Abstract
Utilisation of dissolved organic phosphorus (DOP) by marine microbes as an alternative phosphorus (P) source when phosphate is scarce can help sustain non-Redfieldian carbon:nitrogen:phosphorus ratios and efficient ocean carbon export. However, global spatial patterns and rates of microbial DOP utilisation are poorly investigated. Alkaline phosphatase (AP) is an important enzyme group that facilitates the remineralisation of DOP to phosphate and thus its activity is a good proxy for DOP-utilisation, particularly in P-stressed regions. We present a Global Alkaline Phosphatase Activity Dataset (GAPAD) with 4083 measurements collected from 79 published manuscripts and one database. Measurements are organised into four groups based on substrate and further subdivided into seven size fractions based on filtration pore size. The dataset is globally distributed and covers major oceanic regions, with most measurements collected in the upper 20 m of low-latitude oceanic regions during summer since 1997. This dataset can help support future studies assessing global ocean P supply from DOP utilisation and provide a useful data reference for both field investigations and modelling activities.
Subject terms: Ocean sciences, Element cycles
Background & Summary
Phosphorus (P) is an essential element for marine life1 and the ultimate limiting nutrient of ocean productivity2. Dissolved inorganic phosphorus (DIP), essentially phosphate, is the preferred P source for most microorganisms, but is often scarce in the surface ocean, especially in the North Atlantic Subtropical Gyre and the Mediterranean Sea3–5. Dissolved organic phosphorus (DOP) comprises the majority of the dissolved P pool in the surface open ocean, but is not readily available to many microorganisms6. Alkaline phosphatase (AP), a group of metalloenzymes that catalyses the hydrolysis of a broad spectrum of marine DOP compounds, enables remineralisation of DOP to DIP7,8 and therefore provides the potential to alleviate phosphorus limitation for marine organisms.
Alkaline phosphatase is often induced at extremely low phosphate concentrations, i.e., below a threshold phosphate concentration of ~30 nmol L−1 9, resulting in a high rate of alkaline phosphatase activity (APA) in P-limited oceanic regions10,11. Therefore, APA is an important indicator of P-limitation and a useful proxy to gauge DOP-utilisation by marine microorganisms12. Studies quantifying APA started in the 1970s13 and have greatly improved our understanding of the marine phosphorus cycle. To facilitate better understanding of the role of AP in P supply via microbial DOP-utilisation, we present a Global Alkaline Phosphatase Activity Dataset (GAPAD) including 4083 measurements during the last 50 years, with 4051 measurements from 79 published manuscripts and 32 measurements from 1 database14. Global Alkaline Phosphatase Activity Dataset is the most comprehensive dataset published thus far since it includes not only APA measurements from the global tropical and subtropical oceans, but also their temporal and spatial information, as well as relevant environmental parameters including dissolved inorganic and organic phosphorus concentrations, chlorophyll a concentration, salinity and temperature14. The workflow of the GAPAD compilation is shown in Fig. 1.
Fig. 1.

Work-flow of GAPAD compilation, standardization and quality-control.
Four substrates have been used to measure APA in GAPAD, i.e., 4-methylumbelliferyl phosphate (MUF-P), 6, 8-difluoro-4-methylumbelliferylphosphate (DiFMUP), 3-O-methylfluorescein phosphate (MFP), and paranitrophenyl phosphate (pNPP) (Fig. 2). There are respectively 2919, 232, 233 and 699 measurements collected from 54, 10, 6 and 9 studies applying MUF-P, DiFMUP, MFP and pNPP as substrates14. Although minor differences exist in their experimental methods, rates are often measured at saturating substrate concentrations to obtain the potential activity15, except when concentrations between 30 and 100 nmol L−1 are used to represent in-situ substrate concentration16–19. Furthermore, we have applied statistical methods to flag outliers in order to improve the quality of the dataset (Table 1). The majority of the APA measurements are within a latitudinal span of 50°S–50°N, with a higher density in the northern hemisphere (Fig. 2a). The sampling depths range from 0 to 4000 m, with most sampling depths located within 20 m of the surface (Fig. 2b). Measurements were performed between years 1971 and 2019 (Fig. 2c), and there are more measurements in summer months (400–600 per month) compared to winter months (~200 per month; Fig. 2d).
Fig. 2.
APA measurement distributions in the ocean. (a) Latitudinal, (b) Vertical, (c) Yearly, and (d) Monthly distributions of APA measurements with each substrate colored. Blue, orange, yellow and purple bars represent measurements with the substrates MUF-P, DiFMUP, MFP and pNPP, respectively.
Table 1.
Results of the outlier identification method applying to the substrate MUF-P. Definitions of the different fractions of APA in this table are described in the Methods section.
| APA | Number of measurements | Number of identified outliers | before/after outlier identification (nmol L−1 h−1) | slog before/after outlier identification (nmol L−1 h−1) |
|---|---|---|---|---|
| Bulk | 2266 | 0 | 0.89/0.89 | 1.19/1.19 |
| Dissolved | 292 | 0 | 0.49/0.49 | 1.21/1.21 |
| Particulate | 116 | 2 | 0.0045/−0.047 | 0.97/0.89 |
| Bacterial | 92 | 0 | 1.31/1.31 | 0.77/0.77 |
| Phytoplankton | 93 | 0 | 1.78/1.78 | 0.76/0.76 |
| Trichodesmium | 30 | 0 | 0.37/0.37 | 1.67/1.67 |
: log10-transformed mean values.
slog : log10-transformed standard deviation values.
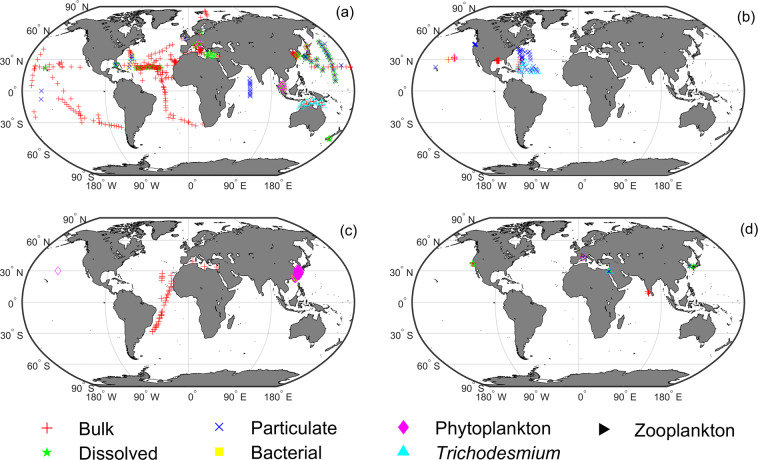
Alkaline phosphatase activity measured with the substrate MUF-P is the most common and widely distributed in global oceans (Fig. 3a). In the North Atlantic and the Northeast Pacific oceans, APA was measured with the substrate DiFMUP, with fractions of particulate APA, phytoplankton APA, and Trichodesmium APA mostly measured in the North Atlantic (Fig. 3b). Average bulk APA (APA measured with unfiltered water) rates in the North Atlantic (2.49 ± 2.34 nmol L−1 h−1, n = 77, mean ± SD) are higher than in the eastern Pacific (0.84 ± 0.38 nmol L−1 h−1, n = 4; Fig. 4b). For the MFP substrate, bulk APA rates are available in the Mediterranean Sea and the Atlantic, while phytoplankton APA was only measured in the East China Sea (Fig. 3c). All APA measurements with pNPP as the substrate are from coastal waters of the Pacific, the Indian Ocean and the Mediterranean Sea (Fig. 3d).
Fig. 3.
Global distribution of APA measurements in GAPAD for the different four substrates. (a) MUF-P, (b) DiFMUP, (c) MFP and (d) pNPP. Each marker represents a fraction, with red plus, green pentagram, blue cross, yellow square, magenta diamond, cyan upward-pointing triangle and black right-pointing triangle representing bulk APA, dissolved APA, particulate APA, bacterial APA, phytoplankton APA, Trichodesmium APA, and zooplankton APA respectively. Definitions of the different fractions of APA in this figure are described in the Methods section.
Fig. 4.
Global distribution of Log10-transformed full-depth-averaged bulk APA measurements in GAPAD for the four different substrates. (a) MUF-P, (b) DiFMUP, (c) MFP and (d) pNPP.
We have also divided APA measured with each substrate according to different fraction types, i.e., bulk APA, dissolved APA, particulate APA, bacterial APA, phytoplankton APA, Trichodesmium APA, and zooplankton APA (Fig. 3). Since APA measured with the substrate MUF-P is the most abundant and widely distributed in GAPAD (Fig. 3a), with bulk APA covering a large part of the Atlantic, the Pacific, and the Mediterranean Sea, we further analyse their distributions and rates (Figs. 4a, 5). The bulk APA rates near the coasts (161.96 ± 523.03 nmol L−1 h−1, n = 1528, defined as water depth less than 1000m in this study) are generally higher than those in the open ocean (2.60 ± 6.94 nmol L−1 h−1, n = 749, defined as water depth>1000m). The highest APA rate (6583 nmol L−1 h−1) is in the northern Adriatic Sea (Fig. 4a). Dissolved APA have been measured in the Northwest Pacific, the Mediterranean Sea, and the North Atlantic (Fig. 5a), whereas particulate APA has been measured in the Northwest Pacific, the Equatorial west Atlantic and the Indian Ocean (Fig. 5b). Bacterial APA has been measured in the North Atlantic and the South China Sea (Fig. 5c), whereas the phytoplankton APA has also been measured mainly in the South China Sea, Adriatic Sea and Bay of Biscay (Fig. 5d) and Trichodesmium APA has been measured in the North Atlantic and near the coast of northern Australia in GAPAD (Fig. 5e).
Fig. 5.
Global distribution of Log10-transformed full-depth-averaged APA measurement of MUF-P in GAPAD for the five different fractions. (a) dissolved APA, (b) particulate APA, (c) bacterial APA, (d) phytoplankton APA and (e) Trichodesmium APA.
In oligotrophic marine environments, AP may contribute a large fraction of DOP utilisation and is therefore important for supporting the non-Redfieldian carbon:nitrogen:phosphorus (C:N:P) ratios of marine organisms and marine carbon export20. Due to the important role of AP in alleviating P-limitation for diazotrophs and supporting N2 fixation, it may also control ecological diversity by giving them an ecological advantage when competing for resources with non-diazotrophs9,20–24. Global Alkaline Phosphatase Activity Dataset will provide a new resource for the study of the global ocean phosphorus cycling, further elucidating impacts on these critical processes.
Methods
Four substrates have been used in the APA measurements, i.e., MUF-P8,9,15–19,25–72, DiFMUP10,12,73–80, MFP13,81–85 and pNPP86–94. Since a fluorescent (or colored) product is released when a substrate is hydrolysed by AP in a seawater sample, APA can be measured by detecting the changes in fluorescence (or color) over time. Measurements were mostly carried out with unfiltered water (bulk APA) and two pre-filtrations with filter sizes of 0.22 µm and 3 µm (size-fractionated APA). The dissolved fraction is often identified as <0.22 µm, even though this might contain nanoparticles, colloidal nanogels and/or viruses95. The particulate fraction is usually identified as >0.22 µm30, except in a few studies in this compilation75,87,88, using >0.25 µm or >0.4 µm. The bacterial fraction, containing heterotrophic bacteria and picocyanobacteria, is often identified as 0.22–3 µm40, except Duhamel et al., Lim et al. and Bogé et al., who used 0.2–0.6 µm12 or 0.2–0.8 µm36, 0.2–2 µm45 and 0.25–5 µm87, respectively. The phytoplankton fraction is often from samples prefiltered through meshes of different pore sizes, e.g., 120 µm83, 200 µm41 and 1 mm81,82 to remove zooplankton. Lim et al. and Bogé et al. identify the phytoplankton fraction as 2–20 µm45 and 5–90 µm87, respectively. Several studies also identify a Trichodesmium fraction49,51,76,94,96,97 and a zooplankton fraction (>90 µm)86–88.
For samples collected on filters with different pore sizes, samples are usually re-suspended in sterile phosphate-free artificial seawater10 or autoclaved pre-filtered seawater47 for several minutes before the start of the experiment. Standard fluorescent products, e.g., MUF (methylumbelliferone), with concentrations typically ranging from 0 to 2000 nM are used to produce the standard curve for converting the rate of change in fluorescence to a substrate hydrolysis rate32. Fluorescence is measured using a fluorometer immediately after substrate addition and at regular intervals (e.g., 30 min). The rate of APA is derived from the changes of fluorescence over time and converted to hydrolysis rate using the calibration curve. To improve the accuracy of the calculation, seawater blanks, boiled samples or ultrapure water are used to correct fluorescence measurements and account for abiotic substrate hydrolysis or degradation12,32,35. Enzyme-kinetic parameters (Michaelis-Menten parameters including the maximum hydrolysis rate (Vmax), and the half-saturation constant (Km)) are also determined in some studies using data from incubations of different substrate concentrations in unfiltered seawater35.
The APA data have been collected by searching published manuscripts with key words ‘alkaline phosphatase; alkaline phosphatase activity; AP; APA; ocean; coast’ in multiple academic service platforms, i.e., the Web of Science (https://www.webofscience.com/), the China National Knowledge Infrastructure (CNKI, https://www.cnki.net/), and the Wanfang Data Knowledge Service Platform (https://www.wanfangdata.com.cn/), as well as available databases, i.e., the Biological & Chemical Oceanography Data Management Office (BCO-DMO) and the British Oceanographic Data Centre (BODC). We reported APA measurements in environmental samples and combined all available measurements to create the most comprehensive global coverage of in-situ APA with the procedures described in Fig. 1. Most data have been obtained directly from the figures and tables in the published manuscripts. Data that could not be obtained directly have been digitized from figures using the Engauge Digitizer 12.1 software or provided by the authors on request25,28,33,37–41,46,47,54,55,82,98,99. Some authors provided unpublished data from their dissertations100–102, which are then included in GAPAD. Data presented in appendices of published manuscripts are also included in this compilation11.
The units of APA are often reported as volumetric rates, e.g., nmol L−1 h−1, µmol L−1 h−1, or nmol L−1 min−1. However, some APA measurements are normalized to other parameters, e.g., chlorophyll a concentration (pmol µg Chl−1 min−1)81,82, cell abundance (nmol cell−1 h−1)12 or Trichodesmium colony abundance (nmol colony−1 h−1)51. We unified the units to the volumetric rates by multiplying them by the in-situ concentrations of the respective parameters. Finally, we transformed all units to nmol L−1 h−1.
Data Records
Global Alkaline Phosphatase Activity Dataset is included in 4 sheets of a dataset file according to substrate type, i.e., MUF-P, DiFMUP, MFP, and pNPP. Each sheet includes the following fields for each record:
Source of data
Latitude (−90° to 90°)
Longitude (−180° to 180°)
Sampling depth (m)
Cruise
Site/Station
Year
Month
APA (nmol L−1 h−1)
Bulk
Dissolved
Particulate
Bacteria
Phytoplankton
Trichodesmium
Zooplankton
Dissolved Inorganic Nitrogen (nmol L−1)
Dissolved Inorganic Phosphorus (nmol L−1)
Dissolved Organic Phosphorus (nmol L−1)
Chlorophyll a (µg L−1)
Colony abundance (colony L−1)
Cell abundance (cell L−1)
Salinity (psu)
Temperature (°C)
Alkaline phosphatase activity measurements are subdivided into seven fractions according to their filtration sizes as outlined in the Methods section described above. In addition, environmental parameters reported to potentially impact rates of APA are also included whenever they are available in published articles or databases, and a summary of detailed sources of APA data is on sheet 5 of the dataset file. The dataset file in Excel Workbook (xlsx) format can be accessed on Figshare using the link (10.6084/m9.figshare.c.6340244.v1)14. ‘– 999’ denotes missing data. The dataset will be updated by the authors when new data are available.
Technical Validation
Alkaline phosphatase activity in the ocean ranges from below the detection limit (denoted by 0, e.g. <=0.002 nmol L-1 h-1 in Yamaguichi et al.68) to very high rates as much as 6583 nmol L−1 h−1 for MUF-P, which is largely controlled by ambient DIP concentration and DOP availability9,36,44. Therefore, APA rates are not normally distributed and show a positively skewed distribution with long tails of high values. However, the collected APA rates are approximately log-normally distributed after excluding the data points of zero.
In order to control the quality of GAPAD, we applied the Chauvenet’s criterion to identify suspicious outliers whose probability of deviation from the mean is less than 1/(2n)103, where n is the number of measurements. Since the APA rates are approximately log-normal distributed, the method is only applied to the log-transformed non-zero data. We use the MATLAB norminv function to calculate the critical value (xlog*) with the mean , the standard deviation slog, and the evaluated probability values in p, where p is calculated from 1-1/(4n) instead of 1/(2n), because the Chauvenet’s criterion is a two-tailed test and only data at the tail with high values will be identified. Then data points with values larger than the critical value xlog* will be flagged. In this study, we apply the method only once in each of the seven fractions of the four groups categorized by substrate respectively, except when it has less than 20 measurements.
We accept all the data which are not flagged by the Chauvenet’s criterion. For the flagged suspicious outliers, we determine whether to exclude them from GAPAD or not after carefully assessing their values to validate that they are very skewed from the approximate log-normal distribution. The results of the quality control applied following this approach are shown in Table 1.
Usage Note
Global alkaline phosphatase activity dataset can serve as a reference to field investigators for assessing their results, and to biogeochemical modelling scientists for model validation. With our APA dataset, the role of environmental factors affecting APA can also be examined to understand the role of global ocean phosphate supply from AP-catalysed DOP utilisation in response to future climate change.
Supplementary information
Acknowledgements
We gratefully acknowledge the very helpful comments from our two anonymous reviewers. We want to thank all the researchers for sharing the data with us and making the compilation of this dataset possible, as well as all the staff of the Biological & Chemical Oceanography Data Management Office (BCO-DMO) and the British Oceanographic Data Centre (BODC) for enabling the access of the data.We would like to thank Dr. Dan Wang, Dr. Gwo-Ching Gong, Prof. Jeng Chang and Prof. Olivier Wurl for their assistance in compiling this dataset. We also acknowledge Dr. Markus Pahlow for his help in improving the writing language of the original manuscript. This research is jointly funded by the Southern Marine Science and Engineering Guangdong Laboratory (Zhuhai) (No. SML2020SP008), the National Key Research and Development Program of China (No. 2020YFA0608304) and the “Fundamental Research Fund of Shandong University” granted to Bei Su.
Author contributions
B.S. and C.M. conceived the study. X.R.S. and B.S. collected metadata, and compiled and analysed the dataset. B.S. and X.R.S. wrote a draft and made figures with substantial inputs from S.D., C.M., C.D., I.I. and J.H.L.
Code availability
The source codes for identifying outliers used in this paper are available at https://github.com/BGM-USD2020/GAPAD_codes.git.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41597-023-02081-7.
References
- 1.Karl DM. Phosphorus, the staff of life. Nature. 2000;406:31–33. doi: 10.1038/35017683. [DOI] [PubMed] [Google Scholar]
- 2.Tyrrell T. The relative influences of nitrogen and phosphorus on oceanic primary production. Nature. 1999;400:525–531. doi: 10.1038/22941. [DOI] [Google Scholar]
- 3.Powley HR, Krom MD, Van Cappellen P. Understanding the unique biogeochemistry of the Mediterranean Sea: Insights from a coupled phosphorus and nitrogen model. Global Biogeochem. Cycles. 2017;31:1010–1031. doi: 10.1002/2017GB005648. [DOI] [Google Scholar]
- 4.Barcelos e Ramos J, et al. Nutrient-specific responses of a phytoplankton community: a case study of the North Atlantic Gyre, Azores. J. Plankton Res. 2017;39:744–761. doi: 10.1093/plankt/fbx025. [DOI] [Google Scholar]
- 5.Pulido-Villena E, et al. Phosphorus cycling in the upper waters of the Mediterranean Sea (PEACETIME cruise): relative contribution of external and internal sources. Biogeosciences. 2021;18:5871–5889. doi: 10.5194/bg-18-5871-2021. [DOI] [Google Scholar]
- 6.Karl, D. M. & Björkman, K. M. in Biogeochemistry of Marine Dissolved Organic Matter (Second Edition) (eds D. A., Hansell & C. A., Carlson) 233–334 (Academic Press, 2015).
- 7.Hoppe HG. Phosphatase activity in the sea. Hydrobiologia. 2003;493:187–200. doi: 10.1023/A:1025453918247. [DOI] [Google Scholar]
- 8.Srivastava A, et al. Enzyme promiscuity in natural environments: alkaline phosphatase in the ocean. ISME J. 2021;15:3375–3383. doi: 10.1038/s41396-021-01013-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mahaffey C, Reynolds S, Davis CE, Lohan MC. Alkaline phosphatase activity in the subtropical ocean: insights from nutrient, dust and trace metal addition experiments. Front. Mar. Sci. 2014;1:73. doi: 10.3389/fmars.2014.00073. [DOI] [Google Scholar]
- 10.Dyhrman ST, Ruttenberg KC. Presence and regulation of alkaline phosphatase activity in eukaryotic phytoplankton from the coastal ocean: Implications for dissolved organic phosphorus remineralization. Limnol. Oceanogr. 2006;51:1381–1390. doi: 10.4319/lo.2006.51.3.1381. [DOI] [Google Scholar]
- 11.Lomas MW, et al. Sargasso Sea phosphorus biogeochemistry: an important role for dissolved organic phosphorus (DOP) Biogeosciences. 2010;7:695–710. doi: 10.5194/bg-7-695-2010. [DOI] [Google Scholar]
- 12.Duhamel S, Dyhrman ST, Karl DM. Alkaline phosphatase activity and regulation in the North Pacific Subtropical Gyre. Limnol. Oceanogr. 2010;55:1414–1425. doi: 10.4319/lo.2010.55.3.1414. [DOI] [Google Scholar]
- 13.Perry MJ. Alkaline phosphatase activity in subtropical Central North Pacific waters using a sensitive fluorometric method. Mar. Biol. 1972;15:113–119. doi: 10.1007/BF00353639. [DOI] [Google Scholar]
- 14.Su B, 2023. A dataset of global ocean alkaline phosphotase activity. Figshare. [DOI] [PMC free article] [PubMed]
- 15.Baltar F, Arístegui J, Gasol JM, Yokokawa T, Herndl GJ. Bacterial versus archaeal origin of extracellular enzymatic activity in the Northeast Atlantic deep waters. Microb. Ecol. 2013;65:277–288. doi: 10.1007/s00248-012-0126-7. [DOI] [PubMed] [Google Scholar]
- 16.Baltar F, Legrand C, Pinhassi J. Cell-free extracellular enzymatic activity is linked to seasonal temperature changes: a case study in the Baltic Sea. Biogeosciences. 2016;13:2815–2821. doi: 10.5194/bg-13-2815-2016. [DOI] [Google Scholar]
- 17.Boge G, Lespilette M, Jamet D, Jamet JL. Role of sea water DIP and DOP in controlling bulk alkaline phosphatase activity in N.W. Mediterranean Sea (Toulon, France) Mar. Pollut. Bull. 2012;64:1989–1996. doi: 10.1016/j.marpolbul.2012.07.028. [DOI] [PubMed] [Google Scholar]
- 18.Sisma-Ventura G, Rahav E. DOP stimulates heterotrophic bacterial production in the oligotrophic southeastern Mediterranean coastal waters. Front. Microbiol. 2019;10:1913. doi: 10.3389/fmicb.2019.01913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang D, Huang B, Liu X, Liu G, Wang H. Seasonal variations of phytoplankton phosphorus stress in the Yellow Sea Cold Water Mass. Acta Oceanologica Sinica. 2014;33:124–135. doi: 10.1007/s13131-014-0547-x. [DOI] [Google Scholar]
- 20.Duhamel S, et al. Phosphorus as an integral component of global marine biogeochemistry. Nat. Geosci. 2021;14:359–368. doi: 10.1038/s41561-021-00755-8. [DOI] [Google Scholar]
- 21.Letscher RT, Moore JK. Preferential remineralization of dissolved organic phosphorus and non-Redfield DOM dynamics in the global ocean: Impacts on marine productivity, nitrogen fixation, and carbon export. Global Biogeochem. Cycles. 2015;29:325–340. doi: 10.1002/2014GB004904. [DOI] [Google Scholar]
- 22.Monteiro FM, Follows MJ. On nitrogen fixation and preferential remineralization of phosphorus. Geophys. Res. Lett. 2012;39:L04606. doi: 10.1029/2012GL050897. [DOI] [Google Scholar]
- 23.Somes CJ, Oschlies A. On the influence of “non-Redfield” dissolved organic nutrient dynamics on the spatial distribution of N2 fixation and the size of the marine fixed nitrogen inventory. Global Biogeochem. Cycles. 2015;29:973–993. doi: 10.1002/2014GB005050. [DOI] [Google Scholar]
- 24.Wang WL, Moore JK, Martiny AC, Primeau FW. Convergent estimates of marine nitrogen fixation. Nature. 2019;566:205–211. doi: 10.1038/s41586-019-0911-2. [DOI] [PubMed] [Google Scholar]
- 25.Ayo B, et al. Imbalanced nutrient recycling in a warmer ocean driven by differential response of extracellular enzymatic activities. Glob Chang Biol. 2017;23:4084–4093. doi: 10.1111/gcb.13779. [DOI] [PubMed] [Google Scholar]
- 26.Azzaro M, et al. Prokaryotic dynamics and heterotrophic metabolism in a deep convection site of Eastern Mediterranean Sea (the Southern Adriatic Pit) Cont Shelf Res. 2012;44:106–118. doi: 10.1016/j.csr.2011.07.011. [DOI] [Google Scholar]
- 27.Boge G, Lespilette M, Jamet D, Jamet JL. Role of DOP on the alkaline phosphatase activity of size fractionated plankton in coastal waters in the NW Mediterranean Sea (Toulon Bay, France) Mar. Pollut. Bull. 2017;117:264–273. doi: 10.1016/j.marpolbul.2016.11.037. [DOI] [PubMed] [Google Scholar]
- 28.Caruso G. Leucine aminopeptidase, β-glucosidase and alkaline phosphatase activity rates and their significance in nutrient cycles in some coastal Mediterranean sites. Marine Drugs. 2010;8:916–940. doi: 10.3390/md8040916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Caruso G, et al. Patterns of prokaryotic activities and abundance among the epi-meso and bathypelagic zones of the southern-central Tyrrhenian Sea. Oceanography. 2013;1:105. [Google Scholar]
- 30.Cotner JB, Ammerman JW, Peele ER, Bentzen E. Phosphorus-limited bacterioplankton growth in the Sargasso Sea. Aquat. Microb. Ecol. 1997;13:141–149. doi: 10.3354/ame013141. [DOI] [Google Scholar]
- 31.Danovaro R, et al. Exo-enzymatic activities and dissolved organic pools in relation with mucilage development in the Northern Adriatic Sea. Sci. Total Environ. 2005;353:189–203. doi: 10.1016/j.scitotenv.2005.09.029. [DOI] [PubMed] [Google Scholar]
- 32.Davis CE, Mahaffey C. Elevated alkaline phosphatase activity in a phosphate-replete environment: Influence of sinking particles. Limnol. Oceanogr. 2017;62:2389–2403. doi: 10.1002/lno.10572. [DOI] [Google Scholar]
- 33.Davis C, et al. Diurnal variability in alkaline phosphatase activity and the potential role of zooplankton. Limnology and Oceanography Letters. 2019;4:71–78. doi: 10.1002/lol2.10104. [DOI] [Google Scholar]
- 34.Downes PP, et al. Phosphorus dynamics in the Barents Sea. Limnol. Oceanogr. 2020;66:S326–342. [Google Scholar]
- 35.Duhamel S, Björkman KM, Van Wambeke F, Moutin T, Karl DM. Characterization of alkaline phosphatase activity in the North and South Pacific Subtropical Gyres: Implications for phosphorus cycling. Limnol. Oceanogr. 2011;56:1244–1254. doi: 10.4319/lo.2011.56.4.1244. [DOI] [Google Scholar]
- 36.Duhamel S, Kim E, Sprung B, Anderson OR. Small pigmented eukaryotes play a major role in carbon cycling in the P-depleted western subtropical North Atlantic, which may be supported by mixotrophy. Limnol. Oceanogr. 2019;64:2424–2440. doi: 10.1002/lno.11193. [DOI] [Google Scholar]
- 37.Hashihama F, et al. Arsenate and microbial dynamics in different phosphorus regimes of the subtropical Pacific Ocean. Prog. Oceanogr. 2019;176:102115. doi: 10.1016/j.pocean.2019.05.007. [DOI] [Google Scholar]
- 38.Hashihama F, et al. Biogeochemical controls of particulate phosphorus distribution across the oligotrophic subtropical Pacific Ocean. Global Biogeochem. Cycles. 2020;34:1–15. doi: 10.1029/2020GB006669. [DOI] [Google Scholar]
- 39.Ivančić I, et al. Alkaline phosphatase activity in relation to nutrient status in the northern Adriatic Sea. Mar. Ecol. Prog. Ser. 2009;378:27–35. doi: 10.3354/meps07851. [DOI] [Google Scholar]
- 40.Ivančić I, et al. Phytoplankton and bacterial alkaline phosphatase activity in the northern Adriatic Sea. Mar. Environ. Res. 2010;69:85–94. doi: 10.1016/j.marenvres.2009.08.004. [DOI] [PubMed] [Google Scholar]
- 41.Ivančić I, et al. Alkaline phosphatase activity related to phosphorus stress of microphytoplankton in different trophic conditions. Prog. Oceanogr. 2016;146:175–186. doi: 10.1016/j.pocean.2016.07.003. [DOI] [Google Scholar]
- 42.Koch MS, Kletou DC, Tursi R. Alkaline phosphatase activity of water column fractions and seagrass in a tropical carbonate estuary, Florida Bay. Estuar. Coast. Shelf Sci. 2009;83:403–413. doi: 10.1016/j.ecss.2009.04.007. [DOI] [Google Scholar]
- 43.Koike I, Nagata T. High potential activity of extracellular alkaline phosphatase in deep waters of the central Pacific. Deep Sea Res. Part II Top. Stud. Oceanogr. 1997;44:2283–2294. doi: 10.1016/S0967-0645(97)00025-8. [DOI] [Google Scholar]
- 44.Landolfi A, Dietze H, Volpe G. Longitudinal variability of organic nutrients in the North Atlantic subtropical gyre. Deep Sea Res. Part I Oceanogr. Res. Pap. 2016;111:50–60. doi: 10.1016/j.dsr.2015.11.009. [DOI] [Google Scholar]
- 45.Lim JH, et al. Distributions of particulate and dissolved phosphorus in aquatic habitats of Peninsular Malaysia. Mar. Pollut. Bull. 2018;128:415–427. doi: 10.1016/j.marpolbul.2018.01.037. [DOI] [PubMed] [Google Scholar]
- 46.Martin P, Dyhrman ST, Lomas MW, Poulton NJ, Van Mooy BA. Accumulation and enhanced cycling of polyphosphate by Sargasso Sea plankton in response to low phosphorus. Proc. Natl. Acad. Sci. USA. 2014;111:8089–8094. doi: 10.1073/pnas.1321719111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Martin P, et al. Particulate polyphosphate and alkaline phosphatase activity across a latitudinal transect in the tropical Indian Ocean. Limnol. Oceanogr. 2018;63:1395–1406. doi: 10.1002/lno.10780. [DOI] [Google Scholar]
- 48.Mather RL, et al. Phosphorus cycling in the North and South Atlantic Ocean subtropical gyres. Nat. Geosci. 2008;1:439–443. doi: 10.1038/ngeo232. [DOI] [Google Scholar]
- 49.Mulholland MR, Floge S, Carpenter EJ, Capone DG. Phosphorus dynamics in cultures and natural populations of Trichodesmium spp. Mar. Ecol. Prog. Ser. 2002;239:45–55. doi: 10.3354/meps239045. [DOI] [Google Scholar]
- 50.Nausch M. Alkaline phosphatase activities and the relationship to inorganic phosphate in the Pomeranian Bight (southern Baltic Sea) Aquat. Microb. Ecol. 1998;16:87–94. doi: 10.3354/ame016087. [DOI] [PubMed] [Google Scholar]
- 51.Orcutt KM, Gundersen K, Ammerman JW. Intense ectoenzyme activities associated with Trichodesmium colonies in the Sargasso Sea. Mar. Ecol. Prog. Ser. 2013;478:101–113. doi: 10.3354/meps10153. [DOI] [Google Scholar]
- 52.Rees AP, et al. Alkaline phosphatase activity in the western English Channel: Elevations induced by high summertime rainfall. Estuar. Coast. Shelf Sci. 2009;81:569–574. doi: 10.1016/j.ecss.2008.12.005. [DOI] [Google Scholar]
- 53.Reynolds S, Mahaffey C, Roussenov V, Williams RG. Evidence for production and lateral transport of dissolved organic phosphorus in the eastern subtropical North Atlantic. Global Biogeochem. Cycles. 2014;28:805–824. doi: 10.1002/2013GB004801. [DOI] [Google Scholar]
- 54.Sato M, Sakuraba R, Hashihama F. Phosphate monoesterase and diesterase activities in the North and South Pacific Ocean. Biogeosciences. 2013;10:7677–7688. doi: 10.5194/bg-10-7677-2013. [DOI] [Google Scholar]
- 55.Sebastián M, Arístegui J, Montero MF, Escanez J, Xavier Niell F. Alkaline phosphatase activity and its relationship to inorganic phosphorus in the transition zone of the North-western African upwelling system. Prog. Oceanogr. 2004;62:131–150. doi: 10.1016/j.pocean.2004.07.007. [DOI] [Google Scholar]
- 56.Sohm JA, Capone DG. Phosphorus dynamics of the tropical and subtropical North Atlantic: Trichodesmium spp. versus bulk plankton. Mar. Ecol. Prog. Ser. 2006;317:21–28. doi: 10.3354/meps317021. [DOI] [Google Scholar]
- 57.Suzumura M, Hashihama F, Yamada N, Kinouchi S. Dissolved phosphorus pools and alkaline phosphatase activity in the euphotic zone of the western North Pacific Ocean. Front. Microbiol. 2012;3:99. doi: 10.3389/fmicb.2012.00099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tamburini C, Garcin J, Ragot M, Bianchi A. Biopolymer hydrolysis and bacterial production under ambient hydrostatic pressure through a 2000m water column in the NW Mediterranean. Deep Sea Res. Part II Top. Stud. Oceanogr. 2002;49:2109–2123. doi: 10.1016/S0967-0645(02)00030-9. [DOI] [Google Scholar]
- 59.Tamburini C, et al. Distribution and activity of bacteria and archaea in the different water masses of the Tyrrhenian Sea. Deep Sea Res. Part II Top. Stud. Oceanogr. 2009;56:700–712. doi: 10.1016/j.dsr2.2008.07.021. [DOI] [Google Scholar]
- 60.Thomson B, Hepburn CD, Lamare M, Baltar F. Temperature and UV light affect the activity of marine cell-free enzymes. Biogeosciences. 2017;14:3971–3977. doi: 10.5194/bg-14-3971-2017. [DOI] [Google Scholar]
- 61.Thomson B, et al. Resolving the paradox: Continuous cell-free alkaline phosphatase activity despite high phosphate concentrations. Mar. Chem. 2019;214:103671. doi: 10.1016/j.marchem.2019.103671. [DOI] [Google Scholar]
- 62.Thomson B, et al. Relative importance of phosphodiesterase vs. phosphomonoesterase (alkaline phosphatase) activities for dissolved organic phosphorus hydrolysis in epi- and mesopelagic waters. Front. Earth Sci. 2020;8:560893. doi: 10.3389/feart.2020.560893. [DOI] [Google Scholar]
- 63.Tsagaraki TM, et al. Atmospheric deposition effects on plankton communities in the eastern Mediterranean: A mesocosm experimental approach. Front. Mar. Sci. 2017;4:210. doi: 10.3389/fmars.2017.00210. [DOI] [Google Scholar]
- 64.Van Wambeke, F., Christaki, U., Giannakourou, A., Moutin, T. & Souvemerzoglou, K. Longitudinal and vertical trends of bacterial limitation by phosphorus and carbon in the Mediterranean Sea. Microb. Ecol.43, 119-133 (2002). [DOI] [PubMed]
- 65.Van Wambeke F, Ghiglione JF, Nedoma J, Mével G, Raimbault P. Bottom up effects on bacterioplankton growth and composition during summer-autumn transition in the open NW Mediterranean Sea. Biogeosciences. 2009;6:705–720. doi: 10.5194/bg-6-705-2009. [DOI] [Google Scholar]
- 66.Wurl O, Shelley RU, Landing WM, Cutter GA. Biogeochemistry of dissolved arsenic in the temperate to tropical North Atlantic Ocean. Deep Sea Res. Part II Top. Stud. Oceanogr. 2015;116:240–250. doi: 10.1016/j.dsr2.2014.11.008. [DOI] [Google Scholar]
- 67.Yamaguchi T, et al. Basin-Scale variations in labile dissolved phosphoric monoesters and diesters in the central North Pacific Ocean. J. Geophys. Res. Oceans. 2019;124:3058–3072. doi: 10.1029/2018JC014763. [DOI] [Google Scholar]
- 68.Yoshimura T, Kudo I. Seasonal phosphorus depletion and microbial responses to the change in phosphorus availability in a subarctic coastal environment. Mar. Chem. 2011;126:182–192. doi: 10.1016/j.marchem.2011.06.003. [DOI] [Google Scholar]
- 69.Zaccone R, et al. Seasonal changes on microbial metabolism and biomass in the euphotic layer of Sicilian Channel. Mar. Environ. Res. 2015;112:20–32. doi: 10.1016/j.marenvres.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 70.Zhang X, et al. Seasonal dynamics of phytoplankton phosphorus stress in temperate Jiaozhou Bay, North China. Cont Shelf Res. 2021;231:104602. doi: 10.1016/j.csr.2021.104602. [DOI] [Google Scholar]
- 71.Zhao L, Tian J, Yu J, Yang G, Jia H. Horizontal and vertical variations of activities of extracellular enzymes in the seawater of the Yellow Sea and the Bohai Sea in late autumn, 2013. Chinese Environmental Science. 2015;35:2171–2181. [Google Scholar]
- 72.Zohary T, Robarts RD. Experimental study of microbial P limitation in the eastern Mediterranean. Limnol. Oceanogr. 1998;43:387–395. doi: 10.4319/lo.1998.43.3.0387. [DOI] [Google Scholar]
- 73.Diaz JM, et al. Polyphosphate dynamics at Station ALOHA, North Pacific subtropical gyre. Limnol. Oceanogr. 2016;61:227–239. doi: 10.1002/lno.10206. [DOI] [Google Scholar]
- 74.Dyhrman S, Jenkins B, Rynearson T. Alkaline phosphatase activities for in situ and incubation samples from RV/Atlantic Explorer cruise AE1812 cruise transect from Bermuda to Rhode Island in May 2018. Biological and Chemical Oceanography Data Management Office (BCO-DMO) 2020 doi: 10.26008/1912/bco-dmo.739973.1. [DOI] [Google Scholar]
- 75.Jakuba RW, Moffett JW, Dyhrman ST. Evidence for the linked biogeochemical cycling of zinc, cobalt, and phosphorus in the western North Atlantic Ocean. Global Biogeochem. Cycles. 2008;22:GB4012. doi: 10.1029/2007GB003119. [DOI] [Google Scholar]
- 76.Orchard ED, Ammerman JW, Lomas MW, Dyhrman ST. Dissolved inorganic and organic phosphorus uptake in Trichodesmium and the microbial community: The importance of phosphorus ester in the Sargasso Sea. Limnol. Oceanogr. 2010;55:1390–1399. doi: 10.4319/lo.2010.55.3.1390. [DOI] [Google Scholar]
- 77.Quigg A, et al. Going west: Nutrient limitation of primary production in the northern gulf of Mexico and the importance of the atchafalaya river. Aquat Geochem. 2011;17:519–544. doi: 10.1007/s10498-011-9134-3. [DOI] [Google Scholar]
- 78.Ruttenberg KC. Temporal and spatial variability of dissolved organic and inorganic phosphorus, and metrics of phosphorus bioavailability in an upwelling-dominated coastal system. J. Geophys. Res. 2005;110:1–22. [Google Scholar]
- 79.Ruttenberg KC, Dyhrman ST. Dissolved organic phosphorus production during simulated phytoplankton blooms in a coastal upwelling system. Front. Microbiol. 2012;3:274. doi: 10.3389/fmicb.2012.00274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Sylvan JB, Quigg A, Tozzi S, Ammerman JW. Eutrophication-induced phosphorus limitation in the Mississippi River plume: Evidence from fast repetition rate fluorometry. Limnol. Oceanogr. 2007;52:2679–2685. doi: 10.4319/lo.2007.52.6.2679. [DOI] [Google Scholar]
- 81.Liu H-C, Gong G-C, Chang J. Lateral water exchange between shelf-margin upwelling and Kuroshio waters influences phosphorus stress in microphytoplankton. Mar. Ecol. Prog. Ser. 2010;409:121–130. doi: 10.3354/meps08603. [DOI] [Google Scholar]
- 82.Liu H-C, et al. Discrimination between the influences of river discharge and coastal upwelling on summer microphytoplankton phosphorus stress in the East China Sea. Cont Shelf Res. 2013;60:104–112. doi: 10.1016/j.csr.2013.04.017. [DOI] [Google Scholar]
- 83.Ou L, et al. Phosphorus stress of phytoplankton in the Taiwan Strait determined by bulk and single-cell alkaline phosphatase activity assays. Mar. Ecol. Prog. Ser. 2006;327:95–106. doi: 10.3354/meps327095. [DOI] [Google Scholar]
- 84.Tanaka T, et al. Lack of P-limitation of phytoplankton and heterotrophic prokaryotes in surface waters of three anticyclonic eddies in the stratified Mediterranean Sea. Biogeosciences. 2011;8:525–538. doi: 10.5194/bg-8-525-2011. [DOI] [Google Scholar]
- 85.Vidal M, Duarte CM, Agustí S, Gasol JM, Vaqué D. Alkaline phosphatase activities in the central Atlantic Ocean indicate large areas with phosphorus deficiency. Mar. Ecol. Prog. Ser. 2003;262:43–53. doi: 10.3354/meps262043. [DOI] [Google Scholar]
- 86.Bogé G, Jamet JL, Richard S, Jamet D, Jean N. Contribution of copepods, cladocerans and cirripeds to phosphatase activity in mediterranean zooplankton. Hydrobiologia. 2002;468:147–154. doi: 10.1023/A:1015266722757. [DOI] [Google Scholar]
- 87.Bogé G, Jean N, Jamet JL, Jamet D, Richard S. Seasonal changes in phosphatase activities in Toulon Bay (France) Mar. Environ. Res. 2006;61:1–18. doi: 10.1016/j.marenvres.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 88.Jean N, Bogé G, Jamet J-L, Richard S, Jamet D. Seasonal changes in zooplanktonic alkaline phosphatase activity in Toulon Bay (France): the role of Cypris larvae. Mar. Pollut. Bull. 2003;46:346–352. doi: 10.1016/S0025-326X(02)00450-2. [DOI] [PubMed] [Google Scholar]
- 89.Kwon HK, Oh SJ, Yang HS. Ecological significance of alkaline phosphatase activity and phosphatase-hydrolyzed phosphorus in the northern part of Gamak Bay, Korea. Mar. Pollut. Bull. 2011;62:2476–2482. doi: 10.1016/j.marpolbul.2011.07.027. [DOI] [PubMed] [Google Scholar]
- 90.Li H, Veldhuis MJW, Post AF. Alkaline phosphatase activities among planktonic communities in the northern Red Sea. Mar. Ecol. Prog. Ser. 1998;173:107–115. doi: 10.3354/meps173107. [DOI] [Google Scholar]
- 91.Mamatha SS, et al. Alkaline phosphatase activity at the southwest coast of India: A comparison of locations differently affected by upwelling. J. Sea Res. 2015;95:196–205. doi: 10.1016/j.seares.2014.06.002. [DOI] [Google Scholar]
- 92.Nicholson D, Dyhrman S, Chavez F, Paytan A. Alkaline phosphatase activity in the phytoplankton communities of Monterey Bay and San Francisco Bay. Limnol. Oceanogr. 2006;51:874–883. doi: 10.4319/lo.2006.51.2.0874. [DOI] [Google Scholar]
- 93.Oh SJ, Yoon YH, Yamamoto T, Matsuyama Y. Alkaline phosphatase activity and phosphatase hydrolyzable phosphorus for phytoplankton in hiroshima bay, Japan. Ocean Sci. J. 2005;40:183–190. doi: 10.1007/BF03023517. [DOI] [Google Scholar]
- 94.Stihl A, Sommer U, Post AF. Alkaline phosphatase activities among populations of the colony-forming diazotrophic cyanobacterium Trichodesmium spp. (cyanobacteria) in the Red Sea. J. Phycol. 2001;37:310–317. doi: 10.1046/j.1529-8817.2001.037002310.x. [DOI] [Google Scholar]
- 95.Baltar F, De Corte D, Thomson B, Yokokawa T. Teasing apart the different size pools of extracellular enzymatic activity in the ocean. Sci. Total Environ. 2019;660:690–696. doi: 10.1016/j.scitotenv.2019.01.083. [DOI] [PubMed] [Google Scholar]
- 96.Moutin T, et al. Phosphate availability controls Trichodesmium spp. biomass in the SW Pacific Ocean. Mar. Ecol. Prog. Ser. 2005;297:15–21. doi: 10.3354/meps297015. [DOI] [Google Scholar]
- 97.Sohm JA, Mahaffey C, Capone DG. Assessment of relative phosphorus limitation of Trichodesmium spp. in the North Pacific, North Atlantic, and the north coast of Australia. Limnol. Oceanogr. 2008;53:2495–2502. doi: 10.4319/lo.2008.53.6.2495. [DOI] [Google Scholar]
- 98.Celussi M, Del Negro P. Microbial degradation at a shallow coastal site: Long-term spectra and rates of exoenzymatic activities in the NE Adriatic Sea. Estuar. Coast. Shelf Sci. 2012;115:75–86. doi: 10.1016/j.ecss.2012.02.002. [DOI] [Google Scholar]
- 99.Wurl O, Zimmer L, Cutter GA. Arsenic and phosphorus biogeochemistry in the ocean: Arsenic species as proxies for P-limitation. Limnol. Oceanogr. 2013;58:729–740. doi: 10.4319/lo.2013.58.2.0729. [DOI] [Google Scholar]
- 100.Wang, D. Ecophysiological responces of phytoplankton to phosphorus in the typical region of the Yellow Sea, Xiamen University, (2008).
- 101.Mather, R. L. Reactivity and nature of organic nutrients in the Atlantic Ocean, University of Liverpool, (2010).
- 102.Nicholson, D. P. Phosphorus status of marine phytoplankton communities in Monterey and San Francisco Bays, Stanford University, (2003).
- 103.Glover, D. M., Jenkins, W. J. & Doney, S. C. Modeling Methods for Marine Science. (Cambridge University Press, 2011).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Su B, 2023. A dataset of global ocean alkaline phosphotase activity. Figshare. [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
The source codes for identifying outliers used in this paper are available at https://github.com/BGM-USD2020/GAPAD_codes.git.