Abstract
The thalamus is composed of multiple nuclei densely connected with the cortex in an organized manner, forming parallel thalamocortical networks critical to sensory, motor, and cognitive functioning. Thalamocortical circuit dysfunction has been implicated in multiple neurodevelopmental disorders, including schizophrenia, which also often exhibit sex differences in prevalence, clinical characteristics, and neuropathology. However, very little is known about developmental and sex effects on thalamocortical networks in youth. The present study characterized the effects of age, sex and psychosis symptomatology in anatomically constrained thalamocortical networks in a large community sample of youth (n = 1100, aged 8–21) from the Philadelphia Neurodevelopmental Cohort (PNC). Cortical functional connectivity of seven anatomically defined thalamic nuclear groups were examined: anterior, mediodorsal, ventral lateral, ventral posterolateral, pulvinar, medial and lateral geniculate nuclear groups. Age and sex effects were characterized using complementary thalamic region-of-interest (ROI) to cortical ROI and voxel-wise analyses. Effects of clinical symptomatology were analyzed by separating youth into three groups based on their clinical symptoms; typically developing youth (n = 298), psychosis spectrum youth (n = 320), and youth with other psychopathologies (n = 482). As an exploratory analysis, association with PRIME scores were used as a dimensional measure of psychopathology. Age effects were broadly characterized by decreasing connectivity with sensory/motor cortical areas, and increasing connectivity with heteromodal prefrontal and parietal cortical areas. This pattern was most pronounced for thalamic motor and sensory nuclei. Females showed greater connectivity between multiple thalamic nuclear groups and the visual cortex compared to males, while males showed greater connectivity with the inferior frontal and orbitofrontal cortices. Youth with psychosis spectrum symptoms showed a subtle decrease in thalamic connectivity with the premotor and prefrontal cortices. Across all youth, greater PRIME scores were associated with lower connectivity between the prefrontal cortex and mediodorsal thalamus. By characterizing typical development in anatomically constrained thalamocortical networks, this study provides an anchor for conceptualizing disruptions to the integrity of these networks observed in neurodevelopmental disorders.
Keywords: Thalamus, Thalamocortical connectivity, Development, Adolescence, Psychosis spectrum, Resting state
1. Introduction
The thalamus is composed of multiple nuclei densely connected with the cortex. Each nucleus has distinct neuroanatomical connections and functions, and can be broadly segregated into sensory/motor and association nuclei based on their neuroanatomical connections (Jones, 2007). Sensory/motor nuclei are connected with sensory organs and primary sensory/motor cortices, while association nuclei are connected to multiple association cortical areas, such as prefrontal and posterior parietal cortices. Historically the thalamus has been conceptualized as a passive relay center, based largely on the organization of sensory systems. However, this view has changed—there is growing evidence that the thalamus, particularly association nuclei, are critically involved in mechanisms of cognitive function, including working memory, cognitive flexibility, and attentional control (Bolkan et al., 2017; Parnaudeau et al., 2013, 2015; Rikhye et al., 2018; Saalmann et al., 2012; Schmitt et al., 2017; Wimmer et al., 2015; Zhou et al., 2016).
The thalamus has been implicated in several psychiatric illnesses, mostly notably schizophrenia. Thalamic dysfunction in schizophrenia is observed in markers of neuronal integrity, reduced thalamus volume, and altered thalamic function during cognitive tasks (Anticevic et al., 2014; Huang et al., 2019, 2020; Kraguljac et al., 2012; Minzenberg et al., 2009; Ragland et al., 2009; van Erp et al., 2016; Woodward et al., 2012). Abnormal functional connectivity of the thalamus, often characterized by a distinct pattern of prefrontal–thalamus hypoconnectivity and sensory/motor–thalamus hyperconnectivity, is one of the most consistent findings in functional neuroimaging studies of schizophrenia (Giraldo-Chica and Woodward, 2017; Ramsay, 2019). Thalamocortical dysconnectivity is present in both non-affective psychosis (i.e. schizophrenia spectrum disorders) and affective psychosis (i.e. bipolar disorder with psychotic features) (Anticevic et al., 2014; Woodward and Heckers, 2016), suggesting that it may be a neurobiological phenotype that is present across the psychosis spectrum.
Evidence of abnormal thalamocortical functional connectivity in early stage psychosis (Woodward and Heckers, 2016) and high-risk individuals (Anticevic et al., 2015; Colibazzi et al., 2017; Dean et al., 2018; Zhu et al., 2019) is consistent with a neurodevelopmental hypothesis of schizophrenia, which posits thalamic abnormalities result from atypical brain development (Andreasen et al., 1998; Cronenwett and Csernansky, 2010; Jones, 1997; Pergola et al., 2015; Sim et al., 2006; Steullet, 2019; Swerdlow, 2010). Many signs of brain dysfunction observed in psychosis manifest during childhood and early adolescence, years before the onset of florid psychosis, and identifying these neurobiological changes allows earlier interventions in those at risk. Youth showing subclinical psychosis symptoms are at elevated risk of converting to a full-blown psychotic disorder, and even those that do not convert demonstrate impaired cognition, elevated mood symptoms and anxiety, higher rates of substance abuse, and reduced global functioning (Marenco and Weinberger, 2000; Seidman et al., 2016). However, neurodevelopmental explanations for thalamocortical dysconnectivity face a significant challenge: relatively little is known about the normal development of thalamocortical functional connectivity. Based largely on an early study of 52 healthy individuals that found greater prefrontal–thalamus connectivity and lower sensory/motor–thalamus connectivity in adults compared to children (Fair et al., 2010), we hypothesized that the pattern of lower frontal and exaggerated somatomotor connectivity with the thalamus in schizophrenia was a consequence of disrupted maturation during adolescence (Woodward et al., 2012). However, a recent study of 95 individuals aged 5–25 years found that except for the cortical dorsal attention network, thalamus connectivity with the cortex generally decreases with age, or remains largely stable (Steiner et al., 2020). The somewhat discrepant findings across studies, which included relatively few individuals, underscore the need for more investigation of age and sex effects using relatively large sample sizes.
Our aims in this study were to: (1) chart age effects of thalamocortical functional connectivity from childhood to young adulthood; (2) characterize sex differences in thalamocortical functional connectivity in youth; and (3) determine whether youth with psychosis spectrum symptoms demonstrate abnormalities in thalamocortical functional connectivity compared to healthy youth and youth with other psychopathologies.
2. Methods
2.1. Study participants
Participants were part of the Philadelphia Neurodevelopmental Cohort (PNC) obtained from the database of Genotypes and Phenotypes (dbGaP). We used the most recent release of the PNC (Study Accession phs000607.v3.p2), consisting of 9498 youth (aged 8–21) including the full neuroimaging sample (n = 1601). Of the 1397 with completed resting state data, 57 were excluded for serious medical conditions, 58 for insufficient clinical data to reach a diagnosis, and 182 for scan quality (e.g. motion, structural image segmentation, normalization). See Table 1 for demographics of our final sample including 1100 youth. Using similar procedures as previously reported (Huang et al., 2020; Roalf et al., 2017), we classified youth into typically developing (n = 298), psychosis spectrum (n = 320), and youth with other psychopathologies (n = 482).
Table 1.
Demographics of study participants.
| Typically Developing (N = 298) | Psychosis Spectrum (N = 320) | Other Psychopathology (N = 482) | Statistics |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| df | X2 /t/F | p | Post-Hoc | |||||||
| Gender (F:M) | 149:149 | 179:141 | 267:215 | 2.78 | .249 | – | ||||
| Ethnicity (W:AA:O) | 168:99:29 | 99:179:41 | 241:198:41 | 47.74 | < 0.001 | – | ||||
| Mean | SD | Mean | SD | Mean | SD | |||||
| Age | 14.8 | 3.6 | 16.1 | 2.9 | 15.5 | 3.3 | 2,1097 | 11.92 | < 0.001 | PS > OP > TD |
| Education | 7.7 | 3.6 | 8.4 | 2.6 | 8.5 | 3.2 | 2,1097 | 5.43 | .004 | PS,OP > TD |
| Parental Education Neuropsychological Functioning |
14.5 | 2.4 | 13.6 | 2.2 | 14.2 | 2.3 | 2,1091 | 13.28 | < 0.001 | TD,OP > PS |
| Global Cognition (z-score) | 0.12 | 0.51 | 0.05 | 0.51 | 0.16 | 0.50 | 2,1097 | 4.11 | .017 | OP > PS |
| WRAT (standard score) | 105.6 | 15.9 | 99.2 | 17.0 | 102.9 | 16.1 | 2,1097 | 11.86 | < 0.001 | TD > OP > PS |
Abbreviations: AA = African American; F = Female; M = Male; O =Other; W = White; WRAT = Wide Range Assessment Test.
Clinical assessments included structured interviews and rating scales previously described (Calkins et al., 2014). All participants underwent clinical assessment with the GOASSESS, a computerized structured clinical interview, collecting demographic data, a timeline of live events, medical history data and including an abbreviated version of the Schedule for Affective Disorders and Schizophrenia for School-Age Children (K-SADS), which assesses a range of psychopathology symptoms. Psychosis symptomatology was assessed with the PRIME Screen-Revised (PS-R), K-SADS psychosis screen, and select items from the Scale for Prodromal Syndromes (SOPS), assessing for negative or disorganized symptoms. Psychosis spectrum youth were identified based on (1) having an age-deviant total PRIME score ≥2 SD above age-matched peers, or rating 1 item at >6, or 3 items >5, (2) endorsing definite or possible hallucinations or delusions on the K-SADS and (3) having an age-deviant total SOPS score ≥2 SD above age-matched peers. Youth with other psychopathologies were identified as youth endorsing sub-threshold psychopathology symptoms and duration consistent with SCID-IV diagnosis on the K-SADS and reporting significant distress, but did not meet psychosis spectrum criteria. Typically developing youth were identified as youth who did not meet either psychosis spectrum or other psychopathologies criteria. See Supplemental Materials for full description of the clinical characteristics.
Study procedures were approved by the institutional review boards at the University of Pennsylvania and the Children’s Hospital of Philadelphia. Written informed consent was obtained from all adult participants and assent was obtained from minors as well as written informed consent from their parent or guardian.
2.2. Neuroimaging data and acquisition and preprocessing
Neuroimage data storage and processing took place on the Vanderbilt University Institute of Imaging Science Center for Computational Imaging XNAT (Harrigan et al., 2016; Huo et al., 2018). The processing pipelines are accessible through github (https://github.com/baxpr/freesurfer-singularity; https://github.com/baxpr/connprep; https://github.com/baxpr/fmriqa; https://github.com/baxpr/fsthalconnMNI-public) and were containerized using Singularity and built at SingularityHub (Sochat et al., 2017) (https://singularity-hub.org).
2.2.1. Neuroimaging acquisition
Structural scan: High resolution T1-weighted structural scans were acquired with the MPRAGE sequence on a Siemens Tim Trio 3T scanner with a 32-channel head coil with 0.93 × 0.93 × 1 mm voxels (160 slices, TR/TE = 1810/3.5, FOV = 180 × 240 × 160, matrix = 192 × 256 × 160; flip angle = 9°).
Resting state scan: A 6:18 min T2∗-weighted resting state BOLD scan was acquired for each participant with a single-shot, interleaved, multi-slice, gradient-echo EPI sequence. 124 volumes were acquired with a voxel resolution 3mm3 isotropic (46 slices, TR/TE = 3000/32, FOV = 192 × 192, matrix = 64 × 64, flip angle = 90°).
2.2.2. Neuroimaging preprocessing
Anatomical images were segmented into gray matter, white matter and cerebrospinal fluid (CSF) with the Computational Anatomy Toolbox 12 (CAT12, version 12.5; http://www.neuro.uni-jena.de/cat/). Resting-state scans were preprocessed in SPM12 and were (1) realigned to a mean scan, (2) coregistered with the native space structural scan, then (3) underwent resting-state denoising procedures: bandpass filter (0.01–0.1 Hz), regression of CSF and white matter signal, regression of 12 motion parameters (6 translation and rotation parameters and their first derivative). All resting state scans went through a quality assurance procedure that included calculating framewise displacement (FD) and temporal signal to noise ratio (tSNR). Scans with a mean FD > 0.5 or a tSNR lower than the 5th percentile of the distribution of the entire sample were excluded from further analysis.
2.3. Regions-of-interest definition
Regions-of-interest (ROIs) used in the functional connectivity analyses described below were defined with FreeSurfer 6 development version including the thalamus segmentation module (Iglesias et al., 2018) using standard parameters (Dale et al., 1999; Fischl et al., 2002). Cortical ROIs were defined for each participant based on the Desikan-Killiany Atlas/DKT atlas. Seven cortical ROIs were created for each hemisphere: prefrontal, parietal, temporal, motor (including the premotor and supplementary motor regions), somatosensory, visual cortices and the hippocampus (see Supplementary Fig. 1 and Supplementary Table 2). Using the thalamus segmentation module from FreeSurfer, we created 7 nuclear groups for each hemisphere: anterior, mediodorsal, pulvinar, ventral lateral (including ventrolateral and ventral anterior nuclei), ventral posterolateral, medial and lateral geniculate nuclear groups (see Supplementary Fig. 2 and Supplementary Table 3). To complement the main analyses using structurally defined thalamic seeds, we used a set of functionally defined thalamic seeds based on a previous study with an independent developmental sample (Woodward et al., 2017).
2.4. Functional connectivity analyses
In order to enhance scientific rigor, comprehensively map thalamocortical connectivity at multiple levels (i.e. cortex, thalamus), and facilitate comparisons of our results with prior studies of thalamic functional connectivity development and abnormalities in psychotic disorders, two approaches were used to examine thalamocortical connectivity. First, to maximize statistical power and ensure we are examining thalamocortical networks with a strong anatomical connectivity basis, we performed ROI-to-ROI analyses that quantified functional connectivity between structurally defined thalamic nuclear groups and cortical regions that are their primary source of anatomical connections (e.g. the mediodorsal nucleus with the prefrontal cortex). We complemented our primary analysis with a voxel-wise analysis of the cortical connectivity pattern with each of our thalamic nuclear groups. These two methods are described in detail below.
2.4.1. ROI-to-ROI analyses
Seven predefined thalamocortical networks were created by pairing each thalamic nuclear group with their primary anatomically connected cortical region. These networks were (1) prefrontal–mediodorsal (2) motor–ventral lateral (3) somatosensory–ventral posterolateral (4) temporal–medial geniculate (5) parietal–pulvinar (6) occipital–lateral geniculate (7) hippocampus–anterior nuclear groups (Behrens et al., 2003; Johansen-Berg et al., 2005; Zhang and Li, 2010). Connectivity (measured as Fisher’s Z) for each of these networks were calculated as follows. Time-courses were extracted from the denoised individual subject space resting state scans for each thalamic and cortical ROI, separately for the left and right hemispheres. For each cortical ROI, connectivity with the thalamic ROI from the same hemisphere was calculated, then connectivity for each cortical ROI–thalamic ROI pair was averaged across left and right hemispheres to obtain a bilateral connectivity value. For validation purposes, we plotted the full connectivity between each cortical with each thalamic ROI (see Supplementary Fig. 3). Statistical analyses in predefined thalamocortical networks were conducted in SPSS (Version 27), as these analyses were repeated over 7 thalamocortical networks, critical alpha was set at a Bonferroni corrected alpha of 0.007.
2.4.2. Thalamic seed-based voxel-wise analyses
Seven separate seed-based voxel-wise functional connectivity analyses were conducted for each of the six thalamic ROIs in SPM12. For each thalamic ROI, the seed time-course was extracted from the thalamic ROI in the denoised individual subject space (native space) resting state scans. Deformation parameters from normalizing T1 structural scans were applied to the resting state scans. The seed time-course was then correlated with every voxel in the denoised normalized resting state scans. Smoothing (6 mm FWHM Gaussian filter) was applied to each resulting connectivity image. Finally, for each thalamic nuclear group, a bilateral connectivity map was created by averaging connectivity values of each voxel across the left and right thalamic ROI for that nuclear group. Cluster significance was determined at p < .05 familywise error (FWE) corrected clusters with a cluster forming threshold of p < .001.
2.5. Statistical analyses
2.5.1. Age effects
In ROI-to-ROI analyses, age effects in thalamocortical networks were modeled as linear and quadratic effects of age using the following linear regression models separately for each thalamocortical network, or thalamus seed:
Linear model
| (1) |
Quadratic model
| (2) |
For all models investigating the effects of age, sex and motion (FD) were included as covariates of no interest. For voxel-wise analyses, one model was applied per voxel, then corrected for multiple comparisons at the cluster level.
2.5.2. Sex effects
In ROI-to-ROI analyses, sex effects were investigated using an omnibus Analysis of Covariance (ANCOVA) with each thalamo-cortical network included as repeated measures, and sex as the independent variable, including age, age2 and motion (FD) as covariates of no interest, followed by one-way univariate ANCOVAs for each thalamocortical network with sex as the independent variable and age, age2 and motion (FD) as covariates of no interest.
ANCOVAs were conducted separately for whole-brain voxel-wise connectivity from each of the 7 thalamus seeds, with sex as an independent variable and age, age2 and motion (FD) included as covariates of no interest, as described in the following equation.
| (3) |
2.5.3. Effects of clinical symptomatology
To investigate the association of clinical symptomatology with thalamocortical networks, we examined clinical symptomatology as a difference between diagnostic groups and also, as an exploratory analysis, in linear regressions including PRIME scores as a dimensional measure of psychopathology.
In ROI-to-ROI analyses, effects of clinical symptomatology were investigated after segregating individuals into diagnostic groups (typically developing, psychosis spectrum, other psychopathologies) using an omnibus ANCOVA with each thalamo-cortical network included as repeated measures, and diagnostic group as the independent variable, including age, age2, sex and motion (FD) as covariates of no interest, followed by a one-way univariate ANCOVAs for each thalamocortical network with diagnostic group as the independent variable and age, age2, sex and motion (FD) as covariates of no interest.
ANCOVAs were conducted separately for whole-brain voxel-wise connectivity from each of the 7 thalamus seeds, with diagnostic group as an independent variable and age, age2, sex and motion (FD) included as covariates of no interest, as described in the following equation.
| (4) |
As a dimensional measure of psychopathology, the sum of PRIME scores was included as a predictor in linear regression models predicting thalamocortical connectivity. In ROI-to-ROI analyses, effects of PRIME score on thalamocortical networks were modeled as linear effects with PRIME score as a predictor as shown in the regression model below.
| (5) |
For all models investigating the effects of PRIME score, age, age2, sex and motion (FD) were included as covariates of no interest.
2.5.4. Interaction effects
Interaction effects between diagnostic group and sex were investigated using a two-way ANCOVA with group and sex as independent variables and age, age2 and motion (FD) as covariates of no interest. Interaction effects between group and age, and sex and age were investigated using linear regression models, motion was also included as a covariate of no interest. Equations for all the interaction effects are presented in the Supplemental Materials.
3. Results
3.1. Thalamocortical functional connectivity: ROI-to-ROI analyses
3.1.1. Age effects
As shown in Fig. 1 A, somatosensory cortex–ventral posterolateral nucleus and occipital cortex–lateral geniculate nucleus connectivity showed significant negative linear associations with age (rpartial = −0.163, p < .001 and rpartial = −0.174, p < .001 respectively). Older youth showed less connectivity within these networks.
Fig. 1.
Age (A), Sex (B) and Group (C) effects in predefined thalamocortical networks across the whole sample. The seven networks tested were prefrontal cortex–mediodorsal nucleus (PFC–MD), motor cortex–ventrolateral nuclear group (MTR–VL), somatosensory cortex–ventral posterolateral nucleus (SS–VPL), temporal cortex–medial geniculate nucleus (TMP–MGN), hippocampus–anterior nuclear group (HIPP–AN), parietal cortex–pulvinar (PAR–PU), occipital cortex–lateral geniculate nucleus (OCC–LGN). † indicates p < .05, uncorrected.
Temporal cortex–medial geniculate nucleus connectivity showed a significant quadratic association with age (rpartial = 0.097, p = .001). Higher connectivity was observed in late childhood/early adolescence and adulthood, while late adolescence showed lower functional connectivity in this network. No other networks showed significant linear or quadratic effects of age on functional connectivity. See Supplementary Materials for results based on functionally defined thalamic ROIs.
3.1.2. Sex effects
An omnibus ANCOVA including all seven networks found no significant interaction between network and sex (F(6,6570) = 1.964, p = .067), main effect of sex (F(1,1095) = 0.114, p = .736) or network (F(6, 6570) = 1.199, p = .304). As seen in Fig. 1B, connectivity between the parietal cortex and pulvinar showed an effect of sex (F(1,1095) = 4.939, p = .026), though this effect did not survive Bonferroni correction (ɑ = 0.007). No other network showed an effect of sex. See Supplementary Materials for results based on functionally defined thalamic ROIs.
3.1.3. Effects of clinical symptomatology
To examine psychosis symptomatology by diagnostic group, an omnibus ANCOVA including all seven networks revealed a significant interaction between network and diagnostic group (F(12,6558) = 2.216, p = .009), but no significant main effect of diagnostic group (F(2,1093) = 1.418, p = .243) or network (F(6,6558) = 1.274, p = .266). Post-hoc ANCOVAs were conducted for each network including age, age2, motion and sex as covariates of no interest. As seen in Fig. 1C, connectivity between the motor cortex and ventrolateral nucleus showed an effect of diagnostic group (F(2,1093) = 3.131, p = .044), though this did not survive Bonferroni correction (ɑ = 0.007). Typically developing youth showed greater connectivity than both the psychosis spectrum youth (p = .021) and youth with other psychopathologies (p = .036). No other network showed any effect of diagnostic group.
As there was a significant difference in age and sex between the three diagnostic groups, we conducted a sensitivity analysis by creating sub-samples of the psychosis spectrum and typically developing youth matched for age, sex and race. Matching was conducted in the MatchIt package (Version 3.0.2) (Ho et al., 2011) in R (Version 3.6.3; R Core Team, 2020) and resulted in a final sample of 83 psychosis spectrum and 99 typically developing youth. See the Supplemental Table 1 for demographics of the matched samples. The sensitivity analysis did not show any significant main effects of diagnostic group (F(1176) = 0.691, p = .407), network (F(6,1056) = 1.054, p = .489) or interaction between network and diagnostic group (F(6,1056) = 1.054, p = .489). None of the 7 predefined thalamocortical networks showed any significant effect of diagnostic group. See Supplemental Material for additional details and results based on functionally defined thalamic ROIs.
In an exploratory analysis of the dimensional effect of psychopathology, we examined the association between connectivity within thalamocortical networks and PRIME scores in a series of linear regressions. As seen in Supplemental Fig. 5, the prefrontal–mediodorsal nucleus network showed a significant negative association with PRIME scores, such that greater PRIME scores were associated with lower connectivity within this network.
3.1.4. Interaction effects of age, sex and diagnostic group
As seen in Supplementary Fig. 4, there was a significant interaction between linear age and sex in the somatosensory–ventral posterolateral nucleus network (rpartial = −0.083, p = .007), showing that males had a more pronounced negative association with age compared to females. No other network showed any significant interaction between linear age, sex and diagnostic group.
3.2. Thalamocortical functional connectivity: voxel-wise analyses
3.2.1. Age effects
As shown in Figs. 2 and 3, linear age effects were characterized by negative associations with sensory/motor cortical regions and anterior insula, together with positive associations with heteromodal prefrontal and parietal regions. These effects were more pronounced with sensory/motor thalamic nuclei.
Fig. 2.
Voxelwise age and sex effects in thalamic association nuclear groups. Freesurfer segmented mediodorsal, pulvinar and anterior nuclear groups were used in a seed-based voxelwise functional connectivity analyses. Linear age, quadratic age and sex effects were masked to only include cortical voxels and thresholded at a cluster level p = .05 FWE corrected, and voxelwise p = .001 uncorrected.
Fig. 3.
Voxelwise age and sex effects in thalamic sensory/motor nuclear groups. Freesurfer segmented ventral lateral, ventral posterolateral, medial geniculate and lateral geniculate nuclear groups were used in seed-based voxelwise functional connectivity analyses. Linear age, quadratic age and sex effects were masked to only include cortical voxels and thresholded at a cluster level p = .05 FWE corrected, and voxelwise p = .001 uncorrected.
As shown in Figs. 2 and 3, quadratic age effects were observed between multiple thalamic nuclei and primarily dorsal attention network regions (including premotor cortex and inferior and superior parietal lobules) characterized by a U-shaped association such that connectivity was higher in early in adolescence and early adulthood, and lower in middle to late adolescence. See Supplemental Figs. 7 and 8 for examples, and see Supplementary Materials for results based on functionally defined thalamic ROIs.
3.2.2. Sex effects
As seen in Figs. 2 and 3, the mediodorsal, pulvinar, ventrolateral and ventral posterolateral nuclei all showed greater connectivity with occipital and parietal visual regions in females compared to males. The pulvinar, ventrolateral and ventral posterolateral nuclei showed greater connectivity with the inferior frontal and orbitofrontal cortices in males compared to females. See Supplementary Materials for results based on functionally defined thalamic ROIs.
3.2.3. Effects of clinical symptomatology
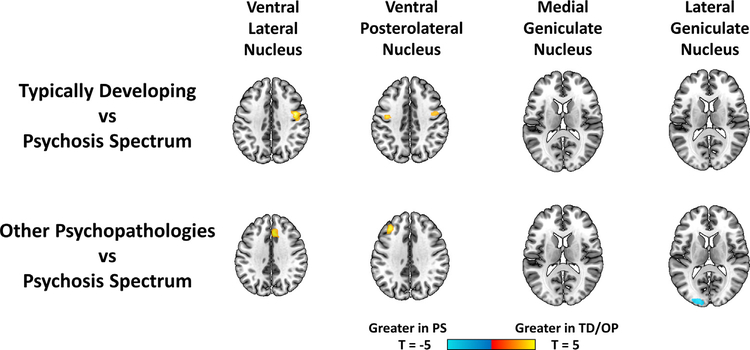
As seen in Fig. 5, psychosis spectrum youth showed lower connectivity compared with typically developing youth in connectivity between somatomotor thalamic nuclei (ventral lateral and ventral posterolateral nuclear groups) and the premotor cortex (extending into the inferior frontal cortex in a sensitivity analysis matched for age, sex and race, as seen in Supplementary Fig. 12). Psychosis spectrum youth showed lower connectivity compared to youth with other psychopathologies in medial prefrontal cortex (with ventral lateral nuclear group), lateral prefrontal cortex (with ventral posterolateral nuclei), and as seen in Fig. 4, inferior temporal cortex (with the pulvinar). Youth with other psychopathologies showed greater connectivity with visual cortex (with the lateral geniculate nucleus) compared to psychosis spectrum youth. See Supplementary Materials for results based on functionally defined thalamic ROIs.
Fig. 5.
Effects of diagnostic groups in thalamic sensory/motor nuclear groups. FreeSurfer segmented ventral lateral, ventral posterolateral, medial geniculate and lateral geniculate nuclear groups were used in seed-based voxel-wise functional connectivity analyses. Connectivity in typically developing youth (TD) and youth with other psychopathologies (OP) were compared with psychosis spectrum youth (PS). Maps were restricted to the cortex and thresholded at a cluster level p = .05 FWE corrected, and voxelwise p = .001 uncorrected.
Fig. 4.
Effects of diagnostic groups in thalamic association nuclear groups. FreeSurfer segmented mediodorsal, pulvinar and anterior nuclear groups were used in seed-based voxelwise functional connectivity analyses. Connectivity in typically developing youth (TD) and youth with other psychopathologies (OP) were compared with psychosis spectrum youth (PS). Maps were restricted to the cortex and thresholded at a cluster level p = .05 FWE corrected, and voxelwise p = .001 uncorrected.
4. Discussion
Thalamocortical dysconnectivity has been implicated in multiple neurodevelopmental disorders, especially schizophrenia. However, development of these networks in typically developed youth and youth atrisk for psychosis remains largely unknown. To address this knowledge gap, we characterized the effects of age, sex and clinical symptomatology on functional connectivity in anatomically constrained thalamocortical circuits in a large sample of youth from the PNC data. Broadly, we found significant age effects in thalamic connectivity along with modest sex differences. Subtle associations with psychopathology were also observed, although these results did not always survive correction for multiple comparisons and sensitivity analyses.
4.1. Thalamocortical network development from childhood to early adulthood
Both ROI-to-ROI and voxel-wise analyses found that thalamic connectivity with sensory cortical areas decreased with age. In the ROI-to-ROI analyses, the ventral posterolateral nuclei and lateral geniculate nuclei demonstrated age-related decreases in connectivity with their primary cortical targets, somatosensory and occipital cortex respectively. Voxel-wise analyses recapitulated these results. These findings are largely consistent with prior resting-state studies by Fair et al. (2010) and Steiner et al. (2020), which, despite using different methods, found lower functional connectivity in sensory thalamocortical networks from late childhood to young adulthood. Interestingly, the human thalamus is dominated by functional connectivity with sensory/motor networks in neonates, though this connectivity decreases rapidly in the first year after birth. Instead, over the first two years after birth, thalamocortical connectivity becomes dominated by the salience network, which includes the anterior insula and dorsal anterior cingulate cortex (Alcauter et al., 2014). Our results show that thalamocortical functional connectivity with both sensory/motor and salience network regions, primarily the anterior insula, also decreases in late childhood and adolescence into early adulthood. This developmental pattern differs from their cortico-cortical counterparts, which show their adult pattern in the first two years of life and appear to be largely mature by late childhood/early adolescence (Gao et al., 2015a, 2015b; Lin et al., 2008; Marek et al., 2015).
In contrast to sensory nuclei, which demonstrated prominent decline in connectivity with their primary sensory cortical targets with age, connectivity between the mediodorsal, pulvinar and ventral lateral nuclear groups with their primary cortical targets were relatively stable. Our findings help clarify prior developmental studies that have produced mixed findings. An early study found that adults showed greater connectivity between the thalamus and prefrontal cortex compared with children (Fair et al., 2010). A recent study investigating thalamus connectivity with multiple cortical networks found no effects of age in executive networks, which are composed primarily of prefrontal and some parietal association regions (Steiner et al., 2020). These differences may be explained by differences in analysis methods. Both our study and the recent study by Steiner and colleagues included larger sample sizes and modelled age as a continuous variable, rather than a categorical variable. The frontal and parietal thalamocortical networks do not have prominent connectivity in the first two years of life (Alcauter et al., 2014), yet our results show that they are prominent and relatively stable in late childhood/early adolescence. This pattern is different from that observed in studies of cortico-cortical networks, which show that frontoparietal networks start developing in the first two years of life and continue to develop through adolescence into early adulthood (Gao et al., 2015a, 2015b; Lin et al., 2008; Marek et al., 2015).
Our voxel-wise analyses revealed decreased within-network and increased cross-network connectivity with age. In addition to lower connectivity with sensory/motor cortices, there was a positive age effect on connectivity between multiple thalamic nuclei, though predominantly sensory and motor nuclei, and regions belonging to the cingulo-opercular and default mode networks. The pattern of lower connectivity with sensory/motor cortices and greater connectivity with association cortices was reminiscent of a sensorimotor–association gradient that has been described in cortical connectivity (Margulies et al., 2016). In particular, development of cortical networks shift from a gradient dominated by the distinction between somatomotor and visual cortices, to a gradient dominated by the distinction between sensorimotor and association cortices in adolescence (Dong et al., 2021). These thalamocortical functional connectivity changes may reflect maturation of thalamocortical white matter microstructure. In this sample, diffusion weighted imaging found greater fractional anisotropy with age, indicating increased myelination, particularly in white matter tracts between the thalamus and association cortices (Avery et al., 2021 Submitted for publication). This shift may also reflect increased integration of sensory/motor thalamus with association cortical networks. In young adults, the thalamus functions as an integrative hub for cortical brain networks, with both sensory/motor and association thalami showing strong functional connectivity with multiple cortical networks, though the cingulo-opercular and default mode networks showed the most connectivity across thalamic nuclei (Hwang et al., 2017). In youth, thalamocortical functional connectivity with salience and default mode networks are positively associated with top down control functions such as selective attention and cognitive flexibility (Steiner et al., 2020). Together with these previous studies, our results suggest that connectivity between the thalamus and cingulo-opercular networks mature during adolescence, possibly reflecting maturation of top-down control processes (Marek et al., 2015).
Our voxel-wise analyses also showed a U-shaped age effect between multiple thalamic nuclear groups with dorsal attention network regions, though this effect was substantially reduced in a sample with stricter motion control. In a recent study examining thalamic connectivity with large cortical networks, the dorsal attention network was the only cortical network that showed a positive effect of age (Steiner et al., 2020), though they did not test whether there were any quadratic effects of age. The dorsal attention network is involved in cognitive functions that are disrupted in neurodevelopmental disorders such as schizophrenia, including the control of attention and working memory (Greene and Soto, 2014; Majerus et al., 2018; Ptak, 2012; Ptak et al., 2017; Spreng et al., 2010). The quadratic effect of age may indicate that there is reorganization of the dorsal attention network during adolescence, making it particularly sensitive to disruption.
4.2. Sex effects in thalamocortical functional networks during development
The ROI-to-ROI analysis showed a trend towards females having higher connectivity in the parietal–pulvinar network. This finding was confirmed in the voxel-wise analyses, which found a consistent pattern across multiple nuclei where females showed greater connectivity with visual regions in both the occipital and parietal cortices. Sex effects have been reported in various visual processes including contrast sensitivity, visual acuity, color vision and visual attention (Gur et al., 2000; Vanston and Strother, 2017; Weiss et al., 2003), though the direction of the effect is not always consistent. In contrast to the greater thalamocortical connectivity we observed, males tend to have larger visual cortices (Amunts et al., 2007; Brun et al., 2009). Previous imaging studies have also observed that males tend to have higher BOLD activation within the visual cortex to visual stimuli (Cowan et al., 2000; Levin et al., 1998), while females showed shorter latencies and larger amplitudes in visual evoked potentials (Dion et al., 2013). Due in part to the inconsistency surrounding sex differences in visual processing, the functional consequences of the thalamocortical functional connectivity differences with visual cortex are unknown and deserve further study.
The voxel-wise analyses further found greater connectivity between the pulvinar, ventral lateral and ventral posterolateral nuclear groups and the orbitofrontal and inferior frontal cortex in males compared with females, which was not tested for in the ROI-to-ROI analyses. The orbitofrontal cortex is implicated in affective decision-making processes, such as reward-motivated decisions (Padoa-Schioppa and Conen, 2017; Rolls, 2004). Males have a small, but consistent advantage on the orbitofrontal dependent Iowa Gambling Task, which measures people’s ability to make decisions that favor smaller rewards that lead to long-term gains over larger rewards that lead to long-term losses (van den Bos et al., 2013). Differences in this task have been attributed to the organizational effects gonadal hormones have on the orbitofrontal cortex during gestation (Overman, 2004). Whether males continue to show greater thalamus connectivity with the orbitofrontal cortex in adulthood or if this is a developmental phenomena deserves further study.
4.3. Thalamocortical functional connectivity in psychosis spectrum youth
As discussed earlier, thalamocortical functional dysconnectivity is a robust finding in schizophrenia and is often characterized by a combination of reduced prefrontal-thalamic connectivity and somatomotorthalamic hyper-connectivity (Giraldo-Chica and Woodward, 2017; Ramsay, 2019). Consistent with this, voxel-wise analyses revealed lower thalamic connectivity with regions of the frontal cortex in psychosis spectrum youth, though this effect was primarily with the somatomotor thalamic nuclei rather than with the mediodorsal nucleus, as has been previously reported in some clinical samples. In addition, our exploratory analysis showed PRIME scores were negatively associated with connectivity in the prefrontal–mediodorsal nucleus network, indicating a dimensional approach to psychopathology may be more sensitive to early dysconnectivity in thalamocortical networks in youth. A previous study in a clinical-high risk sample showed both greater sensory/motor connectivity and lower prefrontal connectivity; this dysconnectivity was greater in individuals that converted compared with those that did not (Anticevic et al., 2015). We find that maturation of thalamocortical functional connectivity is reflected in decreased connectivity with sensory/motor networks and increased connectivity with association networks. Our findings, together with findings from Anticevic and colleagues suggests that this process is arrested in psychosis, supporting a neurodevelopmental origin to thalamocortical dysconnectivity in psychotic disorders, though evidence in cortical functional networks have also shown signs of hypermaturation in psychosis spectrum youth (Truelove-Hill et al., 2020).
Our results differed from a previous study using the PNC sample that did not find thalamocortical connectivity differences in psychosis spectrum youth (Jacobs et al., 2019). Our methodology differed from their study in two major ways. First, we separated youth that show symptoms of other psychopathologies from typically developing youth, which is necessary as thalamus dysconnectivity has been observed in other neurodevelopmental disorders such as autism (Nair et al., 2013; Woodward et al., 2017) and attention deficit hyperactivity disorder (Clerkin et al., 2013; Mills et al., 2012). Second, we defined thalamocortical networks differently, examining both anatomically constrained networks and the cortical connectivity from anatomically defined thalamic nuclei. We found only subtle effects of psychosis symptoms in this sample, suggesting that the appropriate probe of thalamocortical networks is necessary to find effects.
4.4. Strengths and limitations
Strengths of our study include the relatively large, community-ascertained samples; inclusion of youth with other psychopathologies in addition to typically developing and psychosis spectrum groups; use of anatomically defined thalamic sub-regions as seeds for the functional connectivity analyses rather than using the whole thalamus as a seed, as is often done, and complementary ROI-to-ROI and voxel-wise analyses to comprehensively map thalamic connectivity. The use of cross-sectional data to examine age effects is the main limitation of our study. However, we are not aware of a comparable longitudinal dataset comprised of typically developing and psychosis spectrum youth, as well as youth with other psychopathologies. In the future, the on-going Adolescent Brain Cognitive Development (ABCD) study (Casey et al., 2018) may be ideal for examining typical and atypical development of thalamocortical circuitry. Another limitation of our study is that we conducted seven different seed-based analyses for each thalamus ROI. While this allows us to examine different thalamocortical networks, it does inflate the chance of false positives. Future studies that take a gradient approach to examining thalamocortical connectivity may shed greater light on thalamic contribution to the development of the sensorimotor–association axis.
5. Conclusions
By characterizing age-related thalamocortical connectivity, and sex-related differences in thalamocortical networks for the first time, this study provides a framework for neurodevelopmental theories of thalamocortical dysconnectivity in clinical populations. We showed that over development, thalamocortical functional networks appear to integrate less with sensory systems and more with higher-order cortical systems, such as the cingulo-opercular network. This reflects the shift from sensory guided responses to more top-down, cognitively guided processes with maturation. This process may be disrupted in neurodevelopmental disorders, presenting as the pattern of lower-prefrontal, higher-sensory thalamocortical dysconnectivity observed in clinical psychosis samples. We show that subtle signs of this pattern are already present in youth showing psychosis symptomatology who will never convert to a psychotic disorder.
Supplementary Material
Acknowledgments
This work was supported by National Institutes of Health grants R01 MH115000 (awarded to NDW and AA), R01 MH123563–01 (awarded to SV) and the Vanderbilt Institute for Clinical and Translational Research (through grant 1-UL-1-TR000445 from the National Center for Research Resources/NIH). This work was conducted in part using the resources of the Advanced Computing Center for Research and Education at Vanderbilt University, Nashville, TN.
Footnotes
Declaration of Competing Interest
No commercial support was received for the preparation of this manuscript. AA consults, holds equity and is a scientific board member for BlackThorn Therapeutics. All other authors have no conflicts of interest to report.
Data and code availability statement
Participants were part of the Philadelphia Neurodevelopmental Cohort (PNC) obtained from the database of Genotypes and Phenotypes (dbGaP:https://www.ncbi.nlm.nih.gov/gap/). We used the most recent release of the PNC (Study Accession phs000607.v3.p2), consisting of 9498 youth (aged 8–21) including the full neuroimaging sample (n = 1601). Neuroimage data storage and processing took place on the Vanderbilt University Institute of Imaging Science Center for Computational Imaging XNAT (Harrigan et al., 2016; Huo et al., 2018). The processing pipelines are accessible through github (https://github.com/baxpr/freesurfer-singularity; https://github.com/baxpr/connprep; https://github.com/baxpr/fmriqa; https://github.com/baxpr/fsthalconnMNI-public) and were containerized using Singularity and built at SingularityHub (Sochat et al., 2017) (https://singularity-hub.org).
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.neuroimage.2021.118562.
References
- Alcauter S, Lin W, Smith JK, Short SJ, Goldman BD, Reznick JS, Gilmore JH, Gao W, 2014. Development of thalamocortical connectivity during infancy and its cognitive correlations. J. Neurosci 34 (27), 9067–9075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amunts K, Armstrong E, Malikovic A, Hömke L, Mohlberg H, Schleicher A, Zilles K, 2007. Gender-specific left-right asymmetries in human visual cortex. J. Neurosci 27 (6), 1356–1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreasen NC, Paradiso S, O’Leary DS, 1998. “Cognitive dysmetria” as an integrative theory of schizophrenia: a dysfunction in cortical-subcortical-cerebellar circuitry? Schizophr. Bull 24 (2), 203–218. [DOI] [PubMed] [Google Scholar]
- Anticevic A, Cole MW, Repovs G, Murray JD, Brumbaugh MS, Winkler AM, Savic A, Krystal JH, Pearlson GD, Glahn DC, 2014. Characterizing thalamo-cortical disturbances in schizophrenia and bipolar illness. Cereb. Cortex 24 (12), 3116–3130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anticevic A, Haut K, Murray JD, Repovs G, Yang GJ, Diehl C, McEwen SC, Bearden CE, Addington J, Goodyear B, Cadenhead KS, Mirzakhanian H, Cornblatt BA, Olvet D, Mathalon DH, McGlashan TH, Perkins DO, Belger A, Seidman LJ, … Cannon TD, 2015. Association of thalamic dysconnectivity and conversion to psychosis in youth and young adults at elevated clinical risk. JAMA Psychiatry 72 (9), 882–891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avery S, Huang AS, Sheffield JM, Rogers BP, Vandekar S, Anticevic A, & Woodward ND (2021). Development of thalamocortical structural connectivity in typically developing and psychosis spectrum youth. Manuscript submitted for publication [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behrens TEJ, Johansen-Berg H, Woolrich MW, Smith SM, Wheeler-Kingshott CAM, Boulby PA, Barker GJ, Sillery EL, Sheehan K, Ciccarelli O, Thompson AJ, Brady JM, Matthews PM, 2003. Non-invasive mapping of connections between human thalamus and cortex using diffusion imaging. Nat. Neurosci 6 (7), 750–757. [DOI] [PubMed] [Google Scholar]
- Bolkan SS, Stujenske JM, Parnaudeau S, Spellman TJ, Rauffenbart C, Abbas AI, Harris AZ, Gordon JA, Kellendonk C, 2017. Thalamic projections sustain prefrontal activity during working memory maintenance. Nat. Neurosci 20 (7), 987–996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brun CC, Leporé N, Luders E, Chou YY, Madsen SK, Toga AW, Thompson PM, 2009. Sex differences in brain structure in auditory and cingulate regions. Neuroreport 20 (10), 930–935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calkins ME, Moore TM, Merikangas KR, Burstein M, Satterthwaite TD, Bilker WB, Ruparel K, Chiavacci R, Wolf DH, Mentch F, Qiu H, Connolly JJ, Sleiman PA, Hakonarson H, Gur RC, Gur RE, 2014. The psychosis spectrum in a young U.S. community sample: findings from the Philadelphia Neurodevelopmental Cohort. World Psychiatry: Official Journal of the World Psychiatric Association 13 (3), 296–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casey BJ, Cannonier T, Conley MI, Cohen AO, Barch DM, Heitzeg MM, Soules ME, Teslovich T, Dellarco DV, Garavan H, Orr CA, Wager TD, Banich MT, Speer NK, Sutherland MT, Riedel MC, Dick AS, Bjork JM, … Thomas KM ABCD Imaging Acquisition Workgroup, 2018. The Adolescent Brain Cognitive Development (ABCD) study: imaging acquisition across 21 sites. Dev. Cognit. Neurosci 32, 43–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clerkin SM, Schulz KP, Berwid OG, Fan J, Newcorn JH, Tang CY, Halperin JM, 2013. Thalamo-cortical activation and connectivity during response preparation in adults with persistent and remitted ADHD. Am. J. Psychiatry 170 (9), 1011–1019. [DOI] [PubMed] [Google Scholar]
- Colibazzi T, Yang Z, Horga G, Chao-Gan Y, Corcoran CM, Klahr K, Brucato G, Girgis R, Abi-Dargham A, Milham MP, Peterson BS, 2017. Aberrant temporal connectivity in persons at clinical high risk for psychosis. Biol. Psychiatry Cognit. Neurosci. Neuroimaging 2 (8), 696–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan RL, Frederick BB, Rainey M, Levin JM, Maas LC, Bang J, Hennen J, Lukas SE, Renshaw PF, 2000. Sex differences in response to red and blue light in human primary visual cortex: a bold fMRI study. Psychiatry Res. 100 (3), 129–138. [DOI] [PubMed] [Google Scholar]
- Cronenwett WJ, Csernansky J, 2010. Thalamic pathology in schizophrenia. In: Swerdlow NR (Ed.), Behavioral Neurobiology of Schizophrenia and Its Treatment. Springer; Berlin Heidelberg, pp. 509–528. [DOI] [PubMed] [Google Scholar]
- Dale AM, Fischl B, Sereno MI, 1999. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage 9 (2), 179–194. [DOI] [PubMed] [Google Scholar]
- Dean DJ, Walther S, Bernard JA, Mittal VA, 2018. Motor clusters reveal differences in risk for psychosis, cognitive functioning, and thalamocortical connectivity: evidence for vulnerability subtypes. Clin. Psychol. Sci 6 (5), 721–734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dion L−A, Muckle G, Bastien C, Jacobson SW, Jacobson JL, Saint-Amour D, 2013. Sex differences in visual evoked potentials in school-age children: what is the evidence beyond the checkerboard? Int. J. Psychophysiol. Off. J. Int. Organ. Psychophysiol 88 (2), 136–142. [DOI] [PubMed] [Google Scholar]
- Dong H−M, Margulies DS, Zuo XN, Holmes AJ, 2021. Shifting gradients of macroscale cortical organization mark the transition from childhood to adolescence. Proc. Natl. Acad. Sci. U.S.A 118 (28). doi: 10.1073/pnas.2024448118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fair DA, Bathula D, Mills KL, Dias TGC, Blythe MS, Zhang D, Snyder AZ, Raichle ME, Stevens AA, Nigg JT, Nagel BJ, 2010. Maturing thalamocortical functional connectivity across development. Front. Syst. Neurosci 4, 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, van der Kouwe A, Killiany R, Kennedy D, Klaveness S, Montillo A, Makris N, Rosen B, Dale AM, 2002. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33 (3), 341–355. [DOI] [PubMed] [Google Scholar]
- Gao W, Alcauter S, Elton A, Hernandez-Castillo CR, Smith JK, Ramirez J, Lin W, 2015a. Functional network development during the first year: relative sequence and socioeconomic correlations. Cereb. Cortex 25 (9), 2919–2928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao W, Alcauter S, Smith JK, Gilmore JH, Lin W, 2015b. Development of human brain cortical network architecture during infancy. Brain Struct. Funct 220 (2), 1173–1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giraldo-Chica M, Woodward ND, 2017. Review of thalamocortical resting-state fMRI studies in schizophrenia. Schizophr. Res 180, 58–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greene CM, Soto D, 2014. Functional connectivity between ventral and dorsal frontoparietal networks underlies stimulus-driven and working memory-driven sources of visual distraction. Neuroimage 84, 290–298. [DOI] [PubMed] [Google Scholar]
- Gur RC, Alsop D, Glahn D, Petty R, Swanson CL, Maldjian JA, Turetsky BI, Detre JA, Gee J, Gur RE, 2000. An fMRI study of sex differences in regional activation to a verbal and a spatial task. Brain Lang. 74 (2), 157–170. [DOI] [PubMed] [Google Scholar]
- Harrigan RL, Yvernault BC, Boyd BD, Damon SM, Gibney KD, Conrad BN, Phillips NS, Rogers BP, Gao Y, Landman BA, 2016. Vanderbilt university institute of imaging science center for computational imaging XNAT: a multimodal data archive and processing environment. Neuroimage 124 (Pt B), 1097–1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho DE, Imai K, King G, Stuart EA, 2011. MatchIt: nonparametric preprocessing for parametric causal inference. J. Stat. Softw Http://gking. Harvard. Edu/matchit https://r.iq.harvard.edu/docs/matchit/2.4-18/matchit.pdf. [Google Scholar]
- Huang AS, Rogers BP, Sheffield JM, Jalbrzikowski ME, Anticevic A, Blackford JU, Heckers S, Woodward ND, 2020. Thalamic nuclei volumes in psychotic disorders and in youths with psychosis spectrum symptoms. Am. J. Psychiatry appiajp202019101099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang AS, Rogers BP, Woodward ND, 2019. Disrupted modulation of thalamus activation and thalamocortical connectivity during dual task performance in schizophrenia. Schizophr. Res 210, 270–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huo Y, Blaber J, Damon SM, Boyd BD, Bao S, Parvathaneni P, Noguera CB, Chaganti S, Nath V, Greer JM, Lyu I, French WR, Newton AT, Rogers BP, Landman BA, 2018. Towards portable large-scale image processing with high-performance computing. J. Digit. Imaging 31 (3), 304–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang K, Bertolero MA, Liu WB, D’Esposito M, 2017. The human thalamus is an integrative hub for functional brain networks. J. Neurosci 37 (23), 5594–5607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iglesias JE, Insausti R, Lerma-Usabiaga G, Bocchetta M, Van Leemput K, Greve DN, van der Kouwe A, Fischl B, Caballero-Gaudes C, Paz-Alonso PM Alzheimer’s Disease Neuroimaging Initiative, 2018. A probabilistic atlas of the human thalamic nuclei combining ex vivo MRI and histology. Neuroimage 183, 314–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs GR, Ameis SH, Ji JL, Viviano JD, Dickie EW, Wheeler AL, Stojanovski S, Anticevic A, Voineskos AN, 2019. Developmentally divergent sexual dimorphism in the cortico-striatal-thalamic-cortical psychosis risk pathway. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol 44 (9), 1649–1658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansen-Berg H, Behrens TEJ, Sillery E, Ciccarelli O, Thompson AJ, Smith SM, Matthews PM, 2005. Functional-anatomical validation and individual variation of diffusion tractography-based segmentation of the human thalamus. Cereb. Cortex 15 (1), 31–39. [DOI] [PubMed] [Google Scholar]
- Jones EG, 1997. Cortical development and thalamic pathology in schizophrenia. Schizophr. Bull 23 (3), 483–501. [DOI] [PubMed] [Google Scholar]
- Jones EG, 2007. The Thalamus. Cambridge University Press. [Google Scholar]
- Kraguljac NV, Reid M, White D, Jones R, den Hollander J, Lowman D, Lahti AC, 2012. Neurometabolites in schizophrenia and bipolar disorder - a systematic review and meta-analysis. Psychiatry Res. 203 (2–3), 111–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin JM, Ross MH, Mendelson JH, Mello NK, Cohen BM, Renshaw PF, 1998. Sex differences in blood-oxygenation-level-dependent functional MRI with primary visual stimulation. Am. J. Psychiatry 155 (3), 434–436. [DOI] [PubMed] [Google Scholar]
- Lin W, Zhu Q, Gao W, Chen Y, Toh C−H, Styner M, Gerig G, Smith JK, Biswal B, Gilmore JH, 2008. Functional connectivity MR imaging reveals cortical functional connectivity in the developing brain. AJNR Am. J. Neuroradiol 29 (10), 1883–1889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majerus S, Péters F, Bouffier M, Cowan N, Phillips C, 2018. The dorsal attention network reflects both encoding load and top-down control during working memory. J. Cognit. Neurosci 30 (2), 144–159. [DOI] [PubMed] [Google Scholar]
- Marek S, Hwang K, Foran W, Hallquist MN, Luna B, 2015. The contribution of network organization and integration to the development of cognitive control. PLoS Biol. 13 (12), e1002328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marenco S, Weinberger DR, 2000. The neurodevelopmental hypothesis of schizophrenia: following a trail of evidence from cradle to grave. Dev. Psychopathol 12 (3), 501–527. [DOI] [PubMed] [Google Scholar]
- Margulies DS, Ghosh SS, Goulas A, Falkiewicz M, Huntenburg JM, Langs G, Bezgin G, Eickhoff SB, Castellanos FX, Petrides M, Jefferies E, Smallwood J, 2016. Situating the default-mode network along a principal gradient of macroscale cortical organization. Proc. Natl. Acad. Sci. U.S.A 113 (44), 12574–12579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills KL, Bathula D, Dias TGC, Iyer SP, Fenesy MC, Musser ED, Stevens CA, Thurlow BL, Carpenter SD, Nagel BJ, Nigg JT, Fair DA, 2012. Altered cortico-striatal-thalamic connectivity in relation to spatial working memory capacity in children with ADHD. Front. Psychiatry Front. Res. Found 3, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minzenberg MJ, Laird AR, Thelen S, Carter CS, Glahn DC, 2009. Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch. Gen. Psychiatry 66 (8), 811–822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair A, Treiber JM, Shukla DK, Shih P, Müller RA, 2013. Impaired thalamocortical connectivity in autism spectrum disorder: a study of functional and anatomical connectivity. Brain J. Neurol 136 (Pt 6), 1942–1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Overman WH, 2004. Sex differences in early childhood, adolescence, and adulthood on cognitive tasks that rely on orbital prefrontal cortex. Brain Cognit. 55 (1), 134–147. [DOI] [PubMed] [Google Scholar]
- Padoa-Schioppa C, Conen KE, 2017. Orbitofrontal cortex: a neural circuit for economic decisions. Neuron 96 (4), 736–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parnaudeau S, O’Neill P−K, Bolkan SS, Ward RD, Abbas AI, Roth BL, Balsam PD, Gordon JA, Kellendonk C, 2013. Inhibition of mediodorsal thalamus disrupts thalamofrontal connectivity and cognition. Neuron 77 (6), 1151–1162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parnaudeau S, Taylor K, Bolkan SS, Ward RD, Balsam PD, Kellendonk C, 2015. Mediodorsal thalamus hypofunction impairs flexible goal-directed behavior. Biol. Psychiatry 77 (5), 445–453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pergola G, Selvaggi P, Trizio S, Bertolino A, Blasi G, 2015. The role of the thalamus in schizophrenia from a neuroimaging perspective. Neurosci. Biobehav. Rev 54, 57–75. [DOI] [PubMed] [Google Scholar]
- Ptak R, 2012. The frontoparietal attention network of the human brain: action, saliency, and a priority map of the environment. Neurosci. Rev. J. Bring. Neurobiol. Neurol. Psychiatry 18 (5), 502–515. [DOI] [PubMed] [Google Scholar]
- Ptak R, Schnider A, Fellrath J, 2017. The dorsal frontoparietal network: a core system for emulated action. Trends Cognit. Sci. (Regul. Ed.) 21 (8), 589–599. [DOI] [PubMed] [Google Scholar]
- Ragland JD, Laird AR, Ranganath C, Blumenfeld RS, Gonzales SM, Glahn DC, 2009. Prefrontal activation deficits during episodic memory in schizophrenia. Am. J. Psychiatry 166 (8), 863–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramsay IS, 2019. An activation likelihood estimate meta-analysis of thalamocortical dysconnectivity in psychosis. Biol. Psychiatry Cognit. Neurosci. Neuroimaging 4 (10), 859–869. [DOI] [PubMed] [Google Scholar]
- Rikhye RV, Gilra A, Halassa MM, 2018. Thalamic regulation of switching between cortical representations enables cognitive flexibility. Nat. Neurosci 21 (12), 1753–1763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roalf DR, Quarmley M, Calkins ME, Satterthwaite TD, Ruparel K, Elliott MA, Moore TM, Gur RC, Gur RE, Moberg PJ, Turetsky BI, 2017. Temporal lobe volume decrements in psychosis spectrum youths. Schizophr. Bull 43 (3), 601–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rolls ET, 2004. The functions of the orbitofrontal cortex. Brain Cognit. 55 (1), 11–29. [DOI] [PubMed] [Google Scholar]
- Saalmann YB, Pinsk MA, Wang L, Li X, Kastner S, 2012. The pulvinar regulates information transmission between cortical areas based on attention demands. Science 337 (6095), 753–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmitt LI, Wimmer RD, Nakajima M, Happ M, Mofakham S, Halassa MM, 2017. Thalamic amplification of cortical connectivity sustains attentional control. Nature 545 (7653), 219–223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seidman LJ, Shapiro DI, Stone WS, Woodberry KA, Ronzio A, Cornblatt BA, Addington J, Bearden CE, Cadenhead KS, Cannon TD, Mathalon DH, McGlashan TH, Perkins DO, Tsuang MT, Walker EF, Woods SW, 2016. Association of neurocognition with transition to psychosis: baseline functioning in the second phase of the North American prodrome longitudinal study. JAMA Psychiatry 73 (12), 1239–1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sim K, Cullen T, Ongur D, Heckers S, 2006. Testing models of thalamic dysfunction in schizophrenia using neuroimaging. J. Neural Transm 113 (7), 907–928. [DOI] [PubMed] [Google Scholar]
- Sochat VV, Prybol CJ, Kurtzer GM, 2017. Enhancing reproducibility in scientific computing: metrics and registry for Singularity containers. PLoS One 12 (11), e0188511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spreng RN, Stevens WD, Chamberlain JP, Gilmore AW, Schacter DL, 2010. Default network activity, coupled with the frontoparietal control network, supports goal-directed cognition. Neuroimage 53 (1), 303–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steiner L, Federspiel A, Slavova N, Wiest R, Grunt S, Steinlin M, Everts R, 2020. Functional topography of the thalamo-cortical system during development and its relation to cognition. Neuroimage 223, 117361. [DOI] [PubMed] [Google Scholar]
- Steullet P, 2019. Thalamus-related anomalies as candidate mechanism-based biomarkers for psychosis. Schizophr. Res doi: 10.1016/j.schres.2019.05.027. [DOI] [PubMed] [Google Scholar]
- Swerdlow NR, 2010. Integrative circuit models and their implications for the pathophysiologies and treatments of the schizophrenias. In: Swerdlow NR (Ed.), Behavioral Neurobiology of Schizophrenia and Its Treatment. Springer; Berlin Heidelberg, pp. 555–583. [DOI] [PubMed] [Google Scholar]
- Truelove-Hill M, Erus G, Bashyam V, Varol E, Sako C, Gur RC, Gur RE, Koutsouleris N, Zhuo C, Fan Y, Wolf DH, Satterthwaite TD, Davatzikos C, 2020. A multidimensional neural maturation index reveals reproducible developmental patterns in children and adolescents. J. Neurosci 40 (6), 1265–1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Bos R, Homberg J, de Visser L, 2013. A critical review of sex differences in decision-making tasks: focus on the Iowa gambling task. Behav. Brain Res 238, 95–108. [DOI] [PubMed] [Google Scholar]
- van Erp TGM, Hibar DP, Rasmussen JM, Glahn DC, Pearlson GD, Andreassen OA, Agartz I, Westlye LT, Haukvik UK, Dale AM, Melle I, Hartberg CB, Gruber O, Kraemer B, Zilles D, Donohoe G, Kelly S, McDonald C, Morris DW, … Turner JA, 2016. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol. Psychiatry 21 (4), 547–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanston JE, Strother L, 2017. Sex differences in the human visual system. J. Neurosci. Res 95 (1–2), 617–625. [DOI] [PubMed] [Google Scholar]
- Weiss EM, Kemmler G, Deisenhammer EA, Fleischhacker WW, Delazer M, 2003. Sex differences in cognitive functions. Pers. Individ. Differ 35 (4), 863–875. [Google Scholar]
- Wimmer RD, Schmitt LI, Davidson TJ, Nakajima M, Deisseroth K, Halassa MM, 2015. Thalamic control of sensory selection in divided attention. Nature 526 (7575), 705–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward ND, Giraldo-Chica M, Rogers B, Cascio CJ, 2017. Thalamocortical dysconnectivity in autism spectrum disorder: an analysis of the autism brain imaging data exchange. Biol. Psychiatry Cognit. Neurosci. Neuroimaging 2 (1), 76–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward ND, Heckers S, 2016. Mapping thalamocortical functional connectivity in chronic and early stages of psychotic disorders. Biol. Psychiatry 79 (12), 1016–1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodward ND, Karbasforoushan H, Heckers S, 2012. Thalamocortical dysconnectivity in schizophrenia. Am. J. Psychiatry 169 (10), 1092–1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S, Li C-SR, 2010. A neural measure of behavioral engagement: task-residual low-frequency blood oxygenation level-dependent activity in the precuneus. Neuroimage 49 (2), 1911–1918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H, Schafer RJ, Desimone R, 2016. Pulvinar-cortex interactions in vision and attention. Neuron 89 (1), 209–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu F, Liu Y, Liu F, Yang R, Li H, Chen J, Kennedy DN, Zhao J, Guo W, 2019. Functional asymmetry of thalamocortical networks in subjects at ultra-high risk for psychosis and first-episode schizophrenia. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol 29 (4), 519–528. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.