Abstract
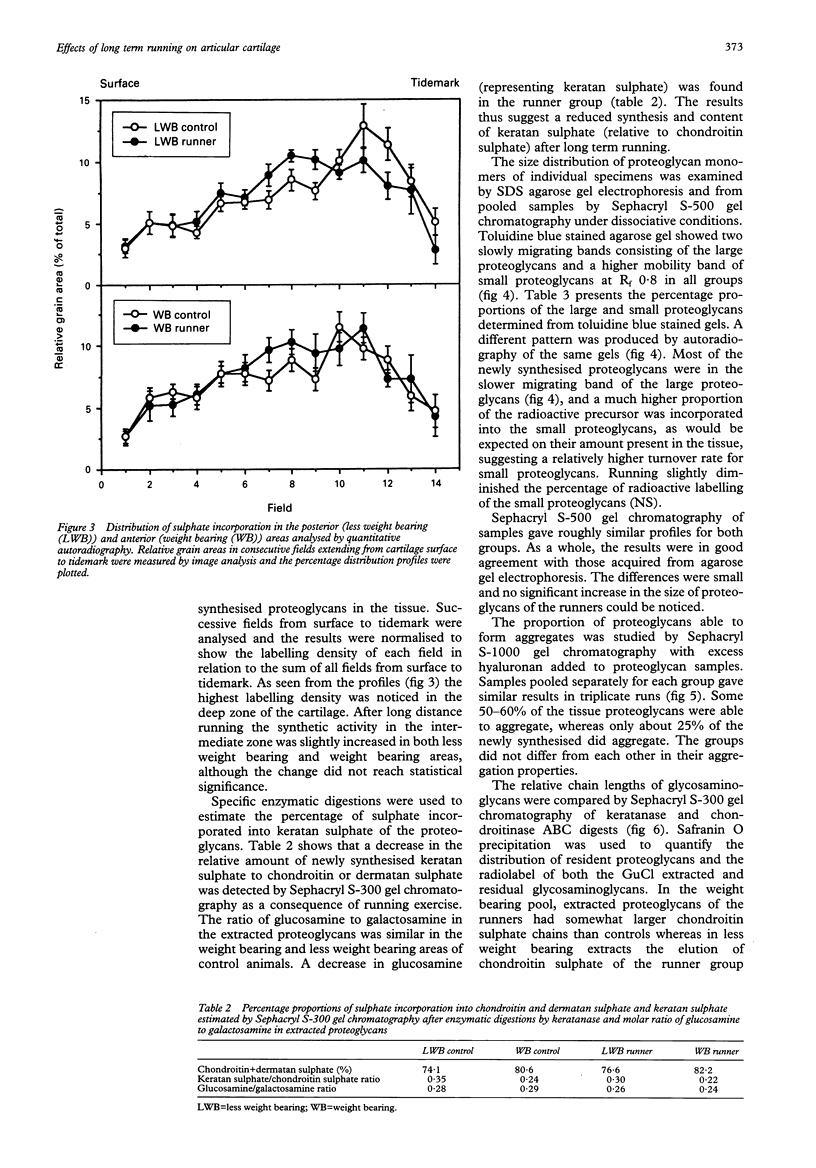
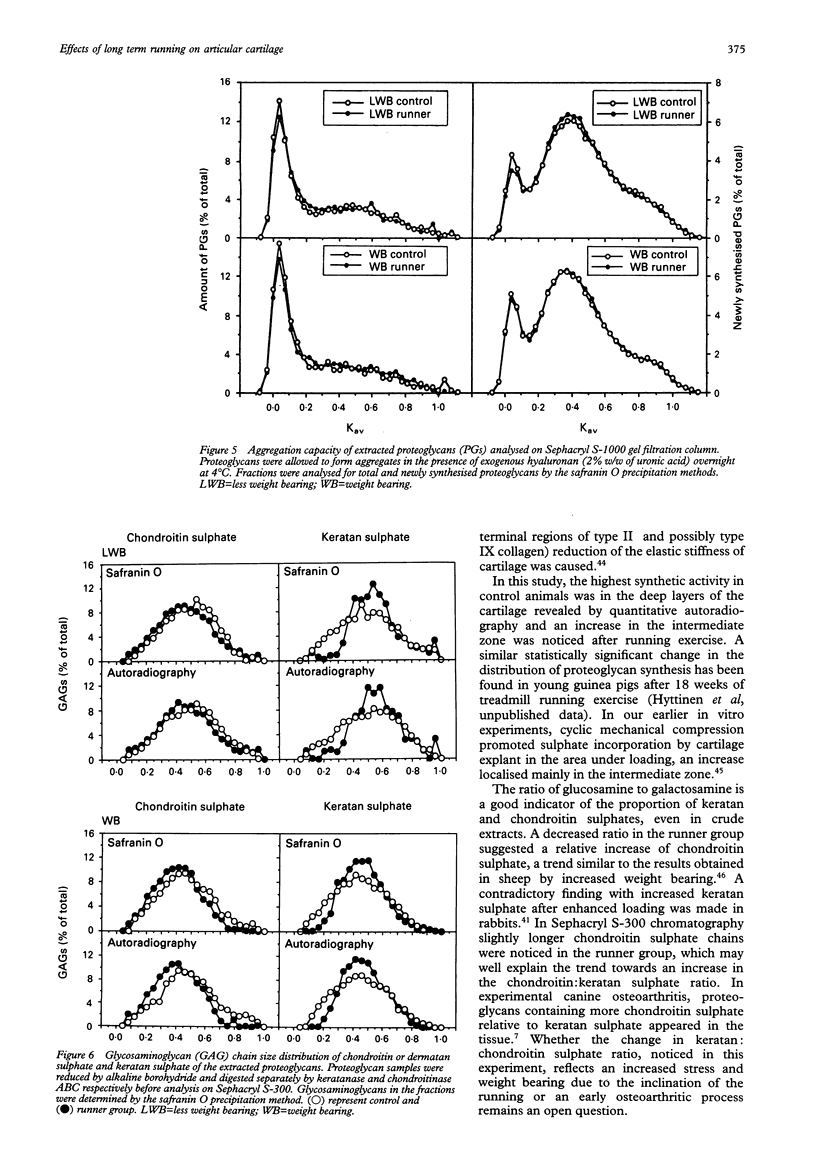
OBJECTIVE--To study the effects of long term (one year), long distance (up to 40 km/day) running on the metabolism of articular cartilage the biosynthesis of proteoglycans was examined by in vitro labelling of anterior (weight bearing) and posterior (less weight bearing) areas of the femoral head from young beagles. METHODS--Total sulphate incorporation rates were determined and distribution of the incorporated sulphate was localised by quantitative autoradiography. Concentration and extractability of the proteoglycans were determined, and proteoglycan structures were investigated by gel filtration chromatography, agarose gel electrophoresis, and chemical determinations. RESULTS--In the less weight bearing area the amount of extractable proteoglycans was decreased (p < or = 0.02), simultaneously with an increased concentration of residual glycosaminoglycans in the tissue after 4 M GuCl extraction (p < or = 0.05). In control animals proteoglycan synthesis was most active in the deep zone of the cartilage, whereas exercise increased synthesis in the intermediate zone. There was a tendency to a lower keratan: chondroitin sulphate ratio in the running dogs. No macroscopical or microscopical signs of articular degeneration or injury were visible in any of the animals. CONCLUSION--The articular cartilage of the femoral head showed a great capacity to adapt to the increased mechanical loading. The reduced proteoglycan extractability in the less weight bearing area changed it similar to the weight bearing area, suggesting that the low extractability of proteoglycans reflects the long term loading history of articular cartilage. The congruency of the femoral head with acetabulum seems to protect the cartilage from the untoward alterations that occur in the femoral condyles subjected to a similar running programme.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Akizuki S., Mow V. C., Müller F., Pita J. C., Howell D. S., Manicourt D. H. Tensile properties of human knee joint cartilage: I. Influence of ionic conditions, weight bearing, and fibrillation on the tensile modulus. J Orthop Res. 1986;4(4):379–392. doi: 10.1002/jor.1100040401. [DOI] [PubMed] [Google Scholar]
- Bader D. L., Kempson G. E., Egan J., Gilbey W., Barrett A. J. The effects of selective matrix degradation on the short-term compressive properties of adult human articular cartilage. Biochim Biophys Acta. 1992 Apr 22;1116(2):147–154. doi: 10.1016/0304-4165(92)90111-7. [DOI] [PubMed] [Google Scholar]
- Bayliss M. T., Venn M., Maroudas A., Ali S. Y. Structure of proteoglycans from different layers of human articular cartilage. Biochem J. 1983 Feb 1;209(2):387–400. doi: 10.1042/bj2090387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergmann G., Siraky J., Rohlmann A., Koelbel R. A comparison of hip joint forces in sheep, dog and man. J Biomech. 1984;17(12):907–921. doi: 10.1016/0021-9290(84)90004-6. [DOI] [PubMed] [Google Scholar]
- Blumenkrantz N., Asboe-Hansen G. New method for quantitative determination of uronic acids. Anal Biochem. 1973 Aug;54(2):484–489. doi: 10.1016/0003-2697(73)90377-1. [DOI] [PubMed] [Google Scholar]
- Bruckner P., Vaughan L., Winterhalter K. H. Type IX collagen from sternal cartilage of chicken embryo contains covalently bound glycosaminoglycans. Proc Natl Acad Sci U S A. 1985 May;82(9):2608–2612. doi: 10.1073/pnas.82.9.2608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eyre D. R., Apon S., Wu J. J., Ericsson L. H., Walsh K. A. Collagen type IX: evidence for covalent linkages to type II collagen in cartilage. FEBS Lett. 1987 Aug 17;220(2):337–341. doi: 10.1016/0014-5793(87)80842-6. [DOI] [PubMed] [Google Scholar]
- Franzén A., Inerot S., Hejderup S. O., Heinegård D. Variations in the composition of bovine hip articular cartilage with distance from the articular surface. Biochem J. 1981 Jun 1;195(3):535–543. doi: 10.1042/bj1950535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh P., Sutherland J. M., Taylor T. K., Bellenger C. R., Pettit G. D. The effect of bilateral medial meniscectomy on articular cartilage of the hip joint. J Rheumatol. 1984 Apr;11(2):197–201. [PubMed] [Google Scholar]
- Ghosh P., Sutherland J., Bellenger C., Read R., Darvodelsky A. The influence of weight-bearing exercise on articular cartilage of meniscectomized joints. An experimental study in sheep. Clin Orthop Relat Res. 1990 Mar;(252):101–113. [PubMed] [Google Scholar]
- Inerot S., Heinegård D., Audell L., Olsson S. E. Articular-cartilage proteoglycans in aging and osteoarthritis. Biochem J. 1978 Jan 1;169(1):143–156. doi: 10.1042/bj1690143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurvelin J., Kiviranta I., Tammi M., Helminen H. J. Effect of physical exercise on indentation stiffness of articular cartilage in the canine knee. Int J Sports Med. 1986 Apr;7(2):106–110. doi: 10.1055/s-2008-1025743. [DOI] [PubMed] [Google Scholar]
- Kempson G. E., Muir H., Pollard C., Tuke M. The tensile properties of the cartilage of human femoral condyles related to the content of collagen and glycosaminoglycans. Biochim Biophys Acta. 1973 Feb 28;297(2):456–472. doi: 10.1016/0304-4165(73)90093-7. [DOI] [PubMed] [Google Scholar]
- Kempson G. E., Spivey C. J., Swanson S. A., Freeman M. A. Patterns of cartilage stiffness on normal and degenerate human femoral heads. J Biomech. 1971 Dec;4(6):597–609. doi: 10.1016/0021-9290(71)90049-2. [DOI] [PubMed] [Google Scholar]
- Kim Y. J., Sah R. L., Doong J. Y., Grodzinsky A. J. Fluorometric assay of DNA in cartilage explants using Hoechst 33258. Anal Biochem. 1988 Oct;174(1):168–176. doi: 10.1016/0003-2697(88)90532-5. [DOI] [PubMed] [Google Scholar]
- Kiviranta I., Tammi M., Jurvelin J., Arokoski J., Sämänen A. M., Helminen H. J. Articular cartilage thickness and glycosaminoglycan distribution in the canine knee joint after strenuous running exercise. Clin Orthop Relat Res. 1992 Oct;(283):302–308. [PubMed] [Google Scholar]
- Kiviranta I., Tammi M., Jurvelin J., Helminen H. J. Topographical variation of glycosaminoglycan content and cartilage thickness in canine knee (stifle) joint cartilage. Application of the microspectrophotometric method. J Anat. 1987 Feb;150:265–276. [PMC free article] [PubMed] [Google Scholar]
- Lammi M. J., Tammi M. Autoradiographic quantitation of radiolabeled proteoglycans. J Biochem Biophys Methods. 1991 May-Jun;22(4):301–310. doi: 10.1016/0165-022x(91)90036-v. [DOI] [PubMed] [Google Scholar]
- Lammi M., Tammi M. Densitometric assay of nanogram quantities of proteoglycans precipitated on nitrocellulose membrane with Safranin O. Anal Biochem. 1988 Feb 1;168(2):352–357. doi: 10.1016/0003-2697(88)90329-6. [DOI] [PubMed] [Google Scholar]
- Lemperg R., Larsson S. E., Hjertquist S. O. The glycosaminoglycans of bovine articular cartilage. I. Concentration and distribution in different layers in relation to age. Calcif Tissue Res. 1974;15(3):237–251. doi: 10.1007/BF02059060. [DOI] [PubMed] [Google Scholar]
- Lust G., Pronsky W. Glycosaminoglycan contents of normal and degenerative articular cartilage from dogs. Clin Chim Acta. 1972 Jul;39(2):281–286. doi: 10.1016/0009-8981(72)90045-9. [DOI] [PubMed] [Google Scholar]
- Manicourt D. H., Pita J. C. Progressive depletion of hyaluronic acid in early experimental osteoarthritis in dogs. Arthritis Rheum. 1988 Apr;31(4):538–544. doi: 10.1002/art.1780310411. [DOI] [PubMed] [Google Scholar]
- McDevitt C. A., Muir H. Biochemical changes in the cartilage of the knee in experimental and natural osteoarthritis in the dog. J Bone Joint Surg Br. 1976 Feb;58(1):94–101. doi: 10.1302/0301-620X.58B1.131804. [DOI] [PubMed] [Google Scholar]
- Palmoski M. J., Brandt K. D. Running inhibits the reversal of atrophic changes in canine knee cartilage after removal of a leg cast. Arthritis Rheum. 1981 Nov;24(11):1329–1337. doi: 10.1002/art.1780241101. [DOI] [PubMed] [Google Scholar]
- Palmoski M. J., Colyer R. A., Brandt K. D. Joint motion in the absence of normal loading does not maintain normal articular cartilage. Arthritis Rheum. 1980 Mar;23(3):325–334. doi: 10.1002/art.1780230310. [DOI] [PubMed] [Google Scholar]
- Parkkinen J. J., Lammi M. J., Helminen H. J., Tammi M. Local stimulation of proteoglycan synthesis in articular cartilage explants by dynamic compression in vitro. J Orthop Res. 1992 Sep;10(5):610–620. doi: 10.1002/jor.1100100503. [DOI] [PubMed] [Google Scholar]
- Pond M. J., Nuki G. Experimentally-induced osteoarthritis in the dog. Ann Rheum Dis. 1973 Jul;32(4):387–388. doi: 10.1136/ard.32.4.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sweet M. B., Thonar E. J., Immelman A. R. Regional distribution of water and glycosaminoglycan in immature articular cartilage. Biochim Biophys Acta. 1977 Nov 7;500(1):173–186. doi: 10.1016/0304-4165(77)90057-5. [DOI] [PubMed] [Google Scholar]
- Sämänen A. M., Tammi M., Jurvelin J., Kiviranta I., Helminen H. J. Proteoglycan alterations following immobilization and remobilization in the articular cartilage of young canine knee (stifle) joint. J Orthop Res. 1990 Nov;8(6):863–873. doi: 10.1002/jor.1100080612. [DOI] [PubMed] [Google Scholar]
- Sämänen A. M., Tammi M., Kiviranta I., Jurvelin J., Helminen H. J. Levels of chondroitin-6-sulfate and nonaggregating proteoglycans at articular cartilage contact sites in the knees of young dogs subjected to moderate running exercise. Arthritis Rheum. 1989 Oct;32(10):1282–1292. doi: 10.1002/anr.1780321014. [DOI] [PubMed] [Google Scholar]
- Sämänen A. M., Tammi M., Kiviranta I., Jurvelin J., Helminen H. J. Maturation of proteoglycan matrix in articular cartilage under increased and decreased joint loading. A study in young rabbits. Connect Tissue Res. 1987;16(2):163–175. doi: 10.3109/03008208709002004. [DOI] [PubMed] [Google Scholar]
- Tammi M., Sämänen A. M., Jauhiainen A., Malminen O., Kiviranta I., Helminen H. Proteoglycan alterations in rabbit knee articular cartilage following physical exercise and immobilization. Connect Tissue Res. 1983;11(1):45–55. doi: 10.3109/03008208309015010. [DOI] [PubMed] [Google Scholar]
- Vasan N. Effects of physical stress on the synthesis and degradation of cartilage matrix. Connect Tissue Res. 1983;12(1):49–58. doi: 10.3109/03008208309005611. [DOI] [PubMed] [Google Scholar]