Abstract
Patients with chronic kidney disease (CKD) exhibit tremendously elevated risk for cardiovascular disease, particularly ischemic heart disease, due to premature vascular and cardiac aging and accelerated ectopic calcification. The presence of cardiovascular calcification associates with increased risk in patients with CKD. Disturbed mineral homeostasis and diverse comorbidities in these patients drive increased systemic cardiovascular calcification in different manifestations with diverse clinical consequences, like plaque instability, vessel stiffening, and aortic stenosis. This review outlines the heterogeneity in calcification patterning, including mineral type and location and potential implications on clinical outcomes. The advent of therapeutics currently in clinical trials may reduce CKD-associated morbidity. Development of therapeutics for cardiovascular calcification begins with the premise that less mineral is better. While restoring diseased tissues to a noncalcified homeostasis remains the ultimate goal, in some cases, calcific mineral may play a protective role, such as in atherosclerotic plaques. Therefore, developing treatments for ectopic calcification may require a nuanced approach that considers individual patient risk factors. Here, we discuss the most common cardiac and vascular calcification pathologies observed in CKD, how mineral in these tissues affects function, and the potential outcomes and considerations for therapeutic strategies that seek to disrupt the nucleation and growth of mineral. Finally, we discuss future patient-specific considerations for treating cardiac and vascular calcification in patients with CKD—a population in need of anticalcification therapies.
Keywords: aortic valve stenosis; minerals; renal insufficiency, chronic; therapeutics; vascular calcification
Cardiovascular calcification is often used as an all-encompassing term to describe the pathological ectopic mineralization of the cardiac valves and arterial walls. However, the deposition of mineral across different cardiac and vascular beds can result from a myriad of pathophysiological mechanisms, and the various presentations of calcification lead to unique clinical issues.1,2 To date, no approved therapeutic strategies exist to prevent or treat cardiovascular calcification. For the general population, calcification in diseased cardiac and vascular tissues develops over the course of decades, complicating potential clinical trials to investigate therapeutic efficacy. Due to the rapidity with which calcification develops in response to renal dysfunction, individuals with chronic kidney disease (CKD) represent a population at need for anticalcification strategies with the potential for completing clinical trials.3 Therapeutics developed for individuals with CKD may then be translatable to other patients at risk for developing ectopic calcification. First, however, potential strategies must consider the effects of mineral across the various vascular beds and off target effects on bone—to avoid unintended deleterious outcomes.
CKD associates with poor outcomes mainly due to a high burden of cardiovascular comorbidity.4 Over the last 2 decades, estimated glomerular filtration rate (eGFR) has emerged as a common measure of renal function and classifier of CKD.5 Stage 1 CKD is classified as mild renal damage with preserved eGFR of over 90 mL/min. Progression of CKD through stage 2 (eGFR, 60–89 mL/min), stage 3a (eGFR, 45–59 mL/min), stage 3b (eGFR, 30–44 mL/min), stage 4 (eGFR, 15–29 mL/min), and end-stage CKD stage 5 (eGFR, <15 mL/min) occurs as renal damage leads to loss of function. Cardiovascular risk increases as CKD progresses, especially with significantly reduced eGFR in later stages,6–8 and vascular calcification correlates strongly with impaired renal function.9 Patients with CKD exhibit >4-fold (CKD stage >3) higher cardiovascular risk compared with the non-CKD cohort, which places about 15% to 20% of adults globally at an excessive cardiovascular risk.10–12 Recent studies have also shown utility in adding urine albumin-creatinine ratio as an additional risk enhancer of cardiovascular risk in CKD.13–15 However, the relationship between albumin-creatinine ratio and cardiovascular calcification remains unclear.
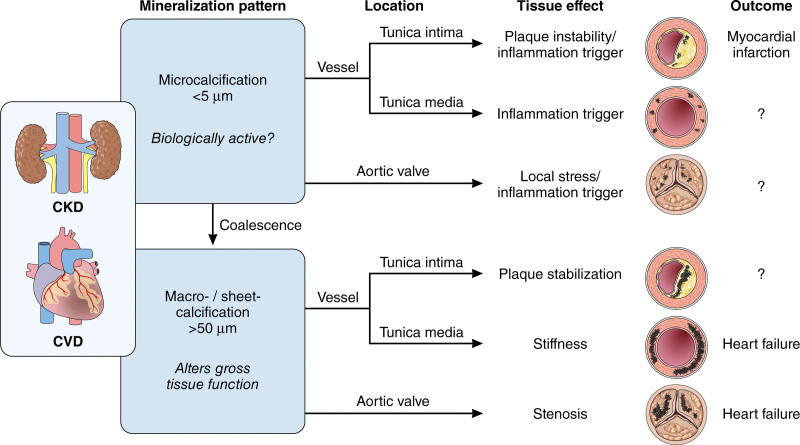
As discussed more throughout this review, the term cardiovascular calcification is used as a general descriptor for the presence of mineral that can occur across different cardiovascular tissues with varied implications to morbidity and mortality depending upon the morphology and location of the mineral. Renal failure greatly increases cardiovascular calcification, both in the tunica intima16–18 and tunica media19 in different vascular beds, as well as heart valves20 with different clinical consequences (Figure 1). Cardiovascular calcification can be seen as a systemic disorder in patients with CKD. Therefore, targeting fibrocalcific remodeling of cardiovascular tissue is being considered to improve cardiovascular outcomes in patients with CKD.
Figure 1.
Heterogeneity of cardiovascular calcification and its clinical consequences. Patients with chronic kidney disease experience accelerated cardiovascular calcification on various cardiovascular sites that are presented in different temporal patterns with diverse clinical consequences. Smaller microcalcifications in the tunica intima may increase plaque instability, promote local stress in the aortic valve leaflets, and trigger inflammation in all vascular beds and the aortic valve. Microcalcifications aggregate to form larger calcifications. Macro/sheet calcification alters gross tissue function: in the tunica intima, it may decrease plaque vulnerability, while it causes vessel stiffening when present in the tunica media and impaired aortic valve function when present in the valve leaflet. Illustration credit: Sceyence Studios.
Increasing evidence suggests that cardiovascular calcification is an independent risk factor for cardiovascular morbidity and mortality in the general population21 and patients with CKD.22 Traditional strategies to reduce this risk are largely ineffective in patients with CKD,23 underscoring the importance of nontraditional CKD-specific cardiovascular risk factors that are hitherto unknown. Atherosclerotic end points like stroke or myocardial infarction are the leading cause of death in the general population.10 Significantly, patients with CKD succumb to sudden cardiac death or ischemic heart disease due to premature vascular and cardiac aging, hallmarked by fibrosis and hypertrophy in the heart and calcification in the vessel wall and cardiac valves.24 CKD associates with coronary heart disease, stroke, peripheral artery disease, arrhythmias, and heart failure4—all associated with cardiovascular calcification. These associations between CKD and different manifestations of cardiovascular disease likely reflect the involvement of several pathophysiological mechanisms and shared risk factors (for example, type 2 diabetes [T2D] and hypertension), changes in bone mineral metabolism, anemia, volume overload, inflammation, and the presence of uremic toxins.
The concept of CKD–mineral and bone disorder was developed to underline the importance of organ cross talk in patients with CKD. The CKD–mineral and bone disorder is a systemic disorder of complex and closely related syndromes of abnormal mineral metabolism (calcium, phosphorus, parathyroid hormone, or vitamin D metabolism), abnormal bone (bone turnover, mineralization volume, and strength), and extraskeletal calcification (vascular, cardiac, and other soft tissue calcification) that together significantly contribute to morbidity and mortality in CKD.25 The dysregulated mineral metabolism with increased phosphate, fibroblast growth factor 23, parathyroid hormone, and 1,25-dihydroxy vitamin D, and various local and systemic factors can contribute to cardiovascular calcification in CKD.26 The mechanisms through which CKD induces cardiovascular calcification seem to be myriad and are still being investigated. Identified triggers and mediators of ectopic vascular mineralization in CKD are well-reviewed elsewhere.27–30 Our focus in the current review is to discuss the outcomes associated with different presentations of calcification and the therapeutic approaches that have been tested or are currently being investigated.
While the focus of the current review is mainly on accelerated ectopic calcification in patients with CKD, the relationship between pathological remodeling in the vasculature and renal dysfunction is not unidirectional. Interestingly, coronary artery calcification (CAC) itself associates with extracardiovascular diseases. Participants of the MESA (Multi-Ethnic Study of Atherosclerosis) cohort with elevated CAC were at increased risk of cancer, CKD, chronic obstructive pulmonary disease, and hip fractures.31 Those without CAC were less likely to develop common age-related comorbidities. Interestingly, cardiovascular calcification might lead to kidney damage that can accelerate adverse renal and associated cardiovascular outcomes. In nonelderly patients free of any cardiovascular disease, renal function declined more rapidly with a high CAC score.32 In patients with CKD, high CAC was associated with an increased risk of adverse kidney outcomes and CKD progression.33 These studies demonstrate the importance of inter-organ interactions in patients with CKD and the potential involvement of ectopic calcification in the cardiorenal syndrome.
No clinically viable therapies for the treatment and prevention of cardiovascular calcification or early biomarkers have been approved to date, partially attributable to the asymptomatic progression of calcification and the lack of high-resolution imaging techniques to identify early calcification before it reaches a point of no return. This emphasizes the need to understand early events in the progression of cardiovascular calcification and its association with other comorbidities in patients with CKD, as those patients often also experience hypertension and T2D.
Here, we review the various presentations of cardiovascular calcification, the associated cardiovascular risk, and therapeutic considerations to improve outcomes in patients with CKD.
Heterogeneity of Cardiovascular Calcification and Its Clinical Implications
As mentioned, patients with CKD are predisposed to developing calcification across various vascular beds. While calcification of the medial layer of central and peripheral arteries represents a common pathology associated with CKD, patients with impaired renal function also commonly exhibit intimal calcification associated with atherosclerosis and calcification of heart valve tissues, which leads to cardiac dysfunction. The relative development of calcific mineral across these tissues in any individual patient remains unpredictable but likely depends on the presence of other risk factors. Further, within any of these tissues, mineral size, shape, and distribution can also vary and lead to different functional outcomes. When we use the term heterogeneity in this review, we intend to capture the variance of calcification at all levels. Individual patients may exhibit differing degrees of calcification across different tissues and within these tissues, the way in which the mineral develops (ie, size, shape, and density) can affect tissue function and cardiovascular outcomes. Therefore, therapeutic approaches targeted to calcification will likely require treatments tailored based upon a particular patient’s specific presentation of mineral. To demonstrate how calcification uniquely affects function depending on location and morphology, we begin with a discussion of the location-specific prevalence of cardiovascular calcification. We then provide separate discussions of the implications of calcification in aortic and mitral valve tissues and intimal and medial layers of the vascular wall. Finally, we discuss the therapeutic implications of modulating mineral considering the effects of differing presentations of calcification and the associated cardiovascular risk.
Location-Specific Prevalence of Cardiovascular Calcification in CKD
In predialysis and late-stage CKD patients, calcification of the coronary artery (64%–77%34–36), abdominal aorta (49%–93%37–40), superficial femoral arteries (91%41,42) and aortic valve (27%–100%35,39) is common.43 Only 7% of dialysis patients with abdominal artery calcification (AAC) had no CAC.40
Plain radiography and ultrasonography in CKD patients on dialysis revealed a mixed pattern of intimal and medial calcification in the common carotid arteries in most patients (53%), while 10% and 24% had isolated medial and intimal calcification, respectively.44 Native radiograms of the pelvis, on the contrary, revealed only 8% with mixed arterial calcification and 27% to 32% and 29% to 45% with isolated medial and intimal calcification.45,46 Medial calcification was observed in young and middle-aged patients who did not have conventional atherosclerotic risk factors and was closely related to duration and adequacy of dialysis, calcium-phosphate disorders, and high serum triglycerides.44–46 The effect on the medial vessel layer may represent a CKD-specific response since calcification of the tunica media rarely occurs in individuals with physiological kidney function under the age of 60 years.47 However, accelerated plaque formation in patients with CKD also reflects increased calcification of the tunica intima, exacerbating remodeling observed in the general population. Indeed, CKD kidney transplant patients with extensive medial calcification detected by histology in epigastric artery biopsies had significantly higher CAC scores.48 Furthermore, calcium in aortic valves and the coronary arteries is more extensive in adults with CKD caused by atherosclerotic renal disease than in patients with nonatherosclerotic causes of CKD,35 suggesting that the calcification process might involve different factors compared with the general population, with a CAC prevalence of 50% in men and women aged 45 to 84 years.49 Even children and young adults on dialysis showed CAC evidence of 20% and 59% to 64%, respectively, indicating that calcification of the coronary artery begins at an early stage even in the absence of traditional cardiovascular risk factors (like elevated blood pressure, male sex, and the presence of T2D).50–52 Studies are needed to better define the temporal association between declining renal function, the appearance and growth of calcification across various cardiovascular beds, the presence of other confounding risk factors, and cardiovascular morbidity.
Calcification of the Cardiac Valves
Heart valve disease mechanisms and resultant pathological remodeling responses are reviewed thoroughly elsewhere.53–55 Here, we focus on the relevant pathologies observed in CKD and the implications for developing new therapeutic strategies. The 4 cardiac valves maintain unidirectional blood flow through both pulmonic and systemic circulation. The unique extracellular matrix structure of thin membranous tissues at the atrioventricular interfaces and the cardiac outflows allows the valves to open and close as pressures change over the course of the cardiac cycle. These thin structures—known as leaflets or cusps—and the muscular annuli from which they protrude must remain compliant enough to allow blood to flow when the valves open and to prevent reverse blood flow when the valves close.
CKD associates with calcification of the aortic valve leaflets and mitral valve annuli.20 In both cases, increased stiffness of the valve apparatus negatively impacts hemodynamics. Once mineralization begins, alterations in the local biomechanics around the hard mineral inclusion can cause neighboring cells to adopt pathological phenotypes,56 resulting in a feedforward mechanism. As mineral deposition progresses to the point where leaflet motion is restricted, calcific aortic valve disease (CAVD) leads to aortic stenosis, restricting blood flow from the heart during systole and resulting in significantly increased cardiac afterload (Figure 1).57 To adapt and maintain cardiac output, the muscle of the left ventricle hypertrophies. Ultimately, however, left ventricular decompensation results in heart failure. Similarly, mitral annular calcification can obstruct blood flow into the left ventricle during diastole, increasing filling pressure and altering cardiac function.58 Although not as well studied as CAVD, the clinical associations between increased mitral annular calcification and worsening cardiovascular morbidity and mortality are evident.59
In a study of 204 CKD patients stages 2 and 3, >30% of the study participants had detectable aortic or mitral valve calcification,60 and other studies have observed even higher rates of aortic valve abnormalities.20 Declining renal function accelerates mineral deposition in these tissues, with severe valvular dysfunction observed in patients significantly younger than the general population.61 Transcatheter approaches have significantly improved outcomes in patients with valvular disease.62 Transcatheter aortic valve replacement (TAVR) procedures can restore physiological cardiac output in patients with CAVD. Similarly, transcatheter mitral valve repair and replacement procedures can reestablish appropriate hemodynamics between the left atrium and ventricle. The presence of calcific mineral, however, can complicate interventions to alleviate the conditions. In TAVR, the replacement valve expands, forcing the diseased aortic valve leaflets against the annulus between the left ventricle and the aorta. As leaflet stiffness increases in severe CAVD, the force required to open the stenosed aortic valve increases. Further, the asymmetric distribution of minerals across the 3 leaflets can result in improper placement of the TAVR valve, leading to perivalvular leakage and necessitating further procedures.63 Similarly, the presence of mitral annular calcification can complicate transcatheter repair or replacement procedures.64
Given the noted positive correlations between valvular calcification and cardiovascular morbidity and the associated complications for minimally invasive interventions, the ultimate therapeutic goal is to minimize the amount of mineral present in the valve apparatus. Once calcification begins in each tissue, however, reversibility and recovery to healthy tissue homeostasis remain unclear. Calcification does not appear as mineral dispersed within the otherwise normal tissue. Instead, calcification most often coincides with—or perhaps follows—other mechanisms of extracellular matrix remodeling, including fibrotic collagen accumulation and elastic fiber degradation.65 In some cases, calcification may represent a final effort to maintain tissue integrity. Therefore, reversing late-stage calcification could exacerbate valvular dysfunction. Prophylactic measures to prevent valvular calcification are also currently unfeasible. Though our mechanistic understanding of valvular calcification has improved,66 no risk calculators exist to accurately predict the onset and progression of disease in an individual patient. An appropriate strategy for developing noninvasive therapeutics will likely require treatment of patients during a small therapeutic window in the early disease stages.
Since no noninvasive therapeutics currently exist for CAVD, clinicians have traditionally taken a conservative approach to CAVD management, waiting until valve function becomes severely diminished before referring patients for replacement. This conservative approach, however, can lead to cardiac and vascular remodeling. Further, worsening aortic stenosis also associates with decreased renal function.67
Patients with moderate CAVD (ie, not severe) have a >50% all-cause 5-year mortality rate,68 and the combination of CAVD-induced aortic stenosis and CKD requiring hemodialysis further increases mortality rates.69
Recent studies have shown better long-term outcomes when valve replacement is performed at earlier stages of CAVD in the general population.70–72 Given the challenges associated with diagnosing CAVD at the earliest (ie, reversible) stages, noninvasive therapeutics may instead focus on delaying disease progression to prolong the window during which an intervention such as TAVR leads to optimal outcomes. Of note, TAVR and the associated improved valve function can also lead to improved renal function, further demonstrating the interaction between the heart and kidney.73,74 Though TAVR can improve long-term renal function, patients with CKD exhibit worse prognosis following TAVR procedures, including in-hospital mortality and bleeding events,75–78 and can associate with acute kidney injury immediately following the procedure.79 More studies are needed to better understand the most appropriate timing of TAVR intervention in the general population but especially in patients with CKD. Delaying the need for intervention is especially relevant in younger patients with CKD. The durability of the bioprosthetic valves currently used in TAVR is still under investigation, but the life span may be shortened in calcification-prone individuals with CKD. Therefore, slowing calcification progression in CKD patients with CAVD could improve the efficacy of current therapeutic approaches to improve cardiovascular and renal health.
Medial Calcification
CKD patients most often exhibit widespread calcification of the tunica media. CKD patients with no detectable vascular calcification have 8-year survival rates of ≈90% compared with 50% survivability in age-matched patients with medial calcification.45 Unlike the mechanisms of CAVD and atherosclerotic calcification (discussed below), medial calcification appears uniformly distributed along elastic fibers and does not generally coexist with lipid and inflammatory-driven remodeling mechanisms observed in atherosclerosis. Instead of narrowing the vessels, medial calcification stiffens them, which increases pulse wave velocity, causes the pulse wave to reflect earlier, and increases cardiac afterload, predisposing to heart failure (Figure 1). The lack of arterial elasticity may also convey greater pulse pressure leading to arteriolosclerosis with resulting vascular insufficiency and tissue damage. Of note, factors other than impaired kidney function can also lead to medial calcification, for example, genetic disease (reviewed elsewhere80).
Individuals with CKD also exhibit peripheral vascular dysfunction, and vascular calcification can occur in peripheral arteries.81–83 Medial calcification is the most common pathological feature observed in the peripheral arteries of patients with limb ischemia and associates with adverse outcomes.84–86 Serious cases of CKD-induced peripheral artery disease can result in limb ischemia, necessitating amputations,83,86 and approximately one-quarter of these patients die within a year of diagnosis.81 Even without peripheral mineral deposition, calcification in larger arterial beds may alter blood flow patterns and perfusion throughout the systemic circulation. However, the relationship between calcification in large, central arterial beds and peripheral blood flow patterns remains unclear. Like valvular calcification, once significant calcification develops, the potential reversibility of mineral deposition also remains unclear; therefore, ongoing clinical studies seek to mitigate vascular calcification in early stages of CKD with the goal of improving cardiovascular outcomes.
The clinical realization of therapeutic approaches will require strategies to detect early signs of vascular remodeling and identify patients at risk for future morbidity. The gold standard for detecting vascular calcification relies on computed tomography–based imaging, but this technique is not well-suited for longitudinal monitoring of vascular calcification due to lack of access, associated expenses, and inherent radiation risks.87 Individuals from underrepresented and underserved populations have disproportionately worse outcomes from peripheral artery disease due to an underreporting of symptoms and lack of access to adequate care.88
Similar to the therapeutic goals for valvular calcification, therapeutic approaches to slow mineral deposition could decrease CKD-associated morbidity and mortality. However, unlike CAVD, the lack of clinically viable management options (such as TAVR) currently means that delaying the progression of mineralization may only postpone rather than reduce future morbidity. In the absence of other available treatments, slowing medial mineral formation along with the management of other cardiovascular risk factors could reduce the overall morbidity and enhance the quality of life in individuals with CKD.
Atherosclerotic Intimal Calcification
In the absence of other risk factors, medial calcification predominates in patients with CKD. However, individuals with CKD often exhibit signs of metabolic syndrome, including obesity, hypercholesterolemia, elevated blood sugars, and hypertension.89 These risk factors predispose individuals to atherosclerosis with subsequent fibrocalcific remodeling of the atherosclerotic plaque leading to plaque calcification (mechanistic details reviewed elsewhere).90 Like other repair mechanisms in adult tissues, infiltrated macrophages stimulate local cells to produce fibrotic collagen and bone-like calcification to stabilize the plaque structure. However, chronic inflammation results in maladaptive repair responses, which destabilize plaques.91 Failure of the collagenous fibrous cap contributes to atherosclerotic plaque rupture—the leading cause of death globally.
Classically, plaque vulnerability associates with low collagen content in the fibrous cap, which compromises its tensile strength.92 Computational studies, however, highlight the presence of destabilizing microcalcifications (≈5 µm), smaller than the resolution limits of traditional clinical imaging modalities, in the cap of vulnerable plaques as a determinant of their biomechanical failure.93,94 Coronary calcium scores predict acute atherosclerotic events better than traditional risk calculators.95 Microcalcifications promote high-stress accumulation within the fibrous cap extracellular matrix as a result of a significant material modulus mismatch between the stiff mineral and the nearby hyperelastic collagen,93 which can result in plaque instability (Figure 1). Data from the MESA study revealed that spotty calcification associates with higher rates of cardiovascular morbidity.96 While the spotty calcifications are larger than plaque-destabilizing microcalcifications, they may suggest ongoing plaque remodeling, including the presence of microcalcifications and a lack of stability.
Current medical practice cannot reliably predict whether or when an acute atherosclerotic rupture event may occur in individual patients. Therefore, physicians use population-based guidelines to assess patient risk and generally prescribe interventions designed to prevent cardiovascular events in high-risk individuals. The first course of action is often to advise lifestyle changes, such as diet, exercise, and smoking cessation, to improve general cardiovascular health.97 Patients are also generally prescribed lipid-lowering statins. Due to their overall efficacy, statins are now among the most prescribed class of drugs in the United States.
The beneficial effects of statins go beyond their lipid-lowering actions. Statin treatment causes macrophages to increase the release of the inflammatory cytokine IL-1β, which stimulates vascular smooth muscle cells to adopt a bone-like osteogenic phenotype.98,99 The resultant mineral that forms may be associated with developing more stable plaques,100 underscoring the fact that this treatment does not immediately reverse existing atherosclerosis but stabilizes existing plaques (Figure 1). This means that the immediate therapeutic goals of atherosclerosis (ie, increasing mineralization to stabilize plaques) are diametrically opposed to the antimineralization strategies required for beneficial effects in valvular and medial calcification. In some cases, delaying the progression of calcification could promote less stable plaque morphologies. As needed anticalcification treatments reach clinical realization, large-scale studies will be required to monitor potential adverse acute events in subsets of patients.
Despite the lack of definitive data, it is difficult to envision that even large intimal calcification is not detrimental to cardiovascular health. Though large calcifications stabilize plaques in the short-term, continued calcification growth can also induce vessel stenosis, reduced blood flow, and elevated systemic resistance. The presence of significant minerals can also stiffen plaques to the point where interventional strategies such as angioplasty and stenting become infeasible. In these cases, the only viable strategy may be invasive bypass procedures, which are associated with poor outcomes in frail patients and suffers from high restenosis rates.101 A novel minimally invasive therapeutic option—intravascular lithotripsy—fractures mineral deposits in severely calcified plaques in coronary and peripheral vessels through endovascular emission of acoustic waves, facilitating lesion dilation.102 However, this technique only removes the mineral physically and does not address the mechanisms that lead to calcification. The ideal therapeutic would induce atherosclerotic plaque regression, including resorption of calcification, to return the arterial wall to its physiological homeostasis. Whether this can be accomplished without causing a highly vulnerable intermediate state with low collagen and microcalcifications remains unclear.
Bone-Vascular Axis in CKD: Bone-Like Remodeling in the Cardiovascular System
Patients with CKD not only experience cardiovascular disease but also disturbed mineral and bone metabolism, referred to as CKD–mineral and bone disorder. A meta-analysis supported the epidemiological association between bone metabolism and cardiovascular disease, showing a 1.65-fold increased risk for coronary artery disease with low bone mineral density (BMD).103 Also, in 1957 predialysis CKD patients, low hip BMD was associated with cardiovascular events.104 Patients with hip fractures are at increased risk for stroke and myocardial infarction for up to 1 year.105 Whether this bidirectional link reflects unrelated coexisting age-related conditions is under debate. However, biological and epidemiological evidence supported a potential causal link between low BMD and cardiovascular mortality and morbidity that cannot be explained by age alone. Epidemiological studies, including older women with postmenopausal osteoporosis,106,107 demonstrated an association between cardiovascular calcification and osteoporosis—the calcification paradox. For individuals with normal kidney function, the link between BMD and valvular and vascular calcification is weak or not evident.107–109 However, an inverse association between skeletal and cardiovascular mineral in individuals with impaired kidney function is reported in several studies, which measured calcification across vascular beds (abdominal aorta, coronary artery, superficial femoral artery, and epigastric artery) by dual-energy X-ray absorptiometry (DXA), computer tomography, and histology and bone parameter measurements (BMD of femur, femoral neck, and spine by DXA; bone formation by bone biopsy; vertebral volumetric BMD by computed tomography) in predialysis CKD patients, late-stage CKD patients on dialysis, and kidney transplant patients.
In predialysis patients, the femoral T score associated negatively with calcification in the superficial femoral arteries but not the abdominal artery.41 In late-stage CKD patients and after kidney transplant, an independent inverse relationship between AAC and lumbar vertebral volumetric BMD or BMD at the proximal femur has been reported,38,110 whereas in CKD patients (mixed stages), AAC was inversely associated with BMD at the femoral neck and spine but not femur and total BMD.111 In young hemodialysis patients, AAC associated inversely with trabecular bone score in the lumbar spine but not other DXA-derived BMD parameters.37 There is conflicting evidence that the lumbar spine BMD assessed by DXA may reveal unreliable data in CKD patients due to extensive AAC.112,113
The association between skeletal mineralization and CAC is also evident. In predialysis CKD patients, low bone formation assessed by iliac crest bone biopsy, low volumetric BMD, and low BMD at the total hip and femur neck but not the lumbar spine were inversely associated with CAC score and progression.36,104,114 In late-stage CKD patients, lower BMD of the leg subregion associated with higher CAC score.48 Another study found this link only in female patients.115 Of note, the association of high CAC with low volumetric BMD is stronger than those based on low BMD by DXA.116 The presence of medial vascular calcification in epigastric artery biopsies from late-stage CKD kidney transplant patients also associated with low BMD.48,117 The link between aortic valve calcification and skeletal mineralization in CKD patients was demonstrated for particular bone sites (femur and femoral neck) and total BMD in only one study.111
Emerging data support observations that cardiovascular calcification and bone mineralization share common cellular and molecular mechanisms (reviewed elsewhere118). Bone-like cells were identified in areas of calcified vascular tissue, and genetic mouse models support the role of bone genes in vascular calcification.119 Cells with an osteoblast phenotype, which express osteogenic markers and build a mineralized matrix,120 and multinucleated cells expressing osteoclast markers like tartrate-resistant acid phosphatase and carbonic anhydrases were identified in cardiovascular tissue close to the calcified area.121–124 However, in vivo proof of vascular osteoclasts capable of mineralized matrix resorption is still lacking. Histological analyses revealed that actual bone-structured matrix—the osteoid metaplasia—was most common in the femoral (45%) and infrapopliteal artery plaques (16%),125 but it was also found in carotid endarterectomy plaques (13%), which was directly correlated with the presence of sheet-like calcifications and the absence of ulceration and hemorrhage.126 Calcified valves from cardiac valve replacement showed mature lamellar bone with hematopoietic elements and active bone remodeling in 13% of the cases.127 However, no study has been conducted solely on tissue from patients with CKD, raising the possibility that the rate of vascular bone formation is actually higher.
The progressive bone loss in CKD may also play a direct role in cardiovascular mineral deposition—a theory known as the mineral redirection hypothesis. Excess bone resorption may cause the release of calcium and phosphate, which, due to renal excretion failure, are then redirected from the bone matrix to the arterial wall or valve leaflet, promoting cardiovascular calcification.128 Indeed, excess bone resorption contributes to hyperphosphatemia.129 Indirect evidence from rodent CKD models suggests that increasing bone formation reduces phosphate levels and vascular calcification and that arteries participate in acute phosphate homeostasis by depositing significant amorphous phosphate.130,131 There is no proof of this causal association hypothesis in humans.
Despite their commonalities, recent studies have shown that cardiovascular calcification and skeletal tissue mineralization are not completely analogous. Cells in calcified cardiovascular tissues may express osteoblast or chondrocyte markers, but the nature of the extracellular matrix, tissue remodeling mechanisms,132–135 and perhaps even composition and crystallinity of the minerals136–139 may differ significantly from that of bone and cartilage.
Nevertheless, drugs treating disturbed bone homeostasis and bone cell function may have anticalcifying effects on the cardiovascular system in patients with CKD. Osteoporosis treatments aim to reduce bone resorption (antiresorptive) or increase bone mineralization (anabolic) to halt disease progression.140 However, more studies are needed to better understand the similarities and differences in skeletal and vascular mineralization to determine how therapeutics targeted to pathologies in one tissue influence remodeling in the other.
Therapeutic Modulation of Mineral Formation, Growth, and Composition and Its Impact on Cardiovascular Outcome
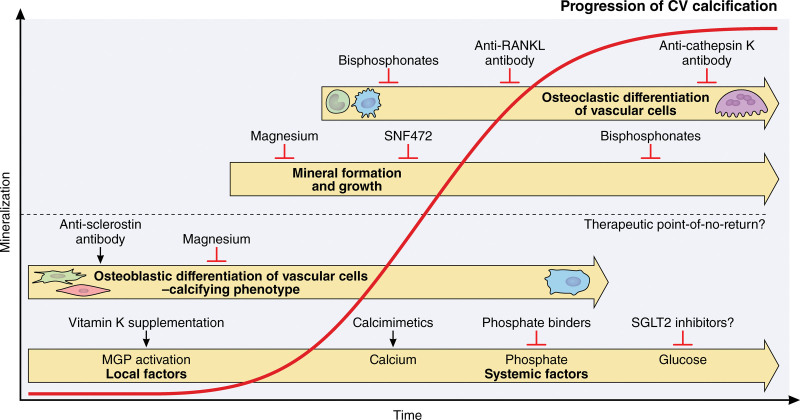
A disturbed mineral homeostasis driven by the kidney and bone pathologies mainly drives cardiovascular calcification in patients with CKD. Therefore, medication that restores mineral homeostasis or improves pathological bone remodeling by cellular targeting may reduce mineral deposition into the cardiovascular system (Figure 2). Here, we review recent and ongoing studies into mineral interactors as therapeutics for CKD-induced valvular and vascular calcification.
Figure 2.
Contributing cellular and noncellular factors to the progression of cardiovascular (CV) calcification and potential therapeutic options for interference. CV calcification starts with changes in local and systemic factors, initiating the osteogenic differentiation of vascular cells to a calcifying phenotype capable of inserting minerals in the extracellular matrix. Calcification progresses with mineral nucleation and growth exponentially to form larger macrocalcification. Once the mineral is formed, hematopoietic lineage cells may differentiate into osteoclast-like cells, but there is no in vivo evidence of mineral-resorbing activity. The therapeutic window with the available medications might be before a point of no return—a time point in the calcification progression when mineralization cannot be halted and irreversible damages the vascular tissue. The indicated drugs are matched to the target mechanism, and the position does not indicate a possible treatment time related to the calcification progression. MGP indicates matrix Gla protein; RANKL, receptor activator of NF-κB ligand; and SGLT2, sodium-glucose cotransporter 2. Illustration credit: Sceyence Studios.
Phosphate Binders
The pathological mineral that forms in diseased heart and vascular tissues resembles a disorganized form of the same hydroxyapatite material observed in bone. When discussing calcification, it is common to refer to the mineral present as calcium. While calcium is a crucial component of the mineral structure, and is the basis for the calcification nomenclature, the crystal unit consists of 5 calcium atoms and 3 phosphate molecules with 2 hydroxide ions. Given the abundance of calcium ions throughout the extracellular environment, the presence of sufficient free phosphate is the rate-limiting factor for mineral formation. To prevent spontaneous ectopic calcification in normal conditions, phosphate availability and the ability to complex with calcium are tightly controlled by various inhibitors.
Healthy kidneys play an important role in helping control phosphate levels, including through the direct removal of excess phosphorous. Increased levels of serum phosphate independently associate with cardiovascular morbidity and continued worsening of renal function.141 Hyperphosphatemia correlates with increased vascular calcification, and studies suggest that mineral formation in the arterial wall can serve as a reservoir to remove circulating phosphate and prevent systemic toxicity.131 Serum proteins such as fetuin-A and albumin can chelate nascent calcium-phosphate complexes in circulation, preventing rapid mineral growth. However, as CKD progresses, the resultant calciprotein particles that form from the serum chelation can induce inflammation and promineralization responses in vascular tissues.142 Novel techniques measuring the maturation of calciprotein particles in patient serum have demonstrated the potential to predict valvular and vascular disease progression.143–145
With the premise that elevated circulating phosphate predicts—and likely contributes to—CKD-induced morbidity, the recently completed IMPROVE-CKD trial (Impact of Phosphate Reduction on Vascular End Points in chronic kidney disease) assessed the effect of phosphate reduction in improving vascular outcomes.146 In this prospective trial, 278 individuals with stage 3b or 4 CKD and serum phosphate >1 mmol/L were randomized into either a treatment group that received the phosphate binder lanthanum carbonate or a placebo control group. At the 8-year end point, the groups did not exhibit significant differences in aortic calcification or serum phosphate. There was also no difference in the number of adverse events between the groups. The lack of differences in serum phosphate suggests that the binder used in the study could not significantly impact the increases caused by renal failure and, consequently, did not exhibit a benefit in vascular outcomes.146 Of note, 2 other, much smaller, placebo-controlled randomized controlled trials (RCTs) with lanthanum carbonate, sevelamer, and calcium acetate also did not observe an effect on vascular calcification at different sites in patients with CKD,147 whereas the majority of RCTs revealed attenuated calcification progression of noncalcium-based phosphate binders when compared with calcium-based phosphate binders.147
Bisphosphonates
Rather than targeting phosphate directly, therapeutic strategies could seek to alter interactions between calcium and phosphate to inhibit or modulate mineral formation. Pyrophosphate, 2 phosphate ions with a P-O-P linkage, is a potent calcification inhibitor. In both pathological and physiological mineralization processes, hydrolysis of pyrophosphate by tissue nonspecific alkaline phosphatase converts this inhibitor into 2 free phosphate groups that can promote mineral formation. Bisphosphonates bind calcium-phosphate mineral and act as a nonhydrolyzable analog of pyrophosphate to prevent continued mineral growth and maturation.148 In bone, bisphosphonates are antiresorptive agents with a high affinity to the mineralized bone matrix close to the resorption lacuna. Nonnitrogenous bisphosphates, like etidronate, are metabolized to a cytotoxic ATP analogue, while nitrogenous bisphosphates, like alendronate and zolendronate, inhibit the farnesyl pyrophosphate synthase—a key enzyme in the mevanolate pathway. The action of both types of bisphosphonates primarily promotes osteoclast apoptosis but can also promote osteoblast differentiation.149
Retrospective data from clinical trials have yielded contradictory results regarding the interaction between bisphosphonates and cardiovascular calcification. In CAVD, some analyses demonstrate that bisphosphonates attenuate disease progression,150,151 whereas others do not. Additionally, in an analysis of women in the MESA cohort, bisphosphonate use correlated with a lower rate of cardiovascular calcification in older (>65 years of age) patients but a greater rate in younger patients.150 The effect of bisphosphonate use on cardiovascular calcification was greater than that which could be attributed to differences in BMD, indicative of a direct interaction between bisphosphonate therapy and the cardiovascular system. Zolendronate did not affect abdominal aortic calcification in postmenopausal women with osteoporosis.152 Small trials assessed the effect of bisphosphonates on vascular calcification in patients with CKD. In dialysis and kidney transplant recipients, etidronate, alendronate, and risedronate attenuated the calcification progression at different vascular sites, while there was no effect of alendronate in patients with CKD stage 3 and 4.147 Further, bisphosphonate treatment inhibited calcification of explanted rat aortas, indicating that bisphosphonates can act directly on vascular tissue, independent of bone metabolism.153 It is unclear whether additional cellular-level mechanisms contribute to the bisphosphate effect seen in cardiovascular tissue because evidence for a direct role of osteoclasts in cardiovascular calcification is limited.
While bisphosphonate effectively reduces fracture risk, there are additional conflicting data on the cardiovascular safety of bisphosphonates. In women with CKD, bisphosphonates decreased mortality risk in patients without a history of cardiovascular disease,154 but certain analyses indicated a paradoxically increased risk of death in patients with a history of a cardiovascular event.155 All-cause mortality was reduced by 28% in patients receiving zolendronate compared with supplementing vitamin D and calcium.156 However, cardiovascular outcome was not the primary outcome in the clinical trial. A meta-analysis of 38 trials could not support the cardioprotective effect of zolendronate and other bisphosphonates.157 Compared with denosumab, zolendronate even increased the risk of atrial fibrillation during the first year of use.158 Other clinical data for zoledronic acid, risedronate, and alendronate indicate no significant differences from placebo in the overall incidence of atrial fibrillation.159 Our recent studies investigated the effects of bisphosphonate treatment on vascular calcification in vitro and in a hyperlipidemic mouse model of atherosclerosis.160,161 Using a collagen hydrogel-based model of calcification formation that mimics aspects of the atherosclerotic fibrous cap, we showed that bisphosphonate treatment given after mineralization begins alters the shape and size of microcalcifications that form as the mineral grows.161 In atherogenic mice, bisphosphonate treatment given before mature plaque formation resulted in larger, more uniform calcifications. Bisphosphonates given to the mice after early signs of mineralization are observed in the plaques, leading to less uniform calcification distribution with a broader range of sizes.160 These data may, in part, explain some of the contradictory clinical outcomes observed with bisphosphonates, where the treatment may result in stable plaque morphologies in some individuals but promote destabilizing microcalcifications in others.
Further studies are needed to clarify the therapeutic effects of bisphosphonates in valvular and vascular calcification, especially in the context of CKD. However, due to the low-grade nephrotoxicity of bisphosphonates, there is a potential concern in patients with impaired kidney function, mainly when administered intravenously,162 but also oral bisphosphonates are associated with the risk of acute kidney injury—a cardiovascular risk factor.163
MGP and Vitamin K Supplementation
First identified in 1983 within the organic matrix of bone,164 MGP (matrix Gla protein) contains the vitamin K–dependent γ-carboxyglutamic acid, which has a high affinity for calcium ions. Subsequent studies demonstrated the distribution of MGP within early development of mineralized tissues,165 suggesting a potential role in directing mineral formation and organization. A decade later, MGP was identified in calcified regions of atherosclerotic plaques166 and is now well recognized as a mineralization inhibitor that can bind developing apatitic mineral.167,168 Under physiological osteochondrogenic conditions, the presence of MGP likely ensures that mineralization proceeds in a controlled, structured manner.
The inactive, dephosphorylated uncarboxylated form of MGP independently predicts both atherosclerotic and medial calcification levels in patients with CKD169,170 and associates with cardiovascular risk in the general population.171,172 When active, the phosphorylated and γ-carboxylated form of MGP chelates calcium-phosphate complexes to inhibit nucleation and growth of hydroxyapatite.173 Both, the conserved glutamic acid residues and N-terminal conserved serine residues contribute to its antimineralization function.174,175
Vitamin K is essential for the activation and mineral inhibitory effects of MGP, and individuals with CKD often exhibit vitamin K deficiency,176 likely contributing to the decreased active MGP in these patients and subsequent vascular calcification. The elevated thromboembolic risk in patients with CKD requires anticoagulation. Warfarin—a commonly used drug—interacts with vitamin K to block the vitamin K–dependent step in clotting factor production but also diminishes posttranslational carboxylation of MGP. Therefore, targeting the vitamin K pathway, as an essential cofactor for MGP carboxylation, may represent a promising treatment strategy.
Recent results from a randomized prospective clinical study demonstrated 56% lower thoracic aortic calcification and 68% lower CAC in hemodialysis patients treated with 5 mg vitamin K1 3× per week compared with patients only receiving standard care.177 Trending differences in valvular calcification were also observed but did not reach statistical significance. The observed differences in calcification associated with significantly lower plasma levels of dephosphorylated uncarboxylated form of MGP, with the vitamin K1–treated group exhibiting only 36% of this inactive form compared with the untreated patients after 4 weeks and persisted to the reported end point at 18 months. While these data demonstrate the potential therapeutic benefits of vitamin K1 supplementation in patients with CKD, larger studies are needed to fully test the efficacy and effects on cardiovascular outcomes. Only 40 patients were included in the final end point analyses of the study.177 Of note, supplementation of vitamin K2 in the form of MK-7 (menaquinone-7) in patients with CKD failed to show any beneficial cardiovascular effects (like calcification progression, pulse wave velocity, or cardiovascular events),178,179 explained by a disturbed lipoprotein-mediated MK-7 transport in patients with CKD.180
Future studies will need to more fully examine whether decreased calcification proves beneficial in reducing cardiovascular events across the wide spectrum of patients with CKD.
SNF472
A novel experimental drug SNF472 selectively binds hydroxyapatite,181 and early clinical trial data demonstrate tremendous potential in preventing ectopic mineral formation in individuals with CKD.182 SNF472 binds to hydroxyapatite and inhibits further crystal formation without affecting free calcium ions. In vitro, 1 µM SNF472 inhibited calcification of vascular smooth muscle cells by 67%.183 The CaLIPSO trial (Cal for calcium and ipso meaning the item itself) randomized 274 patients with end-stage kidney disease into 3 groups, 2 different dosages of SNF472, and a placebo group, for 12 months during hemodialysis.182 Combined data from the patients given the 2 SNF472 dosages exhibited calcium volume scores of 11% and 14% in the coronary arteries and aortic valve, respectively, compared with values of 20% and 98% in the placebo group. No differences in thoracic aortic calcification were observed (23% in the treated groups versus 28% in the placebo group).
These first reports suggest that SNF472 is a promising therapeutic candidate to prevent ectopic calcification—especially in the aortic valve—in patients with CKD. If the treatment can more specifically target CKD-induced CAVD without major effects to arterial mineral, SNF472 may avoid deleterious outcomes caused by elevated atherosclerotic plaque vulnerability. However, larger studies of longer duration are required to test effects on cardiovascular outcomes and potential off-target effects. Though no significant differences in bone fracture were observed, a subanalysis of the CaLIPSO study showed that all 3 groups exhibited decreased BMD over the 12 months, and the reduction appeared larger in patients receiving the highest dosage of SNF472.184 The current formulation of SNF472 limits delivery to intravenous administration, which works well for patients receiving dialysis but may limit broader application to nondialysis patients.
Magnesium
As mentioned, the calcific mineral observed in diseased valvular and vascular tissues consists of poorly crystalline hydroxyapatite. Another form of calcific mineral also appears in these tissues, a highly crystalline form of magnesium-containing whitlockite.139,185 During crystallization, magnesium ions can substitute for calcium, favoring the formation of whitlockite over hydroxyapatite. The relative abundance of these 2 minerals depends on the levels of each of the ions, and high magnesium concentrations can completely prevent hydroxyapatite formation.186 Commonly found in pathological human dental calculus,186 whitlockite appears ubiquitous throughout calcified cardiovascular tissues but is absent from bone.187 Unlike hydroxyapatite, which appears to grow into larger structures as disease progresses, whitlockite appears confined to crystalline spherical structures139; however, its role in physiology and disease remains unclear. Interestingly, serum magnesium demonstrates an inverse relationship with vascular calcification.188 The kidneys help regulate magnesium homeostasis, and although many patients with CKD exhibit hypermagnesemia, a significant subset of patients with proteinuria develop hypomagnesemia.189 Perhaps as a result, arterial calcification in patients with CKD appears to favor hydroxyapatite over whitlockite, whereas the relative abundance of these minerals is similar in individuals with normal renal function.190 Based on its ability to prevent the rapid formation of hydroxyapatite, as observed in the valves and vasculature of patients with CKD, magnesium supplementation has recently been suggested as a potential therapeutic strategy.191
In an open-label clinical study of 96 predialysis CKD patients, magnesium oxide supplementation suppressed coronary artery mineral growth with a calcified volume of 11% compared with 40% for the control group.192 These data suggest a beneficial effect on calcification, but given the uncertain effects of whitlockite crystals, outcome studies are needed to identify patients who would benefit from magnesium-based therapies.
Calcimimetics
Calcimimetics bind to the parathyroid calcium-sensing receptor, which reduces parathyroid hormone release to maintain calcium and phosphate homeostasis and thereby mimic the action of calcium in the tissue. In hemodialysis patients with secondary hyperparathyroidism, cinacalcet reduced the rate of clinical fractures193 and attenuated vascular and valvular calcification194 but did not affect all-cause mortality and cardiovascular events.195 However, a post hoc subgroup analysis revealed a decreased risk of death and major cardiovascular events in older, but not younger, patients.196 Analysis from a health care utilization general CKD cohort with secondary hyperparathyroidism in Sweden showed that cinacalcet reduced cardiovascular events but not all-cause mortality.197
Anti-RANKL Antibody: Denosumab
Rather than targeting mineral interactions directly, targeting mineral-involved cells might be another option to prevent cardiovascular calcification. RANKL (receptor activator of NF-κB ligand) drives osteoclast differentiation, fusion, activation, activity, and survival.198 The central role of RANKL in osteoclast differentiation led to the development of a neutralizing antibody denosumab that inhibits bone resorption, reduces fracture risk,199 and does not affect adverse cardiovascular events during 3-year treatment.200 Denosumab did not affect the progression of aortic calcification in postmenopausal women200 or aortic valve calcification in patients with CAVD.201 Smaller studies investigated the effect of denosumab in hemodialysis patients with low bone mass and showed reduced progression of CAC202 and aortic arch calcification203 after 6- and 30-month treatment and no effect on CAC after 12 months of treatment.204
Denosumab-induced hypocalcemia has been observed in late-stage CKD contraindicating its use in patients with CKD.205 A post hoc analysis of long-term safety in individuals with mild-to-moderate CKD did not reveal hypocalcemia.206 High age, low baseline calcium serum, and 25 hydroxyvitamin D levels were identified as risk factors for hypocalcemia in patients with CKD taking denosumab.207,208 As a result, denosumab treatment in patients with impaired kidney function should be closely monitored by a multidisciplinary integrated care and supplementation of calcium and calcitriol.207,209
Unexplored Therapeutic Modulators of Mineral Handling and Vascular Calcification in Chronic Kidney Disease
Anti-Cathepsin K Antibody: Odanacatib
Acidification by osteoclast mobilizes hydroxyapatite crystals from the bone. It provides an optimal activity environment for proteolytic enzymes like cathepsin K.198 Cathepsin K cleaves key bone matrix proteins and plays a role in degrading the organic phase of bone during bone resorption. This knowledge led to the development of a neutralizing antibody odanacatib that reduced fracture risk in postmenopausal women but increased the risk of cerebrovascular events, causing a stop in drug development210 and, therefore, was never studied in patients with CKD.
Anti-Sclerostin Antibody: Romosozumab
Sclerostin is a glycoprotein that hampers osteogenesis. Osteocytes reduce the release of sclerostin in response to mechanical forces acting on the bone, thereby promoting the Wnt/β-catenin pathway in osteoblasts.198 Romosozumab is a sclerostin monoclonal antibody that increases bone formation and decreases vertebral fracture risk in postmenopausal women with osteoporosis.211–213 The cardiovascular safety of romosozumab is controversially discussed in the literature since ischemic cardiovascular events were observed with romosozumab compared with alendronate treatment during the first year,213 while another placebo-controlled trial did not report adverse cardiovascular events.212 A post hoc analysis of those 2 studies supported those initial findings in postmenopausal women with mild-to-moderate CKD.214 Blocking sclerostin can dysregulate cellular cholesterol homeostasis by the activation of Wnt signaling. Therefore, alendronate may ameliorate the harmful effects of romosozumab through its effects on lipid metabolism.215 Ongoing discussions in the community support the hypothesis that alendronate is cardioprotective and that romosozumab is not harmful.216
There have been no sclerostin-targeted clinical trials in patients with CKD. Treatment in this patient population should be carefully considered due to the risk of accelerated ectopic calcification shown in rodent models.217,218
SGLT2 Inhibitors
SGLT (sodium-glucose cotransporter)-2 inhibitors are a new class of medications that reduce cardiovascular events and improve renal outcomes in patients with CKD and T2D. Three large RCTs documented cardiovascular and renal protective effects of the SGLT2 inhibitors empagliflozin, dapaglizin, and canagliflozin in T2D and CKD.219–222 SGLT2 inhibitors prevent glucose reabsorption within the proximal kidney tubules and decrease blood glucose levels due to increased glucose excretion resulting in weight loss and blood pressure reduction.223 Based on this mechanism of action, SGLT2 inhibitors may affect mineral handling. SGLT2 inhibition mediated calciuria and increased phosphate reabsorption may increase parathormone and fibroblast growth factor 23 levels, respectively, causing decreased active vitamin D.224 This mechanism suggests an effect on bone metabolism supported by data from rodent models. In a nondiabetic rat model, canagliflozin and dapagliflozin induced disorders of cancellous bone microarchitecture and bone mechanical properties.225 In diabetic DBA/2J mice, canagliflozin improved blood glucose but did not affect streptozotocin-induced trabecular and cortical microarchitecture. However, in the control group, canagliflozin caused adverse effects on the femoral trabecular bone.226 In contrast, in streptozotocin-induced diabetic C57Bl/6 mice, canagliflozin increased BMD and trabecular number, while empagliflozin and dapaglizin had no effect.227
The EMPA-REG (Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients–Removing Excess Glucose) and DECLARE (Multicenter Trial to Evaluate the Effect of Dapagliflozin on the Incidence of Cardiovascular Events) outcome trials reported no effect on fracture risk and amputation rate. In contrast, in the CANVAS RCT (CANagliflozin cardioVascular Assessment Study), canagliflozin was associated with increased fractures, especially of the distal extremities, and a higher incidence of amputations of the lower extremities in patients with T2D, which could be explained by the SGLT1-inhibiting properties of canagliflozin and the older study population compared with the other 2 large RCTs.220
A later study compared patients with T2D receiving canagliflozin or glucoagon-like peptide 1 agonist as a control and found no difference in the occurrence of fractures.228 Meta-analysis on all 3 SGLT2 inhibitors revealed no association with a higher fracture incidence in non-T2D and T2D patients.229,230 However, a study with 252 diabetic nephropathy patients (CKD stage 3) reported fractures in 8% of patients in the dapagliflozin group and none in the placebo group during a 104-week follow-up.231
In patients with T2D, the SGLT2 inhibitors tofogliflozin and empagliflozin significantly improved vascular pulse wave velocity, suggesting an effect on arterial stiffness.232,233 Whether this is a consequence of reduced fibrocalcific remodeling has not yet been studied. However, reduced hyperglycemia may prevent glucotoxicity not only in the kidney but also in extrarenal organs, like the arterial wall or cardiac valves.234 A first trial to study the effect of the SGLT2 inhibitor dapagliflozin on the increased risk of heart failure in patients with aortic stenosis undergoing TAVR is ongoing.235
Conclusions
In clinical practice, vascular calcification has been regarded as a surrogate marker for disease and a factor for cardiovascular risk stratification to inform management decisions (eg, whether to begin statin therapy) but not a direct therapeutic target.236,237 Treatment of valvular and vascular calcification in CKD relies on the assumption that it is harmful and that a reduction will be cardiovascular protective. Indeed reduction/slowing of calcification in the valves can preserve function; however, if the same strategy results in more plaque-destabilizing microcalcifications in the intima, the positive valvular effects could be masked by increased risk of acute vascular events. Also, there is a lack of evidence to which extent vascular calcification must be lowered to have clinically substantial benefits on cardiovascular health. CKD patients are complex with several comorbidities, all contributing to cardiovascular calcification at different vascular sites, resulting in a patient-specific heterogeneous picture of clinical consequences. Reduction of microcalcification and macrocalcification may require different strategies. These 2 morphologies often do not exist independently and in some cases may represent part of a continuum. Treatment strategies may need tailoring to a particular patient’s calcification state, especially in patients with CKD where mineralization can change rapidly.
Several drugs to treat mineral disturbance targeting the bone and kidney show protective effects in rodent models (reviewed elsewhere238). However, thus far, no treatments have consistently reduced cardiovascular calcification and cardiovascular outcomes in patients with CKD. Case reports demonstrate that bisphosphonates and thiosulfates have been successfully used to treat rapid calcification associated with CKD in calciphylaxis patients239,240 and general calcification of infancy.241 These extreme cases demonstrate the potential of altering mineral formation for therapeutic benefit; however, more studies are needed to determine how to best interact with mineral to reduce cardiovascular risk in the general CKD population. Most studies showing reduced valvular and vascular calcification fail to demonstrate improved cardiovascular risk. The complete picture of systemic calcification at different vascular beds, including the aortic valve, is missing—most clinical trials focus on 1 to 2 vascular beds and rarely on the aortic valves.
Most studies include patients with CKD undergoing dialysis or renal transplantation—a population with severe multiorgan failure and most likely beyond stages where therapies can induce regeneration of fibrocalcific vascular tissue. The most feasible treatment option based on current knowledge is the attenuation of calcification progression. Prevention is difficult to imagine since there are no specific biomarkers and high-resolution imaging techniques for detecting the first vascular and valvular mineral depositions in CKD. Vascular and valvular mineral resorption after the point of no return may produce smaller harmful and biologically active microcalcifications that may promote inflammation (Figures 1 and 2). Therapeutics will need to balance potential harmful intermediate effects while promoting the regeneration of fibrocalcific tissue to nondiseased vessels and aortic valves.
In general, CKD trials suffer from a small patient number compared with typical cardiovascular outcome trials, and given the multiple diverse comorbidities of patients with CKD, the study population might be too heterogeneous to account for all confounders. The treatment window is another crucial factor to consider. Given the uncertainty regarding reversibility of ectopic mineralization, some otherwise effective therapies may be given too late for some patients. Imaging only detects valvular and vascular calcification when already present in a more extensive form. Most reported adverse cardiovascular events when interfering with mineral handling or treating osteoporosis are acute events, like stroke or myocardial infarction and arterial fibrillation. The underlying mechanisms are unknown.
Different susceptibilities of individual arterial regions to calcification in CKD are well recognized. There is an ongoing debate concerning the differential role of atherosclerotic intimal and medial calcification in patients with CKD.242,243 Coronary arteries in patients with late-stage CKD are characterized by increased media thickness and marked plaque calcification compared with nonrenal patients with cardiovascular disease.16 However, in early CKD stages, isolated medial calcification was also observed in coronary arteries.242 Arterial calcification assessed with standard clinical imaging techniques cannot differentiate between calcification in the tunica intima and media. In addition, the sum of calcification at different cardiovascular beds, including heart valves and myocardium, may contribute to the increased cardiovascular risk of patients with CKD. Therefore, anticalcification therapy in patients with CKD should be motivated by improving cardiovascular health without affecting bone health. The ideal development requires interdisciplinary approaches in a scientific team of cardiology, nephrology, osteology, radiology, cell and molecular biology, chemistry, and mineralogy.
Article Information
Sources of Funding
This work was supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health (1R01HL160740 to J.D. Hutcheson) and the Deutsche Forschungsgemeinschaft (German Research Foundation, Transregional Collaborative Research Centre TRR 219; project ID 322900939, project C02 to C. Goettsch).
Disclosures
None.
Nonstandard Abbreviations and Acronyms
- AAC
- abdominal artery calcification
- BMD
- bone mineral density
- CAC
- coronary artery calcification
- CAVD
- calcific aortic valve disease
- CKD
- chronic kidney disease
- DXA
- dual-energy X-ray absorptiometry
- eGFR
- estimated glomerular filtration rate
- MESA
- Multi-Ethnic Study of Atherosclerosis
- MGP
- matrix Gla protein
- RANKL
- receptor activator of NF-κB ligand
- RCT
- randomized controlled trial
- SGLT
- sodium-glucose cotransporter
- T2D
- type 2 diabetes
- TAVR
- transcatheter aortic valve replacement
For Sources of Funding and Disclosures, see page 1006.
References
- 1.Hutcheson JD, Blaser MC, Aikawa E. Giving calcification its due: recognition of a diverse disease: a first attempt to standardize the field. Circ Res. 2017;120:270–273. doi: 10.1161/CIRCRESAHA.116.310060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sage AP, Tintut Y, Demer LL. Regulatory mechanisms in vascular calcification. Nat Rev Cardiol. 2010;7:528–536. doi: 10.1038/nrcardio.2010.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Palit S, Kendrick J. Vascular calcification in chronic kidney disease: role of disordered mineral metabolism. Curr Pharm Des. 2014;20:5829–5833. doi: 10.2174/1381612820666140212194926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Matsushita K, Ballew SH, Wang AY, Kalyesubula R, Schaeffner E, Agarwal R. Epidemiology and risk of cardiovascular disease in populations with chronic kidney disease. Nat Rev Nephrol. 2022;18:696–707. doi: 10.1038/s41581-022-00616-6 [DOI] [PubMed] [Google Scholar]
- 5.Akbari A, Swedko PJ, Clark HD, Hogg W, Lemelin J, Magner P, Moore L, Ooi D. Detection of chronic kidney disease with laboratory reporting of estimated glomerular filtration rate and an educational program. Arch Intern Med. 2004;164:1788–1792. doi: 10.1001/archinte.164.16.1788 [DOI] [PubMed] [Google Scholar]
- 6.Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296–1305. doi: 10.1056/NEJMoa041031 [DOI] [PubMed] [Google Scholar]
- 7.Matsushita K, Coresh J, Sang Y, Chalmers J, Fox C, Guallar E, Jafar T, Jassal SK, Landman GW, Muntner P, et al. ; CKD Prognosis Consortium. Estimated glomerular filtration rate and albuminuria for prediction of cardiovascular outcomes: a collaborative meta-analysis of individual participant data. Lancet Diabetes Endocrinol. 2015;3:514–525. doi: 10.1016/S2213-8587(15)00040-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weiner DE, Krassilnikova M, Tighiouart H, Salem DN, Levey AS, Sarnak MJ. CKD classification based on estimated GFR over three years and subsequent cardiac and mortality outcomes: a cohort study. BMC Nephrol. 2009;10:26. doi: 10.1186/1471-2369-10-26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Budoff MJ, Rader DJ, Reilly MP, Mohler ER, 3rd, Lash J, Yang W, Rosen L, Glenn M, Teal V, Feldman HI, et al. ; CRIC Study Investigators. Relationship of estimated GFR and coronary artery calcification in the CRIC (Chronic Renal Insufficiency Cohort) study. Am J Kidney Dis. 2011;58:519–526. doi: 10.1053/j.ajkd.2011.04.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Noels H, Boor P, Goettsch C, Hohl M, Jahnen-Dechent W, Jankowski V, Kindermann I, Kramann R, Lehrke M, Linz D, et al. The new SFB/TRR219 research centre. Eur Heart J. 2018;39:975–977. doi: 10.1093/eurheartj/ehy083 [DOI] [PubMed] [Google Scholar]
- 11.Schiffrin EL, Lipman ML, Mann JF. Chronic kidney disease: effects on the cardiovascular system. Circulation. 2007;116:85–97. doi: 10.1161/CIRCULATIONAHA.106.678342 [DOI] [PubMed] [Google Scholar]
- 12.Thompson S, James M, Wiebe N, Hemmelgarn B, Manns B, Klarenbach S, Tonelli M, Alberta Kidney Disease N. Cause of death in patients with reduced kidney function. J Am Soc Nephrol. 2015;26:2504–2511. doi: 10.1681/ASN.2014070714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi Y, Jacobs DR, Jr, Shroff GR, Kramer H, Chang AR, Duprez DA. Progression of chronic kidney disease risk categories and risk of cardiovascular disease and total mortality: coronary artery risk development in young adults cohort. J Am Heart Assoc. 2022;11:e026685. doi: 10.1161/JAHA.122.026685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Matsushita K, van der Velde M, Astor BC, Woodward M, Levey AS, de Jong PE, Coresh J, Gansevoort RT; Chronic Kidney Disease Prognosis Consortium. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet. 2010;375:2073–2081. doi: 10.1016/S0140-6736(10)60674-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nichols GA, Deruaz-Luyet A, Brodovicz KG, Kimes TM, Rosales AG, Hauske SJ. Kidney disease progression and all-cause mortality across estimated glomerular filtration rate and albuminuria categories among patients with vs. without type 2 diabetes. BMC Nephrol. 2020;21:167. doi: 10.1186/s12882-020-01792-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schwarz U, Buzello M, Ritz E, Stein G, Raabe G, Wiest G, Mall G, Amann K. Morphology of coronary atherosclerotic lesions in patients with end-stage renal failure. Nephrol Dial Transplant. 2000;15:218–223. doi: 10.1093/ndt/15.2.218 [DOI] [PubMed] [Google Scholar]
- 17.Raggi P, Boulay A, Chasan-Taber S, Amin N, Dillon M, Burke SK, Chertow GM. Cardiac calcification in adult hemodialysis patients. A link between end-stage renal disease and cardiovascular disease? J Am Coll Cardiol. 2002;39:695–701. doi: 10.1016/s0735-1097(01)01781-8 [DOI] [PubMed] [Google Scholar]
- 18.Kramer H, Toto R, Peshock R, Cooper R, Victor R. Association between chronic kidney disease and coronary artery calcification: the Dallas Heart Study. J Am Soc Nephrol. 2005;16:507–513. doi: 10.1681/Asn.2004070610 [DOI] [PubMed] [Google Scholar]
- 19.Chowdhury UK, Airan B, Mishra PK, Kothari SS, Subramaniam GK, Ray R, Singh R, Venugopal P. Histopathology and morphometry of radial artery conduits: basic study and clinical application. Ann Thorac Surg. 2004;78:1614–1621. doi: 10.1016/j.athoracsur.2004.03.105 [DOI] [PubMed] [Google Scholar]
- 20.Marwick TH, Amann K, Bangalore S, Cavalcante JL, Charytan DM, Craig JC, Gill JS, Hlatky MA, Jardine AG, Landmesser U, et al. ; Conference Participants. Chronic kidney disease and valvular heart disease: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) controversies conference. Kidney Int. 2019;96:836–849. doi: 10.1016/j.kint.2019.06.025 [DOI] [PubMed] [Google Scholar]
- 21.Peng ALW, Dardari ZA, Blumenthal RS, Dzaye O, Obisesan OH, Uddin SMI, Nasir K, Blankstein R, Budoff MJ, Mortensen MB, et al. Very high coronary artery calcium (>= 1000) and association with cardiovascular disease events, non-cardiovascular disease outcomes, and mortality results from MESA. Circulation. 2021;143:1571–1583. doi: 10.1161/Circulationaha.120.050545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen J, Budoff MJ, Reilly MP, Yang W, Rosas SE, Rahman M, Zhang XM, Roy JA, Lustigova E, Nessel L, et al. ; CRIC Investigators. Coronary artery calcification and risk of cardiovascular disease and death among patients with chronic kidney disease. JAMA Cardiol. 2017;2:635–643. doi: 10.1001/jamacardio.2017.0363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Olyaei A, Steffl JL, MacLaughlan J, Trabolsi M, Quadri SP, Abbasi I, Lerma E. HMG-CoA reductase inhibitors in chronic kidney disease. Am J Cardiovasc Drugs. 2013;13:385–398. doi: 10.1007/s40256-013-0041-4 [DOI] [PubMed] [Google Scholar]
- 24.Shamseddin MK, Parfrey PS. Sudden cardiac death in chronic kidney disease: epidemiology and prevention. Nat Rev Nephrol. 2011;7:145–154. doi: 10.1038/nrneph.2010.191 [DOI] [PubMed] [Google Scholar]
- 25.KDIGO Working Group. Chapter 1: introduction and definition of CKD-MBD and the development of the guideline statements. Kidney Int. 2009;76113:S3–S8. doi: 10.1038/ki.2009.189 [DOI] [PubMed] [Google Scholar]
- 26.Global KDI. Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD update work group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD) (vol 7, pg 1, 2017). Kidney Int Suppl. 2017;7:E1–E1. doi: 10.1016/j.kisu.2017.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cannata-Andia JB, Martin-Carro B, Martin-Virgala J, Rodriguez-Carrio J, Bande-Fernandez JJ, Alonso-Montes C, Carrillo-Lopez N. Chronic kidney disease-mineral and bone disorders: pathogenesis and management. Calcif Tissue Int. 2021;108:410–422. doi: 10.1007/s00223-020-00777-1 [DOI] [PubMed] [Google Scholar]
- 28.Rapp N, Evenepoel P, Stenvinkel P, Schurgers L. Uremic toxins and vascular calcification-missing the forest for all the trees. Toxins (Basel). 2020;12:624. doi: 10.3390/toxins12100624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shanahan CM. Mechanisms of vascular calcification in CKD-evidence for premature ageing? Nat Rev Nephrol. 2013;9:661–670. doi: 10.1038/nrneph.2013.176 [DOI] [PubMed] [Google Scholar]
- 30.Yamada S, Giachelli CM. Vascular calcification in CKD-MBD: roles for phosphate, FGF23, and Klotho. Bone. 2017;100:87–93. doi: 10.1016/j.bone.2016.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Handy CE, Desai CS, Dardari ZA, Al-Mallah MH, Miedema MD, Ouyang P, Budoff MJ, Blumenthal RS, Nasir K, Blaha MJ. The association of coronary artery calcium with noncardiovascular disease: the Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc Imaging. 2016;9:568–576. doi: 10.1016/j.jcmg.2015.09.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Park S, Cho NJ, Heo NH, Rhee EJ, Gil H, Lee EY. Vascular calcification as a novel risk factor for kidney function deterioration in the nonelderly. Journal of the American Heart Association. 2021;10:e019300. doi: 10.1161/JAHA.120.019300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yun HR, Joo YS, Kim HW, Park JT, Chang TI, Son NH, Yoo TH, Kang SW, Sung S, Lee KB, et al. Coronary artery calcification score and the progression of chronic kidney disease. J Am Soc Nephrol. 2022;33:1590–1601. doi: 10.1681/asn.2022010080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kestenbaum BR, Adeney KL, de Boer IH, Ix JH, Shlipak MG, Siscovick DS. Incidence and progression of coronary calcification in chronic kidney disease: the Multi-Ethnic Study of Atherosclerosis. Kidney Int. 2009;76:991–998. doi: 10.1038/ki.2009.298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Piers LH, Touw HR, Gansevoort R, Franssen CF, Oudkerk M, Zijlstra F, Tio RA. Relation of aortic valve and coronary artery calcium in patients with chronic kidney disease to the stage and etiology of the renal disease. Am J Cardiol. 2009;103:1473–1477. doi: 10.1016/j.amjcard.2009.01.396 [DOI] [PubMed] [Google Scholar]
- 36.Tomiyama C, Carvalho AB, Higa A, Jorgetti V, Draibe SA, Canziani ME. Coronary calcification is associated with lower bone formation rate in CKD patients not yet in dialysis treatment. J Bone Miner Res. 2010;25:499–504. doi: 10.1359/jbmr.090735 [DOI] [PubMed] [Google Scholar]
- 37.Aleksova J, Kurniawan S, Vucak-Dzumhur M, Kerr P, Ebeling PR, Milat F, Elder GJ. Aortic vascular calcification is inversely associated with the trabecular bone score in patients receiving dialysis. Bone. 2018;113:118–123. doi: 10.1016/j.bone.2018.05.014 [DOI] [PubMed] [Google Scholar]
- 38.Chen TY, Yang J, Zuo L, Wang L, Wang LF. Relationship of abdominal aortic calcification with lumbar vertebral volumetric bone mineral density assessed by quantitative computed tomography in maintenance hemodialysis patients. Arch Osteoporos. 2022;17:24. doi: 10.1007/s11657-022-01059-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kraus MA, Kalra PA, Hunter J, Menoyo J, Stankus N. The prevalence of vascular calcification in patients with end-stage renal disease on hemodialysis: a cross-sectional observational study. Ther Adv Chronic Dis. 2015;6:84–96. doi: 10.1177/2040622315578654 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pencak P, Czerwienska B, Ficek R, Wyskida K, Kujawa-Szewieczek A, Olszanecka-Glinianowicz M, Wiecek A, Chudek J. Calcification of coronary arteries and abdominal aorta in relation to traditional and novel risk factors of atherosclerosis in hemodialysis patients. BMC Nephrol. 2013;14:10. doi: 10.1186/1471-2369-14-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Toussaint ND, Lau KK, Strauss BJ, Polkinghorne KR, Kerr PG. Associations between vascular calcification, arterial stiffness and bone mineral density in chronic kidney disease. Nephrol Dial Transplant. 2008;23:586–593. doi: 10.1093/ndt/gfm660 [DOI] [PubMed] [Google Scholar]
- 42.Toussaint ND, Lau KK, Strauss BJ, Polkinghorne KR, Kerr PG. Relationship between vascular calcification, arterial stiffness and bone mineral density in a cross-sectional study of prevalent Australian haemodialysis patients. Nephrology (Carlton). 2009;14:105–112. doi: 10.1111/j.1440-1797.2008.01056.x [DOI] [PubMed] [Google Scholar]
- 43.Sorensen IMH, Saurbrey SAK, Hjortkjaer HO, Brainin P, Carlson N, Ballegaard ELF, Kamper AL, Christoffersen C, Feldt-Rasmussen B, Kofoed KF, et al. Regional distribution and severity of arterial calcification in patients with chronic kidney disease stages 1-5: a cross-sectional study of the Copenhagen chronic kidney disease cohort. BMC Nephrol. 2020;21:534. doi: 10.1186/s12882-020-02192-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Damjanovic T, Djuric S, Schlieper G, Markovic N, Dimkovic S, Radojicic Z, Kruger T, Floege J, Dimkovic N. Clinical features of hemodialysis patients with intimal versus medial vascular calcifications. J Nephrol. 2009;22:358–366. [PubMed] [Google Scholar]
- 45.London GM, Guerin AP, Marchais SJ, Metivier F, Pannier B, Adda H. Arterial media calcification in end-stage renal disease: impact on all-cause and cardiovascular mortality. Nephrol Dial Transplant. 2003;18:1731–1740. doi: 10.1093/ndt/gfg414 [DOI] [PubMed] [Google Scholar]
- 46.Gelev S, Spasovski G, Trajkovski Z, Damjanovski G, Amitov V, Selim G, Dzekova P, Sikole A. Factors associated with various arterial calcifications in haemodialysis patients. Prilozi. 2008;29:185–199. [PubMed] [Google Scholar]
- 47.Chakrabarti A, Goldstein DR, Sutton NR. Age-associated arterial calcification: the current pursuit of aggravating and mitigating factors. Curr Opin Lipidol. 2020;31:265–272. doi: 10.1097/MOL.0000000000000703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chen Z, Sun J, Haarhaus M, Barany P, Wennberg L, Ripsweden J, Brismar TB, Lindholm B, Wernerson A, Soderberg M, et al. Bone mineral density of extremities is associated with coronary calcification and biopsy-verified vascular calcification in living-donor renal transplant recipients. J Bone Miner Metab. 2017;35:536–543. doi: 10.1007/s00774-016-0788-1 [DOI] [PubMed] [Google Scholar]
- 49.Nasir K, Katz R, Al-Mallah M, Takasu J, Shavelle DM, Carr JJ, Kronmal R, Blumenthal RS, O’Brien K, Budoff MJ. Relationship of aortic valve calcification with coronary artery calcium severity: the Multi-Ethnic Study of Atherosclerosis (MESA). J Cardiovasc Comput Tomogr. 2010;4:41–46. doi: 10.1016/j.jcct.2009.12.002 [DOI] [PubMed] [Google Scholar]
- 50.Al-Biltagi M, ElHafez MAA, El Amrousy DM, El-Gamasy M, El-Serogy H. Evaluation of the coronary circulation and calcification in children on regular hemodialysis. Pediatr Nephrol. 2017;32:1941–1951. doi: 10.1007/s00467-017-3678-4 [DOI] [PubMed] [Google Scholar]
- 51.Goodman WG, Goldin J, Kuizon BD, Yoon C, Gales B, Sider D, Wang Y, Chung J, Emerick A, Greaser L, et al. Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N Engl J Med. 2000;342:1478–1483. doi: 10.1056/NEJM200005183422003 [DOI] [PubMed] [Google Scholar]
- 52.Lumpaopong A, Mathew AV, John E, Jelnin V, Benedetti E, Testa G, Oberholzer J, Sankary H, Ruiz C. Early coronary calcification in children and young adults with end-stage renal disease. Transplant Proc. 2007;39:37–39. doi: 10.1016/j.transproceed.2006.10.198 [DOI] [PubMed] [Google Scholar]
- 53.Dobson LE, Prendergast BD. Heart valve disease: a journey of discovery. Heart. 2022;108:774–779. doi: 10.1136/heartjnl-2021-320146 [DOI] [PubMed] [Google Scholar]
- 54.Iung B, Vahanian A. Epidemiology of valvular heart disease in the adult. Nat Rev Cardiol. 2011;8:162–172. doi: 10.1038/nrcardio.2010.202 [DOI] [PubMed] [Google Scholar]
- 55.Schoen FJ. Morphology, clinicopathologic correlations, and mechanisms in heart valve health and disease. Cardiovasc Eng Technol. 2018;9:126–140. doi: 10.1007/s13239-016-0277-7 [DOI] [PubMed] [Google Scholar]
- 56.Fisher CI, Chen J, Merryman WD. Calcific nodule morphogenesis by heart valve interstitial cells is strain dependent. Biomech Model Mechanobiol. 2013;12:5–17. doi: 10.1007/s10237-012-0377-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rader F, Sachdev E, Arsanjani R, Siegel RJ. Left ventricular hypertrophy in valvular aortic stenosis: mechanisms and clinical implications. Am J Med. 2015;128:344–352. doi: 10.1016/j.amjmed.2014.10.054 [DOI] [PubMed] [Google Scholar]
- 58.Churchill TW, Yucel E, Deferm S, Levine RA, Hung J, Bertrand PB. Mitral valve dysfunction in patients with annular calcification: JACC review topic of the week. J Am Coll Cardiol. 2022;80:739–751. doi: 10.1016/j.jacc.2022.05.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wang TKM, Griffin BP, Xu B, Rodriguez LL, Popovic ZB, Gillinov MA, Pettersson GB, Desai MY. Relationships between mitral annular calcification and cardiovascular events: a meta-analysis. Echocardiogr. 2020;37:1723–1731. doi: 10.1111/echo.14861 [DOI] [PubMed] [Google Scholar]
- 60.Hensen LCR, Mahdiui ME, van Rosendael AR, Smit JM, Jukema JW, Bax JJ, Delgado V. Prevalence and prognostic implications of mitral and aortic valve calcium in patients with chronic kidney disease. Am J Cardiol. 2018;122:1732–1737. doi: 10.1016/j.amjcard.2018.08.009 [DOI] [PubMed] [Google Scholar]
- 61.Urena P, Malergue MC, Goldfarb B, Prieur P, Guedon-Rapoud C, Petrover M. Evolutive aortic stenosis in hemodialysis patients: analysis of risk factors. Nephrologie. 1999;20:217–225. [PubMed] [Google Scholar]
- 62.Nguyen V, Willner N, Eltchaninoff H, Burwash IG, Michel M, Durand E, Gilard M, Dindorf C, Iung B, Cribier A, et al. Trends in aortic valve replacement for aortic stenosis: a French nationwide study. Eur Heart J. 2022;43:666–679. doi: 10.1093/eurheartj/ehab773 [DOI] [PubMed] [Google Scholar]
- 63.Luraghi G, Matas JFR, Beretta M, Chiozzi N, Iannetti L, Migliavacca F. The impact of calcification patterns in transcatheter aortic valve performance: a fluid-structure interaction analysis. Comput Methods Biomech Biomed Engin. 2021;24:375–383. doi: 10.1080/10255842.2020.1817409 [DOI] [PubMed] [Google Scholar]
- 64.Tomsic A, Hiemstra YL, van Brakel TJ, Versteegh MIM, Marsan NA, Klautz RJM, Palmen M. Outcomes of valve repair for degenerative disease in patients with mitral annular calcification. Ann Thorac Surg. 2019;107:1195–1201. doi: 10.1016/j.athoracsur.2018.08.017 [DOI] [PubMed] [Google Scholar]
- 65.Schlotter F, Halu A, Goto S, Blaser MC, Body SC, Lee LH, Higashi H, DeLaughter DM, Hutcheson JD, Vyas P, et al. Spatiotemporal multi-omics mapping generates a molecular atlas of the aortic valve and reveals networks driving disease. Circulation. 2018;138:377–393. doi: 10.1161/CIRCULATIONAHA.117.032291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kraler S, Blaser MC, Aikawa E, Camici GG, Luscher TF. Calcific aortic valve disease: from molecular and cellular mechanisms to medical therapy. Eur Heart J. 2022;43:683–697. doi: 10.1093/eurheartj/ehab757 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shroff GR, Bangalore S, Bhave NM, Chang TI, Garcia S, Mathew RO, Rangaswami J, Ternacle J, Thourani VH, Pibarot P, et al. ; American Heart Association Council on the Kidney in Cardiovascular Disease and Stroke Council. Evaluation and management of aortic stenosis in chronic kidney disease: a scientific statement from the American Heart Association. Circulation. 2021;143:e1088–e1114. doi: 10.1161/CIR.0000000000000979 [DOI] [PubMed] [Google Scholar]
- 68.Strange G, Stewart S, Celermajer D, Prior D, Scalia GM, Marwick T, Ilton M, Joseph M, Codde J, Playford D, et al. ; National Echocardiography Database of Australia contributing sites. Poor long-term survival in patients with moderate aortic stenosis. J Am Coll Cardiol. 2019;74:1851–1863. doi: 10.1016/j.jacc.2019.08.004 [DOI] [PubMed] [Google Scholar]
- 69.Wang Z, Jiang A, Wei F, Chen H. Cardiac valve calcification and risk of cardiovascular or all-cause mortality in dialysis patients: a meta-analysis. BMC Cardiovasc Disord. 2018;18:12. doi: 10.1186/s12872-018-0747-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kang DH, Park SJ, Lee SA, Lee S, Kim DH, Kim HK, Yun SC, Hong GR, Song JM, Chung CH, et al. Early surgery or conservative care for asymptomatic aortic stenosis. N Engl J Med. 2020;382:111–119. doi: 10.1056/NEJMoa1912846 [DOI] [PubMed] [Google Scholar]
- 71.Kang DH, Park SJ, Rim JH, Yun SC, Kim DH, Song JM, Choo SJ, Park SW, Song JK, Lee JW, et al. Early surgery versus conventional treatment in asymptomatic very severe aortic stenosis. Circulation. 2010;121:1502–1509. doi: 10.1161/CIRCULATIONAHA.109.909903 [DOI] [PubMed] [Google Scholar]
- 72.Taniguchi T, Morimoto T, Shiomi H, Ando K, Kanamori N, Murata K, Kitai T, Kawase Y, Izumi C, Miyake M, et al. ; CURRENT AS Registry Investigators. Initial surgical versus conservative strategies in patients with asymptomatic severe aortic stenosis. J Am Coll Cardiol. 2015;66:2827–2838. doi: 10.1016/j.jacc.2015.10.001 [DOI] [PubMed] [Google Scholar]
- 73.Cubeddu RJ, Asher CR, Lowry AM, Blackstone EH, Kapadia SR, Alu MC, Thourani VH, Mack MJ, Kodali SK, Herrmann HC, et al. ; PARTNER Trial Investigators. Impact of transcatheter aortic valve replacement on severity of chronic kidney disease. J Am Coll Cardiol. 2020;76:1410–1421. doi: 10.1016/j.jacc.2020.07.048 [DOI] [PubMed] [Google Scholar]
- 74.Witberg G, Steinmetz T, Landes U, Pistiner Hanit R, Green H, Goldman S, Vaknin-Assa H, Codner P, Perl L, Rozen-Zvi B, et al. Change in kidney function and 2-year mortality after transcatheter aortic valve replacement. JAMA Netw Open. 2021;4:e213296. doi: 10.1001/jamanetworkopen.2021.3296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gargiulo G, Capodanno D, Sannino A, Perrino C, Capranzano P, Stabile E, Trimarco B, Tamburino C, Esposito G. Moderate and severe preoperative chronic kidney disease worsen clinical outcomes after transcatheter aortic valve implantation: meta-analysis of 4992 patients. Circ Cardiovasc Interv. 2015;8:e002220. doi: 10.1161/CIRCINTERVENTIONS.114.002220 [DOI] [PubMed] [Google Scholar]
- 76.Mohananey D, Griffin BP, Svensson LG, Popovic ZB, Tuzcu EM, Rodriguez LL, Kapadia SR, Desai MY. Comparative outcomes of patients with advanced renal dysfunction undergoing transcatheter aortic valve replacement in the United States from 2011 to 2014. Circ Cardiovasc Interv. 2017;10:e005477. doi: 10.1161/CIRCINTERVENTIONS.117.005477 [DOI] [PubMed] [Google Scholar]
- 77.Wang J, Liu S, Han X, Chen Y, Chen H, Dong S, Song B. Impact of chronic kidney disease on the prognosis of transcatheter aortic valve replacement in patients with aortic stenosis: a meta-analysis of 133624 patients. Ann Thorac Cardiovasc Surg. 2022;28:83–95. doi: 10.5761/atcs.ra.21-00187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gupta T, Goel K, Kolte D, Khera S, Villablanca PA, Aronow WS, Bortnick AE, Slovut DP, Taub CC, Kizer JR, et al. Association of chronic kidney disease with in-hospital outcomes of transcatheter aortic valve replacement. JACC Cardiovasc Interv. 2017;10:2050–2060. doi: 10.1016/j.jcin.2017.07.044 [DOI] [PubMed] [Google Scholar]
- 79.Crimi G, De Marzo V, De Marco F, Conrotto F, Oreglia J, D’Ascenzo F, Testa L, Gorla R, Esposito G, Sorrentino S, et al. Acute kidney injury after transcatheter aortic valve replacement mediates the effect of chronic kidney disease. J Am Heart Assoc. 2022;11:e024589. doi: 10.1161/JAHA.121.024589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Rutsch F, Buers I, Nitschke Y. Hereditary disorders of cardiovascular calcification. Arterioscler Thromb Vasc Biol. 2021;41:35–47. doi: 10.1161/ATVBAHA.120.315577 [DOI] [PubMed] [Google Scholar]
- 81.Fowkes FG, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, Norman PE, Sampson UK, Williams LJ, Mensah GA, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet. 2013;382:1329–1340. doi: 10.1016/S0140-6736(13)61249-0 [DOI] [PubMed] [Google Scholar]
- 82.Kullo IJ, Rooke TW. Clinical practice. Peripheral artery disease. N Engl J Med. 2016;374:861–871. doi: 10.1056/NEJMcp1507631 [DOI] [PubMed] [Google Scholar]
- 83.Huang CL, Wu IH, Wu YW, Hwang JJ, Wang SS, Chen WJ, Lee WJ, Yang WS. Association of lower extremity arterial calcification with amputation and mortality in patients with symptomatic peripheral artery disease. PLoS One. 2014;9:e90201. doi: 10.1371/journal.pone.0090201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Narula N, Dannenberg AJ, Olin JW, Bhatt DL, Johnson KW, Nadkarni G, Min J, Torii S, Poojary P, Anand SS, et al. Pathology of peripheral artery disease in patients with critical limb ischemia. J Am Coll Cardiol. 2018;72:2152–2163. doi: 10.1016/j.jacc.2018.08.002 [DOI] [PubMed] [Google Scholar]
- 85.O’Neill WC, Han KH, Schneider TM, Hennigar RA. Prevalence of nonatheromatous lesions in peripheral arterial disease. Arterioscler Thromb Vasc Biol. 2015;35:439–447. doi: 10.1161/ATVBAHA.114.304764 [DOI] [PubMed] [Google Scholar]
- 86.Roijers JP, Rakke YS, Hopmans CJ, Buimer MG, Ho GH, de Groot HGW, Veen EJ, Mulder PGH, van der Laan L. A mortality prediction model for elderly patients with critical limb ischemia. J Vasc Surg. 2020;71:2065–2072.e2. doi: 10.1016/j.jvs.2019.08.245 [DOI] [PubMed] [Google Scholar]
- 87.Smith-Bindman R, Kwan ML, Marlow EC, Theis MK, Bolch W, Cheng SY, Bowles EJA, Duncan JR, Greenlee RT, Kushi LH, et al. Trends in use of medical imaging in US health care systems and in Ontario, Canada, 2000-2016. JAMA. 2019;322:843–856. doi: 10.1001/jama.2019.11456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lefebvre KM, Chevan J. The persistence of gender and racial disparities in vascular lower extremity amputation: an examination of HCUP-NIS data (2002-2011). Vasc Med. 2015;20:51–59. doi: 10.1177/1358863X14565373 [DOI] [PubMed] [Google Scholar]
- 89.Wu M, Shu Y, Wang L, Song L, Chen S, Liu Y, Bi J, Li D, Yang Y, Hu Y, et al. Metabolic syndrome severity score and the progression of CKD. Eur J Clin Invest. 2022;52:e13646. doi: 10.1111/eci.13646 [DOI] [PubMed] [Google Scholar]
- 90.Hutcheson JD, Goettsch C, Rogers MA, Aikawa E. Revisiting cardiovascular calcification: a multifaceted disease requiring a multidisciplinary approach. Semin Cell Dev Biol. 2015;46:68–77. doi: 10.1016/j.semcdb.2015.09.004 [DOI] [PubMed] [Google Scholar]
- 91.Libby P, Buring JE, Badimon L, Hansson GK, Deanfield J, Bittencourt MS, Tokgozoglu L, Lewis EF. Atherosclerosis. Nat Rev Dis Primers. 2019;5:56. doi: 10.1038/s41572-019-0106-z [DOI] [PubMed] [Google Scholar]
- 92.Libby P. Collagenases and cracks in the plaque. J Clin Invest. 2013;123:3201–3203. doi: 10.1172/JCI67526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kelly-Arnold A, Maldonado N, Laudier D, Aikawa E, Cardoso L, Weinbaum S. Revised microcalcification hypothesis for fibrous cap rupture in human coronary arteries. Proc Natl Acad Sci USA. 2013;110:10741–10746. doi: 10.1073/pnas.1308814110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Maldonado N, Kelly-Arnold A, Cardoso L, Weinbaum S. The explosive growth of small voids in vulnerable cap rupture; cavitation and interfacial debonding. J Biomech. 2013;46:396–401. doi: 10.1016/j.jbiomech.2012.10.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Vliegenthart R, Oudkerk M, Hofman A, Oei HH, van Dijck W, van Rooij FJ, Witteman JC. Coronary calcification improves cardiovascular risk prediction in the elderly. Circulation. 2005;112:572–577. doi: 10.1161/CIRCULATIONAHA.104.488916 [DOI] [PubMed] [Google Scholar]
- 96.Criqui MH, Denenberg JO, Ix JH, McClelland RL, Wassel CL, Rifkin DE, Carr JJ, Budoff MJ, Allison MA. Calcium density of coronary artery plaque and risk of incident cardiovascular events. JAMA. 2014;311:271–278. doi: 10.1001/jama.2013.282535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Frodermann V, Rohde D, Courties G, Severe N, Schloss MJ, Amatullah H, McAlpine CS, Cremer S, Hoyer FF, Ji F, et al. Exercise reduces inflammatory cell production and cardiovascular inflammation via instruction of hematopoietic progenitor cells. Nat Med. 2019;25:1761–1771. doi: 10.1038/s41591-019-0633-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ceneri N, Zhao L, Young BD, Healy A, Coskun S, Vasavada H, Yarovinsky TO, Ike K, Pardi R, Qin L, et al. Rac2 modulates atherosclerotic calcification by regulating macrophage interleukin-1beta production. Arterioscler Thromb Vasc Biol. 2017;37:328–340. doi: 10.1161/ATVBAHA.116.308507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Healy A, Berus JM, Christensen JL, Lee C, Mantsounga C, Dong W, Watts JP, Jr, Assali M, Ceneri N, Nilson R, et al. Statins disrupt macrophage Rac1 regulation leading to increased atherosclerotic plaque calcification. Arterioscler Thromb Vasc Biol. 2020;40:714–732. doi: 10.1161/ATVBAHA.119.313832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Puri R, Nicholls SJ, Shao M, Kataoka Y, Uno K, Kapadia SR, Tuzcu EM, Nissen SE. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65:1273–1282. doi: 10.1016/j.jacc.2015.01.036 [DOI] [PubMed] [Google Scholar]
- 101.Cooper WA, O’Brien SM, Thourani VH, Guyton RA, Bridges CR, Szczech LA, Petersen R, Peterson ED. Impact of renal dysfunction on outcomes of coronary artery bypass surgery: results from the Society of Thoracic Surgeons National Adult Cardiac Database. Circulation. 2006;113:1063–1070. doi: 10.1161/CIRCULATIONAHA.105.580084 [DOI] [PubMed] [Google Scholar]
- 102.Hill JM, Kereiakes DJ, Shlofmitz RA, Klein AJ, Riley RF, Price MJ, Herrmann HC, Bachinsky W, Waksman R, Stone GW, et al. ; Disrupt CAD III Investigators. Intravascular lithotripsy for treatment of severely calcified coronary artery disease. J Am Coll Cardiol. 2020;76:2635–2646. doi: 10.1016/j.jacc.2020.09.603 [DOI] [PubMed] [Google Scholar]
- 103.Khandkar C, Vaidya K, Karimi Galougahi K, Patel S. Low bone mineral density and coronary artery disease: a systematic review and meta-analysis. Int J Cardiol Heart Vasc. 2021;37:100891. doi: 10.1016/j.ijcha.2021.100891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Kim H, Lee J, Lee KB, Kim YH, Hong N, Park JT, Han SH, Kang SW, Choi KH, Oh KH, et al. Low bone mineral density is associated with coronary arterial calcification progression and incident cardiovascular events in patients with chronic kidney disease. Clin Kidney J. 2022;15:119–127. doi: 10.1093/ckj/sfab138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Pedersen AB, Ehrenstein V, Szepligeti SK, Sorensen HT. Hip fracture, comorbidity, and the risk of myocardial infarction and stroke: a Danish Nationwide Cohort Study, 1995-2015. J Bone Miner Res. 2017;32:2339–2346. doi: 10.1002/jbmr.3242 [DOI] [PubMed] [Google Scholar]
- 106.Schulz E, Arfai K, Liu X, Sayre J, Gilsanz V. Aortic calcification and the risk of osteoporosis and fractures. J Clin Endocrinol Metab. 2004;89:4246–4253. doi: 10.1210/jc.2003-030964 [DOI] [PubMed] [Google Scholar]
- 107.Simon SP, Fodor D, Muntean L, Poanta L, Cristea P, Rednic S. Bone mineral density, vertebral fractures and body mass index in postmenopausal women with abdominal aortic calcification. Endocr Res. 2014;39:1–6. doi: 10.3109/07435800.2013.794425 [DOI] [PubMed] [Google Scholar]
- 108.Dalla Via J, Sim M, Schousboe JT, Kiel DP, Zhu K, Hodgson JM, Gebre AK, Daly RM, Prince RL, Lewis JR. Association of abdominal aortic calcification with peripheral quantitative computed tomography bone measures in older women: the Perth Longitudinal Study of Ageing Women. Calcif Tissue Int. 2022;111:485–494. doi: 10.1007/s00223-022-01016-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Massera D, Buzkova P, Bortnick AE, Owens DS, Mao S, Li D, De Boer IH, Kestenbaum BR, Budoff MJ, Kizer JR. Bone mineral density and long-term progression of aortic valve and mitral annular calcification: the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis. 2021;335:126–134. doi: 10.1016/j.atherosclerosis.2021.08.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sotomayor CG, Benjamens S, Gomes-Neto AW, Pol RA, Groothof D, Te Velde-Keyzer CA, Chong G, Glaudemans A, Berger SP, Bakker SJL, et al. Bone mineral density and aortic calcification: evidence for a bone-vascular axis after kidney transplantation. Transplantation. 2021;105:231–239. doi: 10.1097/TP.0000000000003226 [DOI] [PubMed] [Google Scholar]
- 111.Uhlinova J, Kuudeberg A, Metskula K, Lember M, Rosenberg M. Significant associations between bone mineral density and vascular calcification in patients with different stages of chronic kidney disease. BMC Nephrol. 2022;23:327. doi: 10.1186/s12882-022-02955-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Campagnaro LS, Carvalho AB, Pina PM, Watanabe R, Canziani MEF. Bone mass measurement by DXA should be interpreted with caution in the CKD population with vascular calcification. Bone Rep. 2022;16:101169. doi: 10.1016/j.bonr.2022.101169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Jorgensen HS, Borghs H, Heye S, Smout D, Claes K, Evenepoel P. Vascular calcification of the abdominal aorta has minimal impact on lumbar spine bone density in patients with chronic kidney disease. Bone. 2022;162:116482. doi: 10.1016/j.bone.2022.116482 [DOI] [PubMed] [Google Scholar]
- 114.Filgueira A, Carvalho AB, Tomiyama C, Higa A, Rochitte CE, Santos RD, Canziani ME. Is coronary artery calcification associated with vertebral bone density in nondialyzed chronic kidney disease patients? Clin J Am Soc Nephrol. 2011;6:1456–1462. doi: 10.2215/CJN.10061110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen Z, Qureshi AR, Brismar TB, Ripsweden J, Haarhaus M, Barany P, Heimburger O, Lindholm B, Stenvinkel P. Differences in association of lower bone mineral density with higher coronary calcification in female and male end-stage renal disease patients. BMC Nephrol. 2019;20:59. doi: 10.1186/s12882-019-1235-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chen Z, Qureshi AR, Ripsweden J, Wennberg L, Heimburger O, Lindholm B, Barany P, Haarhaus M, Brismar TB, Stenvinkel P. Vertebral bone density associates with coronary artery calcification and is an independent predictor of poor outcome in end-stage renal disease patients. Bone. 2016;92:50–57. doi: 10.1016/j.bone.2016.08.007 [DOI] [PubMed] [Google Scholar]
- 117.Dai L, Debowska M, Lukaszuk T, Bobrowski L, Barany P, Soderberg M, Thiagarajan D, Frostegard J, Wennberg L, Lindholm B, et al. Phenotypic features of vascular calcification in chronic kidney disease. J Intern Med. 2020;287:422–434. doi: 10.1111/joim.13012 [DOI] [PubMed] [Google Scholar]
- 118.Thompson B, Towler DA. Arterial calcification and bone physiology: role of the bone-vascular axis. Nat Rev Endocrinol. 2012;8:529–543. doi: 10.1038/nrendo.2012.36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Evenepoel P, Opdebeeck B, David K, D’Haese PC. Bone-vascular axis in chronic kidney disease. Adv Chronic Kidney Dis. 2019;26:472–483. doi: 10.1053/j.ackd.2019.09.006 [DOI] [PubMed] [Google Scholar]
- 120.Johnson RC, Leopold JA, Loscalzo J. Vascular calcification: pathobiological mechanisms and clinical implications. Circ Res. 2006;99:1044–1059. doi: 10.1161/01.RES.0000249379.55535.21 [DOI] [PubMed] [Google Scholar]
- 121.Oksala N, Levula M, Pelto-Huikko M, Kytomaki L, Soini JT, Salenius J, Kahonen M, Karhunen PJ, Laaksonen R, Parkkila S, et al. Carbonic anhydrases II and XII are up-regulated in osteoclast-like cells in advanced human atherosclerotic plaques-Tampere Vascular Study. Ann Med. 2010;42:360–370. doi: 10.3109/07853890.2010.486408 [DOI] [PubMed] [Google Scholar]
- 122.Qiao JH, Mishra V, Fishbein MC, Sinha SK, Rajavashisth TB. Multinucleated giant cells in atherosclerotic plaques of human carotid arteries: identification of osteoclast-like cells and their specific proteins in artery wall. Exp Mol Pathol. 2015;99:654–662. doi: 10.1016/j.yexmp.2015.11.010 [DOI] [PubMed] [Google Scholar]
- 123.Ge Q, Ruan CC, Ma Y, Tang XF, Wu QH, Wang JG, Zhu DL, Gao PJ. Osteopontin regulates macrophage activation and osteoclast formation in hypertensive patients with vascular calcification. Sci Rep. 2017;7:40253. doi: 10.1038/srep40253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Han KH, Hennigar RA, O’Neill WC. The association of bone and osteoclasts with vascular calcification. Vasc Med. 2015;20:527–533. doi: 10.1177/1358863X15597076 [DOI] [PubMed] [Google Scholar]
- 125.Espitia O, Chatelais M, Steenman M, Charrier C, Maurel B, Georges S, Houlgatte R, Verrecchia F, Ory B, Lamoureux F, et al. Implication of molecular vascular smooth muscle cell heterogeneity among arterial beds in arterial calcification. PLoS One. 2018;13:e0191976. doi: 10.1371/journal.pone.0191976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hunt JL, Fairman R, Mitchell ME, Carpenter JP, Golden M, Khalapyan T, Wolfe M, Neschis D, Milner R, Scoll B, et al. Bone formation in carotid plaques: a clinicopathological study. Stroke. 2002;33:1214–1219. doi: 10.1161/01.str.0000013741.41309.67 [DOI] [PubMed] [Google Scholar]
- 127.Mohler ER, 3rd, Gannon F, Reynolds C, Zimmerman R, Keane MG, Kaplan FS. Bone formation and inflammation in cardiac valves. Circulation. 2001;103:1522–1528. doi: 10.1161/01.cir.103.11.1522 [DOI] [PubMed] [Google Scholar]
- 128.Farhat GN, Cauley JA. The link between osteoporosis and cardiovascular disease. Clin Cases Miner Bone Metab. 2008;5:19–34. [PMC free article] [PubMed] [Google Scholar]
- 129.Hruska KA, Mathew S, Lund R, Qiu P, Pratt R. Hyperphosphatemia of chronic kidney disease. Kidney Int. 2008;74:148–157. doi: 10.1038/ki.2008.130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Davies MR, Lund RJ, Mathew S, Hruska KA. Low turnover osteodystrophy and vascular calcification are amenable to skeletal anabolism in an animal model of chronic kidney disease and the metabolic syndrome. J Am Soc Nephrol. 2005;16:917–928. doi: 10.1681/ASN.2004100835 [DOI] [PubMed] [Google Scholar]
- 131.Turner ME, Rowsell TS, Lansing AP, Jeronimo PS, Lee LH, Svajger BA, Zelt JGE, Forster CM, Petkovich MP, Holden RM, et al. Vascular calcification maladaptively participates in acute phosphate homeostasis. Cardiovasc Res. 2022;cvac162. doi: 10.1093/cvr/cvac162 [DOI] [PubMed] [Google Scholar]
- 132.Alves RD, Eijken M, van de Peppel J, van Leeuwen JP. Calcifying vascular smooth muscle cells and osteoblasts: independent cell types exhibiting extracellular matrix and biomineralization-related mimicries. BMC Genomics. 2014;15:965. doi: 10.1186/1471-2164-15-965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Mody N, Parhami F, Sarafian TA, Demer LL. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radical Bio Med. 2001;31:509–519. doi: 10.1016/S0891-5849(01)00610-4 [DOI] [PubMed] [Google Scholar]
- 134.Patel JJ, Bourne LE, Davies BK, Arnett TR, MacRae VE, Wheeler-Jones CPD, Orriss IR. Differing calcification processes in cultured vascular smooth muscle cells and osteoblasts. Exp Cell Res. 2019;380:100–113. doi: 10.1016/j.yexcr.2019.04.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Patel JJ, Zhu D, Opdebeeck B, D’Haese P, Millan JL, Bourne LE, Wheeler-Jones CPD, Arnett TR, MacRae VE, Orriss IR. Inhibition of arterial medial calcification and bone mineralization by extracellular nucleotides: the same functional effect mediated by different cellular mechanisms. J Cell Physiol. 2018;233:3230–3243. doi: 10.1002/jcp.26166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Schmid K, Mcsharry WO, Pameijer CH, Binette JP. Chemical and physicochemical studies on the mineral-deposits of the human atherosclerotic aorta. Atherosclerosis. 1980;37:199–210. doi: 10.1016/0021-9150(80)90005-2 [DOI] [PubMed] [Google Scholar]
- 137.Duer MJ, Friscic T, Proudfoot D, Reid DG, Schoppet M, Shanahan CM, Skepper JN, Wise ER. Mineral surface in calcified plaque is like that of bone: further evidence for regulated mineralization. Arterioscler Thromb Vasc Biol. 2008;28:2030–2034. doi: 10.1161/ATVBAHA.108.172387 [DOI] [PubMed] [Google Scholar]
- 138.Tomazic BB, Brown WE, Queral LA, Sadovnik M. Physiochemical characterization of cardiovascular calcified deposits. I. Isolation, purification and instrumental analysis. Atherosclerosis. 1988;69:5–19. doi: 10.1016/0021-9150(88)90284-5 [DOI] [PubMed] [Google Scholar]
- 139.Bertazzo S, Gentleman E, Cloyd KL, Chester AH, Yacoub MH, Stevens MM. Nano-analytical electron microscopy reveals fundamental insights into human cardiovascular tissue calcification. Nat Mater. 2013;12:576–583. doi: 10.1038/nmat3627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Barnsley J, Buckland G, Chan PE, Ong A, Ramos AS, Baxter M, Laskou F, Dennison EM, Cooper C, Patel HP. Pathophysiology and treatment of osteoporosis: challenges for clinical practice in older people. Aging Clin Exp Res. 2021;33:759–773. doi: 10.1007/s40520-021-01817-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Tonelli M, Sacks F, Pfeffer M, Gao ZW, Curhan G, Invest CT. Relation between serum phosphate level and cardiovascular event rate in people with coronary disease. Circulation. 2005;112:2627–2633. doi: 10.1161/Circulationaha.105.553198 [DOI] [PubMed] [Google Scholar]
- 142.Kutikhin AG, Feenstra L, Kostyunin AE, Yuzhalin AE, Hillebrands JL, Krenning G. Calciprotein particles: balancing mineral homeostasis and vascular pathology. Arterioscler Thromb Vasc Biol. 2021;41:1607–1624. doi: 10.1161/ATVBAHA.120.315697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Chen W, Fitzpatrick J, Monroy-Trujillo JM, Sozio SM, Jaar BG, Estrella MM, Serrano J, Anokhina V, Miller BL, Melamed ML, et al. Associations of serum calciprotein particle size and transformation time with arterial calcification, arterial stiffness, and mortality in incident hemodialysis patients. Am J Kidney Dis. 2021;77:346–354. doi: 10.1053/j.ajkd.2020.05.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kurmann R, Buffle E, Pasch A, Seiler C, de Marchi SF. Predicting progression of aortic stenosis by measuring serum calcification propensity. Clin Cardiol. 2022;45:1297–1302. doi: 10.1002/clc.23922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Pasch A, Block GA, Bachtler M, Smith ER, Jahnen-Dechent W, Arampatzis S, Chertow GM, Parfrey P, Ma X, Floege J. Blood calcification propensity, cardiovascular events, and survival in patients receiving hemodialysis in the EVOLVE trial. Clin J Am Soc Nephrol. 2017;12:315–322. doi: 10.2215/CJN.04720416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Toussaint ND, Pedagogos E, Lioufas NM, Elder GJ, Pascoe EM, Badve SV, Valks A, Block GA, Boudville N, Cameron JD, et al. ; IMPROVE-CKD Trial Investigators. A randomized trial on the effect of phosphate reduction on vascular end points in CKD (IMPROVE-CKD). J Am Soc Nephrol. 2020;31:2653–2666. doi: 10.1681/ASN.2020040411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Xu CL, Smith ER, Tiong MK, Ruderman I, Toussaint ND. Interventions to attenuate vascular calcification progression in chronic kidney disease: a systematic review of clinical trials. J Am Soc Nephrol. 2022;33:1011–1032. doi: 10.1681/asn.2021101327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Cremers S, Papapoulos S. Pharmacology of bisphosphonates. Bone. 2011;49:42–49. doi: 10.1016/j.bone.2011.01.014 [DOI] [PubMed] [Google Scholar]
- 149.Drake MT, Clarke BL, Khosla S. Bisphosphonates: mechanism of action and role in clinical practice. Mayo Clin Proc. 2008;83:1032–1045. doi: 10.4065/83.9.1032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Elmariah S, Delaney JA, O’Brien KD, Budoff MJ, Vogel-Claussen J, Fuster V, Kronmal RA, Halperin JL. Bisphosphonate use and prevalence of valvular and vascular calcification in women MESA (the Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol. 2010;56:1752–1759. doi: 10.1016/j.jacc.2010.05.050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Skolnick AH, Osranek M, Formica P, Kronzon I. Osteoporosis treatment and progression of aortic stenosis. Am J Cardiol. 2009;104:122–124. doi: 10.1016/j.amjcard.2009.02.051 [DOI] [PubMed] [Google Scholar]
- 152.Cai G, Keen HI, Host LV, Aitken D, Laslett LL, Winzenberg T, Wluka AE, Black D, Jones G. Once-yearly zoledronic acid and change in abdominal aortic calcification over 3 years in postmenopausal women with osteoporosis: results from the HORIZON Pivotal Fracture Trial. Osteoporosis Int. 2020;31:1741–1747. doi: 10.1007/s00198-020-05430-z [DOI] [PubMed] [Google Scholar]
- 153.Lomashvili KA, Monier-Faugere MC, Wang X, Malluche HH, O’Neill WC. Effect of bisphosphonates on vascular calcification and bone metabolism in experimental renal failure. Kidney Int. 2009;75:617–625. doi: 10.1038/ki.2008.646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Hartle JE, Tang X, Kirchner HL, Bucaloiu ID, Sartorius JA, Pogrebnaya ZV, Akers GA, Carnero GE, Perkins RM. Bisphosphonate therapy, death, and cardiovascular events among female patients with CKD: a retrospective cohort study. Am J Kidney Dis. 2012;59:636–644. doi: 10.1053/j.ajkd.2011.11.037 [DOI] [PubMed] [Google Scholar]
- 155.Perkins RM, Kirchner HL, Matsushita K, Bucaloiu ID, Norfolk E, Hartle JE. Bisphosphonates and mortality in women with CKD and the presence or absence of cardiovascular disease. Clin J Am Soc Nephrol. 2014;9:874–880. doi: 10.2215/CJN.07790713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Lyles KW, Colon-Emeric CS, Magaziner JS, Adachi JD, Pieper CF, Mautalen C, Hyldstrup L, Recknor C, Nordsletten L, Moore KA, et al. ; HORIZON Recurrent Fracture Trial. Zoledronic acid and clinical fractures and mortality after hip fracture. N Engl J Med. 2007;357:1799–1809. doi: 10.1056/NEJMoa074941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Cummings SR, Lui LY, Eastell R, Allen IE. Association between drug treatments for patients with osteoporosis and overall mortality rates a meta-analysis. Jama Intern Med. 2019;179:1491–1500. doi: 10.1001/jamainternmed.2019.2779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.D’Silva KM, Cromer SJ, Yu EW, Fischer M, Kim SC. Risk of incident atrial fibrillation with zoledronic acid versus denosumab: a propensity score-matched cohort study. J Bone Miner Res. 2021;36:52–60. doi: 10.1002/jbmr.4174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.John Camm A. Review of the cardiovascular safety of zoledronic acid and other bisphosphonates for the treatment of osteoporosis. Clin Ther. 2010;32:426–436. doi: 10.1016/j.clinthera.2010.03.014 [DOI] [PubMed] [Google Scholar]
- 160.Bakhshian Nik A, Ng HH, Garcia Russo M, Iacoviello F, Shearing PR, Bertazzo S, Hutcheson JD. The time-dependent role of bisphosphonates on atherosclerotic plaque calcification. J Cardiovasc Dev Dis. 2022;9:168. doi: 10.3390/jcdd9060168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Ruiz JL, Hutcheson JD, Cardoso L, Bakhshian Nik A, Condado de Abreu A, Pham T, Buffolo F, Busatto S, Federici S, Ridolfi A, et al. Nanoanalytical analysis of bisphosphonate-driven alterations of microcalcifications using a 3D hydrogel system and in vivo mouse model. Proc Natl Acad Sci USA. 2021;118:e1811725118. doi: 10.1073/pnas.1811725118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Rossini M, Adami G, Adami S, Viapiana O, Gatti D. Safety issues and adverse reactions with osteoporosis management. Expert Opin Drug Saf. 2016;15:321–332. doi: 10.1517/14740338.2016.1136287 [DOI] [PubMed] [Google Scholar]
- 163.Oda T, Jodicke AM, Robinson DE, Delmestri A, Keogh RH, Prieto-Alhambra D. Oral bisphosphonates are associated with increased risk of severe acute kidney injury in elderly patients with complex health needs: a self-controlled case series in the United Kingdom. J Bone Miner Res. 2022;37:1270–1278. doi: 10.1002/jbmr.4573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Price PA, Urist MR, Otawara Y. Matrix Gla protein, a new gamma-carboxyglutamic acid-containing protein which is associated with the organic matrix of bone. Biochem Biophys Res Commun. 1983;117:765–771. doi: 10.1016/0006-291x(83)91663-7 [DOI] [PubMed] [Google Scholar]
- 165.Otawara Y, Price PA. Developmental appearance of matrix GLA protein during calcification in the rat. J Biol Chem. 1986;261:10828–10832. [PubMed] [Google Scholar]
- 166.Shanahan CM, Cary NR, Metcalfe JC, Weissberg PL. High expression of genes for calcification-regulating proteins in human atherosclerotic plaques. J Clin Invest. 1994;93:2393–2402. doi: 10.1172/JCI117246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.O’Young J, Liao Y, Xiao Y, Jalkanen J, Lajoie G, Karttunen M, Goldberg HA, Hunter GK. Matrix Gla protein inhibits ectopic calcification by a direct interaction with hydroxyapatite crystals. J Am Chem Soc. 2011;133:18406–18412. doi: 10.1021/ja207628k [DOI] [PubMed] [Google Scholar]
- 168.Viegas CS, Rafael MS, Enriquez JL, Teixeira A, Vitorino R, Luis IM, Costa RM, Santos S, Cavaco S, Neves J, et al. Gla-rich protein acts as a calcification inhibitor in the human cardiovascular system. Arterioscler Thromb Vasc Biol. 2015;35:399–408. doi: 10.1161/ATVBAHA.114.304823 [DOI] [PubMed] [Google Scholar]
- 169.Jaminon AMG, Dai L, Qureshi AR, Evenepoel P, Ripsweden J, Soderberg M, Witasp A, Olauson H, Schurgers LJ, Stenvinkel P. Matrix Gla protein is an independent predictor of both intimal and medial vascular calcification in chronic kidney disease. Sci Rep. 2020;10:6586. doi: 10.1038/s41598-020-63013-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Delanaye P, Krzesinski JM, Warling X, Moonen M, Smelten N, Medart L, Pottel H, Cavalier E. Dephosphorylated-uncarboxylated Matrix Gla protein concentration is predictive of vitamin K status and is correlated with vascular calcification in a cohort of hemodialysis patients. BMC Nephrol. 2014;15:145. doi: 10.1186/1471-2369-15-145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Jespersen T, Mollehave LT, Thuesen BH, Skaaby T, Rossing P, Toft U, Jorgensen NR, Corfixen BL, Jakobsen J, Frimodt-Moller M, et al. Uncarboxylated matrix Gla-protein: a biomarker of vitamin K status and cardiovascular risk. Clin Biochem. 2020;83:49–56. doi: 10.1016/j.clinbiochem.2020.05.005 [DOI] [PubMed] [Google Scholar]
- 172.Mayer O, Jr, Seidlerova J, Wohlfahrt P, Filipovsky J, Vanek J, Cifkova R, Windrichova J, Topolcan O, Knapen MH, Drummen NE, et al. Desphospho-uncarboxylated matrix Gla protein is associated with increased aortic stiffness in a general population. J Hum Hypertens. 2016;30:418–423. doi: 10.1038/jhh.2015.55 [DOI] [PubMed] [Google Scholar]
- 173.Goiko M, Dierolf J, Gleberzon JS, Liao Y, Grohe B, Goldberg HA, de Bruyn JR, Hunter GK. Peptides of Matrix Gla protein inhibit nucleation and growth of hydroxyapatite and calcium oxalate monohydrate crystals. PLoS One. 2013;8:e80344. doi: 10.1371/journal.pone.0080344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Parashar A, Bak K, Murshed M. Prevention of arterial elastocalcinosis: differential roles of the conserved glutamic acid and serine residues of matrix Gla protein. Arterioscler Thromb Vasc Biol. 2022;42:e155–e167. doi: 10.1161/ATVBAHA.122.317518 [DOI] [PubMed] [Google Scholar]
- 175.Schurgers LJ, Spronk HM, Skepper JN, Hackeng TM, Shanahan CM, Vermeer C, Weissberg PL, Proudfoot D. Post-translational modifications regulate matrix Gla protein function: importance for inhibition of vascular smooth muscle cell calcification. J Thromb Haemost. 2007;5:2503–2511. doi: 10.1111/j.1538-7836.2007.02758.x [DOI] [PubMed] [Google Scholar]
- 176.Boxma PY, van den Berg E, Geleijnse JM, Laverman GD, Schurgers LJ, Vermeer C, Kema IP, Muskiet FA, Navis G, Bakker SJ, et al. Vitamin k intake and plasma desphospho-uncarboxylated matrix Gla-protein levels in kidney transplant recipients. PLoS One. 2012;7:e47991. doi: 10.1371/journal.pone.0047991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Saritas T, Reinartz S, Kruger T, Ketteler M, Liangos O, Labriola L, Stenvinkel P, Kopp C, Westenfeld R, Evenepoel P, et al. Vitamin K1 and progression of cardiovascular calcifications in hemodialysis patients: the VitaVasK randomized controlled trial. Clin Kidney J. 2022;15:2300–2311. doi: 10.1093/ckj/sfac184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Witham MD, Lees JS, White M, Band M, Bell S, Chantler DJ, Ford I, Fulton RL, Kennedy G, Littleford RC, et al. Vitamin K supplementation to improve vascular stiffness in CKD: the K4Kidneys randomized controlled trial. J Am Soc Nephrol. 2020;31:2434–2445. doi: 10.1681/ASN.2020020225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.De Vriese AS, Caluwe R, Pyfferoen L, De Bacquer D, De Boeck K, Delanote J, De Surgeloose D, Van Hoenacker P, Van Vlem B, Verbeke F. Multicenter randomized controlled trial of vitamin K antagonist replacement by rivaroxaban with or without vitamin K2 in hemodialysis patients with atrial fibrillation: the Valkyrie Study. J Am Soc Nephrol. 2020;31:186–196. doi: 10.1681/ASN.2019060579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Kaesler N, Schreibing F, Speer T, Puente-Secades S, Rapp N, Drechsler C, Kabgani N, Kuppe C, Boor P, Jankowski V, et al. Altered vitamin K biodistribution and metabolism in experimental and human chronic kidney disease. Kidney Int. 2022;101:338–348. doi: 10.1016/j.kint.2021.10.029 [DOI] [PubMed] [Google Scholar]
- 181.Perello J, Gomez M, Ferrer MD, Rodriguez NY, Salcedo C, Buades JM, Perez MM, Torregrosa JV, Martin E, Maduell F. SNF472, a novel inhibitor of vascular calcification, could be administered during hemodialysis to attain potentially therapeutic phytate levels. J Nephrol. 2018;31:287–296. doi: 10.1007/s40620-018-0471-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Raggi P, Bellasi A, Bushinsky D, Bover J, Rodriguez M, Ketteler M, Sinha S, Salcedo C, Gillotti K, Padgett C, et al. Slowing progression of cardiovascular calcification with SNF472 in patients on hemodialysis: results of a randomized phase 2b study. Circulation. 2020;141:728–739. doi: 10.1161/CIRCULATIONAHA.119.044195 [DOI] [PubMed] [Google Scholar]
- 183.Perello J, Ferrer MD, Del Mar Perez M, Kaesler N, Brandenburg VM, Behets GJ, D’Haese PC, Garg R, Isern B, Gold A, et al. Mechanism of action of SNF472, a novel calcification inhibitor to treat vascular calcification and calciphylaxis. Br J Pharmacol. 2020;177:4400–4415. doi: 10.1111/bph.15163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Bushinsky DA, Raggi P, Bover J, Ketteler M, Bellasi A, Rodriguez M, Sinha S, Garg R, Perello J, Gold A, et al. ; CaLIPSO Investigators*. Effects of myo-inositol hexaphosphate (SNF472) on bone mineral density in patients receiving hemodialysis: an analysis of the randomized, placebo-controlled CaLIPSO study. Clin J Am Soc Nephrol. 2021;16:736–745. doi: 10.2215/CJN.16931020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.You AYF, Bergholt MS, St-Pierre JP, Kit-Anan W, Pence IJ, Chester AH, Yacoub MH, Bertazzo S, Stevens MM. Raman spectroscopy imaging reveals interplay between atherosclerosis and medial calcification in the human aorta. Sci Adv. 2017;3:e1701156. doi: 10.1126/sciadv.1701156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Lagier R, Baud CA. Magnesium whitlockite, a calcium phosphate crystal of special interest in pathology. Pathol Res Pract. 2003;199:329–335. doi: 10.1078/0344-0338-00425 [DOI] [PubMed] [Google Scholar]
- 187.Shah FA. Magnesium whitlockite - omnipresent in pathological mineralisation of soft tissues but not a significant inorganic constituent of bone. Acta Biomater. 2021;125:72–82. doi: 10.1016/j.actbio.2021.02.021 [DOI] [PubMed] [Google Scholar]
- 188.Liu H, Wang R. Associations between the serum magnesium and all-cause or cardiovascular mortality in chronic kidney disease and end-stage renal disease patients: a meta-analysis. Medicine (Baltim). 2021;100:e27486. doi: 10.1097/MD.0000000000027486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Oka T, Hamano T, Sakaguchi Y, Yamaguchi S, Kubota K, Senda M, Yonemoto S, Shimada K, Matsumoto A, Hashimoto N, et al. Proteinuria-associated renal magnesium wasting leads to hypomagnesemia: a common electrolyte abnormality in chronic kidney disease. Nephrol Dial Transplant. 2019;34:1154–1162. doi: 10.1093/ndt/gfy119 [DOI] [PubMed] [Google Scholar]
- 190.Fischer DC, Behets GJ, Hakenberg OW, Voigt M, Vervaet BA, Robijn S, Kundt G, Schareck W, D’Haese PC, Haffner D. Arterial microcalcification in atherosclerotic patients with and without chronic kidney disease: a comparative high-resolution scanning X-ray diffraction analysis. Calcif Tissue Int. 2012;90:465–472. doi: 10.1007/s00223-012-9594-5 [DOI] [PubMed] [Google Scholar]
- 191.Ter Braake AD, Vervloet MG, de Baaij JHF, Hoenderop JGJ. Magnesium to prevent kidney disease-associated vascular calcification: crystal clear?. Nephrol Dial Transplant. 2022;37:421–429. doi: 10.1093/ndt/gfaa222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Sakaguchi Y, Hamano T, Obi Y, Monden C, Oka T, Yamaguchi S, Matsui I, Hashimoto N, Matsumoto A, Shimada K, et al. A randomized trial of magnesium oxide and oral carbon adsorbent for coronary artery calcification in predialysis CKD. J Am Soc Nephrol. 2019;30:1073–1085. doi: 10.1681/ASN.2018111150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Moe SM, Abdalla S, Chertow GM, Parfrey PS, Block GA, Correa-Rotter R, Floege J, Herzog CA, London GM, Mahaffey KW, et al. ; Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) Trial Investigators. Effects of cinacalcet on fracture events in patients receiving hemodialysis: the EVOLVE trial. J Am Soc Nephrol. 2015;26:1466–1475. doi: 10.1681/ASN.2014040414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Raggi P, Chertow GM, Torres PU, Csiky B, Naso A, Nossuli K, Moustafa M, Goodman WG, Lopez N, Downey G, et al. ; ADVANCE Study Group. The ADVANCE study: a randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol Dial Transplant. 2011;26:1327–1339. doi: 10.1093/ndt/gfq725 [DOI] [PubMed] [Google Scholar]
- 195.Investigators ET, Chertow GM, Block GA, Correa-Rotter R, Drueke TB, Floege J, Goodman WG, Herzog CA, Kubo Y, London GM, et al. Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N Engl J Med. 2012;367:2482–2494. doi: 10.1056/NEJMoa1205624 [DOI] [PubMed] [Google Scholar]
- 196.Parfrey PS, Drueke TB, Block GA, Correa-Rotter R, Floege J, Herzog CA, London GM, Mahaffey KW, Moe SM, Wheeler DC, et al. ; Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) Trial Investigators. The effects of cinacalcet in older and younger patients on hemodialysis: the Evaluation of Cinacalcet HCl Therapy to Lower Cardiovascular Events (EVOLVE) trial. Clin J Am Soc Nephrol. 2015;10:791–799. doi: 10.2215/CJN.07730814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Evans M, Methven S, Gasparini A, Barany P, Birnie K, MacNeill S, May MT, Caskey FJ, Carrero JJ. Cinacalcet use and the risk of cardiovascular events, fractures and mortality in chronic kidney disease patients with secondary hyperparathyroidism. Sci Rep. 2018;8:2103. doi: 10.1038/s41598-018-20552-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Rauner M, Jähn K, Hemmatian H, Colditz J, Goettsch C. Cellular contributors to bone homeostasis. Aikawa E, Hutcheson JD, eds. In: Cardiovascular Calcification and Bone Mineralization. Cham: Springer International Publishing; 2020:333–371. [Google Scholar]
- 199.Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, Delmas P, Zoog HB, Austin M, Wang A, et al. ; FREEDOM Trial. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N Engl J Med. 2009;361:756–765. doi: 10.1056/NEJMoa0809493 [DOI] [PubMed] [Google Scholar]
- 200.Samelson EJ, Miller PD, Christiansen C, Daizadeh NS, Grazette L, Anthony MS, Egbuna O, Wang A, Siddhanti SR, Cheung AM, et al. RANKL inhibition with denosumab does not influence 3-year progression of aortic calcification or incidence of adverse cardiovascular events in postmenopausal women with osteoporosis and high cardiovascular risk. J Bone Miner Res. 2014;29:450–457. doi: 10.1002/jbmr.2043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Pawade TA, Doris MK, Bing R, White AC, Forsyth L, Evans E, Graham C, Williams MC, van Beek EJR, Fletcher A, et al. Effect of denosumab or alendronic acid on the progression of aortic stenosis: a double-blind randomized controlled trial. Circulation. 2021;143:2418–2427. doi: 10.1161/CIRCULATIONAHA.121.053708 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Chen CL, Chen NC, Wu FZ, Wu MT. Impact of denosumab on cardiovascular calcification in patients with secondary hyperparathyroidism undergoing dialysis: a pilot study. Osteoporos Int. 2020;31:1507–1516. doi: 10.1007/s00198-020-05391-3 [DOI] [PubMed] [Google Scholar]
- 203.Suzuki S, Suzuki M, Hanafusa N, Tsuchiya K, Nitta K. Denosumab recovers aortic arch calcification during long-term hemodialysis. Kidney Int Rep. 2021;6:605–612. doi: 10.1016/j.ekir.2020.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Iseri K, Watanabe M, Yoshikawa H, Mitsui H, Endo T, Yamamoto Y, Iyoda M, Ryu K, Inaba T, Shibata T. Effects of denosumab and alendronate on bone health and vascular function in hemodialysis patients: a randomized, controlled trial. J Bone Miner Res. 2019;34:1014–1024. doi: 10.1002/jbmr.3676 [DOI] [PubMed] [Google Scholar]
- 205.Thongprayoon C, Acharya P, Acharya C, Chenbhanich J, Bathini T, Boonpheng B, Sharma K, Wijarnpreecha K, Ungprasert P, Gonzalez Suarez ML, et al. Hypocalcemia and bone mineral density changes following denosumab treatment in end-stage renal disease patients: a meta-analysis of observational studies. Osteoporos Int. 2018;29:1737–1745. doi: 10.1007/s00198-018-4533-6 [DOI] [PubMed] [Google Scholar]
- 206.Broadwell A, Chines A, Ebeling PR, Franek E, Huang S, Smith S, Kendler D, Messina O, Miller PD. Denosumab safety and efficacy among participants in the FREEDOM extension study with mild to moderate chronic kidney disease. J Clin Endocrinol Metab. 2021;106:397–409. doi: 10.1210/clinem/dgaa851 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Gopaul A, Kanagalingam T, Thain J, Khan T, Cowan A, Sultan N, Clemens KK. Denosumab in chronic kidney disease: a narrative review of treatment efficacy and safety. Arch Osteoporos. 2021;16:116. doi: 10.1007/s11657-021-00971-0 [DOI] [PubMed] [Google Scholar]
- 208.Horikawa A, Hongo M, Kasukawa Y, Shimada Y, Kodama H, Sano A, Miyakoshi N. The relationship between chronic kidney disease and denosumab-induced hypocalcemia in high-age osteoporotic patients. J Bone Miner Metab. 2022;40:670–676. doi: 10.1007/s00774-022-01331-9 [DOI] [PubMed] [Google Scholar]
- 209.Hsu CT, Deng YL, Chung MC, Tsai SF, Lin SY, Chen CH. Integrated osteoporosis care to reduce denosumab-associated hypocalcemia for patients with advanced chronic kidney disease and end-stage renal disease. Healthcare (Basel). 2023;11:313. doi: 10.3390/healthcare11030313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.McClung MR, O’Donoghue ML, Papapoulos SE, Bone H, Langdahl B, Saag KG, Reid IR, Kiel DP, Cavallari I, Bonaca MP, et al. ; LOFT Investigators. Odanacatib for the treatment of postmenopausal osteoporosis: results of the LOFT multicentre, randomised, double-blind, placebo-controlled trial and LOFT Extension study. Lancet Diabetes Endocrinol. 2019;7:899–911. doi: 10.1016/S2213-8587(19)30346-8 [DOI] [PubMed] [Google Scholar]
- 211.McClung MR, Grauer A, Boonen S, Bolognese MA, Brown JP, Diez-Perez A, Langdahl BL, Reginster JY, Zanchetta JR, Wasserman SM, et al. Romosozumab in postmenopausal women with low bone mineral density. New Engl J Med. 2014;370:412–420. doi: 10.1056/NEJMoa1305224 [DOI] [PubMed] [Google Scholar]
- 212.Cosman F, Crittenden DB, Adachi JD, Binkley N, Czerwinski E, Ferrari S, Hofbauer LC, Lau E, Lewiecki EM, Miyauchi A, et al. Romosozumab treatment in postmenopausal women with osteoporosis. N Engl J Med. 2016;375:1532–1543. doi: 10.1056/NEJMoa1607948 [DOI] [PubMed] [Google Scholar]
- 213.Saag KG, Petersen J, Brandi ML, Karaplis AC, Lorentzon M, Thomas T, Maddox J, Fan M, Meisner PD, Grauer A. Romosozumab or alendronate for fracture prevention in women with osteoporosis. N Engl J Med. 2017;377:1417–1427. doi: 10.1056/NEJMoa1708322 [DOI] [PubMed] [Google Scholar]
- 214.Miller PD, Adachi JD, Albergaria BH, Cheung AM, Chines AA, Gielen E, Langdahl BL, Miyauchi A, Oates M, Reid IR, et al. Efficacy and safety of romosozumab among postmenopausal women with osteoporosis and mild-to-moderate chronic kidney disease. J Bone Miner Res. 2022;37:1437–1445. doi: 10.1002/jbmr.4563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Asadipooya K, Weinstock A. Cardiovascular outcomes of romosozumab and protective role of alendronate. Arterioscler Thromb Vasc Biol. 2019;39:1343–1350. doi: 10.1161/ATVBAHA.119.312371 [DOI] [PubMed] [Google Scholar]
- 216.Adami G, Gatti D, Fassio A, Viapiana O, Rossini M. Cardiovascular safety of romosozumab: new insights from postmenopausal women with chronic kidney disease. J Bone Miner Res. 2022;38:354–355. doi: 10.1002/jbmr.4724 [DOI] [PubMed] [Google Scholar]
- 217.Brandenburg VM, Verhulst A, Babler A, D’Haese PC, Evenepoel P, Kaesler N. Sclerostin in chronic kidney disease-mineral bone disorder think first before you block it!. Nephrol Dial Transpl. 2019;34:408–414. doi: 10.1093/ndt/gfy129 [DOI] [PubMed] [Google Scholar]
- 218.De Mare A, Opdebeeck B, Neven E, D’Haese PC, Verhulst A. Sclerostin protects against vascular calcification development in mice. J Bone Miner Res. 2022;37:687–699. doi: 10.1002/jbmr.4503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, et al. ; DECLARE–TIMI 58 Investigators. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347–357. doi: 10.1056/NEJMoa1812389 [DOI] [PubMed] [Google Scholar]
- 220.Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR, et al. ; CANVAS Program Collaborative Group. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644–657. doi: 10.1056/NEJMoa1611925 [DOI] [PubMed] [Google Scholar]
- 221.Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, Mattheus M, Devins T, Johansen OE, Woerle HJ, et al. ; EMPA-REG OUTCOME Investigators. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–2128. doi: 10.1056/NEJMoa1504720 [DOI] [PubMed] [Google Scholar]
- 222.Heerspink HJL, Stefansson BV, Correa-Rotter R, Chertow GM, Greene T, Hou FF, Mann JFE, McMurray JJV, Lindberg M, Rossing P, et al. ; DAPA-CKD Trial Committees and Investigators. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383:1436–1446. doi: 10.1056/NEJMoa2024816 [DOI] [PubMed] [Google Scholar]
- 223.Fonseca-Correa JI, Correa-Rotter R. Sodium-glucose cotransporter 2 inhibitors mechanisms of action: a review. Front Med (Lausanne). 2021;8:777861. doi: 10.3389/fmed.2021.777861 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Vinke JSJ, Heerspink HJL, de Borst MH. Effects of sodium glucose cotransporter 2 inhibitors on mineral metabolism in type 2 diabetes mellitus. Curr Opin Nephrol Hypertens. 2019;28:321–327. doi: 10.1097/MNH.0000000000000505 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Londzin P, Brudnowska A, Kurkowska K, Wilk K, Olszewska K, Ziembinski L, Janas A, Cegiela U, Folwarczna J. Unfavorable effects of sodium-glucose cotransporter 2 (SGLT2) inhibitors on the skeletal system of nondiabetic rats. Biomed Pharmacother. 2022;155:113679. doi: 10.1016/j.biopha.2022.113679 [DOI] [PubMed] [Google Scholar]
- 226.Takiyama Y, Sera T, Nakamura M, Yanagimachi T, Bessho R, Fujita Y, Hoshino M, Uesugi K, Saijo Y, Yagi N, et al. SGLT2 inhibitor therapy improves blood glucose but does not prevent albuminuria nor glomerular hypertrophy in diabetic db/db mice. Diabetes. 2017;66:A134–A134. [Google Scholar]
- 227.Song P, Chen T, Rui S, Duan X, Deng B, Armstrong DG, Ma Y, Deng W. Canagliflozin promotes osteoblastic MC3T3-E1 differentiation via AMPK/RUNX2 and improves bone microarchitecture in type 2 diabetic mice. Front Endocrinol (Lausanne). 2022;13:1081039. doi: 10.3389/fendo.2022.1081039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Fralick M, Kim SC, Schneeweiss S, Kim D, Redelmeier DA, Patorno E. Fracture risk after initiation of use of canagliflozin: a cohort study. Ann Intern Med. 2019;170:155–163. doi: 10.7326/M18-0567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Lou YK, Yu Y, Duan JC, Bi SN, Swe KNC, Xi ZW, Gao YA, Zhou YJ, Nie XM, Liu W. Sodium-glucose cotransporter 2 inhibitors and fracture risk in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Ther Adv Chronic Dis. 2020;11:2040622320961599. doi: 10.1177/2040622320961599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Tsai WC, Hsu SP, Chiu YL, Yang JY, Pai MF, Ko MJ, Tu YK, Hung KY, Chien KL, Peng YS, et al. Cardiovascular and renal efficacy and safety of sodium-glucose cotransporter-2 inhibitors in patients without diabetes: a systematic review and meta-analysis of randomised placebo-controlled trials. BMJ Open. 2022;12:e060655. doi: 10.1136/bmjopen-2021-060655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Kohan DE, Fioretto P, Tang W, List JF. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int. 2014;85:962–971. doi: 10.1038/ki.2013.356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Katakami N, Mita T, Yoshii H, Shiraiwa T, Yasuda T, Okada Y, Torimoto K, Umayahara Y, Kaneto H, Osonoi T, et al. ; UTOPIA Study Investigators. Effect of tofogliflozin on arterial stiffness in patients with type 2 diabetes: prespecified sub-analysis of the prospective, randomized, open-label, parallel-group comparative UTOPIA trial. Cardiovasc Diabetol. 2021;20:4. doi: 10.1186/s12933-020-01206-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Ikonomidis I, Pavlidis G, Thymis J, Birba D, Kalogeris A, Kousathana F, Kountouri A, Balampanis K, Parissis J, Andreadou I, et al. Effects of glucagon-like peptide-1 receptor agonists, sodium-glucose cotransporter-2 inhibitors, and their combination on endothelial glycocalyx, arterial function, and myocardial work index in patients with type 2 diabetes mellitus after 12-month treatment. J Am Heart Assoc. 2020;9:e015716. doi: 10.1161/JAHA.119.015716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.DeFronzo RA, Norton L, Abdul-Ghani M. Renal, metabolic and cardiovascular considerations of SGLT2 inhibition. Nat Rev Nephrol. 2017;13:11–26. doi: 10.1038/nrneph.2016.170 [DOI] [PubMed] [Google Scholar]
- 235.Amat-Santos IJ, Sanchez-Luna JP, Abu-Assi E, Melendo-Viu M, Cruz-Gonzalez I, Nombela-Franco L, Munoz-Garci AJ, Blas SG, de la Torre Hernandez JM, Romaguera R, et al. ; Dapagliflozin after Transcatheter Aortic Valve Implantation (DapaTAVI) Investigators. Rationale and design of the Dapagliflozin After Transcatheter Aortic Valve Implantation (DapaTAVI) randomized trial. Eur J Heart Fail. 2022;24:581–588. doi: 10.1002/ejhf.2370 [DOI] [PubMed] [Google Scholar]
- 236.Pletcher MJ, Pignone M, Earnshaw S, McDade C, Phillips KA, Auer R, Zablotska L, Greenland P. Using the coronary artery calcium score to guide statin therapy: a cost-effectiveness analysis. Circ Cardiovasc Qual Outcomes. 2014;7:276–284. doi: 10.1161/CIRCOUTCOMES.113.000799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Martin SS, Blaha MJ, Blankstein R, Agatston A, Rivera JJ, Virani SS, Ouyang P, Jones SR, Blumenthal RS, Budoff MJ, et al. Dyslipidemia, coronary artery calcium, and incident atherosclerotic cardiovascular disease: implications for statin therapy from the Multi-Ethnic Study of Atherosclerosis. Circulation. 2014;129:77–86. doi: 10.1161/CIRCULATIONAHA.113.003625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Himmelsbach A, Ciliox C, Goettsch C. Cardiovascular calcification in chronic kidney disease-therapeutic opportunities. Toxins (Basel). 2020;12:181. doi: 10.3390/toxins12030181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Salmhofer H, Franzen M, Hitzl W, Koller J, Kreymann B, Fend F, Hauser-Kronberger C, Heemann U, Berr F, Schmaderer C. Multi-modal treatment of calciphylaxis with sodium-thiosulfate, cinacalcet and sevelamer including long-term data. Kidney Blood Press Res. 2013;37:346–359. doi: 10.1159/000350162 [DOI] [PubMed] [Google Scholar]
- 240.Welte T, Arnold F, Technau-Hafsi K, Neumann-Haefelin E, Wobser R, Zschiedrich S, Walz G, Kramer-Zucker A. Successful management of calciphylaxis in a kidney transplant patient: case report. Transplant Direct. 2016;2:e70. doi: 10.1097/TXD.0000000000000582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Akhtar Ali S, Ng C, Votava-Smith JK, Randolph LM, Pitukcheewanont P. Bisphosphonate therapy in an infant with generalized arterial calcification with an ABCC6 mutation. Osteoporos Int. 2018;29:2575–2579. doi: 10.1007/s00198-018-4639-x [DOI] [PubMed] [Google Scholar]
- 242.Amann K. Media calcification and intima calcification are distinct entities in chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:1599–1605. doi: 10.2215/CJN.02120508 [DOI] [PubMed] [Google Scholar]
- 243.McCullough PA, Agrawal V, Danielewicz E, Abela GS. Accelerated atherosclerotic calcification and Monckeberg’s sclerosis: a continuum of advanced vascular pathology in chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:1585–1598. doi: 10.2215/Cjn.01930408 [DOI] [PubMed] [Google Scholar]