Abstract
Renal cell carcinoma (RCC) is among the top 10 most common cancers in both men and women with an estimated 75 000 cases each year in the US. Over the last decade, the therapeutic landscape for patients with metastatic RCC has significantly evolved, with immunotherapy emerging as the new front-line therapy. Despite significant improvement in toxicity profile and survival outcomes, key concerns such as patient selection, treatment sequencing, and intrinsic and acquired resistance remain unresolved. Emerging options such as antibody-based therapeutics (eg, anti-CD70, anti-CA9, and anti-ENPP3) are being explored in clinical trials for patients with cancer resistant or refractory to current immunotherapies. Despite positive results for hematological cancers, breast cancer, and more recently bladder cancer, most antibody-based therapies failed to improve the outcomes in patients with advanced RCC. This underscores the need to understand the underlying causes of failed responses to this treatment class, which will ultimately support the rational design of more effective and tolerable treatments. In this review, we summarize the evolving landscape of RCC therapeutics and describe recent clinical trials with emerging antibody-based therapeutics. We also describe the challenges that need to be overcome for the successful creation of therapeutic antibodies for treating RCC.
Keywords: renal cell carcinoma, metastatic RCC, antibody-based therapies, immunotherapy, clinical trials, emerging drugs
Despite positive results for other cancers, most antibody-based therapies failed to improve outcomes in patients with advanced renal cell carcinoma (RCC). This review summarizes the evolving landscape of RCC therapeutics and describes recent clinical trials with emerging antibody-based therapeutics, as well as the challenges to be overcome for the successful creation of therapeutic antibodies for treating RCC.
Implications for Practice.
In this review, the reader will receive a thorough overview of the various antibody-based therapeutic options for patients with advanced renal cell carcinoma. As the molecular biology and cytogenetics of RCC being explored more, various targeted medications have been investigated to improve patient outcomes; additionally, they provide physicians the chance to tailor therapies for specific patients. In our review, we highlighted the limitations of previous and most recently conducted clinical trials on antibody-based therapeutics and the approaches put forth to overcome those issues.
Introduction
Renal cell carcinoma (RCC) is the most frequent subtype of kidney cancer with an estimated 400 000 occurrences globally.1 Overall, the lifetime risk for developing kidney cancer in men is approximately 1 in 46 (2.02%), and for women is approximately 1 in 80 (1.03%).2 Nearly 30% of patients present with de novo metastatic disease and almost 30% of those with initial localized disease will eventually develop metastases after surgical resection of the primary tumor.3
Over the past 2 decades, there have been significant changes in the management and treatment of metastatic RCC.4 First-generation immunotherapy with interleukins or interferon which was previously standard of care is now fallen out of favor due to subpar results and high toxicity.5,6 Subsequently, the prognosis for both progression-free survival (PFS) and overall survival (OS) was significantly improved by the introduction of tyrosine kinase inhibitors (TKI), primarily VEGF receptor inhibitors.7 Later the immune checkpoint inhibitors (ICI) emerged which alone or in combination with TKIs have led to better oncologic outcomes.8,9 Because RCC is considered as an immunogenic tumor with high numbers of immune cells, targeted immunotherapy is a best alternative to antiangiogenics.10-12 Currently, multiple therapies are available for patients with de novo metastatic or recurrent RCC, owing to collaborative efforts led by scientists and clinicians. Despite improved response rates to currently available therapies, there is still a large fraction of patients (~50%-70%) who develop treatment resistance and disease progression. Therefore, there is an unmet need to develop new therapeutic options for these patients.7 In our review, we will discuss a new class of therapeutics, namely antibody-based therapeutics, that has been tested in clinical trials.
Materials and Methods
We used MEDLINE to retrieve pertinent literature related to pivotal trials and presentations from the American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO) conferences. We used the keywords “renal cell carcinoma,” “RCC,” “metastatic RCC,” “advanced RCC,” “antibody-based therapies,” “immunotherapy,” “clinical trials,” and “emerging drugs,” and restricted our search to articles published since 2010.
The Evolving Therapeutic Landscape of Metastatic RCC
2005-2015: The “VEGF Inhibitors” Era
Before 2005, first-generation immunotherapy using cytokines such as IL-2 or IFN-γ was the gold standard for the treatment of metastatic RCC, but was associated with severe toxicities and poor response rates.5,6 The discovery of VHL inactivation in RCC and its impact on tumor angiogenesis led to the clinical trials evaluating the efficacy of tyrosine kinase inhibitors (small-molecule inhibitors), particularly those targeting the VEGF receptor.13,14 In metastatic RCC, VEGF inhibitors such as sorafenib, sunitinib, and pazopanib led to an ~30% response rate and overall survival (OS) benefit over first-generation immunotherapy.7 Similarly, genetic alterations of the mTOR signaling pathway frequently observed in RCC have led to phase III trials demonstrating the efficacy of mTOR inhibitors temsirolimus and everolimus in a subset of patients with advanced RCC.15 The treatment paradigm then further evolved with the advent of immune checkpoint inhibitors, which were initially used for treating other aggressive cancers such as melanoma and lung cancer.16,17
2015-Present: The “Immune Checkpoint Inhibitors” Era
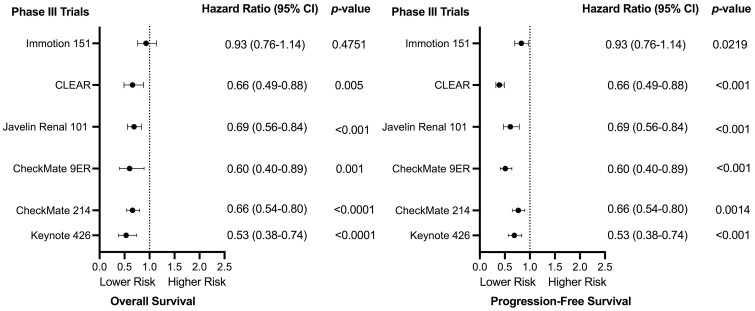
Immune checkpoint inhibitors were used for the treatment of mRCC, given immunogenicity and the high number of infiltrating lymphocytes in these tumors (TIL).11,12,18 Since FDA approval of nivolumab as a second-line therapy for mRCC in 2015, immunotherapy has progressed to become a key treatment for mRCC.19 Following numerous phase III randomized trials, 4 treatments including immune checkpoint inhibitors (Nivolumab, Ipilimumab, Pembrolizumab, and Avelumab) have shown improved survival benefits compared to the tyrosine kinase inhibitor Sunitinib alone and constitute the new backbone for treating advanced RCC. One approach consists of a combination of 2 immune checkpoint inhibitors, Nivolumab (anti-PD1) and Ipilimumab (anti-CTLA4) which was approved in 2018.20 The other treatments include one immune checkpoint inhibitor in combination with a tyrosine kinase inhibitor (Table 1; Fig. 1).
Table 1.
Comparison of the phase III clinical trials investigating immune checkpoint inhibitors.
| Phase III trials | CheckMate 21421 | Keynote 42622 | CheckMate 9ER23 | Javelin Renal 10124 | CLEAR25 | Immotion 15126 | COSMIC-31327 |
|---|---|---|---|---|---|---|---|
| Treatment | Nivo + Ipi vs. Sun | Pembro + Axi vs. Sun | Nivo + Cabo vs. Sun | Ave + Axi vs. Sun | Lenva+Pemb vs. Lenva+Ever vs. Sun | Atezo +Bevaci vs. Sun | Cabo + Nivo +Ipi vs. Nivo + Ipi |
| Number of patients | 1096 | 861 | 651 | 886 | 1069 | 915 | 855 |
| Median follow-up (months) | 32 | 30.6 | 18.1 | 13 | 24 | 24 | 20.2 |
| Median OS (%) | 30Mon: 60 vs. 47 | 12Mon: 90 vs. 78 | 12Mon: 85.7 vs. 75.6 | 12Mon: 86 vs. 83 | 24Mon: 79.2 vs. 66.1 vs. 70.4 | 24Mon: 63 vs. 60 | – |
| Median PFS (months) | 11.6 vs. 8.4 | 15.1 vs. 11.1 | 16.6 vs. 8.3 | 13.8 vs. 7.2 | 23.9 vs. 14.7 vs. 9. | 11.2 vs. 7.7 | NR vs. 11.3 |
| ORR (%) | 42 vs. 34 | 60 vs. 40 | 55.7 vs. 27.1 | 55.2 vs. 25.5 | 71vs. 53.5vs. 36.1 | 37 vs. 33 | 43 vs. 36 |
| CR (%) | 11 vs. 2 | 9 vs. 3 | 8 vs. 4.6 | 3.4 vs. 1.8 | 16.1vs. 9.8vs. 4.2 | 5 vs. 2 | 3 vs. 3 |
| PR (%) | 31 vs. 32 | 51 vs. 37 | 47.7 vs. 22.6 | 48.6 vs. 25.2 | 54.9vs.43.7vs. 31.9 | 31 vs. 31 | 41 vs. 32 |
| SD (%) | 30 vs. 41 | 23 vs. 35 | 32.2 vs. 42.1 | 28.3 vs. 43.7 | 19.2 vs. 33.6vs. 38.1 | 39 vs. 39 | 43 vs. 36 |
| PD (%) | 22 vs. 16 | 11 vs. 17 | 5.6 vs. 13.7 | 12.4 vs. 19.4 | 5.4 vs. 7.3 vs. 14 | 18 vs. 19 | 8 vs. 20 |
| Adverse effects (grade≥3) | 76 vs. 71 | 58 vs. 55 | 75.3 vs. 70.6 | 71.2 vs. 71.5 | 82.4vs. 83.1vs. 71.8 | 40 vs. 54 | 73 vs. 41 |
| Cost-effectiveness | Nivo + Ipi28 | Pembro + Axi29 | Sun23 | Sun30 | – | – | – |
Abbreviations: OS, overall survival; PFS, progressive free survival; ORR, objective response rate; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; Nivo, nivolumab; Ipi, ipilimumab; Sun, sunitinib; Pembro, pembrolizumab; Axi, axitinib; Cabo, cabozantinib; Ave, avelumab; Lenva, lenvatinib; Ever, everolimus; Atezo, atezolizumab; Bevaci, bevacizumab.
Figure 1.
Forest plots showing the association of investigational therapies with risk of mortality and progression-free survival in advanced RCC.
However, the choice of first-line systemic treatment remains challenging as no predictive biomarker to guide treatment selection has been approved to date.31 Patients’ comorbidities and treatment side effects should be considered on a case-by-case basis. Despite improved objective response rates (~40%-70%) with current immunotherapy regimens, the majority of patients develop treatment resistance and disease progression.32,33 Therefore, there is a need to develop novel treatments for patients with refractory or recurrent RCC.3
Emerging Therapeutics for Advanced Renal Cell Carcinoma
Small-Molecule Inhibitors
The growing understanding of RCC biology and progress in bioengineering has enabled the design of novel targeted therapies. This includes small-molecule inhibitors and antibody-based therapies. With the increasing understanding of the VHL/HIF biology in RCC, inhibitors of the HIF-2 transcription factor represent an important line of investigation.34-37 Belzutifan is the first HIF-2 inhibitor to be evaluated in phase III trials for the treatment of advanced ccRCC. It has recently become the first-ever HIF-2 inhibitor approved by the FDA for the treatment of VHL-associated tumors but not yet for non-hereditary renal cell carcinoma.38
Overcoming immunosuppressive barriers within the tumor microenvironment (TME) has become another approach.32 A small-molecule against the adenosine 2A receptor (A2AR) has shown promising antitumor activity in a first-in-human study.39 Adenosine is a nucleoside that can be generated from extracellular ATP through the enzymatic activity of ectonucleotidases such as CD39, ENPP3, and CD73.40 Adenosine binds to A2AR inhibitory receptors expressed by T and NK cells, resulting in immunosuppression. Compared to other solid tumor histology, the adenosine pathway genes ADORA2A (A2AR), ENPP3, and NT5E (CD73) are substantially expressed in RCC.41 Intertumoral hypoxia may also contribute to the formation of extracellular adenosine in RCC tumors by increasing the expression of CD39 and CD73 and encouraging the release of intracellular ATP.42 The phase I trial with the A2AR antagonist CPI-444 (Ciforadenant) included 68 patients with RCC with more than 72% of them having previously unresponsive or refractory anti–PD-1/PD-L1 antibody therapy.43 Ciforadenant was associated with increased recruitment of CD8+ T cells in tumors, diversification of the T-cell receptor (TCR) repertoire in circulating T cells, and durable clinical benefit with a median progression-free survival of 4.1 months and an estimated OS of 69% at 16 months. Therefore, A2AR antagonists represent a promising new form of immune checkpoint inhibitors distinct from PD-1/PD-L1.39 Furthermore, a recent phase II trial was conducted on 69 patients to evaluate the effectiveness of the combination of the mTOR inhibitor (Everolimus) and the oral glutaminase inhibitor (Telaglenastat) versus Everolimus and placebo. Glutaminase is considered a key enzyme in the glutamine-dependent biosynthesis of tumor metabolic intermediates. The combination of Telaglenastat and Everolimus induces synergistic anti-tumor effects and improves progression-free survival (PFS), with a median PFS of 3.8 months compared with 1.9 months in the Everolimus and placebo group.44
Antibody-Based Therapeutics
Antibody-based therapeutics are also emerging as future tools for treating advanced RCC and can be categorized into 2 groups. The first category includes “naked” humanized antibodies which are used directly for therapy. For instance, immune checkpoint inhibitors are monoclonal antibodies that prevent the interaction of immuno-inhibitory ligands with their receptors expressed on the surface of immune cells.9 Some monoclonal antibodies can also induce antibody-dependent cellular cytotoxicity (ADCC) or antibody-dependent cellular phagocytosis (ADCP) naturally. In both mechanisms, natural killer (NK) cells or macrophages recognize the Fc portion of antibodies on the surface of tumor cells leading to tumor cell lysis or phagocytosis respectively.45-47 The second group includes engineered antibodies such as antibody-drug conjugates, radionuclide-conjugated antibodies, bi- and tri-specific antibodies, functionalized liposomes, and chimeric antigen receptor T cells (CAR-T).48-51
The first-ever monoclonal antibody OKT3 (anti-CD3) was approved by the FDA in 1986. Since then, more than 79 therapeutic antibodies have been approved for clinical use, representing a global market of US$115.2 billion in 2018.43 In the following section, we will describe results from recent clinical trials evaluating new antibody-based therapeutics in the treatment of RCC (Fig. 2).
Figure 2.
Recent clinical trials evaluating new antibody-based therapeutics in the treatment of RCC.
Abbreviations: CA9, carbonic anhydrase 9; CAR-T, chimeric antigen receptor T cells; TIM-1, T cell immunoglobulin mucin-1; ADC, antibody-drug conjugate; ENPP3, ectonucleotide pyrophosphatase/phosphodiesterase 3; PFS, progressive-free survival; OS, overall survival.
Antibody-Based Therapeutics in Metastatic RCC: Early Lessons From the Clinic
Anti-CA9
Carbonic anhydrase IX (CA9), a membrane protein that promotes cell proliferation and angiogenesis in response to hypoxia.52 CA9 is overexpressed in different cancers, including RCC, and is activated by hypoxia-inducible factor 1 (HIF1). It is found in more than 80% of primary and metastatic RCC and more than 95% of clear cell RCC.53 As a result, CA9 is a powerful biomarker of RCC and a possible target for RCC immunotherapy.54
Chimeric antigen receptor T cells (CAR-T) models and monoclonal antibodies (mAb) targeting CA9-expressing cells have been recently investigated in clinical trials. CAR-T models are created by inserting the gene for a chimeric T-cell receptor-antibody targeting a specific cancer marker into T cells, enabling the modified cells to target and eliminate cancerous cells.55 In a phase I/II study, 12 patients with metastatic RCC were injected with CAR-T targeting CA9.56 Following infusion, patients generated anti-CAR-T antibodies and anti-CAR-T immune responses. Furthermore, CAR-T infusions resulted in grades 2-4 liver enzyme abnormalities, necessitating therapy discontinuation in 4 out of 12 patients. T-cell infiltration surrounding bile ducts and CA9 expression on bile duct epithelium were found in liver biopsies supporting off-tumor on-target toxicity. The trial was discontinued before achieving therapeutic or maximum tolerated dosage due to the introduction of competitive drugs into the field and the presence of high toxicity.56
Few phases I/II clinical trials have evaluated Girentuximab, a chimeric monoclonal antibody that binds selectively to CA9 and induces ADCC. In the initial safety study, no grade 3 and 4 toxicity was observed and antitumor activity was limited to one complete response out of 36 patients.57 To improve the antitumor activity of Girentuximab in advanced renal cell carcinoma, a second trial tested anti-CA9 antibodies with a low dose of interleukin IL-2.58 Eight patients out of 35 (23%) have shown clinical benefit, which includes 3 patients with partial response and 5 patients with stable disease. Median OS was 22 months. Girentuximab was also tested in a randomized, double-blind, placebo-controlled phase III clinical study as an adjuvant treatment in 864 individuals presenting with RCC and a high risk of postoperative recurrence. However, there was no difference in disease-free survival (DFS) or OS between the 2 groups of patients.59
An alternative therapeutic strategy recently investigated is the use of Girentuximab conjugated with Lutetium-177, a radioligand that delivers radiation to the surrounding environment. Lutetium-177 has recently been found to be useful for treating metastatic castration-refractory prostate cancer.60 In RCC, a phase I trial reported data on 23 patients with metastatic disease treated with 177Lu-labelled Girentuximab and stable disease was observed in 74% of patients.61 In a second trial on 14 patients, most of them developed severe and prolonged grades 3-4 thrombocytopenia which led to treatment discontinuation and only 1 patient demonstrated significant antitumor activity according to Response Evaluation Criteria in Solid Tumors (RECIST) criteria.62 Altogether, these clinical trials indicate that despite CA9 being considered as the most promising tumor target, CAR-T, and monoclonal antibodies against CA9 have failed to demonstrate clinical benefit in patients with mRCC. CA9 is highly expressed in RCC, but normal tissue expression can lead to serious toxicity making it a less clinically useful target. In addition, a recent multi-center phase III trial on 252 mRCC investigated the use of conjugated Girentuximab with Zirconium (TLX250-CDx/ 89Zr-DFO-girentuximab) as a radiolabeled isotope to diagnose clear cell RCC (ccRCC) using the PET scan. The initial results are promising with an 86% sensitivity, 87% specificity, and 93% positive predictive value. All of this indicates that TLX250-CDx has the potential to become a new clinical standard in the diagnosis of ccRCC as a non-invasive diagnostic tool (NCT03849118).
Anti-TIM1
CDX-014 is an antibody-drug conjugate (ADC) that targets T-cell Immunoglobulin Mucin-1 (TIM-1), also known as Kidney Injury Molecule 1 (KIM-1), a transmembrane glycoprotein that is highly expressed in tubular cells in response to ischemia.63 It is found to be highly expressed in 70% of RCC, especially in clear cell and papillary subtypes.64,65 A phase I first-in-human study was conducted on 16 individuals.54 CDX-014 demonstrated anticancer efficacy, resulting in sustained partial response and stable disease in 31% of patients. The 2 patients who had the most significant tumor shrinkage had strong TIM-1 expression on tumor biopsies. Adverse events of grade 3 or higher included hyperglycemia (19%), urosepsis (6%), and one multiple organ failure (6%), which resulted in one treatment-related death. Overall, despite some early evidence of efficacy, this ADC has limited clinical use due to high toxicity profile.66-68
Anti-CD70
CD70, also known as CD27L or TNFSF7, is a type II integral membrane protein found on activated T cells, B cells, and mature dendritic cells.69 Its receptor CD27 is expressed on a high proportion of NK cells as well as resting T and B cells. Signaling through CD27 promotes T-cell and B-cell activation upon CD27L binding.70,71 CD70 is overexpressed in several solid tumors and hematologic malignancies.72-74 Although the causes are unknown, this ligand has characteristics consistent with those of a tumor-associated antigen due to its limited expression in healthy but preferential representation in malignant tissue.73,75 In renal cell carcinoma, CD70 has been found to be overexpressed in a vast majority of cases compared to normal kidney tissue.73,76,77 Importantly, CD70 is rapidly internalized following antibody attachment, making it a viable target for antibody-based cytotoxic drug delivery.75,78
AMG 172 is an ADC constituted of a humanized IgG1 monoclonal antibody (mAb) against CD70 conjugated with a microtubule inhibitor (maytansine).79 It was investigated in a phase I trial of 37 patients with refractory or relapsed RCC. Dose-limiting toxicity that prompted therapy cessation included thrombocytopenia and hepatic damage. Out of 37 patients, 21 reached the maximum tolerated dose and the response was: 2 patients (5.4%) had a partial response, 6 patients (16.2%) had stable disease, and 13 patients (35.1%) had progressive disease. Based on the limited antitumor activity and treatment-related toxicities, no further trials are expected.79
A second ADC, called SGN-CD70A, was also evaluated in a phase I human study.80 SGN-CD70A is an anti-CD70 antibody conjugated with a pyrrolobenzodiazepine which induces DNA breaks and apoptosis. In total, 18 patients with metastatic RCC were enrolled and most patients discontinued treatment because of progressive disease (56%) or adverse events (28%). Like AMG 172, no further trials are ongoing using SGN-CD70A. Further work is needed to understand the causes of treatment-related toxicities and limited antitumor activity.
Anti-ENPP3
Ectonucleotide pyrophosphatase/phosphodiesterase 3 (ENPP3) is one of the most overexpressed surface proteins in RCC, particularly on clear cell histology with up to 94% expression.81,82 In contrast, ENPP3 is found weakly expressed in a fraction of renal tubules and activated basophils and mast cells.83 AGS-16C3F is an ADC against ENPP3 attached to a microtubule inhibitor. Following successful pharmacokinetics and safety study, a phase II trial comparing AGS-16C3F to Axitinib was conducted in the previously 133 treated metastatic RCC.84 Patients were randomized 1:1 and 63% of patients had completed the study at data cutoff. AGS-16C3F did not enhance PFS compared with Axitinib (2.9 months vs. 5.7 months, HR 1.676, P = .015). Similar observations were made for OS (13.1 months for AGS-16C3F versus 15.4 months for Axitinib; HR, 1.079; 95% CI, 0.681-1.707; P = .747). Based on the study findings, no further development of AGS-16C3F in mRCC will be undertaken.84
Anti-CDH6
CDH6 is a cell-adhesion molecule of the cadherin family.85 Protein expression was evaluated in a small subset of primary RCC specimens (39 patients) and CDH6 staining intensity was heterogeneous across samples. In non-human primates, dose-dependent toxicities of HKT288, an ADC against CDH6 carrying a microtubule inhibitor, were observed in corneal epithelium and skin but do not reflect on-target toxicity.85 Data from the first-in-human study in 9 patients with advanced solid tumors was recently reported, including 5 patients with RCC. Three patients developed unexpected grade 2 neurological adverse events (1 seizure, 1 aphasia, and 1 encephalopathy). Further studies investigating the mechanisms of toxicity are critical before further trials with CDH6-targeting agents are pursued.
Current Challenges and Opportunities for Antibody-Based Therapeutics
So far, the therapeutic strategy has been mostly over-simplistic with the idea that a protein highly expressed in RCC is probably the perfect target for antibody-based therapeutics. However, our growing understanding of the cellular and molecular mechanisms of drug resistance and the complex nature of the tumor microenvironment provide important insights to explain the lack of success in recent clinical trials with therapeutic antibodies. In the following section, we describe the biological factors that influence the efficacy of antibody-based therapeutics and propose several strategies to improve the design of more effective and tolerable anticancer drugs (Fig. 3).
Figure 3.
Challenges and opportunities with antibody-based therapeutics.
On-Target Off-Tumor Cytotoxicity
This consists of recognition and destruction of normal tissues that have the shared expression of the targeted protein. The ideal tumor target would be a surface protein only expressed by tumors but, despite tumors accumulating mutations over time, only a few tumor-specific surface proteins have been identified so far (eg, EGFRvIII, mutant FGFR3).86,87 However, overexpression of surface proteins is common in tumors and the expression ratio between normal and tumor tissue can guide the development of high-affinity antibody-based therapeutics. Recent clinical trials with a CAR-T directed against CA9 in RCC illustrate the on-target off-tumor cytotoxicity mechanism.88,89 This highlights the crucial need to establish a full evaluation of target expression in normal human tissues before initiating in-human trials. With the recent advances in profiling technologies, several publicly available databases have been established to interrogate gene and protein expression in normal and tumor tissues (eg, GTEx, TCGA).90 Recently, researchers have developed an interactive CAR clinical trial database combining gene and protein data to show the current “targetable landscape.”91 The GTEx consortium has recently released protein expression profiles of 12 000 genes across 32 normal human tissues using advanced mass spectrometry.92 These resources are helpful to interrogate the normal expression of druggable surface proteins (surfaceome) and anticipate potential on-target off-tumor cytotoxicity.93 Such initiative will significantly drive the discovery of novel targets for next-generation immunotherapy.94
Tumor Uptake
Another challenge to overcome in antibody-based therapies is ensuring that the antibody reaches the intended target. Solid tumors are characterized by a disorganized vasculature and a dense extracellular matrix that limits the tissue penetration of therapeutic antibodies. One approach is to produce antibody fragments with smaller sizes and higher diffusion rates.95,96 There is poor blood flow and hypoxia because the blood vessels within solid tumors are largely immature and disordered.97,98 In addition, plasma leakage into the interstitial space is caused by structural defects in solid tumor blood vessels and an overexpression of pro-angiogenic factors that raise the permeability of the vasculature.98 High interstitial oncotic pressure can constrain the convective flow of therapeutic antibodies from the blood into the interstitial fluid of tumors and can cause tumor blood vessels to collapse.99 One method to improve tumor uptake of therapeutic antibodies is to use pharmacological agents to modify tumor vasculature and tissue permeability.96 Tumor absorption of therapeutic antibodies increased between 40% and 200% when given concomitantly with the vasculature-promoting drugs such as angiotensin II, tumor necrosis factor-alpha, interferon, and interleukin 2.100-103
Tumor Heterogeneity
Intratumoral heterogeneity consists of the co-existence of several tumor clones within the same patient’s tumor, and it can be categorized as spatial heterogeneity or temporal heterogeneity.104 Intratumoral heterogeneity has been well documented in RCC.105-107 Intertumoral heterogeneity defines genotypic and phenotypic differences between different tumors within the same patients (ie, primary tumor versus metastasis). It can be attributed to multiple factors including somatic mutations, environmental pressures, tumor-immune interactions, and prior systemic therapies.104 Intertumoral heterogeneity can be a major cause of resistance to therapy in metastatic cancer patients as patient selection for antibody-based therapeutics is usually based on target protein expression in resected primary tumor specimens. However, throughout the past few years, several studies have demonstrated molecular differences between primary RCC tumors and patient-matched metastases.108-111 One group showed a discordance rate of 27% and 22% for PD-1 and PD-L1, respectively between patient-matched primary and metastatic ccRCC.112 Importantly, both proteins were less expressed in metastatic lesions compared to primary tumors. One important caveat in clinical trials evaluating therapeutic antibodies for the treatment of RCC is the lack of information on protein expression in metastatic specimens. In the failed trial with SGN-CD70A, the authors acknowledged that it is uncertain if CD70 expression differed across primary and metastatic locations, or whether CD70 expression changed throughout therapy.80 For CA9, one study has shown a very poor concordance rate (<50%) in metastatic lesions within the same patient.113 Altogether, this data highlights the need to select patients based on target protein expression in metastatic lesions. While obtaining metastatic biopsy samples can be challenging due to their invasiveness, a promising way to overcome this is through utilizing liquid biopsies.114,115 In the context of antibody-based therapeutics, circulating tumor cells (CTCs) and extracellular vesicles (EVs) hold significant potential as they carry surface proteins expressed by the tumor they originate from. CTCs are disseminating cells from a primary tumor and are thought to form metastases at remote sites. Patients with metastatic disease usually have a higher CTC blood count than those with localized disease.116 In the blood of RCC patients, CA9 has been detected on the surface of CTCs, which provides a proof of feasibility for isolation of CTCs from patient blood to assess the expression of targets in metastasis-derived cells.117
Extracellular vesicles (EVs) are small particles (~30 nm to 5 µm) released naturally by all cell types including tumor cells. They contain molecular cargo (DNA, RNA, proteins) from donor cells and their detection in blood and urine offers new opportunities for cancer diagnosis and early detection of disease recurrence. Organ- or cancer-specific EVs can be detected directly from biofluids using cutting-edge technologies such as nanoscale flow cytometry and interferometric imaging.118 Several proteins upregulated in RCC have been found in urine-derived EVs, including ENPP3, PODXL, and AQP1.119 Other groups have reported the presence of CA9 and CD70 in blood- and urine-derived EVs. isolated from RCC patients.120-122 These findings support the potential of liquid biopsies to guide treatment decisions in biomarker-driven clinical trials. However, CTC and EV research is still in an exploratory phase and further studies are needed to fully characterize the molecular profile of RCC-derived CTCs and EVs. and assess their clinical value for patient selection and predictive markers of treatment response.
Another interesting approach to determining target protein expression in inaccessible metastatic lesions is positron emission tomography (PET). Radiotracers can be conjugated into antibodies or ligands specific to tumors. Several radiotracers are currently under investigation, including 89Zr-Geruntuximab (anti-CA9) and 89Zr-Atezolizumab (anti-PD-L1).123,124 A clinical study on 195 patients showed a sensitivity and a specificity of 86.2% and 85.9% for the detection of malignant renal masses with 89Zr-Geruntuximab.125
Circulating Molecular Decoys
The primary target for the therapeutic antibodies is a protein expressed on the surface of tumor cells. However, alternative forms of the protein target can be found in circulation in the form of soluble protein or on the surface of extracellular vesicles. As described above, tumor cells release EVs as an intrinsic mechanism of cargo elimination and the EV proteome reflects the cell of origin.126 Within EVs, surface proteins can preserve their structural conformation and topology. Interestingly, engineered EVs with specific cytokine receptors have been developed to reduce systemic inflammation by capturing pro-inflammatory cytokines IL-6 and TNF-a.127 Similarly, circulating EVs expressing the virus entry receptor ACE2 were effective to neutralize SARS-Cov2 and inhibit its infectivity.128-130 While EVs can be used as a therapeutic tool, they can also serve as a mechanism of resistance to antibody-based therapies. SARS-CoV-2 infected cells can release EVs carrying the virus spike protein and act as a decoy target for convalescent patient serum-derived neutralizing antibodies.131 Therefore, we can assume that circulating EVs can capture monoclonal therapeutic antibodies and prevent their accumulation in the target site (ie, metastatic lesions).132-134 In line with this, a recent study has demonstrated the predictive value of circulating PD-1+-EVs in melanoma patients treated with nivolumab (anti-PD-1).135 PD-1+-EVs can bind to nivolumab and higher levels of PD-1+-EVs were found in non-responder patients, which supports the hypothesis that EVs can act as molecular decoys to antibodies resulting in therapeutic resistance. Soluble proteins can be produced through proteolytic cleavage or released upon cell death. By being functional, they can also contribute to resistance to therapeutic antibodies through mechanisms similar to EVs. To date, there is no FDA-approved clinical intervention to inhibit EV biogenesis. Recently, it has been demonstrated the feasibility of eliminating soluble PD-L1 and PD-L1-positive EVs from patient’s blood by therapeutic plasma exchange (TPE).136 During TPE the blood is passed through an apheresis machine separating plasma from cellular components. After one single treatment, ~70% of soluble PD-L1 and PD-L1-positive EVs were eliminated from the patient’s blood. A phase I trial is currently ongoing to investigate the therapeutic benefit of plasma exchange in melanoma patients treated with immunotherapy (NCT04581382).
Conclusions
Over the last decade, the therapeutic landscape for patients with metastatic RCC has greatly evolved, with tailored immunotherapy emerging as a new pillar in front-line treatment. Despite these major advances, critical issues like optimal patient selection, treatment sequencing, and overcoming intrinsic and acquired resistance remain to be solved. Antibody-based therapeutics have shown promise in other malignancies, but the lack of success in recent clinical trials in kidney cancer underscores the need to better understand resistance mechanisms and other biological barriers to their efficient use. Importantly, a systematic evaluation of the expression and regulation of targetable proteins in tumors and normal tissue is essential to anticipate treatment-related toxicities and to develop new drugs.
Contributor Information
Ahmed M Mahmoud, Department of Urology, Mayo Clinic, Rochester, MN, USA.
Reza Nabavizadeh, Department of Urology, Mayo Clinic, Rochester, MN, USA.
Rodrigo Rodrigues Pessoa, Department of Urology, Mayo Clinic, Rochester, MN, USA.
Ishita Garg, Department of Immunology, Mayo Clinic, Rochester, MN, USA.
Jacob Orme, Department of Oncology, Mayo Clinic, Rochester, MN, USA.
Brian A Costello, Department of Urology, Mayo Clinic, Rochester, MN, USA; Department of Oncology, Mayo Clinic, Rochester, MN, USA.
John Cheville, Department of Anatomic Pathology, Mayo Clinic, Rochester, MN, USA.
Fabrice Lucien, Department of Urology, Mayo Clinic, Rochester, MN, USA.
Funding
This research received no external funding.
Conflict of Interest
The authors indicated no financial relationships.
Author Contributions
Conception and design: F.L., B.A.C., J.C. Collection of data: A.M.M., J.O., I.G. Manuscript writing: A.M.M., R.N., R.R.P., I.G., J.O., B.A.C., F.L. Final approval of the manuscript: All authors. Accountable for all aspects of the work: All authors.
Data Availability
No new data were generated or analyzed in support of this research.
References
- 1. Siegel RL, Miller KD, Fuchs HE, Jemal A.. Cancer statistics, 2021. CA Cancer J Clin. 2021;71(1):7-33. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33433946. [DOI] [PubMed] [Google Scholar]
- 2. Lifetime risk estimates, CSR 1975-2016. Available from: https://seer.cancer.gov/csr/1975_2016/results_merged/topic_lifetime_risk.pdf
- 3. Ljungberg B, Campbell SC, Choi HY, et al. The epidemiology of renal cell carcinoma. Eur Urol. 2011;60(4):615-621. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21741761. [DOI] [PubMed] [Google Scholar]
- 4. Albiges L, Powles T, Staehler M, et al. Updated European Association of Urology Guidelines on renal cell carcinoma: immune checkpoint inhibition is the new backbone in first-line treatment of metastatic clear-cell renal cell carcinoma. Eur Urol. 2019;76(2):151-156. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31151678. [DOI] [PubMed] [Google Scholar]
- 5. Motzer RJ, Bacik J, Murphy BA, Russo P, Mazumdar M.. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. 2002;20(1):289-296. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11773181. [DOI] [PubMed] [Google Scholar]
- 6. Fyfe G, Fisher RI, Rosenberg SA, et al. Results of treatment of 255 patients with metastatic renal cell carcinoma who received high-dose recombinant interleukin-2 therapy. J Clin Oncol. 1995;13(3):688-696. Available from: http://www.ncbi.nlm.nih.gov/pubmed/7884429. [DOI] [PubMed] [Google Scholar]
- 7. Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356(2):115-124. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17215529. [DOI] [PubMed] [Google Scholar]
- 8. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. 2018;378(14):1277-1290. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29562145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015;373(19):1803-1813. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26406148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Leite KRM, Reis ST, Junior JP, et al. PD-L1 expression in renal cell carcinoma clear cell type is related to unfavorable prognosis. Diagn Pathol. 2015;10:189. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26470780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Thompson RH, Dong H, Kwon ED.. Implications of B7-H1 expression in clear cell carcinoma of the kidney for prognostication and therapy. Clin Cancer Res. 2007;13(2 Pt 2):709s-715s. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17255298 [DOI] [PubMed] [Google Scholar]
- 12. Thompson RH, Dong H, Lohse CM, et al. PD-1 is expressed by tumor-infiltrating immune cells and is associated with poor outcome for patients with renal cell carcinoma. Clin Cancer Res. 2007;13(6):1757-1761. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17363529. [DOI] [PubMed] [Google Scholar]
- 13. Haas NB, Manola J, Uzzo RG, et al. Adjuvant sunitinib or sorafenib for high-risk, non-metastatic renal-cell carcinoma (ECOG-ACRIN E2805): a double-blind, placebo-controlled, randomised, phase 3 trial. Lancet (London, England). 2016;387(10032):2008-2016. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26969090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Ravaud A, Motzer RJ, Pandha HS, et al. Adjuvant Sunitinib in high-risk renal-cell carcinoma after nephrectomy. N Engl J Med. 2016;375(23):2246-2254. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27718781. [DOI] [PubMed] [Google Scholar]
- 15. Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet (London, England). 2008;372(9637):449-456. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18653228 [DOI] [PubMed] [Google Scholar]
- 16. Barrios DM, Do MH, Phillips GS, et al. Immune checkpoint inhibitors to treat cutaneous malignancies. J Am Acad Dermatol. 2020;83(5):1239-1253. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32461079 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Melosky B, Cheema PK, Brade A, et al. Prolonging survival: the role of immune checkpoint inhibitors in the treatment of extensive-stage small cell lung cancer. Oncologist. 2020;25(11):981-992. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32860288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Malouf GG, Compérat E, Yao H, et al. Unique transcriptomic profile of collecting duct carcinomas relative to upper tract urothelial carcinomas and other kidney carcinomas. Sci Rep. 2016;6:30988. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27484008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hsieh JJ, Purdue MP, Signoretti S, et al. Renal cell carcinoma. Nat Rev Dis Prim. 2017 Mar 9;3:17009. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28276433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Gao X, McDermott DF.. Ipilimumab in combination with nivolumab for the treatment of renal cell carcinoma. Expert Opin Biol Ther. 2018;18(9):947-957. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30124333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Motzer RJ, Rini BI, McDermott DF, et al. Nivolumab plus ipilimumab versus sunitinib in first-line treatment for advanced renal cell carcinoma: extended follow-up of efficacy and safety results from a randomised, controlled, phase 3 trial. Lancet Oncol. 2019;20(10):1370-1385. Available from:http://www.ncbi.nlm.nih.gov/pubmed/31427204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380(12):1116-1127. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30779529. [DOI] [PubMed] [Google Scholar]
- 23. Liao W, Lei W, Feng M, et al. Cost-effectiveness analysis of first-line nivolumab plus cabozantinib for advanced renal cell carcinoma in the United States. Adv Ther. 2021;38(12):5662-5670. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34664194. [DOI] [PubMed] [Google Scholar]
- 24. Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380(12):1103-1115. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30779531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Motzer R, Alekseev B, Rha S-Y, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384(14):1289-1300. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33616314 [DOI] [PubMed] [Google Scholar]
- 26. Rini BI, Powles T, Atkins MB, et al. Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet (London, England). 2019;393(10189):2404-2415. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31079938 [DOI] [PubMed] [Google Scholar]
- 27. Choueiri TK, Powles TB, Albiges L, et al. COSMIC_313. Available from: https://oncologypro.esmo.org/meeting-resources/esmo-congress/phase-iii-study-of-cabozantinib-c-in-combination-with-nivolumab-n-and-ipilimumab-i-in-previously-untreated-advanced-renal-cell-carcinoma-arc
- 28. Reinhorn D, Sarfaty M, Leshno M, et al. A cost-effectiveness analysis of nivolumab and ipilimumab versus sunitinib in first-line intermediate- to poor-risk advanced renal cell carcinoma. Oncologist. 2019;24(3):366-371. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30710066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ding D, Hu H, Shi Y, et al. Cost-effectiveness of pembrolizumab plus axitinib versus sunitinib as first-line therapy in advanced renal cell carcinoma in the U.S. Oncologist. 2021;26(2):e290-e297. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32918790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lu P, Liang W, Li J, et al. A cost-effectiveness analysis: first-line avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. Front Pharmacol. 2020;11:619. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32457618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Deleuze A, Saout J, Dugay F, et al. Immunotherapy in renal cell carcinoma: the future is now. Int J Mol Sci. 2020;21(7):1-22. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32260578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A.. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. 2017;168(4):707-723. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28187290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Anderson AC, Joller N, Kuchroo VK.. Lag-3, Tim-3, and TIGIT: co-inhibitory receptors with specialized functions in immune regulation. Immunity. 2016;44(5):989-1004. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27192565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Chen W, Hill H, Christie A, et al. Targeting renal cell carcinoma with a HIF-2 antagonist. Nature. 2016;539(7627):112-117. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27595394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Xu R, Wang K, Rizzi JP, et al. 3-[(1S,2S,3R)-2,3-Difluoro-1-hydroxy-7-methylsulfonylindan-4-yl]oxy-5-fluorobenzonitrile (PT2977), a hypoxia-inducible factor 2α (HIF-2α) inhibitor for the treatment of clear cell renal cell carcinoma. J Med Chem. 2019;62(15):6876-6893. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31282155. [DOI] [PubMed] [Google Scholar]
- 36. Courtney KD, Ma Y, Diaz de Leon A, et al. HIF-2 complex dissociation, target inhibition, and acquired resistance with PT2385, a first-in-class HIF-2 inhibitor, in patients with clear cell renal cell carcinoma. Clin Cancer Res. 2020;26(4):793-803. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31727677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Choueiri TK, Kaelin WG.. Targeting the HIF2-VEGF axis in renal cell carcinoma. Nat Med. 2020;26(10):1519-1530. 10.1038/s41591-020-1093-z. [DOI] [PubMed] [Google Scholar]
- 38. Jonasch E, Donskov F, Iliopoulos O, et al. Belzutifan for renal cell carcinoma in von Hippel-Lindau Disease. N Engl J Med. 2021;385(22):2036-2046. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34818478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Fong L, Hotson A, Powderly JD, et al. Adenosine 2A receptor blockade as an immunotherapy for treatment-refractory renal cell cancer. Cancer Discov. 2020;10(1):40-53. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31732494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Perrot I, Michaud H-A, Giraudon-Paoli M, et al. Blocking antibodies targeting the CD39/CD73 immunosuppressive pathway unleash immune responses in combination cancer therapies. Cell Rep. 2019;27(8):2411-2425.e9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31116985 [DOI] [PubMed] [Google Scholar]
- 41. Linehan WM, Ricketts CJ.. The cancer genome atlas of renal cell carcinoma: findings and clinical implications. Nat Rev Urol. 2019;16(9):539-552. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31278395 [DOI] [PubMed] [Google Scholar]
- 42. Sitkovs.ky MV, Hatfield S, Abbott R, et al. Hostile, hypoxia-A2-adenosinergic tumor biology as the next barrier to overcome for tumor immunologists. Cancer Immunol Res. 2014;2(7):598-605. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24990240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Lu R-M, Hwang Y-C, Liu I-J, et al. Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci. 2020;27(1):1. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31894001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Meric-Bernstam F, Tannir NM, Iliopoulos O, et al. Telaglenastat plus cabozantinib or everolimus for advanced or metastatic renal cell carcinoma: an open-label phase I trial. Clin Cancer Res. 2022;28(8):1540-1548. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35140121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Saunders KO. Conceptual approaches to modulating antibody effector functions and circulation half-life. Front Immunol. 2019;10:1296. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31231397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Kelley RF, Meng YG.. Methods to engineer and identify IgG1 variants with improved FcRn binding or effector function. Methods Mol Biol. 2012;901:277-293. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22723108 [DOI] [PubMed] [Google Scholar]
- 47. Liu Z, Gunasekaran K, Wang W, et al. Asymmetrical Fc engineering greatly enhances antibody-dependent cellular cytotoxicity (ADCC) effector function and stability of the modified antibodies. J Biol Chem. 2014;289(6):3571-3590. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24311787 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Beck A, Goetsch L, Dumontet C, Corvaïa N.. Strategies and challenges for the next generation of antibody-drug conjugates. Nat Rev Drug Discov. 2017;16(5):315-337. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28303026 [DOI] [PubMed] [Google Scholar]
- 49. Labrijn AF, Janmaat ML, Reichert JM, Parren PWHI.. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov. 2019;18(8):585-608. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31175342 [DOI] [PubMed] [Google Scholar]
- 50. Ohradanova-Repic A, Nogueira E, Hartl I, et al. Fab antibody fragment-functionalized liposomes for specific targeting of antigen-positive cells. Nanomedicine. 2018;14(1):123-130. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28939491 [DOI] [PubMed] [Google Scholar]
- 51. Feins S, Kong W, Williams EF, Milone MC, Fraietta JA.. An introduction to chimeric antigen receptor (CAR) T-cell immunotherapy for human cancer. Am J Hematol. 2019;94(S1):S3-S9. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30680780. [DOI] [PubMed] [Google Scholar]
- 52. Wykoff CC, Beasley NJ, Watson PH, et al. Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res. 2000;60(24):7075-7083. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11156414 [PubMed] [Google Scholar]
- 53. Uemura H, Nakagawa Y, Yoshida K, et al. MN/CA IX/G250 as a potential target for immunotherapy of renal cell carcinomas. Br J Cancer. 1999;81(4):741-746. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10574265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. McGregor BA, Gordon M, Flippot R, et al. Safety and efficacy of CDX-014, an antibody-drug conjugate directed against T cell immunoglobulin mucin-1 in advanced renal cell carcinoma. Invest New Drugs. 2020;38(6):1807-1814. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32472319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. June CH, Sadelain M.. Chimeric antigen receptor therapy. N Engl J Med. 2018;379(1):64-73. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29972754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Lamers CHJ, Klaver Y, Gratama JW, Sleijfer S, Debets R.. Treatment of metastatic renal cell carcinoma (mRCC) with CAIX CAR-engineered T-cells-a completed study overview. Biochem Soc Trans. 2016 Jun 15;44(3):951-959. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27284065 [DOI] [PubMed] [Google Scholar]
- 57. Bleumer I, Knuth A, Oosterwijk E, et al. A phase II trial of chimeric monoclonal antibody G250 for advanced renal cell carcinoma patients. Br J Cancer. 2004;90(5):985-990. Available from: http://www.ncbi.nlm.nih.gov/pubmed/14997194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Bleumer I, Oosterwijk E, Oosterwijk-Wakka JC, et al. A clinical trial with chimeric monoclonal antibody WX-G250 and low dose interleukin-2 pulsing scheme for advanced renal cell carcinoma. J Urol. 2006;175(1):57-62. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16406869 [DOI] [PubMed] [Google Scholar]
- 59. Chamie K, Donin NM, Klöpfer P, et al. Adjuvant weekly girentuximab following nephrectomy for high-risk renal cell carcinoma: the ARISER randomized clinical trial. JAMA Oncol. 2017 Jul 1;3(7):913-920. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27787547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Sartor O, de Bono J, Chi KN, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med. 2021;385(12):1091-1103. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34161051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Stillebroer AB, Boerman OC, Desar IME, et al. Phase 1 radioimmunotherapy study with lutetium 177-labeled anti-carbonic anhydrase IX monoclonal antibody girentuximab in patients with advanced renal cell carcinoma. Eur Urol. 2013;64(3):478-485. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22980441 [DOI] [PubMed] [Google Scholar]
- 62. Muselaers CHJ, Boers-Sonderen MJ, van Oostenbrugge TJ, et al. Phase 2 study of lutetium 177-labeled anti-carbonic anhydrase IX monoclonal antibody girentuximab in patients with advanced renal cell carcinoma. Eur Urol. 2016;69(5):767-770. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26706103 [DOI] [PubMed] [Google Scholar]
- 63. Han WK, Bailly V, Abichandani R, Thadhani R, Bonventre JV.. Kidney Injury Molecule-1 (KIM-1): a novel biomarker for human renal proximal tubule injury. Kidney Int. 2002;62(1):237-244. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12081583 [DOI] [PubMed] [Google Scholar]
- 64. Han WK, Alinani A, Wu C-L, et al. Human kidney injury molecule-1 is a tissue and urinary tumor marker of renal cell carcinoma. J Am Soc Nephrol. 2005;16(4):1126-1134. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15744000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Lin F, Zhang PL, Yang XJ, et al. Human kidney injury molecule-1 (hKIM-1): a useful immunohistochemical marker for diagnosing renal cell carcinoma and ovarian clear cell carcinoma. Am J Surg Pathol. 2007 Mar;31(3):371-381. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17325478 [DOI] [PubMed] [Google Scholar]
- 66. Ye L, Zhang Q, Cheng Y, et al. Tumor-derived exosomal HMGB1 fosters hepatocellular carcinoma immune evasion by promoting TIM-1+ regulatory B cell expansion. J ImmunoTher Cancer. 2018;6(1):145. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30526680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Kong X, Fu M, Niu X, Jiang H.. Comprehensive analysis of the expression, relationship to immune infiltration and prognosis of TIM-1 in cancer. Front Oncol. 2020;10:1086. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33014768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Thomas LJ, Vitale L, O’Neill T, et al. Development of a novel antibody-drug conjugate for the potential treatment of ovarian, lung, and renal cell carcinoma expressing TIM-1. Mol Cancer Ther. 2016;15(12):2946-2954. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27671527 [DOI] [PubMed] [Google Scholar]
- 69. Goodwin RG, Alderson MR, Smith CA, et al. Molecular and biological characterization of a ligand for CD27 defines a new family of cytokines with homology to tumor necrosis factor. Cell. 1993;73(3):447-456. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8387892 [DOI] [PubMed] [Google Scholar]
- 70. Bullock TNJ, Yagita H.. Induction of CD70 on dendritic cells through CD40 or TLR stimulation contributes to the development of CD8+ T cell responses in the absence of CD4+ T cells. J Immunol. 2005;174(2):710-717. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15634890 [DOI] [PubMed] [Google Scholar]
- 71. Jin L, Ge H, Long Y, et al. CD70, a novel target of CAR T-cell therapy for gliomas. Neuro Oncol. 2018;20(1):55-65. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28651374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Held-Feindt J, Mentlein R.. CD70/CD27 ligand, a member of the TNF family, is expressed in human brain tumors. Int J Cancer. 2002;98(3):352-356. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11920585 [DOI] [PubMed] [Google Scholar]
- 73. Junker K, Hindermann W, von Eggeling F, et al. CD70: a new tumor specific biomarker for renal cell carcinoma. J Urol. 2005;173(6):2150-2153. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15879877 [DOI] [PubMed] [Google Scholar]
- 74. Pich C, Sarrabayrouse G, Teiti I, et al. Melanoma-expressed CD70 is involved in invasion and metastasis. Br J Cancer. 2016;114(1):63-70. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26671750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Law C-L, Gordon KA, Toki BE, et al. Lymphocyte activation antigen CD70 expressed by renal cell carcinoma is a potential therapeutic target for anti-CD70 antibody-drug conjugates. Cancer Res. 2006;66(4):2328-2337. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16489038. [DOI] [PubMed] [Google Scholar]
- 76. Ryan MC, Kostner H, Gordon KA, et al. Targeting pancreatic and ovarian carcinomas using the auristatin-based anti-CD70 antibody-drug conjugate SGN-75. Br J Cancer. 2010;103(5):676-684. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20664585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Jilaveanu LB, Sznol J, Aziz SA, et al. CD70 expression patterns in renal cell carcinoma. Hum Pathol. 2012;43(9):1394-1399. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22401771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Adam PJ, Terrett JA, Steers G, et al. CD70 (TNFSF7) is expressed at high prevalence in renal cell carcinomas and is rapidly internalised on antibody binding. Br J Cancer. 2006;95(3):298-306. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16892042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Massard C, Soria J-C, Krauss J, et al. First-in-human study to assess safety, tolerability, pharmacokinetics, and pharmacodynamics of the anti-CD27L antibody-drug conjugate AMG 172 in patients with relapsed/refractory renal cell carcinoma. Cancer Chemother Pharmacol. 2019;83(6):1057-1063. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30915497. [DOI] [PubMed] [Google Scholar]
- 80. Pal SK, Forero-Torres A, Thompson JA, et al. A phase 1 trial of SGN-CD70A in patients with CD70-positive, metastatic renal cell carcinoma. Cancer. 2019;125(7):1124-1132. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30624766. [DOI] [PubMed] [Google Scholar]
- 81. Ricketts CJ, De Cubas AA, Fan H, et al. The cancer genome atlas comprehensive molecular characterization of renal cell carcinoma. Cell Rep. 2018;23(1):313-326.e5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29617669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Doñate F, Raitano A, Morrison K, et al. AGS16F Is a novel antibody drug conjugate directed against ENPP3 for the treatment of renal cell carcinoma. Clin Cancer Res. 2016;22(8):1989-1999. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26589436 [DOI] [PubMed] [Google Scholar]
- 83. Schrödter S, Braun M, Syring I, et al. Identification of the dopamine transporter SLC6A3 as a biomarker for patients with renal cell carcinoma. Mol Cancer. 2016;15:10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/26831905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Kollmannsberger C, Choueiri TK, Heng DYC, et al. A randomized phase II study of AGS-16C3F versus axitinib in previously treated patients with metastatic renal cell carcinoma. Oncologist. 2021;26(3):182-e361. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33289953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Bialucha CU, Collins SD, Li X, et al. Discovery and optimization of HKT288, a cadherin-6-targeting ADC for the treatment of ovarian and renal cancers. Cancer Discov. 2017;7(9):1030-1045. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28526733. [DOI] [PubMed] [Google Scholar]
- 86. An Z, Aksoy O, Zheng T, Fan Q-W, Weiss WA.. Epidermal growth factor receptor and EGFRvIII in glioblastoma: signaling pathways and targeted therapies. Oncogene. 2018;37(12):1561-1575. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29321659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Gust KM, McConkey DJ, Awrey S, et al. Fibroblast growth factor receptor 3 is a rational therapeutic target in bladder cancer. Mol Cancer Ther. 2013;12(7):1245-1254. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23657946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Lamers CHJ, Sleijfer S, Vulto AG, et al. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J Clin Oncol. 2006;24(13):e20-e22. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16648493. [DOI] [PubMed] [Google Scholar]
- 89. Lamers CH, Sleijfer S, van Steenbergen S, et al. Treatment of metastatic renal cell carcinoma with CAIX CAR-engineered T cells: clinical evaluation and management of on-target toxicity. Mol Ther. 2013;21(4):904-912. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23423337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. GTEx Consortium. The Genotype-Tissue Expression (GTEx) project. Nat Genet. 2013 Jun;45(6):580-585. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23715323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. MacKay M, Afshinnekoo E, Rub J, et al. The therapeutic landscape for cells engineered with chimeric antigen receptors. Nat Biotechnol. 2020;38(2):233-244. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31907405 [DOI] [PubMed] [Google Scholar]
- 92. Jiang L, Wang M, Lin S, et al. A quantitative proteome map of the human body. Cell. 2020;183(1):269-283.e19. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32916130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Lavoie RR, Gargollo PC, Ahmed ME, et al. Surfaceome profiling of rhabdomyosarcoma reveals B7-H3 as a mediator of immune evasion. Cancers (Basel). 2021;13(18). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Kuhlmann L, Cummins E, Samudio I, Kislinger T.. Cell-surface proteomics for the identification of novel therapeutic targets in cancer. Expert Rev Proteomics. 2018;15(3):259-275. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29343144. [DOI] [PubMed] [Google Scholar]
- 95. Cruz E, Kayser V.. Monoclonal antibody therapy of solid tumors: clinical limitations and novel strategies to enhance treatment efficacy. Biologics. 2019;13:33-51. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31118560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Lu G, Fakurnejad S, Martin BA, et al. Predicting therapeutic antibody delivery into human head and neck cancers. Clin Cancer Res. 2020;26(11):2582-2594. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31980465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Jain RK. Normalizing tumor vasculature with anti-angiogenic therapy: a new paradigm for combination therapy. Nat Med. 2001;7(9):987-989. Available from: http://www.ncbi.nlm.nih.gov/pubmed/11533692. [DOI] [PubMed] [Google Scholar]
- 98. Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58-62. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15637262 [DOI] [PubMed] [Google Scholar]
- 99. Baxter LT, Jain RK.. Transport of fluid and macromolecules in tumors. II. Role of heterogeneous perfusion and lymphatics. Microvasc Res. 1990;40(2):246-263. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2250603 [DOI] [PubMed] [Google Scholar]
- 100. LeBerthon B, Khawli LA, Alauddin M, et al. Enhanced tumor uptake of macromolecules induced by a novel vasoactive interleukin 2 immunoconjugate. Cancer Res. 1991;51(10):2694-2698. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2021947 [PubMed] [Google Scholar]
- 101. Li J, Merton DA, Duggaraju R, Thakur ML.. Augmenting of tumor uptake of anti-melanoma antibody MEM136: influence of interferon. Nucl Med Biol. 1996;23(7):873-879. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8971854 [DOI] [PubMed] [Google Scholar]
- 102. Folli S, Pèlegrin A, Chalandon Y, et al. Tumor-necrosis factor can enhance radio-antibody uptake in human colon carcinoma xenografts by increasing vascular permeability. Int J Cancer. 199;53(5):829-836. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8449608 [DOI] [PubMed] [Google Scholar]
- 103. Netti PA, Hamberg LM, Babich JW, et al. Enhancement of fluid filtration across tumor vessels: implication for delivery of macromolecules. Proc Natl Acad Sci U S A. 1999;96(6):3137-3142. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10077650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Dagogo-Jack I, Shaw AT.. Tumour heterogeneity and resistance to cancer therapies. Nat Rev Clin Oncol. 2018;15(2):81-94. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29115304. [DOI] [PubMed] [Google Scholar]
- 105. Gerlinger M, Rowan AJ, Horswell S, et al. Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med. 2012 8;366(10):883-892. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22397650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Okegawa T, Morimoto M, Nishizawa S, et al. Intratumor heterogeneity in primary kidney cancer revealed by metabolic profiling of multiple spatially separated samples within tumors. EBioMedicine. 2017;19:31-38. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28408240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Beksac AT, Paulucci DJ, Blum KA, et al. Heterogeneity in renal cell carcinoma. Urol Oncol. 2017;35(8):507-515. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28551412 [DOI] [PubMed] [Google Scholar]
- 108. Lee C, Park J-W, Suh JH, Nam KH, Moon KC.. Histologic variations and immunohistochemical features of metastatic clear cell renal cell carcinoma. Korean J Pathol. 2013;47(5):426-432. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24255630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Barr ML, Jilaveanu LB, Camp RL, et al. PAX-8 expression in renal tumours and distant sites: a useful marker of primary and metastatic renal cell carcinoma? J Clin Pathol. 2015;68(1):12-17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25315900 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Bissig H, Richter J, Desper R, et al. Evaluation of the clonal relationship between primary and metastatic renal cell carcinoma by comparative genomic hybridization. Am J Pathol. 1999;155(1):267-274. Available from: http://www.ncbi.nlm.nih.gov/pubmed/10393858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Zhang J-P, Yuan H-X, Kong W-T, et al. Increased expression of chitinase 3-like 1 and microvessel density predicts metastasis and poor prognosis in clear cell renal cell carcinoma. Tumour Biol. 2014;35(12):12131-12137. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25142236. [DOI] [PubMed] [Google Scholar]
- 112. Eckel-Passow JE, Ho TH, Serie DJ, et al. Concordance of PD-1 and PD-L1 (B7-H1) in paired primary and metastatic clear cell renal cell carcinoma. Cancer Med. 2020;9(3):1152-1160. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31829518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Kim SH, Park WS, Park EY, Joo J, Chung J.. Analysis of the concordance of 20 immunohistochemical tissue markers in metastasectomy lesions in patients with metastatic renal cell carcinoma: a retrospective study using tissue microarray. Investig Clin Urol. 2020;61(4):372-381. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32665993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Cimadamore A, Massari F, Santoni M, et al. Molecular characterization and diagnostic criteria of renal cell carcinoma with emphasis on liquid biopsies. Expert Rev Mol Diagn. 2020;20(2):141-150. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31498685. [DOI] [PubMed] [Google Scholar]
- 115. Lakshminarayanan H, Rutishauser D, Schraml P, Moch H, Bolck HA.. Liquid biopsies in renal cell carcinoma-recent advances and promising new technologies for the early detection of metastatic disease. Front Oncol. 2020;10:582843. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33194717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Palmela Leitão T, Miranda M, Polido J, et al. Circulating tumor cell detection methods in renal cell carcinoma: a systematic review. Crit Rev Oncol Hematol. 2021 May;161:103331. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33862248. [DOI] [PubMed] [Google Scholar]
- 117. Naoe M, Kusaka C, Ohta M, et al. Development of a highly sensitive technique for capturing renal cell cancer circulating tumor cells. Diagnostics (Basel, Switzerland). 2019;9(3):1–10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/31416266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Arab T, Mallick ER, Huang Y, et al. Characterization of extracellular vesicles and synthetic nanoparticles with four orthogonal single-particle analysis platforms. J Extracell Vesicles. 2021;10(6):e12079. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33850608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Dhondt B, Geeurickx E, Tulkens J, et al. Unravelling the proteomic landscape of extracellular vesicles in prostate cancer by density-based fractionation of urine. J Extracell Vesicles. 2020;9(1):1736935. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32284825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Raimondo F, Morosi L, Corbetta S, et al. Differential protein profiling of renal cell carcinoma urinary exosomes. Mol Biosyst. 2013;9(6):1220-1233. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23511837. [DOI] [PubMed] [Google Scholar]
- 121. Vergori L, Martinez MC, Bigot P.. Circulating large extracellular vesicles carrying CA9 in the diagnosis and prognosis of clear-cell renal cell carcinoma. Clin Transl Med. 2021;11(3):e358. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33783991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Himbert D, Zeuschner P, Ayoubian H, et al. Characterization of CD147, CA9, and CD70 as tumor-specific markers on extracellular vesicles in clear cell renal cell carcinoma. Diagnostics (Basel, Switzerland). 2020;10(12):1–17. Available from: http://www.ncbi.nlm.nih.gov/pubmed/33276608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Lindenberg L, Mena E, Choyke PL, Bouchelouche K.. PET imaging in renal cancer. Curr Opin Oncol. 2019;31(3):216-221. Available from: http://www.ncbi.nlm.nih.gov/pubmed/30747736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Hekman MCH, Rijpkema M, Aarntzen EH, et al. Positron emission tomography/computed tomography with 89Zr-girentuximab can aid in diagnostic dilemmas of clear cell renal cell carcinoma suspicion. Eur Urol. 2018;74(3):257-260. Available from: http://www.ncbi.nlm.nih.gov/pubmed/29730017. [DOI] [PubMed] [Google Scholar]
- 125. Divgi CR, Uzzo RG, Gatsonis C, et al. Positron emission tomography/computed tomography identification of clear cell renal cell carcinoma: results from the REDECT trial. J Clin Oncol. 2013;31(2):187-194. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23213092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Hurwitz SN, Rider MA, Bundy JL, et al. Proteomic profiling of NCI-60 extracellular vesicles uncovers common protein cargo and cancer type-specific biomarkers. Oncotarget. 2016;7(52):86999-87015. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27894104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Gupta D, Wiklander OPB, Görgens A, et al. Amelioration of systemic inflammation via the display of two different decoy protein receptors on extracellular vesicles. Nat Biomed Eng. 2021;5(9):1084-1098. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34616047. [DOI] [PubMed] [Google Scholar]
- 128. El-Shennawy L, Hoffmann AD, Dashzeveg NK, et al. Circulating ACE2-expressing extracellular vesicles block broad strains of SARS-CoV-2. Nat Commun. 2022;13(1):405. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35058437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Kim HK, Cho J, Kim E, et al. Engineered small extracellular vesicles displaying ACE2 variants on the surface protect against SARS-CoV-2 infection. J Extracell Vesicles. 2022;11(1):e12179. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34982509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Xie F, Su P, Pan T, et al. Engineering extracellular vesicles enriched with palmitoylated ACE2 as COVID-19 therapy. Adv Mater. 2021;33(49):e2103471. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34665481. [DOI] [PMC free article] [PubMed]
- 131. Troyer Z, Alhusaini N, Tabler CO, et al. Extracellular vesicles carry SARS-CoV-2 spike protein and serve as decoys for neutralizing antibodies. J Extracell Vesicles. 2021;10(8):e12112. Available from: http://www.ncbi.nlm.nih.gov/pubmed/34188786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Ifergan I, Scheffer GL, Assaraf YG.. Novel extracellular vesicles mediate an ABCG2-dependent anticancer drug sequestration and resistance. Cancer Res. 2005;65(23):10952-10958. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16322243. [DOI] [PubMed] [Google Scholar]
- 133. Ciravolo V, Huber V, Ghedini GC, et al. Potential role of HER2-overexpressing exosomes in countering trastuzumab-based therapy. J Cell Physiol. 2012;227(2):658-667. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21465472 [DOI] [PubMed] [Google Scholar]
- 134. Aung T, Chapuy B, Vogel D, et al. Exosomal evasion of humoral immunotherapy in aggressive B-cell lymphoma modulated by ATP-binding cassette transporter A3. Proc Natl Acad Sci USA. 2011;108(37):15336-15341. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21873242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Serratì S, Guida M, Di Fonte R, et al. Circulating extracellular vesicles expressing PD1 and PD-L1 predict response and mediate resistance to checkpoint inhibitors immunotherapy in metastatic melanoma. Mol Cancer. 2022;21(1):20. Available from: http://www.ncbi.nlm.nih.gov/pubmed/35042524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Orme JJ, Enninga EAL, Lucien-Matteoni F, et al. Therapeutic plasma exchange clears circulating soluble PD-L1 and PD-L1-positive extracellular vesicles. J ImmunoTher Cancer. 2020;8(2):e001113. 10.1136/jitc-2020-001113. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32817395 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were generated or analyzed in support of this research.