Cardiol 1996;27:277–284 [DOI] [PubMed] [Google Scholar]
- 235.Juonala M, Magnussen CG, Venn A, et al. Influence of age on associations between childhood risk factors and carotid intima-media thickness in adulthood: the Cardiovascular Risk in Young Finns Study, the Childhood Determinants of Adult Health Study, the Bogalusa Heart Study, and the Muscatine Study for the International Childhood Cardiovascular Cohort (i3C) Consortium. Circulation 2010;122:2514–2520 [DOI] [PubMed] [Google Scholar]
- 236.Gungor N, Thompson T, Sutton-Tyrrell K, Janosky J, Arslanian S. Early signs of cardiovascular disease in youth with obesity and type 2 diabetes. Diabetes Care 2005;28:1219–1221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Shah AS, Dolan LM, Kimball TR, et al. Influence of duration of diabetes, glycemic control, and traditional cardiovascular risk factors on early atherosclerotic vascular changes in adolescents and young adults with type 2 diabetes mellitus. J Clin Endocrinol Metab 2009;94:3740–3745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Wadwa RP, Urbina EM, Anderson AM, et al.; SEARCH Study Group . Measures of arterial stiffness in youth with type 1 and type 2 diabetes: the SEARCH for Diabetes in Youth study. Diabetes Care 2010;33:881–886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Bacha F, Edmundowicz D, Sutton-Tyrell K, Lee S, Tfayli H, Arslanian SA. Coronary artery calcification in obese youth: what are the phenotypic and metabolic determinants? Diabetes Care 2014;37:2632–2639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Levitt Katz L, Gidding SS, Bacha F, et al.; TODAY Study Group . Alterations in left ventricular, left atrial, and right ventricular structure and function to cardiovascular risk factors in adolescents with type 2 diabetes participating in the TODAY clinical trial. Pediatr Diabetes 2015;16:39–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Bacha F, Gidding SS, Pyle L, et al.; Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) Study Group . Relationship of cardiac structure and function to cardiorespiratory fitness and lean body mass in adolescents and young adults with type 2 diabetes. J Pediatr 2016;177:159–166.e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Juutilainen A, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Type 2 diabetes as a “coronary heart disease equivalent”: an 18-year prospective population-based study in Finnish subjects. Diabetes Care 2005;28:2901–2907 [DOI] [PubMed] [Google Scholar]
- 243.Tancredi M, Rosengren A, Svensson A-M, et al. Excess mortality among persons with type 2 diabetes. N Engl J Med 2015;373:1720–1732 [DOI] [PubMed] [Google Scholar]
- 244.Waernbaum I, Blohmé G, Ostman J, et al. Excess mortality in incident cases of diabetes mellitus aged 15 to 34 years at diagnosis: a population-based study (DISS) in Sweden. Diabetologia 2006;49:653–659 [DOI] [PubMed] [Google Scholar]
- 245.Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care 2014;37:436–443 [DOI] [PubMed] [Google Scholar]
- 246.Al-Saeed AH, Constantino MI, Molyneaux L, et al. An inverse relationship between age of type 2 diabetes onset and complication risk and mortality: the impact of youth-onset type 2 diabetes. Diabetes Care 2016;39:823–829 [DOI] [PubMed] [Google Scholar]
- 247.Reid GJ, Irvine MJ, McCrindle BW, et al. Prevalence and correlates of successful transfer from pediatric to adult health care among a cohort of young adults with complex congenital heart defects. Pediatrics 2004;113:e197–e205 [DOI] [PubMed] [Google Scholar]
- 248.Blum RW, Garell D, Hodgman CH, et al. Transition from child-centered to adult health-care systems for adolescents with chronic conditions. A position paper of the Society for Adolescent Medicine. J Adolesc Health 1993;14:570–576 [DOI] [PubMed] [Google Scholar]
- 249.American Academy of Pediatrics; American Academy of Family Physicians; American College of Physicians–American Society of Internal Medicine A consensus statement on health care transitions for young adults with special health care needs. Pediatrics 2002;110:1304–1306 [PubMed] [Google Scholar]
- 250.Turner JR, Schatz DA, Cusi K, Strumph P. Healthcare transition from pediatric to adult medical homes in diabetes mellitus. Endocr Pract 2014;20:715–721 [DOI] [PubMed] [Google Scholar]
- 251.Reiss J, Gibson R. Health care transition: destinations unknown. Pediatrics 2002;110:1307–1314 [PubMed] [Google Scholar]
- 252.Peters A, Laffel L; American Diabetes Association Transitions Working Group . Diabetes care for emerging adults: recommendations for transition from pediatric to adult diabetes care systems. A position statement of the American Diabetes Association, with representation by the American College of Osteopathic Family Physicians, the American Academy of Pediatrics, the American Association of Clinical Endocrinologists, the American Osteopathic Association, the Centers for Disease Control and Prevention, Children with Diabetes, The Endocrine Society, the International Society for Pediatric and Adolescent Diabetes, Juvenile Diabetes Research Foundation International, the National Diabetes Education Program, and the Pediatric Endocrine Society (formerly Lawson Wilkins Pediatric Endocrine Society). Diabetes Care 2011;34:2477–2485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Chiang JL, Kirkman MS, Laffel LMB, Peters AL; Type 1 Diabetes Sourcebook Authors . Type 1 diabetes through the life span: a position statement of the American Diabetes Association. Diabetes Care 2014;37:2034–2054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Chiang JL, Maahs DM, Garvey KC, et al. Type 1 diabetes in children and adolescents: a position statement by the American Diabetes Association. Diabetes Care 2018;41:2026–2044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Lotstein DS, Seid M, Klingensmith G, et al.; SEARCH for Diabetes in Youth Study Group . Transition from pediatric to adult care for youth diagnosed with type 1 diabetes in adolescence. Pediatrics 2013;131:e1062–e1070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Van Walleghem N, MacDonald CA, Dean HJ. Building connections for young adults with type 1 diabetes mellitus in Manitoba: feasibility and acceptability of a transition initiative. Chronic Dis Can 2006;27:130–134 [PubMed] [Google Scholar]
- 257.Van Walleghem N, MacDonald CA, Dean HJ. Evaluation of a systems navigator model for transition from pediatric to adult care for young adults with type 1 diabetes. Diabetes Care 2008;31:1529–1530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Van Walleghem N, MacDonald CA, Dean HJ. The Maestro Project: a patient navigator for the transition of care for youth with type 1 diabetes. Diabetes Spectr 2011;24:9–13 [Google Scholar]
- 259.Salamon KS, Brouwer AM, Fox MM, et al. Experiencing type 2 diabetes mellitus: qualitative analysis of adolescents’ concept of illness, adjustment, and motivation to engage in self-care behaviors. Diabetes Educ 2012;38:543–551 [DOI] [PubMed] [Google Scholar]
- 260.Arslanian S, Kim JY, Nasr A, et al. Insulin sensitivity across the lifespan from obese adolescents to obese adults with impaired glucose tolerance: who is worse off? Pediatr Diabetes 2018;19:205–211 [DOI] [PubMed] [Google Scholar]
- 261.Chen ME, Chandramouli AG, Considine RV, Hannon TS, Mather KJ. Comparison of β-cell function between overweight/obese adults and adolescents across the spectrum of glycemia. Diabetes Care 2018;41:318–325 [DOI] [PMC free article] [PubMed] [Google Scholar]


